Abstract
Purpose
There is growing evidence that stress and other behavioral factors may affect cancer progression and patient survival. The underlying mechanisms for this association are poorly understood. The purpose of this study is to determine the effects of stress-associated hormones norepinephrine, epinephrine, and cortisol on the invasive potential of ovarian cancer cells.
Experimental Design
The ovarian cancer cells EG, SKOV3, and 222 were exposed to increasing levels of either norepinephrine, epinephrine, or cortisol, and the in vitro invasive potential was determined using the membrane invasion culture system. Additionally, the effects of these stress hormones on matrix metalloproteinase-2 (MMP-2) and MMP-9 were determined by ELISA. The effects of the β-adrenergic agonist isoproterenol on in vivo tumor growth were determined using nude mice.
Results
Stress levels of norepinephrine increased the in vitro invasiveness of ovarian cancer cells by 89% to 198%. Epinephrine also induced significant increases in invasion in all three cell lines ranging from 64% to 76%. Cortisol did not significantly affect invasiveness of the EG and 222 cell lines but increased invasion in the SKOV3 cell line (P = 0.01). We have previously shown that ovarian cancer cells express β-adrenergic receptors. The β-adrenergic antagonist propanolol (1 µmol/L) completely blocked the norepinephrine-induced increase in invasiveness. Norepinephrine also increased tumor cell expression of MMP-2 (P = 0.02 for both SKOV3 and EG cells) and MMP-9 (P = 0.01 and 0.04, respectively), and pharmacologic blockade of MMPs abrogated the effects of norepinephrine on tumor cell invasive potential. Isoproterenol treatment resulted in a significant increase in tumor volume and infiltration in the SKOV3ip1 in vivo model, which was blocked by propranolol.
Conclusions
These findings provide direct experimental evidence that stress hormones can enhance the invasive potential of ovarian cancer cells. These effects are most likely mediated by stimulation of MMPs.
There is extensive evidence supporting stress-immune relationships in healthy adults (1) and a growing body of literature demonstrating these relationships in cancer patients (2–4). Meta-analyses and reviews have reported alterations in cellular immunity (decreased T-cell response to mitogen stimulation, decreased natural killer cell cytotoxicity, and altered production of cytokines) in association with chronic stress and/or depressed affect (5, 6). Among cancer patients, behavioral factors may serve as predictors of clinical outcome, such as response to therapy and overall survival (7–11). These findings suggest that psychosocial stress factors not only affect the immune system adversely but also contribute to poor outcome in cancer patients. However, no study has shown that stress-induced changes in cancer outcomes are mediated by changes in immune system function. Here, we consider the alternative hypothesis that stress hormones directly affect tumor cells to alter their malignant potential.
Immune system cells express receptors for glucocorticoids from the hypothalamic-pituitary-adrenal axis and catecholamines from the sympathetic nervous system. These signals alter several aspects of immune cell function, including cellular activation, cytokine production, and cell trafficking (4, 12). However, the direct effects of stress hormones (from the sympathetic nervous system and hypothalamic-pituitary-adrenal axis) on cancer cells have not been well characterized. We recently examined ovarian carcinoma cells to assess the expression of receptors for stress hormones and the functional consequences of their ligation (13). Those studies found significant effects of catecholamines on production of proangiogenic cytokines, which are mediated by β-adrenergic activation of the cyclic AMP/protein kinase A signaling pathway (13). The catecholamines epinephrine and norepinephrine are components of the sympathetic response that are manufactured by nerves of the sympathetic nervous system and the adrenal medulla. Both norepinephrine and epinephrine are elevated in individuals with acute or chronic stress (14, 15) and all major catecholamine neurotransmitters are present in the ovary (16, 17). Among them, norepinephrine is the most abundant and plays functionally relevant roles in ovarian steroidogenesis and follicular development (18–21). Catecholamine concentrations are substantially higher in the ovarian tissue than in circulating plasma (22). We have previously shown that norepinephrine and the β-adrenergic agonist isoproterenol can enhance the production of a proangiogenic cytokine, vascular endothelial growth factor, by ovarian cancer cells (13). These effects were mediated by β-adrenergic receptors and were completely blocked by β-adrenergic antagonist propranolol. Increased production of proangiogenic factors suggests that catecholamines could potentially facilitate tumor metastasis in vivo. However, metastasis is a complex process involving several sequential, interrelated steps, each of which can be rate limiting. To produce clinically relevant lesions, metastatic cells must complete all the steps of the process (23). Invasion is a key step in the pathogenesis of metastasis (24, 25) but little is known about how catecholamines might affect the invasive potential of tumor cells. Indirect evidence suggests that catecholamines could potentially enhance invasive capacity because norepinephrine has been shown to affect tumor cell motility in vitro (26–28) and circulating catecholamines have been associated in vivo with tissue expression of matrix metalloproteinases (MMP) that facilitate invasion (29, 30). Those data provide circumstantial evidence suggesting that catecholamines could potentially affect the cellular and molecular processes involved in tumor cell invasion but no studies have directly evaluated this hypothesis by examining invasive function (i.e., capacity of tumor cells to penetrate extracellular matrix; ref. 31).
In this study, we examined the effects of catecholamines and cortisol on the invasive potential of ovarian carcinoma cells and their production of key MMP proteins involved in tumor cell penetration of extracellular matrix. Results show that β-adrenergic signaling significantly up-regulates the invasive capacity of three different ovarian cancer cell lines via β-adrenergic regulation of MMP proteins.
Materials and Methods
Cell culture
The ovarian cancer cell lines used in this study were SKOV3, SKOV3ip1, EG, and 222. The derivation and sources of the cell lines have been reported previously (25, 31, 32). These cells were maintained and propagated in vitro by serial passage in RPMI 1640 supplemented with 15% fetal bovine serum and 0.1% gentamicin sulfate (Gemini Bioproducts, Calabasas, CA). All of the cell lines are routinely screened for Mycoplasma species (GenProbe detection kit; Fisher, Itasca, IL). All of the experiments were done with 70% to 80% confluent cultures.
Invasion assay
The membrane invasion culture system chamber was used to measure the in vitro invasiveness of all cell lines used in this study (25, 31, 33). Briefly, a polycarbonate membrane with 10 µmol/L pores (Osmonics, Livermore, CA) was uniformly coated with a defined basement membrane matrix consisting of human laminin/type IV collagen/gelatin and used as the intervening barrier to invasion. The defined matrix was prepared (stored at 4°C) in a 10 mL stock solution as follows: laminin (50 µg/mL), 1 mL + type IV collagen (50 µg/mL), 0.2 mL + gelatin (2 mg/mL), and 4 mL + 4.8 mL PBS. Using a disposable pipette, 1 mL of the matrix solution was dispensed across a long side of the membrane. An 8 mm glass rod was used to spread the matrix across the membrane and allowed to dry for 30 minutes. The matrix-coated filter was placed coated side up on the lower plate followed by carefully attaching the upper plate. Both upper and lower wells of the chamber were filled with serum-free RPMI containing 1 × MITO+ (Collaborative Biomedical, Bedford, MA). Single-cell tumor suspensions were seeded into the upper wells at a concentration of 1 × 105 cells per well. Following a 24-hour incubation in a humidified incubator at 37°C with 5% CO2, cells that had invaded through the basement membrane were collected through the side port by replacing the medium in the lower chamber with 2 mmol/L EDTA/PBS (pH 7.4) for 20 minutes at 37°C. The cells recovered from the bottom of the filter were then loaded onto a dot blot manifold containing 3 µm pore polycarbonate filters, fixed, stained, and counted by light microscopy (25, 31). Invasiveness was calculated as the percentage of cells that had successfully invaded through the matrix-coated membrane to the lower wells relative to the total number of cells seeded into the upper wells. The invasion assays were done in triplicate and repeated once.
Determination of MMP concentration
Serum-free conditioned medium from cultures of ovarian cancer cells was collected at 3, 6, 12, and 24 hours following exposure to catecholamines or cortisol. The supernatants were microfuged to remove debris and then stored at −80°C. The samples were thawed only once for determining the MMP concentration. An identical number of cells were plated without the three-dimensional matrix for comparison. The protein concentration of total MMP-2 (pro-MMP-2 and active MMP-2) and total MMP-9 (92 kDa pro-MMP2 and 82 kDa active forms) were determined using Quantikine immunoassays (R&D Systems, Minneapolis, MN) as per the protocol of the manufacturer. The concentrations of active MMP-2 and MMP-9 were determined using the Biotrak Activity Assay System (Amersham Biosciences, Piscataway, NJ) as per the protocol of the manufacturer. The MMP experiments were done in triplicate and were repeated once.
MMP inhibition
Inhibition of the MMPs was accomplished using a chemically modified tetracycline (CMT-3; CollaGenex Pharmaceuticals, Newton, PA). CMT-3 retains broad-spectrum inhibitory activity against MMPs but does not possess antimicrobial properties (34, 35). CMT-3 (5 µg/mL) was added to the different wells of the chamber postseeding (34).
In vivo tumor model
Female nude mice were purchased from the National Cancer Institute-Frederick Cancer Research Facility (Frederick, MD). The mice were housed and maintained under specific pathogen-free conditions in facilities approved by the American Association for Accreditation of Laboratory Animal Care in accordance with current regulations and standards of the U.S. Department of Agriculture, DHHS, and the NIH. The mice were used according to institutional guidelines when they were 8 to 12 weeks of age. The tumor cells (SKOV3ip1) were harvested from subconfluent cultures by a brief exposure to 0.25% trypsin and 0.02% EDTA. Trypsinization was stopped with medium containing 10% fetal bovine serum. The cells were then washed once in serum-free medium and resuspended in HBSS (serum free). Only single-cell suspensions with >95% viability, as determined by trypan blue exclusion, were used for the injections. To produce tumors, 1 × 106 SKOV3ip1 cells (0.1 mL) were injected s.c. into the right flank of the nude mice. A total of five mice per group were used. Starting 24 hours after tumor cell injection, mice were treated with daily i.p. injections of PBS, isoproterenol (10 mg/kg), or isoproterenol (10 mg/kg) in combination with propranolol (2 mg/kg) for 7 days. All treatments were administered in a total volume of 200 µL. Eight days after tumor cell injection, mice were euthanized by cervical dislocation. Tumors were measured in two dimensions, dissected, and fixed in formalin. Tumor volume was calculated as (length / 2) × (width2). Tumor samples were analyzed using H&E staining. Representative images were taken from each tumor using a light microscope at ×40 and ×100 magnification.
Statistical analysis
The χ2 test was used to determine differences between cell counts using SPSS (SPSS, Inc., Chicago, IL). Hormone-mediated changes in MMP concentrations over time were analyzed using a dose × time ANOVA with both factors treated as repeated measures. Differences in tumor volume were determined using Student’s t test. P < 0.05 was considered statistically significant.
Results
Catecholamines stimulate ovarian cancer invasion
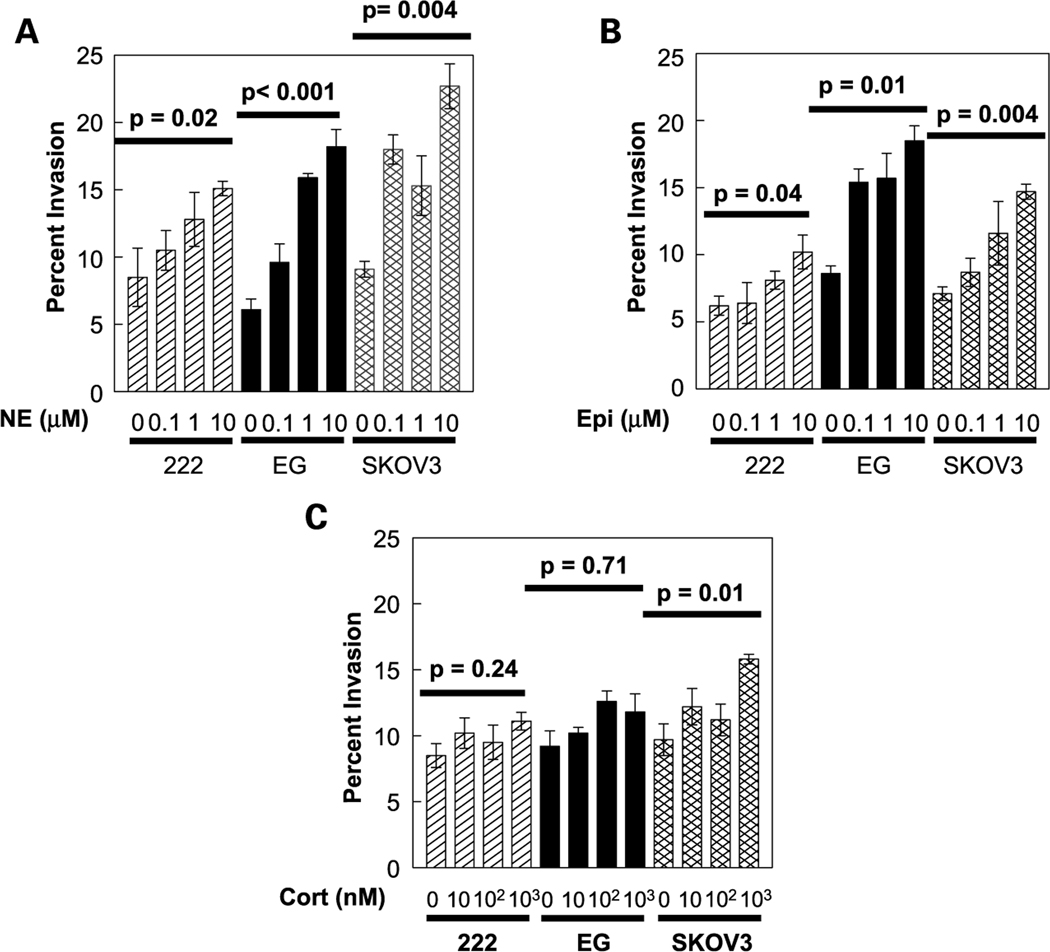
Tumor cell invasion is a key step in the metastatic process. To determine whether catecholamines might enhance ovarian cancer invasion, the SKOV3, EG, and 222 cells were exposed to a range of catecholamine doses and assayed 24 hours later for penetration of a basement membrane barrier using the membrane invasion culture system assay (31). Baseline invasion rates of these ovarian cancer cell lines have been reported previously (31). Under basal conditions, 6.1% of EG cells penetrated the basement membrane barrier in 24 hours. Norepinephrine induced a dose-dependent increase in invasive capacity that peaked with an ~3-fold enhancement in the frequency of penetrating cells at 10 µmol/L norepinephrine (P < 0.001; Fig. 1A). Similar effects were observed for the other cell lines studied, with norepinephrine producing peak increases in invasive capacity ranging from 2-fold in 222 cells (P = 0.02) to 2.8-fold in SKOV3 cells (P = 0.004; Fig. 1A). The catecholamine epinephrine also enhanced ovarian cancer cell invasive potential, although its effects were less pronounced than those of norepinephrine (Fig. 1B). Epinephrine induced dose-dependent increases in invasive capacity for all three cell lines with a 1.6-fold maximum increase in SKOV3 cells (P = 0.004), 1.8-fold in EG cells (P = 0.01), and 1.7-fold in the 222 cells (P = 0.04; Fig. 1B).
Fig. 1.
Invasion profile of ovarian cancer cell lines (222, EG, and SKOV3) in the presence or absence of norepinephrine (NE; A), epinephrine (Epi; B), or cortisol (Cort; C). Bars, SE.
Cortisol is a glucocorticoid hormone released by the adrenal cortex in response to stress (36, 37) and some studies have found that it can potentiate the effects of catecholamines on cancer cells (38). In the present studies, cortisol weakly stimulated ovarian cancer cell invasive potential (Fig. 1C), but these results failed to reach statistical significance in two of the three cell lines studied. Only in the SKOV3 cells did cortisol induce significant increases in invasive capacity and the peak 1.6-fold increase was substantially smaller than that observed for the catecholamines epinephrine and norepinephrine (Fig. 1C). Because stress can increase levels of both cortisol and catecholamines, we also examined the invasive potential of ovarian cancer cells following costimulation with cortisol and either norepinephrine or epinephrine. Results showed increases in invasion that paralleled the effects of catecholamines alone, but no additive or synergistic effects were noted (data not shown).
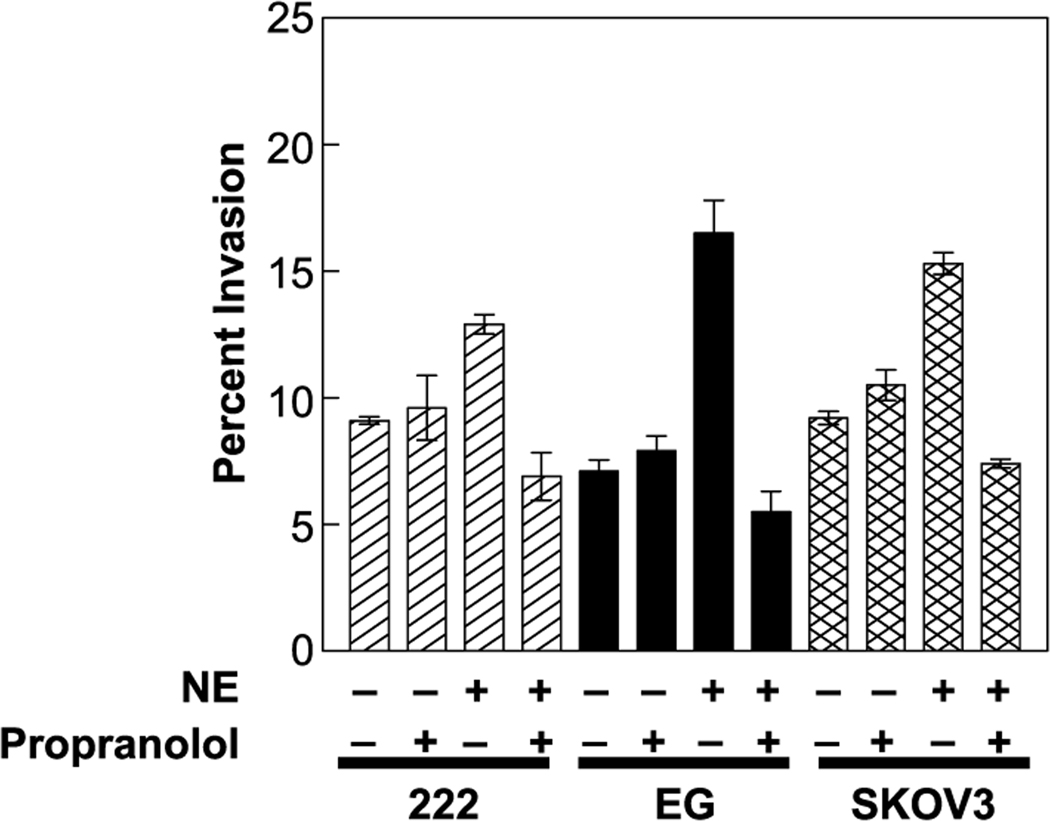
We have previously shown that the ovarian cancer cells used in this study express both β1 and β2 adrenergic receptors (13). To determine whether those receptors mediate catecholamine effects on invasive potential, we treated ovarian cancer cells with the broad β-blocker propranolol immediately following their introduction into the membrane invasion culture system chamber and introduced catecholamines 30 minutes later. Propranolol (1 µmol/L) completely blocked norepinephrine-mediated increases in invasion (Fig. 2), but propranolol alone had no effect on invasive capacity (data not shown).
Fig. 2.
Effect of β-blocker (propranolol) on ovarian cancer invasion in the presence or absence of norepinephrine. Bars, SE.
Catecholamine mediated up-regulation of MMPs
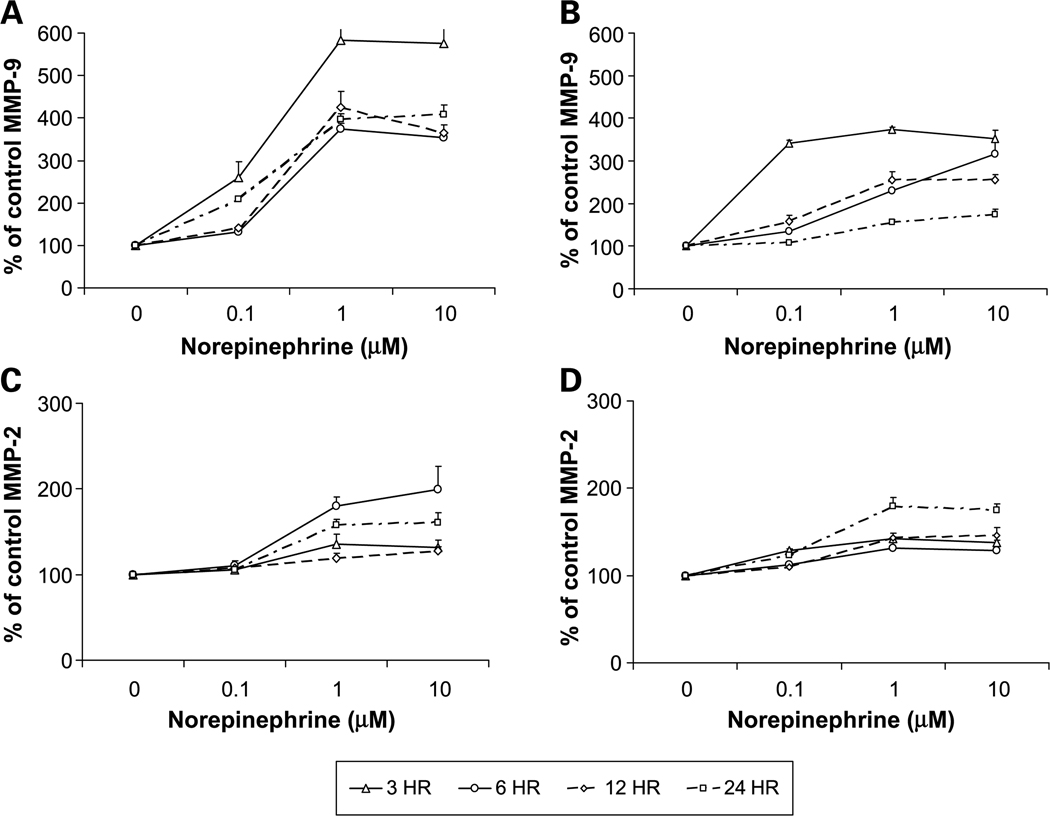
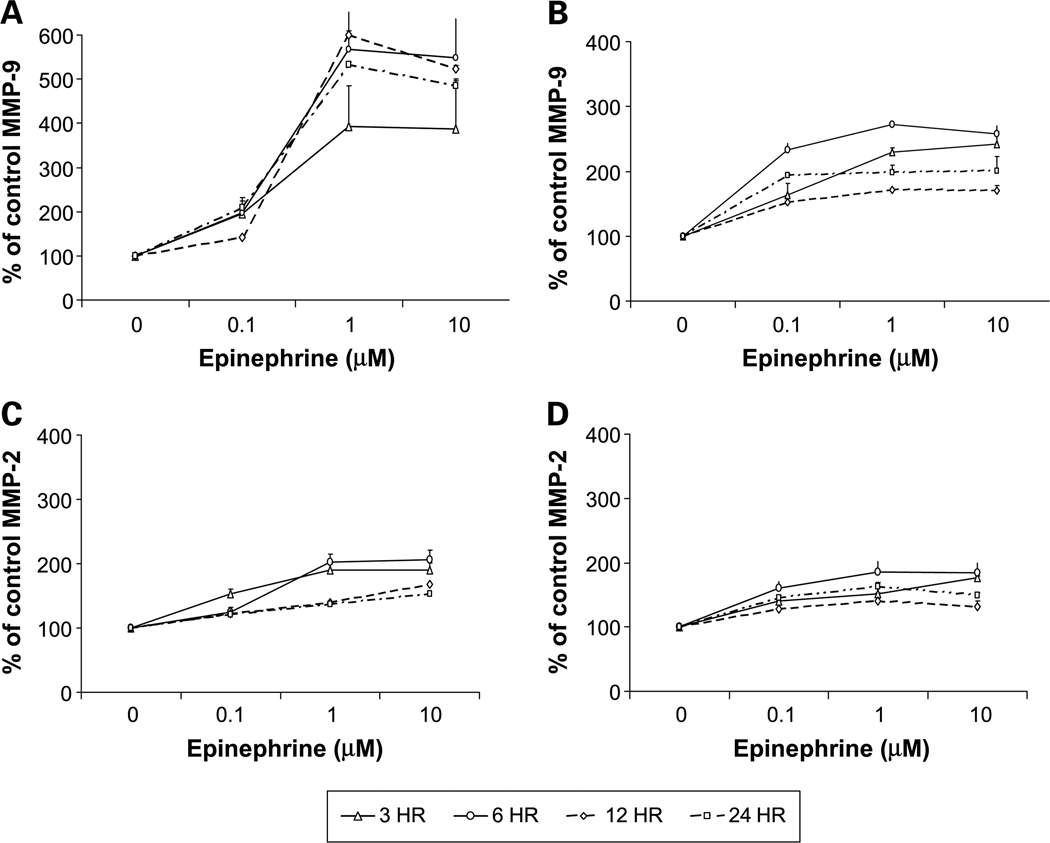
MMPs degrade most components of the extracellular matrix (39, 40) and MMP-2 and MMP-9 are believed to play a key role in ovarian cancer invasion (36, 41–45). To determine whether altered MMP expression mediated catecholamine effects on ovarian cancer cell invasive potential, we treated cultured EG or SKOV3 cells with the same hormone doses used in the invasion assays and determined supernatant MMP levels 3, 6, 12, and 24 hours later. Norepinephrine (1 µmol/L) induced a peak 5-fold increase in MMP-9 levels in the SKOV3 supernatants 3 hours poststimulation, and a 4-fold increase for EG cells at the same time point (P < 0.001 for both cell lines; Fig. 3A–B). Norepinephrine also significantly increased MMP-2 levels, although these effects were smaller in magnitude and peaked later than for MMP-9 (P < 0.01 for both cell lines). Norepinephrine (1 µmol/L) induced a 25% to 55% increase in active MMP-9 and 35% to 48% increase in active MMP-2 levels in both cell lines, 6 hours after treatment (data not shown). Epinephrine also enhanced total MMP-9 levels to a peak 5-fold increase at 12 hours for SKOV3 cells (Fig. 4A) and a peak 2.7-fold increase at 6 hours (Fig. 4B). Total MMP-2 levels also showed epinephrine-induced peaks at 6 hours in both cell lines (Fig. 4C–D). Epinephrine 1 µmol/L resulted in a 22% to 53% increase in active MMP-9 and 33% to 63% increase in active MMP-2 levels in both cell lines, 6 hours posttreatment (data not shown). Cortisol had much weaker effects, inducing a slight increase in both MMP-2 and MMP-9 levels at physiologic stress levels but a slight decrease at pharmacologic levels (data not shown). Effects of cortisol were not statistically significant at any time point for either cell line.
Fig. 3.
MMP-9 (A and B) and MMP-2 (C and D) concentrations in conditioned medium from SKOV3 (A and C) and EG(B and D) cell lines after treatment with norepinephrine. Bars, SE.
Fig. 4.
MMP-9 (A and B) and MMP-2 (C and D) concentrations in conditioned medium from SKOV3 (A and C) and EG (B and D) cell lines after treatment with epinephrine. Bars, SE.
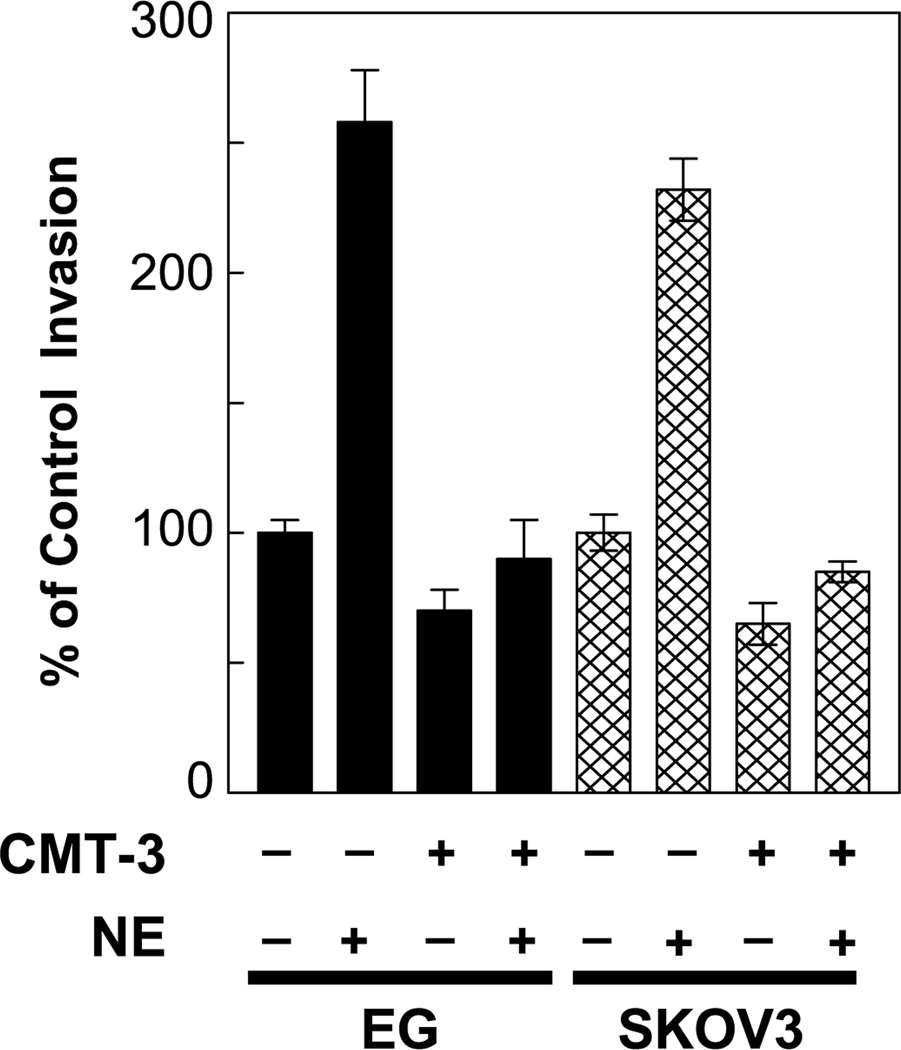
To determine whether increased MMP expression was responsible for the effect of norepinephrine on the invasive potential of ovarian cancer cells, we blocked MMP production with CMT-3, which is a chemically modified tetracycline (34). CMT-3 alone decreased invasion by approximately one third in both EG and SKOV3 cells (Fig. 5). Treatment of either EG or SKOV3 cells with 5 µg/mL CMT-3 completely blocked the norepinephrine(1 µmol/L)–induced increases in invasion (Fig. 5). CMT-3 also blocked the effects of epinephrine on invasive capacity (data not shown). These results suggest that MMPs play a critical role in mediating catecholamine effects on the ability of ovarian cancer cells to invade the basement membrane in the membrane invasion culture system model.
Fig. 5.
The in vitro invasive potential of ovarian cancer cell lines (EG and SKOV3) through a defined basement membrane matrix over 24 hours was normalized to a value of 100%, and the invasive potential cells treated with 1 µmol/L norepinephrine and/or 5 µg/mL CMT-3 during the assay compared with the control value of 100%. Bars, SE.
Isoproterenol promotes tumor growth and infiltration in vivo
To discern the effects of catecholamines in vivo, female nude mice were injected s.c. with SKOV3ip1 cells and treated according to the following groups (n = 5 per group); (a) daily PBS i.p.; (b) daily isoproterenol (10 mg/kg) i.p.; (c) daily isoproterenol (10 mg/kg) i.p. plus propranolol (2 mg/kg) i.p. for 7 days. The tumor volume in the PBS group was 31.1 ± 7.5 mm3 (Table 1) and was significantly increased in the isoproterenol-only group (P = 0.04). Treatment with propranolol blocked the isoproterenol-induced increase in tumor volume (Table 1). Although the depth of invasion could not be measured specifically because of lack of a consistent landmark, four of five tumors in the isoproterenol group infiltrated deeply into the s.c. tissues, including muscle (Fig. 6C). In contrast, all of the tumors in the PBS only or the isoproterenol plus propranolol groups remained encapsulated above the muscle layer. The difference in the incidence of deep infiltration between the PBS and isoproterenol groups was statistically significant with P < 0.05. These observations suggest that β-adrenergic agonists can promote invasion in vivo, which can be abrogated by a β-blocker.
Table 1.
Incidence and final tumor volume of SKOV3ip1 cells following s.c. injection
| Treatment group | Incidence | Final tumor volume (±SE; mm3) |
|---|---|---|
| PBS | 4 of 5 | 31.1 ± 7.5 |
| Isoproterenol | 5 of 5 | 129.5 ± 34.9* |
| Isoproterenol + propranolol | 4 of 5 | 23.3 ± 4.4 |
P < 0.05.
Fig. 6.
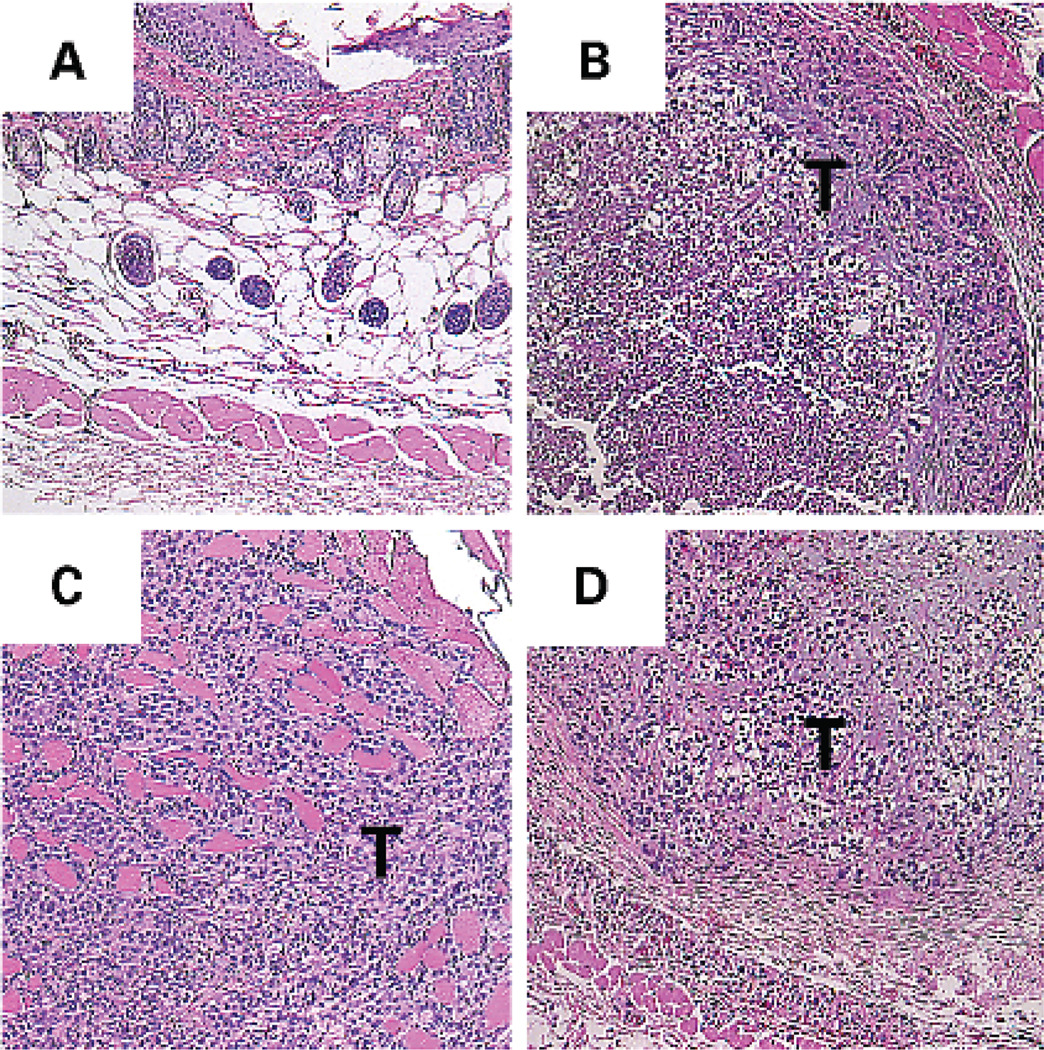
Effect of β-adrenergic agonist isoproterenol on in vivo growth of SKOV3ip1 cells injected s.c. in nude mice. H&E sections (original magnification, ×40) were obtained from (A) normal mouse skin without tumor and from tumor-bearing mice treated daily i.p. for 7 days with (B) PBS, (C) isoproterenol (10 mg/kg), or (D) isoproterenol (10 mg/kg) plus propranolol (2 mg/kg). The tumor (T) is seen infiltrating deeply through the muscle layer after daily isoproterenol treatment (C), but remained encapsulated in the other two groups.
Discussion
In this study, we addressed the effects of selected stress hormones on ovarian cancer invasion, which is a critical component of the metastatic cascade. Our data show that physiologically relevant concentrations of norepinephrine and epinephrine can significantly enhance the capacity of ovarian tumor cells to invade the extracellular matrix that is characteristic of the basement membrane. These effects are mediated via β-adrenergic receptors on cancer cells, which can enhance production of MMP-2 and MMP-9. Changes in MMP expression facilitate ovarian cancer cell penetration of extracellular matrix, and pharmacologic blockade of those effects can inhibit catecholamine-mediated increases in tumor cell invasion. Furthermore, a β-adrenergic agonist (isoproterenol) promoted in vivo tumor growth and infiltration. In addition to documenting a novel mechanism by which stress biology might effect the pathogenesis of ovarian cancer, these data identify two key molecular mediators of such effects (β-adrenergic receptors and MMP expression).
The effects observed here take place at physiologically relevant concentrations of catecholamines. Basal circulating levels of norepinephrine range between 10 pmol/L and 1 nmol/L, with stress increasing these levels to 100 nmol/L (46). Circulating epinephrine levels range from 1 to 10 pmol/L, and increase up to 10 nmol/L under stress (46). Stress can also increase tissue catecholamine levels in the ovary via increased sympathetic activity, which has been shown to trigger precystic follicles (47–50). However, in addition to the sympathetic innervation, the primate ovary contains the endogenous enzymatic machinery necessary for catecholamine biosynthesis (51). As a result, ovarian tissue levels of catecholamines can substantially exceed those in plasma (22). Therefore, the in vitro doses of catecholamines used in our experiments cover the spectrum of stress and nonstress levels that the tumor cells would be exposed to in vivo and are consistent with doses that promote biological effects in other in vitro studies (46, 52–57). Thus, the present data suggest that catecholamine effects on ovarian cancer invasive potential occur at physiologically relevant levels of those hormones.
The ability to invade extracellular matrices plays an important role in metastasis and in development of blood flow to tumors. The process of tumor cell penetration of the host basement membrane consists of attachment, matrix dissolution, motility, and penetration (58). MMPs play a key role in these dynamics by degrading components of the extracellular matrix, such as collagen, laminins, fibronectins, elastins, and the protein core of proteoglycans (39, 40). Given their destructive potential, MMPs are normally expressed only when and where they are needed to support physiologic processes, such as embryonic development, wound healing, and placental development (59, 60). Aberrant expression of MMPs contributes to several pathologic conditions, including tumor cell invasion and metastasis. MMPs associated with ovarian carcinomas include MMP-2 and MMP-9 (32, 41–45). MMP-2 is the primary gelatinolytic MMP secreted by ovarian cancer cells (32, 43) and MMP-9 contributes to the angiogenic switch that occurs during carcinogenesis (61, 62). Recently, Huang et al. (63) have shown that host-derived MMP-9 expression plays a critical role in angiogenesis and progressive growth of human ovarian tumors in mice. We have previously shown the critical role of specific MMPs in the in vitro matrix remodeling by ovarian cancer cells (31, 32). The present data show that physiologic stress hormones can significantly enhance the expression and activity of these key MMPs by ovarian cancer cells.
These data provide the first indication that MMPs might play a role in stress hormone–mediated changes in ovarian cancer cell function. Previous studies have linked hormone dynamics to MMP production by other cell types. Yang et al. (29) examined the effects of stress on MMP levels using a blister chamber wound model on UV-B–exposed human forearm skin. In their study, plasma norepinephrine levels were correlated with high MMP-2 protein levels in damaged dermal tissue. Recent studies in mice also suggest that psychological stress can elevate MMP activity. Wu et al. stressed mice by social isolation and found increased expression of mRNA for MMP-2, MMP-9, matrix-type MMP-1, and urokinase-type plasminogen activator in colon tumors and liver tissues in stressed versus control mice (30). The present study shows similar effects in the context of ovarian cancer and it extends those findings to show the functional significance of increased MMP expression in facilitating tumor cell invasion of basement membrane-like structures. It is possible that other factors may also be involved in stress hormone–stimulated invasion and will be examined in ongoing work in our laboratory.
In summary, our results show that stress hormones can directly increase the invasive potential of ovarian cancer cells via β-adrenergic up-regulation of MMP-2 and MMP-9. In conjunction with previous studies showing β-adrenergic regulation of other molecular processes involved in the metastatic cascade (26, 64), these results suggest that physiologic catecholamine activity could represent a new target for adjunctive therapies that seek to block metastatic processes during and after primary therapy. Understanding the neuroendocrine influences on cancer growth and progression might allow development of strategies to delay or prevent malignant disease.
Acknowledgments
Grant support: National Cancer Institute grants CA11079301 and CA10929801 and the Donna Marie Cimitile-Fotheringham Award for Ovarian Cancer Research (A.K. Sood) and grant AI 52737 (S.W. Cole).
References
- 1.Ader R. Psychoneuroimmunology. 3rd ed. San Diego (California): Academic Press; 2001. [Google Scholar]
- 2.Andersen BL, Kiecolt-Glaser JK, Glaser R. A biobehavioral model of cancer stress and disease course. Am Psychol. 1994;49:389–404. doi: 10.1037//0003-066x.49.5.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Andersen BL, Farrar WB, Golden-Kreutz DM, et al. Psychological, behavioral, and immune changes after a psychological intervention: a clinical trial. J Clin Oncol. 2004;22:3570–3580. doi: 10.1200/JCO.2004.06.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Glaser R, Kiecolt-Glaser JK. Stress-induced immune dysfunction: implications for health. Nat Rev Immunol. 2005;5:243–251. doi: 10.1038/nri1571. [DOI] [PubMed] [Google Scholar]
- 5.Herbert TB, Cohen S. Stress and immunity in humans: a meta-analytic review. Psychosom Med. 1993;55:364–379. doi: 10.1097/00006842-199307000-00004. [DOI] [PubMed] [Google Scholar]
- 6.Herbert TB, Cohen S. Depression and immunity: a meta-analytic review. Psychol Bull. 1993;113:472–486. doi: 10.1037/0033-2909.113.3.472. [DOI] [PubMed] [Google Scholar]
- 7.Levy SM, Herberman RB, Lippman M, et al. Immunological and psychosocial predictors of disease recurrence in patients with early-stage breast cancer. Behav Med. 1991;17:67–75. doi: 10.1080/08964289.1991.9935161. [DOI] [PubMed] [Google Scholar]
- 8.Stommel M, Given BA, Given CW. Depression and functional status as predictors of death among cancer patients. Cancer. 2002;94:2719–2727. doi: 10.1002/cncr.10533. [DOI] [PubMed] [Google Scholar]
- 9.Coates A, Porzsolt F, Osoba D. Quality of life in oncology practice: prognostic value of EORTC QLQ-C30 scores in patients with advanced malignancy. Eur J Cancer. 1997;33:1025–1030. doi: 10.1016/s0959-8049(97)00049-x. [DOI] [PubMed] [Google Scholar]
- 10.Ganz PA, Lee JJ, Siau J. Quality of life assessment. An independent prognostic variable for survival in lung cancer. Cancer. 1991;67:3131–3135. doi: 10.1002/1097-0142(19910615)67:12<3131::aid-cncr2820671232>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 11.Seidman AD, Portenoy R, Yao TJ, et al. Quality of life in phase II trials: a study of methodology and predictive value in patients with advanced breast cancer treated with paclitaxel plus granulocyte colony-stimulating factor. J Natl Cancer Inst. 1995;87:1316–1322. doi: 10.1093/jnci/87.17.1316. [DOI] [PubMed] [Google Scholar]
- 12.Sanders VM, Straub RH. Norepinephrine, the β-adrenergic receptor, and immunity. Brain Behav Immun. 2002;16:290–332. doi: 10.1006/brbi.2001.0639. [DOI] [PubMed] [Google Scholar]
- 13.Lutgendorf SK, Cole S, Costanzo E, et al. Stress-related mediators stimulate vascular endothelial growth factor secretion by two ovarian cancer cell lines. Clin Cancer Res. 2003;9:4514–4521. [PubMed] [Google Scholar]
- 14.Schmidt C, Kraft K. β-endorphin and catecholamine concentrations during chronic and acute stress in intensive care patients. Eur J Med Res. 1996;1:528–532. [PubMed] [Google Scholar]
- 15.Rupp H, Dhalla KS, Dhalla NS. Mechanisms of cardiac cell damage due to catecholamines: significance of drugs regulating central sympathetic outflow. J Cardiovasc Pharmacol. 1994;24 Suppl 1:S16–S24. doi: 10.1097/00005344-199424001-00004. [DOI] [PubMed] [Google Scholar]
- 16.Ben-Jonathan N, Arbogast LA, Rhoades TA, et al. Norepinephrine in the rat ovary: ontogeny and de novo synthesis. Endocrinology. 1984;115:1426–1431. doi: 10.1210/endo-115-4-1426. [DOI] [PubMed] [Google Scholar]
- 17.Aguado LI, Ojeda SR. Prepubertal ovarian function is finely regulated by direct adrenergic influences. Role of noradrenergic innervation. Endocrinology. 1984;114:1845–1853. doi: 10.1210/endo-114-5-1845. [DOI] [PubMed] [Google Scholar]
- 18.Aguado LI, Petrovic SL, Ojeda SR. Ovarian β-adrenergic receptors during the onset of puberty: characterization, distribution, and coupling to steroidogenic responses. Endocrinology. 1982;110:1124–1132. doi: 10.1210/endo-110-4-1124. [DOI] [PubMed] [Google Scholar]
- 19.Hernandez ER, Jimenez JL, Payne DW, et al. Adrenergic regulation of ovarian androgen biosynthesis is mediated via β2-adrenergic theca-interstitial cell recognition sites. Endocrinology. 1988;122:1592–1602. doi: 10.1210/endo-122-4-1592. [DOI] [PubMed] [Google Scholar]
- 20.Lara HE, McDonald JK, Ojeda SR. Involvement of nerve growth factor in female sexual development. Endocrinology. 1990;126:364–375. doi: 10.1210/endo-126-1-364. [DOI] [PubMed] [Google Scholar]
- 21.Mayerhofer A, Dissen GA, Costa ME, et al. A role for neurotransmitters in early follicular development: induction of functional follicle-stimulating hormone receptors in newly formed follicles of the rat ovary. Endocrinology. 1997;138:3320–3329. doi: 10.1210/endo.138.8.5335. [DOI] [PubMed] [Google Scholar]
- 22.Lara HE, Porcile A, Espinoza J, et al. Release of norepinephrine from human ovary: coupling to steroidogenic response. Endoc J. 2001;15:187–192. doi: 10.1385/ENDO:15:2:187. [DOI] [PubMed] [Google Scholar]
- 23.Fidler IJ. The pathogenesis of cancer metastasis: the “seed and soil” hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 24.Fidler IJ. Critical determinants of metastasis. Semin Cancer Biol. 2002;12:89–96. doi: 10.1006/scbi.2001.0416. [DOI] [PubMed] [Google Scholar]
- 25.Sood AK, Coffin JE, Schneider GB, et al. Biological significance of focal adhesion kinase in ovarian cancer : role in migration and invasion. Am J Pathol. 2004;165:1087–1095. doi: 10.1016/S0002-9440(10)63370-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Entschladen F, Lang K, Drell TL, et al. Neurotransmitters are regulators for the migration of tumor cells and leukocytes. Cancer Immunol Immunother. 2002;51:467–482. doi: 10.1007/s00262-002-0300-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Masur K, Niggemann B, Zanker KS, et al. Norepinephrine-induced migration of SW 480 colon carcinoma cells is inhibited by β-blockers. Cancer Res. 2001;61:2866–2869. [PubMed] [Google Scholar]
- 28.Joseph J, Niggemann B, Zaenker KS, et al. The neurotransmitter γ-aminobutyric acid is an inhibitory regulator for the migration of SW 480 colon carcinoma cells. Cancer Res. 2002;62:6467–6469. [PubMed] [Google Scholar]
- 29.Yang EV, Bane CM, MacCallum RC, et al. Stress-related modulation of matrix metalloproteinase expression. J Neuroimmunol. 2002;133:144–150. doi: 10.1016/s0165-5728(02)00270-9. [DOI] [PubMed] [Google Scholar]
- 30.Wu W, Yamaura T, Murakami K, et al. Involvement of TNF-α in enhancement of invasion and metastasis of colon 26-L5 carcinoma cells in mice by social isolation stress. Oncol Res. 1999;11:461–469. [PubMed] [Google Scholar]
- 31.Sood AK, Seftor EA, Fletcher MS, et al. Molecular determinants of ovarian cancer plasticity. Am J Pathol. 2001;158:1279–1288. doi: 10.1016/S0002-9440(10)64079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sood AK, Fletcher MS, Coffin JE, et al. Functional role of matrix metalloproteinases in ovarian tumor cell plasticity. Am J Obstet Gynecol. 2004;190:899–909. doi: 10.1016/j.ajog.2004.02.011. [DOI] [PubMed] [Google Scholar]
- 33.Chambers SK. In vitro invasion assays. In: Bartlett JS, editor. Ovarian cancer methods and protocols. Totowa (New Jersey): Humana Press; 2000. pp. 179–185. [Google Scholar]
- 34.Seftor RE, Seftor EA, De Larco JE, et al. Chemically modified tetracyclines inhibit human melanoma cell invasion and metastasis. Clin Exp Metastasis. 1998;16:217–225. doi: 10.1023/a:1006588708131. [DOI] [PubMed] [Google Scholar]
- 35.Ryan ME, Ramamurthy S, Golub LM. Matrix metalloproteinases and their inhibition in periodontal treatment. Curr Opin Periodontol. 1996;3:85–96. [PubMed] [Google Scholar]
- 36.Sapolsky RM, Romero LM, Munck AU. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev. 2000;21:55–89. doi: 10.1210/edrv.21.1.0389. [DOI] [PubMed] [Google Scholar]
- 37.Chrousos GP, Gold PW. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA. 1992;267:1244–1252. [PubMed] [Google Scholar]
- 38.Nakane T, Szentendrei T, Stern L, et al. Effects of IL-1 and cortisol on β-adrenergic receptors, cell proliferation, and differentiation in cultured human A549 lung tumor cells. J Immunol. 1990;145:260–266. [PubMed] [Google Scholar]
- 39.Curran S, Murray GI. Matrix metalloproteinases in tumour invasion and metastasis. J Pathol. 1999;189:300–308. doi: 10.1002/(SICI)1096-9896(199911)189:3<300::AID-PATH456>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- 40.Kleiner DE, Stetler-Stevenson WG. Matrix metalloproteinases and metastasis. Cancer Chemother Pharmacol. 1999;43 Suppl:S42–S51. doi: 10.1007/s002800051097. [DOI] [PubMed] [Google Scholar]
- 41.Ellerbroek SM, Fishman DA, Kearns AS, et al. Ovarian carcinoma regulation of matrix metalloproteinase-2 and membrane type 1 matrix metalloproteinase through β1 integrin. Cancer Res. 1999;59:1635–1641. [PubMed] [Google Scholar]
- 42.Ellerbroek SM, Wu YI, Overall CM, et al. Functional interplay between type I collagen and cell surface matrix metalloproteinase activity. J Biol Chem. 2001;276:24833–24842. doi: 10.1074/jbc.M005631200. [DOI] [PubMed] [Google Scholar]
- 43.Fishman DA, Bafetti LM, Stack MS. Membrane-type matrix metalloproteinase expression and matrix metalloproteinase-2 activation in primary human ovarian epithelial carcinoma cells. Invasion Metastasis. 1996;16:150–159. [PubMed] [Google Scholar]
- 44.Davidson B, Goldberg I, Gotlieb WH, et al. High levels of MMP-2, MMP-9, MT1-MMP and TIMP-2 mRNA correlate with poor survival in ovarian carcinoma. Clin Exp Metastasis. 1999;17:799–808. doi: 10.1023/a:1006723011835. [DOI] [PubMed] [Google Scholar]
- 45.Stack MS, Ellerbroek SM, Fishman DA. The role of proteolytic enzymes in the pathology of epithelial ovarian carcinoma. Int J Oncol. 1998;12:569–576. doi: 10.3892/ijo.12.3.569. [DOI] [PubMed] [Google Scholar]
- 46.Bierhaus A, Wolf J, Andrassy M, et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc Natl Acad Sci U S A. 2003;100:1920–1925. doi: 10.1073/pnas.0438019100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Greenwald G, Roy S. Follicular development and its control. In: Knobil E, Neill J, editors. The physiology of reproduction. New York: Raven Press; 1994. pp. 629–724. [Google Scholar]
- 48.Nankova B, Kvetnansky R, Hiremagalur B, et al. Immobilization stress elevates gene expression for catecholamine biosynthetic enzymes and some neuropeptides in rat sympathetic ganglia: effects of adrenocorticotropin and glucocorticoids. Endocrinology. 1996;137:5597–5604. doi: 10.1210/endo.137.12.8940389. [DOI] [PubMed] [Google Scholar]
- 49.Paredes A, Galvez A, Leyton V, et al. Stress promotes development of ovarian cysts in rats: the possible role of sympathetic nerve activation. Endocrine. 1998;8:309–315. doi: 10.1385/ENDO:8:3:309. [DOI] [PubMed] [Google Scholar]
- 50.Lara HE, Dorfman M, Venegas M, et al. Changes in sympathetic nerve activity of the mammalian ovary during a normal estrous cycle and in polycystic ovary syndrome: studies on norepinephrine release. Microsc Res Tech. 2002;59:495–502. doi: 10.1002/jemt.10229. [DOI] [PubMed] [Google Scholar]
- 51.Mayerhofer A, Smith GD, Danilchik M, et al. Oocytes are a source of catecholamines in the primate ovary: evidence for a cell-cell regulatory loop. Proc Natl Acad Sci U S A. 1998;95:10990–10995. doi: 10.1073/pnas.95.18.10990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fredriksson JM, Lindquist JM, Bronnikov GE, et al. Norepinephrine induces vascular endothelial growth factor gene expression in brown adipocytes through a β-adrenoreceptor/cAMP/protein kinase A pathway involving Src but independently of Erk1/2. J Biol Chem. 2000;275:13802–13811. doi: 10.1074/jbc.275.18.13802. [DOI] [PubMed] [Google Scholar]
- 53.Daunt DA, Hurt C, Hein L, et al. Subtype-specific intracellular trafficking of α2-adrenergic receptors. Mol Pharmacol. 1997;51:711–720. doi: 10.1124/mol.51.5.711. [DOI] [PubMed] [Google Scholar]
- 54.Murphy CJ, Campbell S, Araki-Sasaki K, et al. Effect of norepinephrine on proliferation, migration, and adhesion of SV-40 transformed human corneal epithelial cells. Cornea. 1998;17:529–536. doi: 10.1097/00003226-199809000-00011. [DOI] [PubMed] [Google Scholar]
- 55.Yamanaka Y, Mammoto T, Kirita T, et al. Epinephrine inhibits invasion of oral squamous carcinoma cells by modulating intracellular cAMP. Cancer Lett. 2002;176:143–148. doi: 10.1016/s0304-3835(01)00764-9. [DOI] [PubMed] [Google Scholar]
- 56.Maloney JP, Silliman CC, Ambruso DR, et al. In vitro release of vascular endothelial growth factor during platelet aggregation. Am J Physiol. 1998;275:H1054–H1064. doi: 10.1152/ajpheart.1998.275.3.H1054. [DOI] [PubMed] [Google Scholar]
- 57.Huber D, Cramer EM, Kaufmann JE, et al. Tissue-type plasminogen activator (t-PA) is stored in Weibel-Palade bodies in human endothelial cells both in vitro and in vivo. Blood. 2002;99:3637–3645. doi: 10.1182/blood.v99.10.3637. [DOI] [PubMed] [Google Scholar]
- 58.Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411:375–379. doi: 10.1038/35077241. [DOI] [PubMed] [Google Scholar]
- 59.Woessner JF., Jr Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J. 1991;5:2145–2154. [PubMed] [Google Scholar]
- 60.Rundhaug JE. Matrix metalloproteinases, angiogenesis, and cancer: commentary re: A.C. Lockhart et al., Reduction of wound angiogenesis in patients treated with BMS-275291, a broad spectrum matrix metalloproteinase inhibitor. Clin Cancer Res. 2003;9:551–554. [PubMed] [Google Scholar]
- 61.Coussens LM, Tinkle CL, Hanahan D, et al. MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell. 2000;103:481–490. doi: 10.1016/s0092-8674(00)00139-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bergers G, Brekken R, McMahon G, et al. Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat Cell Biol. 2000;2:737–744. doi: 10.1038/35036374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Huang S, Van Arsdall M, Tedjarati S, et al. Contributions of stromal metalloproteinase-9 to angiogenesis and growth of human ovarian carcinoma in mice. [comment] J Natl Cancer Inst. 2002;94:1134–1142. doi: 10.1093/jnci/94.15.1134. [DOI] [PubMed] [Google Scholar]
- 64.Enserink JM, Price LS, Methi T, et al. The cAMP-Epac-Rap1 pathway regulates cell spreading and cell adhesion to laminin-5 through the α3β1 integrin but not the α6β4 integrin. J Biol Chem. 2004;279:44889–44896. doi: 10.1074/jbc.M404599200. [DOI] [PubMed] [Google Scholar]