Abstract
In low-phosphorus (P) marine systems, phytoplankton replace membrane phospholipids with non-phosphorus lipids, but it is not known how rapidly this substitution occurs. Here, when cells of the model diatom Thalassiosira pseudonana were transferred from P-replete medium to P-free medium, the phospholipid content of the cells rapidly declined within 48 h from 45±0.9 to 21±4.5% of the total membrane lipids; the difference was made up by non-phosphorus lipids. Conversely, when P-limited T. pseudonana were resupplied with P, cells reduced the percentage of their total membrane lipids contributed by a non-phosphorus lipid from 43±1.5 to 7.3±0.9% within 24 h, whereas the contribution by phospholipids rose from 2.2±0.1 to 44±3%. This dynamic phospholipid reservoir contained sufficient P to synthesize multiple haploid genomes, suggesting that phospholipid turnover could be an important P source for cells. Field observations of phytoplankton lipid content may thus reflect short-term changes in P supply and cellular physiology, rather than simply long-term adjustment to the environment.
Keywords: betaine lipids, DGCC, lipid substitution, non-phosphorus lipids, phosphatidylcholine, diatoms
Phosphate is chronically scarce in oligotrophic oceans (Krom et al., 1991; Karl et al., 1997; Wu et al., 2000), and phytoplankton appear to posses highly effective physiological mechanisms to reduce their phosphorus (P) quota in these environments (Twining et al., 2010). For example, all phytoplankton groups examined to date, both in culture and in situ, substitute non-phosphorus (sulfur- and nitrogen-containing) lipids for phospholipids in their membranes under low-P conditions (Van Mooy et al., 2009). Such lipid substitution in bacteria (Minnikin et al., 1974; Benning et al., 1995) can take place within hours (Zavaleta-Pastor et al., 2010). However, it is not known how rapidly phytoplankton remodel their membranes, and whether field observations (Van Mooy et al., 2009) reflect long-term adjustment to the environment or an immediate cellular response to low-P conditions.
The diatom Thalassiosira pseudonana and other eukaryotic phytoplankton contain the phospholipids phosphatidylcholine (PC), phosphatidylglycerol (PG) and phosphatidylethanolamine (PE). Under low-P conditions, T. pseudonana substitutes PC with the betaine lipid diacylglycerylcarboxyhydroxymethylcholine (DGCC), and PG with the sulfolipid sulfoquinovosyldiacylglycerol (SQDG) (Van Mooy et al., 2009).
Two culture experiments were conducted to test how rapidly lipid substitution occurs in T. pseudonana upon changes in phosphate concentration. In the first experiment, T. pseudonana CCMP1335 was grown in P-replete medium (in triplicate, Supplementary Methods) until early log-phase. Cells were then gently filtered onto 0.2 μm polycarbonate membranes and resuspended in either phosphate-free (−P) or phosphate-replete (+P, 36 μmol l−1 phosphate) medium. However, as the −P cultures contained 0.5±0.2 μmol l−1 phosphate at 0 h, representing a carryover of the original culture medium on the order of 1%, the cells did not experience limiting levels of P instantaneously. Samples for intact polar lipids (IPLs) and cell counts were taken over 48 h (Supplementary Methods).
The cells responded quickly after transfer, and grew equally well in the −P and +P treatments over 48 h (Figure 1a). However, despite the minor carryover of P in the culture medium, the DGCC:PC ratio increased sharply in the −P treatments after 24 h, relative to the +P controls, indicating lipid substitution in response to low-P conditions (Figure 1b, Supplementary Figure 1, note that DGCC was undetectable before 24 h). Similarly, the SQDG:PG ratio rose in the −P compared with the +P treatments (Figure 1c). The molar percentage contribution by phospholipids to total IPLs fell from 45±0.9% at 0 h to 21±4.5% at 48 h in the −P cultures (Figure 1d). Concomitantly, the contribution by DGCC rose from undetectable levels to 11.5±1.7%, and by SQDG from 38±2 to 52±6% (Supplementary Figure 1). The contribution by the lipids monogalactosyldiacylglycerol and digalactosyldiacylglycerol did not differ between treatments (Supplementary Figure 1). This contrasts with higher plants, which have been shown to substitute PC with digalactosyldiacylglycerol (Tjellström et al., 2008).
Figure 1.
A time course of parameters in an experiment where P-replete T. pseudonana cells were harvested and transferred into either P-replete (+P) or P-free (−P) medium. (a) Cell growth in +P and −P treatments; (b) DGCC:PC ratio (note that DGCC was undetectable before 24 h); (c) SQDG:PG ratio; (d) combined molar percentage of total IPLs contributed by the phospholipids PC, PG and PE; (e) combined content of the three phospholipids PC, PG and PE per cell; (f) PE:PC ratio. All data are treatment mean±1 s.d. of n=3.
The per-cell content of the phospholipids PC, PG and PE dropped from 1.8±0.2 fmol per cell at 6 h to 0.92±0.12 fmol per cell (Figure 1e). Moreover, the amount of phospholipids per ml of culture medium decreased by 160±30 pmol ml−1 in the −P treatments between 24 and 48 h (Supplementary Figure 1), indicating that the per-cell shift in phospholipids was caused by net breakdown of these molecules. This breakdown of phospholipids represented a quantity of P equivalent to 3.0±0.6 × 108 atoms P per cell for the new cells that grew during the 24 to 48 h time period. As T. pseudonana contains 7.2 × 107 P atoms per haploid genome (von Dassow et al., 2008; Supplementary Methods), this is equivalent to the amount of P needed for each cell to synthesize about four haploid genomes. Clearly, the phospholipid pool represents a significant amount of P that is rapidly mobilized in response to low-P conditions.
The relatively constant PE:PC ratio (Figure 1f) underscores that the breakdown rates of these two lipids were relatively equal. This supports the finding of Riekhof et al. (2005) that betaine lipid synthesis is linked to the cleavage of phospholipid headgroups (for example, via indiscriminant phospholipase C activity).
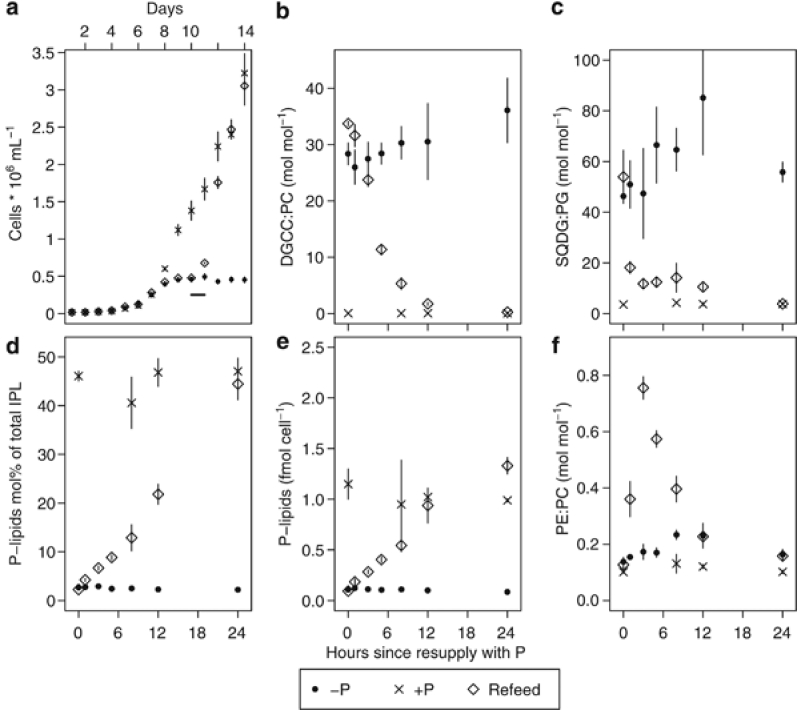
To test the response time of P-limited T. pseudonana to P resupply, triplicate flasks of T. pseudonana were grown (Supplementary Methods) until P-limited, and then resupplied with 36 μmol l−1 phosphate (Refeed treatment), and tracked relative to triplicate P-replete (+P) and P-limited (−P) controls. The cells in the Refeed cultures resumed growth within 24 h after phosphate addition (Figure 2a). The DGCC:PC ratio in the Refeed cultures fell from an initial value of 33.7±0.4 to 1.7±0.3 at 12 h, with a discernable change within the first hour (Figure 2b), and this was driven both by the breakdown of DGCC and the synthesis of new PC. DGCC per cell decreased after 5 h (Supplementary Figure 2), and the molar percentage of IPLs contributed by DGCC fell from 43±1.5% initially to 7.3±0.9% at 24 h (Supplementary Figure 2). The SQDG:PG ratio differed dramatically between +P and −P cultures, and in the Refeed cultures it dropped to values identical to the +P cultures by 24 h (Figure 2c). Change in the SQDG:PG ratio was driven by changes in PG content; the percentage contribution by SQDG (and by monogalactosyldiacylglycerol and digalactosyldiacylglycerol) remained largely constant and identical between treatments (Supplementary Figure 2). The PE:PC ratio spiked sharply in the first 12 h (Figure 2f), which is consistent with the activation of a synthesis pathway where PC is synthesized from PE as has been observed in yeast (but not in plants; Birner and Daum, 2003).
Figure 2.
A time course of parameters in an experiment where P-limited T. pseudonana cells (−P) were resupplied with phosphate (Refeed) and compared to replete (+P) treatments. (a) Cell growth curve. The solid horizontal line between Days 10 and 11 indicates the 24 h period over which lipids were sampled, starting from the time of phosphate resupply. (b–f) share a common x-axis corresponding to the horizontal line shown in (a). (b) DGCC:PC ratio; (c) SQDG:PG ratio; (d) combined molar percentage of total IPLs contributed by PC, PG and PE; (e) combined per-cell content of PC, PG and PE; (f) PE:PC ratio. All data are mean±1 s.d. of n=3.
In the Refeed treatments, the percentage contribution by phospholipids to total IPLs increased from 2.2±0.1% at 0 h to 44±3% at 24 h (Figure 2d). The overall phospholipid content increased by 1.2±0.1 fmol per cell, or 7.4±0.5 × 108 atoms P per cell, which is enough P to synthesize 10 haploid genomes (Figure 2e). Overall, T. pseudonana responds rapidly to P resupply, increasing phospholipid synthesis and degrading DGCC. As DGCC contains nitrogen, there may be a physiological benefit to actively recycling it.
These data demonstrate that the IPL composition of T. pseudonana is dynamic and changes within one cell division or less in response to changes in external P concentrations. The breakdown of phospholipids appears to liberate a significant quantity of P, enough for the synthesis of several genomes. Likewise, cells can sequester a significant amount of P in phospholipids when P concentrations increase suddenly. Recycling of P from phospholipids may be able to support several cell divisions in low-P conditions. This suggests that recent observations of elevated betaine lipid:PC and SQDG:PG ratios in low-P marine systems (Van Mooy et al., 2006, 2009) at least partly reflect a short-term physiological response to low P. Most marine phytoplankton do appear to have inherently low phospholipid contents, even when P is replete (Van Mooy et al., 2009; Van Mooy and Fredericks, 2010). Although it has not yet been tested whether P enrichment of low-P ocean samples leads to changes in whole-community lipid composition, at least one phytoplankton taxon, the ubiquitous cyanobacterium Synechococcus (which undertakes SQDG–PG substitution; Van Mooy et al., 2009), can increase its total cellular P-quota upon short-term P supply by mesoscale eddies (Twining et al., 2010). Our data indicate that membrane remodelling may be important for such a response, and that it is an important and dynamic process for phytoplankton in the sea.
Acknowledgments
We thank Helen Fredericks for help with chromatography and mass spectrometry, and two anonymous reviewers for constructive criticism. This research was funded by NSF OCE-0646944 and OCE-1045670 (BVM), OCE-0723677 and OCE-0549794 (STD), and by the Graduate School, National Oceanography Centre, Southampton (PM).
Footnotes
Supplementary Information accompanies the paper on The ISME Journal website (http://www.nature.com/ismej)
Supplementary Material
References
- Benning C, Huang ZH, Gage DA. Accumulation of a novel glycolipid and a betaine lipid in cells of Rhodobacter sphaeroides grown under phosphate limitation. Arch Biochem Biophys. 1995;317:103–111. doi: 10.1006/abbi.1995.1141. [DOI] [PubMed] [Google Scholar]
- Birner R, Daum G. Biogenesis and cellular dynamics of aminoglycerophospholipids. Int Rev Cytol. 2003;225:273–323. doi: 10.1016/s0074-7696(05)25007-6. [DOI] [PubMed] [Google Scholar]
- Karl D, Letelier R, Tupas L, Dore J, Christian J, Hebel D. The role of nitrogen fixation in biogeochemical cycling in the subtropical North Pacific Ocean. Nature. 1997;388:533–538. [Google Scholar]
- Krom MD, Kress N, Brenner S, Gordon LI. Phosphorus limitation of primary productivity in the Eastern Mediterranean Sea. Limnol Oceanogr. 1991;36:424–432. [Google Scholar]
- Minnikin DE, Abdolrahimzadeh H, Baddiley J. Replacement of acidic phospholipids by acidic glycolipids in Pseudomonas diminuta. Nature. 1974;249:268–269. doi: 10.1038/249268a0. [DOI] [PubMed] [Google Scholar]
- Riekhof WR, Andre C, Benning C. Two enzymes, BtaA and BtaB, are sufficient for betaine lipid biosynthesis in bacteria. Arch Biochem Biophys. 2005;441:96–105. doi: 10.1016/j.abb.2005.07.001. [DOI] [PubMed] [Google Scholar]
- Tjellström H, Andersson MX, Larsson KE, Sandelius AS. Membrane phospholipids as a phosphate reserve: the dynamic nature of phospholipid-to-digalactosyl diacylglycerol exchange in higher plants. Plant Cell Environ. 2008;31:1388–1398. doi: 10.1111/j.1365-3040.2008.01851.x. [DOI] [PubMed] [Google Scholar]
- Twining BS, Nuñez-Milland D, Vogt S, Johnson RS, Sedwick PN. Variations in Synechococcus cell quotas of phosphorus, sulfur, manganese, iron, nickel, and zinc within mesoscale eddies in the Sargasso Sea. Limnol Oceanogr. 2010;55:492–506. [Google Scholar]
- Van Mooy BAS, Fredericks HF. Bacterial and eukaryotic intact polar lipids in the eastern subtropical South Pacific: water-column distribution, planktonic sources, and fatty acid composition. Geochim Cosmochim Acta. 2010;74:6499–6516. [Google Scholar]
- Van Mooy BAS, Fredricks HF, Pedler BE, Dyhrman ST, Karl DM, Koblížek M, et al. Phytoplankton in the ocean use non-phosphorus lipids in response to phosphorus scarcity. Nature. 2009;458:69–72. doi: 10.1038/nature07659. [DOI] [PubMed] [Google Scholar]
- Van Mooy BAS, Rocap G, Fredricks HF, Evans CT, Devol AH. Sulfolipids dramatically decrease phosphorus demand by picocyanobacteria in oligotrophic marine environments. Proc Natl Acad Sci USA. 2006;103:8607–8612. doi: 10.1073/pnas.0600540103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Dassow P, Petersen TW, Chepurnov VA, Armbrust EV. Inter- and intraspecific relationships between nuclear DNA content and cell size in selected members of the centric diatom genus Thalassiosira (Bacillariophyceae) J Phycol. 2008;44:335–349. doi: 10.1111/j.1529-8817.2008.00476.x. [DOI] [PubMed] [Google Scholar]
- Wu JF, Sunda W, Boyle EA, Karl DM. Phosphate depletion in the western North Atlantic Ocean. Science. 2000;289:759–762. doi: 10.1126/science.289.5480.759. [DOI] [PubMed] [Google Scholar]
- Zavaleta-Pastor M, Sohlenkamp C, Gao JL, Ziqiang G, Zaheer R, Finan TM, et al. Sinorhizobium meliloti phospholipase C required for lipid remodeling during phosphorus limitation. Proc Natl Acad Sci USA. 2010;107:302–307. doi: 10.1073/pnas.0912930107. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.