Abstract
HSCs are rare cells that have the unique ability to self-renew and differentiate into cells of all hematopoietic lineages. The lack of donors and current inability to rapidly and efficiently expand HSCs are roadblocks in the development of successful cell therapies. Thus, the challenge of ex vivo human HSC expansion remains a fertile and critically important area of investigation. Here, we show that either SALL4A- or SALL4B-transduced human HSCs obtained from the mobilized peripheral blood are capable of rapid and efficient expansion ex vivo by >10 000-fold for both CD34+/CD38− and CD34+/CD38+ cells in the presence of appropriate cytokines. We found that these cells retained hematopoietic precursor cell immunophenotypes and morphology as well as normal in vitro or vivo potential for differentiation. The SALL4-mediated expansion was associated with enhanced stem cell engraftment and long-term repopulation capacity in vivo. Also, we demonstrated that constitutive expression of SALL4 inhibited granulocytic differentiation and permitted expansion of undifferentiated cells in 32D myeloid progenitors. Furthermore, a TAT-SALL4B fusion rapidly expanded CD34+ cells, and it is thus feasible to translate this study into the clinical setting. Our findings provide a new avenue for investigating mechanisms of stem cell self-renewal and achieving clinically significant expansion of human HSCs.
Introduction
HSCs, rare cells capable of differentiating into blood lineages, have been identified in fetal bone marrow and liver, umbilical cord blood, adult bone marrow, and peripheral blood. HSCs are used in clinical transplantation protocols to treat a variety of diseases, including leukemia, inborn anomalies of the blood and immune system, aplastic anemia, and hemoglobinopathies. However, finding a matching donor is difficult, time-consuming, and especially problematic for African Americans, Hispanics, Native Americans, and people of mixed ethnicity. Currently, only 1 in 3 patients will find a suitable donor; thus, many patients die before finding an appropriate match.
The lack of donors and the current inability to rapidly and efficiently expand HSCs are key roadblocks in the development of successful therapies. Investigators have been trying to expand human HSCs for the last 3 decades but have not succeeded in a clinically meaningful way. Thus, the challenge of ex vivo human HSC expansion remains a fertile and critically important area of investigation. Clinical trials have used cord blood-derived HSCs expanded through Notch ligands;1 stromal coculture;2 and automated, continuous perfusion culture systems, or “bioreactors.”3 Other approaches to expand HSCs include the use of aryl hydrocarbon receptor antagonists4 and transduction of NF-Ya to activate HOXB4 using cell-penetrating peptides such as TAT.5 Of these techniques, the highest expansion efficiencies have been obtained with Notch signaling,1 which produced a 160-fold increase in CD34+ cells compared with controls; however, long-term engraftment was not achieved.1,6 Therefore, identification of additional potent HSC-expanding factors and development of a robust method for expansion of HSCs from the bone marrow or cord blood is urgently needed for bone marrow transplantation and genetic therapy.7
SALL4 is a zinc-finger transcriptional factor and a member of the SALL gene family, originally cloned based on their DNA-sequence homolog to Drosophila homeotic gene, sal.8,9 Recently, our group and others have shown that the embryonic stem (ES) cell factor SALL4 plays a vital role in maintaining ES cell pluripotency and in governing decisions affecting the fate of ES cells through transcriptional modulation and interaction with Oct4 and Nanog.10–16 We have demonstrated that SALL4 is expressed constitutively in human leukemia cell lines and primary acute myeloid leukemia cells.17–21 To our best knowledge, SALL4 may be one of a few genes creating a connection between leukemic stem cells and the self-renewal properties of normal HSCs and ESCs.10,18–20
Here, we demonstrated that SALL4 is a robust stimulator for the expansion of human HSCs. We demonstrated that overexpression of SALL4 isoforms ex vivo leads to a >10 000-fold expansion of human HSCs in the presence of appropriate cytokines. More importantly, these expanded HSC cells were associated with enhanced stem cell repopulation capacity in vivo. Finally, we developed a recombinant TAT-SALL4B protein that is capable of human HSC expansion ex vivo. Our findings may stimulate investigations of the mechanisms of stem cell self-renewal and clinically significant expansion of human HSCs.
Methods
SALL4-lentiviral infection of human CD34+ cells
Normal human bone marrow CD34+ cells were purchased (AllCells) and single-cell suspensions were generated in StemSpan SFEM (StemCell Technologies) for 24 hours. Next, 105 cells/well were placed in a 12-well plate. The cells were cultured in StemSpan SFEM containing 10% FBS and 1% pen/strep (Invitrogen). The media were supplemented with 100 ng/mL FLT-3, 100 ng/mL thrombopoietin (TPO), and 100 ng/mL SCF (ProSpec). SALL4 lentivirus particles at an MOI between 10 and 20 were added to the bone marrow cells at 37°C. For controls, green fluorescent protein (GFP)–only lentivirus particles were added to the bone marrow cells at similar MOIs. The cells were infected overnight for 12-15 hours and then recovered in culture medium.
Expansion of SALL4-transduced human CD34+ cells
The SALL4-transduced CD34+ cells were expanded in 12-well plates under normal culture conditions (StemSpan SFEM containing 10% FBS and 1% pen/strep supplemented with 100 ng/mL FLT-3, 100 ng/mL TPO, and 100 ng/mL SCF). In addition, cells were cultured in more stringent conditions in which recombinant cytokine concentrations were decreased to see whether the SALL4-transduced cells were still capable of surviving and expanding. In one experiment, cells were cultured in media containing 50% less cytokines (50 ng/mL FLT-3, 50 ng/mL TPO, and 50 ng/mL SCF). In an additional trial, cells were cultured in media containing 75% less growth factors (25 ng/mL FLT-3, 25 ng/mL TPO, and 25 ng/mL SCF). Cells were monitored for 2 months and observed with bright field and fluorescent microscopy (microscopes: Olympus CKX41, Olympus BX51TF; objectives: 10×/0.25 Php, 20×/0.40 Php, 40×/0.55 Php, 100×/1.25 Oil; cameras: Olympus DP72, Q-Imaging QlCam Fast.1394; image-acquisition: Q Capture Pro Version 6.0.0.412).
Expansion of SALL4-transduced isolated CD34+ cells from patient samples
Peripheral blood stem cells were obtained from the Blood and Marrow Stem Cell Laboratory at Stony Brook University Medical Center. CD34+ cells were isolated from the stem cell pool using the CD34 human Microbead kit and MiniMACS separation columns (Miltenyi Biotec). After the CD34+ cells were isolated, the cells were transfected with lentivirus and cultured in the same manner as mentioned for SALL4-transduced human CD34+ cells.
CFU assay of bone marrow cells
Tubes of MethoCult (StemCell Technologies) medium were thawed overnight in a 4°C refrigerator. The next morning, SALL4-transduced or GFP-transduced CD34+ cells were prepared at ×10 the final concentration required. Cell suspensions of 1 × 106 cells/mL were prepared and duplicated with different concentrations (2- to 4-fold). Then, 0.3 mL of cells was added to 3 mL of MethoCult medium for duplicate cultures. A 16-gauge blunt-end needle attached to a 3-cc syringe was used to dispense the cells and MethoCult medium into culture dishes. Then, 1.1 mL of cells was dispensed per 35-mm dish. The 2 dishes were placed into a 100-mm petri dish and a third, uncovered 35-mm dish containing 3 mL of sterile water also was added. All 3 dishes were then covered within the 100-mm petri dish. The cells were incubated for 14 to 18 days at 37°C under 5% CO2 and ≥ 95% humidity. The burst-forming unit-erythroid (BFU-E), CFU-GM, and CFU-granulocyte-erythrocyte-macrophage-megakaryocyte (GEMM) colonies were observed with bright field and fluorescent microscopy. CFUs were counted under the microscope 13-18 days after the cells were plated in MethoCult medium. A colony with >100 cells was counted as a positive colony.
Flow cytometry and phenotyping of cells
Flow cytometry was conducted with PE-conjugated antibody to CD34 and CD19; allophycocyanin (APC)–conjugated antibody to CD38, CD15, and CD33; and PerCP-Cy5.5–conjugated antibody to CD45 for cell sorting (BD Biosciences). The presence of human cells in NOD/SCID mouse bone marrow was determined using CD45-PE antibody. Myeloid cells were tracked by CD15-APC or CD33-APC antibody, and lymphoid cells were tracked by CD19-PE or CD3-PE antibody.
Cell counting and growth curve experiments
SALL4A-, SALL4B-transduced, and control cells were counted every 24 hours for 7 days using a hemacytometer. Before counting, cells were gently aspirated with a 200-μL pipette tip to dissociate cell clusters into individual cells.
LTC-IC assays
Long-term culture-initiating cell (LTC-IC) assays using human CD34+ cells were conducted under bulk or limiting dilution conditions in MethoCult media. To calculate the total LTC-IC number, the frequency of LTC-ICs was determined from secondary cultures. This calculated number was then multiplied by the total number of cells present after 4-week primary long-term cultures.
NOD/SCID mice repopulating cell assays
We injected 4 × 103, 8 × 103, 2 × 104, or 4 × 104 2-day-old GFP-only or 14-day-old cultured SALL4A- or SALL4B-expressing human CD34+ bone marrow cells along with 2 × 105 CD34− accessory cells into irradiated (2.5-Gy) NOD-LtSz-scid/scid (NOD/SCID) mice on day 0. Seven weeks after transplantation, peripheral blood samples were analyzed by flow cytometry for the presence of CD45+ cells. Mice were scored as positive for human engraftment when at least 0.5% CD45+ human cells were detected among mouse peripheral blood cells. Stem cell–initiating cell frequency was determined by the reciprocal of the concentration of test cells that gave 37% negative mice. Animal experiments were performed according to the investigator's protocols approved by the Stony Brook University Institutional Animal Care and Use Committee.
Serially transplanted studies
Mice bone marrow cells were harvested from the tibiae and femurs of highly engrafted primary recipient mice 16 weeks after transplantation. After removal of red blood cells by lysis buffer, half of the bone marrow cells from each recipient mouse were transplanted into 1 secondary sublethally irradiated (2.5-Gy) NOD/SCID mouse. Five weeks after transplantation, the percentage of human CD45+ cells in peripheral blood of the secondary recipient mouse was analyzed by flow cytometry as described under “Flow cytometry and phenotyping of cells.” Tertiary transplants were conducted in the same manner, and flow cytometry was conducted on bone marrow cells to analyze CD45+, CD33+, CD19+, and CD3+ cells 10 weeks after transplantation.
Expression and purification of His-TAT-SALL4A/B
For expression of TAT-SALL4 in Escherichia coli, the human SALL4B gene was doubly digested with the restriction endonucleases SalI and NotI and ligated into a pTAT-pET28b vector, a 6xHis-taq protein expression vector bearing the T7 promoter, kanamycin resistance, and pTAT peptide/protein-transducing domain. The plasmids with the correct gene sequences were then transformed into E coli strain BL21 (DE3). The 6xHis-fused human SALL4B was expressed with 0.1 mM isopropyl β-d-thiogalactoside induction for 3 h and then purified using a nickel-nitrilotriacetic acid affinity column (QIAGEN). The purity of the purified His-TAT-SALL4B was further determined based on SDS-PAGE and liquid chromatography-tandem mass spectrometry (LC-MS/MS).
SDS-PAGE and Western blot
Proteins were loaded on 12.5% 1-dimensional SDS-PAGE for protein separation, followed by staining with Coomassie Brilliant Blue R-250 and destained in 10% methanol/7% acetic acid. For Western blot analysis, the SDS-PAGE was transferred to polyvinylidene difluoride membranes (Millipore). After transfer, the membranes were saturated with 5% (wt/vol) nonfat dry milk in TBS/0.1% Tween 20 at 4°C overnight, followed by incubation with the primary antibodies overnight at 4°C. Primary antibodies against human SALL4 and 6xHis-tag were purchased from Abcam. After 3 washes with TBS/0.1% Tween 20, the membranes were incubated with a solution of peroxidase-conjugated secondary antibodies (Abcam). After 1-hour incubation at room temperature, the membranes were washed 3 times with TBS/0.1% Tween 20, and the membrane blots were developed using ECL substrates (Millipore).
In-gel digestion and LC-MS/MS
Gel bands were cut and digested in-gel with trypsin. Peptides were resuspended in 50 μL 0.1% formic acid/2% acetonitrile. Then, 10 μL of the peptide was injected into an Orbitrap mass spectrometer at Stony Brook University Proteomics Center for 1-dimensional LC-MS/MS analysis.
Statistical analysis
Results are reported as means + SD. Values with P < .05 were considered to be statistically significant.
Results
Robust ex vivo expansion of human HSCs by overexpression of SALL4 isoforms
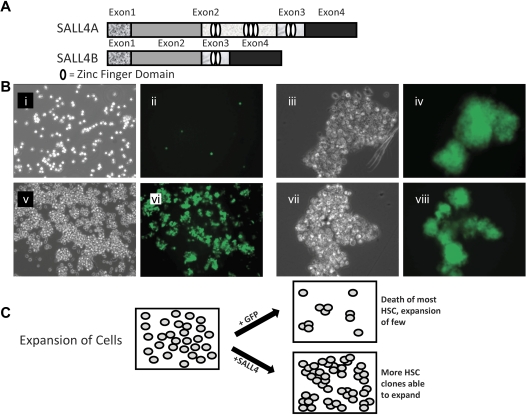
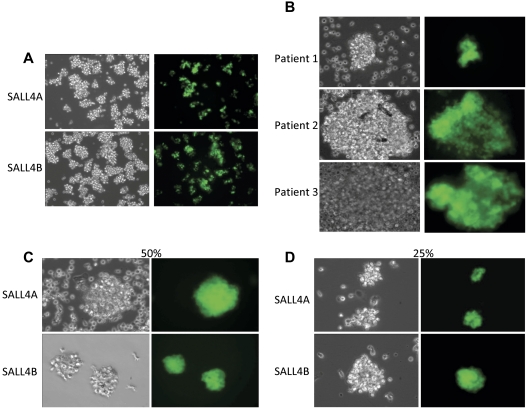
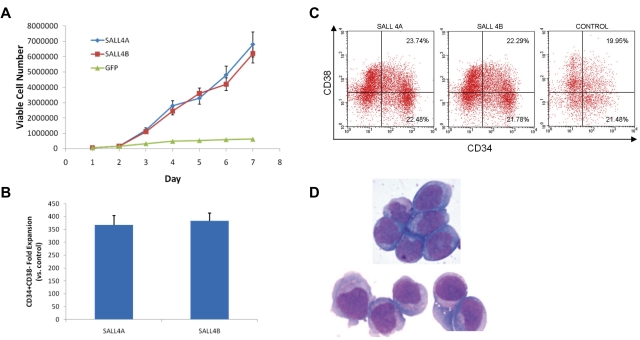
A series of experiments were carried out to determine the effects of SALL4 isoforms on hematopoietic precursors, human bone marrow CD34+ cells. The cells were transduced with lentiviruses expressing either SALL4A or SALL4B cDNAs that were combined with a GFP reporter gene to facilitate isolation and tracking of transduced cells. SALL4A and -B are 2 splicing isoforms generated through internal splicing (Figure 1A). CD34+ cells transduced with either SALL4A or -B and grown in cell culture containing SCF, TPO, and FLT-3L cytokines without stromal support expanded dramatically and regularly formed GFP-positive cell clusters (Figure 1Biii-viii,C). In contrast, control cells transduced with control GFP did not form many large clusters. In these cultures, single cells or few small clusters were visible (Figure 1Bi-ii,C). The efficiency of lentiviral infection was ∼ 40% (supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). After 7 days, the expansion cells seemed to be observed solely in the SALL4-transduced cells (Figure 2A). Similar experiments were carried out using CD34+ cells isolated from mobilized peripheral blood stem cells from patients who were collected, and samples were cryopreserved. Three samples from different patients showed promising results in CD34+ cell expansion (Figure 2B). Cells transduced with SALL4A or SALL4B expanded approximately 130-fold, whereas control cells expanded 12-fold at most (Figure 3A). Furthermore, when these SALL4-transduced cells were grown for 14 days, the CD34+/CD38− population increased 368-fold for SALL4A and 384-fold for SALL4B (Figure 3B) and maintained stem cell immunophenotypes (n = 4; supplemental Figure 2). At 31 days of cell culture, the SALL4-transduced expanding cells still retained progenitor or stem cell immunophenotypes (n = 2; Figure 3C) and primitive cell morphology (Figure 3D). At the end of expansion, approximately 90% of the SALL4-transduced cells were expressing GFP. In contrast, control cells ceased to proliferate or were no longer viable after 2 weeks after infection. SALL4-induced cells continued to expand with CD34+/CD38− or CD34+/CD38+ ratios similar to that of the original cells with which the culture began (Figure 3C). The expansion experiments were repeated at least 12 times using a variety of sources for the CD34+ cells (supplemental Table 1). In all trials, the SALL4A- or SALL4B-transduced human CD34 cells were able to expand in the liquid culture medium for > 2 months and demonstrated similar percentages of cell populations (15% CD34+/CD38−, 17% CD34+/CD38+) to 31-day-old cells demonstrated by flow cytometry.
Figure 1.
SALL4 isoforms and SALL4-transduced HSC expansion. (A) Schematic diagram of the SALL4A and SALL4B isoforms demonstrating the variable number of zinc-finger domains possessed by each. The HSCs were transduced with either the SALL4A or SALL4B gene using a lentiviral transfection system. (B) Bright field and fluorescent images of human bone marrow CD34+ cells transduced with GFP (i-ii, 10×) or representative SALL4 isoform B (v-vi, 10×) 9 days after infection. Initially, 50 000 CD34+/CD38− cells were plated. High magnification of SALL4B-transduced HSC clusters (iii,iv,vii,viii, 40×). The GFP cell clusters signified positive overexpression of SALL4B. With SALL4B overexpression, HSC cell clusters are able to survive and are rapidly expanding at 9 days after infection. (C) Model of SALL4-mediated ex vivo HSC expansion. The primary culture was divided and transduced with a SALL4 or GFP control. The viable HSCs without SALL4 overexpression decreased in number because of differentiation or death leading to a net HSC decline. In contrast, HSCs in which SALL4 was overexpressed, many clones were able to survive and expand in the culture. A net HSC expansion was exhibited with numerous expanding clusters throughout the culture.
Figure 2.
Expansion of SALL4-transduced HSCs. (A) HSCs transduced with SALL4A and SALL4B are able to survive and expand rapidly 7 days after lentiviral infection (10×). (B) CD34+ cells isolated from peripheral blood stem cells of 3 different patients. CD34+ cells were isolated from the stem cell pool using magnetic anti-CD34+ human microbeads. The CD34+-enriched cells were transduced with SALL4A and imaged under bright field and fluorescent microscopy (40×). All 3 samples from the various patients were successfully transduced with SALL4A and expanded rapidly in culture. In addition, SALL4-induced HSCs are able to expand when growth factor concentrations are decreased. When SALL4-transduced HSCs were cultured in growth media containing 50% less cytokines (C), they were still able to survive and expand 6 days after lentiviral infection (40×). Furthermore, when growth factor concentrations were decreased to 25% of original values (D), the SALL4-transduced HSCs continued to proliferate (40×). In contrast, control cells had undergone cell death by day 6.
Figure 3.
Characterization of SALL4-transduced HSC cells. (A) Growth curves of CD34+ cells transduced with SALL4A, SALL4B, or GFP and cultured in media containing 75% less cytokines. After transduction, 50 000 cells of each group were cultured in stringent conditions in which normal cytokine concentrations were decreased by 75%. HSCs transduced with SALL4A or SALL4B continued to survive and expand over 7 days, whereas control cells growth halted at day 5. (B) Fold expansion of CD34+/CD38− cells 14 days after infection of lenti-SALL4A or -SALL4B versus control. Cells transduced with SALL4A demonstrated a 368-fold increase of CD34+/CD38− cells over control, whereas those transduced with SALL4B showed a 384-fold increase. Values are means + SD. (C) Phenotypic analysis of SALL4-induced hematopoietic stem cells 31 days after lentiviral infection. Human-specific antibodies CD34-PE and CD38-APC were used to compare SALL4-transduced HSCs versus 3-day control cells. Thirty-one days after lentiviral infection, the aged SALL4-induced cells continued to demonstrate similar phenotypic ratios compared with control cells for CD34+/C38−. FLOW analysis was carried out on 3 separate samples. Therefore, many of these aged cells still attained progenitor characteristics and had the ability to differentiate into various cells lines. (D) Thirty-one-day old SALL4-induced HSCs attain blastlike morphology. Aged 31-day-old SALL4-induced HSCs were Wright-Giemsa–stained. Many cells showed blastlike morphology including large nuclei and scant cytoplasm (100×). These cells represented a population of undifferentiated cells still visible 31 days after SALL4-lentiviral infection and expansion.
The expansion of SALL4-transduced cells occurred quite rapidly, with significant CD34+ cell growth in just a few days. To see whether SALL4-induced clusters could serve as a seed for further expansion, single-cell clusters were picked from the parental plate and placed into individual wells of a 48-well plate. The cluster was gently dissociated by repeated aspiration with a pipette tip. The next day, new small GFP-positive cell clusters began to form again (supplemental Figure 3). These clusters were allowed to expand for 5 days and began to grow at a similar rate as that seen in the initial parental plates.
To determine whether SALL4 was able to expand CD34+ cells under stringent conditions, we conducted cell cultures in which recombinant growth factor concentrations were decreased to 50 or 25% of cytokines for both SALL4-induced HSCs and control cells. Cell growth was monitored during the first week. Interestingly, cells transduced with SALL4A or SALL4B were still able to survive and expand beyond 7 days of culture (Figures 2C-D,3A). In contrast, control cells had ceased to proliferate and expansion slowed by day 7 (Figure 3A). In other experiments, SALL4-transduced cells were grown in culture with various combinations of the growth factors SCF, TPO, and FLT-3L, which have been reported to be present at low levels in the marrow niche. After 14 days of culture, it was noted that the SALL4-transduced cells growth and survival were independent of FLT-3L, partially dependent of TPO, and dependent of SCF (supplemental Table 2). This finding is potentially important because the niche in the normal marrow does not bear excessive special cytokines and is delicately regulated, unlike the high artificial levels in our cell cultures. This study may indicate that SALL4 works in conjunction with the available cytokine levels in its environment to regulate cell expansion.
Blocking differentiation and permitting proliferation by SALL4 isoforms
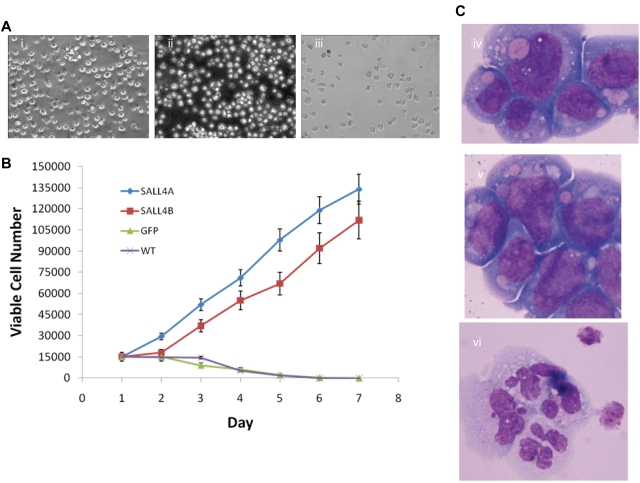
To investigate the mechanism by which SALL4 effects stem cell growth and identity, we extended our studies to the myeloid progenitor cell line 32D. SALL4-transformed 32D cells grew indefinitely in culture when IL-3 was removed and replaced with G-CSF (Figure 4Ai-ii). In contrast, control 32D cells died after 5 to 6 days in culture with G-CSF in the absence of IL-3 (Figure 4Aiii). In addition, 32D cells transformed with SALL4A or SALL4B proliferated at a 3- to 6-fold higher rate than the control counterparts in G-CSF culture without IL-3 (Figure 4B). 32D cells proliferate as undifferentiated blasts when maintained in IL-3 (Figure 4Civ), but they differentiate into mature neutrophilic granulocytes when stimulated with G-CSF (Figure 4Cvi). SALL4-transduced 32D cells grew indefinitely without IL-3, and they retained undifferentiated blast morphology when given G-CSF (Figure 4Cv). This study indicates that constitutive expression of SALL4 inhibits differentiation and permits expansion of undifferentiated cells. These findings are consistent with that of other known HSC expanding factors such as HOXB4 and Notch ligand.
Figure 4.
Overexpression of SALL4-immortalized 32D cells and blocks G-CSF–dependent differentiation. (A) SALL4-induced expansion of 32D cells proliferate after the removal of IL-3 and addition of G-CSF. Three days after the removal of IL-3 and addition of G-CSF to the growth media of the cells, the SALL4A- and SALL4B-induced cells continue to expand, whereas the GFP-induced cells exhibit a decrease in cell number. At 7 days, the SALL4A- (i, 20×) and SALL4B-induced (ii, 20×) cells continue to proliferate, whereas the control cells (iii, 20×) have undergone cell death. (B) Growth curves of SALL4-induced 32D cells cultured only with G-CSF. 32D cells were transduced with SALL4A, SALL4B, or GFP lentivirus and then cultured for 3 days in growth media containing IL-3. On the day 4, 15 000 cells were aliquoted from each group and placed in new growth media with G-CSF and without IL-3. Cell growth was monitored daily, and the viable number of cells in each group was recorded. In cells that were transduced with SALL4A, an 8-fold increase in the number of cells was observed from day 1 to day 7. Cells that were transduced with SALL4B exhibited a 7-fold expansion of cells. In contrast, cells that were only transduced with GFP and wild type (WT, no lentiviral infection) demonstrated a decrease in the number of cells over the same period with almost all the cells undergoing cell death by day 5. (C) Wright-Giemsa staining of 32D cells (100×). Morphology of 32D cells with IL-3 alone (iv), transduced with SALL4A with G-CSF (v), and with G-CSF alone (vi). Cells given IL-3 or transduced with SALL4A continue to demonstrate blastlike morphology (iv and v), whereas the cells not transduced with SALL4A and given G-CSF exhibit neutrophil morphology (vi).
Functional analysis of SALL4-induced CD34+ cells in vitro
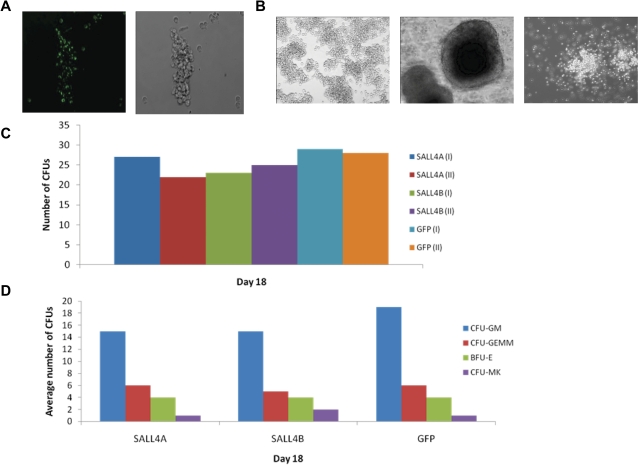
To further study the proliferation potential of the SALL4-transduced HSCs without an excess of special cytokines, CFU assays were conducted. The CFU progenitors used for the study were initially GFP-positive, signifying the expression of SALL4 protein (Figure 5A) and were cultured for at least 1 month in media containing the cytokines SCF, TPO, and FLT-3L. The CFU assays, conducted in MethoCult media without these cytokines, revealed that these cells could form various colonies, including CFU-GM, CFU-GEMM, and BFU-E (Figure 5B). In addition, the 1-month-old transduced cells had the capability to form similar numbers of CFU colonies compared with 2-day-old GFP-transduced control cells when counted 18 days after the CFU assay was initialized (Figure 5C-D). This study indicates that SALL4 works in conjunction with other cytokines to block HSC differentiation; therefore, other cytokines are needed to inhibit differentiation.
Figure 5.
CFU assays of SALL4-transduced cells. (A) CFU progenitors are GFP-positive. HSC cells selected for CFU assays were GFP-positive, which verified that the cells were successfully transduced and overexpressing SALL4 (10×). (B) Various CFU colonies are able to differentiate from SALL4-induced HSCs. Aged SALL4-induced HSCs were plated in MethoCult and observed for CFU colonies. Numerous lineages were observed in CFU assays using the SALL4-induced HSCs, including CFU-GEMM, BFU-E, and CFU-GM colonies (10×). These data demonstrated that the aged HSCs transduced with SALL4 were capable of differentiating into different blood cell lineages. (C) Number of CFU colonies formed from SALL4-induced hematopoietic stem cells. The number of CFU colonies was counted 13-18 days after SALL4-induced or GFP-induced cells were cultured in CFU MethoCult media. The representative data from day 18 are shown. (D) Types of CFU colonies formed at day 18.
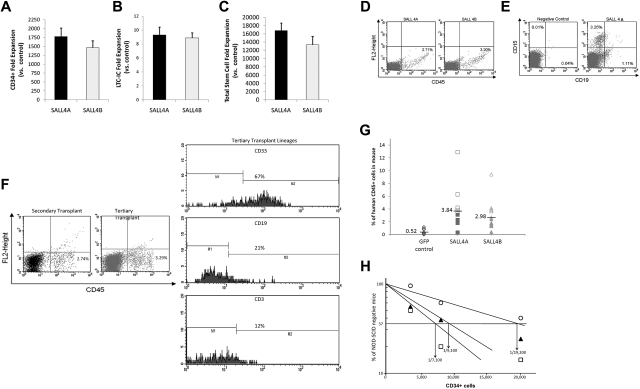
To further test whether the transfer of SALL4 can lead to the long-term expansion of human hematopoietic progenitors, we cultured SALL4A- and SALL4B-transduced cells for 1 month. After 31 days of culture, there was a 1780-fold increase for SALL4A and 1463-fold increase for SALL4B in total CD34+ cells numbers compared with controls (Figure 6A; control cells ceased to expand after 10-12 days). Furthermore, SALL4-transduced cells showed 9.32-fold increases for SALL4A and 8.88-fold increases for SALL4B versus controls for the total number of LTC-ICs after 1 month (Figure 6B). Overall, SALL4A-transduced cells had a total fold CD34+/CD38− stem cell expansion of 16 700 over control, whereas SALL4B-transduced cells showed a 13 300-fold increase, although there was not a statistically significant difference between the 2 SALL4 isoforms (Figure 6C).
Figure 6.
Relative amplification of total stem cells and human cell engraftment into NOD/SCID mice. (A) After 1 month of cell culture, CD34+ cells transduced with SALL4A or SALL4B had 1780- and 1463-fold increases, respectively, relative to control cells. Values are means + SD. (B) Furthermore, SALL4-transduced cells showed 9.32-fold increases for SALL4A and 8.88-fold increases for SALL4B versus controls for the total number of LTC-ICs after 1 month. Values are means + SD. (C) Overall, SALL4A-transduced cells had a total fold CD34+/CD38− stem cell expansion 16 776 over control, whereas SALL4B-transduced cells showed 13 320-fold increases. Values are means + SD. (D) Representative flow cytometry analysis 4 weeks after injection for CD45+ human leukocytes from peripheral blood of NOD/SCID recipients transplanted with SALL4A- or SALL4B-transduced HSCs. (E) Representative flow cytometry profile 4 weeks after injection of a mouse exhibiting multilineage repopulation of human cells by engrafted cells. Although the negative control animal showed no engraftment of human cells, the experimental animal showed both CD15+ myeloid and CD19+ lymphoid human cell engraftment. (F) Flow analysis of secondary and tertiary bone marrow transplant NOD/SCID mice. The animals were positive for CD45+ cells in both secondary (2.74%) and tertiary (3.29%) transplants (left panel). The threshold bars for positive and negative populations were set according to independent staining controls. When the CD45+ population in the tertiary transplant was analyzed further for specific lineages, CD33 myeloid and CD19/CD3 lymphoid cells were positively measured (right panel). (G) Amount of human engraftment in the peripheral blood of NOD/SCID mice transplanted with 20 000 (SALL4A, ■; SALL4B, ▴, or GFP, ♦) or 40 000 (SALL4A, □; SALL4B, ▵; or GFP, ◊) initial human CD34+ cells. (H) Limiting dilution analysis of CD34+ bone marrow cells injected into NOD/SCID mice (n = 72) after lentiviral transfection with SALL4A (□), SALL4B (▴), or GFP (○).
Expansion was associated with enhanced SCR capacity in vivo
To test whether SALL4-mediated cells are able to override the regulatory machinery in the marrow niche to control their differentiation, repopulation capacity, and stem cell output, we performed xenotransplantation assays. Phenotypic analysis revealed that both SALL4A- (n = 9) and SALL4B-transduced (n = 10) cells were capable of positive cell engraftment into NOD/SCID mice (Figure 6D) 4 weeks after injection. In addition, experimental animals (n = 8) demonstrated myeloid (CD15+) and lymphoid (CD19+) lineages 4 weeks after injection (Figure 6E), indicative of a myeloid/lymphoid differentiation process. Bone marrow from animals (n = 4) 15 weeks after injection was analyzed using flow cytometry and still exhibited 3 lineage differentiation, including CD3+/CD19+ lymphoid, CD15+ myeloid, and Glycophorin-4A+ erythroid lineages (supplemental Figure 4).
To further determine whether SALL4-induced cells bear a long-term engraftable property, secondary and tertiary transplantations were conducted. The secondary and tertiary animals were injected with bone marrow cells that had ∼ 1000 human CD34+ cells based on flow analysis. Flow analysis demonstrated that 8 weeks after injection, animal marrows still had CD45+ cells (2.74%; Figure 6F) but did not attain significant CD34+ cells (< 0.24%), showing no leukemic effect. In addition, the CD45+ population were positive for CD3+ (16.5%)/CD19+ (4.9%) lymphoid, and CD15+ (78.5%) myeloid. An additional subpopulation in the marrow showed Glycophorin-4A+ (5.6%) lineages. This showed that successful bone marrow transplantation from 1 animal to another was possible and that cells could differentiate properly in the bone marrow niche. In addition, we conducted tertiary transplantations with bone marrow harvested from secondary transplant animals. Interestingly, 10 weeks after transplantation, the animals still exhibited CD45+ (3.29%) cells within their bone marrow (Figure 6F). Further analysis of the CD45+ population demonstrated the cells were positive for myeloid and lymphoid lineages similar to that of the second transplant (Figure 6F). These percentages are similar to the range seen in previous studies.1,22
After 4 weeks of culture, the ability of SALL4A or SALL4B to increase the repopulating capacity of human cells into NOD/SCID was demonstrated by a strong enhancement in the level of engraftment in mice transplanted with SALL4A- or SALL4B-transduced cells. The proportion of CD45+ cells with SALL4A was 3.84% (0.34-12.89%; n = 12) and with SALL4B was 2.98% (0.41-9.41%; n = 12) compared with 0.52% (0.03-1.18%; n = 12) for control cells (Figure 6G). Furthermore, long-term engraftment with SALL4A- or SALL4B-transduced cells was also evident by detecting human CD45+ cells in the mouse peripheral blood at 14 weeks (n = 6) or 19 weeks (n = 3).
To quantitatively measure the effects of SALL4-transduced CD34+ cells, we conducted limiting-dilution experiments to determine the NOD/SCID repopulating assay (SRC) frequency. At 7 weeks after injection, the SRC frequency increased from 1 in 19 200 CD34+ cells (range defined by ± SE, 14 100-25 900) to 1 in 7100 (range defined by ± SE, 5200-9800) for SALL4A and 1 in 9100 (range defined by ± SE: 6 700-12 400) for SALL4B. The total SRC content was expanded by 1080-fold for SALL4A and 844-fold for SALL4B (n = 72) compared with uncultured cells by taking into account the increase in the SRC frequencies and total cell numbers (Figure 6H). Our results suggest that overexpression of SALL4 solely in HSCs does not override the regulatory mechanisms involved in the control of stem cell output in vivo.
TAT-SALL4B protein induced CD34+ cell expansion
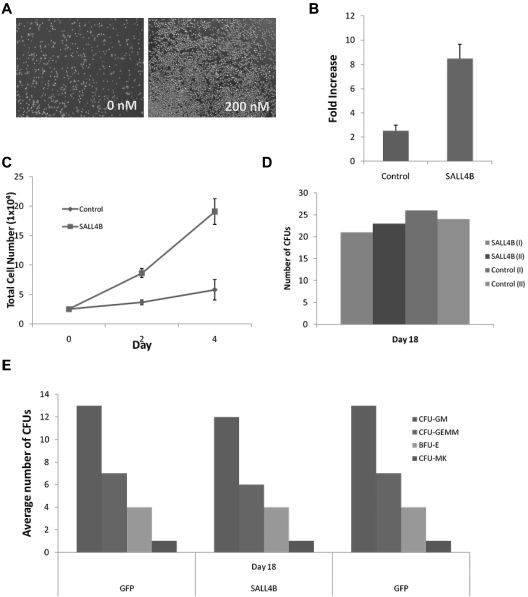
Lentiviral expression of SALL4 is very efficient, but its clinical application is not ideal because of difficulties in controlling the level and duration of expression of the transgene in vivo as well as the potential for insertional leukemogenesis. Protein transduction using cell-penetrating peptides can overcome the limitations of lentiviral vectors and has been widely used recently.5,23,24 To use a different approach to demonstrate the role of SALL4 in HSC expansion, we produced TAT-6xHis-SALL4B protein expressed in E coli, and we purified the protein using nickel-nitrilotriacetic acid agarose (supplemental Figure 5A-B). The recombinant protein of TAT-SALL4B was confirmed by a Western blot with an anti-6xHis tag mouse monoclonal antibody and mass spectrometry analysis (supplemental Figure 5C-D). We focused on SALL4B because it is a shorter form and expressed a high level of protein in E coli. After 3 days of TAT-SALL4B treatment, the CD34+ cells expanded rapidly (Figure 7A). Human CD34+ cells cultured 3 to 4 days with SALL4 fusion protein (200 nM) along with TPO, SCF, and FLT-3L showed > 10- and 8-fold increases of total mononuclear cells and CD34+ cells, respectively (Figure 7B-C). TAT-SALL4B protein (200 nM) was added twice a day and seemed to be sufficient to expand the cells. In addition, CFU assays demonstrated that these cells could form various colonies including CFU-GM, CFU-GEMM, and BFU-E (Figure 7D-E). By taking the fold increase of the TAT-SALL4B–treated cells versus control times the CFU numbers, it was noted that the overall CFU number increased by approximately 10-fold.
Figure 7.
Human bone marrow CD34+ cells expand at a higher rate when treated with TAT-SALL4B protein. (A) Bright field images of CD34+ cells after 3 days of protein treatment (10×). (B) Fold increase and total cell number (C) of TAT-SALL4B–treated bone marrow cells versus control cells treated solely with BSA. (D) Number of CFU colonies formed from hematopoietic stem cells treated with TAT-SALL4B protein compared with unmanipulated CD34+ cells. (E) Types of CFU colonies formed after treatment with TAT-SALL4B protein.
Discussion
We have developed a new therapeutic strategy using cytokine-dependent SALL4 technology for the dramatic 10 000- to 15 000-fold ex vivo expansion of human HSCs without significant differentiation over 4 weeks. In addition, even after 8 weeks of cell culture, 37% of the CD34+ cells were still CD34+/CD38− (data not shown). In xenotransplantation models, the stem cell frequency of cells that had been induced by SALL4 for 4 weeks ex vivo was only 2- to 2.5-fold higher than fresh CD34+ cells (Figure 6H). This is consistent with our own results showing that SALL4 expansion is cytokine-dependent (supplemental Table 2), although the total SRC content was quite high (expanded by 1080-fold for SALL4A and 844-fold for SALL4B). Thus, the in vivo growth of SALL4-induced HSCs might be dissimilar to those in cell culture conditions (ex vivo) where an excess of special cytokines is present. In vivo growth is dependent on the tissue niche (supplemental Table 2 and Figure 6) consistent with the findings of other HSC expanding factors, HOXB4, and Notch signaling.
Like other approaches used for HSC expansion, the SALL4-transduced cells growth and survival were dependent of special cytokines. The magnitude of HSC expansion is unprecedentedly high at 10 000-fold for CD34+/CD38− and CD34+/ CD38+ populations, and seems to be far superior to existing approaches such as Notch ligands that demonstrated only 160-fold increases for the CD34+ population. In addition, the expansion of engraftable long-term HSCs by our SALL4 approach seemed to be achievable and was supported by evidence of the secondary and tertiary transplantation studies. In contrast, the 2 main approaches for HSC expansion using HOXb4 and Notch ligands do not.1,6
Our current findings may be extremely valuable for clinical therapy. Massive ex vivo expansion of CD34+ cells can be achieved without differentiation. These expanded cells retain long-term engraftment properties similar to those of unmanipulated cells in vivo. The SALL4-expanded cells sustained a long-term engraftment demonstrated by serial xenotransplant models and repopulation assays. Most critically, after transplantation they do not override the niche-induced regulatory controls, allowing these expanded stem cells to avoid leukemic formation. In our studies, no evidence of leukemia was evident in transplanted mice in either serially xenotransplanted animals or by >10 months after transplantation (n = 12), although additional long-term follow up is needed (data not shown). In addition, no leukemic formation was exhibited for 12 months after syngeneic transplantation when either SALL4A or -B was expressed and introduced into mouse stem/progenitor hematopoietic cells (n = 6). However, in our previous transgenic mouse studies of SALL4B overexpression controlled by a universal promoter, CMV, there was an increased incidence of leukemic formation in older mice.17 Yet, this phenotype was not seen with SALL4A overexpression (n = 170; 5 transgenic mouse lines; data not shown) after 2 years of observation. This could be because of an abnormal niche resulting from dysregulated SALL4B expression with this universal CMV promoter and/or SALL4B may bear oncogenic potential. A further study should be conducted to explore this possibility. As an interesting note, Notch signaling has been used for expansion of HSCs, and the expanded cells have been used in human clinical trials. Notch signaling also is involved in a variety of cancer stem cells because both cancer and adult stem cells share functional properties in self-renewal. Nevertheless, we have developed a safer approach: a TAT-SALL4 protein to avoid constitutive expression of SALL4. CD34+ cells exposed to this TAT-SALL4 protein were able to rapidly expand ex vivo. In the future, it will be important to optimize TAT-fusion protein technology, enabling maximal cell expansion. This may open a new avenue to study the mechanisms of stem cell self-renewal and achieve clinically significant expansion of human HSCs for therapy.
Supplementary Material
Acknowledgments
This work is supported in part by a grant from the National Institutes of Health (NIH R01HL087948; Y.M.) and an NIH research fellowship for postdoctoral training (NIH 7R01HL087948-04; J.A.).
Footnotes
The online version of the article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: J.R.A., W.L., and Y.M. designed the research; J.R.A. and W.L. performed the research; J.R.A., W.L., J.Y., N.H., L.S., and Y.M. analyzed data; and J.R.A., W.L., J.Y., C.A., N.H., L.S., and Y.M. wrote the paper.
Conflict-of-interest disclosure: Y.M. is a scientific consultant to MarrowSource Therapeutics International LLC. The remaining authors declare no competing financial interests.
Correspondence: Y. Ma, Department of Pathology, SUNY at Stony Brook, Stony Brook, NY 11794; e-mail: yupo.ma@stonybrook.edu.
References
- 1.Delaney C, Heimfeld S, Brashem-Stein C, Voorhies H, Manger RL, Bernstein ID. Notch-mediated expansion of human cord blood progenitor cells capable of rapid myeloid reconstitution. Nat Med. 2010;16(2):232–236. doi: 10.1038/nm.2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kelly SS, Sola CB, de Lima M, Shpall E. Ex vivo expansion of cord blood. Bone Marrow Transplant. 2009;44(10):673–681. doi: 10.1038/bmt.2009.284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Astori G, Adami V, Mambrini G, et al. Evaluation of ex vivo expansion and engraftment in NOD-SCID mice of umbilical cord blood CD34+ cells using the DIDECO “Pluricell System”. Bone Marrow Transplant. 2005;35(11):1101–1106. doi: 10.1038/sj.bmt.1704964. [DOI] [PubMed] [Google Scholar]
- 4.Boitano AE, Wang J, Romeo R, et al. Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science. 2010;329(5997):1345–1348. doi: 10.1126/science.1191536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Domashenko AD, Danet-Desnoyers G, Aron A, Carroll MP, Emerson SG. TAT-mediated transduction of NF-Ya peptide induces the ex vivo proliferation and engraftment potential of human hematopoietic progenitor cells. Blood. 2010;116(15):2676–83. doi: 10.1182/blood-2010-03-273441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang XB, Beard BC, Beebe K, Storer B, Humphries RK, Kiem HP. Differential effects of HOXB4 on nonhuman primate short- and long-term repopulating cells. PLoS Med. 2006;3(5):e173. doi: 10.1371/journal.pmed.0030173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McNiece I. Ex vivo expansion of hematopoietic cells. Part II: control of proliferation and differentiation of hematopoietic stem cells. Exp Hematol. 2004;32(8):692. doi: 10.1016/j.exphem.2004.05.025. [DOI] [PubMed] [Google Scholar]
- 8.Kohlhase J, Altmann M, Archangelo L, Dixkens C, Engel W. Genomic cloning, chromosomal mapping, and expression analysis of msal-2. Mamm Genome. 2000;11(1):64–68. doi: 10.1007/s003350010012. [DOI] [PubMed] [Google Scholar]
- 9.Kohlhase J, Schubert L, Liebers M, et al. Mutations at the SALL4 locus on chromosome 20 result in a range of clinically overlapping phenotypes, including Okihiro syndrome, Holt-Oram syndrome, acro-renal-ocular syndrome, and patients previously reported to represent thalidomide embryopathy. J Med Genet. 2003;40(7):473–478. doi: 10.1136/jmg.40.7.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yang J, Chai L, Fowles TC, et al. Genome-wide analysis reveals Sall4 to be a major regulator of pluripotency in murine-embryonic stem cells. Proc Natl Acad Sci U S A. 2008;105(50):19756–61. doi: 10.1073/pnas.0809321105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu Q, Chen X, Zhang J, et al. Sall4 interacts with Nanog and co-occupies Nanog genomic sites in embryonic stem cells. J Biol Chem. 2006;281(34):24090–24094. doi: 10.1074/jbc.C600122200. [DOI] [PubMed] [Google Scholar]
- 12.Warren M, Wang W, Spiden S, et al. A Sall4 mutant mouse model useful for studying the role of Sall4 in early embryonic development and organogenesis. Genesis. 2007;45(1):51–58. doi: 10.1002/dvg.20264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xu D, Alipio Z, Fink LM, et al. Phenotypic correction of murine hemophilia A using an iPS cell-based therapy. Proc Natl Acad Sci U S A. 2009;106(3):808–813. doi: 10.1073/pnas.0812090106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang J, Tam WL, Tong GQ, et al. Sall4 modulates embryonic stem cell pluripotency and early embryonic development by the transcriptional regulation of Pou5f1. Nat Cell Biol. 2006;8(10):1114–1123. doi: 10.1038/ncb1481. [DOI] [PubMed] [Google Scholar]
- 15.Lim CY, Tam WL, Zhang J, et al. Sall4 regulates distinct transcription circuitries in different blastocyst-derived stem cell lineages. Cell Stem Cell. 2008;3(5):543–554. doi: 10.1016/j.stem.2008.08.004. [DOI] [PubMed] [Google Scholar]
- 16.Yang J, Gao C, Chai L, Ma Y. A novel SALL4/OCT4 transcriptional feedback network for pluripotency of embryonic stem cells. PLoS ONE. 2010;5(5):e10766. doi: 10.1371/journal.pone.0010766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ma Y, Cui W, Yang J, et al. SALL4, a novel oncogene, is constitutively expressed in human acute myeloid leukemia (AML) and induces AML in transgenic mice. Blood. 2006;108(8):2726–2735. doi: 10.1182/blood-2006-02-001594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yang J, Chai L, Gao C, et al. SALL4 is a key regulator of survival and apoptosis in human leukemic cells. Blood. 2008;112(3):805–13. doi: 10.1182/blood-2007-11-126326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang J, Chai L, Liu F, et al. Bmi-1 is a target gene for SALL4 in hematopoietic and leukemic cells. Proc Natl Acad Sci U S A. 2007;104(25):10494–10499. doi: 10.1073/pnas.0704001104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lu J, Jeong H, Kong N, et al. Stem cell factor SALL4 represses the transcriptions of PTEN and SALL1 through an epigenetic repressor complex. PLoS ONE. 2009;4(5):e5577. doi: 10.1371/journal.pone.0005577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang P, Sun H, Liu YF, Wang GY, Yin YF. [Expression of SALL4 and BMI-1 mRNA in acute leukemia]. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2008;16(6):1271–1274. [PubMed] [Google Scholar]
- 22.Delaney C, Varnum-Finney B, Aoyama K, Brashem-Stein C, Bernstein ID. Dose-dependent effects of the Notch ligand Delta1 on ex vivo differentiation and in vivo marrow repopulating ability of cord blood cells. Blood. 2005;106(8):2693–2699. doi: 10.1182/blood-2005-03-1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krosl J, Austin P, Beslu N, Kroon E, Humphries RK, Sauvageau G. In vitro expansion of hematopoietic stem cells by recombinant TAT-HOXB4 protein. Nat Med. 2003;9(11):1428–1432. doi: 10.1038/nm951. [DOI] [PubMed] [Google Scholar]
- 24.Varnum-Finney B, Xu L, Brashem-Stein C, et al. Pluripotent, cytokine-dependent, hematopoietic stem cells are immortalized by constitutive Notch1 signaling. Nat Med. 2000;6(11):1278–1281. doi: 10.1038/81390. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.