Abstract
OBJECTIVE
The blood perfusion of pancreatic islets is highly variable and tightly regulated by the blood glucose concentration. Thus, oxygen levels are considered crucial for islet metabolism and function. Although islet oxygenation has been extensively studied in vitro, little is known about it in vivo. The current study aimed to investigate the oxygenation of the endocrine pancreas in vivo.
RESEARCH DESIGN AND METHODS
The reductive metabolism of 2-nitroimidazoles, such as pimonidazole, has previously been extensively used in studies of oxygen metabolism both in vitro and in vivo. At tissue oxygen levels <10 mmHg, pimonidazole accumulates intracellularly and may thereafter be detected by means of immunohistochemistry. Islet oxygenation was investigated in normal, 60% partially pancreatectomized, as well as whole-pancreas–transplanted rats. Moreover, leucine-dependent protein biosynthesis was performed using autoradiography to correlate islet oxygenation with metabolic activity.
RESULTS
In vivo, 20–25% of all islets in normal rats showed low oxygenation (pO2 <10 mmHg). Changes in the islet mass, by means of whole-pancreas transplantation, doubled the fraction of low-oxygenated islets in the endogenous pancreas of transplanted animals, whereas this fraction almost completely disappeared after a 60% partial pancreatectomy. Moreover, oxygenation was related to metabolism, since well-oxygenated islets in vivo had 50% higher leucine-dependent protein biosynthesis, which includes (pro)insulin biosynthesis.
CONCLUSIONS
The current study suggests a novel subpopulation of dormant low-oxygenated islets, which seems to constitute a functional reserve of endocrine cells. This study establishes a novel perspective on the use of the endocrine pancreas in glucose homeostasis.
The pancreatic islets have a vascular endothelial growth factor–dependent dense glomerular-like angioarchitecture (1–3), which assures that no portion of an islet is more than one cell away from arterial blood (4,5). Although the pancreatic islets only constitute 1–2% of the pancreatic volume, they receive 5–10% of the whole pancreatic blood flow (6,7). Moreover, the oxygen tension of large superficial pancreatic islets has been reported to be ∼40 mmHg, which is much higher than in other visceral organs (8,9). This may reflect high needs of oxygen for adequate glucose sensing and metabolism of the pancreatic β-cells (10,11). High vascularization may also be important for β-cell replication (12,13) and to facilitate fast disposal of secreted islet hormones.
The vascular tree of the endocrine pancreas is notably heterogeneous. Larger islets receive blood from one to three afferent arterioles, which penetrate into the islet and divide into numerous capillaries, whereas smaller islets seem to be integrated into the exocrine capillary system (14). Moreover, the degree of blood perfusion in different islets seems to be highly variable. When islet blood flow is measured by microsphere techniques, a small fraction of islets may contain more than six microspheres, whereas ∼90% of the islets are totally devoid of microspheres (15). A reasonable interpretation is that islets without microspheres have a blood perfusion below the detection limit, i.e., ∼375 nL/min. Repeated microsphere injections also show that up to 65% of islets may contain microspheres, but a substantial fraction still remains devoid of microspheres (16). Primarily the islets containing microspheres in the first injection contain microspheres after repeated microsphere injections, suggesting that the heterogeneity in blood perfusion is fairly constant over time.
We hypothesized that the highly variable islet blood perfusion observed in the rodent pancreas may result in a heterogeneous islet oxygenation, reflecting differences in islet function. For this purpose, we used pimonidazole, which is known to accumulate in tissues with an oxygen tension <10 mmHg (17–20), in order to assess tissue areas/cells low in oxygen tension within the endocrine pancreas. Furthermore, we studied leucine-dependent protein biosynthesis, which includes (pro)insulin biosynthesis, in order to assess whether differences in islet oxygenation in situ were associated with changed cellular activity.
RESEARCH DESIGN AND METHODS
Inbred male 3-month-old Wistar-Furth rats were purchased from B&K, Sollentuna, Sweden. All experimental procedures were approved by the animal ethics committee of Uppsala University.
Islet isolation and culture.
Pancreatic islets from Wistar-Furth rats were prepared by collagenase digestion and cultured free-floating in RPMI 1640 medium (Sigma-Aldrich, Irvine, U.K.) in groups of 150 islets (21).
Threshold for intracellular pimonidazole incorporation in vitro.
Islets were dispersed by trypsin digestion and placed on cover glasses (diameter 13 mm), precoated with poly-l-lysine (Sigma-Aldrich), and incubated for 30 min at 37°C to allow the cells to attach. RPMI 1640 medium was then changed for Krebs Ringer bicarbonate buffer (KRBH; 5.6 mmol/L glucose) with 200 μmol/L pimonidazole (Hypoxiaprobe-1; Chemicon International, Temecula, CA). Dispersed islet cells from each animal were incubated at 37°C for 4 h in airtight chambers containing gas mixtures of 0.5, 1.0, 1.5, or 2.0% oxygen (Supplementary Data). To assure complete equilibrium between the chamber’s gas mixture and the incubation medium, the KRBH was pregassed for 10 min, and the incubation time was doubled compared with standard in vitro experiments (compared below). All oxygen gas mixtures were produced by a certified manufacturer (Air Liquid Gas, Stockholm, Sweden) and the oxygen level within the airtight chambers was continuously monitored with an oxygen sensor with a drift of <0.05% per hour (Dräger PAC III, Lübeck, Germany). After incubation at different oxygen pressures, the dispersed islet cells were fixed in 4% paraformaldehyde for 5 min, washed in phosphate buffered saline (pH 7.4), and immunostained for pimonidazole. Because of practical reasons, these cover slides were stained for pimonidazole manually. The manual staining protocol differed from the automatized protocol (described below) by permeabilization of cells using 0.2% Triton-X for 10 min prior to antigen retrieval and incubation with the antipimonidazole monoclonal antibody (1:50) overnight at 4°C. In control experiments, the manual and automatic staining procedures produced similar results on tissue sections (data not shown).
Moreover, islet cell viability after incubation at 0.5, 1.0, 1.5, 2.0, or 20% O2 was evaluated by stainings with propidium iodide (Sigma-Aldrich; 20 μg/mL) and bisbenzimide (Sigma-Aldrich; 5 mg/mL), or immunostained for caspase-3 (Cell Signaling Technology, Danvers, MA). Captures of stained cells in a fluorescence microscope were processed in a computerized system for morphometry (ImageJ 1.3v; National Institutes of Health, Bethesda, MD). Total numbers of cells (nuclei stained with bisbenzimide), dead cells (cytoplasm stained with propidium iodide), or apoptotic cells (caspase-3 positive) were counted separately, followed by calculations of the percentage of vital cells.
Induction of decreased oxygenation in vitro.
Isolated islets were cultured for 24 h in excessive (15 mL) or normal amounts of culture medium (5 mL), followed by a 2-h incubation with pimonidazole at 95% air/5% CO2. The rationale for this experimental set-up was that excess nonstirred culture medium markedly increases the diffusion distance of oxygen from air to the pancreatic islets, thus decreasing the oxygenation of the cultured islets (22), which was confirmed by oxygen measurements using modified Clark microelectrodes.
Induction of decreased tissue oxygenation in vivo.
The animals were anesthetized with thiobutabarbital sodium (120 mg/kg i.p.; Inactin; Research Biochemicals International, Natick, MA), and maintained at 37°C. Polyethylene catheters were inserted into the right femoral artery and vein. The pancreas was immobilized over a cylindrical plastic block and sterile-filtered neutral red (0.8 mL; 2% wt/vol; Kebo Grave, Stockholm, Sweden) was administered intravenously to selectively stain the islets (23). Neutral red does not affect islet function, blood flow, or oxygen tension (8,23). The tissue oxygen tension of a superficial pancreatic islet was then continuously monitored with a Clark microelectrode (8,24). Pimonidazole was injected intravenously and a graded aortic vascular clamp was positioned superior to the celiac artery to allow an arterial blood pressure of ∼20 mmHg, resulting in an islet oxygen tension of 5–10 mmHg for 2 h.
Pancreaticoduodenal transplantation.
The whole-donor pancreas was retrieved and transplanted, as previously described (25). Three weeks after transplantation, the animals were allocated to measurements of pancreatic tissue oxygenation.
Partial pancreatectomy.
A transverse incision was made in the left hypochondrium to visualize the pancreas. Thereafter, the splenic 60% of the pancreas (approximately 500 mg) was removed, the abdomen was closed, and the animals were left to recover. One week or 4 weeks later, the animals were allocated to measurements of pancreatic tissue oxygenation.
Measurements of tissue oxygenation.
Islet oxygenation was studied in dispersed or isolated islets, native pancreatic islets of awake rats (n = 8), native and transplanted islets of whole-pancreas–transplanted rats (n = 8), and native islets of partially (60%) pancreatectomized rats 1 week (n = 8) or 4 weeks (n = 8) postsurgery and in native pancreatic islets during induced hypoxia by graded vascular clamping (n = 2). The oxygenation marker pimonidazole was added to the culture medium (200 μmol/L), or injected intravenously into the animals’ tail vein (60 mg/kg). Two hours later, islets were retrieved from culture, or the animals were killed and their pancreata along with part of their livers were removed and prepared for histological analysis. In all animals, blood glucose concentration was measured by test reagent strips (MediSense; Baxter Travenol, Deerfield, IL).
Immunohistochemical staining for pimonidazole.
Paraffin-embedded tissue sections, 5 μm thick, were mounted on poly-l-lysine coated glass slides. Antigens were retrieved from the sections by incubation with 0.01% (wt/vol) pronase (Dako, Glostrup, Denmark) for 40 min at 40°C. An automatic staining procedure (Dako Autostainer; Dako) was applied to facilitate reproducible staining at room temperature. The slides were incubated with Dako blocking solution (Dako) for 30 min and incubated with primary antibody against pimonidazole, diluted 1:25 in Tris-buffered saline (TBS) with 0.2% (wt/vol) Brij 35, for 40 min. Thereafter, the slides were washed in TBS and incubated with a biotinylated secondary goat F(ab’)2 anti-mouse antibody (Southern Biotechnology, Birmingham, AL), diluted 1:500 in TBS with 0.2% Brij 35 and 0.035% (vol/vol) Dako blocking solution (Dako) for 30 min. The slides were washed and incubated with Alkaline Phosphatase Standard ABC kit (Vector Laboratories, Burlingame, CA) for 30 min, followed by incubation with Vector Red Alkaline Phosphatase substrate kit (Vector Laboratories) for 25 min. Liver tissue was used as positive control slides (26), whereas animals not given an intravenous injection of pimonidazole were used as negative controls. In each pancreas, islets in ≥10 tissue sections (86 ± 13; n = 32) were evaluated for presence of pimonidazole by an examiner unaware of the origin of the samples.
Estimation of islet mass.
The fractional volume of the islets in each pancreas was determined by a point-counting technique (27). Because the densities of the exocrine and endocrine pancreas differ less than 1% (28), the islet mass was estimated by multiplying the pancreatic weight by the islet volume fraction of the whole pancreas.
Islet oxygenation and leucine-dependent protein biosynthesis in vivo.
A previous study protocol for [3H]-l-leucine autoradiography (29) was modified. Briefly, pimonidazole (60 mg/kg) was injected into the tail vein of awake rats. After 90 min, the same rats were given an intravenous injection of 25 μCi 3,4,5-[3H]-l-leucine (American Radiolabeled Chemicals, St. Louis, MO; 1 mCi/ml) dissolved in saline. The animals were killed 30 min later and the pancreata and livers were retrieved for histological evaluation of both pimonidazole incorporation and [3H]-l-leucine autoradiography. Tissue sections were first immunostained for pimonidazole (described above). Thereafter, wet slides were dipped in 50% autoradiography film emulsion (Eastman Kodak Co, Rochester, NY), and dried overnight in a light-proof chamber. The slides were kept for 1 week at 4°C before being developed. In a light microscope (1000×) digital pictures were made of both islets positive and negative for pimonidazole. An examiner unaware of the origin of the samples counted the number of autoradiographic silver grains in pimonidazole-positive and pimonidazole-negative islets, respectively. In each animal, >0.02 mm2 islet tissue were evaluated using a computerized system for morphometry (ImageJ). Leucine-dependent protein biosynthesis was then expressed as silver grains per millimeter squared islet tissue.
Pimonidazole immunostaining and blood flow in vivo.
In anesthetized animals (n = 3), 2 hours after an intravenous injection of pimonidazole (compared above) simultaneous islet blood flow measurements were performed using a microsphere technique, as described elsewhere (15).
Statistical analysis.
All values are given as means ± SEM. Student t test or χ2 test was used for comparisons between only two experimental groups, whereas multiple comparisons between data were performed by using ANOVA and Bonferroni post hoc test. For all comparisons, P < 0.05 was considered statistically significant.
RESULTS
Body weights, pancreatic weights, total islet mass, and blood glucose concentrations.
All animals allocated to this study weighed ∼300 g and were normoglycemic, although the control animals had slightly lower blood glucose concentrations (Table 1). Whole-pancreas transplantation roughly doubled the islet mass, and after partial pancreatectomy the islet mass was 40–50% of that in control animals.
TABLE 1.
Body weights, pancreatic weights, total islet mass, and blood glucose concentrations in control, whole-pancreas–transplanted, and partially pancreatectomized rats subjected to measurements of pancreatic oxygenation by intravenous pimonidazole injections
| Control animals | Whole-pancreas–transplanted animals |
Partially pancreatectomized animals |
||||||
|---|---|---|---|---|---|---|---|---|
| 1 Week |
4 Weeks |
|||||||
| Before | After | Before | After | Before | After | |||
| Body weight (g) | 310 ± 6 | 318 ± 2 | 335 ± 6* | 319 ± 3 | 299 ± 5* | 308 ± 5 | 336 ± 5* | |
| Total pancreas mass (mg) | 915 ± 36 | — | 1915 ± 92† | — | 382 ± 14† | — | 386 ± 31† | |
| Weight of caput (mg) | 356 ± 16 | — | 450 ± 31 (Native) | 429 ± 23 (Tx) | — | 382 ± 14 | — | 386 ± 31 |
| Weight of cauda (mg) | 558 ± 28 | — | 583 ± 40 (Native) | 454 ± 30 (Tx) | 523 ± 13 | — | 498 ± 28 | — |
| Total islet mass (mg) | 8.1 ± 0.7 | — | 15.4 ± 0.9† | — | 3.2 ± 0.4† | — | 3.8 ± 0.4† | |
| Blood glucose (mmol/L) | 4.7 ± 0.1 | — | 5.8 ± 0.2† | 5.6 ± 0.2† | 6.0 ± 0.3† | 5.0 ± 0.1 | 5.8 ± 0.2*† | |
All values are given as means ± SEM for 8 experiments in each group.
*P < 0.05 when compared with rats before surgery.
†P < 0.05 when compared with control rats.
Threshold for intracellular pimonidazole incorporation in vitro.
Dispersed islet cells were seeded on poly-l-lysine–coated cover glasses to exclude oxygen tension gradients within islets. It was, however, technically impossible to fully avoid areas with high densities of cells. Dispersed cells in these “high density” areas were often high in pimonidazole-positive cells, most likely due to higher oxygen consumption in that particular area of the cover glass. These cells constituted the low fraction of pimonidazole-positive cells (∼10%) after incubations at oxygen tension levels >10 mmHg (Fig. 1). The observed percentage of islet cells positive for pimonidazole at oxygen tension levels <10 mmHg is similar to previously reported data, when oxygenation was measured in clamped tumors with microelectrodes and thereafter correlated to the fraction of pimonidazole-accumulating cells (18). In dispersed islet cells, pimonidazole accumulation was induced at 1.0% O2 (pO2 ∼8 mmHg), as detected by immunohistochemistry. Islet cell viability, as measured by means of propidium iodide stainings, was similar after incubation at 0.5, 1.0, 1.5, and 2.0% O2, respectively (97 ± 2, 95 ± 2, 97 ± 2, or 95 ± 2% vital cells, n = 4). Hyperoxia (20% O2; pO2 ∼150 mmHg) did not improve cell viability (96 ± 3% vital cells, n = 4). Moreover, the number of apoptotic caspase-3 positive free islet cells was similar and below 5% after incubation at 0.5, 1.0, 1.5, or 2.0% O2 (n = 3).
FIG. 1.
Percentages of dispersed islet cells, which stain positive for a marker of low oxygenation (pimonidazole; black bars), necrosis (propidium iodide; hatched bars), or apoptosis (caspase-3; white bars) after incubation at 37°C and 0.5, 1.0, 1.5, or 2.0% O2 for 4 h. Values are given as means ± SEM for 3–4 paired experiments in each group. *P < 0.05 when compared with 0.5 and 1.0% O2.
Induction of decreased oxygenation in vitro.
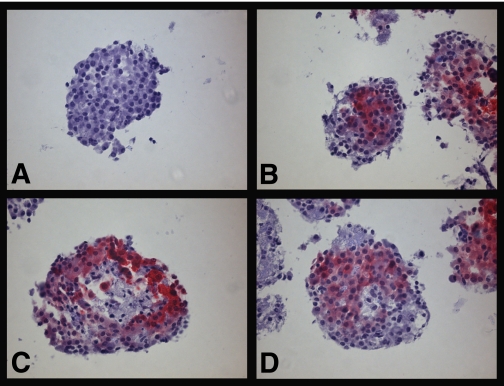
The oxygen tension at the surface of cultured islets, incubated at standard culture conditions (95% air/5% CO2, 5 mL culture medium), was almost twice that in culture dishes with excessive (15 mL) culture media (24.4 ± 0.6 vs. 14.9 ± 0.3 mmHg; n = 3, P < 0.05), when measured by Clark microelectrodes. A majority of isolated pancreatic islets incubated at normal culture conditions stained negative for pimonidazole (Fig. 2A), irrespective of preceding culture period (57 ± 2% of islets; n = 3 for 1, 5, and 10 days, respectively). In islets that stained positive for pimonidazole, an obvious gradient with pronounced central staining and unstained periphery of the islets was discerned (Fig. 2B). In some larger islets, especially when investigated without preceding culture, there was central necrosis surrounded by pimonidazole staining (Fig. 2C). Incubation of islets with excessive culture medium (15 mL) increased the fraction of pimonidazole-positive islets (77 ± 7 vs. 43 ± 2%; n = 3, P < 0.05). Isolated large pancreatic islets incubated with excessive culture medium regularly developed central necrosis, which was surrounded by cells staining positive for pimonidazole (Fig. 2D).
FIG. 2.
Micrographs of pancreatic islets exposed to pimonidazole for 2 h during culture and thereafter stained with antibody (red) for adducts of pimonidazole incorporated intracellularly. A: Islet negative for pimonidazole. B: Islets positive for pimonidazole. C: Freshly isolated islet positive for pimonidazole with central necrosis. D: Islet incubated with excessive culture medium. Scale bars 100 μm. (A high-quality digital representation of this figure is available in the online issue.)
Pancreas oxygenation in vivo.
In untreated control rats, 20–25% of the pancreatic islets stained positive for pimonidazole (Fig. 3), whereas the exocrine pancreatic parenchyma remained negative. Pimonidazole-positive islets were in all cases homogenously stained (Fig. 4A). There were no differences in frequency of islets positive for pimonidazole between the head and tail of the pancreas (27 ± 6 and 20 ± 3%, respectively; n = 8). Pimonidazole staining did not correlate to islet size or blood glucose concentration.
FIG. 3.
Fraction of islets that stain positive for pimonidazole in control (native pancreas of nontransplanted animals), whole-pancreas–transplanted, and 60% pancreatectomized rats. Values are given as means ± SEM for eight experiments in each group. *P < 0.05 when compared with controls; †P < 0.05 when compared with the native pancreas of whole-pancreas–transplanted animals.
FIG. 4.
Pancreatic islets of normal animals compared with rats subjected to graded aortic vascular clamping applied cranially to the celiac artery for 2 h to induce islet hypoxia. Pimonidazole was injected 2 h before the pancreas was retrieved for histological sectioning followed by staining with antibody (red) for pimonidazole. A: Normal pancreatic islets staining negative (arrow) and positive (arrow head) for pimonidazole, respectively. B: An islet with distinct pimonidazole positivity (arrow) after induced hypoxia by vascular clamping. Note also the pimonidazole staining in the periphery of an exocrine lobulus (arrow head). Scale bars 100 µm. (A high-quality digital representation of this figure is available in the online issue.)
Whole-pancreas–transplanted animals were used to investigate the importance of islet innervation and mass for the variable oxygenation of pancreatic islets. The transplanted whole pancreas had similar frequency of islets that stained positive for pimonidazole as the pancreas of nontransplanted animals (Fig. 3). However, the intact native pancreas of these transplanted animals had an increased number of pimonidazole-positive islets (Fig. 3). This increase of pimonidazole-stained islets was similar in both the head and tail region of these pancreata (data not shown). Up to four weeks after ∼60% partial pancreatectomy pimonidazole-positive islets were rare in the remaining pancreas (Fig. 3).
All pancreatic islets in the animals subjected to a graded aortic vascular clamping applied cranially to the celiac artery stained strongly positive for pimonidazole and had pronounced dilatation of their capillaries (Fig. 4B). In contrast to control animals, the exocrine pancreas of vascular clamped animals displayed pimonidazole stainings with the peripheral margins of each pancreatic lobulus strongly positive, which probably reflect that these regions are supplied by the most distal parts of the pancreatic vascular tree.
Islet oxygenation and leucine-dependent protein biosynthesis in vivo.
Previous studies have reported heterogeneity of β-cell protein biosynthesis (30,31). In the current study, the resolution of the assay was, however, not sufficient to analyze protein biosynthesis on the cellular level, and the data were therefore expressed per millimeter squared islet tissue (Fig. 5). Leucine-dependent protein biosynthesis in islets unstained for pimonidazole was 50% higher than in pimonidazole-positive islets (12,949 ± 1,882 vs. 8,794 ± 9,62 silver grains/mm2; n = 7, P < 0.05). The use of twice the amount of [3H]-l-leucine (50 μCi/animal, n = 2) gave similar values for protein biosynthesis in both pimonidazole-positive and pimonidazole-negative islets (data not shown). Furthermore, [3H]-l-leucine in plasma was observed to be cleared by 70% within 10 min (n = 2), which suggests that [3H]-l-leucine is rapidly taken up by the cells.
FIG. 5.
Protein biosynthesis as measured by [3H]-l-leucine autoradiography in low-oxygenated pimonidazole-positive (pO2 <10 mmHg) vs. pimonidazole-negative islets. Values are given as means ± SEM for 7 animals. *P < 0.05 when compared with islets positive for pimonidazole.
Pimonidazole immunostaining and blood flow in vivo.
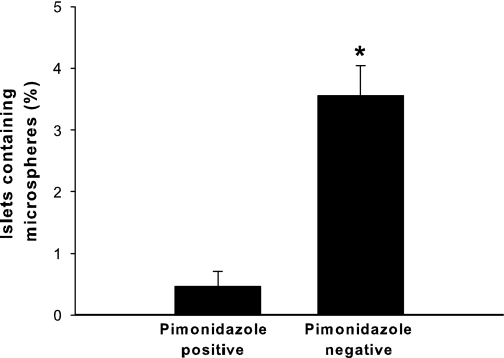
Islet pimonidazole immunostainings were dependent on islet blood flow (Table 2), and the percentage of pimonidazole-negative islets containing microspheres was higher than in pimonidazole-positive islets (3.6 ± 0.8 vs. 0.5 ± 0.4%; n = 3, P < 0.05) (Fig. 6).
TABLE 2.
Cumulative distribution of islets from three rats after simultaneous islet oxygenation (pimonidazole immunostaining) and blood flow measurements (microspheres)
| Pimonidazole immunostaining |
||
|---|---|---|
| Negative | Positive | |
| Islets containing microspheres | ||
| No | 1,292 | 378 |
| Yes | 48 | 2 |
The pimonidazole immunostaining was dependent on the simultaneous blood flow measurements (χ2 test, P < 0.05), suggesting that pimonidazole preferentially accumulates in islets with low blood perfusion.
FIG. 6.
Percentages of microsphere-containing islets among pimonidazole-positive or pimonidazole-negative islets. All values are given as means ± SEM for 3 animals. *P < 0.05 when compared with islets positive for pimonidazole.
DISCUSSION
In healthy subjects most visceral organs have a functional overcapacity, which means that full metabolic activity of all cells in that organ is not needed to maintain homeostasis. Moreover, there are large variations in metabolic activity and oxygen consumption within as well as between different organs. In line with this, the current study is the first to report a great heterogeneity in oxygenation of endogenous pancreatic islets. The novel data also suggest that a normally low-oxygenated (<10 mmHg) subpopulation of islets (20–25%) may constitute a functional reserve of insulin-producing cells.
Our laboratory has previously reported an oxygen tension of ∼40 mmHg in endogenous islets, as measured by modified Clark microelectrodes (8,9). However, in order to visualize the pancreatic islets in vivo an intravital islet staining (neutral red) is needed. Since the intravital stain is delivered through the vascular system, and the microelectrodes only can assess the oxygenation of large subcapsular islets (>100 μm), belonging to an islet subgroup that do not form any insuloacinar portal system (23), it is, however, likely that these measurements do not reflect the true oxygenation of the whole endocrine pancreas.
The oxygen-dependent metabolism of 2-nitroimidazoles by nitroreductases may be used to detect low-oxygenated cells (18) because reduced forms of 2-nitroimidazoles bind to intracellular proteins and may thereafter be visualized by immunohistochemistry (32). We now report that the 2-nitroimidazole pimonidazole is feasible for measurements of islet oxygenation both in vitro and in vivo, and similarly to that observed in other tissues pimonidazole accumulates in islet cells at oxygen tensions <10 mmHg. At normal culture conditions most small islets were unstained for pimonidazole, whereas large islets were often observed with pronounced gradients of pimonidazole staining toward the islet core, reflecting an oxygen tension gradient due to oxygen consumption and/or diffusion limitations. In some larger islets an unstained central necrosis surrounded by pimonidazole staining was noted due to loss of nitroreductase activity in dead cells (33). Notably, during normal culture conditions there was a major drop in oxygen tension from the surface of the culture medium (∼150 mmHg) to the vicinity of the islets (∼25 mmHg). Given that the oxygen consumption rate of isolated islet cells is similar to cells in intact islets, the pitfalls with oxygen diffusion and consumption properties may be limited by the decreased cellular density as well as the minimal diffusion distance when incubating dispersed islet cells in airtight chambers with known oxygen levels. Thus, this experimental set-up was chosen to elucidate the “true” threshold for pimonidazole accumulation in islet cells, and these experiments confirmed that pimonidazole accumulates in islet cells below 10 mmHg without any concomitant signs of increased rates of necrosis or apoptosis. Moreover, islet pimonidazole accumulation was inducible in vitro (excessive culture medium) as well as in vivo, where a subphrenic vascular clamp in anesthetized rats caused both islet and exocrine tissues to display strong pimonidazole positivity. Accumulation of metabolically derived substances such as adenosine has been reported to dilate islet blood vessels (34), and in line with this the islet capillaries were markedly widened, probably because of islet afferent arteriole relaxation in an attempt to compensate for the decreased islet blood perfusion during vascular clamping.
Following these control experiments, we investigated the oxygenation of endogenous islets measured by pimonidazole in normal awake rats. Surprisingly, 20–25% of the pancreatic islets accumulated pimonidazole. In contrast to the investigated isolated islets (compared above) pimonidazole-positive islets in vivo always displayed homogenous staining, which suggests that the low oxygen tension in islets in vivo was not due to limitations in diffusion from islet capillaries. The pattern with homogenously pimonidazole-stained islets rather indicates vasoconstriction at the islet arteriolar level, resulting in very low blood perfusion of the entire islet. Islet blood perfusion is normally regulated at the islet arteriolar level (23) independently from the exocrine pancreas by hormonal, neural, and vasoregulatory factors (35). Moreover, in normal animals no pimonidazole staining in the exocrine pancreas was observed, suggesting a homogenous oxygenation above 10 mmHg, which is supported by previous measurements of oxygen tension within the exocrine pancreas (8,36,37).
Questions have been raised concerning the potential influence of variable glucose concentrations on 2-nitroimidazole binding to low-oxygenated cells (38). An experimental design in which normoglycemic animals with differences in islet functional load through variations in islet mass was therefore chosen for the current study. For this purpose, experimental set-ups with whole-pancreas–transplanted or partially pancreatectomized animals were designed. The doubled islet mass after whole-pancreas transplantation resulted in increased numbers of islets staining positive for pimonidazole in the endogenous pancreas, whereas the islets in the transplanted pancreas had similar fraction of pimonidazole-positive islets as in normal animals. This suggests that an increase in islet mass resulting from decreased metabolic demands recruits more islets to the low-oxygenated subpopulation. Moreover, since the transplanted pancreas is denervated, it is most likely that this phenomenon is mediated by nervous mechanisms, which may downregulate islet metabolism followed by a compensatory decrease in blood perfusion and oxygenation because a high blood perfusion to islets with resting activity would be wasted.
Up to 4 weeks after decreasing the islet mass to 40–50% by partial pancreatectomy almost no islets stained positive for pimonidazole. These experiments demonstrate that the pimonidazole staining in the intact pancreas was not irreversibly inherent to some islets. Instead the data indicate that all islets became well-perfused and that all accessible islet function was needed to maintain normoglycemia in these animals. The mechanism for the recruitment of pimonidazole-positive islets is most likely to reflect local mediators or local nervous mechanisms and not the extrinsic nervous mechanism involved in the closing down of excess islets, since the transplanted denervated pancreas had similar fraction of pimonidazole-positive islets as normal animals.
It seems unlikely that positive pimonidazole staining would be associated with increased metabolic function and oxygen consumption, since increased islet mass resulted in augmented staining and decreased islet mass almost completely abolished the fraction of pimonidazole-accumulating islets. Therefore, we hypothesized that the pimonidazole-positive islets are metabolically less active with, for example, decreased protein synthesis. Indeed, a modified protocol for [3H]-l-leucine autoradiography to measure leucine-dependent protein biosynthesis, of which (pro)insulin biosynthesis normally accounts for approximately 25%, in endogenous islets (29), revealed decreased protein biosynthesis in pimonidazole-positive compared with unstained islets. This did not seem to reflect differences in blood perfusion, since varying the dosage of [3H]-l-leucine in control experiments did not affect the absolute values for protein biosynthesis in neither pimonidazole-positive nor pimonidazole-negative islets. However, it is reasonable to believe that islets with low metabolic activity and low oxygenation also have low blood perfusion, and simultaneous pimonidazole immunostainings and blood flow measurements, using a microsphere technique, confirmed that pimonidazole preferentially accumulates in islets with low blood flow.
The present findings imply a low-oxygenated and functionally less active subpopulation of islets. This underlines the previously observed anatomic and functional heterogeneity of individual β-cells and islets described both in vitro and in vivo (14,30,31,39,40). The interplay between islet mass, insulin resistance and blood glucose may regulate the size of the proposed metabolically dormant islet subpopulation, which may be of functional importance when an increased β-cell mass is needed such as in pregnant (12,41) or insulin-resistant subjects (42).
The metabolism of normal β-cells is well-known to be more or less strictly aerobic (43), and pathological conditions with anaerobic metabolism have been observed to result in inappropriate insulin secretion (44,45). Thus, the observed low degree of islet oxygenation is probably sufficient to maintain basal functions and viability of islet cells, and we deem it highly unlikely that β-cells in vivo are restricted by their oxygen supply. Indeed, islet cells exposed to oxygen tension levels <12 mmHg in vitro have a decreased second phase of glucose-stimulated insulin secretion (46), whereas in vivo other mechanisms than oxygenation seem to regulate insulin secretion (47).
In conclusion, the current study reports that there is a markedly heterogeneous oxygenation of endogenous pancreatic islets. During normal circumstances 20–25% of all islets accumulate the low-oxygen tension marker pimonidazole, the size of the subpopulation varies with the size of the total islet mass, and the protein biosynthesis of these low-oxygenated islets is concomitantly decreased. The present findings propose a dormant subpopulation of pancreatic islets, which may constitute a functional reserve of insulin-producing cells.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by grants from The Swedish Research Council (72XD-15043), the Juvenile Diabetes Research Foundation, the Swedish Diabetes Association, the Novo Nordisk Foundation, AFA Insurance, European Foundation for the Study of Diabetes/Novo, the Family Ernfors Fund, the Swedish Society for Medical Research, and the David and Astrid Hageléns Foundation. No other potential conflicts of interest relevant to this article were reported.
R.O. researched data and wrote the manuscript. P.-O.C. researched data, contributed to discussion, and reviewed the manuscript.
Birgitta Bodin, Astrid Nordin, Eva Törnelius, and Guangxiang Zang (Department of Medical Cell Biology, Uppsala University, Sweden) are gratefully acknowledged for their skilled technical assistance.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db09-0877/-/DC1.
See accompanying commentary, p. 2018.
REFERENCES
- 1.Lammert E, Gu G, McLaughlin M, et al. Role of VEGF-A in vascularization of pancreatic islets. Curr Biol 2003;13:1070–1074 [DOI] [PubMed] [Google Scholar]
- 2.Kamba T, Tam BY, Hashizume H, et al. VEGF-dependent plasticity of fenestrated capillaries in the normal adult microvasculature. Am J Physiol Heart Circ Physiol 2006;290:H560–H576 [DOI] [PubMed] [Google Scholar]
- 3.Brissova M, Shostak A, Shiota M, et al. Pancreatic islet production of vascular endothelial growth factor–A is essential for islet vascularization, revascularization, and function. Diabetes 2006;55:2974–2985 [DOI] [PubMed] [Google Scholar]
- 4.Bonner-Weir S. Morphological evidence for pancreatic polarity of beta-cell within islets of Langerhans. Diabetes 1988;37:616–621 [DOI] [PubMed] [Google Scholar]
- 5.Brunicardi FC, Stagner J, Bonner-Weir S, et al. ; Long Beach Veterans Administration Regional Medical Education Center Symposium Microcirculation of the islets of Langerhans. Diabetes 1996;45:385–392 [DOI] [PubMed] [Google Scholar]
- 6.Lifson N, Lassa CV, Dixit PK. Relation between blood flow and morphology in islet organ of rat pancreas. Am J Physiol 1985;249:E43–E48 [DOI] [PubMed] [Google Scholar]
- 7.Jansson L, Carlsson PO. Graft vascular function after transplantation of pancreatic islets. Diabetologia 2002;45:749–763 [DOI] [PubMed] [Google Scholar]
- 8.Carlsson PO, Liss P, Andersson A, Jansson L. Measurements of oxygen tension in native and transplanted rat pancreatic islets. Diabetes 1998;47:1027–1032 [DOI] [PubMed] [Google Scholar]
- 9.Carlsson PO, Palm F, Andersson A, Liss P. Markedly decreased oxygen tension in transplanted rat pancreatic islets irrespective of the implantation site. Diabetes 2001;50:489–495 [DOI] [PubMed] [Google Scholar]
- 10.Hellerström C. Effects of carbohydrates on the oxygen consumption of isolated pancreatic islets of mice. Endocrinology 1967;81:105–112 [DOI] [PubMed] [Google Scholar]
- 11.Hutton JC, Malaisse WJ. Dynamics of O2 consumption in rat pancreatic islets. Diabetologia 1980;18:395–405 [DOI] [PubMed] [Google Scholar]
- 12.Johansson M, Mattsson G, Andersson A, Jansson L, Carlsson PO. Islet endothelial cells and pancreatic beta-cell proliferation: studies in vitro and during pregnancy in adult rats. Endocrinology 2006;147:2315–2324 [DOI] [PubMed] [Google Scholar]
- 13.Nikolova G, Jabs N, Konstantinova I, et al. The vascular basement membrane: a niche for insulin gene expression and beta cell proliferation. Dev Cell 2006;10:397–405 [DOI] [PubMed] [Google Scholar]
- 14.Bonner-Weir S. The microvasculature of the pancreas, with special emphasis on that of the islets of Langerhans. Anatomy and functional implications. In The Pancreas. Biology, pathophysiology and disease, 2nd ed. Go VLW, DiMagno EP, Gardner JD, Lebenthal E, Reber HA, Scheele GA, Eds. New York, Raven Press 1993, p. 759–768
- 15.Jansson L. Microsphere distribution in the pancreas of anesthetized rats. Alloxan stimulates the blood flow to all islets whereas glucose only affects the blood perfusion of a subgroup of islets. Int J Pancreatol 1996;20:69–74 [DOI] [PubMed] [Google Scholar]
- 16.Carlsson PO, Källskog O, Bodin B, Andersson A, Jansson L. Multiple injections of coloured microspheres for islet blood flow measurements in anaesthetised rats: influence of microsphere size. Ups J Med Sci 2002;107:111–120 [DOI] [PubMed] [Google Scholar]
- 17.Hodgkiss RJ. Use of 2-nitroimidazoles as bioreductive markers for tumour hypoxia. Anticancer Drug Des 1998;13:687–702 [PubMed] [Google Scholar]
- 18.Raleigh JA, Chou SC, Arteel GE, Horsman MR. Comparisons among pimonidazole binding, oxygen electrode measurements, and radiation response in C3H mouse tumors. Radiat Res 1999;151:580–589 [PubMed] [Google Scholar]
- 19.Gardiner TA, Gibson DS, de Gooyer TE, de la Cruz VF, McDonald DM, Stitt AW. Inhibition of tumor necrosis factor-alpha improves physiological angiogenesis and reduces pathological neovascularization in ischemic retinopathy. Am J Pathol 2005;166:637–644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Franco M, Man S, Chen L, et al. Targeted anti-vascular endothelial growth factor receptor-2 therapy leads to short-term and long-term impairment of vascular function and increase in tumor hypoxia. Cancer Res 2006;66:3639–3648 [DOI] [PubMed] [Google Scholar]
- 21.Andersson A. Isolated mouse pancreatic islets in culture: effects of serum and different culture media on the insulin production of the islets. Diabetologia 1978;14:397–404 [DOI] [PubMed] [Google Scholar]
- 22.Andersson A. Tissue culture of isolated pancreatic islets. Acta Endocrinol Suppl (Copenh) 1976;205:283–294 [PubMed] [Google Scholar]
- 23.Carlsson PO, Jansson L, Ostenson CG, Källskog O. Islet capillary blood pressure increase mediated by hyperglycemia in NIDDM GK rats. Diabetes 1997;46:947–952 [DOI] [PubMed] [Google Scholar]
- 24.Revsbech NP. An oxygen microsensor with guard cathode. Limnol Oceanogr 1989;34:474–478 [Google Scholar]
- 25.Jansson L, Korsgren O, Wahlberg J, Andersson A. Pancreatic islet blood flow after syngeneic pancreaticoduodenal transplantation in rats. Differences between the blood perfusion of the native and the transplanted gland. Transplantation 1992;53:517–521 [DOI] [PubMed] [Google Scholar]
- 26.Arteel GE, Thurman RG, Yates JM, Raleigh JA. Evidence that hypoxia markers detect oxygen gradients in liver: pimonidazole and retrograde perfusion of rat liver. Br J Cancer 1995;72:889–895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weibel E. Practical methods for biological morphometry. In Stereological Methods. Wiebel ER, Ed. London, Academic Press, 1979 [Google Scholar]
- 28.Chadwick DR, Robertson GS, Toomey P, et al. Pancreatic islet purification using bovine serum albumin: the importance of density gradient temperature and osmolality. Cell Transplant 1993;2:355–361 [DOI] [PubMed] [Google Scholar]
- 29.Logothetopoulos J, Jain K. In vivo incorporation of [3H]leucine and [3H]tryptophan into proinsulin-insulin and other islet cell proteins in normoglycemic, hyperglycemic, and hypoglycemic rats. Diabetes 1980;29:801–805 [DOI] [PubMed] [Google Scholar]
- 30.Schuit FC, In’t Veld PA, Pipeleers DG. Glucose stimulates proinsulin biosynthesis by a dose-dependent recruitment of pancreatic beta cells. Proc Natl Acad Sci USA 1988;85:3865–3869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kiekens R, In ’t Veld P, Mahler T, Schuit F, Van De Winkel M, Pipeleers D. Differences in glucose recognition by individual rat pancreatic B cells are associated with intercellular differences in glucose-induced biosynthetic activity. J Clin Invest 1992;89:117–125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhong Z, Arteel GE, Connor HD, et al. Cyclosporin A increases hypoxia and free radical production in rat kidneys: prevention by dietary glycine. Am J Physiol 1998;275:F595–F604 [DOI] [PubMed] [Google Scholar]
- 33.Rasey JS, Grunbaum Z, Magee S, et al. Characterization of radiolabeled fluoromisonidazole as a probe for hypoxic cells. Radiat Res 1987;111:292–304 [PubMed] [Google Scholar]
- 34.Carlsson PO, Olsson R, Källskog O, Bodin B, Andersson A, Jansson L. Glucose-induced islet blood flow increase in rats: interaction between nervous and metabolic mediators. Am J Physiol Endocrinol Metab 2002;283:E457–E464 [DOI] [PubMed] [Google Scholar]
- 35.Jansson L. The regulation of pancreatic islet blood flow. Diabetes Metab Rev 1994;10:407–416 [DOI] [PubMed] [Google Scholar]
- 36.Harper SL, Pitts VH, Granger DN, Kvietys PR. Pancreatic tissue oxygenation during secretory stimulation. Am J Physiol 1986;250:G316–G322 [DOI] [PubMed] [Google Scholar]
- 37.Carlsson PO, Palm F. Oxygen tension in isolated transplanted rat islets and in islets of rat whole-pancreas transplants. Transpl Int 2002;15:581–585 [DOI] [PubMed] [Google Scholar]
- 38.Ling LL, Sutherland RM. Modulation of the hypoxic toxicity and binding of misonidazole by glucose. Br J Cancer 1986;54:911–917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pipeleers DG. Heterogeneity in pancreatic beta-cell population. Diabetes 1992;41:777–781 [DOI] [PubMed] [Google Scholar]
- 40.Aizawa T, Kaneko T, Yamauchi K, et al. Size-related and size-unrelated functional heterogeneity among pancreatic islets. Life Sci 2001;69:2627–2639 [DOI] [PubMed] [Google Scholar]
- 41.Zhang H, Zhang J, Pope CF, et al. Gestational diabetes mellitus resulting from impaired beta-cell compensation in the absence of FoxM1, a novel downstream effector of placental lactogen. Diabetes 2010;59:143–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lavine JA, Raess PW, Stapleton DS, et al. Cholecystokinin is up-regulated in obese mouse islets and expands beta-cell mass by increasing beta-cell survival. Endocrinology 2010;151:3577–3588 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hellman B, Idahl LA, Sehlin J, Täljedal IB. Influence of anoxia on glucose metabolism in pancreatic islets: lack of correlation between fructose-1,6-diphosphate and apparent glycolytic flux. Diabetologia 1975;11:495–500 [DOI] [PubMed] [Google Scholar]
- 44.Otonkoski T, Jiao H, Kaminen-Ahola N, et al. Physical exercise-induced hypoglycemia caused by failed silencing of monocarboxylate transporter 1 in pancreatic beta cells. Am J Hum Genet 2007;81:467–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Otonkoski T, Kaminen N, Ustinov J, et al. Physical exercise-induced hyperinsulinemic hypoglycemia is an autosomal-dominant trait characterized by abnormal pyruvate-induced insulin release. Diabetes 2003;52:199–204 [DOI] [PubMed] [Google Scholar]
- 46.Dionne KE, Colton CK, Yarmush ML. Effect of oxygen on isolated pancreatic tissue. ASAIO Trans 1989;35:739–741 [DOI] [PubMed] [Google Scholar]
- 47.Carlsson PO, Jansson L, Palm F. Unaltered oxygen tension in rat pancreatic islets despite dissociation of insulin release and islet blood flow. Acta Physiol Scand 2002;176:275–281 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.