Abstract

Dynamic nuclear polarization (DNP) of 13C-labeled metabolic substrates in vitro and their subsequent intravenous administration allow both the location of the hyperpolarized substrate and the dynamics of its subsequent conversion into other metabolic products to be detected in vivo. We report here the hyperpolarization of [1-13C]-ascorbic acid (AA) and [1-13C]-dehydroascorbic acid (DHA), the reduced and oxidized forms of vitamin C, respectively, and evaluate their performance as probes of tumor redox state. Solution-state polarization of 10.5 ± 1.3% was achieved for both forms at pH 3.2, whereas at pH 7.0, [1-13C]-AA retained polarization of 5.1 ± 0.6% and [1-13C]-DHA retained 8.2 ± 1.1%. The spin–lattice relaxation times (T1's) for these labeled nuclei are long at 9.4 T: 15.9 ± 0.7 s for AA and 20.5 ± 0.9 s for DHA. Extracellular oxidation of [1-13C]-AA and intracellular reduction of [1-13C]-DHA were observed in suspensions of murine lymphoma cells. The spontaneous reaction of DHA with the cellular antioxidant glutathione was monitored in vitro and was approximately 100-fold lower than the rate observed in cell suspensions, indicating enzymatic involvement in the intracellular reduction. [1-13C]-DHA reduction was also detected in lymphoma tumors in vivo. In contrast, no detectable oxidation of [1-13C]-AA was measured in the same tumors, consistent with the notion that tumors maintain a reduced microenvironment. This study demonstrates that hyperpolarized 13C-labeled vitamin C could be used as a noninvasive biomarker of redox status in vivo, which has the potential to translate to the clinic.
Introduction
Dynamic nuclear polarization (DNP) of 13C-labeled cell substrates enhances their sensitivity to detection in vivo by over 10,000-fold. DNP has shown great promise for metabolic imaging of cancer in preclinical studies, with a wide range of substrates now available(1) including hyperpolarized [1-13C]-pyruvate, which enables early detection of tumor response to drug treatment2,3 and noninvasive assessment of tumor grade;(4) [2-13C]-fructose, phosphorylation of which is influenced by both glycolytic and pentose phosphate pathway activity;(5) [1,4-13C2]-fumarate, which is emerging as a marker of cell death following chemotherapy;2,6 and [13C]-bicarbonate, which allows assessment of tumor pH.(7) Although these hyperpolarized substrates probe a wide range of biological processes, there are currently no hyperpolarized substrates that report directly on tumor redox status. Rapid cancer cell proliferation promotes intracellular accumulation of reducing equivalents such as glutathione and NADPH(8) to counteract the effects of reactive oxygen species (ROS) and to maintain redox balance.(9) The ability of certain cancer cells to maintain a highly reduced intracellular environment is strongly correlated with aggressiveness and drug resistance.10,11 Noninvasive imaging of tumor redox status may therefore indicate prognosis and give an early readout of treatment response. Recently, 13C-benzoylformic acid (BFA) was shown to report specifically on hydrogen peroxide concentration in vitro, but its application in vivo is likely to be limited by a lack of sensitivity.(12) To be used in vivo, 13C-labeled DNP substrates must exhibit long spin–lattice relaxation times (T1's), be rapidly transported (or react in the extracellular space), and produce an observable chemical shift change between the injected substrate and its product, to allow spectroscopic imaging of both metabolites. The chemical and biological properties of vitamin C make it a prime candidate for in vivo detection of redox status: the C1 carbonyl group has a long T1, its oxidation produces a chemical shift difference of ∼5 ppm, the oxidized form is transported rapidly by the same membrane transporters utilized by glucose,(13) and its redox state is coupled to two primary cellular antioxidants, glutathione and NADPH.(14)
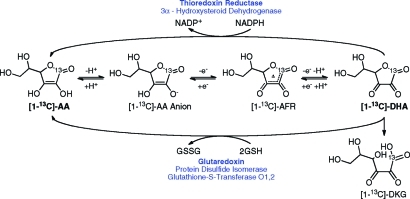
The reduced form of vitamin C, ascorbic acid (AA), is an essential water-soluble antioxidant and cofactor of numerous enzymes involved in biosynthetic reactions.(14) AA is reversibly oxidized (Figure 1), forming the stable ascorbic free radical (AFR) intermediate with the loss of one electron or dehydroascorbic acid (DHA) with the loss of two electrons. AFR decays by disproportionation to AA and DHA, whereas DHA spontaneously hydrolyses under physiological conditions, with the opening of the lactone ring to form diketogulonic acid (DKG).(15) While epithelial cells of the intestine, liver, and kidney can transport AA directly through sodium-dependent vitamin C transporters (SVCT1, 2(16)), the primary means by which most tissues acquire vitamin C is through transport of DHA, which forms a stable bicyclic hydrated hemiacetal structure in aqueous solution.17,18 Studies in tumor cell lines have found that they either lack the capacity to transport AA, or if they do have the capability, the rate of uptake of DHA is at least an order of magnitude faster.(19) DHA is taken up by the glucose transporters13,20 GLUT1 (Km = 1.1 mM) and GLUT3 (Km = 1.7 mM) (Figure 2), which display a similar affinity for both glucose and DHA and are overexpressed in most tumors compared to normal tissue.(21) This phenomenon is widely exploited in detection and staging of tumors by [18F]-fluorodeoxyglucose positron emission tomography (FDG PET).9,22 Once inside the cell, DHA must be reduced rapidly to avoid hydrolysis(23) and maintain antiscorbutic activity. This can occur spontaneously or enzymatically, by reaction with glutathione (GSH) or GSH-dependent enzymes (Km = 2.2 mM) and through NADPH-dependent reactions, primarily involving the enzyme thioredoxin reductase (Km = 0.7 mM).(14) Glutathione-dependent enzymes include the thiol-disulfide oxidoreductases, glutaredoxin and protein disulfide isomerase, while the predominant NADPH-dependent enzyme is the selenoprotein, thioredoxin reductase (Figure 1). In both cases, rapid intracellular reduction of DHA will maintain a favorable gradient for transport.(19)
Figure 1.
Primary intracellular oxidation, reduction, and degradation pathways for vitamin C. Enzymes involved in GSH- and NADPH-dependent reduction of dehydroascorbic acid (DHA) are highlighted, with the primary cytosolic enzymes in bold.(14) Abbreviations: AA, ascorbic acid; AFR, ascorbate free radical (also known as semidehydroascorbic acid); DHA, dehydroascorbic acid; DKG, diketogulonic acid; GSH, reduced glutathione; GSSG, oxidized glutathione.
Figure 2.

Mechanism of vitamin C transport, in both reduced (AA) and oxidized (DHA) forms, by sodium-dependent vitamin C transporters 1 and 2 (SVCT 1, 2) and glucose transporters (primarily GLUT1, 3 and also GLUT 4), respectively. This is dominated in cancer cells by GLUT1 transport of DHA, with SVCTs either not expressed or having low activity due to the acidic extracellular pH in tumors.19,21
The upregulation of the glutathione and thioredoxin antioxidant systems in tumors has been implicated in both chemoresistance and increased metastatic potential.10,24−26 While the dominant mechanism of DHA reduction remains unresolved in cancer cells, NADPH provides the reducing power for both of these systems, and therefore the rate of intracellular reduction of DHA is determined ultimately by the availability of NADPH and, in turn, pentose phosphate pathway activity, reflecting oxidative stress.27,28 Furthermore, oxidative stress is associated with almost all major diseases and in addition to cancer has been implicated in numerous neurodegenerative and cardiovascular conditions.(11) Oxidative stress has been assessed in the clinic, at a systemic level, from measurements of plasma ubiquinol and ubiquinone(29) and from measurements of serum antioxidants and the presence of oxidized protein and lipid.(30) Although imaging regions of oxidative stress in tissues should provide a more sensitive readout in a range of diseases, this has been limited to date by the lack of available probes and there are currently no methods available to image the spatial distribution of redox state in the clinic. Imaging measurements of nitroxide reduction, either directly using EPR imaging31,32 or indirectly from T1-weighted MRI measurements, have shown great promise in preclinical studies,(33) but these techniques have yet to translate to the clinic. The rapid interconversion of the reduced and oxidized forms of vitamin C in vivo makes them ideal candidates for hyperpolarized redox imaging, with AA primarily an extracellular probe and DHA an intracellular redox probe. The goal of this study was to evaluate the performance of [1-13C]-AA and [1-13C]-DHA as hyperpolarized imaging probes using cancer models in vitro and in vivo.
Methods
Hyperpolarization of [1-13C]-Ascorbic Acid and [1-13C]-Dehydroascorbic Acid
A 1.5 M solution of [1-13C]-AA (Omicron Biochemicals, Inc., South Bend, IN) in 60:40 H2O/glycerol or a 1.8 M solution of [1-13C]-DHA (Supporting Information) in DMSO-d6, containing 14.8 mM trityl radical (OX063; GE Healthcare, Amersham, UK) and 1.4 mM of gadolinium chelate (Dotarem; Guerbet, Roissy, France), were polarized as described previously.(3) Briefly, the sample was rapidly cooled under liquid helium in a GE Healthcare DNP prototype hyperpolarizer at a pressure of ∼1 mBar to ∼1.2 K. Polarization of the electron spins on the trityl radical were transferred to the 13C label using microwave irradiation at 94.010 GHz (100 mW) over 1 and 1.5 h for [1-13C]-AA and [1-13C]-DHA, respectively. The frozen samples were dissolved in 5 mL of water (pH ∼3.2). For in vivo administration, samples were neutralized by addition of a further 1 mL of phosphate buffer (200 mM; pH 7.8) containing EDTA (1.8 mM) and NaCl (400 mM) to yield a final pH and osmolality in the physiological range (pH 7.0–7.2; >310 mOsm/kg). This procedure prevented significant sample degradation and loss of polarization.
Hyperpolarized 13C NMR Spectroscopy in Vitro at 9.4 T
NMR studies of reaction kinetics in vitro were performed on a 9.4 T vertical wide-bore spectrometer (100 MHz 13C, Oxford Instruments, Abingdon, U.K.) using a 10 mm 13C broadband probe (Varian NMR Instruments, Palo Alto, CA) and temperature-controlled at 37 °C. Samples of the [1-13C]-AA (46 μL; 14 mM on dissolution) or [1-13C]-DHA (39 μL; 14 mM on dissolution) preparations were hyperpolarized, and measurements of the percentage polarization for both substrates were made with a liquid-state polarimeter (Amersham Health R & D, Malmö, Sweden) approximately 8 s after the dissolution, at both pH 3.2 and 7.0. For [1-13C]-AA, the reading was confirmed by acquisition of 25 pulse and acquire 13C NMR spectra at approximately 10 s post dissolution (number of transients (nt) = 1, 32 kHz spectral window, 5 s pulse repetition time (TR), 65° flip angle pulse, total acquisition time of 105 s) followed by acquisition of spectra at thermal equilibrium from the same solution (nt = 16, 32 kHz spectral window, TR = 200 s, 65° flip angle pulse, total acquisition time of 50 min). The polarization was calculated by comparing the first hyperpolarized spectrum with the spectrum at thermal equilibrium, correcting for differences in the number of transients. This measurement could not be performed with [1-13C]-DHA due to the rapid hydrolysis of the sample, with a half-life of approximately 10 min at neutral pH.(23) The T1 of the hyperpolarized 13C label was determined in a separate experiment at pH 7.0 using 180 spectra acquired with an 8° flip angle pulse (nt = 1, 16 kHz spectral window, TR = 1 s, total acquisition time of 3 min) and fitting the decay of the integrated peak intensity to eq 1:
where S0 is the initial integrated peak intensity, TR is the repetition time in seconds, T1 is the spin–lattice relaxation time in seconds, α is the flip angle, and n is the number of preceding RF pulses.
To determine the reaction kinetics of [1-13C]-DHA with glutathione (GSH), hyperpolarized [1-13C]-DHA (2 mL) was injected into a 10 mm NMR tube containing a solution of GSH (2 mL, 100 mM) in phosphate buffer (100 mM, pH 7.4) with EDTA (0.3 mM), to yield final concentrations of 5 mM [1-13C]-DHA and 50 mM GSH at pH 7.3; 240 spectra were then recorded using an 8° pulse (nt = 1, 24 kHz spectral window, TR = 1 s, total acquisition time of 4 min). This was repeated for final GSH concentrations of 1, 5, 10, and 25 mM. The rate of [1-13C]-AA production, kDHA, was determined by fitting the peak integrals to the modified Bloch equations for two-site exchange2,3,6 with the back-flux of the reaction, kAA, set to zero. The relationship between the forward flux ([DHA] × kDHA) and the rate of production of AA determined spectrophotometrically (see Supporting Information) was established using linear regression in Prism (Graphpad, San Diego, CA).
For spectroscopic studies of vitamin C turnover in cancer cells, EL-4 murine lymphoma cells were grown in RPMI 1640 medium supplemented with 10% fetal calf serum and 2 mM l-glutamine (culture media from Invitrogen). Cell number and viability were assessed using trypan blue dye staining. Cells (1 × 108) were suspended in RPMI 1640 medium (2 mL) in a 10-mm NMR tube. For the experiments with AA the dissolution was performed within 2.5 min of resuspension of the cells to allow for generation of extracellular ROS and for the DHA experiments within 1.5 min in order to minimize ROS generation. The hyperpolarized substrate under study (2 mL, 14 mM) was injected into the cell suspension at approximately 10 s post dissolution. RPMI 1640 medium was found to be sufficient to buffer the hyperpolarized sample dissolved in water to pH 7.0. Spectra (180) were recorded using an 8° flip angle pulse (nt = 1, 16 kHz spectral window, TR = 0.5 s, total acquisition time of 3 min) starting at the beginning of hyperpolarized substrate injection.
Hyperpolarized 13C Spectroscopy (MRS) in Vivo at 9.4 T
For experiments in vivo, a 24-mm diameter surface coil tuned to 13C (100 MHz) was used in combination with a quadrature 1H-tuned volume coil (Varian NMR Instruments, Palo Alto, CA), as described previously.(2) Transverse 1H spin–echo images were acquired to localize the tumor and plan voxels for MRS (TR = 500 ms, TE = 10 ms, field-of-view = 35 × 35 mm2 in a data matrix of 256 × 256; 21 slices of 2 mm thickness). After administration of [1-13C]-AA or [1-13C]-DHA (0.2 mL; 30 mM, 1.1 mg/kg), 200 spectra were acquired from a 6 mm tumor slice using a nominal flip angle of 10° (every fifth spectrum was non-slice selective), with TR = 1 and 0.25 s for AA and DHA, respectively. The T1 of each substrate was estimated by fitting the peak integrals of the injected substrate in the non-slice selective spectra to eq 1. Over 250 mg/kg intravenous AA, a 250-fold excess compared to the dose here, is administered in pharmacological studies without side effects.(34) High dose infusions of DHA (up to 60 mg/kg) can cause elevated blood pressure and CNS responses in healthy rats,(35) but this level is 60-fold in excess of that used in this study, and adverse effects on blood pressure were not observed here.
Results and Discussion
The measured polarizations and T1's for the labeled C1 position in both ascorbic acid and dehydroascorbic acid at 9.4 T are given in Table 1. [1-13C]-DHA and [1-13C]-AA exhibit a similar level of polarization at pH 3.2, but at neutral pH, [1-13C]-DHA retains a degree of polarization significantly higher than that of [1-13C]-AA (p = 0.05; n = 6). The loss of [1-13C]-AA polarization at physiological pH (5.1 ± 0.6% vs 10.5 ± 1.3% at pH 3.2, p = 0.008; n = 6) can be attributed to the fact that the hydroxyl group at the C3 position has a pKa of 4.2 and so is completely dissociated at pH 7.0. The polarization of [1-13C]-DHA is also slightly lower at neutral pH (p = 0.12; n = 8), relating in part to hydrolysis of the sample prior to measurement. The polarization values reported here are lower than those commonly reported for [1-13C]-pyruvate, which are in the range of 20–35%, but are within the range of other molecules imaged in vivo including [13C]-bicarbonate and [2-13C]-fructose. As expected, the C1 position in [1-13C]-DHA displays a longer T1 than that of [1-13C]-AA in vitro (p = 0.003; n = 3), but there was no significant difference in the estimated T1 values of the two species in vivo, which are in agreement with those measured in erythrocyte suspensions(36) and of a similar order to those found for hyperpolarized [13C]-bicarbonate and [2-13C]-fructose.
Table 1. T1 Relaxation Times Measured in Vitro and in Vivo at 9.4 T and Percent Polarization Measured at Acidic and Neutral pH Approximately 8 s Post Dissolution.
| polarization (%) |
T1 (s) |
|||
|---|---|---|---|---|
| vitamin C form | pH 3.2 | pH 7.0 | in vitro (pH 7.0) | in vivo |
| [1-13C]-ascorbic acid | 10.5 ± 1.3 | 5.1 ± 0.6 | 15.9 ± 0.7 | 8.6 ± 0.7 |
| [1-13C]-dehydroascorbic acid | 10.7 ± 1.3 | 8.2 ± 1.1 | 20.5 ± 0.9 | 11.9 ± 0.1 |
The intracellular reduction of DHA depends on NADPH, both through glutathione-dependent reactions, which require NADPH to replenish the reduced glutathione pool, and directly through NADPH-dependent reactions. The observed reduction of hyperpolarized [1-13C]-DHA in vitro and in vivo is likely to depend in large part on the reaction with reduced glutathione,(14) proceeding via both spontaneous and enzymatic reactions. The spontaneous reaction of DHA with glutathione is thermodynamically favorable at physiological pH (Keq = 2.5 × 105 M–1 at pH 7) but proceeds slowly at endogenous concentrations of DHA (micromolar) and glutathione (few millimolar).10,11 Therefore, we sought to establish the rate at which higher concentrations of [1-13C]-DHA react spontaneously with glutathione to determine whether this could contribute to the [1-13C]-DHA reduction observed in cancer cell suspensions in vitro and tumor xenografts in vivo. The reaction of [1-13C]-DHA with glutathione yielded [1-13C]-AA at 179 ppm (Figure 3a) immediately following addition of [1-13C]-DHA to the glutathione-containing buffer. The [1-13C]-AA signal reached maximal intensity within 7–16 s depending on the concentration of glutathione (Figure 3b). The ratio of the peak integrals of AA and DHA in the spectrum containing the maximum AA signal was calculated and found to be linear with both glutathione concentration and the rate of AA production (data not shown). The rate of [1-13C]-AA production was established by multiplying the amount of DHA (micromoles) with the apparent rate constant of the reaction kDHA (s–1), and this showed a linear correlation with the rate of AA production measured spectrophotometrically under equivalent experimental conditions (Figure 3c, see also Supporting Information; y = (1.00 ± 0.04)x; r2 = 0.992). The second order rate constant for the reaction derived from the spectrophotometric data, 0.34 ± 0.01 M–1 s–1, was in excellent agreement with previously reported data.(37)
Figure 3.
(a) 13C NMR spectra of hyperpolarized [1-13C]-DHA (175 ppm) at the peak of [1-13C]-AA (179 ppm) production following reaction of 5 mM [1-13C]-DHA with 1, 5, 10, 25, and 50 mM glutathione. Peaks at 173.5 and 175.4 ppm are from diketogulonic acid and an unknown product, respectively. (b) Time course of [1-13C]-AA peak intensity at various glutathione concentrations, normalized to the maximum [1-13C]-DHA intensity. (c) Correlation of the rate of [1-13C]-AA production, [DHA] × kDHA, with the rate of AA production measured spectrophotometrically at equivalent concentrations of DHA and glutathione.
Oxidation of [1-13C]-AA to [1-13C]-DHA was observed at a rate of 327 ± 41 μmol/s ([AA] x kAA) in suspensions of EL4 lymphoma cells at 2.5 min after resuspension of the cells in fresh RPMI medium (Figure 4b, e). No oxidation was observed when [1-13C]-AA was added to either RPMI medium alone (Figure 4a) or to cells at 1 min after resuspension, although a low level became apparent at 2 min after cell resuspension (data not shown). Cell culture imposes a state of oxidative stress on cells, with antioxidants provided primarily in supplemented serum.38,39 Cancer cells in vitro constitutively release hydrogen peroxide,(38) which readily penetrates the cell membrane and is relatively stable in most culture media compared to reactive oxygen species.(40) Following resuspension, cells are not only deprived of serum antioxidants, leading to an increased rate of intracellular superoxide generation(38) and in turn, a higher release of hydrogen peroxide, but will also rapidly deplete the medium of oxygen and nutrients, leading to an acutely hypoxic state. Furthermore, the pharmacological effects of high concentrations of AA (>0.3 mM) have been attributed to the generation of hydrogen peroxide through the reduction of catalytic metals such as Cu(II), which are present in cell culture media, by AA.(41) AA is known to be oxidized by hydrogen peroxide,(42) so the observation of [1-13C]-AA oxidation at increasing time after resuspension is likely due to accumulation of hydrogen peroxide and is consistent with the notion that this is an extracellular reaction.
Figure 4.
Oxidation of [1-13C]-AA (a, b, e) and reduction of [1-13C]-DHA (c, d, f) in EL4 lymphoma cell suspensions. Hyperpolarized substrates were added to either RPMI medium alone (a, c) or a suspension of 108 EL4 lymphoma cells in RPMI medium. (b) Spectrum obtained at 2.5 min post cell resuspension and (d) at 1.5 min post cell resuspension. Only every fourth spectrum is displayed in the interests of clarity (e, f; time course over 46 s). Peaks at 172.6 and 176.4 ppm are from diketogulonic acid and the C3 carbon of AA,(50) respectively.
Conversely, the reduction of [1-13C]-DHA (Figure 4d, f) was observed only at early time points (<1 min) after cell resuspension and returned to baseline by 2.5 min. Reduction of [1-13C]-DHA was observed in RPMI medium alone, which contains 0.0033 mM glutathione.(43) The medium also contains other factors that have been documented to both reduce and hydrolyze DHA, including sodium bicarbonate(44) and cystine,(45) as well as 11 mM d-glucose, which may competitively inhibit DHA transport. The final concentrations of glutathione, cystine, bicarbonate and glucose in these experiments were 0.0017, 0.104, 11.9, and 5.5 mM respectively, with a final concentration of 7 mM [1-13C]-DHA. Based on the measurements made above, the rate of [1-13C]-AA production due to spontaneous reaction with the glutathione in RPMI would be 0.15 nmol/s. The rates observed in RPMI medium and EL4 lymphoma cell suspensions were 99 ± 6 nmol/s (n = 2) and 223 ± 18 nmol/s (n = 3), respectively. The contribution of the spontaneous reaction with glutathione may therefore be assumed to be negligible in EL4 cell suspensions, indicating that other constituents of RPMI contribute to the reduction of DHA in vitro and that a further reaction, enzymatic or otherwise, occurs within EL4 cells. The shorter T1 of DHA in cell suspensions compared to RPMI medium, 20.8 ± 0.7 s (n = 5) versus 17.9 ± 1.7 s (n = 3), may be due to the intracellular transport of this substrate.
The in vivo metabolism of [1-13C]-AA and [1-13C]-DHA is demonstrated in Figure 5. Intravenous administration of 30 mM [1-13C]-AA yielded no observable [1-13C]-DHA resonance in EL4 lymphoma tumors (Figure 5a; n = 4), whereas administration of 30 mM [1-13C]-DHA resulted in readily detectable production of [1-13C]-AA (Figure 5b; n = 4) and hydrolysis to [1-13C]-DKG. The apparent rate constant for the in vivo reduction of [1-13C]-DHA was kDHA = 0.020 ± 0.004 s–1. The ratio of the peak integrals of [1-13C]-AA and [1-13C]-DHA calculated within the first 1 s of data acquisition (summation of the first 4 FIDs) was 0.35 ± 0.08, with an average signal-to-noise ratio for the [1-13C]-DHA peak of 67 ± 20 over the same period. By comparison, the peak ratio in the non-slice selective spectrum taken at 1.25 s was 0.17 ± 0.03, indicating that a significant portion of the overall reduction of [1-13C]-DHA occurs in the tumor. The EL4 tumor concentration of glutathione was determined by the method of Tietze et al.(46) to be 1.18 ± 0.03 μmol/g wet wt, corresponding to approximately 1.2 mM in the tumor tissue.46,47 Given the results in Figure 3, at this concentration the spontaneous reaction of DHA with glutathione would be expected to proceed at a rate of only 0.0004 s–1 and produce a maximum relative signal intensity of 0.013 for [1-13C]-AA. These data provide further evidence that an enzymatic reaction is required to account for both the rate and level of [1-13C]-AA produced in EL4 cells. The lack of observable oxidation of [1-13C]-AA in vivo, despite the high level seen in cell suspensions, together with the rapid reduction of [1-13C]-DHA, indicate that upregulation of antioxidant systems allows tumors to maintain a reduced microenvironment.(48)
Figure 5.
13C MR spectra acquired from EL4 murine lymphoma tumors following injection of 30 mM [1-13C]-AA (a) and [1-13C]-DHA (b; every second spectrum shown for clarity). Sequential spectra were collected from a 6-mm tumor slice starting at 15 s post injection over a period of 16 s (a) and 32 s (b). Whereas there was no observable flux of hyperpolarized 13C label from [1-13C]-AA (179 ppm) to [1-13C]-DHA (175 ppm) (a), significant flux was observed from [1-13C]-DHA to [1-13C]-AA, indicating rapid reduction of the injected [1-13C]-DHA in the tumor.
Conclusions
In this study, we have demonstrated hyperpolarization of [1-13C]-ascorbic acid and [1-13C]-dehydroascorbic acid, the reduced and oxidized forms of vitamin C, to relatively high levels. Both compounds have been administered previously in high doses in clinical studies with little or no observed toxicity and were shown here to exhibit sufficiently long T1's to allow observation of their signals in vitro and in vivo. The extracellular pool of [1-13C]-AA was oxidized rapidly in hypoxic EL4 cell suspensions but not in vivo, as might be expected given that tumors maintain a reduced microenvironment. [1-13C]-AA may find application in diseases that lead to a high level of extracellular ROS, for example, for imaging of inflammation, where high levels of superoxide and hydrogen peroxide are generated by immune cells.(38) Rapid intracellular reduction of [1-13C]-DHA was observed, both in EL4 tumor cell suspensions in vitro and in EL4 tumor xenografts in vivo. The rate of reduction could not be accounted for by the direct reaction with glutathione. [1-13C]-DHA is transported by the glucose transporters GLUT1 and 3, which are often overexpressed in tumor cells but are also abundant at the blood–brain barrier. Given the association of oxidative stress with neurodegenerative diseases and the avid utilization of vitamin C in the brain,(49) [1-13C]-DHA may provide a hyperpolarized probe for clinical imaging of redox status in vivo not only in cancer but in a wide range of pathologies and could enable resolution of the role of vitamin C in both physiological and pharmacological situations.
Acknowledgments
This work was supported by a Cancer Research UK program grant to K.M.B. (C197/A3514) and by a Translational Resesarch Program Award from The Leukemia and Lymphoma Society. F.A.G. received a Cancer Research UK & Royal College of Radiologists (UK) clinical research training fellowship, and B.W.C.K. received a Cancer Research UK studentship.
Supporting Information Available
Method of oxidation of [1-13C]-AA to [1-13C]-DHA, as well as the UV spectrophotometric data and animal procedures in full. This material is available free of charge via the Internet at http://pubs.acs.org.
Supplementary Material
References
- Kurhanewicz J.; Vigneron D. B.; Brindle K.; Chekmenev E. Y.; Comment A.; Cunningham C. H.; Deberardinis R. J.; Green G. G.; Leach M. O.; Rajan S. S.; Rizi R. R.; Ross B. D.; Warren W. S.; Malloy C. R. Neoplasia 2011, 13, 81–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohndiek S. E.; Kettunen M. I.; Hu D. E.; Witney T. H.; Kennedy B. W. C.; Gallagher F. A.; Brindle K. M. Mol. Cancer Ther. 2010, 9, 3278–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day S. E.; Kettunen M. I.; Gallagher F. A.; Hu D. E.; Lerche M.; Wolber J.; Golman K.; Ardenkjaer-Larsen J. H.; Brindle K. M. Nat. Med. 2007, 13, 1382–7. [DOI] [PubMed] [Google Scholar]
- Albers M. J.; Bok R.; Chen A. P.; Cunningham C. H.; Zierhut M. L.; Zhang V. Y.; Kohler S. J.; Tropp J.; Hurd R. E.; Yen Y. F.; Nelson S. J.; Vigneron D. B.; Kurhanewicz J. Cancer Res. 2008, 68, 8607–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keshari K. R.; Wilson D. M.; Chen A. P.; Bok R.; Larson P. E.; Hu S.; Van Criekinge M.; Macdonald J. M.; Vigneron D. B.; Kurhanewicz J. J. Am. Chem. Soc. 2009, 131, 17591–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallagher F. A.; Kettunen M. I.; Hu D. E.; Jensen P. R.; Zandt R. I.; Karlsson M.; Gisselsson A.; Nelson S. K.; Witney T. H.; Bohndiek S. E.; Hansson G.; Peitersen T.; Lerche M. H.; Brindle K. M. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 19801–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallagher F. A.; Kettunen M. I.; Day S. E.; Hu D. E.; Ardenkjaer-Larsen J. H.; Zandt R.; Jensen P. R.; Karlsson M.; Golman K.; Lerche M. H.; Brindle K. M. Nature 2008, 453, 940–3. [DOI] [PubMed] [Google Scholar]
- Sattler U. G.; Mueller-Klieser W. Int. J. Radiat. Biol. 2009, 85, 963–71. [DOI] [PubMed] [Google Scholar]
- Cairns R.; Harris I.; Mak T. Nat. Rev. Cancer 2011, 11, 85–95. [DOI] [PubMed] [Google Scholar]
- Balendiran G. K.; Dabur R.; Fraser D. Cell Biochem. Funct. 2004, 22, 343–52. [DOI] [PubMed] [Google Scholar]
- Ballatori N.; Krance S. M.; Notenboom S.; Shi S.; Tieu K.; Hammond C. L. Biol. Chem. 2009, 390, 191–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lippert A. R.; Keshari K. R.; Kurhanewicz J.; Chang C. J. J. Am. Chem. Soc. 2011, 133, 3776–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vera J. C.; Rivas C. I.; Fischbarg J.; Golde D. W. Nature 1993, 364, 79–82. [DOI] [PubMed] [Google Scholar]
- Linster C. L.; Van Schaftingen E. FEBS J. 2007, 274, 1–22. [DOI] [PubMed] [Google Scholar]
- Bode A.; Cunningham L.; Rose R. Clin. Chem. 1990, 36, 1807–9. [PubMed] [Google Scholar]
- Tsukaguchi H.; Tokui T.; Mackenzie B.; Berger U. V.; Chen X. Z.; Wang Y.; Brubaker R. F.; Hediger M. A. Nature 1999, 399, 70–5. [DOI] [PubMed] [Google Scholar]
- Deutsch J. C. J. Chromatogr. A 2000, 881, 299–307. [DOI] [PubMed] [Google Scholar]
- Kerber R. J. Chem. Educ. 2008, 85, 1237–42. [Google Scholar]
- Corti A.; Casini A. F.; Pompella A. Arch. Biochem. Biophys. 2010, 500, 107–15. [DOI] [PubMed] [Google Scholar]
- Rumsey S. C.; Kwon O.; Xu G. W.; Burant C. F.; Simpson I.; Levine M. J. Biol. Chem. 1997, 272, 18982–9. [DOI] [PubMed] [Google Scholar]
- Rumsey S.; Levine M. J. Nutr. Biochem. 1998, 9, 116–30. [Google Scholar]
- Levine A.; Puzio-Kuter A. Science 2010, 330. [DOI] [PubMed] [Google Scholar]
- Deutsch J. C. Anal. Biochem. 1998, 260, 223–9. [DOI] [PubMed] [Google Scholar]
- Ceccarelli J.; Delfino L.; Zappia E.; Castellani P.; Borghi M.; Ferrini S.; Tosetti F.; Rubartelli A. Int. J. Cancer 2008, 123, 1770–8. [DOI] [PubMed] [Google Scholar]
- Mena S.; Benlloch M.; Ortega A.; Carretero J.; Obrador E.; Asensi M.; Petschen I.; Brown B. D.; Estrela J. M. Clin. Cancer Res. 2007, 13, 2658–66. [DOI] [PubMed] [Google Scholar]
- Simons A. L.; Parsons A. D.; Foster K. A.; Orcutt K. P.; Fath M. A.; Spitz D. R. J. Oncol. 2009, 2009, 519–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong X.; Zhao F.; Thompson C. B. Curr. Opin. Genet. Dev. 2009, 19, 32–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vander Heiden M. G.; Cantley L. C.; Thompson C. B. Science 2009, 324, 1029–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita S.; Yamamoto Y. Anal. Biochem. 1997, 250, 66–73. [DOI] [PubMed] [Google Scholar]
- Battisti V.; Maders L. D.; Bagatini M. D.; Santos K. F.; Spanevello R. M.; Maldonado P. A.; Brule A. O.; Araujo Mdo C.; Schetinger M. R.; Morsch V. M. Clin. Biochem. 2008, 41, 511–8. [DOI] [PubMed] [Google Scholar]
- Ilangovan G.; Li H.; Zweier J. L.; Kuppusamy P. Mol. Cell. Biochem. 2002, 234–235, 393–8. [PubMed] [Google Scholar]
- Kuppusamy P.; Li H.; Ilangovan G.; Cardounel A. J.; Zweier J. L.; Yamada K.; Krishna M. C.; Mitchell J. B. Cancer Res. 2002, 62, 307–12. [PubMed] [Google Scholar]
- Hyodo F.; Soule B. P.; Matsumoto K.; Matsumoto S.; Cook J. A.; Hyodo E.; Sowers A. L.; Krishna M. C.; Mitchell J. B. J. Pharm. Pharmacol. 2008, 60, 1049–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Q.; Espey M. G.; Sun A. Y.; Lee J. H.; Krishna M. C.; Shacter E.; Choyke P. L.; Pooput C.; Kirk K. L.; Buettner G. R.; Levine M. Proc. Natl. Acad. Sci. U.S.A. 2007, 104, 8749–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascorbic Acid: Chemistry, Metabolism, and Uses; Seib P., Tolbert B., Eds.; American Chemical Society: Washington, DC, 1982; Vol. 200. [Google Scholar]
- Himmelreich U.; Drew K. N.; Serianni A. S.; Kuchel P. W. Biochemistry 1998, 37, 7578–88. [DOI] [PubMed] [Google Scholar]
- Winkler B. S.; Orselli S. M.; Rex T. S. Free Radical Biol. Med. 1994, 17, 333–49. [DOI] [PubMed] [Google Scholar]
- Burdon R. H. Free Radical Biol. Med. 1995, 18, 775–94. [DOI] [PubMed] [Google Scholar]
- Halliwell B. FEBS Lett. 2003, 540, 3–6. [DOI] [PubMed] [Google Scholar]
- Gille J. J.; Joenje H. Mutat. Res. 1992, 275, 405–14. [DOI] [PubMed] [Google Scholar]
- Buettner G. R.; Jurkiewicz B. A. Radiat. Res. 1996, 145, 532–41. [PubMed] [Google Scholar]
- Deutsch J. C. Anal. Biochem. 1998, 255, 1–7. [DOI] [PubMed] [Google Scholar]
- Moore G. E.; Gerner R. E.; Franklin H. A. J. Am. Med. Assoc. 1967, 199, 519–24. [PubMed] [Google Scholar]
- Koshiishi I.; Mamura Y.; Imanari T. Biochim. Biophys. Acta 1998, 1379, 257–63. [DOI] [PubMed] [Google Scholar]
- Park J. B. Biochim. Biophys. Acta 2001, 1525, 173–9. [DOI] [PubMed] [Google Scholar]
- Tietze F. Anal. Biochem. 1969, 27, 502–22. [DOI] [PubMed] [Google Scholar]
- Pilatus U.; Shim H.; Artemov D.; Davis D.; van Zijl P. C.; Glickson J. D. Magn. Reson. Med. 1997, 37, 825–32. [DOI] [PubMed] [Google Scholar]
- Hayes J. D.; McMahon M. Trends Biochem. Sci. 2009, 34, 176–88. [DOI] [PubMed] [Google Scholar]
- Agus D. B.; Gambhir S. S.; Pardridge W. M.; Spielholz C.; Baselga J.; Vera J. C.; Golde D. W. J. Clin. Invest. 1997, 100, 2842–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benetis N.; Holmén K.; Kowalewski J.; Nordenskiöld L.; Wahlberg O. Acta Chem. Scand. A 1981, 35, 513–20. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.