Abstract
Utilization of polymers as biomaterials has greatly impacted the advancement of modern medicine. Specifically, polymeric biomaterials that are biodegradable provide the significant advantage of being able to be broken down and removed after they have served their function. Applications are wide ranging with degradable polymers being used clinically as surgical sutures and implants. In order to fit functional demand, materials with desired physical, chemical, biological, biomechanical and degradation properties must be selected. Fortunately, a wide range of natural and synthetic degradable polymers has been investigated for biomedical applications with novel materials constantly being developed to meet new challenges. This review summarizes the most recent advances in the field over the past 4 years, specifically highlighting new and interesting discoveries in tissue engineering and drug delivery applications.
INTRODUCTION
A biomaterial is defined as any natural or synthetic substance engineered to interact with biological systems in order to direct medical treatment.1 Biomaterials must be biocompatible meaning that they perform their function with an appropriate host response.2 In order to meet the needs of the biomedical community, materials composed of everything from metals and ceramics to glasses and polymers have been researched. Polymers possess significant potential since flexibility in chemistry gives rise to materials with great physical and mechanical property diversity. Degradable polymers are of utmost interest since these biomaterials are able to be broken down and excreted or resorbed without removal or surgical revision.
While natural polymers like collagen have been used biomedically for thousands of years, research into biomedical applications of synthetic degradable polymers is relatively new, starting in the 1960s.3,4 In the fifty years since, successes have been numerous, but grand challenges still exist in both the basic and translational elements of biomaterial design. From a basic science perspective, the capacity to modulate biomaterial chemistry to convey unique material properties is endless yet requires significant time and resources to complete the research. As biomaterials are applied in the clinical setting, numerous issues arise that cannot be adequately identified and addressed in previous in vitro and model in vivo experiments. The host response to both tissue engineering and drug delivery devices depends on the chemical, physical and biological properties of the biomaterials. When these materials are also biodegradable, there exists the additional issue of continuing changes in the material properties induced by degradation over time. These changes can cause long-term host responses to these biomaterials to be greatly different than the initial response. These issues are non-trivial and have contributed to the slow evolution of biodegradable polymer biomaterials as a field of research.
In order to better address the many issues in biomaterial design and expedite progress, biomaterial scientists have fundamentally changed their approach to the research. Especially in the last ten years, there has been a shift in paradigm from investigators working independently on narrow research goals to collaborative teams that facilitate solving greater objectives. By combining researchers with expertise in chemistry, biology, materials, engineering and clinical practice, biomaterials research has been able to advance more rapidly in the past few years.5–13
In the design of biodegradable biomaterials, many important properties must be considered. These materials must (1) not evoke a sustained inflammatory response; (2) possess a degradation time coinciding with their function; (3) have appropriate mechanical properties for their intended use; (4) produce non-toxic degradation products that can be readily resorbed or excreted; and (5) include appropriate permeability and processability for designed application.14 These properties are greatly affected by a number of features of degradable polymeric biomaterials including, but not limited to: material chemistry, molecular weight, hydrophobicity, surface charge, water adsorption, degradation and erosion mechanism. Due to the wide-ranging use of polymeric biomaterials, a single, ideal polymer or polymeric family does not exist. Instead a library of materials is available to researchers that can be synthesized and engineered to best match the specifications of the material’s desired biomedical function.
Biomaterial applications of biodegradable polymers have already been extensively reviewed in the past so no attempt will be made to provide a further exhaustive review. Instead, the reader is referred to comprehensive articles in Advances in Biochemical Engineering/Biotechnology15 and Progress in Polymer Science16 which include research prior to and including 2006. This review will focus on the numerous advancements made in the development of hydrolytically and enzymatically degradable polymers over the past 4 years.
HYDROLYTICALLY DEGRADABLE POLYMERS
Hydrolytically degradable polymers are materials that possess hydrolytically labile chemical bonds in their backbone and can be broken down without secondary influence as shown in Fig. 1. The broken bond yields two species with one product gaining a hydrogen atom and the other gaining a hydroxyl group. A number of degradable polymers possess bonds that are susceptible to hydrolysis including esters, anhydrides, acetals, carbonates, amides, urethanes and phosphates. One of the major features that conveys significant impact on the capacity of these polymeric families to function as biomaterials is their relative degradation rates and erosion mechanisms. An extensive investigation into a number of different degradable polymeric families showed that the degradation rates (Table 1) can vary twelve-fold from very hydrolytically unstable (polyphosphazenes) to extremely hydrolytically stable (polyamides).17 It should be noted with certain families’ (polyphosphazenes and polyanhydrides) degradation rate can be greatly modulated based on polymer chemistry conveying significant flexibility in material properties for these families. Degradation rates are incorporated with other factors like water diffusion, monomer solubility and diffusion, and device geometry and size, to determine how a degradable polymeric biomaterial will erode. Erosion is typically categorized as surface erosion, bulk erosion or a combination of the two.18 Surface erosion is characterized by the rate of polymer degradation and mass relief at the water-device interface being much greater than the rate at which water diffuses into the bulk of the material leading to a device that degrades almost entirely at its surface. Bulk erosion is characterized by the reverse in which water diffusion is much faster than degradation leading to degradation and subsequent mass loss occurring throughout the bulk of the material. These categorizations are extremely important in determining which material is best for a desired application. For example, in sustained drug delivery a material that can undergo surface erosion may be desired since stable, near zeroth-order release can be maintained and payload release kinetics can be more easily tailored.19 Whereas for applications requiring a permeable membrane like in tissue engineering, bulk eroding materials would allow for necessary hydrolytic diffusion.20 The following sections discuss a number of hydrolytically sensitive polymers and their biomedical applications.
Figure 1.
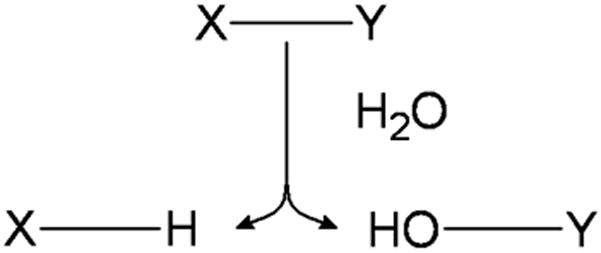
The hydrolytically sensitive bond X-Y is cleaved by a water molecule yielding the products of X-H and HO-Y.
Table 1.
Summary of different polymeric families’ applications, advantages, disadvantages, degradation rate and structure.
| Polymer | Applications | Advantages | Disadvantages | λ, Degradation Rate Constant (s−1) | Structure |
|---|---|---|---|---|---|
| Polyphosphazenes | Tissue Engineering Vaccine Adjuvant |
Synthetic Flexibility Controllable Mechanical Properties |
Complex Synthesis | 4.5 × 10−2 −1.4 × 10−7 Ref. 776,13 |
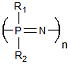
|
| Polyanhydrides | Drug Delivery Tissue Engineering |
Significant Monomer Flexibility Controllable Degradation Rates |
Low Molecular Weights Week Mechanical Properties |
1.9 × 10−3 − 9.4 × 10−9 Ref. 17,777 |
|
| Polyacetals | Drug Delivery | Mild pH Degradation Products pH Sensitive Degradation |
Low Molecular Weights Complex Synthesis |
6.4 × 10−5 Ref. 17 |
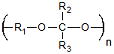
|
| Poly(ortho esters) | Drug Delivery | Controllable Degradation Rates pH Sensitive Degradation |
Week Mechanical Properties Complex Synthesis |
4.8 × 10−5 Ref. 17 |
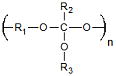
|
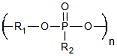
| Polyphosphoesters | Drug Delivery Tissue Engineering |
Biomolecule Compatibility Highly Biocompatible Degradation Products |
Complex Synthesis | 1.4 × 10−6 Ref. 778,779 |
|
| Polycaprolactone | Tissue Engineering | Highly Processable Many Commercial Vendors Available |
Limited Degradation | 3.5 × 10−8 Ref. 17 |
|
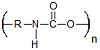
| Polyurethanes | Prostheses Tissue Engineering |
Mechanically Strong Handle Physical Stresses Well |
Limited Degradation Require Copolymerization with Other Polymers |
8.3 × 10−9 Ref. 780 |
|
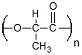
| Polylactide | Tissue Engineering Drug Delivery |
Highly Processable Many Commercial Vendors Available |
Limited Degradation Highly Acidic Degradation Products |
6.6 × 10−9 Ref. 17 |
|
| Polycarbonates | Drug Delivery Tissue Engineering Fixators |
Chemistry-Dependent Mechanical Properties Surface Eroding |
Limited Degradation Require Copolymerization with Other Polymers |
4.1 × 10−10 Ref. 285 |
|
| Polyamides | Drug Delivery | Conjugatable Side Group Highly Biocompatible Degradation Products |
Very Limited Degradation Charge Induced Toxicity |
2.6 × 10−13 Ref. 17 |
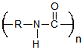
|
Poly(α-esters)
Poly(α-esters) are a class of polymers that contain an aliphatic ester bond in their backbone. While a number of polyesters are commercially available and all are theoretically degradable, the hydrolytically stable nature of the ester bond (Table 1) means only polyesters with reasonably short aliphatic chains can be utilized as degradable polymers for biomedical applications. While these polymers are often mildly hydrophobic, ester bond stability causes them to undergo bulk erosion.21 Due to the relative ease of their synthesis (via ring-opening or condensation polymerization) and commercial availability, poly(α-esters) have been the most heavily researched degradable biomaterials to date.22
Polyglycolide
Polyglycolide or poly(glycolic acid) (PGA) was one of the very first degradable polymers ever investigated for biomedical use. With a melting point (Tm) greater than 200 °C, a glass transition temperature (Tg) of 35 – 40 °C and very high tensile strength (12.5 GPa),23 PGA found favor as the degradable suture DEXON® which has been actively used since 1970.24 From 1984 to 1996, PGA was marketed as an internal bone pin under the name Biofix®, but since 1996 Biofix has been converted to a poly(L-lactide) base for better long-term stability.25,26
Due to PGA’s rapid degradation and insolubility in many common solvents, limited research has been conducted with PGA-based drug delivery devices. Instead, most recent research has focused on short-term tissue engineering scaffolds and the utilization of PGA as a filler material coupled with other degradable polymer networks. PGA is often fabricated into a mesh network and has been used as a scaffold for bone,27–30 cartilage,31–33 tendon,34,35 tooth,36 vaginal,37 intestinal,38 lymphatic,39 and spinal regeneration.40 While there has been research conducted into a wide range of applications, there exists significant issues with PGA. Rapid degradation leads to loss of mechanical strength and significant local production of glycolic acid. While glycolic acid is bioresorbable by cells via the citric acid cycle,41 high level of glycolic acid have been linked to a strong, undesired inflammatory response.42–44 In addition, PGA has mechanically failed as a biomaterial when used to facilitate colonic anastomosis formation38 and prevent intrapericardial adhesions.45
Polylactide
Since polylactide (PLA) possesses chiral molecules, polylactides come in four forms: poly(L-lactic acid) (PLLA), poly(D-lactic acid) (PDLA), poly(D,L-lactic acid) (PDLLA) – a racemic mixture of PLLA and PDLA, and meso-poly(lactic acid). As far as use in biomedical research, only PLLA and PDLLA have shown promise and have been extensively studied.
PLLA has a Tg of 60 – 65 °C, a melting temperature of around 175 °C and a mechanical strength of 4.8 GPa.46 The additional methyl group in PLA causes the polymer to be much more hydrophobic and stable against hydrolysis than PGA. High molecular weight PLLA has been shown to take greater than 5 years to be completely resorbed in vivo.47 Due to the slow degradation time, limited research has been recently conducted into drug delivery by PLLA systems alone.48–51 In order to reduce degradation time, investigators have either developed modification techniques or have blended or copolymerized PLLA with other degradable polymers. One interesting modification technique has been through the use of radiation.52,53 This process works by creating radicals in the ester alpha carbon which upon rearrangement shortens the polymer backbone through the removal of an ester bond and the release of carbon dioxide. Recombination of carbon radicals induces branching and cross-linking causing a decrease in crystallinity due to the modified polymers possessing unique molecular architectures. Shortening of the polymer and decrease in crystallinity work in concert to facilitate more rapid device degradation. While this process is very predictable, allowing for fine tuning of PLLA degradation behavior, even heavily irradiated PLLA is not completely absorbed until months after being delivered in vivo. This holds promise for drug eluting depots (i.e. birth control delivery devices), but more research must be done in order for PLLA to be more widely used for short-term controlled delivery applications. Under the product name Fixsorb®, PLLA has been used as a bone fixator.54 PLLA has also been extensively utilized in tissue engineering applications ranging from scaffolds for bone,55–58 cartilage,59,60 tendon,61 neural,62,63 and vascular 64 regeneration. Specifically, Dr. Peter Ma’s group has produced some very exciting research on the design of PLLA-based patient specific scaffolds (Fig. 2).65 A CT image of a digit was converted to a 3-D structured wax mold through layer-by-layer printing. By utilizing a solvent-extraction process and paraffin spheres, a 3-D PLLA scaffold that matched the structure of the digit was manufactured. The authors were able to uniquely control the nano-, micro- and macro-structure of the scaffold. This promising technique has the potential to be used for a number of different degradable polymers and parameters could be optimized for a wide-range of applications. Composite materials include PLLA combined with PDLLA,66 poly(lactide-co-glycolide),67 poly(caprolactone),68,69 poly(ethylene glycol),70 collagen,71 and chitosan.72
Figure 2.

Conversion of CT images into micro- and nanostructure controlled PLA scaffolds. A CT image of a hand (left) with a non-traditional defect (shown in purple) is converted into a wax mold which can be filled with PLA to create a scaffold with controllable pore size on the micro scale (center) and fiber size on the nano scale (right). (reprinted from 65 with permission from Elsevier.)
PDLLA is an amorphous polymer due to the random positions of its two isomeric monomers within the polymer chain yielding a slightly lower Tg of 55 – 60 °C and lower mechanical strength of 1.9 GPa.23 While possessing more desirable degradation properties than PLLA, PDLLA still takes over a year to properly erode which has kept it from being researched as a particle-based delivery vehicle. Instead PDLLA has been commonly used as a drug delivery film for inorganic implants,73–76 and as a tissue engineering scaffold.77–79 Like PLLA, PDLLA has been often combined with other degradable polymer like poly(lactide-co-glycolide),80 poly(ethylene glycol),81,82 and chitosan83 to create composites with desirable material properties.
Poly(lactide-co-glycolide)
Random copolymerization of PLA (both L- and D,L-lactide forms) and PGA, known as poly(lactide-co-glycolide) (PLGA), is the most investigated degradable polymer for biomedical applications and has been used in sutures, drug delivery devices and tissue engineering scaffolds. With a number of commercial manufacturers and easy polymer processability, researchers do not have to be polymer synthesis experts in order to utilize PLGA in their work. One particular advantage is that since PLA and PGA have significantly different properties, careful choice of copolymer composition allows for optimization of PLGA for intended applications. Property modulation is even more significant for PLGA copolymers since with 25 – 75% lactide composition, PLGA forms amorphous polymers which are very hydrolytically unstable compared to the more stable homopolymers.41,84 This is evident in the degradation times of 50:50 PLGA, 75:25 PLGA, and 85:15 PLGA being 1–2 months, 4–5 months and 5–6 months, respectively.85
PLGA has been used as a suture material since 197486 under the product name Vicryl® (Ethicon), a 10:90 PLGA braided construct. More recently a modified version, Vicryl Rapide®, has come to market. Vicryl Rapide® degrades much more quickly than traditional Vicryl® since it is irradiated during production. Panacryl® (Ethicon) is another product, which has a higher LA/GA ratio (90:10) than Vicryl®, which undergoes more rapid degradation. Unfortunately, Panacryl® has seen a significant drop in recent use due to public concern that it induces significant inflammation after implantation even though a recent report refutes this argument.87 While Ethicon produces the most widely used PLGA sutures, Polysorb® (Syneture) and Purasorb® (Purac Biomaterials) are also commonly used suture materials composed of PLGA.
With rapid degradation compared to other polyesters, PLGA has been utilized extensively in drug delivery applications. PLGA has been used to deliver chemotherapeutics,88,89 proteins,90–92 vaccines,93–95 antibiotics,96–98 analgesics,99,100 anti-inflammatory drugs101,102 and siRNA.103–105 Most often PLGA is fabricated into microspheres,90–95,97,98,101 microcapsules,106–108 nanospheres88,89,95,96,100 or nanofibers109,110 to facilitate controlled delivery of encapsulated or adsorbed payloads. Depending on the composition of the PLGA used and the interactions between payload and polymer, drug or protein release profiles can vary.54 Unfortunately, bulk erosion of the polymer prevents significant modulation of the release rate. Water diffusion in, payload dissolution and subsequent diffusion out is not controlled by polymer degradation rate and often PLGA delivery devices have a significant bolus release of their payload. Additionally, hydration of the entire matrix can often damage or deactivate hydrolytically sensitive encapsulated materials through constant water exposure and the high acidity of PLGA degradation products.111,112 The use of surface eroding polymers is better for zeroth-order and controlled release kinetics as well as payload protection.
PLGA demonstrates great cell adhesion and proliferation properties making it an excellent candidate for application in tissue engineering. PLGA has been fabricated into scaffolds by a number of different techniques including gas foaming,113,114 microsphere sintering,115–117 porogen leaching,118–120 electrospinning,121–124 polymer printing,125,126 or a combination of these techniques80,127,128 in order to create unique nano- and microstructured materials that can facilitate tissue development. Polymer printing in particular is a novel technique that holds great promise in the design of tissue engineering scaffolds. Dr. James Dunn’s group has demonstrated the capacity of 3D printing with PLGA.126,129 As shown in Fig. 3, very complex designs with controllable features can be generated to mimic structured tissue like villi for smooth muscle tissue engineering.126 The ability to utilize this technology with other degradable polymers holds promise in allowing for the design of organ-like structures that until now have been impossible to replicate. PLGA scaffolds have been used in the engineering of bone,10,79,115,116 cartilage,60,92,117 tendon,117,123,130,131 skin,108,122,132 liver,133–135 and nerve tissue.136–138
Figure 3.

PLGA scaffolds with villi architecture generated by indirect three-dimensional printing with villus diameter, height and intervillus spacing of (a) 0.5, 1, 0.5 mm; (b) 0.5, 1, 1 mm; (c) 1, 1, 1 mm, respectively. (reprinted from 126 with permission from Wiley.)
Polyhydroxyalkanoates
Polyhydroxyalkanoates are biodegradable polyesters that can be produced by both bacterial and synthetic routes. The most common polymer is poly(3-hydroxybutyrate) (PHB), a semi-crystalline isotactic polymer that undergoes surface erosion due to the hydrophobicity of the backbone and its crystallinity.139 PHB has a glass transition temperature around 5 °C and a melting temperature from 160 – 180 °C.140 Hydrolytic degradation of PHB results in the formation of D-(−)-3-hydroxybutyric acid, a normal blood constituent.141 The biocompatibility, processibility and degradability of PHB make it an excellent candidate for use in long-term tissue engineering applications142–147 Unfortunately, the stability of PHB makes it a poor candidate for controlled delivery applications.
To widen the applicability of PHB as a biomaterial, most commonly PHB is copolymerized with 3-hydroxyvalerate to create PHBV. PHBV is less crystalline than PHB with a lower melting temperature of 80 – 160 °C and a glass transition temperature in the range of −5 – 20 °C depending on HV content.148 PHBV has been used in tissue engineering of bone,147,149,150 cartilage,151 tendon,152 skin,143,153 and nerves.143,147 While the addition of HV content improves the biomaterial potential of PHB, rate of degradation is still too low for other biomedical applications. Significant research is underway to speed degradation rates through copolymerizing or blending PHB or PHBV with PLLA,153 PDLLA,154,155 PLGA,114,156–158 poly(dioxanone),159 poly(caprolactone),160–162 and polyethers.163–165
Polycaprolactone
Polycaprolactone (PCL) is a semicrystalline polyester with great organic solvent solubility, a melting temperature of 55 – 60 °C and glass transition temperature of −54 °C.166 Due to PCL’s very low in vivo degradation rate and high drug permeability, it has found favor as a long-term implant delivery device. Capronor® is a commercial contraceptive PCL product that is able to deliver levonorgestrel in vivo for over a year and has been on the market for over 25 years.167 Current research is being conducted into the development of micro- and nano-sized drug delivery vehicles, but the degradation rate (2–3 years) is a significant issue for pure PCL products to be FDA approved for this use. Instead PCL is often blended or copolymerized with other polymers like PLLA,68,168,169 PDLLA,170,171 PLGA104,172,173 and polyethers174–176 to expedite overall polymer erosion.
While somewhat limited in drug delivery applications, tissue engineering implications of PCL are numerous. PCL has low tensile strength (~23 MPa), but very high elongation at breakage (4700%) making it a very good elastic biomaterial.41 PCL’s processability allows for the formation of scaffolds composed of adhered microspheres177,178 electrospun fibers,179–181 or through porous networks created by porogen leaching.182–184 PCL and PCL composites have been used as tissue engineering scaffolds for regeneration of bone,182,185,186 ligament,187,188 cartilage,133,189 skin,177,181,190 nerve,184,191,192 and vascular tissues.183,193,194 A recent advancement using PCL hybrid scaffolds has been used in interfacial tissue engineering. Lee and coworkers have shown that if distinct scaffold regions are seeded with appropriate cells harvested from cartilage or ligament sources (Fig. 4), complex tissue interfaces like the bone-ligament interface can be regenerated.195
Figure 4.
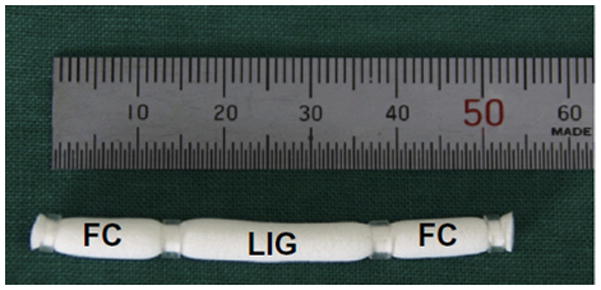
Cylindrical porous poly(L-lactide-co-caprolactone) scaffold loaded with fibrochondryocytes (fibrocartilage sections) on either end with fibroblasts (ligament section) in the center in order to mimic the ligament-bone interfacial tissues. (reprinted from 195 with permission from Elsevier.)
Poly(propylene fumarate)
Poly(propylene fumarate) (PPF) is a high-strength polymeric biomaterial that while technically a polyester, it possesses the unique ability to be cross-linked through the unsaturated bonds in its backbone. Since PPF can be cross-linked, polymer degradation is dependent on molecular weight, cross-linker and cross-linking density.196 PPF is a liquid injectable which becomes solid upon cross-linking, therefore it has found favor in biomedical applications such as filling bone defects197–199 and the depot, long-term delivery of ocular drugs.200–202 For osteogenic tissue engineering, PPF is often mixed with ceramics like hydroxyapatite203–205 or alumoxane206–208 to create stronger, more-bioactive scaffolds. Recent research has focused on the use of PPF to fill irregular shaped bone defects like ear ossicle209 or mandibular defects.210 In both circumstances PPF-based scaffolds allow the design of structures that may not be attainable from non-cross-linkable degradable polymers.
Polyanhydrides
Polyanhydrides are a class of surface eroding polymers that contain two carbonyl groups bound together by an ether bond and have been almost exclusively studied for biomedical applications. While originally developed as textile fibers in the 1930s, their hydrolytic instability precluded their wide-spread usage.211 Beginning in the 1980s, polyanhydrides were investigated for the biomaterial potential,212 eventually leading to their FDA approval as drug delivery vehicles in 1996.213 One particularly unique property of polyanhydrides is that the degradation of the anhydride bond is highly dependent on polymer backbone chemistry. In fact degradation rate can vary by over six orders of magnitude based on monomer chemistry (Table 1). Surface erosion and control over degradation rate allows for precision tuning of payload release rate which is why polyanhydrides have found significant favor in drug delivery applications. Polyanhydrides have been used for the delivery of chemotherapeutics,214,215 antibiotics,216,217 vaccines,218–221 and proteins.106,222,223 Polyanhydrides are often fabricated into microparticles106,218,220,222 or nanoparticles214,215,219,221,223 to allow for injectable, oral or aerosol delivery. Aliphatic homo-polyanhydrides, such as poly(sebacic anhydride) (pSA), have been found to have limited applications due to their rapid degradation. In order to retard polymer degradation and extend payload delivery, aliphatic diacid monomers have been copolymerized with hydrophobic aromatic diacid monomers218,222,224–226 or aliphatic fatty acid dimers (FAD).227,228 By varying aromatic monomer content, Determan and coworkers have fabricated polyanhydride particles that release their payload over a few days to a couple of years, as shown in Fig. 5.19 Additional research in polyanhydrides has focused on novel monomer development to allow for the replacement of SA due to its high acidity in solution (pH 4.2).229 A monomer of interest is 1,8-bis-(p-carboxyphenoxy)-3,6-dioxaoctane which is an aromatic diacid monomer that contains a triethylene glycol backbone that together convey amphiphiliticity.230,231 Copolymerizing this polymer with traditional aromatic diacid monomers yields degradable polymers that can be used as long-term delivery vehicles that have shown to stabilize acid-sensitive payloads like recombinant proteins.220,223
Figure 5.
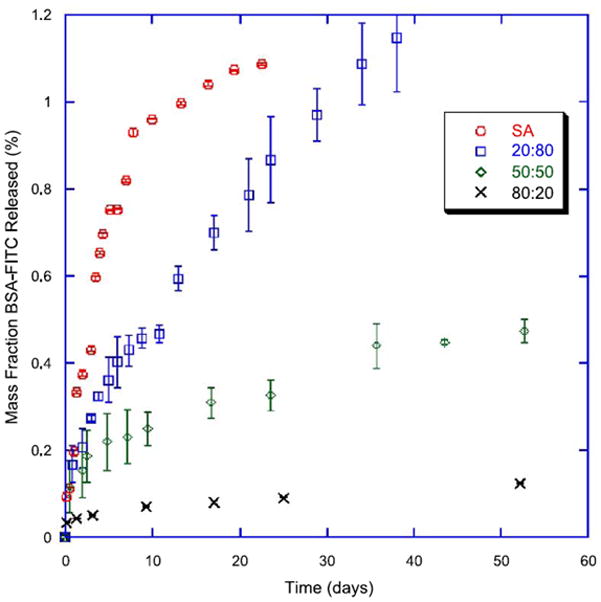
In vitro release of bovine serum albumin from poly(sebacic anhydride-co-1,6-bis-p-carboxyphenoxy hexane) microparticles in phosphate-buffered saline (pH 7.4). (reprinted from 19 with permission from Elsevier.)
While polyanhydrides have been extensively investigated for drug delivery applications, their low molecular weights yield poor mechanical properties precluding their use in tissue engineering. In order to increase their strength, methacrylated polyanhydrides have been studied as injectable, cross-linkable biomaterials. Methacrylic groups are typically incorporated by reacting diacids with methacryloyl chloride to create dimethacrylate monomers.232 Dimethacrylate monomers exist as liquids (i.e. dimethacrylic sebacic anhydride) or soft solids (i.e. dimethacrylic 1,3-bis-p-(carboxyphenoxy)hexane) which can be injected and cross-linked into solid scaffolds for tissue engineering applications.233 Cross-linked polyanydrides have been mostly studied for utilization in drug delivery and structural support for bone tissue engineering,234,235 but due to monomer flexibility have the potential to be used in other tissue engineering applications as well.
Polyacetals
Polyacetals are degradable polymers in which two ether bonds are connected to the same carbon molecule (geminal). The molecular closeness of the normally stable ether bonds conveys hydrolytic instability close to that seen for polyanhydrides (Table 1) and gives polyacetals surface eroding properties. Polyacetals are normally subdivided into two subgroups: polyacetals and polyketals. While all geminal-diether polymers are technically polyacetals, the namesake is normally reserved for polymers with only one of the two other geminal bonds possessing an R group. Polyketals instead have both other geminal bonds with R groups. Both polyacetals and polyketals have gained traction in biomedical research recently since their degradation products possess no carboxylic acids yielding significantly milder pH microenvironments236 and their degradation is acid-catalyzed.236–238 Milder pH microenvironments allow for the delivery of acid- and hydrolytically-sensitive payloads. Dr. Niren Murthy’s group has shown acid-catalyzed degradation allows for intracellular payload delivery, since particle-based delivery vehicles are stable under normal physiological pH (7.4), but rapidly degrade when they reach lysosomal pH (4 – 5), as shown in Fig. 6.237 So far polyketal microparticles and nanoparticles have been used to directionally deliver siRNA,238 DNA,239 and proteins237,240–244 in the treatment of acute inflammatory disease,237,242 ischemic heart disease,244 and cancer,241 as well as in the delivery of vaccines.240,243
Figure 6.
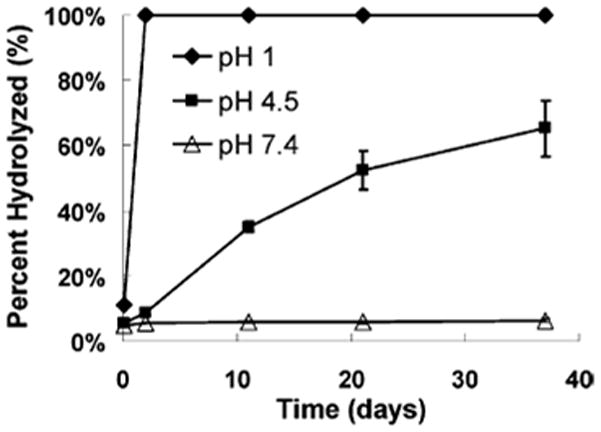
In vitro hydrolysis of poly(cyclohexane-1,4-diyl acetone dimethyl ketal) is greatly influenced by surrounding pH evidenced by its half-life of 24.1 days in pH 4.5 and 4 years in pH 7.4. (reprinted from 237 with permission from the American Chemical Society.)
For most implant applications, polyacetals have found limited use since they are often unable to be synthesized at high enough molecular weights to meet mechanical strength needs. A notable exception is Delrin® (polyoxymethylene) which is the homopolymer of formaldehyde that can be polymerized by acid or anionic catalysis to high molecular weights. Delrin®-based implants found favor as tilting disc valves in the repair of faulty heart valves in the late 1960s.245 Unfortunately, it was found that these implants swelled when they were used in vivo and other materials have since replaced Delrin® in artificial valves.246 Also, the degradation product of Delrin® is formaldehyde which is toxic. In order to create tissue engineering scaffolds from polyacetals, cyclic polyacetal monomers with two ester acrylate end groups have been synthesized that can then be crosslinked.247 Cyclic polyacetal homopolymers and those copolymerized with poly(ethylene glycol) diacrylate have shown preliminary promise as osteogenic biomaterials for bone tissue engineering.248,249
Poly(ortho esters)
Poly(ortho esters) are hydrophobic, surface eroding polymers that have three geminal ether bonds. Like polyacetals, control of poly(ortho ester) backbone chemistry allows for the synthesis of polymers with varied acid-catalyzed degradation rates and material properties. They have been specifically developed for drug delivery applications by the ALZA Corporation in the early 1970’s.250 Research in poly(ortho esters) is still ongoing in the academic community. While four classes of poly(ortho esters) have been developed, inherent issues with POE I – III have led to nearly all research focusing on POE IV. POE IV incorporates short segments of lactic or glycolic acid into the polymer backbone in order to expedite degradation since POE I – III possess much too slow erosion rates to be clinically relevant as drug delivery vehicles.250 POE IV polymers have been used for the delivery of analgesics,251 DNA vaccines252,253 and antiproliferative drugs.254 There capacity to be used as tissue engineering scaffolds is limited due to their weak mechanical properties and their capacity to induce a mild to moderate inflammatory response.255
Polycarbonates
Polycarbonates are linear polymers that have two geminal ether bonds and a carbonyl bond. While this bond is extremely hydrolytically stable (Table 1), research has shown in vivo degradation to be much more rapid presumably due to enzymatic degradation which causes these polymers to be surface eroding.256 The most extensively studied polycarbonate is poly(trimethylene carbonate) (PTMC) which has a Tg of −17 °C.257 PTMC is an elastomeric aliphatic polymer with great flexibility and a slow degradation profile, but poor mechanical strength. Its degradation into biocompatible, non-acidic 1,3-propanediol and carbonic acid make it an ideal candidate for drug delivery applications. PTMC has been fabricated into microparticles,258,259 discs,260,261 and gels262–264 for the delivery of angiogenic agents264 and antibiotics.260,261 To enhance the delivery potential of PTMC it is often copolymerized with PLA,265,266 PCL,265 polyether,266–268 or poly(L-glutamic acid)269,270 to allow for the fabrication of sutures,265 micelles,266–268 and polymersomes267,269,270 with superior mechanical and degradation properties for delivery of chemotherapeutics266–268,270 and antibiotics.265
In addition to PTMC, new polycarbonates have been recently researched for tissue engineering applications. One approach in order to create stiffer polycarbonates than PTMC is the use of cyclohexane or propylene instead of trimethylene in the monomer backbone.271 Another approach has been attaching bulky side groups through an ester bond to the β-carbon of the backbone.272 A particularly interesting novel polycarbonate has been created using the glucose metabolism intermediate dihydroxacetone (DHA).273 When DHA is copolymerized with methyl poly(ethylene glycol), the resulting rapidly-gelating, rapidly-degrading (100% mass loss within days) copolymer has been shown to assist the body in clotting through the development of new vascular tissue274 and the prevention of seromas, fluid filled gaps commonly created following ablative or reconstructive surgeries.275 The continued exploration of new polycarbonates holds potential for the expansion of this class of degradable polymers in biomedical applications.
Other polycarbonates that are used as fixators and in tissue engineering scaffolds are tyrosine-derived polycarbonates. These polymers are variations of poly(amino acids) in which amino acid like backbones are connected by carbonate bonds giving them strong mechanical properties while maintaining the biocompatibility of their degradation products. The most extensively studied of these pseudo poly(amino acids) are poly(desaminotyrosyl-tyrosine alkyl ester carbonates) (PDTEs). Due to aromatic groups in the polymer backbone, PDTEs possess significant mechanical strength allowing for their use in load-bearing applications. PDTEs have a variable pendant alkyl chain allowing for modulation of their thermal and mechanical properties with Tgs of 50 – 81 °C, Tms of 75 – 118 °C, tensile strengths of 50 – 70 MPa and stiffnesses of 1 – 2 GPa.276 Their processibility has allowed for fabrications of scaffolds composed of films,276–281 fibers,282 and gels.283,284 PDTEs have been investigated for their potential in tissue engineering of bone,277,278,280 vasculature,283 and muscle.281 Slow degradation (Mw half life of over 200 days285) and minimal mass loss of PDTEs allows for them to maintain their physical properties for very long times making them good candidates for slow regenerative processes.
Polyurethanes
Polyurethanes are biocompatible, biostable, moldable, strong polymers that possess ester bonds with geminal amide bonds that have a degradation rate similar to polyesters and polycarbonates (Table 1). They are typically synthesized by polycondensation of diisocyanates with alcohols and amines.286 Polyurethanes are composed of hard and soft segments that can undergo microphase separation allowing for these polymers to handle physical stresses very well.287 Polyurethanes have been used extensively in prostheses like cardiac assist devices,288 small vascular shunts 289 and tracheal tubes.290,291 A commercial polyurethane product, NovoSorb™ (PolyNovo®), is a two component system that cures in situ. The self setting system is an injectable liquid that polymerizes at physiological temperatures creating a biomaterial that has been shown to be mechanically similar to bone cements, but also promotes favorable cell adhesion and proliferation.292 Under most conditions pure polyurethanes are degradation resistant making them poor candidates for drug delivery and many tissue engineering applications. In order to expand the biomedical potential of polyurethanes they have been utilized in multi-degradable group or combination polymers which will be discussed later in this review.
Polyphosphazenes
Polyphosphazenes are a unique class of degradable polymers in that their backbone is completely inorganic consisting of phosphorous and nitrogen bonded linearly through alternating single and double bonds. While these polymers have been synthesized with high molecular weights since the mid 1960s,293 only in the past two decades have they been investigated in biomedical research.294 What distinguishes these materials is that they are highly flexible both physically and chemically. With two phosphorous side groups open to conjugation via esterification, etherification or amidification, over 500 different polyphosphazenes have been synthesized to date.295 While the phosphonitrilic backbone is not intrinsically hydrolytically sensitive, careful choice of side groups greatly impacts the degradation rate (Table 1). Certain side groups, like amino acid esters, glucosyl, glyceryl, glycolate, lactate and imidazole, have been found to sensitize hydrolysis of the backbone to allow for the design of clinically relevant biomaterials. In addition to control over degradation rates, physical properties of the polymer are also greatly affected by side group substitution. By changing disubsituted polyphosphazene side groups for one particular system, thermal and mechanical properties were greatly varied with Tg −10 – 35 °C, contact angle 63° – 107°, tensile strength 2.4 – 7.6 MPa, and modulus of elasticity 31.4 – 455.9 MPa.296 Another unique feature is that polyphosphazenes degrade into neutral products that have been found to have a pH buffering effect when combined with polymers, like polyesters, that have highly acidic degradation products.297 A commercially available product Polyzene-F® (poly[bis(trifluoroethoxy)phosphazene], CeloNova BioSciences) has shown tremendous potential as stent coatings298 and embolizing microspheres,299 and was FDA approved in 2008. While original research found the material to cause limited inflammation,300 a more recent study showed a significant, sustained foreign body response that is of concern.301 More research into this phenomenon is warranted before Polyzene-F® can be used clinically.
Polyphosphazenes have shown significant promise in drug delivery and tissue engineering applications. The large library of side groups and the processibility of polyphosphazenes has allowed for them to be fabricated into particles,302–304 micelles,305–309 microneedle coatings,310,311 and gels.312–315 They have been used in the delivery of anti-inflammatory drugs,306 chemotherapeutics,307,309,315 growth factors,313,316 DNA,302–304 proteins,312,314 and vaccines.305,308,310,311 One particular promising application has been in the design of biodegradable microneedle coatings to deliver vaccines.310 Andrianov and coworkers have shown polyphosphazene-vaccine covered metallic cones can be used to pierce the skin to deposit rapidly degrading polymer films (~90% in 15 minutes) into the dermis layer (Fig. 7). An added benefit of utilizing polyphosphazene as the delivery vehicle is that prior research has found that certain polyphosphazenes are strongly immunoactivating and hold great potential as adjuvants, non-specific immune boosting substances.305,308 This research has shown the induction of stronger immune responses than comparable intramuscular injections without the pain and strong inflammation seen with traditional needle use providing promise for this new technology.
Figure 7.
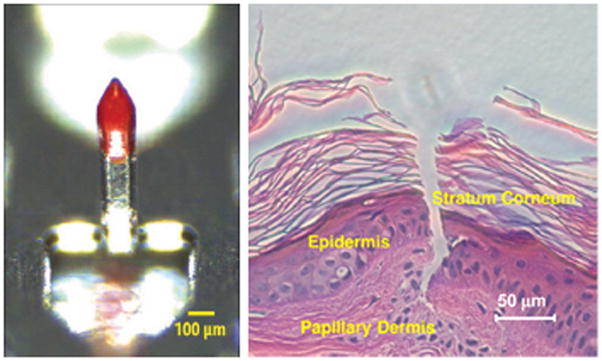
Optical microscopy image of a polyphosphazene coated metallic microneedle (left) and histological section of porcine skin after coated microneedle insertion (right). (reprinted from 310)
While many rapidly degrading polyphosphazenes have shown promise in drug delivery applications, more hydrophobic side group substitutions have allowed for the use of polyphosphazenes in tissue engineering applications. Polyphosphazene scaffolds have been composed of films,317,318 fibers,319–322 and sintered microspheres.323 These matrices have been used to assist in nerve regeneration,317,319,322 but recent research has almost exclusively focused on orthopedic applications of polyphosphazenes.318,320,321,323 Often polyphosphazenes have been blended with polyesters to increase mechanical strength and provide a moderate pH microenvironment for developing tissues. Interestingly, miscible blends of poly[(glycine ethyl glycinato)1 (phenylphenoxy)1 phosphazene] and PLGA that are originally cast as films undergo varied rates of degradation of the principal components under aqueous conditions.13 As the PLGA degrades and erodes from the matrix the more hydrophobic polyphosphazene reforms into a scaffold with a microstructure very similar to adhered microspheres. Porous scaffolds of adhered microspheres have been used heavily in tissue engineering applications since they allow for the infiltration of host cells into the scaffold that will eventually become new tissue. Having biomaterials with the strength and flexibility of films that can form into porous scaffolds over time greatly enhances the biomedical potential of these materials.
Polyphosphoesters
Polyphosphoesters form another interesting class of biomaterials that is composed of phosphorous-incorporated monomers. These polymers consist of phosphates with two R groups (one in the backbone and one side group) and can be synthesized by a number of routes including ring opening polymerization, polycondensation, and polyaddition. Originally developed in the 1970s,324,325 polyphosphoesters have great biocompatibility and similarity to biomacromolecules like RNA and DNA. Relatively rapid hydrolytic cleavage (Table 1) of the phosphate bonds in the backbone leads to the production of bioresorbable or excretable phosphates, alcohols and diols. While a commercial polyphosphoester-based microsphere delivery device (PACLIMER®) has shown promise in Phase I/II trials for the treatment of ovarian and lung disease, MGI Pharma discontinued further research with the product after purchasing the original development company, Guilford Pharmaceuticals. Polyphosphoesters are divided into two different classes: polyphosphonates (alkyl/aryl R groups) and polyphosphates (alkoxy/aryloxy R groups). Due to the flexibility in choosing R groups, polymers of significantly varying physical properties and degradation rates can be synthesized. In order to enhance physical properties, polyphosphoesters are commonly copolymerized with polyethers and polyesters. Polyphosphoesters and polyphosphoester composites have shown significant promise as chemotherapy326–328 and DNA,329–331 delivery devices. Polyphosphoesters have also been utilized as scaffolds in the engineering of bone tissue.328,329,332–335 These polymers have been formed into particles,328,330 micelles,326,327 films329,331 and gels332–335 for these applications. Recent research utilizing polyphosphoesters has been limited, but their chemical flexibility and similarity to biomacromolecules gives them great promise for future applications.
Combination Polymers
A growing trend in degradable polymer research is the development of combination polymers. These are polymers in which monomers contain multiple degradable groups. Unlike copolymerization of different monomers, the molecular proximity of these groups yields functionally novel biomaterials. These materials often have properties that cannot be obtained by single degradable group polymer families or through simple copolymerization. With the goal of developing new polymers, this research area has yielded a large number of new families which will not be completely reviewed in this section. Below are a couple examples of interesting degradable combination polymer families that have been recently developed.
Poly(ester ether)s are an interesting class of degradable polymers in which typically an ether bond in incorporated into the backbone of a polyester in order to expedite hydrolytic cleavage of the ester bond. One very common poly(ester ether) is polydioxanone (PDO). PDO is a colorless, semicrystalline polymer synthesized by ring- opening polymerization of p-dioxanone that has a Tg about −10 to 0 °C and a Tm of 115 °C.336 While quicker degrading than longer aliphatic polyesters of similar backbone length, PDO can still be considered a slow degrading polymer (6 – 12 months for complete mass loss).23 With a low modulus (1.5 GPa),23 but good flexibility and strength maintenance (1 – 2 months) PDO has been commercialized as the monofilament suture PDS® for nearly 30 years.337 In an effort to create faster degrading polyesters, research has been conducted into the synthesis of poly(ether ester)s via polycondensation. This is carried out by the use of a catalyst to create alternating blocks from dicarboxylic acids and diols. Oligomeric ethylene glycol (n = 2 – 4) and trans-β-hydromuconic acid are condensed to create low molecular weight poly(ether ester)s (4,000 – 6,000 g/mol) that are amorphous and have low Tgs (−36 – −32 °C).338,339 These polymers are liquid at room temperature and possess cross-linkable double bonds in their backbone making them promising biomaterials for filling non-uniform defects. By creating random copolymers between cross-linkable and non-cross-linkable (adipic acid) monomers, control over cross-linking density can be obtained. These materials are able to be synthesized with Young’s Moduli varying three orders of magnitude (0.02 – 20 MPa).339 Also, the liquid poly(ester ether)s are able to be fabricated into complex architectures not easily obtainable by solid polyesters.339
Poly(amide ester)s are cationic, degradable polymers originally investigated for their biomedical potential in the 1990s by Robert Langer.340 The most widely studied sub-group of these polymers are poly(β-amino esters) (PBAEs) which are synthesized by a Michael Addition reaction of diester diacrylates and primary or secondary amines.341,342 Due to having control over both components’ backbone chemistry a wide range of PBAEs can be synthesized. The fidelity of the reaction and the library of polymers that can be synthesized have led to the use of combinatorial research methods to identify polymers that show particular promise as biomaterials.343–346 In one particular study, over 2,000 polymers were synthesized and screened combinatorially.345 PBAEs show tremendous promise in DNA delivery due to their positively charged amide bonds347–350 and in tissue engineering since they can be synthesized with high molecular weights and hydrolytically stable bonds allowing for long-term maintenance of their mechanical strength.351–353
While copolymers and blends of polyesters and polyanhydrides have been studied for a few decades, the synthesis of poly(anhydride ester)s for biomedical applications have only been investigated for the last decade.354 The original impetus for their development was the creation of prodrug polymers. If commonly used aromatic diacid monomers have two internal ester bonds instead of ether bonds, their degradation products are resorbable carboxylic diacids and salicylic acid, a non-steroidal anti-inflammatory drug and the active compound of aspirin. Polycondensation of salicylate-containing diester diacids yields poly(anhydride ester)s very similar to classical polyanhydrides with moderate hydrophobicity, surface erosion and good processability.354,355 These polymers have been found to have Tgs lower than physiological temperatures (12 – 34 °C),354,356,357 and are often copolymerized with traditional aromatic diacid monomers to improve mechanical properties.356,358 Since this original discovery, salicylic acid-derived poly(anhydride ester)s and their composites have since been fabricated into particles,357,359 fibers,358 and films355,356,360,361 for biomedical application. They have been used in the prevention of biofilm formation,360,361 the prevention of bone resorption,356,359 and the creation of a local anti-inflammatory effect.356,360
Polyurethanes have shown some applicability in biomedical research, but their hydrolytic stability limits their potential. In order to expedite degradation, ester bonds have been introduced into the polymer backbone. Poly(ester urethane)s are typically synthesized by reacting diisocyanates with polyester diols or triols composed of glycolide, lactide or caprolactone to create the soft segments of the polymer.362 Often the hard segments are composed of polypeptides or diols or triols of 3-hydroxybutyrate in order to give biomaterials that are degradable, but still relatively hydrolytically stable.362,363 In order to better commercialize the aforementioned PolySorb™, more recent research has focused on including degradable ester groups into the injectable prepolymers. This has allowed for development of biomaterials that has shown promise in bone364 and articular cartilage regeneration.365 Another commercialized poly(ester urethane) is the highly porous Degrapol® (Ab Medica)which has shown promise in engineering tracheal soft tissue366,367 and bone tissue.368
ENZYMATICALLY DEGRADABLE POLYMERS
Enzymatically degradable polymers are materials that possess bonds that while technically hydrolytically sensitive, in reality require catalysis to undergo meaningful degradation under physiological conditions. Most of these polymers contain ether or amide bonds which have hydrolytic degradation rates much lower than the polymers discussed in the previous section (Table 1). This section details a number of these polymeric families and their application as biomaterials.
Synthetic Polyethers
Synthetically-derived polyethers are highly biocompatible polymers that have been used in polymeric drug delivery and tissue engineering for over 30 years.369 Nearly all biomedical research with synthetic polyethers has focused on the use of poly(ethylene glycol) (PEG) and poly(propylene glycol) (PPG), while a limited amount of work has been conducted using poly(tetrahydrofuran).370–373 Polyethers do not readily undergo hydrolytic degradation and while bacterial etherases have been discovered,374,375 human equivalents of these enzymes have yet to be identified. Instead polyether chains are normally dissociated from the biomaterial and removed via the excretory system. Due to a near absence of in vivo degradation and the fear of accumulation, it is recommended that polyethers with lower molecular weights be utilized for biomedical applications. In addition to issues associated with high molecular weight PEG, very low molecular weight PEG has been found to induce bodily harm. Specifically, tetraethylene glycol and PEG 200 have been shown to induce clastogenic effects (chromosomal disruption) in Chinese hamster epithelial liver cells.376 Most research with synthetic polyethers has focused on triblock Pluronic ([PEG]n-[PPG]m-[PEG]n), cross-linkable oligomers or these polymers in union with many of the previously mentioned hydrolytically degradable polymers.
Pluronic is of particular interest because PEG is hydrophilic while PPG is hydrophobic allowing for the formation of very small micelles (10 to 100 nm in diameter)377 by self assembly in water. These micelles allow for high payload loading (30 wt%)378 of hydrophobic drugs into the core while the hydrophilic shell makes the particles easy to administer. Drug delivery devices composed of Pluronic micelles have been used to deliver chemotherapeutics,379–381 antibacterials,382,383 antidiuretics,384 anti-inflammatory drugs,385 and DNA.386 Pluronic has also been formulated into hydrogels. These hydrogels possess relatively weak mechanical properties (maximum shear storage modulus of 13.7 kPa at 20 wt% Pluronic)387 making them candidates for drug delivery388–391 and soft tissue engineering.392,393
In order to enhance the mechanical strength of polyether-based hydrogels, cross-linkable oligomers are synthesized by reacting PEG with excess acryloyl chloride or methacryloyl chloride yielding the products PEG diacrylate (PEGDA) or PEG dimethacrylate (PEGDMA), respectively. PEGDA and PEGDMA can then be polymerized by their double bond yielding cross-linked networks which upon degradation yield poly(acrylic acid) or poly(methacrylic acid) and PEG. PEGDA hydrogels (shear storage modulus of 68 kPa at 20 wt%)394 and PEGDMA hydrogels (shear storage modulus of 125 kPa at 20 wt%)395 are stronger than their Pluronic counterparts. PEGDA, PEGDMA and PEGDA/PEGDMA hydrogels have been used in the delivery of chemotherapeutics,396 hormones,397 antibacterials398 and anti-inflammatory drugs399 as well as scaffolds in the engineering of cartilage,400,401 bone,402–404 endothelial,405 and vascular tissues.406
While synthetic polyethers have shown some promise when utilized by themselves, they have much greater biomedical potential when utilized in combination with other degradable polymers. PEG is commonly used to cap (PEGylation) or coat other degradable polymers in order to convey steric stabilization limiting the interactions between the device and the host. This is especially important in preventing phagocytosis, cellular uptake, of particle-based delivery vehicles.407–409 Unlike polyanhydrides and polyesters, PEG does not initiate the complement cascade which is known to facilitate particle phagocytosis.410 PEG incorporation has been used to enhance the biocompatibility and delivery properties of polyanhydrides,411–414 poly(ortho esters),415 PLA,416–419 PLGA,420–423 and PCL.424–427 Of particular interest is the work being conducted with poly(ether anhydride)s. The addition of a low molecular weight PEG shell to polyanhydride micro- and nanoparticles conveys “virus-like” behavior and allows for them to pass through mucosal membranes much more rapidly than uncoated particles.428,429 Being able to transport drug delivery devices across mucosal barriers is important in improving therapeutic efficacy against diseases like cystic fibrosis430 and lung cancer 414 (lung mucosa) and HIV431 (vaginal mucosa).
PEGDA and PEGDMA have also been heavily researched in composite systems with other acrylated and methacrylated degradable polymers, most commonly polyesters,432–434 polyanhydrides,232,234,235,435,436 and chitosan,437,438 a polysaccharide that will be discussed later in this review. The polyether diacrylate monomers give the cross-linked network hydrophilicity while the other degradable components convey significantly greater mechanical strength (compressive moduli as great as ~ 100 MPa)232 than cross-linked polyether diacrylate homopolymers. Greater strength and component flexibility has lead to the use of these composites in a wide range of tissue engineering applications including in the regeneration of ligament,433 cartilage,434 bone, 232,432 and epithelial tissue.437
Proteins and Poly(amino acids)
Proteins are essentially high molecular weight polymers composed of amino acid monomers joined by amide bonds. They often occur in three-dimensional folded structures and are one of the most common materials found in the human body. Proteins and amino acid-derived polymers have been utilized as biomaterials in sutures, scaffolds and drug delivery devices. While amide bonds are hydrolytically stable, the body possesses a wide-array of proteases that can rapidly degrade proteins.
Collagen
Collagen is the most abundant protein in the human body and is a major component of ligament, cartilage, tendon, skin and bone. It also forms the structural network of other tissues like blood vessels. Collagen is composed of polypeptide strands bearing tri-amino acid blocks of Glycine-X-Y where X and Y can be any of a number of different amino acids and are most commonly proline and hydroxyproline.439 These polypeptides are formed into left-handed triple helix microfibrils that organize together in a number of different architectures to create collagen fibers with appropriate mechanical properties for their function. To date, at least 28 different types of collagen have been identified, however types I, II, III, and IV are the most heavily investigated with over 90% of all collagen being type I.440
Collagen has been extensively researched for various medical applications due to its biocompatibility, mechanical strength and enzymatic degradability by collagenases and metalloproteinases.441 In addition, it is very processable with high solubility in acidic aqueous solutions allowing for the fabrication of collagen sponges,442–444 tubes,445,446 sheets,447 powders,448 and injectables.449–452 Collagen has been used for centuries as a suture material of which one form, catgut, is still sometimes utilized in surgery,453,454 but due to collagen suture’s increased infection rates and inflammation, synthetic sutures are much more commonly used today. Collagen has also been used as a depot payload delivery device in the local extended release of antibiotics,455,456 DNA,457,458 siRNA,459,460 and proteins.461–463 In each case collagen had a burst release due to bulk erosion making it less ideal than the aforementioned surface eroding hydrolytically-sensitive polymers for drug delivery applications.
More recently collagen has found use as a haemostatic sealant.464 Collagen is highly thrombogenic and plays a role in the body’s natural clotting process by activating fibrogen conversion into fibrin, heavily cross-linked mesh networks of fibrogen. Fibrin captures activated platelets to make a clot. By creating a collagen based sealent, wounds can be coated and blood flow halted much more quickly. The FDA has approved a couple of collagen-containing solutions, including Helistat® (Integra Life Sciences) and FloSeal® (Baxter), for the treatment of bleeding during surgery.
Due to collagen’s structural integrity conveyed by its fibrous nature, a majority of biomedical research using collagen as a biomaterial has focused on its potential as a tissue engineering scaffold, specifically in load bearing applications. Collagen sponges have been used as tissue supports and scaffolds for nearly 50 years.465 Due to collagen’s ability to withstand high tensile loads (92.5 MPa ultimate tensile strength),466 it has often been used in bone tissue engineering.467–469 Composites of hydroxyapatite and collagen are utilized since these materials closely mimic the composition of natural bone.470–472 Collagraft® (Angiotech Pharmaceuticals) is a synthetic bone-graft substitute composed of bovine type I collagen and hydroxyapatite/tricalcium phosphate granules which has been approved by the FDA and used clinically.473 Collagen has also been widely researched as a tissue engineering scaffold for cartilage,474–476 tendon,477–479 and ligament.480,481
While often used for load bearing applications, collagen’s biocompatibility and ease of use has lead to its investigation as a scaffold in skin engineering as well.482–484 Due to collagen’s ability to improve cellular adhesion and proliferation, burn treatment and reconstructive surgery is often conducted with an acellular collagen matrix taken from human cadavers which is marketed as Alloderm® (LifeCell). While Alloderm is the most commonly used commercial product for skin engineering and wound healing, several other FDA collagen-based products exist in the market place including Promogran® (Johnson & Johnson), Biobrane® (UDL Laboratories) and OrCel® (Ortec International) for this application. While these products provide excellent support to regenerating tissue, they lack many of the constituent structures found within skin like hair, nerves and glands as well as tissue layer separation necessary to truly replicate skin’s three-dimensional structure. Several approaches to these problems have been proposed, but two concepts of particular interest are outlined. In order to develop sweat glands within regenerated skin, Huang and coworkers have developed growth-factor releasing collagen microspheres that can support differentiating sweat gland cells.485 These constructs are then embedded into a keratinocyte/fibroblast co-culture with structural maintenance provided by a collagen matrix to create substructures within the developing skin that are maintained even after implantation in vivo (Fig. 8). In order to create keratinocyte-fibroblast separated layers, keratinocytes are normally cultured on top of a preformed fibroblast layer which is time-consuming and inefficient. A new method has been developed for creating controlled large pore scaffolds that allow for keratinocyte and fibroblast structures to separate naturally creating better skin mimicking constructs which are also well integrated into the collage scaffold.486 These advances hold promise not only for collagen-based biomaterials and skin engineering but for tissue engineering as a whole since as the field moves towards creating and regenerating more complex structures like organs three-dimensional patterning of multiple tissue types will be paramount to continued success.
Figure 8.
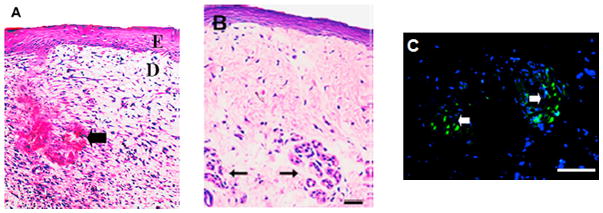
Photomicrographs of keratinocyte-fibroblast co-culture engineered skin tissue with the addition of collagen microsphere supported sweat gland cell constructs. (A) Hemotoxylin and Eosin (H&E) staining after two weeks of in vitro co-cultivation showed differentiated tissue layers (E: epithelium and D: dermis) with a bud-like structure (black arrow) where sweat gland constructs were loaded. (B) H&E staining of six week post-implantation in vivo skin tissue showed the continued presence of bud-like structures in the dermis layer (black arrow). (C) Fluorescence microscope observation showed DiO-positive cells (green) confirming the presence of still viable sweat glands. (reprinted from 485 with permission from Elsevier.)
In order to improve collagen’s potential as a biomaterial it has often been modified or combined with other degradable polymers. Modifications such as crosslinking,487–489 association of bioactive molecules,490,491 and enzymatically pre-treatment492–494 have all led to novel collagen-based materials with expanded functionality. In composite materials, collagen has been combined with PLA,60,495,496 PLGA,60,497,498 PCL,60,497,499 and chitosan.500–502 These multi-polymer constructs are typically either polymeric blends or intermixed devices (ex: microparticles of one polymer dispersed in a fibrous scaffold of another polymer).
While heavily researched, collagen possesses many negative attributes limiting its biomedical potential and clinical utility. Collagen-based biomaterials have been known to induce a moderate immunological response in vivo due to its terminal region composition and a series of antigenic sites in the central helix.503 The degree and nature of this response greatly depends on the source and post-processing of the collagen used. Other issues include the high cost of pure collagen, significantly varying physico-chemical properties and risk of transmitted infection from the grafting source. While current research is underway to produce recombinant human collagen,504 animals and cadavers still remain the most common sources. Until alternative biological sources are created or purely synthetic collagen can be synthesized, wide spread clinical acceptance and use of collagen-based biomaterials does not seem likely.
Elastin & Elastin-like Polypeptides
Elastin is an insoluble, highly elastic polymer composed of heavily cross-linked tropoelastin molecules that is a major component of vascular and lung tissue and is responsible for the contraction of these tissues following stress. Soluble tropoelastin molecules are produced intracellularly by smooth muscle cells and fibroblasts and are cross-linked extracellularly to form their elastic polymeric network.505 Since it is prevalent in vascular tissue, elastin has been found to not activate platelets making it a promising material for synthetic vascular grafts.506,507 While some biomaterials research has been conducted with elastin, its ability to elicit an immune response and its insolubility have limited its use.508
In order to overcome the limitations inherent to elastin, synthetic elastins have been developed. Soluble recombinant human tropoelastin can be molded, coaservated and cross-linked at 37 °C to create soluble elastin with controlled architecture.509,510 Also, tropoelastin undergoes an irreversible temperature transition (ITT) above 25 °C where its molecular organization goes from a disordered to ordered state. This transition gives synthetic elastin promise as a smart, injectable drug delivery system.511,512
Another attempt to utilize elastin-based materials as biomaterials has been in the investigation of elastin-like polypeptides (ELPs). ELPs are artificial polypeptides that are composed of pentapeptide repeats (VPGXG) similar to those found in elastin where X can be any of a number of different amino acids except proline.513 While very flexible like elastin, ELPs are biocompatible and non-immunogenic. They also can be synthesized to undergo ITT, as well as respond to pH, ionic strength and light based on which amino acid is synthesized in the X position.514 While once synthesized chemically, ELPs have been more recently produced in E. Coli.515 Due to the variety of phase transitions that ELPs can undergo, they have been investigated as delivery vehicles for chemotherapeutics,516,517 antibiotics,518 and proteins.519,520 Also, when cross-linked ELPs are seeded with chondrocytes they have been shown to possess dynamic sheer moduli (~ 1.7 kPa)521 similar to normal cartilage. The elastic behavior of ELPs makes them uniquely suited for the engineering of soft tissues.522–524
Albumin
Albumin is an abundant water soluble blood protein comprising almost 50% of total plasma mass in the body. Albumin carries hydrophobic fatty acids in the blood stream as well as carefully maintains blood pH. Since Albumin is essentially ubiquitous in the body, nearly all tissues have enzymes that can degrade it making it a promising polymer for biomedical applications.525 Albumin’s solubility allows for the protein to be easily processed into a number of different shapes including fibers,526,527 microparticles,528,529 and nanoparticles.530–532 Due to its serological compatibility and weak mechanical strength, albumin has been primarily investigated for payload delivery,529–531 coating,533,534 and suturing applications.535,536 Currently, a bovine albumin-based adhesive marketed as BioGlue® (CryoLife) is FDA approved for vascular surgery.535
Fibrin
Fibrin, a large cross-linked biopolymer composed of fibronectin, is involved in the natural clotting process. In the presence of the enzyme thrombin, cleavage of an internal fibrin linker yields linear fibrils that laterally associate into nanofibers (10 – 200 nm) that form a clot. This clot is able to be degraded by a complex cascade of enzymes.537 The use of fibrin as a biomaterial goes back centuries and it has been shown to be biocompatible, biodegradable, injectable and able to enhance cell proliferation.538 Fibrin glues has been studied as a surgical supplement under the market name Evicil® (Ethicon) which has been FDA approved as a tissue sealant and haemostatic agent. Fibrin glues are prepared as solutions containing thrombin and fibronectin separately that are mixed right before application. Thrombin rapidly crosslinks the fibronectin into a fibrin clot closing the wound. Fibrin has also been investigated for use as a drug delivery device539–542 and cell carrier.543–545 Due to its potential for cross-linking, fibrin can be uniquely modified so that its material properties can be tailored to the desired application.546
Natural Poly(amino acids)
Natural poly(amino acids) are biodegradable, ionic polymers similar to proteins in that they possess amide linkages, but poly(amino acids) are only composed of one type of amino acid. The two most commonly studied natural poly(amino acids) as biomaterials are poly(γ-glutamic acid) and poly(L-lysine).
Poly(γ-glutamic acid) (γPGA) is a water soluble, biodegradable polyamide composed of both enantiomeric D- and L-glutamic acid units commonly produced by a number of different bacteria.547–549 The biomaterial promise of γPGA centers on its reactive side carboxylate which allows for the covalent attachment of other functional groups and drugs. Benzyl ester,550 sulfonate,551 sulfide,552 and chemotherapeutic attachment553,554 have all allowed for its further development as a biomaterial. It has also been used in particle-based555–558 delivery of antibiotics,556 vaccines,557,558 DNA,555 and proteins.556 As a homopolymer it is too physically weak to be used in supportive tissue engineering scaffolds, but as a cross-linked hydrogel it has found some promise in soft tissue engineering.559,560 Often it is blended with other polymers like PLA,561,562 PLGA,563 PCL,564 collagen,565 and chitosan566–568 to give composites that are mechanically strong but possess some hydrophilicity. While promising, research into γPGA as a biomaterial has been limited due to its scarcity.
Like γPGA, poly(L-lysine) is generated by bacteria and is currently being investigated as a tissue engineering scaffold and drug delivery device. It has been shown to possess intrinsic antibacterial,569 antiviral,570 and antitumor activity571 that make it a very promising polymer. Unfortunately, it’s very high positive charge causes it to be rather toxic which has limited its applications. It has found some use being blended with other degradable polymer like PLA,572 PLGA,573,574 PCL,575 γPGA,576,577 and chitosan.578
Synthetic Poly(amino acids)
Synthetic poly(amino acids) have been investigated for a number of biomedical applications due to their similarity to naturally occurring proteins. While several homo-and co-poly(amino acids) have been synthesized and evaluated; high crystallinity, low degradation rate, unfavorable mechanical properties and immunogenicity have kept a majority of these polymers from being utilized clinically.579 However, two poly(amino acids) have been found to function as promising biomaterials: poly(L-glutamic acid) and poly(aspartic acid).
Poly(L-glutamic acid) (L-PGA) is different than γPGA since its amide linkage is made with the α-carbon amine group instead of the γ-carbon amine group. The shorter distance between amide bonds in L-PGA makes it more flexible than its γPGA counterpart. Also, synthetic techniques allow for L-PGA to be more easily produced. While L-PGA is typically synthesized as a linear polymer, the development of new synthesis techniques allows for the creation of unique architectures like dendrimers.580 L-PGA is very biocompatible and non-immunogenic and has been shown to be highly susceptible to degradation by lysosomal enzymes.581,582 Due to its negative charge at physiological pH L-PGA has found significant promise as a DNA delivery device.555,583 Also L-PGA’s negative charge allows for the construction of layer-by-layer (LBL) film assembly with negatively charged polymers like poly(L-lysine),584,585 and chitosan.586 LBLs are relatively new drug delivery devices which alternate layers of charged polymer allowing for the repeated delivery of either positively or negatively charged payloads holding significant promise for future biomedical applications. Like γPGA, the reactive side carboxylate of L-PGA allows for conjugation. So far this has been used for the creation of soluble, long-lasting polymer-chemotherapy conjugates,587,588 one of which is a Paclitaxel-conjugate marketed as OPAXIO® (Cell Therapeutics) that has shown promise in phase III clinical trials.589 Conjugates with small molecules have yielded degradable MRI contrast agents.590–592 Tian and coworkers have demonstrated the capacity of a L-PGA-gadolinium complex whose molecular weight keeps the contrast agent from diffusing out of the vasculature. When tested in rhesus monkeys, the vasculature contrast of subjects given the complex was much greater than those given a small molecular weight contrast agent (Magnevist) at 2 hours post-injection (Fig. 9).593 This complex allows for the use of less contrast agent while getting clearer MRI of small vasculature which can lead to earlier detection of tumors, atherosclerosis, and gross hemorrhage. L-PGA has also been combined with other degradable polymers like PCL594,595 and PTMC269,270 for drug delivery applications and collagen596 and chitosan597 to create novel tissue engineering scaffolds.
Figure 9.
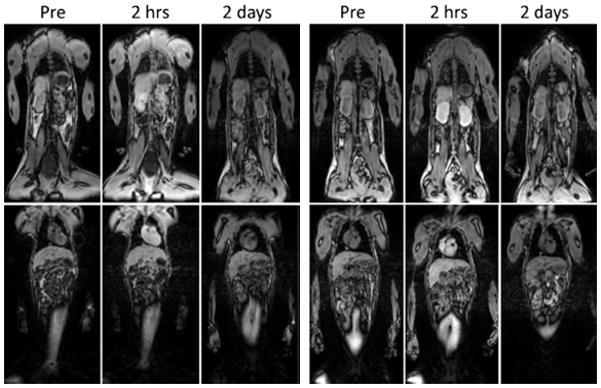
Magnetic resonance imaging of rhesus monkeys before contrast injection and at 2 h and 2 days after injection of Magnevist at 0.1 mmol Gd/kg (left) and PG-Gd 0.01 mmol Gd/kg (right). Enhancements of blood vessel, heart, kidney and liver are clearly visualized at 2 h after PG-Gd injection at a tenth of the dose of Magnevist. By 2 days, the contrast agent has been mostly cleared with both contrast agents. (reprinted from 593 with permission from Wiley.)
Poly(aspartic acid) (PAA) is a highly water-soluble ionic polymer with a greater carboxylate content than PGA or L-PGA since it has one less carbon atom in its backbone. Like poly(glutamic acid), PAA has been found to be degradable by lysosomal enzymes.598 It has been copolymerized with a number of degradable polymers (PLA,599;600 PCL,601 PEG,600–602 etc.) to create materials that form micellar structures which have shown promise as smart delivery vehicles. PAA can also be easily converted to a hydrogel by high energy radiation which has shown promise in biomedical applications.603
Polysaccharides
Polysaccharides are polymers composed of monosaccharide units joined together by glycosidic linkages, a type of ether bond. Their use as biomaterials has become much more common as new biological functions are identified for these materials. Also, the array of materials that can be investigated has increased due to new synthetic routes that have been developed for modifying polysaccharides. Their biodegradability, processability and bioactivity make polysaccharides very promising natural biomaterials.
Human Origin
Hyaluronic acid (HA) was originally isolated by Meyer and Palmer in 1934604 and has shown significant promise as a biomaterial. HA is a linear anionic polysaccharide consisting of alternating units of N-acetyl-D-glucosamine and glucuronic acid making it a member of the glycosaminoglycan family. HA is the largest polymer in the family and has been found in molecular weights up to a few million.605 It has been traditionally isolated from rooster combs and bovine vitreous humor. However, recent advances in microbiological techniques have led to the production of the first animal-free sodium hyaluronate which is synthesized by Bacillius subtilis and has been patented by Novozymes Biopharma. HA is water soluble and forms highly viscous solutions. Synovial fluid and vitreous humor have a large quantity of HA contributing to these tissues’ viscoelastic properties. HA also plays an important structural role in articular cartilage and skin.
HA possesses several properties that make it unique. It has been shown to scavenge free radicals,606 cause bacteriostasis607 and assist in tissue repair.608 These factors have made it a promising material for tissue engineering applications, but HA homopolymer is too weak and structurally fluid to create a supportive scaffold. To overcome this limitation, HA has been cross-linked with ethyl esters or benzyl esters yielding hydrogels which have been commercialized as HYAFF® (Fidia Farmaceutici). HYAFF® undergoes hydrolytic degradation that causes scission of the ester bond converting it back to hyaluronic acid. Depending on the extent of esterification, degradation rate can be varied from 1–2 weeks609,610 to 4–5 months.611,612 HYAFF® hydrogels are extremely versatile as shown by their capacity to be fabricated into sheets,613 membranes,614 sponges,615 tubes612,616,617 or fibers618,619 and their usefulness as scaffolds for wound healing618 and the regeneration of the trachea,613 articular cartilage,619 nasal cartilage,615 respiratory epithelium,614 vasculature,612,616 and nerve tissue.617 Other hyaluronic acid tissue engineering devices include injectables like SYNVISC ONE® (Genzyme) and ORTHOVISC® (Johnson & Johnson) which have been developed to be injected into the knee to relieve pain from osteoarthritis and improve joint mobility. A particularly novel injectable regenerative tissue scaffold has been developed by reacting hyaluronic acid with methacrylic anhydride creating injectable, cross-linkable methacrylated HA (MeHA). Dr. Jason Burdick’s group has shown MeHA possesses tremendous promise as a supportive network for heart tissue that is recovering from infarction.620 Figure 10 shows the ease of injecting the polymer into the heart wall as well as the thicker resulting walls from sheep given these scaffolds over untreated controls. Injectable, cross-linkable polymers like MeHA hold tremendous promise for a number of regenerative therapies especially in soft tissue engineering since they can fit defects or structural abnormalities in vivo.
Figure 10.
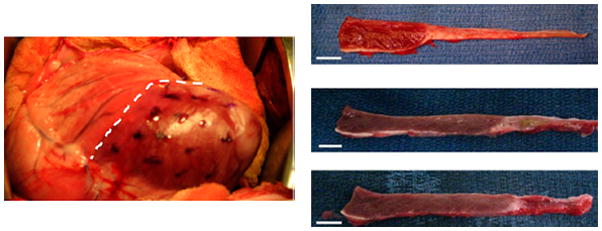
MeHA injection into a sheet heart after infarction helps prevent subsequent myocardial damage. Multiple injections (dots) of MeHA were given into the infarcted area (right of dashed line) (left). Representative images of myocardial wall thickness 8 weeks post treatment with no scaffold (top right), MeHA High scaffold (middle right), and MeHA Low scaffold (bottom right). (reprinted from 620)
HA has also shown promise as a payload delivery vehicle and has been formed into nanoparticles621–624 and hydrogels625–628 for this application. These constructs have been used to deliver chemotherapeutics,622,624 antibiotics,625 analgesics,626 siRNA,621 and proteins.623,627,628 Composites of HA with PLA,629,630 PLGA,630–632 PCL,633,634 PLL,635–637 and chitosan.638–641 have all been developed to create delivery vehicles with enhanced mechanical properties while retaining excellent biocompatibility.
Another human carbohydrate that has shown promise as a biomaterial is chondroitin sulfate (CS). CS is a glycosaminoglycan with very similar structure to HA (CS has a sulfate group in at least one of its side groups). It has been found to be a major component of the body’s natural, hydrophilic would healing matrix produced by fibroblasts.642 CS has also been shown to stimulate the metabolic response of cartilage,643 possess anti-inflammatory properties644 and connect cells to extracellular matrix components.645 Due to CS’s role in natural wound healing and chondrogenesis, it has been studied extensively as a hydrogel for wound dressings135,646 and cartilage tissue engineering. In cartilage tissue engineering, the successful regeneration of cartilage requires a scaffold that causes the correct phenotypic development of seeded cells. Because CS plays a crucial role in the natural development of cartilage, CS647,648 and CS composites with PCL,649 PEG,650,651 collagen,649,652 hyaluronic acid653 and chitosan654 have been used to direct proper cellular chondrogenesis and successful regeneration of cartilage tissue.655
Other carbohydrates of human origin that are being considered as potential biomaterials include heparin, keratin and dermatan.
Non-Human Origin
In addition to polysaccharides of human origin, there exist a number of molecules from other sources that have shown promise as degradable polymeric biomaterials. While a number of candidates have been identified, two of particular interest are chitosan found in crustacean skeletons and alginic acid found in brown algae.
Chitin is a linear polysaccharide consisting of β-1,4 linked N-acetylglucosamine units that forms the exoskeletons of many arthropods. Chitin is structurally similar to hyaluronic acid and has shown a similar capacity to accelerate wound healing.656 Chitin fibers,657,658 sponges659,660 and membranes661 have all been investigated as wound dressing materials. Unfortunately, chitin is insoluble in many common solvents limiting its processability and potential in biomedical applications.
To overcome insolubility issues, chitosan, a chitin derivative, is created by deacetylation of chitin giving a polysaccharide composed of randomly located units of D-glucosamine and N-acetylglucosamine. A number of enzymes have been found in vitro to degrade chitosan including chitosanase, lysozyme and papain.662 Physiologically, lysozyme is the primary degrading enzyme and chitosan degradation rate is dependent on the degree of acetylation and crystallinity.663 Chitosan with lower acetylation percentages have been shown to last in vivo up to several months.664 Side group modification has been found to be a secondary technique for modulating degradation rate. Chitosan undergoes significant hydrogen bonding which can be disrupted by the inclusion of bulky side groups like isobutyl leading to faster polymer degradation.665 Due to chitosan’s processability and versatility, it has been used in a wide range of biomedical applications.
Since chitosan is water absorptive, oxygen permeable and haemostatic, it has been extensively studied as a wound dressing over the past 20 years.666–668 In addition, chitosan is much more bioactive than many of its degradable polymer counterparts leading to acceleration of wound healing. Induction of interleukin-8 production from fibroblasts,669 stimulatation of macrophages,670,671 and chemoattraction of neutrophils672 by chitosan are all important steps necessary to initiate the body’s wound healing cascade. Chitosan’s intrinsic antibacterial property,673 minimal foreign body reaction664 and polymer degradation into N-acetylglucosamine (a major component of dermal tissue) also assist in rapid wound healing. Since chitosan by itself is mechanically weak, it is often crosslinked669,674 or combined with other degradable polymers like PLA,675 PLGA,676,677 PEG,437,678 collagen,679,680 and alginate681,682 which are formed into films,437 membranes,667,682 sponges,673,678,679 particles,676 fibers,675,677,680 and gels668,669,681 to create stronger bandages. Often times, antibiotics are trapped within chitosan and chitosan-based composite wound healing materials to prevent bacterial infection.683,684 HemCon® dressings (HemCon Medical Technologies) are a FDA-approved chitosan-based wound dressing that is often used in combat,685 emergency medicine686 and dentistry.687
Chitosan has also been investigated as a delivery device. Chitosan is so hydrophilic that it almost always is either crosslinked688,689 or blended with other degradable polymers to yield materials with physiologically relevant release rates. Some blended systems include chitosan with PLA,690–692 PLGA,690,692–695 PEG,692,696–699 collagen,700–702 γPGA,703,704 and alginate.705–707 Formation of the polymer composites into particles,690–696,701,703,704,706,707 micelles,698,699 fibers,697 hydrogels,705 and porous scaffolds700–702 has allowed for the delivery of chemotherapeutics,693,694,698 antibiotics,704 anti-inflammatory drugs,697; antipsychotics,690 immunosuppressants,691 vaccines,695,706 DNA,696,699,702,703 siRNA,692 and proteins.700,701,705,707 Due to chitosan’s positive charge and significantly high charge density, its ability to condense DNA has made it most promising as a DNA/gene delivery material. Also, chitosan has been found to be very mucoadhesive, so it has great potential for pulmonary drug delivery.708–710
The ease with which chitosan can be processed into porous matrices gives it promise as a tissue engineering scaffold. So far chitosan and chitosan-composites have been formed into membranes,500,711–713 sponges,443,714–716 fibers,717–721 fused microspheres,722–724 and hydrogels725–729 for regenerative applications. Cellular and acellular scaffolds have been used for the engineering of bone,443,719,722–724 tendon,717 ligament,717,721 cartilage,428,726,728 nerve714,718,725,729 skin,500,713,716,720 and vascular tissue.711,712,715 By side group modification, deacetylation and polymer blending, chitosan can be incorporated into scaffolds with a wide range of physical properties but still retain the novel bioactive function of chitosan itself.
Alginate is a linear copolymer composed of β-D-mannuronic acid and α-L-guluronic acid joined by a 1–4 glycosidic bond. The composition and patterning of the monomers is dependent on the source of the polysaccharide. The most common source of alginate is the cell wall of brown algae and it is normally extracted via a basic solution followed by acidic precipitation to achieve alginic acid. These polymers have been found to have molecular weights up to 500 kDa. Alginate has shown great promise in biomedical applications due to its capacity to form spontaneous gelation when exposed to divalent cations like calcium and the reactivity of its carboxylate side groups.
The simple and mild reaction conditions necessary for alginate hydrogel formation has led to their extensive use as drug and cell delivery devices, wound healing dressings and tissue engineering scaffolds. While some pure alginate hydrogel-based drug delivery devices have been used to deliver payloads,730–733 most research has focused on composite systems of blends or intermixed constructs (e.g. particles within a hydrogel). Alginate-based drug delivery composites include the incorporation of PLGA,734–737 PCL,738,739 polyethers,740–742 and chitosan705,743–745 in order to effectively deliver chemotherapeutics,735,739 antibiotics,741 anti-inflammatory drugs,734,738,740 calcium channel blockers,744 and proteins.705,736,737,742,745 Supportive alginate-based hydrogels have also been researched for the delivery of chondrocytes,746 osteoblasts,747 myoblasts,748 fibroblasts,749 keratinocytes,749 and adipose-derived stem cells.750 Alginate wound healing dressings have been FDA approved and marketed as AlgiDERM® (Bard Medical Division), AlgiSite M (Smith & Nephew), Hyperion Advanced Alginate Dressing (Hyperion Medical), KALTOSTAT® (ConvaTec), and Tegaderm® (3M).
While alginate alone is too mechanically weak to be used as a structural tissue engineering scaffold, it has shown significant promise when blended or copolymerized with other degradable polymers. Composites of PLGA,751–753 collagen,754–757 PLL,758–762 and chitosan746,753,763–765 with alginate have been formed into scaffolds composed of films,760 sponges,759 fibers,763 adhered microspheres,751,755,757,758,761,762 gel,746,753,754,756,764 and freeze casted porous networks.752,765 These constructs have been used in the regenerative engineering of bone,752,757,759 cartilage,746,751,753,756,765 corneal,764 liver,758,760 nerve,761 vascular,755 pancreas762 and connective tissue.754
Modification of alginate through reactions with its carboxylate side groups have yielded novel, bioactive materials. Specifically, adding acetal aldehyde crosslinker yields a pH-sensitive contracting gel,766 and adding laminin peptides causes better adhesion, cell spreading and neurite outgrowth.767 Further investigation into adding functional groups is underway and will hopefully yield promising hybrid materials that can overcome some of the current limitations found with using alginate.
While possessing many great properties, alginate has two significant drawbacks as a biomaterial: limited in vivo degradation and poor cellular adhesion. Mammals do not produce alginate lyase, the enzyme that cleaves alginate polymers,768 so the in vivo degradation of alginate is very slow. In order to overcome this issue, alginate is often either irradiated or oxidized through the use of gamma radiation or periodate, respectively. Low level radiation causes cleavage at the mannuronic-gluronic glycoside bond allowing for more rapid polymer degradation and material solvation.769 Oxidation by periodate, an ion composed of iodine and oxygen, causes the formation of a hemiacetal ring with an open urinate residue with a hydrolytic bond that can cleave much more quickly than the glycoside bond normally does.770 In most tissue engineering applications the scaffold is designed to provide structure support as well as mimic natural extracellular matrix, so poor cell attachment is highly undesirable.771 Most cells that do not interact with their surroundings tend to not differentiate and eventually die off. Side group modification of alginate with the peptides RGD772,773 and GRGDSP774,775 have lead to the development of scaffolds that interact more favorably with transplanted cells and host cells.
In addition to the polymers above, a variety of other polysaccharides, such as dextran, agarose, mannan and inulin have been investigated for potential as biomaterials.
CONCLUSIONS
There currently exists a wide range of degradable polymers that hold potential as biomaterials. With advancements in polymer synthesis techniques, the paradigm of utilizing a few well characterized polymers (e.g. PLGA and collagen) for all biomedical applications has shifted to using polymers, both heavily researched and newly developed, that can fit certain niches (e.g. DNA and RNA association with phosphoesters and inherent bioactivity of chitosan). In addition, the emergence of combination polymers holds promise for the creation of novel materials that possess desired properties for highly specific applications. The further development of processing techniques, especially with the assistance of computer-aided technology, is allowing for the formation of particles and scaffolds with extremely complex architectures that can mimic their biological counterparts. While these developments in polymer research have been critical, it should be noted that the advancements in biological research has led to a better understanding of how biomaterials interact with the host on cellular, tissue, organ and systemic levels. The field of degradable polymeric biomaterials will only continue to progress if the recent creation of strong collaboration teams composed of chemists, biologists, material scientists, engineers and clinicians is encouraged.
Biographies
Dr. Bret Ulery received his B.S. and B.S.E. in Biochemistry and Chemical Engineering, respectively, from the University of Iowa in 2006. He was a graduate student in the Chemical and Biological Engineering department at Iowa State University from 2006 until he earned his Ph.D. in 2010. Since September 2010, he has served as a PostDoctoral Fellow in the Orthopaedic Surgery department and Institute for Regenerative Engineering at the University of Connecticut Health Center. His research interests include novel degradable polymer synthesis, composite tissue engineering scaffolds, and immunomodulatory biomaterials.

Dr. Lakshmi Nair received her Ph.D. from SCTIMST, India in Polymer Chemistry and Biomaterials in 2000. After finishing her graduate work, she completed post-doctoral training at Drexel University and the University of Virginia. In 2006, she became a research assistant professor in the department of Orthopaedic Surgery at the University of Virginia. She has served as an assistant professor in the departments of Orthopaedic Surgery and Chemical, Materials and Biomolecular Engineering at the University of Connecticut and as a core member of the Institute for Regenerative Engineering since 2008. Her research interests include cell-biomaterial interactions, tissue regeneration devices, immunomodulatory biomaterials and cell delivery devices.

Dr. Cato T. Laurencin is the Van Dusen Endowed Chair in Academic Medicine, Distinguished Professor of Orthopaedic Surgery, and Professor of Chemical, Materials and Biomolecular Engineering at the University of Connecticut. Dr. Laurencin is Vice President for Health Affairs at the University of Connecticut, and Dean of the UConn School of Medicine. Dr. Laurencin is an elected member of the Institute of Medicine of the National Academy of Sciences. He is also an elected member of the National Academy of Engineering. Dr. Laurencin earned his B.S.E. in chemical engineering from Princeton, his M.D., Magna Cum Laude from Harvard Medical School, and his Ph.D. in biochemical engineering/biotechnology from the Massachusetts Institute of Technology.

References
- 1.Williams DF. Biomaterials. 2009;30:5897–5909. doi: 10.1016/j.biomaterials.2009.07.027. [DOI] [PubMed] [Google Scholar]
- 2.Williams DF. The Williams Dictionary of Biomaterials. Liverpool University Press; Liverpool: 1999. [Google Scholar]
- 3.Schmitt EE, Polistina RA USPTO. United States; 1967. [Google Scholar]
- 4.Schmitt EE, Polistina RA USPTO. United States; 1963. [Google Scholar]
- 5.Ibim SEM, Ambrosio AMA, Kwon MS, El-Amin SF, Allcock HR, Laurencin CT. Biomaterials. 1997;18:1565–1569. doi: 10.1016/s0142-9612(97)80009-9. [DOI] [PubMed] [Google Scholar]
- 6.Vehof JWM, Fisher JP, Dean D, Waerden J-PCMvd, Spauwen PHM, Mikos AG, Jansen JA. Journal of Biomedical Materials Research. 2002;60:241–251. doi: 10.1002/jbm.10073. [DOI] [PubMed] [Google Scholar]
- 7.Pangas SA, Saudye H, Shea LD, Woodruff TK. Tissue Engineering. 2003;9:1013–1021. doi: 10.1089/107632703322495655. [DOI] [PubMed] [Google Scholar]
- 8.Holland TA, Bodde EWH, Baggett LS, Tabata Y, Mikos AG, Jansen JA. Journal of Biomedical Materials Research Part A. 2005;75A:156–167. doi: 10.1002/jbm.a.30379. [DOI] [PubMed] [Google Scholar]
- 9.Greish YE, Bender JD, Nair LS, Brown PW, Allcock HR, Laurencin CT. Journal of Biomedical Materials Research Part A. 2006;77A:416–425. doi: 10.1002/jbm.a.30145. [DOI] [PubMed] [Google Scholar]
- 10.West ER, Xu M, Woodruff TK, Shea LD. Biomaterials. 2007;28:4439–4448. doi: 10.1016/j.biomaterials.2007.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu M, Banc A, Woodruff TK, Shea LD. Biotechnology and Bioengineering. 2009;103:378–386. doi: 10.1002/bit.22250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kretlow JD, Spicer PP, Jansen JA, Vacanti CA, Kasper FK, Mikos AG. Tissue Engineering Part A. 2010;16:3555–3568. doi: 10.1089/ten.tea.2010.0471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Deng M, Nair LS, Nukavarapu SP, Kumbar SG, Jiang T, Weikel AL, Krogman NR, Allcock HR, Laurencin CT. Advanced Functional Materials. 2010;20:2794–2806. doi: 10.1002/adfm.201090073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lloyd AW. Medical Device Technology. 2002;13:18–21. [PubMed] [Google Scholar]
- 15.Nair LS, Laurencin CT. Advances in Biochemical Engineering Biotechnology. 2006;102:47–90. doi: 10.1007/b137240. [DOI] [PubMed] [Google Scholar]
- 16.Nair LS, Laurencin CT. Progress in Polymer Science. 2007;32:762–798. [Google Scholar]
- 17.Burkersroda Fv, Schedl L, Gopferich A. Biomaterials. 2002;23:4221–4231. doi: 10.1016/s0142-9612(02)00170-9. [DOI] [PubMed] [Google Scholar]
- 18.Tamada JA, Langer R. Proceedings of the National Academy of Sciences of the United States of America. 1993;90:552–556. doi: 10.1073/pnas.90.2.552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Determan AS, Trewyn BG, Lin VS-Y, Nilsen-Hamilton M, Narasimhan B. Journal of Controlled Release. 2004;100:97–109. doi: 10.1016/j.jconrel.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 20.Wen X, Tresco PA. Biomaterials. 2006;27:3800–3809. doi: 10.1016/j.biomaterials.2006.02.036. [DOI] [PubMed] [Google Scholar]
- 21.Gopferich A. Macromolecules. 1997;30:2598–2604. [Google Scholar]
- 22.Coulembier O, Degee P, Hedrick JL, Dubois P. Progress in Polymer Science. 2006;31:723–747. [Google Scholar]
- 23.Maurus PB, Kaeding CC. Operative Techniques in Sports Medicine. 2004;12:158–160. [Google Scholar]
- 24.Katz AR, Turner RJ. Surgery, Gynecology & Obstetrics. 1970;131:701–716. [PubMed] [Google Scholar]
- 25.Burns AE. The Journal of Foot and Ankle Surgery. 1995;34:276–282. doi: 10.1016/S1067-2516(09)80060-4. [DOI] [PubMed] [Google Scholar]
- 26.Reed TM. The Journal of Foot and Ankle Surgery. 1999;38:14–23. doi: 10.1016/s1067-2516(99)80083-0. [DOI] [PubMed] [Google Scholar]
- 27.Knight S, Erggelet C, Endres M, Sittinger M, Kaps C, Stussi E. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2007;83B:50–57. doi: 10.1002/jbm.b.30765. [DOI] [PubMed] [Google Scholar]
- 28.Wang L, Dormer NH, Bonewald LF, Detamore MS. Tissue Engineering Part A. 2010;16:1937–1948. doi: 10.1089/ten.TEA.2009.0706. [DOI] [PubMed] [Google Scholar]
- 29.Dunne N, Jack V, O’Hara R, Farrar D, Buchanan F. Journal of Materials Science. Materials in Medicine. 2010;21:2263–2270. doi: 10.1007/s10856-010-4021-9. [DOI] [PubMed] [Google Scholar]
- 30.Pihlajamaki HK, Salminen ST, Tynninen O, Bostman OM, Laitinen O. Calcified Tissued International. 2010;87:90–98. doi: 10.1007/s00223-010-9374-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erggelet C, Neumann K, Endres M, Haberstroh K, Sittinger M, Kaps C. Biomaterials. 2007;28:5570–5580. doi: 10.1016/j.biomaterials.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 32.Frisbie DD, Lu Y, Kawcak CE, DiCarlo EF, Binette F, McIlwraith CW. The Americal Journal of Sports Medicine. 2009;37:71S–80S. doi: 10.1177/0363546509348478. [DOI] [PubMed] [Google Scholar]
- 33.Mahmoudifar N, Doran PM. Biomaterials. 2010;31:3858–3867. doi: 10.1016/j.biomaterials.2010.01.090. [DOI] [PubMed] [Google Scholar]
- 34.Pihlajamaki H, Tynninen O, Karjalainen P, Rokkanen P. Journal of Biomedical Materials Research Part A. 2007;81:987–993. doi: 10.1002/jbm.a.31144. [DOI] [PubMed] [Google Scholar]
- 35.Xu L, Cao D, Liu W, Zhou G, Zhang WJ, Cao Y. Biomaterials. 2010;31:3894–3902. doi: 10.1016/j.biomaterials.2010.01.106. [DOI] [PubMed] [Google Scholar]
- 36.Ohara T, Itaya T, Usami K, Ando Y, Sakurai H, Honda MJ, Ueda M, Kagami H. Journal of Biomedical Materials Research Part A. 2010;94:800–805. doi: 10.1002/jbm.a.32749. [DOI] [PubMed] [Google Scholar]
- 37.Sayasneh A, Johnson H. Journal of Obstetrics & Gynaecology. 2010;30:721–724. doi: 10.3109/01443615.2010.501921. [DOI] [PubMed] [Google Scholar]
- 38.Aysan E, Bektas H, Ersoz F, Sari S, Kaygusuz A. International Journal of Clinical and Experimental Medicine. 2010;3:341–346. [PMC free article] [PubMed] [Google Scholar]
- 39.Dai Tt, Jiang Zh, Li Sl, Zhou Gd, Kretlow JD, Cao Wg, Liu W, Cao Yl. Journal of Biotechnology. 2010;150:182–189. doi: 10.1016/j.jbiotec.2010.07.028. [DOI] [PubMed] [Google Scholar]
- 40.Abbushi A, Endres M, Cabraja M, Kroppenstedt SN, Thomale UW, Sittinger M, Hegewald AA, Morawietz L, Lemke A-J, Bansemer V-G, Kaps C, Woiciechowsky C. Spine. 2008;33:1527–1532. doi: 10.1097/BRS.0b013e3181788760. [DOI] [PubMed] [Google Scholar]
- 41.Gunatillake P, Mayadunne R, Adhikari R. Biotechnology Annual Review. 2006;12:301–347. doi: 10.1016/S1387-2656(06)12009-8. [DOI] [PubMed] [Google Scholar]
- 42.Ceonzo K, Gaynor A, Shaffer L, Kojima K, Vacanti CA, Stahl GL. Tissue Engineering. 2006;12:301–308. doi: 10.1089/ten.2006.12.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pihlajamaki H, Salminen S, Laitinen O, Tynninen O, Bostman O. Journal of Orthopaedic Research. 2006;24:1597–1606. doi: 10.1002/jor.20191. [DOI] [PubMed] [Google Scholar]
- 44.Otto J, Binnebosel M, Pietsch S, Anurov M, Titkova S, Ottinger AP, Jansen M, Rosch R, Kammer D, Klinge U. Journal of Investigative Surgery: the Official Journal of the Academy of Surgical Research. 2010;23:190–196. doi: 10.3109/08941931003739741. [DOI] [PubMed] [Google Scholar]
- 45.Patila T, Jokinen JJ, Salminen J, Kankuri E, Harjula A. Journal of Surgical Research. 2008;148:181–184. doi: 10.1016/j.jss.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 46.Middleton JC, Tipton AJ. Biomaterials. 2000;21:2335–2346. doi: 10.1016/s0142-9612(00)00101-0. [DOI] [PubMed] [Google Scholar]
- 47.Suuronen R, Pohjonen T, Hietanen J, Lindqvist C. Journal of Oral and Maxillofacial Surgery. 1998;56:604–614. doi: 10.1016/s0278-2391(98)90461-x. [DOI] [PubMed] [Google Scholar]
- 48.Zielhuis SW, Nijsen JFW, Seppenwoolde J-H, Bakker CJG, Krijger GC, Dullens HFJ, Zonnenbery BA, Rijk PPv, Hennink WE, Schip ADvg. Biomaterials. 2007;28:4591–4599. doi: 10.1016/j.biomaterials.2007.07.012. [DOI] [PubMed] [Google Scholar]
- 49.Lu J, Jackson JK, Gleave ME, Burt HM. Cancer Chemotherapy and Pharmacology. 2008;61:997–1005. doi: 10.1007/s00280-007-0557-x. [DOI] [PubMed] [Google Scholar]
- 50.Chen A-Z, Li Y, Chau F-T, Lau T-Y, Hu J-Y, Zhao Z, Mok DK-w. Acta Biomaterialia. 2009;5:2913–2919. doi: 10.1016/j.actbio.2009.04.032. [DOI] [PubMed] [Google Scholar]
- 51.Lensen D, Breukelen Kv, Vriezema DM, Hest JCMv. Macromolecular Bioscience. 2010;10:475–480. doi: 10.1002/mabi.200900404. [DOI] [PubMed] [Google Scholar]
- 52.Loo JSC, Oooi CP, Boey FYC. Biomaterials. 2005;26:1359–1367. doi: 10.1016/j.biomaterials.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 53.Loo SCJ, Tan HT, Ooi CP, Boey YCF. Acta Biomaterialia. 2006;2:287–296. doi: 10.1016/j.actbio.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 54.Ueda H, Tabata Y. Advanced Drug Delivery Reviews. 2003;55:501–518. doi: 10.1016/s0169-409x(03)00037-1. [DOI] [PubMed] [Google Scholar]
- 55.Chan P-C, Liu B-Y, Liu C-M, Chou H-H, Ho M-H, Liu H-C, Wang D-M, Hou L-T. Journal of Biomedical Materials Research Part A. 2007;81A:771–780. doi: 10.1002/jbm.a.31031. [DOI] [PubMed] [Google Scholar]
- 56.Schofer MD, Fuchs-Winkelmann S, Grabedunkel C, Wack C, Dersch R, Rudisile M, Wendorff JH, Greiner A, Paletta JRJ, Boudriot U. Scientific World Journal. 2008;25:1269–1279. doi: 10.1100/tsw.2008.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shim IK, Jung MR, Kim KH, Seol YJ, Park YJ, Park WH, Lee SJ. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2010;95B:150–160. doi: 10.1002/jbm.b.31695. [DOI] [PubMed] [Google Scholar]
- 58.Cai YZ, Wang LL, Cai HX, Qi YY, Zou XH. Journal of Biomedical Materials Research Part A. 2010;95A:49–57. doi: 10.1002/jbm.a.32816. [DOI] [PubMed] [Google Scholar]
- 59.Ju YM, Park K, Son JS, Kim J-J, Rhie J-W, Han DK. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2007;85B:252–260. doi: 10.1002/jbm.b.30943. [DOI] [PubMed] [Google Scholar]
- 60.Tanaka Y, Yamaoka H, Nishizawa S, Nagata S, Ogasawara T, Asawa Y, Fujihara Y, Takato T, Hoshi K. Biomaterials. 2010;31:4506–4516. doi: 10.1016/j.biomaterials.2010.02.028. [DOI] [PubMed] [Google Scholar]
- 61.Inui A, Kokubu T, Makino T, Nagura I, Toyokawa N, Sakata R, Kotera M, Nishino T, Fujioka H, Kurosaka M. Internation Orthopaedics. 2010;34:1327–1332. doi: 10.1007/s00264-009-0917-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hu J, Sun X, Ma H, Xie C, Chen YE, Ma PX. Biomaterials. 2010;31:7971–7977. doi: 10.1016/j.biomaterials.2010.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang HB, Mullins ME, Cregg JM, McCarthy CW, Gilbert RJ. Acta Biomaterialia. 2010;6:2970–2978. doi: 10.1016/j.actbio.2010.02.020. [DOI] [PubMed] [Google Scholar]
- 64.Francois S, Chakfe N, Durand B, Laroche G. Acta Biomaterialia. 2009;5:2418–2428. doi: 10.1016/j.actbio.2009.03.013. [DOI] [PubMed] [Google Scholar]
- 65.Wang P, Hu J, Ma PX. Biomaterials. 2009;30:2735–2740. doi: 10.1016/j.biomaterials.2009.01.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wright LD, Young RT, Andric T, Freeman JW. Biomedical Materials. 2010;5:055006. doi: 10.1088/1748-6041/5/5/055006. [DOI] [PubMed] [Google Scholar]
- 67.Wei G, Jin Q, Giannobile WV, Ma PX. Biomaterials. 2007;28:2087–2096. doi: 10.1016/j.biomaterials.2006.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lu XL, Sun ZJ, Cai W, Gao ZY. Journal of Materials Science. Materials in Medicine. 2008;19:395–399. doi: 10.1007/s10856-006-0100-3. [DOI] [PubMed] [Google Scholar]
- 69.Idris SB, Arvidson K, Plikk P, Ibrahim S, Finne-Wistrand A, Albertsson A-C, Bolstad AI, Mustafa K. Journal of Biomedical Materials Research Part A. 2010;94A:631–639. doi: 10.1002/jbm.a.32726. [DOI] [PubMed] [Google Scholar]
- 70.Luo X, Qiu D, He B, Wang L, Luo J. Macromolecular Bioscience. 2006;6:373–381. doi: 10.1002/mabi.200500236. [DOI] [PubMed] [Google Scholar]
- 71.Pu F, Rhodes NP, Bayon Y, Chen R, Brans G, Benne R, Hunt JA. Biomaterials. 2010;31:4330–4340. doi: 10.1016/j.biomaterials.2010.02.010. [DOI] [PubMed] [Google Scholar]
- 72.Tan H, Wu J, Huang D, Gao C. Macromolecular Bioscience. 2010;10:156–163. doi: 10.1002/mabi.200900160. [DOI] [PubMed] [Google Scholar]
- 73.Grundmann S, Royen Nv, Pasterkamp G, Gonzalez N, Tijsma EJ, Piek JJ, Hoefer IE. Journal of the American College of Cardiology. 2007;50:351–358. doi: 10.1016/j.jacc.2007.03.046. [DOI] [PubMed] [Google Scholar]
- 74.Luderer F, Lobler M, Rohm HW, Gocke C, Kunna K, Kock K, Kroemer HK, Weitschies W, Schmitz K-P, Sternberg K. Journal of Biomaterials Applications. 2010 doi: 10.1177/0885328209360696. [DOI] [PubMed] [Google Scholar]
- 75.Blaker JJ, Bismarck A, Boccaccinni AR, Oung YAM, Nazhat SN. Acta BIomaterialia. 2010;6:756–762. doi: 10.1016/j.actbio.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 76.Tang L, Zhao C, Xiong Y, Wang A. International Orthopaedics. 2010;34:755–759. doi: 10.1007/s00264-009-0808-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hasegawa S, Neo M, Tamura J, Fujibayashi S, Takemoto M, Shikinami Y, Okazaki K, Nakamura T. Journal of Biomedical Materials Research Part A. 2007;81A:930–938. doi: 10.1002/jbm.a.31109. [DOI] [PubMed] [Google Scholar]
- 78.Carletti E, Endogan T, Hasirci N, Hasirci V, Maniglio D, Motta A, Migliaresi C. Journal of Tissue Engineering and Regenerative Medicine. 2010 doi: 10.1002/term.349. [DOI] [PubMed] [Google Scholar]
- 79.Yu NY, Schindeler A, Peacock L, Mikulec K, Baldock PA, Ruys AJ, Little DG. European Cells & Materials. 2010;22:431–442. doi: 10.22203/ecm.v020a35. [DOI] [PubMed] [Google Scholar]
- 80.Leung L, Chan C, Baek S, Naguib H. Biomedical Materials. 2008;3:025006. doi: 10.1088/1748-6041/3/2/025006. [DOI] [PubMed] [Google Scholar]
- 81.Tsallas A, Jackson J, Burt H. Cancer Chemotherapy and Pharmacology. 2010 doi: 10.1007/s00280-010-1499-2. [DOI] [PubMed] [Google Scholar]
- 82.Seck TM, Melchels FPW, Feijen J, Grijpma DW. Journal of Controlled Release. 2010;148:34–41. doi: 10.1016/j.jconrel.2010.07.111. [DOI] [PubMed] [Google Scholar]
- 83.Cai K, Yao K, Yang Z, Qu Y, Li X. Journal of Materials Science. Materials in Medicine. 2007;18:2017–2024. doi: 10.1007/s10856-007-3151-1. [DOI] [PubMed] [Google Scholar]
- 84.Miller RA, Brady JM, Cutright DE. Journal of Biomedical Materials Research. 1977;11:711–719. doi: 10.1002/jbm.820110507. [DOI] [PubMed] [Google Scholar]
- 85.Middleton JC, Tipton AJ. Medical Plastics and Biomaterials. 1998:31–38. [Google Scholar]
- 86.Conn J, Oyasu R, Welsh M, Beal JM. American Journal of surgery. 1974;128:19–23. doi: 10.1016/0002-9610(74)90228-1. [DOI] [PubMed] [Google Scholar]
- 87.Clavert P, Warner JJP. Arthroscopy: The Journal of Arthroscopic & Related Surgery. 2005;21:200–203. doi: 10.1016/j.arthro.2004.10.015. [DOI] [PubMed] [Google Scholar]
- 88.Betancourt T, Brown B, Brannon-Peppas L. Nanomedicine. 2007;2:219–232. doi: 10.2217/17435889.2.2.219. [DOI] [PubMed] [Google Scholar]
- 89.Liu J, Qiu Z, Wang S, Zhou L, Zhang S. Biomedical Materials. 2010;5:065002. doi: 10.1088/1748-6041/5/6/065002. [DOI] [PubMed] [Google Scholar]
- 90.Gavenis K, Schneider U, Groll J, Schmidt-Rohlfing B. The International Journal of Artificial Organs. 2010;33:45–53. [PubMed] [Google Scholar]
- 91.Ye M, Kim S, Park K. Journal of Controlled Release. 2010;146:241–260. doi: 10.1016/j.jconrel.2010.05.011. [DOI] [PubMed] [Google Scholar]
- 92.Andreas K, Zehbe R, Kazubek M, Grzeschik K, Sternberg N, Baumler H, Schubert H, Sittinger M, Ringe J. Acta Biomaterialia. 2011 doi: 10.1016/j.actbio.2010.12.014. [DOI] [PubMed] [Google Scholar]
- 93.Jiang W, Schwendeman SP. Molecular Pharmaceuticals. 2008;5:808–817. doi: 10.1021/mp800027f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Quintilio W, Takata CS, Sant’Anna OA, Costa MHBd, Raw I. Current Drug Delivery. 2009;6:297–304. doi: 10.2174/156720109788680886. [DOI] [PubMed] [Google Scholar]
- 95.Thomas C, Gupta V, Ahsan F. Pharmaceutical Research. 2010;27:905–919. doi: 10.1007/s11095-010-0094-x. [DOI] [PubMed] [Google Scholar]
- 96.Jeong Y-I, Na H-S, Seo D-H, Kim D-G, Lee H-C, Jang M-K, Na S-K, Roh S-H, Kim S-I, Nah J-W. International Journal of Pharmaceutics. 2008;352:317–323. doi: 10.1016/j.ijpharm.2007.11.001. [DOI] [PubMed] [Google Scholar]
- 97.Jhunjhunwala S, Raimondi G, Thomson AW, Little SR. Journal of Controlled Release. 2009;133:191–197. doi: 10.1016/j.jconrel.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Farazuddin M, Alam M, Khan AA, Khan N, Parvez S, Dutt GU, Mohammad O. Journal of Drug Targeting. 2010;18:45–52. doi: 10.3109/10611860903156401. [DOI] [PubMed] [Google Scholar]
- 99.Tang Y, Singh J. International Journal of Pharmaceutics. 2008;357:119–125. doi: 10.1016/j.ijpharm.2008.01.053. [DOI] [PubMed] [Google Scholar]
- 100.Vega E, Gamisans F, Garcia ML, Chauvet A, Lacoulonche F, Egea MA. Journal of Pharmaceutical Sciences. 2008;97:5306–5317. doi: 10.1002/jps.21383. [DOI] [PubMed] [Google Scholar]
- 101.Zolnik BS, Burgess DJ. Journal of Controlled Release. 2008;127:137–145. doi: 10.1016/j.jconrel.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 102.Eperon S, Bossy-Nobs L, Petropoulos IK, Gurny R, Guex-Crosier Y. International Journal of Pharmaceutics. 2008;352:240–247. doi: 10.1016/j.ijpharm.2007.10.054. [DOI] [PubMed] [Google Scholar]
- 103.Murata N, Takashima Y, Toyoshima K, Yamamoto M, Okada H. Journal of Controlled Release. 2008;126:246–254. doi: 10.1016/j.jconrel.2007.11.017. [DOI] [PubMed] [Google Scholar]
- 104.Singh A, Nie H, Ghosn B, Qin H, Kwak LW, Roy K. Molecular Therapy. 2008;16:2011–2021. doi: 10.1038/mt.2008.206. [DOI] [PubMed] [Google Scholar]
- 105.Patil Y, Panyam J. International Journal of Pharmaceutics. 2009;367:195–203. doi: 10.1016/j.ijpharm.2008.09.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Berkland C, Pollauf E, Varde N, Pack DW, Kim K. Pharmaceutical Research. 2007;24:1007–1013. doi: 10.1007/s11095-006-9197-9. [DOI] [PubMed] [Google Scholar]
- 107.Kim BS, Oh JM, Hyun H, Kim KS, Lee SH, Kim YH, Park K, Lee HB, Kim MS. Molecular Pharmaceuticals. 2009;6:353–365. doi: 10.1021/mp800087t. [DOI] [PubMed] [Google Scholar]
- 108.Haddadi A, Aboofazeli R, Erfan M, Farboud ES. Journal of Microencapsulation. 2008;25:379–386. doi: 10.1080/02652040802000714. [DOI] [PubMed] [Google Scholar]
- 109.Xie J, Wang C-H. Pharmaceutical Research. 2006;23:1817–1826. doi: 10.1007/s11095-006-9036-z. [DOI] [PubMed] [Google Scholar]
- 110.Ionescu LC, Lee GC, Sennett BJ, Burdick JA, Mauck RL. Biomaterials. 2010;31:4113–4120. doi: 10.1016/j.biomaterials.2010.01.098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Fu K, Pack DW, Klibanov AM, Langer R. Pharmaceutical Research. 2000;17:100–106. doi: 10.1023/a:1007582911958. [DOI] [PubMed] [Google Scholar]
- 112.Ding AG, Schwendeman SP. Pharmaceutical Research. 2008;25:2041–2052. doi: 10.1007/s11095-008-9594-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chung HJ, Kim IK, Kim TG, Park TG. Tissue Engineering Part A. 2008;14:607–615. doi: 10.1089/tea.2007.0263. [DOI] [PubMed] [Google Scholar]
- 114.Zhu XH, Lee LY, Jackson JSH, Tong YW, Wang C-H. Biotechnology and Bioengineering. 2008;100:998–1009. doi: 10.1002/bit.21824. [DOI] [PubMed] [Google Scholar]
- 115.Simpson RL, Wiria FE, Amis AA, Chua CK, Leong KF, Hansen UN, Chandrasekaran M, Lee MW. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2008;84B:17–25. doi: 10.1002/jbm.b.30839. [DOI] [PubMed] [Google Scholar]
- 116.Jabbarzadeh E, Starnes T, Khan YM, Jiang T, Wirtel AJ, Deng M, Lv Q, Nair LS, Doty SB, Laurencin CT. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:11099–11104. doi: 10.1073/pnas.0800069105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Spalazzi JP, Vyner MC, Jacobs MT, Moffat KL, Lu HH. Clinical Orthopaedics and Related Research. 2008;466:1938–1948. doi: 10.1007/s11999-008-0310-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Arnold MM, Gorman EM, Schieber LJ, Munson EJ, Berkland C. Journal of Controlled Release. 2007;121:100–109. doi: 10.1016/j.jconrel.2007.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Narayan D, Venkatraman SS. Journal of Biomedical Materials Research Part A. 2008;87A:710–718. doi: 10.1002/jbm.a.31749. [DOI] [PubMed] [Google Scholar]
- 120.Ren J, Ren T, Zhao P, Huang Y, Pan K. Journal of Biomaterials Science. Polymer Edition. 2007;18:505–517. doi: 10.1163/156856207780852578. [DOI] [PubMed] [Google Scholar]
- 121.Bashur CA, Dahlgren LA, Goldstein AS. Biomaterials. 2006;27:5681–5688. doi: 10.1016/j.biomaterials.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 122.Kumbar SG, Nukavarapu SP, James R, Nair LS, Laurencin CT. Biomaterials. 2008;29:4100–4107. doi: 10.1016/j.biomaterials.2008.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Moffat KL, Kwei AS-P, Spalazzi JP, Doty SB, Levine WN, Lu HH. Tissue Engineering Part A. 2009;15:115–126. doi: 10.1089/ten.tea.2008.0014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Aviss KJ, Gough JE, Downes S. European Cells & Materials. 2010;19:193–204. doi: 10.22203/ecm.v019a19. [DOI] [PubMed] [Google Scholar]
- 125.Ge Z, Wang L, Heng BC, Tian X-F, Lu K, Fan VTW, Yeo JF, Cao T, Tan E. Journal of Biomaterials Applications. 2008;23:533–547. doi: 10.1177/0885328208094301. [DOI] [PubMed] [Google Scholar]
- 126.Lee M, Wu BM, Dunn JCY. Journal of Biomedical Materials Research Part A. 2008;87A:1010–1016. doi: 10.1002/jbm.a.31816. [DOI] [PubMed] [Google Scholar]
- 127.Yoon JJ, Chung HJ, Lee HJ, Park TG. Journal of Biomedical Materials Research Part A. 2006;79A:934–942. doi: 10.1002/jbm.a.30843. [DOI] [PubMed] [Google Scholar]
- 128.Perron JK, Naguib HE, Daka J, Chawla A, Wilkins R. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2009;91B:876–886. doi: 10.1002/jbm.b.31470. [DOI] [PubMed] [Google Scholar]
- 129.Lee M, Dunn JCY, Wu BM. Biomaterials. 2005;26:4281–4289. doi: 10.1016/j.biomaterials.2004.10.040. [DOI] [PubMed] [Google Scholar]
- 130.Xie J, Li X, Lipner J, Manning CN, Schwartz AG, Thomopoulos S, Xia Y. Nanoscale. 2010;2:923–926. doi: 10.1039/c0nr00192a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Stoll C, John T, Endres M, Rosen C, Kaps C, Kohl B, Sittinger M, Ertel W, Schulze-Tanzil G. Journal of Orthopaedic Research. 2010;28:1170–1177. doi: 10.1002/jor.21109. [DOI] [PubMed] [Google Scholar]
- 132.Blackwood KA, McKean R, Canton I, Freeman CO, Franklin KL, Cole D, Brook I, Farthing P, Rimmer S, Haycock JW, Ryan AJ, MacNeil S. Biomaterials. 2008;29:3091–3104. doi: 10.1016/j.biomaterials.2008.03.037. [DOI] [PubMed] [Google Scholar]
- 133.Li J, Li L, Yu H, Cao H, Gao C, Gong Y. ASAIO Journal. 2006;52:321–327. doi: 10.1097/01.mat.0000217794.35830.4a. [DOI] [PubMed] [Google Scholar]
- 134.Wen F, Chang S, Toh YC, Arooz T, Zhuo L, Teoh SH, Yu H. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2008;87:154–162. doi: 10.1002/jbm.b.31086. [DOI] [PubMed] [Google Scholar]
- 135.Wang W, Zhang M, Lu W, Zhang X, Ma D, Rong X, Yu C, Jin Y. Tissue Engineering Part C: Methods. 2010;16:269–279. doi: 10.1089/ten.TEC.2009.0161. [DOI] [PubMed] [Google Scholar]
- 136.Bhang SH, Lim JS, Choi CY, Kwon YK, Kim B-S. Journal of Biomaterials Science. Polymer Edition. 2007;18:223–239. doi: 10.1163/156856207779116711. [DOI] [PubMed] [Google Scholar]
- 137.He L, Zhang Y, Zeng C, Ngiam M, Liao S, Quan D, Zeng Y, Lu J, Ramakrishna S. Tissue Engineering Part C: Methods. 2009;15:243–255. doi: 10.1089/ten.tec.2008.0255. [DOI] [PubMed] [Google Scholar]
- 138.Olson HE, Rooney GE, Gross L, Nesbitt JJ, Galvin KE, Knight A, Chen B, Yaszemski MJ, Windebank AJ. Tissue Engineering Part A. 2009;15:1797–1805. doi: 10.1089/ten.tea.2008.0364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Abe H, Doi Y. International Journal of Biological Macromolecules. 1999;25:185–192. doi: 10.1016/s0141-8130(99)00033-1. [DOI] [PubMed] [Google Scholar]
- 140.Zhijiang C, Zhihong W. Journal of Materials Science. 2007;42:5886–5890. [Google Scholar]
- 141.Laeger T, Metges CC, Kuhla B. Appetite. 2010;54:450–455. doi: 10.1016/j.appet.2010.04.005. [DOI] [PubMed] [Google Scholar]
- 142.Zhijiang C. Journal of Materials Science: Materials in Medicine. 2006;17:1297–1303. doi: 10.1007/s10856-006-0604-x. [DOI] [PubMed] [Google Scholar]
- 143.Suwantong O, Waleetorncheepsawat S, Sanchavanakit N, Pavasant P, Cheepsunthorn P, Bunaprasert T, Supaphol P. International Journal of Biological Macromolecules. 2007;40:217–223. doi: 10.1016/j.ijbiomac.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 144.Kalbermatten DF, Erba P, Mahay D, Wiberg M, Pierer G, Terenghi G. The Journal of hand surgery, European volume. 2008;33:587–594. doi: 10.1177/1753193408090755. [DOI] [PubMed] [Google Scholar]
- 145.Bernd HE, Kunze C, Freier T, Sternberg K, Kramer S, Behrend D, Prall F, Donat M, Kramp B. Acta Oto-laryngologica. 2009;129:1010–1017. doi: 10.1080/00016480802552493. [DOI] [PubMed] [Google Scholar]
- 146.Gredes T, Spassov A, Mai R, Mack H, Loster BW, Mazurkiewicz-Janik M, Kubein-Meesenburg D, Fanghanel J, Gedrange T. Journal of Physiology and Pharmacology. 2009;60:77–81. [PubMed] [Google Scholar]
- 147.Ahmed T, Marcal H, Lawless M, Wanandy NS, Chiu A, Foster LJR. Biomacromolecules. 2010;11:2707–2715. doi: 10.1021/bm1007579. [DOI] [PubMed] [Google Scholar]
- 148.Pouton CW, Akhtar S. Advanced Drug Delivery Reviews. 1996;18:133–162. [Google Scholar]
- 149.Cool SM, Kenny B, Wu A, Nurcombe V, Trau M, Cassady AI, Grondahl L. Journal of Biomedical Materials Research Part A. 2007;82:599–610. doi: 10.1002/jbm.a.31174. [DOI] [PubMed] [Google Scholar]
- 150.Ke Y, Wang YJ, Ren L, Zhao QC, Huang W. Acta Biomaterialia. 2010;6:1329–1336. doi: 10.1016/j.actbio.2009.10.026. [DOI] [PubMed] [Google Scholar]
- 151.Liu J, Zhao B, Zhang Y, Lin Y, Hu P, Ye C. Journal of Biomedical Materials Research Part A. 2010;94A:603–610. doi: 10.1002/jbm.a.32730. [DOI] [PubMed] [Google Scholar]
- 152.Rathbone S, Furrer P, Lubben J, Zinn M, Cartmell S. Journal of Biomedical Materials Research Part A. 2010;93:1391–1403. doi: 10.1002/jbm.a.32641. [DOI] [PubMed] [Google Scholar]
- 153.Ji Y, Li X-T, Chen G-Q. Biomaterials. 2008;29:3807–3814. doi: 10.1016/j.biomaterials.2008.06.008. [DOI] [PubMed] [Google Scholar]
- 154.Zorlutana P, Tezcaner A, Hasirci V. Journal of Biomaterials Science. Polymer Edition. 2008;19:399–410. doi: 10.1163/156856208783720976. [DOI] [PubMed] [Google Scholar]
- 155.Yucel D, Kose GT, Hasirci V. Biomaterials. 2010;31:1596–1603. doi: 10.1016/j.biomaterials.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 156.Yang C, Plackett D, Needham D, Burt HM. Pharmaceutical Research. 2009;26:1644–1656. doi: 10.1007/s11095-009-9875-5. [DOI] [PubMed] [Google Scholar]
- 157.Errico C, Bartoli C, Chiellini F, Chiellini E. Journal of Biomedicine and Biotechnology. 2009;2009:571702. doi: 10.1155/2009/571702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Huang W, Shi X, Ren L, Du C, Wang Y. Biomaterials. 2010;31:4278–4285. doi: 10.1016/j.biomaterials.2010.01.059. [DOI] [PubMed] [Google Scholar]
- 159.Dias M, Antunes MCM, Santos AR, Felisberti MI. Journal of Materials Science: Materials in Medicine. 2008;19:3535–3544. doi: 10.1007/s10856-008-3531-1. [DOI] [PubMed] [Google Scholar]
- 160.Dai S, Li Z. Biomacromolecules. 2008;9:1883–1893. doi: 10.1021/bm8001396. [DOI] [PubMed] [Google Scholar]
- 161.Wu L, Wang L, Wang X, Xu K. Acta Biomaterialia. 2010;6:1079–1089. doi: 10.1016/j.actbio.2009.08.014. [DOI] [PubMed] [Google Scholar]
- 162.Xue L, Dai S, Li Z. Biomaterials. 2010;31:8132–8140. doi: 10.1016/j.biomaterials.2010.07.043. [DOI] [PubMed] [Google Scholar]
- 163.Chen C, Yu CH, Cheng YC, Yu PHF, Cheung MK. Biomaterials. 2006;27:4804–4814. doi: 10.1016/j.biomaterials.2006.04.039. [DOI] [PubMed] [Google Scholar]
- 164.Chen C, Cheng YC, Yu CH, Chan SW, Cheung MK, Yu PHF. Journal of Biomedical Materials ResearchPart A. 2008;87A:290–298. doi: 10.1002/jbm.a.31719. [DOI] [PubMed] [Google Scholar]
- 165.Li X, Liu KL, Wang M, Wong SY, Tjiu WC, He CB, Goh SH, Li J. Acta Biomaterialia. 2009;5:2002–2012. doi: 10.1016/j.actbio.2009.01.035. [DOI] [PubMed] [Google Scholar]
- 166.Patlolla A, Collins G, Arinzeh TL. Acta Biomaterialia. 2010;6:90–101. doi: 10.1016/j.actbio.2009.07.028. [DOI] [PubMed] [Google Scholar]
- 167.Darney PD, Monroe SE, Klaisle CM, Alvarado A. American Journal of Obstetrics and Gynecology. 1989;160:1292–1295. doi: 10.1016/s0002-9378(89)80015-8. [DOI] [PubMed] [Google Scholar]
- 168.Ito Y, Ochii Y, Fukushima K, Sugioka N, Takada K. International Journal of Pharmaceutics. 2010;384:53–59. doi: 10.1016/j.ijpharm.2009.09.032. [DOI] [PubMed] [Google Scholar]
- 169.Lee WL, Foo WL, Widjaja E, Loo SCJ. Acta Biomaterialia. 2010;6:1342–1352. doi: 10.1016/j.actbio.2009.10.028. [DOI] [PubMed] [Google Scholar]
- 170.Rieger J, Freichels H, Imberty A, Putaux J-L, Delair T, Jerome C, Auzely-Velty R. Biomacromolecules. 2009;10:651–657. doi: 10.1021/bm801492c. [DOI] [PubMed] [Google Scholar]
- 171.Richter A, Olbrich C, Krause M, Kissel T. International Journal of Pharmaceutics. 2010;389:244–253. doi: 10.1016/j.ijpharm.2010.01.032. [DOI] [PubMed] [Google Scholar]
- 172.Singh J, Pandit S, Bramwell VW, Alpar HO. Methods. 2006;38:96–105. doi: 10.1016/j.ymeth.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 173.Mundargi RC, Srirangarajan S, Agnihotri SA, Patil SA, Ravindra S, Setty SB, Aminabhavi TM. Journal of Controlled Release. 2007;119:59–68. doi: 10.1016/j.jconrel.2007.01.008. [DOI] [PubMed] [Google Scholar]
- 174.Huang MJ, Gou ML, Qian ZY, Dai M, Li XY, Cao M, Wang K, Zhao J, Yang JL, Lu Y, Tu MJ, Wei YQ. Journal of Biomedical Materials Research Part A. 2008;86A:979–986. doi: 10.1002/jbm.a.31704. [DOI] [PubMed] [Google Scholar]
- 175.Hassan AS, Sapin A, Lamprecht A, Emond E, Ghazouani FE, Maincent P. European Journal of Pharmaceutics and Biopharmaceutics. 2009;73:337–344. doi: 10.1016/j.ejpb.2009.07.009. [DOI] [PubMed] [Google Scholar]
- 176.Huang M-H, Chou A-H, Lien S-P, Chen H-W, Huang C-Y, Chen W-W, Chong P, Liu S-J, Leng C-H. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2009;90B:832–841. doi: 10.1002/jbm.b.31352. [DOI] [PubMed] [Google Scholar]
- 177.Garkhal K, Verma S, Tikoo K, Kumar N. Journal of Biomedical Materials Research Part A. 2007;82A:747–756. doi: 10.1002/jbm.a.31150. [DOI] [PubMed] [Google Scholar]
- 178.Danhier F, Vroman B, Lecouturier N, Crokart N, Pourcelle V, Freichels H, Jerome C, Marchand-Brynaert J, Feron O, Preat V. Journal of Controlled Release. 2009;140:166–173. doi: 10.1016/j.jconrel.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 179.Li W-J, Cooper JA, Mauck RL, Tuan RS. Acta Biomaterialia. 2006;2:377–385. doi: 10.1016/j.actbio.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 180.Luciani A, Coccoli V, Orsi S, Ambrosio L, Netti PA. Biomaterials. 2008;29:4800–4807. doi: 10.1016/j.biomaterials.2008.09.007. [DOI] [PubMed] [Google Scholar]
- 181.Chung S, Ingle NP, Montero GA, Kim SH, King MW. Acta Biomaterialia. 2010;6:1958–1967. doi: 10.1016/j.actbio.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 182.Pankajakshan D, Philipose LP, Palakkal M, Krishnan K, Krishnan LK. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2008;87B:570–579. doi: 10.1002/jbm.b.31146. [DOI] [PubMed] [Google Scholar]
- 183.Chen H, Huang J, Yu J, Liu S, Ge P. International Journal of Biological Macromolecules. 2011;48:13–19. doi: 10.1016/j.ijbiomac.2010.09.019. [DOI] [PubMed] [Google Scholar]
- 184.Guarino V, Ambrosio L. Acta Biomaterialia. 2008;4:1778–1787. doi: 10.1016/j.actbio.2008.05.013. [DOI] [PubMed] [Google Scholar]
- 185.Plikk P, Malberg S, Albertsson A-C. Biomacromolecules. 2009;10:1259–1264. doi: 10.1021/bm900093r. [DOI] [PubMed] [Google Scholar]
- 186.Zuo Y, Yang F, Wolke JGC, Li Y, Jansen JA. Acta Biomaterialia. 2010;6:1238–1247. doi: 10.1016/j.actbio.2009.10.036. [DOI] [PubMed] [Google Scholar]
- 187.Mountziaris PM, Tzouanas SN, Mikos AG. Biomaterials. 2010;31:1666–1675. doi: 10.1016/j.biomaterials.2009.11.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Hayami JWS, Surrao DC, Waldman SD, Amsden BG. Journal of Biomedical Materials Research Part A. 2010;92A:1407–1420. doi: 10.1002/jbm.a.32472. [DOI] [PubMed] [Google Scholar]
- 189.Vaquette C, Kahn C, Frochot C, Nouvel C, Six J-L, Isla ND, Luo L-H, Cooper-White J, Rahouadj R, Wang X. Journal of Biomedical Materials Research Part A. 2010;94A:1270–1282. doi: 10.1002/jbm.a.32801. [DOI] [PubMed] [Google Scholar]
- 190.Li W-J, Chiang H, Kuo T-F, Lee H-S, Jiang C-C, Tuan RS. Journal of Tissue Engineering and Regenerative Medicine. 2009;3:1–10. doi: 10.1002/term.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Jeong SI, Lee A-Y, Lee YM, Shin H. Journal of Biomaterials Science. Polymer Edition. 2008;19:339–357. doi: 10.1163/156856208783721029. [DOI] [PubMed] [Google Scholar]
- 192.Nisbet DR, Rodda AE, Horne MK, Forsythe JS, Finkelstein DI. Biomaterials. 2009;30:4573–4580. doi: 10.1016/j.biomaterials.2009.05.011. [DOI] [PubMed] [Google Scholar]
- 193.Liu J-J, Wang C-Y, Wang J-G, Ruan H-J, Fan C-Y. Journal of Biomedical Materials Research Part A. 2011;96A:13–20. doi: 10.1002/jbm.a.32946. [DOI] [PubMed] [Google Scholar]
- 194.Heydarkhan-Hagvall S, Schenke-Layland K, Dhanasopon AP, Rofail F, Smith H, Wu BM, Shemin R, Beygui RE, MacLellan WR. Biomaterials. 2008;29:2907–2914. doi: 10.1016/j.biomaterials.2008.03.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Lee J, Choi WI, Tae G, Kim YH, Kang SS, Kim SE, Kim S-H, Jung Y, Kim SH. Acta Biomaterialia. 2011;7:244–257. doi: 10.1016/j.actbio.2010.08.017. [DOI] [PubMed] [Google Scholar]
- 196.He S, Timmer MD, Yaszemski MJ, Yasko AW, Engel PS, Mikos AG. Polymer. 2001;42:1251–1260. [Google Scholar]
- 197.Christenson EM, Soofi W, Holm JL, Cameron NR, Mikos AG. Biomacromolecules. 2007;8:3806–3814. doi: 10.1021/bm7007235. [DOI] [PubMed] [Google Scholar]
- 198.Kim CW, Talac R, Lu L, Moore MJ, Currier BL, Yaszemski MJ. Journal of Biomedical Materials Research Part A. 2008;85A:1114–1119. doi: 10.1002/jbm.a.31633. [DOI] [PubMed] [Google Scholar]
- 199.Young S, Patel ZS, Kretlow JD, Murphy MB, Mountziaris PM, Baggett LS, Ueda H, Tabata Y, Jansen JA, Wong M, Mikos AG. Tissue Engineering Part A. 2009;15:2347–2362. doi: 10.1089/ten.tea.2008.0510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Haesslein A, Ueda H, Hacker MC, Jo S, Ammon DM, Borazjani RN, Kunzler JF, Salamone JC, Mikos AG. Journal of Controlled Release. 2006;114:251–260. doi: 10.1016/j.jconrel.2006.05.024. [DOI] [PubMed] [Google Scholar]
- 201.Ueda H, Hacker MC, Haesslein A, Jo S, Ammon DM, Borazjani RN, Kunzler JF, Salamone JC, Mikos AG. Journal of Biomedical Materials Research Part A. 2007;83A:656–666. doi: 10.1002/jbm.a.31226. [DOI] [PubMed] [Google Scholar]
- 202.Hacker MC, Haesslein A, Ueda H, Foster WJ, Garcia CA, Ammon DM, Borazjani RN, Kunzler JF, Salamone JC, Mikos AG. Journal of Biomedical Materials Research Part A. 2009;88A:976–989. doi: 10.1002/jbm.a.31942. [DOI] [PubMed] [Google Scholar]
- 203.Lee K-W, Wang S, Yaszemski MJ, Lu L. Biomaterials. 2008;29:2839–2848. doi: 10.1016/j.biomaterials.2008.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Jayabalan M, Shalumon KT, Mitha MK, Ganesan K, Epple M. Acta Biomaterialia. 2010;6:763–775. doi: 10.1016/j.actbio.2009.09.015. [DOI] [PubMed] [Google Scholar]
- 205.Lee K-W, Wang S, Dadsetan M, Yaszemski MJ, Lu L. Biomacromolecules. 2010;11:682–689. doi: 10.1021/bm901260y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Mistry AS, Mikos AG, Jansen JA. Journal of Biomedical Materials Research Part A. 2007;83A:940–953. doi: 10.1002/jbm.a.31280. [DOI] [PubMed] [Google Scholar]
- 207.Mistry AS, Cheng SH, Yeh T, Christenson E, Jansen JA, Mikos AG. Journal of Biomedical Materials Research Part A. 2009;89A:68–79. doi: 10.1002/jbm.a.32010. [DOI] [PubMed] [Google Scholar]
- 208.Mistry AS, Pham QP, Schouten C, Yeh T, Christenson EM, Mikos AG, Jansen JA. Journal of Biomedical Materials Research Part A. 2010;92A:451–462. doi: 10.1002/jbm.a.32371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Danti S, D’Alessandro D, Pietrabissa A, Petrini M, Berrettini S. Journal of Biomedical Materials Research Part A. 2010;92A:1343–1356. doi: 10.1002/jbm.a.32447. [DOI] [PubMed] [Google Scholar]
- 210.Nguyen C, Young S, Kretlow JD, Mikos AG, Wong M. Journal of Oral and Maxillofacial Surgery. 2011;69:11–18. doi: 10.1016/j.joms.2010.02.026. [DOI] [PubMed] [Google Scholar]
- 211.Hill JW. Journal of the American Chemical Society. 1932;54:4105–4106. [Google Scholar]
- 212.Rosen HB, Chang J, Wnek GE, Linhardt RJ, Langer R. Biomaterials. 1983;4:131–133. doi: 10.1016/0142-9612(83)90054-6. [DOI] [PubMed] [Google Scholar]
- 213.Katti DS, Lakshmi S, Langer R, Laurencin CT. Advanced Drug Delivery Reviews. 2002;54:933–961. doi: 10.1016/s0169-409x(02)00052-2. [DOI] [PubMed] [Google Scholar]
- 214.Agueros M, Ruiz-Gaton L, Vauthier C, Bouchemal K, Espuelas S, Ponchel G, Irache JM. European Journal of Pharmaceutical Sciences. 2009;38:405–413. doi: 10.1016/j.ejps.2009.09.010. [DOI] [PubMed] [Google Scholar]
- 215.Agueros M, Zabaleta V, Espuelas S, Campanero MA, Irache JM. Journal of Controlled Release. 2010;145:2–8. doi: 10.1016/j.jconrel.2010.03.012. [DOI] [PubMed] [Google Scholar]
- 216.Krasko MY, Golenser J, Nyska A, Nyska M, Brin YS, Domb AJ. Journal of Controlled Release. 2007;117:90–96. doi: 10.1016/j.jconrel.2006.10.010. [DOI] [PubMed] [Google Scholar]
- 217.Brin YS, Golenser J, Mizrahi B, Maoz G, Domb AJ, Peddada S, Tuvia S, Nyska A, Nyska M. Journal of Controlled Release. 2008;131:121–127. doi: 10.1016/j.jconrel.2008.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kipper MJ, Wilson JH, Wannemuehler MJ, Narasimhan B. Journal of Biomedical Materials Research Part A. 2006;76A:798–810. doi: 10.1002/jbm.a.30545. [DOI] [PubMed] [Google Scholar]
- 219.Salman HH, Irache JM, Gamazo C. Vaccine. 2009;27:4784–4790. doi: 10.1016/j.vaccine.2009.05.091. [DOI] [PubMed] [Google Scholar]
- 220.Carrillo-Conde B, Schiltz E, Yu J, Minion FC, Phillips GJ, Wannemuehler MJ, Narasimhan B. Acta Biomaterialia. 2010;6:3110–3119. doi: 10.1016/j.actbio.2010.01.040. [DOI] [PubMed] [Google Scholar]
- 221.Tamayo I, Irache JM, Mansilla C, Ochoa-Reparaz J, Lasarte JJ, Gamazo C. Clinical and Vaccine Immunology. 2010:1356–1362. 9. doi: 10.1128/CVI.00164-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Sun L, Zhou S, Wang W, Su Q, Li X, Weng J. Journal of Materials Science: Materials in Medicine. 2009;20:2035–2042. doi: 10.1007/s10856-009-3765-6. [DOI] [PubMed] [Google Scholar]
- 223.Petersen LK, Sackett CK, Narasimhan B. Acta Biomaterialia. 2010;6:3873–3881. doi: 10.1016/j.actbio.2010.04.004. [DOI] [PubMed] [Google Scholar]
- 224.Ulery BD, Phanse Y, Sinha A, Wannemuehler MJ, Narasimhan B, Bellaire BH. Pharmaceutical Research. 2009;26:683–690. doi: 10.1007/s11095-008-9760-7. [DOI] [PubMed] [Google Scholar]
- 225.Petersen LK, Xue L, Wannemuehler MJ, Rajan K, Narasimhan B. Biomaterials. 2009;30:5131–5142. doi: 10.1016/j.biomaterials.2009.05.069. [DOI] [PubMed] [Google Scholar]
- 226.Manoharan C, Singh J. Journal of Pharmaceutical Sciences. 2009;98:4237–4250. doi: 10.1002/jps.21741. [DOI] [PubMed] [Google Scholar]
- 227.Tian Y, Li L, Gao X, Deng J, Stephens D, Robinson D, Chang H. Drug Development and Industrial Pharmacy. 2002;28:897–903. doi: 10.1081/ddc-120006422. [DOI] [PubMed] [Google Scholar]
- 228.Jain JP, Modi S, Kumar N. Journal of Biomedical Materials Research Part A. 2008;84A:740–752. doi: 10.1002/jbm.a.31456. [DOI] [PubMed] [Google Scholar]
- 229.Determan AS, Wilson JH, Kipper MJ, Wannemuehler MJ, Narasimhan B. Biomaterials. 2006;27:3312–3320. doi: 10.1016/j.biomaterials.2006.01.054. [DOI] [PubMed] [Google Scholar]
- 230.Torres MP, Vogel BM, Narasimhan B, Mallapragada SK. Journal of Biomedical Materials Research Part A. 2006;76A:102–110. doi: 10.1002/jbm.a.30510. [DOI] [PubMed] [Google Scholar]
- 231.Torres MP, Determan AS, Anderson GL, Mallapragada SK, Narasimhan B. Biomaterials. 2007;28:108–116. doi: 10.1016/j.biomaterials.2006.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Weiner AA, Shuck DM, Bush JR, Shastri VP. Biomaterials. 2007;28:5259–5270. doi: 10.1016/j.biomaterials.2007.08.022. [DOI] [PubMed] [Google Scholar]
- 233.Tarcha PJ, Su L, Baker T, Langridge D, Shastri V, Langer R. Journal of Polymer Science Part A: Polymer Chemistry. 2001;39:4189–4195. [Google Scholar]
- 234.Weiner AA, Bock EA, Gipson ME, Shastri VP. Biomaterials. 2008;29:2400–2407. doi: 10.1016/j.biomaterials.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 235.Weiner AA, Moore MC, Walker AH, Shastri VP. International Journal of Pharmaceutics. 2008;360:107–114. doi: 10.1016/j.ijpharm.2008.04.037. [DOI] [PubMed] [Google Scholar]
- 236.Heffernan MJ, Murthy N. Bioconjugate Chemistry. 2005;16:1340–1342. doi: 10.1021/bc050176w. [DOI] [PubMed] [Google Scholar]
- 237.Lee S, Yang SC, Heffernan MJ, Taylor WR, Murthy N. Bioconjugate Chemistry. 2007;18:4–7. doi: 10.1021/bc060259s. [DOI] [PubMed] [Google Scholar]
- 238.Lee S, Yang SC, Kao C-Y, Pierce RH, Murthy N. Nucleic Acids Research. 2009:37. doi: 10.1093/nar/gkp758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Goh SL, Murthy N, Xu M, Frechet JMJ. Bioconjugate Chemistry. 2004;15:2004. doi: 10.1021/bc034159n. [DOI] [PubMed] [Google Scholar]
- 240.Murthy N, Xu M, Schuck S, Kunisawa J, Shastri N, Frechet JMJ. Proceedings of the National Academy of Sciences of the United States of America. 2003;100:4995–5000. doi: 10.1073/pnas.0930644100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Standley SM, Kwon YJ, Murthy N, Kunisawa J, Shastri N, Guillaudeu SJ, Lau L, Frechet JMJ. Bioconjugate Chemistry. 2004;15:1281–1288. doi: 10.1021/bc049956f. [DOI] [PubMed] [Google Scholar]
- 242.Yang SC, Bhide M, Crispe IN, Pierce RH, Murthy N. Bioconjugate Chemistry. 2008;19:1164–1169. doi: 10.1021/bc700442g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Heffernan MJ, Kasturi SP, Yang SC, Pulendran B, Murthy N. Biomaterials. 2009;30:910–918. doi: 10.1016/j.biomaterials.2008.10.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Seshadri G, Sy JC, Brown M, Dikalov S, Yang SC, Murthy N, Davis ME. Biomaterials. 2010;31:1372–1379. doi: 10.1016/j.biomaterials.2009.10.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Bjork VO. Scandinavian journal of thoracic and cardiovascular surgery. 1969;3:1–10. doi: 10.3109/14017436909131912. [DOI] [PubMed] [Google Scholar]
- 246.Larmi TK, Karkola P. The Journal of Thoracic and Cardiovascular Surgery. 1974;68:66–69. [PubMed] [Google Scholar]
- 247.Moreau JL, Kesselman D, Fisher JP. Journal of Biomedical Materials Research Part A. 2007;81A:594–602. doi: 10.1002/jbm.a.31104. [DOI] [PubMed] [Google Scholar]
- 248.Betz MW, Modi PC, Caccamese JF, Coletti DP, Sauk JJ, Fisher JP. Journal of Biomedical Materials Research Part A. 2008;86A:662–670. doi: 10.1002/jbm.a.31640. [DOI] [PubMed] [Google Scholar]
- 249.Falco EE, Roth JS, Fisher JP. Journal of Surgical Research. 2008;149:76–83. doi: 10.1016/j.jss.2007.08.016. [DOI] [PubMed] [Google Scholar]
- 250.Choi NS, Heller J USPTO. United States; 1978. [Google Scholar]
- 251.Qi M, Li X, Yang Y, Zhou S. European Journal of Pharmaceutics and Biopharmaceutics. 2008;70:445–452. doi: 10.1016/j.ejpb.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 252.Wang C, Ge Q, Ting D, Nguyen D, Shen H-R, Chen J, Eisen HN, Heller J, Langer R, Putnam D. Nature Materials. 2004;3:190–196. doi: 10.1038/nmat1075. [DOI] [PubMed] [Google Scholar]
- 253.Nguyen DN, Raghavan SS, Tashima LM, Lin EC, Fredette SJ, Langer RS, Wang C. Biomaterials. 2008;29:2783–2793. doi: 10.1016/j.biomaterials.2008.03.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Polak MB, Valamanesh F, Felt O, Torriglia A, Jeanny J-C, Bourges J-L, Rat P, Thomas-Doyle A, BenEzra D, Gurny R, Behar-Cohen F. Investigative Opththalmology & Visual Science. 2008;49:2993–3003. doi: 10.1167/iovs.07-0919. [DOI] [PubMed] [Google Scholar]
- 255.Ekholm M, Helander P, Hietanen J, Lindqvist C, Salo A, Kellomaki M, Suuronen R. International Journal of Oral and Maxillofacial Surgery. 2006;35:631–635. doi: 10.1016/j.ijom.2006.01.029. [DOI] [PubMed] [Google Scholar]
- 256.Zhang Z, Kuijer R, Bulstra SK, Grijpma DW, Feijen J. Biomaterials. 2006;27:1741–1748. doi: 10.1016/j.biomaterials.2005.09.017. [DOI] [PubMed] [Google Scholar]
- 257.Pego AP, Luyn MJAv, Brouwer LA, Wachem PBv, Poot AA, Grijpma DW, Feijen J. Journal of Biomedical Materials Research Part A. 2003;67A:1044–1054. doi: 10.1002/jbm.a.10121. [DOI] [PubMed] [Google Scholar]
- 258.Zhang Z, Foks MA, Grijpma DW, Feijen J. Journal of Controlled Release. 2005;101:392–394. [PubMed] [Google Scholar]
- 259.Habraken WJEM, Zhang Z, Wolke JGC, Grijpma DW, Mikos AG, Feijen J, Jansen JA. Biomaterials. 2008;29:2464–2476. doi: 10.1016/j.biomaterials.2008.02.012. [DOI] [PubMed] [Google Scholar]
- 260.Kluin OS, Mei HCvd, Busscher HJ, Neut D. Biomaterials. 2009;30:4738–4742. doi: 10.1016/j.biomaterials.2009.05.012. [DOI] [PubMed] [Google Scholar]
- 261.Neut D, Kluin OS, Crielaard BJ, Mei HCvd, Busscher HJ, Grijpma DW. Acta Orthopaedica. 2009;80:514–519. doi: 10.3109/17453670903350040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Timbart L, Tse MY, Pang SC, Babasola O, Amsden BG. Macromolecular Bioscience. 2009;9:786–794. doi: 10.1002/mabi.200800318. [DOI] [PubMed] [Google Scholar]
- 263.Bat E, Feijen J, Grijpma DW. Biomacromolecules. 2010;11:2692–2699. doi: 10.1021/bm1007234. [DOI] [PubMed] [Google Scholar]
- 264.Amsden BG, Timbart L, Marecak D, Chapanian R, Tse MY, Pang SC. Journal of Controlled Release. 2010;145:109–115. doi: 10.1016/j.jconrel.2010.03.029. [DOI] [PubMed] [Google Scholar]
- 265.Zurita R, Puiggali J, Rodriguez-Galan A. Macromolecular Bioscience. 2006;6:767–775. doi: 10.1002/mabi.200600084. [DOI] [PubMed] [Google Scholar]
- 266.Chen W, Meng F, Li F, Ji S-J, Zhong Z. Biomacromolecules. 2009;10:1727–1735. doi: 10.1021/bm900074d. [DOI] [PubMed] [Google Scholar]
- 267.Chen W, Meng F, Cheng R, Zhong Z. Journal of Controlled Release. 2010;142:40–46. doi: 10.1016/j.jconrel.2009.09.023. [DOI] [PubMed] [Google Scholar]
- 268.Kim SH, Tan JPK, Nederberg F, Fukushima K, Colson J, Yang C, Nelson A, Yang Y-Y, Hedrick JL. Biomaterials. 2010;31:8063–8071. doi: 10.1016/j.biomaterials.2010.07.018. [DOI] [PubMed] [Google Scholar]
- 269.Sanson C, Schatz C, Meins J-FL, Brulet A, Soum A, Lecommandoux S. Langmuir. 2010;26:2751–2760. doi: 10.1021/la902786t. [DOI] [PubMed] [Google Scholar]
- 270.Sanson C, Schatz C, Meins J-FL, Soum A, Thevenot J, Garanger E, Lecommandoux S. Journal of Controlled Release. 2010;147:428–435. doi: 10.1016/j.jconrel.2010.07.123. [DOI] [PubMed] [Google Scholar]
- 271.Welle A, Kroger M, Doring M, Niederer K, Pindel E, Chronakis IS. Biomaterials. 2007;28:2211–2219. doi: 10.1016/j.biomaterials.2007.01.024. [DOI] [PubMed] [Google Scholar]
- 272.Suriano F, Pratt R, Tan JPK, Wiradharma N, Nelson A, Yang Y-Y, Dubois P, Hedrick JL. Biomaterials. 2010;31:2637–2645. doi: 10.1016/j.biomaterials.2009.12.022. [DOI] [PubMed] [Google Scholar]
- 273.Zelikin AN, Zawaneh PN, Putnam D. Biomacromolecules. 2006;7:3239–3244. doi: 10.1021/bm060544e. [DOI] [PubMed] [Google Scholar]
- 274.Henderson PW, Kadouch DJM, Singh SP, Zawaneh PN, Wieser J, Yazdi S, Weinstein A, Krotscheck U, Wechsler B, Putnam D, Spector JA. Journal of Biomedical Materials Research Part A. 2010;93A:776–782. doi: 10.1002/jbm.a.32586. [DOI] [PubMed] [Google Scholar]
- 275.Zawaneh PN, Singh SP, Padera RF, Henderson PW, Spector JA, Putnam D. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:11014–11019. doi: 10.1073/pnas.0811529107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Ertel SI, Kohn J. Journal of Biomedical Materials Research. 1994;28:919–930. doi: 10.1002/jbm.820280811. [DOI] [PubMed] [Google Scholar]
- 277.Asikainen AJ, Noponen J, Mesimaki K, Laitinen O, Peltola J, Pelto M, Kellomaki M, Ashammakhi N, Lindqvist C, Suuronen R. Journal of Materials Science: Materials in Medicine. 2005;16:753–758. doi: 10.1007/s10856-005-2613-6. [DOI] [PubMed] [Google Scholar]
- 278.Asikainen AJ, Noponen J, Lindqvist C, Pelto M, Kellomaki M, Juuti H, Pihlajamaki H, Suuoronen R. Journal of the Royal Society, Interface. 2006;3:629–635. doi: 10.1098/rsif.2006.0119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Bailey LO, Becker ML, Stephens JS, Gallant ND, Mahoney CM, Washburn NR, Rege A, Kohn J, Amis EJ. Journal of Biomedical Materials Research. 2006;76A:491–502. doi: 10.1002/jbm.a.30527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Briggs T, Treiser MD, Holmes PF, Kohn J, Moghe PV, Arinzeh TL. Journal of Biomedical Materials Research Part A. 2009;91A:975–984. doi: 10.1002/jbm.a.32310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Johnson PA, Luk A, Demtchouk A, Patel H, Sung H-J, Treiser MD, Gordonov S, Sheihet L, Bolikal D, Kohn J, Moghe PV. Journal of Biomedical Materials Research Part A. 2010;93A:505–514. doi: 10.1002/jbm.a.32544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Meechaisue C, Dubin R, Supaphol P, Hoven VP, Kohn J. Journal of Biomaterials Science. Polymer Edition. 2006;17:1039–1056. doi: 10.1163/156856206778365988. [DOI] [PubMed] [Google Scholar]
- 283.Sung HJ, Labazzo KMS, Bolikal D, Weiner MJ, Zimnisky R, Kohn J. European Cells & Materials. 2008;25:77–87. doi: 10.22203/ecm.v015a06. [DOI] [PubMed] [Google Scholar]
- 284.Costache MC, Qu H, Ducheyne P, Devore DI. Biomaterials. 2010;31:6336–6343. doi: 10.1016/j.biomaterials.2010.04.065. [DOI] [PubMed] [Google Scholar]
- 285.Tangpasuthadol V, Pendharkar SM, Peterson RC, Kohn J. Biomaterials. 2000;21:2379–2387. doi: 10.1016/s0142-9612(00)00105-8. [DOI] [PubMed] [Google Scholar]
- 286.Guelcher SA. Tissue Engineering Part B: Reviews. 2008;14:3–17. doi: 10.1089/teb.2007.0133. [DOI] [PubMed] [Google Scholar]
- 287.Wen J, Somorjai G. Macromolecules. 1997;30:7206–7213. [Google Scholar]
- 288.Asai T, Lee M-H, Arrecubieta C, Bayern MPv, Cespedes CA, Baron HM, Cadeiras M, Sakguchi T. The Journal of Thoracic and Cardiovascular Surgery. 2007;133:1147–1153. doi: 10.1016/j.jtcvs.2006.10.084. [DOI] [PubMed] [Google Scholar]
- 289.Uttayarat P, Perets A, Li M, Pimpton P, Stachelek SJ, Alferiev I, Composto RJ, Levy RJ, Lelkes PI. Acta Biomaterialia. 2010;6:4229–4237. doi: 10.1016/j.actbio.2010.06.008. [DOI] [PubMed] [Google Scholar]
- 290.Poelaert J, Depuydt P, Wolf AD, Velde SVd, Herck I, Blot S. The Journal of Thoracic and Cardiovascular Surgery. 2008;135:771–776. doi: 10.1016/j.jtcvs.2007.08.052. [DOI] [PubMed] [Google Scholar]
- 291.Backman S, Bjorling G, Johansson U-B, Lysdahl M, Markstrom A, Schedin U, Aune RE, Frostell C, Karlsson S. The Laryngoscope. 2009;119:657–664. doi: 10.1002/lary.20048. [DOI] [PubMed] [Google Scholar]
- 292.Bonzani IC, Adhikari R, Houshyar S, Mayadunne R, Gunatillake P, Stevens MM. Biomaterials. 2007;28:423–433. doi: 10.1016/j.biomaterials.2006.08.026. [DOI] [PubMed] [Google Scholar]
- 293.Allcock HR, Kugel RL. Journal of the American Chemical Society. 1965;87:4216–4217. [Google Scholar]
- 294.Laurencin CT, Koh HJ, Neenan TX, Allcock HR, Langer R. Journal of Biomedical Materials Research. 1987;21:1231–1246. doi: 10.1002/jbm.820211006. [DOI] [PubMed] [Google Scholar]
- 295.Lakshmi S, Katti DS, Laurencin CT. Advanced Drug Delivery Reviews. 2003;55:467–482. doi: 10.1016/s0169-409x(03)00039-5. [DOI] [PubMed] [Google Scholar]
- 296.Singh A, Krogmas NR, Sethuraman S, Nair LS, Sturgeon JL, Brown PW, Laurencin CT, Allcock HR. Biomacromolecules. 2006;7:914–918. doi: 10.1021/bm050752r. [DOI] [PubMed] [Google Scholar]
- 297.Weikel AL, Owens SG, Morozowich NL, Deng M, Nair LS, Laurencin CT, Allcock HR. Biomaterials. 2010;31:8507–8515. doi: 10.1016/j.biomaterials.2010.07.094. [DOI] [PubMed] [Google Scholar]
- 298.Radeleff B, Thierjung H, Stampfl U, Stampfl S, Lopez-Benitez R, Sommer C, Berger I, Richter GM. Cardiovascular and Interventional Radiology. 2008;31:971–980. doi: 10.1007/s00270-007-9243-y. [DOI] [PubMed] [Google Scholar]
- 299.Smeets AJ, Nijenhuis RJ, Rooij WJv, Lampmann LEH, Boekkooi PF, Vervest HAM, Vries JD, Lohle PNM. Journal of Vascular and Interventional Radiology. 2010;21:1830–1834. doi: 10.1016/j.jvir.2010.09.014. [DOI] [PubMed] [Google Scholar]
- 300.Stampfl U, Sommer C-M, Thierjung H, Stampfl S, Lopez-Benitez R, Radeleff B, Berger I, Richter GM. Cardiovascular and Interventional Radiology. 2008;31:1184–1192. doi: 10.1007/s00270-008-9392-7. [DOI] [PubMed] [Google Scholar]
- 301.Verret V, Wasser M, Pelage J-P, Ghegediban SH, Jouneau L, Moine L, Labarre D, Golzarian J, Schwartz-Cornil I, Laurent A. Biomaterials. 2011;32:339–351. doi: 10.1016/j.biomaterials.2010.09.001. [DOI] [PubMed] [Google Scholar]
- 302.Jun YJ, Kim JH, Choi SJ, Lee HJ, Jun MJ, Sohn YS. Bioorganic & Medicinal Chemistry Letters. 2007;17:2975–2978. doi: 10.1016/j.bmcl.2007.03.069. [DOI] [PubMed] [Google Scholar]
- 303.Yang Y, Xu Z, Chen S, Gao Y, Gu Y, Chen L, Pei Y, Li Y. International Journal of Pharmaceutics. 2008;353:277–282. doi: 10.1016/j.ijpharm.2007.11.041. [DOI] [PubMed] [Google Scholar]
- 304.Yang Y, Zhang Z, Chen L, Gu W, Li Y. Bioconjugate Chemistry. 2010;21:419–426. doi: 10.1021/bc900267g. [DOI] [PubMed] [Google Scholar]
- 305.Andrianov AK, Marin A, Chen J. Biomacromolecules. 2006;7:394–399. doi: 10.1021/bm050790a. [DOI] [PubMed] [Google Scholar]
- 306.Zhang JX, Li XJ, Qiu LY, Li XH, Yan MQ, Jin Y, Zhu KJ. Journal of Controlled Release. 2006;116:322–329. doi: 10.1016/j.jconrel.2006.09.013. [DOI] [PubMed] [Google Scholar]
- 307.Qiu LY, Wu XL, Jin Y. Pharmaceutical Research. 2009;26:946–957. doi: 10.1007/s11095-008-9797-7. [DOI] [PubMed] [Google Scholar]
- 308.Kovacs-Nolan J, Mapletoft JW, Lawman Z, Babiuk LA, Hurk SvDL-vd. The Journal of General Virology. 2009;90:1892–1905. doi: 10.1099/vir.0.011684-0. [DOI] [PubMed] [Google Scholar]
- 309.Zheng C, Qiu L, Yao X, Zhu K. International Journal of Pharmaceutics. 2009;373:133–140. doi: 10.1016/j.ijpharm.2009.01.025. [DOI] [PubMed] [Google Scholar]
- 310.Andrianov AK, DeCollibus DP, Gillis HA, Kha HH, Marin A, Prausnitz MR, Babiuk LA, Townsend H, Mutwiri G. Proceedings of the National Academy of Sciences of the United States of America. 2009;106:18936–18941. doi: 10.1073/pnas.0908842106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Andrianov AK, Marin A, DeCollibus DP. Pharmaceutical Research. 2011;28:58–65. doi: 10.1007/s11095-010-0133-7. [DOI] [PubMed] [Google Scholar]
- 312.Seong J-Y, Jun YJ, Kim BM, Park YM, Sohn YS. International Journal of Pharmaceutics. 2006;314:90–96. doi: 10.1016/j.ijpharm.2006.02.009. [DOI] [PubMed] [Google Scholar]
- 313.Park M-R, Chun C, Ahn S-W, Ki M-H, Cho C-S, Song S-C. Journal of Controlled Release. 2010;147:359–367. doi: 10.1016/j.jconrel.2010.07.126. [DOI] [PubMed] [Google Scholar]
- 314.Potta T, Chun C, Song S-C. Biomaterials. 2009;30:6178–6192. doi: 10.1016/j.biomaterials.2009.08.015. [DOI] [PubMed] [Google Scholar]
- 315.Chun C, Lee SM, Kim CW, Hong K-Y, Kim SY, Yang HK, Song S-C. Biomaterials. 2009;30:4752–4762. doi: 10.1016/j.biomaterials.2009.05.031. [DOI] [PubMed] [Google Scholar]
- 316.Oredein-McCoy O, Krogman NR, Weikel AL, Hindenlang MD, Allcock HR, Laurencin CT. Journal of Microencapsulation. 2009;26:544–555. doi: 10.1080/02652040802500473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Aldini NN, Fini M, Rocca M, Giavaresi G. Internation Orthopaedics. 2000;24:121–125. doi: 10.1007/s002640000142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Deng M, Nair LS, Nukavarapu SP, Kumbar SG, Jiang T, Krogman NR, Singh A, Allcock HR, Laurencin CT. Biomaterials. 2008;29:337–349. doi: 10.1016/j.biomaterials.2007.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Conconi MT, Lora S, Baiguera S, Boscolo E, Folin M, Scienza R, Rebuffat P, Parnigotto PP, Nussdorfer GG. Journal of Biomedical Materials Research Part A. 2004;71A:669–674. doi: 10.1002/jbm.a.30198. [DOI] [PubMed] [Google Scholar]
- 320.Nair LS, Bhattacharyya S, Bender JD, Greish YE, Brown PW, Allcock HR, Laurencin CT. Biomacromolecules. 2004;5:2212–2220. doi: 10.1021/bm049759j. [DOI] [PubMed] [Google Scholar]
- 321.Conconi MT, Lora S, Menti AM, Carampin P, Parnigotto PP. Tissue Engineering. 2006;12:811–819. doi: 10.1089/ten.2006.12.811. [DOI] [PubMed] [Google Scholar]
- 322.Zhang QS, Yan YH, Li SP, Feng T. Biomedical Materials. 2009;4:035008. doi: 10.1088/1748-6041/4/3/035008. [DOI] [PubMed] [Google Scholar]
- 323.Nukavarapu SP, Kumbar SG, Brown JL, Krogman NR, Weikel AL, Hindenlang MD, Nair LS, Allcock HR, Laurencin CT. Biomacromolecules. 2008;9:1818–1825. doi: 10.1021/bm800031t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Lapienis G, Penczek S. Macromolecules. 1974;7:166–174. [Google Scholar]
- 325.Kaluzynski K, Libisowski J, Penczek S. Macromolecules. 1976;9:365–367. [Google Scholar]
- 326.Wang Y-C, Liu X-Q, Sun T-M, Xiong M-H, Wang J. Journal of Controlled Release. 2008;128:32–40. doi: 10.1016/j.jconrel.2008.01.021. [DOI] [PubMed] [Google Scholar]
- 327.Wang Y-C, Tang L-Y, Sun T-M, Li C-H, Xiong M-H, Wang J. Biomacromolecules. 2008;9:388–395. doi: 10.1021/bm700732g. [DOI] [PubMed] [Google Scholar]
- 328.Liu J, Huang W, Pang Y, Zhu X, Zhou Y, Yan D. Biomacromolecules. 2010;11:1564–1570. doi: 10.1021/bm100188h. [DOI] [PubMed] [Google Scholar]
- 329.Lu Z-Z, Wu J, Sun T-M, Ji J, Yan L-F, Wang J. Biomaterials. 2008;29:733–741. doi: 10.1016/j.biomaterials.2007.10.033. [DOI] [PubMed] [Google Scholar]
- 330.Ditto AJ, Shah PN, Gump LR, Yun YH. Molecular Pharmaceutics. 2009;6:986–995. doi: 10.1021/mp9000316. [DOI] [PubMed] [Google Scholar]
- 331.Yang X-Z, Sun T-M, Dou S, Wu J, Wang Y-C, Wang J. Biomacromolecules. 2009;10:2213–2220. doi: 10.1021/bm900390k. [DOI] [PubMed] [Google Scholar]
- 332.Li Q, Wang J, Shahani S, Sun DDN, Sharma B, Elisseeff JH, Leong KW. Biomaterials. 2005;27:1027–1034. doi: 10.1016/j.biomaterials.2005.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Du J-Z, Sun T-M, Weng S-Q, Chen X-S, Wang J. Biomacromolecules. 2007;8:3375–3381. doi: 10.1021/bm700474b. [DOI] [PubMed] [Google Scholar]
- 334.Zhang Z, Mao J, Feng X, Xiao J, Qiu J. Journal of Huazhong University of Science and Technology -Medical Sciences. 2008;28:604–607. doi: 10.1007/s11596-008-0526-6. [DOI] [PubMed] [Google Scholar]
- 335.Qiu J-J, He Z-X, Liu C-M, Guo X-D, Zheng Q-X. Biomedical Materials. 2008;3:044107. doi: 10.1088/1748-6041/3/4/044107. [DOI] [PubMed] [Google Scholar]
- 336.Qiu Z-C, Zhang J-J, Zhou Y, Song B-Y, Chang J-J, Yang K-K, Wang Y-Z. Polymers for Advanced Technologies. 2009 [Google Scholar]
- 337.Malnati GA, Stone EA. Veterinary Surgery. 1983;12:24–25. [Google Scholar]
- 338.Olson DO, Sheares VV. Macromolecules. 2006;39:2808–2814. [Google Scholar]
- 339.Olson DA, Gratton SEA, DeSimone JM, Sheares VV. Journal of the American Chemical Society. 2006;128:13625–13633. doi: 10.1021/ja063092m. [DOI] [PubMed] [Google Scholar]
- 340.Barrera DA, Zylstra E, Lansbury PT, Langer R. Journal of the American Chemical Society. 1993;115:11010–11011. [Google Scholar]
- 341.Putnam D, Langer R. Macromolecules. 1999;32:3658–3662. [Google Scholar]
- 342.Green JJ, Zugates GT, Langer R, Anderson DG. Macromolecular Drug Delivery. 2009;480:53–63. doi: 10.1007/978-1-59745-429-2_4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Lynn DM, Anderson DG, Putnam D, Langer R. Journal of the American Chemical Society. 2001;123:8155–8156. doi: 10.1021/ja016288p. [DOI] [PubMed] [Google Scholar]
- 344.Akinc A, Lynn DM, Anderson DG, Langer R. Journal of the American Chemical Society. 2003;125:5316–5323. doi: 10.1021/ja034429c. [DOI] [PubMed] [Google Scholar]
- 345.Anderson DG, Lynn DM, Langer R. Angewandte Chemie International Edition. 2003;42:3153–3158. doi: 10.1002/anie.200351244. [DOI] [PubMed] [Google Scholar]
- 346.Jere D, Yoo M-K, Arote R, Kim T-H, Cho M-H, Nah J-W, Choi Y-J, Cho C-S. Pharmaceutical Research. 2008;25:875–885. doi: 10.1007/s11095-007-9448-4. [DOI] [PubMed] [Google Scholar]
- 347.Little SR, Lynn DM, Puram SV, Langer R. Journal of Controlled Release. 2005;107:449–462. doi: 10.1016/j.jconrel.2005.04.022. [DOI] [PubMed] [Google Scholar]
- 348.Greenland JR, Liu H, Berry D, Anderson DG, Kim W-K, Irvine DJ, Langer R, Letvin NL. Molecular Therapy. 2005;12:164–170. doi: 10.1016/j.ymthe.2005.01.021. [DOI] [PubMed] [Google Scholar]
- 349.Zugates GT, Peng W, Zumbuehl A, Jhunjhunwala S, Huang YH, Langer R, Sawicki JA, Anderson DG. Molecular Therapy. 2007;15:1306–1312. doi: 10.1038/sj.mt.6300132. [DOI] [PubMed] [Google Scholar]
- 350.Brito L, Little S, Langer R, Amiji M. Biomacromolecules. 2008;9:1179–1187. doi: 10.1021/bm7011373. [DOI] [PubMed] [Google Scholar]
- 351.Bettinger CJ, Bruggeman JP, Borenstein JT, Langer RS. Biomaterials. 2008;29:2315–2325. doi: 10.1016/j.biomaterials.2008.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Brey DM, Chung C, Hankenson KD, Garino JP, Burdick JA. Journal of Biomedical Materials Research Part A. 2010;93A:807–816. doi: 10.1002/jbm.a.32769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Horwitz JA, Shum KM, Bodle JC, Deng M, Chu C-C, Reinhart-King CA. Journal of Biomedical Materials Research Part A. 2010;95A:371–380. doi: 10.1002/jbm.a.32858. [DOI] [PubMed] [Google Scholar]
- 354.Erdmann L, Uhrich KE. Biomaterials. 2000;21:1941–1946. doi: 10.1016/s0142-9612(00)00073-9. [DOI] [PubMed] [Google Scholar]
- 355.Whitaker-Brothers K, Uhrich K. Journal of Biomedical Materials Research Part A. 2006;76A:470–479. doi: 10.1002/jbm.a.30356. [DOI] [PubMed] [Google Scholar]
- 356.Erdmann L, Macedo B, Uhrich KE. Biomaterials. 2000;21:2507–2512. doi: 10.1016/s0142-9612(00)00118-6. [DOI] [PubMed] [Google Scholar]
- 357.Yeagy BA, Prudencio A, Schmeltzer RC, Uhrich KE, Cook TJ. Journal of Microencapsulation. 2006;23:643–653. doi: 10.1080/02652040600776481. [DOI] [PubMed] [Google Scholar]
- 358.Whitaker-Brothers K, Uhrich K. Journal of Biomedical Materials Research Part A. 2004;70A:309–318. doi: 10.1002/jbm.a.30083. [DOI] [PubMed] [Google Scholar]
- 359.Harten RD, Svach DJ, Schmeltzer R, Uhrich KE. Journal of Biomedical Materials Research Part A. 2005;72A:354–362. doi: 10.1002/jbm.a.30184. [DOI] [PubMed] [Google Scholar]
- 360.Bryers JD, Jarvis RA, Lebo J, Prudencio A, Kyriakides TR, Uhrich K. Biomaterials. 2006;27:5039–5048. doi: 10.1016/j.biomaterials.2006.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Rosenberg LE, Carbone AL, Romling U, Uhrich KE, Chikindas ML. Letters in Applied Microbiology. 2008;46:593–599. doi: 10.1111/j.1472-765X.2008.02356.x. [DOI] [PubMed] [Google Scholar]
- 362.Henry JA, Simonet M, Pandit A, Neuenschwander P. Journal of Biomedical Materials Research Part A. 2007;82A:669–679. doi: 10.1002/jbm.a.31094. [DOI] [PubMed] [Google Scholar]
- 363.Zhang JY, Beckman EJ, Piesco NP, Agarwal S. Biomaterials. 2000;21:1247–1258. doi: 10.1016/s0142-9612(00)00005-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Adhikari R, Gunatillake PA, Griffiths I, Tatai L, Wichramaratna M, Houshyar S, Moore T, Mayadunne RTM, Field J, McGee M, Carbone T. Biomaterials. 2008;29:3762–3770. doi: 10.1016/j.biomaterials.2008.06.021. [DOI] [PubMed] [Google Scholar]
- 365.Werkmeister JA, Adhikari R, White JF, Tebb TA, Le TPT, Taing HC, Mayadunne R, Gunatillake PA, Danon SJ, Ramshaw JAM. Acta Biomaterialia. 2010;6:3471–3481. doi: 10.1016/j.actbio.2010.02.040. [DOI] [PubMed] [Google Scholar]
- 366.Yang L, Korom S, Welti M, Hoerstrup SP, Zund G, Jung FJ, Neuenschwander P, Weder W. European Journal of Cardio-Thoracic Surgery. 2003;24:201–207. doi: 10.1016/s1010-7940(03)00263-x. [DOI] [PubMed] [Google Scholar]
- 367.Brizzola S, Eguileor Md, Brevini T, Grimaldi A, Congiu T, Neuenschwander P, Acocella F. Interactive Cardiovascular and Thoracic Surgery. 2009;8:610–614. doi: 10.1510/icvts.2008.197012. [DOI] [PubMed] [Google Scholar]
- 368.Riboldi SA, Sadr N, Pigini L, Neuenschwander P, Simonet M, Mognol P, Sampaolesi M, Cossu G, Mantero S. Journal of Biomedical Materials Research Part A. 2008;84A:1094–1101. doi: 10.1002/jbm.a.31534. [DOI] [PubMed] [Google Scholar]
- 369.Kaetsu I, Yoshida M, Yamada A. Journal of Biomedical Materials Research. 1980;14:185–197. doi: 10.1002/jbm.820140302. [DOI] [PubMed] [Google Scholar]
- 370.Park JH, Bae YH. Journal of Biomaterials Science. Polymer Edition. 2002;13:527–542. doi: 10.1163/15685620260178382. [DOI] [PubMed] [Google Scholar]
- 371.Park JH, Bae YH. Journal of Biomedical Materials Research Part A. 2003;64A:309–319. doi: 10.1002/jbm.a.10424. [DOI] [PubMed] [Google Scholar]
- 372.Hsu S-H, Tseng H-J. Journal of Biomaterials Applications. 2004;19:135–146. doi: 10.1177/0885328204044160. [DOI] [PubMed] [Google Scholar]
- 373.Pomel C, Leborgne C, Cheradame H, Scherman D, Kichler A, Guegan P. Pharmaceutical Research. 2008;25:2963–2971. doi: 10.1007/s11095-008-9698-9. [DOI] [PubMed] [Google Scholar]
- 374.Frings J, Schramm E, Schink B. American Society for Microbiology. 1992;58:2164–2167. doi: 10.1128/aem.58.7.2164-2167.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Ohta T, Tani A, Kimbara K, Kawai F. Applied Microbiology and Biotechnology. 2005;68:639–646. doi: 10.1007/s00253-005-1936-z. [DOI] [PubMed] [Google Scholar]
- 376.Biondi O, Motta S, Mosesso P. Mutagenesis. 2002;17:261–264. doi: 10.1093/mutage/17.3.261. [DOI] [PubMed] [Google Scholar]
- 377.Kabanov AV, Nazarova IR, Astafieva I, Batrakova EV, Alakhov VY, Yaroslavov AA, Kabanov VA. Macromolecules. 1995;28:2303–2314. [Google Scholar]
- 378.Kozlov MY, Melik-Nubarov NS, Batrakova EV, Kabanov AV. Macromolecules. 2000;33:3305–3313. [Google Scholar]
- 379.Sezgin Z, Yuksel N, Baykara T. European Journal of Pharmaceutics and Biopharmaceutics. 2006;64:261–268. doi: 10.1016/j.ejpb.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 380.Kwon SH, Kim SY, Ha HW, Kang MJ, Huh JS, Jong IT, Kim YM, Park YM, Kang HH, Lee S, Chang JY, Lee J, Choi YW. Archives of Pharmacal Research. 2007;30:1138–1143. doi: 10.1007/BF02980249. [DOI] [PubMed] [Google Scholar]
- 381.Zhang W, Shi Y, Chen Y, Yu S, Hao J, Luo J, Sha X, Fang X. European Journal of Pharmaceutics and Biopharmaceutics. 2010;75:341–353. doi: 10.1016/j.ejpb.2010.04.017. [DOI] [PubMed] [Google Scholar]
- 382.Chen F, Liu X-M, Rice KC, Li X, Yu F, Reinhardt RA, Bayles KW, Wang D. Antimicrobial agents and chemotherapy. 2009;53:4898–4902. doi: 10.1128/AAC.00387-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Chen F, Rice KC, Liu X-M, Reinhardt RA, Bayles KW, Wang D. Pharmaceutical Research. 2010;27:2356–2364. doi: 10.1007/s11095-010-0119-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Kadam Y, Yerramilli U, Bahadur A, Bahadur P. Colloids and Surfaces B: Biointerfaces. 2011;83:49–57. doi: 10.1016/j.colsurfb.2010.10.041. [DOI] [PubMed] [Google Scholar]
- 385.Foster B, Cosgrove T, Hammouda B. Langmuir. 2009;25:6760–6766. doi: 10.1021/la900298m. [DOI] [PubMed] [Google Scholar]
- 386.Roques C, Bouchemal K, Ponchel G, Fromes Y, Fattal E. Journal of Controlled Release. 2009;138:71–77. doi: 10.1016/j.jconrel.2009.04.030. [DOI] [PubMed] [Google Scholar]
- 387.Jones DS, Bruschi ML, Freitas Od, Fremiao MPD, Lara EHG, Andrews GP. International Journal of Pharmaceutics. 2009;372:49–58. doi: 10.1016/j.ijpharm.2009.01.006. [DOI] [PubMed] [Google Scholar]
- 388.Liu Y, Lu W-L, Wang J-C, Zhang X, Zhang H, Wang X-Q, Zhou T-Y, Zhang Q. Journal of Controlled Release. 2007;117:387–395. doi: 10.1016/j.jconrel.2006.11.024. [DOI] [PubMed] [Google Scholar]
- 389.Derakhshandeh K, Fashi M, Seifoleslami S. Drug Design, Development and Therapy. 2010;24:255–262. doi: 10.2147/dddt.s13289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Lee JW, Lim T-H, Park JB. Journal of Biomedical Materials Research Part A. 2010;92A:378–385. doi: 10.1002/jbm.a.32377. [DOI] [PubMed] [Google Scholar]
- 391.Aka-Any-Grah A, Bouchemal K, Koffi A, Agnely F, Zhang M, Djabourov M, Ponchel G. European Journal of Pharmaceutics and Biopharmaceutics. 2010;76:296–303. doi: 10.1016/j.ejpb.2010.07.004. [DOI] [PubMed] [Google Scholar]
- 392.Cortiella J, Nichols JE, Kojima K, Bonassar LJ, Dargon P, Roy AK, Vacant MP, Niles JA, Vacanti CA. Tissue Engineering. 2006;12:1213–1225. doi: 10.1089/ten.2006.12.1213. [DOI] [PubMed] [Google Scholar]
- 393.Vashi AV, Keramidaris E, Abberton KM, Morrison WA, Wilson JL, O’Connor AJ, Cooper-White JJ, Thompson EW. Biomaterials. 2008;29:573–579. doi: 10.1016/j.biomaterials.2007.10.017. [DOI] [PubMed] [Google Scholar]
- 394.Beamish JA, Zhu J, Kottke-Marchant K, Marchant RE. Journal of Biomedical Materials Research Part A. 2010;92A:441–450. doi: 10.1002/jbm.a.32353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Pfister PM, Wendlandt M, Neuenschwander P, Suter UW. Biomaterials. 2007;28:567–575. doi: 10.1016/j.biomaterials.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 396.Ainslie KM, Kraning CM, Desai TA. Lab on a Chip. 2008;8:1042–1047. doi: 10.1039/b800604k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.DiRamio JA, Kisaalita WS, Majetich GF, Shimkus JM. Biotechnology Progress. 2005;21:1281–1288. doi: 10.1021/bp0495670. [DOI] [PubMed] [Google Scholar]
- 398.Fu Y, Kao WJ. Pharmaceutical Research. 2009;26:2115–2124. doi: 10.1007/s11095-009-9923-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Woldum HS, Larsen KL, Madsen F. Drug Delivery. 2008;15:69–80. doi: 10.1080/10717540701829267. [DOI] [PubMed] [Google Scholar]
- 400.Hwang NS, Varghese S, Elisseeff J. Methods in Molecular Biology. 2007;407:351–373. doi: 10.1007/978-1-59745-536-7_24. [DOI] [PubMed] [Google Scholar]
- 401.Buxton AN, Zhu J, Marchant R, West JL, Yoo JU, Johnstone B. Tissue Engineering. 2007;13:2549–2560. doi: 10.1089/ten.2007.0075. [DOI] [PubMed] [Google Scholar]
- 402.Nuttelman CR, Benoit DSW, Tripod MC, Anseth KS. Biomaterials. 2006;27:1377–1386. doi: 10.1016/j.biomaterials.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 403.Zhou Z, Ren Y, Yang D, Nie J. Biomedical Materials. 2009;4:035007. doi: 10.1088/1748-6041/4/3/035007. [DOI] [PubMed] [Google Scholar]
- 404.Betz MW, Yeatts AB, Richbourg WJ, Caccamese JF, Coletti DP, Falco EE, Fisher JP. Biomacromolecules. 2010;11:1160–1168. doi: 10.1021/bm100061z. [DOI] [PubMed] [Google Scholar]
- 405.Moon JJ, Hahn MS, Kim I, Nsiah BA, West JL. Tissue Engineering Part A. 2009;15:579–585. doi: 10.1089/ten.tea.2008.0196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Jimenez-Vergara AC, Guiza-Arguello V, Becerra-Bayona S, Munoz-Pinto DJ, McMahon RE, Morales A, Cubero-Ponce L, Hahn MS. Annals of Biomedical Engineering. 2010;38:2885–2895. doi: 10.1007/s10439-010-0049-8. [DOI] [PubMed] [Google Scholar]
- 407.Lacasse FX, Filion MC, Phillips NC, Escher E, McMullen JN, Hildgen P. Pharmaceutical Research. 1998;15:312–317. doi: 10.1023/a:1011935222652. [DOI] [PubMed] [Google Scholar]
- 408.Romberg B, Hennink WE, Storm G. Pharmaceutical Research. 2008;25:55–71. doi: 10.1007/s11095-007-9348-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Zhang X, He H, Yen C, Ho W, Lee LJ. Biomaterials. 2008;29:4253–4259. doi: 10.1016/j.biomaterials.2008.07.032. [DOI] [PubMed] [Google Scholar]
- 410.Shan X, Yuan Y, Liu C, Tao X, Sheng Y, Xu F. Biomedical Microdevices. 2009;11:1187–1194. doi: 10.1007/s10544-009-9336-2. [DOI] [PubMed] [Google Scholar]
- 411.Yoncheva K, Guembe L, Campanero MA, Irache JM. International Journal of Pharmaceutics. 2007;334:156–165. doi: 10.1016/j.ijpharm.2006.10.016. [DOI] [PubMed] [Google Scholar]
- 412.Hou S, McCauley LK, Ma PX. Macromolecular Bioscience. 2007;7:620–628. doi: 10.1002/mabi.200600256. [DOI] [PubMed] [Google Scholar]
- 413.Yoncheva K, Centelles MN, Irache JM. Journal of Microencapsulation. 2008;25:82–89. doi: 10.1080/02652040701776083. [DOI] [PubMed] [Google Scholar]
- 414.Tang BC, Fu J, Watkins DN, Hanes J. Biomaterials. 2010;31:339–344. doi: 10.1016/j.biomaterials.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Schacht E, Toncheva V, Vandertaelen K, Heller J. Journal of Controlled Release. 2006;116:219–225. doi: 10.1016/j.jconrel.2006.07.026. [DOI] [PubMed] [Google Scholar]
- 416.Gao X, Tao W, Lu W, Zhang Q, Zhang Y, Jiang X, Fu S. Biomaterials. 2006;27:3482–3490. doi: 10.1016/j.biomaterials.2006.01.038. [DOI] [PubMed] [Google Scholar]
- 417.Lee W-C, Li Y-C, Chu I-M. Macromolecular Bioscience. 2006;6:846–854. doi: 10.1002/mabi.200600101. [DOI] [PubMed] [Google Scholar]
- 418.Xu X, Chen X, Wang Z, Jing X. European Journal of Pharmaceutics and Biopharmaceutics. 2009;72:18–25. doi: 10.1016/j.ejpb.2008.10.015. [DOI] [PubMed] [Google Scholar]
- 419.Zhang Y, Wu X, Han Y, Mo F, Duran Y, Li S. International Journal of Pharmaceutics. 2010;386:15–22. doi: 10.1016/j.ijpharm.2009.10.045. [DOI] [PubMed] [Google Scholar]
- 420.Cheng J, Teply BA, Sherifi I, Sung J, Luther G, Gu FX, Levy-Nissenbaum E, Radovic-Moreno AF, Langer R, Farokhzad OC. Biomaterials. 2007;28:869–876. doi: 10.1016/j.biomaterials.2006.09.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Cu Y, Saltzman WM. Molecular Pharmaceuticals. 2009;6:173–181. doi: 10.1021/mp8001254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Gao Y, Sun Y, Ren F, Gao S. Drug Development and Industrial Pharmacy. 2010;36:1131–1138. doi: 10.3109/03639041003680826. [DOI] [PubMed] [Google Scholar]
- 423.Song Z, Feng R, Sun M, Guo C, Gao Y, Li L, Zhai G. Journal of Colloid and Interface Science. 2011;354:116–123. doi: 10.1016/j.jcis.2010.10.024. [DOI] [PubMed] [Google Scholar]
- 424.Liu J, Zeng F, Allen C. European Journal of Pharmaceutics and Biopharmaceutics. 2007;65:309–319. doi: 10.1016/j.ejpb.2006.11.010. [DOI] [PubMed] [Google Scholar]
- 425.Yanez JA, Forrest ML, Ohgami Y, Kwon GS, Davies NM. Cancer Chemotherapy and Pharmacology. 2008;61:133–144. doi: 10.1007/s00280-007-0458-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Forrest ML, Yanez JA, Remsberg CM, Ohgami Y, Kwon GS, Davies NM. Pharmaceutical Research. 2008;25:194–206. doi: 10.1007/s11095-007-9451-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Feng M, Cai Q, Shi X, Huang H, Zhou P, Guo X. Journal of Drug Targeting. 2008;16:502–508. doi: 10.1080/10611860802200938 . [DOI] [PubMed] [Google Scholar]
- 428.Wang Y-Y, Lai SK, Suk JS, Pace A, Cone R, Hanes J. Angewandte Chemie International Edition. 2008;47:9726–9729. doi: 10.1002/anie.200803526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Tang BC, Dawson M, Lai SK, Wang Y-Y, Suk JS, Yang M, Zetilin P, Boyle MP, Fu J, Hanes J. Proceedings of the National Academy of Sciences of the United States of America. 2009;106:19268–19273. doi: 10.1073/pnas.0905998106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Suk JS, Lai SK, Wang Y-Y, Ensign LM, Zeitlin PL, Boyle MP, Hanes J. Biomaterials. 2009;30:2591–2597. doi: 10.1016/j.biomaterials.2008.12.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Whaley KJ, Hanes J, Shattock R, Cone RA, Friend DR. Antiviral Research. 2010;88:S55–S66. doi: 10.1016/j.antiviral.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 432.Benoit DSW, Durney AR, Anseth KS. Tissue Engineering. 2006;12:1663–1673. doi: 10.1089/ten.2006.12.1663. [DOI] [PubMed] [Google Scholar]
- 433.Freeman JW, Woods MD, Cromer DA, Ekwueme EC, Atiemo TAEA, Bijoux CH, Laurencin CT. Journal of Biomechanics. 2010 doi: 10.1016/j.jbiomech.2010.10.043. [DOI] [PubMed] [Google Scholar]
- 434.Papadopoulos A, Bichara DA, Zhao X, Ibusuki S, Randolph MA, Anseth KS, Yaremchuk MJ. Tissue Engineering Part A. 2011;17:161–169. doi: 10.1089/ten.tea.2010.0253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Kim J, Lee K-W, Hefferan TE, Currier BL, Yaszemski MJ, Lu L. Biomacromolecules. 2008;9:149–157. doi: 10.1021/bm700924n. [DOI] [PubMed] [Google Scholar]
- 436.Poon YF, Cao Y, Zhu Y, Judeh ZMA, Chan-Park MB. Biomacromolecules. 2009;10:2043–2052. doi: 10.1021/bm801367n. [DOI] [PubMed] [Google Scholar]
- 437.Zhang X, Yang D, Nie J. Internation Journal of Biological Macromolecules. 2008;43:456–462. doi: 10.1016/j.ijbiomac.2008.08.010. [DOI] [PubMed] [Google Scholar]
- 438.Ma G, Zhang X, Han J, Song G, Nie J. International Journal of Biological Macromolecules. 2009;45:499–503. doi: 10.1016/j.ijbiomac.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 439.Ramshaw JAM, Shah NK, Brodsky B. Journal of Structural Biology. 1998;122:86–91. doi: 10.1006/jsbi.1998.3977. [DOI] [PubMed] [Google Scholar]
- 440.Przybyla DE, Chmielewski J. Biochemistry. 2010;49:4411–4419. doi: 10.1021/bi902129p. [DOI] [PubMed] [Google Scholar]
- 441.Krane SM. Amino Acids. 2008;35:703–710. doi: 10.1007/s00726-008-0073-2. [DOI] [PubMed] [Google Scholar]
- 442.Matsuno T, Nakamura T, Kuremoto K-i, Notazawa S, Nakahara T, Hashimoto Y, Satoh T, Shimizu Y. Dental Materials Journal. 2006;25:138–144. doi: 10.4012/dmj.25.138. [DOI] [PubMed] [Google Scholar]
- 443.Oliveira SM, Amaral IF, Barbosa MA, Teixeira CC. Tissue Engineering Part A. 2008;15:625–634. doi: 10.1089/ten.tea.2008.0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 444.Fukushima K, Enomoto M, Tomizawa S, Takahashi M, Wakabayashi Y, Itoh S, Kuboki Y, Shinomiya K. Journal of Medical and Dental Sciences. 2008;55:71–79. [PubMed] [Google Scholar]
- 445.Bushnell BD, McWilliams AD, Whitener GB, Messer T. The Journal of Hand Surgery. 2008;33:1081–1087. doi: 10.1016/j.jhsa.2008.03.015. [DOI] [PubMed] [Google Scholar]
- 446.Soltysiak P, Hollwarth ME, Saxena AK. Bio-Medical Materials and Engineering. 2010;20:1–11. doi: 10.3233/BME-2010-0610. [DOI] [PubMed] [Google Scholar]
- 447.Komura M, Komura H, Kanamori Y, Tanaka Y, Suzuki K, Sugiyama M, Nakahara S, Kawashima H, Hatanaka A, Hoshi K, Ikada Y, Tabata Y, Iwanaka T. Journal of Pediatric Surgery. 2008;43:2141–2146. doi: 10.1016/j.jpedsurg.2008.08.038. [DOI] [PubMed] [Google Scholar]
- 448.Choi JS, Yang H-J, Kim BS, Kim JD, Kim JY, Yoo B, Park K, Lee HY, Cho YW. Journal of Controlled Release. 2009;139:2–7. doi: 10.1016/j.jconrel.2009.05.034. [DOI] [PubMed] [Google Scholar]
- 449.Huang Z, Tian J, Yu B, Xu Y, Feng Q. Biomedical Materials. 2009;4:055005. doi: 10.1088/1748-6041/4/5/055005. [DOI] [PubMed] [Google Scholar]
- 450.Sulica L, Rosen CA, Postma GN, Simpson B, Amin M, Courey M, Merati A. The Laryngoscope. 2010;120:319–325. doi: 10.1002/lary.20737. [DOI] [PubMed] [Google Scholar]
- 451.Wang F, Li Z, Khan M, Tamara K, Kuppusamy P, Wagner WR, Sen CK, Guan J. Acta Biomaterialia. 2010;6:1978–1991. doi: 10.1016/j.actbio.2009.12.011. [DOI] [PubMed] [Google Scholar]
- 452.Liu X, Wang X-M, Chen Z, Cui F-Z, Liu H-Y, Mao K, Wang Y. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2010;94B:72–79. doi: 10.1002/jbm.b.31625. [DOI] [PubMed] [Google Scholar]
- 453.Lamphongsai S, Eshraghi Y, Totonchi A, Midler J, Abdul-Karim FW, Guyuron B. Aesthetic Surgery Journal. 2010;29:93–97. doi: 10.1016/j.asj.2009.01.016. [DOI] [PubMed] [Google Scholar]
- 454.Yilmaz N, Inal S, Muglali M, Guvenc T, Bas B. Medicina Oral, Patologia Oral y Cirugia Bucal. 2010;15:e526–530. [PubMed] [Google Scholar]
- 455.Kleinmann G, Larson S, Hunter B, Stevens S, Mamalis N, Olson RJ. Ophthalmologica. 2007;221:51–56. doi: 10.1159/000096523. [DOI] [PubMed] [Google Scholar]
- 456.Friberg O, Dahlin L-G, Kallman J, Kihlstrom E, Soderquist B, Svedjeholm R. Interactive Cardiovascular and Thoracic Surgery. 2009;9:454–458. doi: 10.1510/icvts.2009.207514. [DOI] [PubMed] [Google Scholar]
- 457.Holladay C, Keeney M, Greiser U, Murphy M, O’Brein T, Pandit A. Journal of Controlled Release. 2009;136:220–225. doi: 10.1016/j.jconrel.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 458.Peng L, Cheng X, Zhuo R, Lan J, Wang Y, Shi B, Li S. Journal of Biomedical Materials Research Part A. 2009;90A:564–576. doi: 10.1002/jbm.a.32117. [DOI] [PubMed] [Google Scholar]
- 459.Krebs MD, Jeon O, Alsberg E. Journal of the American Chemical Society. 2009;131:9204–9206. doi: 10.1021/ja9037615. [DOI] [PubMed] [Google Scholar]
- 460.Castells RV, Holladay C, Luca Ad, Diaz VM, Pandit A. Bioconjugate Chemistry. 2009;20:2262–2269. doi: 10.1021/bc900241w. [DOI] [PubMed] [Google Scholar]
- 461.Wang L, Cao J, Lei D-l, Cheng X-b, Yang Y-w, Hou R, Zhao Y-h, Cui F-z. Journal of Craniofacial Surgery. 2009;20:2188–2192. doi: 10.1097/SCS.0b013e3181bf04c7. [DOI] [PubMed] [Google Scholar]
- 462.Maehara H, Sotome S, Yoshii T, Torigoe I, Kawasaki Y, Sugata Y, Yuasa M, Hirano M, Mochizuki N, Kikuchi M, Shinomiya K, Okawa A. Journal of Orthopaedic Research. 2010;28:677–686. doi: 10.1002/jor.21032. [DOI] [PubMed] [Google Scholar]
- 463.Solorio L, Zwolinski C, Lund AW, Farrell MJ, Stegemann JP. Journal of Tissue Engineering and Regenerative Medicine. 2010;4:514–523. doi: 10.1002/term.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Baik SH, Kim JH, Cho HH, Park S-N, Kim YS, Suh H. Journal of Surgical Research. 2010;164:e221–e228. doi: 10.1016/j.jss.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 465.Bellucci RJ, Wolff D. Laryngoscope. 1964;74:668–688. doi: 10.1288/00005537-196405000-00005. [DOI] [PubMed] [Google Scholar]
- 466.Sionkowska A, Skopinska-Wisniewska J, Gawron M, Kozlowska J, Planecka A. International Journal of Biological Macromolecules. 2010;47:570–577. doi: 10.1016/j.ijbiomac.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 467.Kakudo N, Shimotsuma A, Miyake S, Kushida S, Kusumoto K. Journal of Biomedical Materials Research Part A. 2008;84:191–197. doi: 10.1002/jbm.a.31311. [DOI] [PubMed] [Google Scholar]
- 468.Dong S-W, Ying D-J, Duan X-J, Xie Z, Yu Z-J, Zhu C-H, Yang B, Sun J-S. Bioscience, Biotechnology, and Biochemistry. 2009;73:2226–2233. doi: 10.1271/bbb.90329. [DOI] [PubMed] [Google Scholar]
- 469.Hesse E, Hefferan TE, Tarara JE, Haasper C, Meller R, Krettek C, Lu L, Yaszemski MJ. Journal of Biomedical Materials Research Part A. 2010;94:442–449. doi: 10.1002/jbm.a.32696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 470.Sachlos E, Gotora D, Czernuszka JT. Tissue Engineering. 2006;12:2479–2487. doi: 10.1089/ten.2006.12.2479. [DOI] [PubMed] [Google Scholar]
- 471.Venugopal J, Low S, Choon AT, Kumar TSS, Ramakrishna S. Journal of Materials Science: Materials in Medicine. 2008;19:2039–2046. doi: 10.1007/s10856-007-3289-x. [DOI] [PubMed] [Google Scholar]
- 472.Gleeson JP, Plunkett NA, O’Brien FJ. European Cells & Materials. 2010;20:218–230. doi: 10.22203/ecm.v020a18. [DOI] [PubMed] [Google Scholar]
- 473.Grimes JS, Bocklage TJ, Pitcher JD. Orthopedics. 2006;29:145–148. doi: 10.3928/01477447-20060201-03. [DOI] [PubMed] [Google Scholar]
- 474.Minura T, Imai S, Kubo M, Isoya E, Ando K, Okumura N, Matsusue Y. Osteoarthritis and Cartilage. 2008;16:1083–1091. doi: 10.1016/j.joca.2008.02.003. [DOI] [PubMed] [Google Scholar]
- 475.Pulkkinen HJ, Tiitu V, Valonen P, Jurvelin JS, Lammi MJ, Kiviranta I. Osteoarthritis and Cartilage. 2010;18:1077–1087. doi: 10.1016/j.joca.2010.05.004. [DOI] [PubMed] [Google Scholar]
- 476.Mueller-Rath R, Gavenis K, Andereya S, Mumme T, Albrand M, Stoffel M, Weichert D, Schneider U. Bio-Medical Materials and Engineering. 2010;20:317–328. doi: 10.3233/BME-2010-0645. [DOI] [PubMed] [Google Scholar]
- 477.Kuo CK, Tuan RS. Tissue Engineering Part A. 2008;14:1615–1627. doi: 10.1089/ten.tea.2006.0415. [DOI] [PubMed] [Google Scholar]
- 478.Butler DL, Gooch C, Kinneberg KR, Boivin GP, Galloway MT, Nirmalanandhan VS, Shearn JT, Dyment NA, Juncosa-Melvin N. Nature Protocols. 2010;5:849–863. doi: 10.1038/nprot.2010.14. [DOI] [PubMed] [Google Scholar]
- 479.Gurkan UA, Cheng X, Kishore V, Uquillas JA, Akkus O. Journal of Biomedical Materials Research Part A. 2010;94A:1070–1079. doi: 10.1002/jbm.a.32783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Zeugolis DI, Paul GR, Attenburrow G. Journal of Biomedical Materials Research Part A. 2009;89A:895–908. doi: 10.1002/jbm.a.32031. [DOI] [PubMed] [Google Scholar]
- 481.Robayo LM, Moulin VJ, Tremblay P, Cloutier R, Lamontagne J, Larkin A-M, Chabaud S, Simon F, Islam N, Goulet F. Wound Repair and Regeneration. 2011;19:38–48. doi: 10.1111/j.1524-475X.2010.00640.x. [DOI] [PubMed] [Google Scholar]
- 482.George J, Onodera J, Miyata T. Journal of Biomedical Materials Research Part A. 2008;87A:1103–1111. doi: 10.1002/jbm.a.32277. [DOI] [PubMed] [Google Scholar]
- 483.Helary C, Bataille I, Abed A, Illoul C, Anglo A, Louedec L, Letourneur D, Meddahi-Pelle A, Giraud-Guille MM. Biomaterials. 2010;31:481–490. doi: 10.1016/j.biomaterials.2009.09.073. [DOI] [PubMed] [Google Scholar]
- 484.Reckenrich AK, Hopfner U, Krotz F, Zhang Z, Koch C, Kremer M, Machens H-G, Plank C, Egana JT. Biomaterials. 2011;32:1996–2003. doi: 10.1016/j.biomaterials.2010.11.022. [DOI] [PubMed] [Google Scholar]
- 485.Huang S, Xu Y, Wu C, Sha D, Fu X. Biomaterials. 2010;31:5520–5525. doi: 10.1016/j.biomaterials.2010.03.060. [DOI] [PubMed] [Google Scholar]
- 486.Ahn S, Yoon H, Kim G, Kim Y, Lee S, Chun W. Tissue Engineering Part C: Methods. 2010;16:813–820. doi: 10.1089/ten.tec.2009.0511. [DOI] [PubMed] [Google Scholar]
- 487.Bastiaansen-Jenniskens YM, Koevoet W, Bart ACWd, Linden JCvd, Zuurmond AM, Weinans H, Verhaar JAN, Osch GJVMv, DeGroot J. Osteoarthritis and Cartilage. 2008;16:359–366. doi: 10.1016/j.joca.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 488.Lu X, Zhai W, Zhou Y, Zhang YZH, Chang J. Journal of Materials Science: Materials in Medicine. 2010;21:473–480. doi: 10.1007/s10856-009-3924-9. [DOI] [PubMed] [Google Scholar]
- 489.Yan L-P, Wang Y-J, Ren L, Wu G, Caridade SG, Fan J-B, Wang L-Y, Ji P-H, Oliveira JM, Oliveria JT, Mano JF, Reis RL. Journal of Biomedical Materials Research Part A. 2010;95A:465–475. doi: 10.1002/jbm.a.32869. [DOI] [PubMed] [Google Scholar]
- 490.Casper CL, Yang W, Farach-Carson MC, Rabolt JF. Biomacromolecules. 2007;8:1116–1123. doi: 10.1021/bm061003s. [DOI] [PubMed] [Google Scholar]
- 491.Sahoo S, Ang L-T, Goh JC-H, Toh S-L. Differentiation. 2010;79:102–110. doi: 10.1016/j.diff.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 492.Spurlin TA, Bhadriraju K, Chung K-H, Tona A, Plant AL. Biomaterials. 2009;30:5486–5496. doi: 10.1016/j.biomaterials.2009.07.014. [DOI] [PubMed] [Google Scholar]
- 493.Torres-Giner S, Gimeno-Alcaniz JV, Ocio MJ, Lagaron JM. ACS Applied Materials & Interfaces. 2009;1:218–223. doi: 10.1021/am800063x. [DOI] [PubMed] [Google Scholar]
- 494.Ciardelli G, Gentile P, Chiono V, Mattioli-Belmonte M, Vozzi G, Barbani N, Giusti P. Journal of Biomedical Materials Research Part A. 2010;92A:137–151. doi: 10.1002/jbm.a.32344. [DOI] [PubMed] [Google Scholar]
- 495.Zhang C, Hu Y-Y, Cui F-Z, Zhang S-M, Ruan D-K. Biomedical Materials. 2006;1:56–62. doi: 10.1088/1748-6041/1/2/002. [DOI] [PubMed] [Google Scholar]
- 496.Liu X, Huang C, Feng Y, Liang J, Fan Y, Gu Z, Zhang X. Journal of Biomaterials Science. Polymer Edition. 2010;21:963–977. doi: 10.1163/156856209X461034. [DOI] [PubMed] [Google Scholar]
- 497.Sahoo S, Cho-Hong JG, Siew-Lok T. Biomedical Materials. 2007;2:169–173. doi: 10.1088/1748-6041/2/3/001. [DOI] [PubMed] [Google Scholar]
- 498.Tatekawa Y, Kawazoe N, Chen G, Shirasaki Y, Komuro H, Kaneko M. Pediatric Surgery International. 2010;26:575–580. doi: 10.1007/s00383-010-2609-2. [DOI] [PubMed] [Google Scholar]
- 499.Ladd MR, Lee SJ, Stitzel JD, Atala A, Yoo JJ. Biomaterials. 2011;32:1549–1559. doi: 10.1016/j.biomaterials.2010.10.038. [DOI] [PubMed] [Google Scholar]
- 500.Chen K-Y, Liao W-J, Kuo S-M, Tsai F-J, Chen Y-S, Huang C-Y, Yao C-H. Biomacromolecules. 2009;10:1642–1649. doi: 10.1021/bm900238b. [DOI] [PubMed] [Google Scholar]
- 501.Sun LP, Wang S, Zhang ZW, Wang XY, Zhang QQ. Biomedical Materials. 2009;4:055008. doi: 10.1088/1748-6041/4/5/055008. [DOI] [PubMed] [Google Scholar]
- 502.Wang L, Stegemann JP. Biomaterials. 2010;31:3976–3985. doi: 10.1016/j.biomaterials.2010.01.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 503.Ye Q, Harmsen MC, Luyn MJAv, Bank RA. Biomaterials. 2010;31:9192–9201. doi: 10.1016/j.biomaterials.2010.08.049. [DOI] [PubMed] [Google Scholar]
- 504.Xu C, Yu Z, Inouye M, Brodsky B, Mirochnitchenko O. Biomacromolecules. 2010;11:348–356. doi: 10.1021/bm900894b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 505.Kothapalli CR, Ramamurthi A. Acta Biomaterialia. 2010;6:170–178. doi: 10.1016/j.actbio.2009.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 506.Kurane A, Simionescu DT, Vyavahare NR. Biomaterials. 2007;28:2830–2838. doi: 10.1016/j.biomaterials.2007.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 507.Kurane A, Vyavahare N. Journal of Tissue Engineering and Regenerative Medicine. 2009;3:280–289. doi: 10.1002/term.164. [DOI] [PubMed] [Google Scholar]
- 508.Mithieux SM, Rasko JEJ, Weiss AS. Biomaterials. 2004;25:4921–4927. doi: 10.1016/j.biomaterials.2004.01.055. [DOI] [PubMed] [Google Scholar]
- 509.Nivison-Smith L, Rnjak J, Weiss AS. Acta Biomaterialia. 2010;6:354–359. doi: 10.1016/j.actbio.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 510.Tu Y, Wise SG, Weiss AS. Micron. 2010;41:268–272. doi: 10.1016/j.micron.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 511.Kaufmann D, Weberskirch R. Macromolecular Bioscience. 2006;6:952–958. doi: 10.1002/mabi.200600117. [DOI] [PubMed] [Google Scholar]
- 512.Liu Y, Jia Z, Li L, Chen F. Medical Hypotheses. 2011;76:239–240. doi: 10.1016/j.mehy.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 513.Floss DM, Schallau K, Rose-John S, Conrad U, Scheller J. Trends in Biotechnology. 2010;28:37–45. doi: 10.1016/j.tibtech.2009.10.004. [DOI] [PubMed] [Google Scholar]
- 514.MacEwan SR, Chilkoti A. Peptide Science. 2010;94:60–77. doi: 10.1002/bip.21327. [DOI] [PubMed] [Google Scholar]
- 515.Chu H-S, Park J-E, Kim D-M, Kim B-G, Won J-I. Protein Expression and Purification. 2010;74:298–303. doi: 10.1016/j.pep.2010.07.006. [DOI] [PubMed] [Google Scholar]
- 516.Massodi I, Thomas E, Raucher D. Molecules. 2009;14:1999–2015. doi: 10.3390/molecules14061999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 517.Bidwell GL, Fokt I, Priebe W, Raucher D. Biochemical Pharmacology. 2007;73:620–631. doi: 10.1016/j.bcp.2006.10.028. [DOI] [PubMed] [Google Scholar]
- 518.Adams SB, Shamji MF, Nettles DL, Hwang P, Setton LA. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2009;90B:67–74. doi: 10.1002/jbm.b.31254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Betre H, Liu W, Zalutsky MR, Chilkoti A, Kraus VB, Setton LA. Journal of Controlled Release. 2006;115:175–182. doi: 10.1016/j.jconrel.2006.07.022. [DOI] [PubMed] [Google Scholar]
- 520.Bessa PC, Machado R, Nurnberger S, Dopler D, Banerjee A, Cunha AM, Rodriquez-Cabello JC, Redl H, Griensven Mv, Reis RL, Casal M. Journal of Controlled Release. 2010;142:312–318. doi: 10.1016/j.jconrel.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 521.Betre H, Ong SR, Guilak F, Chilkoti A, Fermor B, Setton LA. Biomaterials. 2006;27:91–99. doi: 10.1016/j.biomaterials.2005.05.071. [DOI] [PubMed] [Google Scholar]
- 522.Lim DW, Nettles DL, Setton LA, Chilkoti A. Biomacromolecules. 2008;9:222–230. doi: 10.1021/bm7007982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Srokowski EM, Woodhouse KA. Journal of Biomaterials Science, Polymer Edition. 2008;19:785–799. doi: 10.1163/156856208784522038. [DOI] [PubMed] [Google Scholar]
- 524.Nettles DL, Chilkoti A, Setton LA. Advanced Drug Delivery Reviews. 2010;62:1479–1485. doi: 10.1016/j.addr.2010.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 525.Prinsen BHCMT, Velden MGMdS-vd. Clinica Chimica Acta. 2004;347:1–14. doi: 10.1016/j.cccn.2004.04.005. [DOI] [PubMed] [Google Scholar]
- 526.Dror Y, Ziv T, Makarov V, Wolf H, Admon A, Zussman E. Biomacromolecules. 2008;9:2749–2754. doi: 10.1021/bm8005243. [DOI] [PubMed] [Google Scholar]
- 527.Regev O, Khalgin R, Zussman E, Cohen Y. International Journal of Biological Macromolecules. 2010;47:261–265. doi: 10.1016/j.ijbiomac.2010.04.012. [DOI] [PubMed] [Google Scholar]
- 528.Gayakwad SG, Bejugam NK, Akhavein N, Uddin NA, Oettinger CE, D’Souza MJ. Journal of Microencapsulation. 2009;26:692–700. doi: 10.3109/02652040802666910. [DOI] [PubMed] [Google Scholar]
- 529.Okoroukwu ON, Green GR, D’Souza MJ. Journalof Microencapsulation. 2010;27:142–149. doi: 10.3109/02652040903052028. [DOI] [PubMed] [Google Scholar]
- 530.Kapoor DN, Manvi FV, Doijad RC, Dhawan S. PDA Journal of Pharmaceutical Science and Technology. 2008;62:111–124. [PubMed] [Google Scholar]
- 531.Li J-m, Chen W, Wang H, Jin C, Yu X-j, Lu W-y, Cui L, Fu D-l, Ni Q-x, Hou H-m. Acta Pharmacologica Sinica. 2009;30:1337–1343. doi: 10.1038/aps.2009.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 532.Shen Z, Li Y, Kohama K, Oneill B, Bi J. Pharmacological Research. 2011;63:51–58. doi: 10.1016/j.phrs.2010.10.012. [DOI] [PubMed] [Google Scholar]
- 533.Khan W, Kapoor M, Kumar N. Acta Biomaterialia. 2007;3:541–549. doi: 10.1016/j.actbio.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 534.Wei Q, Li B, Yi N, Su B, Yin Z, Zhang F, Li J, Zhao C. Journal of Biomedical Materials Research Part A. 2011;96A:38–45. doi: 10.1002/jbm.a.32956. [DOI] [PubMed] [Google Scholar]
- 535.Zehr KJ. The Annals of Thoracic Surgery. 2007;84:1048–1052. doi: 10.1016/j.athoracsur.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 536.Somer FD, Delanghe J, Somers P, Debrouwere M, Nooten GV. Journal of Biomedical Materials Research Part A. 2008;86A:1106–1112. doi: 10.1002/jbm.a.31705. [DOI] [PubMed] [Google Scholar]
- 537.Rijken DC, Lijnen HR. Journal of Thrombosis and Haemostasis. 2008;7:4–13. doi: 10.1111/j.1538-7836.2008.03220.x. [DOI] [PubMed] [Google Scholar]
- 538.Shaikh FM, Callanan A, Kavanagh EG, Burke PE, Grace PA, McGloughlin TM. Cells, Tissues, Organs. 2008;188:333–346. doi: 10.1159/000139772. [DOI] [PubMed] [Google Scholar]
- 539.Schillinger U, Wexel G, Hacker C, Kullmer M, Koch C, Gerg M, Vogt S, Ueblacker P, Tischer T, Hensler D, Wilisch J, Aigner J, Walch A, Stemberger A, Plank C. Pharmaceutical Research. 2008;25:2946–2962. doi: 10.1007/s11095-008-9719-8. [DOI] [PubMed] [Google Scholar]
- 540.Zhibo X, Miaobo Z. Aesthetic Surgery Journal. 2009;29:32–34. doi: 10.1016/j.asj.2008.10.008. [DOI] [PubMed] [Google Scholar]
- 541.Yang HS, Bhang SH, Hwang JW, Kim D-I, Kim B-S. Tissue Engineering Part A. 2010;16:2113–2119. doi: 10.1089/ten.TEA.2009.0673. [DOI] [PubMed] [Google Scholar]
- 542.Kuehn C, Graf K, Mashaqi B, Pichlmaier M, Heuer W, Hilfiker A, Stiesch M. Journal of Surgical Research. 2010;164:e185–e191. doi: 10.1016/j.jss.2010.05.029. [DOI] [PubMed] [Google Scholar]
- 543.Hou T, Xu J, Li Q, Feng J, Zen L. Tissue Engineering Part A. 2008;14:1173–1182. doi: 10.1089/ten.tea.2007.0159. [DOI] [PubMed] [Google Scholar]
- 544.Dickhut A, Dexheimer V, Martin K, Lauinger R, Heisel C, Richter W. Tissue Engineering Part A. 2010;16:453–464. doi: 10.1089/ten.TEA.2009.0168. [DOI] [PubMed] [Google Scholar]
- 545.Peura M, Siltanen A, Saarinen I, Soots A, Bizik J, Vuola J, Harjula A, Kankuri E. Journal of Biomedical Materials Research Part A. 2010;95A:658–664. doi: 10.1002/jbm.a.32881. [DOI] [PubMed] [Google Scholar]
- 546.Mana M, Cole M, Cox S. Wound Repair and Regeneration. 2006;14:72–80. doi: 10.1111/j.1743-6109.2005.00091.x. [DOI] [PubMed] [Google Scholar]
- 547.Candela T, Moya M, Haustant M, Fouet A. Canadian Journal of Microbiology. 2009;55:627–632. doi: 10.1139/w09-003. [DOI] [PubMed] [Google Scholar]
- 548.Yeh C-M, Wang J-P, Lo S-C, Chan W-C, Lin M-Y. Biotechnology Progress. 2010;26:1001–1007. doi: 10.1002/btpr.417. [DOI] [PubMed] [Google Scholar]
- 549.Liu J, He D, Li X-z, Gao S, Wu H, Liu W, Gao X, Zhou T. International Journal of Food Microbiology. 2010;142:190–197. doi: 10.1016/j.ijfoodmicro.2010.06.023. [DOI] [PubMed] [Google Scholar]
- 550.Jeong Y-I, Na H-S, Cho K-O, Lee H-C, Nah J-W, Cho C-S. International Journal of Pharmaceutics. 2009;365:150–156. doi: 10.1016/j.ijpharm.2008.08.011. [DOI] [PubMed] [Google Scholar]
- 551.Matsusaki M, Akashi M. Biomacromolecules. 2005;6:3351–3356. doi: 10.1021/bm050369m. [DOI] [PubMed] [Google Scholar]
- 552.Yoshida H, Klinkhammer K, Matsusaki M, Moller M, Klee D, Akashi M. Macromolecular Bioscience. 2009;9:568–574. doi: 10.1002/mabi.200800334. [DOI] [PubMed] [Google Scholar]
- 553.Homsi J, Simon GR, Garrett CR, Springett G, Conti RD, Chiappori AA, Munster PN, Burton MK, Stromatt S, Allievi C, Angiuli P, Eisenfeld A, Sullivan DM, Daud AI. Clinical Cancer Research. 2007;13:5855–5861. doi: 10.1158/1078-0432.CCR-06-2821. [DOI] [PubMed] [Google Scholar]
- 554.Van S, Das SK, Wang X, Feng Z, Jin Y, Hou Z, Chen F, Pham A, Jiang N, Howell SB, Yu L. International Journal of Nanomedicine. 2010;5:825–837. doi: 10.2147/IJN.S13482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 555.Sun Y, Tang Y, Chu M, Song S, Xin Y. International Journal of Nanomedicine. 2008;3:249–256. doi: 10.2147/ijn.s2532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 556.Portilla-Arias JA, Camargo B, Garcia-Alzarez M, IIarduya AMd, Munoz-Guerra S. Journal of Biomaterials Science, Polymer Edition. 2009;20:1065–1079. doi: 10.1163/156856209X444420. [DOI] [PubMed] [Google Scholar]
- 557.Okamoto S, Matsuura M, Akagi T, Akashi M, Tanimoto T, Ishikawa T. Vaccine. 2009;27:5896–5905. doi: 10.1016/j.vaccine.2009.07.037. [DOI] [PubMed] [Google Scholar]
- 558.Matsuo K, Ishii Y, Matsuo K, Yoshinaga T, Akashi M, Mukai Y, Yoshioka Y, Okada N, Nakagawa S. Biological & Pharmaceutical Bulletin. 2010;33:2003–2007. doi: 10.1248/bpb.33.2003. [DOI] [PubMed] [Google Scholar]
- 559.Izumi Y, Yamamoto M, Kawamura M, Adachi T, Kobayashi K. Surgery. 2007;141:678–681. doi: 10.1016/j.surg.2006.12.010. [DOI] [PubMed] [Google Scholar]
- 560.Matsusaki M, Yoshida H, Akashi M. Biomaterials. 2007;28:2729–2737. doi: 10.1016/j.biomaterials.2007.02.015. [DOI] [PubMed] [Google Scholar]
- 561.Deng C, Tian H, Zhang P, Sun J, Chen X, Jing X. Biomacromolecules. 2006;7:590–596. doi: 10.1021/bm050678c. [DOI] [PubMed] [Google Scholar]
- 562.Sun J, Deng C, Chen X, Yu H, Tian H, Sun J, Jing X. Biomacromolecules. 2007;8:1013–1017. doi: 10.1021/bm0609792. [DOI] [PubMed] [Google Scholar]
- 563.Gryparis EC, Mattheolabakis G, Bikiaris D, Avgoustakis K. Drug Delivery. 2007;14:371–380. doi: 10.1080/10717540701202937. [DOI] [PubMed] [Google Scholar]
- 564.Cao N, Cheng D, Zou S, Ai H, Gao J, Shuai X. Biomaterials. 2011;32:2222–2232. doi: 10.1016/j.biomaterials.2010.11.061. [DOI] [PubMed] [Google Scholar]
- 565.Hsu F-Y, Cheng Y-Y, Tsai S-W, Tsai W-B. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2010;95B:29–35. doi: 10.1002/jbm.b.31679. [DOI] [PubMed] [Google Scholar]
- 566.Colonna C, Conti B, Perugini P, Pavanetto F, Modena T, Dorati R, Genta I. Journal of Microencapsulation. 2007;24:553–564. doi: 10.1080/02652040701449608. [DOI] [PubMed] [Google Scholar]
- 567.Wei L, Cai C, Lin J, Chen T. Biomaterials. 2009;30:2606–2613. doi: 10.1016/j.biomaterials.2009.01.006. [DOI] [PubMed] [Google Scholar]
- 568.Peng S-F, Tseng MT, Ho Y-C, Wei M-C, Liao Z-X, Sung H-W. Biomaterials. 2011;32:239–248. doi: 10.1016/j.biomaterials.2010.08.081. [DOI] [PubMed] [Google Scholar]
- 569.Takehara M, Hibino A, Saimura M, Hirohara H. Biotechnology Letters. 2010;32:1299–1303. doi: 10.1007/s10529-010-0294-9. [DOI] [PubMed] [Google Scholar]
- 570.Couffin-Hoarau A-C, Aubertin A-M, Boustta M, Schmidt S, Fehrentz J-A, Martinez J, Vert M. Biomacromolecules. 2009;10:865–876. doi: 10.1021/bm801376v. [DOI] [PubMed] [Google Scholar]
- 571.Tang CK, Sheng K-C, Pouniotis D, Esparon S, Son H-Y, Kim C-W, Pietersz GA, Apostolopoulos V. Vaccine. 2008;26:3827–3834. doi: 10.1016/j.vaccine.2008.05.008. [DOI] [PubMed] [Google Scholar]
- 572.Yu H, Guo X, Qi X, Liu P, Shen X, Duan Y. Journal of Materials Science: Materials in Medicine. 2008;19:1275–1281. doi: 10.1007/s10856-007-3237-9. [DOI] [PubMed] [Google Scholar]
- 573.Bertram JP, Jay SM, Hynes SR, Robinson R, Criscione JM, Lavik EB. Acta Biomaterialia. 2009;5:2860–2871. doi: 10.1016/j.actbio.2009.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 574.Tahara K, Furukawa S, Yamamoto H, Kawashima Y. International Journal of Pharmaceutics. 2010;392:311–313. doi: 10.1016/j.ijpharm.2010.03.042. [DOI] [PubMed] [Google Scholar]
- 575.Nottelet B, Ghzaoui AE, Coudane J, Vert M. Biomacromolecules. 2007;8:2594–2601. doi: 10.1021/bm700449c. [DOI] [PubMed] [Google Scholar]
- 576.Itoh K, Tokumi S, Kimura T, Nagase A. Langmuir. 2008;24:13426–13433. doi: 10.1021/la8015832. [DOI] [PubMed] [Google Scholar]
- 577.Isaksson K, Akerberg D, Andersson R, Tingstedt B. European Surgical Research. 2010;44:17–22. doi: 10.1159/000258654. [DOI] [PubMed] [Google Scholar]
- 578.Zheng Z, Zhang L, Kong L, Wang A, Gong Y, Zhang X. Journal of Biomedical Materials Research Part A. 2009;89A:453–465. doi: 10.1002/jbm.a.31979. [DOI] [PubMed] [Google Scholar]
- 579.Bourke SL, Kohn J. Advanced Drug Delivery Reviews. 2003;55:447–466. doi: 10.1016/s0169-409x(03)00038-3. [DOI] [PubMed] [Google Scholar]
- 580.Yuan H, Luo K, Lai Y, Pu Y, He B, Wang G, Wu Y, Gu Z. Molecular Pharmaceutics. 2010;7:953–962. doi: 10.1021/mp1000923. [DOI] [PubMed] [Google Scholar]
- 581.Chiu H-C, Kopeckova P, Deshmane SS, Kopecek J. Journal of Biomedical Materials Research. 1997;34:381–392. doi: 10.1002/(sici)1097-4636(19970305)34:3<381::aid-jbm13>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 582.Wadhwa S, Mumper RJ. Molecular Pharmaceutics. 2010;7:854–862. doi: 10.1021/mp1000058. [DOI] [PubMed] [Google Scholar]
- 583.Chen L, Tian H, Chen J, Chen X, Huang Y, Jing X. The Journal of Gene Medicine. 2010;12:64–76. doi: 10.1002/jgm.1405. [DOI] [PubMed] [Google Scholar]
- 584.Jessel N, Oulad-Abdelghani M, Meyer F, Lavalle P, Haikel Y, Schaaf P. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:8618–8621. doi: 10.1073/pnas.0508246103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 585.Pilbat A-M, Ball V, Schaaf P, Voegel J-C, Szalontai B. Langmuir. 2006;22:5753–5759. doi: 10.1021/la060454v. [DOI] [PubMed] [Google Scholar]
- 586.Song Z, Yin J, Luo K, Zheng Y, Yang Y, Li Q, Yan S, Chen X. Macromolecular Bioscience. 2009;9:268–278. doi: 10.1002/mabi.200800164. [DOI] [PubMed] [Google Scholar]
- 587.Feng X, Lv F, Liu L, Tang H, Xing C, Yang Q, Wang S. ACS Applied Materials & Interfaces. 2010;2:2429–2435. doi: 10.1021/am100435k. [DOI] [PubMed] [Google Scholar]
- 588.Huang W, Wang W, Wang P, Tian Q, Zhang C, Wang C, Yuan Z, Liu M, Wan H, Tang H. Acta Biomaterialia. 2010;6:3927–3935. doi: 10.1016/j.actbio.2010.04.021. [DOI] [PubMed] [Google Scholar]
- 589.O’Brien MER, Socinski MA, Popovich AY, Bondarenko IN, Tomova A, Bilynskyi BT, Hotko YS, Ganul VL, Kostinsky IY, Eisenfeld AJ, Sandalic L, Oldham FB, Bandstra B, Sandler AB, Singer JW. Journal of Thoracic Oncology. 2008;3:728–734. doi: 10.1097/JTO.0b013e31817c6b68. [DOI] [PubMed] [Google Scholar]
- 590.Ke T, Feng Y, Guo J, Parker DL, Lu Z-R. Magnetic Resonance Imaging. 2006;24:931–940. doi: 10.1016/j.mri.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 591.Zhang G, Zhang R, Wen X, Li L, Li C. Biomacromolecules. 2008;9:36–42. doi: 10.1021/bm700713p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 592.Melancon MP, Lu W, Huang Q, Thapa P, Zhou D, Ng C, Li C. Biomaterials. 2010;31:6567–6573. doi: 10.1016/j.biomaterials.2010.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 593.Tian M, Wen X, Jackson EF, Ng C, Uthamanthil R, Liang D, Gelovani JG, Li C. Contrast Media & Molecular Imaging. 2011 doi: 10.1002/cmmi.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 594.Deng M, Wang R, Rong G, Sun J, Zhang X, Chen X, Jing X. BIomaterials. 2004;25:3553–3558. doi: 10.1016/j.biomaterials.2003.10.018. [DOI] [PubMed] [Google Scholar]
- 595.Pang Z, Lu W, Gao H, Hu K, Chen J, Zhang C, Gao X, Jiang X, Zhu C. Journal of Controlled Release. 2008;128:120–127. doi: 10.1016/j.jconrel.2008.03.007. [DOI] [PubMed] [Google Scholar]
- 596.Layman H, Spiga M-G, Brooks T, Pham S, Webster KA, Anderopoulos FM. Biomaterials. 2007;28:2646–2654. doi: 10.1016/j.biomaterials.2007.01.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 597.Cao B, Yin J, Yan S, Cui L, Chen X, Xie Y. Macromolecular Bioscience. 2010 doi: 10.1002/mabi.201000389. [DOI] [PubMed] [Google Scholar]
- 598.Kakizawa Y, Furukawa S, Kataoka K. Journal of Controlled Release. 2004;97:345–356. doi: 10.1016/j.jconrel.2004.03.031. [DOI] [PubMed] [Google Scholar]
- 599.Arimura H, Ohya Y, Ouchi T. Biomacromolecules. 2005;6:720–725. doi: 10.1021/bm0494491. [DOI] [PubMed] [Google Scholar]
- 600.Karal-Yilmaz O, Kayaman-Apohan N, Misirli Z, Baysal K, Baysal BM. Journal of Materials Science: Materials in Medicine. 2006;17:213–227. doi: 10.1007/s10856-006-7307-1. [DOI] [PubMed] [Google Scholar]
- 601.Cha E-J, Kim JE, Ahn C-H. European Journal of Pharmaceutical Sciences. 2009;38:341–346. doi: 10.1016/j.ejps.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 602.Ponta A, Bae Y. Pharmaceutical Research. 2010;27:2330–2342. doi: 10.1007/s11095-010-0120-z. [DOI] [PubMed] [Google Scholar]
- 603.Pitarresi G, Saiano F, Cavallaro G, Mandracchia D, Palumbo FS. International Journal of Pharmaceutics. 2007;335:130–137. doi: 10.1016/j.ijpharm.2006.11.012. [DOI] [PubMed] [Google Scholar]
- 604.Meyer K, Palmer JW. Journal of Biological Chemistry. 1934;107:629–634. [Google Scholar]
- 605.Fraser JRE, Laurent TC, Laurent UBG. Journal of Internal Medicine. 1997;242:27–33. doi: 10.1046/j.1365-2796.1997.00170.x. [DOI] [PubMed] [Google Scholar]
- 606.Rapta P, Valachova K, Gemeiner P, Soltes L. Chemistry & Biodiversity. 2009;6:162–169. doi: 10.1002/cbdv.200800075. [DOI] [PubMed] [Google Scholar]
- 607.Carlson GA, Dragoo JL, Samimi B, Bruckner DA, Bernard GW, Hedrick M, Benhaim P. Biochemical and Biophysical Research Communications. 2004;321:472–478. doi: 10.1016/j.bbrc.2004.06.165. [DOI] [PubMed] [Google Scholar]
- 608.Lloyd LL, Kennedy JF, Methacanon P, Paterson M, Knill CJ. Carbohydrate Polymers. 1998;37:315–322. [Google Scholar]
- 609.Benedetti L, Cortivo R, Berti T, Berti A, Pea F, Mazzo M, Moras M, Abatangelo G. Biomaterials. 1993;14:1154–1160. doi: 10.1016/0142-9612(93)90160-4. [DOI] [PubMed] [Google Scholar]
- 610.Avitabile T, Marano F, Castiglione F, Bucolo C, Cro M, Ambrosio L, Ferrauto C, Reibaldi A. Biomaterials. 2001;22:195–200. doi: 10.1016/s0142-9612(00)00169-1. [DOI] [PubMed] [Google Scholar]
- 611.Lepidi S, Abatangelo G, Vindigni V, Deriu GP, Zavan B, Tonello C, Cortivo R. FASEB Journal. 2006;20:103–105. doi: 10.1096/fj.05-4802fje. [DOI] [PubMed] [Google Scholar]
- 612.Zavan B, Vindigni V, Lepidi S, Iacopetti I, Avruscio G, Abatangelo G, Cortivo R. FASEB Journal. 2008;22:2853–2861. doi: 10.1096/fj.08-107284. [DOI] [PubMed] [Google Scholar]
- 613.Temiz A, Kazikdas KC, Ergur B, Tugyan K, Bozok S, Kaya D, Guneli E. Otolarygology -Head and Neck Surgery. 2010;143:772–778. doi: 10.1016/j.otohns.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 614.Huang T-W, Cheng P-W, Chan Y-H, Yeh T-H, Young Y-H, Young T-H. Biomaterials. 2010;31:6701–6709. doi: 10.1016/j.biomaterials.2010.05.054. [DOI] [PubMed] [Google Scholar]
- 615.Wolf F, Haug M, Farhadi J, Candrian C, Martin I, Barbero A. eCells & Materials Journal. 2008;15:1–10. doi: 10.22203/ecm.v015a01. [DOI] [PubMed] [Google Scholar]
- 616.Pandis L, Zavan B, Abatangelo G, Lepidi S, Cortivo R, Vindigni V. Journal of Biomedical Materials Research. 2010;93A:1289–1296. doi: 10.1002/jbm.a.32626. [DOI] [PubMed] [Google Scholar]
- 617.Zavan B, Abatangelo G, Mazzoleni F, Bassetto F, Cortivo R, Vindigni V. Neurological Research. 2008;30:190–196. doi: 10.1179/174313208X281082. [DOI] [PubMed] [Google Scholar]
- 618.Pasquinelli G, Orrico C, Foroni L, Bonafe F, Carboni M, Guarnieri C, Raimondo S, Penna C, Geuna S, Pagliaro P, Freyrie A, Stella A, Caldarera CM, Muscari C. Journal of Anatomy. 2008;213:520–530. doi: 10.1111/j.1469-7580.2008.00974.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 619.Grigolo B, Lisignoli G, Desando G, Cavallo C, Marconi E, Tschon M, Giavaresi G, Fini M, Giardino R, Facchini A. Tissue Engineering Part C: Methods. 2009;15:647–658. doi: 10.1089/ten.TEC.2008.0569. [DOI] [PubMed] [Google Scholar]
- 620.Ifkovits JL, Tous E, Minakawa M, Morita M, Robb JD, Koomalsingh KJ, Gorman JH, Gorman RC, Burdick JA. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:11507–11512. doi: 10.1073/pnas.1004097107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 621.Lee H, Mok H, Lee S, Oh Y-K, Park TG. Journal of Controlled Release. 2007;119:245–252. doi: 10.1016/j.jconrel.2007.02.011. [DOI] [PubMed] [Google Scholar]
- 622.Al-Ghananeem AM, Malkawi AH, Mauammer YM, Balko JM, Black EP, Mourad W, Romoud E. AAPS PharmSciTech. 2009;10:410–417. doi: 10.1208/s12249-009-9222-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 623.Zavan B, Vindigni V, Vezzu K, Zorzato G, Luni C, Abatangelo G, Elvassore N, Cortivo R. Journal of Materials Science: Materials in Medicine. 2009;20:235–247. doi: 10.1007/s10856-008-3566-3. [DOI] [PubMed] [Google Scholar]
- 624.Choi KY, Chung H, Min KH, Yoon HY, Kim K, Park JH, Kwon IC, Jeong SY. Biomaterials. 2010;31:106–114. doi: 10.1016/j.biomaterials.2009.09.030. [DOI] [PubMed] [Google Scholar]
- 625.Heijink A, Yaszemski MJ, Patel R, Rouse MS, Lewallen DG, Hanssen AD. Clinical Orthopaedics and Related Research. 2006;451:29–33. doi: 10.1097/01.blo.0000229319.45416.81. [DOI] [PubMed] [Google Scholar]
- 626.Gianolio DA, Philbrook M, Avila LZ, Young LE, Plate L, Santos MR, Bernasconi R, Liu H, Ahn S, Sun W, Jarrett PK, Miller RJ. Bioconjugate Chemistry. 2008;19:1767–1774. doi: 10.1021/bc8000479. [DOI] [PubMed] [Google Scholar]
- 627.Lee F, Chung JE, Kurisawa M. Journal of Controlled Release. 2009;134:186–193. doi: 10.1016/j.jconrel.2008.11.028. [DOI] [PubMed] [Google Scholar]
- 628.Hirakura T, Yasugi K, Nemoto T, Sato M, Shimoboji T, Aso Y, Morimoto N, Akiyoshi K. Journal of Controlled Release. 2010;142:483–489. doi: 10.1016/j.jconrel.2009.11.023. [DOI] [PubMed] [Google Scholar]
- 629.Pitarresi G, Palumbo FS, Albanese A, Fiorica C, Picone P, Giammona G. Journal of Drug Targeting. 2010;18:264–276. doi: 10.3109/10611860903434027. [DOI] [PubMed] [Google Scholar]
- 630.Zille H, Paquet J, Henrionnet C, Scala-Bertola J, Leonard M, Six JL, Deschamp F, Netter P, Verges J, Gillet P, Grossin L. Bio-Medical Materials and Engineering. 2010;20:235–242. doi: 10.3233/BME-2010-0637. [DOI] [PubMed] [Google Scholar]
- 631.Lee H, Ahn C-H, Park TG. Macromolecular Bioscience. 2009;9:336–342. doi: 10.1002/mabi.200800229. [DOI] [PubMed] [Google Scholar]
- 632.Mondalek FG, Ashley RA, Roth CC, Kibar Y, Shakir N, Ihnat MA, Fung K-M, Grady BP, Kropp BP, Lin H-K. Journal of Biomedical Materials Research Part A. 2010;94A:712–719. doi: 10.1002/jbm.a.32748. [DOI] [PubMed] [Google Scholar]
- 633.Yadav AK, Mishra P, Jain S, Mishra P, Mishra AK, Agrawal GP. Journal of Drug Targeting. 2008;16:464–478. doi: 10.1080/10611860802095494 . [DOI] [PubMed] [Google Scholar]
- 634.Yenice I, Mocan MC, Palaska E, Bochot A, Bilensoy E, Vural I, Irkec M, Hincal AA. Experimental Eye Research. 2008;87:162–167. doi: 10.1016/j.exer.2008.04.002. [DOI] [PubMed] [Google Scholar]
- 635.Lee H, Jeong Y, Park TG. Biomacromolecules. 2007;8:3705–3711. doi: 10.1021/bm700854j. [DOI] [PubMed] [Google Scholar]
- 636.Zhang X, Sharma KK, Boeglin M, Ogier J, Mainard D, Voegel J-C, Mely Y, Benkirane-Jessel N. Nano Letters. 2008;8:2432–2436. doi: 10.1021/nl801379y. [DOI] [PubMed] [Google Scholar]
- 637.Wang X, Ji J. Langmuir. 2009;25:11664–11671. doi: 10.1021/la9013575. [DOI] [PubMed] [Google Scholar]
- 638.Fuente Mdl, Seijo B, Alonso MJ. Investigative Opththalmology & Visual Science. 2008;49:2016–2024. doi: 10.1167/iovs.07-1077. [DOI] [PubMed] [Google Scholar]
- 639.Luppi B, Bigucci F, Mercolini L, Musenga A, Sorrenti M, Catenacci L, Zecchi V. Journal of Pharmacy and Pharmacology. 2009;61:151–157. doi: 10.1211/jpp/61.02.0003. [DOI] [PubMed] [Google Scholar]
- 640.Oyarzun-Ampuero FA, Brea J, Loza MI, Torres D, Alonso MJ. International Journal of Pharmaceutics. 2009;381:122–129. doi: 10.1016/j.ijpharm.2009.04.009. [DOI] [PubMed] [Google Scholar]
- 641.Ravina M, Cubillo E, Olmeda D, Novoa-Carballal R, Fernandez-Megia E, Riguera R, Sanchez A, Cano A, Alonso MJ. Pharmaceutical Research. 2010;27:2544–2555. doi: 10.1007/s11095-010-0263-y. [DOI] [PubMed] [Google Scholar]
- 642.Kosir MA, Quinn CCV, Wang W, Tromp G. Journal of Surgical Research. 2000;92:45–52. doi: 10.1006/jsre.2000.5840. [DOI] [PubMed] [Google Scholar]
- 643.Chan PS, Caron JP, Rosa GJM, Orth MW. Osteoarthritis and Cartilage. 2005;13:387–394. doi: 10.1016/j.joca.2005.01.003. [DOI] [PubMed] [Google Scholar]
- 644.Souich PD, Garcia AG, Verges J, Montell E. Journal of Cellular and Molecular Medicine. 2009;13:1451–1463. doi: 10.1111/j.1582-4934.2009.00826.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 645.Malavaki C, Mizumoto S, Karamanos N, Sagahara K. Connective Tissue Research. 2008;49:133–139. doi: 10.1080/03008200802148546. [DOI] [PubMed] [Google Scholar]
- 646.Liu Y, Cai S, Shu XZ, Shelby J, Prestwich GD. Wound Repair and Regeneration. 2007;15:245–251. doi: 10.1111/j.1524-475X.2007.00211.x. [DOI] [PubMed] [Google Scholar]
- 647.Wang D-A, Varghese S, Sharma B, Strehin I, Fermanian S, Gorham J, Fairbrother DH, Cascio B, Elisseeff JH. Nature Materials. 2007;6:385–392. doi: 10.1038/nmat1890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 648.Park JS, Yang HJ, Woo DG, Yang HN, Na K, Park K-H. Journal of Biomedical Materials Research Part A. 2010;92A:806–816. doi: 10.1002/jbm.a.32388. [DOI] [PubMed] [Google Scholar]
- 649.Chang K-Y, Hung L-H, Chu I-M, Ko C-S, Lee Y-D. Journal of Biomedical Materials Research Part A. 2010;92A:712–723. doi: 10.1002/jbm.a.32198. [DOI] [PubMed] [Google Scholar]
- 650.Villanueva I, Gladem SK, Kessler J, Bryant SJ. Matrix Biology. 2010;29:51–62. doi: 10.1016/j.matbio.2009.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 651.Nguyen LH, Kudva AK, Guckert NL, Linse KD, Roy K. Biomaterials. 2011;32:1327–1338. doi: 10.1016/j.biomaterials.2010.10.009. [DOI] [PubMed] [Google Scholar]
- 652.Liang W-H, Kienitz BL, Penick KJ, Welter JF, Zawodzinski TA, Baskaran H. Journal of Biomedical Materials Research Part A. 2010;94A:1050–1060. doi: 10.1002/jbm.a.32774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Shu XZ, Ahmad S, Liu Y, Prestwich GD. Journal of Biomedical Materials Research Part A. 2006;79A:902–912. doi: 10.1002/jbm.a.30831. [DOI] [PubMed] [Google Scholar]
- 654.Chen Y-L, Lee H-P, Chan H-Y, Sung L-Y, Chen H-C, Hu Y-C. Biomaterials. 2007;28:2294–2305. doi: 10.1016/j.biomaterials.2007.01.027. [DOI] [PubMed] [Google Scholar]
- 655.Susante JLCv, Pieper J, Buma P, Kuppevelt THv, Beuningen Hv, Kraan PMvd, Veerkamp JH, Berg WBvd, Veth RPH. Biomaterials. 2001;22:2359–2369. doi: 10.1016/s0142-9612(00)00423-3. [DOI] [PubMed] [Google Scholar]
- 656.Kojima K, Okamoto Y, Kojima K, Miyatake K, Fujise H, Shigemasa Y, Minami S. Journal of Veterinary Medical Science. 2004;66:1595–1598. doi: 10.1292/jvms.66.1595. [DOI] [PubMed] [Google Scholar]
- 657.Min B-M, Lee SW, Lee JW, Lim JN, You Y, Lee TS, Kang PH. Polymer. 2004;45:7137–7142. [Google Scholar]
- 658.Noh HK, Lee SW, Kim J-M, Oh J-E, Kim K-H, Chung C-P, Choi S-C, Park WH, Min B-M. Biomaterials. 2006;27:3934–3944. doi: 10.1016/j.biomaterials.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 659.Suzuki D, Takahashi M, Abe M, Sarukawa J, Tamura H, Tokura S, Kurahashi Y, Nagano A. Journal of Materials Science:Materials in Medicine. 2008;19:1307–1315. doi: 10.1007/s10856-007-3245-9. [DOI] [PubMed] [Google Scholar]
- 660.Ehrlich H, Steck E, Ilan M, Maldonado M, Muricy G, Bavestrello G, Kljajic Z, Carballo JL, Schiaparelli S, Ereskovsky A, Schupp P, Born R, Worch H, Bazhenov VV, Kurek D, Varlamov V, Vyalikh D, Kummer K, Sivkov VV, Molodtsov SL, Meissner H, Richter G, Hunoldt S, Kammer M, Paasch S, Krasokhin V, Patzke G, Brunner E, Richter W. International Journal of Biological Macromolecules. 2010;47:141–145. doi: 10.1016/j.ijbiomac.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 661.Nagahama H, Nwe N, Jayakumar R, Koiwa S, Furuike T, Tamura H. Carbohydrate Polymers. 2008;73:295–302. [Google Scholar]
- 662.Xia W, Liu P, Liu J. Bioresource Technology. 2008;99:6751–6762. doi: 10.1016/j.biortech.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 663.Ren D, Yi H, Wang W, Ma X. Carbohydrate Polymers. 2005;340:2403–2410. doi: 10.1016/j.carres.2005.07.022. [DOI] [PubMed] [Google Scholar]
- 664.Patois E, Cruz SO-d, Tille J-C, Walpoth B, Gurny R, Jordan O. Journal of Biomedical Materials Research Part A. 2009;91 A:324–330. doi: 10.1002/jbm.a.32211. [DOI] [PubMed] [Google Scholar]
- 665.Li D-H, Liu L-M, Tian K-L, Liu J-C, Fan X-Q. Carbohydrate Polymers. 2007;67:40–45. [Google Scholar]
- 666.Biagini G, Bertani A, Muzzarelli R, Damadei A, Benedetto GD, Belligolli A, Riccotti G, Zucchini C, Rizzoli C. Biomaterials. 1991;12:281–286. doi: 10.1016/0142-9612(91)90035-9. [DOI] [PubMed] [Google Scholar]
- 667.Clasen C, Wilhelms T, Kulicke W-M. Biomacromolecules. 2006;7:3210–3222. doi: 10.1021/bm060486x. [DOI] [PubMed] [Google Scholar]
- 668.Ribiero MP, Espiga A, Silva D, Baptista P, Henriques J, Ferreira C, Silva JC, Borges JP, Pires E, Chaves P, Correia IJ. Wound Repair and Regeneration. 2009;17:817–824. doi: 10.1111/j.1524-475X.2009.00538.x. [DOI] [PubMed] [Google Scholar]
- 669.Ishihara M, Nakanishi K, Ono K, Sato M, Kikuchi M, Saito Y, Yura H, Matsui T, Hattori M, Uenoyama M, Kurita A. Biomaterials. 2002;23:833–840. doi: 10.1016/s0142-9612(01)00189-2. [DOI] [PubMed] [Google Scholar]
- 670.Peluso G, Petillo O, Ranieri M, Santin M, Ambrosic L, Calabro D, Avallone B, Balasamo G. Biomaterials. 1994;15:1215–1220. doi: 10.1016/0142-9612(94)90272-0. [DOI] [PubMed] [Google Scholar]
- 671.Bianco ID, Balsinde J, Beltramo DM, Castagna LF, Landa CA, Dennis EA. FEBS Letters. 2000;466:292–294. doi: 10.1016/s0014-5793(00)01089-9. [DOI] [PubMed] [Google Scholar]
- 672.Park CJ, Gabrielson NP, Pack DW, Amison JRD, Johnson AJW. Biomaterials. 2009;30:436–444. doi: 10.1016/j.biomaterials.2008.09.060. [DOI] [PubMed] [Google Scholar]
- 673.Burkatovskaya M, Tegos GP, Swietlik E, Demidova TN, Castano AP, Hamblin MR. Biomaterials. 2006;27:4157–4164. doi: 10.1016/j.biomaterials.2006.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 674.Azab AK, Orkin B, Doviner V, Nissan A, Klein M, Srebnik M, Rubinstein A. Journal of Controlled Release. 2006;111:281–289. doi: 10.1016/j.jconrel.2005.12.014. [DOI] [PubMed] [Google Scholar]
- 675.Ignatova M, Manolova N, Markova N, Rashkov I. Macromolecular Bioscience. 2009;9:102–111. doi: 10.1002/mabi.200800189. [DOI] [PubMed] [Google Scholar]
- 676.Rahmani-Neishaboor E, Jackson J, Burt H, Ghahary A. Pharmaceutical Research. 2009;26:2002–2014. doi: 10.1007/s11095-009-9916-0. [DOI] [PubMed] [Google Scholar]
- 677.Wu L, Li H, Li S, Li X, Yuan X, Li X, Zhang Y. Journal of Biomedical Materials Research Part A. 2010;92A:563–574. doi: 10.1002/jbm.a.32393. [DOI] [PubMed] [Google Scholar]
- 678.Kim IY, Yoo MK, Seo JH, Park SS, Na HS, Lee HC, Kim SK, Cho CS. International Journal of Pharmaceutics. 2007;341:35–43. doi: 10.1016/j.ijpharm.2007.03.042. [DOI] [PubMed] [Google Scholar]
- 679.Chen R-N, Wang G-M, Chen C-H, Ho H-O, Sheu M-T. Biomacromolecules. 2006;7:1058–1064. doi: 10.1021/bm050754b. [DOI] [PubMed] [Google Scholar]
- 680.Liu B-S, Yao C-H, Fang S-S. Macromolecular Bioscience. 2008;8:432–440. doi: 10.1002/mabi.200700211. [DOI] [PubMed] [Google Scholar]
- 681.Murakami K, Aoki H, Nakamura S, Nakamura S-i, Takikawa M, Hanzawa M, Kishimoto S, Hattori H, Tanaka Y, Kiyosawa T, Sato Y, Ishihara M. Biomaterials. 2010;31:83–90. doi: 10.1016/j.biomaterials.2009.09.031. [DOI] [PubMed] [Google Scholar]
- 682.Meng X, Tian F, Yang J, He C-N, Xing N, Li F. Journal of Materials Science: Materials in Medicine. 2010;21:1751–1759. doi: 10.1007/s10856-010-3996-6. [DOI] [PubMed] [Google Scholar]
- 683.Aoyagi S, Onishi H, Machida Y. International Journal of Pharmaceutics. 2007;330:138–145. doi: 10.1016/j.ijpharm.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 684.Campos MGN, Rawls HR, Innocentini-Mei LH, Satsangi N. Journal of Materials Science: Materials in Medicine. 2009;20:537–542. doi: 10.1007/s10856-008-3611-2. [DOI] [PubMed] [Google Scholar]
- 685.Wedmore I, McManus JG, Pusateri AE, Holcomb JB. Journal of Trauma-Injury Infection & Critical Care. 2006;60:655–658. doi: 10.1097/01.ta.0000199392.91772.44. [DOI] [PubMed] [Google Scholar]
- 686.Brown MA, Daya MR, Worley JA. Journal of Emergency Medicine. 2009;37:1–7. doi: 10.1016/j.jemermed.2007.05.043. [DOI] [PubMed] [Google Scholar]
- 687.Malmquist JP, Clemens SC, Oien HJ, Wilson SL. Journal of Oral and Maxillofacial Surgery. 2008;66:1177–1183. doi: 10.1016/j.joms.2007.12.023. [DOI] [PubMed] [Google Scholar]
- 688.Gan Q, Wang T. Colloids andSurfaces B: Biointerfaces. 2007;59:24–34. doi: 10.1016/j.colsurfb.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 689.Amsden BG, Sukarto A, Knight DK, Shapka SN. Biomacromolecules. 2007;8:3758–3766. doi: 10.1021/bm700691e. [DOI] [PubMed] [Google Scholar]
- 690.Budhian A, Siegel SJ, Winey KI. International Journal of Pharmaceutics. 2008;346:151–159. doi: 10.1016/j.ijpharm.2007.06.011. [DOI] [PubMed] [Google Scholar]
- 691.Yuan X-B, Yuan Y-B, Jiang W, Liu J, Tian E-J, Shun H-M, Huang D-H, Yuan X-Y, Li H, Sheng J. International Journal of Pharmaceutics. 2008;349:241–248. doi: 10.1016/j.ijpharm.2007.07.045. [DOI] [PubMed] [Google Scholar]
- 692.Wang J, Feng S-S, Wang S, Chen Z-y. International Journal of Pharmaceutics. 2010;400:194–200. doi: 10.1016/j.ijpharm.2010.08.026. [DOI] [PubMed] [Google Scholar]
- 693.Yang R, Shim W-S, Cui F-D, Cheng G, XuHan, Jin Q-R, Kim D-D, Chung S-J, Shim C-K. International Journal of Pharmaceutics. 2009;371:142–147. doi: 10.1016/j.ijpharm.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 694.Chen H, Yang W, Chen H, Liu L, Gao F, Yang X, Jiang Q, Zhang Q, Wang Y. Colloids and Surfaces B: Biointerfaces. 2009;73:212–218. doi: 10.1016/j.colsurfb.2009.05.020. [DOI] [PubMed] [Google Scholar]
- 695.Slutter B, Bal S, Keijzer C, Mallants R, Hagenaars N, Que I, Kaijzel E, Eden Wv, Augustihns P, Lowik C, Bouwstra J, Broere F, Jiskoot W. Vaccine. 2010;28:6282–6291. doi: 10.1016/j.vaccine.2010.06.121. [DOI] [PubMed] [Google Scholar]
- 696.Zhang Y, Chen J, Zhang Y, Pan Y, Zhao J, Ren L, Liao M, Hu Z, Kong L, Wang J. Biotechnology and Applied Biochemistry. 2007;46:197–204. doi: 10.1042/BA20060163. [DOI] [PubMed] [Google Scholar]
- 697.Wang Q, Zhang N, Hu X, Yang J, Du Y. Journal of Biomedical Materials Research Part A. 2008;85A:881–887. doi: 10.1002/jbm.a.31544. [DOI] [PubMed] [Google Scholar]
- 698.Hu F-Q, Meng P, Dai Y-Q, Du Y-Z, You J, Wei X-H, Yuan H. European Journal of Pharmaceutics and Biopharmaceutics. 2008;70:749–757. doi: 10.1016/j.ejpb.2008.06.015. [DOI] [PubMed] [Google Scholar]
- 699.Germershaus O, Mao S, Sitterberg J, Bakowsky U, Kissel T. Journal of Controlled Release. 2008;125:145–154. doi: 10.1016/j.jconrel.2007.10.013. [DOI] [PubMed] [Google Scholar]
- 700.Shi H, Han C, Mao Z, Ma L, Gao C. Tissue Engineering Part A. 2008;14:1775–1785. doi: 10.1089/ten.tea.2007.0007. [DOI] [PubMed] [Google Scholar]
- 701.Niu X, Feng Q, Wang M, Guo X, Zheng Q. Journal of Controlled Release. 2009;134:111–117. doi: 10.1016/j.jconrel.2008.11.020. [DOI] [PubMed] [Google Scholar]
- 702.Mao Z, Shi H, Guo R, Ma M, Gao C, Han C, Shen J. Acta Biomaterialia. 2009;5:2983–2994. doi: 10.1016/j.actbio.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 703.Peng S-F, Yang M-J, Chun-Jen, Chen H-L, Lee P-W, Wei M-C, Sung H-W. Biomaterials. 2009;30:1797–1808. doi: 10.1016/j.biomaterials.2008.12.019. [DOI] [PubMed] [Google Scholar]
- 704.Chang C-H, Lin Y-H, Yeh C-L, Chen Y-C, Chiou S-F, Hsu Y-M, Chen Y-S, Wang C-C. Biomacromolecules. 2009;11:133–142. doi: 10.1021/bm900985h. [DOI] [PubMed] [Google Scholar]
- 705.Matsusaki M, Sakaguchi H, Serizawa T, Akashi M. Journal of Biomaterials Science, Polymer Edition. 2007;18:775–783. doi: 10.1163/156856207781034160. [DOI] [PubMed] [Google Scholar]
- 706.Borges O, Silva M, Sousa Ad, Borchard G, Junginger HE, Cordeiro-da-Silva A. International Immunopharmacology. 2008;8:1773–1780. doi: 10.1016/j.intimp.2008.08.013. [DOI] [PubMed] [Google Scholar]
- 707.Woitiski CB, Neufeld RJ, Ribeiro AJ, Veiga F. Acta Biomaterialia. 2009;5:2475–2484. doi: 10.1016/j.actbio.2009.03.007. [DOI] [PubMed] [Google Scholar]
- 708.Perioli L, Ambrogi V, Pagano C, Scuota S, Rossi C. International Journal of Pharmaceutics. 2009;377:120–127. doi: 10.1016/j.ijpharm.2009.05.016. [DOI] [PubMed] [Google Scholar]
- 709.Patil S, Babbar A, Mathur R, Mishra A, Sawant K. Journal of Drug Targeting. 2010;18:321–331. doi: 10.3109/10611861003663523. [DOI] [PubMed] [Google Scholar]
- 710.Pawar D, Goyal AK, Mangal S, Mishra N, Vaidya B, Tiwari S, Jain AK, Vyas SP. The AAPS Journal. 2010;12:130–137. doi: 10.1208/s12248-009-9169-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 711.Zhang L, Ao Q, Wang A, Lu G, Kong L, Gong Y, Zhao N, Zhang X. Journal of Biomedical Materials Research Part A. 2006;77A:277–284. doi: 10.1002/jbm.a.30614. [DOI] [PubMed] [Google Scholar]
- 712.Ni R-F, Kranokpiraksa P, Pavcnik D, Kakizawa H, Uchida BT, Keller FS, Rosch J. Cardiovascular and Interventional Radiology. 2009;32:313–316. doi: 10.1007/s00270-008-9426-1. [DOI] [PubMed] [Google Scholar]
- 713.Lu G, Ling K, Zhao P, Xu Z, Deng C, Zheng H, Huang J, Chen J. Wound Repair and Regeneration. 2010;18:70–79. doi: 10.1111/j.1524-475X.2009.00557.x. [DOI] [PubMed] [Google Scholar]
- 714.Ishikawa N, Suzuki Y, Dezawa M, Kataoka K, Ohta M, Cho H, Ide C. Journal of BIomedical Materials Research Part A. 2009;89A:1118–1124. doi: 10.1002/jbm.a.32389. [DOI] [PubMed] [Google Scholar]
- 715.Fan J, Shang Y, Yuan Y, Yang J. Journal of Materials Science: Materials in Medicine. 2010;21:319–327. doi: 10.1007/s10856-009-3833-y. [DOI] [PubMed] [Google Scholar]
- 716.Deshpande MS, Kuchroo PV. Biotechnology Progress. 2010;26:1424–1430. doi: 10.1002/btpr.458. [DOI] [PubMed] [Google Scholar]
- 717.Majima T, Irie T, Sawaguchi N, Funakoshi T, Iwasaki N, Harada K, Minami A, Nishimura S. Proceedings of the Institution of Mechanical Engineers. Part H, Journal of Engineering in Medicine. 2007;221:537–546. doi: 10.1243/09544119JEIM203. [DOI] [PubMed] [Google Scholar]
- 718.Wang W, Itoh S, Konno K, Kikkawa T, Ichinose S, Sakai K, Ohkuma T, Watabe K. Journal of Biomedical Materials Research Part A. 2008;91A:994–1005. doi: 10.1002/jbm.a.32329. [DOI] [PubMed] [Google Scholar]
- 719.Weir MD, Xu HHK. Acta Biomaterialia. 2010;6:4118–4126. doi: 10.1016/j.actbio.2010.04.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 720.Dhandayuthapani B, Krishnan UM, Sethuraman S. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2010;94B:264–277. doi: 10.1002/jbm.b.31651. [DOI] [PubMed] [Google Scholar]
- 721.Sarukawa J, Takahashi M, Abe M, Suzuki D, Tokura S, Furuike T, Tamura H. Journal of Biomaterials Science, Polymer Edition. 2011;22:717–732. doi: 10.1163/092050610X491067. [DOI] [PubMed] [Google Scholar]
- 722.Chesnutt BM, Yuan Y, Buddington K, Haggard WO, Bumgardner JD. Tissue Engineering Part A. 2009;15:2571–2579. doi: 10.1089/ten.tea.2008.0054. [DOI] [PubMed] [Google Scholar]
- 723.Li LH, Kommareddy KP, Pilz C, Zhou CR, Fratzl P, Manjubala I. Acta Biomaterialia. 2010;6:2525–2531. doi: 10.1016/j.actbio.2009.03.028. [DOI] [PubMed] [Google Scholar]
- 724.Jiang T, Nukavarapu SP, Deng M, Jabbarzadeh E, Kofron MD, Doty SB, Abdel-Fattah WI, Laurencin CT. Acta Biomaterialia. 2010;6:3457–3470. doi: 10.1016/j.actbio.2010.03.023. [DOI] [PubMed] [Google Scholar]
- 725.Pfister LA, Papaloizos M, Merkle HP, Gander B. Journal of Biomedical Materials Research Part A. 2007;80A:932–937. doi: 10.1002/jbm.a.31052. [DOI] [PubMed] [Google Scholar]
- 726.Tan H, Chu CR, Payne KA, Marra KG. Biomaterials. 2009;30:2499–2506. doi: 10.1016/j.biomaterials.2008.12.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 727.Wang P-Y, Chow H-H, Lai J-Y, Liu H-L, Tsai W-B. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2009;91B:143–152. doi: 10.1002/jbm.b.31384. [DOI] [PubMed] [Google Scholar]
- 728.Hao T, Wen N, Cao J-K, Wang H-B, Lu S-H, Liu T, Lin Q-X, Duan C-M, Wang C-Y. Osteoarthritis and Cartilage. 2010;18:257–265. doi: 10.1016/j.joca.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 729.Zuidema JM, Pap MM, Jaroch DB, Morrison FA, Gilbert RJ. Acta Biomaterialia. 2011 doi: 10.1016/j.actbio.2010.11.039. [DOI] [PubMed] [Google Scholar]
- 730.Tan CS, Jejurikar A, Rai B, Bostrom T, Lawrie G, Grondahl L. Journal of Biomedical Materials Research Part A. 2009;91A:866–877. doi: 10.1002/jbm.a.32297. [DOI] [PubMed] [Google Scholar]
- 731.Ruvinov E, Leor J, Cohen S. Biomaterials. 2010;31:4573–4582. doi: 10.1016/j.biomaterials.2010.02.026. [DOI] [PubMed] [Google Scholar]
- 732.Rabbany SY, Pastore J, Yamamoto M, Miller T, Rafii S, Aras R, Penn M. Cell Transplantation. 2010;19:399–408. doi: 10.3727/096368909X481782. [DOI] [PubMed] [Google Scholar]
- 733.Chan AW, Neufeld RJ. Biomaterials. 2010;31:9040–9047. doi: 10.1016/j.biomaterials.2010.07.111. [DOI] [PubMed] [Google Scholar]
- 734.Kim D-H, Martin DC. Biomaterials. 2006;27:3031–3037. doi: 10.1016/j.biomaterials.2005.12.021. [DOI] [PubMed] [Google Scholar]
- 735.Ranganath SH, Kee I, Krantz WB, Chow PK-H, Wang C-H. Pharmaceutical Research. 2009;26:2101–2114. doi: 10.1007/s11095-009-9922-2. [DOI] [PubMed] [Google Scholar]
- 736.Lee J, Bhang SH, Park H, Kim B-S, Lee KY. Pharmaceutical Research. 2010;27:767–774. doi: 10.1007/s11095-010-0067-0. [DOI] [PubMed] [Google Scholar]
- 737.Choi DH, Park CH, Kim IH, Chun HJ, Park K, Han DK. Journal of Controlled Release. 2010;147:193–201. doi: 10.1016/j.jconrel.2010.07.103. [DOI] [PubMed] [Google Scholar]
- 738.Colinet I, Dulong V, Mocanu G, Picton L, Cerf DL. European Journal of Pharmaceutics and Biopharmaceutics. 2009;73:345–350. doi: 10.1016/j.ejpb.2009.07.008. [DOI] [PubMed] [Google Scholar]
- 739.Gou M, Men K, Shi H, Xiang M, Zhang J, Song J, Long J, Wan Y, Luo F, Zhao X, Quan Z. Nanoscale. 2011 doi: 10.1039/c0nr00758g. [DOI] [PubMed] [Google Scholar]
- 740.Giovagnoli S, Tsai T, DeLuca PP. AAPS PharmSciTech. 2010;11:212–220. doi: 10.1208/s12249-009-9361-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 741.Shastri DH, Prajapati ST, Patel LD. Current Drug Delivery. 2010;7:238–243. doi: 10.2174/156720110791560928. [DOI] [PubMed] [Google Scholar]
- 742.Lim SM, Oh SH, Lee HH, Yuk SH, Im GI, Lee JH. Journal of Materials Science: Materials in Medicine. 2010;21:2593–2600. doi: 10.1007/s10856-010-4118-1. [DOI] [PubMed] [Google Scholar]
- 743.Dai Y-N, Li P, Zhang J-P, Wang A-Q, Wei Q. Biopharmaceutics & Drug Disposition. 2008;29:173–184. doi: 10.1002/bdd.590. [DOI] [PubMed] [Google Scholar]
- 744.Meng X, Li P, Wei Q, Zhang H-X. Pharmaceutical Development and Technology. 2011;16:22–28. doi: 10.3109/10837450903479947. [DOI] [PubMed] [Google Scholar]
- 745.Zhao L, Tang M, Weir MD, Detamore MS, Xu HHK. Tissue Engineering Part A. 2011 doi: 10.1089/ten.tea.2010.0521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 746.Marsich E, Borgogna M, Donati I, Mozetic P, Strand BL, Salvador SG, Vittur F, Paoletti S. Journal of Biomedical Materials Research Part A. 2008;84A:364–376. doi: 10.1002/jbm.a.31307. [DOI] [PubMed] [Google Scholar]
- 747.Xu HHK, Weir MD, Simon CG. Dental Materials. 2008;24:1212–1222. doi: 10.1016/j.dental.2008.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 748.Orive G, Castro MD, Kong H-J, Hernandez RM, Ponce S, Mooney DJ, Pedraz JL. Journal of Controlled Release. 2009;135:203–210. doi: 10.1016/j.jconrel.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 749.Hunt NC, Shelton RM, Grover L. Biotechnology Journal. 2009;4:730–737. doi: 10.1002/biot.200800292. [DOI] [PubMed] [Google Scholar]
- 750.Moyer H, Kinney R, Singh KA, Williams JK, Schwartz Z, Boyan BD. Annals of Plastic Surgery. 2010;65:497–503. doi: 10.1097/SAP.0b013e3181d37713. [DOI] [PubMed] [Google Scholar]
- 751.Jin XB, Sun YS, Zhang K, Wang J, Shi TP, Ju XD, Lou SQ. Journal of Biomedical Materials Research Part A. 2008;86A:1077–1087. doi: 10.1002/jbm.a.31656. [DOI] [PubMed] [Google Scholar]
- 752.Qi X, Ye J, Wang Y. Journal of Biomedical Materials Research Part A. 2009;89A:980–987. doi: 10.1002/jbm.a.32054. [DOI] [PubMed] [Google Scholar]
- 753.Wang Q, Jamal S, Detamore MS, Berkland C. Journal of Biomedical Materials Research Part A. 2011;96A:520–527. doi: 10.1002/jbm.a.33000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 754.Hahn MS, Teply BA, Stevens MM, Zeitels SM, Langer R. BIomaterials. 2006;27:1104–1109. doi: 10.1016/j.biomaterials.2005.07.022. [DOI] [PubMed] [Google Scholar]
- 755.Jay SM, Shepherd BR, Andrejecsk JW, Kyriakides TR, Pober JS, Saltzman WM. Biomaterials. 2010;31:3054–3062. doi: 10.1016/j.biomaterials.2010.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 756.Zheng L, Fan HS, Sun J, Chen XN, Wang G, Zhang L, Fan YJ, Zhang XD. Journal of Biomedical Materials Research Part A. 2009;93A:783–792. doi: 10.1002/jbm.a.32588. [DOI] [PubMed] [Google Scholar]
- 757.Tan R, Feng Q, Jin H, Li J, Yu X, She Z, Wang M, Liu H. Journal of Biomaterials Science, Polymer Edition. 2011 doi: 10.1163/092050610X528561. [DOI] [PubMed] [Google Scholar]
- 758.Maguire T, Novik E, Schloss R, Yarmush M. Biotechnology and Bioengineering. 2006;93:581–591. doi: 10.1002/bit.20748. [DOI] [PubMed] [Google Scholar]
- 759.Olmez SS, Korkusuz P, Bilgili H, Senel S. Die Pharmazie. 2007;62:423–431. [PubMed] [Google Scholar]
- 760.Wittmer CR, Phelps JA, Lepus CM, Saltzman WM, Harding MJ, Tassel PRV. Biomaterials. 2008;29:4082–4090. doi: 10.1016/j.biomaterials.2008.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 761.Purcell EK, Singh A, Kipke DR. Tissue Engineering Part C: Methods. 2009;15:541–550. doi: 10.1089/ten.tec.2008.0302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 762.Liu XY, Nothias J-M, Scavone A, Garfinkel M, Millis JM. ASAIO Journal. 2010;56:241–245. doi: 10.1097/MAT.0b013e3181d7b8e3. [DOI] [PubMed] [Google Scholar]
- 763.Wang J-Z, Huang X-B, Xiao J, Yu W-T, Wang W, Xie W-Y, Zhang Y, Ma X-J. Journal of Biomedical Materials Research Part A. 2010;93A:910–919. doi: 10.1002/jbm.a.32590. [DOI] [PubMed] [Google Scholar]
- 764.Liang Y, Liu W, Han B, Yang C, Ma Q, Song F, Bi Q. Colloids and Surfaces B: Biointerfaces. 2011;82:1–7. doi: 10.1016/j.colsurfb.2010.07.043. [DOI] [PubMed] [Google Scholar]
- 765.Qi J, Chen A, You H, Li K, Zhang D, Guo F. Biomedical Materials. 2011;6:015006. doi: 10.1088/1748-6041/6/1/015006. [DOI] [PubMed] [Google Scholar]
- 766.Chan AW, Whitney RA, Neufeld RJ. Biomacromolecules. 2009;10:609–616. doi: 10.1021/bm801316z. [DOI] [PubMed] [Google Scholar]
- 767.Yamada Y, Hozumi K, Katagiri F, Kikkawa Y, Nomizu M. Peptide Science. 2010;94:711–720. doi: 10.1002/bip.21429. [DOI] [PubMed] [Google Scholar]
- 768.Wong TY, Preston LA, Schiller NL. Annual Review of Microbiology. 2000;54:289–340. doi: 10.1146/annurev.micro.54.1.289. [DOI] [PubMed] [Google Scholar]
- 769.Simmons CA, Alsberg E, Hsiong S, Kim WJ, Mooney DJ. Bone. 2004;35:562–569. doi: 10.1016/j.bone.2004.02.027. [DOI] [PubMed] [Google Scholar]
- 770.Boontheekul T, Kong H-J, Mooney DJ. Biomaterials. 2005;26:2455–2465. doi: 10.1016/j.biomaterials.2004.06.044. [DOI] [PubMed] [Google Scholar]
- 771.Pokrywczynska M, Drewa T, Jundzill A, Lysik J. Transplantation Proceedings. 2009;40:1664–1667. doi: 10.1016/j.transproceed.2008.03.135. [DOI] [PubMed] [Google Scholar]
- 772.Evangelista MB, Hsiong SX, Fernandes R, Sampaio P, Kong H-J, Barrias CC, Salema R, Barbosa MA, Mooney DJ, Granja PL. Biomaterials. 2007;28:3644–3655. doi: 10.1016/j.biomaterials.2007.04.028. [DOI] [PubMed] [Google Scholar]
- 773.Lee JW, Park YJ, Lee SJ, Lee SK, Lee KY. Biomaterials. 2010;31:5545–5551. doi: 10.1016/j.biomaterials.2010.03.063. [DOI] [PubMed] [Google Scholar]
- 774.Hill E, Boontheekul T, Mooney DJ. Tissue Engineering. 2006;12:1295–1304. doi: 10.1089/ten.2006.12.1295. [DOI] [PubMed] [Google Scholar]
- 775.Jeong SI, Krebs MD, Bonino CA, Khan SA, Alsberg E. Macromolecular Bioscience. 2010;10:934–943. doi: 10.1002/mabi.201000046. [DOI] [PubMed] [Google Scholar]
- 776.Crommen J, Vandorpe J, Schacht E. Journal of Controlled Release. 1993;24:167–180. [Google Scholar]
- 777.Leong KW, Brott BC, Langer R. Journal of Biomedical Materials Research. 1985;19:941–955. doi: 10.1002/jbm.820190806. [DOI] [PubMed] [Google Scholar]
- 778.Richards M, Dahiyat BI, Arm DM, Brown PR, Leong KW. Journal of Biomedical Materials Research. 1991;25:1151–1167. doi: 10.1002/jbm.820250908. [DOI] [PubMed] [Google Scholar]
- 779.Huang S-W, Wang J, Zhang P-C, Mao H-Q, Zhuo R-X, Leong KW. Biomacromolecules. 2004;5:306–311. doi: 10.1021/bm034241l. [DOI] [PubMed] [Google Scholar]
- 780.Chapman TM. Journal of Polymer Science Part A: Polymer Chemistry. 1989;27:1993–2005. [Google Scholar]


