Abstract
Pleural effusion is an accumulation of fluid in the pleural space that is classified as transudate or exudate according to its composition and underlying pathophysiology. Empyema is defined by purulent fluid collection in the pleural space, which is most commonly caused by pneumonia. A lung abscess, on the other hand, is a parenchymal necrosis with confined cavitation that results from a pulmonary infection. Pleural effusion, empyema, and lung abscess are commonly encountered clinical problems that increase mortality. These conditions have traditionally been managed by antibiotics or surgical placement of a large drainage tube. However, as the efficacy of minimally invasive interventional procedures has been well established, image-guided small percutaneous drainage tubes have been considered as the mainstay of treatment for patients with pleural fluid collections or a lung abscess. In this article, the technical aspects of image-guided interventions, indications, expected benefits, and complications are discussed and the published literature is reviewed.
Keywords: Pleural effusion, empyema, lung abscess, malignant pleural effusion, interventional radiology
PLEURAL EFFUSION AND EMPYEMA
Approximately 1.5 million patients are diagnosed with pleural effusion each year in the United States.1 Pleural effusion is defined as abnormal fluid collection in the pleural space. The pleural space is normally filled with ~5 to 10 mL of serous fluid, which is secreted mainly from the parietal pleura at a rate of 0.01 mL/kg/h and absorbed through the lymphatics in the parietal pleura.2 In certain clinical conditions, the balance between the secretion and absorption can be disturbed and the fluid starts accumulating in the pleural space.
Pleural effusion is classically divided into transudate and exudate based on Light's criteria (Table 1).3 In transudate, fluid accumulates in the pleural space due to increased hydrostatic pressure or decreased oncotic pressure across the intact capillary beds of pleural membranes.4 However, in exudate, the capillary beds themselves are diseased and its increased permeability results in fluid leak into the pleural space.5 In adults, congestive heart failure and liver cirrhosis are the most common causes of transudative pleural effusions. On the other hand, pneumonia, malignant pleural disease, pulmonary embolism, and gastrointestinal disease account for ~90% of exudative pleural effusions.6 In the pediatric population, congenital heart disease, pneumonia, and malignancy are the most common causes of pleural effusions.2
Table 1.
Differentiation between Transudate and Exudate
| Transudate | Exudate | |
|---|---|---|
| Appearance | Serous | Cloudy |
| Leukocyte count | < 10,000/mm3 | > 50,000/mm3 |
| pH | > 7.2 | < 7.2 |
| Protein | < 3.0 g/dL | > 3.0 g/dL |
| Ratio of protein in pleural fluid to serum | < 0.5 | > 0.5 |
| Lactate dehydrogenase (LDH) | < 200 IU/L | > 200 IUL |
| Ratio of LDH in pleural fluid to serum | < 0.6 | > 0.6 |
| Glucose | ≥ 60 mg/dL | < 60 mg/dL |
LDH, Lactate dehydrogenase.
Parapneumonic effusion is referring to a pleural fluid collection resulting from bacterial pneumonia, lung abscess, and bronchiectasis.7 The most common source of exudative effusion is parapneumonic effusion.8 Parapneumonic effusions are usually resolved with appropriate treatment. However, it may be infected and develop an empyema. Empyema is a collection of purulent fluid in the pleural space. The most common cause of empyema is pneumonia. Lung abscess, bronchopleural fistula, esophageal perforation, postsurgical complications, and trauma may also result in empyema.6 There are three stages in the evolution of empyema. The first stage is the exudative stage where only a small amount of sterile fluid is accumulated in the pleural space. The second stage is the transitional fibropurulent stage. This stage is characterized by higher neutrophil counts and fibrin deposition due to an infection. In this stage, the fluid tends to be loculated (Fig. 1). The final stage is the organized stage where fibroblasts grow into the pleural walls and produce a thick pleural peel that prevents the lung from reexpansion (Fig. 2).9
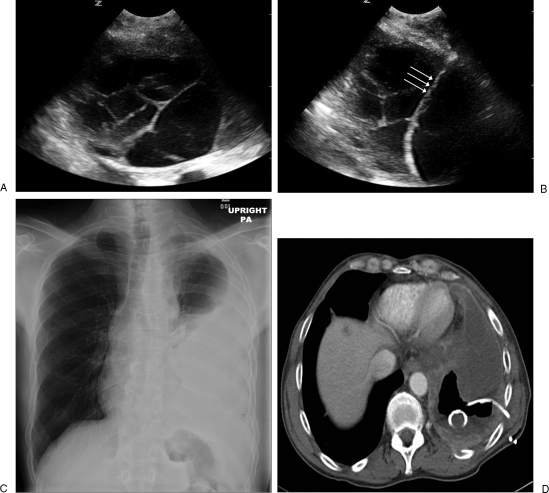
Figure 1.
(A) An ultrasound image shows a multiloculated pleural effusion. (B) A guidewire (triple arrows) is inserted through the initial access needle into the pleural effusion for drainage catheter placement. (C) A chest radiograph shows a large amount of left-sided pleural effusion. (D) An axial computed tomography (CT) image shows large amount of pleural effusion with a 10F pigtail catheter placed percutaneously under CT guidance. The posterior part of the effusion is removed and replaced with air.
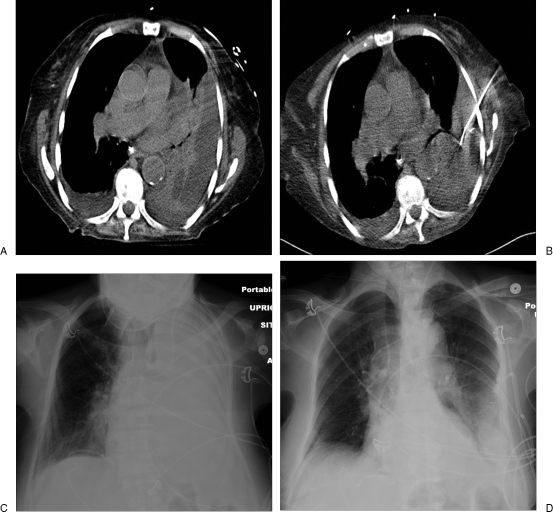
Figure 2.
(A) An axial computed tomography (CT) image shows a complicated pleural effusion with inner septations and adjacent atelectatic lung parenchyma. (B) A 10F nontunneled pigtail catheter is placed percutaneously under CT guidance. (C) A chest radiograph shows a complete opacification in the left hemothorax due to pleural effusion. (D) Follow-up chest radiograph after placement of pigtail drainage catheter shows decreased effusion with reexpanded lung parenchyma.
The symptoms of pleural effusion include dyspnea, pleuritic chest pain, cough, fever, chills, and weight loss. Clinical manifestations of pleural effusion are largely dependent on the underlying lung disease. Physical examination findings of pleural effusion itself can be subtle or normal if the amount of fluid is less than 300 mL.2 Furthermore, if the respiratory function and the compliance of the lung and chest walls are normal, it is rare for pleural effusion to develop significant hypoxemia. This finding is probably due to a decrease in ventilation and perfusion at the same time in the compressed lung parenchyma. Cardiac function, however, can be affected by a large amount of pleural effusion due to diminished cardiac ability for distension and filling. A large amount of fluid in the left pleural space may cause clinical manifestations similar to cardiac tamponade.6
The chest radiograph has been the initial diagnostic tool for the detection and evaluation of pleural effusion. For the detection of pleural effusion, more than 175 mL of fluid is required; this can obliterate the costophrenic angle on upright posteroanterior chest radiograph.10 However, the lateral decubitus chest radiograph can demonstrate as little as 10 mL of free pleural fluid.11 Ultrasound is useful for the evaluation of a small amount of pleural fluid and as guidance for the thoracentesis or drainage catheter placement.12 Over the last decade, ultrasound has played a major role in the management of patients with pleural effusion. The advent of affordable and portable ultrasound has made bedside procedures possible not only in hospital rooms, but in intensive care units and emergency departments.13,14 On the other hand, computed tomography (CT) is the imaging study of choice for the evaluation of pleural pathology and underlying lung disease. CT is more accurate to separate empyema from underlying compressed lung than a plain chest radiograph. CT with contrast enhancement may differentiate empyema from lung abscess and transudate from exudate.15 As a guidance tool, CT is especially useful to locate a skin entry site for thoracentesis or drainage catheter placement when ultrasound has a limited role due to adjacent bony structures, large patients, or air in the lung parenchyma.16,17
The goal in the management of pleural effusion is to provide symptomatic relief by removing fluid from the pleural space and to allow the treatment of the underlying disease. The management options often depend on the type of pleural effusion, stage in the evolution, and underlying disease.2 The first step for the treatment of pleural effusion is to determine whether the fluid is a transudate or an exudate (Table 1). Light and Rodriguez have proposed a classification and treatment scheme for pleural effusion based on the amount of fluid, gross and biochemical characteristics of fluid, and whether the fluid is loculated.18 According to their classification, a transudate is considered as uncomplicated effusion, which can be managed by conservative treatment or antibiotics alone. However, an exudative effusion or a large amount of loculated effusion, which is classified as complicated effusion, should be managed by drainage.9,18 Complicated effusions also include empyema, malignant effusion, and hemothorax. For a complicated effusion, it is important to remove the pleural fluid to expand lung parenchyma for a good prognosis.19 The treatment options include therapeutic thoracentesis, drainage catheter placement, fibrinolytic therapy, pleurodesis, and surgery.
THORACENTESIS
Thoracentesis is a basic and valuable procedure not only to obtain a fluid sample for differentiating transudate from exudate, but to remove the fluid in a patient with a large volume of effusion for symptomatic relief. The most common indication of diagnostic thoracentesis is a fluid in the pleural space more than 10 mm in thickness on lateral decubitus chest radiograph with unknown etiology.20 If there is an obvious underlying disease that is likely to cause the effusion, thoracentesis can be postponed until the underlying process is managed first. For instance, bilateral symmetrical pleural effusion in a patient with known congestive heart failure without fever or chest pain should be treated with diuretics before trying thoracentesis. Approximately 75% of pleural effusion resulting from congestive heart failure is resolved within 2 days by diuretics.21 However, if the pleural effusion in a patent with congestive heart failure is persistent for more than 3 days, then thoracentesis should be performed. If the patient has a shortness of breath at rest, up to 1500 mL of fluid should be removed to relieve the symptom.20
The procedure may be performed at bedside without image guidance by an experienced operator. However, it is generally recommended to use ultrasonographic guidance to obtain a fluid sample from a small or loculated effusion and to avoid potential complications. Ultrasound saves time and improves the first-puncture success of thoracentesis.13 A study by Diacon et al demonstrated that a puncture site selection with bedside ultrasound increased the yield and decreased the risk of thoracentesis compared to that without image guidance.22 In patients who are receiving positive-pressure ventilation, the procedure should be performed with caution due to the risk of tension pneumothorax by puncturing lung parenchyma. In this situation, ultrasound may be essential.
Complications of thoracentesis include pneumothorax, hemothorax, reexpansion pulmonary edema, and organ laceration.23 The incidences of pneumothorax and hemothorax are 2–6% and 1%, respectively. Only a half of pneumothorax cases require chest tube insertion.24 Reexpansion pulmonary edema or organ laceration is a rare complication.25
To avoid bleeding complications in patients with coagulopathy or thrombocytopenia, the procedure can be performed after transfusion of fresh frozen plasma or platelets.
Thoracentesis under ultrasound guidance is usually performed with the patient in a sitting position on the edge of the bed, leaning forward with the patient's arms resting on a bedside table. When the patient is not able to be placed in a sitting position, the lateral decubitus or supine position can be used. Preprocedural ultrasound evaluation can localize the pleural fluid pocket and skin entry site at the posterior intercostal space, which is prepared and draped in a sterile manner. A skin entry site is then anesthetized using 1% lidocaine with epinephrine. The access site should be along the superior margin of the rib to avoid the injury to the intercostal artery, which runs along the inferior border of the rib. After making a small skin incision, an 18-gauge over-the-needle sheath is then advanced into the pleural fluid under continuous ultrasound guidance. A 4- or 5F Valved One-Step Centesis® catheter (Merit Medical Systems, South Jordan, UT) is one of the sheathed needles that is available either in a Luer-locking or slip-fit outer sheath. The outer sheath of this needle has an integrated self-sealing valve to minimize the risk of air infiltration and fluid leakage. The other sheathed needle is a 4- or 5F Yueh needle (Cook, Bloomington, IN), which is available in a slip-fit outer sheath. There are multiple side holes at the distal portion of the sheath to facilitate the fluid aspiration. Once the needle tip is safely positioned inside the fluid pocket, the outer soft sheath can be advanced over the metal stylet needle, which is later removed. As the metal stylet is removed, cover the open hub of the sheath if there is no valve. A three-way stopcock is then connected to the hub and a fluid sample can be obtained without the risk of air introduction into the pleural space. Fifty milliliters of fluid are usually required for diagnostic thoracentesis. A fluid sample should be placed immediately in the appropriate specimen containers and sent for an analysis; the sample should include protein and LDH levels for comparison to those in serum (Table 1).
If therapeutic thoracentesis is necessary, the sheath is attached to an extension tubing system, which is connected to a vacuum bottle. In general, removal of < 1500 mL pleural effusion is recommended to avoid the risk of reexpansion pulmonary edema.25 Once thoracentesis is completed, the sheath is removed at end expiration; the skin entry site should be covered using an antimicrobial ointment with sterile occlusive dressing.
Continuous ultrasound guidance is essential for a safe thoracentesis with a higher success rate. Grogan et al demonstrated a significant reduction in the pneumothorax rate when thoracentesis was performed under ultrasound guidance (0% vs ~29%).26 Raptopoulos et al also reported a similar reduction in the rate of pneumothorax (18% vs 3%).27 With ultrasound guidance, the success rate in thoracentesis is increased even after unsuccessful blind thoracentesis. Several studies have reported up to an 88% success rate after failed blind thoracentesis.13
A routine chest radiograph is usually obtained immediately after the procedure to exclude a possible complication, such as pneumothorax. However, a previous prospective study by Petersen et al showed that the chest radiograph had only a limited value in the evaluation of postprocedural complications.24 Therefore, it is generally not recommended to obtain a chest radiograph immediately after thoracentesis in the absence of suspicion or clinical indication of complications.
NONTUNNELED PIGTAIL DRAINAGE CATHETER PLACEMENT
Complicated pleural effusion refers to fluid collections that are not resolved without drainage of the pleural fluid. Exudate, empyema, and hemothorax are considered as complicated effusions; they are the most common indications for drainage catheter placement.19 Other indications include malignant effusion, recurrent effusion, chylothorax, pneumothorax, hemopneumothorax, and leakage into the pleural space from esophageal or gastric rupture.
Empyema is defined as infected fluid collection in the pleural space; it is associated with significant morbidity and mortality in adults and children.28 Along with antibiotic therapy and treatment of underlying disease, early and complete drainage of infected fluid is essential in the successful management of empyema. Traditionally, empyema has been managed by the surgical placement of large (22F–34F) drainage catheters in the pleural space. However, small drainage catheters have successfully been used in the management of empyema. In one study of 103 patients with empyema, 80 patients were successfully treated by placing small (< 14F) drainage catheters under ultrasound or CT guidance.29 This result is comparable to those of previous studies using a large catheter placement for treating empyema.6
The most recent prospective study by Rahman et al also demonstrated that small (< 14F) drainage catheters were as effective as large (> 14F) catheters in the management of infected pleural fluid collections.30 There was no significant difference in clinical outcome in patients with different-sized drainage catheters. In this study, patients with smaller drainage catheters experienced much less pain than those with large catheters, which were mainly placed by blunt dissection.
In general, recurrent chronic pleural infection is considered as a contraindication for long-term indwelling catheter placement. The treatment of choice, therefore, has been open surgical drainage of infected fluid. Davies et al, however, presented cases with chronically infected pleural effusions that were successfully treated by small (12F) ambulatory indwelling pigtail catheters for 8–21 months.31 The authors also stated that small long-term drainage catheters were especially beneficial for patients who were not surgical candidates.
Pierrepoint et al compared the clinical outcome in 24 pediatric patients with empyema who were treated by surgical open thoracotomy with pleural debridement, conventional stiff chest drain catheter, or pigtail drain catheter placement.32 Pigtail catheters were placed under image guidance and were considerably smaller than conventional stiff drains. Patients who were treated by pigtail catheters or surgical thoracotomy showed better outcomes compared to those with conventional stiff drains. The outcomes included shorter drain time, earlier clinical improvement, earlier resolution of fever, and earlier discharge. Because pigtail catheter placement was less painful, less uncomfortable, and cosmetically favorable, the authors concluded that pigtail drainage catheters were preferable to thoracotomy.
There is no absolute contraindication for nontunneled pigtail drainage catheter placement. Relative contraindications include coagulopathy and thrombocytopenia, which should be corrected before the procedure.
After explaining details of the procedure, risk, and benefits to the patient or next of kin, written informed consent is obtained. The procedure is performed with the patient in a sitting, lateral decubitus, or semidecubitus position. After localization of the pleural fluid and skin entry site using ultrasound, the patient is prepared and draped in a sterile manner. Afterward, local anesthesia is performed using 1% lidocaine with epinephrine and a small skin incision is made. Access to the pleural fluid collection using an 18-gauge entry needle under continuous ultrasound guidance is similar to that of thoracentesis. If ultrasound is not able to identify fluid collection due to its location, surrounding anatomic structures, or loculated nature, CT can be an alternative option for localization of fluid and procedure guidance. Once the needle tip is safely positioned inside the pleural space, a 0.038-inch Newton-J or 0.035-inch Bentson (AngioDynamics, Queensbury, NY) guidewire is advanced through the needle into the pleural fluid collection. Then the needle is exchanged over a guidewire for an 8F or 10F dilator, which is utilized to dilate the skin and soft tissue tract. The dilator is then exchanged for an 8F or 10F locking pigtail nontunneled drainage catheter, which is advanced over a guidewire and coiled in the pleural space (Fig. 1,2). The catheter is secured to the skin using a suture at the exit site and covered by sterile dressing. The external tip of the catheter is connected to a sterile pleural drainage system, such as a Pleur-evac® (Teleflex Medical, Research Triangle Park, NC) plastic compartmentalized drainage system.
Pleural fluid drainage should to be started immediately and up to 1500 mL of fluid can be removed. After removing the pleural fluid, a chest radiograph or postprocedural CT scan should be obtained to confirm the appropriate position of the pigtail catheter and evaluate possible complications including pneumothorax. Other complications include hemothorax, intercostal artery injury, perforation of major organs, perforation of major arteries, intercostal neuralgia due to injury to the neurovascular bundles, subcutaneous emphysema, and reexpansion pulmonary edema.33
The drainage catheter should be managed by periodic flushing with sterile saline to maintain the catheter patency. The drainage catheter for empyema should be left in place until the volume of daily output is less than 50 mL and until the draining fluid becomes clear yellow.6 On a follow-up chest radiograph, if the lung is reexpanded and the patient's clinical status is improved, then the drainage catheter can be safely removed.
TUNNELED DRAINAGE CATHETER PLACEMENT
Malignant pleural effusion is the second most common cause of an exudative pleural effusion and the most common cause in patients over 60 years of age.34 The fluid may accumulate due to overproduction from diseased pleura, obstruction of lymphatic channels, or atelectasis of adjacent lung. It is associated with significant morbidity and mortality. The 30-day mortality is ~29–50% with a median survival of 3–12 months. Approximately 50% of patients with disseminated malignancy eventually develop malignant effusion in the course of the disease.35,36,37
The etiology is lung cancer, breast cancer, lymphoma, ovarian cancer, and gastric cancer. Among these cancers, lung and breast cancers account for ~75% of malignant pleural effusion. In patients with malignant pleural effusion, dyspnea is the most common symptom that compromises their quality of life. Other clinical symptoms include cough and chest discomfort.38
Treatment of malignant effusion is usually palliative and the main goal of management is rapid and effective relief of symptoms with minimal discomfort or inconvenience, minimal disruption of daily activity, and cost effectiveness. Traditional treatment options for malignant effusion are repeated thoracentesis, chest tube drainage with pleurodesis, pleuroperitoneal shunt, pleurectomy, and thoracoscopy.39
Thoracentesis may be the first and simple option not only for diagnosis, but for immediate symptomatic relief. However, repeated thoracentesis is not an optimal option for palliative management of rapidly reaccumulating malignant effusion. Thoracentesis only provides short-term relief of symptoms and requires repeated visits to the hospital for the procedure. Therefore, thoracentesis may be an option only for patients with expected response to systemic therapy, short survival expectance, or slow reaccumulation of effusion.40 Chest tube insertion with pleurodesis and thoracoscopy usually require a long-term hospitalization of ~4–7 days, which is not desirable or cost effective as a palliative management in patients with short-life expectancy.38
As an alternative treatment option, a tunneled drainage catheter has been successfully used for a long-term management of malignant pleural effusion. Tunneled drainage catheters have several advantages over other treatment options. Tunneled drainage catheter placement is safe, comfortable, and less expensive. It makes long-term outpatient management of effusion and symptoms possible.34,38,39,40,41
In 1997, the United States Food and Drug Administration has approved the Pleurx® catheter (Denver Biomedical, Golden, CO) for management of malignant pleural effusion. The catheter is 15.5F in diameter, 66 cm in length, and made of silicone, which is soft and comfortable for long-term use. There are multiple side holes along the distal ⅓ of the catheter, which allow easy drainage of the fluid with low risk of obstruction. The risks for catheter dislodgement, tunnel or pleural infections, and pericatheter fluid leakage are low because of fibrous tissue developing around a polyester cuff at the proximal portion of the catheter. The risks for transcatheter fluid leakage and pneumothorax are also low due to one-way valve system at the proximal hub of the catheter (Fig. 3).
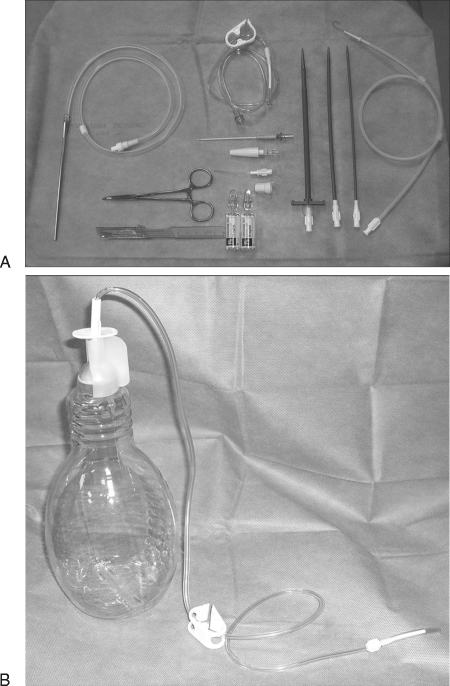
Figure 3.
(A) Complete Pleurx® kit showing Pleurx® catheter with a metal tunneler, guidewire, peel-away sheath, dilators, access needle, connecting tube, and cap. (B) Drainage bottle with connector. The end of the connecting tube fits in the one-way valve at the hub of the Pleurx® catheter.
The procedure is similar to that of nontunneled catheter placement for the most part. After sterile preparation and draping, local anesthesia is achieved using 1% lidocaine with epinephrine. Through a small skin incision, an 18-gauge needle is inserted into the pleural space along the superior margin of the low rib slightly above the diaphragm under ultrasound guidance. A lateral approach with slight posterior direction of the needle is usually recommended to position the catheter tip in the posterior costophrenic angle for free-flowing fluid. From 5–8 cm anterior and inferior to the initial entry site, the catheter is tunneled through the subcutaneous space. The needle is then exchanged over a 0.038 inch Newton-J or 0.035-inch Bentson (AngioDynamics, Queensbury, NY) guidewire for a dilator and the tract is serially dilated to 16F. The dilator is again exchanged for a 16F peel-away sheath through which a tunneled catheter is advanced into the pleural space. The initial pleural puncture site and tunnel exit site are closed using a suture and covered with sterile dressing. A final fluoroscopic spot image or routine chest radiograph should be obtained to evaluate the catheter position, residual fluid, pneumothorax, and underlying lung parenchyma. After the procedure, up to 1500 mL of fluid can be safely removed using wall suction to achieve immediate symptomatic relief without reexpansion pulmonary edema. The catheter is subsequently connected to the special vacuum bottle system.
Pollack et al reported the successful placement of 31 Pleurx® catheters in 28 patients with malignant pleural effusion in 2001.34 Symptomatic relief was achieved in 94% and 91% at 48 hours and 30 days, respectively. Malignant pleural effusion was successfully controlled in 90% of the patients; spontaneous pleurodesis occurred in 42% of the patients and effective control of effusion without pleurodesis in 48%. There were three complications in two patients, which included extrapleural migration of the catheter with tumor spread through the tract and infection of the pleural effusion. Most patients experienced transient pain after the first full drainage of effusion.
In 2006, Tremblay et al, in a larger retrospective study with 250 Pleurx® catheter placements in 233 patients with malignant pleural effusion, demonstrated complete and partial symptom control in 38.8% and 50%, respectively, at 2-week follow-up.40 Only 3.6% of patients showed no response to the Pleurx® catheters. Spontaneous pleurodesis occurred in 42.9%. Repeat procedures were performed in 9.9%. Complications included loculation of fluid, empyema, cellulitis, dislodgement, bleeding, tumor seeding, and extrapleural migration of catheter. Symptomatic loculation of fluid was the most common complication, which occurred in 8.4%.
After failed pleurodesis, recurrent malignant effusion can also be effectively managed by Pleurx® catheter placement. In 2010, a retrospective study of 63 cases of failed pleurodesis showed successful management of recurrent malignant pleural effusion in 95%.41
If the lung is entrapped by tumor encasement or thick pleural fibrosis, pleurodesis is generally not indicated because the pleural apposition cannot be achieved. Therefore, pleurodesis via thoracoscopic assistance or thoracostomy tube is usually recommended to patients with fully reexpandable lung parenchyma. Ohm et al tried to find an alternative option for the management of malignant pleural effusion in patients with entrapped lung syndrome.42 In their study of 34 patients with trapped lung, spontaneous pleurodesis developed in 12% of patients with Pleurx® catheter placement; the catheters were found to be easy to use, less expensive, and provided a better quality of life. In another smaller study with 11 patients with trapped lung syndrome, Pien et al reported symptomatic benefit, defined as improved dyspnea and exercise tolerance in 10 patients following Pleurx® catheter placement.43
Spontaneous pleurodesis developed after Pleurx® catheter placement in over 40%, mostly within one month of the procedure.34 Intrapleural instillation of a sclerosing agent can also be considered at an outpatient setting to facilitate pleurodesis and removal of the catheter. Once the drainage volume decreases to 50 mL for three consecutive drainage attempts at 3-day intervals without evidence of symptoms or reaccumulation of fluid on chest radiograph, the drainage catheter can be removed.38
INTRAPLEURAL FIBRINOLYTIC THERAPY
In conjunction with drainage catheter placement, intrapleural administration of fibrinolytic agents has been used to increase the drain in patients with multiloculated parapneumonic effusion or empyema. In the transitional fibropurulent stage of empyema, drainage catheter placement only is usually not sufficient for the complete removal of fluid due to its multiloculated nature. Fibrinolytics are therefore used to improve the drainage of complicated pleural fluid by lysis of fibrinous septations.28,44 Streptokinase, urokinase, and recombinant tissue-type plasminogen activator (r-tPA) are commonly used fibrinolytics.
In a small retrospective study in 1996, Temes et al described the use of streptokinase or urokinase for fibrinolysis in 26 patients with empyema. A complete and partial resolution of symptoms and radiographic findings were achieved in 62% and 8%, respectively. Bleeding complications occurred in 4%.45 In 2009, Zuckerman et al reported the feasibility and effectiveness of fibrinolytic therapy with r-tPA in 25 patients with loculated parapneumonic effusion. A complete clinical and radiographic resolution of loculated effusion was achieved in 72% of cases. They injected 6 mg of r-tPA diluted in 50 mL of saline through the existing drainage catheter, which was clamped for 2 hours. Afterward, the catheter was unclamped and suctioned for 8–12 hours. There were no bleeding complications.46
Fibrinolytics may also be useful in the drainage of multiloculated malignant pleural effusion. Davies et al reported the use of streptokinase in 10 patients with malignant multiloculated pleural effusion in a small retrospective study in 1999. There was a significant increase in the drainage pleural fluid and improvement of radiographic findings after instillation of streptokinase in all patients.47
However, recently the Multicenter Intrapleural Sepsis Trial, the largest double-blinded randomized controlled study in the United Kingdom, demonstrated that intrapleural administration of streptokinase in patients with infected pleural effusion was not effective in reducing mortality, the need for surgical drainage, or the length of the hospital stay. The authors concluded that intrapleural streptokinase should be avoided in pleural infection.48 In a meta-analysis of five randomized trials comparing intrapleural fibrinolytic agents with placebo, Tokuda et al reported that there was no evidence that the intrapleural administration of fibrinolytic agents reduced mortality and the need for surgery of patients with empyema and complicated parapneumonic effusion.49
PLEURODESIS
Pleurodesis is the procedure that obliterates the pleural space to prevent pleural effusion from reaccumulation. Approximately ⅔ of patients with malignant pleural effusion do not respond to therapeutic thoracentesis or drainage catheters. Treatment options for these patients include pleurodesis via chest tube, video-assisted thoracoscopy with pleurodesis, open thoracotomy with pleurectomy, and pleuroperitoneal shunting. Among these options, pleurodesis via chest tube is the best method to control recurrent malignant pleural effusion.50
Pleurodesis is usually performed in patients with recurrent malignant pleural effusion after one or two consecutive drainages of fluid by thoracentesis or drainage catheters. Early pleurodesis immediately after initial diagnosis of malignant pleural effusion may also be performed to reduce the risk of developing a trapped lung. The recently published International Survey of Pleurodesis Practice (ISPP) reported that the pleurodesis practice was widely variable in its techniques and effectiveness with an average success rate of ~66%.51
The size of chest tubes for pleurodesis varies from less than 14F to over 32F. Traditionally, pleurodesis has been performed via a conventional large chest tube that results in pain, discomfort, and prolonged hospital stay. However, small drainage catheters have also been proven to be as effective as large chest tubes for pleurodesis. It results in less pain and discomfort during and after the procedure, and makes ambulatory outpatient-based pleurodesis possible.52
Sclerosing agents for pleurodesis include talc, tetracycline, doxycycline, and bleomycin. Although there has been no consensus worldwide on the best sclerosing agent to date, ISPP showed that talc was perceived as the most effective agent and therefore most commonly used by 68% of survey respondents.51 A recent meta-analysis of 36 randomized controlled studies also confirmed that talc was the most effective agent compared with other agents. However, talc was also found to be associated with more pain, nausea, and fever. Respiratory failure was also observed more frequently in patients with talc pleurodesis than other sclerosing agents.53
The pleural cavity should be dried as much as possible for a better apposition of visceral and parietal pleura to improve the efficacy of pleurodesis. Daily output of a drainage catheter < 150 mL was traditionally recommended as an indicator for the initiation of pleurodesis. However, full expansion of the lung on chest radiograph may also be used as an indicator regardless of the amount of daily output. Once the pleural fluid is completely removed, a sclerosing agent is instilled in the pleural cavity through the drainage catheter, which is then clamped for ~1– 4 hours. Rotation of the patients following instillation of a sclerosing agent had been thought to be helpful for dispersion of the agent in the pleural space. However, a study by Mager et al reported that rotation of the patients did not affect the overall dispersion of talc suspension and there was no difference in success rates of pleurodesis between patients with and without rotation.54 Pleurodesis with tetracycline was also found not to be affected by rotation of the patients in a study by Dryzer et al.55
Fever and pain are the most common complications following pleurodesis. Other complications include nausea, respiratory failure, and death. In the ISPP report, talc was the agent that resulted in significantly more pain, fever, and nausea than other agents. Respiratory failure was also observed more frequently with talc pleurodesis than other agents.51
LUNG ABSCESS
A lung abscess develops when a bacterial infection causes necrosis and produces cavities in the lung parenchyma. A primary lung abscess occurs when one or two cavities with air-fluid levels form in the lung parenchyma as the result of an aspiration of pathogen-laden secretion (Fig. 4). Although many organisms can cause lung abscesses, anaerobic mouth flora is the most common pathogen in a primary lung abscess. A secondary lung abscess develops from predisposing conditions, such as congenital lung abnormalities, obstructing neoplasm, a foreign body, and bronchiectasis. In necrotizing pneumonia, multiple small cavities (< 2 cm in diameter) develop in contiguous areas of the lung.56
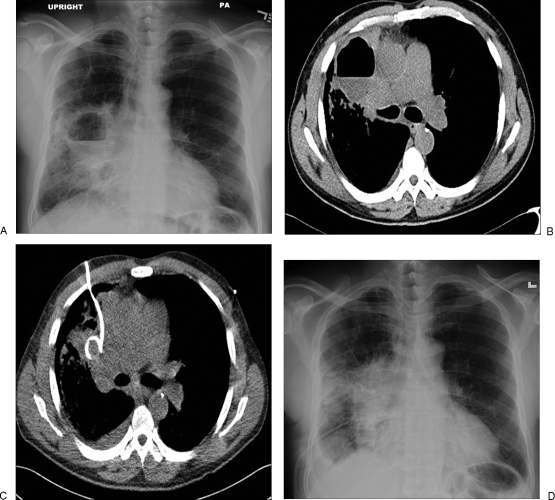
Figure 4.
(A) Posteroanterior chest radiograph shows a 7-cm lung abscess with an air-fluid level in the right middle lobe. (B) An axial computed tomography (CT) image shows an abscess with an irregular outer margin and inner air-fluid level. (C) An axial CT image shows a 10F nontunneled pigtail drainage catheter placed percutaneously in the lung abscess. (D) Follow-up chest radiograph shows a pigtail catheter in the abscess with decreased size without an air-fluid level.
Patients usually present with nonspecific symptoms including fevers, night sweats, cough, putrid sputum, hemoptysis, pleuritic chest pain, and fatigue. A diagnosis is made based on clinical symptoms, physical examination, predisposing conditions, and radiographic findings. A typical finding of a lung abscess on a plain chest radiograph is a thick-walled cavity with an air-fluid level. This finding, however, may also be seen in other pathologic conditions including infected bulla, cavitary tumor, mycobacterial infection, pulmonary infarction, pulmonary sequestration, and vasculitis. Overlying lung disease and an unfavorable location also make image findings nondiagnostic of a lung abscess. Therefore, contrast-enhanced CT is essential to differentiate a lung abscess from other similar pathologic conditions.56 Characteristic CT findings of lung abscess include a round and thick-walled cavity with an irregular inner wall and air-fluid levels in areas of destroyed lung parenchyma. Bronchial trees and pulmonary vessels usually terminate abruptly at the irregular outer margin of the lung abscess.57
Lung abscesses are most commonly managed by medical treatment with prolonged antibiotics. The duration of medical treatment varies from 1 month to 3 months. However, medical treatment can be failed if the patient has poor prognostic factors, such as a large abscess cavity (> 6 cm), compromised immunity, neoplasm, advanced age, reduced level of consciousness, or infection with certain aerobic pathogens (Klebsiella pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus).58 Eleven to 21% of patients with lung abscesses eventually require surgical or percutaneous drainage due to medical treatment failure.50 Surgery is usually associated with high morbidity and mortality. The success rate of CT-guided drainage of a lung abscess is up to 90%. Therefore, CT-guided percutaneous drainage of a lung abscess should be considered as the initial treatment of choice for patients with failed medical treatment.50
The procedure is usually performed under conscious sedation using fentanyl and midazolam. In pediatric patients, general anesthesia is required. The procedure can be performed with the patients in a supine, prone, decubitus, or semidecubitus position. The initial puncture site is localized using preprocedural CT scan after which the patient is prepared and draped in a sterile manner. Once the skin entry site is anesthetized using 1% lidocaine with epinephrine, a small incision is made. Although a Trocar technique can be used to access the cavity directly, a Seldinger technique over a guidewire is usually recommended because it entails less pain, less discomfort, and has a lower rate of complications. When an initial 18-gauge access needle is advanced into the lung abscess, normal lung parenchyma should be avoided to prevent the development of a bronchopleural fistula or pyopneumothorax. The needle should be traversed through the affected lung parenchyma into the lung abscess. Once the needle is successfully placed in the center of the abscess cavity, fluid can be aspirated for diagnostic or therapeutic purposes. The needle is then exchanged for a dilator over a 0.038-inch Newton-J or 0.035 inch Bentson (AngioDynamics, Queensbury, NY) guidewire and the tract is serially dilated. The dilator is then exchanged for a 10F or 12F locking pigtail catheter over a guidewire. The drainage catheter should be secured to the skin using suture and covered using a sterile dressing. The drainage catheter is connected to a self-contained suction system, such as a Jackson-Pratt (JP) or Hemovac drain. A Pleur-evac® can also be used for underwater seal drainage. Afterward, a postprocedural CT scan should be obtained to confirm optimal position of the drainage catheter and to evaluate possible complications. The drainage catheter should be flushed daily with 5–15 mL of sterile saline.
The sepsis symptoms are usually resolved in 48 hours after drainage catheter placement. The lung abscess itself may be resolved after 10–15 days. The possible complications of percutaneous drainage catheters include pneumothorax, pyopneumothorax, and bronchopleural fistula.50
CONCLUSION
The role of interventional radiology in the management of pleural effusions, empyema, and lung abscess is becoming more important. As imaging and percutaneous interventional techniques are improving, in cases of pleural fluid collection they are considered the mainstay of treatment with less morbidity and mortality than surgery. Ultrasound-guided thoracentesis is an effective and safe procedure not only for differentiating transudate from exudate, but for achieving symptomatic relief by removing fluid. Complicated effusions should be managed by the placement of a nontunneled pigtail catheter under ultrasound or CT guidance, which is less painful, produces less discomfort and has less complications and a shorter hospital stay. For managing patients with recurrent malignant pleural effusion, a tunneled Pleurx® catheter is widely used because it is a soft and more comfortable catheter with less risk for infection and dislodgement, and may be used in the outpatient-based management of drainage and symptoms. A lung abscess is usually managed by medical treatment with antibiotics. However, if medical treatment fails, CT-guided pigtail catheter placement should be considered as an initial procedure to improve the patient's morbidity and mortality risk.
References
- Light R W. Pleural Diseases. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2001. [Google Scholar]
- Chung J, Perrot M D. Pleural effusion and empyema thoracis. In: Bope E T, Rakel R E, Kellerman R D, editors. Conn's Current Therapy 2010. 1st ed. Philadelphia: Saunders/ Elsevier; 2010. pp. 263–265. [Google Scholar]
- Light R W, Macgregor M I, Luchsinger P C, Ball W C., Jr Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med. 1972;77(4):507–513. doi: 10.7326/0003-4819-77-4-507. [DOI] [PubMed] [Google Scholar]
- Sahn S A. State of the art. The pleura. Am Rev Respir Dis. 1988;138(1):184–234. doi: 10.1164/ajrccm/138.1.184. [DOI] [PubMed] [Google Scholar]
- Miller E J, Idell S. Interleukin-8: an important neutrophil chemotaxin in some cases of exudative pleural effusions. Exp Lung Res. 1993;19(5):589–601. doi: 10.3109/01902149309031730. [DOI] [PubMed] [Google Scholar]
- Broaddus V C, Light R W. Pleural Effusion. In: Mason R J, Broaddus V C, Martin T R, et al., editors. Textbook of Respiratory Medicine. 5th ed. Philadelphia: Saunders/Elsevier; 2010. pp. 1719–1763. [Google Scholar]
- Bouros D, Plataki M, Antoniou K M. Parapneumonic effusion and empyema: best therapeutic approach. Monaldi Arch Chest Dis. 2001;56(2):144–148. [PubMed] [Google Scholar]
- Schiza S, Siafakas N M. Clinical presentation and management of empyema, lung abscess and pleural effusion. Curr Opin Pulm Med. 2006;12(3):205–211. doi: 10.1097/01.mcp.0000219270.73180.8b. [DOI] [PubMed] [Google Scholar]
- Light R W. Parapneumonic effusions and empyema. Clin Chest Med. 1985;6(1):55–62. [PubMed] [Google Scholar]
- Colins J D, Burwell D, Furmanski S, Lorber P, Steckel R J. Minimal detectable pleural effusions. A roentgen pathology model. Radiology. 1972;105(1):51–53. doi: 10.1148/105.1.51. [DOI] [PubMed] [Google Scholar]
- Moskowitz H, Platt R T, Schachar R, Mellins H. Roentgen visualization of minute pleural effusion. An experimental study to determine the minimum amount of pleural fluid visible on a radiograph. Radiology. 1973;109(1):33–35. doi: 10.1148/109.1.33. [DOI] [PubMed] [Google Scholar]
- Eibenberger K L, Dock W I, Ammann M E, Dorffner R, Hörmann M F, Grabenwöger F. Quantification of pleural effusions: sonography versus radiography. Radiology. 1994;191(3):681–684. doi: 10.1148/radiology.191.3.8184046. [DOI] [PubMed] [Google Scholar]
- Feller-Kopman D. Ultrasound-guided thoracentesis. Chest. 2006;129(6):1709–1714. doi: 10.1378/chest.129.6.1709. [DOI] [PubMed] [Google Scholar]
- Liu Y H, Lin Y C, Liang S J, et al. Ultrasound-guided pigtail catheters for drainage of various pleural diseases. Am J Emerg Med. 2010;28(8):915–921. doi: 10.1016/j.ajem.2009.04.041. [DOI] [PubMed] [Google Scholar]
- Bressler E L, Francis I R, Glazer G M, Gross B H. Bolus contrast medium enhancement for distinguishing pleural from parenchymal lung disease: CT features. J Comput Assist Tomogr. 1987;11(3):436–440. doi: 10.1097/00004728-198705000-00013. [DOI] [PubMed] [Google Scholar]
- Moulton J S, Benkert R E, Weisiger K H, Chambers J A. Treatment of complicated pleural fluid collections with image-guided drainage and intracavitary urokinase. Chest. 1995;108(5):1252–1259. doi: 10.1378/chest.108.5.1252. [DOI] [PubMed] [Google Scholar]
- Keeling A N, Leong S, Logan P M, Lee M J. Empyema and effusion: outcome of image-guided small-bore catheter drainage. Cardiovasc Intervent Radiol. 2008;31(1):135–141. doi: 10.1007/s00270-007-9197-0. [DOI] [PubMed] [Google Scholar]
- Light R W, Rodriguez R M. Management of parapneumonic effusions. Clin Chest Med. 1998;19(2):373–382. doi: 10.1016/s0272-5231(05)70084-8. [DOI] [PubMed] [Google Scholar]
- Moulton J S. Image-guided management of complicated pleural fluid collections. Radiol Clin North Am. 2000;38(2):345–374. doi: 10.1016/s0033-8389(05)70167-6. [DOI] [PubMed] [Google Scholar]
- Light R W. Clinical practice. Pleural effusion. N Engl J Med. 2002;346(25):1971–1977. doi: 10.1056/NEJMcp010731. [DOI] [PubMed] [Google Scholar]
- Shinto R A, Light R W. Effects of diuresis on the characteristics of pleural fluid in patients with congestive heart failure. Am J Med. 1990;88(3):230–234. doi: 10.1016/0002-9343(90)90147-6. [DOI] [PubMed] [Google Scholar]
- Diacon A H, Brutsche M H, Solèr M. Accuracy of pleural puncture sites: a prospective comparison of clinical examination with ultrasound. Chest. 2003;123(2):436–441. doi: 10.1378/chest.123.2.436. [DOI] [PubMed] [Google Scholar]
- Thomsen T W, DeLaPena J, Setnik G S. Videos in clinical medicine. Thoracentesis. N Engl J Med. 2006;355(15):e16. doi: 10.1056/NEJMvcm053812. [DOI] [PubMed] [Google Scholar]
- Petersen W G, Zimmerman R. Limited utility of chest radiograph after thoracentesis. Chest. 2000;117(4):1038–1042. doi: 10.1378/chest.117.4.1038. [DOI] [PubMed] [Google Scholar]
- Trachiotis G D, Vricella L A, Aaron B L, Hix W R. As originally published in 1988: Reexpansion pulmonary edema. Updated in 1997. Ann Thorac Surg. 1997;63(4):1206–1207. doi: 10.1016/s0003-4975(97)00193-8. [DOI] [PubMed] [Google Scholar]
- Grogan D R, Irwin R S, Channick R, et al. Complications associated with thoracentesis. A prospective, randomized study comparing three different methods. Arch Intern Med. 1990;150(4):873–877. doi: 10.1001/archinte.150.4.873. [DOI] [PubMed] [Google Scholar]
- Raptopoulos V, Davis L M, Lee G, Umali C, Lew R, Irwin R S. Factors affecting the development of pneumothorax associated with thoracentesis. AJR Am J Roentgenol. 1991;156(5):917–920. doi: 10.2214/ajr.156.5.2017951. [DOI] [PubMed] [Google Scholar]
- Colice G L, Curtis A, Deslauriers J, et al. Medical and surgical treatment of parapneumonic effusions : an evidence-based guideline. Chest. 2000;118(4):1158–1171. doi: 10.1378/chest.118.4.1158. [DOI] [PubMed] [Google Scholar]
- Shankar S, Gulati M, Kang M, Gupta S, Suri S. Image-guided percutaneous drainage of thoracic empyema: can sonography predict the outcome? Eur Radiol. 2000;10(3):495–499. doi: 10.1007/s003300050083. [DOI] [PubMed] [Google Scholar]
- Rahman N M, Maskell N A, Davies C W, et al. The relationship between chest tube size and clinical outcome in pleural infection. Chest. 2010;137(3):536–543. doi: 10.1378/chest.09-1044. [DOI] [PubMed] [Google Scholar]
- Davies H E, Rahman N M, Parker R J, Davies R J. Use of indwelling pleural catheters for chronic pleural infection. Chest. 2008;133(2):546–549. doi: 10.1378/chest.07-1742. [DOI] [PubMed] [Google Scholar]
- Pierrepoint M J, Evans A, Morris S J, Harrison S K, Doull I J. Pigtail catheter drain in the treatment of empyema thoracis. Arch Dis Child. 2002;87(4):331–332. doi: 10.1136/adc.87.4.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dev S P, Nascimiento B, Jr,Simone C, Chien V. Videos in clinical medicine. Chest-tube insertion. N Engl J Med. 2007;357(15):e15. doi: 10.1056/NEJMvcm071974. [DOI] [PubMed] [Google Scholar]
- Pollak J S, Burdge C M, Rosenblatt M, Houston J P, Hwu W J, Murren J. Treatment of malignant pleural effusions with tunneled long-term drainage catheters. J Vasc Interv Radiol. 2001;12(2):201–208. doi: 10.1016/s1051-0443(07)61826-0. [DOI] [PubMed] [Google Scholar]
- Zimmerman L H. Pleural effusions. In: Goldstein R H, O'Connell J J, Karlinsky J B, editors. A Practical Approach to Pulmonary Medicine. Philadelphia: Lippincott-Raven; 1997. pp. 195–205. [Google Scholar]
- Grossi F, Pennucci M C, Tixi L, Cafferata M A, Ardizzoni A. Management of malignant pleural effusions. Drugs. 1998;55(1):47–58. doi: 10.2165/00003495-199855010-00004. [DOI] [PubMed] [Google Scholar]
- Tattersall M. Management of malignant pleural effusion. Aust N Z J Med. 1998;28(3):394–396. doi: 10.1111/j.1445-5994.1998.tb01972.x. [DOI] [PubMed] [Google Scholar]
- Musani A I. Treatment options for malignant pleural effusion. Curr Opin Pulm Med. 2009;15(4):380–387. doi: 10.1097/MCP.0b013e32832c6a8a. [DOI] [PubMed] [Google Scholar]
- Pollak J S. Malignant pleural effusions: treatment with tunneled long-term drainage catheters. Curr Opin Pulm Med. 2002;8(4):302–307. doi: 10.1097/00063198-200207000-00010. [DOI] [PubMed] [Google Scholar]
- Tremblay A, Michaud G. Single-center experience with 250 tunnelled pleural catheter insertions for malignant pleural effusion. Chest. 2006;129(2):362–368. doi: 10.1378/chest.129.2.362. [DOI] [PubMed] [Google Scholar]
- Thornton R H, Miller Z, Covey A M, et al. Tunneled pleural catheters for treatment of recurrent malignant pleural effusion following failed pleurodesis. J Vasc Interv Radiol. 2010;21(5):696–700. doi: 10.1016/j.jvir.2010.01.021. [DOI] [PubMed] [Google Scholar]
- Ohm C, Park D, Vogen M, et al. Use of an indwelling pleural catheter compared with thorascopic talc pleurodesis in the management of malignant pleural effusions. Am Surg. 2003;69(3):198–202. discussion 202. [PubMed] [Google Scholar]
- Pien G W, Gant M J, Washam C L, Sterman D H. Use of an implantable pleural catheter for trapped lung syndrome in patients with malignant pleural effusion. Chest. 2001;119(6):1641–1646. doi: 10.1378/chest.119.6.1641. [DOI] [PubMed] [Google Scholar]
- Davies C W, Gleeson F V, Davies R J, Pleural Diseases Group, Standards of Care Committee, British Thoracic Society BTS guidelines for the management of pleural infection. Thorax. 2003;58(Suppl 2):ii18–ii28. doi: 10.1136/thorax.58.suppl_2.ii18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temes R T, Follis F, Kessler R M, Pett S B, Jr, Wernly J A. Intrapleural fibrinolytics in management of empyema thoracis. Chest. 1996;110(1):102–106. doi: 10.1378/chest.110.1.102. [DOI] [PubMed] [Google Scholar]
- Zuckerman D A, Reed M F, Howington J A, Moulton J S. Efficacy of intrapleural tissue-type plasminogen activator in the treatment of loculated parapneumonic effusions. J Vasc Interv Radiol. 2009;20(8):1066–1069. doi: 10.1016/j.jvir.2009.04.067. [DOI] [PubMed] [Google Scholar]
- Davies C W, Traill Z C, Gleeson F V, Davies R J. Intrapleural streptokinase in the management of malignant multiloculated pleural effusions. Chest. 1999;115(3):729–733. doi: 10.1378/chest.115.3.729. [DOI] [PubMed] [Google Scholar]
- Maskell N A, Davies C W, Nunn A J, et al. First Multicenter Intrapleural Sepsis Trial (MIST1) Group U.K. Controlled trial of intrapleural streptokinase for pleural infection. N Engl J Med. 2005;352(9):865–874. doi: 10.1056/NEJMoa042473. [DOI] [PubMed] [Google Scholar]
- Tokuda Y, Matsushima D, Stein G H, Miyagi S. Intrapleural fibrinolytic agents for empyema and complicated parapneumonic effusions: a meta-analysis. Chest. 2006;129(3):783–790. doi: 10.1378/chest.129.3.783. [DOI] [PubMed] [Google Scholar]
- Erasmus J J, Goodman P C, Patz E F., Jr Management of malignant pleural effusions and pneumothorax. Radiol Clin North Am. 2000;38(2):375–383. doi: 10.1016/s0033-8389(05)70168-8. [DOI] [PubMed] [Google Scholar]
- Lee Y C, Baumann M H, Maskell N A, et al. Pleurodesis practice for malignant pleural effusions in five English-speaking countries: survey of pulmonologists. Chest. 2003;124(6):2229–2238. doi: 10.1378/chest.124.6.2229. [DOI] [PubMed] [Google Scholar]
- Putnam J B, Jr, Light R W, Rodriguez R M, et al. A randomized comparison of indwelling pleural catheter and doxycycline pleurodesis in the management of malignant pleural effusions. Cancer. 1999;86(10):1992–1999. [PubMed] [Google Scholar]
- Shaw P, Agarwal R. Pleurodesis for malignant pleural effusions. Cochrane Database Syst Rev. 2004;(1):CD002916. doi: 10.1002/14651858.CD002916.pub2. [DOI] [PubMed] [Google Scholar]
- Mager H J, Maesen B, Verzijlbergen F, Schramel F. Distribution of talc suspension during treatment of malignant pleural effusion with talc pleurodesis. Lung Cancer. 2002;36(1):77–81. doi: 10.1016/s0169-5002(01)00475-5. [DOI] [PubMed] [Google Scholar]
- Dryzer S R, Allen M L, Strange C, Sahn S A. A comparison of rotation and nonrotation in tetracycline pleurodesis. Chest. 1993;104(6):1763–1766. doi: 10.1378/chest.104.6.1763. [DOI] [PubMed] [Google Scholar]
- Morrow L E, Jarrett J. Primary Lung Abscess. In: Bope E T, Rakel R E, Kellerman R D, editors. Conn's Current Therapy 2010. 1st ed. Philadelphia: Saunders/Elsevier; 2010. pp. 266–267. [Google Scholar]
- Stark D D, Federle M P, Goodman P C, Podrasky A E, Webb W R. Differentiating lung abscess and empyema: radiography and computed tomography. AJR Am J Roentgenol. 1983;141(1):163–167. doi: 10.2214/ajr.141.1.163. [DOI] [PubMed] [Google Scholar]
- Mwandumba H C, Beeching N J. Pyogenic lung infections: factors for predicting clinical outcome of lung abscess and thoracic empyema. Curr Opin Pulm Med. 2000;6(3):234–239. doi: 10.1097/00063198-200005000-00012. [DOI] [PubMed] [Google Scholar]