Abstract
Inhibition is a physiological process that decreases the probability of a neuron generating an action potential. The two main mechanisms that have been proposed for inhibition are hyperpolarization and shunting. Shunting results from increased membrane conductance, and it reduces the neuron-firing probability. Here we show that ambient GABA, the main inhibitory neurotransmitter in the brain, can excite adult hippocampal interneurons. In these cells, the GABAA current reversal potential is depolarizing, making baseline tonic GABAA conductance excitatory. Increasing the tonic conductance enhances shunting-mediated inhibition, which eventually overpowers the excitation. Such a biphasic change in interneuron firing leads to corresponding changes in the GABAA-mediated synaptic signalling. The described phenomenon suggests that the excitatory or inhibitory actions of the current are set not only by the reversal potential, but also by the conductance.
 Ambient levels of the neurotransmitter GABA tonically activate GABAA. Song et al. show that GABA can have both excitatory and inhibitory effects on hippocampal interneurons and find that low levels of GABA-mediated conductance are excitatory, whereas higher levels result in shunting inhibition.
Ambient levels of the neurotransmitter GABA tonically activate GABAA. Song et al. show that GABA can have both excitatory and inhibitory effects on hippocampal interneurons and find that low levels of GABA-mediated conductance are excitatory, whereas higher levels result in shunting inhibition.
GABA is the main inhibitory neurotransmitter in the adult brain. Synaptically released GABA activates postsynaptic GABAA receptors, which trigger fast inhibitory postsynaptic currents (IPSCs). Ambient GABA produces persistent activation of extrasynaptic GABAA receptors, which are responsible for the tonic current1,2,3. The tonic current is mediated by heterogeneous and plastic GABAA receptors4,5 and is expressed in a cell-type specific manner in various brain structures, such as the hippocampus6, thalamus7, neocortex8 and striatum9. Cell type-specific expression of the tonic current indicates the importance of this phenomenon for neuronal network operation, especially when the extracellular GABA concentration changes. Indeed, changes in ambient GABA levels are reported in both physiological (for example, during exploratory behaviour) and pathological (for example, epilepsy, pain) conditions10,11,12.
The effect of GABA on cell excitability is largely determined by the reversal potential for Cl−, which is the major ion responsible for the GABAA current. In adult neurons, the intracellular Cl− concentration is kept low by developmentally regulated cotransporters13,14 and sets a reversal potential for GABAA currents (EGABA) close to the resting membrane potential (RMP). The RMP and EGABA vary among cell types and even cell compartments15,16; therefore, the effect of GABA can be either hyperpolarizing or depolarizing. Whereas hyperpolarization is the primary synaptic action of GABA on cortical principal neurons17, depolarizing effects of GABA are observed in interneurons in numerous circuits, including the hippocampus18,19,20,21, cerebellum22, and basolateral amygdala23; as well as in striatal neurons9,24 and peripheral nervous system neurons25. Because this depolarization is set by a small driving force for Cl−, the predominant effect of GABAA activation is thought to be shunting inhibition.
Shunting inhibition is a conductance-dependent phenomenon and may not provide strong inhibitory effects on the cell at low GABAA conductances. In contrast, even a small depolarization of the neuronal membrane can trigger activation of some voltage dependent channels. In turn, activation of these channels can potentially bring the RMP to the action potential threshold. This raises the possibility that low depolarizing GABAA conductance can be excitatory whereas high conductance is inhibitory though shunting26. Previous reports suggested that voltage-dependent oscillations of the membrane potential in hippocampal interneurons could reach the action potential threshold and trigger cell firing27,28,29. Indeed, hippocampal CA1 stratum (str.) radiatum interneurons could fire spontaneous action potentials even when excitatory neurotransmission was blocked. Here we found that the firing frequency of these cells is bidirectionally regulated by ambient GABA concentrations. Low GABA concentrations increased both membrane potential fluctuations and cell firing; high concentrations decreased these two parameters below the control values.
Results
Biphasic effect of tonic conductance on interneuron firing
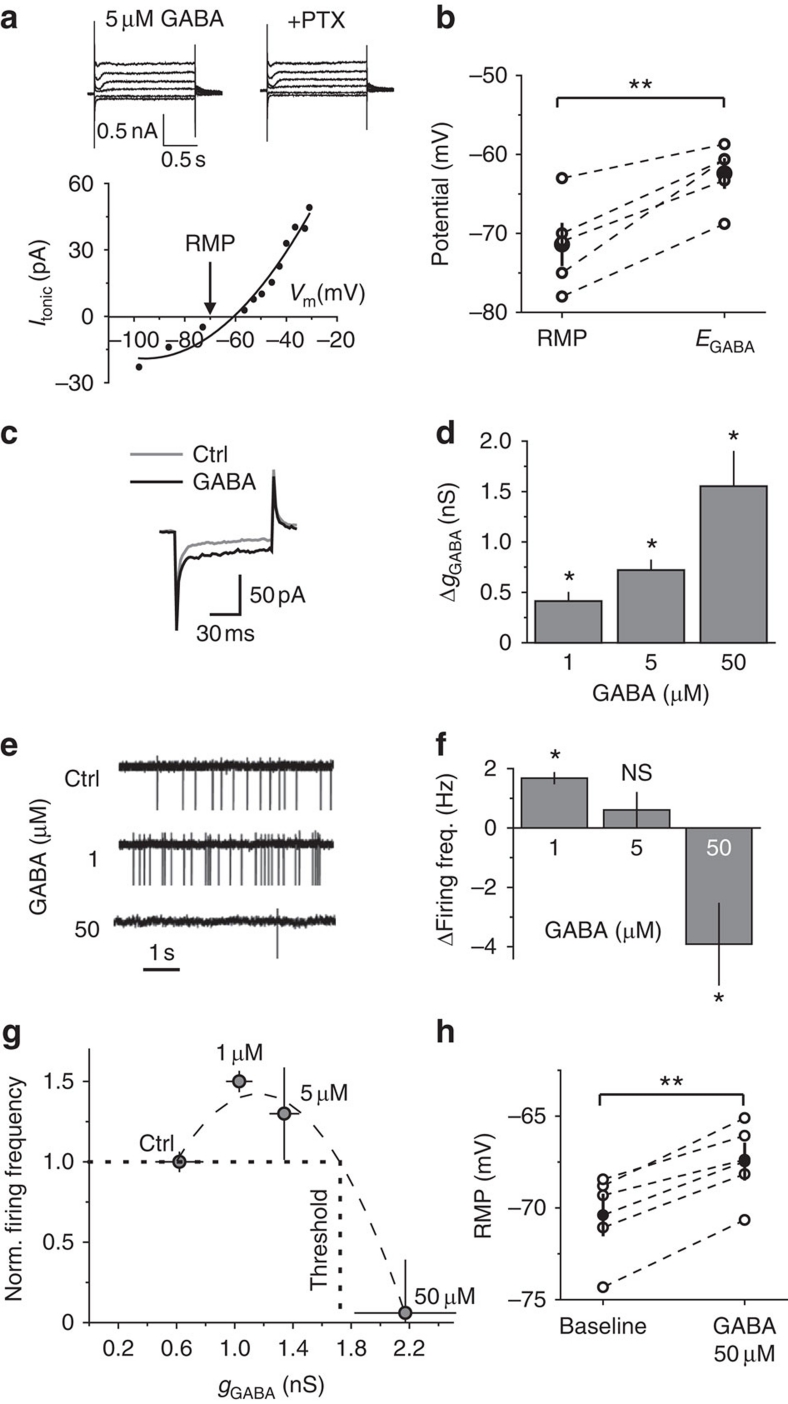
We investigated the effect of tonic GABAA conductance on CA1 str. radiatum interneurons in mouse hippocampal slices and blocked ionotropic glutamate and GABAB receptors. Consistent with previous reports, gramicidin-perforated patch recordings showed that these cells have depolarizing EGABA (RMP, −71.4±2.7 mV n=5; EGABA, −62.4±2.7, n=5, P=0.003 paired t-test; Fig. 1a,b)18,19,20,21. As expected, exogenous GABA produced a concentration-dependent increase in the tonic GABAA conductance (ΔgGABA, 0.41±0.08 nS, 0.72±0.09 nS, and 1.55±0.34 nS for 1, 5, and 50 μM GABA, respectively, n=8; Fig. 1c,d)3,6. In cell-attached recordings, the interneurons could fire spontaneous action potentials despite the blockade of ionotropic glutamate receptors (Supplementary Fig. S1). In contrast to the commonly held notion that GABA mediates inhibitory tonic conductance1,2,30, however, 1 μM GABA increased interneuron firing by 1.68±0.18 Hz (n=7, P=0.028 paired t-test). Cell firing did not significantly change with 5 μM GABA (increase by 0.6±0.6 Hz, n=5, P=0.2 paired t-test), and significantly decreased with 50 μM GABA by 3.9±1.3 Hz (n=5, P=0.023 paired t-test; Fig. 1e,f). These findings suggest a biphasic effect of tonic GABAA conductance on cell excitability. Therefore, we established the inhibitory threshold conductance at which GABA switches its action from excitatory to inhibitory and the peak excitation conductance at which GABA produces maximal excitatory effect. We plotted the cell firing rate versus the corresponding tonic GABAA conductance (gGABA=baseline tonic conductance+ΔgGABA). The inhibitory threshold conductance (1.73 nS) and the peak excitation conductance (1.16 nS) were obtained from a second-order polynomial fit (Fig. 1g). These two parameters characterize the cell response to the change in ambient GABA concentration and are potentially important for computation within individual interneurons and local networks.
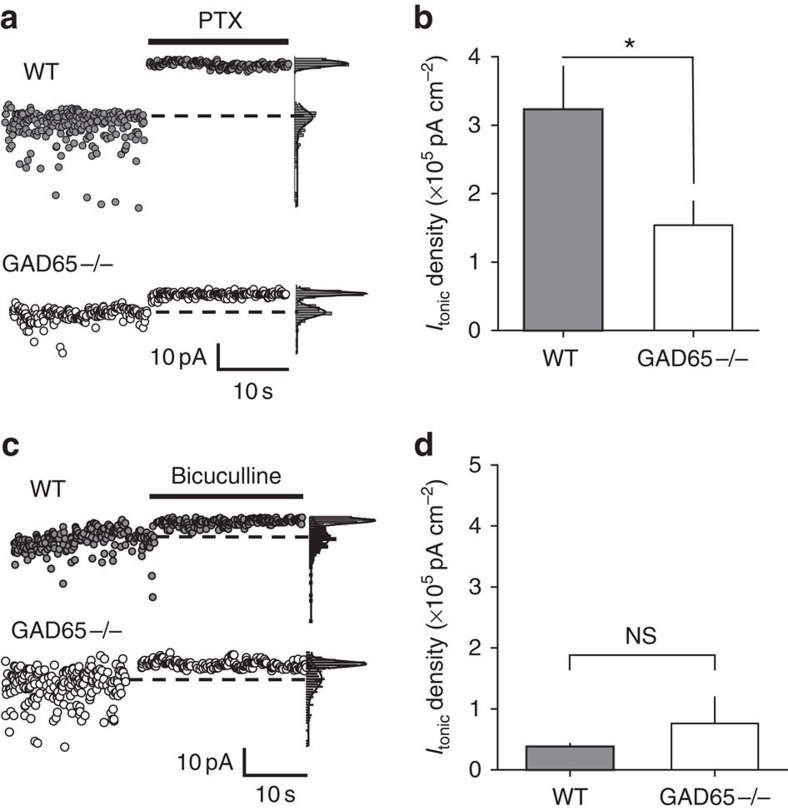
Figure 1. The effect of ambient GABA on interneuron membrane properties and firing.
(a) Top, Gramicidin-perforated patch recordings at voltage steps from −90 to −20 mV in 5 μM GABA and, thereafter 100 μM picrotoxin (+PTX) was added. Tonic GABAA currents at different voltages were calculated by subtracting the '+PTX'-currents from corresponding '5 μM GABA'-currents. Bottom: Tonic current (Itonic) versus membrane potential (Vm). Points were fitted with a second-order polynomial function to determine EGABA. Arrow marks RMP. (b) Summary data of RMP and EGABA showing data from individual cells (n=5, empty circles) and mean values (black circles). (c) Five averaged current traces induced by −5 mV voltage step in control (ctrl) and in 50 μM GABA, demonstrating change in membrane conductance. (d) Summary plot of the GABA-induced change in membrane conductance (ΔgGABA) (n=7 for 1 μM GABA, n=5 for 5 μM GABA, n=5 for 50 μM GABA). (e) Cell-attached recordings from interneurons in control and in 1 and 50 μM GABA. (f) Summary plot of the change in firing frequency induced by GABA. (g) Summary plot of normalized firing frequency versus tonic GABAA conductance (gGABA) with a second order polynomial fit (dashed line). Dotted lines indicate the threshold conductance. (h) Effect of 50 μM GABA on RMP in individual cells (n=5, empty circles) and mean values (black circles). Error bars, s.e.m., **P<0.005, *P<0.05, NS P>0.05, paired t-test.
According to the above definition of the peak excitation conductance an increase in the tonic GABAA conductance further from this point should decrease the cell firing rate and vice versa. Indeed, a low dose of the GABAA antagonist picrotoxin (1 μM) reversed the inhibitory effect of 10 μM GABA by partial reduction of the tonic GABAA conductance, presumably setting it closer to the peak excitation (Supplementary Fig. S2). It should be noted that picrotoxin at this concentration has only a minor effect on IPSCs6. This finding is consistent with our previous report demonstrating an increase in hippocampal interneuron firing in guinea pigs by partially reducing the tonic conductance6. Baseline tonic GABAA conductance in guinea pig interneurons is almost twice that in mice (guinea pigs, 1.1±0.3 nS, n=7 versus mice, 0.62±0.16 nS, n=7)6 and could be above the excitation peak conductance in these animals, even if still below the inhibitory threshold conductance (excitatory GABA action).
The inactivation of Na+ channels is an alternative explanation for the biphasic effect of tonic GABAA depolarization. This explanation for the inhibitory effect of depolarizing GABA was previously suggested for neurons in the peripheral nervous system31. In fact, GABA could not provide strong depolarization to hippocampal interneurons, even at high conductances (EGABA ~10 mV above RMP). 50 μM GABA produced only 3.1±0.1 mV (n=5, P=0.001 paired t-test) of depolarization in perforated-patch experiments (Fig. 1h). In whole-cell current clamp recordings, we tested the effect of a depolarizing block on the interneuron firing by injecting different amounts of constant current. At first, cell firing increased with the depolarization, but after the depolarization reached −38.7±4.1 mV (~26 mV above RMP, n=5), the firing rate began to decrease, presumably due to inactivation of Na+ channels. This finding indicates that the small amount of depolarization produced by ambient GABA is unlikely to produce a significant depolarizing block in hippocampal interneurons. We cannot completely rule out this possibility, however, because the depolarizing effect of GABA can be larger in the axon initial segment, as it has been demonstrated in principle cells in the context of axo-axonic synaptic connections16,32.
The small depolarization produced by tonic GABAA conductances raises the question that whether it is sufficient to reach the action potential threshold. Consistent with previous reports, the action potential threshold (the value of membrane potential when the first derivative of potential was 30 mV/ms) estimated in whole-cell current clamp recordings was −43.9±2.3 mV (n=8)27. Indeed, the small depolarization produced by tonic GABAA conductance cannot reach this value. The action potential measurements, however, are commonly obtained in the cell soma although the action potential threshold can be lower in the axon trigger zone. Moreover, neuron firing can be triggered not by constant depolarization, but through the enhancement of voltage-dependent membrane potential fluctuations characteristic of interneurons29,33. Indeed, GABA produced a biphasic effect on the standard deviation (s.d.) of the RMP recorded in perforated-patch experiments: 1 μM GABA increased s.d. by 25±8% (n=5, P=0.04 paired t-test), 5 μM produced an insignificant decrease of 10±10% (n=7, P=0.19, paired t-test), and 50 μM decreased the s.d. by 24±9% (n=6, P=0.03 paired t-test). The observed decrease in the RMP fluctuations produced by 50 μM GABA was consistent with the previously reported observations in cell attached recordings21.
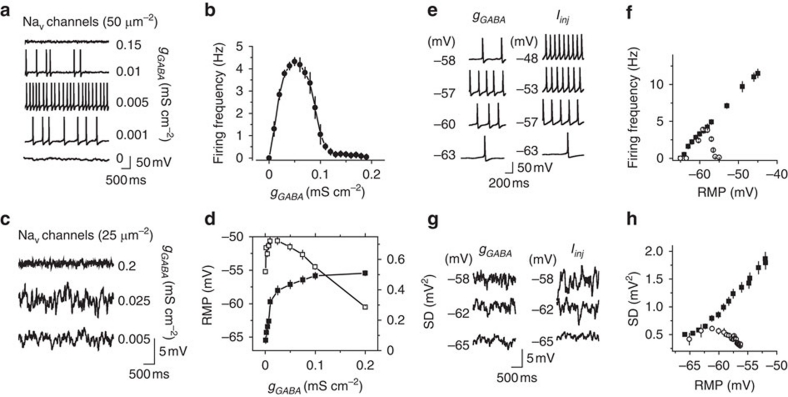
Thus, our results point that low tonic GABAA conductance depolarizes the cell and enhances voltage-dependent fluctuations of the RMP, which can trigger action potentials. When the conductance increases, however, it shunts the fluctuations and prevents cell firing. If this is the case, such a mechanism should work not only in hippocampal interneurons, but also in any neuron with depolarizing GABA. We addressed this issue by developing a simple computer simulation on a cylindrical model cell lacking any interneuron-specific features. The excitability of this cell was based on the Hodgkin–Huxley model with the kinetics of Na+ and K+ channels assuming stochastic opening and closing (Methods). The cell also contained stochastic A-type K+ channels34. Under baseline conditions, the cell received no synaptic input and generated no action potentials, but did show small fluctuations in the RMP (Fig. 2a). A tonic GABAA conductance (gGABA=0–0.2 mS cm-2) with depolarizing EGABA (10.2 mV above RMP) was then introduced. This produced a biphasic change in the cell firing frequency that was dependent on the gGABA amplitude, similar to the effect observed in hippocampal interneurons (Fig. 2a,b). The density of Na+ channels was then reduced from 50 μm−2 (in initial model) to 25 μm−2 to prevent the model cell from spiking, which allowed us to observe the effect of gGABA on the RMP fluctuations (Fig. 2c). The RMP s.d. initially increased with cell depolarization, and then decreased regardless of the membrane potential (Fig. 2d). Interestingly, the biphasic changes in the cell firing frequency and in the RMP fluctuations were not due to the inactivation of Na+ channels. The depolarizing current injections did not produce any biphasic effect on the above parameters at the same RMP range that was produced by gGABA (Fig. 2e–h). This model demonstrates that the described phenomenon does not require any specific cell anatomy, subcellular gradients of EGABA, or inactivation of Na+ channels; and can be achieved with a limited number of voltage-dependent conductances.
Figure 2. The effect of depolarizing tonic GABAA conductance on a model neuron.
(a) Sample traces of the cell firing induced by gGABA. (b) Summary plot of the cell firing frequency versus gGABA (n=10) (c) Sample traces of the effect of gGABA on RMP fluctuations set by stochastic channel openings. The Na+ channel density was reduced by half compared with panel a to avoid cell firing. (d) Summary plot of RMP (black squares, n=10) and RMP standard deviation (s.d., white squares, n=10) versus gGABA. (e) Representative traces of membrane potential at different RMPs produced by gGABA (left panel) and by Iinj (right panel). (f) Summary data of the change in the model cell firing at different RMPs set by gGABA (empty circles, n=10) and by Iinj (black squares, n=10). (g) Representative traces of membrane potential at different RMPs produced by gGABA (left panel) and by Iinj (right panel). gNa was reduced by half to prevent the cell from firing. (h) Mean standard deviation of RMP (s.d.) at different RMPs set by gGABA (empty circles, n=10) and by Iinj (black squares, n=10). Error bars, s.e.m.
Synaptic GABAA currents are inhibitory
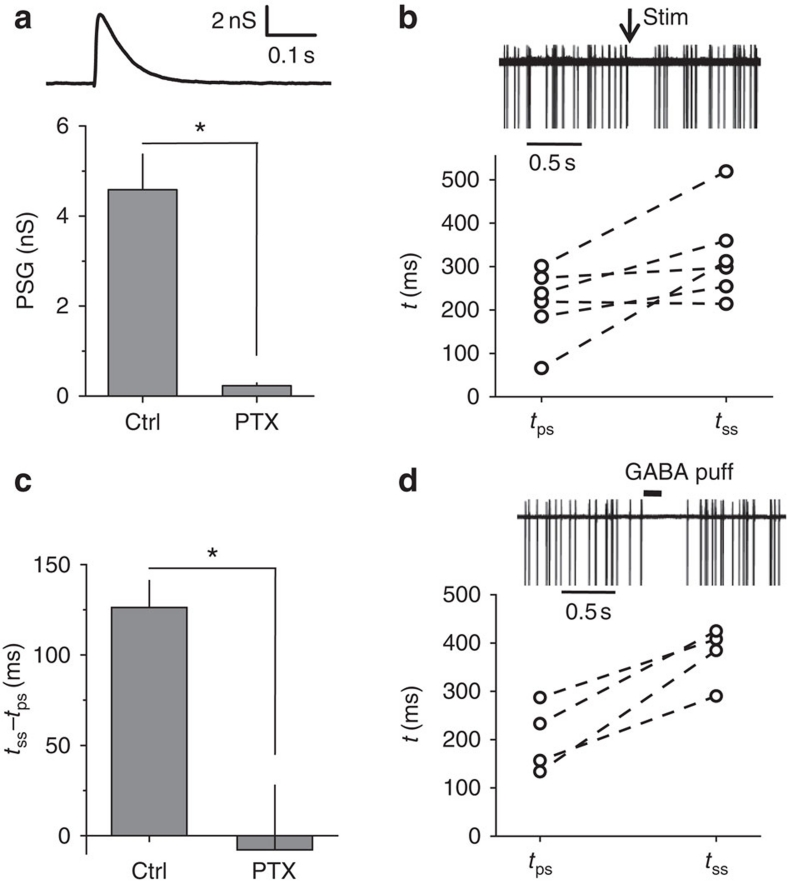
Our findings suggest that whereas a low tonic GABAA conductance can be excitatory, synaptic signalling mediated by high GABAA conductance should provide shunting inhibition in hippocampal interneurons. To test this, we produced stimulations with an extracellular electrode placed proximally to the recorded cell in the str. radiatum. The recordings were performed in the presence of ionotropic glutamate and GABAB receptor antagonists. First, in whole-cell mode, we recorded the evoked GABAA-mediated postsynaptic current (PSC) induced in CA1 interneurons by 50-μA, 200-μs stimulus (Supplementary Fig. S3). We then estimated the postsynaptic conductance (PSG) required to generate such a PSC according using the following formula: PSG=PSC/(EGABA−Vhold), where Vhold is the holding potential (−70 mV). The PSG peak conductance 4.6±0.8 nS (n=7, Fig. 3a) was significantly larger than the conductance required to reach the inhibitory threshold conductance from the baseline tonic GABAA conductance (1.1 nS, calculated as the difference between the inhibitory threshold conductance and baseline tonic GABAA conductance, Fig. 1g). Moreover, the synaptic conductance calculated from PSCs, recorded in the soma, was probably underestimated because some synaptic currents originate from the distal dendrites. Second, we recorded CA1 interneuron firing in the cell-attached mode and produced extracellular stimulation as described above. The time between the stimulus and preceding spike (tps) was assumed to be random; the time between the stimulus and the succeeding spike (tss) was influenced by the PSG. The mean tps and tss were compared for each cell (Fig. 3b). Consistent with the inhibitory synaptic effect, the tss was almost twice as large as the tps (tss/tps, 1.96±0.55, n=6, P=0.011 paired t-test). The difference between tps and tss was completely blocked by 100 μM picrotoxin (tss−tps, 126±14.7 ms in control and −7.8±35 ms in picrotoxin, n=5, P=0.02 paired t-test; Fig. 3c). These data support the notion that GABAA-mediated PSGs in interneurons are inhibitory (IPSGs). Therefore, we use the term spontaneous inhibitory PSCs (sIPSCs) in the following sections for consistency. An alternative explanation for the inhibitory effect of synaptic GABA is that the site of action is different from that of tonic conductance. In a separate set of experiments, we tested the effect of a GABA puff (100 μM×100 ms). Similar to synaptic stimulation, the GABA puff increased the tss by a factor of two (tss/tps, 1.99±0.31, n=4, P=0.005 paired t-test; Fig. 3d). Because puff application produces a local transient increase in GABA, which activates both synaptic and extrasynaptic GABAA receptors, this finding suggests that the conductance magnitude is more important than the site of action.
Figure 3. Evoked GABAA PSG and 100 μM GABA puff are inhibitory.
(a) Top: sample trace of calculated GABAA mediated PSG. Bottom, Summary plot of averaged peak PSG in control (ctrl) and in 100 μM picrotoxin (PTX) (n=7). (b) Top: Five superimposed cell-attached recordings with synaptic stimulation (arrow). Bottom: Plot of averaged preceding spike (tps) and succeeding spike (tss) intervals in 6 individual cells. (c) Difference between tss and tps in control (ctrl) and in 100 μM picrotoxin (PTX) (n=5). (d) Top: 5 superimposed cell-attached recordings with 100 μM GABA puff. Bottom: plot of averaged preceding spike (tps) and succeeding spike (tss) intervals in four individual cells. Error bars, s.e.m., *P<0.05, paired t-test.
Biphasic changes in GABAergic synaptic signalling
Tonic GABAA conductance is commonly referred as 'tonic inhibition'1,2,30 and therefore its function in neuronal networks is thought to be unidirectional. Here we demonstrate that tonic GABAA conductance has a biphasic effect on the hippocampal interneuron firing rate. Interneurons are a diverse group of cells and each class of interneurons has a different function in the local network35. The amount of tonic GABAA conductance also varies among interneuron types8. Thus, our sample of interneurons does not characterize the tonic GABAA current in all classes of interneurons and cannot be used to predict the effect of ambient GABA on the CA1 network. Therefore, we recorded the sIPSC frequency directly in CA1 pyramidal neurons, which are the final targets of the CA1 interneurons and generate the major hippocampal output.
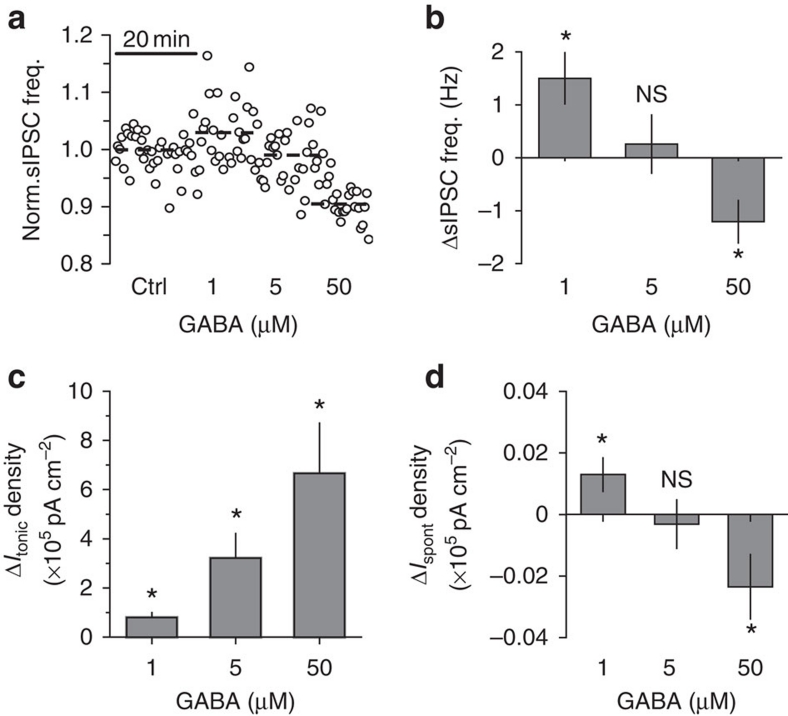
Consistent with the biphasic change in interneuron firing, 1 μM GABA increased sIPSC frequency in the pyramidal cells by 1.5±0.5 Hz (n=11, P=0.005 paired t-test), 5 μM GABA did not produce a significant change (increase by 0.25±0.55 Hz; n=9, P=0.34 paired t-test), and 50 μM GABA reduced the frequency by 1.2±0.4 Hz (n=6, P=0.036 paired t-test; Fig. 4a,b). In this set of experiments, we used relatively high concentrations of exogenous GABA (up to 50 μM). Although it is unlikely that all the GABA reached the neurons deep in the tissue because of its efficient uptake, it is still possible that some of the synaptic GABAA receptors were recruited or desensitised, and the activation of presynaptic GABAA receptors affected the probability of GABA release. To rule out these possibilities, we recorded action-potential independent miniature IPSCs (mIPSCs) under control conditions in the presence of 1 μM tetrodotoxin (Na+ channel blocker) and then added 1, 5 or 50 μM GABA (Supplementary Fig. S4). Neither the amplitude nor frequency of the mIPSCs changed significantly, suggesting the lack of synaptic GABAA receptor activation/desensitisation and the lack of an effect on GABA release probability, respectively.
Figure 4. Effect of ambient GABA on sIPSC and tonic current in CA1 pyramidal neurons.
(a) Normalized sIPSC frequency in CA1 pyramidal neuron in control (ctrl) and after application of 1, 5 and 50 μM GABA. (b) Summary plot of the change in sIPSC frequency induced by GABA (n=11 for 1 μM GABA, n=9 for 5 μM GABA, n=6 for 50 μM GABA). (c) Summary plot of the change in tonic GABAA current (ΔItonic) density recorded in 1, 5 and 50 μM GABA (n=10). (d) Summary plot of the change in time-averaged phasic current (ΔIspont) density recorded in 1, 5 and 50 μM GABA (n=10). Error bars, s.e.m., *P<0.05, NS P>0.05, paired t-test.
Because of efficient GABA uptake, the tonic GABAA current is either absent in pyramidal cells or significantly smaller than that in interneurons4,6. Ambient GABA, however, can increase tonic GABAA current in these cells. Indeed, 1, 5 and 50 μM GABA produced a dose-dependent increase in the tonic GABAA current (Fig. 4c). This increase was two orders of magnitude larger than the change in the time-averaged current mediated by sIPSCs (Ispont), which was calculated as mean charge transfer of sIPSCs multiplied by their frequency6 (Fig. 4d). Thus, the overall effect of bath-applied GABA on the excitability of CA1 pyramidal neurons was dominated by tonic inhibition (Discussion). We next investigated the conditions under which ambient GABA is decreased.
Reduced tonic conductance correlates with low firing rate
We used a mouse lacking one of the two major GABA synthesizing enzymes, 65 kDa glutamic acid decarboxylase (GAD65−/−). This mouse develops an ambient GABA deficit in the brain after the first month of life36,37. Consistent with the reduced extracellular GABA, the tonic GABAA current density was two times smaller in CA1 str. radiatum interneurons of GAD65−/− mice than in wild-type (WT) mice interneurons (GAD65−/−, 1.5±0.3×105 pA cm−2, n=7 versus WT, 3.2±0.6×105 pA cm−2, n=10; P=0.037; Fig. 5a,b). Surprisingly, we detected no difference in the magnitude of the tonic currents between the WT and GAD65−/− CA1 pyramidal neurons (Fig. 5c,d). It is possible that under baseline conditions, ambient GABA does not reach the receptors on these cells6 and the small tonic current is mediated by agonist-independent spontaneous openings of GABAA receptors38,39.
Figure 5. Tonic GABAA current in GAD65−/− interneurons and pyramidal cells.
(a) Change in Ihold in WT and GAD65−/− interneurons produced by 100 μM picrotoxin (PTX). Right panels: Current histograms with Gaussian fits of recorded current. Dashed lines indicate the Ihold obtained from the peaks of the fits. (b) Summary plot of tonic GABAA current (Itonic) density in interneurons (n=7 for GAD65−/−, n=10 for WT). (c) Change in Ihold in WT and GAD65−/− pyramidal cells produced by 10 μM bicuculline. Right panels: same as in a. (d) Summary plot of tonic GABAA current (Itonic) density in WT (n=6) and GAD65−/− (n=4) pyramidal cells. Error bars, s.e.m., NS P>0.05, *P<0.05, unpaired t-test.
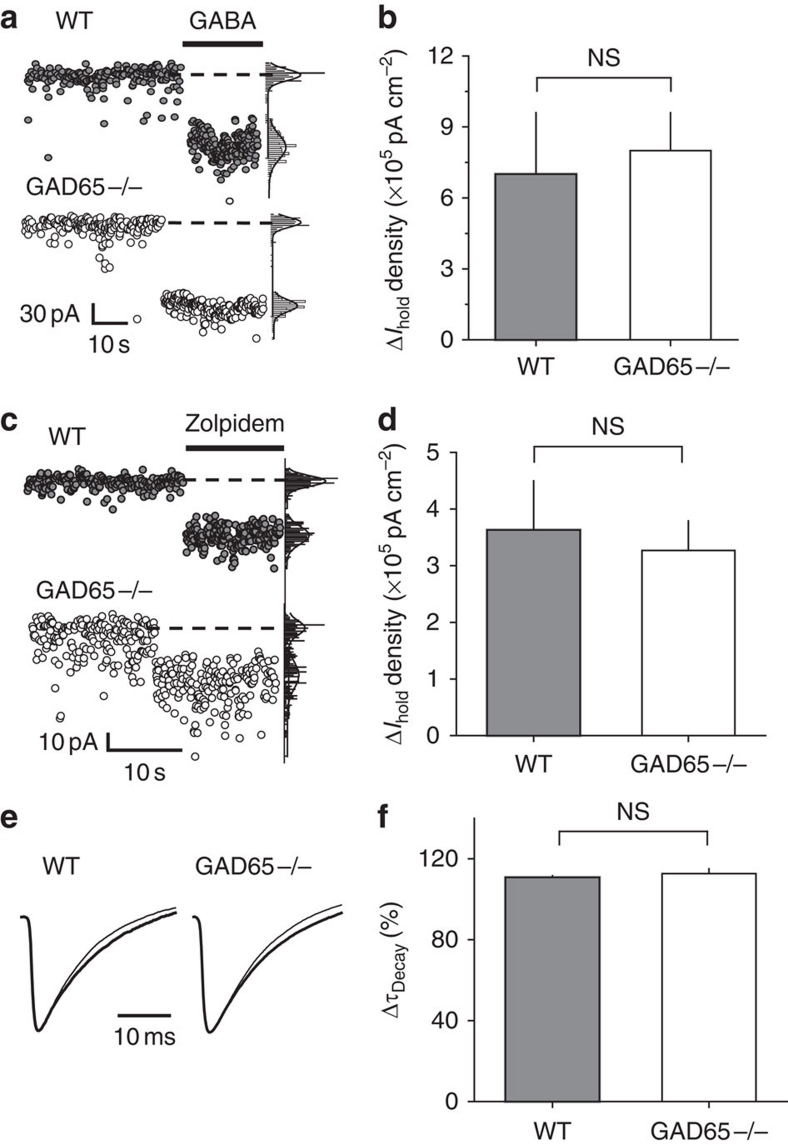
GABA (5 μM) induced a similar rise in the holding current in GAD65−/− and WT interneurons, indicating that the smaller tonic current in knockout animals was not due to a change in their sensitivity to ambient GABA (Fig. 6a,b). Consistent with our previous report6, 100 nM zolpidem, a benzodiazepine site agonist, enhanced the tonic GABAA current and increased the sIPSC decay time in WT interneurons, suggesting that these currents were mediated by γ2-subunit-containing GABAA receptors (Fig. 6c–f). Zolpidem had a similar effect in GAD65−/− interneurons, pointing to an unchanged pharmacological profile of GABAA receptors in these animals40. Also consistent with a previous report41, the amplitude, kinetics and frequency of mIPSCs was not significantly different in GAD65−/− interneurons compared with WT interneurons (Supplementary Fig. S5), suggesting that synaptic signalling was intact under resting conditions. This finding does not exclude, however, the possibility that synaptic GABAA signalling differs between the two genotypes during sustained synaptic activation induced by prolonged electrical stimulation, as previously reported41. Because we did not deliver prolonged stimulation to the slices, this phenomenon is unlikely to contribute to the difference in the tonic currents.
Figure 6. WT and GAD65−/− interneurons equally respond to 5 μM GABA and 100 nM zolpidem.
(a) Change in Ihold in WT and GAD65−/− interneurons produced by 5 μM GABA. Right panels: current histograms with Gaussian fits of recorded current. Dashed lines indicate the Ihold obtained from the peaks of the fits. (b) Summary plot of the change in Ihold density induced by 5 μM GABA in WT (n=10) and GAD65−/− (n=5) interneurons. (c) Change in Ihold in WT and GAD65−/− interneurons produced by 100 nM zolpidem. Right panels: same as in a. (d) Summary plot of the change in Ihold density induced by zolpidem in WT (n=7) and GAD65−/− (n=7) interneurons. (e) Mean peak scaled sIPSCs recorded from WT and GAD65−/− interneurons without (thin trace) or with 100 nM zolpidem (thick trace). (f) Summary plot of the change in the sIPSC decay time constant ( decay) induced by zolpidem in WT (n=7) and GAD65−/− (n=7) interneurons. Error bars, s.e.m., NS P>0.05, unpaired t-test.
decay) induced by zolpidem in WT (n=7) and GAD65−/− (n=7) interneurons. Error bars, s.e.m., NS P>0.05, unpaired t-test.
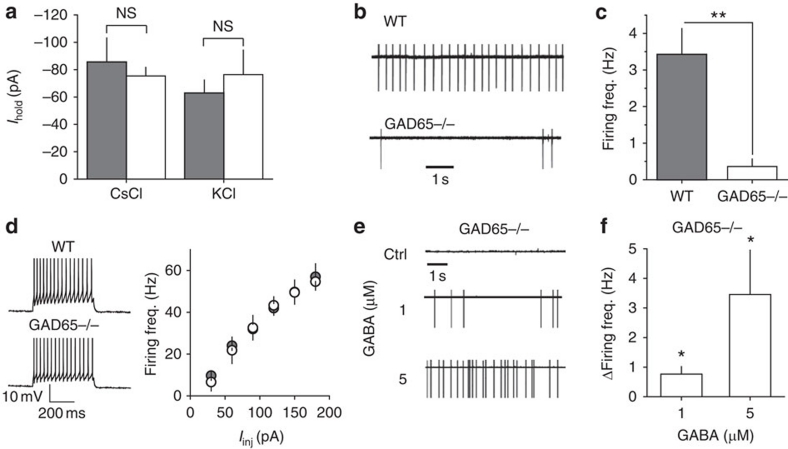
In contrast to GABAA receptor knockout animals42, we found no changes in the non-GABA-mediated holding current in GAD65−/− interneurons, which suggests a lack of compensatory changes in the resting membrane conductances (Fig. 7a). The RMP (−70.6±2.5 mV, n=5) and EGABA (−59.2±1.8 mV, n=5, P=0.039 compared with RMP, paired t-test) in GAD65−/− interneurons were not significantly different from the RMP and EGABA in WT cells.
Figure 7. Analysis of GAD65−/− interneurons.
(a) Summary of Ihold recorded in WT (shaded columns) and GAD65−/− (open columns) interneurons voltage-clamped at −60 mV loaded with CsCl (GAD65−/− n=8; WT n=13) and with KCl (GAD65−/− n=5; WT n=7) in the presence of 100 μM picrotoxin. (b) Cell-attached recordings from WT and GAD65−/− interneurons (without picrotoxin). (c) Summary plot of firing frequency in WT and GAD65−/− interneurons (n=7) obtained under the same conditions as for b. (d) Left panel: Action potentials recorded in response to depolarizing current steps (90 pA; 500 ms) in whole-cell current-clamp mode from WT and GAD65−/− interneurons in the presence of 100 μM picrotoxin. Right panel: Summary plot of action potential frequency recorded during depolarizing steps varying from 30 to 180 pA (input–output characteristics) in GAD65−/− (n=5) and WT (n=4) interneurons. (e) Cell-attached recordings from GAD65−/− interneuron in control (ctrl) and in 1 and 5 μM GABA (without picrotoxin). (f) Summary plot of the change in firing frequency induced by GABA in GAD65−/− interneurons obtained under the same conditions as for e. Error bars, s.e.m., **P<0.005, *P<0.05, NS P>0.05, unpaired (a and c) and paired (f) t-test.
Consistent with the excitatory effect of baseline tonic GABAA conductance, the firing frequency of GAD65−/− interneurons (0.023±0.01 Hz, n=7) was significantly lower than that of WT cells (3.2±0.7 Hz, n=7; P<0.001 compared with WT) (Fig. 7b,c). When GABAA receptors were blocked, the excitability of the GAD65−/− interneurons was not different from the WT cells, ruling out the possibility that compensatory changes were responsible for the reduced firing rate of these cells (Fig. 7d). If a reduced firing rate of GAD65−/− interneurons results from the decreased tonic GABAA conductance, exogenous GABA should rescue it. Indeed, bath application of 1 μM GABA increased the firing rate of GAD65−/− interneurons by 0.8±0.3 Hz (n=6, P=0.015 paired t-test). In contrast to WT interneurons, 5 μM GABA further increased the firing rate of GAD65−/− interneurons by 3.5±1.5 Hz (n=7, P=0.03 paired t-test; Fig. 7e,f). This effect was due to lower baseline tonic conductance in GAD65−/− (0.31±0.12 nS, n=9) than that in WT (0.62±0.16 nS, n=7, P=0.042 for difference with GAD65−/−) interneurons. Both 1 and 5 μM GABA resulted in an increase in tonic conductance (ΔgGABA, 0.48±0.07 nS, n=9 and 1.07±0.15 nS, n=9, respectively) within the range of excitatory GABA action (Fig. 1g). This finding further supports the notion that the decreased firing rates of GAD65−/− interneurons are due to decreased tonic GABAA excitation.
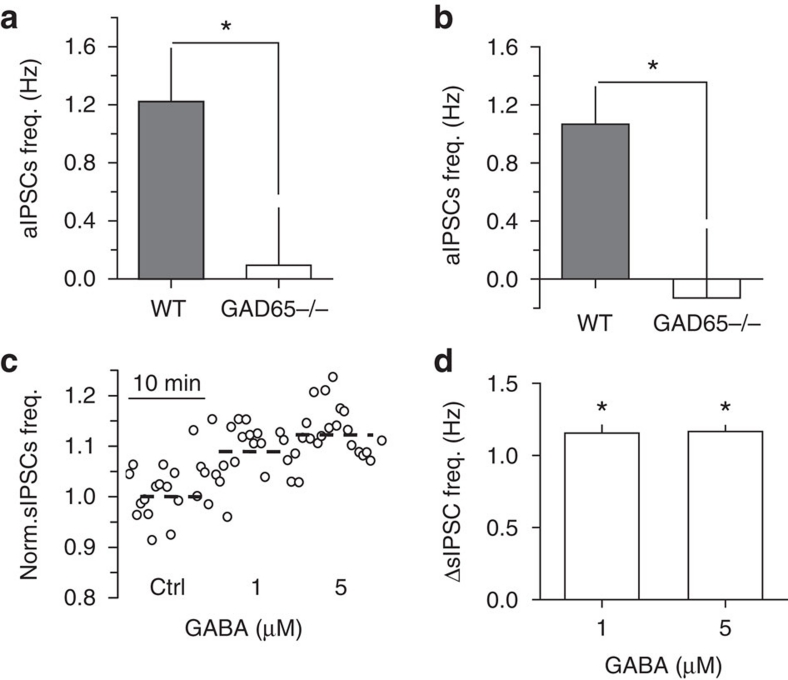
We then compared the baseline frequency of spontaneous action potential-dependent IPSCs (aIPSCs=sIPSCs—mIPSCs) in WT and GAD65−/− neurons to estimate the effect of an ambient GABA deficit on synaptic GABAA signalling driven by interneuron firing. Consistent with interneuron firing rates, the frequency of aIPSCs in WT pyramidal cells (1.2±0.4 Hz, n=4) was significantly higher than the frequency of the practically absent aIPSCs in GAD65−/− pyramidal cells (0.1±0.4 Hz, n=6, P=0.041 compared to WT; Fig. 8a). A similar difference in the frequency of the aIPSCs was observed in the interneurons (WT, 1.1±0.3 Hz, n=13; GAD65−/−, −0.1±0.5 Hz, n=9, P=0.013; Fig. 8b).
Figure 8. GABAA mediated synaptic signalling in GAD65−/− CA1 neurons.
(a, b) Summary plots of aIPSCs frequency in WT and GAD65−/− pyramidal cells (n=4 for WT, n=6 for GAD65−/−) and interneurons (n=13 for WT, n=9 for GAD65−/−), respectively. (c) Normalized sIPSC frequency in GAD65−/− pyramidal neuron in control (ctrl) and after 1 and 5 μM GABA application. (d) Summary plot of the change in sIPSC frequency by GABA in GAD65−/− pyramidal cells (n=8 for 1 μM GABA, n=6 for 5 μM GABA). Error bars, s.e.m., *P<0.05, unpaired (a, b) and paired (d) t-test.
Exogenous GABA increased the frequency of sIPSCs in GAD65−/− pyramidal cells (1 μM, by 1.15±0.05 Hz, n=8, P<0.001 paired t-test, and 5 μM, by 1.13±0.04 Hz, n=6, P<0.001 paired t-test, Fig. 8c,d). Interestingly, this increase in the frequency of sIPSCs was not proportional to the increase in GAD65−/− interneuron firing, whereas 5 μM GABA increased firing frequency several times more than 1 μM (Fig. 7e,f). There are at least two explanations for such a discrepancy. First, using somatic recordings, we were unable to detect sIPSCs from distal dendrites, and thus the frequency change might be underestimated. Second, interneurons target both pyramidal cells and other interneurons. The output of interconnected interneurons to pyramidal cells might not exactly follow the average firing rate of individual interneurons recorded in the str. radiatum. These considerations point to the complexity of the interneuron network and the need for future studies to dissect the effect of tonic excitation on different classes of interneurons. The present data, however, are sufficient to draw the general conclusion that resting levels of ambient GABA are set to maintain the tone of synaptic GABA-mediated signalling in the hippocampal CA1 region.
Discussion
Changes in extracellular GABA determine the magnitude of tonic GABAA conductance, which has a biphasic effect on the excitability of hippocampal interneurons. A low-tonic GABAA conductance depolarizes the cell and results in the onset of cell firing. This depolarization also enhances voltage-dependent RMP fluctuations, which are driven by stochastic Na+ channel opening29. It is likely that these fluctuations are involved in the increase in the cell firing rate, because they get larger following GABAA-mediated depolarization. When the tonic GABAA conductance increases, the excitatory effect of depolarization is overpowered by inhibition. Both cell firing frequency and RMP fluctuations decrease. This inhibition can be explained by the shunting effect, which increases with the tonic GABAA conductance.
Consistent with the effect of shunting, evoked synaptic GABAA signalling was inhibitory in interneurons, because of the large peak conductance. This finding, however, does not rule out the possible excitatory effect of relatively small spontaneous GABAA-mediated postsynaptic currents. Nevertheless, the present finding adds one more distinction to the functional role of tonic and phasic GABA-mediated signalling in the brain1,2,3.
A biphasic effect of tonic GABAA conductance on neuronal firing has important implications for computation within a neuronal network. The classic view holds that neurons generate a variable output in response to the integration of synaptic inputs. Here we show that hippocampal interneurons can generate a variable output in response to changes in extrasynaptic GABA, regardless of excitatory synaptic drive. Because these neurons are part of the local circuit, extrasynaptic input can be considered as one of the inputs into the local neuronal network that determines the network operation and output.
An increase in ambient GABA has been reported for particular types of behaviour (for example, exploration11) or in pathological conditions10,12. We used bath application of GABA to mimic such an increase. Exogenous GABA, however, also induced a dose-dependent tonic current with a magnitude of two orders larger than the time-averaged current produced by the change in sIPSCs in CA1 pyramidal neurons. Because these cells are reported to have hyperpolarizing EGABA (ref. 17), the tonic GABAA current may be their main source of inhibition. This raises the question of whether a biphasic change in sIPSC frequency is of any importance for the excitability of pyramidal neurons. In fact, bath application of GABA may not faithfully reproduce the changes in ambient GABA concentrations under physiological conditions. A recent report using an innovative GABA imaging technique demonstrated layer-specific changes in ambient GABA resulting from increased neuronal activity43. Moreover, the functional roles of tonic and phasic GABAA signalling are largely different2,30. For example, tonic GABAA conductance determines the membrane time constant and characteristic length constant30, and modulates neuronal offset in CA1 pyramidal neurons44. GABAA-mediated IPSCs interact in a time-dependent manner with glutamatergic EPSCs, which has a specific impact on neuronal computation (for example, integration time window of neuronal inputs45), and are involved in network synchronization and oscillatory behaviour46. Thus, the actual effect of physiological changes in ambient GABA concentration on the hippocampal network function requires further investigation.
In summary, we suggest that the described phenomenon has a role in different types of excitable cells. The amount of conductance acts to switch between excitation and inhibition when certain conditions are met: the current has low depolarizing reversal potential and the produced depolarization is amplified by other voltage-dependent conductances. Here we demonstrated these conditions in hippocampal interneurons, but they can be potentially observed in the other neurons with depolarizing EGABA (refs 9,22,23,24,25). It is also likely that EGABA determines the excitation peak conductance and the inhibitory threshold conductance. Thus, the biphasic action of tonic GABAA conductance should be considered in the broader context of the determinants of EGABA. First, the expression of the main Cl− transporters (NKCC1 and KCC2) is developmentally regulated, which differs in various cell types. For example, in hippocampal pyramidal neurons, the ratio of KCC2/NKCC1 increases during development, making GABA hyperpolarizing14. In contrast, this ratio is fixed in hippocampal interneurons and GABA continues to have depolarizing effects throughout development19. Second, activity-dependent regulation of Cl− transporters contributes to EGABA in different cells. A recent report demonstrated that NMDA receptor activity downregulates KCC2, resulting in a depolarizing GABAA-mediated current in rat dissociated neurons47.
Additionally, the described phenomenon can have a role at subcellular level. We did not systematically investigate the effect of low GABA concentrations at different sites within interneurons. A previous report, however, demonstrated that depolarizing GABA can produce inhibition in the soma and excitation in dendrites of cortical pyramidal cells48. This can be explained that shunting inhibition of the axon initial segment is larger for soma than for more remote dendritic sites. That is, the inhibitory threshold conductance increases with distance from the axon initial segment. This observation prompts further investigation of tonic GABAA conductances at the subcellular level.
Methods
Electrophysiology in hippocampal slices
All experimental procedures were performed in accordance with the guidelines of the Animal Experiment Committee of the RIKEN Brain Science Institute. Hippocampal slices (350–380 μm) were obtained from 4- to 6-week-old male WT or GAD65−/− littermates49. Heterozygous knockout mice with a C57BL/6J genetic background were interbred to produce WT and GAD65−/− mice in a specific pathogen-free facility. Mice were housed at 24 °C with a 12/12-h light/dark cycle and ad libitum access to food and water. Genomic PCR was used to differentiate the mice.
Hippocampal slices were maintained at room temperature in a submerged chamber with solution containing (in mM): 119 NaCl, 2.5 KCl, 1.3 MgSO4, 1 CaCl2, 26.2 NaHCO3, 1 NaH2PO4, and 11 glucose, and saturated with 95% CO2/5% O2. After 1 h of incubation, slices were transferred to the recording chamber and superfused at 34 °C with the solution described above, but containing 2.5 mM CaCl2. Except for the experiment shown in Supplementary Figure S1, AMPA/kainate, NMDA, and GABAB receptors were blocked with 25 μM NBQX, 50 μM APV, and 5 μM CGP 52432 (Tocris Cookson), respectively.
CA1 pyramidal cells and str. radiatum interneurons were visually identified with an infrared differential interference contrast microscope (Olympus BX51WI). The data were acquired with a Multiclamp700B (Axon Instruments), filtered at 2 kHz, and digitized at 5 kHz using a NI PCI-6221 data acquisition card (National Instruments). The data were analysed without further re-sampling. Sample traces were also taken at the same rate, except for the long traces (>30 s), which were re-sampled at 10 Hz. Whole-cell, gramicidin-perforated patch, and cell-attached recordings were performed in separate cells.
Whole-cell experiments
Whole-cell pipettes used in voltage-clamp recordings contained (in mM): 130 CsCl, 8 NaCl, 10 Cs-HEPES, 2 EGTA, 0.2 MgCl2, 2 MgATP, 0.3 Na3GTP, and 5 QX314Br (pH 7.2, osmolarity 295 mOsm, liquid junction potential 4.1 mV). In the experiments for Figure 7a, KCl was used instead of CsCl (liquid junction potential 3.4 mV). Whole-cell current-clamp recordings were performed with pipettes containing (in mM): 132.3 K-gluconate, 7.7 KCl, 4 NaCl, 0.5 CaCl2, 10 HEPES, 5 EGTA, 2 MgATP, and 0.5 Na3GTP.
Series resistance (Rs), input resistance (Ri), and membrane capacitance (cm) were monitored throughout the recordings. These parameters were obtained from the current in response to hyperpolarizing voltage steps (Vstep=−5 mV).
| 1 |
| 2 |
| 3 |
where, Ipeak is the peak amplitude of the current transient immediately after the step is applied,  is the decay time constant of the current, and Iss is the steady-state current50. The Rs was not compensated and data were discarded if Rs changed by more than 20% during the recording. There was no significant difference in cm between GAD65−/− (70.9±7.9 pF, n=9) and WT (69.4±6.9 pF, n=11) interneurons (P=0.445).
is the decay time constant of the current, and Iss is the steady-state current50. The Rs was not compensated and data were discarded if Rs changed by more than 20% during the recording. There was no significant difference in cm between GAD65−/− (70.9±7.9 pF, n=9) and WT (69.4±6.9 pF, n=11) interneurons (P=0.445).
Tonic GABAA current (Itonic) was calculated as the difference between the baseline Ihold and the Ihold in the presence of a GABAA receptor antagonist. The change in the tonic GABAA current (ΔItonic) was calculated as the difference between the baseline Ihold and the Ihold in the presence of exogenous GABA. Itonic density was obtained by dividing Itonic by the surface area of the neuron. The surface area was obtained by dividing cm by the membrane capacitance constant (0.9 μF cm-2)50. Tonic GABAA conductance was calculated as the difference between the baseline conductance (1/Ri) and the conductance in the presence of a GABAA receptor antagonist (gGABA) or exogenous GABA (ΔgGABA).
Synaptic GABAA conductance in response to electric stimulation (PSG, Fig. 3a) was calculated according to
| 4 |
where PSC—postsynaptic current in the presence of ionotropic glutamate and GABAB receptor antagonists; Vhold=70 mV—holding current; EGABA=−0.7 mV was calculated by
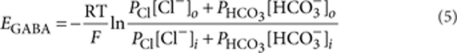 |
5 |
where PCl=1 and PHCO3 = 0.2−relative permeabilities for Cl− and HCO3−, respectively51.
mISPCs were recorded in the presence of 1 μM tetrodotoxin. sIPSCs and mIPSCs were completely blocked by 100 μM picrotoxin. Frequency, mean amplitude, and decay time constant of sIPSCs and mIPSCs were analysed off-line with the MiniAnalysis (Synaptosoft) and Clampfit (MDS Analytical Technologies). aIPSC frequency was calculated as the difference between the sIPSC and mIPSC frequencies. Time-averaged current mediated by sIPSCs (Ispont) was calculated as the mean charge transfer of sIPSCs multiplied by their frequency6.
Gramicidin-perforated patch experiments
The pipette tips were filled with the solution containing (in mM) 145 KCl, 10 HEPES, 5 ATP-Mg, 0.2 GTP-Na, 2 QX314, and 2 EGTA to prevent leakage of the antibiotic while approaching the cell. The pipettes were back-filled with the same solution that also contained 20 μg ml−1 gramicidin-D (Sigma). Fresh gramicidin solution was made every 2 h. After the pipette tip contacted the cell membrane, the Rs was monitored. Recordings were started when the Rs stabilized at 30–45 MΩ (usually within 15–20 min). All measurements were corrected for a liquid junction potential and voltage drop across the Rs (ref. 52). RMP was measured in current clamp mode (Ihold was 0 pA). Mean RMP and its standard deviation were obtained under control conditions and in the presence of GABA. The depolarizing effect of GABA was calculated as difference between mean RMPs. In some experiments, the cells were voltage-clamped at −65 mV. Membrane I–V characteristics were then obtained with 2,000-ms voltage steps delivered from −90 to −70 mV with 10 mV intervals, and from −55 to −20 mV with 5-mV intervals. I–V characteristics for GABAA tonic current were obtained as the difference between membrane I–V characteristics in 5 μM GABA and in 100 μM picrotoxin, added sequentially.
Cell-attached experiments
Pipettes were filled with superfusion solution and loosely attached to the cell membrane to measure interneuron firing without perturbing intracellular ionic concentrations. Recordings were performed in the voltage-clamp mode from the interneurons in which action currents were detectable (approximately 50% of cells)21,27. The command potential was set to the potential at which the holding current was 0 pA to avoid direct cell stimulation by the electrode53. The evoked GABAA-mediated PSGs were triggered by 200-μs current pulses at 50 μA delivered by a tungsten monopolar electrode placed in the str. radiatum. GABAA activation was also produced with a 100-μM GABA puff (100 ms, 50 psi). Preceding (tps) and succeeding (tss) spike intervals were calculated from the beginning of the synaptic stimulation or GABA puff.
Computational model
The cylindrical neuron (length 20 μm; diameter 10 μm; total surface 628 μm2) was created with the Neuron 7 simulator. The cell excitability was described according to Hodgkin–Huxley's model with corresponding Na+ (Nav) and K+ (Kv) channels (single-channel conductance: γNa=20 pS and γK=15 pS; channel density: 50 and 0.667 μm−2, giving a total number of channels of NNa=31,400 and NK=419). Additionally, A-type K+ channels (KA) were introduced (single-channel conductance γKA=10 pS; channel density: 8 μm−2 giving total number of channels: NKA=5,024)34. The transition of the channels between different conformational states was described as a Markov process, whereas the rate constants varied with the voltage54,55. The membrane also included the linear deterministic tonic GABAA conductance (gGABA), which was varied from 0 to 0.2 mS cm-2 with 0.001 mS cm-2 steps. On the basis of this set of conductances, we used the following equation:
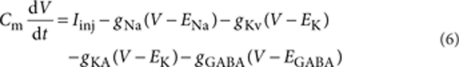 |
6 |
where Cm—membrane capacitance (1 μF cm-2), Iinj—injected current (0 pA for baseline conditions), ENa—Na+ equilibrium potential (50 mV), and EK—K+ equilibrium potential (−77 mV). The reversal potential for GABAA current (EGABA=−55 mV) was set 10.2 mV above the mean RMP without GABAA conductance (65.2 mV) to match experimental data. gNa, gK, and gKA—NaV, KV, and KA conductances, respectively, were controlled by continuous variables that range between 0 and 1 (m, h, n, k, l).
| 7 |
| 8 |
| 9 |
where [m3h1], [n4], and [l1k1] are the probabilities of the NaV, KV, and KA channels to be in the open state according to their respective kinetic schemes54,55. The variables m, h, n, k, l obeyed first-order differential equations with voltage dependent rate constants (αm, βm, αh, βh, αn, βn, αk, βk, αl).
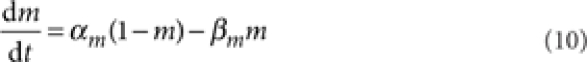 |
10 |
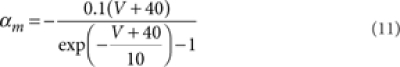 |
11 |
 |
12 |
 |
13 |
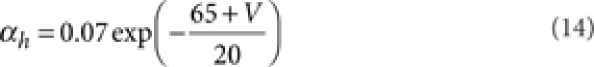 |
14 |
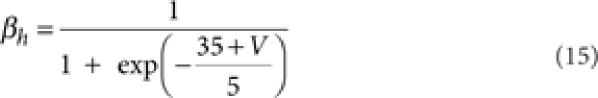 |
15 |
 |
16 |
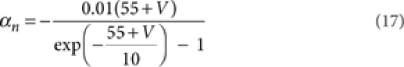 |
17 |
| 18 |
 |
19 |
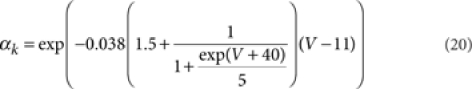 |
20 |
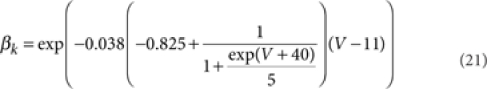 |
21 |
 |
22 |
| 23 |
The effect of gGABA on fluctuations of RMP (RMP standard deviation) was studied when the density of Na+ channels was reduced from 50 to 25 μm−2 to prevent action potential generation.
The effect of membrane depolarization on the cell firing and the RMP fluctuations was studied with the current injections (Iinj) from 0 to 1 nA and gGABA=0 nS.
All simulations were run for 1,000 ms, and the data were collected from the interval between 500 ms and 1,000 ms when the model reached a quasi-steady state. All the model data are means from 10 independent simulations. To avoid the correlation between data, each computation was performed with a different seed obtained from the random number generator.
Statistical analysis
Data are presented as means±s.e.m. Error bars on the graphs represent s.e.m. Statistical comparisons were made using unpaired Student's t-test unless a paired t-test is stated. A P-value of less than 0.05 was considered statistically significant.
Author contribution
A.S. and I.S. designed the study and planned the experiments; A.S., I.S., and L.S. analysed the data and prepared the manuscript; I.S. performed all the experiments; L.S. performed the modelling.
Additional information
How to cite this article: Song, I. et al. Tonic excitation or inhibition is set by GABAA conductance in hippocampal interneurons. Nat. Commun. 2:376 doi: 10.1038/ncomms1377 (2011).
Supplementary Material
Supplementary Figures S1–S5.
Acknowledgments
We thank Kunihiko Obata for providing us GAD65−/− mice; Kaiyu Zheng for providing the multimode cluster for computations; Dimiti Kullmann, Matthew Walker, Hajime Hirase, Ivan Pavlov, Karri Lamsa, Thomas McHugh, Kirill Volynski, Stephen F. Heinemann, Dmitri Rusakov and all members of the AS laboratory for reading and commenting on the manuscript. This work was supported by RIKEN BSI and The Wellcome Trust.
References
- Glykys J. & Mody I. Activation of GABAA receptors: views from outside the synaptic cleft. Neuron 56, 763–770 (2007). [DOI] [PubMed] [Google Scholar]
- Farrant M. & Nusser Z. Variations on an inhibitory theme: phasic and tonic activation of GABA(A) receptors. Nat. Rev. Neurosci. 6, 215–229 (2005). [DOI] [PubMed] [Google Scholar]
- Semyanov A., Walker M. C., Kullmann D. M. & Silver R. A. Tonically active GABA A receptors: modulating gain and maintaining the tone. Trends Neurosci. 27, 262–269 (2004). [DOI] [PubMed] [Google Scholar]
- Scimemi A., Semyanov A., Sperk G., Kullmann D. M. & Walker M. C. Multiple and plastic receptors mediate tonic GABAA receptor currents in the hippocampus. J. Neurosci. 25, 10016–10024 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glykys J., Mann E. O. & Mody I. Which GABA(A) receptor subunits are necessary for tonic inhibition in the hippocampus? J. Neurosci. 28, 1421–1426 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semyanov A., Walker M. C. & Kullmann D. M. GABA uptake regulates cortical excitability via cell type-specific tonic inhibition. Nat. Neurosci. 6, 484–490 (2003). [DOI] [PubMed] [Google Scholar]
- Jia F. et al. An extrasynaptic GABAA receptor mediates tonic inhibition in thalamic VB neurons. J. Neurophysiol. 94, 4491–4501 (2005). [DOI] [PubMed] [Google Scholar]
- Vardya I., Drasbek K. R., Dosa Z. & Jensen K. Cell type-specific GABA A receptor-mediated tonic inhibition in mouse neocortex. J. Neurophysiol. 100, 526–532 (2008). [DOI] [PubMed] [Google Scholar]
- Ade K. K., Janssen M. J., Ortinski P. I. & Vicini S. Differential tonic GABA conductances in striatal medium spiny neurons. J. Neurosci. 28, 1185–1197 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamoto Y. et al. In vivo microdialysis of amino acid neurotransmitters in the hippocampus in amygdaloid kindled rat. Brain Res. 573, 345–348 (1992). [DOI] [PubMed] [Google Scholar]
- Bianchi L. et al. Investigation on acetylcholine, aspartate, glutamate and GABA extracellular levels from ventral hippocampus during repeated exploratory activity in the rat. Neurochem. Res. 28, 565–573 (2003). [DOI] [PubMed] [Google Scholar]
- Kupers R., Danielsen E. R., Kehlet H., Christensen R. & Thomsen C. Painful tonic heat stimulation induces GABA accumulation in the prefrontal cortex in man. Pain 142, 89–93 (2009). [DOI] [PubMed] [Google Scholar]
- Dzhala V. I. et al. NKCC1 transporter facilitates seizures in the developing brain. Nat. Med. 11, 1205–1213 (2005). [DOI] [PubMed] [Google Scholar]
- Rivera C. et al. The K+/Cl− co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 397, 251–255 (1999). [DOI] [PubMed] [Google Scholar]
- Hausselt S. E., Euler T., Detwiler P. B. & Denk W. A dendrite-autonomous mechanism for direction selectivity in retinal starburst amacrine cells. PLoS Biol. 5, e185 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khirug S. et al. GABAergic depolarization of the axon initial segment in cortical principal neurons is caused by the Na-K-2Cl cotransporter NKCC1. J. Neurosci. 28, 4635–4639 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glickfeld L. L., Roberts J. D., Somogyi P. & Scanziani M. Interneurons hyperpolarize pyramidal cells along their entire somatodendritic axis. Nat. Neurosci. 12, 21–23 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michelson H. B. & Wong R. K. Excitatory synaptic responses mediated by GABAA receptors in the hippocampus. Science 253, 1420–1423 (1991). [DOI] [PubMed] [Google Scholar]
- Banke T. G. & McBain C. J. GABAergic input onto CA3 hippocampal interneurons remains shunting throughout development. J. Neurosci. 26, 11720–11725 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vida I., Bartos M. & Jonas P. Shunting inhibition improves robustness of gamma oscillations in hippocampal interneuron networks by homogenizing firing rates. Neuron 49, 107–117 (2006). [DOI] [PubMed] [Google Scholar]
- Verheugen J. A., Fricker D. & Miles R. Noninvasive measurements of the membrane potential and GABAergic action in hippocampal interneurons. J. Neurosci. 19, 2546–2555 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chavas J. & Marty A. Coexistence of excitatory and inhibitory GABA synapses in the cerebellar interneuron network. J. Neurosci. 23, 2019–2031 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodruff A. R., Monyer H. & Sah P. GABAergic excitation in the basolateral amygdala. J. Neurosci. 26, 11881–11887 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bracci E. & Panzeri S. Excitatory GABAergic effects in striatal projection neurons. J. Neurophysiol. 95, 1285–1290 (2006). [DOI] [PubMed] [Google Scholar]
- Bowery N. G. et al. Indirect effects of amino-acids on sympathetic ganglion cells mediated through the release of gamma-aminobutyric acid from glial cells. Br. J. Pharmacol. 57, 73–91 (1976). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morita K., Tsumoto K. & Aihara K. Possible effects of depolarizing GABAA conductance on the neuronal input-output relationship: a modeling study. J. Neurophysiol. 93, 3504–3523 (2005). [DOI] [PubMed] [Google Scholar]
- Fricker D., Verheugen J. A. & Miles R. Cell-attached measurements of the firing threshold of rat hippocampal neurones. J. Physiol. 517 (Part 3), 791–804 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourdeau M. L., Morin F., Laurent C. E., Azzi M. & Lacaille J. C. Kv4.3-mediated A-type K+ currents underlie rhythmic activity in hippocampal interneurons. J. Neurosci. 27, 1942–1953 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman C. A. & Lacaille J. C. Intrinsic theta-frequency membrane potential oscillations in hippocampal CA1 interneurons of stratum lacunosum-moleculare. J. Neurophysiol. 81, 1296–1307 (1999). [DOI] [PubMed] [Google Scholar]
- Walker M. C. & Semyanov A. Regulation of excitability by extrasynaptic GABA(A) receptors. Results Probl. Cell Differ. 44, 29–48 (2008). [DOI] [PubMed] [Google Scholar]
- Brown D. A. & Marsh S. Axonal GABA-receptors in mammalian peripheral nerve trunks. Brain Res. 156, 187–191 (1978). [DOI] [PubMed] [Google Scholar]
- Rojas P., Akrouh A., Eisenman L. N. & Mennerick S. Differential effects of axon initial segment and somatodendritic GABAA receptors on excitability measures in rat dentate granule neurons. J. Neurophysiol. 105, 366–379 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin F., Haufler D., Skinner F. K. & Lacaille J. C. Characterization of voltage-gated K+ currents contributing to subthreshold membrane potential oscillations in hippocampal CA1 interneurons. J. Neurophysiol. 103, 3472–3489 (2010). [DOI] [PubMed] [Google Scholar]
- Migliore M., Hoffman D. A., Magee J. C. & Johnston D. Role of an A-type K+ conductance in the back-propagation of action potentials in the dendrites of hippocampal pyramidal neurons. J. Comput. Neurosci. 7, 5–15 (1999). [DOI] [PubMed] [Google Scholar]
- Klausberger T. & Somogyi P. Neuronal diversity and temporal dynamics: the unity of hippocampal circuit operations. Science 321, 53–57 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kash S. F. et al. Epilepsy in mice deficient in the 65-kDa isoform of glutamic acid decarboxylase. Proc. Natl Acad. Sci. USA 94, 14060–14065 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stork O. et al. Postnatal development of a GABA deficit and disturbance of neural functions in mice lacking GAD65. Brain Res. 865, 45–58 (2000). [DOI] [PubMed] [Google Scholar]
- Birnir B., Everitt A. B., Lim M. S. & Gage P. W. Spontaneously opening GABA(A) channels in CA1 pyramidal neurones of rat hippocampus. J. Membr. Biol. 174, 21–29 (2000). [DOI] [PubMed] [Google Scholar]
- McCartney M. R., Deeb T. Z., Henderson T. N. & Hales T. G. Tonically active GABAA receptors in hippocampal pyramidal neurons exhibit constitutive GABA-independent gating. Mol. Pharmacol. 71, 539–548 (2007). [DOI] [PubMed] [Google Scholar]
- Kash S. F., Tecott L. H., Hodge C. & Baekkeskov S. Increased anxiety and altered responses to anxiolytics in mice deficient in the 65-kDa isoform of glutamic acid decarboxylase. Proc. Natl Acad. Sci. USA 96, 1698–1703 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian N. et al. The role of the synthetic enzyme GAD65 in the control of neuronal gamma-aminobutyric acid release. Proc. Natl Acad. Sci. USA 96, 12911–12916 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brickley S. G., Revilla V., Cull-Candy S. G., Wisden W. & Farrant M. Adaptive regulation of neuronal excitability by a voltage-independent potassium conductance. Nature 409, 88–92 (2001). [DOI] [PubMed] [Google Scholar]
- Morishima T. et al. GABA imaging in brain slices using immobilized enzyme-linked photoanalysis. Neurosci. Res. 67, 347–353 (2010). [DOI] [PubMed] [Google Scholar]
- Pavlov I., Savtchenko L. P., Kullmann D. M., Semyanov A. & Walker M. C. Outwardly rectifying tonically active GABAA receptors in pyramidal cells modulate neuronal offset, not gain. J. Neurosci. 29, 15341–15350 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pouille F. & Scanziani M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science 293, 1159–1163 (2001). [DOI] [PubMed] [Google Scholar]
- Bartos M., Vida I. & Jonas P. Synaptic mechanisms of synchronized gamma oscillations in inhibitory interneuron networks. Nat. Rev. Neurosci. 8, 45–56 (2007). [DOI] [PubMed] [Google Scholar]
- Lee H. H., Deeb T. Z., Walker J. A., Davies P. A. & Moss S. J. NMDA receptor activity downregulates KCC2 resulting in depolarizing GABA(A) receptor-mediated currents. Nat. Neurosci. 14, 736–743 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulledge A. T. & Stuart G. J. Excitatory actions of GABA in the cortex. Neuron 37, 299–309 (2003). [DOI] [PubMed] [Google Scholar]
- Asada H. et al. Mice lacking the 65 kDa isoform of glutamic acid decarboxylase (GAD65) maintain normal levels of GAD67 and GABA in their brains but are susceptible to seizures. Biochem. Biophys. Res. Commun. 229, 891–895 (1996). [DOI] [PubMed] [Google Scholar]
- Gentet L. J., Stuart G. J. & Clements J. D. Direct measurement of specific membrane capacitance in neurons. Biophys. J. 79, 314–320 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaila K. Ionic basis of GABAA receptor channel function in the nervous system. Prog. Neurobiol. 42, 489–537 (1994). [DOI] [PubMed] [Google Scholar]
- Ulrich D. & Huguenard J. R. Nucleus-specific chloride homeostasis in rat thalamus. J. Neurosci. 17, 2348–2354 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins K. L. Cell-attached voltage-clamp and current-clamp recording and stimulation techniques in brain slices. J. Neurosci. Methods 154, 1–18 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeFelice L. J. & Isaac A. Chaotic states in a random world: Relationship between the nonlinear differential equations of excitability and the stochastic properties of ion channels. J. Stat. Phys. 70, 339–354 (1993). [Google Scholar]
- Savtchenko L. P., Gogan P., Korogod S. M. & Tyc-Dumont S. Imaging stochastic spatial variability of active channel clusters during excitation of single neurons. Neurosci. Res. 39, 431–446 (2001). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figures S1–S5.