Abstract
Cyanobacteria in the genus Acaryochloris are the only known oxyphototrophs that have exchanged chlorophyll a (Chl a) with Chl d as their primary photopigment, facilitating oxygenic photosynthesis with near infrared (NIR) light. Yet their ecology and natural habitats are largely unknown. We used hyperspectral and variable chlorophyll fluorescence imaging, scanning electron microscopy, photopigment analysis and DNA sequencing to show that Acaryochloris-like cyanobacteria thrive underneath crustose coralline algae in a widespread endolithic habitat on coral reefs. This finding suggests an important role of Chl d-containing cyanobacteria in a range of hitherto unexplored endolithic habitats, where NIR light-driven oxygenic photosynthesis may be significant.
Keywords: cyanobacteria, Acaryochloris marina, chlorophyll d, endolithic, bioimaging
Chlorophyll d (Chl d) is only found in the cyanobacterium Acaryochloris marina, where it has replaced Chl a as the dominant pigment in the antennae and in the reaction centers extending the range of photosynthetic active radiation into the near infrared (NIR) light region (700–740 nm; Miyashita et al., 1996; Kühl et al., 2007). Only four A. marina strains have so far been isolated (Table 1), and they have been subject to detailed biochemical and genomic analysis (for example, Miller et al., 2005; Swingley et al., 2008). Although there is increasing evidence that Chl d is more widely distributed in both aquatic and terrestrial systems (Kashiyama et al., 2008; Table 1), the ecology of Chl d-containing cyanobacteria is still largely unknown and in only one case, has a detailed description of its habitat been considered with actual in situ measurements of photosynthetic activity (Kühl et al., 2005). Among cyanobacteria, A. marina has an unusually large genome with a significant proportion of its DNA localized to 9 plasmids (Swingley et al., 2008), and it was speculated that such high capacity for genetic mobility and genome expansion could facilitate its adaptation to a variety of ecological niches. In this study, we show that Chl d-containing cyanobacteria can colonize widespread endolithic habitats on coral reefs, where they occupy a distinct optically defined niche deprived of visible light.
Table 1. Habitats for Chlorophyll d-containing and/or Acaryochloris marina-like phototrophs.
| Habitat | Evidence | Geographic location |
|---|---|---|
| Marine | ||
| Coral reefs and mediterranean: | ||
| Epizoic biofilms on didemnid ascidians | A. marina isolate (MBIC11017); absorbance and fluorescence spectra; pigment extraction; cell morphology. | Palau (Miyashita et al., 1996) and Australia (Kühl et al., 2005) |
| Epizoic biofilms on colonial ascidian | Fluorescence microscopy, pigment extraction, cell morphology. | Mediteranean sea (Martínez-Garcia et al., 2010) |
| Epilitihic biofilms on dead coral | A. marina. isolate (HICR111A); absorbance and fluorescence spectra; pigment extraction; cell morphology; genetic analysis. | Australia (Mohr et al., 2010) |
| Red algae | ||
| Epiphytic patches on thallus | A. marina isolate (AWAJI-1); absorbance and fluorescence spectra; pigment extraction; 16S rRNA gene sequence. | Japan (Murakami et al., 2004; Ohkubo et al., 2006) |
| Endolithic biofilm under crustose coralline algae | Absorbance and fluorescence spectra; pigment extraction; 16S rRNA gene sequence; SEM, cell morphology | Australia (this study) |
| Brown and green algae | 16S rRNA gene sequence | Japan (Ohkubo et al., 2006) |
| Sediments | ||
| Temperate coastal basins | Pigment extraction | Japan (Kashiyama et al., 2008) |
| Subarctic and arctic basins | Pigment extraction | Bering Sea and Arctic Sea (Kashiyama et al., 2008) |
| Stromatolites | 16S rRNA gene sequence | Australia (Goh et al., 2009) |
| Saline and freshwater | ||
| Sediments/biofilms | ||
| Hypertrophic lake | A. marina isolate (CCMEE 5410); absorbance and fluorescence spectra; pigment extraction; morphology; 16S rRNA gene sequence | USA (Miller et al., 2005) |
| Temperate freshwater lake | Pigment extraction | Japan (Kashiyama et al., 2008) |
| Saline lakes | Pigment extraction | Antarctica (Kashiyama et al., 2008) |
| High altitude lakes | 16S rRNA gene sequence | Bolivia (Fleming and Prufert-Bebout, 2010) |
| Terrestrial | ||
| Epilithic biofilms | 16S rRNA gene sequence | Mexico (McNamara et al., 2006) |
| Epi- and endolithic biofilms | TEM; SEM; 16S rRNA gene sequence; cell morphology | Antarctica (de los Rios et al., 2007) |
Abbreviations: rRNA, ribosomal RNA; SEM, scanning electron microscopy; TEM, transmission electron microscopy.
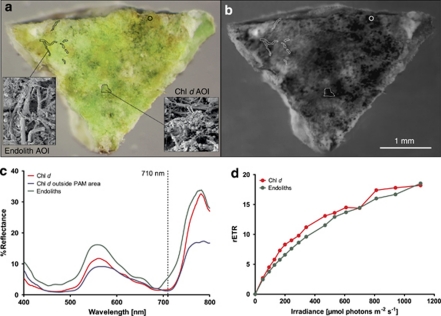
We combined hyperspectral and variable chlorophyll fluorescence imaging (Kühl and Polerecky, 2008) with pigment extraction (Mohr et al., 2010), scanning electron microscopy and DNA pyrosequencing (see Materials and Methods in Supplementary Online Material) to investigate the occurrence of Chl d-containing phototrophs on patches of dead coral branches from the reef flat and shallow outer reef crest off Heron Island, Australia. Such branches were almost completely covered by a diversity of crustose coralline algae (predominantly Porolithon spp. and Lithophyllum spp.; Ringeltaube and Harvey, 2000) and other epiphytic macroalgae, sponges and didemnid ascidians. Careful mechanical removal of the coralline algal layer revealed a thin yellow–green, biofilm closely associated with the carbonate skeleton immediately below (Figures 1a, 2a and Supplementary Figure S1). Hyperspectral imaging in combination with variable chlorophyll fluorescence imaging and scanning electron microscopy confirmed that this biofilm contained photosynthetically active Chl d in patches of A. marina-like cells (1–2 μm) intermixed with other endolithic filamentous cyanobacteria and the green alga Ostreobium sp. (Figures 1b–d and Figures 2b–f).
Figure 1.
Endolithic habitat of Chl d -containing cyanobacteria. (a) Typical Chl d-containing yellow–greenish biofilm found directly under encrusting coralline algae. Labels indicate two different areas of interest (AOI) where pulse-amplitude modulated (PAM) variable chlorophyll fluorescence imaging was carried out: (1) Endolith AOI is dominated by filamentous cyanobacteria and green algae, and (2) Chl d AOI is dominated by Acaryochloris-like Chl d-containing cyanobacteria. Another circular AOI indicates a Chl d-containing area where no PAM analysis was carried out. Inserts show scanning electron microscopy pictures for the respective AOI; note the presence of round 1–2 μm large bacteria, similar in size and shape to Acaryochloris marina. (b) Hyperspectral reflectance image at 710 nm, that is, the peak absorbance of Chl d in situ. Dark areas indicate strong Chl d absorption. (c) Reflectance spectra of the two AOI showing distinct spectral signatures of Chl d around 710 nm. (d) Photosynthetic activity versus irradiance measured as the PSII related relative electron transport rate (rETR) in the two AOI.
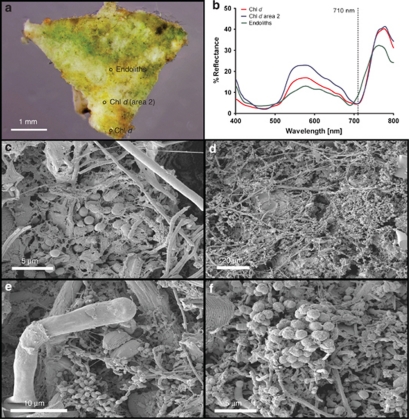
Figure 2.
Scanning electron microscopy and hyperspectral measurements performed on a dead coral sample with the crustose coralline algal layer removed. (a) Biofilm found under encrusting coralline algae. (b) Reflectance spectra extracted from hyperspectral imaging of three AOI in (a) showing Chl d absorption around 710 nm. (c–f) Scanning electron microscopy pictures portraying biofilms growing below encrusting corraline algae. Note frequent occurrence of 1–2 μm large A. marina like cells (c–f), along with filamentous cyanobacteria (c, d) and Ostreobium-like filamentous green algae (e).
We did not attempt a quantification of light attenuation in the crustose coralline layer, but earlier studies of the light microenvironment in coral skeleton (Magnusson et al., 2007) showed a very strong light attenuation of visible light (down to 0.1–1% of the incident irradiance) in the uppermost millimetres, whereas NIR light penetrated significantly deeper reaching 10–100 times the level of visible wavelengths in the skeleton matrix. However, coral skeleton density varies with species affecting light penetration (Enriquez et al., 2005; Magnusson et al., 2007).
Vertical slices through the encrusted coralline layer further revealed that Chl d is found in a thin layer in between the coralline alga and deeper parts of the coral skeleton (Supplementary Figures S1A, B), which was harboring other phototrophic endoliths, such as the green algae Ostreobium sp. These siphonalean green algae can thrive on NIR (Halldal, 1968) due to antenna chlorophylls bound to the Lhca1-type protein (Koehne et al., 1999) that absorb light beyond the typical absorption maximum of Chl d (Kühl et al., 2007). We found shoulders between 710 and 725 nm in the reflectance spectra indicative of such far-red antenna pigments (Figures 1a, 2b, Supplementary Figure S1C). Pigment extraction analysis of crustose coralline samples with ∼1–2 mm of attached skeleton showed a distinct spectral signal of Chl d, albeit on a large background of coralline algal photopigments (Supplementary Figure S2). Pyrosequencing analysis of DNA extracted from similar samples revealed an endolithic microbial community with cyanobacteria as the dominant oxyphototrophs, of which ∼5% could be assigned to A. marina (Supplementary Figure S3); A. marina accounted for ∼1.8% of the total microbial community. In this first attempt of quantifying the presence of A. marina in a natural habitat, a total of five operational taxonomic units (OTUs) showed significant homology to known Acaryochloris species.
Crustose coralline algae are ubiquitous on coral reefs and are essential for stabilizing corals against mechanical erosion by sealing the porous carbonate skeleton matrix and consolidating the reef framework. Inside shallow water patches of coralline encrusted corals NIR light prevails (Kühl et al., 2007) and our data indicate that A. marina-like Chl d-containing cyanobacteria are much more common on coral reefs than previously thought. Interestingly, a new A. marina-like isolate was reported from similar habitats (Mohr et al., 2010). We have recently started a survey of Chl d underneath crustose coralline algae from different climatic zones, and first preliminary results obtained with pigment extraction and hyperspectral imaging (data not shown) confirmed the presence of Chl d underneath coralline specimens encrusting sandstone rock platforms around Sydney in more temperate coastal waters. Our results can also explain several sequence-based findings of A. marina and/or Chl d in other endolithic habitats (Table 1) and thus provide strong evidence for a more widespread occurrence of A. marina-like phototrophs, supporting the hypothesis that these unique oxyphototrophs exhibit a large capacity for niche adaptation (Swingley et al., 2008). Recently, a new chlorophyll, that is, Chl f, has been identified in cyanobacteria from stromatolites (Chen et al., 2010), and this pigment shows an even more red-shifted absorption than Chl d. It is not known whether this new pigment is indeed involved in active photosynthesis, but Chl f-containing phototrophs may colonize similar habitats where Chl d-containing cyanobacteria thrive. Thus Chl d-containing cyanobacteria and other phototrophs with far-red absorbing chlorophylls may have an important yet unexplored role as NIR-driven oxygenic primary producers in both terrestrial and aquatic ecosystems.
Acknowledgments
The work was conducted under a Marine Parks Permit (G06/178151.1) from the Great Barrier Reef Authority and was supported by the Danish Natural Science Research Council and the Australian Research Council. We thank the Heron Island Research Station staff, Martin Schliep and Waleed Abu Al-Soud for technical assistance, HPLC and sequencing analysis.
Footnotes
Supplementary Information accompanies the paper on The ISME Journal website (http://www.nature.com/ismej)
Supplementary Material
References
- Chen M, Schliep M, Willows RD, Cai Z-L, Neilan BA, Scheer H. A red-shifted chlorophyll. Science. 2010;329:1318–1319. doi: 10.1126/science.1191127. [DOI] [PubMed] [Google Scholar]
- Enriquez S, Méndez ER, Iglesias-Prieto R. Multiple scattering on coral skeletons enhances light absorption by symbiotic algae. Limnol Oceanogr. 2005;50:1025–1031. [Google Scholar]
- Fleming ED, Prufert-Bebout L. Characterization of cyanobacterial communities form high-elevation lakes in the Bolivian Andes. J Geophys Res. 2010;115:G00D07. [Google Scholar]
- Goh F, Allen MA, Leuko S, Kawaguchi T, Decho AW, Burns BP, et al. Determining the specific microbial populations and their spatial distribution within the stromatolite ecosystem of Shark Bay. ISME J. 2009;3:383–396. doi: 10.1038/ismej.2008.114. [DOI] [PubMed] [Google Scholar]
- Halldal P. Photosynthetic capacities and photosynthetic action spectra of endozoic algae of the massive coral Favia. Biol Bull. 1968;134:411–424. [Google Scholar]
- Kashiyama Y, Miyashita H, Ohkubo S, Ogawa NO, Chikaraishi Y, Takano Y, et al. Evidence for global chlorophyll d. Science. 2008;321:658. doi: 10.1126/science.1158761. [DOI] [PubMed] [Google Scholar]
- Koehne B, Elli GG, Jennings R, Wilhelm C, Trissl H-W. Spectroscopic and molecular characterisation of a long wavelengths absorbing antenna of Ostreobium sp. Biochim Biophys Acta. 1999;1412:94–107. doi: 10.1016/s0005-2728(99)00061-4. [DOI] [PubMed] [Google Scholar]
- Kühl M, Chen M, Larkum AWD.2007Biology of the chlorophyll d-containing cyanobacterium Acaryochloris marinaIn: Seckbach J (ed).Algae and Cyanobacteria in Extreme Environments Springer: Berlin; 101–123. [Google Scholar]
- Kühl M, Chen M, Ralph PJ, Schreiber U, Larkum AWD. A niche for cyanobacteria containing chlorophyll d. Nature. 2005;433:820. doi: 10.1038/433820a. [DOI] [PubMed] [Google Scholar]
- Kühl M, Polerecky L. Functional and structural imaging of phototrophic microbial commmunities and symbioses. Aq Microb Ecol. 2008;53:99–118. [Google Scholar]
- Magnusson SH, Fine M, Kühl M. Light microclimate of endolithic phototrophs in the scleractinian corals Montipora monasteriata and Porites cylindrica. Mar Ecol Progr Ser. 2007;332:119–128. [Google Scholar]
- Martínez-Garcia M, Koblízek M, López-Legentil S, Antón J.2010Epibiosis of oxygenic phototrophs containing chlorophylls a, b, c, and d on the colonial ascidian Cystodytes dellechajei Microb Ecolpublished ahead of print, doi:10 1007/s00248 1010-9694-6. [DOI] [PubMed]
- McNamara CJ, Perry TD, VI, Bearce KA, Hernandez-Duque G, Mitchell R. Epilithic and endolithic bacterial communities in limestone from a Mayan archaeological site. Microb Ecol. 2006;51:51–64. doi: 10.1007/s00248-005-0200-5. [DOI] [PubMed] [Google Scholar]
- Miller SM, Augustine S, Olson TL, Blankenship RE, Selker J, Wood AM. Discovery of a free-living chlorophyll d-producing cyanobacterium with a hybrid proteobacterial/cyanobacterial small-subunit rRNA gene. Proc Natl Acad Sci USA. 2005;102:850–855. doi: 10.1073/pnas.0405667102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyashita H, Ikemoto H, Kurano N, Adachi K, Chihara M, Miyachi S. Chlorophyll d as a major pigment. Nature. 1996;383:402. [Google Scholar]
- Mohr R, Voss B, Schliep M, Kurz T, Maldener I, Adams DG, et al. A new chlorophyll d-containing cyanobacterium: evidence for niche adaptation in the genus Acaryochloris. ISME J. 2010;4:1456–1469. doi: 10.1038/ismej.2010.67. [DOI] [PubMed] [Google Scholar]
- Murakami A, Miyashita H, Iseki M, Adachi K, Mimuro M. Chlorophyll d in an epiphytic cyanobacterium of red algae. Science. 2004;303:1633. doi: 10.1126/science.1095459. [DOI] [PubMed] [Google Scholar]
- Ohkubo S, Miyashita H, Murakami A, Takeyama H, Tsuchiya T, Mimuro M. Molecular detection of epiphytic Acaryochloris spp. on marine macroalgae. Appl Environ Microbiol. 2006;72:7912–7915. doi: 10.1128/AEM.01148-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringeltaube P, Harvey A. Non-geniculate coralline algae (Corallinales, Rhodophyta) on Heron Reef, Great Barrier Reef (Australia) Bot Marina. 2000;43:431–454. [Google Scholar]
- de los Rios A, Grube M, Sancho LG, Ascaso C. Ultrastructural and genetic characteristics of endolithic cyanobacterial biofilms colonizing Antarctic granite rocks. FEMS Microbiol Ecol. 2007;59:386–395. doi: 10.1111/j.1574-6941.2006.00256.x. [DOI] [PubMed] [Google Scholar]
- Swingley WD, Chen M, Cheung PC, Conrad AL, Dejesa LC, Hao J, et al. Niche adaptation and genome expansion in the chlorophyll d-producing cyanobacterium Acaryochloris marina. Proc Natl Acad Sci USA. 2008;105:2005–2010. doi: 10.1073/pnas.0709772105. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.