Abstract
Aims
Neutrophil gelatinase-associated lipocalin (NGAL) is a measure of acute kidney injury. Renal dysfunction portends significant risk after discharge from acute heart failure (AHF). Thus, a sensitive marker of renal injury might also help to risk stratify HF patients.
Methods and results
GALLANT [NGAL EvaLuation Along with B-type NaTriuretic Peptide (BNP) in acutely Decompensated Heart Failure] was a multicentre, prospective study to assess the utility of plasma NGAL, alone and in combination with BNP, as an early risk marker of adverse outcomes. We studied 186 patients (61% male). There were 29 events (AHF readmissions and all-cause mortality) at 30 days (16%). Patients with events had higher levels of NGAL than those without (134 vs. 84 ng/mL, P < 0.001). The area under the receiver operating characteristic curve was higher for NGAL (0.72) than BNP (0.65), serum creatinine (0.57), or estimated glomerular filtration rate (eGFR; 0.55). In multivariable analyses, NGAL predicted events (P= 0.001), BNP approached significance (P= 0.052 and 0.070 without creatinine and GFR, respectively) while neither serum creatinine nor eGFR were significant. The addition of discharge NGAL over BNP alone improved classification by a net 10.3% in those with events and 19.5% in those without events, for a net reclassification improvement of 29.8% (P= 0.010). Subjects with both BNP and NGAL elevated were at significant risk [hazard ratio (HR) = 16.85, P= 0.006], as were subjects with low BNP and high NGAL (HR = 9.95, P= 0.036).
Conclusions
Plasma NGAL is a measure of kidney injury that at the time of discharge is a strong prognostic indicator of 30 days outcomes in patients admitted for AHF.
Clinical trial registration number: NCT 00693745
Keywords: Renal injury, Acute heart failure, Biomarkers, BNP
See page 820 for the editorial comment on this article (doi:10.1093/eurjhf/hfr092)
Introduction
Renal dysfunction is common in patients with acute heart failure (AHF) and is associated with significant early and late morbidity and mortality.1,2 The fact that creatinine rise is a marker of renal function and not renal injury makes it less sensitive to early injury that might occur secondary to diuretic treatment, or other causes of decreased renal perfusion, including elevated central venous pressure.3 This and the fact that it can take up to 2 days for creatinine to increase after an ‘insult’ to the renal tubules make it less appealing than a potential specific early rising marker of renal injury and, therefore, as a risk predictor in these patients.3,4
Neutrophil gelatinase-associated lipocalin (NGAL) is a member of the lipocalin family of proteins. Neutrophil gelatinase-associated lipocalin is produced in the kidney following ischaemic or nephrotoxic injury in both animals and humans.5–11 Dent et al.12 demonstrated that NGAL was effective in identifying acute kidney injury (AKI) 2–3 h after cardiopulmonary bypass surgery as compared with 2–3 days for creatinine. In chronic HF urinary NGAL levels are elevated,1 while Damman et al. demonstrated that admission serum NGAL levels were associated with heightened risk of subsequent development of worsening renal function (WRF) in patients admitted for AHF.1,3
B-type natriuretic peptide (BNP) is a well-established marker for the diagnosis of AHF.13–15 Importantly, it has been clearly demonstrated that BNP levels at discharge are highly predictive of short-term outcomes.16 Ideally if BNP, which is a marker for ventricular stretch and a surrogate for pulmonary congestion,17 could be matched with a marker of renal injury, the combination would be quite useful for risk stratification.
The GALLANT trial was a multicentre prospective pilot clinical study to assess the utility plasma NGAL—alone and in conjunction with the BNP—as an aid to early risk assessment for HF-related adverse clinical outcomes (deaths, readmissions, and additional emergent outpatient visits) in patients presenting with acutely decompensated HF.
Methods
Patient population
We studied patients who were >18 years of age, and who presented to the emergency department (ED) or hospital with symptoms of new or decompensated HF in whom diuretics were an expected part of therapy. B-type natriuretic peptide levels were required to be ≥100 pg/mL and patients were also required to have systolic or diastolic dysfunction as documented on an echocardiogram within the past 12 months or during hospitalization. Patients were excluded if there was evidence of acute myocardial infarction or active ischaemia, cardiogenic shock, or any other clinical condition that might preclude administration of diuretics. Patients were also excluded if they had renal failure (serum creatinine ≥3.0, on dialysis (either acute or chronic) or in imminent need of dialysis). Patients who had received intravenous diuretic treatment for >90 min prior to enrolment were also excluded. Finally, patients with known or suspected infection with human immunodeficiency virus (HIV) or infectious hepatitis were excluded.
Ethylenediaminetetraacetic acid (EDTA) anticoagulated whole blood was collected for the NGAL and BNP measurements. Specimens were centrifuged (10 min at 2000 g) within 1h of collection and the plasma was frozen at <70°C prior to analysis. Creatinine was measured on serum specimens.
Once written informed consent had been obtained, baseline demographic data were recorded and blood samples were obtained for measurements of creatinine, NGAL, and BNP. These were repeated daily for up to 5 days as well as at discharge. Discharge samples were used for the analysis of the prognostic value of NGAL and BNP for adverse HF-related outcomes. Patients were followed for 30 days following discharge for HF readmission or all-cause mortality. The combined endpoint was considered to have been met if the patient experienced AHF readmission or all-cause mortality during the follow-up period. The institutional review boards of the enrolling institutions approved this study. The five participating study centres and corresponding patient numbers are given in Table 1.
Table 1.
Demographic and baseline characteristics
| With event (n= 29) |
Without event (n= 159) |
P | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Age (years) | 68.2 | 15.3 | 70.8 | 14.1 | 0.381 |
| BMI | 28.5 | 8.8 | 30.9 | 8.9 | 0.171 |
| BP systolic (mmHg) | 123.1 | 30.7 | 142.8 | 30.0 | 0.001 |
| BP diastolic (mmHg) | 72.2 | 12.8 | 81.8 | 17.7 | 0.006 |
| Heart rate (bpm) | 88.7 | 21.7 | 86.9 | 22.0 | 0.695 |
| With event (n= 29) | Without Event (n= 159) | P | |||
| n | % | n | % | ||
| Male | 19 | 65.5 | 96 | 60.4 | 0.682 |
| White race | 17 | 58.6 | 101 | 63.5 | 0.678 |
| Black race | 7 | 24.1 | 39 | 24.5 | 1.000 |
| Orthopnoea | 16 | 55.2 | 90 | 56.6 | 1.000 |
| PND | 10 | 34.5 | 44 | 27.7 | 0.505 |
| Dyspnoea on exertion | 22 | 75.9 | 121 | 76.1 | 1.000 |
| Dyspnoea at rest | 10 | 34.5 | 51 | 32.1 | 0.831 |
| Rales | 5 | 17.2 | 28 | 17.6 | 1.000 |
| Wheezing | 5 | 17.2 | 30 | 18.9 | 1.000 |
| S3 | 1 | 3.4 | 4 | 2.5 | 0.572 |
| S4 | 0 | 0.0 | 1 | 0.6 | 1.000 |
| JVP | 9 | 31.0 | 34 | 21.4 | 0.335 |
| Peripheral oedema | 21 | 72.4 | 120 | 75.5 | 0.816 |
| EF < 45% | 11/20 | 55.0 | 39/77 | 50.6 | 0.729 |
Note: Study centres and patient numbers were as follows: San Diego VA Hospital (n= 41), Ohio State University (n= 11), New York Methodist Hospital (n= 91), University Medical Center, Groningen (n= 23), and Universitätsspital, Basel (n= 22).
Measurements
The Triage NGAL Test (Alere Inc, San Diego, CA, USA) is an immunoassay in a single-use plastic cartridge that contains a fluorescently labelled monoclonal antibody against NGAL labelled with a fluorescent dye and NGAL. There are built-in control features, including control immunoassays, to ensure that the test performs properly and the reagents are functionally active. The test procedure involves the addition of several drops of whole blood or plasma to the sample port on the test device. After addition of the sample, the cells are automatically separated from the plasma via a filter. The sample reacts with fluorescent antibody conjugates within the reaction chamber and flows down the device detection lane by capillary action. The fluorescent antibody conjugates are captured on discrete solid-phase zones resulting in binding immunoassays that are specific for NGAL or the control antigens. B-type natriuretic peptide testing was also performed on the Triage platform using a standard commercially available assay (Triage Assay, Alere Inc.). Estimated glomerular filtration rate (eGFR) was calculated as MDRD as per Smilde et al.16
Statistical analysis
Continuous variables are expressed as mean (standard deviation) and compared using t-tests for independent groups if normally distributed; otherwise, they are expressed as medians [quartiles] and compared with Mann–Whitney U tests. Receiver operating characteristic (ROC) analysis was performed to evaluate individual marker prognostic utility for the combined endpoint and to ascertain cut-points yielding 80% sensitivity. The areas under the ROC curves (C-statistics) were compared using the method of DeLong et al.17 Relationships between biomarkers were evaluated with Spearman rank-order correlation. Kaplan–Meier methods were employed to generate survival plots that were compared using a log-rank test. Cox proportional hazards regression was employed to evaluate survival models using biomarkers with common log transformations for NGAL and BNP to achieve normality, and using biomarker cut-points. Net reclassification improvement20 was calculated based on risk levels of 6 and 20% as cut-points. A significance criterion of 0.05 without correction for multiple comparisons was employed throughout.
Results
There were 194 patients who met the inclusion criteria, of whom six were excluded due to loss to follow-up (n= 3) or withdrawal (n= 1), or HF not being the final diagnosis (n= 2). Twenty-nine subjects reached an endpoint of all-cause mortality or HF readmission by 30 days (16%). Table 1 shows the demographics of patients with or without events. The overall sample was 61% male with a mean age of 71 ± 13.8 years. Subjects with events were not different from subjects without events with respect to age, gender, symptoms, and physical findings. Only systolic and diastolic blood pressures were significantly different between groups.
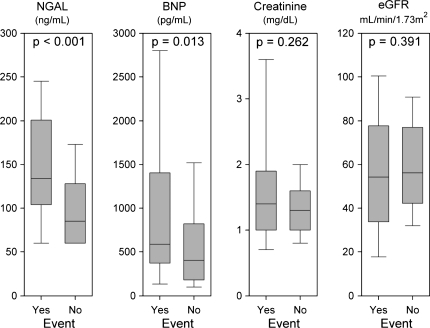
Figure 1 depicts discharge levels of NGAL, BNP, serum creatinine, and eGFR in those with and without 30 days events. Those with events had higher levels of NGAL than those without events (134 ng/mL [104–181] vs. 84 ng/mL [59–128], P < 0.001). Patients with events also had higher discharge BNP levels (585 pg/mL [375–1380]) than those without (384 pg/mL [172–818] P = 0.013). Neither discharge creatinine nor eGFR were significantly different between patients with and without events. At baseline, NGAL levels were already different for those with events than those without (112 ng/mL [75–153] vs. 72 ng/mL [60–120], P= 0.006) while BNP levels did not differ (837 pg/mL [500–1465] vs. 672 pg/mL [359–1350], P = 0.180).
Figure 1.
Box plots of markers at discharge. Boxes indicate medians and quartiles, whiskers indicate 10th and 90th percentiles.
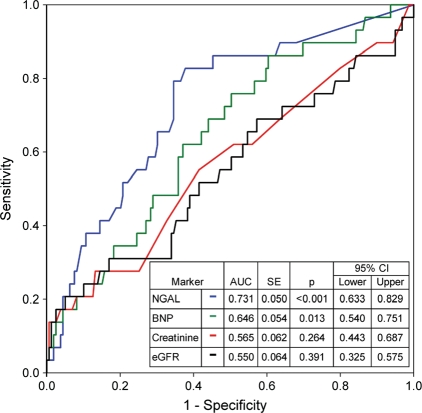
Figure 2 shows the area under the ROC curve for NGAL, BNP, creatinine, and eGFR for predicting 30 days HF-related adverse events. The area under the ROC curve (or C-statistic) was higher for NGAL (0.73) than for BNP (0.65), serum creatinine (0.57), or eGFR (0.55). Comparing C-statistics, NGAL was superior to creatinine (P = 0.004) and eGFR (P = 0.002), but not to BNP (P = 0.21). B-type natriuretic peptide, however, was not significantly different to creatinine (P = 0.33) or eGFR (P = 0.24). Discharge eGFR and creatinine were also not significantly different (P = 0.50).
Figure 2.
Receiver operating characteristic curve analysis for markers predicting heart failure re-hospitalization or all-cause mortality.
The correlation of NGAL levels was modest with creatinine (r = 0.42, P < 0.001) and eGFR (r = 0.44, P< 0.001) and weak with BNP (r = 0.22, P= 0.002). Multiple linear regression was used to predict NGAL levels from these variables.
Table 2 shows Cox regression models with either eGFR or creatinine in the model. In these multivariable analyses, neither log serum creatinine nor eGFR were significant for 30 days events, but log NGAL (P< 0.001 for both models) was a significant predictor of events and the log BNP values approached significance (P = 0.052 and 0.070). Using risk cut-points of 6 and 20%, subjects were divided into 30 days risk groups using log BNP alone or log BNP and log NGAL. The addition of NGAL to the model improved classification by a net 10.3% in those with events and 19.5% in those without events, for a net reclassification improvement of 29.8% (P= 0.010).
Table 2.
Multivariable Cox models for risk of heart failure re-hospitalization or all-cause mortality
| Biomarker | P | HR | 95% CI for HR |
|
|---|---|---|---|---|
| Lower | Upper | |||
| Model with eGFR | ||||
| lg BNP (pg/mL) | 0.052 | 2.47 | 0.99 | 6.14 |
| lg NGAL (ng/mL) | 0.000 | 29.83 | 5.43 | 163.96 |
| eGFR (mL/min/1.73 m2) | 0.284 | 1.01 | 0.99 | 1.02 |
| Model with creatinine | ||||
| lg BNP (pg/mL) | 0.070 | 2.327 | 0.934 | 5.795 |
| lg NGAL (ng/mL) | 0.001 | 19.912 | 3.472 | 114.189 |
| lg creatinine (mg/dL) | 0.891 | 1.177 | 0.115 | 12.064 |
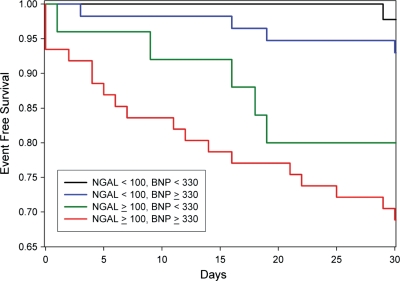
Using cut-points at the level of 80% sensitivity for BNP (330pg/mL) and NGAL (100 ng/mL), subjects were sorted by high/low BNP and high/low NGAL. Table 3 shows a Cox regression model with the four groups. As can be seen, when NGAL is low, a high BNP delineates no significant risk. However, when BNP is low, a high NGAL still portends significant risk. When both markers were elevated the risk was substantial (HR = 16.9, 95% CI = 2.3–195.9, P = 0.006). Figure 3 depicts Kaplan–Meier curves for these four groups. As shown, high-BNP/high-NGAL had the worst outcomes and survival rate was high when both markers were low. While the risk in the low-BNP/high-NGAL group was significant, the high-BNP/low-NGAL group had outcomes similar to when both markers were low.
Table 3.
Cox models for risk of heart failure re-hospitalization or all-cause mortality in groups based on neutrophil gelatinase-associated lipocalin and B-type natriuretic peptide cut-offs
| Group | P | HR | 95.0% CI for HR |
|
|---|---|---|---|---|
| Lower | Upper | |||
| NGAL < 100, BNP < 330 | 0.002 | 1.00 | reference | |
| NGAL < 100, BNP > 330 | 0.294 | 3.23 | 0.36 | 28.93 |
| NGAL > 100, BNP < 330 | 0.036 | 9.95 | 1.16 | 85.19 |
| NGAL > 100, BNP > 330 | 0.006 | 16.85 | 2.26 | 125.94 |
Figure 3.
Kaplan–Meier plots of heart failure re-hospitalization free survival based on cut-offs of 100 ng/mL for neutrophil gelatinase-associated lipocalin and 330 pg/mL for B-type natriuretic peptide. Log rank test P< 0.001.
Discussion
The main finding of the present study is that plasma NGAL at the time of hospital discharge is a powerful predictor of 30 days outcome in patients with AHF. It is stronger than BNP, which is considered to be the standard for risk prediction, and is substantially superior to conventional measures of renal function such as serum creatinine and eGFR. Thus, NGAL is not only a risk predictor for renal injury but is an overall strong risk marker for cardiac events in the setting of HF.
Physicians now recognize that HF entails a disorder at the level of both the heart and the kidneys. Worsening renal function is especially common during admission for acute decompensated HF and is associated with a poor prognosis.19–31 While the underlying causes of WRF are multifactorial, the combination of angiotensin-converting enzyme-inhibitors and intravenous diuretics likely are large contributors. The shortcomings of creatinine as a predictor of acute renal dysfunction are well known. Serum creatinine lacks the sensitivity to detect changes in GFR, as its increase is delayed by the need for accumulation once there is a reduction in GFR. Even if not present at the time of admission, kidney injury can ensue during diuresis at the tail end of the hospital period, and may not be manifest (in terms of elevated serum creatinine) at the time of discharge. While serum creatinine and eGFR are clearly prognostic at the time of discharge, a specific marker of tubular injury that rises early should reflect the state of tubular function at the time of discharge.
Neutrophil gelatinase-associated lipocalin is one of the earliest markers produced in the kidney following ischaemic or nephrotoxic injury in animal models, and it can be detected in the blood and urine of humans soon after AKI6–12 Several studies have confirmed these findings. In intensive care adult patients with AKI secondary to sepsis, ischaemia, or nephrotoxins, NGAL is significantly increased in the plasma and urine when compared with normal controls.12 In children undergoing cardiopulmonary bypass, NGAL was increased significantly in plasma and urine 2–6 h after surgery in those patients who subsequently developed AKI; the rise in creatinine was not evident until 48–72 h later. In this setting, both urine and plasma NGAL were powerful independent predictors of AKI, with an outstanding area under the curve of 0.998 for 2 h urine NGAL levels and 0.91 for 2 h plasma NGAL measurement. Similar findings have been reported in adults who develop AKI after cardiac surgery, with NGAL elevation occurring 1–3 h after surgery.6
Neutrophil gelatinase-associated lipocalin and B-type natriuretic peptide: implications for risk stratification
Assessment of blood BNP levels is now commonly performed in the ED to aid in the diagnosis of AHF in patients presenting with acute dyspnoea. The clinical utility of assessing BNP levels in the ED has been confirmed and described in detail in the scientific literature.13–16,30 B-type natriuretic peptide concentrations measured during hospital admission and/or at discharge in patients with HF have also been shown to provide prognostic information about the patient's subsequent risk of death or rehospitalization.16,17
While BNP is a marker of ventricular stretch, and therefore a surrogate of volume overload, NGAL is a marker of renal tubular injury that is both diagnostic and prognostic. Haase et al.31 recently presented a meta-analysis consisting of 1217 patients, with clinical outcomes of AKI patients according to NGAL and creatinine status. A major finding was that 41% of patients diagnosed with AKI would have been missed by creatinine alone. Neutrophil gelatinase-associated lipocalin tended to identify increased risk even when the creatinine was not significantly changed. Damman et al.1 demonstrated that structural tubular damage, as measured by increased concentrations of NGAL, is highly prevalent in patients with chronic HF. While the Damman study differed from ours in that urinary NGAL was not prognostic, the acute nature of our patients and the use of blood rather than urine, may have accounted for the difference in prognostic effect observed. Thus, across the spectrum of HF and healthy controls, NGAL levels are not only associated with different indices of renal dysfunction, but also positively associated with increased levels of natriuretic peptides.
In our study, subjects with the combination of high BNP/high NGAL had the worst outcomes; survival rate was high when both markers were low. While the risk in the low BNP/high NGAL group was significant, the high BNP/low NGAL group had outcomes similar to when both markers were low. This may be due to the fact that even though the BNP was high, it likely represented, ‘dry’ not ‘wet’ BNP. Thus, at the time of discharge, two easy to measure biomarkers: one of ventricular stretch (and volume overload) and one of renal tubular injury offer powerful risk stratification. Perhaps treatments to effectively lower BNP levels to euvolaemic ‘dry’ states without leading to decreases in renal perfusion, subsequent tubular ischaemia, and rising NGAL levels might offer the patient a better chance of improved survival and improved quality of life once they are discharged from the hospital. One might speculate that a high NGAL at the time of discharge might at a minimum, be a signal for early and perhaps more frequent follow-up and a judicious use of outpatient diuretics.
Funding
This study was sponsored by Alere. All data were analysed at the Veterans Affairs Medical Centre in San Diego.
Conflict of interest: A.M., consultant for Alere, speaker fees from Alere; C.M., consultant for Alere; R.F., none; R.B., none; B.C.H., none; N.I., none; P.C., none; DJvD, consultant for Alere.
References
- 1.Damman K, van Veldhuisen DJ, Navis G, Voors AA, Hillege HL. Urinary neutrophil gelatinase associated lipocalin (NGAL), a marker of tubular damage, is increased in patients with chronic heart failure. Eur J Heart Fail. 2008;10:997–1000. doi: 10.1016/j.ejheart.2008.07.001. [DOI] [PubMed] [Google Scholar]
- 2.Damman K, Voors AA, Hilldge HL, Navis G, Lechat P, van Veldhuisen DJ, Dargie HJ CIBIS-2 Investigators and Committees. Congestion in chronic systolic heart failure is related to renal dysfunction and increased mortality. Eur J Heart Fail. 2010;12:974–982. doi: 10.1093/eurjhf/hfq118. [DOI] [PubMed] [Google Scholar]
- 3.Chertow G, Burdick E, Honour M, Bonventre J, Bates D. Acute Kidney injury, mortality, length of stay and costs in hospitalized patients. J Am Soc Nephrol. 2005;16:3365–3370. doi: 10.1681/ASN.2004090740. [DOI] [PubMed] [Google Scholar]
- 4.Mishra J, Mori K, Ma Q, Kelly C, Barasch J, Devarajan P. Neutrophil gelatinase-associated lipocalin: a novel early urinary biomarker for cisplatin nephrotoxicity. Am J Nephrol. 2004;24:307–315. doi: 10.1159/000078452. [DOI] [PubMed] [Google Scholar]
- 5.Mori K, Lee HT, Rapoport D, Drexler IR, Foster K, Yang J, Schmidt-Ott KM, Chen X, Li JY, Weiss S, Mishra J, Cheema FH, Markowitz G, Suganami T, Sawai K, Mukoyama M, Kunis C, D'Agati V, Devarajan P, Barasch J. Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury. J Clin Invest. 2005;115:610–621. doi: 10.1172/JCI23056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schmidt-Ott KM, Mori K, Li JY, Kalandadze A, Cohen DJ, Devarajan P, Barasch J. Dual action of neutrophil gelatinase-associated lipocalin. J Am Soc Nephrol. 2007;18:407–413. doi: 10.1681/ASN.2006080882. [DOI] [PubMed] [Google Scholar]
- 7.Mori K, Nakao K. Neutrophil gelatinase-associated lipocalin as the real-time indicator of active kidney damage. Kidney Int. 2007;71:967–970. doi: 10.1038/sj.ki.5002165. [DOI] [PubMed] [Google Scholar]
- 8.Hirsch R, Dent C, Pfriem H, Allen J, Beekman RH, Ma Q, Dastrala S, Bennett M, Mitsnefes S, Devarajan P. NGAL is an early predictive biomarker of contrast-induced nephropathy in children. Pediatr Nephrol. 2007;22:2089–2095. doi: 10.1007/s00467-007-0601-4. [DOI] [PubMed] [Google Scholar]
- 9.Mishra J, Dent C, Tarabishi R, Tarabishi R, Mitsnefes M, Kelly C, Ruff S, Zahedl K, Shao M, Bean J, Mori K, Barasch J, Devarajan P. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet. 2005;365:1231–1238. doi: 10.1016/S0140-6736(05)74811-X. [DOI] [PubMed] [Google Scholar]
- 10.Dent CL, Ma Q, Dastrala S, Bennett M, Mitsnefes MM, Barasch J, Devarajan P. Plasma neutrophil gelatinase-associated lipocalin predicts acute kidney injury, morbidity and mortality after pediatric cardiac surgery: a prospective uncontrolled cohort study. Crit Care. 2007;11:R127. doi: 10.1186/cc6192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maisel AS, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, Omland T, Storrow AB, Abraham WT, Wu AHB, Clopton P, Steg PG, Westheim A, Knudsen CW, Perez A, Kazanegra R, Herrmann HC, McCullough PA. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med. 2002;347:161–167. doi: 10.1056/NEJMoa020233. [DOI] [PubMed] [Google Scholar]
- 12.McCullough PA, Nowak RM, McCord J, Hollander JE, Herrmann HC, Steg PG, Duc P, Westheim A, Omland T, Knudsen CW, Storrow AB, Abraham WT, Lamba S, Wu AHB, Perez A, Clopton P, Krishnaswamy P, Kazanegra R, Maisel AS. B-type natriuretic peptide and clinical judgment in emergency diagnosis of heart failure: analysis from Breathing Not Properly (BNP) Multinational Study. Circulation. 2002;106:416–422. doi: 10.1161/01.cir.0000025242.79963.4c. [DOI] [PubMed] [Google Scholar]
- 13.Januzzi JL, Jr, Camargo CA, Anwaruddin S, Baggish AL, Chen AA, Krauser DG, Tung R, Cameron R, Nagurney JT, Chae CU, Lloyd-Jones DM, Brown DF, Foran-Melanson S, Sluss PM, Lee-Lewandrowski E, Lewandrowski KB. The N-terminal pro-BNP investigation of dyspnea in the emergency department (PRIDE) study. Am J Cardiol. 2005;95:948–954. doi: 10.1016/j.amjcard.2004.12.032. [DOI] [PubMed] [Google Scholar]
- 14.Cheng V, Kazanegra R, Garcia A, Lenert L, Krishnaswamy P, Gardetto N, Clopton P, Maisel AS. A rapid bedside test for B-type peptide predicts treatment outcomes in patients admitted for decompensated heart failure—a pilot study. J Am Coll Cardiol. 2001;37:386–391. doi: 10.1016/s0735-1097(00)01157-8. [DOI] [PubMed] [Google Scholar]
- 15.Kazanegra R, Chen V, Garcia A, Krishnaswamy P, Gardetto N, Clopton P, Maisel AS. A rapid test for B-type natriuretic peptide correlates with falling wedge pressures in patients treated for decompensated heart failure: a pilot study. J Cardiac Fail. 2001;7:21–29. doi: 10.1054/jcaf.2001.23355. [DOI] [PubMed] [Google Scholar]
- 16.Smilde TDJ, van Veldhuisen DJ, Navis G. Drawbacks and prognostic value of formulas estimating renal function in patients with chronic heart failure and systolic dysfunction. Circulation. 2006;114:1572–1580. doi: 10.1161/CIRCULATIONAHA.105.610642. [DOI] [PubMed] [Google Scholar]
- 17.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44:837–845. [PubMed] [Google Scholar]
- 18.Pencina MJ, D'Agostino RB, Sr, D'Agostino RB, Jr, Vasan RS. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med. 2008;27:157–172. doi: 10.1002/sim.2929. [DOI] [PubMed] [Google Scholar]
- 19.Aghel A, Shrestha K, Mullens W, Borowski A, Tang WH. Serum neutrophil gelatinase-associated lipocalin (NGAL) in predicting worsening renal function in acute decompensated heart failure. J Card Fail. 2010;16:49–54. doi: 10.1016/j.cardfail.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smith GL, Lichtman JH, Bracken MB, Shlipak MG, Phillips CO, DiCapua P, Krumholz HM. Renal impairment and outcomes in heart failure: systematic review and meta-analysis. J Am Coll Cardiol. 2006;47:1987–1996. doi: 10.1016/j.jacc.2005.11.084. [DOI] [PubMed] [Google Scholar]
- 21.Heywood JT. The cardiorenal syndrome: lessons from the ADHERE database and treatment options. Heart Fail Rev. 2004;9:195–201. doi: 10.1007/s10741-005-6129-4. [DOI] [PubMed] [Google Scholar]
- 22.Devarajan P. Emerging biomarkers of acute kidney injury. Contrib Nephrol. 2007;156:203–212. doi: 10.1159/000102085. [DOI] [PubMed] [Google Scholar]
- 23.Dries DL, Exner DV, Domanski MJ, Greenberg B, Stevenson LW. The prognostic implications of renal insufficiency in asymptomatic and symptomatic patients with left ventricular systolic dysfunction. J Am Coll Cardiol. 2000;35:681–689. doi: 10.1016/s0735-1097(99)00608-7. [DOI] [PubMed] [Google Scholar]
- 24.McAlister FA, Ezekowitz J, Tonelli M, Armstrong PW. Renal insufficiency and heart failure: prognostic and therapeutic implications from a prospective cohort study. Circulation. 2004;109:1004–1009. doi: 10.1161/01.CIR.0000116764.53225.A9. [DOI] [PubMed] [Google Scholar]
- 25.Shlipak MG, Massie BM. The clinical challenge of cardiorenal syndrome. Circulation. 2004;110:1514–1517. doi: 10.1161/01.CIR.0000143547.55093.17. [DOI] [PubMed] [Google Scholar]
- 26.Weinfeld MS, Chertow GM, Stevenson LW. Aggravated renal dysfunction during intensive therapy for advanced chronic heart failure. Am Heart J. 1999;138:285–290. doi: 10.1016/s0002-8703(99)70113-4. [DOI] [PubMed] [Google Scholar]
- 27.Krumholz HM, Chen YT, Vaccarino V, Wang Y, Radford MJ, Bradford WG, Horwitz RI. Correlates and impact on outcomes of worsening renal function in patients ≥65 years of age with heart failure. Am J Cardiol. 2000;85:1110–1113. doi: 10.1016/s0002-9149(00)00705-0. [DOI] [PubMed] [Google Scholar]
- 28.Metra M, Nodari S, Parrinello G, Bordonali T, Bugatti S, Danesi R, Fontanella B, Lombardi C, Milani P, Verzura G, Cotter G, Dittrich H, Massie BM, Dei Cas L. Worsening renal function in patients hospitalized for acute heart failure: clinical implications and prognostic significance. Eur J Heart Fail. 2008;10:188–195. doi: 10.1016/j.ejheart.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 29.Butler J, Forman DE, Abraham WT, Gottleib SS, Loh E, Massie BM, O'Connor CM, Rich MW, Stephenson LW, Wang Y, Young JB, Krumholz HM. Relationship between heart failure treatment and development of worsening renal function among hospitalized patients. Am Heart J. 2004;147:331–338. doi: 10.1016/j.ahj.2003.08.012. [DOI] [PubMed] [Google Scholar]
- 30.Rogers K, Stehlik J, Stoddard G, Greene T, Collins S, Peacock WF, Maisel A, Clopton P, Michaels AD. Adjusting for clinical covariates improves the ability of B-type natriuretic peptide to distinguish cardiac from non-cardiac dyspnoea: a Sub-study of HEARD-IT. Eur J Heart Fail. 2009;11:1043–1049. doi: 10.1093/eurjhf/hfp127. [DOI] [PubMed] [Google Scholar]
- 31.Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009;6:1012–1024. doi: 10.1053/j.ajkd.2009.07.020. [DOI] [PubMed] [Google Scholar]