Abstract
Hereditary phosphofructokinase (PFK) deficiency was diagnosed in two Wachtelhund dogs and suspected in three related Wachtelhund dogs with exercise intolerance, hemolytic anemia, and pigmenturia. Severe, persistent reticulocytosis in light of only mild anemia together with hemoglobinuria after strenuous exercise suggested PFK deficiency. Low erythrocyte PFK activity together with low 2,3-diphosphoglycerate concentrations and a high hemoglobin-oxygen affinity confirmed the diagnosis. The PFK deficiency is due to a single missense mutation in the muscle-type PFK M-PFK gene in English springer and American cocker spaniels, whippets, and mixed-breed dogs; however, these PFK-deficient Wachtelhunds do not have the same PFK mutation.
Introduction
Phosphofructokinase (PFK) is a key regulatory enzyme in the anaerobic glycolysis of erythrocytes. Characteristic findings of PFK deficiency in dogs include a well-compensated hemolytic disorder with hemolytic crises and metabolic myopathy. Hemolytic crises are often associated with strenuous exercise, barking, heat, and panting.1,2 Breeds previously known to be affected by this inborn error of metabolism include English springer spaniels, American cocker spaniels, whippets, and several mixed-breed dogs.1,3–6 As with most enzymopathies, PFK is an autosomal recessively inherited disorder and a single missense mutation has been found in all previously described PFK-deficient dogs of any breed. This report describes the clinical, hematologic, and biochemical features of PFK deficiency in the Wachtelhund that appears to be caused by a different mutation than in previously described affected breeds.
Case Report
Case 1
A 2.5 yr old intact female Wachtelhund was presented in 2004 to the Strömsholm Referral Animal Hospital with anorexia, red urine, and lethargy after intensive running while hunting. On physical examination, the dog was lethargic and icteric, had a systolic heart murmur, and a body temperature of 40°C. A presumptive diagnosis of immune-mediated hemolytic anemia (IMHA) was made. The dog was treated with prednisolone and azathioprine. Clinical signs regressed gradually over the following days, and the medications were tapered over several months. Half a year later, the dog was represented with similar signs after hunting and again she was treated with prednisolone and azathioprine and recovered uneventfully. PFK deficiency was then suspected, but DNA testing for the known mutation in English springer spaniels at PennGen Laboratorya was negative.7 The owners were instructed to restrict the dog from vigorous exercise, including hunting, which diminished the recurrence of hemolytic episodes. The dog had several follow-up examinations and had, at the time of this report, no serious clinical signs according to the owners.
Case 2
In 2008, a 2 yr old intact female Wachtelhund was presented to the University Animal Hospital in Uppsala, Sweden with lethargy and red urine after running for over 6 hr while hunting moose. She had pale and icteric mucus membranes and a mild systolic heart murmur. PFK deficiency was suspected and the dog recovered uneventfully. Exercise restriction was recommended, but no drug therapy was initiated. The dog had occasional episodes of pigmenturia and lethargy during the next 2 yr and sometimes was unwilling to exercise.
Additional Testing
Both dogs described in this report had a distant common ancestor according to a six generation pedigree (Figure 1). Case 1 had one littermate and case 2 had two littermates with clinical signs of lethargy and pigmenturia after exercise. Two of these littermates had been treated with prednisolone and azathioprine for a presumed IMHA and typically recovered rapidly from the episodes of lethargy and pigmenturia. There was also one related male dog that was believed to have IMHA with frequent hemolytic crises even when receiving immunosuppressive treatment. That dog was euthanized at the age of 1.5 yr.
FIGURE 1.
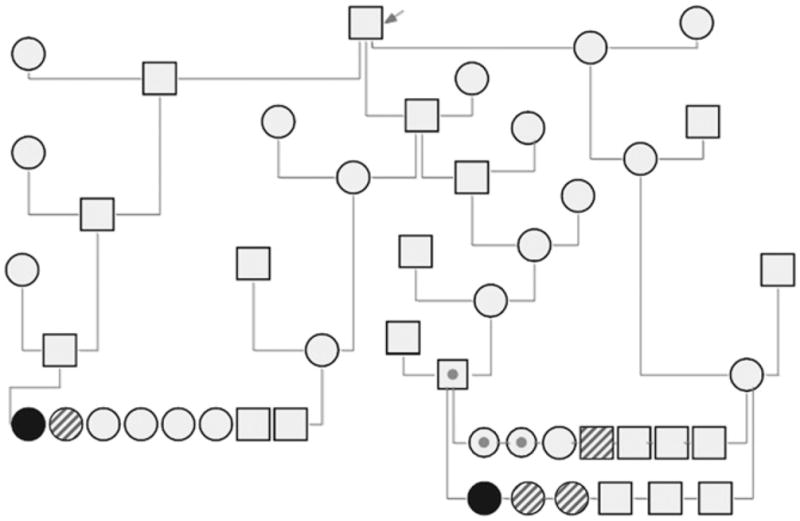
Pedigree of two PFK-deficient Wachtelhund dogs with a common ancestor (arrow). Black circle, affected female dog; shadowed circle or square, suspected PFK-deficient female or male, respectively, with hematologic findings suggesting PFK deficiency; open circle or square, untested female or male, respectively; open circle or square with a black dot, female or male, respectively, with no hematologic findings suggesting PFK deficiency.
Laboratory evaluation of cases 1 and 2, the three related dogs with signs compatible with PFK deficiency, and three related, but asymptomatic, Wachtelhunds included routine hematology, serum chemistry, and urinalysis. An impedance hematology instrumentb was used for case 1 and a laser type hematology analyzerc was used in case 2 and the six related dogs. Reticulocyte counts in case 1 were measured by manual counting whereas instrumental reticulocyte counts were used for the other seven cases. Chemistry testing was performed with routine methods on an automated analyzerd. For cases 1 and 2, analyses were performed within 4 hr from collection. The samples from the other six dogs were assessed 1 day after collection. At the time of sampling, the related dogs had no reported signs of disease.
The hematologic evaluations of case 1 (performed between 2004 and 2009) and case 2 (performed between 2008 and 2009) revealed a severe to fully compensated, highly regenerative hemolytic disorder (Table 1). Case 1 had a persistent, moderate to marked reticulocytosis, and a mild anemia. An extreme reticulocytosis of 1,650×109/L or 90% reticulocytes (reference range, <60×109/L and <0.6%) occurred during an initial severe hemolytic crisis when the hematocrit was 18% (reference range, 39–59%) (Figure 2). At another time, the reticulocyte count was 810×109/L, despite only having a mild anemia (hematocrit was 31%).
TABLE 1.
Hematologic Parameters from Two Wachtelhund Dogs with PFK Deficiency
| Red blood cell parameter | Case 1 mean (range) | Case 2 mean (range) | Reference range |
|---|---|---|---|
| Hemoglobin (g/L) | 104 (53–125) | 117 (103–137) | 120–199 |
| Hematocrit (%) | 29 (18–34) | 39 (34–44) | 39–59 |
| MCV (fL) | 82* (77–90) | 84 (82–86) | 64–75 |
| MCHC (g/L) | 339* (327–353) | 302 (287–313) | 320–363 |
| Reticulocytes (× 109/L) | 787 (206–1650) | 433 (312–489) | < 60 |
| Nucleated red blood cell (×106/L) | 660 (68–2363) | 815 (721–981) | Rare |
Mean and range of hematologic parameters from two Wachtelhund dogs with PFK deficiency measured on five (*three) different occasions
MCV, mean corpuscle volume; MCHC, mean corpuscular hemoglobin concentration; PFK, phosphofructokinase
FIGURE 2.
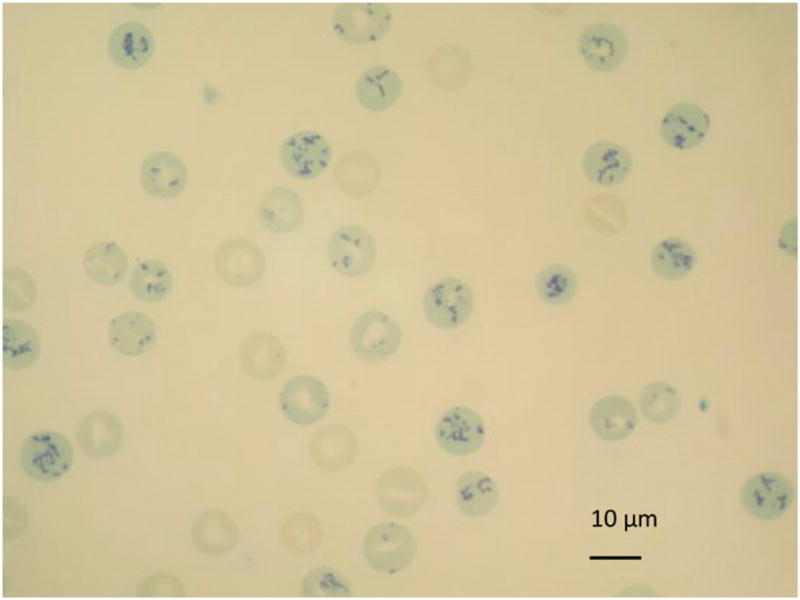
Blood smear from case 1 with 90% reticulocytes. New methylene blue, original magnification ×100, oil objective.
Case 2 had a moderate to marked reticulocytosis despite having normal to slightly low hematocrit values. Blood smear examinations from both dogs described in this report revealed evidence of regeneration such as marked polychromasia, anisocytosis, and metarubricytosis, but no signs of other types of hemolytic anemias such as spherocytes, autoagglutionation, Heinz bodies, eccentrocytes, or blood parasites were seen (Figure 2). The direct Coombs’ test was negative in case 1 and was not determined in case 2. Platelet counts were normal in case 1 except on two occasions when the platelet counts were 120×109/L and 146×109/L (reference range, 150–450×109/L) after two separate hemolytic crises. Case 2 had a mild thrombocytopenia on all five occasions that the dog was examined (platelet counts ranged from 119–137×109/L); however, mild platelet clumping was seen microscopically on three occasions.
All routine chemistry screening results were within laboratory reference limits for both dogs described in this report. The urine from case 1 was consistently strongly positive for bilirubin and intermittently positive for hemoglobin (blood) by a reagent stripe.5 Zero to only few erythrocytes in the urine sediment suggested hemoglobinuria and intravascular hemolysis. During an intravascular hemolytic crisis, the urine sediment from case 1 had hemoglobinuria with many hemoglobin droplets (Figure 3). One urine sediment from case 2 had many bilirubin-stained casts per high-power field.
FIGURE 3.
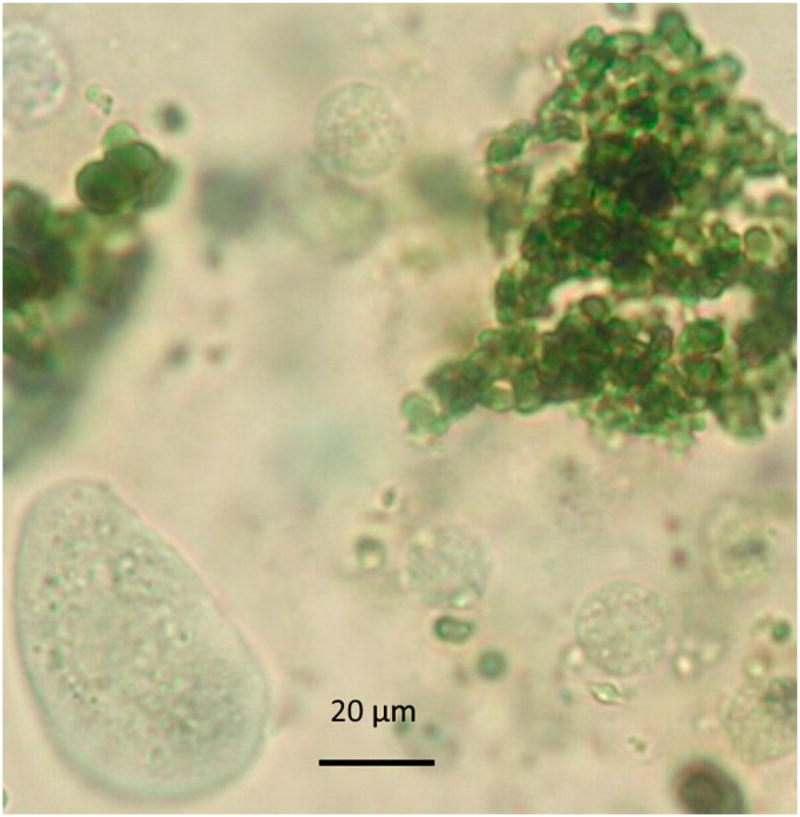
Microscopy of urine sediment from the PFK-deficient Wachtelhund described in case 2. Hemoglobin droplets appear light green when stained with new methylene blue. Original magnification × 40.
Both dogs described in this report had systolic heart murmurs on initial presentation. In case 1, echocardiography was unremarkable, and no murmur was noted on subsequent examinations when the hematocrit values were near normal. In case 2, the heart was not specifically examined by ultrasound, but serum troponin concentrations measured by an enzyme-labeled chemiluminescent immunometric assayf were repeatedly normal.
One littermate of case 1 with intermittent clinical signs of PFK deficiency had a mild anemia with marked reticulocytosis. The hemoglobin was 117 g/L (reference range, 120–199 g/L) and the reticulocyte count was 658×109/L. This littermate had mildly increased serum creatine kinase (CK, 8.7 μkat/L; reference range, <4 μkat/L) and aspartate aminotransferase activity (AST; 0.9 μkat/L; reference range, <0.7 μkat/L). Other serum biochemical results were normal.
The two littermates of case 2 with clinical signs of PFK deficiency had mildly decreased hemoglobin concentrations of 105 g/L and 138 g/L, respectively and greatly increased reticulocyte counts of 1,610×109/L and 847×109/L, respectively. The serum chemistry results were within reference limits except for hyperkalemia (11 mmol/L and 12.7 mmol/L, respectively; reference range, 4.0–5.4 mmol/L) and elevated CK activity (17.1 and 7.6 μkat/L, respectively). One of the littermates to case 2 had a mildly elevated AST (1.6 μkat/L). Serum samples from these two littermates appeared slightly hemolyzed. EDTA and serum samples from three additional related, but asymptomatic, dogs had no findings suggestive of PFK deficiency.
Erythrocyte PFK activity in cases 1 and 2 was determined at Uppsala University Hospital.8 Erythrocyte concentrations of 2,3-diphosphoglycerate (DPG) in both dogs, erythrocyte PFK activity, and the hemoglobin-oxygen (Hb-O2) dissociation curve performed in case 2, as well as all DNA studies were done at the PennGen Laboratorya as previously described.3,9,10 Erythrocyte PFK activity and DPG content were lower in cases 1 and 2 than in healthy control beagles. A marked left shift of the Hb-O2 dissociation curve indicated high Hb-O2 affinity (Table 2). Results of the polymerase chain reaction-based restriction enzyme test and sequencing around the site of the known mutation used to diagnose PFK deficiency in English springer spaniels did not reveal the known mutation in any tested Wachtelhund dog.7
TABLE 2.
Erythrocyte PFK Activity, DPG Concentration, and Hb-O2 Dissociation Curve Results in Two Wachtelhund Dogs and Healthy Controls
| Erythrocyte parameter | Case 1 | Healthy control to case 1 | Case 2 | Healthy control to case 2 |
|---|---|---|---|---|
| PFK activity* IU/g Hb (%) | 0.78 (11) | 6.8 (100) | 0.78 (12) | 6.5 (100) |
| PFK activity† IU/g Hb (%) | — | — | 1.35 (9) | 15.0 (100) |
| DPG content mmol/mL RBC (%) | 0.56 (8) | 7.14 (102) | 1.67 (24) | 7.0 (100) |
| Hb-O2 dissociation (P50)§ mm Hg | — | — | 19.6 | 28 |
Wachtelhund dogs and healthy controls were studied simultaneously (% of healthy controls).
PFK activity measured at Uppsala University Hospital
PFK activity measured at University of Pennsylvania
P50 refers to the oxygen tension at which 50% of the Hb is bound to oxygen.
DPG, 2,3-diphosphoglycerate; Hb, hemoglobin; Hb-O2, hemoglobin-oxygen; RBC, red blood cell
Discussion
This clinical report documents PFK deficiency in a family of Wachtelhunds. The hemolytic disorder was clinically very similar to that reported in English springer spaniels, but was apparently due to a different PFK mutation. PFK deficiency was confirmed in two of the Wachtelhunds based on low PFK activity, high Hb-O2 affinity, and decreased concentrations of DPG in erythrocytes. Episodes of intravascular hemolysis occurred in both dogs after prolonged running (hunting). The association of hemolytic crises with strenuous exercise is characteristic of PFK deficiency in dogs. Low erythrocyte PFK activity causes low erythrocyte DPG concentrations, which in turn results in an increased erythrocyte pH value. Canine erythrocytes are known to lyse at slightly alkaline pH values and PFK-deficient erythrocytes exhibit increased alkaline fragility.11,12 It is likely that exercise- and/or heat-induced hyperventilation caused respiratory alkalosis leading to accelerated intravascular hemolysis in the affected dogs. An excessive reticulocyte response despite a normal to only mildly reduced hematocrit is another typical feature of PFK deficiency, which was seen in these two probands and also other affected relatives. Part of the reticulocyte response is due to the constantly ongoing hemolysis of PFK-deficient erythrocytes; however, PFK-deficient dogs appear to exhibit tissue hypoxia even at normal hematocrit values. The low concentrations of DPG cause an increased Hb-O2 affinity in the PFK-deficient erythrocytes, illustrated by a left shift in the Hb-O2 dissociation curve shown in one the Wachtelhunds described in this report and previously in English springer spaniels.9
Erythrocyte PFK activity was analyzed at two different laboratories (Table 2). The absolute erythrocyte PFK activities for both normal and PFK-deficient dogs were slightly lower when measured in the Swedish laboratory than the American laboratory. These differences may be due to variation in techniques used by the two laboratories and the fact that frozen, rather than fresh, red cell lysates were analyzed at the Swedish laboratory. Most importantly, PFK activities from affected dogs were similarly reduced when compared with that of normal control dogs.
There are three isoforms of the PFK enzyme: muscle (M)-, liver-, and platelet-type.13 PFK-deficient English springer spaniels have a single missense mutation in the M-PFK gene causing truncation of the M-PFK protein, which completely abolishes M-PFK enzyme activity. Due to the expression of other isoforms of the PFK enzyme, the residual PFK activity in erythrocytes is 8–22% compared with normal, but there is no PFK activity in skeletal muscle.9,14 Despite this complete muscle enzyme deficiency, signs of myopathy are relatively mild. Severe exertional myopathies rarely occur in PFK-deficient dogs, which probably is due to the high oxidative rate of canine skeletal muscle.15 Nevertheless, the muscles of PFK-deficient dogs have reduced working capacity and may cramp, which is problematic especially in hunting breeds like the Wachtelhund and English springer spaniel.16,17 The two anemic Wachtelhund dogs in this study had signs of exercise intolerance while hunting, but did not seem to have any other signs of a metabolic myopathy such as cramping. The exercise intolerance may have been caused by their hemolytic anemia. The only other potential sign of myopathy in the Wachtelhund dogs studied in this report was a mildly increased serum CK activity in one of the littermates. Two whippets with PFK deficiency were recently reported to also have cardiac involvement, which could be due to cardiac PFK deficiency.6 Cardiac involvement has not been reported in either English springer spaniels or cocker spaniels and neither was it evident in any affected Wachtelhunds in this report.
Hyperkalemia was present in two hemolyzed samples which had been shipped to the laboratory that were collected from dogs with suspected PFK deficiency, but not in nonhemolyzed samples from the other dogs included in this report. Both of the samples with hyperkalemia had high reticulocyte counts. Thus, pseudo-hyperkalemia due to the in vitro leakage from reticulocytes was suspected. Mature canine erythrocytes (except for some Asian breeds) have potassium concentrations similar to serum, whereas reticulocytes have a higher potassium content.18 Intravascular hemolysis is unlikely to cause life-threatening hyperkalemia in PFK-deficient dogs because PFK-deficient reticulocytes are not alkaline fragile as they have DPG concentrations similar to that of normal canine erythrocytes.12 Thus, mainly mature erythrocytes not containing high intracellular potassium would be lysed. Other cell damage, such as active muscle damage, can also release intracellular potassium into the blood and hyperkalemia has been reported in PFK-deficient dogs with potassium levels up to 8.4 mmol/L.9 Serum CK activity was mildly elevated in the two hemolyzed samples as well, but hemolysis is reported to falsely increase CK activity in dogs.19
Diagnosis of PFK deficiency in Wachtelhund dogs is currently complicated by the fact that the DNA test developed for English springer spaniels, which also identifies PFK deficiency in American cocker spaniels, whippets and mixed-breed dogs, did not reveal the same mutation and thus cannot be applied to affected Wachtelhunds. This suggests that a different mutation in the PFK gene is causing PFK deficiency in Wachtelhunds. Sequencing of the entire PFK cDNA or genomic DNA would be required to discover this breed specific mutation. English springer spaniels and cocker spaniels are closely related breeds, and it was surprising to find affected whippets to have the same PFK mutation, suggesting a very ancestral mutation rather than the same mutation occurring twice.6 The Wachtelhund is not closely related to the previously reported PFK-deficient breeds. Thus, PFK deficiency may well have been caused by a different mutation. Indeed, there are many PFK mutations described in humans.20 Similarly several different mutations in the red cell pyruvate kinase (R-PK) gene have been identified to cause pyruvate kinase deficiency in different dog breeds.1 In dogs, such mutations are typically breed specific, and it is likely that all affected Wachtelhunds have the same mutation. A genetic test capable of identifying the mutant gene in PFK-deficient Wachtelhund dogs would simplify diagnosis because currently, the diagnosis of PFK deficiency in Wachtelhunds requires measuring PFK activity in purified erythrocytes in a specialized laboratory. A DNA test would also make it possible to identify carriers (heterozygotes), which have no clinical signs and routine laboratory test abnormalities, but can pass on the mutant allele to their offspring. Molecular genetic testing could limit the further spread of this disease in this breed and thus would be highly desirable. In the absence of a specific genetic test, laboratory evidence of an excessive reticulocyte response compared with the severity of the anemia together with hemoglobinuria after strenuous exercise could be used to identify likely affected dogs.
Conclusion
The frequency of PFK deficiency in the Wachtelhund dog breed is currently unknown, but there are likely more affected dogs than currently suspected. The two confirmed cases described in this report were not closely related to each other, and three littermates of these two cases also seemed to be affected. The Wachtelhund dog is gaining popularity in Sweden, with 517 and 530 puppies registered by the Swedish Kennel Club in 2008 and 2009, respectively. These versatile dogs are used for many different types of hunting, which includes seeking and chasing animals like roe deer, moose, and wild pigs over short to moderate distances. This intense exercise often lasts several hours and causes dogs to pant and become hyperthermic, both of which can trigger a hemolytic crisis in affected animals. Signs of severe hemolytic anemia likely obscure any evidence of the concurrent exertional myopathy. Fortunately, hemolytic crises can be avoided if excessive exercise, heat, panting, and barking are avoided, and affected dogs may nearly reach their expected lifespan for the breed. It is not recommended to hunt with affected dogs. In contrast, PFK carriers are clinically unaffected and can hunt, but may pass the mutant allele to their offspring.
Acknowledgments
The authors thank Gunnar Ronquist and Gunilla Frenne for performing the PFK analyzes at the Uppsala University Hospital and Osheiza Abdulmalik and Angela Erat-Huff for the laboratory studies performed at the University of Pennsylvania. The authors would also like to thank the laboratory technicians and veterinary staff for hematology testing and advice at Strömsholm and University Animal Hospital, Swedish University of Agricultural Sciences (SLU). This study was supported in part by NIH RR02512.
Glossary
- AST
aspartate aminotransferase
- CK
creatine kinase
- DPG
2,3-di-phosphoglycerate
- Hb-O2
hemoglobin-oxygen
- IMHA
immune-mediated hemolytic anemia
- M-PFK
muscle-type phosphofructokinase
- PFK
phosphofructokinase
Footnotes
PennGen Laboratory; University of Pennsylvania, Philadelphia, PA
Medonic CA 620; Boule Medical AB, Stockholm, Sweden
Advia 2120; Siemens Health Care Diagnostics, Deerfield, IL
Konelab 30; Thermo Fisher Scientific Inc, Waltham, MA
Combur Test D; Roche Diangostics Ltd., Sussex, UK
Immulite 2000 Troponin I; Siemens Medical Solutions Diagnostics, Los Angeles, CA
References
- 1.Giger U. Erythrocyte phosphofructokinase and pyruvate kinase deficiencies. In: Feldman BF, Zinkel JG, Jain NC, editors. Schalm’s veterinary hematology. 5. Baltimore (MD): Lippincott Williams & Wilkins; 2000. pp. 1020–5. [Google Scholar]
- 2.Harvey JW. Pathogenesis, laboratory diagnosis, and clinical implications of erythrocyte enzyme deficiencies in dogs, cats, and horses. Vet Clin Pathol. 2006;35(2):144–56. doi: 10.1111/j.1939-165x.2006.tb00108.x. [DOI] [PubMed] [Google Scholar]
- 3.Giger U, Reilly MP, Asakura T, et al. Autosomal recessive inherited phosphofructokinase deficiency in English springer spaniel dogs. Anim Genet. 1986;17(1):15–23. doi: 10.1111/j.1365-2052.1986.tb03184.x. [DOI] [PubMed] [Google Scholar]
- 4.Skibild E, Dahlgaard K, Rajpurohit Y, et al. Haemolytic anaemia and exercise intolerance due to phosphofructokinase deficiency in related springer spaniels. J Small Anim Pract. 2001;42(6):298–300. doi: 10.1111/j.1748-5827.2001.tb02043.x. [DOI] [PubMed] [Google Scholar]
- 5.Giger U, Smith BF, Woods CB, et al. Inherited phosphofructokinase deficiency in an American cocker spaniel. J Am Vet Med Assoc. 1992;201(10):1569–71. [PubMed] [Google Scholar]
- 6.Gerber K, Harvey JW, D’Agorne S, et al. Hemolysis, myopathy, and cardiac disease associated with hereditary phosphofructokinase deficiency in two Whippets. Vet Clin Pathol. 2009;38(1):46–51. doi: 10.1111/j.1939-165X.2008.00089.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Smith BF, Stedman H, Rajpurohit Y, et al. Molecular basis of canine muscle type phosphofructokinase deficiency. J Biol Chem. 1996;271(33):20070–4. doi: 10.1074/jbc.271.33.20070. [DOI] [PubMed] [Google Scholar]
- 8.Ronquist G, Rudolphi O, Engström I, et al. Familial phosphofructokinase deficiency is associated with a disturbed calcium homeostasis in erythrocytes. J Intern Med. 2001;249(1):85–95. doi: 10.1046/j.1365-2796.2001.00780.x. [DOI] [PubMed] [Google Scholar]
- 9.Giger U, Harvey JW. Hemolysis caused by phosphofructokinase deficiency in English springer spaniels: seven cases (1983–1986) J Am Vet Med Assoc. 1987;191(4):453–9. [PubMed] [Google Scholar]
- 10.Beutler E. Red cell metabolism: a manual of biochemical methods. 3. Orlando (FL): Grune & Stratton; 1984. pp. 42–5.pp. 146–8. [Google Scholar]
- 11.Giger U, Harvey JW, Yamaguchi RA, et al. Inherited phosphofructokinase deficiency in dogs with hyperventilation-induced hemolysis: increased in vitro and in vivo alkaline fragility of erythrocytes. Blood. 1985;65(2):345–51. [PubMed] [Google Scholar]
- 12.Harvey JW, Sussman WA, Pate MG. Effect of 2,3-diphosphogly-cerate concentration on the alkaline fragility of phosphofructokinase-deficient canine erythrocytes. Comp Biochem Physiol B. 1988;89(1):105–9. doi: 10.1016/0305-0491(88)90269-6. [DOI] [PubMed] [Google Scholar]
- 13.Vora S, Giger U, Turchen S, et al. Characterization of the enzymatic lesion in inherited phosphofructokinase deficiency in the dog: an animal analogue of human glycogen storage disease type VII. Proc Natl Acad Sci USA. 1985;82(23):8109–13. doi: 10.1073/pnas.82.23.8109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Giger U, Kelly AM, Teno PS. Biochemical studies of canine muscle phosphofructokinase deficiency. Enzyme. 1988;40(1):25–9. doi: 10.1159/000469137. [DOI] [PubMed] [Google Scholar]
- 15.Snow DH, Billeter R, Mascarello F, et al. No classical type IIB fibres in dog skeletal muscle. Histochemistry. 1982;75(1):53–65. doi: 10.1007/BF00492533. [DOI] [PubMed] [Google Scholar]
- 16.Giger U, Argov Z, Schnall M, et al. Metabolic myopathy in canine muscle-type phosphofructokinase deficiency. Muscle Nerve. 1988;11(12):1260–5. doi: 10.1002/mus.880111210. [DOI] [PubMed] [Google Scholar]
- 17.Brechue WF, Gropp KE, Ameredes BT, et al. Metabolic and work capacity of skeletal muscle of PFK-deficient dogs studied in situ. J Appl Physiol. 1994;77(5):2456–67. doi: 10.1152/jappl.1994.77.5.2456. [DOI] [PubMed] [Google Scholar]
- 18.Kirk RG, Andrews SB, Lee P. The correlation of composition and morphology during the high to low potassium transition in single erythropoietic cells. J Membr Biol. 1983;76(3):281–7. doi: 10.1007/BF01870370. [DOI] [PubMed] [Google Scholar]
- 19.O’Neill SL, Feldman BF. Hemolysis as a factor in clinical chemistry and hematology of the dog. Vet Clin Pathol. 1989;18(3):58–68. doi: 10.1111/j.1939-165x.1989.tb00519.x. [DOI] [PubMed] [Google Scholar]
- 20.Toscano A, Musumeci O. Tarui disease and distal glycogenoses: clinical and genetic update. Acta Myol. 2007;26(2):105–7. [PMC free article] [PubMed] [Google Scholar]


