Abstract
Abnormal innate immune response contributes to inflammatory bowel disease (IBD) and experimental mouse colitis. Colitis studies have focused primarily on key regulators of innate immunity, like pathogen recognition receptors and cytoplasmic mediators. Extracellular matrix (ECM) proteins are emerging as modulators of inflammatory responses by virtue of their interactions with pathogen associated molecular patterns (PAMPs), cytokines, growth factors, receptors, and ECM fragments that mimic pathogens or cytokines. The ECM proteins have not been investigated in IBD at great depth from this standpoint. We have shown previously that the ECM protein lumican modulates host sensing of bacterial lipopolysaccharides (LPS) by toll like receptor (TLR) 4, and neutrophil chemotaxis via integrins. Here we investigated the role of lumican in the development of colitis mediated by intra-rectal administration of the hapten 2-4-5, trinitrobenzene sulfonic acid (TNBS) in Lum+/+ and Lum−/− mice. The TNBS-treated Lum+/+ mouse colons showed marked increases in CXCL1, TNF-α and neutrophil infiltration; while these responses were significantly dampened in the Lum−/− mice. The NF-κB transcription factor, known to regulate inflammatory genes, showed a robust increase after TNBS treatment in Lum+/+ but not in Lum−/− colons. Also, nuclear translocation of NF-κB was delayed in LPS-stimulated Lum−/− primary peritoneal macrophages. Thus, the Lum−/− mice have low innate immune and inflammatory responses, but more severe body weight loss and tissue damage, a phenomenon seen in the innate immune impaired Tlr4−/− and MyD88−/− mice. Therefore, lumican promotes intestinal homeostasis by aiding innate immune and inflammatory responses that are beneficial in the early stages of colitis.
Keywords: Extracellular matrix, lumican, TLR4, CD14, innate immune response, inflammation, murine colitis, neutrophils, tri-nitrobenzene sulfonic acid, dextran sodium sulfate
Introduction
Inflammatory bowel disease (IBD), Crohn’s disease and ulcerative colitis are chronic inflammatory diseases with certain common inflammatory changes but distinct etiology and pathogenesis (1, 2). In Crohn’s disease, transmural damage can affect any segment of the gastrointestinal tract, while ulcerative colitis manifests mucosal damage of the colon. It is now generally recognized that abnormal mucosal immune response to the commensal flora plays a major role in the etiopathology of IBD (3). In agreement with this view, IBD has been associated with changes in the toll-like receptors (TLR) and the nucleotide-binding oligomerization domain containing (Nod) pathogen recognition receptors (PRR) at the cell surface and cytoplasm, respectively (4, 5). Innate immune response is the first response of host cells to pathogen associated molecular patterns (PAMPs), through TLRs and the Nod like receptors (6). Activations of these PRR ultimately lead to degradation of cytoplasmic inhibitors of NF-κB (IκB), nuclear translocation and activation of NF-κB and up regulation of pro-inflammatory cytokine genes (7, 8). Studies investigating innate immune response have focused largely on these cell surface and cytoplasmic regulators. However, the magnitude of innate immune response is modulated by a number of other factors beyond the specific PAMPs and their host receptors. A few studies have identified extracellular matrix (ECM) components and their degradation products as having the potential to modulate innate immune response either by mimicking PAMPs, or by interacting with PAMPs or their receptors. For example, hyaluronan, an ECM glycosaminoglycan (9), and biglycan, an ECM protein, stimulate TLR 2 and 4 mediated innate immune response (10). Mindin, an ECM protein of the spondin family, recognizes PAMPs and modulates recruitment of inflammatory cells (11). However, such modulations of innate immune response by non-immune components have not been investigated in IBD.
In a previous study, we developed a mouse model of chronic colitis by administering low doses of 2-4-5, trinitrobenzene sulfonic acid (TNBS) as weekly enemas (12). The TNBS colitis model develops transmural colonic inflammation and tissue damage as seen in Crohn’s colitis. We previously investigated gene expression patterns at the early and late stages of this model. As expected, during the early stage (first six weeks) of active inflammation, several pro-inflammatory and innate immune response genes, such as Il1b, Cxcl1, several IFN-γ inducible genes and Cd14, were upregulated (13). Surprisingly, genes for several ECM proteins were also upregulated at this time. Among these, lumican, a leucine rich repeat ECM protein, was upregulated together with Collagen I and III. Our earlier study shows that lumican binds to bacterial lipopolysaccharides (LPS) and to CD14, the glycerol phsophatidyl inositol-linked cell surface protein that promotes host recognition of LPS through TLR 4 (14). Gene targeted lumican-null mice (Lum−/−) are hypo-responsive to LPS mediated septic shock and produce lower levels of pro-inflammatory cytokines. At the cellular level, Lum−/− macrophages in culture produce lower levels of proinflammatory cytokines in response to LPS. This connection between lumican and innate immune response prompted us in the current study to examine the development of colonic inflammation in the Lum−/− mice.
Materials and Methods
Mice
Gene targeted lumican-null mice that we developed earlier and maintained in the outbred CD1 background (15) were bred into the C57BL/6J background by backcrossing for more than 12 generations. Wild type (Lum+/+) and Lum−/− C57BL/6J mice, 8–10 weeks of age, of both genders were used in most experiments. In a few experiments, as indicated in specific figures, the Lum+/+ and Lum−/− mice in the CD1 background were used. The Rag1tm1Mom (Rag1−/−) C57BL/6J and wildtype C57BL/6J mice at 8 weeks of age were purchased from the Jackson Laboratories.
Colitis models
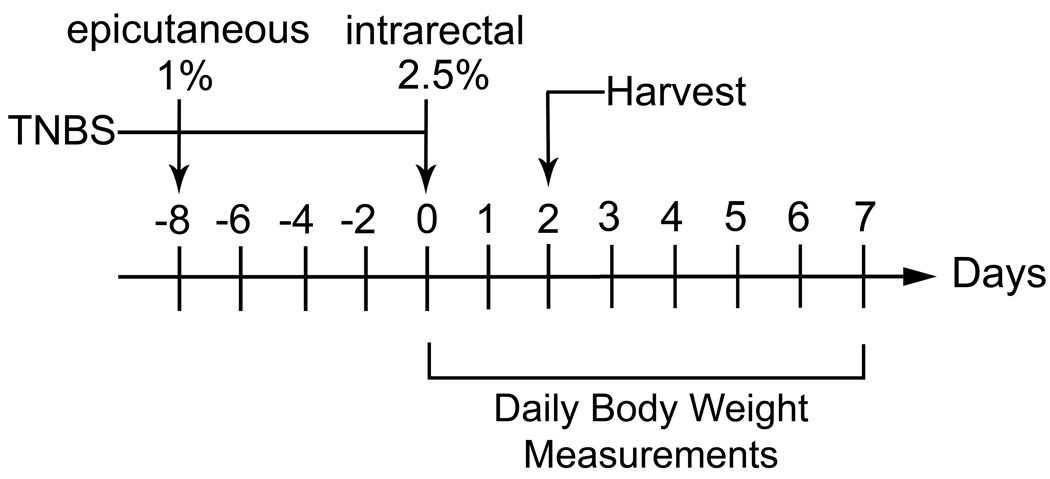
Lum+/+ and Lum−/− C57BL/6J mice were randomized into control and TNBS groups. The TNBS mice were sensitized with an epicutaneous application of 1% TNBS (Sigma-Aldrich) in 80% ethanol and the control group was given an epicutaneous treatment of saline (Fig. 1). Seven days after sensitization, the mice were anesthetized with avertin (350 mg/kg bodyweight) and weighed. The TNBS group received 5 µl/g body weight of 2.5% TNBS in 50% ethanol by intra-rectal administration using a syringe fitted with a 20 gauge gavage feeding needle (Fine Science Tools, 18060-20). The control group was treated in a similar manner with 5 µl/g body weight of saline or 50% ethanol. For histology and extraction of proteins, the mice were euthanized 48 hours after intra-rectal administration of TNBS or saline. The TNBS treatment described above was also used for the wild type and the Rag1−/− C57BL/6J mice. In a few of our earlier experiments, the Lum+/+ and Lum−/− in the CD1 background were not sensitized epicutaneously before intra-rectal administration of TNBS as described (12). To determine survival and body weight loss, the mice were weighed daily without anesthesia for 8–10 days after intra-rectal administration of TNBS. Intra-rectal administration of saline or 50% ethanol served as controls. Animals that died by 24 hours of treatment were not included in determining mortality.
FIGURE 1.
TNBS Colitis model. The mice were pre-sensitized by epicutaneous application of 1% TNBS in 80% ethanol. Seven days later 5 µl/g body weight of 2.5% TNBS in 50% ethanol was administered intra-rectally under anesthesia. The mice were euthanized 48 hours later for tissue harvest, or followed up to 8 days after TNBS treatment for daily body weight measurements.
To develop the dextran sulfate sodium (DSS) colitis model, Lum+/+ and Lum−/− in the CD1 outbred strain were given 5% DSS in the drinking water. The mice were weighed daily and harvested on the 8th day.
Histology
Colon segments ~2 mm in length were taken 4 cm away from the anus and fixed in 10% buffered formalin, paraffin embedded, cut into 7 µm thick cross sections and stained with H&E. To examine thickening of the colon associated with colitis, images of H&E stained colon cross sections were captured with a Nikon Eclipse E400 microscope fitted with a DXM1200 Nikon Digital camera. The areas of colon cross sections were measured using the software NIS-Elements. An increase in cross sectional area was taken as an indication of inflammation and thickening of the colon. The pathology of the H&E stained tissue sections was scored using a scale of 0–4 as follows: No significant lesions = 0, minimal to mild inflammatory infiltrates in the lamina propria or the submucosa = 1, mild to moderate inflammatory infiltrates, edema, occasional limited focal necrosis/ulceration = 2, moderate to marked transmural inflammation, edema, necrosis affecting up to 70% of the mucosa = 3, marked transmural inflammation, edema, necrosis affecting 70–100% of the mucosa= 4.
Protein extraction and ELISA
A colon segment of approximately 2 mm length was taken from each control and experimental animal. Total protein was extracted by homogenization, followed by a brief sonication (10 seconds at 20% amplitude, 3 times) of the tissue in T-PER containing the Halt proteinase inhibitor cocktail (Thermo Scientific) and centrifuged to remove debris. Protein concentrations were estimated using the BCA Protein Assay kit (Thermo Scientific).
The following ELISA kits were used to assay specific proteins and cytokines : mouse MPO kit (HK210) from Hycult Biotechnology, R&D mouse quantikine TNF-α kit (MTA00), R&D Quantikine mouse IL-4 (M4000B), R&D mouse KC/CXCL1 (MKC00B), total NF-κB p65 Sandwich ELISA kit (#7174) from Cell Signaling.
Electrophoretic mobility gel shift assay (EMSA)
NF-κB gel binding oligonuceotides (SC2505) and mutant oligonuceotides (SC2511) were purchased from Santa Cruz and end labeled with γ32P-ATP and T4 polynuceotide kinase (Fermentas). The labeled oligonuceotides were incubated with macrophage nuclear extracts and resolved by SDS PAGE in a 6% polyacrylamide gel pre-run for 20 minutes. The gels were dried and exposed to film overnight at −80°C.
Statistical analysis
Measurements taken in control and TNBS treated animals were compared using the student t test when results were consistent with normally distributed values. Experiments that involved unequal sample size and did not necessarily follow normal distributions were compared using the Mann-Whitney test with ≤ 0.05 considered significant. The GraphPad PRISM software (v4.02) was used to perform these analyses.
Ethical considerations
All mouse procedures were evaluated and approved by the Johns Hopkins Animal Care and Use Committee. In case of extreme lethargy and >25% loss in bodyweight after inducing colitis, the mice were taken out of the study and euthanized.
Results
Macroscopic disease and the course of colitis
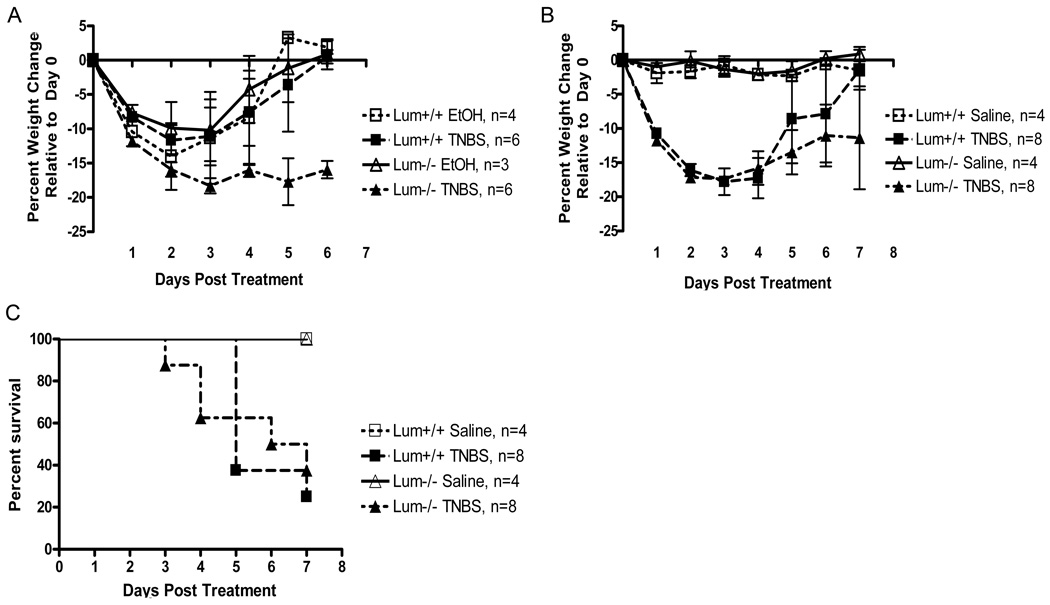
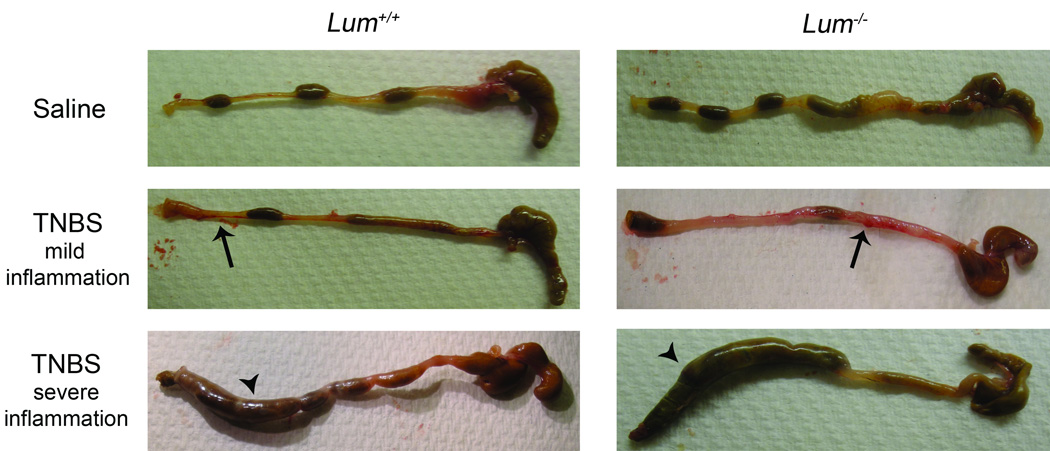
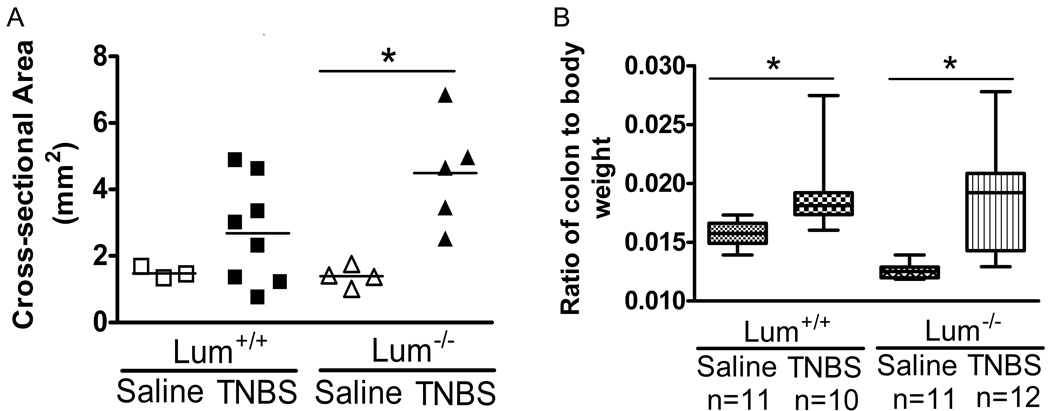
TNBS treated mice of both genotypes showed signs of piloerection, lethargy and diarrhea which were not seen in the saline treated mice (data not shown). Ethanol, often used as a vehicle control for the TNBS treatment, leads to colonic tissue damage, and in our experience changes the expression of pro-inflammatory genes (13). Here we show that intra-rectal administration of ethanol leads to similar trends in body weight loss in the Lum+/+ and the Lum−/− mice. The results further show that all ethanol treated mice and the TNBS treated Lum+/+ mice recovered their initial body weight by the sixth day after treatment (Fig. 2A). In contrast, the Lum−/− TNBS treated mice did not recover by day 6, suggesting that the TNBS hapten had additional detrimental effects on lumican deficiency. Intra-rectal administration of saline did not lead to loss in body weight (Fig. 2B). In the same experiment, the Lum+/+ and Lum−/− TNBS treated mice showed up to 18% loss in body weight. However, after seven to eight days the Lum+/+ TNBS group had regained most of the lost weight, while the Lum−/− TNBS group still displayed on average an 11% body weight reduction (Fig. 2B). We also tested the TNBS colitis model in the CD1 outbred strain, and as in the C57BL/6J background, the Lum−/− CD1 mice showed slower recovery from body weight loss (data not shown). As is typical for the TNBS model, we found an overall survival of 30% with no significant difference between Lum+/+ and Lum−/− mice, while saline treated controls showed 100% survival (Fig. 2C). Macroscopic appearance of the colon was normal in the saline treated animals (Fig. 3 top panels), while in the TNBS treated wild type and Lum−/− mice it was frequently swollen and bloody, with soft stool (Fig. 3 middle panels). To gauge edema and inflammation of the colon, we used two parameters: 1) cross sectional areas of H&E stained colon sections, and 2) colon to body weight ratios. The cross-sectional areas were higher for the TNBS over saline–treated controls, with the difference being more pronounced and significant (p= 0.007) in the TNBS- Lum−/− group over its respective saline control (Fig. 4A). The colon to body weight ratio was higher in the TNBS over saline treated mice of both genotypes. However, the increase was more pronounced in the Lum−/− TNBS treated mice (Fig. 4B). These results suggest that colonic edema and swelling is higher in the inflamed Lum−/− mice.
FIGURE 2.
Response to intra-rectal TNBS treatment. A. Lum+/+ and Lum−/− in the C57BL/6J mice were challenged with 2.5%TNBS in 50% ethanol (ETOH) or ETOH alone. Change in body weight was expressed as a percent weight change relative to their weight on day 0. The Lum−/− TNBS challenged mice failed to recover the lost body weight, whereas the ETOH challenged Lum−/− and the wild type TNBS and ETOH treated mice recovered by day 6. B. TNBS colitis using saline treatment as a control. The saline treated mice did not lose weight while the TNBS treated mice lost weight. C. Survival presented as a Kaplan Meier survival plot. Lum+/+ and Lum−/− C57BL/6J mice challenged with or without TNBS as in Figure 1, and observed for survival for 8 days. There was no significant difference in survival between the two genotypes. The data shown here is one of two representative experiments.
FIGURE 3.
Macroscopic appearance of the colon in TNBS-treated Lum+/+ and Lum−/− C57BL/6J mice. The colons were harvested 48 hours after intra-rectal TNBS administration and photographed. Saline treated colons (top two panels) showed no inflammation. TNBS treated colons with mild inflammation (middle two panels) appeared bloody in areas (arrows), while colons showing significant inflammation (bottom panels) were macroscopically thickened and necrotic (arrow heads). The highly inflamed colons were seen more frequently in the Lum+/+ TNBS-treated mice. This is one of three representative experiments.
FIGURE 4.
Increased thickening of TNBS treated colons. A. Cross-sectional areas of colons, taken as a measure of colon thickening and inflammation for Lum+/+ and Lum−/− C57BL/6J mice treated with TNBS. Colon cross-sectional areas were increased significantly in Lum−/− TNBS treated mice compared to saline controls. * p=0.02 (Mann-Whitney test). The data shown is one of two representative experiments. B. Increased colon to body weight ratio in Lum+/+ and Lum−/− CD1 mice treated with TNBS. The increase in colon to body weight ratio was significantly increased in TNBS challenged Lum+/+ (p= 0.005) and Lum−/− (p=0.0002) mice compared to their saline treated controls. * A p value ≤ 0.05 was considered statistically significant based on Mann-Whitney test. Representative data from two similar experiments is shown here.
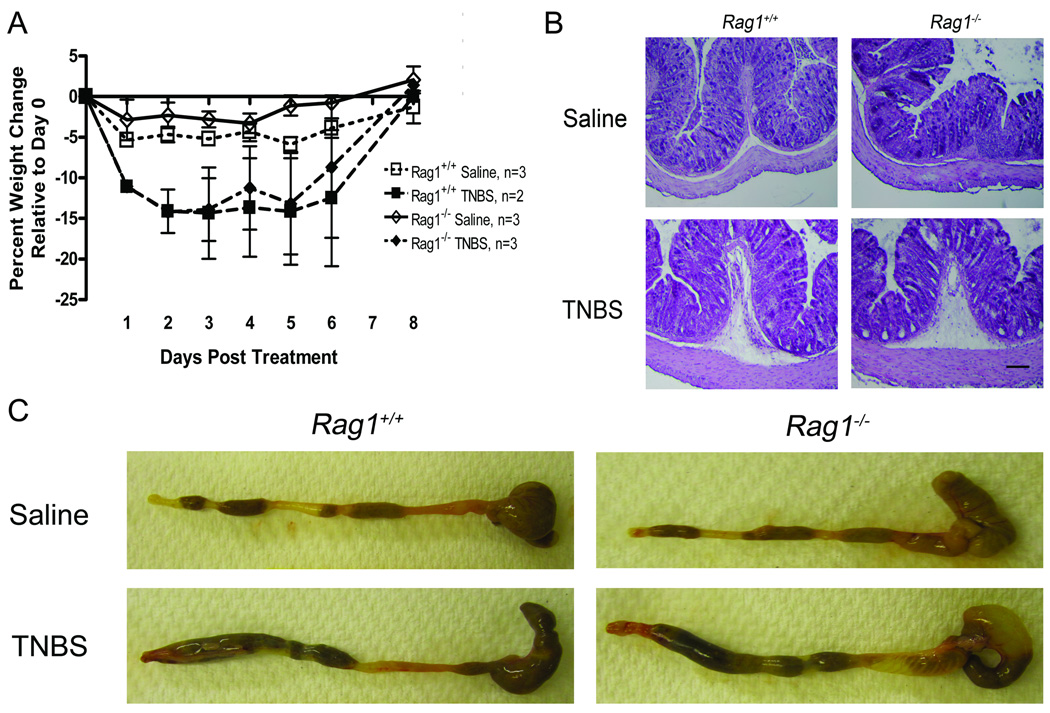
The TNBS colitis model is considered to involve a T-cell mediated immune response in the colonic mucosa (16). However, the early acute response to the TNBS treatment may be largely innate immune-driven. To test this we used the TNBS colitis model in the Rag1tm1Mom mice (Rag1−/−). The Rag1−/− mice are deficient in mature B and T cells (17) and often used as recipients in T-cell transfer colitis models (18) but not directly in TNBS colitis models. In our TNBS model weight loss in the Rag1−/− was similar to that of the wildtype mice (Fig. 5A), and the colon, harvested 2 days after the intra-rectal TNBS treatment, appeared equally inflamed in both (Fig. 5B). Histology also indicated similar degrees of inflammation in the wild types and the Rag1−/− mice (Fig. 5C). These results suggest that the early stage of TNBS colitis involves an innate immune inflammatory response.
FIGURE 5.
TNBS colitis in the Rag1−/− mice. A. The mice were given an intra-rectal treatment of TNBS or saline and weighed daily. Weight loss in the TNBS treated wild type and the Rag1−/− was similar. B. Macroscopic appearance of the colon. The colon appeared equally inflamed in the wild type and the Rag1−/− mice. C. Histology. Colonic sections of the TNBS treated Rag1−/− mice showed signs of inflammation that were comparable to those seen in the wild type. The submucosa was inflamed and edematous and inflammatory cells were visible in the submucosa and the lamina propria.
Given that the TNBS colitis model as used here is primarily an injury and innate immune response, we considered if the increased morbidity in the TNBS-challenged Lum−/− mice was entirely due to epithelial injury and delayed healing. To test this possibility we used the DSS-colitis model where inflammation is initiated by epithelial injury and loss of barrier integrity. Administration of 5% DSS in the drinking water caused diarrhea and equivalent weight loss in the Lum+/+ and the Lum−/− mice (data not shown). This suggests that in the TNBS-colitis model the TNBS hapten itself has an additional detrimental effect in the Lum−/− mice.
Colonic tissue damage and inflammatory cell infiltration in the TNBS treated lumican-deficient mice
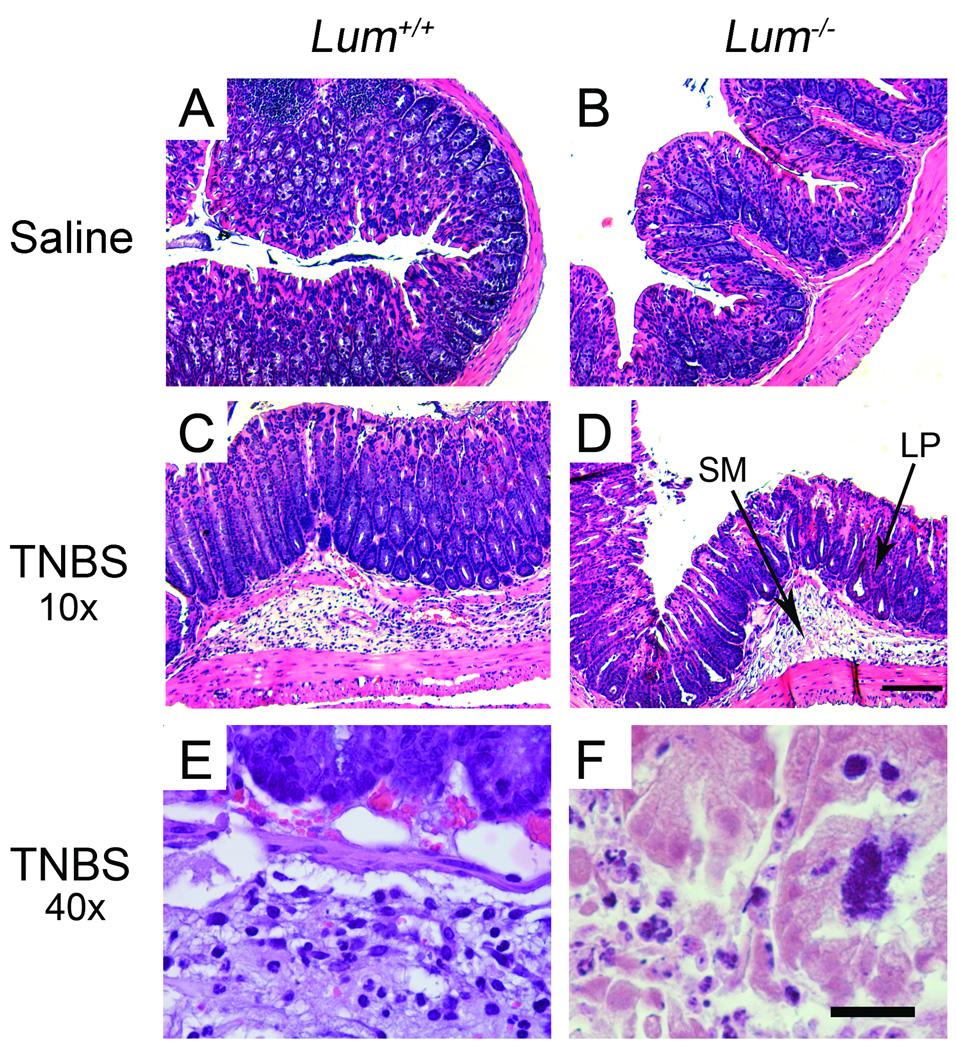
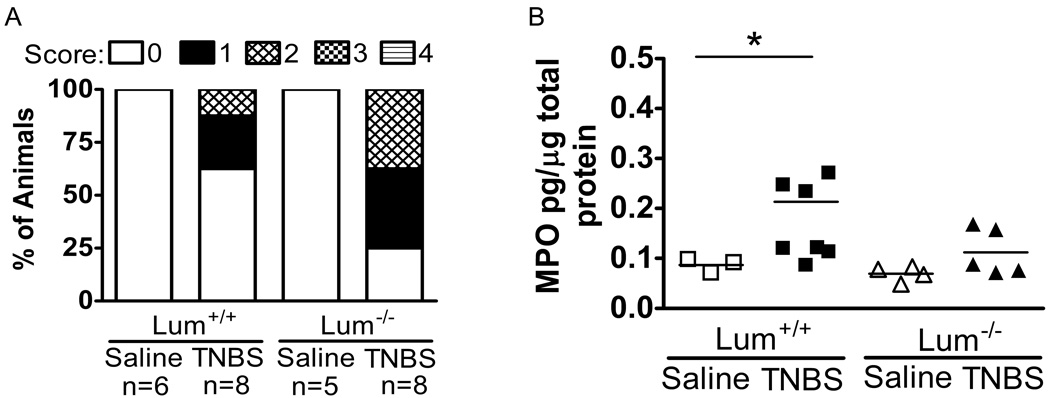
Histology showed healthy tissue architecture in the saline treated mice (Fig. 6A and B). The TNBS-wild type group showed infiltration of inflammatory cells, moderate edema and ulceration (Fig. 6C). The Lum−/− TNBS group tended to have more edema, ulceration and frequent necrosis (Fig. 6D and F). However, the histology was quite variable, with some sections showing very little inflammation and tissue damage in the TNBS-treated animals. Therefore, to compare the overall response in the Lum+/+ and the Lum−/− TNBS challenged mice, the slides were scored using a scale of 0 to 4 (methods). The pathology scores from one set of experiments shows higher inflammation scores in a larger proportion of the TNBS challenged Lum−/− compared to the Lum+/+ mice (Fig. 7A). Measurements of myeloperoxidase (MPO) released by inflammatory cells, taken as a measure of inflammatory cell infiltrates, showed less than normal increase in colonic extracts of the TNBS-treated Lum−/− mice (Fig. 7B). Taken together, the results suggest increased edema, tissue damage and necrosis, but overall lower inflammatory cell influx in the Lum−/− TNBS treated mice.
FIGURE 6.
Histology of colon cross sections. A. Lum+/+ and B. Lum−/− saline and TNBS treated mice. C. Lum+/+ and D. Lum−/− TNBS treated colons under low magnification show inflammation of the submucosa (SM) and some inflammatory cell infiltrates in the lamina propria (LP). Necrosis and tissue damage is shown under high magnification in E. Lum+/+ and F. Lum−/− TNBS treated mice.
FIGURE 7.
Reduced influx of neutrophils in Lum−/− mice compared to Lum+/+ TNBS treated colons but increased overall tissue damage. A. Tiled bar graph showing percent of animals with 0–4 inflammation scores (Methods). In this experimental set there were no mice that displayed scores of 3 or 4. Overall, the Lum−/− TNBS challenged group showed more animals with an inflammation score of 2. B. Measurements of MPO by ELISA to assess neutrophil infiltration after TNBS challenge. TNBS treated Lum−/− showed no significant increases in MPO, whereas Lum+/+ TNBS treated colons showed significant increases (*p=0.048, Mann-Whitney test).
Induction of pro-inflammatory factors in inflamed colonic tissues
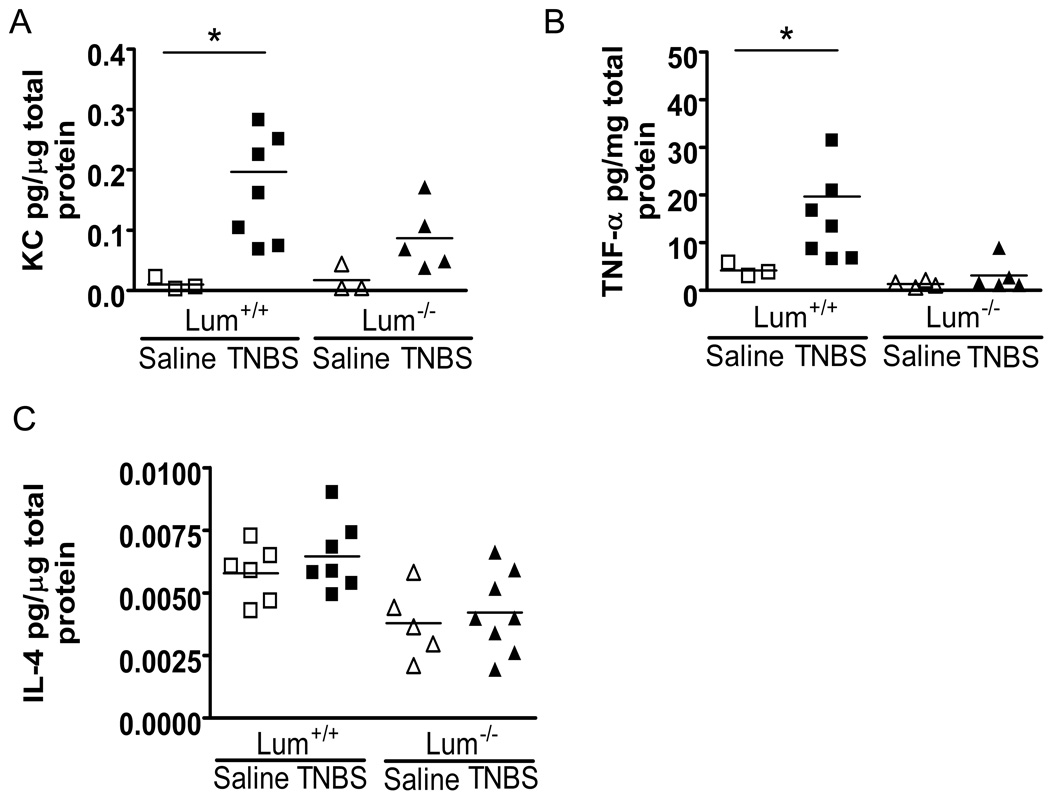
To further examine inflammation at the molecular level, we measured levels of selected cytokines in colonic protein extracts by ELISA. Compared to basal levels in saline-treated mice, the chemokine CXCL1/KC was markedly increased in the TNBS-treated wild type mice (12 fold over basal level, p=0.027). By comparison, the increase in Lum−/− TNBS was modest (5 fold, p=0.04) (Fig. 8A). The pro-inflammatory cytokine TNF-α was increased significantly in the TNBS-wild type mice over saline controls, while the TNBS-treated Lum−/− mice showed very little induction of TNF-α (Fig. 8B). We next investigated IL-4 levels as a Th2 response-related cytokine; no significant change in IL-4 reaffirms a lack of Th2-related immune response in this colitis model (Fig. 8C).
FIGURE 8.
Cytokine levels in colonic protein extracts. A. CXCL1/KC was increased significantly (p=0.01) in colonic extracts of Lum+/+ TNBS-treated mice compared to saline treated controls. The Lum−/− mice showed little increase in KC after TNBS treatment. B. TNFα levels were increased significantly (p=0.012) in the Lum+/+ TNBS mice, while there was no significant increase in TNF-α in Lum−/− TNBS compared to saline-treated controls. C. IL-4 levels showed no significant increase after TNBS treatment in the Lum+/+ or the Lum−/− mice. * A p value ≤ 0.05 was considered statistically significant based on Mann-Whitney test. The result shown is one of two similar experiments.
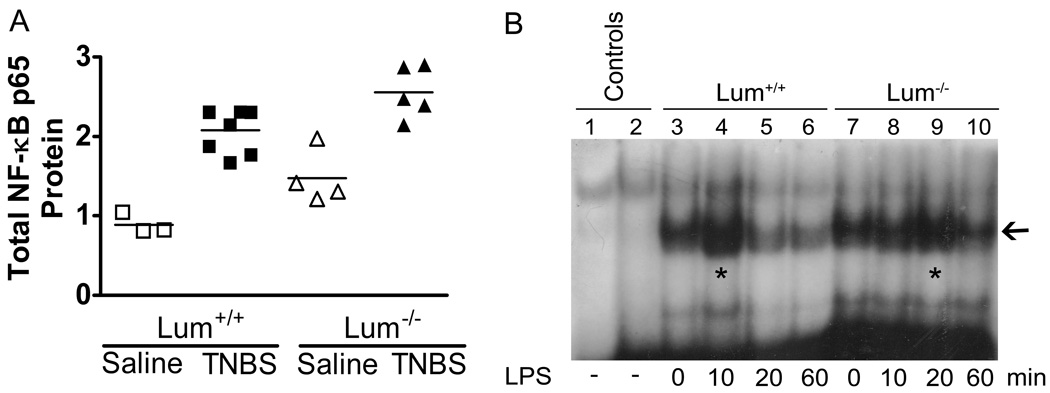
Since NF-κB plays a central role in innate immune response and induction of pro-inflammatory cytokines and chemokines, we measured total NF-κB p65 levels in colonic protein extracts of each animal using ELISA methods. While total NF-κB p65 increased in both genotypes after TNBS treatment, the increase in TNBS treated wild types was higher (2.5 fold) than that seen in TNBS-treated Lum−/− mice (1.7 fold) (Fig. 9A). To further test if there is a delay in the activation of NF-κB in lumican deficiency, we performed an EMSA test on primary peritoneal macrophages. Lum+/+ and Lum−/− macrophages in culture were exposed to LPS for different times. Nuclear extracts of these macrophages were incubated with radio-labeled NF-κB specific and nonspecific oligonucleotides and analyzed by SDSPAGE. Maximal specific binding of nuclear proteins with the target oligonucleotide occurred in extracts of Lum+/+ macrophages that were treated with LPS for 10 min. In the Lum−/− macrophage extracts, comparable nuclear protein binding occurred after 20 min of LPS treatment. Thus nuclear localization of NF-κB in Lum−/− peritoneal macrophages was delayed by 10 minutes compared to wild types in response to bacterial LPS treatment (Fig. 9B).
FIGURE 9.
Poor NF-κB response in the absence of lumican. A. Total NF-κB level was assessed in colonic extracts by ELISA measurements of the p65 subunit. The increase in total NF-κB in TNBS treated mice of both genotypes was significant (p ≤ 0.05) compared to the saline controls. However, the increase was much lower in the Lum−/− (1.7X) compared to that seen in the Lum+/+ (2.5X) mice. B. Delayed nuclear localization of NF-κB in peritoneal macrophages of Lum−/− mice compared to Lum+/+ after exposure to 10 ng/ml LPS. Nuclear extracts were tested by EMSA (Methods). Lum−/− peritoneal macrophages show maximal NF-κB binding after 20 min LPS exposure, while this was achieved by 10 min in Lum+/+ macrophages.
Discussion
Here we report distinct differences in the response of wild type and lumican-deficient mice to an intra-rectal challenge of TNBS that speak of weak innate immune and inflammatory responses, and increased morbidity in the absence of lumican. Lumican is an extracellular matrix proteoglycan present in large amounts in connective tissues rich in fibrillar collagens such as the intestinal submucosa (19). The lumican core protein has leucine-rich repeat (LRR) motifs, similar to those found in TLR proteins and other members of the LRR superfamily (20). Lumican deficiency disrupts collagen fibril assembly and impairs normal functions of connective tissues (15). In addition, we have shown that lumican promotes innate immune response through its interactions with the CD14-TLR4 pathway and neutrophil migration by interacting with β2 integrin, a subunit of the neutrophil migration receptor MAC1 or CD11b/CD18 (14, 21). We also know that lumican expression is induced in epithelial cells after injury (22), and epithelial wound healing in the cornea is delayed in Lum−/− mice (23, 24). The emerging role of lumican, and possibly some of the other members of this group of ECM proteins, is one of modulating innate immune signals during inflammation and wound repair and maintaining extracellular matrix homeostasis.
We used the TNBS-colitis model to follow disease manifestation and inflammatory responses during acute and active inflammation of the colon. MPO measurements from colonic protein extracts suggest very little influx of neutrophils in the TNBS-treated Lum−/− mice. CXCL1/KC was induced in all TNBS-colons, but the increase in Lum−/− mice was markedly lower than that seen in the Lum+/+ mice. Mouse KC, a functional equivalent of human IL-8, is a strong chemoattractant for neutrophils; its weaker induction in Lum−/− TNBS treated colons is consistent with low MPO measurements in these animals. The pro-inflammatory cytokine TNF-α was also not induced to significant levels in the Lum−/− TNBS treated colons. Lower inductions of KC and TNF-α in the Lum−/− mice suggest weak innate immune response from epithelial cells and macrophages in the colon. An ELISA that detects the p65 subunit of NF-κB showed lower levels of this subunit in the Lum−/− compared to Lum+/+ TNBS treated colons. By gel shift assays we found that in Lum−/− primary peritoneal macrophage cultures there was a delay in nuclear localization of NF-κB in response to an LPS challenge. The NF-κB transcription factor is a common switch that responds to several cell surface and cytoplasmic pathogen recognition receptors, including TLR4, and mediates the upregulation of many inflammatory genes (25). NF-κB blockade in vivo has been shown to effectively ameliorate inflammation in the TNBS mouse colitis model (26). In a previous study we used this NF-κB blockade treatment in the chronic TNBS colitis model and found effective suppression of Cxcl1 (13). Lumican was also inhibited by NF-κB blockade indicating that its regulation closely mimics that of the inflammatory genes, rather than ECM collagens (13). Our observations in the current study further support the idea that lumican has an inflammation-regulatory role in colitis. The Lum−/− mice show subtle but clearly discernible differences in overall disease response that relate to the differences seen in innate immune signals at the molecular level. While there is no dramatic difference in mortality between wild type and Lum−/− mice challenged with TNBS, the Lum−/− mice show significantly slower recovery of lost body weight. Histology of the inflamed colons, on average, show more edema and tissue damage in the Lum−/− TNBS colon. Also, there were more instances of necrosis in the Lum−/− TNBS colon that could be due to increased bacterial colonization of the colon.
The TNBS colitis first tested in rats (27) and then adapted in the mouse (16, 28) is considered to be driven by T-cell mediated immune response as TNBS haptenates host proteins and makes them immunogenic. However, response to the TNBS-ETOH challenge also involves an innate immune response that in the short term can produce pro-inflammatory cytokines and an inflammatory response without significant antigen presentation to T cells and the induction of Th1 cells. Thus, in the Rag1−/− mice, deficient in mature T and B lymphocytes, we also detected wild type–like inflammatory response within 6 days after TNBS treatment. The DSS colitis model, which is T-cell independent and mediated by epithelial injury and inflammatory cell infiltration, has been shown by others to be equally effective in the Rag1−/− mice (29). We tested the DSS colitis model in the Lum−/− mice, and compared to the wild type mice, found no difference in disease manifestation. It appears that the DSS-mediated epithelial injury and healing may not be affected by lumican-deficiency. In contrast, the TNBS-haptenated host proteins seem to be stimulating an early innate immune response that is affected by lumican deficiency.
In the Lum−/− TNBS treated mice cytokine induction and inflammatory cell infiltration was reduced, yet disease severity in terms of weight loss and tissue damage was elevated. A more extreme version of reduced inflammatory response but aggravated disease is seen in the Tlr4−/− and MyD88−/− mice. These mutant mice show poor neutrophil infiltration, aggravated commensal gram-negative bacteria in the lamina propria and overall increased disease (30). In the same vein, MyD88−/− mice showed increased bacterial burden in a Citrobacter rodentium infection model (31). TLR4 and MYD88 dependent innate immune responses play a central role in epithelial and mucosal health (32–34). The exact protective mechanism is not well understood, but a combination of processes is credited. First, TLR activation by commensal bacteria promotes epithelial integrity and homeostasis. TLR mediated activation of pro-inflammatory cytokines help epithelial survival and cell migration for efficacious healing of wounds. Second, infiltrating neutrophils and macrophages are protective by restricting microbial (commensal and pathogenic) overgrowth.
Innumerable studies of IBD point to an etiopathogensis of dysregulated immune response and altered response to environmental and microbial factors, with the concept of altered host innate immune response gaining significant traction (1, 3, 35). NOD2/CARD15, a key regulator of innate immune response has been established as a Crohn’s disease susceptibility gene; TLR4 and CD14 have also been associated with IBD genetics. Investigations of the major innate immune signaling nodes will no doubt continue to elucidate IBD pathogenesis and significant susceptibility genes. The ECM genes may modify IBD pathogenesis to affect disease severity and possibly susceptibility. ECM proteins bind growth factors, cytokines and cell surface receptors, and can either enhance or attenuate signals (24, 36, 37). Clearly, the absence of lumican in the mouse affects the outcome of colitis in a small but significant way. Other related ECM proteins that may have similar functions are fibromodulin, decorin and biglycan. We believe that as more extracellular matrix proteins become better characterized, many will be identified as modifiers of key innate immune and inflammatory genes and as having an effect on IBD pathogenesis.
Acknowledgments
Funding support: Crohn’s and Colitis Foundation of America Senior Investigator Award and NEI/NIH EY 11654 to Shukti Chakravarti
References
- 1.Kaser A, Zeissig S, Blumberg RS. Inflammatory bowel disease. Annu Rev Immunol. 2010;28:573–621. doi: 10.1146/annurev-immunol-030409-101225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417–429. doi: 10.1056/NEJMra020831. [DOI] [PubMed] [Google Scholar]
- 3.Danese S, Fiocchi C. Etiopathogenesis of inflammatory bowel diseases. World J Gastroenterol. 2006;12:4807–4812. doi: 10.3748/wjg.v12.i30.4807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ogura Y, Bonen DK, Inohara N, et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature. 2001;411:603–606. doi: 10.1038/35079114. [DOI] [PubMed] [Google Scholar]
- 5.Strober W, Fuss I, Mannon P. The fundamental basis of inflammatory bowel disease. J Clin Invest. 2007;117:514–521. doi: 10.1172/JCI30587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010;11:373–384. doi: 10.1038/ni.1863. [DOI] [PubMed] [Google Scholar]
- 7.Karin M. The beginning of the end: IkappaB kinase (IKK) and NF-kappaB activation. J Biol Chem. 1999;274:27339–27342. doi: 10.1074/jbc.274.39.27339. [DOI] [PubMed] [Google Scholar]
- 8.Sen R, Smale ST. Selectivity of the NF-{kappa}B response. Cold Spring Harb Perspect Biol. 2010;2 doi: 10.1101/cshperspect.a000257. a000257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jiang D, Liang J, Fan J, et al. Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med. 2005;11:1173–1179. doi: 10.1038/nm1315. [DOI] [PubMed] [Google Scholar]
- 10.Schaefer L, Babelova A, Kiss E, et al. The matrix component biglycan is proinflammatory and signals through Toll-like receptors 4 and 2 in macrophages. J Clin Invest. 2005;115:2223–2233. doi: 10.1172/JCI23755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.He YW, Li H, Zhang J, et al. The extracellular matrix protein mindin is a pattern-recognition molecule for microbial pathogens. Nat Immunol. 2004;5:88–97. doi: 10.1038/ni1021. [DOI] [PubMed] [Google Scholar]
- 12.Lawrance IC, Wu F, Leite AZ, et al. A murine model of chronic inflammation-induced intestinal fibrosis down-regulated by antisense NF-kappa B. Gastroenterology. 2003;125:1750–1761. doi: 10.1053/j.gastro.2003.08.027. [DOI] [PubMed] [Google Scholar]
- 13.Wu F, Chakravarti S. Differential Expression of Inflammatory and Fibrogenic Genes and Their Regulation by NF-{kappa}B Inhibition in a Mouse Model of Chronic Colitis. J Immunol. 2007;179:6988–7000. doi: 10.4049/jimmunol.179.10.6988. [DOI] [PubMed] [Google Scholar]
- 14.Wu F, Vij N, Roberts L, et al. A novel role of the lumican core protein in bacterial lipopolysaccharide-induced innate immune response. J Biol Chem. 2007;282:26409–26417. doi: 10.1074/jbc.M702402200. [DOI] [PubMed] [Google Scholar]
- 15.Chakravarti S, Magnuson T, Lass JH, et al. Lumican regulates collagen fibril assembly: skin fragility and corneal opacity in the absence of lumican. J Cell Biol. 1998;141:1277–1286. doi: 10.1083/jcb.141.5.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Neurath M, Fuss I, Strober W. TNBS-colitis. Int Rev Immunol. 2000;19:51–62. doi: 10.3109/08830180009048389. [DOI] [PubMed] [Google Scholar]
- 17.Mombaerts P, Iacomini J, Johnson RS, et al. RAG-1-deficient mice have no mature B and T lymphocytes. Cell. 1992;68:869–877. doi: 10.1016/0092-8674(92)90030-g. [DOI] [PubMed] [Google Scholar]
- 18.Pavlick KP, Ostanin DV, Furr KL, et al. Role of T-cell-associated lymphocyte function-associated antigen-1 in the pathogenesis of experimental colitis. Int Immunol. 2006;18:389–398. doi: 10.1093/intimm/dxh378. [DOI] [PubMed] [Google Scholar]
- 19.Chakravarti S. Functions of lumican and fibromodulin: lessons from knockout mice. Glycoconj J. 2002;19:287–293. doi: 10.1023/A:1025348417078. [DOI] [PubMed] [Google Scholar]
- 20.Chakravarti S, Stallings RL, SundarRaj N, et al. Primary structure of human lumican (keratan sulfate proteoglycan) and localization of the gene (LUM) to chromosome 12q21.3-q22. Genomics. 1995;27:481–488. doi: 10.1006/geno.1995.1080. [DOI] [PubMed] [Google Scholar]
- 21.Lee S, Bowrin K, Hamad AR, et al. Extracellular matrix lumican deposited on the surface of neutrophils promotes migration by binding to beta2 integrin. J Biol Chem. 2009;284:23662–23669. doi: 10.1074/jbc.M109.026229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Saika S, Shiraishi A, Liu CY, et al. Role of lumican in the corneal epithelium during wound healing. J Biol Chem. 2000;275:2607–2612. doi: 10.1074/jbc.275.4.2607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vij N, Roberts L, Joyce S, et al. Lumican suppresses cell proliferation and aids Fas-Fas ligand mediated apoptosis: implications in the cornea. Exp Eye Res. 2004;78:957–971. doi: 10.1016/j.exer.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 24.Vij N, Roberts L, Joyce S, et al. Lumican regulates corneal inflammatory responses by modulating Fas-Fas ligand signaling. Invest Ophthalmol Vis Sci. 2005;46:88–95. doi: 10.1167/iovs.04-0833. [DOI] [PubMed] [Google Scholar]
- 25.Neurath MF, Becker C, Barbulescu K. Role of NF-kappaB in immune and inflammatory responses in the gut. Gut. 1998;43:856–860. doi: 10.1136/gut.43.6.856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Neurath MF, Pettersson S, Meyer zum Buschenfelde KH, et al. Local administration of antisense phosphorothioate oligonucleotides to the p65 subunit of NF-kappa B abrogates established experimental colitis in mice. Nat Med. 1996;2:998–1004. doi: 10.1038/nm0996-998. [DOI] [PubMed] [Google Scholar]
- 27.Yamada Y, Marshall S, Specian RD, et al. A comparative analysis of two models of colitis in rats. Gastroenterology. 1992;102:1524–1534. doi: 10.1016/0016-5085(92)91710-l. [DOI] [PubMed] [Google Scholar]
- 28.Neurath M, Fuss I, Kelsall B, et al. Antibodies to interleukin 12 abrogate established experimental colitis in mice. J Exp Med. 1995;182:1281–1290. doi: 10.1084/jem.182.5.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sainathan SK, Hanna EM, Gong Q, et al. Granulocyte macrophage colony-stimulating factor ameliorates DSS-induced experimental colitis. Inflamm Bowel Dis. 2008;14:88–99. doi: 10.1002/ibd.20279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fukata M, Michelsen KS, Eri R, et al. Toll-like receptor-4 is required for intestinal response to epithelial injury and limiting bacterial translocation in a murine model of acute colitis. Am J Physiol Gastrointest Liver Physiol. 2005;288:G1055–G1065. doi: 10.1152/ajpgi.00328.2004. [DOI] [PubMed] [Google Scholar]
- 31.Gibson DL, Ma C, Bergstrom KS, et al. MyD88 signalling plays a critical role in host defence by controlling pathogen burden and promoting epithelial cell homeostasis during Citrobacter rodentium-induced colitis. Cell Microbiol. 2008;10:618–631. doi: 10.1111/j.1462-5822.2007.01071.x. [DOI] [PubMed] [Google Scholar]
- 32.Abreu MT, Fukata M, Arditi M. TLR signaling in the gut in health and disease. J Immunol. 2005;174:4453–4460. doi: 10.4049/jimmunol.174.8.4453. [DOI] [PubMed] [Google Scholar]
- 33.Rakoff-Nahoum S, Hao L, Medzhitov R. Role of toll-like receptors in spontaneous commensal-dependent colitis. Immunity. 2006;25:319–329. doi: 10.1016/j.immuni.2006.06.010. [DOI] [PubMed] [Google Scholar]
- 34.Rakoff-Nahoum S, Paglino J, Eslami-Varzaneh F, et al. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell. 2004;118:229–241. doi: 10.1016/j.cell.2004.07.002. [DOI] [PubMed] [Google Scholar]
- 35.Yamamoto-Furusho JK, Podolsky DK. Innate immunity in inflammatory bowel disease. World J Gastroenterol. 2007;13:5577–5580. doi: 10.3748/wjg.v13.i42.5577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ruoslahti E, Yamaguchi Y, Hildebrand A, et al. Extracellular matrix/growth factor interactions. Cold Spring Harb Symp Quant Biol. 1992;57:309–315. doi: 10.1101/sqb.1992.057.01.035. [DOI] [PubMed] [Google Scholar]
- 37.Beattie J, McIntosh L, van der Walle CF. Cross-talk between the insulin-like growth factor (IGF) axis and membrane integrins to regulate cell physiology. J Cell Physiol. 2010;224:605–611. doi: 10.1002/jcp.22183. [DOI] [PubMed] [Google Scholar]