Abstract
In polarized epithelial cells measles virus (MV) is predominantly released at the apical cell surface, irrespective of the sorting of its two envelope glycoproteins F and H. It has been reported previously that the viral matrix (M) protein modulates the fusogenic capacity of the viral envelope glycoproteins. Here, extant MV mutants and chimeras were used to determine the role of M protein in the transport of viral glycoproteins and release of progeny virions in polarized epithelial CaCo2 cells. In the absence of M, envelope glycoproteins are sorted to the basolateral surface, suggesting that they possess intrinsic basolateral sorting signals. However, interactions of M with the glycoprotein cytoplasmic tails allow M–glycoprotein co-segregation to the apical surface, suggesting a vectorial function of M to retarget the glycoproteins for apical virion release. Whereas this may allow virus airway shedding, the intrinsic sorting of the glycoproteins to the basolateral surface may account for systemic host infection by allowing efficient cell–cell fusion.
Keywords: apical sorting/envelope glycoproteins/epithelial cells/matrix protein/measles virus
Introduction
Measles is a highly contagious acute disease, and is still a major killer of children in developing countries. Measles virus (MV) belongs to the Morbillivirus genus of the Paramyxoviridae family, in the order Mononegavirales, and possesses a non-segmented, single-stranded, minus-sense RNA genome tightly encapsidated into a helical ribonucleoprotein (RNP) structure (ter Meulen and Billeter, 1995; Griffin and Bellini, 1996). The virus acquires its envelope at the late stages of virus assembly, immediately before egress from the cell. During this process, the matrix (M) protein and the RNP (Udem and Cook, 1984) become progressively incorporated into a cell-derived membrane that is highly enriched in virally encoded envelope glycoproteins (Wild and Buckland, 1995). The envelope glycoproteins, fusion protein (F) and hemagglutinin (H) are integral transmembrane proteins mediating virus attachment and cell fusion. F, a type I transmembrane protein, is synthesized as an inactive precursor, F0, and gains its fusogenic activity when it is cleaved by a cellular protease into disulfide-linked F1 and F2 moieties. H, a type II transmembrane protein, mediates virus–cell attachment via interaction with the cell surface receptor CD46 (Dörig et al., 1993; Naniche et al., 1993). M protein, which lines the inner surface of the virion membrane of Paramyxoviridae, has been suggested to play a key role in virion morphogenesis. M appears to act by concentrating viral envelope proteins, and probably the RNPs, to the sites of virus assembly (Buechi and Bachi, 1982). Indeed, an interaction between M and the cytoplasmic tail of F has been demonstrated in chimeric MV particles (Spielhofer et al., 1998). Abrogation of M function dramatically alters the assembly of MV, and is likely to be a factor responsible for subacute sclerosing panencephalitis (SSPE), a chronic disorder associated with defective MV genomes (Billeter et al., 1994). M proteins of Mononegavirales are essential for membrane–nucleocapsid interaction (Yoshida et al., 1976; Chong and Rose, 1994), virus budding, virus morphology (Mebatsion et al., 1999) and infectivity (Garoff et al., 1998). In addition, M protein of MV has been shown to control the fusion function of the viral envelope glycoproteins (Cathomen et al., 1998a).
Epithelial cells line the cavities of the human body, and are the primary target for initial MV infection of the respiratory tract. In culture, epithelial cells form monolayers in which adjacent cells are joined by junctional complexes that physically separate the outer leaflet of the plasma membrane into two functionally distinct domains: the apical and basolateral (Louvard et al., 1992; Rodriguez-Boulan and Powell, 1992). In such cells, specialized vectorial functions such as transport and secretion are made possible by the targeting of proteins and lipids into these distinct surface domains (van Meer and Simons, 1988; Simons and Wandinger-Ness, 1990; Matlin, 1992). The polarized distribution results from selective delivery to and retention at the appropriate domain. Many viruses infect epithelial cells specifically at either the apical or the basolateral surface, and are also released in polarized fashion (Rodriguez-Boulan and Sabatini, 1978; Fuller et al., 1984; Compans, 1995). It is postulated that the envelope glycoproteins are the major determinants for polarized budding and release of enveloped viruses such as influenza, vesicular stomatitis (VSV), Sendai and Sindbis virus. MV enters from the apical surface, and is mainly released from that surface (Blau and Compans, 1995; Maisner et al., 1998). The release of MV from MDCK cells has been postulated to be independent of its envelope glycoprotein sorting (Maisner et al., 1998). Despite the involvement of M protein in multiple interactions with cellular and viral proteins (Bohn et al., 1986; Moyer et al., 1990), and its regulatory function for fusion (Cathomen et al., 1998b), a possible implication in sorting and release of the virus remains obscure.
To investigate the determinants that control the traffic and sorting of MV envelope and membrane proteins as well as virus budding in polarized epithelial cells, a human colon carcinoma cell line, CaCo2, was used. Taking advantage of our MV reverse-genetics system, MV mutants have been generated in which M protein was deleted, or the cytoplasmic domains of F and H were truncated, and chimeras in which envelope proteins were substituted with the VSV-G protein. In this study with polarized epithelial cells we demonstrate that the M protein is apically targeted and plays a crucial role in determining normal assembly of MV at the apical domain. We also show that the envelope glycoproteins are sorted to the basolateral domain in the absence of M protein, indicating that they possess basolateral sorting signals, and that these signals are largely overridden in the presence of M protein.
Results
Mutant and chimeric measles viruses
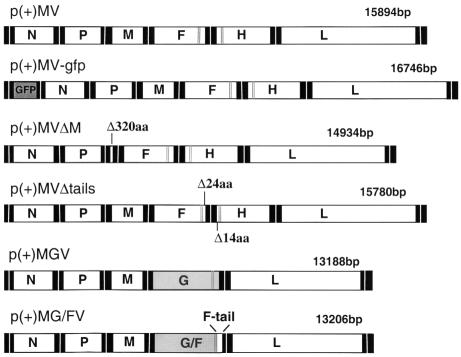
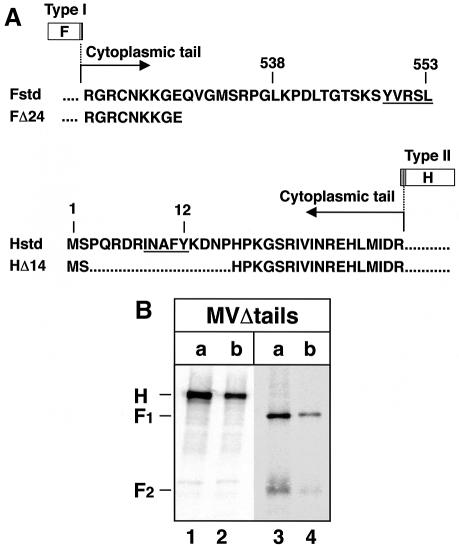
The construction of chimeric MV–VSV viruses (MGV and MG/FV) and mutant MV viruses (MVΔtails and MVΔM) (Figure 1) has been described previously. In MGV, the MV envelope glycoproteins are replaced by the single envelope glycoprotein G of VSV. MG/FV is similar to MGV but encodes a G/F hybrid in which the VSV G cytoplasmic tail was substituted by that of MV F. We demonstrated that replacement of the envelope glyco proteins H and F by the VSV envelope protein G results in the exclusion of M protein from viable mature virions. Conversely, substitution of the cytoplasmic tail of G by that of F (G/F) leads to the incorporation of M into the virions (Spielhofer et al., 1998). In MVΔtails and its fluorescence-inducing derivative MVΔtails–green fluorescent protein (GFP), 24 amino acids of F and 14 amino acids of H were deleted from their cytoplasmic tails. These viruses grew to similar titers to standard MV, but induced faster cell-to-cell fusion (Cathomen et al., 1998b). In the case of MVΔM and its derivative MVΔM–GFP, where the M protein is deleted, the viruses grew to very low titers in Vero cells, but also caused faster cell-to-cell fusion (Cathomen et al., 1998a).
Fig. 1. Structures of MV cDNAs encoding standard, recombinant, mutant and chimeric viruses. The portions of the plasmids giving rise to anti-genomic RNAs are shown. The constructions of p(+)MV and its derivatives p(+)MVΔM, p(+)MVΔtails, p(+)MGV and p(+)MG/FV were reported (Radecke et al., 1995; Cathomen et al., 1998a,b; Spielhofer et al., 1998). The white boxes represent the reading frames of the six MV genes N, P, M, F, H and L. The black boxes represent the non-translated gene boundary regions. The gray boxes represent reading frames of VSV-G and the hybrid VSV-G/MV-F, respectively. The extent of deletions (Δ) of M, as well as F and H tails, and the replacement of the G tail by that of F are indicated. The dark gray box represents the position of the GFP reading frame in the MV genome sequence. Strong expression of GFP was ensured by creating an additional independent transcription unit upstream of N; a duplicate of the N/P intergenic region was placed between the GFP and the N reading frames. The vertical gray lines represent the transmembrane regions in F, H, G and G/F reading frames.
Release of infectious MV particles occurs preferentially at the apical domain
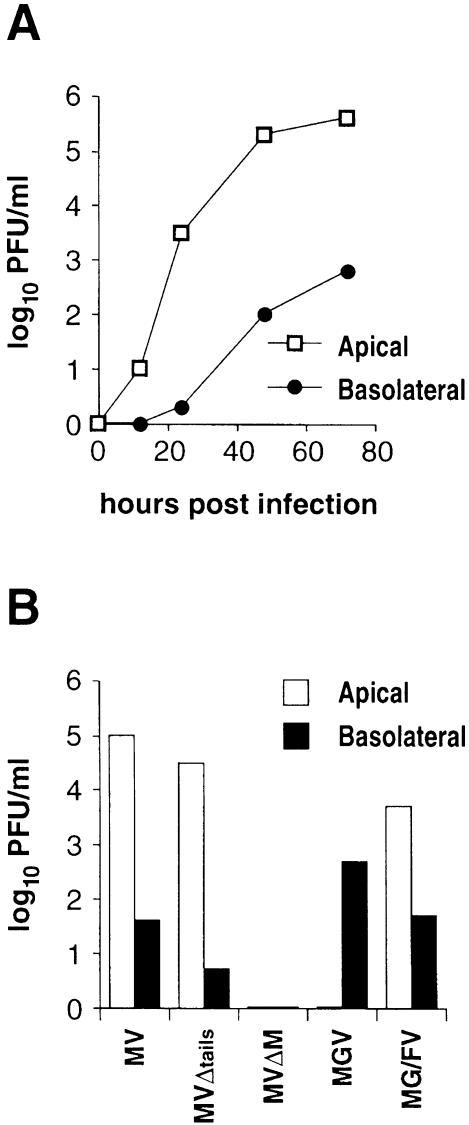
MV enters polarized epithelial cells at the apical membrane and is mainly released from that side. To investigate the mechanism of MV assembly at the apical domain, we first examined whether the pattern of virus release is reproducible in our system using the CaCo2 cell line. Cell monolayers on transwell filters, grown to complete confluence to establish tight cell–cell junctions, were infected with standard MV at an m.o.i. of 0.1 p.f.u./cell. In parallel, the extent of infection and syncytia formation was visualized by using recombinant MV–GFP that expresses enhanced GFP (not shown). At various times post-infection, media from the apical and the basolateral domains were collected, and the released infectivity was titered by plaque assay on Vero cells. Relative titers of apically and basolaterally released infectivity with time are shown in Figure 2A. Throughout the infection 500- to 1000-fold more infectious particles were released from the apical than from the basolateral domain. This predominant apical release of MV is not due to a possible filter impediment to virus release from the basolateral domain. Control, non-polarized, infected Vero cells grown on such filters released equal amounts of infectivity from both sides (not shown). These results are consistent with previous reports (Blau and Compans, 1995; Maisner et al., 1998), and corroborate the notion that MV particles are shed in a polarized fashion from epithelial cells.
Fig. 2. Polarized release of MV and mutant viruses in CaCo2 cells. (A) Release of MV at increasing times p.i. CaCo2 cells were grown on transwell filters until a tight monolayer was reached. Cells were then infected with standard MV, and medium was collected from the apical and the basolateral domains at 12, 24, 48 and 70 h p.i. Infectious viruses released to either side were plaque assayed. (B) Effects of F- and H-tail truncation, M deletion and envelope glycoprotein exchange on virus budding. At 50 h p.i. (MV, MVΔtails and MVΔM) and 70 h p.i. (MGV and MG/FV) virus shed into the media from the apical and basolateral domains was determined by plaque assays.
Apical virus budding fully depends on the M protein
It is suggested that M protein interacts with the cytoplasmic tails of the envelope glycoproteins F and possibly H (Cathomen et al., 1998b; Spielhofer et al., 1998) and recruits them into intracellular microdomains (Cathomen et al., 1998a; Manié et al., 2000). To investigate the nature of virus budding upon the deletion of M protein, deletion of F and H cytoplasmic tails or glycoprotein swapping, cultures of CaCo2 cells on filters were infected with MV, MVΔtails, MVΔM, MGV and MG/FV viruses at an m.o.i. of 0.1 p.f.u./cell. Media were collected at 50 hours post-infection (h p.i.) in the case of MV, MVΔtails and MVΔM, and at 70 h p.i. in the case of MGV and MG/FV. Virus release from apical and basolateral domains was monitored by plaque assay on Vero cells. Figure 2B depicts the apical and basolateral release of the different viruses. In standard MV infection, apically released infectivity was always 500- to 1000-fold higher than basolaterally released infectivity, as is also shown in Figure 2A. When the cytoplasmic tails of F and H were partially deleted, the ratio of apical to basolateral release of infectivity even increased by 2-fold. Total infectivity was slightly reduced for MVΔtails, which is likely to be due to inherent defects in truncated F and H proteins. Compared with standard MV, an M-deficient virus (MVΔM) has been shown to be more efficient at inducing cell-to-cell fusion when grown in non-polarized Vero cells, although endpoint titers were ∼250-fold lower than standard MV (Cathomen et al., 1998a). In CaCo2 cells, however, deletion of the M protein completely abolished release of infectious particles from both domains. In MGV-infected cells, no detectable infectivity was recovered from the apical side, but 3 × 102 p.f.u./ml from the basolateral side (Figure 2B). With cells infected by MG/FV, 5 × 103 p.f.u./ml were recovered from the apical and ∼50 p.f.u./ml from the basolateral side [as experienced with non-polarized Vero cells, MGV was consistently released less efficiently than MG/FV (Spielhofer et al., 1998)]. Since MGV was relatively poorly released, but exclusively from the basolateral domain, and MG/FV was released more efficiently, and predominantly from the apical domain, these results indicate that M allowed better release of MG/FV in CaCo2 cells, as previously shown in non-polarized cells. MGV is released devoid of M (Spielhofer et al., 1998) due to the intrinsic vesiculation function of G protein (Mebatsion et al., 1996). MVΔM, however, is not released at all in polarized cells, indicating that M protein is the key player for the apical release of MV.
Sorting of the envelope glycoproteins F and H is regulated by the M protein
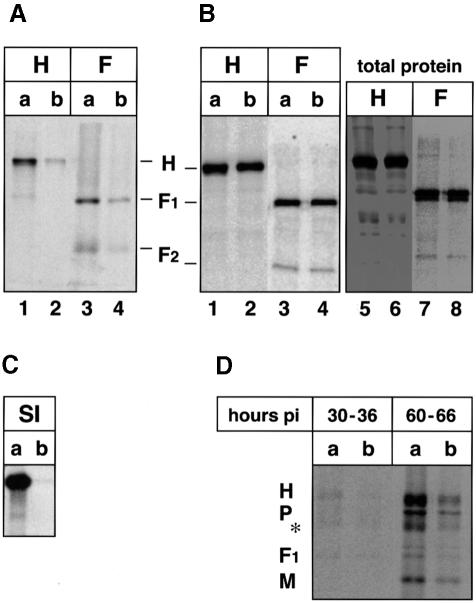
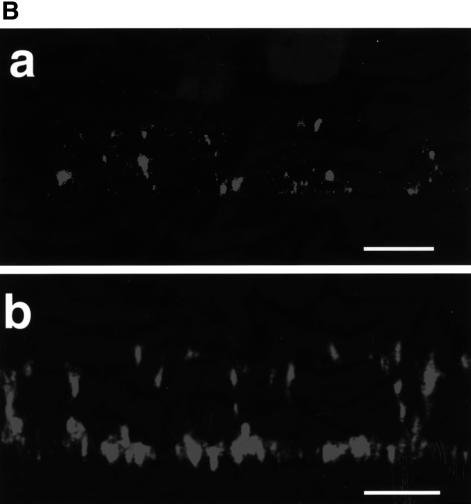
Viruses acquire their envelope glycoproteins usually at the site of budding. Since most MV infectivity is released from the apical domain, the envelope glycoproteins are expected to be concentrated there. To test this, confluent CaCo2 cell monolayers inoculated with MV (as above) were pulse-labeled at 30 and 60 h p.i., chased, and cell surface proteins were immunoprecipitated. At early times of infection (30–36 h p.i.) both envelope glycoproteins were predominantly sorted to the apical domain (Figure 3A; 80% of the H protein apical, lane 1, and 20% basolateral, lane 2; similarly, 70% of F apical, lane 3, 30% basolateral, lane 4). However, at late times of infection (60–66 h p.i.) the percentages shifted to nearly equal levels in both domains (Figure 3B; H 55% apical, lane 1, 45% basolateral, lane 2; F 50% both apical and basolateral, lanes 3 and 4). To compare the fraction of surface capture with the total amount of labeled proteins in the same experiment, cells were lysed and immunoprecipitated by additional antibody (Figure 3B, lanes 5–6). Apical H was 46% of the total (Figure 3B, lanes 1 and 5) and basolateral H was 39% of the total (lanes 2 and 6); apical and basolateral F were 40% of the total each (lanes 3, 7 and lanes 4, 8, respectively). This indicates that the majority of H and F (80–85% of the total) was precipitated by surface capture. The remaining uncaptured 15–20% of H or F must be intracellular (note the different glycosylation pattern, comparing lanes 3 and 4 with 7 and 8). To test whether cells remained polarized during the experiment, sucrase–isomaltase (SI), an endogenous apical protein (Matter et al., 1990), was used as a marker. Indeed, SI remained exclusively at the apical surface (Figure 3C), indicating that the cells sorted their proteins in a polarized fashion even at 60–66 h after MV infection. To verify the sorting of the envelope proteins by immunofluorescence, CaCo2 cells were infected by standard MV and fixed at either 36 or 66 h p.i. Surface proteins were stained with monoclonal anti-F antibody and labeled with Cy3 (red). The results are consistent with the biochemical data; at 36 h p.i. F was mainly apical (Figure 3E, a), and at 66 h p.i. F was sorted equally to both domains (Figure 3E, b). The temporal changes in the envelope glycoprotein sorting at a later time of infection (60–66 h p.i.) could be due to an efficient virus release from the apical surface (as shown in Figure 2A), which results in shedding of more labeled proteins into virions, whereas proteins directed to the basolateral surface accumulate there. To verify this possibility, media from MV-infected cultures were collected after 3 h pulse and 3 h chase at 30–36 h p.i. or at 60–66 h p.i., and analyzed on SDS–PAGE after purification on a sucrose cushion. Figure 3D shows that at 30–33 h p.i. only small amounts of labeled virus particles were released from the apical surface, whereas at 60–66 h p.i. labeled particles were released predominantly from the apical domain (higher exposure of the gel was necessary to detect those released from the basolateral domain). Apically released particles were 10- to 15-fold more numerous than those released from the basolateral surface, and contained 10–12% of the surface apical glycoproteins; basolateral particles contained <1%. The efficient release of labeled particles from the apical surface and the continuous bleed of F and H to the basolateral surface could explain the shifts in glycoprotein sorting at later times of infection. Since the release of infectivity from the basolateral surface at 50–70 h p.i. was ∼4.2 × 102 p.f.u./ml, compared with 2.0 × 105 p.f.u./ml from the apical surface (Figure 2B), and the increase in amounts of envelope glycoproteins at the basolateral surface did not quantitatively enhance release of infectivity, the presence of MV glycoproteins at a cell surface is not sufficient to specify the budding site for infectious virions.
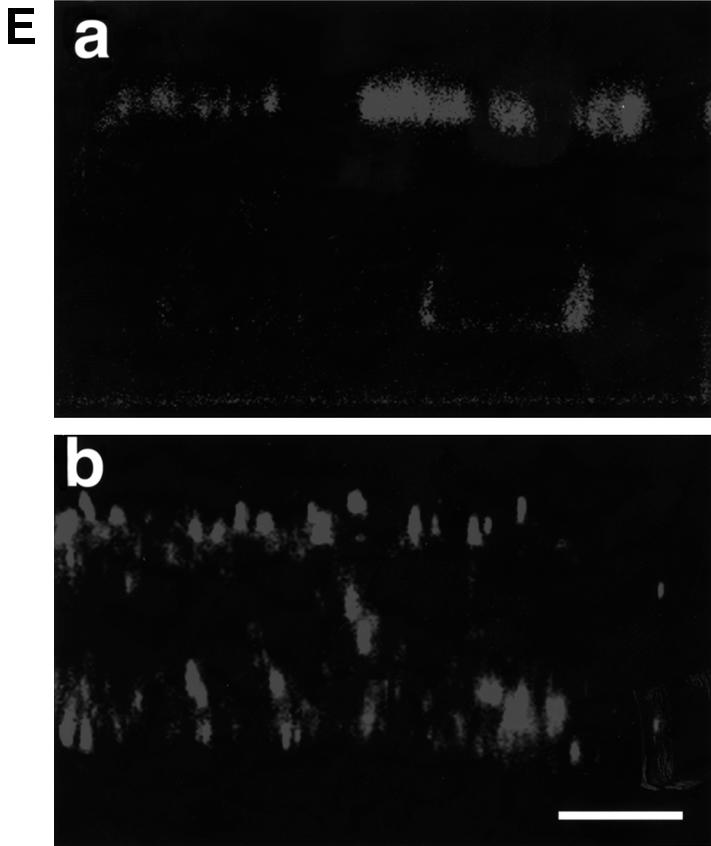
Fig. 3. Sorting of infectious MV and the envelope glycoproteins F and H in CaCo2 cells. Infected cells were radiolabeled at (A) 30 h p.i. and (B) 60 h p.i., for 3 h and chased for another 3 h. Monoclonal anti-F or anti-H antibodies were applied from either the apical or the basolateral domains; captured proteins were immunoprecipitated by protein A–Sepharose. To analyze total proteins, additional antibodies were added to the corresponding cell lysates and immunoprecipitated (B, lanes 5–8). (C) Monoclonal anti-SI antibody was used to capture surface SI at the apical surface. (D) Labeled particles were isolated from infected cultures after 3 h pulse and 3 h chase at 30–36 h p.i. and at 60–66 h p.i. Viruses were partially purified by pelleting through a 30% sucrose cushion; the asterisk indicates the position of N and possibly F proteins. Proteins were analyzed by 10–12% SDS–PAGE and quantitated by phosphoimaging. (E) Localization of viral glycoproteins at the surface by immunofluorescence and confocal vertical sections at 36 h p.i. (a) and 66 h p.i. (b). Monoclonal anti-F antibody coupled to Cy3 was used. Bar, 10 µm.
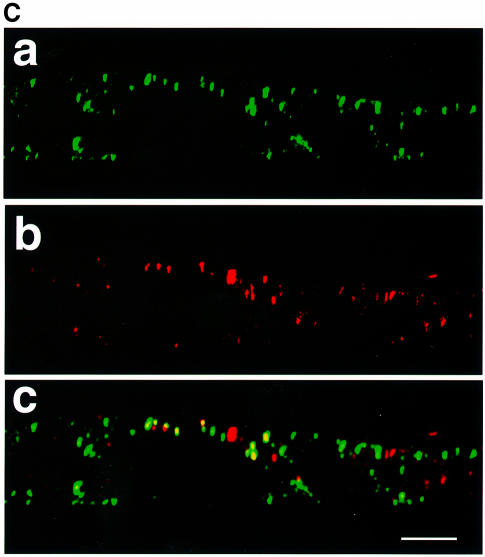
To determine possible consequences of deleting the M protein on envelope glycoprotein sorting, monolayers of CaCo2 cells were infected with MVΔM and protein distribution was analyzed biochemically and by immunofluorescence. At 30 and 60 h p.i. cells were labeled, and viral envelope glycoproteins expressed at the cell surface were captured from the apical and the basolateral domains and analyzed as above. Figure 4A shows that under these conditions both F and H proteins were predominantly sorted to the basolateral domain (75% of H at 36 h p.i. and 65% at 66 h p.i.; similarly, 75–80% of F protein, sum of F0, F1 and F2, both at 36 and 66 h p.i.). To assess further the sorting of MV envelope glycoproteins in the absence of M, surface proteins were captured by monoclonal anti-F antibody and stained with Cy3 (red) at 33 and at 66 h p.i., and analyzed by immunofluorescence and confocal microscopy. The results are in accordance with the biochemical data of Figure 4A (Figure 4B). In contrast to standard MV, F protein was predominantly localized at the basolateral surface at early (Figure 4B, a) and late (Figure 4B, b) times of infection. Taken together these results indicate that the localization of the envelope glycoproteins within the cell is strongly influenced by M protein, and that M protein is the key player in the regulation of envelope glycoprotein sorting.
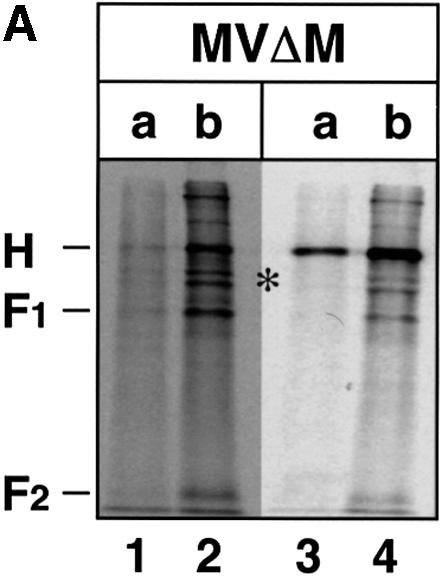
Fig. 4. Sorting of F and H in MVΔM-infected CaCo2 cells. Infection and pulse–chase were exactly as for standard MV. (A) In this experi ment both anti-H and anti-F antibodies were added consecutively from either the apical or the basolateral domains. The asterisk indicates uncleaved F0 protein (shown as double bands). (B) Localization of viral proteins by immunofluorescence and confocal vertical sections at 36 h p.i. (a) and 66 h p.i. (b). Monoclonal anti-F antibody coupled to Cy3 was used. Bar, 10 µm.
M protein is targeted to the apical domain
The results presented so far show that M is a key mediator for virus budding, and that it regulates sorting of the envelope glycoproteins to the apical cell surface. To investigate whether M itself could be targeted to the apical cell surface and drags along glycoproteins that are able to interact with it, CaCo2 cells infected either with standard MV, MGV or MG/FV were used first to analyze the sorting of the envelope proteins biochemically, and then to correlate their sorting in comparison with that of M by indirect immunofluorescence and confocal scanning.
In MGV, the G protein contains a basolateral sorting signal that efficiently directs the protein to the basolateral surface (Thomas and Roth, 1994). In addition, we knew that MGV particles, in contrast to MG/FV particles, do not contain M protein, an indication of the lack of interaction between G and M (Spielhofer et al., 1998). If M protein is apically targeted then replacement of the G cytoplasmic tail by that of F should give rise to a protein that is sorted predominantly to the apical domain in the presence of M. At 70 h p.i., surface protein expression was determined by antibody capture. Figure 5A, lanes 1–4 shows the sorting pattern of the proteins. Whereas unmodified G was sorted mainly to the basolateral domain (20% apical, lane 1; 80% basolateral, lane 2), G/F glycoprotein was sorted mainly to the apical domain (65% apical, lane 3; 35% basolateral, lane 4). Compared with the total immunoprecipitated proteins (Figure 5A, lanes 5–8), surface captured G was 80% of the total, and G/F was 46% of the total (the intracellular transport rate of G/F to the cell surface is slower; note the different glycosylation pattern of total and surface G/F, lanes 7 and 8).
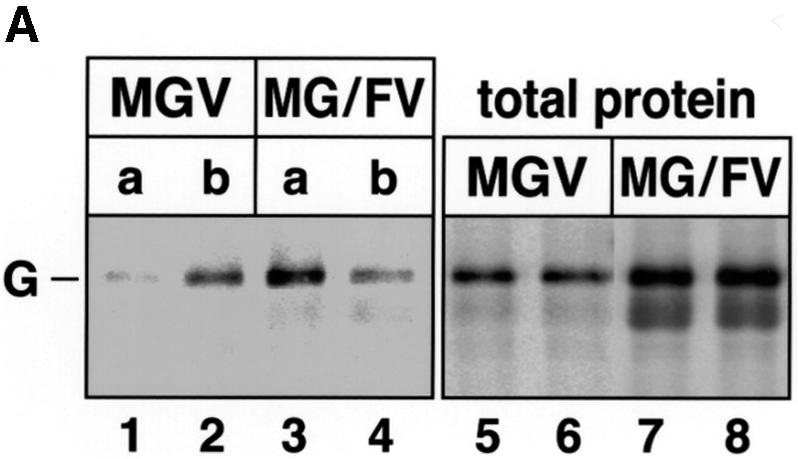
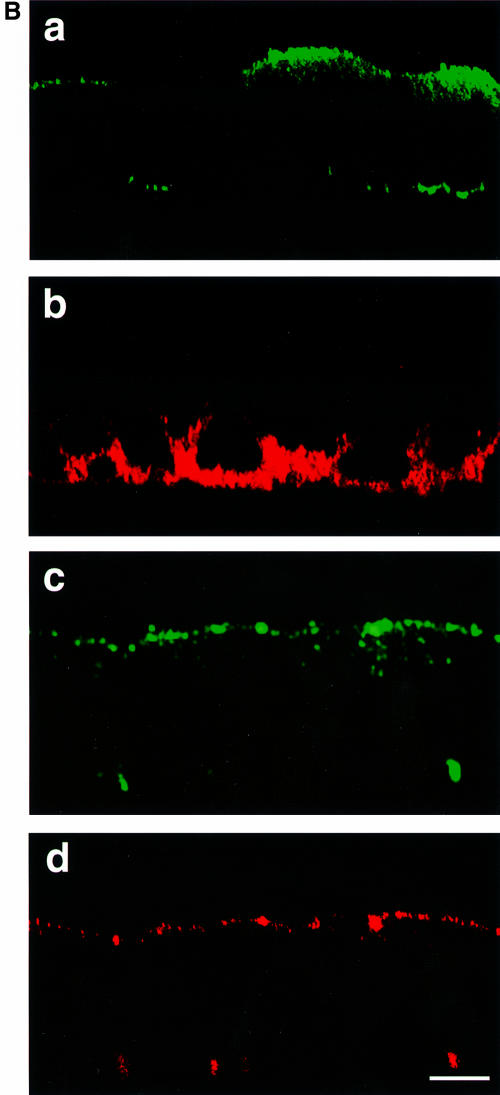
Fig. 5. Envelope and M protein sorting in MGV, MG/FV and standard MV-infected CaCo2 cells. (A) MGV- and MG/FV-infected cells were pulsed and chased at 70 h p.i., and polyclonal anti-VSV antibody was used. To analyze total G and G/F proteins, additional antibodies were added to the corresponding cell lysates and immunoprecipitated (lanes 5–8); the faster bands in lanes 7 and 8 represent the immature high-mannose population. (B) Localization of M, G and G/F by confocal vertical sections; M was stained with FITC (green), G and G/F were stained with Cy3 (red). (C) Localization of M and H in CaCo2 cells infected with standard MV. Cells were fixed and permeabilized at 66 h p.i. and double stained first with polyclonal anti-H coupled to FITC (green signal in b and c), then with monoclonal anti-M coupled to Cy3 (red signal in a and c). To demonstrate the polarization of the CaCo2 cells, antibody to DPPIV, which sorts to the apical domain (d), and antibody to gp120 (e), which sorts to the basolateral domain, were used in MV-infected cells. Bar, 10 µm.
To localize M as well as G and G/F proteins, immunofluorescence and confocal analysis were performed. The surface viral envelope glycoproteins were first captured by polyclonal anti-G antibody before permeabilization (to avoid staining of intracellular G), then cells were permeabilized and M was captured by anti-M monoclonal antibody. G was mainly sorted to the basolateral surface (Figure 5B, b), whereas G/F was biased to the apical surface (Figure 5B, d), consistent with the biochemical data (Figure 5A). Interestingly, M protein was mainly sorted to the apical surface in both MGV and MG/FV infections (Figure 5B, a and c). In addition, as expected, M and G/F co-localized in several areas of the apical surface (Figure 5B, compare c and d), an indication of G/F–M interaction.
The sorting pattern of M protein in standard MV-infected CaCo2 cells was determined by immunofluorescence at 66 h p.i. Anti-H polyclonal antibody coupled to fluorescein isothiocyanate (FITC; green) was used first, then cells were permeabilized, and anti-M monoclonal antibody coupled to Cy3 (red) was used. H was sorted to both domains equally (Figure 5C, a). Conversely, M protein was preferentially localized at the apical domain (Figure 5C, panel b), where some of it co-localized with H at that surface (Figure 5C, arrows in a–c). The integrity and polarity of CaCo2 cells during these experiments were assessed using two internal controls, dipeptidyl aminopeptidase IV (DPPIV) and gp120. DPPIV is known to be sorted to the apical domain, and gp120 to the basolateral domain (Matter et al., 1990; Casanova et al., 1991). Figure 5C shows that DPPIV (d) and gp120 (e) were indeed present at the expected domain, indicating that the infected cells retained complete polarization under the conditions used. Taken together, these results demonstrate that the M protein is preferentially sorted to the apical domain and this provides the basis for budding of MV from the apical cell surface.
The cytoplasmic tails of F and H contribute partially to their intrinsic basolateral sorting
Since F and H were preferentially sorted to the basolateral domain in the absence of M, it is likely that their cytoplasmic domains contain sequences that could potentially serve as basolateral sorting signals. Potential sequence candidates are 549YVRSL553 and 8INAFY12 in the cytoplasmic tails of F and H, respectively (Figure 6A). To determine the possible role of these segments of the cytoplasmic tails in basolateral sorting, we used the MVΔtails mutant described previously, in which 24 amino acids are deleted from the F tail and 14 amino acids from the H tail (Figure 6A) (Cathomen et al., 1998b). If the cytoplasmic tails of F and H contain intrinsic basolateral sorting signals, then the deletions that include the sequence candidates should give rise to dominant apically targeted proteins, even at later times of infection, in the presence of M.
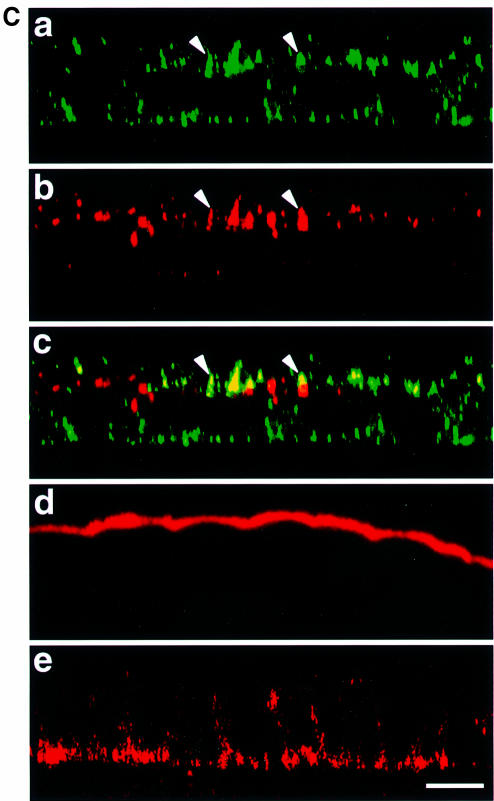
Fig. 6. Sorting of F and H with cytoplasmic tail deletion. (A) Scheme of F- and H-tail deletions. The underlined letters highlight the potential basolateral sorting signals and their positions in the cytoplasmic domains. (B) Polarity of FΔtail and HΔtail at 66 h p.i. Proteins were expressed by MVΔtails, labeled and immunoprecipitated as with standard MV in Figure 3. (C) Immunofluorescence analysis at 66 h p.i. of HΔtail coupled to FITC (green, b and c) and M with Cy3 (red, a and c). Bar, 10 µm.
At 66 h p.i., infected CaCo2 cells were pulse labeled and cell surface glycoproteins were immunoprecipitated. In contrast to MV-infected cells, both truncated glycoproteins remained mainly at the apical domain even late in infection (Figure 6B). For HΔtail the apical population was 70–75%, and for FΔtail 75–80% (based on the total immunoprecipitated proteins, as in standard MV). Immunofluorescence analyses are in accordance with the biochemical data, and revealed a more biased distribution of H to the apical surface (Figure 6C, a); M protein was again sorted to the apical domain (b). The interaction of H and M persisted at the apical domain as shown in Figure 6C (compare a–c). These results suggest that the deleted portions of the cytoplasmic tails contain determinants that at least partially contribute to the intrinsic sorting of F and H to the basolateral domain. The presence of additional signals in the undeleted regions could not be ruled out.
Discussion
The identification of the mechanisms by which MV matures and assembles in polarized epithelial cells is essential to understand its interactions with the host and its pathogenic potential; in addition, this might provide clues to the progression of the rare and invariably lethal disease, SSPE.
The results presented here reveal a previously unrecognized role of M protein during MV infection. In addition to its structural role in virion morphogenesis, we have found that M protein also specifies the vectorial release of virus from the apical surface of polarized epithelial cells. M protein accumulates apically and reverts the transport of glycoproteins F and H to the apical cell surface. Only in the presence of M, carrying along both F and H, will MV properly bud from the apical surface. In the absence of M, F and H prevalently accumulate basolaterally, and by themselves do not promote viral budding.
Budding of enveloped viruses usually occurs at the site at which their envelope glycoproteins are mostly concentrated (Simons and Garoff, 1980; Owens et al., 1991); thus, the envelope glycoproteins determine the site of virus release in epithelial cells. Since budding of MV devoid of M is not detectable from the basolateral surface, and the shedding of infectious MV and chimeric MG/FV virions at the apical surface occurs only in the presence of M, an interaction between F, H and M is likely to occur to retarget and segregate F and H, and to allow the release of infectious virions from the apical cell surface. The intracellular co-localization of envelope proteins and M strongly suggests, as previously shown for Sendai virus (Sanderson et al., 1994, 1995), that interaction of H and F with M occurs intracellularly, in specific membrane domains. This adds to the evidence that M protein has multiple regulatory functions. M functions not only as a regulator for virus-induced cell–cell fusion (Cathomen et al., 1998a) but also as a regulator for viral glycoprotein sorting and virion release (this study).
M proteins of other negative strand RNA viruses, influenza virus and VSV apparently play a minor role in affecting virus polarity. In contrast, mutated Sendai virus M protein of a pantropic strain altered virus budding from the apical to become random (Tashiro et al., 1996). The accumulation of MV M at the apical surface and its involvement in glycoprotein sorting is likely to be mediated by the interaction of M with putative intracellular transporter proteins. So far, M protein is shown to associate with the cellular cytoskeleton, actin, which could be involved in the budding process (Bohn et al., 1986). Little is known, however, about actin involvement in MV sorting. Thus, determining the cellular partner with which M interacts, near the apical surface, could explain mechanistically how M is able to override the innate tendencies of F and H to sort to the basolateral surface.
Several mechanisms and sorting signals have been discovered that are responsible for apical sorting of membrane and secretory proteins, underlining the complexity of this pathway compared with the basolateral pathway. Glycolipid (Lisanti and Rodriguez-Boulan, 1991; Brown and Rose, 1992) and transmembrane domains (Lin et al., 1998), N- and O-linked glycans (Fiedler et al., 1994; Scheiffele et al., 1995; Yeaman et al., 1997; Alfalah et al., 1999) and also protein motifs in the ectodomains of proteins (Jacob and Naim, 1995; Gut et al., 1998) have been shown to direct proteins to the apical surface. In many cases targeting occurs through interaction of these signals in the trans-Golgi network with detergent-insoluble lipid rafts enriched in glycosphingolipids and cholesterol (Simons and Ikonen, 1997; Lin et al., 1998; Alfalah et al., 1999). Our data present a novel apical sorting pathway by which MV envelope glycoproteins are chaperoned by the M protein to initiate virus release at that surface. This mechanism may implicate lipid rafts since M, F and H are found partially associated with these structures (20–40%) (unpublished observation and Manié et al., 2000). Therefore, the potential roles of lipid rafts, and probably actin-associated proteins (Salas et al., 1997) in the apical sorting of MV remain to be determined. MV envelope glycoproteins F and H apparently contain intrinsic basolateral sorting determinants in their cytoplasmic tails. This is evident in MVΔM-infected cells. These signals, however, do not function properly in the presence of M protein due to the direct interaction of cytoplasmic tails with M. A continuous bleed of F and H to the basolateral surface allows conspicuous accumulation of the envelope glycoproteins at that surface, especially since virus budding is inefficient and the half-life of these envelope proteins at the cell surface, ∼10–15 h (Fujinami et al., 1981), is high. However, truncations in the cyto plasmic domains of F and H shift the ratio of sorting to the apical side and only slightly reduce their interaction with M protein, suggesting that these domains could be involved in directing the proteins to the basolateral surface. The identity of the basolateral sorting signals in the cytoplasmic tails of F and H and the mechanism by which these proteins are sorted to the basolateral cell surface have yet to be elucidated. This requires the construction of various MV mutants with deleted M, and sequential truncations and mutations in the cytoplasmic tails of F and H.
The intrinsic preference of the envelope glycoproteins to sort to the basolateral surface may be of great importance for MV pathogenesis. Other viruses, like Sendai virus, simian virus 5 and influenza virus (Rodriguez-Boulan and Sabatini, 1978; Roth et al., 1979) enter and mature from the apical surface in polarized cells, and in vivo produce a localized infection, restricted to the respiratory tract. MV enters and is mainly released from the apical surface as well. However, in contrast to these viruses, MV always produces systemic infections in vivo. It is known that cells of the immune system are infectable by MV and recently macrophages have been found to be infected soon after intranasal inoculation of CD46-transgenic mice with MV (Mrkic et al., 2000). Our findings in CaCo2 cells add to the understanding of MV pathogenesis and may help explain the transmission of the virus to the lymph and blood-stream. While M protein drives the apical assembly of MV and leads to release of infectious particles early after infection, the envelope glycoproteins escaping the capture by M are sorted to the basolateral domain, at which they accumulate at prolonged time of infection. Infection of macrophages is likely to occur upon contact with the basal surface of epithelial cells by direct cell–cell fusion, which is not inhibited by the presence of M.
Materials and methods
Cells and viruses
The human embryonic kidney 293, monkey kidney fibrobast (Vero) and human colon carcinoma (CaCo2) cell lines were maintained in Dulbecco’s modified essential medium (DMEM) supplemented with 10% fetal calf serum (FCS). For virus rescue, 293-3-46 cells were used. These cells are helper cells stably expressing MV N and P proteins as well as T7 RNA polymerase (Radecke et al., 1995).
CaCo2 cells were maintained according to established protocols (Jacob et al., 1999). For experiments where the sorting of the viral envelope glycoproteins was measured, cells were cultured on transwell filters of pore size 1 µm. Cells were fed daily by changing their medium. Cell monolayers were infected when trans-monolayer resistance reached 650–680 µohm/cm2, 5–7 days post-plating.
Measles virus Edmonston B (MV EdB) and all mutant derivatives were rescued according to Radecke et al. (1995). Propagation and titration were as described earlier (Udem, 1984). Gradient purification and amplification of all viruses were carried out according to our previously published reports (Naim and Roth, 1993). The construction of plasmids allowing the rescue of mutant MV, MVΔtails, MVΔM and chimeric MV–VSV (MGV and MG/FV) was described earlier (Cathomen et al., 1998a,b; Spielhofer et al., 1998). To obtain MV–GFP, eliciting strong fluorescence, a cDNA encoding the enhanced GFP (Clontech) followed by a duplicate N/P gene boundary region was inserted upstream of the N reading frame of the standard plasmid p(+)MV (Hangartner, 1998). To construct p(+)MVΔtails–GFP and p(+)MVΔM–GFP plasmids, which were used to monitor the extent of virus infection, a fragment resulting from cleavage of p(+)MV–GFP with SacII and SfiI was ligated to complementary fragments derived from p(+)MVΔtails and p(+)MVΔM, respectively (not shown). All experiments with live mutant and chimeric viruses were performed at biosafety level 2, complying with the rules of the Swiss Interdisciplinary Committee for Biosafety in Research and Technology (SICBRT).
Immunofluorescence
To visualize expression of proteins, infected cells on coverslips or filters were fixed with 3% paraformaldehyde in phosphate-buffered saline (PBS), and permeabilized with 0.1% Triton X-100 in PBS, washed with blocking solution (PBS containing 1% bovine serum albumin) for 1 h, and stained with the specific antibody (Naim and Roth, 1994). Polyclonal anti-MV H or anti-MV F peptide antibodies were used at a dilution of 1:300 v/v followed by TRITC, Cy3 or FITC-conjugated goat anti-rabbit serum. The MV-specific monoclonal antibodies anti-F and anti-H were used in a dilution of 1:300 v/v and anti-M in a dilution of 1:100 v/v followed by TRITC-, Cy3- or FITC-conjugated goat anti-mouse serum. Polyclonal anti-VSV antibody was used in a dilution of 1:500 v/v. Cells were analyzed on a confocal microscope.
Confocal microscopy
The imaging system consisted of a Leica inverted microscope DM IRB/E, a Leica true confocal scanner TCS NT and a Silicon Graphics workstation. The images were recorded using a Leica PL APO 63× oil or a PL APO 40× oil immersion objective. The system was equipped with an argon/krypton mixed gas laser. Image processing was performed on a Silicon Graphics workstation using ‘Imaris’ (Bitplane AG, Zurich, Switzerland), 3D multi-channel image processing software specialized for confocal microscopy images.
Radiolabeling and polyacrylamide gel electrophoresis
CaCo2 cells were grown on Costar (Cambridge, MA) transparent transwell filter inserts with 1 µm pores, in 36-mm six-well plates, and were infected with standard MV (MV-tag), MV mutants (MVΔtails, MVΔM) and MV chimeras (MGV and MG/FV) at an m.o.i. of 0.1 p.f.u./cell. At 30 or 60 h after MV and MV mutant infection and 70 h post MV chimera infection, cells were washed four times with Dulbecco’s PBS supplemented with Ca2+ and Mg2+ at room temperature to remove excess cysteine and methionine, pre-starved with cysteine- and methionine-free DMEM for 45 min, and pulse-labeled for 3 h from the basal side with 500 µCi/ml Trans35S-label (ICN Pharmaceuticals Inc., Irvine, CA). Transwell inserts were placed on 1 ml of labeling medium, using 0.5 ml of the same medium without label to keep the apical side wet. Cells were washed once with warm PBS and chased in prewarmed DMEM, containing additional 50 mM methionine and cysteine, for 3 h. The chase was terminated by washing cells with ice-cold DMEM. Cell surface proteins were captured, immunoprecipitated (see below) and proteins were eluted by SDS–Laemmli buffer and analyzed by 10 or 12.5% SDS–PAGE (Laemmli, 1970).
Cell surface arrival of envelope glycoproteins and immunoprecipitation
CaCo2 cells grown on transwell filter inserts were infected with MV and MV mutants from the apical domain (Blau and Compans, 1995), and with MGV and MG/FV from the basolateral domain, similarly to VSV infection protocols in polarized epithelial cells (Roth and Compans, 1981; Pfeiffer et al., 1985). Inoculum was removed after an incubation period of 2 h. Cells were washed twice with PBS, then warm DMEM containing 10% FCS was added and incubated at 37°C for various times. Protein labeling and chase protocols are basically described above; additional modification of the protocol is described for individual experiments. At the end of the chase, transwell filters were placed on ice, and monoclonal anti-H or anti-F antibodies were added from either the apical or the basolateral domains and incubated on ice for 2–3 h with occasional shaking every 30 min (to distribute the antibody evenly). After a series of washing with ice-cold DMEM or PBS, cells were lysed with 1 ml of lysis buffer (50 mM Tris pH 8.0, 10 mM EDTA, 0.1 U/ml aprotinin, 1% v/v NP-40). Lysates were clarified by centrifugation at 10 000 g for 10 min at 4°C. One half of the lysates were frozen for further use, and the remainder were combined with NET-GEL (150 mM NaCl, 5 mM EDTA pH 8.0, 50 mM Tris–HCl pH 7.4, 0.05% NP-40, 0.25% gelatin, 0.02% azide) containing 60 µl of 10% protein A–Sepharose CL-4B (Amersham Pharmacia Biotech AB, Sweden). Antibody-bound surface glycoproteins were immunoprecipitated with the 10% protein A–Sepharose overnight at 4°C. Immunoprecipitates were washed with NET-GEL containing 150 mM NaCl, then with NET-GEL containing 0.1% SDS and finally with 10 mM Tris–HCl pH 8.0 (Naim and Roth, 1994), and analyzed on 12% SDS–polyacryamide gels. Monoclonal anti-MV H and anti-MV F antibodies were used in a dilution of 1:200 v/v and the polyclonal anti-VSV antibody was used in a dilution of 1:500 v/v; all antibodies were diluted in DMEM (without serum).
Growth analysis of apical and basolateral viruses
CaCo2 cells were infected with MV, MV mutants or chimera for 2 h at 37°C. Inocula were collected to control virus adsorption. Infected cells were washed twice with PBS, from both domains, and DMEM containing 10% FCS was added, 2 ml from the apical and 2 ml from the basolateral, and incubated for various times at 37°C. At each time point, media from both apical and basolateral chambers were collected, and clarified by centrifugation for 2 min at 8000 r.p.m. in an Eppendorf model 5415C centrifuge. Infectivity was determined by plaque assays on Vero cells.
Acknowledgments
Acknowledgements
We would like to thank Dr Hassan Naim and his group, School of Veterinary Medicine, Hannover, Germany, for material exchange and helpful suggestions, Dr M.G.Roth, UT-Southwestern Medical Center, Dallas, TX, and Dr L.Martin for critical reading of the manuscript. Drs R.Cattaneo, T.Cathomen and B.Roscic-Mrkic made MVΔM and MVΔtails expressing GFP available to us. Dr F.Wild, Institute Pasteur de Lyon, France, supplied MV monoclonal antibodies, Dr E.Sterchi, University of Bern, CaCo2 cells, and Dr H.-P.Hauri, Biocenter, Basel, Switzerland, anti-DPPIV and anti-gp120 antibodies. We thank F.Ochsenbein for the photography. Dr J.-C.Perriard supported E.E. This work was supported by grants from the Swiss National Science Foundation, 31.45900-95 (to M.A.B.) and 31.52417-97 (to J.-C.Perriard), and by the Commission for Technology and Innovation (CTI), 3786.1.
References
- Alfalah M., Jacob,R., Preuss,U., Zimmer,K.P., Naim,H. and Naim,H.Y. (1999) O-linked glycans mediate apical sorting of human intestinal sucrase-isomaltase through association with lipid rafts. Curr. Biol., 9, 593–596. [DOI] [PubMed] [Google Scholar]
- Billeter M.A., Cattaneo,R., Spielhofer,P., Kaelin,K., Huber,M., Schmid,A., Baczko,K. and ter Meulen,V. (1994) Generation and properties of measles virus mutations typically associated with subacute sclerosing panencephalitis. Ann. N Y Acad. Sci., 724, 367–377. [DOI] [PubMed] [Google Scholar]
- Blau D.M. and Compans,R.W. (1995) Entry and release of measles virus are polarized in epithelial cells. Virology, 210, 91–99. [DOI] [PubMed] [Google Scholar]
- Bohn W., Rutter,G., Hohenberg,H., Mannweiler,K. and Nobis,P. (1986) Involvement of actin filaments in budding of measles virus: studies on cytoskeletons of infected cells. Virology, 149, 91–106. [DOI] [PubMed] [Google Scholar]
- Brown D.A. and Rose,J.K. (1992) Sorting of GPI-anchored proteins to glycolipid-enriched membrane subdomains during transport to the apical cell surface. Cell, 68, 533–544. [DOI] [PubMed] [Google Scholar]
- Buechi M. and Bachi,T. (1982) Microscopy of internal structures of Sendai virus associated with the cytoplasmic surface of host membranes. Virology, 120, 349–359. [DOI] [PubMed] [Google Scholar]
- Casanova J.E., Mishumi,Y., Ikehara,Y., Hubbard,A.L. and Mostov,K.E. (1991) Direct apical sorting of rat liver dipeptidylpeptidase IV expressed in Madin–Darby canine kidney cells. J. Biol. Chem., 266, 24428–24432. [PubMed] [Google Scholar]
- Cathomen T., Mrkic,B., Spehner,D., Drillien,R., Naef,R., Pavlovic,J., Aguzzi,A., Billeter,M.A. and Cattaneo,R. (1998a) A matrix-less measles virus is infectious and elicits extensive cell fusion: consequences for propagation in the brain. EMBO J., 17, 3899–3908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cathomen T., Naim,H.Y. and Cattaneo,R. (1998b) Measles viruses with altered envelope protein cytoplasmic tails gain cell fusion competence. J. Virol., 72, 1224–1234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chong L.D. and Rose,J.K. (1994) Interactions of normal and mutant vesicular stomatitis virus matrix proteins with the plasma membrane and nucleocapsids. J. Virol., 68, 441–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Compans R.W. (1995) Virus entry and release in polarized epithelial cells. Curr. Top. Microbiol. Immunol., 202, 209–219. [DOI] [PubMed] [Google Scholar]
- Dörig R.E., Marcil,A., Chopra,A. and Richardson,C.D. (1993) The human CD46 molecule is a receptor for measles virus (Edmonston strain). Cell, 75, 295–305. [DOI] [PubMed] [Google Scholar]
- Fiedler K., Parton,R.G., Kellner,R., Etzold,T. and Simons,K. (1994) VIP36, a novel component of glycolipid rafts and exocytic carrier vesicles in epithelial cells. EMBO J., 13, 1729–1740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujinami R.S., Sissons,J.G. and Oldstone,M.B. (1981) Immune reactive measles virus polypeptides on the cell’s surface: turnover and relationship of the glycoproteins to each other and to HLA determinants. J. Immunol., 127, 935–940. [PubMed] [Google Scholar]
- Fuller S., von Bonsdorff,C.H. and Simons,K. (1984) Vesicular stomatitis virus infects and matures only through the basolateral surface of the polarized epithelial cell line, MDCK. Cell, 38, 65–77. [DOI] [PubMed] [Google Scholar]
- Garoff H., Hewson,R. and Opstelten,D.J.E. (1998) Virus maturation by budding. Microbiol. Mol. Biol. Rev., 62, 1171–1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin D.E. and Bellini,W.J. (1996) Measles Virus. Lippincott-Raven, Philadelphia, PA. [Google Scholar]
- Gut A., Balda,M.S. and Matter,K. (1998) The cytoplasmic domains of a β1 integrin mediate polarization in Madin–Darby canine kidney cells by selective basolateral stabilization. J. Biol. Chem., 273, 29381–29388. [DOI] [PubMed] [Google Scholar]
- Hangartner L. (1998) Development of Measles Virus as a Vector. Diploma thesis, University of Zurich, Zurich, Switzerland, p. 77. [Google Scholar]
- Jacob R. and Naim,H.Y. (1995) Analysis of the putative cleavage site in human lactase-phlorizin hydrolase. Biochem. Soc. Trans., 23, 305S. [DOI] [PubMed] [Google Scholar]
- Jacob R., Preuss,U., Panzer,P., Alfalah,M., Quack,S., Roth,M.G., Naim,H. and Naim,H.Y. (1999) Hierarchy of sorting signals in chimeras of intestinal lactase-phlorizin hydrolase and the influenza virus hemagglutinin. J. Biol. Chem., 274, 8061–8067. [DOI] [PubMed] [Google Scholar]
- Laemmli U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685. [DOI] [PubMed] [Google Scholar]
- Lin S., Naim,H.Y., Rodriguez,A.C. and Roth,M.G. (1998) Mutations in the middle of the transmembrane domain reverse the polarity of transport of the influenza virus hemagglutinin in MDCK epithelial cells. J. Cell Biol., 142, 51–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisanti M.P. and Rodriguez-Boulan,E. (1991) Polarized sorting of GPI-linked proteins in epithelia and membrane microdomains. Cell Biol. Int. Rep., 15, 1023–1049. [DOI] [PubMed] [Google Scholar]
- Louvard D., Kedinger,M. and Hauri,H.P. (1992) The differentiating intestinal epithelial cell: establishment and maintenance of functions through interactions between cellular structures. Annu. Rev. Cell Biol., 8, 157–195. [DOI] [PubMed] [Google Scholar]
- Maisner A., Klenk,H. and Herrler,G. (1998) Polarized budding of measles virus is not determined by viral surface glycoproteins. J. Virol., 72, 5276–5278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manié S.N., Debreyne,S.V., Vincent,S. and Gerlier,D. (2000) Measles virus structural components are enriched into lipid raft microdomains: a potential cellular location for virus assembly. J. Virol., 74, 305–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matlin K.S. (1992) W(h)ither default? Sorting and polarization in epithelial cells. Curr. Opin. Cell Biol., 4, 623–628. [DOI] [PubMed] [Google Scholar]
- Matter K., Brauchbar,M., Bucher,K. and Hauri,H.P. (1990) Sorting of endogenous plasma membrane proteins occurs from two sites in cultured human intestinal epithelial cells (Caco-2). Cell, 60, 429–437. [DOI] [PubMed] [Google Scholar]
- Mebatsion T., Konig,M. and Conzelmann,K.K. (1996) Budding of rabies virus particles in the absence of the spike glycoprotein. Cell, 84, 941–951. [DOI] [PubMed] [Google Scholar]
- Mebatsion T., Weiland,F. and Conzelmann,K.K. (1999) Matrix protein of rabies virus is responsible for the assembly and budding of bullet-shaped particles and interacts with the transmembrane spike glycoprotein G. J. Virol., 73, 242–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyer S.A., Baker,S.C. and Horikami,S.M. (1990) Host cell proteins required for measles virus reproduction. J. Gen. Virol., 71, 775–783. [DOI] [PubMed] [Google Scholar]
- Mrkic B., Odermatt,B., Klein,M., Billeter,M.A., Pavlovic,J. and Cattaneo,R. (2000) Lymphatic dissemination and comparative pathology of recombinant measles virus in genetically modified mice. J. Virol., 74, 1364–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naim H.Y. and Roth,M.G. (1993) Basis for selective incorporation of glycoproteins into the influenza virus envelope. J. Virol., 67, 4831–4841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naim H.Y. and Roth,M.G. (1994) SV40 virus expression vectors. Methods Cell Biol., 43, 113–136. [DOI] [PubMed] [Google Scholar]
- Naniche D., Varior-Krishnan,G., Cervoni,F., Wild,T.F., Rossi,B., Rabourdin-Combe,C. and Gerlier,D. (1993) Human membrane cofactor protein (CD46) acts as a cellular receptor for measles virus. J. Virol., 67, 6025–6032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens R.J., Dubay,J.W., Hunter,E. and Compans,R.W. (1991) Human immunodeficiency virus envelope protein determines the site of virus release in polarized epithelial cells. Proc. Natl Acad. Sci. USA, 88, 3987–3991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeiffer S., Fuller,S.D. and Simons,K. (1985) Intracellular sorting and basolateral appearance of the G protein of vesicular stomatitis virus in Madin–Darby canine kidney cells. J. Cell Biol., 101, 470–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radecke F., Spielhofer,P., Schneider,H., Kaelin,K., Huber,M., Dötsch,C., Christiansen,G. and Billeter,M.A. (1995) Rescue of measles viruses from cloned DNA. EMBO J., 14, 5773–5784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Boulan E. and Powell,S.K. (1992) Polarity of epithelial and neuronal cells. Annu. Rev. Cell Biol., 8, 395–427. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Boulan E. and Sabatini,D.D. (1978) Asymmetric budding of viruses in epithelial monolayers: a model system for study of epithelial polarity. Proc. Natl Acad. Sci. USA, 75, 5071–5075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth M.G. and Compans,R.W. (1981) Delayed appearance of pseudotypes between vesicular stomatitis virus influenza virus during mixed infection of MDCK cells. J. Virol., 40, 848–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth M.G., Fitzpatrick,J.P. and Compans,R.W. (1979) Polarity of influenza and vesicular stomatitis virus maturation in MDCK cells: lack of a requirement for glycosylation of viral glycoproteins. Proc. Natl Acad. Sci. USA, 76, 6430–6434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salas P.J., Rodriguez,M.L., Viciana,A.L., Vega-Salas,D.E. and Hauri,H.P. (1997) The apical submembrane cytoskeleton participates in the organization of the apical pole in epithelial cells. J. Cell Biol., 137, 359–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanderson C.M., Wu,H.H. and Nayak,D.P. (1994) Sendai virus M protein binds independently to either the F or the HN glycoprotein in vivo.J. Virol., 68, 69–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanderson C.M., Avalos,R., Kundu,A. and Nayak,D.P. (1995) Interaction of Sendai viral F, HN and M proteins with host cytoskeletal and lipid components in Sendai virus-infected BHK cells. Virology, 209, 701–707. [DOI] [PubMed] [Google Scholar]
- Scheiffele P., Peranen,J. and Simons,K. (1995) N-glycans as apical sorting signals in epithelial cells. Nature, 378, 96–98. [DOI] [PubMed] [Google Scholar]
- Simons K. and Garoff,H. (1980) The budding mechanisms of enveloped animal viruses. J. Gen. Virol., 50, 1–21. [DOI] [PubMed] [Google Scholar]
- Simons K. and Ikonen,E. (1997) Functional rafts in cell membranes. Nature, 387, 569–572. [DOI] [PubMed] [Google Scholar]
- Simons K. and Wandinger-Ness,A. (1990) Polarized sorting in epithelia. Cell, 62, 207–210. [DOI] [PubMed] [Google Scholar]
- Spielhofer P., Bachi,T., Fehr,T., Christiansen,G., Cattaneo,R., Kaelin,K., Billeter,M.A. and Naim,H.Y. (1998) Chimeric measles viruses with a foreign envelope. J. Virol., 72, 2150–2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tashiro M., McQueen,N.L., Seto,J.T., Klenk,H.D. and Rott,R. (1996) Involvement of the mutated M protein in altered budding polarity of a pantropic mutant, F1-R, of Sendai virus. J. Virol., 70, 5990–5997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ter Meulen V. and Billeter,M.A. (1995) Measles Virus. Curr. Top. Microbiol. Immunol., 191. Springer-Verlag, Berlin, Germany. [Google Scholar]
- Thomas D.C. and Roth,M.G. (1994) The basolateral targeting signal in the cytoplasmic domain of glycoprotein G from vesicular stomatitis virus resembles a variety of intracellular targeting motifs related by primary sequence but having diverse targeting activities. J. Biol. Chem., 269, 15732–15739. [PubMed] [Google Scholar]
- Udem S.A. (1984) Measles virus: conditions for the propagation and purification of infectious virus in high yield. J. Virol. Methods, 8, 123–136. [DOI] [PubMed] [Google Scholar]
- Udem S.A. and Cook,K.A. (1984) Isolation and characterization of measles virus intracellular nucleocapsid RNA. J. Virol., 49, 57–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Meer G. and Simons,K. (1988) Lipid polarity and sorting in epithelial cells. J. Cell. Biochem., 36, 51–58. [DOI] [PubMed] [Google Scholar]
- Wild T.F. and Buckland,R. (1995) Functional Aspects of Envelope-Associated Measles Virus Proteins. Springer-Verlag KG, Berlin, Germany. [DOI] [PubMed] [Google Scholar]
- Yeaman C., Le Gall,A.H., Baldwin,A.N., Monlauzeur,L., Le Bivic,A. and Rodriguez-Boulan,E. (1997) The O-glycosylated stalk domain is required for apical sorting of neurotrophin receptors in polarized MDCK cells. J. Cell. Biol., 139, 929–940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida T., Nagai Y’Yoshii,S., Maeno,K. and Matsumoto,T. (1976) Membrane (M) protein of HVJ (Sendai virus): its role in virus assembly. Virology, 71, 143–161. [DOI] [PubMed] [Google Scholar]