Abstract
Calvatia lilacina (CL), Pleurotus ostreatus (PO) and Volvariella volvacea (VV) are widely distributed worldwide and commonly eaten as mushrooms. In this study, cell viabilities were evaluated for a human colorectal adenocarcinoma cell line (SW480 cells) and a human monocytic leukemia cell line (THP-1 cells). Apoptotic mechanisms induced by the protein extracts of PO and VV were evaluated for SW480 cells. The viabilities of THP-1 and SW480 cells decreased in a concentration-dependent manner after 24 h of treatment with the protein extracts of CL, PO or VV. Apoptosis analysis revealed that the percentage of SW480 cells in the SubG1 phase (a marker of apoptosis) was increased upon PO and VV protein-extract treatments, indicating that oligonucleosomal DNA fragmentation existed concomitantly with cellular death. The PO and VV protein extracts induced reactive oxygen species (ROS) production, glutathione (GSH) depletion and mitochondrial transmembrane potential (ΔΨm) loss in SW480 cells. Pretreatment with N-acetylcysteine, GSH or cyclosporine A partially prevented the apoptosis induced by PO protein extracts, but not that induced by VV extracts, in SW480 cells. The protein extracts of CL, PO and VV exhibited therapeutic efficacy against human colorectal adenocarcinoma cells and human monocytic leukemia cells. The PO protein extracts induced apoptosis in SW480 cells partially through ROS production, GSH depletion and mitochondrial dysfunction. Therefore, the protein extracts of these mushrooms could be considered an important source of new anti-cancer drugs.
1. Introduction
Calvatia lilacina (CL), Pleurotus ostreatus (PO) and Volvariella volvacea (VV) are three common medicinal or edible mushrooms. Among them, CL was widely used by CAM practitioners in China and Taiwan for treatment of traumatic bleeding, hemorrhagic vomiting, nasal bleeding, throat and nasopharyngeal pain, loss of voice and coughing. PO showed antimicrobial and antineoplastic activities [1]. Our previous study demonstrated that the protein extracts of CL induce apoptosis through glutathione (GSH) depletion in human colorectal cancer SW480 cells [2]. The extraction of PO suppresses the proliferation of breast and colon cancer cells via p53-dependent and p53-independent pathways [3]. The water-soluble polysaccharide of PO is capable of enhancing concanavalin A- or lipopolysaccharide-induced lymphocyte proliferation [4, 5] and induces apoptosis in human PC-3 androgen-independent cancer cells as well as human HT-29 colon cancer cells [6, 7]. DNA isolated from the fruit body of PO augmented NK cytotoxic activity in vitro and significantly increased the life span of mice with solid Ehrlich carcinoma [8]. Despite the observation of anti-cancer effects for PO, the effects of the protein extract of PO on cancer are unclear thus far; moreover, there have been no anti-cancer studies on VV protein extracts. The anti-cancer effects of the protein extracts of PO and VV are worth studying. Colorectal cancer and leukemia are two common cancers in human. The aim of this study was to evaluate the anti-cancer effects of three protein extracts from CL, PO and VV on human colorectal cancer SW480 cells and monocytic leukemia THP-1 cells. The relationship between apoptosis and intracellular oxidative stress in cancer cells treated with PO and VV protein extracts was also evaluated.
2. Methods
2.1. Experimental Material
CL was collected from the National Chiayi University, Chiayi, Taiwan, in May 2008. PO and VV were bought from Carrefour Supermarket, Chiayi, Taiwan, in June 2008.
2.2. Extraction and Isolation of Proteins
CL, PO and VV (400–450 g) were washed twice with distilled water and then homogenized in cold extraction buffer (50 mM KH2PO4, 150 mM NaCl and 1 mM EDTA, pH 7.3) at maximum speed in a Waring blender for 10 min. The water-soluble protein extract homogenate was filtered through two layers of surgical gauze, and the filtrate was centrifuged at 8900 g for 20 min. The supernatants were precipitated by addition of ammonium sulphate to 80% saturation, incubated at 4°C overnight, and then centrifuged at 8900 g for 30 min. The protein pellets were resuspended in a phosphate buffer solution (123 mM NaCl, 10.4 mM Na2HPO4 and 3.16 mM KH2PO4, pH 7.3) (weight/volume = 1 : 1) and centrifuged. The supernatants were dialyzed against a 50-fold volume of phosphate buffer solution three times at 4°C and clarified by centrifugation. The supernatants were filtered through 0.45 μm Millex filter units (Millipore) and concentrated using Amicon Ultra-4 (10 kDa cut-off, Millipore) tubes at 4000 g for 20 min. The 1 mL concentrate was sterilized using 0.22 μm Millex filter units (Millipore) in a laminar flow chamber; it was then freeze-dried to obtain a powder and stored at −80°C as stock. The Bio-Rad protein assay reagent (Bio-Rad Lab., Hercules, CA), 12% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and Coomassie Brilliant Blue staining were utilized for the detection of CL, PO and VV proteins.
2.3. Cell Lines and Reagents
The human colorectal cancer cell line SW480 and the monocytic leukemia cell line THP-1 were obtained from the Bioresource Collection and Research Center (Hsinchu, Taiwan, ROC). Dulbecco's modified Eagle's medium (DMEM), RPMI-1640 and foetal bovine serum (FBS) were obtained from Hyclone (South Logan, UT, USA). Chloromethylflourescein diacetate (CMF-DA) was acquired from Invitrogen Corporation (Carlsbad, CA, USA). Propidium iodide (PI), N-acetylcysteine (NAC), GSH, cyclosporine A (CyA), 2′,7′-dichlorodihydrofluorescein-diacetate (DCFH-DA), rhodamine 123, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), DNase-free RNase A and other chemicals were purchased from Sigma Chemical Co. (St. Louis, MO, USA).
2.4. Cell Culture and Treatment
SW480 and THP-1 cells were cultured in DMEM and RPMI-1640 medium, respectively, containing 10% FBS. The stock solutions of CL, PO and VV were dissolved in the phosphate buffer saline (PBS) (136.89 mM NaCl, 2.68 mM KCl, 10.14 mM Na2HPO4 and 1.76 mM KH2PO4, pH 7.4), and different concentrations (μg mL−1) were prepared in DMEM or RPMI-1640 media.
2.5. Cell Viability Assays
Cell viability was evaluated using the MTT assay. Yellow MTT was reduced to purple formazan by dehydrogenases in the mitochondria of living cells. A solubilization agent, such as dimethyl sulfoxide (DMSO) or SDS, was used to dissolve the insoluble purple formazan product into a colored solution. The absorbance of this colored solution was quantified at a certain wavelength (usually 570 nm) with a spectrophotometer. The effectiveness of the protein extracts in causing cell death was evaluated by comparing the amount of purple formazan produced by cells treated with the extracts to that produced by untreated cells. Cells (1 × 106) were cultured in 60 mm tissue-culture dishes for 24 h. The culture medium was replaced with a new medium and the cells were exposed to various concentrations of CL, PO or VV for 24 h. After the indicated treatments, cells were incubated for 2 h with 0.5 mg mL−1 of MTT reagent and then lysed with DMSO. Absorbance was measured at 570 nm in a microplate reader (Bio-Rad, Richmond, CA, USA).
2.6. Cell Cycle and DNA Damage Analysis
The cell cycle and DNA damage were evaluated with PI staining and flow cytometry. PI is a specific fluorescent dye that stains the double-stranded DNA. In methanol-fixed cells, the PI molecules translocate into the nucleus and bind to the double-stranded DNA. Using flow cytometry, PI was excited at 488 nm and emitted fluorescence at 580 ∼ 630 nm. The G1 and G2/M phases are represented by the diploid and the tetraploid regions, respectively, of the DNA distribution histograms. Cells in the S phase were found between the diploid and the tetraploid regions. The length of double-stranded DNA in cells with DNA damage was smaller than that of cells in the G1 phase. Fewer PI molecules were found in DNA-damaged cells as compared with the G1 phase; the fluorescent intensity of PI in DNA-damaged cells was weaker than that of cells in the G1 phase. The percentage of DNA-damaged cells was characterized as the percentage of cells in the SubG1 region of the DNA distribution histograms. Cells (1 × 106) were cultured in 60-mm tissue-culture dishes for 24 h. The culture medium was replaced by a new medium, and then cells were exposed or not to various concentrations of the protein extracts of PO and VV for 24 h. After treatment, adherent and floating cells were pooled, washed with PBS, fixed in PBS-methanol (1 : 2, volume/volume) solution, and then maintained at 4°C for at least 18 h. After one wash with PBS, the cell pellets were stained with a fluorescent probe solution containing PBS, 50 μg mL−1 PI and 50 μg mL−1 DNase-free RNase A for 30 min at room temperature in the dark. The DNA fluorescence of PI-stained cells was analyzed by excitation at 488 nm and monitored through a 630/22 nm band-pass filter using a Becton-Dickinson FACScan flow cytometer (Franklin Lakes, NJ, USA). A minimum of 10 000 cells was counted per sample, and the DNA histograms were evaluated further using Modfit software on a PC workstation to calculate the percentage of cells in various phases of the cell cycle.
2.7. Measurement of Intracellular Reactive Oxygen Species by Flow Cytometry
Production of intracellular Reactive oxygen species (ROS) was detected by flow cytometry using DCFH-DA. DCFH-DA is a ROS probe that undergoes intracellular deacetylation to DCFH by esterase in cytosol, followed by ROS-mediated oxidation to a DCF fluorescent species that is excited at 485 nm and emits fluorescence at 530 nm. DCFH-DA can be used to measure ROS generation in the cytoplasm and in cellular organelles such as the mitochondria. Fluorescence intensity is quantified using flow cytometry or a microplate spectrophotometer. After treatment, cells were treated with 20 μM DCFH-DA for 30 min in the dark, washed once with PBS, detached by trypsinization, collected by centrifugation and suspended in PBS. The intracellular ROS levels, as indicated by the fluorescence of dichlorofluorescein (DCF), were measured with a Becton-Dickinson FACScan flow cytometer.
2.8. Measurement of GSH Depletion
The intracellular GSH levels were evaluated using CMF-DA. After treatment, the cells were incubated with 25 μM CMF-DA for 20 min at 37°C in a CO2 incubator. Once CMF-DA is internalized into the cell, cytosolic esterase cleaves the acetates, and the chloromethyl group reacts with intracellular thiols, altering the probe into a cell-impermeable fluorescent dye-thioether adduct, CMF. The CMF fluorescence is directly associated with intracellular GSH level. After CMF-DA staining, the cells were washed with PBS, collected by centrifugation and then fluorescence was measured with a Becton-Dickinson FACSan flow cytometer.
2.9. Measurement of ΔΨm by Flow Cytometry
After treatment, the cells were trypsinized and then incubated with 5 μM rhodamine 123 for 30 min at 37°C in a CO2 incubator. Rhodamine 123, a lipophilic cation, is specifically and selectively internalized into the mitochondria of mammalian cells. The collection of rhodamine 123 is dependent on the proton gradient produced across mitochondrial membranes. The accumulation of rhodamine 123 within cells can thus be used as an indicator of mitochondrial membrane potential and mitochondrial function. Following the incubation step, ΔΨm, as indicated by the change in the fluorescence level of rhodamine 123, was analyzed using a Becton-Dickinson FACScan flow cytometer.
2.10. Statistical Analysis
Data are presented as means ± SD from at least three independent experiments and were analyzed using Student's t-test. P < .05 was considered as statistically significant [9].
3. Results
3.1. The Number of Major Proteins and Their Molecular Weights Were Evaluated in the Protein Extracts of CL, PO and VV
To detect the number of major proteins and their molecular weights in the protein extracts of CL, PO and VV, 12% SDS-PAGE gel electrophoresis and Coomassie Brilliant Blue staining were used. The profiles of protein extracts from CL, PO and VV used in this study are presented in Figure 1. Major proteins with various molecular weights around 70, 55, 45, 40, 28, 24, 20, 18 and 11 kDa were visualized after Coomassie Brilliant Blue staining in 36 and 48 μg of protein extracts of CL. Major proteins with various molecular weights around 175, 96, 95, 72, 68, 32, 23 and 11 kDa were visualized in 36 and 48 μg of protein extracts of VV. Major proteins with various molecular weights around 170, 130, 95, 72, 43, 42, 28, 20 and 11 kDa were visualized in 36 and 48 μg of protein extracts of PO.
Figure 1.
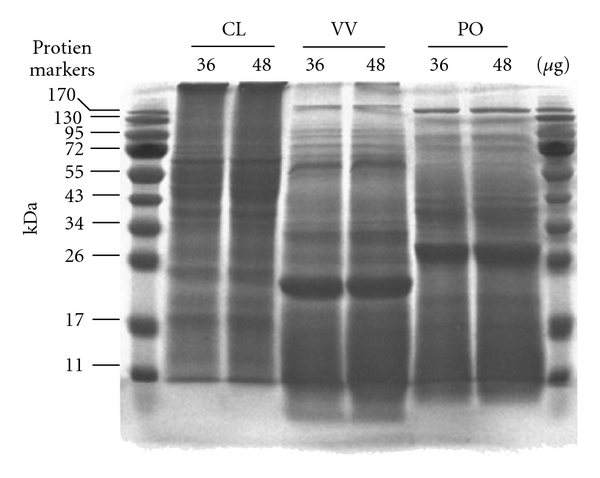
Protein extract analysis for CL, VV and PO. Concentrations of proteins in extracts of CL, VV and PO were quantified with Bio-Rad protein assay reagent. BSA was used as a standard for protein analysis. Protein extracts (36 and 48 μg) of CL, VV and PO were subjected to SDS-PAGE (12% (w/v) polyacrylamide). CL, VV and PO protein extracts were stained with Coomassie Brilliant Blue. The positions of molecular mass markers (in kilodaltons) are shown on the left and on the right.
3.2. CL, PO and VV Decreased the Viabilities of THP-1 and SW480 Cells
To evaluate the effects of protein extracts of CL, PO and VV on the cell viability of human monocytic leukemia THP-1 cells and human colorectal SW480 cells, a concentration of 500 μg mL−1 was used for the MTT assay. As indicated in Figure 2(a), the viability of THP-1 cells was 52%, 3% and 3% after 24 h of treatment with CL, PO and VV, respectively. As indicated in Figure 2(b), the viability of SW480 cells was 48%, 2% and 7% after 24 h of treatment with CL, PO and VV, respectively. These results suggest that the degree of cell viability decrease observed for PO and VV was larger than that for CL in THP-1 and SW480 cells. We further selected PO and VV for evaluation of the effects of lower concentrations on the cell viability of SW480 cells. As indicated in Figure 2(c), the cell viability decreased to 39% after a 24-h treatment with 10 and 25 μg mL−1 of PO protein extracts. The cell viability further decreased to 10 and 7% after treatment with 50 and 100 μg mL−1 of PO protein extracts, respectively. Similarly to the decrease in cell viability induced by the PO protein extracts, the protein extracts of VV also decreased the viability of SW480 cells. As shown in Figure 2(d), treatment with 10 μg mL−1 of VV protein extracts decreased cell viability by 20%; however, the decrease in cell viability was less than 10% after treatment with 25, 50 and 100 μg mL−1 of VV protein extracts.
Figure 2.
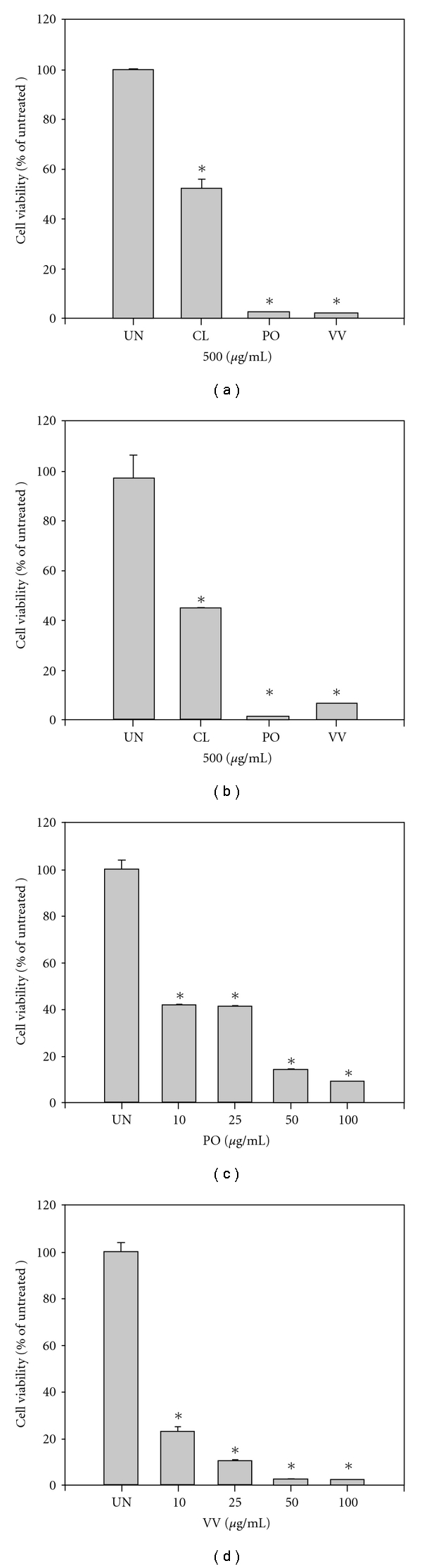
Cell viability analysis of CL, PO and VV protein extract-treated THP-1 and SW480 cells. (a) THP-1 and (b) SW480 cells were treated with CL, PO and VV protein extracts (500 μg mL−1) for 24 h. SW480 cells were treated with 0, 10, 25, 50 and 100 μg mL−1 of (c) PO and (d) VV protein extracts for 24 h. After treatment, cell viabilities were evaluated using the MTT assay. The values shown are the means ± SD (n = 5–8). *P < .05 (significant differences from the untreated group).
3.3. PO and VV Induced DNA Damage in SW480 Cancer Cells
The DNA damage (SubG1) induced by CL protein extracts in SW480 cells was reported in our previous study [2]. The DNA damage induced by PO and VV protein-extract treatments in SW480 cells has not been extensively studied. We therefore evaluated whether the protein extracts of PO and VV could induce apoptosis. SW480 cells were exposed to 100 μg mL−1 of PO and VV for 24 h, and the DNA damage of the cells was evaluated by flow cytometry. The total cells were separated into a damaged DNA group (cells in the SubG1 phase) and a normal cell cycle group (G1, S and G2/M phases). As shown in Figure 3, 59.0% untreated cells were in G1, 29.1% in S, 11.9% in G2/M and 1.9% in SubG1. The proportion of cells in SubG1 increased to 28.2% after treatment of SW480 cells with 100 μg mL−1 PO protein extracts for 24 h. Following treatment with the protein extracts of VV (100 μg mL−1) for 24 h, the proportion of cells in SubG1 markedly increased to 97.8% in SW480 cells.
Figure 3.
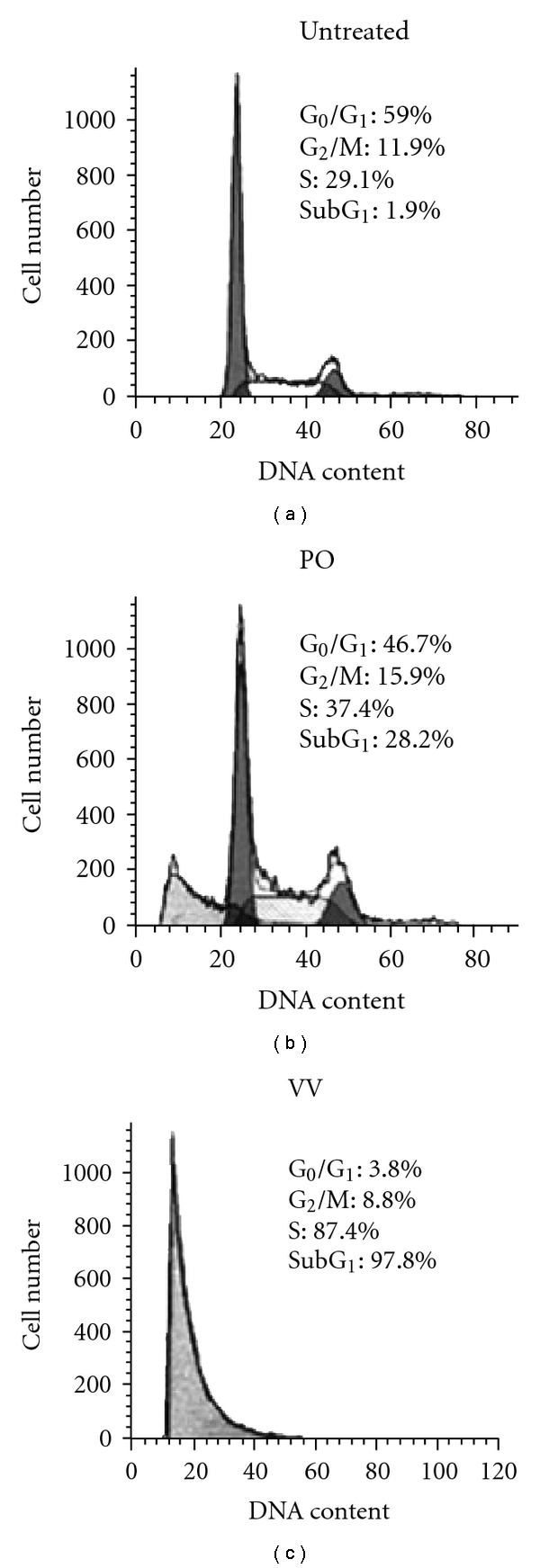
DNA damage analysis of PO and VV protein extract-treated SW480 cells. SW480 cells were treated with PO and VV protein extracts (100 μg mL−1) for 24 h. After treatment, cells were fixed and then stained with propidium iodide, followed by flow cytometric analysis. Data in each panel show the percentages of cells in SubG1 (DNA-damaged cells), G1, S and G2/M phases of the cell cycle. These experiments were performed at least three times and a representative experiment is presented.
3.4. PO and VV Induced ROS in SW480 Cells
ROS are known to be important mediators of apoptosis and are induced by various stimuli, including many drugs. For this reason, intracellular ROS production was measured using a DCF fluorescence assay following treatment with PO and VV protein extracts. As indicated in Figure 4, SW480 cells were exposed to PO and VV protein extracts at 100 μg mL−1 of for 24 h. The DCF fluorescence increased significantly from 128.47 ± 736 to 555.01 ± 9.6 and 311.61 ± 6.1 after treatment with PO and VV protein extracts, respectively.
Figure 4.
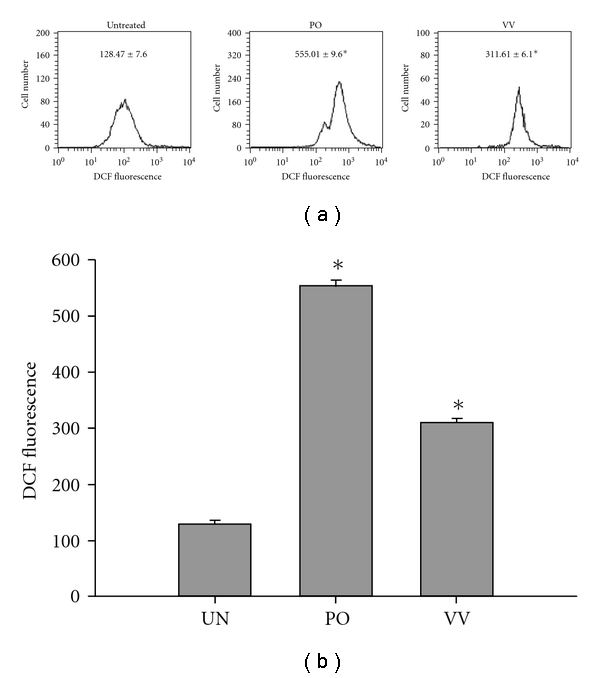
Intracellular ROS evaluation of PO and VV protein extract-treated SW480 cells. SW480 cells were exposed to PO and VV protein extracts (100 μg mL−1) for 24 h. Production of intracellular ROS was detected by flow cytometry using DCFH-DA. The intracellular fluorescence of DCF was measured with a Becton-Dickinson FACScan flow cytometer. Data in each panel represent the DCF fluorescence intensity within the cells. The values shown are the means ± SD (n = 5–8 of individual experiments). *P < .05 (significant differences from the untreated group).
3.5. PO and VV Induced GSH Depletion in SW480 Cells
GSH plays an important role in protection against oxidative stress-induced injury. Intracellular GSH depletion results in apoptosis. We therefore analyzed changes in the intracellular GSH level of SW480 cells after treatment with the protein extracts of PO and VV; we utilized flow cytometric analysis of CMF-DA fluorescence. SW480 cells were treated with protein extracts of PO and VV at 100 μg mL−1 for 24 h. As shown in Figure 5, the fractions of cells with intracellular GSH depletion significantly increased to 59.11 ± 4.3 and 99.58 ± 0.4% after treatment with the protein extracts of PO and VV, respectively, as compared with untreated cells (0.34 ± 0.2%).
Figure 5.
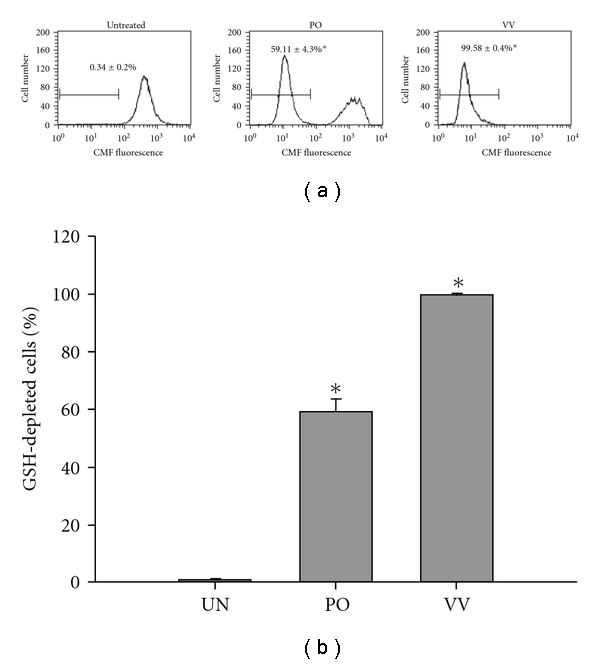
GSH depletion analysis of PO and VV protein extract-treated SW480 cells. SW480 cells were exposed to PO and VV protein extracts (100 μg mL−1) for 24 h. After treatment, the cells were incubated with 25 μM CMF-DA for 30 min in a 37°C CO2 incubator and fluorescence was then measured with a flow cytometer. Data in each panel represent the percentage of GSH-depleted cells with respect to the total cells. The values shown are means ± SD (n = 5–8). *P < .05 (significant differences from the untreated group).
3.6. PO and VV Disrupted ΔΨm in the SW480 Cells
Mitochondrial dysfunction usually triggers specific cellular signaling to induce apoptosis. Loss of ΔΨm is commonly used to measure mitochondrial dysfunction. To evaluate the effects of the protein extracts of PO and VV on ΔΨm, SW480 cells were exposed to the protein extracts of PO and VV at 100 μg mL−1 for 24 h. The ΔΨm was detected by staining of rhodamine 123, which specifically accumulated within the mitochondrial compartment in a ΔΨm-dependent manner. As indicated in Figure 6, rhodamine 123 fluorescence was significantly decreased to 153 ± 3 and 41 ± 2 after treatment with the protein extracts of PO and VV, respectively, as compared with untreated cells (514 ± 10).
Figure 6.
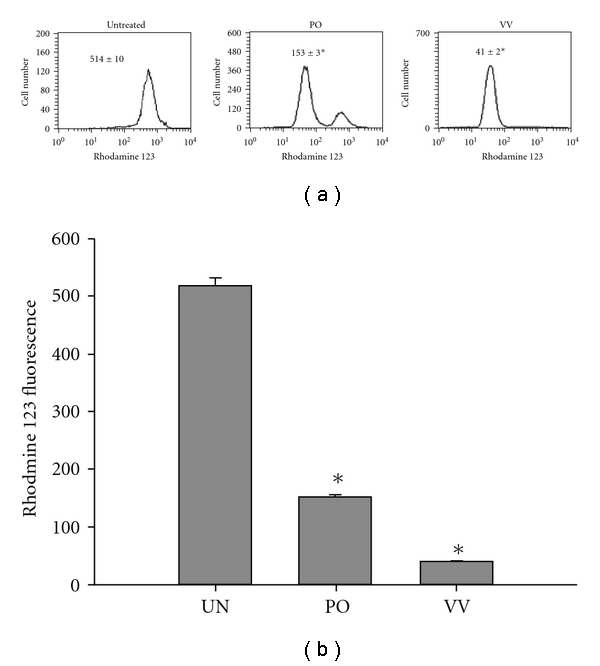
ΔΨm analysis of PO and VV protein extract-treated SW480 cells. SW480 cells were treated with PO and VV protein extracts (100 μg mL−1) for 24 h. After treatment, the culture medium was replaced with a new medium containing 5 μM rhodamine 123. Cultures were then incubated for 20 min in the dark. Values in each panel represent the rhodamine 123 fluorescence intensity within the cells. The values shown are means ± SD (n = 5–8). *P < .05 (significant differences from the untreated group).
3.7. Evaluation of the Critical Pathway of Apoptosis Induced by PO and VV
To evaluate the critical pathway of apoptosis induced by the protein extracts of PO and VV, an extract concentration of 10 μg mL−1 was used. As shown in Figure 7, 16.9% and 18.5% of cells were apoptotic (SubG1 phase) after treatment with the protein extracts of PO and VV for 24 h as compared with 2.3% and 2.9% of untreated cells, respectively. To evaluate the critical pathway, several agents were employed: (i) NAC, an intracellular GSH synthetic agent; (ii) GSH, were used to discover the GSH depletion pathway; and (iii) CyA, a mitochondrial permeability transition opening inhibitor, was used to determine the pathway involved in decreased ΔΨm. SW480 cells were treated with these agents for 1 h before the addition of the protein extracts of PO and VV. As shown in Figure 7(a), NAC and GSH inhibited PO protein extract-induced apoptosis; the fractions of apoptotic cells decreased to 6.6 and 8.2%, respectively. CyA displayed a moderate inhibitory effect; the fraction of apoptotic cells was 11.7%. Figure 7(b) shows that the apoptotic effect induced by the protein extracts of VV was not prevented by NAC, GSH or CyA; the fractions of apoptotic cells were 19.3, 20.8 and 20.0%, respectively.
Figure 7.
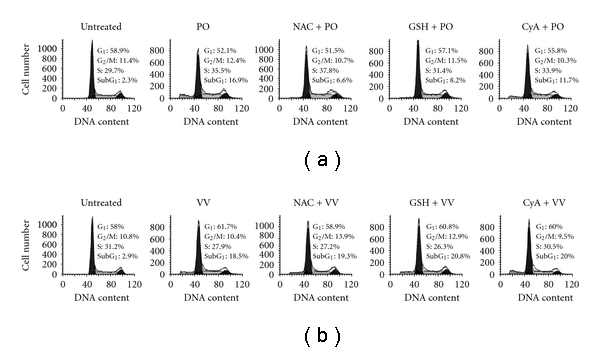
Evaluation of the critical mechanism in PO and VV protein extract-induced apoptosis. (a) The SW480 cells were treated with (a) PO and (b) VV protein extracts (0 (untreated) or 10 μg mL−1) for 24 h, pretreated with 10 mM NAC, 10 mM GSH or 5 μM CyA for 1 h, followed by (a) PO and (b) VV protein extracts (10 μg mL−1) treatment for 24 h, and then analyzed for DNA damage. Data in each panel show the percentages of cells in the SubG1 (DNA-damaged cells), G1, S and G2/M phases of cell cycle. These experiments were performed at least three times and a representative experiment is presented.
4. Discussion
Currently, the prevalent choices for treating human cancer are surgical resection, general chemotherapy, gene therapy and radiation therapy. These treatments are costly and almost always performed in the hospital. It is necessary to develop new therapeutic strategies, such as integrative and complementary medicine, for cancer treatment. One approach, as highlighted in this study, looks for effective adjuvant substances from edible or medical mushrooms that are capable of activating the cellular apoptotic response in cancer cells [10]. Many edible and culinary mushrooms have become important medicinal and biotechnological species. For example, Antrodia camphorata, a unique mushroom of Taiwan, has been used as a traditional medicine for the prevention of various health-related conditions. It can be considered an efficient alternative phytotherapeutic agent or a synergizer for the treatment of cancer [11]. 4,7-dimethoxy-5-methyl-l,3-benzodioxole isolated from the fruiting body of Antrodia camphorata inhibits anchorage-independent proliferation and G0/G1 cell-cycle regulation in human colorectal carcinoma cells [12]. Another study reported that the basidiomycete mushroom Agaricus blazei Murrill, a mushroom native to Brazil, is traditionally used to treat many common diseases, such as atherosclerosis, hepatitis, hyperlipidemia, diabetes, dermatitis and cancer [13]. Agaricus blazei extract, which increases apoptosis via ROS-dependent JNK activation and inhibition of constitutive NF-κB in leukemia THP-1 cells [14], has been reported to have anti-tumor and anti-mutagenic effects [15]. The water-extract of Chaga mushroom (Inonotus obliquus) has potent inhibitory effects on B16-F10 melanoma that include anti-proliferation, induction of differentiation and apoptosis [16]. It also induces G0/G1 arrest of the cell cycle and apoptosis in the human hepatoma Hep G2 cell line [17]. Several indian medicinal mushrooms, such as Ganoderma lucidum, Phellinus rimosus, Pleurotus florida and Pleurotus pulmonarius, have shown strong antioxidant and anti-cancer effects [18]. The results of the present study suggest that CL, PO and VV may be useful for integrative and complementary medicine by promoting apoptotic cell death in human colorectal cancer cells via induction of ROS production, GSH depletion and mitochondrial dysfunction.
In the present study, the protein extracts of CL, PO and VV were used to evaluate anti-cancer effects. The protein extracts of PO and VV induced marked apoptosis in SW480 cells (Figure 3). The apoptotic mechanism of the PO protein extract in SW480 cells was partially related to ROS production, GSH depletion and ΔΨm loss (Figure 7(a)). However, the apoptotic mechanism of the VV protein extract is not clear because NAC, GSH and CyA did not prevent VV protein extract-induced apoptosis (Figure 7(b)). This mechanism should be further studied in the future.
In 1963, related proteins and amino acids of PO were studied [19]. The first application of PO for antineoplastic activity utilized its polysaccharide [20]. Another study demonstrated that PO expresses marked antihepatoma and antisarcoma activities [21]. Treatment with the water extract of PO (150 μg mL−1) induced phosphatidyl serine translocation from the inner membrane to the outer membrane after as little as 2 h and then induced apoptosis in human prostate cancer PC-3 cells [6]. Recently, the water-soluble polysaccharide extracted from the fruiting bodies of PO was shown to have obvious antitumor activity; they inhibited human HeLa cells in a concentration-dependent manner [11]. The water-soluble polysaccharide extracted from PO exhibited significantly lower cytotoxicity in human embryonic kidney 293T cells than in HeLa tumor cells when compared with 5-Fu [11]. These results suggest that the water-soluble polysaccharide extracted from PO may be a potential candidate for the development of a novel low-toxicity antitumor agent.
It is interesting that the protein extracts of VV had a stronger apoptotic effect than those of PO (Figure 3). Despite the lack of clear understanding of the detailed apoptotic mechanism of the protein extracts from VV, several other reports have observed anti-cancer effects for VV. The cold-alkali extracts and the polysaccharide of VV expressed antitumor activity [22–24]. Another study demonstrated that the lectin of VV exhibits immunomodulatory effects, stimulating the expression of Th1 cytokines and the proliferative activity of mouse splenocytes [25]. The lectin of VV also induces rapid expression of CD69, CD25, nuclear factor of activated T cells, IL-2 and proliferating cell nuclear antigen in a concentration- and time-dependent manner, leading to lymphocyte proliferation in a calcium-dependent manner [26]. The VV lectin also arrested S180 mouse sarcoma cell proliferation by blocking cell cycle progression in the G2/M phase via activation of the expression of the cyclin kinase inhibitors p21, p27, p53 and Rb [27].
The regulation of apoptosis is related to the susceptibility of cancer cells to chemotherapy. Many anticancer drugs can slow the progression of leukemia and other cancers through induction of apoptosis [28]. For this reason, apoptosis is considered to be a very important event for the development of anti-cancer drugs. The present results demonstrated that the protein extracts of PO and VV not only induced apoptosis but also resulted in ROS production, GSH depletion and ΔΨm loss. These phenomena are similar to effects observed for some clinical and pre-clinical anticancer drugs. For example, cisplatin can change the intracellular ROS and ΔΨm in human Hep G2 hepatoma cells [29]. Curcumin, a pre-clinical anticancer drug, can increase ROS, induce GSH depletion, decrease ΔΨm, release cytochrome c and activate caspases to induce apoptosis in cancer cells [30]. Our previous study also demonstrated that the protein extracts of CL-induced apoptosis in human colorectal SW480 cancer cells through γ-glutamylcysteine synthase inhibition and GSH depletion [2]. ROS production, GSH depletion and ΔΨm loss are highly correlated with PO protein-extract-induced apoptosis in SW480 cells. Pretreatment with NAC, GSH and CyA did not affect the fraction of cells in the SubG1 phase after treatment with VV protein extracts; thus, the mechanism of VV-induced apoptosis is unknown. This is the first finding that the proteins of PO and VV extracted by ammonia sulfate show marked anti-cancer effects in human colorectal cancer cells and human monocytic leukemia cells. A hypothetical diagram that illustrates the anti-cancer effect of CL, PO and VV protein extracts is depicted in Figure 8.
Figure 8.
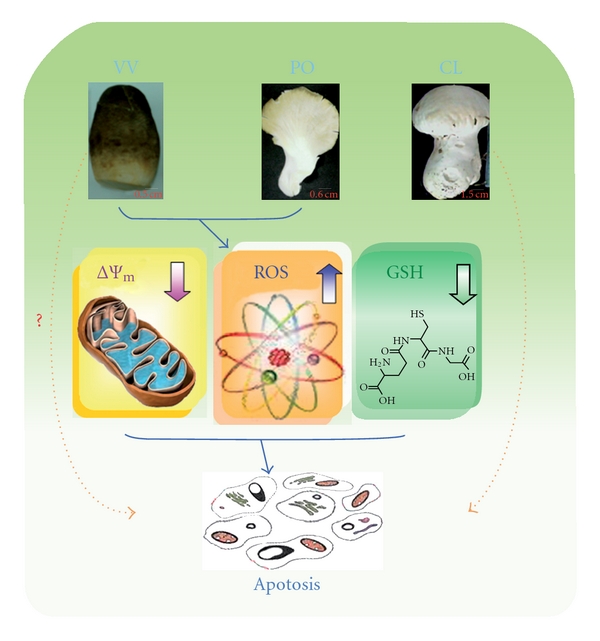
A hypothetical diagram for the anti-cancer effect of CL, PO and VV protein extracts.
5. Conclusions
The protein extracts of PO induced ROS production, GSH depletion and ΔΨm loss; these events were correlated with apoptosis in SW480 cells. The protein extracts of CL, PO and VV can be considered as a source of potential cancer therapeutic agents.
Funding
Grant No. (NSC 96-2320-B-415-002-MY3) to C.-H.C. from the National Science Council, Taiwan, ROC.
References
- 1.Wolff ERS, Wisbeck E, Silveira MLL, Gern RMM, Pinho MSL, Furlan SA. Antimicrobial and antineoplasic activity of Pleurotus ostreatus . Applied Biochemistry and Biotechnology. 2008;151(2-3):402–412. doi: 10.1007/s12010-008-8208-1. [DOI] [PubMed] [Google Scholar]
- 2.Tsay JG, Chung KT, Yeh CH, Chen WL, Chen CH, Lin MH. Calvatia lilacina protein-extract induces apoptosis through glutathione depletion in human colorectal carcinoma cells. Journal of Agricultural and Food Chemistry. 2009;57:1579–1588. doi: 10.1021/jf8030265. [DOI] [PubMed] [Google Scholar]
- 3.Jedinak A, Sliva D. Pleurotus ostreatus inhibits proliferation of human breast and colon cancer cells through p53-dependent as well as p53-independent pathway. International Journal of Oncology. 2008;33:1307–1313. [PMC free article] [PubMed] [Google Scholar]
- 4.Sarangi I, Ghosh D, Bhutia SK, Mallick SK, Maiti TK. Anti-tumor and immunomodulating effects of Pleurotus ostreatus mycelia-derived proteoglycans. International Immunopharmacology. 2006;6(8):1287–1297. doi: 10.1016/j.intimp.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 5.Sun Y, Liu J. Purification, structure and immunobiological activity of a water-soluble polysaccharide from the fruiting body of Pleurotus ostreatus . Bioresource Technology. 2009;100(2):983–986. doi: 10.1016/j.biortech.2008.06.036. [DOI] [PubMed] [Google Scholar]
- 6.Gu Y-H, Sivam G. Cytotoxic effect of oyster mushroom Pleurotus ostreatus on human androgen-independent prostate cancer PC-3 cells. Journal of Medicinal Food. 2006;9(2):196–204. doi: 10.1089/jmf.2006.9.196. [DOI] [PubMed] [Google Scholar]
- 7.Lavi I, Friesem D, Geresh S, Hadar Y, Schwartz B. An aqueous polysaccharide extract from the edible mushroom Pleurotus ostreatus induces anti-proliferative and pro-apoptotic effects on HT-29 colon cancer cells. Cancer Letters. 2006;244(1):61–70. doi: 10.1016/j.canlet.2005.12.007. [DOI] [PubMed] [Google Scholar]
- 8.Shlyakhovenko V, Kosak V, Olishevsky S. Application of DNA from mushroom Pleurotus ostreatus for cancer biotherapy: a pilot study. Experimental Oncology. 2006;28(2):132–135. [PubMed] [Google Scholar]
- 9.Lucke JF. Student’s t test and the glasgow coma scale. Annals of Emergency Medicine. 1996;28(4):408–413. doi: 10.1016/s0196-0644(96)70006-9. [DOI] [PubMed] [Google Scholar]
- 10.Sullivan R, Smith JE, Rowan NJ. Medicinal mushrooms and cancer therapy: translating a traditional practice into Western medicine. Perspectives in Biology and Medicine. 2006;49(2):159–170. doi: 10.1353/pbm.2006.0034. [DOI] [PubMed] [Google Scholar]
- 11.Geethangili M, Tzeng YM. Review of pharmacological effects of Antrodia camphorata and its bioactive compounds. Evidence-Based Complementary and Alternative Medicine. doi: 10.1093/ecam/nep108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lien HM, Lin HW, Wang YJ, Chen LC, Yang DY, Lai YY. Inhibition of anchorage-independent proliferation and G0/G1 cell-cycle regulation in human colorectal carcinoma cells by 4,7-dimethoxy-5-methyl-l,3-benzodioxole isolated from the fruiting body of Antrodia camphorata . Evidence-Based Complementary and Alternative Medicine. doi: 10.1093/ecam/nep020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Firenzuoli F, Gori L, Lombardo G. The medicinal mushroom Agaricus blazei murrill: review of literature and pharmaco-toxicological problems. Evidence-Based Complementary and Alternative Medicine. 2008;5(1):3–15. doi: 10.1093/ecam/nem007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim MO, Moon DO, Jung JM, Lee WS, Choi YH, Kim GY. Agaricus blazei extract induces apoptosis through ROS-dependent JNK activation involving the mitochondrial pathway and suppression of constitutive NF-κB in THP-1 cells. Evidence-Based Complementary and Alternative Medicine. doi: 10.1093/ecam/nep176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kawamura M, Kasai H. Delayed cell cycle progression and apoptosis induced by hemicellulase-treated Agaricus blazei. Evidence-Based Complementary and Alternative Medicine. 2007;4(1):83–94. doi: 10.1093/ecam/nel059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tong H, Xia F, Feng K, et al. Structural characterization and in vitro antitumor activity of a novel polysaccharide isolated from the fruiting bodies of Pleurotus ostreatus . Bioresource Technology. 2009;100(4):1682–1686. doi: 10.1016/j.biortech.2008.09.004. [DOI] [PubMed] [Google Scholar]
- 17.Youn MJ, Kim JK, Park SY, Kim Y, Kim SJ, Lee JS. Chaga mushroom (Inonotus obliquus) induces G0/G1 arrest and apoptosis in human hepatoma HepG2 cells. World Journal of Gastroenterology. 2008;14:511–517. doi: 10.3748/wjg.14.511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ajith TA, Janardhanan KK. Indian medicinal mushrooms as a source of antioxidant and antitumor agents. Journal of Clinical Biochemistry and Nutrition. 2007;40(3):157–162. doi: 10.3164/jcbn.40.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bano Z, Srinivasan KS, Srivastava HC. Amino acid composition of the protein from a mushroom (Pleurotus sp.) Applied Microbiology. 1963;11:184–187. doi: 10.1128/am.11.3.184-187.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Watanabe T. Antineoplastic activity of polysaccharides of Pleurotus ostreates . Nippon Rinsho. 1969;27(6):1759–1761. [PubMed] [Google Scholar]
- 21.Wang H, Gao J, Ng TB. A new lectin with highly potent antihepatoma and antisarcoma activities from the oyster mushroom Pleurotus ostreatus . Biochemical and Biophysical Research Communications. 2000;275(3):810–816. doi: 10.1006/bbrc.2000.3373. [DOI] [PubMed] [Google Scholar]
- 22.Kishida E, Sone Y, Misaki A. Purification of an antitumor-active, branched (1–3)-β-d-glucan from Volvariella volvacea, and elucidation of its fine structure. Carbohydrate Research. 1989;193(C):227–239. doi: 10.1016/0008-6215(89)85121-3. [DOI] [PubMed] [Google Scholar]
- 23.Sone Y, Shibata S, Kishida E, Misaki A. Alteration of beta-D-glucan from edible mushroom after injection into mouse peritoneal cavity. Journal of Nutritional Science and Vitaminology. 1994;40:335–341. doi: 10.3177/jnsv.40.335. [DOI] [PubMed] [Google Scholar]
- 24.Kishida E, Kinoshita C, Sone Y, Misaki A. Structures and antitumor activities of polysaccharides isolated from mycelium of Volvariella volvacea . Bioscience, Biotechnology, and Biochemistry. 1992;56(8):1308–1309. doi: 10.1271/bbb.56.1308. [DOI] [PubMed] [Google Scholar]
- 25.She QB, Ng TB, Liu WK. A novel lectin with potent immunomodulatory activity isolated from both fruiting bodies and cultured mycelia of the edible mushroom Volvariella volvacea . Biochemical and Biophysical Research Communications. 1998;247:106–111. doi: 10.1006/bbrc.1998.8744. [DOI] [PubMed] [Google Scholar]
- 26.Sze SCW, Ho JCK, Liu WK. Volvariella volvacea lectin activates mouse T lymphocytes by a calcium dependent pathway. Journal of Cellular Biochemistry. 2004;92(6):1193–1202. doi: 10.1002/jcb.20153. [DOI] [PubMed] [Google Scholar]
- 27.Liua W-K, Ho JCK, Ng T-B. Suppression of cell cycle progression by a fungal lectin: activation of cyclin-dependent kinase inhibitors. Biochemical Pharmacology. 2001;61(1):33–37. doi: 10.1016/s0006-2952(00)00533-5. [DOI] [PubMed] [Google Scholar]
- 28.Sellers WR, Fisher DE. Apoptosis and cancer drug targeting. Journal of Clinical Investigation. 1999;104(12):1655–1661. doi: 10.1172/JCI9053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen WQ, Shen W, Shen DM. The changes of ROS and mitochondria membrane potential in HepG2 cells on the pressure of cisplatin. Zhonghua Gan Zang Bing Za Zhi. 2005;13(7):531–533. [PubMed] [Google Scholar]
- 30.Sandur SK, Ichikawa H, Pandey MK, et al. Role of pro-oxidants and antioxidants in the anti-inflammatory and apoptotic effects of curcumin (diferuloylmethane) Free Radical Biology and Medicine. 2007;43(4):568–580. doi: 10.1016/j.freeradbiomed.2007.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]


