Abstract
Mycobacterium tuberculosis (Mtb) has evolved into a highly successful human pathogen. It deftly subverts the bactericidal mechanisms of alveolar macrophages, ultimately inducing granuloma formation and establishing long-term residence in the host. These hallmarks of Mtb infection are facilitated by the metabolic adaptation of the pathogen to its surrounding environment and the biosynthesis of molecules that mediate its interactions with host immune cells. The sulfate assimilation pathway of Mtb produces a number of sulfur-containing metabolites with important contributions to pathogenesis and survival. This pathway is regulated by diverse environmental cues and regulatory proteins that mediate sulfur transactions in the cell. Here, we discuss the transcriptional and biochemical mechanisms of sulfur metabolism regulation in Mtb and potential small molecule regulators of the sulfate assimilation pathway that are collectively poised to aid this intracellular pathogen in its expert manipulation of the host. From this global analysis, we have identified a subset of sulfur-metabolizing enzymes that are sensitive to multiple regulatory cues and may be strong candidates for therapeutic intervention.
Introduction
Mycobacterium tuberculosis (Mtb), the bacterium that causes tuberculosis in humans, infects roughly 2 billion people worldwide [1]. However, less than 1% of those infected with Mtb show signs of active disease [2]. The majority of infected individuals have a latent infection characterized by dormant, nonreplicating bacteria that persist within a mass of immune cells in the lung [3]. These cells form a protective barrier between the bacteria and surrounding tissue known as the granuloma [4], [5]. When host immunity is compromised, the granuloma deteriorates, liberating the confined bacteria and reactivating the disease.
In order to mount a latent infection, Mtb must withstand phagocytosis by alveolar macrophages, the host's primary line of defense against airborne pathogens [5]. By evading typical bactericidal processes, Mtb is able to replicate and eventually induce granuloma formation [3]. The mechanisms by which Mtb persists in the hostile phagosomal environment and orchestrates the transition to latency are ill defined. Elucidating the molecular machinery and metabolic pathways that facilitate these pivotal events is crucial to identifying new therapeutic targets for this global human pathogen.
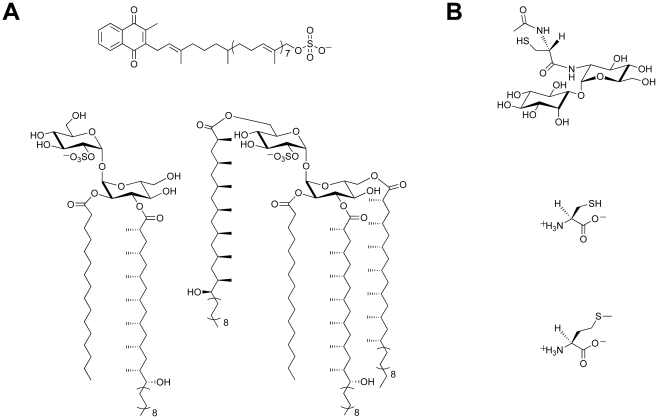
There is a growing body of evidence that supports a role for sulfur-containing metabolites in Mtb pathogenesis [6], [7]. Sulfated molecules from Mtb have been intimately linked to bacterial virulence (Figure 1A) [8]–[10]. For example, the prominent cell wall-associated glycolipid Sulfolipid-1 is only produced by pathogenic species of mycobacteria [8], [11], and its biosynthetic precursor SL1278, named for its observed mass, has been shown to elicit cytokine production in human tuberculosis patients [10]. On the contrary, biosynthesis of the sulfated menaquinone S881 suppresses bacterial virulence [9]. Reduced sulfur compounds (Figure 1B), such as cysteine and methionine, also contribute to Mtb pathogenesis [6]. Disabling their biosynthesis dramatically attenuates bacterial virulence and persistence during the chronic phase of infection in mice [12]. Mycothiol (MSH), the primary thiol-containing small molecule of mycobacteria [13], is instrumental in the detoxification of numerous bactericidal agents and confers protection against oxidative stress [14]–[16]. Biosynthesis of these important sulfur-containing metabolites relies on the sulfate assimilation pathway [17].
Figure 1. Sulfur-containing metabolites from Mtb.
(A) Sulfated compounds, clockwise from top: S881, Sulfolipid-1, SL1278. (B) Reduced sulfur compounds, from top to bottom: MSH, cysteine, methionine.
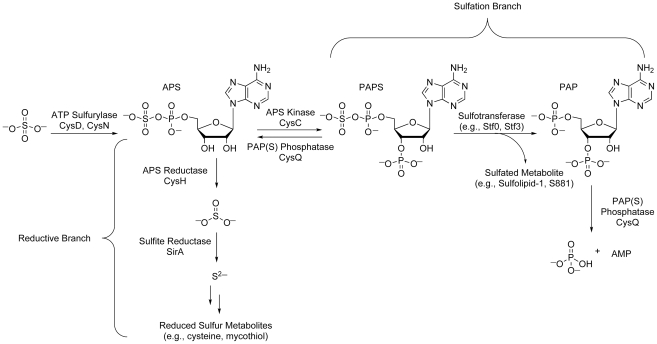
The sulfate assimilation pathway (Figure 2) is composed of a group of enzymes that catalyze the uptake and metabolism of inorganic sulfate from the host [7]. This pathway commences with the active import of sulfate, which is subsequently adenylated by ATP sulfurylase, an enzyme containing both GTPase and sulfurylase domains [18]. The resulting product, adenosine 5′-phosposulfate (APS), can be reduced to sulfite by APS reductase for the biosynthesis of reduced sulfur species via the reductive branch of the pathway [19]. Alternatively, because APS constitutes a metabolic branchpoint, it can be phosphorylated at the 3′-position by APS kinase to generate 3′-phosphoadenosine 5′-phosphosulfate (PAPS), the universal sulfate donor in the cell [17]. PAPS is a substrate for sulfotransferases, enzymes that catalyze the transfer of its sulfate group onto bacterial metabolites [11]. Collectively, these reactions constitute the sulfation branch of the sulfate assimilation pathway.
Figure 2. The sulfate assimilation pathway of Mtb.
Inorganic sulfate from the host is converted to APS by ATP sulfurylase. APS is either sequentially reduced by APS reductase and sulfite reductase for the biosynthesis of essential reduced sulfur metabolites (reductive branch), or phosphorylated by APS kinase to generate PAPS, a substrate for sulfotransferases (sulfation branch). PAP, a byproduct of sulfotransferase reactions, is degraded by the PAP(S) phosphatase CysQ, which also converts PAPS to APS.
The regulation of Mtb sulfur metabolism relies on the transcriptional response of sulfate assimilation enzymes to diverse environmental cues and regulatory proteins that influence flux through the sulfate assimilation pathway. Further, small molecule regulation of sulfur metabolism in other bacteria suggests similar mechanisms may modulate related transcriptional circuits in Mtb. Here, we review each of these regulatory elements in an effort to guide our understanding of how the sulfate assimilation pathway facilitates bacterial adaptation to the host.
Transcriptional Regulation of Sulfur Metabolism Genes
Transcriptional profiling has demonstrated that Mtb sulfur metabolism genes are dynamically regulated by diverse environmental cues (Table 1) [20]–[29]. This finding is perhaps not surprising, given the demonstrated importance of the sulfate assimilation pathway to bacterial survival [6] and the hostile conditions encountered by bacteria in the phagosome [30]. For example, upon phagocytosis, bacteria are exposed to oxidative stress and deprived of essential nutrients [30], [31]. Microarray analysis has shown that these conditions elicit the upregulation of key genes from the sulfate assimilation pathway (Table 1) [21], [22], [26], [27]. Genes encoding the primary sulfate transport complex of Mtb are induced following treatment with hydrogen peroxide (cysT) and nutrient starvation (cysA1, cysT, cysW, subI), conditions that also induce the ATP sulfurylase genes cysD and cysN, as well as the cysC gene that encodes APS kinase (in Mtb, cysC is fused to cysN, yielding a bifunctional cysNC gene [18]). Given that these genes coordinate the first few steps of the sulfate assimilation pathway, their induction is most likely accompanied by an increase in the biosynthesis of sulfur-containing metabolites that protect the pathogen during the course of infection, though this remains to be demonstrated. Notably, cysD and cysNC are also induced during macrophage infection, underscoring the importance of sulfur metabolism to intracellular survival [21], [25]. Indeed, reduced sulfur compounds have been shown to play a critical role in facilitating bacterial persistence in vivo [12]. Genes contributing to the biosynthesis of these metabolites, such as cysH and sirA, which direct the reduction of APS to sulfide [19], [32], and cysM, which is involved in cysteine biosynthesis [33], [34], are also among those induced by hydrogen peroxide and nutrient starvation. Collectively, these genes are likely to facilitate bacterial adaptation to the phagosomal environment.
Table 1. Sulfur metabolism genes from M. tuberculosis induced by various conditions of environmental stress.
| Stress Condition | Genes Induced | Reference |
| 5-Chloropyrazinamide | cysN | [20] |
| Hydrogen peroxide | cysD, cysNC a, cysK2, cysM, cysT | [21], [22] |
| Hypoxia | cysD, cysK2, cysN, cysM | [23], [24] |
| Macrophage infection | cysD, cysN | [21], [25] |
| Menadione | cysA1, cysT, cysW, subI | [20] |
| Nutrient starvation | cysA1, cysD, cysH, cysN, cysQ, cysT, cysW, sirA, subI | [26], [27] |
| SDS | cysD, cysN | [28] |
| Vancomycin | cysD, cysK2, cysNC | [29] |
The cysN and cysC genes of Mtb are fused into a single, bifunctional cysNC gene [22]. However, the transcript is often reported as cysN in microarray data.
SDS, sodium dodecyl sulfate.
Hypoxia is another environmental cue that has been correlated with the upregulation of genes from the sulfate assimilation pathway [23], [24]. Transcription of cysD, cysNC, cysK2, and cysM is induced by hypoxia, suggesting ATP sulfurylase and cysteine biosynthesis may facilitate bacterial adaptation to oxygen-limiting conditions. Because the granuloma has been shown to harbor regions of low oxygen tension [4], it is plausible that sulfur metabolism may be important in the transition to latency. The finding that reduced sulfur compounds are critical to the onset of chronic Mtb infection in mice lends additional support to this hypothesis [12].
Certain antibiotics have also been shown to affect the transcription of sulfur metabolism genes. Vancomycin, an inhibitor of peptidoglycan biosynthesis, induces expression of cysK2, cysD, and cysNC [29]. The cysNC gene is also induced by 5-chloropyrazinamide, a pyrazinamide analog that irreversibly inhibits fatty acid biosynthesis in vitro [20], [35]. Furthermore, cysA1, cysT, cysW, and subI are collectively induced by menadione, a synthetic vitamin K precursor that promotes the formation of reduced oxygen species [20], [36]. These data suggest that sulfur metabolism may modulate the bacterial response to certain drugs. Evaluating Mtb susceptibility to these drugs upon overexpression of related sulfur metabolism genes may reveal a more direct role for the encoded enzymes in antibiotic resistance.
Indeed, several studies point to a role for sulfur-containing compounds in bacterial detoxification pathways. In Salmonella typhimurium, cysteine biosynthesis is required for the enhanced antibiotic resistance of migrating swarm cells [37]. Similarly, dysregulation of cysteine metabolism in Bacillus subtilis and Staphylococcus aureus sensitizes the bacterial response to oxidative stress and tellurite exposure [38], [39]. Further, MSH exhibits well-documented protective effects against free radicals, potent oxidizing agents, alkylating agents, and certain antibiotics [14]–[16], [40], [41], though is essential for ethionamide susceptibility and was recently shown to be dispensable for Mtb growth [42]. Overall, these findings underscore the importance of bacterial sulfur metabolism to the detoxification of toxic species in the cell and are consistent with the enhanced expression of Mtb sulfur metabolism genes in response to oxidative stress and exposure to certain antibiotics.
Transcriptional Regulators of Bacterial Sulfur Metabolism
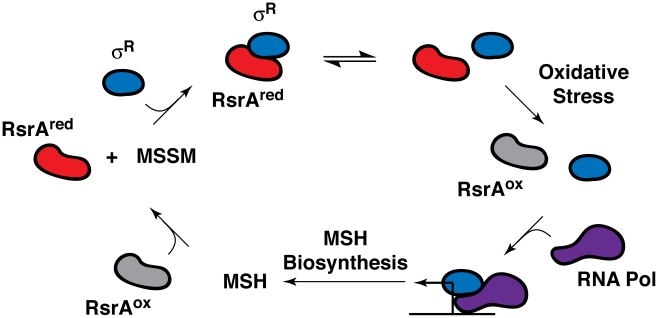
In all likelihood, one or more transcriptional regulators mediate the transcriptional response of the sulfate assimilation pathway to extracellular signals. The alternative sigma factor SigH, which is induced by phagocytosis and stress conditions, including acidic pH and exposure to diamide, a thiol oxidizer, has been shown to regulate the transcription of several sulfur metabolism genes (e.g., cysA1, cysT, cysW, cysD, and cysNC) following diamide treatment [43]. SigH activity is regulated by the redox-sensitive anti-sigma factor RshA [44]. Interestingly, the alternative sigma factor SigR from Streptomyces coelicolor, a related actinomycete, is also induced by diamide and regulated by the redox-sensitive anti-sigma factor RsrA (Figure 3) [45]. SigR and RsrA have been shown to modulate MSH production in S. coelicolor in response to intracellular MSH levels [45]. The strong dependence of MSH biosynthesis on the sulfate assimilation pathway suggests that SigH and RshA might similarly regulate the biosynthesis of this ubiquitous thiol in mycobacteria.
Figure 3. Feedback regulation of RsrA activity and SigR (σR)-mediated transcription by MSH in S. coelicolor.
The anti-sigma factor RsrA binds the alternative sigma factor σR in its reduced state (RsrAred), preventing the association of σR with RNA polymerase (RNA Pol). Under conditions of oxidative stress (e.g., hydrogen peroxide or diamide treatment), RsrA is oxidized (RsrAox) and no longer binds σR, enabling it to form a complex with RNA polymerase, which induces the transcription of MSH biosynthetic genes. The reduction of oxidized RsrA by MSH facilitates sequestration of σR, which, in turn, suppresses MSH biosynthesis. Importantly, when oxidizing or alkylating agents deplete MSH levels, RsrA remains oxidized, which stimulates σR-mediated transcription and MSH biosynthesis [45], [67].
In Escherichia coli, the majority of sulfate assimilation genes belong to the cysteine (cys) regulon that is positively regulated by the transcription factor CysB [46]. In the absence of inorganic sulfate, a second transcription factor, Cbl, induces a series of sulfate starvation-inducible (SSI) genes that coordinate the uptake and subsequent metabolism of organosulfur compounds for the generation of sulfite [47], [48]. Comparable transcriptional regulators have not yet been identified in mycobacteria.
Though the enzymes involved in sulfur metabolism are largely conserved between these two prokaryotes, there are some notable differences in the organization of their respective sulfate assimilation pathways that caution against using the transcriptional regulatory networks of E. coli as a blueprint for those of Mtb. For example, the reductive branch of the Mtb sulfate assimilation pathway emanates from APS, whereas E. coli initiates the biosynthesis of reduced sulfur metabolites from PAPS [17]. Mtb also encodes four eukaryotic-like sulfotransferases (stf0, stf1, stf2, and stf3), which are absent from E. coli [11], [49]. Further, Mtb encodes two independent pathways for cysteine biosynthesis: the canonical pathway, which utilizes L-serine (as in E. coli), and a novel route that utilizes O-phospho-L-serine [33], [50], [51]. While these distinctions do not rule out the possibility of a CysB-like master regulator in Mtb, they do suggest that the transcriptional regulation of mycobacterial sulfur-metabolizing enzymes may diverge considerably from that of E. coli. Additional studies are needed to elucidate the transcriptional machinery mediating the expression of these enzymes in Mtb. Notably, a functional analog of CysB was recently identified in Corynebacterium glutamicum, a gram-positive bacterium belonging to the same suborder as Mtb, which will likely facilitate the search for related proteins in mycobacteria [52].
Biochemical Regulation of Sulfur Metabolism
Sulfate Transporters
In addition to the various environmental conditions that modulate sulfur metabolism at the transcriptional level, several proteins regulate the metabolic pipeline by mediating the availability of sulfate and its flux through the sulfate assimilation pathway. Most notable are the sulfate transporters, which enable the uptake of extracellular sulfate. The primary sulfate permease of Mtb is an ABC transporter encoded by the subI-cysTWA1 operon [53]. Genetic disruption of the cysA1 gene in M. bovis completely inhibits sulfate uptake in vitro and renders the mutant auxotrophic for methionine, yet does not impair bacterial survival in mice [53]. This implies either a strong reliance on methionine transport in vivo, or the presence of other sulfate transporters that can compensate for the loss of SubI-CysTWA1 activity during infection. Notably, Mtb encodes two additional putative sulfate transporters, Rv1739c and Rv1707, whose genes are induced 24 h postinfection of activated macrophages [21], [54]. Rv1739c expression in E. coli has been shown to enhance sulfate uptake, though complementation of the M. bovis cysA1 mutant with the Rv1739c gene is not sufficient to restore sulfate prototrophy [54]. In contrast, little is known about the Rv1707 gene product. Assuming all three of these transporters are functional, it is possible they modulate sulfate uptake in response to discrete environmental signals.
Sulfatases
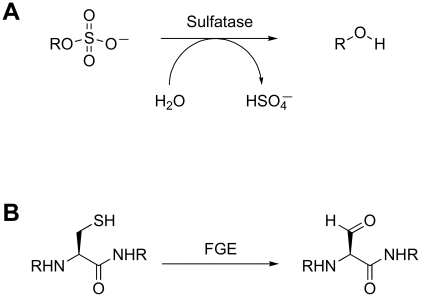
The sulfatases are a second class of proteins that likely contribute to the intracellular concentration of free sulfate. These enzymes catalyze the hydrolysis of sulfate esters from sulfated proteins, peptides, and small molecules (Figure 4A) [55]. Type I sulfatases are characterized by an active-site formylglycine residue that is critical for catalysis [56]. The formylglycine is either co- or post-translationally installed by a formylglycine-generating (FGE) enzyme via oxidation of a conserved cysteine (Figure 4B). The Mtb genome encodes six type I sulfatases, yet little is known about their biochemistry [56]. One of these, AtsG, was recently shown to possess arylsulfatase activity, but its native substrate has not been identified [57]. In the absence of FGE, Mtb exhibits residual sulfatase activity that may be attributed to FGE-independent type II and type III sulfatases [56]. It is possible that some of these enzymes are secreted and facilitate sulfate scavenging from the host. Alternatively, they may hydrolyze sulfate from endogenous metabolites to redirect the biosynthesis of sulfur-containing compounds in response to evolving cellular needs. In either scenario, these sulfatases are likely to regulate sulfur metabolism by influencing sulfate availability.
Figure 4. Sulfatase biochemistry.
(A) Sulfatase-catalyzed hydrolysis of a sulfated metabolite. (B) Modification of the active site cysteine of type I sulfatases by FGE.
The 3′-Phosphoadenosine-5′-Phosphatase CysQ
Another regulator of Mtb sulfur metabolism is CysQ, a 3′-phosphoadenosine-5′-phosphatase. Unlike sulfate permeases and sulfatases, whose activity directly influences the availability of free sulfate, CysQ has the ability to modulate the levels of intermediates that may affect flux through the sulfate assimilation pathway. Specifically, CysQ dephosphorylates 3′-phosphoadenosine 5′-phosphate (PAP), a byproduct of sulfotransferase reactions, and its sulfated counterpart, PAPS [58]. Since PAP inhibits at least one Mtb sulfotransferase [59], and PAPS accumulation is believed to be cytotoxic [60], the degradation of these pathway intermediates by CysQ is likely to promote sulfation and balance sulfur transactions in the cell. Interestingly, CysQ activity is inhibited by alkali metal cations in vitro, including physiological concentrations of sodium, and a homologous enzyme from Streptococcus mutans confers resistance to superoxide stress [58], [61]. These findings suggest that CysQ may be sensitive to changes in the ionic composition of the cytosol or to oxidative stress encountered during the course of infection. If so, CysQ may modulate sulfur metabolism in response to evolving environmental conditions.
Molecular Mechanisms of Sulfur Metabolism Regulation
Small molecule regulation of sulfur metabolism is another important mechanism directing the biosynthesis of sulfur-containing metabolites in bacteria. Though this phenomenon has not been thoroughly investigated in Mtb, several paradigms have emerged from other bacteria that may prove functional in mycobacteria. While it is important to consider genus- and species-specific variations in sulfur metabolism when drawing such parallels [62], regulatory networks from other bacteria have often guided the discovery of comparable pathways in Mtb [63]–[65] and thus warrant further discussion.
In S. typhimurium, cysteine, sulfide, and thiosulfate have all been shown to inhibit sulfate assimilation [46]. Cysteine does so by inhibiting serine acetyltransferase, which catalyzes the formation of O-acetylserine, the biosynthetic precursor of cysteine. In contrast, sulfide and thiosulfate bind the CysB transcriptional regulator, repressing the transcription of genes that facilitate sulfide biosynthesis or in the case of thiosulfate, its uptake from outside the cell. In E. coli, APS has been shown to inhibit Cbl-mediated transcription, preventing the induction of SSI genes that facilitate sulfur metabolism in the absence of inorganic sulfate [66]. Thus, APS may serve as a molecular barometer of sulfate assimilation that modulates gene expression in response to sulfate availability. While similar feedback mechanisms remain to be elucidated in Mtb, they are likely to play an important role in regulating sulfur metabolism. Consistent with this hypothesis, transcription of the cysDNC operon of Mtb increases in response to sulfur limitation and decreases following treatment with exogenous cysteine [22].
MSH is also likely to trigger transcriptional regulation of sulfur-metabolizing enzymes in Mtb. As mentioned previously, the alternative sigma factor SigR from S. coelicolor has been shown to modulate the biosynthesis of MSH in response to the intracellular MSH concentration [45], [67]. The current model of MSH regulation revolves around the redox-sensitive anti-sigma factor RsrA, which binds SigR in its reduced state, sequestering the sigma factor and preventing it from binding RNA polymerase (Figure 3). MSH facilitates this activity by maintaining RsrA in its reduced state. Thus, when MSH is abundant, RsrA remains active, suppressing MSH biosynthesis. However, when thiol-reactive toxins overwhelm the cell's MSH supply, RsrA is no longer maintained in its reduced form, freeing SigR to induce MSH biosynthesis. Given the importance of MSH in maintaining the reducing environment of the cytosol and promoting resistance to toxic oxidants [68], it is likely that a similar regulatory network is in place to regulate its biosynthesis in mycobacteria.
Identifying Nodes of Regulatory Convergence in Mtb Sulfur Metabolism
A global assessment of the transcriptional, biochemical, and molecular mechanisms of sulfur metabolism regulation described herein suggests nodes of regulatory convergence, or enzymes that are sensitive to multiple regulatory cues. These enzymes are apparently poised to tune flux through the sulfate assimilation pathway in response to substrate availability and a broad range of environmental signals. While many sulfur-metabolizing enzymes are promising drug targets (please see the review by Bhave et al. [6] for a comprehensive discussion), those that occupy key positions in the regulatory landscape may be particularly strong candidates for therapeutic intervention.
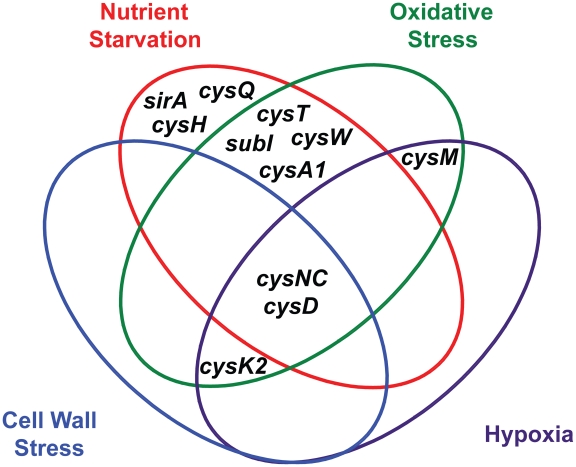
ATP sulfurylase and APS kinase, encoded by the cysDNC operon, are the most prominent regulatory nodes to emerge from this analysis. As illustrated in Figure 5, the cysD and cysNC genes are consistently upregulated by diverse environmental cues, including nutrient starvation [26], [27], hypoxia [23], [24], oxidative stress [21], [22], and cell wall stress [28], [29]. These genes are also induced upon phagocytosis [21], [25], a process that integrates many of these signals in vivo. Moreover, the catalytic activities of ATP sulfurylase and APS kinase make them particularly susceptible to biochemical regulatory cues. Together, these enzymes form the sulfate-activating complex (SAC) of Mtb, which converts free sulfate into PAPS [18]. Sulfate transporters, and possibly sulfatases, indirectly regulate this complex by determining sulfate availability in the cell. CysQ may also influence SAC activity by affecting the equilibrium between PAPS and APS. Finally, both cysteine and sulfate have been shown to modulate cysDNC transcription [22], indicating these genes are subject to small molecule regulation. In sum, ATP sulfurylase and APS kinase are highly regulated enzymes whose inhibition may critically impair Mtb sulfur metabolism.
Figure 5. Venn diagram illustrating the convergent transcriptional regulation of Mtb sulfur metabolism genes by various conditions of environmental stress.
Notably, ATP sulfurylase, a heterodimeric enzyme with both GTPase (CysN) and sulfurylase (CysD) domains, possesses several unique features that make it a particularly compelling drug target [6]. Since this enzyme catalyzes the first committed step in the Mtb sulfate assimilation pathway [18], its inhibition would eliminate flux through the pathway. Further, significant structural, mechanistic, and genetic features (e.g., the presence of a GTPase domain; the fusion of cysN with cysC) distinguish ATP sulfurylase from its human counterpart, which may facilitate the discovery of selective inhibitors [6], [18], [69]. Consistent with these findings, CysD was classified as a high-confidence drug target by an in silico target identification program following an extensive series of systems-, sequence-, and structure-based analyses [70].
Additional regulatory nodes suggested by the combined transcriptional data (Figure 5) are cysK2, cysM, and the subI-cysTWA1 operon. Both cysK2 and cysM are annotated as putative O-acetyl-L-serine sulfhydrylases (OAS), enzymes that catalyze the PLP-dependent biosynthesis of L-cysteine from O-acetyl-L-serine and a sulfur donor such as sulfide [34]. In fact, a third gene, cysK1, encodes the true OAS of Mtb [71]. CysM has been shown to catalyze an alternate cysteine biosynthetic pathway that uses O-phosphoserine as a substrate [33], [51], while the role of CysK2 in sulfur metabolism remains an enigma. Both cysK2 and cysM are transcriptionally regulated by multiple environmental cues (Table 1) [21], [24], [29], [72]. Similarly, the subI-cysTWA1 operon, which encodes the primary sulfate transport system of Mtb, is sensitive to diverse stress conditions [20], [21], [26], [27]. CysT, CysW, and CysK2 have also been classified as high-confidence drug targets [70]. Thus, these enzymes warrant further biochemical investigation and may prove attractive targets for the inhibition of Mtb sulfur metabolism.
Conclusions and Perspectives
The sulfate assimilation pathway of Mtb is responsible for the biosynthesis of sulfur-containing metabolites that influence bacterial pathogenesis. The transcriptional regulation of this pathway in response to multifarious environmental cues, including those typically encountered in the phagosome, likely facilitates adaptation to host immune cells. In addition, several proteins that mediate the flux of sulfate through the pathway, such as sulfate permeases, sulfatases, and the phosphatase CysQ, modulate the biosynthesis of sulfur-containing compounds in response to the evolving metabolic demands of the cell. Finally, the potential for small molecule regulation of Mtb sulfur metabolism abounds; the metabolites produced by the sulfate assimilation pathway are themselves candidate regulators of this network, as evidenced by cysteine and MSH in other bacteria. Realizing the extent of this regulation remains an outstanding challenge in the field, as does identifying the transcriptional proteins that orchestrate this pathway's response to the bacterial environment. Characterization of CysK2 activity and the design of selective inhibitors targeting it and other nodes of regulatory convergence, particularly ATP sulfurylase, also warrant exploration. Addressing these aims will not only advance our understanding of sulfur metabolism in Mtb, but may also reveal a molecular linchpin whose inhibition could dismantle an essential metabolic pathway in this ubiquitous human pathogen.
Acknowledgments
We thank Michael Boyce for critical evaluation of this manuscript.
Footnotes
The authors have declared that no competing interests exist.
This work was supported by National Institutes of Health Grant AI51622 to CRB. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.World Health Organization. Tuberculosis (TB). Available: www.who.int/tb/en/
- 2.Ginsberg AM, Spigelman M. Challenges in tuberculosis drug research and development. Nat Med. 2007;13:290–294. doi: 10.1038/nm0307-290. [DOI] [PubMed] [Google Scholar]
- 3.Lin PL, Flynn JL. Understanding latent tuberculosis: a moving target. J Immunol. 2010;185:15–22. doi: 10.4049/jimmunol.0903856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Russell DG. Who puts the tubercle in tuberculosis? Nat Rev Microbiol. 2007;5:39–47. doi: 10.1038/nrmicro1538. [DOI] [PubMed] [Google Scholar]
- 5.Russell DG. Mycobacterium tuberculosis: here today, and here tomorrow. Nat Rev Mol Cell Biol. 2001;2:569–577. doi: 10.1038/35085034. [DOI] [PubMed] [Google Scholar]
- 6.Bhave DP, Muse WB, 3rd, Carroll KS. Drug targets in mycobacterial sulfur metabolism. Infect Disord Drug Targets. 2007;7:140–158. doi: 10.2174/187152607781001772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schelle MW, Bertozzi CR. Sulfate metabolism in mycobacteria. Chembiochem. 2006;7:1516–1524. doi: 10.1002/cbic.200600224. [DOI] [PubMed] [Google Scholar]
- 8.Gangadharam PR, Cohn ML, Middlebrook G. Infectivity, pathogenicity and sulpholipid fraction of some Indian and British strains of tubercle bacilli. Tubercle. 1963;44:452–455. doi: 10.1016/s0041-3879(63)80087-2. [DOI] [PubMed] [Google Scholar]
- 9.Mougous JD, Senaratne RH, Petzold CJ, Jain M, Lee DH, et al. A sulfated metabolite produced by stf3 negatively regulates the virulence of Mycobacterium tuberculosis. Proc Natl Acad Sci U S A. 2006;103:4258–4263. doi: 10.1073/pnas.0510861103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gilleron M, Stenger S, Mazorra Z, Wittke F, Mariotti S, et al. Diacylated sulfoglycolipids are novel mycobacterial antigens stimulating CD1-restricted T cells during infection with Mycobacterium tuberculosis. J Exp Med. 2004;199:649–659. doi: 10.1084/jem.20031097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mougous JD, Green RE, Williams SJ, Brenner SE, Bertozzi CR. Sulfotransferases and sulfatases in mycobacteria. Chem Biol. 2002;9:767–776. doi: 10.1016/s1074-5521(02)00175-8. [DOI] [PubMed] [Google Scholar]
- 12.Senaratne RH, De Silva AD, Williams SJ, Mougous JD, Reader JR, et al. 5′-Adenosinephosphosulphate reductase (CysH) protects Mycobacterium tuberculosis against free radicals during chronic infection phase in mice. Mol Microbiol. 2006;59:1744–1753. doi: 10.1111/j.1365-2958.2006.05075.x. [DOI] [PubMed] [Google Scholar]
- 13.Newton GL, Arnold K, Price MS, Sherrill C, Delcardayre SB, et al. Distribution of thiols in microorganisms: mycothiol is a major thiol in most actinomycetes. J Bacteriol. 1996;178:1990–1995. doi: 10.1128/jb.178.7.1990-1995.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Buchmeier NA, Newton GL, Koledin T, Fahey RC. Association of mycothiol with protection of Mycobacterium tuberculosis from toxic oxidants and antibiotics. Mol Microbiol. 2003;47:1723–1732. doi: 10.1046/j.1365-2958.2003.03416.x. [DOI] [PubMed] [Google Scholar]
- 15.Jothivasan VK, Hamilton CJ. Mycothiol: synthesis, biosynthesis and biological functions of the major low molecular weight thiol in actinomycetes. Nat Prod Rep. 2008;25:1091–1117. doi: 10.1039/b616489g. [DOI] [PubMed] [Google Scholar]
- 16.Rawat M, Johnson C, Cadiz V, Av-Gay Y. Comparative analysis of mutants in the mycothiol biosynthesis pathway in Mycobacterium smegmatis. Biochem Biophys Res Commun. 2007;363:71–76. doi: 10.1016/j.bbrc.2007.08.142. [DOI] [PubMed] [Google Scholar]
- 17.Williams SJ, Senaratne RH, Mougous JD, Riley LW, Bertozzi CR. 5′-Adenosinephosphosulfate lies at a metabolic branch point in mycobacteria. J Biol Chem. 2002;277:32606–32615. doi: 10.1074/jbc.M204613200. [DOI] [PubMed] [Google Scholar]
- 18.Sun M, Andreassi JL, 2nd, Liu S, Pinto R, Triccas JA, et al. The trifunctional sulfate-activating complex (SAC) of Mycobacterium tuberculosis. J Biol Chem. 2005;280:7861–7866. doi: 10.1074/jbc.M409613200. [DOI] [PubMed] [Google Scholar]
- 19.Carroll KS, Gao H, Chen H, Stout CD, Leary JA, et al. A conserved mechanism for sulfonucleotide reduction. PLoS Biol. 2005;3:e250. doi: 10.1371/journal.pbio.0030250. doi: 10.1371/journal.pbio.0030250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boshoff HI, Myers TG, Copp BR, McNeil MR, Wilson MA, et al. The transcriptional responses of Mycobacterium tuberculosis to inhibitors of metabolism: novel insights into drug mechanisms of action. J Biol Chem. 2004;279:40174–40184. doi: 10.1074/jbc.M406796200. [DOI] [PubMed] [Google Scholar]
- 21.Schnappinger D, Ehrt S, Voskuil MI, Liu Y, Mangan JA, et al. Transcriptional adaptation of Mycobacterium tuberculosis within macrophages: insights into the phagosomal environment. J Exp Med. 2003;198:693–704. doi: 10.1084/jem.20030846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pinto R, Tang QX, Britton WJ, Leyh TS, Triccas JA. The Mycobacterium tuberculosis cysD and cysNC genes form a stress-induced operon that encodes a tri-functional sulfate-activating complex. Microbiology. 2004;150:1681–1686. doi: 10.1099/mic.0.26894-0. [DOI] [PubMed] [Google Scholar]
- 23.Voskuil MI, Visconti KC Schoolnik GK. Mycobacterium tuberculosis gene expression during adaptation to stationary phase and low-oxygen dormancy. Tuberculosis (Edinb) 2004;84:218–227. doi: 10.1016/j.tube.2004.02.003. [DOI] [PubMed] [Google Scholar]
- 24.Rustad TR, Harrell MI, Liao R, Sherman DR. The enduring hypoxic response of Mycobacterium tuberculosis. PLoS ONE. 2008;3:e1502. doi: 10.1371/journal.pone.0001502. doi: 10.1371/journal.pone.0001502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fontan P, Aris V, Ghanny S, Soteropoulos P, Smith I. Global transcriptional profile of Mycobacterium tuberculosis during THP-1 human macrophage infection. Infect Immun. 2007;76:717–725. doi: 10.1128/IAI.00974-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hampshire T, Soneji S, Bacon J, James BW, Hinds J, et al. Stationary phase gene expression of Mycobacterium tuberculosis following a progressive nutrient depletion: a model for persistent organisms? Tuberculosis (Edinb) 2004;84:228–238. doi: 10.1016/j.tube.2003.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Betts JC, Lukey PT, Robb LC, McAdam RA, Duncan K. Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Mol Microbiol. 2002;43:717–731. doi: 10.1046/j.1365-2958.2002.02779.x. [DOI] [PubMed] [Google Scholar]
- 28.Manganelli R, Voskuil MI Schoolnik GK, Smith I. The Mycobacterium tuberculosis ECF sigma factor sigmaE: role in global gene expression and survival in macrophages. Mol Microbiol. 2001;41:423–437. doi: 10.1046/j.1365-2958.2001.02525.x. [DOI] [PubMed] [Google Scholar]
- 29.Provvedi R, Boldrin F, Falciani F, Palu G, Manganelli R. Global transcriptional response to vancomycin in Mycobacterium tuberculosis. Microbiology. 2009;155:1093–1102. doi: 10.1099/mic.0.024802-0. [DOI] [PubMed] [Google Scholar]
- 30.Rohde K, Yates RM, Purdy GE, Russell DG. Mycobacterium tuberculosis and the environment within the phagosome. Immunol Rev. 2007;219:37–54. doi: 10.1111/j.1600-065X.2007.00547.x. [DOI] [PubMed] [Google Scholar]
- 31.Appelberg R. Macrophage nutriprive antimicrobial mechanisms. J Leukoc Biol. 2006;79:1117–1128. doi: 10.1189/jlb.0206079. [DOI] [PubMed] [Google Scholar]
- 32.Pinto R, Harrison JS, Hsu T, Jacobs WR, Jr, Leyh TS. Sulfite reduction in mycobacteria. J Bacteriol. 2007;189:6714–6722. doi: 10.1128/JB.00487-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.O'Leary SE, Jurgenson CT, Ealick SE, Begley TP. O-phospho-L-serine and the thiocarboxylated sulfur carrier protein CysO-COSH are substrates for CysM, a cysteine synthase from Mycobacterium tuberculosis. Biochemistry. 2008;47:11606–11615. doi: 10.1021/bi8013664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schnell R, Schneider G. Structural enzymology of sulphur metabolism in Mycobacterium tuberculosis. Biochem Biophys Res Commun. 2010;396:33–38. doi: 10.1016/j.bbrc.2010.02.118. [DOI] [PubMed] [Google Scholar]
- 35.Boshoff HI, Mizrahi V, Barry CE, 3rd Effects of pyrazinamide on fatty acid synthesis by whole mycobacterial cells and purified fatty acid synthase I. J Bacteriol. 2002;184:2167–2172. doi: 10.1128/JB.184.8.2167-2172.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Criddle DN, Gillies S, Baumgartner-Wilson HK, Jaffar M, Chinje EC, et al. Menadione-induced reactive oxygen species generation via redox cycling promotes apoptosis of murine pancreatic acinar cells. J Biol Chem. 2006;281:40485–40492. doi: 10.1074/jbc.M607704200. [DOI] [PubMed] [Google Scholar]
- 37.Turnbull AL, Surette MG. L-Cysteine is required for induced antibiotic resistance in actively swarming Salmonella enterica serovar Typhimurium. Microbiology. 2008;154:3410–3419. doi: 10.1099/mic.0.2008/020347-0. [DOI] [PubMed] [Google Scholar]
- 38.Hullo MF, Martin-Verstraete I, Soutourina O. Complex phenotypes of a mutant inactivated for CymR, the global regulator of cysteine metabolism in Bacillus subtilis. FEMS Microbiol Lett. 2010;309:201–207. doi: 10.1111/j.1574-6968.2010.02043.x. [DOI] [PubMed] [Google Scholar]
- 39.Soutourina O, Dubrac S, Poupel O, Msadek T, Martin-Verstraete I. The pleiotropic CymR regulator of Staphylococcus aureus plays an important role in virulence and stress response. PLoS Pathog. 2010;6:e1000894. doi: 10.1371/journal.ppat.1000894. doi: 10.1371/journal.ppat.1000894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Miller CC, Rawat M, Johnson T, Av-Gay Y. Innate protection of Mycobacterium smegmatis against the antimicrobial activity of nitric oxide is provided by mycothiol. Antimicrob Agents Chemother. 2007;51:3364–3366. doi: 10.1128/AAC.00347-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Newton GL, Buchmeier N, Fahey RC. Biosynthesis and functions of mycothiol, the unique protective thiol of Actinobacteria. Microbiol Mol Biol Rev. 2008;72:471–494. doi: 10.1128/MMBR.00008-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vilcheze C, Av-Gay Y, Attarian R, Liu Z, Hazbon MH, et al. Mycothiol biosynthesis is essential for ethionamide susceptibility in Mycobacterium tuberculosis. Mol Microbiol. 2008;69:1316–1329. doi: 10.1111/j.1365-2958.2008.06365.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mehra S, Kaushal D. Functional genomics reveals extended roles of the Mycobacterium tuberculosis stress response factor sigmaH. J Bacteriol. 2009;191:3965–3980. doi: 10.1128/JB.00064-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Park ST, Kang CM, Husson RN. Regulation of the SigH stress response regulon by an essential protein kinase in Mycobacterium tuberculosis. Proc Natl Acad Sci U S A. 2008;105:13105–13110. doi: 10.1073/pnas.0801143105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Park JH, Roe JH. Mycothiol regulates and is regulated by a thiol-specific antisigma factor RsrA and sigma(R) in Streptomyces coelicolor. Mol Microbiol. 2008;68:861–870. doi: 10.1111/j.1365-2958.2008.06191.x. [DOI] [PubMed] [Google Scholar]
- 46.Kredich NM. The molecular basis for positive regulation of cys promoters in Salmonella typhimurium and Escherichia coli. Mol Microbiol. 1992;6:2747–2753. doi: 10.1111/j.1365-2958.1992.tb01453.x. [DOI] [PubMed] [Google Scholar]
- 47.Iwanicka-Nowicka R, Hryniewicz MM. A new gene, cbl, encoding a member of the LysR family of transcriptional regulators belongs to Escherichia coli cys regulon. Gene. 1995;166:11–17. doi: 10.1016/0378-1119(95)00606-8. [DOI] [PubMed] [Google Scholar]
- 48.Stec E, Witkowska-Zimny M, Hryniewicz MM, Neumann P, Wilkinson AJ, et al. Structural basis of the sulphate starvation response in E. coli: crystal structure and mutational analysis of the cofactor-binding domain of the Cbl transcriptional regulator. J Mol Biol. 2006;364:309–322. doi: 10.1016/j.jmb.2006.06.033. [DOI] [PubMed] [Google Scholar]
- 49.Mougous JD, Petzold CJ, Senaratne RH, Lee DH, Akey DL, et al. Identification, function and structure of the mycobacterial sulfotransferase that initiates Sulfolipid-1 biosynthesis. Nat Struct Mol Biol. 2004;11:721–729. doi: 10.1038/nsmb802. [DOI] [PubMed] [Google Scholar]
- 50.Burns KE, Baumgart S, Dorrestein PC, Zhai H, McLafferty FW, et al. Reconstitution of a new cysteine biosynthetic pathway in Mycobacterium tuberculosis. J Am Chem Soc. 2005;127:11602–11603. doi: 10.1021/ja053476x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Agren D, Schnell R, Oehlmann W, Singh M, Schneider G. Cysteine synthase (CysM) of Mycobacterium tuberculosis is an O-phosphoserine sulfhydrylase: evidence for an alternative cysteine biosynthesis pathway in mycobacteria. J Biol Chem. 2008;283:31567–31574. doi: 10.1074/jbc.M804877200. [DOI] [PubMed] [Google Scholar]
- 52.Ruckert C, Milse J, Albersmeier A, Koch DJ, Puhler A, et al. The dual transcriptional regulator CysR in Corynebacterium glutamicum ATCC 13032 controls a subset of genes of the McbR regulon in response to the availability of sulphide acceptor molecules. BMC Genomics. 2008;9:483. doi: 10.1186/1471-2164-9-483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wooff E, Michell SL, Gordon SV, Chambers MA, Bardarov S, et al. Functional genomics reveals the sole sulphate transporter of the Mycobacterium tuberculosis complex and its relevance to the acquisition of sulphur in vivo. Mol Microbiol. 2002;43:653–663. doi: 10.1046/j.1365-2958.2002.02771.x. [DOI] [PubMed] [Google Scholar]
- 54.Zolotarev AS, Unnikrishnan M, Shmukler BE, Clark JS, Vandorpe DH, et al. Increased sulfate uptake by E. coli overexpressing the SLC26-related SulP protein Rv1739c from Mycobacterium tuberculosis. Comp Biochem Physiol A Mol Integr Physiol. 2008;149:255–266. doi: 10.1016/j.cbpa.2007.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bojarova P, Williams SJ. Sulfotransferases, sulfatases and formylglycine-generating enzymes: a sulfation fascination. Curr Opin Chem Biol. 2008;12:573–581. doi: 10.1016/j.cbpa.2008.06.018. [DOI] [PubMed] [Google Scholar]
- 56.Carlson BL, Ballister ER, Skordalakes E, King DS, Breidenbach MA, et al. Function and structure of a prokaryotic formylglycine generating enzyme. J Biol Chem. 2008;283:20117–20125. doi: 10.1074/jbc.M800217200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hossain MM, Kawarabayasi Y, Kimura M, Kakuta Y. Expression and functional analysis of a predicted AtsG arylsulphatase identified from Mycobacterium tuberculosis genomic data. J Biochem. 2009;146:767–769. doi: 10.1093/jb/mvp141. [DOI] [PubMed] [Google Scholar]
- 58.Hatzios SK, Iavarone AT, Bertozzi CR. Rv2131c from Mycobacterium tuberculosis is a CysQ 3′-phosphoadenosine-5′-phosphatase. Biochemistry. 2008;47:5823–5831. doi: 10.1021/bi702453s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pi N, Hoang MB, Gao H, Mougous JD, Bertozzi CR, et al. Kinetic measurements and mechanism determination of Stf0 sulfotransferase using mass spectrometry. Anal Biochem. 2005;341:94–104. doi: 10.1016/j.ab.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 60.Neuwald AF, Krishnan BR, Brikun I, Kulakauskas S, Suziedelis K, et al. cysQ, a gene needed for cysteine synthesis in Escherichia coli K-12 only during aerobic growth. J Bacteriol. 1992;174:415–425. doi: 10.1128/jb.174.2.415-425.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang J, Biswas I. 3′-Phosphoadenosine-5′-phosphate phosphatase activity is required for superoxide stress tolerance in Streptococcus mutans. J Bacteriol. 2009;191:4330–4340. doi: 10.1128/JB.00184-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ung KS, Av-Gay Y. Mycothiol-dependent mycobacterial response to oxidative stress. FEBS Lett. 2006;580:2712–2716. doi: 10.1016/j.febslet.2006.04.026. [DOI] [PubMed] [Google Scholar]
- 63.Rodriguez GM, Smith I. Mechanisms of iron regulation in mycobacteria: role in physiology and virulence. Mol Microbiol. 2003;47:1485–1494. doi: 10.1046/j.1365-2958.2003.03384.x. [DOI] [PubMed] [Google Scholar]
- 64.Krawczyk J, Kohl TA, Goesmann A, Kalinowski J, Baumbach J. From Corynebacterium glutamicum to Mycobacterium tuberculosis--towards transfers of gene regulatory networks and integrated data analyses with MycoRegNet. Nucleic Acids Res. 2009;37:e97. doi: 10.1093/nar/gkp453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Fan F, Vetting MW, Frantom PA, Blanchard JS. Structures and mechanisms of the mycothiol biosynthetic enzymes. Curr Opin Chem Biol. 2009;13:451–459. doi: 10.1016/j.cbpa.2009.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bykowski T, van der Ploeg JR, Iwanicka-Nowicka R, Hryniewicz MM. The switch from inorganic to organic sulphur assimilation in Escherichia coli: adenosine 5′-phosphosulphate (APS) as a signaling molecule for sulphate excess. Mol Microbiol. 2002;43:1347–1358. doi: 10.1046/j.1365-2958.2002.02846.x. [DOI] [PubMed] [Google Scholar]
- 67.Newton GL, Fahey RC. Regulation of mycothiol metabolism by sigma(R) and the thiol redox sensor anti-sigma factor RsrA. Mol Microbiol. 2008;68:805–809. doi: 10.1111/j.1365-2958.2008.06222.x. [DOI] [PubMed] [Google Scholar]
- 68.Buchmeier NA, Newton GL, Fahey RC. A mycothiol synthase mutant of Mycobacterium tuberculosis has an altered thiol-disulfide content and limited tolerance to stress. J Bacteriol. 2006;188:6245–6252. doi: 10.1128/JB.00393-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mougous JD, Lee DH, Hubbard SC, Schelle MW, Vocadlo DJ, et al. Molecular basis for G protein control of the prokaryotic ATP sulfurylase. Mol Cell. 2006;21:109–122. doi: 10.1016/j.molcel.2005.10.034. [DOI] [PubMed] [Google Scholar]
- 70.Raman K, Yeturu K, Chandra N. targetTB: a target identification pipeline for Mycobacterium tuberculosis through an interactome, reactome and genome-scale structural analysis. BMC Syst Biol. 2008;2:109. doi: 10.1186/1752-0509-2-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schnell R, Oehlmann W, Singh M, Schneider G. Structural insights into catalysis and inhibition of O-acetylserine sulfhydrylase from Mycobacterium tuberculosis. Crystal structures of the enzyme alpha-aminoacrylate intermediate and an enzyme-inhibitor complex. J Biol Chem. 2007;282:23473–23481. doi: 10.1074/jbc.M703518200. [DOI] [PubMed] [Google Scholar]
- 72.Voskuil MI. Mycobacterium tuberculosis gene expression during environmental conditions associated with latency. Tuberculosis (Edinb) 2004;84:138–143. doi: 10.1016/j.tube.2003.12.008. [DOI] [PubMed] [Google Scholar]