Abstract
OBJECTIVE
The role of reactive oxygen species (ROS) and their dissipation in type 1 diabetes pathogenesis have garnered considerable controversy. Our recent work has demonstrated the importance of NADPH oxidase (NOX) activity for type 1 diabetes development and modulating T-cell autoreactivity. We previously linked decreased monocyte ROS with diabetes resistance in the alloxan-resistant mouse, and NOD-Ncf1m1J mice with a genetic ablation of NOX activity had reduced and delayed type 1 diabetes compared with NOD mice.
RESEARCH DESIGN AND METHODS
To determine the required cellular sources of ROS that are necessary for type 1 diabetes initiation, we used antibody depletion and adoptive transfer experiments into NOD and NOD-Scid females, respectively. After receiving treatment, female mice were monitored for hyperglycemia and overt diabetes.
RESULTS
Depletion of macrophages and neutrophils fully protected NOD mice from type 1 diabetes. However, elimination of neutrophils alone showed no significant reduction or delay. Type 1 diabetes induction in NOD-Scid mice by adoptive transfer with NOD-Ncf1m1J splenocytes was significantly delayed compared with NOD splenocytes, suggesting macrophage ROS and modulation of effector responses are critical for diabetes. The adaptive immune response was also altered by the absence of NOX activity, as purified T cells from NOD-Ncf1m1J mice exhibited delayed transfer kinetics. Cotransfer experiments demonstrated the defect was intrinsic to NOX-deficient CD8+ T cells. After stimulation, cytotoxic T cells exhibited decreased effector function in the absence of superoxide production.
CONCLUSIONS
These data demonstrate that the impaired autoreactive response of NOX-deficient NOD-Ncf1m1J immune system results from an alteration in the antigen-presenting cell–T-cell axis rather than failure of neutrophils to act as effector cells and that ROS signaling is important for the initiation of β-cell–directed autoimmunity by T cells.
Destruction of pancreatic β-cells is mediated by aberrant immune responses against islet antigens resulting in the development of type 1 diabetes. During the early stages of disease, an insulitic infiltrate consisting of macrophages, dendritic cells, T-cell subsets, and B cells accumulates in the pancreatic islets (1,2). Effector mechanisms, including direct T-cell cytotoxicity and indirect methods mediated by leukocytes, contribute to β-cell destruction and overt diabetes. Production of reactive oxygen species (ROS) has been proposed to be an important contributor to β-cell loss during type 1 diabetes pathogenesis. Endogenous free radicals produced by the β-cell in response to cytokines are one source of cytotoxic ROS: γ-interferon (IFN-γ) in combination with interleukin (IL)-1β and tumor necrosis factor-α (TNF-α) are cytotoxic to β-cells as a result of increased production of nitric oxide (NO) and superoxide. However, the roles of cellular sources of ROS during the development of spontaneous type 1 diabetes have not been fully defined (3,4).
NADPH oxidase (NOX), a multicomponent enzymatic complex, is a major source of free radicals and important for the effector function of neutrophils and macrophages (5–7). Islet-infiltrating macrophages release high levels of ROS, including superoxide via NOX. Defects in NOX function have been associated with increased susceptibility to autoimmunity in experimental allergic encephalomyelitis and collagen-induced arthritis (8–12). In addition to contributing to toxicity, free radicals are also potent signaling molecules and are important in adaptive immune responses (13–16). Recent work has demonstrated that exogenous as well as endogenous sources of ROS are involved in initiating and dictating cytokine responses of CD4+ T cells (17). Likewise, antioxidant regulation significantly modifies proliferative (15,18) and effector responses of CD8+ cytotoxic T cells, reducing cytolytic function and cytokine production (15). To study the role of superoxide production via NOX in a type 1 diabetes–prone mouse model, a mutation in p47phox subunit was congenically introduced into the NOD mouse (NOD-Ncf1m1J) (19).
Genetic ablation of NOX protects against type 1 diabetes development, as NOD-Ncf1m1J mice have reduced incidence and delayed type 1 diabetes onset. To determine the innate immune cellular sources of ROS essential for type 1 diabetes pathogenesis, we depleted neutrophils and macrophages from NOD mice. Here we show that macrophages are essential for type 1 diabetes induction, whereas neutrophils are dispensable. In addition, NOD-Ncf1m1J mice were protected from type 1 diabetes after adoptive transfer of BDC-2.5 T cells, demonstrating a requirement of NOX during CD4+ T-cell–mediated autoreactivity. NOX-deficient β-cells were not protected as NOD-Ncf1m1J islets were susceptible to cytokine-mediated damage and mice developed diabetes induced by AI4 CD8+ T cells. We have previously shown that NOX function is important for T-helper cell lineage development and cytokine synthesis (15,16). NOX deficiency resulted in decreased production of Th1-associated cytokines, including IFN-γ, TNF-α, IL-1β, and IL-12 p70. However, there was a marked increase in Th17 cytokines, including IL-17 and IL-10 (19). Here we report the reduced diabetogenic capabilities of NOX-deficient leukocytes as purified splenocytes and T cells from NOD-Ncf1m1J mice had delayed transfer kinetics of type 1 diabetes in contrast to NOD mice. It is noteworthy that this delay was associated with the source of CD8+ T cells, suggesting an important role for NOX in propagation of islet-directed cytotoxic T-cell activity. Examination of cytotoxic T-cell function associated decreased production of IFN-γ and granzyme B with NOX deficiency, demonstrating reduced effector responses when superoxide is lacking. Therefore, ROS production by macrophages and T cells is essential for the development and effector function of proinflammatory responses mediating β-cell destruction.
RESEARCH DESIGN AND METHODS
Animals.
NOD/ShiLtJ, NOD.B6-Ncf1m1J/m1J (NOD-Ncf1m1J, generated as previously described) (19) mice were bred and housed in the University of Florida or the Children’s Hospital of Pittsburgh facility under specific pathogen-free conditions. NOD.CB17-Prkdcscid/J (NOD-Scid), C57BL/6-Ncf1m1J (B6-Ncf1), and C57BL/6.NOD-(D17Mit21-D17Mit10)/LtJ (B6-H2g7) mice were purchased from the Jackson Laboratory (Bar Harbor, ME). NOD.129S7 (B6)-Rag1tm1Mom/J (NOD.Rag), NOD.Cg-Rag1tm1MomTg (TcraAI4)1Dvs/DvsJ (NOD-AI4a), and NOD.Cg-Rag1tm1MomTg (TcrbAI4)1Dvs/DvsJ (NOD-AI4b) mice were purchased from The Jackson Laboratory and bred in our mouse facility. F1 hybrid progeny from matings of NOD-AI4a with NOD-AI4b (NOD-AI4a/b) developed diabetes between 3 and 5 weeks of age. All mice were housed in specific pathogen-free facilities and approved by the institution animal care and use committee at the University of Florida or the Children’s Hospital of Pittsburgh.
Materials.
Depleting antibodies anti-Gr-1 (RB6–8C5) and anti-Ly6G (1A8) purchased from Bio X Cell (West Lebanon, NH) are both Rat IgG2 antibodies. Fluorescently labeled antibodies, Pacific blue–labeled anti-CD11b (M1/70), allophycocyanin (APC)-labeled anti-F4/80 (BM8), and phycoerythrin (PE)-labeled anti-CD8 (53–6.7) were purchased from eBioscience (San Diego, CA). PE-labeled anti-Ly6G (1A8), APC-labeled anti-CD11b (M1/70), and fluorescein isothiocyanate–labeled anti-CD11c (HL3) were purchased from BD Pharmingen (San Jose, CA). Pacific blue–labeled anti-CD4 (GK1.5) and APC/Cy7-labeled anti-B220 (RA3–6B2) were purchased from Biolegend (San Diego, CA).
Measurement of superoxide production.
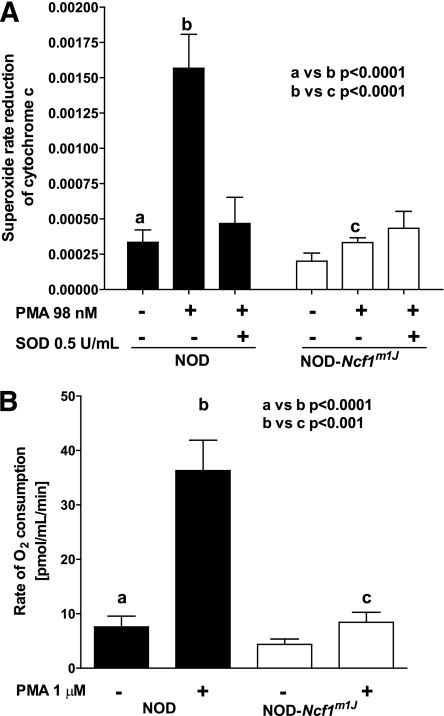
Bone marrow was collected from age-matched NOD and NOD-Ncf1m1J female mice, and erythrocytes were removed on a Histopaque-1119 gradient as previously described (20). In Hanks’ balanced salt solution (HBSS) containing 145 μmol/L cytochrome c, 1 × 105 cells were stimulated with phorbol myristic acid (PMA; 98 nmol/L) at 37°C. Reduction of cytochrome c was measured at 550 nm at 1-min intervals for 45 min. The maximal rate of reduction was calculated by linear regression analysis. Purified superoxide dismutase 1 (0.5 units/mL) was used to confirm specificity of superoxide production.
Measurement of oxygen consumption.
Bone marrow was collected from age-matched NOD and NOD-Ncf1m1J female mice, and erythrocytes were removed on a Histopaque-1119 gradient as previously described (20). After calibration, 2 × 107 cells were loaded into chamber of Oxygraph-2 K (Oroboros Instruments, Innsbruck, Austria) at 37°C. Samples were stimulated with 1 μmol/L PMA and oxygen concentration and rate of consumption monitored for 30 min.
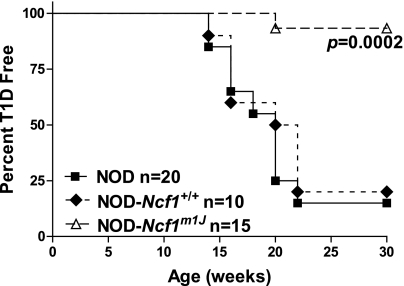
Spontaneous incidence.
At 8 weeks of age, NOD and NOD-Ncf1m1J female mice were enrolled and monitored for diabetes onset with weekly urinalysis using Diastix (Bayer, Elkhart, IN). Mice positive for glucosuria were monitored daily thereafter for hyperglycemia using One Touch Ultra 2 glucometers (LifeScan, Inc., Milpitas, CA). Any mouse with measured blood glucose levels on consecutive days >250 mg/dL was diagnosed with type 1 diabetes.
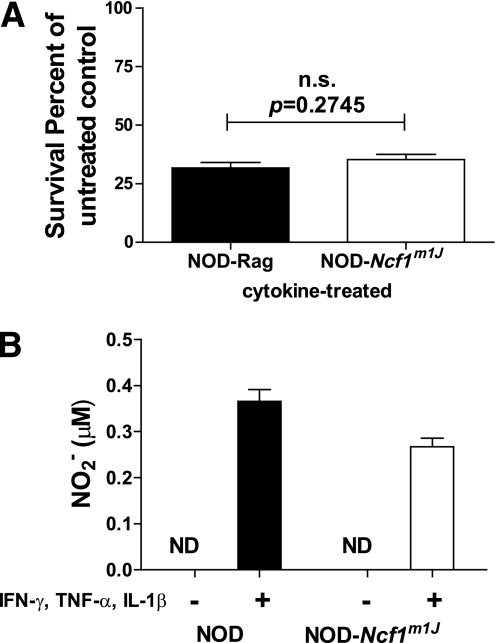
Islet isolation and culture with cytokines.
Pancreatic islets from age-matched female NOD.Rag and NOD-Ncf1m1J mice were isolated (21) and cultured with IFN-γ, IL-1β, and TNF-α as previously described (22). Islet NO production was measured and cell viability was assessed via MTT assay as previously reported (23).
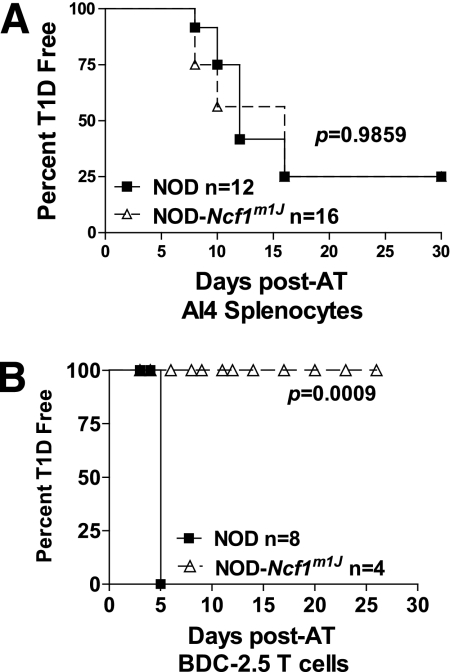
Transfer of AI4 splenocytes.
F1 progeny of NOD-AI4a crossed with NOD-AI4b (NOD-AI4a/b) were killed, and splenocytes were harvested. After the erythrocytes were lysed with Gey’s solution (24), live cells were counted and diluted to a concentration of 108 cells per milliliter. A total of 2 × 107 cells in sterile PBS were injected via the tail vein into sublethally irradiated 8-week-old NOD and NOD-Ncf1m1J female mice (dose of 7.5 Gy delivered using X-RAD 320 [Precision X-ray, North Branford, CT]) as previously described (25). Mice were monitored every other day for the onset of diabetes as described above.
Transfer of BDC-2.5 T-cell clones.
Cultures of BDC-2.5 T cells were collected and expanded in vitro as previously described for adoptive transfer experiments (26). NOD and NOD-Ncf1m1J mice were injected intraperitoneally with 107 BDC-2.5 T cells between 3 and 7 days of age. Mice were monitored daily by urinalysis, using Diastix, for the onset of diabetes.
Adoptive transfer.
Diabetic (20 weeks old) or prediabetic (8 weeks old) NOD and NOD-Ncf1m1J donors were used for adoptive transfer experiments. Splenocytes were harvested, and erythrocytes were lysed using Gey’s solution. Splenocytes, 2 × 107 cells in PBS, were transferred intraperitoneally to 8-week-old NOD-Scid recipients. In addition, CD4+ and CD8+ T cells were purified by negative selection with magnetic beads according to the manufacturer’s protocol using a CD4+ T-cell isolation kit or a CD8+ T-cell isolation kit (Miltenyi Biotec, Auburn, CA), respectively. Purity, >96%, was confirmed by flow-cytometric analysis. CD4+ and CD8+ T cells were mixed at a ratio of 3:1, and 107 cells were transferred intraperitoneally to 8-week-old NOD-Scid recipients. Transfers were divided into four groups receiving 1) NOD CD4+ + NOD CD8+, 2) NOD-Ncf1m1J CD4+ + NOD-Ncf1m1J CD8+, 3) NOD-Ncf1m1J CD4+ + NOD CD8+, or 4) NOD CD4+ + NOD-Ncf1m1J CD8+. Mice were monitored weekly for diabetes onset as described above. Engraftment of cells was confirmed by flow cytometry.
Antibody depletion studies.
Depletion studies were initiated at 4 weeks of age and were carried out until the onset of type 1 diabetes, as described above, or until 35 weeks of age. Female NOD mice were treated intraperitoneally with antibody diluted in sterile PBS three times per week. The dose for either the anti-Gr-1 (RB6–8C5) or anti-Ly6G (1A8) depleting antibodies was 500 µg on Monday and Wednesday as well as 750 µg on Friday of each week. The volume of all injections was 100 µL. Depletion of macrophages and neutrophils in peripheral blood was confirmed monthly by flow cytometry. Bone marrow, splenocytes, and peripheral blood were analyzed via flow cytometry after killing.
Purification and stimulation of CD8+ T cells.
Splenic CD8+ T cells from age-matched NOD and NOD-Ncf1m1J mice were purified by negative selection with magnetic beads according to the manufacturer’s protocol with the CD8+ T-cell isolation kit (Miltenyi Biotec). Purity, >90%, was confirmed by flow cytometric analysis. A total of 5 × 104 cells or 5 × 105 cells were used for proliferation or intracellular cytokine staining, respectively. Purified cells were stimulated with plate-bound anti-CD3ε (0.1 μg/mL) and anti-CD28 (1 μg/mL) for 72 h (27). Proliferation was measured by [3H]TdR incorporation (27). ROS production by CD8+ T cell was performed as previously described (19).
Intracellular cytokine staining of stimulated CD8+ T cells.
After 72 h of polyclonal stimulation, CD8+ T cells were further stimulated with 50 ng/mL PMA and 50 ng/mL ionomycin, followed by treatment with GolgiStop for 6 h at 37°C in a 5% CO2, humid air incubator. Cells were surface stained with allophycocyanin-labeled anti-CD8, fixed in BD Cytofix/Cytoperm buffer, washed in BD Perm/Wash buffer, and then stained with fluorescein isothiocyanate–labeled rat anti–IFN-γ (XMG1.2; BD Biosciences, San Jose, CA) and PE-labeled rat anti–granzyme B (16G6; eBioscience) or isotype controls, according to the manufacturers’ protocol. Fluorescence was measured on an LSR Fortessa (BD Biosciences), and results were analyzed with FACSDiva 6.0 software (BD Biosciences).
Flow cytometry.
Peripheral blood samples were lysed with Gey’s solution twice and resuspended in 100 µl HBSS for staining. Gey’s treated splenocytes, Ficoll-separated bone marrow suspensions, or purified T cells were counted and resuspended in HBSS at 2 × 107 cells per milliliter. Approximately 106 cells were labeled with antibodies at the proper dilution. Fluorescence was measured using LSR Fortessa and analyzed using FACSDiva 6.0 software.
Statistics.
GraphPad Prism (GraphPad Software, Inc., La Jolla, CA) was used for calculating statistical differences. Significance between mean values was determined using the Student t test, with P < 0.05 considered significant. Kaplan-Meier survival analysis was used to evaluate diabetes onset.
RESULTS
Ncf1 mutation ablates NOX superoxide production from bone marrow populations.
To study the role of ROS in the pathogenesis of type 1 diabetes, we generated NOD mice with genetic ablation of NOX superoxide production, as previously described (19), by congenic introduction of a mutant p47phox subunit (Ncf1) of NOX (28). PMA stimulation of NOD bone marrow cells exhibited elevated superoxide production (P < 0.0001); however, there was no increase in superoxide production after PMA stimulation of cells isolated from NOD-Ncf1m1J mice (Fig. 1A), confirming ablation of NOX function. Oxygen consumption by NOD-Ncf1m1J bone marrow cells after the addition of PMA was not elevated; however, bone marrow cells from NOD consumed significant amounts of oxygen (P < 0.001) (Fig. 1B).
FIG. 1.
Congenic introduction Ncf1m1J mutation ablates bone marrow cell superoxide production and oxygen consumption in response to PMA. Bone marrow from NOD (black bars) and NOD-Ncf1m1J (white bars) mice were (A) stimulated with PMA (98 nmol/L) and superoxide production was evaluated by measuring rate reduction of cytochrome C. Purified superoxide dismutase 1 (SOD1; 0.5 units/mL) was added to confirm production of superoxide. B: Oxygen consumption was measured with an oxygen electrode for 30 min. Results are from five independent experiments.
Ablation of NOX activity protects NOD congenic mice from type 1 diabetes onset.
Development of spontaneous diabetes was assessed by following cohorts of female NOD-Ncf1m1J and NOD-Ncf1+/+ littermate controls for the development of glucosuria. By 30 weeks of age, 93% of NOD-Ncf1m1J mice were diabetes-free compared with 20% of NOD-Ncf1+/+ littermate controls and 15% of NOD (Fig. 2), demonstrating significant protection (P = 0.0002).
FIG. 2.
NOD-Ncf1m1J mice are protected from spontaneous autoimmune diabetes. Age-matched female NOD (black square), NOD-Ncf1+/+ littermate controls (black diamond), and NOD-Ncf1m1J (open triangle) mice were monitored by glucosuria for the onset of spontaneous type 1 diabetes (T1D).
Islets from NOD-Ncf1m1J mice exhibit no enhanced protection against proinflammatory cytokine toxicity.
To determine whether the enhanced diabetes resistance in NOD-Ncf1m1J mice was attributed to the inherent inability of islets to generate ROS and maintain β-cell integrity, islets from NOD.Rag and NOD-Ncf1m1J mice were isolated and treated with cytokines to evaluate cell viability and NO production. Treatment with proinflammatory cytokines significantly reduced cell viability of NOD.Rag- and NOD-Ncf1m1J–cultured islets (Fig. 3A). NOX-deficient islets produced NO after treatment with cytokines (Fig. 3B), demonstrating no change in the synthesis of reactive nitrogen species.
FIG. 3.
Cultured islets from NOD.Rag (black bars) and NOD-Ncf1m1J (white bars) mice were treated with cytokines IFN-γ, IL-1β, and TNF-α. Islets were (A) susceptible to proinflammatory cytokine-mediated damage and (B) produced NO after treatment. Data representative of three independent experiments performed in triplicate. ND, none detected.
NOD-Ncf1m1J mice are fully susceptible to AI4 T-cell transfer.
Intracellular production of ROS within islet cells contributes to β-cell killing (29–31), but whether the synthesis of ROS also functions as accessory molecules to facilitate T-cell autoreactivity within the islet has not been well studied. β-Cells express a NOX complex that can produce superoxide upon stimulation (29,31). To determine whether genetic ablation of NOX at the β-cell level would prevent diabetes induction with diabetogenic CD8+ T cells, we transferred highly pathogenic splenocytes from NOX-intact NOD-AI4 transgenic donors to sublethally irradiated NOD and NOD-Ncf1m1J female mice. There was no significant difference in the rate or overall incidence of diabetes (Fig. 4A) as 75% of NOD and NOD-Ncf1m1J mice developed hyperglycemia by 15 days after transfer.
FIG. 4.
AI4 transgenic T cells, but not BDC-2.5 cells, are able to induce diabetes in NOX-deficient mice. A: Splenocytes (2 × 107 cells) from NOD-AI4 donors were transferred to sublethally (7.5 Gy) irradiated NOD (black square) and NOD-Ncf1m1J (open triangle) recipients. B: BDC-2.5 CD4+ T cells were harvested, activated, and transferred to age-matched NOD and NOD-Ncf1m1J recipients. Mice were monitored by glucosuria for the onset of type 1 diabetes (T1D). Mice were considered diabetic after two consecutive readings of blood glucose above 250 mg/dL. AT, adoptive transfer.
NOD-Ncf1m1J mice are resistant to BDC-2.5 T-cell transfer.
In addition to their function in perpetuating cytotoxic responses, macrophages also serve as vital APCs. Depletion or inactivation of phagocytes and macrophages has prevented diabetes progression in mouse and rat models (32–35), and recent reports suggest that endogenous macrophages are essential for T-cell autoreactivity in the BDC-2.5 transfer models (36–38). To determine whether the absence of phagocyte ROS production is essential for mediating type 1 diabetes resistance in the NOD-Ncf1m1J mouse, we transferred BDC-2.5 T cells to NOX-deficient NOD-Ncf1m1J mice. CD4+ T cells from BDC-2.5 transgenic mice were transferred into age-matched NOD and NOD-Ncf1m1J female recipients. All NOD recipients developed diabetes 5 days after transfer; however, after 30 days, NOD-Ncf1m1J mice had still not developed diabetes (Fig. 4B).
Macrophages are essential whereas neutrophils are superfluous for type 1 diabetes pathogenesis.
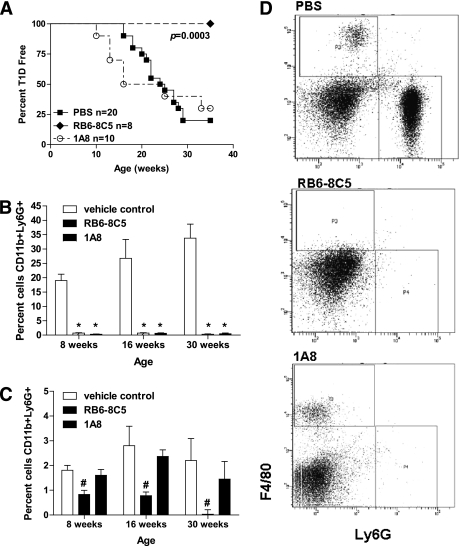
NOX respiratory burst is well described in neutrophils and macrophages and is an important mechanism by which these innate immune cells mediate killing (5,7). Macrophages have previously been reported as critical to the development of spontaneous type 1 diabetes; however, direct assessment of neutrophils has not been investigated. To elucidate the involvement of neutrophils in type 1 diabetes pathogenesis, the use of Gr-1–specific monoclonal antibodies RB6–8C5 and 1A8 were used for depletion studies. RB6–8C5 clone binds to Ly6G and Ly6C, whereas 1A8 is Ly6G specific (39). NOD female mice were treated with either RB6–8C5 antibodies to deplete macrophages and neutrophils or 1A8 antibodies to deplete only neutrophils. Depletion with RB6–8C5 provided 100% protection from diabetes development, whereas treatment with 1A8 provided no protection from type 1 diabetes after 35 weeks as compared with control-treated animals (Fig. 5A). Weekly systemic administration of the antibodies began at 4 weeks of age, prior to advancing insulitis, and continued throughout the course of the study to maintain reduction of the target cell types. Peripheral blood samples were collected monthly and monitored via flow cytometry to determine depletion of macrophages (CD11b+F4/80+) and neutrophils (CD11b+Ly6G+). During the course of the study, RB6–8C5 substantially depleted both macrophage and neutrophil populations (Fig. 5B–D), whereas 1A8 only depleted neutrophils with no significant affect on macrophage numbers (Fig. 5B–D). CD11b+ cells were gated and analyzed for F4/80 and Ly6G expression. Representative samples from 16-week-old mice are shown in Fig. 5D. There were no changes in dendritic cell or lymphocyte populations from either treatment (data not shown).
FIG. 5.
Macrophages, but not neutrophils, are critical for type 1 diabetes (T1D) induction. A: Age-matched NOD female mice were treated with 500 μg 2 days a week and 750 μg 1 day a week anti-Gr-1 RB6–8C5 (black diamond), 1 mg 3 days a week anti-Gr-1 1A8 (open triangle), or PBS vehicle control (black square) starting at 4 weeks of age. Mice were monitored by glucosuria for the onset of diabetes and considered diabetic after two consecutive readings of blood glucose above 250 mg/dL. Peripheral blood was used to confirm depletion of (B) neutrophils (CD11b+Ly6G+) and (C) macrophages (CD11b+F4/80+) by flow cytometry. D: Blood samples were gated on CD11b+ cells and analyzed for Ly6G and F4/80 expression (representative plot of treated mice at 16 weeks of age). Statistical differences noted above signify *P < 0.0001 or #P < 0.002, respectively.
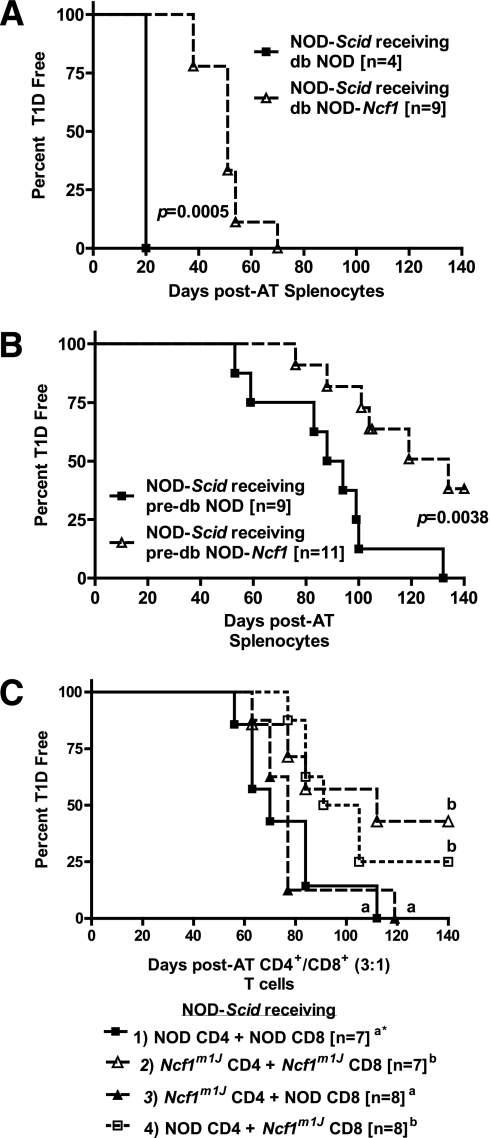
Delayed transfer of diabetes with NOD-Ncf1m1J splenocytes and purified CD4+ and CD8+ T cells.
To assess internal defects in the diabetogenicity of APC and T cells lacking intracellular superoxide production, whole splenocytes or purified T cells collected from diabetic and prediabetic female NOD and NOD-Ncf1m1J donors were adoptively transferred into NOD-Scid recipients. Splenocytes from diabetic (Fig. 6A) and prediabetic (Fig. 6B) NOD-Ncf1m1J mice exhibited delayed diabetes onset compared with NOD cells. As the phagocyte NOX is expressed in T cells and activated after T-cell receptor stimulation (17,40), we also sought to determine whether NOX-deficient T cells would lack the ability to transfer disease. To study potential defects specifically in NOX-deficient T cells, CD4+ and CD8+ T cells from donor animals were purified and adoptively transferred into NOX-intact NOD-Scid recipients. There were a total of four groups (Fig. 6C): group 1) NOD CD4+ + NOD CD8+ cells and group 2) NOD-Ncf1m1J CD4+ + NOD-Ncf1m1J CD8+ cells, as well as the cotransfer groups receiving either group 3) NOD-Ncf1m1J CD4+ and NOD CD8+ cells or group 4) NOD CD4+ and NOD-Ncf1m1J CD8+ cells. These experiments allowed us to determine whether the defect was present in all NOX-deficient T cells or specific to NOX-deficient CD4+ or CD8+ T cells. A significantly delayed transfer of disease (P < 0.05) was observed in hosts receiving group 2 NOD-Ncf1m1J CD4+ and NOD-Ncf1m1J CD8+ in addition to group 4 NOD CD4+ and NOD-Ncf1m1J CD8+ cells compared with the other two recipient groups. The common cell type in the groups with delayed transfer was the NOD-Ncf1m1J CD8+ cells, indicating that in this model, NOX-deficient CD8+ T cells were less efficient in inducing type 1 diabetes.
FIG. 6.
NOX-deficient NOD-Ncf1m1J immune system has a reduced capacity to transfer type 1 diabetes (T1D) to immune-deficient hosts. Splenocytes (2 × 107 cells) from (A) 20-week-old diabetic or (B) 8-week-old prediabetic donor NOD (black square) and NOD-Ncf1m1J (open triangle) female mice were transferred intraperitoneally to NOD-Scid hosts. C: A total of 1 × 107 purified CD4+ and CD8+ T cells at a ratio of 3:1 were transferred intraperitoneally to NOD-Scid recipients. Transfers were divided into four groups: group 1) NOD CD4+ + NOD CD8+, group 2) NOD-Ncf1m1J CD4+ + NOD-Ncf1m1J CD8+, group 3) NOD-Ncf1m1J CD4+ + NOD CD8+, or group 4) NOD CD4+ + NOD-Ncf1m1J CD8+. *Letters denote significance. Groups/lines with different letters were statistically significant (P < 0.05), whereas those with the same letter were not statistically different. Comparisons of the groups led to the following P values: group 1 vs. group 2, P = 0.047; group 1 vs. group 3, P = 0.67; group 1 vs. group 4, P = 0.049; group 2 vs. group 3, P = 0.043; group 2 vs. group 4, P = 0.64; and group 3 vs. group 4, P = 0.021. In all three adoptive transfer (AT) experiments, mice were monitored by glucosuria for the onset of diabetes and confirmed by blood glucose measurement. Mice were considered diabetic after two consecutive readings of blood glucose above 250 mg/dL.
Decreased production of proinflammatory markers by NOD-Ncf1m1J CD8+ T cells.
As adoptive transfer kinetics were dependent on the NOX status of cytotoxic T cells during transfer, we further examined the effector function of purified CD8+ T cells. Anti-CD3 stimulation of CD8+ T cells confirmed the inability of NOD-Ncf1m1J cytotoxic T cells to produce ROS (Supplementary Fig. 1). Proliferation and intracellular cytokine synthesis was then evaluated using polyclonal and PMA/ionomycin-stimulated CD8+ T cells from NOD and NOD-Ncf1m1J mice. NOX-deficient cytotoxic T cells had significantly decreased production of IFN-γ and granzyme B compared with NOD cytotoxic T cells (Table 1), indicating a defect in effector function.
TABLE 1.
NOD-Ncf1m1J CD8+ T cells exhibit a decrease in synthesis of the proinflammatory cytokine IFN-γ and the cytotoxic effector molecule granzyme B. Proliferation by [3H]TdR incorporation and intracellular cytokine staining for CD8+/IFN-γ+ and CD8+/GzmB+ after 72-h stimulation of purified naïve CD8+ T cells from the spleen of NOD and NOD-Ncf1m1J mice
| NOD | NOD-Ncf1m1J | |
|---|---|---|
| CPM | 20,080 ± 2,668a | 35,260 ± 4,472b |
| CD8+/IFN-γ+ (%) | 31.05 ± 1.29c | 22.65 ± 1.91d |
| CD8+/GzmB+b (%) | 77.34 ± 7.05e | 60.68 ± 7.52f |
Data are means ± SD. CPM, counts per minute.
P = 0.014 for a vs. b; P = 0.0003 for c vs. d; P = 0.011 for e vs. f.
DISCUSSION
Free radical generation, either exogenous or endogenous to the β-cell, is thought to contribute to β-cell demise and diabetes (3,22,23,26,41). Oxidative stress and the generation of free radicals are directly toxic to β-cells, but they also have a dual role as potent signaling molecules to induce proinflammatory cytokine synthesis that can also directly affect β-cell destruction (4,13,14). Genetic analyses investigating diabetes resistance in the alloxan-resistant mouse linked increased free radical dissipation and decreased ROS production with type 1 diabetes protection (3,20,41,42). NOD-Ncf1m1J mice were generated to evaluate the role of superoxide production in the pathogenesis of type 1 diabetes in the diabetes-prone NOD mouse model. Genetic ablation of NOX-mediated superoxide production (Fig. 1) was significantly protective when congenically introgressed into the NOD genetic background (Fig. 2); therefore, ROS production is critical in the induction of type 1 diabetes. Recent work suggests NOX complex subunits are expressed in β-cells and produce superoxide upon stimulation (29–31). In the absence of β-cell–specific ROS synthesis, NOD-Ncf1m1J cultured islets were still susceptible to cytokine-mediated cell death (Fig. 3A) and capable of producing NO in response to cytokines (Fig. 3B), demonstrating no defect in the production of reactive nitrogen species. NOD-Ncf1m1J mice were equally susceptible as NOD mice to diabetes induction upon adoptive transfer of AI4 splenocytes (Fig. 4A), a highly diabetogenic CD8+ T cell. Preactivated AI4 CD8+ T cells are efficient effectors capable of inducing type 1 diabetes without leukocyte help (43,44). These results demonstrate that NOX-deficient β-cells are susceptible to immune-mediated destruction.
Macrophage and neutrophil production of superoxide via NOX is an important mechanism used during immune responses and is a significant source of ROS during inflammation. Macrophages have been shown to be important in the pathogenesis of type 1 diabetes (32–37); however, neutrophils have not been as extensively studied. The contributions of ROS-producing innate immune cells to type 1 diabetes pathogenesis were investigated with depleting antibodies specific for macrophages and neutrophils (RB6–8C5) or neutrophils alone (1A8). When both macrophage and neutrophil populations were depleted, 100% of the animals were free of type 1 diabetes out to 35 weeks; however, neutrophil depletion alone did not alter the course of type 1 diabetes development (Fig. 5). Although neutrophils may be involved in β-cell killing and autoreactivity, effector contributions are not essential and may be redundant in the process of β-cell damage. Both neutrophils and macrophages exhibit important phagocytic and direct cytotoxic effector function, but macrophages are also involved in antigen presentation and stimulation of T cells. The protection afforded by NOX inhibition may partially be explained at the macrophage level as observed with the adoptive transfer studies with BDC-2.5 CD4+ T cells into NOD-Ncf1m1J recipients (Fig. 4B). Reports by Calderon and colleagues show that macrophages are essential for effector function and diabetes induction in BDC-2.5 transfer models (36,38). Our data suggest that NOX-deficient macrophages are unable to mediate pancreatic β-cell destruction after adoptive transfer with BDC-2.5 T cells. Taken together, not only are macrophages required but NOX-dependent ROS production by macrophages also is likely essential for CD4+ T-cell effector function and autoreactivity.
Diabetogenic defects resulting from NOX ablation were assessed by adoptive transfer of whole splenocytes or purified T cells collected from diabetic and prediabetic female NOD and NOD-Ncf1m1J donors. Adoptive transfer experiments demonstrated delayed transfer kinetics to NOX-intact hosts (Fig. 6), suggesting a defect intrinsic to the NOX-deficient T cell. This delay occurred with the transfer of whole splenocytes and with purified CD4+ and CD8+ T cells, where antigen presentation would be in the context of a NOX-intact host APC. Transfer with splenocytes harvested from diabetic donors, where the islet-reactive population would be expanded and activated, was still delayed compared with control NOD donor cells. This indicates a fundamental defect in the ability of NOX-deficient leukocytes to respond and execute sufficient effector function. It also demonstrates that ROS production by T-cell subsets is important for initiating islet-directed autoimmunity. Redox status and redox molecules contribute to transcriptional and posttranscriptional regulation of transcription factors and cytokine synthesis (45–47). We have previously shown that redox balance and superoxide production are critical in the initiation of a pathogenic cytokine response (16), with NOD-Ncf1m1J macrophages and CD4+ T cells skewed toward a Th17 profile (19). When adoptive transfer experiments were performed into NOD-Scid mice with a NOX-intact environment, NOD-Ncf1m1J splenocytes gained effector function and induced type 1 diabetes (Fig. 6A–C). This observation highlights the ability of NOX-intact APCs to initiate Th1 effector function, as we previously demonstrated (19). This was highlighted by the cotransfer system where NOX-deficient CD4+ T cells gained full effector function in the presence of NOX-intact APC and CD8+ T cells. In contrast, the NOX-deficient CD8+ T cells exhibited defective reactivity even when cotransferred with NOX-intact CD4+, suggesting the defect was intrinsic in cytotoxic T cells (Fig. 6C). Earlier reports highlight an essential role for NOX and ROS generation in CD8+ T-cell expansion, survival, and activation (18,40,48,49), with altered cytokine production from gp91phox- or p47phox-deficient CD8+ T cells (40). Here, we show that CD8+ T cells exhibit reduced effector function when NOX activity is ablated, in that diabetes transfer is delayed when cytotoxic T cells are from NOD-Ncf1m1J donors (Fig. 6C). This delay was associated with a significant decrease in the production of IFN-γ and granzyme B (Table 1) after stimulation of purified CD8+ T cells, suggesting NOX deficiency was reducing cytotoxic capability of cytotoxic T cells. Proinflammatory mechanisms are not globally dysfunctional in NOX-deficient animals. Ncf1 mutations have previously been associated with increased severity of experimental allergic encephalomyelitis and collagen-induced arthritis (9–12). In fact, NOD-Ncf1m1J mice demonstrated severe and relapsing paralysis after challenge with myelin oligodendrocyte glycoprotein peptide sequence 35–55 (MOG35–55) (19). Therefore, we propose that type 1 diabetes is reduced and delayed by the Ncf1m1J mutation as a result of a combination of altered Th1 T-cell activation by APCs (19) and diminished cytotoxic T-cell functions that are critical for β-cell killing (Fig. 6C and Table 1).
Our data describe a role for ROS produced by macrophages and T cells in the initiation of autoimmune diabetes. Macrophage ROS-mediated effector function is essential for CD4+ T-cell autoreactivity and type 1 diabetes pathogenesis. In addition, intracellular ROS produced by T cells is important for executing efficient islet reactivity. The loss of ROS synthesis provided by the Ncf1 mutation significantly alters effector function of macrophages and T-cell subsets. This demonstrates the importance of ROS-dependent mechanisms in controlling activation of autoimmune effectors and in turn mediating aberrant reactivity toward β-cells.
Supplementary Material
ACKNOWLEDGMENTS
M.D. has received a T32 training grant in burns and trauma (T32 GM-008721-13) awarded by the National Institutes of Health. L.E.P. was supported by a training grant (T32 AI-007051) awarded by the National Institutes of Health. H.M.T. has received a grant from the American Diabetes Association. J.D.P. has received a grant from the American Diabetes Association. L.L.M. has received grants from the National Institutes of Health (GM-40586-22 and GM-81923-03). C.E.M. has received grants from the Juvenile Diabetes Foundation and from the National Institutes of Health (AI-056374 and DK-074656). This study was also supported by the Sebastian Family Endowment for Diabetes Research.
No potential conflicts of interest relevant to this article were reported.
T.C.T. and M.D. designed the research plan, performed experiments, analyzed data, and wrote the manuscript. C.L. assisted in design and performed the coadoptive transfer experiments as well as the studies into cytotoxic T-cell effector function. J.C. assisted in AI4 transfers. L.E.P. assayed cytotoxic T-cell ROS production. H.M.T. assayed cytotoxic T-cell ROS production, designed and performed the BDC-2.5 adoptive transfers, and edited the manuscript. M.A. performed experiments of islet exposure to proinflammatory cytokines. J.D.P. designed and performed the BDC-2.5 adoptive transfers and edited the manuscript. L.L.M. participated in the experimental design and edited the manuscript. C.E.M. designed the research plan, performed experiments, analyzed data, and wrote the manuscript.
Parts of this study were presented in abstract form at the 70th Scientific Sessions of the American Diabetes Association, Orlando, Florida, 25–29 June 2010, and were published in abstract form in Diabetes 2010;59(Suppl. 1):A361.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db10-1222/-/DC1.
REFERENCES
- 1.Bouma G, Coppens JM, Mourits S, et al. Evidence for an enhanced adhesion of DC to fibronectin and a role of CCL19 and CCL21 in the accumulation of DC around the pre-diabetic islets in NOD mice. Eur J Immunol 2005;35:2386–2396 [DOI] [PubMed] [Google Scholar]
- 2.Delovitch TL, Singh B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune dysregulation gets the NOD. Immunity 1997;7:727–738 [DOI] [PubMed] [Google Scholar]
- 3.Chen J, Gusdon AM, Thayer TC, Mathews CE. Role of increased ROS dissipation in prevention of T1D. Ann N Y Acad Sci 2008;1150:157–166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Griffiths HR. ROS as signalling molecules in T cells—evidence for abnormal redox signalling in the autoimmune disease, rheumatoid arthritis. Redox Rep 2005;10:273–280 [DOI] [PubMed] [Google Scholar]
- 5.Nauseef WM. Biological roles for the NOX family NADPH oxidases. J Biol Chem 2008;283:16961–16965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rossi F, Zatti M. Biochemical aspects of phagocytosis in polymorphonuclear leucocytes. NADH and NADPH oxidation by the granules of resting and phagocytizing cells. Experientia 1964;20:21–23 [DOI] [PubMed] [Google Scholar]
- 7.Vignais PV. The superoxide-generating NADPH oxidase: structural aspects and activation mechanism. Cell Mol Life Sci 2002;59:1428–1459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bäckdahl L, Guo JP, Jagodic M, et al. Definition of arthritis candidate risk genes by combining rat linkage-mapping results with human case-control association data. Ann Rheum Dis 2009;68:1925–1932 [DOI] [PubMed] [Google Scholar]
- 9.Hultqvist M, Olofsson P, Holmberg J, Bäckström BT, Tordsson J, Holmdahl R. Enhanced autoimmunity, arthritis, and encephalomyelitis in mice with a reduced oxidative burst due to a mutation in the Ncf1 gene. Proc Natl Acad Sci USA 2004;101:12646–12651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hultqvist M, Holmdahl R. Ncf1 (p47phox) polymorphism determines oxidative burst and the severity of arthritis in rats and mice. Cell Immunol 2005;233:97–101 [DOI] [PubMed] [Google Scholar]
- 11.Olofsson P, Holmdahl R. Positional cloning of Ncf1—a piece in the puzzle of arthritis genetics. Scand J Immunol 2003;58:155–164 [DOI] [PubMed] [Google Scholar]
- 12.Olofsson P, Holmberg J, Tordsson J, Lu S, Akerström B, Holmdahl R. Positional identification of Ncf1 as a gene that regulates arthritis severity in rats. Nat Genet 2003;33:25–32 [DOI] [PubMed] [Google Scholar]
- 13.Babior BM, Kipnes RS, Curnutte JT. Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J Clin Invest 1973;52:741–744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lander HM, Milbank AJ, Tauras JM, et al. Redox regulation of cell signalling. Nature 1996;381:380–381 [DOI] [PubMed] [Google Scholar]
- 15.Sklavos MM, Tse HM, Piganelli JD. Redox modulation inhibits CD8 T cell effector function. Free Radic Biol Med 2008;45:1477–1486 [DOI] [PubMed] [Google Scholar]
- 16.Tse HM, Milton MJ, Schreiner S, Profozich JL, Trucco M, Piganelli JD. Disruption of innate-mediated proinflammatory cytokine and reactive oxygen species third signal leads to antigen-specific hyporesponsiveness. J Immunol 2007;178:908–917 [DOI] [PubMed] [Google Scholar]
- 17.Kwon J, Shatynski KE, Chen H, et al. The nonphagocytic NADPH oxidase Duox1 mediates a positive feedback loop during T cell receptor signaling. Sci Signal 2010;3:ra59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Laniewski NG, Grayson JM. Antioxidant treatment reduces expansion and contraction of antigen-specific CD8+ T cells during primary but not secondary viral infection. J Virol 2004;78:11246–11257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tse HM, Thayer TC, Steele C, et al. NADPH oxidase deficiency regulates Th lineage commitment and modulates autoimmunity. J Immunol 2010;185:5247–5258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mathews CE, Dunn BD, Hannigan MO, Huang CK, Leiter EH. Genetic control of neutrophil superoxide production in diabetes-resistant ALR/Lt mice. Free Radic Biol Med 2002;32:744–751 [DOI] [PubMed] [Google Scholar]
- 21.Ablamunits V, Elias D, Cohen IR. The pathogenicity of islet-infiltrating lymphocytes in the non-obese diabetic (NOD) mouse. Clin Exp Immunol 1999;115:260–267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mathews CE, Graser RT, Savinov A, Serreze DV, Leiter EH. Unusual resistance of ALR/Lt mouse beta cells to autoimmune destruction: role for beta cell-expressed resistance determinants. Proc Natl Acad Sci USA 2001;98:235–240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mathews CE, Suarez-Pinzon WL, Baust JJ, Strynadka K, Leiter EH, Rabinovitch A. Mechanisms underlying resistance of pancreatic islets from ALR/Lt mice to cytokine-induced destruction. J Immunol 2005;175:1248–1256 [DOI] [PubMed] [Google Scholar]
- 24.Walker JM. Molecular diagnosis of genetic diseases. In Methods in Molecular Medicine, Elles E., Ed Totowa, NJ, Humana Press, 1996 [Google Scholar]
- 25.Christianson SW, Shultz LD, Leiter EH. Adoptive transfer of diabetes into immunodeficient NOD-scid/scid mice. Relative contributions of CD4+ and CD8+ T-cells from diabetic versus prediabetic NOD.NON-Thy-1a donors. Diabetes 1993;42:44–55 [DOI] [PubMed] [Google Scholar]
- 26.Piganelli JD, Flores SC, Cruz C, et al. A metalloporphyrin-based superoxide dismutase mimic inhibits adoptive transfer of autoimmune diabetes by a diabetogenic T-cell clone. Diabetes 2002;51:347–355 [DOI] [PubMed] [Google Scholar]
- 27.Tse HM, Josephy SI, Chan ED, Fouts D, Cooper AM. Activation of the mitogen-activated protein kinase signaling pathway is instrumental in determining the ability of Mycobacterium avium to grow in murine macrophages. J Immunol 2002;168:825–833 [DOI] [PubMed] [Google Scholar]
- 28.Huang CK, Zhan L, Hannigan MO, Ai Y, Leto TL. P47(phox)-deficient NADPH oxidase defect in neutrophils of diabetic mouse strains, C57BL/6J-m db/db and db/+. J Leukoc Biol 2000;67:210–215 [DOI] [PubMed] [Google Scholar]
- 29.Morgan D, Oliveira-Emilio HR, Keane D, et al. Glucose, palmitate and pro-inflammatory cytokines modulate production and activity of a phagocyte-like NADPH oxidase in rat pancreatic islets and a clonal beta cell line. Diabetologia 2007;50:359–369 [DOI] [PubMed] [Google Scholar]
- 30.Newsholme P, Morgan D, Rebelato E, et al. Insights into the critical role of NADPH oxidase(s) in the normal and dysregulated pancreatic beta cell. Diabetologia 2009;52:2489–2498 [DOI] [PubMed] [Google Scholar]
- 31.Oliveira HR, Verlengia R, Carvalho CR, Britto LR, Curi R, Carpinelli AR. Pancreatic beta-cells express phagocyte-like NAD(P)H oxidase. Diabetes 2003;52:1457–1463 [DOI] [PubMed] [Google Scholar]
- 32.Lee KU, Amano K, Yoon JW. Evidence for initial involvement of macrophage in development of insulitis in NOD mice. Diabetes 1988;37:989–991 [DOI] [PubMed] [Google Scholar]
- 33.Lee KU, Kim MK, Amano K, et al. Preferential infiltration of macrophages during early stages of insulitis in diabetes-prone BB rats. Diabetes 1988;37:1053–1058 [DOI] [PubMed] [Google Scholar]
- 34.Lee KU, Pak CY, Amano K, Yoon JW. Prevention of lymphocytic thyroiditis and insulitis in diabetes-prone BB rats by the depletion of macrophages. Diabetologia 1988;31:400–402 [DOI] [PubMed] [Google Scholar]
- 35.Oschilewski U, Kiesel U, Kolb H. Administration of silica prevents diabetes in BB-rats. Diabetes 1985;34:197–199 [DOI] [PubMed] [Google Scholar]
- 36.Calderon B, Suri A, Unanue ER. In CD4+ T-cell-induced diabetes, macrophages are the final effector cells that mediate islet beta-cell killing: studies from an acute model. Am J Pathol 2006;169:2137–2147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jun HS, Yoon CS, Zbytnuik L, van Rooijen N, Yoon JW. The role of macrophages in T cell-mediated autoimmune diabetes in nonobese diabetic mice. J Exp Med 1999;189:347–358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cantor J, Haskins K. Recruitment and activation of macrophages by pathogenic CD4 T cells in type 1 diabetes: evidence for involvement of CCR8 and CCL1. J Immunol 2007;179:5760–5767 [DOI] [PubMed] [Google Scholar]
- 39.Daley JM, Thomay AA, Connolly MD, Reichner JS, Albina JE. Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J Leukoc Biol 2008;83:64–70 [DOI] [PubMed] [Google Scholar]
- 40.Jackson SH, Devadas S, Kwon J, Pinto LA, Williams MS. T cells express a phagocyte-type NADPH oxidase that is activated after T cell receptor stimulation. Nat Immunol 2004;5:818–827 [DOI] [PubMed] [Google Scholar]
- 41.Mathews CE, Leiter EH. Constitutive differences in antioxidant defense status distinguish alloxan-resistant and alloxan-susceptible mice. Free Radic Biol Med 1999;27:449–455 [DOI] [PubMed] [Google Scholar]
- 42.Mathews CE, Graser RT, Bagley RJ, et al. Genetic analysis of resistance to Type-1 Diabetes in ALR/Lt mice, a NOD-related strain with defenses against autoimmune-mediated diabetogenic stress. Immunogenetics 2003;55:491–496 [DOI] [PubMed] [Google Scholar]
- 43.DiLorenzo TP, Lieberman SM, Takaki T, et al. During the early prediabetic period in NOD mice, the pathogenic CD8(+) T-cell population comprises multiple antigenic specificities. Clin Immunol 2002;105:332–341 [DOI] [PubMed] [Google Scholar]
- 44.Graser RT, DiLorenzo TP, Wang F, et al. Identification of a CD8 T cell that can independently mediate autoimmune diabetes development in the complete absence of CD4 T cell helper functions. J Immunol 2000;164:3913–3918 [DOI] [PubMed] [Google Scholar]
- 45.Gloire G, Piette J. Redox regulation of nuclear post-translational modifications during NF-kappaB activation. Antioxid Redox Signal 2009;11:2209–2222 [DOI] [PubMed] [Google Scholar]
- 46.Oliveira-Marques V, Marinho HS, Cyrne L, Antunes F. Role of hydrogen peroxide in NF-kappaB activation: from inducer to modulator. Antioxid Redox Signal 2009;11:2223–2243 [DOI] [PubMed] [Google Scholar]
- 47.Sun Y, Oberley LW. Redox regulation of transcriptional activators. Free Radic Biol Med 1996;21:335–348 [DOI] [PubMed] [Google Scholar]
- 48.Donaldson M, Antignani A, Milner J, et al. p47phox-deficient immune microenvironment signals dysregulate naive T-cell apoptosis. Cell Death Differ 2009;16:125–138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sauer H, Wartenberg M, Hescheler J. Reactive oxygen species as intracellular messengers during cell growth and differentiation. Cell Physiol Biochem 2001;11:173–186 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.