Abstract
BACKGROUND & AIMS
Current surveillance guidelines for Barrett’s esophagus (BE) recommend extensive biopsies to minimize sampling error. Biopsy practice patterns for BE surveillance in the community have not been well-described. We used a national community-based pathology database to analyze adherence to guidelines and to determine whether adherence was associated with dysplasia detection.
METHODS
We identified 10,958 cases of established BE in the Caris Diagnostics pathology database from January 2002–April 2007. Demographic, pathologic, and endoscopic data were recorded. Dysplasia was categorized as low grade, high grade, or adenocarcinoma. Adherence was defined as ≥4 esophageal biopsies per 2 cm BE or a ratio ≥2.0. Generalized estimating equation multivariable analysis was performed to assess factors associated with adherence, adjusted for clustering by individual gastroenterologist.
RESULTS
A total of 2245 BE surveillance cases were identified with linked endoscopy reports that recorded BE length and could be assessed for adherence. Adherence to guidelines was seen in 51.2% of cases. In multivariable analysis, longer segment BE was associated with significantly reduced adherence (3–5 cm, odds ratio [OR] 0.14, 95% confidence interval [CI] 0.10 – 0.19; 6 – 8 cm, OR 0.06, 95% CI 0.03– 0.09; ≥9 cm, OR 0.03, 95% CI 0.01– 0.07). Stratified by BE length, nonadherence was associated with significantly decreased dysplasia detection (summary OR 0.53, 95% CI 0.35– 0.82).
CONCLUSIONS
Adherence to BE biopsy guidelines in the community is low, and nonadherence is associated with significantly decreased dysplasia detection. Future studies should identify factors underlying nonadherence as well as mechanisms to increase adherence to guidelines to improve early detection of dysplasia.
The incidence of esophageal adenocarcinoma (EAC) has increased rapidly in the Western world during the past 3 decades.1,2 Barrett’s esophagus (BE), the presence of columnar mucosa with intestinal metaplasia in the esophagus, is associated with a 30- to 40-fold increased risk of EAC.3 The overall 5-year survival for patients with EAC in the United States is ~15%4 and depends heavily on the stage of diagnosis.5 Endoscopic surveillance is recommended for patients with established BE, with the goal of detecting high-grade dysplasia or early cancer before the development of advanced EAC.6
The presence of dysplasia within Barrett’s mucosa is often patchy,7–10 and thus esophageal biopsies are associated with significant sampling error. A biopsy protocol of random 4 quadrant biopsies every 2 cm results in increased detection of dysplasia.10–12 As part of its first guidelines for the surveillance of patients with BE, the American College of Gastroenterology (ACG) in 1998 recommended the use of this sampling method,13 also known as the Seattle biopsy protocol. This technique has remained the standard sampling method for endoscopic surveillance in patients with BE.
Endoscopic surveillance has been shown to result in the detection of adenocarcinoma at earlier stages as well as improved survival in patients with EAC who underwent endoscopic surveillance, as compared with those who did not.14–20 However, according to several survey studies, only 26%–77% of endoscopists adhere to the Seattle protocol in clinical practice.21–26 Therefore, we decided to use a national communitybased pathology database to analyze adherence to surveillance biopsy guidelines in patients with established BE, to assess for factors associated with adherence to guidelines, and to determine whether detection of dysplasia is associated with adherence to biopsy guidelines.
Methods
Study Design and Database
We identified patients who underwent surveillance endoscopy for previously diagnosed BE by using a database that had been maintained prospectively by Caris Diagnostics (Irving, TX). This database is derived from all patients referred to Caris Diagnostics, a provider of gastrointestinal pathology services for physicians from community-based freestanding endoscopy centers from 34 states throughout the United States.
We used WinSURGE anatomic pathology software (Computer Trust Corporation, Boston, MA) to perform all database queries. An initial free-text search of clinical indications was performed to find upper endoscopy cases between January 1, 2002, and April 30, 2007 in which the term Barrett was present. We then further refined that query by doing a free-text search for several terms that would indicate patients undergoing surveillance endoscopy for BE: “follow-up, history, established,” and “surveillance.” Subsequently, we manually reviewed the results of the refined query to include those upper endoscopy cases performed for surveillance and to exclude those cases without a clear prior diagnosis of BE (eg, rule out Barrett’s esophagus). All data were entered into Microsoft Office Excel 2003 (Microsoft Corporation, Redmond, WA), and MySQL (Sun Microsystems, Santa Clara, CA) was used to store the master database.
Demographic information for all patients is present on the pathology requisitions and was entered into the Caris database. Corresponding endoscopy reports, when available, were manually reviewed, and endoscopic data, including length of BE and presence and size of hiatal hernia, were recorded. Individual gastroenterologists and endoscopy centers were assigned unique identification numbers. All pathology and endoscopy data were deidentified before analysis.
This study was approved by the Columbia University Institutional Review Board.
Pathology Review
The number of tissue biopsies listed on the pathology reports reflected the number of biopsy pieces that were counted and recorded before fixation in paraffin. When the specific number of specimen fragments was not clearly recorded or when the term multiple was used, the archived case slides were obtained for manual review and counting.
The presence or absence of intestinal metaplasia and dysplasia was noted. The degree of dysplasia for a particular case was defined as the highest degree (low grade, high grade, or adenocarcinoma) present on any of the biopsies.
The diagnosis of dysplasia or carcinoma (low grade, high grade, EAC) required confirmation by at least 2 pathologists. When the diagnosis was indefinite for dysplasia, the case was classified in the low-grade dysplasia group.
Biopsies are interpreted by a group of gastrointestinal pathologists who share a common approach to biopsy evaluation. This relative uniformity is achieved via predetermined approach to specimen handling, diagnostic criteria, terminology, and diagnosis-specific guidelines. In an effort to reduce interobserver variability, Caris Diagnostics guidelines require that all diagnoses of low-grade dysplasia or greater in specimens from BE are confirmed by a second pathologist, and in case of disagreement, that the case is presented to all pathologists during the daily consensus conference. When this study was begun, Caris used 8 pathologists; Caris currently has 23 pathologists on staff at the Irving location (where the cases were derived from).
Assessment of Adherence to Biopsy Guidelines
The 1998, 2002, and 2008 ACG Guidelines for BE Surveillance6,13,27 all recommend 4 quadrant biopsies taken every 2 cm throughout the length of BE. We therefore calculated a ratio for each case by using the following formula: (Number of esophageal biopsies) ÷ (Length of BE in cm). If the ratio was <2.0, then the case was defined as nonadherent, and if the ratio was ≥2.0, then the case was defined as adherent.
Statistical Methods and Analysis
We used Fisher exact test to analyze categorical variables. Student t tests were used to compare continuous variables with normal distributions; Wilcoxon rank sum tests were used to compare all other continuous variables. All tests were two-sided.
Generalized estimating equation (GEE) multivariable modeling was performed, adjusted for clustering by individual gastroenterologist. Presumably, adherence to guidelines is correlated with an individual gastroenterologist’s practice pattern; GEE modeling allows for multivariable analysis, taking into account the likelihood that the observed data are not independent. Statistical significance was defined as P < .05 or a confidence interval (CI) that did not cross 1.00. All analyses were performed by using Stata 9.2 (StataCorp, College Station, TX).
Results
Biopsies were received from 278,259 upper endoscopies between January 2002 and April 2007. There were 10,958 cases of BE surveillance performed in 9418 unique patients. Cases were submitted by 668 individual gastroenterologists from 214 community-based endoscopy centers in 34 states. The mean age was 62.3 years (standard deviation, 12.9), and 64.3% of the patients were male.
Endoscopy reports were available and reviewed for 4069 cases (37.1%) (Figure 1). Of these, the length of BE was recorded in 2448 (60.2%). The precise number of biopsies taken could not be determined for an additional 203 cases, and these were excluded from the analyses of adherence. Of these 2245 cases, 29 (1.3%) had a documented history of dysplasia (indefinite or greater). The median BE length was 3 cm (interquartile range, 2–5), and long segment BE (≥3 cm) was present in 53.3% of these cases. The median number of biopsies per case was 5 (interquartile range, 3– 8), and the median ratio of biopsies taken per centimeter of BE was 2.0 (interquartile range, 1.0 – 3.0). As compared with the excluded cases, subjects assessed for adherence were older (62.5 vs 61.7 years, P = .02) and had a higher proportion of men (69.2% vs 63.4%, P < .001).
Figure 1.
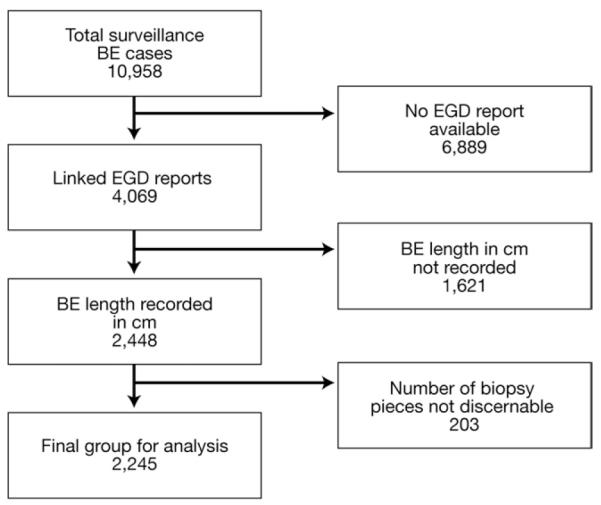
Flow chart of surveillance cases used for analysis of adherence to biopsy guidelines in patients with BE.
Pathology Results
Intestinal metaplasia was identified in 6633 cases (60.5%). Of these cases, low-grade dysplasia (including indefinite for dysplasia) was detected in 478 (7.2%), high-grade dysplasia in 80 (1.2%), and adenocarcinoma in 31 cases (0.5%). Patients with any dysplasia were more likely to be older (68.0 vs 61.7 years, P < .0001) and male (74.0% vs 64.5%, P < .0001). Of the cases analyzed for adherence, a significantly higher proportion of cases had intestinal metaplasia, as compared with those excluded from adherence analysis (70.6% vs 58.0%, P < .0001). Among patients with intestinal metaplasia, there was no difference in the proportion of patients with dysplasia between the cases analyzed for adherence to guidelines and those excluded from adherence analyses (P = .88) (Table 1). Both intestinal metaplasia and dysplasia were detected more frequently with longer segment BE (Table 2).
Table 1.
Presence of Dysplasia From Cases of Surveillance for BE in Which Intestinal Metaplasia Was Detected, 2002–2007
| All (n = 6633) |
Not assessed for adherence (n = 5049) |
Assessed for adherence (n = 1584) |
|||||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | P valuea | |
| Dysplasia | 588 | 8.9 | 447 | 8.9 | 142 | 9.0 | .88 |
| Indefinite or low grade | 478 | 7.2 | 355 | 7.0 | 123 | 7.8 | .34 |
| High grade | 80 | 1.2 | 68 | 1.4 | 12 | 0.8 | .06 |
| Adenocarcinoma | 31 | 0.5 | 24 | 0.5 | 7 | 0.4 | 1.00 |
For comparison of adherence analysis vs not in adherence analysis.
Table 2.
Detection of Intestinal Metaplasia and Dysplasia by Length of BE
| Intestinal metaplasia |
Dysplasia |
|||
|---|---|---|---|---|
| % |
P valuea |
% |
P valuea |
|
| BE length (cm) |
<.001 | <.001 | ||
| <3 | 53.6 (562/1049) | 2.8 (29/1049) | ||
| 3–5 | 79.5 (601/756) | 6.2 (47/756) | ||
| 6–8 | 96.2 (226/235) | 12.8 (30/235) | ||
| ≥9 | 95.1 (195/205) | 17.6 (36/205) | ||
Overall P value.
Impact of Endoscopy Documentation
Length of Barrett’s esophagus
Intestinal metaplasia was detected in 71.6% of cases in which length of BE (in centimeters) was recorded, as compared with 46.8% of cases in which length was not recorded (P < .001). Dysplasia was also detected in a significantly higher proportion of cases in which the length of BE was recorded (6.9% vs 3.1%, P < .001). After adjusting for patient age and sex, documentation of BE length was associated with a significantly increased odds of detecting both intestinal metaplasia (odds ratio [OR], 2.73; 95% CI, 2.38 – 3.11) and dysplasia (OR, 2.13; 95% CI, 1.54 –2.95).
We also compared those cases in which BE length was recorded as short segment (n = 157) with cases of BE < 3 cm. There were higher rates of detection of intestinal metaplasia (54.0% vs 42.7%, P = .008) and dysplasia (3.0% vs 0.6%, P = .11) in the cases with quantified BE length < 3 cm, as compared with those with BE length recorded as short segment.
Size of hiatal hernia
The hiatal hernia size was recorded in 42.5% of the endoscopy reports. Intestinal metaplasia was detected in a higher proportion of cases in which the size of hiatal hernia was recorded (66.8% vs 57.9%, P < .001). Dysplasia was detected in 7.1% of cases in which the size of hiatal hernia was recorded (vs 4.2% when not recorded, P = .0001). After adjusting for documentation of the length of BE, recording hiatal hernia size was not significantly associated with detection of intestinal metaplasia (OR, 1.05; 95% CI, 0.92–1.21), but it was still associated with dysplasia detection (OR, 1.41; 95% CI, 1.06 –1.87). Larger hiatal hernia size was significantly associated with the detection of intestinal metaplasia (P < .001) and dysplasia (P = .008).
Adherence to Biopsy Guidelines
A total of 1149 cases (51.2%) were adherent to biopsy guidelines. Adherence to biopsy guidelines was inversely and significantly associated with increased length of BE (P for trend < .001) (Figure 2). There was a significant trend toward increased adherence over time (P for trend = .03) (Figure 3), but there was no change in detection of dysplasia over time (P for trend = .38). When stratified by length of BE and excluding patients with a history of dysplasia, nonadherence was associated with significantly decreased odds of detection of dysplasia (Mantel-Haenszel summary OR, 0.53; 95% CI, 0.35– 0.82) (Figure 4).
Figure 2.

Adherence to the Seattle biopsy protocol by length of BE. Trend P value < .001.
Figure 3.

Adherence to the Seattle biopsy protocol by year of procedure. P value for trend = .03.
Figure 4.

Detection of dysplasia (low grade, high grade, or EAC) in cases adherent and nonadherent to the Seattle biopsy protocol, excluding patients with a history of dysplasia. Mantel-Haenszel summary OR for the association between nonadherence and dysplasia detection, stratified by BE length, equals 0.53; 95% CI, 0.35–0.82.
GEE multivariable modeling was performed, adjusted for clustering by individual gastroenterologist. Increased lengths of BE were strongly associated with reduced odds of adherence to biopsy guidelines (Table 3). Male sex was associated with significantly increased odds of adherence to guidelines, but patient age was not associated with adherence.
Table 3.
GEE Multivariable Model of Factors Associated With Adherence to Seattle Protocol Biopsy Guidelines, Adjusted for Clustering by Individual Gastroenterologist and Region
| OR | 95% CI | |
|---|---|---|
| Sex | ||
| Female | 1.00 | Referent |
| Male | 1.36 | 1.02–1.81 |
| Age (y) | ||
| <50 | 1.00 | Referent |
| 50–59 | 0.98 | 0.72–1.34 |
| 60–69 | 0.85 | 0.65–1.10 |
| ≥70 | 0.87 | 0.66–1.14 |
| BE length (cm) | ||
| <3 | 1.00 | Referent |
| 3–5 | 0.14 | 0.10–0.19 |
| 6–8 | 0.06 | 0.03–0.09 |
| ≥9 | 0.03 | 0.01–0.07 |
| Year of procedure | ||
| 2002 | 1.00 | Referent |
| 2003 | 0.47 | 0.25–0.87 |
| 2004 | 0.56 | 0.34–0.92 |
| 2005 | 0.57 | 0.34–0.96 |
| 2006 | 0.67 | 0.41–1.09 |
| 2007 | 0.80 | 0.44–1.46 |
Discussion
In this study of a large national community-based pathology database of nearly 11,000 patients with established BE, we found that (1) adherence to the recommended BE surveillance biopsy guidelines in the community is poor, with endoscopists following guidelines only half of the time, and (2) this failure to adhere to the guidelines is associated with reduced detection of dysplasia. These results in and of themselves are not proof that nonadherence to guidelines has an adverse impact on patient survival and overall outcomes in BE. However, the findings of this study do support the notion that adherence to these guidelines might result in earlier stage diagnosis of esophageal cancer and potentially improved survival.14–20
Prior survey studies have consistently highlighted suboptimal BE surveillance in clinical practice. In 3 survey studies in Europe, only 41%–58% of endoscopists responded that they took 4 biopsies every 2 cm in patients with BE.23–25 Mandal et al24 reported that 62% of responding gastroenterologists in the United Kingdom believed that it was not appropriate to perform endoscopic surveillance for short segment BE, and an additional 24% believed that endoscopic surveillance as a whole was not “worthwhile.” A survey study from the US reported 77% adherence to the Seattle biopsy protocol, although this figure might be an overestimate as a result of a relatively low response rate (45%) compared with the European studies.26 Although our study did not examine adherence to guidelines regarding time intervals in BE surveillance endoscopy, it is interesting to point out that adherence to appropriate BE surveillance time intervals has been reported to be much higher (84%–98%).25,26
We found a strong inverse association between length of BE and adherence to biopsy guidelines. Although the risk of EAC is proportional to the length of BE,28,29 an endoscopy with Seattle protocol biopsies for a patient with long segment BE can be time-consuming. It seems fair to assert that strict adherence to biopsy protocol increases the procedure time per upper endoscopy.
Another factor that might impact adherence to biopsy guidelines is the physician’s perception of an individual patient’s risk of neoplastic progression. Although epidemiologic associations with the presence of both BE and EAC have been relatively well-described,3,30 much less is known with regard to risk factors for neoplastic progression in patients with BE. In the present study, male sex was associated with increased odds of adherence to guidelines, possibly as a result of endoscopists’ perceptions of increased preventive benefit for male patients. Patient race and ethnicity, potentially associated with adherence to guidelines, were not captured in this study.
In 2006, the American Society for Gastrointestinal Endoscopy/ACG Taskforce on Quality in Endoscopy listed documentation of the length of BE as a quality indicator.31 The results of our study showed that documentation of length of BE was associated with increased detection of dysplasia, effectively validating the Taskforce’s recommendation. Endoscopists who record BE length might be more conscientious about performing BE surveillance per the recommended guidelines and might have stronger convictions with regard to the efficacy of endoscopic surveillance. Given the current lack of standard quality of care indicators for upper endoscopy in patients with BE, perhaps, at a minimum, Seattle protocol biopsies and documentation of the length of BE in centimeters should be included in future measures of quality.
The present study does have certain limitations. We had limited information on the presence of nodules or other focal lesions in the esophagus. Endoscopists might target biopsies of these areas, taking fewer biopsies overall, with the idea that this strategy will have the highest diagnostic yield. With the assumption that focal lesions were most likely to be present in patients with high-grade dysplasia or cancer, we performed repeat analyses excluding these cases and found no qualitative changes in the results (data not shown). An additional limitation to consider is the fact that we relied on the endoscopists’ discretion regarding the diagnosis of previously established BE.
The dysplasia end point consisted of the detection of low-grade dysplasia (including indefinite), high-grade dysplasia, or adenocarcinoma. At least 2 pathologists confirmed the diagnosis of all cases with low-grade dysplasia or greater. However, even among pathologists experienced in BE, there is poor interobserver and intraobserver agreement in the diagnosis of low-grade dysplasia.32 Other confounding variables potentially related to the detection of dysplasia and adenocarcinoma, such as obesity,33–35 smoking,36 and Helicobacter pylori,37 were not captured in our analyses.
There has been a large body of literature during the past several years investigating the potential of enhanced imaging techniques, such as chromoendoscopy and narrow band imaging, to improve the diagnostic yield of surveillance biopsies in BE.38 It is unknown whether the use of these techniques among community gastroenterologists could have impacted the results with regard both to adherence to biopsy guidelines and to detection of dysplasia.
The endoscopy reports were available for review in only 37% of the cases. Inclusion of a greater number of endoscopy reports in our analyses would have certainly strengthened the findings and subsequent conclusions. Although the dataset used in the present study contained cases from community-based gastroenterologists in 34 states, the average number of BE cases per gastroenterologist is low, potentially limiting the generalizability of the results.
In our analysis of patients with a prior clinical diagnosis of BE, intestinal metaplasia was identified in only 61% of cases. This figure is consistent with prior published data on detection of intestinal metaplasia in patients with BE.39 We suspect that a combination of undersampling plus the patchy presence of intestinal metaplasia within columnar mucosa explains this finding. However, we cannot rule out the possibility that some of these patients might have been initially misdiagnosed with BE.
The assessment of adherence was based on a ratio generated from variables in the dataset. The number of biopsies was determined by counting the tissue fragments in each bottle. The result of biopsy fragmentation would be an increase in the biopsy:BE length ratio and misclassification of some cases as adherent. However, a ratio < 2.0, regardless of fragmentation or reasons for or targets of biopsies, is less than indicated by the Seattle biopsy protocol. As such, the nonadherent group should be appropriately classified. We believe that it is likely that only a small proportion of the adherent group is misclassified, although this is difficult to prove or disprove on the basis of the available data.
More than half of the BE patients in this dataset had long segment BE (≥3 cm). Previous studies have reported that the proportion of long segment BE among prevalent cases is only 8%–21%.40–42 In all of these studies, there was a standard biopsy protocol for patients with suspected BE. In our communitybased study, such a protocol was not adhered to uniformly. Patients with shorter segments of BE were possibly less likely to have intestinal metaplasia successfully detected, and therefore a lower proportion of true short segment BE (compared with long segment) would be captured in our cohort. In addition, we suspect that BE length is more likely measured and recorded in patients with long segment BE. Both of these factors could explain the higher proportion of long segment BE observed in the present study compared with prior studies.
Given the high prevalence of nonadherence to recommended biopsy guidelines and the associated decreased detection of dysplasia, steps should be taken to identify factors responsible for nonadherence. There is a potential financial disincentive to adhere to guidelines in long segment BE, a high-risk population for progression to EAC. Endoscopists’ attitudes with regard to the efficacy of endoscopic surveillance, particularly in specific patient subpopulations, might also impact the decision to adhere to biopsy guidelines. Several studies have shown associations between physician characteristics, clinical practice patterns, and patient outcomes.43–45
In our analysis of a national community-based pathology database, adherence to endoscopic biopsy guidelines for BE surveillance was approximately 50%, and this nonadherence was associated with reduced detection of dysplasia. The presence of long segment BE was strongly associated with nonadherence to biopsy guidelines. In addition, although it seems fair to acknowledge that strict adherence to biopsy protocol might increase procedure time, one must also keep in mind that the application of developing endoscopic imaging techniques also might increase procedure time, in addition to the cost of the new technology. It seems reasonable to conclude that our study should encourage endoscopists to follow established biopsy practice guidelines, because this might have an immediate impact on improving our ability to identify early dysplasia in BE. Future studies should be aimed at identifying factors underlying nonadherence as well as mechanisms to improve adherence to BE biopsy guidelines in the community.
Supplementary Material
Acknowledgments
The authors acknowledge the following gastroenterology centers for their participation in the study: Ambulatory Endoscopy of Dallas, Dallas, TX; Arapahoe Gastroenterology, Littleton, CO; Arizona Digestive, Glendale, AZ; Atherton Endoscopy Center, Atherton, CA; Bergen Gastroenterology, Emerson, NJ; Central Maine Endoscopy, Waterville, ME; Charlotte Endoscopy, Port Charlotte, FL; Danbury Surgery Center, Danbury, CT; East Bay Endosurgery Inc, Oakland, CA; Fairfield Endoscopy Center, Fairfield, OH; Grossmont Surgery Center, La Mesa, CA; Gastroenterology Specialty, Inc, Tulsa, OK; Intercoastal Medical Group, Sarasota, FL; Midwest Gastro Center, Lee’s Summit, MO; Mountain West Endoscopy Center, Salt Lake City, UT; Southern Endoscopy Suite, Lawrenceville, GA; Triangle Endoscopy, Durham, NC; Valdosta Endoscopy Center, Valdosta, GA; and West Coast Endoscopy, Clearwater, FL.
The authors thank Mike Elam and Brittany Dodd at Caris Diagnostics, Irving, TX, for all their time and effort in assembling the dataset for this study.
Funding Dr Abrams is supported in part by a K07 award from the National Cancer Institute (CA132892). Dr Neugut is supported in part by a grant from the American Cancer Society (RSGT-01-024-04-CPHPS).
Abbreviations used in this article
- ACG
American College of Gastroenterology
- BE
Barrett’s esophagus
- CI
confidence interval
- EAC
esophageal adenocarcinoma
- GEE
generalized estimating equation
- OR
odds ratio
Footnotes
This article has an accompanying continuing medical education activity on page 710. Learning Objectives–After completing this CME activity, the learner should be able to understand further the epidemiology of Barrett’s esophagus and adenocarcinoma, and the impact of adherence to biopsy guidelines for Barrett’s esophagus in a community setting.
Supplementary Data Note: To access the supplementary material accompanying this article, visit the online version of Clinical Gastroenterology and Hepatology at www.cghjournal.org, and atdoi:10.1016/j.cgh.2008.12.027.
Conflicts of interest The authors disclose the following: Dr Kapel is a consultant for Caris Diagnostics. Drs Lindberg, Saboorian, and Genta are pathologists for Caris Diagnostics. The remaining authors disclose no conflicts.
References
- 1.Kubo A, Corley DA. Marked regional variation in adenocarcinomas of the esophagus and the gastric cardia in the United States. Cancer. 2002;95:2096–2102. doi: 10.1002/cncr.10940. [DOI] [PubMed] [Google Scholar]
- 2.Pohl H, Welch HG. The role of overdiagnosis and reclassification in the marked increase of esophageal adenocarcinoma incidence. J Natl Cancer Inst. 2005;97:142–146. doi: 10.1093/jnci/dji024. [DOI] [PubMed] [Google Scholar]
- 3.Shaheen NJ. Advances in Barrett’s esophagus and esophageal adenocarcinoma. Gastroenterology. 2005;128:1554–1566. doi: 10.1053/j.gastro.2005.03.032. [DOI] [PubMed] [Google Scholar]
- 4.Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2007. CA Cancer J Clin. 2007;57:43–66. doi: 10.3322/canjclin.57.1.43. [DOI] [PubMed] [Google Scholar]
- 5.Ries LAG, Melbert D, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2005. Volume 2008. National Cancer Institute; Bethesda, MD: [Accessed: May 27, 2008]. 2008. Available at http://seer.cancer.gov/statfacts/html/esoph.html?statfacts_page=esoph.html&x=14&y=11. [Google Scholar]
- 6.Wang KK, Sampliner RE. Updated guidelines 2008 for the diagnosis, surveillance and therapy of Barrett’s esophagus. Am J Gastroenterol. 2008;103:788–797. doi: 10.1111/j.1572-0241.2008.01835.x. [DOI] [PubMed] [Google Scholar]
- 7.McArdle JE, Lewin KJ, Randall G, et al. Distribution of dysplasias and early invasive carcinoma in Barrett’s esophagus. Hum Pathol. 1992;23:479–482. doi: 10.1016/0046-8177(92)90123-k. [DOI] [PubMed] [Google Scholar]
- 8.Jego M, Volant A, Faycal J, et al. Prevalence and topography of intestinal metaplasia in columnar lined esophagus. Gastroenterol Clin Biol. 2007;31:601–606. doi: 10.1016/s0399-8320(07)89437-1. [DOI] [PubMed] [Google Scholar]
- 9.Cameron AJ, Carpenter HA. Barrett’s esophagus, high-grade dysplasia, and early adenocarcinoma: a pathological study. Am J Gastroenterol. 1997;92:586–591. [PubMed] [Google Scholar]
- 10.Reid BJ, Weinstein WM, Lewin KJ, et al. Endoscopic biopsy can detect high-grade dysplasia or early adenocarcinoma in Barrett’s esophagus without grossly recognizable neoplastic lesions. Gastroenterology. 1988;94:81–90. doi: 10.1016/0016-5085(88)90613-0. [DOI] [PubMed] [Google Scholar]
- 11.Peters FP, Curvers WL, Rosmolen WD, et al. Surveillance history of endoscopically treated patients with early Barrett’s neoplasia: nonadherence to the Seattle biopsy protocol leads to sampling error. Dis Esophagus. 2008;21:475–479. doi: 10.1111/j.1442-2050.2008.00813.x. [DOI] [PubMed] [Google Scholar]
- 12.Abela JE, Going JJ, Mackenzie JF, et al. Systematic four-quadrant biopsy detects Barrett’s dysplasia in more patients than nonsystematic biopsy. Am J Gastroenterol. 2008;103:850–855. doi: 10.1111/j.1572-0241.2007.01746.x. [DOI] [PubMed] [Google Scholar]
- 13.Sampliner RE. Practice guidelines on the diagnosis, surveillance, and therapy of Barrett’s esophagus: the Practice Parameters Committee of the American College of Gastroenterology. Am J Gastroenterol. 1998;93:1028–1032. doi: 10.1111/j.1572-0241.1998.00362.x. [DOI] [PubMed] [Google Scholar]
- 14.Streitz JM, Jr, Andrews CW, Jr, Ellis FH., Jr Endoscopic surveillance of Barrett’s esophagus: does it help? J Thorac Cardiovasc Surg. 1993;105:383–388. [PubMed] [Google Scholar]
- 15.Peters JH, Clark GW, Ireland AP, et al. Outcome of adenocarcinoma arising in Barrett’s esophagus in endoscopically surveyed and nonsurveyed patients. J Thorac Cardiovasc Surg. 1994;108:813–822. [PubMed] [Google Scholar]
- 16.van Sandick JW, van Lanschot JJ, Kuiken BW, et al. Impact of endoscopic biopsy surveillance of Barrett’s oesophagus on pathological stage and clinical outcome of Barrett’s carcinoma. Gut. 1998;43:216–222. doi: 10.1136/gut.43.2.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Incarbone R, Bonavina L, Saino G, et al. Outcome of esophageal adenocarcinoma detected during endoscopic biopsy surveillance for Barrett’s esophagus. Surg Endosc. 2002;16:263–266. doi: 10.1007/s00464-001-8161-3. [DOI] [PubMed] [Google Scholar]
- 18.Ferguson MK, Durkin A. Long-term survival after esophagectomy for Barrett’s adenocarcinoma in endoscopically surveyed and nonsurveyed patients. J Gastrointest Surg. 2002;6:29–36. doi: 10.1016/s1091-255x(01)00052-x. [DOI] [PubMed] [Google Scholar]
- 19.Corley DA, Levin TR, Habel LA, et al. Surveillance and survival in Barrett’s adenocarcinomas: a population-based study. Gastroenterology. 2002;122:633–640. doi: 10.1053/gast.2002.31879. [DOI] [PubMed] [Google Scholar]
- 20.Fountoulakis A, Zafirellis KD, Dolan K, et al. Effect of surveillance of Barrett’s oesophagus on the clinical outcome of oesophageal cancer. Br J Surg. 2004;91:997–1003. doi: 10.1002/bjs.4591. [DOI] [PubMed] [Google Scholar]
- 21.Ofman JJ, Shaheen NJ, Desai AA, et al. The quality of care in Barrett’s esophagus: endoscopist and pathologist practices. Am J Gastroenterol. 2001;96:876–881. doi: 10.1111/j.1572-0241.2001.03637.x. [DOI] [PubMed] [Google Scholar]
- 22.Das D, Ishaq S, Harrison R, et al. Management of Barrett’s esophagus in the UK: overtreated and underbiopsied but improved by the introduction of a national randomized trial. Am J Gastroenterol. 2008;103:1079–1089. doi: 10.1111/j.1572-0241.2008.01790.x. [DOI] [PubMed] [Google Scholar]
- 23.Amamra N, Touzet S, Colin C, et al. Current practice compared with the international guidelines: endoscopic surveillance of Barrett’s esophagus. J Eval Clin Pract. 2007;13:789–794. doi: 10.1111/j.1365-2753.2006.00754.x. [DOI] [PubMed] [Google Scholar]
- 24.Mandal A, Playford RJ, Wicks AC. Current practice in surveillance strategy for patients with Barrett’s oesophagus in the UK. Aliment Pharmacol Ther. 2003;17:1319–1324. doi: 10.1046/j.1365-2036.2003.01586.x. [DOI] [PubMed] [Google Scholar]
- 25.van Sandick JW, Bartelsman JF, van Lanschot JJ, et al. Surveillance of Barrett’s oesophagus: physicians’ practices and review of current guidelines. Eur J Gastroenterol Hepatol. 2000;12:111–117. [PubMed] [Google Scholar]
- 26.Falk GW, Ours TM, Richter JE. Practice patterns for surveillance of Barrett’s esophagus in the United States. Gastrointest Endosc. 2000;52:197–203. doi: 10.1067/mge.2000.107728. [DOI] [PubMed] [Google Scholar]
- 27.Sampliner RE. Updated guidelines for the diagnosis, surveillance, and therapy of Barrett’s esophagus. Am J Gastroenterol. 2002;97:1888–1895. doi: 10.1111/j.1572-0241.2002.05910.x. [DOI] [PubMed] [Google Scholar]
- 28.Weston AP, Badr AS, Hassanein RS. Prospective multivariate analysis of clinical, endoscopic, and histological factors predictive of the development of Barrett’s multifocal high-grade dysplasia or adenocarcinoma. Am J Gastroenterol. 1999;94:3413–3419. doi: 10.1111/j.1572-0241.1999.01602.x. [DOI] [PubMed] [Google Scholar]
- 29.Weston AP, Krmpotich PT, Cherian R, et al. Prospective long-term endoscopic and histological follow-up of short segment Barrett’s esophagus: comparison with traditional long segment Barrett’s esophagus. Am J Gastroenterol. 1997;92:407–413. [PubMed] [Google Scholar]
- 30.Wong A, Fitzgerald RC. Epidemiologic risk factors for Barrett’s esophagus and associated adenocarcinoma. Clin Gastroenterol Hepatol. 2005;3:1–10. doi: 10.1016/s1542-3565(04)00602-0. [DOI] [PubMed] [Google Scholar]
- 31.Cohen J, Safdi MA, Deal SE, et al. Quality indicators for esophagogastroduodenoscopy. Am J Gastroenterol. 2006;101:886–891. doi: 10.1111/j.1572-0241.2006.00676.x. [DOI] [PubMed] [Google Scholar]
- 32.Montgomery E, Bronner MP, Goldblum JR, et al. Reproducibility of the diagnosis of dysplasia in Barrett esophagus: a reaffirmation. Hum Pathol. 2001;32:368–378. doi: 10.1053/hupa.2001.23510. [DOI] [PubMed] [Google Scholar]
- 33.Reeves GK, Pirie K, Beral V, et al. Cancer incidence and mortality in relation to body mass index in the Million Women Study: cohort study. BMJ. 2007;335:1134. doi: 10.1136/bmj.39367.495995.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Merry AH, Schouten LJ, Goldbohm RA, et al. Body mass index, height and risk of adenocarcinoma of the oesophagus and gastric cardia: a prospective cohort study. Gut. 2007;56:1503–1511. doi: 10.1136/gut.2006.116665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Corley DA, Kubo A, Levin TR, et al. Abdominal obesity and body mass index as risk factors for Barrett’s esophagus. Gastroenterology. 2007;133:34–41. doi: 10.1053/j.gastro.2007.04.046. quiz 311. [DOI] [PubMed] [Google Scholar]
- 36.Gammon MD, Schoenberg JB, Ahsan H, et al. Tobacco, alcohol, and socioeconomic status and adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst. 1997;89:1277–1284. doi: 10.1093/jnci/89.17.1277. [DOI] [PubMed] [Google Scholar]
- 37.Weston AP, Badr AS, Topalovski M, et al. Prospective evaluation of the prevalence of gastric Helicobacter pylori infection in patients with GERD, Barrett’s esophagus, Barrett’s dysplasia, and Barrett’s adenocarcinoma. Am J Gastroenterol. 2000;95:387–394. doi: 10.1111/j.1572-0241.2000.01758.x. [DOI] [PubMed] [Google Scholar]
- 38.Bergman JJ, Tytgat GN. New developments in the endoscopic surveillance of Barrett’s oesophagus. Gut. 2005;54(Suppl 1):i38–i42. doi: 10.1136/gut.2004.041590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harrison R, Perry I, Haddadin W, et al. Detection of intestinal metaplasia in Barrett’s esophagus: an observational comparator study suggests the need for a minimum of eight biopsies. Am J Gastroenterol. 2007;102:1154–1161. doi: 10.1111/j.1572-0241.2007.01230.x. [DOI] [PubMed] [Google Scholar]
- 40.Ward EM, Wolfsen HC, Achem SR, et al. Barrett’s esophagus is common in older men and women undergoing screening colonoscopy regardless of reflux symptoms. Am J Gastroenterol. 2006;101:12–17. doi: 10.1111/j.1572-0241.2006.00379.x. [DOI] [PubMed] [Google Scholar]
- 41.Hirota WK, Loughney TM, Lazas DJ, et al. Specialized intestinal metaplasia, dysplasia, and cancer of the esophagus and esophagogastric junction: prevalence and clinical data. Gastroenterology. 1999;116:277–285. doi: 10.1016/s0016-5085(99)70123-x. [DOI] [PubMed] [Google Scholar]
- 42.Rex DK, Cummings OW, Shaw M, et al. Screening for Barrett’s esophagus in colonoscopy patients with and without heartburn. Gastroenterology. 2003;125:1670–1677. doi: 10.1053/j.gastro.2003.09.030. [DOI] [PubMed] [Google Scholar]
- 43.Lee SJ, Joffe S, Artz AS, et al. Individual physician practice variation in hematopoietic cell transplantation. J Clin Oncol. 2008;26:2162–2170. doi: 10.1200/JCO.2007.15.0169. [DOI] [PubMed] [Google Scholar]
- 44.Abdel-Malek N, Chiarelli AM, Sloan M, et al. Influence of physician and patient characteristics on adherence to breast cancer screening recommendations. Eur J Cancer Prev. 2008;17:48–53. doi: 10.1097/CEJ.0b013e32809b4cef. [DOI] [PubMed] [Google Scholar]
- 45.Hershman DL, Buono D, McBride RB, et al. Surgeon characteristics and receipt of adjuvant radiotherapy in women with breast cancer. J Natl Cancer Inst. 2008;100:199–206. doi: 10.1093/jnci/djm320. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


