Abstract
Objective
To examine the efficacy and durability of a behavioral therapy (BT) protocol for pediatric TTM compared to a minimal attention control (MAC) condition. It was hypothesized that the BT condition would be superior to MAC at the end of acute treatment, and would also demonstrate durability of gains through the maintenance treatment phase.
Method
A randomized controlled trial in which twenty-four youths were assigned to either a pilot-tested BT protocol, consisting of eight weekly sessions, or to MAC, consisting of three sessions and five phone calls over eight weeks. Independent evaluators assessed outcome at pre-treatment (week 0) and post-treatment (week 8) for BT and MAC, and again at week 16 for BT patients only. The primary outcome measure was the NIMH Trichotillomania Severity Scale (NIMH-TSS).
Results
The BT condition’s week 8 mean NIMH-TSS score was significantly lower than that of the MAC condition. The BT condition’s mean week 8 score was also significantly lower than their own mean week 0 score, whereas no such reductions were observed for the MAC condition. Upon completion of acute treatment at week 8, the BT group’s gains were maintained through an 8-week maintenance treatment phase.
Conclusions
BT produced superior outcome compared to a condition that controlled for participation in a pediatric TTM research study, non-specific therapist contact effects, repeated assessments, and the passage of time. Maintenance of gains following acute BT provides preliminary support for the durability of treatment gains.
Keywords: Trichotillomania, Behavior Therapy, TTM
Trichotillomania (TTM), classified as an impulse-control disorder in DSM IV TR,1 involves repetitive hair pulling that results in significant hair loss. In adults, TTM has been associated with significant functional impairment,2,3 psychiatric comorbidity3,4,5,6 and, in some cases, medical complications such as skin irritation, infections and repetitive use injuries to the hands.7 The disorder also appears to take a significant toll on affected children and adolescents in terms of associated impairment and comorbidity8, but small sample sizes and other methodological shortcomings of studies in the extant literature highlight the need for replication and extension. Individuals who ingest the hair are also at risk for gastrointestinal complications stemming from trichobezoars (i.e., hairballs) if the individual ingests the hair after pulling.8,9 TTM may also be more common than previously believed:2,10,11,12 hair pulling and associated distress has been found in 1% – 3% of college samples,13,14 which further underscores the need for early identification and treatment of this often disabling condition.
With the exception of recently published randomized controlled studies supporting the efficacy of olanzapine15 and N-acetylcysteine,16 pharmacotherapy for TTM in adults has generally not proven efficacious.17,18 Behavior therapy (BT) and cognitive behavioral therapy (CBT) protocols have been found efficacious in randomized controlled trials in the acute treatment of adult TTM,8, 19, 20 yet relapse appears to be common following treatment discontinuation.21, 22, 23 Keuthen and colleagues24 found that while pulling symptoms did not significantly decrease from post-treatment to follow-up in a sample of adults with TTM, self-esteem declined and there was a downward trend among psychosocial impact scores. Moreover, despite the fact that clinical and survey studies converge to indicate that pediatric onset is the norm, there are no published randomized controlled trials (RCTs) of any pharmacotherapy or psychotherapy regimens for TTM in youth to guide clinical practice.
The primary purpose of the present study was to demonstrate the feasibility of conducting a RCT of BT for pediatric TTM and to develop patient recruitment, assessment, and treatment strategies toward the end of conducting a larger future study to examine the efficacy of BT more fully. The design involved randomization to: 1) a weekly BT package for developed for pediatric TTM; or 2) minimal attention control (MAC). The control group was selected to allow for estimation of the non-specific effects of participation in a research study for pediatric TTM, minimal contact with a therapist, repeated assessments and the passage of time. For ethical reasons, MAC participants were discontinued from the study at week 8 and offered open treatment by the study team; all patients randomized to BT participated in a maintenance treatment phase (4 visits over 8 weeks) that allowed for examination of the durability of gains following participation in acute BT. Based on our review of the adult literature on patient response to behavioral treatment for TTM, we generated an exploratory hypothesis that CBT, but not MAC, would yield statistically significant reductions in hair pulling symptoms at the end of the acute treatment phase (week 8), and that gains made during acute BT would be maintained through the maintenance treatment phase (week 16).
Method
Participants
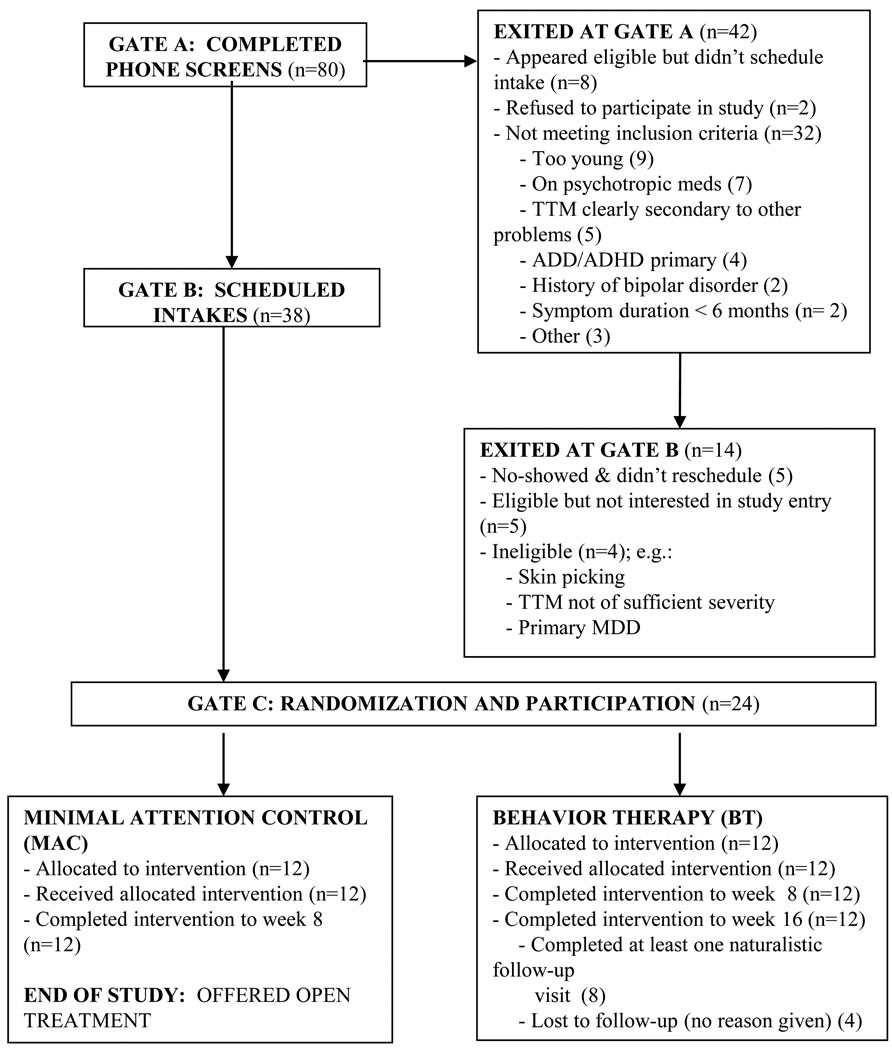
Participants were recruited into a randomized controlled trial examining the efficacy of behavior therapy for pediatric TTM that was being conducted at the University of Pennsylvania School of Medicine. Twenty-four children and adolescents with TTM participated in the present study, with 12 participants assigned to each condition. Inclusion criteria were age 7–17 (inclusive), primary diagnosis of TTM, minimum IQ of 80 and minimum symptom duration of 6 months. Exclusion criteria were a primary diagnosis other than TTM, current bipolar illness, developmental disorder, thought disorder; current psychotherapy or current psychotropic medication. Provided that TTM was determined by the evaluator to be the primary concern, comorbid conditions were allowed (see Table 1 for sample demographic and clinical characteristics). As has been the case in other recent investigations of TTM in youth,4 criteria B and C (increasing and decreasing tension) of the DSM-IV-TR criteria for TTM were optional for inclusion, as these criteria have been found to exclude patients with clinically significant hair pulling.21,25,26 All 24 patients completed the acute phase (week 8) of treatment, and all 12 patients randomized to BT completed the 8-week maintenance phase. Because all MAC patients were offered open BT at week 8 on ethical grounds (withholding of treatment), no maintenance data on MAC are available. Patient flow from initial telephone screening through study completion is depicted in Figure 1.
Table 1.
Demographic and Clinical Information
| Demographic Characteristics | |||
|---|---|---|---|
| BT Group (12) | MAC Group (12) | Total Sample (24) | |
| Mean Age | 12.2 (3.2) | 12.8 (2.2) | 12.5 (2.7) |
| % of Sample < 10 years old | 3 (25%) | 1 (8.3%) | 4 (16.7%) |
| Mean Age of Onset of Pulling Symptoms | 7.7 (3.7) | 10.0 (2.2) | 8.8 (3.2) |
| Gender | |||
| Female | 9 (75%) | 7 (58.3%) | 16 (67%) |
| Male | 3 (25%) | 5 (41.7%) | 8 (33%) |
| Ethnicity | |||
| Caucasian | 9 (75%) | 9 (75%) | 18 (75%) |
| African-American | 2 (16.7%) | 2 (16.7%) | 4 (16.7%) |
| Hispanic | 0% | 1 (8.3%) | 1 (4.2%) |
| Asian | 1 (8.3%) | 0% | 1 (4.2%) |
| Clinical Characteristics | |||
| BT Group | MAC Group | Total Sample | |
| Pulling Sitea | |||
| Scalp | 10 (83.3%) | 9 (75.0%) | 19 (79.2%) |
| Eyelashes | 6 (50.0%) | 4 (33.3%) | 10 (41.7%) |
| Eyebrows | 2 (16.7%) | 4 (33.3%) | 6 (25.0%) |
| Pubic | 0% | 2 (16.7%) | 2 (8.3%) |
| Arms/Legs | 0% | 1 (8.3%) | 1 (4.2%) |
| Comorbid Disordersb | |||
| Number of participants who met criteria for one or more comorbid conditions | 4 (33.3%) | 7 (58.3%) | 11 (45.8%) |
| At least one anxiety disorder (i.e., GAD) | 4 (33.3%) | 4 (33.3%) | 8 (33.3%) |
| OCD | 1 (8.3%) | 0% | 1 (4.2%) |
| At least one externalizing disorder (i.e., ADHD/ADD, ODD, CD) | 0% | 3 (25%) | 3 (12.5%) |
| Mood disorder | 1 (8.3%) | 2 (16.7%) | 3 (12.5%) |
| Pre-Treatment CDI Mean Total Score | 9.5 (7.7) | 7.5 (6.9) | 8.5 (7.2) |
Note: ADD = Attention Deficit Disorder; ADHD = Attention Deficit Hyperactivity Disorder; BT = behavioral therapy; CD = Conduct Disorder; CDI = Child Depression Inventory; GAD = Generalized Anxiety Disorder; MAC = minimal attention control; OCD = Obsessive Compulsive Disorder; ODD = Oppositional Defiant Disorder.
33.3 % of participants had more than one current pulling site
None of the participants presented with Post-Traumatic Stress Disorder or a history of interpersonal violence, as queried by the Anxiety Disorders Interview Schedule (ADIS)
Figure 1.
CONSORT Diagram
Note: ADD = Attention Deficit Disorder; ADHD = Attention-Deficit/Hyperactive Disorder; MDD = Major Depressive Disorder; TTM = Trichotillomania.
Consenting Procedure, Randomization, and Trial Oversight
At the intake meeting, study personnel provided a detailed description of the trial and its procedures, discussed associated risks and benefits, and encouraged parents/guardians and participating children to ask questions prior to providing informed consent (parent/guardian) and assent (child/adolescent) participant. Patients were randomly assigned to either BT or MAC using a computer-generated, randomized permuted blocking procedure. As is the case in most psychotherapy outcome studies, patients and treating clinicians were aware of the assignment to condition; masking was maintained for the primary and secondary dependent measures by use of a trained independent evaluator who was not otherwise involved in the patient’s care. The study was approved by the Institutional Review Board at the University of Pennsylvania School of Medicine, and a Data and Safety Monitoring Plan was developed in concert with program officers at the National Institute of Mental Health and was implemented during the trial.
Measures
Diagnostic criteria for TTM were assessed using the Trichotillomania Diagnostic Interview (TDI),12 which examined TTM diagnosis according to DSM IV criteria. Diagnostic criteria for other psychiatric disorders were surveyed using the Anxiety Disorders Interview Schedule for Children (ADIS-C),27 a semi-structured interview with established psychometric properties.27,28 The TDI and ADIS-C were used to assess diagnostic inclusion criteria, and were conducted at intake by evaluators trained to criteria in their use. The primary measure of treatment outcome was the NIMH Trichotillomania Severity Scale (NIMH TSS),29 which has demonstrated adequate psychometric properties in prior studies of TTM treatment.(see 30) The NIMH TSS total score ranges from 0 (no symptoms) to 25 (severe symptoms), and is comprise of items that survey time spent pulling in the past week, time spent pulling the previous day, resistance to pulling, associated distress, and functional impairment. The 7-point Clinical Global Impression – Improvement scale31 was used to categorize the degree of change in TTM symptoms over time. In the version used in the current study: 1 = much improved; 2 = improved; 3 = minimally improved; 4 =no change; 5 =minimally worse; 6 = worse; 7 = much worse; consistent with other clinical outcome studies in pediatrics32 the patient had to be assigned a CGI-I score a 1 or a 2 from the IE at post-treatment to be classified as a responder.
Independent Evaluations and Psychometrics of the NIMH-TSS in the Current Sample
Trained independent evaluators blind to treatment assignment conducted NIMH TSS interviews at weeks 0, 4, and 8 during the acute treatment phase; CGI-I scores were assigned by these same IEs at weeks 4 and 8. Notably, although the specific language of the questions was not formally adapted for use with this pediatric sample, study evaluators were highly experienced in conducting symptom assessments with children and adolescents with OC spectrum disorders including TTM, and their assessments were supervised for quality throughout the study. For the purpose of the current report, data from weeks 0 and 8 (end of acute phase) will be presented for both conditions; data from week 16 and from the naturalistic follow-up phase of the study were collected for the CBT condition only. At baseline, patients were assigned to be evaluated by the same evaluator throughout their participation in the study in order to minimize interviewer effects. In the current sample, test-retest reliability on all subjects completing both intake and baseline assessments (n=24) was acceptable (.70), even with different raters and interviews. Inter-rater agreement between the IE’s NIMH-TSS total score at baseline and the NIMH-TSS total score of that same videotape rated by a trained, independent rater was calculated for 50% of the sample and was good (.88).
Sample Size Justification
Power calculations for estimating sample size in this preliminary feasibility study reflected our interest in detecting only a large effect size, and were based on data from an adult study of behavior therapy conducted by our research group.33,34 Using change scores calculated from the pre- to post-treatment results from that study, a sample size of 12 in each group afforded us 91% power to detect a difference in NIMH-TSS means of 4.6 (the difference between a CBT mean of 8.6 and a conservative MAC change score mean of 4.0), assuming that the common standard deviation is 3.2 using a two group t-test with a 0.05 two-tailed alpha level. Power for a dichotomous outcome (CGI-I responder vs. nonresponder) was estimated using a continuity corrected chi-square test. Specifically, a two group continuity corrected2 test with a 0.05 two-tailed alpha level afforded us 80% power to detect the difference between a CBT proportion of 0.80 and a MAC proportion of 0.10 (odds ratio of 0.028) with a sample size of 10 in each group.
Treatments
Behavior therapy was conducted in accordance with a manual developed in the context of a treatment development grant, the manual from which has now been published.33 The acute treatment phase for behavior therapy lasted eight weeks and was conducted weekly; core elements of treatment included: 1) psychoeducation about the nature and treatment of TTM; 2) awareness training, in which participants were taught to become more aware of pulling behavior and pulling urges; 3) stimulus control, in which barriers to pulling were created based on participants’ report of high-risk situations; and 4) competing response training, in which participants were taught to engage in behaviors that were physically incompatible with pulling. Ancillary strategies were also permitted and included: progressive muscle relaxation (Session 5) and cognitive restructuring (Session 6); inclusion of these strategies was discussed in weekly supervision meetings with the Principal Investigator (MEF). Minimal attention control (MAC) was employed in this treatment development project to control for the effects of time and of repeated assessment; participants who received MAC were assigned to a therapist for 30 minute visits at week 0 and met again at weeks 4 and 8. Brief (10 – 20 minute) telephone conversations were conducted at weeks 1, 2, 3, 5, 6 and 7. MAC procedures were manualized, and included session outlines for both telephone and office sessions. These sessions included therapist inquiry about TTM symptom worsening and overall functioning, provision of emotional support to the family in the absence of specific treatment recommendations, and developmentally appropriate conversation or play depending on the child’s age. Notably, MAC did not match BT in the amount of clinical contact, nor was it intended to be an active intervention. MAC participants were offered open no-cost BT from the study team at week 8, thus no comparisons of BT and MAC were possible beyond week 8.
Clinical Supervision
Doctoral level psychologists experienced in BT for TTM treated the majority of cases; supervised doctoral students treated the remainder. Clinical supervision for both conditions was conducted by the study’s principal investigator (MEF), an expert in BT for TTM and related conditions across the developmental spectrum. For BT supervision, weekly meetings were conducted to review clinical case material, view selected segments of BT sessions conducted in the previous week, and discuss the implementation of BT for all patients randomized to this condition. Although not assigned to psychotherapy per se, patients assigned to MAC were also discussed in these weekly meetings to review their current clinical status and ensure that their symptoms were not worsening. To prevent “bleeding,” care was taken to remind therapists not to recommend any proscribed therapy techniques for MAC patients.
Maintenance Treatment Phase for BT
All patients randomized to the BT condition entered an 8-week maintenance treatment phase upon completion of the acute phase of BT. Four in-person sessions (weeks 10, 12, 14, and 16) and four brief scheduled telephone contacts (weeks 9, 11, 13, 15) were scheduled. The in-person meetings include inspection of self-monitoring information, review of treatment procedures utilized, evaluation of current pulling behavior and urges to pull, and troubleshooting any problems that may have occurred; these sessions lasted approximately 30 – 40 minutes. The telephone contacts were designed to solicit similar information but typically took 10 – 15 minutes to complete. Upon completion of the maintenance phase of BT, all participants were provided with clinical recommendations as needed and encouraged to re-contact the study team if symptoms returned.
Naturalistic Follow-up for Participants Randomized to BT
In order to assess maintenance of treatment gains, all BT participants (n = 12) were invited to participate in a re-evaluation of their TTM symptoms at 3 and 6 months post-maintenance treatment; they were also asked about whether they had initiated any psychiatric or psychological treatment since completing the maintenance phase of the study. To maximize participation, these evaluations were conducted either in person or by telephone depending on the family’s preference. Eight of the 12 patients assigned to BT participated in at least one follow-up evaluation; four were lost to follow-up and did not provide a reason not to participate (see Figure 1). None of the families who participated in the follow-up phase reported having initiated psychiatric or psychological treatment during the follow-up phase.
Results
Acute Outcomes
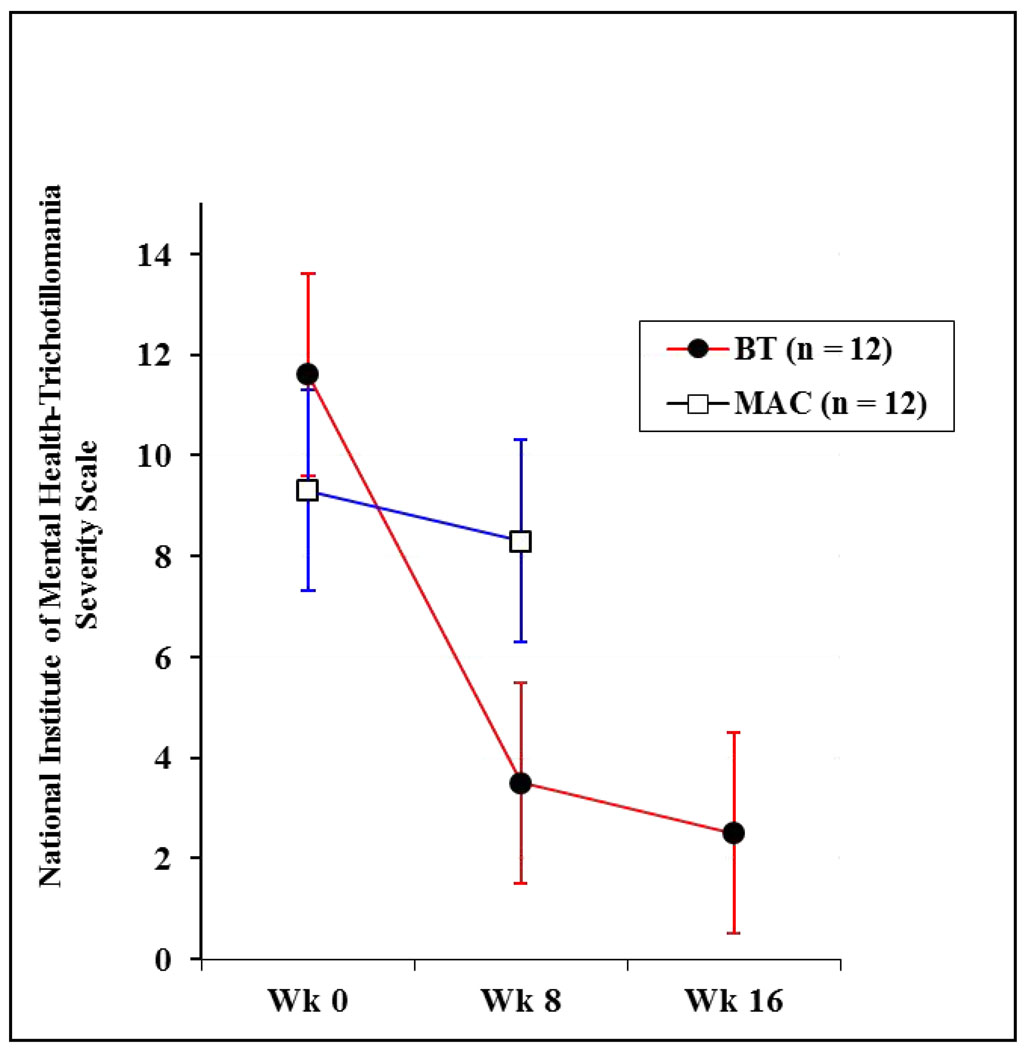
As depicted in Figure 2, the means and standard deviations on the IE-rated NIMH-TSS scores at week 0 were 11.6 (6.7) and 9.3 (4.5) for BT and MAC, respectively. At week 8, the NIMH-TSS scores were 3.5 (3.7) and 8.3 (4.0) for BT and MAC, respectively. To examine the effects of treatment, a 2 (condition: BT, MAC) × 2 (time: week 0, week 8) mixed model ANOVA was conducted, yielding a significant main effect for time, F (1, 22) = 17.7, p. < .01), and a significant group by time interaction, F (1, 22) = 10.7, p. < .01. Given the significant interaction, post hoc analyses were conducted to compare changes in NIMH-TSS scores from week 0 to week 8 separately by condition. NIMH-TSS scores in the BT group were significantly lower at week 8 than at week 0; t (11) = 4.1, p. < .01, whereas no such effect was found for MAC; t (11) = 1.1, p. > .05. Post-hoc tests comparing BT and MAC at week 0 and at week 8 indicated that the two groups did not differ significantly at week 0; t (22) = 1.0, p > .05, but did differ at week 8; t (22) = 3.0, p. < .01). Notably, an ANCOVA comparing BT and MAC on week 8 NIMH-TSS scores using pre-treatment NIMH-TSS scores as a covariate was also significant; F (1,21) =13.5, p. < .01. With respect to the percentage of patients meeting the definition of treatment responder (CGI-I of 1 or 2) at week 8, 75% of those assigned to BT and 0% of those assigned to MAC met this criterion.
Figure 2.
Behavior Therapy and Minimal Attention Control Outcomes on National Institute of Mental Health-Trichotillomania Severity Scale
Note: BT = behavioral therapy; MAC = minimal attention control
Durability of BT Gains Through Maintenance Treatment Phase
All 12 of the patients randomized to BT also completed the maintenance phase; planned comparisons of NIMH-TSS scores from week 0 to week 16 were significant; t (11) = 4.2, p. < .01, indicating that NIMH-TSS scores were significantly lower at the end of the maintenance phase than at week 0. The difference between week 8 and week 16 scores was not significant; t (11) = 1.4, p. >.05, suggesting no further improvement or relapse (see Figure 2). Seventy-five percent of BT participants at week 16 met the criterion for treatment responder (CGI-I = 1 or 2), which is the same percentage that did so as at week 8. Notably, although most week 8 responders were also responders at week 16, one non-responder at week 8 had achieved this criterion at week 16, and one responder at week 8 no longer met this criterion at week 16.
Exploratory Analysis: Durability of BT Gains through Naturalistic Follow-up
All patients randomized to BT were asked to participate in naturalistic follow-up assessments using the NIMH-TSS at weeks 28 and 40; 8 of 12 BT patients (67.3%) participated in at least one of those assessments. Notably, three of the four patients who did not participate in any follow-up assessments were week 8 CGI-I non-responders. To maximize use of all available data for these exploratory analyses of treatment durability, the week 28 score was carried forward to week 40 for one patient, and mean substitution at week 40 (M = 3.5) was used to replace missing values for the four BT patients who did not participate in either follow-up assessment. Planned comparisons were then conducted on NIMH-TSS scores (week 0 vs. follow-up, week 16 vs. follow-up) using these data. Means and SDs were: 11.6 (6.7), 2.5 (3.4), and 3.3 (3.3) for week 0, 16, and follow-up, respectively. Results indicated that NIMH-TSS scores at week 40 were significantly lower than at week 0; t (11) = 3.8, p < .01, and that week 16 and follow-up NIMH-TSS scores did not differ significantly; t (11) = 0.7, p < .05. Seven out of eight (87.5%) BT participants who completed at least one follow-up assessment were classified as responders at follow-up on the CGI-I; the CGI-I response rate for BT drops to 7 out of 12 participants (58.3%), however, if those lost to follow-up are assumed to have relapsed. Notably, follow-up analyses conducted using more conservative substitutions of week 0 NIMH-TSS means (assuming relapse to baseline symptom severity levels) yielded similar findings (data not shown) to those presented here.
Discussion
To the best of our knowledge the current study is the first randomized controlled trial that has examined the efficacy of any treatment for pediatric TTM, and its findings are encouraging with respect to the potential clinical utility of BT. BT was superior to MAC at post-treatment; moreover, acute treatment gains in BT were on average maintained over the course of an eight-week maintenance phase and through a naturalistic follow-up period. The absence of evidence for relapse following BT alone contrasts somewhat with findings from adult trials of behavioral approaches that included awareness training, stimulus control, and competing response procedures,35,36 and provides some preliminary support for the possibility that treatment of TTM during childhood or adolescence may be associated with more durable outcomes than does treatment during adulthood. Recent examinations of TTM phenomenology across the developmental spectrum have indicated a preponderance of automatic pulling in children and adolescents relative to focused pulling, which is not the same pattern typically observed in adults.37 Thus, it could be the case that treating TTM with BT before it takes on more complex affect regulation functions may be why treatment gains appear to be more robust and durable in the current sample than has been observed in the adult TTM treatment literature.4 In light of such developmental considerations, we also performed secondary analyses on the current sample to explore whether treatment outcome differences could be observed between the younger children and the older children who received behavior therapy in the current sample. A mixed model ANOVA, discussed in detail elsewhere,38 indicated that children ages 7–9 experienced comparable symptom reduction to the children ages 10–17 based on the NIMH-TSS; moreover, visual inspection of the plotted NIMH-TSS data between week 0 and week 8 suggested greater symptom reduction among the younger participants. Although low statistical power precludes drawing definitive conclusions, these findings suggest that even manualized BT can be tailored to the child’s appropriate developmental level.
The current sample is notable for phenomenological similarity to a sample collected as part of the Child-Adolescent Trichotillomania Impact Project,8 especially with respect to the pulling site information: scalp hair was by far the most common pulling site in both groups, followed by eyebrows and eyelashes. These two samples differed though in the number of active pulling sites reported: 57% of the CA-TIP sample reported more than one active pulling site, whereas only 33% of the current sample did. It is notable that the mean age of the current sample is about two years younger than reported for the CA-TIP sample. It is possible that the difference in the number of current pulling sites reflects a developmental trajectory for TTM in that the vast majority (97%) of the adult sample from the TIP reported multiple current pulling sites. Data on pulling styles collected from clinical samples across the full developmental spectrum will be critically important to examine in order to look at whether there are differences in the predominance of focused pulling (pulling with full awareness of the behavior and a clearly identifiable affective trigger) versus automatic pulling (pulling outside of awareness), as these different styles of pulling may necessitate different intervention strategies. New instruments have been developed for use with adults39 and children/adolescents40 that will facilitate a careful examination of the relationship between pulling styles and treatment outcome.
There are, of course, many limitations to what can be learned from a small treatment development project given its inherently restricted budget, duration, and scope. First, the small sample size of the present study limits the degree to which strong conclusions can be drawn about treatment effects. Second, while the MAC condition did control for the effects of repeated assessments and for the non-specific effects of participating in a clinical trial, it failed to account for the potential effects of therapist contact time, psychoeducation about TTM, and patient and parent expectancies of outcome. Third, the naturalistic follow-up period was relatively short, included only a subset (75%) of those who had been randomized to BT, and three of the four BT participants who did not complete a follow-up assessment had been week 8 non-responders, so it is possible that the data on durability of gains overestimate the long-term benefits of BT. Fourth, the absence of a treatment outcome measure adapted specifically for use with younger samples raises questions about the validity of conclusions; work is currently underway to examine the psychometrics of the NIMH-TSS with developmental adaptations to the language used to solicit information about pulling behavior and related phenomena (e.g., urges). Finally, information about pulling style in this sample was not collected, and thus the potential for differential response to BT depending on the preponderance of focused versus automatic pulling cannot be determined. A psychometrically sound measure of pulling style among children and adolescents with TTM has since been created,40 and will be valuable in the search for moderators and mediators of treatment response. Our research group is now taking the next steps to address some of these key limitations in the context of an ongoing NIMH-funded project41 in which we have: 1) increased the sample size from 24 in the present RCT to 60 or more in the ongoing study, which will allow us to observe whether the BT outcome can be replicated in a larger sample; 2) included an active comparison condition (Psychoeducation/Supportive Counseling) that will control for therapist contact time, psychoeducation, and other non-specific factors to ascertain whether the encouraging outcome achieved in the current trial is due to the specific components of the BT protocol; and 3) continued to treat and formally evaluate patients who receive open BT following participation in the control treatment, to permit greater statistical power to explore moderators and mediators of outcome, including pulling styles as measured by the Milwaukee Inventory of Styles of Pulling – Child Version (MIST-C).40 Recent encouraging findings for NAC in adults,16 a glutamate modulator, now open a potential avenue for exploration of its efficacy and safety in youth. Future studies should examine the relative and combined efficacy of BT and NAC, which would provide further information to guide clinical practice for the fairly large number of youth who suffer from TTM.
Acknowledgments
This research was supported by research grant R21 MH 61457 from the National Institute of Mental Health (M.E.F.)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure: Drs. Franklin, Ledley, and Cahill, and Ms. Edson report no biomedical financial interests or potential conflicts of interest.
Clinical Trial Registration Information – Cognitive Behavioral Treatment of Pediatric Trichotillomania; http://www.clinicaltrials.gov; R21 MH 61457;
Contributor Information
Martin E. Franklin, University of Pennsylvania School of Medicine
Aubrey L. Edson, University of Pennsylvania School of Medicine
Deborah A. Ledley, University of Pennsylvania School of Medicine
Shawn P. Cahill, University of Wisconsin-Milwaukee
References
- 1.American Psychiatric Association. Diagnostic and statistical manual of mental disorders, Fourth edition Text Revision. Washington, DC: American Psychiatric Association; 2000. [Google Scholar]
- 2.Christenson GA, Pyle RL, Mitchell JE. Estimated lifetime prevalence of trichotillomania in college students. J Clin Psychol. 1991;52:415–417. [PubMed] [Google Scholar]
- 3.Woods DW, Flessner CA, Franklin ME, et al. The trichotillomania impact project (TIP): Exploring phenomenology, functional impairment, and treatment utilization. J Clin Psychiat. 2006;67(12):1877–1888. doi: 10.4088/jcp.v67n1207. [DOI] [PubMed] [Google Scholar]
- 4.Stein DJ, Christenson GA, Hollander E. Trichotillomania. Washington, DC: American Psychiatric Press; 1991. [Google Scholar]
- 5.Mansueto CS, Townsley-Stemberger RM, McCombs-Thomas A, Goldfinger-Golomb R. Trichotillomania: A comprehensive behavioral model. Clin Psych Rev. 1991;17:567–577. doi: 10.1016/s0272-7358(97)00028-7. [DOI] [PubMed] [Google Scholar]
- 6.du Toit PL, van Kradenburg J, Niehaus DHJ, Stein DJ. Characteristics and phenomenology of hair-pulling: An exploration of subtypes. Compr Psychiat. 2001;42:247–256. doi: 10.1053/comp.2001.23134. [DOI] [PubMed] [Google Scholar]
- 7.Bouwer C, Stein DJ. Trichobezoars in trichotillomania: Case report and literature overview. Psychosom Med. 1998;60:658–660. doi: 10.1097/00006842-199809000-00025. [DOI] [PubMed] [Google Scholar]
- 8.Franklin ME, Flessner CA, Woods DW, et al. The Child and Adolescent Trichotillomania Impact Project: Descriptive psychopathology, comorbidity, functional impairment, and treatment utilization. J Dev Behav Pediatr. 2008;29(6):493–500. doi: 10.1097/DBP.0b013e31818d4328. [DOI] [PubMed] [Google Scholar]
- 9.Swedo SE, Leonard HL. Trichotillomania: An obsessive compulsive spectrum disorder? Psychiatr Clin North Am. 1992;15(4):777–790. [PubMed] [Google Scholar]
- 10.Christenson GA, Mansueto CS. Trichotillomania: Descriptive characteristics and phenomenology. In: Stein DJ, Christenson GA, Hollander E, editors. Trichotillomania. Washington, DC: American Psychiatric Press, Inc.; 1999. pp. 1–42. [Google Scholar]
- 11.Hajcak G, Franklin ME, Simons RF, Keuthen NJ. Hairpulling and skin picking in relation to affective distress and obsessive-compulsive symptoms. J Psychopathol Behav. 2006;28(3):179–187. [Google Scholar]
- 12.Rothbaum BO, Ninan PT. The assessment of trichotillomania. Behav Res Ther. 1994;32(6):651–662. doi: 10.1016/0005-7967(94)90022-1. [DOI] [PubMed] [Google Scholar]
- 13.Reeve E. Hair pulling in children and adolescents. In: Stein DJ, Christenson GA, Hollander E, editors. Trichotillomania. Washington, DC: American Psychiatric Press, Inc.; 1999. pp. 201–224. [Google Scholar]
- 14.Diefenbach GJ, Tolin DF, Hannan S, Crocetto J, Worhunsky P. Trichotillomania: Impact on psychosocial functioning and quality of life. Behav Res Ther. 2005;43:869–884. doi: 10.1016/j.brat.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 15.Van Ameringen M, Mancini C, Patterson B, Bennett M, Oakman J. A randomized, double blind, placebo- controlled trial of olanzapine in the treatment of trichotillomania. J Clin Psychiat. 2010;71(10):1336–1343. doi: 10.4088/JCP.09m05114gre. [DOI] [PubMed] [Google Scholar]
- 16.Grant JE, Odlaug BL, Kim SW. N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: A double-blind, placebo-controlled study. Arch Gen Psychiat. 2009;66(7):756–763. doi: 10.1001/archgenpsychiatry.2009.60. [DOI] [PubMed] [Google Scholar]
- 17.Woods DW, Friman PC, Teng E. Physical and social functioning in persons with repetitive behavior disorders. In: Woods DW, Miltenberger RG, editors. Tic disorders, trichotillomania, and other repetitive behavior disorders: Behavioral approaches to analysis and treatment. Norwell, MA: Kluwer Academic Publishers; 2001. pp. 33–52. [Google Scholar]
- 18.O’Sullivan RL, Keuthen NJ, Jenike MA, Gumley G. Trichotillomania and carpal tunnel syndrome. J Clin Psychiat. 1996;57:174. [PubMed] [Google Scholar]
- 19.Wetterneck CT, Woods DW, Norberg MM, Begotka AM. The social and economic impact of trichotillomania. Behav Intervent. 2006;21(2):97–109. [Google Scholar]
- 20.Stemberger RMT, Thomas AM, Mansueto CS, Carter JG. Personal toll of trichotillomania: Behavioral and interpersonal sequelae. J Anxiety Disord. 2000;14:97–104. doi: 10.1016/s0887-6185(99)00028-6. [DOI] [PubMed] [Google Scholar]
- 21.Schlosser S, Black DW, Blum N, Goldstein RB. The demography, phenomenology, and family history of 22 persons with compulsive hair pulling. Annals Clin Psychiatry. 1994;6:147–152. doi: 10.3109/10401239409148996. [DOI] [PubMed] [Google Scholar]
- 22.Friman PC, Blum N, Rostain A. Is hair pulling benign? J Am Acad Child Psy. 1992;31:991–992. doi: 10.1097/00004583-199209000-00040. [DOI] [PubMed] [Google Scholar]
- 23.Greer JM, Capecchi MR. Hoxb8 is required for normal grooming behavior in mice. Neuron. 1994;33:23–34. doi: 10.1016/s0896-6273(01)00564-5. [DOI] [PubMed] [Google Scholar]
- 24.Keuthen NJ, Fraim C, Deckersbach T, Dougherty DD, Baer L, Jenike MA. Longitudinal follow-up of naturalistic treatment outcome in patients with trichotillomania. J Clin Psychiat. 2001;62(2):101–107. doi: 10.4088/jcp.v62n0205. [DOI] [PubMed] [Google Scholar]
- 25.Christenson GA, Mackenzie TB, Mitchell JE, Callies AL. A placebo-controlled, double-blind crossover study of fluoxetine in trichotillomania. Amer J Psychiat. 1991;148:1566–1571. doi: 10.1176/ajp.148.11.1566. [DOI] [PubMed] [Google Scholar]
- 26.Hanna GL. Trichotillomania and related disorders in children and adolescents. Child Psychiat Hum D. 1997;27:255–268. doi: 10.1007/BF02353354. [DOI] [PubMed] [Google Scholar]
- 27.Silverman WK, Albano AM. Anxiety disorders interview schedule for DSM-IV: child and parent versions. San Antonio, TX: The Psychological Corporation; 1996. [Google Scholar]
- 28.Silverman WK, Saavedra LM, Pina AA. Test-retest reliability of anxiety symptoms and diagnoses with anxiety disorders interview schedule for DSM-IV : Child and parent versions. J Am Acad Child Psy. 2001;40(8):937–944. doi: 10.1097/00004583-200108000-00016. [DOI] [PubMed] [Google Scholar]
- 29.Swedo SE, Leonard HL, Rapoport JL, Lenane M, Goldberger EL, Cheslow D. A double blind comparison of clomipramine and desipramine in the treatment of trichotillomania (hair pulling) N Engl J Med. 1998;32:497–501. doi: 10.1056/NEJM198908243210803. [DOI] [PubMed] [Google Scholar]
- 30.Diefenbach GJ, Tolin DF, Crocetto J, Maltby N, Hannan S. Assessment of trichotillomania: A psychometric evaluation of hair-pulling scales. J Psychopathol Behav Assess. 2005;27(3):169–178. [Google Scholar]
- 31.Guy W. Clinical Global Impressions. Rockville, MD: National Institute for Mental Health; 1976. pp. 218–222. ECDEU Assessment Manual for Psychopharmacology (Revised DHEW Pub) [Google Scholar]
- 32.Piacentini J, Woods DW, Scahill L, et al. Behavior therapy for children with tourette disorder: A randomized controlled trial. JAMA. 2010;303(19):1929–1937. doi: 10.1001/jama.2010.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Franklin ME, Tolin DF. Treating trichotillomania: Cognitive-behavioral therapy for hairpulling and related problems. New York: Springer Science + Business Media; 2007. [Google Scholar]
- 34.Lerner J, Franklin ME, Meadows EA, Hembree E, Foa EB. Effectiveness of a cognitive behavioral treatment program for trichotillomania: An uncontrolled evaluation. Behav Ther. 1998;29(1):157–171. [Google Scholar]
- 35.Diefenbach GJ, Tolin DR, Hannan S, Maltby N, Crocetto J. Group treatment for trichotillomania: Behavior therapy versus supportive therapy. Behav Ther. 2006;37(4):353–363. doi: 10.1016/j.beth.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 36.Keijsers GPJ, van Minnen A, Hoogduin CAL, Klassen BNW, Hendriks MJ, Tanis-Jacobs J. Behavioural treatment of trichotillomania: Two year follow-up results. Behav Res Ther. 2006;44(3):359–370. doi: 10.1016/j.brat.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 37.Flessner CA, Woods DW, Franklin ME, Keuthen NJ, Piacentini J. Cross-sectional study of women with trichotillomania: A preliminary examination of pulling styles, severity, phenomenology, and functional impact. Child Psychiat Hum D. 2009;40(1):153–167. doi: 10.1007/s10578-008-0118-5. [DOI] [PubMed] [Google Scholar]
- 38.Franklin ME, Edson AL, Freeman JB. Behavior therapy for pediatric trichotillomania: Exploring the effects of age on treatment outcome. Child and Adolescent Psychiatry and Mental Health. 2010;4(18) doi: 10.1186/1753-2000-4-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Flessner CA, Woods DW, Franklin ME, Cashin SE, Keuthen NJ. The Milwaukee Inventory for Subtypes of Trichotillomania-Adult Version (MIST-A): Development of an instrument for the assessment of "focused" and "automatic" hair pulling. J Psychopathol Behav. 2008;30(1):20–30. [Google Scholar]
- 40.Flessner CA, Woods DW, Franklin ME, et al. The Milwaukee Inventory for Styles of Trichotillomania-Child Version (MIST-C): Initial development and psychometric properties. Behav Modif. 2007;31(6):896–918. doi: 10.1177/0145445507302521. [DOI] [PubMed] [Google Scholar]
- 41.Weiss B, DiLillo D. ABCT members Franklin and Woods receive first ever NIMH R01 grants for behavioral treatment of trichotillomania. Beh Therapist. 2009:189–190. [Google Scholar]