Abstract
Context
Since HSV-2 antibody tests have become commercially available, an increasing number of persons learn that they have genital herpes through serologic testing. The course of natural history of HSV-2 in asymptomatic, seropositive persons is uncertain.
Objective
To evaluate the virologic and clinical course of HSV genital shedding among participants with symptomatic and asymptomatic HSV-2 infection.
Design, Setting and Participants
Cohort of 498 immunocompetent HSV-2 seropositive persons enrolled in prospective studies of genital HSV shedding at the University of Washington Virology Research Clinic, Seattle, Washington, and Westover Heights Clinic in Portland, Oregon, between 1992 and 2008. Each participant obtained daily self-collected swabs of genital secretions for ≥ 30 days.
Main Outcome Measurement
The rate of viral shedding measured by quantitative real-time fluorescence polymerase chain reaction (PCR) for HSV DNA from genital swabs.
Results
HSV was detected on 4,753 of 23,683 days (20.1%; 95% CI, 18.3 to 22.0) in persons with symptomatic genital HSV-2 infection compared with 519 of 5,070 days (10.2%; 95% CI, 7.7 to 13.6) in persons with asymptomatic infection, p<0.001. Subclinical shedding rates were higher in persons with symptomatic infection compared with asymptomatic infection (2,708 of 20,735 days (13.1%; 95% CI, 11.5 to14.6) vs. 434 of 4,929 days (8.8%; 95% CI, 6.3 to 11.5), p<0.001. However, the amount of HSV detected during subclinical shedding episodes was similar (median 4.3 [IQR 3.1-5.6] log10 copies in the symptomatic infection group vs. 4.2 [IQR, 2.9-5.5], p=0.27 in the asymptomatic infection group). Days with lesions accounted for 2,045 of 4,753 days (43.0%; 95% CI, 39.8 to 46.5) with genital viral shedding among persons with symptomatic genital HSV-2 infection compared with 85 of 519 days (16.4%; 95% CI, 11.2 to 23.9) among persons with asymptomatic infection, p<0.001.
Conclusions
Persons with asymptomatic HSV-2 infection shed virus in the genital tract less frequently than persons with symptomatic infection, but much of the difference is attributable to less frequent genital lesions, as lesions are accompanied by frequent viral shedding.
INTRODUCTION
Herpes simplex virus type-2 (HSV-2) is one of the most frequent sexually transmitted infections worldwide with global estimates of 536 million infected persons and an annual incidence of 23.6 million cases among persons 15 to 49 years of age.1-3 In the United States, 16% of adults are HSV-2 seropositive,4 but only 10 to 25 percent of persons with HSV-2 infection have recognized genital herpes.5 Moreover, most HSV-2 infections are acquired from persons without a clinical history of genital herpes.6 Thus, the risk of sexual transmission does not correlate with the recognition of clinical signs and symptoms of HSV-2, but most likely correlates with viral mucosal shedding.7
Past work that characterized genital shedding in seropositive asymptomatic persons suggested that most, if not all, persons with HSV-2 intermittently shed virus on the genital skin/ and or mucosa, and most such persons recognized recurrences after learning the clinical signs and symptoms caused by HSV-2.5, 8 However, the genital shedding rate was lower and the duration of lesional episodes was shorter among persons who were unaware of their HSV-2 status compared with those who had a diagnosis of genital herpes. Whether these differences constitute perception, anatomic site, quantity of latently infected ganglionic cells, viral strain or host immunity characteristics remains unclear. To better understand the biological differences between persons who do and do not recognize their HSV-2 infection, we compared the rates and patterns of genital HSV shedding in what is to date the largest cohort of HSV-2 seropositive persons with and without a history of genital herpes, using daily sampling and an extensively characterized HSV PCR assay.
METHODS
Participants and clinical procedures
Participants were enrolled into prospective studies of natural history of genital HSV infection at University of Washington (UW) Virology Research Clinic and the Westover Heights Clinic in Portland, Oregon between March 1992 and April 2008. Inclusion criteria included age 18 or older, HSV-2 infection as defined by the University of Washington Western Blot, and good general health. None of the participants received antiviral medication during the study period, and all participants obtained 30 or more days of daily swabs of the genital skin/mucosa. Participants were recruited from the community by word of mouth and advertisements. Asymptomatic participants were identified as potential participants in a study of a candidate prophylactic HSV-2 vaccine, but were unexpectedly found to be HSV-2 seropositive, through screening for HSV antibodies by their providers as part of medical care, or had partners with genital HSV-2 but were thought to be uninfected. All participants signed informed consent and the University of Washington IRB approved the study protocol.
Demographic information, medical and sexual history were collected using standardized forms. Race and ethnicity information was gathered from participants using self-report. Participants attended individual educational sessions on HSV-2, were shown pictures of both typical and atypical lesions, and were instructed to inspect the genital region for lesions daily, and obtain swabs of their genital area, including any genital lesions. Men were instructed to swab first the penile skin and then the perineum and the perianal areas.9 Women were instructed to insert the swab into the vagina, then swab the vulva, the perineum and the perianal areas.10 Swabs were placed into vials containing 1 mL of PCR transport medium and stored at 4°C until laboratory processing. Each participant kept a diary of genital lesions and symptoms.10 Participants visited the clinic every 2 weeks for symptom review and collection of swabs.
Laboratory Methods
HSV serostatus was determined with the University of Washington Western Blot.11 A real-time fluorescence based quantitative PCR assay using primers to glycoprotein B was used to detect HSV DNA.12 A result was considered positive if greater than 150 copies of HSV DNA per mL of PCR buffer were detected.13 In order to detect contamination, 15 HSV negative controls were included in each 96-well plate; no contaminants were detected.
Definitions
Participants were classified as having ‘symptomatic HSV-2 infection’ if they had a clinical history of genital herpes at the time of their diagnosis.14 Participants had ‘asymptomatic HSV-2 infection’ if they did not have a history of symptomatic genital herpes prior to the serologic diagnosis of HSV-2, and thus the diagnosis was based on the positive antibody test for HSV-2. For most participants the time of the serologic diagnosis coincided with study entry, but some participants may have tested HSV-2 seropositive prior to study entry. We further subdivided the participants who lacked genital herpes but had a serologic HSV-2 diagnosis into those that remained asymptomatic and those that developed manifestations of genital herpes during the follow up. Genital shedding was defined as detection of HSV in the genital area. Clinical viral shedding was defined as shedding on days with lesions consistent with genital herpes. Subclinical genital shedding was defined as days on which HSV was detected in the absence of lesions. We defined shedding episodes as consecutive HSV positive swabs; those that were preceded and followed by two consecutive negative swabs were deemed to have known duration.15
Statistical Analysis
Genital shedding rates were calculated using the number of days with positive swabs divided by the total number of days with swabs collected for each subject. Confidence intervals for average shedding and lesion rates were computed using an intercept-only Poisson regression model, which correctly accounts for multiple samples per person. In cases where raw rates and model-predicted rates differ by more than 1%, both are provided. The amount of virus shed per sample was quantified as log10 copies per mL. The annualized episode rate was calculated by scaling the number of shedding episodes to the follow-up period. Categories for variables such as age and recurrences per year were determined by using quantiles, as graphical investigation demonstrated a lack of linear relationship. Years since acquisition were categorized based on prior work showing that that shedding decreases the first year after HSV-2 acquisition, and then stabilizes16. The Wilcoxon rank sum was used when continuous data were not normally distributed to calculate p-values. The chi-square test was used for categorical data, and the t-test was used for normally distributed continuous data. To characterize the predictors of genital shedding, Poisson regression analyses were performed, including a scale parameter for overdispersion.17 Variables that were hypothesized to be predictors of genital shedding included, age, sex, race, sexual preference, HSV-1 seropositivity, frequency of herpes recurrences and a history of symptomatic genital HSV-2 infection.15, 18 The multivariate model was determined through backwards elimination. Variables with p<0.10 in the univariate model were included in the initial multivariate model, then removed individually, if p>0.05. A two-sided p-value of 0.05 was considered to be statistically significant. A post-hoc, two-sample test for repeated measures binary data was performed to determine power: with outcome rates of 10-20% we had at least 80% power to detect risk ratios equal or less than 0.7 and equal or greater than 1.2.17 Statistical analyses were performed using SAS® version 9.2 (Cary, NC).
RESULTS
Characteristics of Study Participants
Four hundred and ninety-eight HSV-2 seropositive, healthy participants collected at least 30 days of genital swabs and were included in the analysis. The mean age of study participants was 40.7 years (SD, 11.0) (Table 1). Two hundred and fifteen (43.2%) participants were men and 283 (56.8%) participants were women. Two hundred and sixty-eight (53.8%) participants were HSV-2 seropositive only, while 230 (46.2%) participants were HSV-1 and HSV-2 co-infected. Most participants were white (401; 80.5%), 32 (6.4%) were African American, and 65 (13.1%) were other races or unknown. Most participants (325; 65.3%), were heterosexual; 68 (13.7%) were men who had sex with men; sexual behavior was not collected for 104 (20.9%). The mean age at sexual debut was 16.6 years (SD, 2.9).
Table 1.
Demographics, Clinical Characteristics, Frequency and Quantity of Genital Viral Shedding Rates and Characteristics of Shedding Episodes (N=498)
| Characteristic | Na (% Total or range) | Asymptomatic HSV-2 Infectionb (N=88) | Symptomatic HSV-2 Infectionc (N=410) | P-Value |
|---|---|---|---|---|
| Age, mean (SD)d | 40.7 (11.0) | 42.5 (9.6) | 40.2 (11.3) | 0.060 |
| Sex | ||||
| Men | 215 (43.2) | 54 (61.4) | 161 (39.3) | <0.001 |
| Women | 283 (56.8) | 34 (38.6) | 249 (60.7) | |
| Race | 0.059 | |||
| White | 401 (80.5) | 73 (82.9) | 328 (80.0) | |
| African-American | 32 (6.4) | 9 (10.2) | 23 (5.6) | |
| Other or Unknown | 65 (13.1) | 6 (6.8) | 59(14.4) | |
| Sexual Preference | <0.001 | |||
| Heterosexual Men and Women | 325 (65.3) | 48 (54.6) | 277 (67.6) | |
| Men who have Sex with Men | 68 (13.7) | 36 (40.9) | 32 (7.8) | |
| Women who have Sex with Women | 1 (0.2) | 0 (0) | 1 (0.2) | |
| Unknown | 104 (20.9) | 4 (4.6) | 100 (24.4) | |
| Age at First Sex, mean (SD) | 16.6 (2.9) | 16.0 (3.1) | 16.7 (2.9) | <0.001 |
| HSV serology | 0.027 | |||
| HSV-2 only | 268(53.8) | 38 (43.2) | 230 (56.1) | |
| HSV-1&2 | 230(46.2) | 50 (56.8) | 180 (43.9) | |
| Years since Initial Infectione, median (IQR) | 8.3(2.5-16.8) | 8.3 (2.5-16.8) | ||
| Recurrences in year prior to first visit, median (IQR) | 4.0(2-8) | 0.0 (0-4)f | 4.0 (2-8) | <0.001 |
| Days Sampled, median (IQR) | 57.0(47-62) | 56.5 (43-67.5) | 57.0 (50-62) | 0.83 |
| PCR-Positive Days/Total PCR Swabs (%) | 5272/28,753 (18.3) | 519/5,070 (10.2) | 4,753/23,683 (20.1) | <0.001 |
| Subclinical PCR-Positive Days/Subclinical Swabs (%) | 3142/25,664 (12.2) | 434/4,929 (8.8) | 2,708/20,735 (13.1) | <0.001 |
| PCR-Positive Lesional Swabs/Total Swabs With Lesions Present (%) | 2,130/ 3,089(68.9) | 85/141 (60.3) | 2,045/2,948 (69.4) | 0.025 |
| Genital log10 Copies, Median (IQR)g | 4.9 (3.4-6.3) | 4.5 (3.2-5.8) | 4.9 (3.5-6.3) | <0.001 |
| Subclinical Genital log10 Copies, Median (IQR)g | 4.3 (3.1-5.6) | 4.2 (2.9-5.5) | 4.3 (3.1-5.6) | 0.27 |
| Clinical Genital log10 Copies, Median (IQR)g | 5.6 (4.3-6.7) | 5.7 (4.2-6.9) | 5.6 (4.3-6.7) | 0.21 |
| Genital Shedding Episode Duration in Days, Median (IQR) | 3.0 (1.0-5.5) | 2.0(1.0-5.0) | 3.0(1.0-5.5) | 0.33 |
Numbers may not sum to total due to missing data, and percentages may not sum to 100% due to rounding.
Asymptomatic HSV-2 infection is defined as diagnosis of HSV-2 infection made by serologic testing in the absence of a history of genital lesions.
Symptomatic HSV-2 infection is defined as clinical history of genital herpes at the time of the diagnosis.
Age was unknown for 41 persons.
For people with history of genital herpes. Unknown for 88 persons with a history of genital herpes.
One person who was asymptomatic at the time of diagnosis by a positive HSV-2 antibody test developed recurrences prior to entry to our study.
Among positive samples.
Four-hundred ten participants (82.3%) reported symptomatic genital herpes infection, while 88 (17.6%) participants were asymptomatic at the time they had serologically documented HSV-2 infection. Among persons with symptomatic genital herpes, the median time since HSV-2 infection was 8.3 years (IQR, 2.5-16.8) and the median number of self-reported recurrences per year was 4 (IQR, 2-8).
Genital shedding and lesion rate
Overall, there were 33,113 days of observation; of these, 28,753 had genital swabs available for analysis. HSV DNA was detected in 5,272 of 28,753 swabs (18.3%; 95% CI 16.7 to 20.1). Participants were observed for a median of 57 days (IQR, 47-62) (Table 1). The overall genital shedding rate was significantly higher in persons with symptomatic infection than with asymptomatic infection, 4,753 of 23,683 days (20.1%; 95% CI, 18.3 to 22.0 ) vs. 519 of 5,070 days (10.2%; 95% CI, 7.7 to 13.6, p<0.001). Genital HSV was detected at least once in 342 of 410 (83.4%; 95% CI, 79.8 to 87.0) of persons with symptomatic HSV-2 infection and in 60 of 88 (68.2%; 95% CI 58.5 to 77.9) persons with asymptomatic HSV-2 infection (Figure 1).
Figure 1.
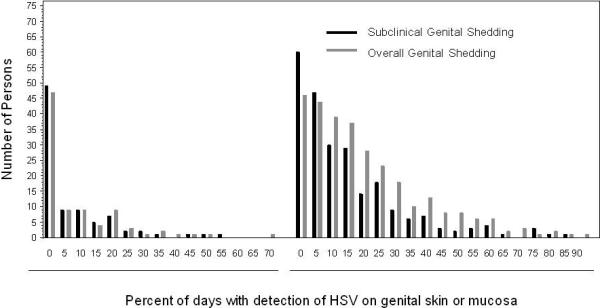
Distribution of Genital Shedding Rate among Asymptomatic and Symptomatic Infection Groups. Left panel represents persons with asymptomatic HSV-2 infection; right panel represents persons with symptomatic HSV-2 infection.
No genital lesions were reported on 25,664 days of follow up (89.6%) (vs. the model predicted per-person rate of 87.9%; 95%CI, 86.3 to 89.3), and HSV was detected on 3,142 (12.2%; 95% CI, 10.8 to 13.5) of those days. Subclinical genital shedding rates were higher in persons with symptomatic infection compared to asymptomatic infection, (2,708 of 20,735 [13.1%; 95% CI, 11.5 to 14.6] vs. 434 of 4,929 [8.8%; 95% CI, 6.3 to 11.5], p<0.001).
Genital lesions were reported on 3,089 days with PCR swabs (10.7%, versus model-predicted lesion rate of 12.1; 95% CI, 10.7 to 13.7), including 2,130 days (68.9%; 95% CI, 58.4 to 72.7) on which genital HSV was detected. During prospective follow-up, 262 (63.9%; 95% CI, 59.2 to 68.5) persons enrolled with symptomatic HSV-2 infection reported genital lesions at least once. In addition, 19 (21.6%; 95% CI, 13.0 to 30.2) persons who had asymptomatic, serologically diagnosed HSV-2 infection, recognized lesions during the follow-up. The frequency of lesions during the study was higher among persons with symptomatic vs. asymptomatic genital HSV-2, (3,259 of 26,777 days [12.2%; 95% CI, 12.2 to 15.6]) compared to 194 of 6,336 days [3.1%; 95% CI, 2.3 to 6.4], p<0.001).
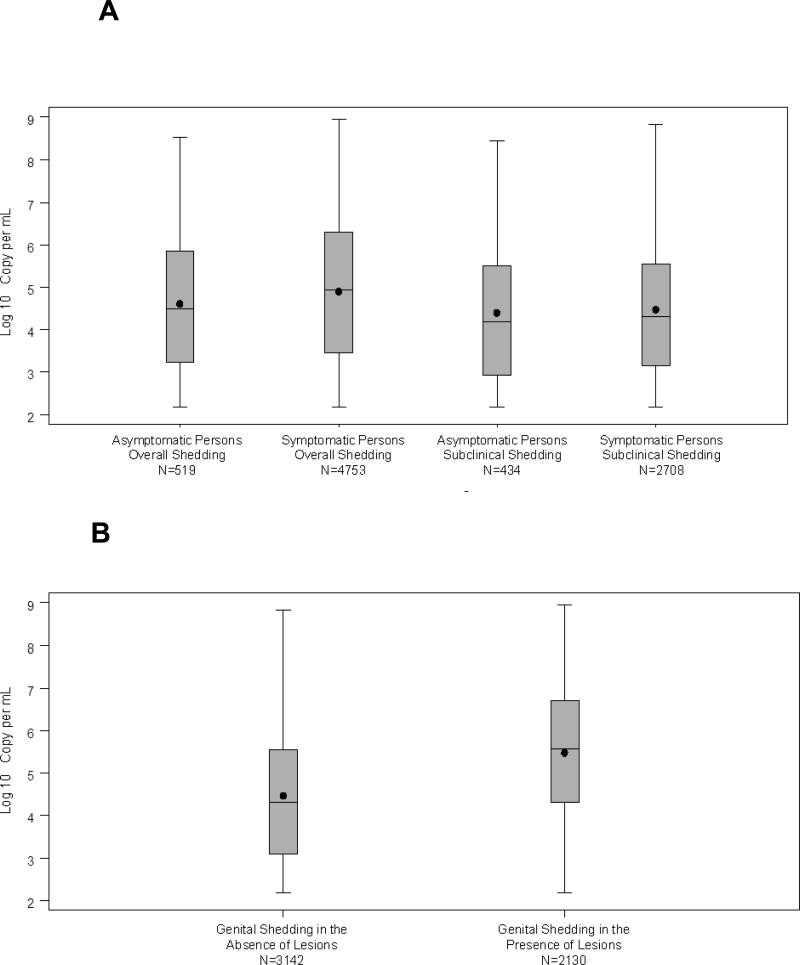
Overall, on days with detectable HSV, the median log10 copy number was 4.9 (IQR, 3.4-6.3). The median HSV log10 copy number was significantly higher among persons with symptomatic than asymptomatic infection, 4.9 (IQR, 3.5-6.3) vs. 4.5 (IQR, 33.2-5.8), respectively (p<0.001; Figure 2A). The median amount of HSV detected in the presence of lesions was also higher compared to the amount of HSV detected in the absence of lesions, 5.6 log10 (IQR, 4.3-6.7) vs. 4.3 log10 (IQR, 3.1-5.6) copies/mL, respectively, (p<0.001; Figure 2B). However, the median amount of HSV detected during subclinical genital shedding episodes was similar in those with symptomatic and asymptomatic infection (4.3 [IQR, 3.1-5.6] vs. 4.2 [IQR, 2.9-5.5] log10 copies/mL, p=0.27). Among persons with symptomatic genital herpes, 2,708 (56.9%; 95% CI, 53.4 to 59.6) of days of shedding were subclinical compared with 434 (83.6%; 95% CI, 69.4 to 92.3) of days among persons with asymptomatic HSV-2 infection.
Figure 2.
HSV Copy Number During Genital Shedding
A) HSV Copy Number During Overall and Subclinical Genital Shedding and History of HSV-2 Infection.
B) HSV Copy Number During Genital Shedding and Lesion Status.
Footnote: Boxes represent interquartile range; dots represent means; whiskers represent the minimum and maximum and horizontal lines represent the median. The numbers indicate the number of swabs in each category.
Shedding Episode Frequency and Duration
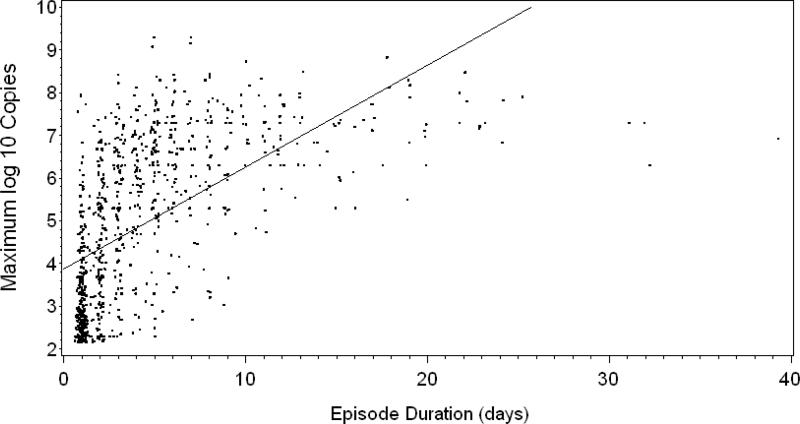
Viral shedding episodes associated with genital lesions were significantly longer than those without lesions (median 5.0 [IQR 3.0-9.0] vs. 2.0 [IQR, 1.0-3.5] days, p<0.001). Persons with symptomatic infection had more frequent genital shedding episodes as compared with persons with asymptomatic infection (median 17.9 [IQR, 11.9-27.1] vs. 12.5 [IQR, 8.5-19.8] episodes per year, p=0.004). In all persons, longer shedding episode duration was associated with higher maximum log10 copy number detected (R2 value=0.34; eFigure 1).
eFigure 1.
Episode Duration and Maximum Amount of HSV Shed During 922 Episodes. Footnote: The curve represents the R2 regression line. R2 Value=0.34.
Predictors of Genital Shedding
In univariate analyses, persons with asymptomatic HSV-2 infection had a shedding rate of 10.2% compared to a rate of 20.1% among persons with symptomatic genital herpes (RR=0.51; 95% CI, 0.38 to 0.68, p<0.001; Table 2). A higher number of recurrences per year was found to be a predictor of genital shedding. Persons with 8 or more recurrences per year had a shedding rate of 30.7% compared to 19.1% among persons with persons with 1-7 recurrences per year (RR = 1.61; 95% CI, 1.27 to 2.03, p<0.001). Persons of white race had a higher viral shedding rate than persons reporting non-white race, (20.0% vs. 10.8%, RR=1.78; 95% CI, 1.22 to 2.60, p=0.003). HSV-1 infection did not influence the risk of HSV-2 genital shedding, with a shedding rate of 18.2% for persons with HSV-1 and HSV-2 vs. 18.4% for persons with HSV-2 alone (RR = 0.99; 95% CI, 0.83 to 1.19, p=0.91).
Table 2.
Predictors of HSV-2 Genital Shedding Rate
| Overall Genital Shedding Ratea | Subclinical Genital Shedding Rateb | |||||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Unadjusted Risk Ratio (95% CI) | Unadjusted P-Value | Adjusted Risk Ratio (95% CI) | Adjusted P-value | Unadjusted Risk Ratio (95% CI) | Unadjusted P-Value | Adjusted Risk Ratio (95% CI) | Adjusted P-value |
| History of Infection of HSV-2 | ||||||||
| Symptomatic | Ref | Ref | ||||||
| Asymptomatic | 0.51 (0.38, 0.68) | <0.001 | 0.57 (0.42, 0.77) | <0.001 | 0.66 (0.48, 0.91) | 0.011 | 0.72 (0.51, 1.02) | 0.064 |
| Age | Ref | Ref | ||||||
| Age ≥ 41 years | ||||||||
| Age 31-40 years | 1.21 (0.976, 1.50) | 0.0819 | 1.15 (0.87, 1.52) | 0.327 | ||||
| Age ≤ 30 years | 1.67 (1.34, 2.08) | <0.001 | 1.88 (1.44, 2.46) | <0.001 | ||||
| Age Unknownc | 0.38 (0.24, 0.60) | <0.001 | 0.43 (0.26, 0.71) | <0.001 | ||||
| Gender | ||||||||
| Male | Ref | Ref | ||||||
| Female | 1.17 (0.98, 1.41) | 0.090 | 1.25 (0.99, 1.56) | 0.059 | ||||
| Race | ||||||||
| Other | Ref | Ref | ||||||
| White | 1.78 (1.22, 2.60) | 0.003 | 1.74 (1.31, 2.31) | <0.001 | 1.53 (1.11, 2.11) | 0.010 | 1.45 (1.05, 2.00) | 0.026 |
| Years Since Acquisition | ||||||||
| 11-40 Years Since | Ref | Ref | ||||||
| Acquisition | ||||||||
| < 1 Year Since Acquisition | 2.20 (1.74, 2.77) | <0.001 | 3.06 (2.30, 4.09) | <0.001 | ||||
| 1-10 Years Since Acquisition | 1.32 (1.07, 1.63) | 0.0096 | 1.48 (1.12, 1.95) | 0.006 | ||||
| Unknown Number of Years | 0.30 (0.33,0.41) | <0.001 | 0.32 (0.24, 0.43) | <0.001 | ||||
| Since Acquisitiond | ||||||||
| Sexual Preference | ||||||||
| MSM and Bisexual Men | Ref | Ref | ||||||
| Heterosexual Mene | 1.37 (1.02, 1.84) | 0.034 | 1.40 (0.97, 2.02) | 0.074 | ||||
| Women | 1.41 (1.09, 1.83) | <0.001 | 1.51 (1.10, 2.08) | 0.012 | ||||
| HSV Serology | ||||||||
| HSV-2 Only | Ref | Ref | ||||||
| HSV 1 & 2 | 0.99 (0.83, 1.19) | 0.91 | 0.98 (0.79, 1.23) | 0.89 | ||||
| Recurrences Per Year | ||||||||
| 1-7 recurrences per year | Ref | Ref | ||||||
| ≥8 Recurrences per year | 1.61 (1.27, 2.03) | <0.001 | 1.57 (1.25, 1.97) | <0.001 | 1.49 (1.09, 2.03) | 0.013 | 1.46 (1.07,1.99) | 0.016 |
| Unknown # of Recurrences | 0.77 (0.63, 0.94) | <0.001 | 0.96 (0.78,1.19) | 0.71 | 0.81 (0.63, 1.03) | 0.090 | 0.94 (0.72, 1.23) | 0.65 |
The genital shedding rate was calculated per participant using the number of days in which HSV DNA was detected divided by the total number of days with genital swabs.
The subclinical genital shedding rate was calculated per participant using the number of subclinical days in which HSV DNA was detected divided by the number of subclinical days with genital swabs.
Age unknown for 41 persons.
Years since acquisition unknown for 136 persons, including all 88 persons reporting asymptomatic acquisition.
34 men with unknown sexual preference are categorized in this group.
In the adjusted multivariate model, the risk for genital shedding remained lower among persons with asymptomatic HSV-2 infection compared to those with symptomatic infection, RR = 0.57 (95% CI, 0.42 to 0.77, p<0.001). Higher number of recurrences per year and white race also remained predictive of higher genital shedding rate in the adjusted analysis (Table 2).
Predictors of the Subclinical Genital Shedding
Asymptomatic infection was associated with lower rates of shedding in univariate analyses, with a shedding rate of 8.5% vs. 12.9% among persons with symptomatic infection (RR= 0.66; 95% CI, 0.48 to 0.91, p=0.011; Table 2). Women had a slightly increased subclinical genital shedding rate compared to men, 13.2% of days vs. 10.6% (RR=1.25; 95% CI, 0.99 to 1.56, p=0.059). Subclinical viral shedding was also more frequent among persons of white race compared to persons of other races, 12.9% vs. 8.4% (RR=1.53; 95% CI, 1.11 to 2.11, p=0.010). Persons with 8 or greater recurrences per year had a viral shedding rate of 18.9% vs. 12.7% among person reporting 1 to 7 recurrences per year (RR=1.49; 95% CI, 1.09 to 2.03, p=0.013). HSV-1 infection did not influence the risk of subclinical HSV-2 genital shedding, 11.9% for persons with HSV-1 and HSV-2 vs. 12.2% for HSV-2 only seropositive persons (RR=0.98; 95% CI, 0.79 to 1.23, p=0.89).
In the multivariate model, the risk for subclinical genital shedding among persons with asymptomatic HSV-2 infection remained lower compared to those with symptomatic infection (RR=0.72; 95% CI, 0.51 to 1.02, p=0.064). White race and frequent recurrences also remained significant predictors of higher subclinical shedding (Table 2).
Predictors of Genital Lesions
In univariate analyses, persons with asymptomatic HSV-2 infection had lower rates of genital lesions compared with those with symptomatic HSV-2 infection, 3.8% vs. 13.8%, respectively, (RR=0.28; 95% CI, 0.17 to 0.47, p<0.001; Table 3). No differences in lesion rates were noted by HSV-1 status, with lesion rates of 11.4% for persons infected with both HSV-1 and HSV-2 compared to 12.5% for persons with HSV-2 only (RR=0.92; 95% CI, 0.72 to 1.18, p=0.50). Persons of white race had a higher genital lesion rate compared to persons of other races, 13.3% vs. 6.5% (RR=2.06; 95% CI, 1.37 to 3.10, p=0.001).
Table 3.
Predictors of HSV-2 Genital Lesion Ratea
| Parameter | Unadjusted Risk Ratio (95% CI) | Unadjusted P-Value | Adjusted Risk Ratio (95% CI) | Adjusted P-value |
|---|---|---|---|---|
| History of Infection of HSV-2 | ||||
| Symptomatic | Ref | |||
| Asymptomatic | 0.28 (0.17, 0.47) | <0.001 | 0.27 (0.16, 0.45) | <0.001 |
| Age | ||||
| Age 41+ years | Ref | |||
| Age 31-40 years | 1.25 (0.94, 1.66) | 0.13 | ||
| Age under 30 years | 1.36 (0.99, 1.86) | 0.056 | ||
| Age Unknownb | 0.38 (0.19, 0.75) | 0.006 | ||
| Gender | ||||
| Male | Ref | |||
| Female | 0.99 (0.77, 1.26) | 0.92 | ||
| Race | ||||
| Other | Ref | |||
| White | 2.06 (1.37, 3.10) | 0.001 | 2.15 (1.44, 3.21) | <0.001 |
| Years Since Acquisition | ||||
| 11-40 Years Since Acquisition | Ref | |||
| 1-10 Years Since Acquisition | 1.24 (0.88, 1.77) | 0.23 | ||
| <1 Year Since Acquisition | 1.18 (0.90, 1.55) | 0.24 | ||
| Unknown Number of Years | 0.29 (0.18, 0.46) | <0.001 | ||
| Since Acquisitionc | ||||
| Sexual Preference | ||||
| MSM and Bisexual Men | Ref | |||
| Heterosexual Mend | 1.42 (0.97, 2.09) | 0.073 | ||
| Women | 1.22 (0.86, 1.72) | 0.27 | ||
| HSV Serology | ||||
| HSV-2 only | Ref | |||
| HSV 1 & 2 | 0.92 (0.72, 1.18) | 0.50 |
The genital shedding rate was calculated per participant using the number of days in which HSV DNA was detected divided by the total number of days with genital swabs.
Age unknown for 41 persons.
Years since acquisition unknown for 136 persons, including all 88 persons reporting asymptomatic infection.
34 men with unknown sexual preference are categorized in this group.
In the multivariate model, the risk for genital lesions among persons with asymptomatic HSV-2 infection remained lower compared to those with symptomatic infection (RR=0.27; 95% CI, 0.16 to 0.45, p<0.001). Persons reporting white race also had a persistently higher genital lesion rate.
Recognition of clinical genital herpes among asymptomatic persons with HSV-2 infection
Among the 88 person who had asymptomatic HSV-2 infection, 19 persons reported genital signs and symptoms during follow up. The participants who recognized lesions were slightly older that those who remained asymptomatic (mean age 45 ,SD 9.6 vs. 42, SD 9.5 years of age, p<0.001), but there were no difference in gender, race or frequency of HSV-1 coinfection. The genital HSV shedding rate was 18.7% (227 of 1211 days) among persons in the asymptomatic group who reported genital lesions during the follow-up compared to 7.6% (292 of 3859 days) among the persons who remained without any clinical manifestations of HSV-2 infection (RR=2.47; 95% CI, 1.46 to 4.19, p<0.001). The median log10 copy number among the participants who reported lesions was 4.8 (IQR, 3.7-6.6) compared with 4.1 (IQR, 2.9-5.4) among those who remained asymptomatic (p<0.001). Among persons in the asymptomatic group who reported genital lesion during follow up, the subclinical viral shedding rate was 13.3% (142 of 1070 days) compared to 7.6 % (292 of 3859) days among persons who remained without any clinical manifestations of HSV-2 infection (RR=1.86; 95% CI, 1.03 to 3.38, p=0.041), but the copy number on subclinical shedding days was similar (data not shown).
Comment
The uncertainty regarding the optimal management of asymptomatic persons who test HSV-2 seropositive derives in part from lack of information regarding the natural history of such infection. To address this gap, we prospectively evaluated a large cohort of HSV-2 seropositive persons to determine the patterns of genital viral shedding among persons with symptomatic and asymptomatic genital HSV-2 infection. We found that the risk for genital shedding was twice as high, and the risk for lesions almost three times as high among persons with symptomatic genital HSV-2 infection. However, even among persons with asymptomatic HSV-2 infection, genital HSV shedding occurred on 10% of days, and almost all of it – 84% – was subclinical. The quantity of virus (the median genital log10 copy number) shed subclinically was similar in persons with symptomatic and asymptomatic infection. Lesions were associated with a higher quantity of virus shed compared to subclinical shedding. In addition, higher quantity of virus shed was associated with longer shedding episode duration.
These findings extend earlier observation that genital HSV shedding in persons who are seropositive for this virus is likely universal, but that the clinical manifestations of disease differ widely.5, 8 Recent reports of CD4+ and CD8+ T cell infiltrate into genital lesions that appears to persist for many months in the genital tract even in the absence of clinically evident ulcerations suggest that the immune system is actively surveilling the sites of HSV-2 reactivation.19 However, the immune containment is imperfect, reflecting either inability of the immune response to control the reactivation, or the almost constant release of herpes simplex virions from neuronal endings,7 or both.
Despite the observation that biologically HSV-2 infection is best characterized along a continuum, we dichotomized our population into “symptomatic” and “asymptomatic” to mirror how the diagnosis is made in clinical practice: mostly on the basis of clinical presentation, but increasingly by type-specific serologic assays, which became commercially available in US in 1999. A similar dichotomy is often used in serological surveys that assess whether HSV-2 antibody is associated with prior diagnosis of genital herpes; usually only 10-25% of HSV-2 seropositive persons will have a history of genital herpes.5, 20, 21 Further work is required to define to what extent this dichotomy reflects viral or host factors. However, even among persons with a history of genital HSV-2 infection, the spectrum of clinical disease is large, and our findings show that virological spectrum is also broad, with substantial overlap across the two groups. In addition, a substantial proportion of initially asymptomatic persons will recognize recurrent genital herpes once diagnosed and educated,5 as evidenced by the participants in this study with “asymptomatic” infection who noted genital lesions during the follow up period. The lowest rates of viral shedding were among those who remained asymptomatic throughout the study, although HSV-2 was still detected on more than 5% of days.
Our study collected over 28,000 genital swabs from 498 persons infected with HSV-2. Because the collection of genital swabs is time consuming, it is possible that participants who were concerned about genital HSV-2 infection were more likely to participate in this study. Since participants with asymptomatic HSV-2 infection were recruited in the absence of symptoms, it is unlikely that the asymptomatic cohort was inherently biased towards individuals with more severe HSV-2 infections. However, it is possible that a population-based study of genital herpes would have found milder disease among participants with a history of genital herpes. All genital swabs used for analysis were self-collected, as we have shown previously that such swabs are as likely to yield virus as those collected by clinicians.22 Our prior work has established that despite variations in the number of swabs collected by each participant, the shedding rates accurately represent virologic behavior.8
Our large cohort allowed us to detect several associations between demographic and clinical characteristics of the participants and the viral shedding. Several of the associations were of small magnitude and their clinical relevance is uncertain. Of note is the similar shedding rate between men and women, both for overall and subclinical shedding, confirming that men have subclinical shedding on normal appearing genital skin. In addition, infection with HSV-1 does not impact the frequency of HSV-2 reactivation, outside the early initial genital HSV-2 acquisition.23 In this cohort, non-white persons had a lower viral shedding rate, but they represented a relatively small proportion of the cohort. Other studies have also suggested a differential response of African-Americans to famciclovir.24 As host genetic predictors of severity of HSV disease are identified, further work is needed to understand the potential for the gene-environment interactions in this infection.25, 26
The finding that the quantity of virus shed during subclinical episodes is comparable in the symptomatic and asymptomatic persons underscores the epidemiologic observations that the risk of HSV-2 transmission is high from persons with unrecognized HSV-2 infection. Most likely, there is a relationship between the risk of HSV-2 transmission and frequency and quantity of virus shed. In HIV, where this relationship has been characterized, the risk is higher at higher HIV RNA, but the population that has very high viral load may be relatively small.27, 28 Thus the risk of transmission is likely a function of both the frequency of shedding and the amount of virus present. The inoculum necessary for HSV-2 transmission to sexual partners is unknown, but neonatal HSV transmission as well as findings from the valacyclovir transmission trial suggest that even a quantitatively moderate shedding episode can result in transmission.7, 29, 30
Our findings suggest that “best practices” management of HSV-2 infected persons who learn that they are infected from serologic testing should include anticipatory guidance with regard to genital symptoms, as well as counseling regarding the potential for transmission. The issue of infectivity is both a patient management and a public health concern. The primary concern of many HSV-2 seropositive persons is the risk of transmission to sexual partners; and in our experience this is the main source of angst in patients with genital herpes.31 Several methods have been identified that partly reduce the risk of HSV-2 transmission to sexual partners. Condom use, daily valacyclovir therapy, and disclosure of HSV-2 serostatus each approximately halve the risk of HSV-2 transmission.30, 32, 33 However, these approaches reach a small portion of the population, and have not had an impact on HSV-2 seroprevalence in the last decade 4. One of the reasons for such a limited impact is that few people are aware of their genital HSV-2 infection, and routine serologic testing, while available commercially, is recommended only in very limited settings.34 We hope that these data will result in further discussions regarding control programs for HSV-2 in the United States.
Acknowledgments
We are indebted to the study participants, as well as the study clinicians. This work was supported by NIH Grant PO1 AI-30731 and K24 AI 071113. The senior author, AW, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The funding source, NIH, has had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
References
- 1.Wald A, Corey L, Cone R, Hobson A, Davis G, Zeh J. Frequent genital herpes simplex virus 2 shedding in immunocompetent women. Effect of acyclovir treatment. J Clin Invest. 1997 Mar 1;99(5):1092–1097. doi: 10.1172/JCI119237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mertz KJ, Trees D, Levine WC, et al. Etiology of genital ulcers and prevalence of human immunodeficiency virus coinfection in 10 US cities. The Genital Ulcer Disease Surveillance Group. J Infect Dis. 1998 Dec;178(6):1795–1798. doi: 10.1086/314502. [DOI] [PubMed] [Google Scholar]
- 3.Looker KJ, Garnett GP, Schmid GP. An estimate of the global prevalence and incidence of herpes simplex virus type 2 infection. Bulletin of the World Health Organization. 2008;86:805–812A. doi: 10.2471/BLT.07.046128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Seroprevalence of herpes simplex virus type 2 among persons aged 14-49 years--United States, 2005-2008. MMWR Morb Mortal Wkly Rep. 2010 Apr 23;59(15):456–459. [PubMed] [Google Scholar]
- 5.Wald A, Zeh J, Selke S, et al. Reactivation of genital herpes simplex virus type 2 infection in asymptomatic seropositive persons. N Engl J Med. 2000 Mar 23;342(12):844–850. doi: 10.1056/NEJM200003233421203. [DOI] [PubMed] [Google Scholar]
- 6.Mertz GJ. Asymptomatic shedding of herpes simplex virus 1 and 2: implications for prevention of transmission. J Infect Dis. 2008 Oct 15;198(8):1098–1100. doi: 10.1086/591914. [DOI] [PubMed] [Google Scholar]
- 7.Schiffer JT, Abu-Raddad L, Mark KE, et al. Frequent Release of Low Amounts of Herpes Simplex Virus From Neurons: Results of a Mathematical Model. Sci Transl Med. 2009 Nov 1;1(7):7ra16. doi: 10.1126/scitranslmed.3000193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Magaret AS, Johnston C, Wald A. Use of the designation “shedder” in mucosal detection of herpes simplex virus DNA involving repeated sampling. Sex Transm Infect. 2009 Aug;85(4):270–275. doi: 10.1136/sti.2008.034751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mark K, Wald A, Magaret A, et al. Rapidly Cleared Episodes of Herpes Simplex Virus Reactivation in Immunocompetent Adults. The Journal of Infectious Diseases. 2008;198(8):1141–1149. doi: 10.1086/591913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wald A, Zeh J, Barnum G, Davis LG, Corey L. Suppression of subclinical shedding of herpes simplex virus type 2 with acyclovir. Ann Intern Med. 1996 Jan 1;124(1 Pt 1):8–15. doi: 10.7326/0003-4819-124-1_part_1-199601010-00002. [DOI] [PubMed] [Google Scholar]
- 11.Ashley RL, Militoni J, Lee F, Nahmias A, Corey L. Comparison of Western blot (immunoblot) and glycoprotein G-specific immunodot enzyme assay for detecting antibodies to herpes simplex virus types 1 and 2 in human sera. J Clin Microbiol. 1988 Apr;26(4):662–667. doi: 10.1128/jcm.26.4.662-667.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ryncarz AJ, Goddard J, Wald A, Huang ML, Roizman B, Corey L. Development of a high-throughput quantitative assay for detecting herpes simplex virus DNA in clinical samples. J Clin Microbiol. 1999 Jun;37(6):1941–1947. doi: 10.1128/jcm.37.6.1941-1947.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Magaret AS, Wald A, Huang ML, Selke S, Corey L. Optimizing PCR positivity criterion for detection of herpes simplex virus DNA on skin and mucosa. J Clin Microbiol. 2007 May;45(5):1618–1620. doi: 10.1128/JCM.01405-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Corey L, Holmes KK. Genital herpes simplex virus infections: current concepts in diagnosis, therapy, and prevention. Ann Intern Med. 1983 Jun;98(6):973–983. doi: 10.7326/0003-4819-98-6-973. [DOI] [PubMed] [Google Scholar]
- 15.Benedetti J, Corey L, Ashley R. Recurrence rates in genital herpes after symptomatic first-episode infection. Ann Intern Med. 1994 Dec 1;121(11):847–854. doi: 10.7326/0003-4819-121-11-199412010-00004. [DOI] [PubMed] [Google Scholar]
- 16.Phipps WSM, Magaret A, Selke S, Remington M, Huang ML, Warren T, Casper C, Corey L, Wald A. Persistent genital herpes simplex virus-2 shedding years following the first clinical episode. The Journal of Infectious Diseases. 2011 doi: 10.1093/infdis/jiq035. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Diggle P. Analysis of longitudinal data. 2nd ed. Oxford University Press; New York, NY: 2002. [Google Scholar]
- 18.Mertz GJ, Benedetti J, Ashley R, Selke SA, Corey L. Risk factors for the sexual transmission of genital herpes. Ann Intern Med. 1992 Feb 1;116(3):197–202. doi: 10.7326/0003-4819-116-3-197. [DOI] [PubMed] [Google Scholar]
- 19.Zhu J, Koelle DM, Cao J, et al. Virus-specific CD8+ T cells accumulate near sensory nerve endings in genital skin during subclinical HSV-2 reactivation. J Exp Med. 2007 Mar 19;204(3):595–603. doi: 10.1084/jem.20061792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fleming DT, McQuillan GM, Johnson RE, et al. Herpes simplex virus type 2 in the United States, 1976 to 1994. N Engl J Med. 1997 Oct 16;337(16):1105–1111. doi: 10.1056/NEJM199710163371601. [DOI] [PubMed] [Google Scholar]
- 21.Cowan FM, Johnson AM, Ashley R, Corey L, Mindel A. Antibody to herpes simplex virus type 2 as serological marker of sexual lifestyle in populations. Bmj. 1994 Nov 19;309(6965):1325–1329. doi: 10.1136/bmj.309.6965.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wald A, Zeh J, Selke S, Ashley RL, Corey L. Virologic characteristics of subclinical and symptomatic genital herpes infections. N Engl J Med. 1995 Sep 21;333(12):770–775. doi: 10.1056/NEJM199509213331205. [DOI] [PubMed] [Google Scholar]
- 23.Sucato G, Wald A, Wakabayashi E, Vieira J, Corey L. Evidence of latency and reactivation of both herpes simplex virus (HSV)-1 and HSV-2 in the genital region. J Infect Dis. 1998 Apr;177(4):1069–1072. doi: 10.1086/515261. [DOI] [PubMed] [Google Scholar]
- 24.Leone P, Abudalu M, Mitha E, Gani M, Zhou W, Hamed K. One-day famciclovir vs. placebo in patient-initiated episodic treatment of recurrent genital herpes in immunocompetent Black patients. Curr Med Res Opin. 2010 Mar;26(3):653–661. doi: 10.1185/03007990903554471. [DOI] [PubMed] [Google Scholar]
- 25.Bochud PY, Magaret AS, Koelle DM, Aderem A, Wald A. Polymorphisms in TLR2 are associated with increased viral shedding and lesional rate in patients with genital herpes simplex virus Type 2 infection. J Infect Dis. 2007 Aug 15;196(4):505–509. doi: 10.1086/519693. [DOI] [PubMed] [Google Scholar]
- 26.Casrouge A, Zhang SY, Eidenschenk C, et al. Herpes simplex virus encephalitis in human UNC-93B deficiency. Science. 2006 Oct 13;314(5797):308–312. doi: 10.1126/science.1128346. [DOI] [PubMed] [Google Scholar]
- 27.Celum C, Wald A, Lingappa JR, et al. Acyclovir and transmission of HIV-1 from persons infected with HIV-1 and HSV-2. N Engl J Med. 2010 Feb 4;362(5):427–439. doi: 10.1056/NEJMoa0904849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lingappa JR, Hughes JP, Wang RS, et al. Estimating the impact of HIV-1 plasma RNA reductions on heterosexual HIV-1 transmission risk. PLoS One. 2010 13 Sep;5(9):e12598. doi: 10.1371/journal.pone.0012598. 2010. doi:12510.11371/journal.pone.0012598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cone RW, Hobson AC, Brown Z, et al. Frequent detection of genital herpes simplex virus DNA by polymerase chain reaction among pregnant women. Jama. 1994 Sep 14;272(10):792–796. [PubMed] [Google Scholar]
- 30.Corey L, Wald A, Patel R, et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med. 2004 Jan 1;350(1):11–20. doi: 10.1056/NEJMoa035144. [DOI] [PubMed] [Google Scholar]
- 31.Melville J, Sniffen S, Crosby R, et al. Psychosocial impact of serological diagnosis of herpes simplex virus type 2: a qualitative assessment. Sex Transm Infect. 2003 Aug;79(4):280–285. doi: 10.1136/sti.79.4.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wald A, Langenberg AG, Link K, et al. Effect of condoms on reducing the transmission of herpes simplex virus type 2 from men to women. Jama. 2001 Jun 27;285(24):3100–3106. doi: 10.1001/jama.285.24.3100. [DOI] [PubMed] [Google Scholar]
- 33.Wald A, Krantz E, Selke S, Lairson E, Morrow RA, Zeh J. Knowledge of partners’ genital herpes protects against herpes simplex virus type 2 acquisition. J Infect Dis. 2006 Jul 1;194(1):42–52. doi: 10.1086/504717. [DOI] [PubMed] [Google Scholar]
- 34.Guerry SL, Bauer HM, Klausner JD, et al. Recommendations for the selective use of herpes simplex virus type 2 serological tests. Clin Infect Dis. 2005 Jan 1;40(1):38–45. doi: 10.1086/426438. [DOI] [PubMed] [Google Scholar]