Abstract
Background:
Evaluate the adjunctive use of locally delivered green tea catechin with scaling and root planing, as compared to scaling and root planing alone in the management of chronic periodontitis.
Materials and Methods:
Fourteen patients with two sites in the contralateral quadrants with probing pocket depth of 5–8mm were selected. Each of the sites was assessed for the plaque index, gingival index, and probing pocket depth at baseline and 21 days and for microbiological analysis at baseline, 1 week and 21 days. Test sites received scaling and root planing along with green tea catechin strips and control sites received scaling and root planning alone.
Results:
The result showed intercomparison of the plaque index and gingival index for test and control groups at 21 days was not significant with P>0.05, whereas the probing depth at 21 days was significant with P<0.001. Intercomparison between microbial results demonstrated a considerable reduction of occurrence of Aggregatibacter actinomycetemcomitans, Prevotella intermedia, Fusobacterium species and Capnocytophaga in test.
Conclusion:
Green tea catechin local delivery along with scaling and root planing is more effective than scaling and root planing alone.
Keywords: Chronic periodontitis, green tea catechin, local drug delivery, root planing, scaling
INTRODUCTION
Green tea is one of the most popularly consumed beverage worldwide contains a number of bioactive chemicals, it is particularly rich in flavonoids which include catechins (flavan-2-ols) such as epicatechin (EC), epicatechin-3-gallate (ECG), epigallocatechin (EGC), and epigallocatechin-3-gallate (EGCG). Green tea also contains carotenoids, tocopherols, ascorbic acid (vitamin C), minerals such as Cr, Mn, Se or Zn, and certain phytochemical compounds.[1,2] The most abundant polyphenolic compound epigallocatechin gallate (EGCG) is thought to contribute to the beneficial effects of tea.[3]
Numerous studies in a variety of experimental animal models have demonstrated that catechin possess antioxidant, antimutagenic, antidiabetic, anti-inflammatory, antibacterial and antiviral, and above all, cancer-preventive properties.[1–3]
Periodontal diseases are considered as infections affecting the periodontium because there is a bacterial etiology, an immune response and tissue destruction.[4] Green tea showed antimicrobial effects against black pigmented Gram-negative anerobic rods and its catechin also inhibited collagenase activity, thus limiting tissue destruction.[5]
Green tea catechin (EGCG) significantly reduced the expression of matrix metalloproteinase-9 in osteoblasts and also inhibits the formation of osteoclasts. Thus, EGCG may prevent alveolar bone resorption that occurs in periodontal disease.[6] Another study showed an inhibitory effect of green tea catechins on the adherence of Porphyromonas gingivalis onto the buccal epithelial cells.[7]
The aim of the present study is to evaluate the therapeutic effect of scaling and root planing along with the adjunctive use of locally delivered green tea catechin, as compared to scaling and root planing alone in patients with chronic periodontitis.
MATERIALS AND METHODS
Study design
Fourteen patients (Both males and females, age range 30–55 years) were selected from the outpatients who visited the Department of Periodontics, Jaipur Dental College, Jaipur.
The nature and design of the clinical trial was explained to the patients and written consent was obtained for their participation. A split mouth design was followed, where two sites in the contralateral quadrants with probing pocket depth of 5–8 mm at baseline were chosen.
Patients diagnosed to have chronic periodontitis with pockets of 5–8 mm depth bilaterally, who had not undergone any form of non surgical or surgical periodontal therapy in the last 6 months and who have not received any topical or systemic antimicrobial treatment for the past 6 months were included in the study. Exclusion criteria included patients having systemic diseases, pregnant and lactating mothers, smokers, and patients with less than 20 scorable teeth.
Selected sites were divided randomly under test group and control group. Test sites received scaling and root planing along with Green tea catechin and control sites received scaling and root planning alone.
Each of the sites (both Test and Control) were assessed for plaque index (Silness and Loe, 1967), gingival index ( Loe and Silness, 1963) and probing pocket depth using UNC 15 probe at baseline and twenty one days.
Microbiological samples were collected using a sterile Gracey Curette by inserting it subgingivally into the deepest portion of the periodontal pocket and moved coronally by scraping along the root surface. The samples were then transferred to a bottle containing thioglycollate transport media. The procedure was again repeated after one week and at twenty one days. The periodontal pathogens microbiologically assessed were Porphyromonas gingivalis, Prevotella intermedia, Aggregatibacter actinomycetemcomitas, Fusibacterium speciesand Capnocytophaga species.
Green tea catechin
Green tea catechin strips were manufactured by Department of Pharmaceutics, KLES College of pharmacy, Hubli, Karnataka. Strips are transparent rectangular shaped (size- 2 mm in width, length- 4 mm and 0.3 mm in thickness) with catechin used from green tea powder and hydroxypropyl cellulose (HPC) as the carrier. Gamma radiation (dose of 5 kGy) was used to sterilize the strips [Figure 1].
Figure 1.
Green tea catechin chips
Application of local drug delivery
After the clinical parameters were recorded, full mouth scaling was done using piezoelectric ultrasonic scalers. Root planing was performed at the test site and control site using area specific Gracey curettes. Then the area was isolated with cotton rolls and dried. Green tea catechin chip was grasped with tweezer and inserted deep in the periodontal pocket so that the chip rested subgingivally at the base of the pocket, and not exposed [Figure 2]. The patients were then instructed not to use any other chemical plaque control methods other than normal brushing and rinsing. The patient was informed to report to the clinic in case of any irritation and discomfort.
Figure 2.
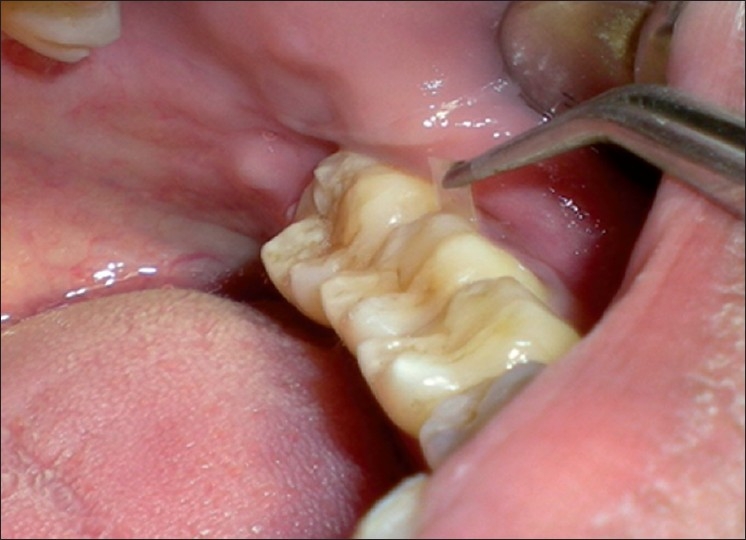
Placement of green tea catechin chip
Statistical analyses were performed by applying the mean, standard deviation (S.D.), Student's t-test (paired and unpaired).
RESULT
The present study was conducted to assess the clinical and efficacy of Green tea catechin (Camellia sinensis) local drug delivery system as an adjunct to scaling and root planing in the treatment of patients with chronic periodontitis. The comparisons of the clinical parameters were done between control and test group at baseline and 21 days.
Plaque index
The mean plaque index in control group at baseline was 2.25±0.53 and the mean plaque index for test group was 2.11±0.55 [Table 1].
Table 1.
Mean±SD in plaque index from baseline to different intervals of control and test group
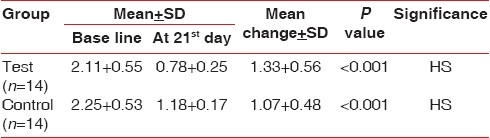
The result showed statistically highly significant difference in plaque index at 21 days in the control group as well as in the test group when compared to baseline [Table 1].
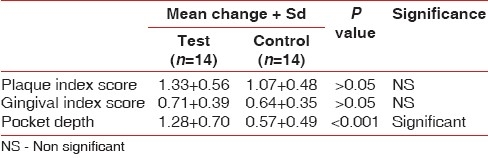
The intercomparison of the plaque index for test and control group at 21 days was not significant with P>0.05 [Table 2].
Table 2.
Mean change + SD of various parameters of test and control group patients
Gingival index
The mean gingival index score for the control group at baseline was 1.75 0.40 and for the test group was 1.67±0.37 [Table 3].
Table 3.
Mean±SD in Gingival index from baseline to different intervals of control and test group
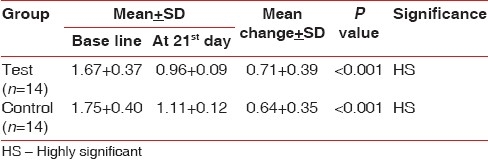
The result depicted the mean change in the gingival index at 21 days to be highly significant in control and test group compared to the baseline [Table 3].
The intercomparison of the gingival index for test and control groups at 21 days was not significant with P>0.05 [Table 2].
Probing depth
The mean probing depth of the control group at baseline was 5.71±0.45 and the mean probing depth of the test group was 6.43±0.49 [Table 4].
Table 4.
Mean±SD in probing depth from baseline to different intervals of control and test group

The result showed the mean change in the probing depth at 21 days was highly significant in control as well as in test groups compared to the baseline [Table 4].
The intercomparison of the probing depth for test and control group at 21 days was significant with P<0.001 [Table 2].
Microbiological analysis
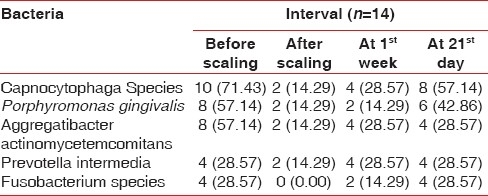
When the microbiological scores were compared for the Porphyromonas gingivalis in the control group it showed no percentage difference from base line at 1 week. It showed 28.57% difference from base line to 21 days and from 1 week to 21 days [Table 5].
Table 5.
Distribution of type of bacteria presents at various time interval in the control group patients
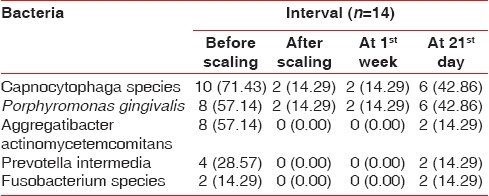
The comparison of Porphyromonas gingivalis values for test group showed no difference at base line to 1 week and 28.57% level at base line to 21 days. Same difference of 28.57% at 1 week to 21 days was seen [Table 6].
Table 6.
Distribution of type of bacteria presents at various time interval in the test group patients
There was same percentage difference in occurrence of Prevotella intermedia values in the control group at base line to 1 week and base line to 21 days; it was 14.28%. There was no difference at 1 week to 21 days [Table 5].
The percentage difference in occurrence of Prevotella intermedia values in the test group at base line to 1 week was nil, whereas it was 14.29% from base line to 21 days and from 1 week to 21 days [Table 6].
The percentage difference in occurrence of Aggregatibacter actinomycetemcomitans values in the control group was 14.28% from base line to 1 week and from base line to 21 days and no difference at 1 week to 21 days [Table 5].
The percentage difference in occurrence of Aggregatibcter actinomycetemcomitans values in the test group was 14.29% at baseline to 21 days and at 1 week to 21 days, whereas no difference was observed at baseline to 1 week [Table 6].
The percentage difference in occurrence of Capnocytophaga organism value in the control group at 1 week to baseline was 14.28%, and at 21 days to baseline was 42.85%. There was 28.57% difference at 21days to 1 week [Table 5].
The percentage difference in occurrence of Capnocytophaga organism value in the test group was 28.55% at 21 days to baseline and at 21 days to 1 week. There was no difference in percentage value at 1 week to base line [Table 6].
Percentage difference of occurrence of Fusobacterium species in control group at 1week to base line was 14.29%, at 21 days to baseline was 28.57%, and at 21 days to 1 week was 14.28% [Table 5].
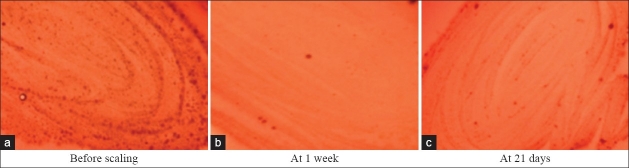
The percentage difference of occurrence of Fusobacterium species in test group at 21days to base line and 21 days to 1 week was 14.29%, whereas there was no difference at 1 week to baseline value [Table 6, Figure 3].
Figure 3.
Fusobacterium colonies in supplemented blood agar
DISCUSSION
Numerous health benefits of green tea and its constituents have been reported. Green tea is a powerful antioxidant and has anti-inflammatory properties.[8]
Treatment of chronic periodontitis is routinely based on oral hygiene procedures and root debridement, which reduces inflammation, pocket depth, and increases clinical attachment level. Mechanical debridement also disturbs the biofilm and reduces the bacterial load. Elimination or adequate suppression of putative periodontopathogenic microorganisms in the subgingival microbiota is essential for periodontal healing.[9]
Locally applied antimicrobial agents, primarily in the form of oral rinses have been used for many years as adjuncts to plaque control in periodontal therapy. Since oral rinses and irrigation at the gingival margin do not reach subgingival areas on a predictable basis, local delivery to the subgingival compartment requires alternative approaches.[10] Local delivery has the advantage over systemic antibiotics in providing highly concentrated drug delivery, improved compliance and fewer propensities for development of bacterial resistance.[11]
The antimicrobial effects of green tea catechins on periodon-topathogens have been studied by Hirasawa et al.[5] They showed bactericidal activity against black pigmented organism at a minimum inhibitory concentration (MIC) of 1 mg/ml. Therefore, the concentration of green tea catechins used in the chips prepared was decided to be maintained at 1 mg/ml, and was prepared by the Department of Pharmaceutics, KLES College of pharmacy, Hubli, Karnataka.
The present study was carried out with the objective to evaluate the efficacy of green tea catechin containing local delivery system over scaling and root planing for a period of 21 days.
The clinical parameter was recorded at baseline and 21 days as the flora is supposedly said to return to pretreatment patterns following 3 weeks of scaling and root planning.[12–14]
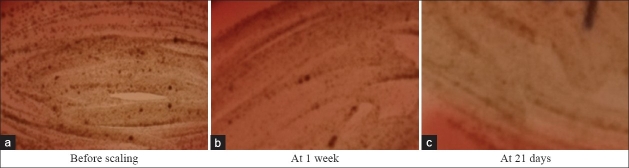
Highly significant reduction in the gingival index and plaque index scores were seen in both the control and test groups from baseline to 21 days. There was also highly significant reduction of 0.57±0.49 mm in probing pocket depth in the control group and of 1.28±0.70 mm in test group from baseline to 21days [Figures 4 and 5]. Probing depth value when compared between the test and control group showed a statistically significant difference [Figure 6]. The results are in accordance with the study conducted by Hirasawa et al.[5], which demonstrated a clinical and microbial improvement following insertion of green tea catechin using a slow release local delivery system when compared to subgingival debridement alone. A study by Kushiyama et al.[15] showed that the intake of green tea was inversely correlated with the mean probing depth, mean clinical attachment level, and bleeding on probing. Also, Okamoto et al.[16] in 2004 has suggested that the green tea catechin may have the potential to reduce periodontal breakdown resulting from the potent proteinase activity of Porphyromonas gingivalis. Green tea catechin (EGCG) inhibits the expression of gelatinse (MMP-9 RNA), and the formation of osteoclasts, thereby reduces the alveolar bone resorption that occurs in periodontal disease [Figure 7].[6]
Figure 4.
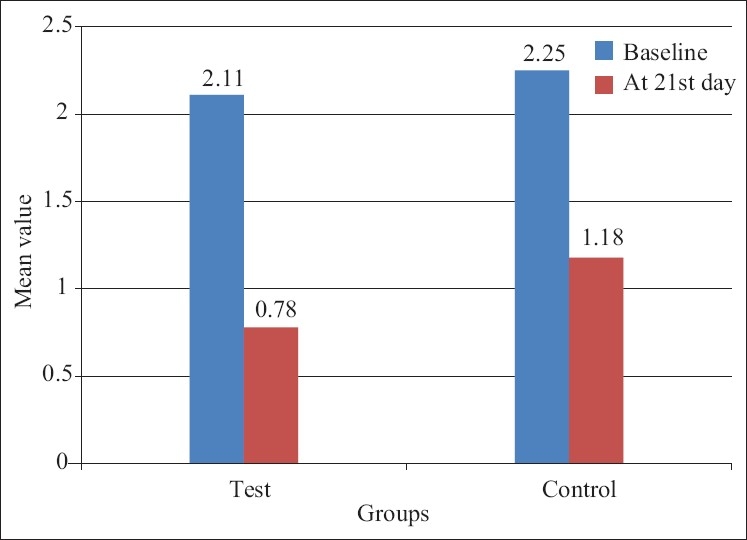
Mean+SD of plaque index score of test and control group patients
Figure 5.
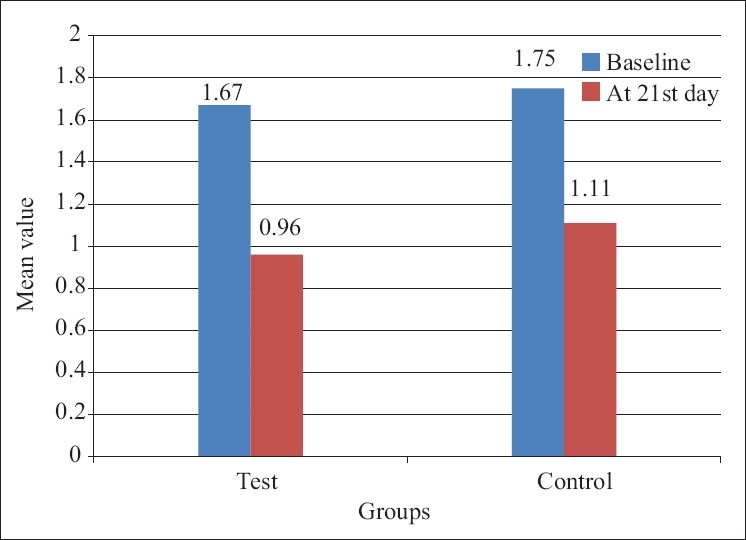
Mean+SD of gingival index score of test and control group patients
Figure 6.
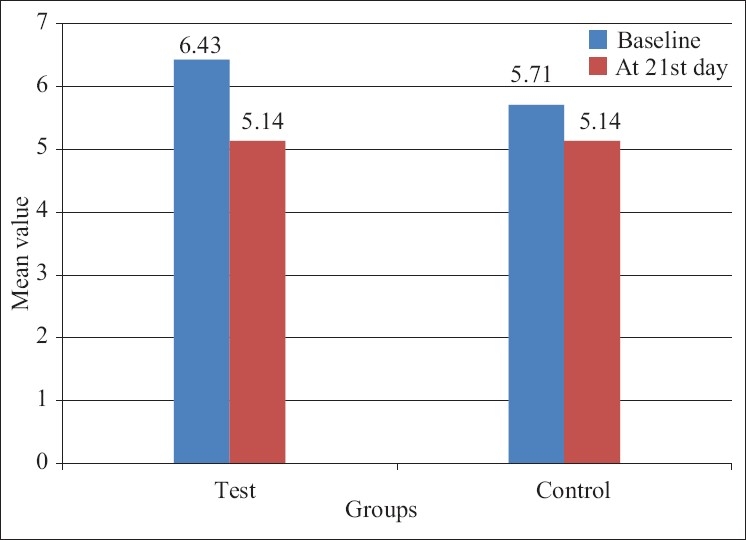
Mean+SD of pocket depth score of test and control group patients
Figure 7.
Porphyromonas gingivalis colonies in supplemented Blood agar
Scaling and root planing is accompanied by histological alteration like diminution in size of inflammatory infiltrate and laying down of new collagen. These alterations may be responsible for reduced probing pocket depth and improvement of clinical parameters.[14]
Studies by Cobb CM in 1996 and Bruce et al.[12,17] demonstrated that mechanical treatment, i.e., scaling and root planing are effective in altering the flora. Green tea catechin has been shown to be effective in altering the flora and acting as adjunct to scaling and root planing.
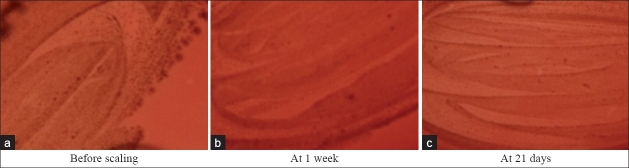
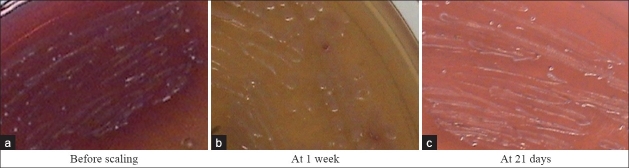
The intercomparison of microbiological results obtained between control and test sites showed a higher reduction in occurrence of Aggregatibacter actinomycetemcomitans, Fusobacterium species, Capnocytophaga species, and Prevotella intermedia in test group [Figures 8–10]. These results are in accordance with study done by Hirasawa et al.[5] on the antimicrobial effect of green tea catechin on periodontopathogens. They showed bactericidal activity against black pigmented organism at an MIC of 1 mg/ml. A study by Okamoto et al.[18] reported that epigallocatechin gallate (EGCG) and its derivative inhibit protein-tyrosine phosphatase activity in Prevotella intermedia. Intercomparison of microbiological results showed reduction of P. gingivalis in both groups at 21 days as compared to baseline with no significant reduction between the two groups. This is contradictory to the study done by Sakanaka et al.[7] who found that green tea polyphenols inhibit the growth and adherence of Porphyromonas gingivalis on to human oral epithelial surface at concentration of 250–1000 μg/ml [Figures 11 and 12].
Figure 8.
Prevotella intermedia colonies in supplemented Blood agar
Figure 10.
Capnocytophaga colonies in TSBV agar
Figure 11.
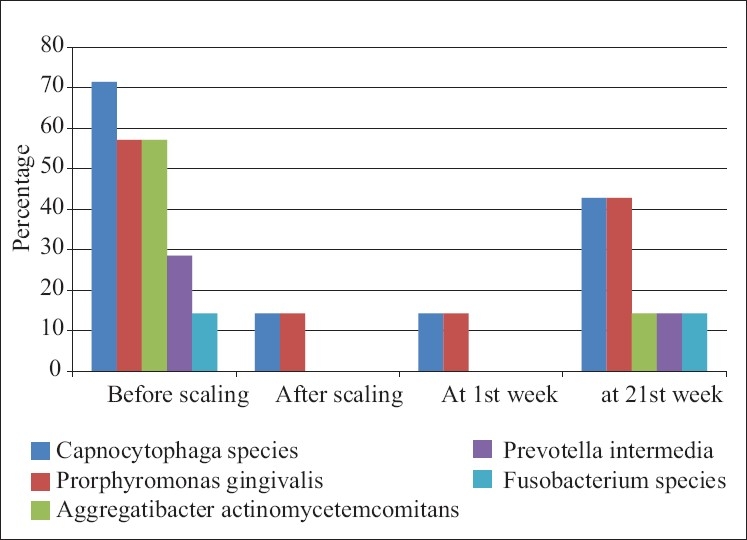
Distribution of type of bacteria presents at various time interval in the test group patients
Figure 12.
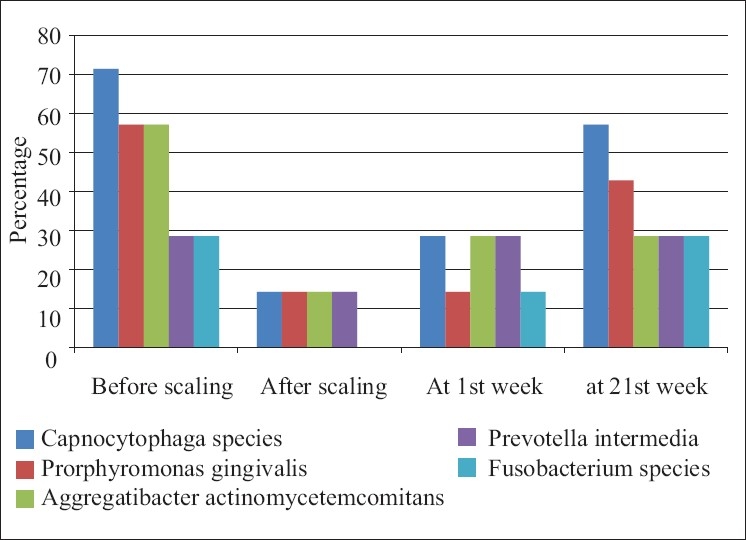
Distribution of type of bacteria presents at various time interval in the control group patients
Figure 9.
Aggregatibacter actinomycetem comitans in Dentaid media
As evident from this study, the results demonstrate clinical and microbiological improvements following the use of Green tea catechin chip as an adjunct to scaling and root planing.
Further research with relatively large sample size, longer follow-up period, and use of advanced periodontal probes are advocated to further study the efficacy of green tea as an effective local drug delivery agent in periodontal therap
Acknowledgments
This study was supported by the Department of Periodontics, Jaipur Dental College.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- 1.Mckey DL, Blumberg JB. The role of tea in human health: An update. J Am Coll Nutr. 2002;21:1–13. doi: 10.1080/07315724.2002.10719187. [DOI] [PubMed] [Google Scholar]
- 2.Wiseman SA, Balentine DA, Frei B. Antioxidants in tea. Crit Rev Food Sci Nutr. 1997;37:705–18. doi: 10.1080/10408399709527798. [DOI] [PubMed] [Google Scholar]
- 3.Higdon JV, Frei B. Tea catechins and polyphenols: Health effects, metabolism and antioxidant effects. Crit Rev Food Sci Nutr. 2003;43:89–143. doi: 10.1080/10408690390826464. [DOI] [PubMed] [Google Scholar]
- 4.Greenstein G, Polson A. The role of local drug delivery in the management of periodontal diseases: A comprehensive review. J Periodontol. 1998;69:507–20. doi: 10.1902/jop.1998.69.5.507. [DOI] [PubMed] [Google Scholar]
- 5.Hirasawa M, Takada K, Makimura M, Otake S. Improvement of periodontal status by green tea catechin using a local delivery system: A clinical pilot study. J Periodontal Res. 2002;37:433–8. doi: 10.1034/j.1600-0765.2002.01640.x. [DOI] [PubMed] [Google Scholar]
- 6.Yun JH, Pang EK, Kim CS, Yoo YJ, Cho KS, Chai JK. Inhibitory effects of green tea polyphenol (-)-epigallocatechin gallate on the expression of matrix metalloproteinase-9 and on the formation of osteoclasts. J Periodontal Res. 2004;39:300–7. doi: 10.1111/j.1600-0765.2004.00743.x. [DOI] [PubMed] [Google Scholar]
- 7.Sakanaka S, Aiwaza M, Kim M, Yamamoto T. Inhibitory effects of green tea polyphenols on growth and cellular adherence of an oral bacterium, Porphyromonas gingivalis. Biosci Biotechnol Biochem. 1996;60:745–9. doi: 10.1271/bbb.60.745. [DOI] [PubMed] [Google Scholar]
- 8.Cabrera C, Artacho R, Giménez R. Beneficial effects of green tea: A review. J Am Coll Nutr. 2006;25:79–99. doi: 10.1080/07315724.2006.10719518. [DOI] [PubMed] [Google Scholar]
- 9.Petersilka GJ, Ehmke B, Flemmig TF. Antimicrobial effects of mechanical debridement. Periodontol 2000. 2002;28:56–71. doi: 10.1034/j.1600-0757.2002.280103.x. [DOI] [PubMed] [Google Scholar]
- 10.Kornman KS. Controlled-release local delivery antimicrobials in periodontics: Prospects for future. J Periodontol. 1993;64:782–91. doi: 10.1902/jop.1993.64.8s.782. [DOI] [PubMed] [Google Scholar]
- 11.Wilson TJ, Kornman KS. 2nd ed. Chicago: Quintessence publishing; 2003. Fundamentals on periodontics. [Google Scholar]
- 12.Bruce LP, William FA. American academy of periodontology: Treatment of gingivitis and periodontitis (Position paper) J Periodontol. 1997;68:1246–53. [PubMed] [Google Scholar]
- 13.Roxane AL, Greenstein G. Clinical and microbiological response to nonsurgical mechanical periodontal therapy. Periodontol 2000. 1995;9:14–22. doi: 10.1111/j.1600-0757.1995.tb00052.x. [DOI] [PubMed] [Google Scholar]
- 14.Mousques T, Listgarten MA, Phillips RW. Effect of scaling and root planing on the compostion of the human sublingual microbial flora. J Periodontal Res. 1980;15:144–51. doi: 10.1111/j.1600-0765.1980.tb00268.x. [DOI] [PubMed] [Google Scholar]
- 15.Kushiyama M, Shimazaki Y, Murakami M, Yamashita Y. Relationship between intake of green tea and periodontal disease. J Periodontol. 2009;80:372–7. doi: 10.1902/jop.2009.080510. [DOI] [PubMed] [Google Scholar]
- 16.Okamoto M, Sugimoto A, Leung KP, Nakayama K, Kamaguchi A, Maeda N. Inhibitory effect of green tea catechin on cysteinases in Porphyromonas gingivalis. Oral Microbiol Immunol. 2004;19:118–20. doi: 10.1046/j.0902-0055.2003.00112.x. [DOI] [PubMed] [Google Scholar]
- 17.Cobb CM. Non surgical pocket theraphy: Mechanical. Ann Periodontol. 1996;1:443–90. doi: 10.1902/annals.1996.1.1.443. [DOI] [PubMed] [Google Scholar]
- 18.Okamoto M, Takano K, Maeda N. Distribution of the tetracycline resitance determinant tetQ gene in oral isolates of black pigmented anaerobes in Japan. Oral Microbiol Immunol. 2001;16:224–8. doi: 10.1034/j.1399-302x.2001.160405.x. [DOI] [PubMed] [Google Scholar]