Abstract
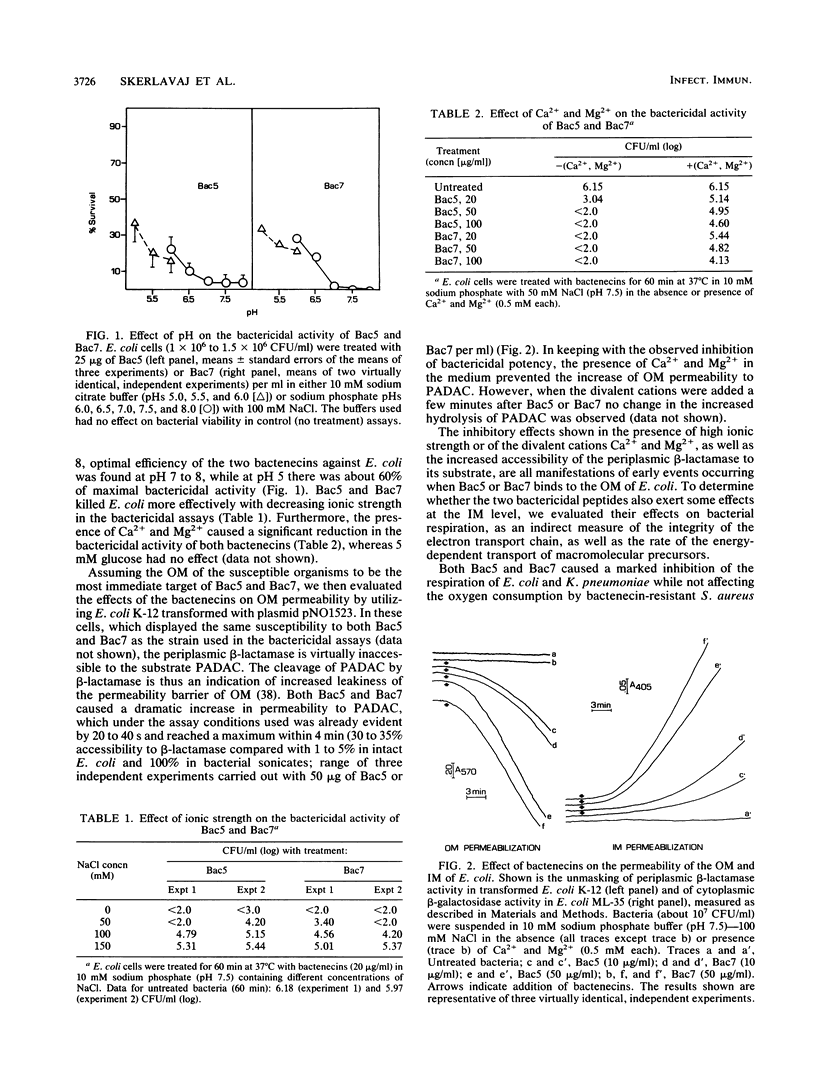
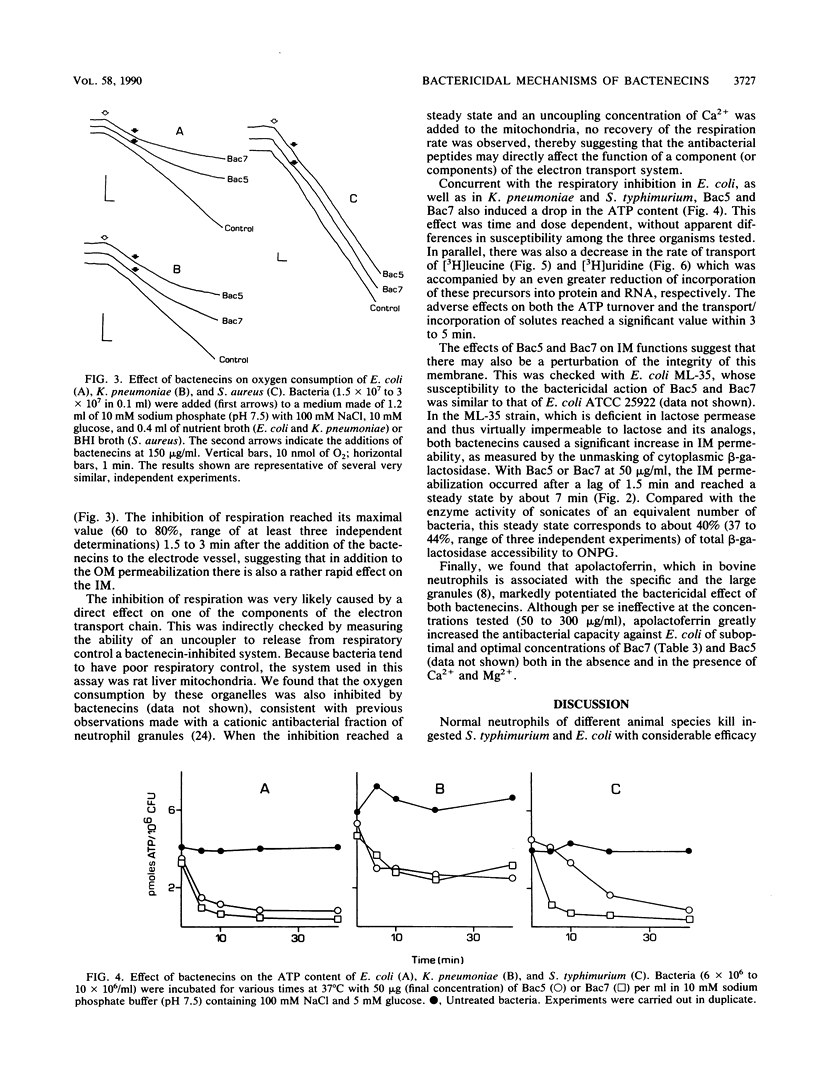
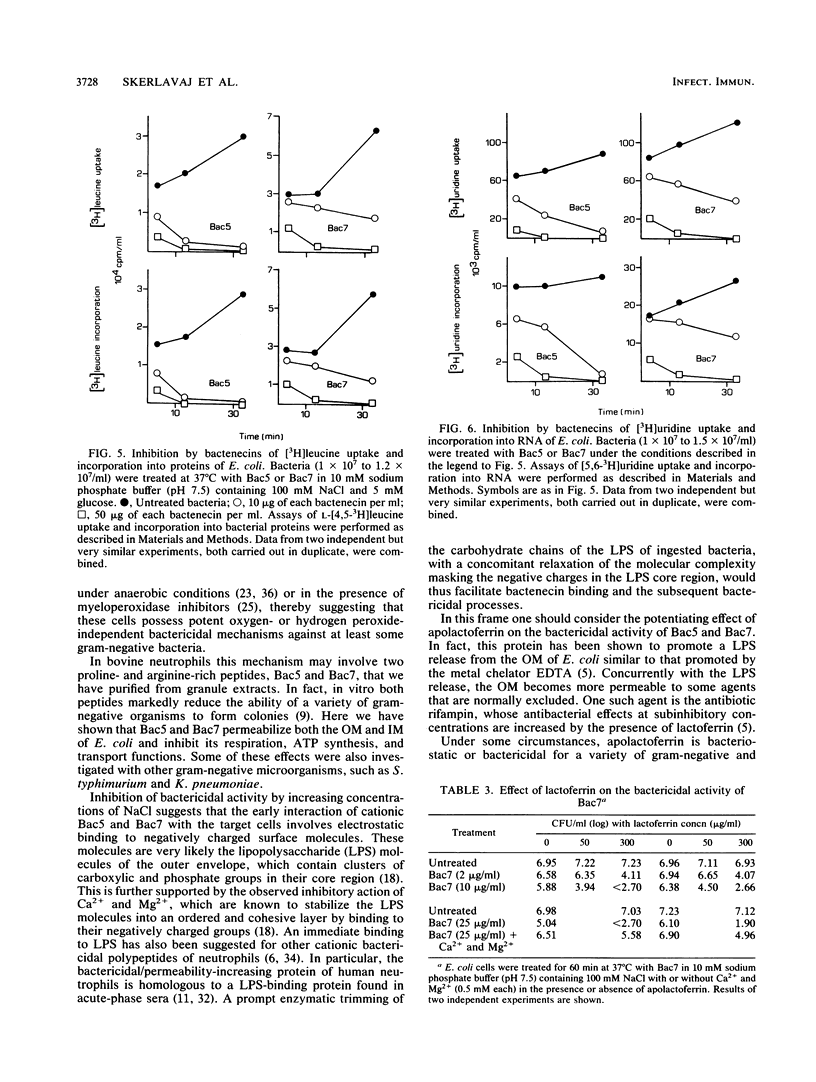
Bactenecins are a class of arginine-rich antibacterial peptides of bovine neutrophil granules. Two bactenecins with approximate molecular weights of 5,000 and 7,000 designated Bac5 and Bac7, respectively, exert in vitro a potent bactericidal activity toward several gram-negative bacteria (R. Gennaro, B. Skerlavaj, and D. Romeo, Infect. Immun. 57:3142-3146, 1989). We have now found that this activity shows an inverse relationship to the ionic strength of the medium and is inhibited by divalent cations and greatly potentiated by lactoferrin. Under conditions supporting marked bactericidal activity, the two peptides cause a rapid increase in the permeability of both the outer and inner membranes of Escherichia coli, as shown by unmasking of periplasmic beta-lactamase and of cytoplasmic beta-galactosidase. In addition, the two bactenecins inhibit the respiration of E. coli and Klebsiella pneumoniae but not of Bac5- and Bac7-resistant Staphylococcus aureus. Furthermore, they induce a drop in ATP content in E. coli, K. pneumoniae, and Salmonella typhimurium and a marked decrease in the rates of transport and incorporation of [3H]leucine and [3H]uridine into E. coli protein and RNA, respectively. In general, all these effects become evident within 1 to 2 min and reach their maximal expression within about 5 min. Overall, these data strongly suggest that the decrease in bacterial viability is causally related to the increase in membrane permeability and the subsequent fall in respiration-linked proton motive force, with the attendant loss of cellular metabolites and macromolecular biosynthesis ability.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anraku Y. Bacterial electron transport chains. Annu Rev Biochem. 1988;57:101–132. doi: 10.1146/annurev.bi.57.070188.000533. [DOI] [PubMed] [Google Scholar]
- Arnold R. R., Brewer M., Gauthier J. J. Bactericidal activity of human lactoferrin: sensitivity of a variety of microorganisms. Infect Immun. 1980 Jun;28(3):893–898. doi: 10.1128/iai.28.3.893-898.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baggiolini M., De Duve C., Masson P. L., Heremans J. F. Association of lactoferrin with specific granules in rabbit heterophil leukocytes. J Exp Med. 1970 Mar 1;131(3):559–570. doi: 10.1084/jem.131.3.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortner C. A., Miller R. D., Arnold R. R. Bactericidal effect of lactoferrin on Legionella pneumophila. Infect Immun. 1986 Feb;51(2):373–377. doi: 10.1128/iai.51.2.373-377.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellison R. T., 3rd, Giehl T. J., LaForce F. M. Damage of the outer membrane of enteric gram-negative bacteria by lactoferrin and transferrin. Infect Immun. 1988 Nov;56(11):2774–2781. doi: 10.1128/iai.56.11.2774-2781.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farley M. M., Shafer W. M., Spitznagel J. K. Lipopolysaccharide structure determines ionic and hydrophobic binding of a cationic antimicrobial neutrophil granule protein. Infect Immun. 1988 Jun;56(6):1589–1592. doi: 10.1128/iai.56.6.1589-1592.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganz T., Selsted M. E., Szklarek D., Harwig S. S., Daher K., Bainton D. F., Lehrer R. I. Defensins. Natural peptide antibiotics of human neutrophils. J Clin Invest. 1985 Oct;76(4):1427–1435. doi: 10.1172/JCI112120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gennaro R., Dewald B., Horisberger U., Gubler H. U., Baggiolini M. A novel type of cytoplasmic granule in bovine neutrophils. J Cell Biol. 1983 Jun;96(6):1651–1661. doi: 10.1083/jcb.96.6.1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gennaro R., Skerlavaj B., Romeo D. Purification, composition, and activity of two bactenecins, antibacterial peptides of bovine neutrophils. Infect Immun. 1989 Oct;57(10):3142–3146. doi: 10.1128/iai.57.10.3142-3146.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray P. W., Flaggs G., Leong S. R., Gumina R. J., Weiss J., Ooi C. E., Elsbach P. Cloning of the cDNA of a human neutrophil bactericidal protein. Structural and functional correlations. J Biol Chem. 1989 Jun 5;264(16):9505–9509. [PubMed] [Google Scholar]
- Holak T. A., Engström A., Kraulis P. J., Lindeberg G., Bennich H., Jones T. A., Gronenborn A. M., Clore G. M. The solution conformation of the antibacterial peptide cecropin A: a nuclear magnetic resonance and dynamical simulated annealing study. Biochemistry. 1988 Oct 4;27(20):7620–7629. doi: 10.1021/bi00420a008. [DOI] [PubMed] [Google Scholar]
- Lehrer R. I., Barton A., Daher K. A., Harwig S. S., Ganz T., Selsted M. E. Interaction of human defensins with Escherichia coli. Mechanism of bactericidal activity. J Clin Invest. 1989 Aug;84(2):553–561. doi: 10.1172/JCI114198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugtenberg B., Van Alphen L. Molecular architecture and functioning of the outer membrane of Escherichia coli and other gram-negative bacteria. Biochim Biophys Acta. 1983 Mar 21;737(1):51–115. doi: 10.1016/0304-4157(83)90014-x. [DOI] [PubMed] [Google Scholar]
- Marion D., Zasloff M., Bax A. A two-dimensional NMR study of the antimicrobial peptide magainin 2. FEBS Lett. 1988 Jan 18;227(1):21–26. doi: 10.1016/0014-5793(88)81405-4. [DOI] [PubMed] [Google Scholar]
- Mazurier J., Spik G. Comparative study of the iron-binding properties of human transferrins. I. Complete and sequential iron saturation and desaturation of the lactotransferrin. Biochim Biophys Acta. 1980 May 7;629(2):399–408. doi: 10.1016/0304-4165(80)90112-9. [DOI] [PubMed] [Google Scholar]
- Nonnecke B. J., Smith K. L. Inhibition of mastitic bacteria by bovine milk apo-lactoferrin evaluated by in vitro microassay of bacterial growth. J Dairy Sci. 1984 Mar;67(3):606–613. doi: 10.3168/jds.S0022-0302(84)81345-4. [DOI] [PubMed] [Google Scholar]
- Okamura N., Spitznagel J. K. Outer membrane mutants of Salmonella typhimurium LT2 have lipopolysaccharide-dependent resistance to the bactericidal activity of anaerobic human neutrophils. Infect Immun. 1982 Jun;36(3):1086–1095. doi: 10.1128/iai.36.3.1086-1095.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penniall R., Zeya H. I. The effects of cationic proteins of rabbit polymorphonuclear leukocyte lysosomes on the respiratory activity of liver mitochondria. Biochem Biophys Res Commun. 1971 Oct 1;45(1):6–13. doi: 10.1016/0006-291x(71)90042-8. [DOI] [PubMed] [Google Scholar]
- Rainard P. Assessment of attachment, ingestion, and killing of Escherichia coli by bovine polymorphonuclear cells with combined micromethods. Vet Immunol Immunopathol. 1985 Nov;10(2-3):155–165. doi: 10.1016/0165-2427(85)90043-1. [DOI] [PubMed] [Google Scholar]
- Rice W. G., Ganz T., Kinkade J. M., Jr, Selsted M. E., Lehrer R. I., Parmley R. T. Defensin-rich dense granules of human neutrophils. Blood. 1987 Sep;70(3):757–765. [PubMed] [Google Scholar]
- Romeo D., Skerlavaj B., Bolognesi M., Gennaro R. Structure and bactericidal activity of an antibiotic dodecapeptide purified from bovine neutrophils. J Biol Chem. 1988 Jul 15;263(20):9573–9575. [PubMed] [Google Scholar]
- Segal A. W., Geisow M., Garcia R., Harper A., Miller R. The respiratory burst of phagocytic cells is associated with a rise in vacuolar pH. Nature. 1981 Apr 2;290(5805):406–409. doi: 10.1038/290406a0. [DOI] [PubMed] [Google Scholar]
- Selsted M. E., Harwig S. S. Purification, primary structure, and antimicrobial activities of a guinea pig neutrophil defensin. Infect Immun. 1987 Sep;55(9):2281–2286. doi: 10.1128/iai.55.9.2281-2286.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shafer W. M., Martin L. E., Spitznagel J. K. Late intraphagosomal hydrogen ion concentration favors the in vitro antimicrobial capacity of a 37-kilodalton cationic granule protein of human neutrophil granulocytes. Infect Immun. 1986 Sep;53(3):651–655. doi: 10.1128/iai.53.3.651-655.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorne K. J., Oliver R. C., Barrett A. J. Lysis and killing of bacteria by lysosomal proteinases. Infect Immun. 1976 Aug;14(2):555–563. doi: 10.1128/iai.14.2.555-563.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tobias P. S., Mathison J. C., Ulevitch R. J. A family of lipopolysaccharide binding proteins involved in responses to gram-negative sepsis. J Biol Chem. 1988 Sep 25;263(27):13479–13481. [PubMed] [Google Scholar]
- Viljanen P., Koski P., Vaara M. Effect of small cationic leukocyte peptides (defensins) on the permeability barrier of the outer membrane. Infect Immun. 1988 Sep;56(9):2324–2329. doi: 10.1128/iai.56.9.2324-2329.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss J., Hutzler M., Kao L. Environmental modulation of lipopolysaccharide chain length alters the sensitivity of Escherichia coli to the neutrophil bactericidal/permeability-increasing protein. Infect Immun. 1986 Feb;51(2):594–599. doi: 10.1128/iai.51.2.594-599.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss J., Olsson I. Cellular and subcellular localization of the bactericidal/permeability-increasing protein of neutrophils. Blood. 1987 Feb;69(2):652–659. [PubMed] [Google Scholar]
- Weiss J., Victor M., Stendhal O., Elsbach P. Killing of gram-negative bacteria by polymorphonuclear leukocytes: role of an O2-independent bactericidal system. J Clin Invest. 1982 Apr;69(4):959–970. doi: 10.1172/JCI110535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zerial A., Skerlavaj B., Gennaro R., Romeo D. Inactivation of herpes simplex virus by protein components of bovine neutrophil granules. Antiviral Res. 1987 Jul;7(6):341–352. doi: 10.1016/0166-3542(87)90016-7. [DOI] [PubMed] [Google Scholar]
- Zimmermann W., Rosselet A. Function of the outer membrane of Escherichia coli as a permeability barrier to beta-lactam antibiotics. Antimicrob Agents Chemother. 1977 Sep;12(3):368–372. doi: 10.1128/aac.12.3.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- in't Veld G., Mannion B., Weiss J., Elsbach P. Effects of the bactericidal/permeability-increasing protein of polymorphonuclear leukocytes on isolated bacterial cytoplasmic membrane vesicles. Infect Immun. 1988 May;56(5):1203–1208. doi: 10.1128/iai.56.5.1203-1208.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]



