Abstract
There are two important mechanisms of activation of invariant natural killer T cells (iNKT cells) by microbes: direct activation of the invariant T-cell receptor (TCR) by microbial glycolipids presented by CD1d and indirect activation, mediated by the responses of antigen-presenting cells to microbes. In this study, we provide evidence for a novel CD1d-independent direct activation of iNKT cells involving a microbial protein superantigen presented in the context of major histocompatibility complex class II (MHC-II), which plays a critical role in pathogenesis, thereby redefining the role of iNKT cells. Intranasal exposure to staphylococcal enterotoxin B (SEB) in C57BL/6 wild-type mice caused acute lung injury (ALI) characterized by vascular leak, cytokine storm, and infiltration of mononuclear cells in the lungs. In contrast, the vascular leak and inflammation were decreased by ∼50% in NKT cell-deficient Jα18−/− and CD1d−/− mice following SEB exposure, which was reversed following adoptive transfer of iNKT cells into CD1d−/− mice. In vitro, SEB could directly stimulate iNKT cells in a CD1d-independent manner via MHC-II/TCR interaction, specifically involving Vβ8. These studies not only demonstrate that iNKT cells can be activated directly by a bacterial protein superantigen independent of CD1d but also indicate that in addition to the conventional T cells, iNKT cells play a critical role in SEB-mediated ALI.
INTRODUCTION
Natural killer T (NKT) cells are a unique subset of T lymphocytes, which coexpress the T cell receptor (TCR) and NK cell marker (NK1.1 or CD161c). NKT cells are the first responders to microbial antigens, and within a few hours of activation, they secret a variety of pro- and anti-inflammatory cytokines, modulating innate and adaptive immune responses (23). There are two main types of NKT cells: invariant or CD1d-dependent and CD1d-independent NKT cells (14, 23). Invariant NKT (iNKT) cells were named as such because they carry TCRs with a specific α-chain (Vα14/Jα18 in mice and Vα24 in humans) paired with limited β chains (Vβ8.2 and Vβ2 in mice and Vβ11 in humans) (37). The TCR of the CD1d-independent NKT cells is variable, and the antigen-presenting molecule, CD1d, is not required for the activation of the CD1d-independent NKT cells (2, 5). NKT cells arise in the thymus and diverge from the progenitor T cells at the CD4+ CD8+ double-positive stage (15). The mature NKT cells can be found in the spleen (approximately 2.5% of T cells), liver (up to 30% of the lymphocytes), lymph nodes, and bone marrow of mice, and in humans, about 1% of the lymphocytes in the liver are NKT cells (14, 27).
iNKT cells are activated when a glycolipid is presented in the context of CD1d. CD1d is a nonclassical major histocompatibility complex class I (MHC-I)-like antigen presentation molecule with a hydrophobic groove, and even though the cellular glycosylphosphatidylinositol (GPI) is a natural ligand for CD1d, this ligand does not activate iNKT cells (35, 37). Alpha-galactosylceramide (α-GalCer) is a molecule derived from the marine sponge Agelas mauritianus and, when presented by CD1d, is a potent activator of iNKT cells in both humans and mice (5, 23, 33). CD1d can also present other glycolipid ligands to iNKT cells such as bacterial glycolipids (i.e., phosphatidylinositol mannoside and α-galacturonsylceramide) and synthetic compounds (i.e., analogs of α-GalCer) (4). Secretion of several cytokines by other cells such as interleukin-12 (IL-12), IL-18, and type 1 interferons (IFNs) can also activate iNKT cells, and this is called the indirect pathway of activation. Once activated, iNKT cells produce a plethora of cytokines including IFN-γ, IL-4, and IL-2.
Staphylococcal enterotoxin B (SEB) belongs to a group of potent antigens called superantigens, and SEB can lead to a robust activation of the immune system (7). This toxin is produced by the Gram-positive Staphylococcus aureus; when ingested, it causes food poisoning, and when inhaled, it results in acute lung injury (ALI), acute respiratory distress syndrome, toxic shock, and, in severe cases, death (9). SEB is listed as a category B priority agent by the Centers for Disease Control and Prevention as it can be used in biological warfare (9). SEB is a 28-kDa protein, and unlike regular antigens, this protein activates the immune system as an intact molecule without being processed by the antigen-presenting cell. In addition, while a conventional antigen activates only about 0.1% of the T cell population in a host, SEB can activate 5 to 30% of the T cell population (12). The intact protein binds both to the nonpolymorphic parts of the MHC-II and to the variable region of the β chain of the TCR (such as Vβ8 TCR). This binding activates the potent CD4+ or CD8+ T cells and results in a cytokine storm (3).
The invariant receptor of the iNKT cells also possesses Vβ8, Vβ7, or Vβ2 specificities, and even though it has been well established that the direct pathway of iNKT cell activation is triggered by glycolipids presented by CD1d, we hypothesized that SEB, as a protein, can directly activate iNKT cells through the TCR in a CD1d-independent manner, thereby resulting in acute lung injury. In the current study, we report two novel findings: (i) in addition to the importance of T cells, we identify the direct involvement of iNKT cells as innate responders in SEB-induced acute lung injury, and (ii) we demonstrate for the first time that iNKT cells can be activated through the TCR by a microbial protein antigen such as SEB, in the context of MHC-II and independent of CD1d.
MATERIALS AND METHODS
Animals.
Female C57BL/6 mice at the age of 6 to 8 weeks were purchased from the National Cancer Institute and housed at the animal facility of University of South Carolina, School of Medicine (Columbia, SC). Jα18−/− and CD1d−/− mice on a C57BL/6 background were age matched to wild-type (WT) mice for each experiment. A Vα14 transgenic (Tg) breeding pair was a kind gift from Albert Bendelac (University of Chicago, Chicago, IL). All procedures were approved by the Institutional Animal Care and Use Committee and were in agreement with the guidelines of the National Institutes of Health.
Evans blue dye extraction.
Vascular leak in the lungs of mice was measured as described previously (8, 21). Briefly, we administered SEB (50 μg/mouse) that was dissolved in sterile phosphate-buffered saline (PBS) through the intranasal route. The control mice received intranasal administration of sterile PBS. After 24 to 72 h, the mice were injected intravenously (i.v.) with 1% Evans blue. Two hours after dye injection, the mice were exsanguinated and perfused through the heart with heparinized PBS. After the complete removal of peripheral blood was ensured, the lungs were collected and placed in formamide. The organs were incubated at 37°C for 24 h, and the absorbance of formamide was measured at 620 nm. The percent increase in vascular leak was calculated with the following formula: (ODsample − ODcontrol)/ODcontrol × 100, where OD is optical density.
Adoptive transfer.
We purified NKT cells from the spleens of Vα14 Tg mice by labeling the cells with CD1d/PBS-57 tetramer and sorting the cells with an Aria fluorescence-activated cell sorter (FACS). CD1d−/− mice received 2 × 106 NKT cells per mouse intravenously immediately before they received the intranasal administration of SEB. The mice were kept for 48 h, and Evans blue dye extraction studies were followed as described above.
Histology.
Sterile PBS (50 μl) or SEB (50 μg/mouse in 50 μl sterile PBS) was administered through the intranasal route into WT, Jα18−/−, and CD1d−/− mice, and 48 h later the lungs were collected for histopathology. The organs were immediately placed in histological grade 10% formalin and fixed overnight. The tissues were processed with paraffin embedding, Vibratome sectioning, and staining for hematoxylin and eosin (H&E). The pictures were taken at magnifications of ×4 and ×100 with a light microscope.
Isolation of lung-infiltrating cells and flow cytometry.
Mice received intranasal administration of PBS or SEB (50 μg/mouse); 48 h later the lungs were excised, and a single-cell suspension was obtained with a tissue homogenizer. The cells were then washed and layered on Histopaque in order to isolate mononuclear cells. The mononuclear cells were counted and stained with anti-Vβ8-fluorescein isothiocyanate (FITC) and phycoerythrin (PE)-labeled CD1d/PBS-57 tetramer (CD1d/PBS-57–PE). PBS-57 is an analog of α-GalCer, and CD1d/PBS-57 tetramer was provided by the NIH tetramer facility.
Measurement of cytokines in serum and BALF.
Sterile PBS (50 μl) or SEB (50 μg/mouse in 50 μl sterile PBS) was administered through the intranasal route into WT, Jα18−/−, and CD1d−/− mice. On day 2, blood was collected, and serum was separated. In order to collect bronchoalveolar lavage fluid (BALF), mice were euthanized, the trachea was tied, and the lungs were excised as a whole set. Then, 1 ml of cold sterile PBS was pushed through the opening of the trachea several times, and the supernatants were collected. Cytokine analysis was performed with individual enzyme-linked immunosorbent assay (ELISA) kits for IL-2, tumor necrosis alpha (TNF-α), and IFN-γ.
DC and iNKT cell coculture experiments.
The dendritic cells (DCs) were generated from the bone marrow of WT mice by incubating the cells in the presence of granulocyte-macrophage colony-stimulating factor (GM-CSF) for 10 days, as described previously (18). The DCs were collected and placed in 96-well culture plates in the presence of antigen (α-GalCer at 200 ng/ml or SEB at 1 μg/ml) and purified iNKT cells as described below. In some experiments, the cultures were incubated with isotype control (clone A95-1, catalog item 553986; BD Biosciences), anti-MHC-II (clone M5/114.15.2, catalog item 556999; BD Biosciences), or anti-TCR (clone KJ16-133.18, catalog item 118401; Biolegend) (10 μg/ml) antibody for 2 h prior to addition of SEB into the cultures. To obtain iNKT cells, splenocytes of Vα14 transgenic mice were stained with CD1d/PBS-57 tetramer. The iNKT cells were then sorted with a FACS Aria. The purity of the DCs and the iNKT cells was determined with flow cytometry. Sorted iNKT cells were added to the DC cultures (1 × 104 DCs to 1 × 105 NKT cells) and cultured at 37°C for 72 h. The supernatants were collected, and the levels of IFN-γ and IL-4 were measured with ELISA.
Statistics.
The data that are shown in this paper represent at least three independent experiments. The means ± standard errors of the means (SEM) are shown for experiments that are applicable. The statistical difference was calculated with a Student's t test and analysis of variance (ANOVA) with a posthoc Tukey test in applicable experiments. A P value of ≤0.05 was considered to be statistically significant.
RESULTS
NKT cells regulate SEB-induced vascular injury in the lungs of mice.
Exposure to staphylococcal enterotoxins leads to toxic shock syndrome characterized by damage to the endothelial cells, capillary leak, hypotension, and shock, which together contribute to death of the host (17). In this study, we used two different murine knockout (KO) models in order to investigate the role of NKT cells in SEB-induced vascular leak and lung injury. The first type of knockout mice lacks Jα18 (Jα18KO) and therefore fails to form the invariant TCR of the iNKT cells. The second model consists of mice that lack the antigen-presenting molecule CD1d (CD1dKO), and as a result, the NKT cells fail to get positively selected during development. It is important to note that while Jα18−/− mice lack only the iNKT cells, CD1d−/− mice are deficient in both types of NKT cells.
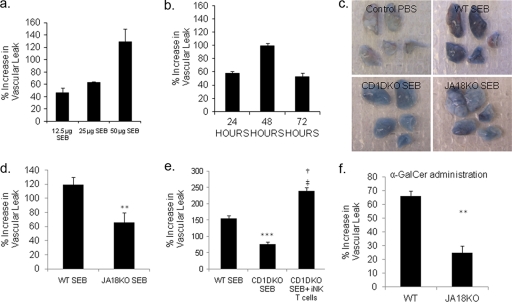
We administered different doses of SEB (12.5, 25, and 50 μg) into wild-type mice through the intranasal route and measured the vascular leak at different time intervals, as described previously (8, 21, 22). The data demonstrated that vascular injury in the lungs induced by SEB was dose dependent, peaked at 48 h, and started to decline at 72 h (Fig. 1 a and b). Next, we administered PBS or SEB (50 μg/mouse) into wild-type (WT), Jα18−/−, or CD1d−/− mice and measured the vascular leak in the lungs after 48 h. Figure 1c depicts leakage of Evans blue dye into the interstitium of the lungs after 48 h of SEB exposure. The lungs from WT mice exposed to SEB appeared darker, thereby showing increased vascular leak compared with WT mice administered PBS. Interestingly, the lungs from SEB-exposed Jα18−/− and CD1d −/− mice showed significantly less vascular leak than SEB-exposed WT mice.
Fig. 1.
NKT cells regulate SEB-induced vascular injury in the lungs of mice. Groups of five C57BL/6 wild-type mice were administered SEB or PBS intranasally, and vascular leak in the lungs was measured as described in Materials and Methods. (a) Analysis of vascular leak in the lungs of mice using different doses of SEB and detection of peak response at 50 μg/mouse. (b) Time course for the vascular injury induced by SEB at 24, 48, and 72 h after SEB administration. (c) Set of representative lungs from each group showing the leakage of Evans blue into the interstitium 48 h after SEB or PBS exposure. (d and e) Quantification of vascular injury in lungs of WT mice compared to Jα18−/− and CD1d −/− mice after 48 h of exposure to SEB. (f) Quantification of vascular injury in lungs of WT mice compared to Jα18−/− mice after intranasal α-GalCer (10 μg/mouse) administration (**, P < 0.01 by a Student t test; ANOVA, P < 0.001 with a Tukey test; ***, P < 0.05 versus WT SEB; ‡, P < 0.05 versus WT SEB; †, P < 0.05 versus CD1dKO SEB).
Quantification of vascular leak also demonstrated that at 48 h, Jα18−/−, and CD1d−/− mice had significantly less vascular leak than SEB-exposed WT mice (Fig. 1d and e). The residual vascular leak seen in Jα18−/− and CD1d−/− mice is likely to result from the activation of T cells. To further corroborate the role of iNKT cells, we adoptively transferred CD1d/PBS-57 tetramer-sorted iNKT cells into CD1d−/− mice and found that the vascular leak was not only restored but also significantly increased compared to the degree of vascular leak in SEB-exposed WT mice (Fig. 1d). We also administered α-GalCer (10 μg/mouse) through the intranasal route into WT and Jα18−/− mice and found that specific activation of iNKT cells resulted in a significant increase in vascular leak in WT mice (Fig. 1f). α-GalCer treatment of WT mice resulted in reduced vascular leak compared to SEB administration because, unlike α-GalCer, SEB also activates T cells. Jα18−/− mice administered α-GalCer were used as a negative control in this study (Fig. 1f).
Absence of NKT cells leads to decreased inflammation in the lungs upon SEB exposure.
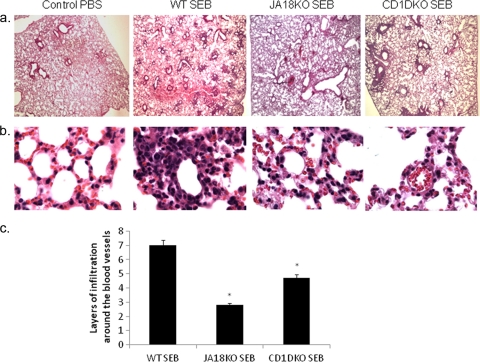
We next investigated whether the decreased vascular injury seen in SEB-exposed Jα18−/− and CD1d−/− mice resulted from altered levels of inflammation in the lungs. To this end, mice were exposed to PBS or SEB (50 μg/mouse), and 48 h later, lungs were harvested and analyzed using H&E staining. Images taken at a magnification of ×4 showed that SEB exposure led to dramatic infiltration of immune cells into the lungs of WT mice (Fig. 2 a). The infiltration was diffused through the whole section of the lungs, and all of the blood vessels had pericapillary rings of infiltrating cells. On the other hand, Jα18−/− and CD1d−/− mice had fewer blood vessels with layers of infiltration after SEB inhalation (Fig. 2a). At a magnification of ×100, the signs of acute lung injury were evident in SEB-exposed WT mice, where the airways were collapsed and the hyaline membranes were evident. On the other hand, the structure of the lungs of SEB-exposed Jα18−/− and CD1d−/− mice appeared normal, with distinct airways (Fig. 2b). In order to quantify the degree of infiltration, we counted the layers of cells around 10 different capillaries from each group. The numbers of layers of infiltration were significantly reduced in Jα18−/− and CD1d−/− mice compared to the number in WT mice after SEB exposure (Fig. 2c). The fact that both the vascular leak (Fig. 1) and inflammation (Fig. 2) in NKT cell-deficient mice exposed to SEB were reduced by ∼50% was a surprising finding, considering that SEB is known to activate only the conventional T cells, and these data pointed toward the critical role of iNKT cells in SEB-mediated ALI.
Fig. 2.
Absence of NKT cells leads to decreased infiltration in the lungs upon SEB exposure. Mice were administered 50 μg/mouse SEB through the intranasal route. (a and b) The lung sections from SEB-exposed animals were stained with H&E, and the photographs were taken at magnifications of ×4 (a) and ×100 (b). Cellular infiltration is indicated by arrows. (c) The layers of infiltrating cells were counted around 10 different capillaries from each group and averaged for quantification (*, P < 0.05 versus WT SEB by a Student t test).
Cytokine storm induced by SEB is alleviated in mice that lack NKT cells.
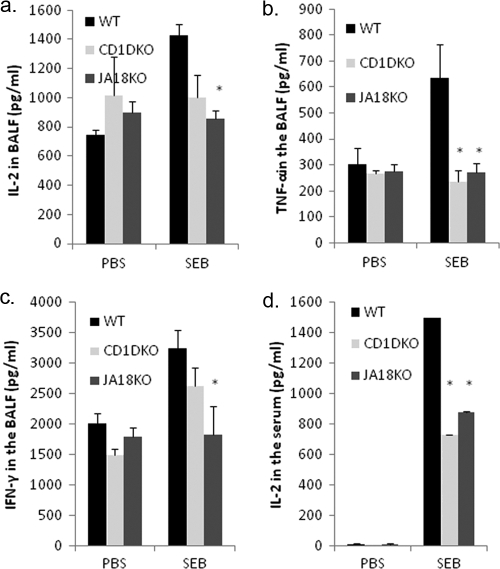
It is well established that as a superantigen, SEB activates large numbers of T cells expressing certain Vβ specificities, such as Vβ8, which triggers a plethora of cytokines such as IL-2, IFN-γ, and TNF-α. We tested the role of iNKT cells in the production of such cytokines following exposure to SEB by quantifying their levels both in the bronchoalveolar lavage fluid (BALF) and serum in WT, Jα18−/−, and CD1d−/− mice. SEB exposure significantly increased the amount of IL-2, TNF-α, and IFN-γ in the BALF of WT mice compared to levels in PBS-exposed WT mice. In the BALF of Jα18−/− mice, all of the three cytokines were significantly reduced compared to levels in SEB-exposed WT animals (Fig. 3 a, b, and c). However, in the BALF of CD1d−/− mice, only TNF-α levels were significantly reduced compared to levels in SEB-exposed WT mice (Fig. 3b). This can be explained by the differences in the nature of NKT cell deficiency in Jα18−/− and CD1d−/− mice. We further measured the circulating cytokine levels in the serum, and the only cytokine that was detectable after SEB exposure was IL-2. In WT SEB-exposed mice, there was a dramatic increase in IL-2 levels in the serum while SEB-exposed Jα18−/− and CD1d−/− mice had significantly lower levels of this cytokine (Fig. 3d). It should be noted that the bronchoalveolar lavage fluid from PBS-exposed mice had higher levels of background cytokines than serum probably because of direct exposure of the lungs to environmental antigens.
Fig. 3.
Cytokine storm induced by SEB is alleviated in mice that lack NKT cells. Groups of five mice were exposed to PBS or SEB through the intranasal route, and 48 h later, IL-2, TNF-α, and IFN-γ were measured in the BALF (a, b, c) and in the serum (d) of mice (*, P < 0.05 versus WT SEB by a Student t test).
Administration of α-GalCer or SEB results in increased iNKT cells in the lungs.
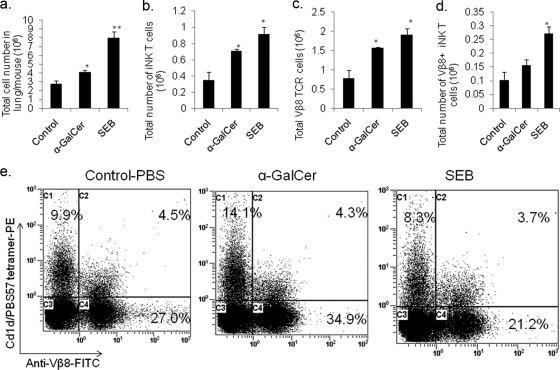
In order to study the local immune response to α-GalCer or SEB, we administered these antigens through the intranasal route and counted the number of lung-infiltrating cells. Control mice received intranasal administration of sterile PBS. Inhalation of α-GalCer resulted in an increase in total number of cells in the lungs; however, SEB administration had an even greater effect, with the greatest number of cells recruited into the lungs (Fig. 4 a). We then labeled the cells with an iNKT cell-specific tetramer (CD1d/PBS-57) as well as anti-Vβ8. The data from a representative experiment are shown in Fig. 4e. Based on the percentages of various subsets (Fig. 4e) and the total number of infiltrating cells (Fig. 4a), we calculated the absolute numbers of various subsets of cells infiltrating the lungs. The numbers of iNKT cells and Vβ8+ cells (includes T cells and iNKT cells) were significantly increased in both α-GalCer- and SEB-treated mice compared to numbers in control mice (Fig. 4b and c). Interestingly, the number of cells that were positive for both CD1d-PBS-57 and Vβ8-TCR was significantly increased relative to control levels only in SEB-treated mice (Fig. 4d).
Fig. 4.
Intranasal administration of α-GalCer or SEB results in the infiltration of lymphocytes into the lungs of WT mice. (a) The lung-infiltrating cells were collected 48 h after antigen administration. The cells were then stained with CD1d/PBS-57 tetramer (iNKT cell marker) and antibodies against Vβ8 TCR. The percentages were applied to the total number of cells in each group in order to obtain the total number of each population. (b, c, and d) Absolute cell numbers in the lungs of mice; data are shown for iNKT cells, Vβ8 TCR+ cells, and iNKT cells, as indicated, that are Vβ8 TCR+ positive. (e) Flow cytometry data where the lung-infiltrating cells were stained with CD1d/PBS-57–PE tetramer and anti-Vβ8 TCR-FITC (ANOVA, P < 0.02 with a Tukey test; *, P < 0.05 versus control; ***, P < 0.01 versus control).
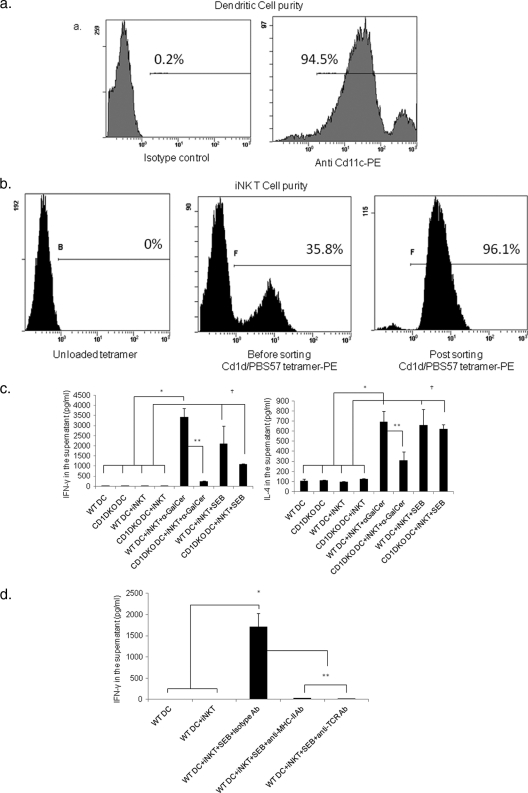
SEB directly activates iNKT cells.
In order to demonstrate the direct activation of iNKT cells with SEB, we cocultured bone marrow-derived dendritic cells (BMDCs) with sorted iNKT cells in the presence or absence of SEB. The purity of the cells that were used in cocultures is shown in Fig. 5 a and b. Dendritic cells from WT mice cultured with α-GalCer were used as positive controls in stimulating iNKT cells. In the first set of experiments, we utilized BMDCs from WT as well as CD1d−/− mice. We demonstrated that α-GalCer-pulsed WT BMDCs were able to stimulate iNKT cells very efficiently to produce high levels of IFN-γ and IL-4. Interestingly, SEB-pulsed WT BMDCs were also able to stimulate iNKT cells to produce significantly high levels of IFN-γ and IL-4. As expected, CD1d−/− BMDCs were unable to stimulate cytokine production in the presence of α-GalCer; however, in the presence of SEB, CD1d−/− BMDCs stimulated iNKT cells to produce levels of IFN-γ and IL-4 similar to those of the WT BMDCs (Fig. 5c). In the second set of experiments, we cocultured WT BMDCs with iNKT cells and used antibodies against MHC-II and TCR. We found that antibodies against MHC-II and TCR completely suppressed IFN-γ production compared to the isotype control antibodies (Fig. 5d). Together, these data conclusively demonstrated that SEB-induced activation of iNKT cells is independent of CD1d and is restricted by MHC-II–TCR interactions.
Fig. 5.
SEB can directly activate iNKT cells through TCR involving MHC-II but independent of CD1d. (a and b) Purity of the dendritic cells and iNKT cells was determined before the cells were placed in cocultures. iNKT cells for these experiments were purified from the spleens of Vα14 Tg mice. (c) Purified BMDCs from WT and CD1d−/− mice were mixed with purified iNKT cells from Vα14 Tg mice in the presence of α-GalCer or SEB for 72 h. Next, the culture supernatants were analyzed for IFN-γ and IL-4 (ANOVA, P < 0.005 with Tukey test *, **, ‡, P < 0.05). (d) WT BMDCs were pulsed with SEB and placed in coculture with iNKT cells in the presence of isotype control antibodies or antibodies against MHC-II or TCR (10 μg/ml) (ANOVA: *, P < 0.003 with a Tukey test; **, P < 0.05).
DISCUSSION
NKT cells have been called many things such as “double-edged sword of the immune system,” “Swiss-army knife of the immune system,” and “bridge between innate and adaptive immune systems” (5, 20, 33). This stems from the fact that NKT cells can up- or downregulate the immune functions and play important roles in many disease states such as diabetes, multiple sclerosis, allergy, transplantation, and microbial immunity (11, 25, 31, 36). In the case of lung inflammation, NKT cells can exacerbate or ameliorate the disease, depending on the microorganism and the strain (6). While previous studies have explored the role of NKT cells in the regulation of the T cell response to microbes or their toxins (19, 26), thus far, no studies have demonstrated whether iNKT cells directly respond to bacterial enterotoxins such as SEB. In the current study, we investigated the role of iNKT cells in SEB-induced ALI and demonstrated that mice that lack either the iNKT cells or the whole NKT cell population had significantly reduced levels of lung injury upon SEB exposure compared to WT mice. Moreover, adoptive transfer of purified iNKT cells into CD1d−/− mice significantly increased vascular leak in the lungs of these mice following SEB exposure. We used CD1d−/− mice for the adoptive transfer experiments in order to rule out activation via any endogenous NKT cell ligands. In addition, the use of CD1d−/− mice confirmed that SEB activates iNKT cells in a CD1d-independent manner. Furthermore, our in vitro studies using purified iNKT cells cultured with WT DCs or CD1d−/− DCs and antibody blocking studies against MHC-II and TCR demonstrated conclusively that activation of iNKT cells by SEB was CD1d independent and regulated by MHC-II.
In the current study, we used SEB intranasally to induce ALI. It should be noted that mice are more resistant to SEB than humans because human MHC-II can bind SEB better than mouse MHC-II (32). Thus, mice can survive even a high dose of SEB, such as 100 μg/mouse. Interestingly, however, it was recently reported that mice were highly sensitive if they were given two low doses of SEB within 2 h (10, 13). Thus, in this study, the first intranasal dose consisted of 5 μg of SEB/mouse, and the second dose consisted of intraperitoneal (i.p.) injection of 2 μg of SEB/mouse. Such mice died within 96 to 120 h while exhibiting severe lung injury. These studies suggested that while mice are more resistant to SEB, they can develop severe lung inflammation and die following exposure to relatively low concentrations of SEB through the intranasal route (10, 13). Thus, the binding of MHC-II to SEB may not be the sole cause of increased resistance to SEB seen in mice. The authors attributed this to increased concentrations of MCP-1 within the lungs of SEB-treated mice, leading to leukocyte recruitment into this organ.
Staphylococcal enterotoxin B is known to activate T cells primarily bearing Vβ8 as well as the Vβ2 and Vβ7 regions of the TCR, which results in rapid expansion of these cells, massive release of cytokines, and lethality in mice (19, 26). Interestingly, a majority of the iNKT cells express a TCR with a Vβ8 chain and, to a lesser extent, with Vβ2 and Vβ7 (14). These findings are consistent with our observation that SEB exposure in the lungs led to significant expansion of iNKT cells expressing Vβ8. The hallmark of the iNKT cell response is the rapid and copious production of cytokines, specifically, secretion of the prototypical Th1 and Th2 cytokines, IFN-γ and IL-4, respectively (14). Activation of iNKT cells also leads to the immediate activation of other cell types. For example, within hours of α-GalCer stimulation of iNKT cells, NK cells are stimulated to secrete IFN-γ (14). Thus, the lung injury may result from a combination of cytokines produced by iNKT cells and T cells that are directly activated by SEB, as well as other cells such as NK cells, which are activated indirectly by the cytokine storm. The impact of SEB administration on cytokine production in vivo has also been addressed in the context of the presence or absence of NKT cells (30). It was shown that NKT cells positively regulated the secretion of IL-4 and IFN-γ production in response to SEB while negatively regulating the secretion of TNF-α (30). While these studies showed the potential indirect role played by NKT cells in the regulation of the T cell response to SEB challenge, the authors did not address whether NKT cells would respond directly to SEB.
The underlying mechanism of vascular injury is initiated by the infiltration of mononuclear cells into the lungs, and we noted that Jα18−/− and CD1d−/− mice had decreased levels of cellular infiltration into the lungs compared with WT mice after SEB exposure. These data also correlated with decreased levels of endothelial cell damage, as evident from our confocal microscopy studies (data not shown). Moreover, we also noted that in the BALF, the NKT cell-deficient mice showed decreased levels of inflammatory cytokines including IL-2, IFN-γ, and TNF-α. Our group has previously demonstrated that IL-2 cytokine therapy subsequently mediates endothelial cell injury through induction of LAK cells. The mechanism involves interaction between CD44 expressed on LAK cells and hyaluronic acid on the endothelial cells, which activate the LAK cells, produce perforin and FasL, and kill the endothelial cells (28, 29). iNKT cells have also been shown to secrete perforin as well as Fas ligand to induce cytotoxicity of target cells (24). Thus, the decreased endothelial cell injury seen in iNKT cell-deficient mice (Fig. 3) may result from lack of the cytolytic activity by iNKT cells as well as from reduced cytokine production (Fig. 4).
iNKT cells participate in the development of detrimental immune responses, including the induction of airway hypersensitivity, which requires IL-4 and IL-13 (1). The role of iNKT cells in acute lung injury and lethal shock caused by lipopolysaccharide (LPS) were also indicated by demonstrating that sensitization of mice with α-GalCer followed by LPS administration leads to an increased iNKT cell population in the lungs of mice as well as increased mortality (34). In this study, we compared intranasal administration of α-GalCer and SEB and demonstrated that administration of both antigens results in a significant increase of iNKT cells in the lungs. Interestingly, administration of α-GalCer alone was sufficient to trigger vascular leak in the lungs of WT but not NKT cell-deficient mice. These findings suggested that activation of iNKT cells alone in the lungs is sufficient to cause significant lung injury. Thus, SEB, which activates both iNKT cells and conventional T cells, is likely to cause a robust cytokine storm and lung injury.
iNKT cells are activated by diverse microbial infections. There are two important mechanisms of activation of iNKT cells by microbes: direct activation of the invariant TCR by microbial glycolipids presented by CD1d and indirect activation, which is mediated by the responses of antigen-presenting cells (APC) to microbes (16). The APCs produce cytokines such as IL-12 and/or present self-glycolipid antigens in the context of CD1d to activate iNKT cells. The indirect mechanism of activation allows iNKT cells to respond rapidly to diverse microbes despite a limited TCR diversity. In the current study, we provide evidence for an additional direct mechanism of iNKT cell activation involving a microbial protein superantigen, SEB, via cross-linking of MHC-II and TCR, thereby redefining the role of these cells in bacterial pathogenesis. We demonstrate that iNKT cells are important players in SEB-induced ALI, and their direct activation by SEB plays a key role in the initiation and amplification of the immune response that results in acute lung injury.
ACKNOWLEDGMENTS
This study was supported in part by NIH grants P01-AT03961, R01-ES09098, R01ES019313-02, and R01-DA016545.
Footnotes
Published ahead of print on 31 May 2011.
REFERENCES
- 1. Akbari O., et al. 2003. Essential role of NKT cells producing IL-4 and IL-13 in the development of allergen-induced airway hyperreactivity. Nat. Med. 9:582–588 [DOI] [PubMed] [Google Scholar]
- 2. Arrenberg P., Halder R., Kumar V. 2009. Cross-regulation between distinct natural killer T cell subsets influences immune response to self and foreign antigens. J. Cell. Physiol. 218:246–250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Baker M. D., Acharya K. R. 2004. Superantigens: structure-function relationships. Int. J. Med. Microbiol. 293:529–537 [DOI] [PubMed] [Google Scholar]
- 4. Balato A., Unutmaz D., Gaspari A. A. 2009. Natural killer T cells: an unconventional T-cell subset with diverse effector and regulatory functions. J. Invest. Dermatol. 129:1628–1642 [DOI] [PubMed] [Google Scholar]
- 5. Berzins S. P., Smyth M. J., Godfrey D. I. 2005. Working with NKT cells—pitfalls and practicalities. Curr. Opin. Immunol. 17:448–454 [DOI] [PubMed] [Google Scholar]
- 6. Boyton R. 2008. The role of natural killer T cells in lung inflammation. J. Pathol. 214:276–282 [DOI] [PubMed] [Google Scholar]
- 7. Florquin S., Aaldering L. 1997. Superantigens: a tool to gain new insight into cellular immunity. Res. Immunol. 148:373–386 [DOI] [PubMed] [Google Scholar]
- 8. Guan H., Nagarkatti P. S., Nagarkatti M. 2007. Blockade of hyaluronan inhibits IL-2-induced vascular leak syndrome and maintains effectiveness of IL-2 treatment for metastatic melanoma. J. Immunol. 179:3715–3723 [DOI] [PubMed] [Google Scholar]
- 9. Henghold W. B., 2nd 2004. Other biologic toxin bioweapons: ricin, staphylococcal enterotoxin B, and trichothecene mycotoxins. Dermatol. Clin. 22:257–262 [DOI] [PubMed] [Google Scholar]
- 10. Huzella L. M., Buckley M. J., Alves D. A., Stiles B. G., Krakauer T. 2009. Central roles for IL-2 and MCP-1 following intranasal exposure to SEB: a new mouse model. Res. Vet. Sci. 86:241–247 [DOI] [PubMed] [Google Scholar]
- 11. Iwamura C., Nakayama T. 2007. Role of α-galactosylceramide-activated Vα14 natural killer T cells in the regulation of allergic diseases. Allergol. Int. 56:1–6 [DOI] [PubMed] [Google Scholar]
- 12. Krakauer T. 2005. Chemotherapeutics targeting immune activation by staphylococcal superantigens. Med. Sci. Monit. 11:RA290–RA295 [PubMed] [Google Scholar]
- 13. Krakauer T., Buckley M. J., Huzella L. M., Alves D. A. 2009. Critical timing, location and duration of glucocorticoid administration rescue mice from superantigen-induced shock and attenuate lung injury. Int. Immunopharmacol. 9:1168–1174 [DOI] [PubMed] [Google Scholar]
- 14. Kronenberg M. 2005. Toward an understanding of NKT cell biology: progress and paradoxes. Annu. Rev. Immunol. 23:877–900 [DOI] [PubMed] [Google Scholar]
- 15. Kronenberg M., Engel I. 2007. On the road: progress in finding the unique pathway of invariant NKT cell differentiation. Curr. Opin. Immunol. 19:186–193 [DOI] [PubMed] [Google Scholar]
- 16. Kronenberg M., Kinjo Y. 2009. Innate-like recognition of microbes by invariant natural killer T cells. Curr. Opin. Immunol. 21:391–396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lee P. K., Vercellotti G. M., Deringer J. R., Schlievert P. M. 1991. Effects of staphylococcal toxic shock syndrome toxin 1 on aortic endothelial cells. J. Infect. Dis. 164:711–719 [DOI] [PubMed] [Google Scholar]
- 18. Lutz M. B., et al. 1999. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J. Immunol. Methods 223:77–92 [DOI] [PubMed] [Google Scholar]
- 19. Marrack P., Blackman M., Kushnir E., Kappler J. 1990. The toxicity of staphylococcal enterotoxin B in mice is mediated by T cells. J. Exp. Med. 171:455–464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Matsuda J. L., Mallevaey T., Scott-Browne J., Gapin L. 2008. CD1d-restricted iNKT cells, the “Swiss-Army knife” of the immune system. Curr. Opin. Immunol. 20:358–368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. McKallip R. J., et al. 2003. Targeted deletion of CD44v7 exon leads to decreased endothelial cell injury but not tumor cell killing mediated by interleukin-2-activated cytolytic lymphocytes. J. Biol. Chem. 278:43818–43830 [DOI] [PubMed] [Google Scholar]
- 22. Melencio L., et al. 2006. Role of CD4+ CD25+ T regulatory cells in IL-2-induced vascular leak. Int. Immunol. 18:1461–1471 [DOI] [PubMed] [Google Scholar]
- 23. Mercer J. C., Ragin M. J., August A. 2005. Natural killer T cells: rapid responders controlling immunity and disease. Int. J. Biochem. Cell Biol. 37:1337–1343 [DOI] [PubMed] [Google Scholar]
- 24. Metelitsa L. S., et al. 2001. Human NKT cells mediate antitumor cytotoxicity directly by recognizing target cell CD1d with bound ligand or indirectly by producing IL-2 to activate NK cells. J. Immunol. 167:3114–3122 [DOI] [PubMed] [Google Scholar]
- 25. Nagarajan N. A., Kronenberg M. 2007. Invariant NKT cells amplify the innate immune response to lipopolysaccharide. J. Immunol. 178:2706–2713 [DOI] [PubMed] [Google Scholar]
- 26. Newell K. A., Ellenhorn J. D., Bruce D. S., Bluestone J. A. 1991. In vivo T-cell activation by staphylococcal enterotoxin B prevents outgrowth of a malignant tumor. Proc. Natl. Acad. Sci. U. S. A. 88:1074–1078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Notas G., Kisseleva T., Brenner D. 2009. NK and NKT cells in liver injury and fibrosis. Clin. Immunol. 130:16–26 [DOI] [PubMed] [Google Scholar]
- 28. Rafi A. Q., et al. 1998. Evidence for the involvement of Fas ligand and perforin in the induction of vascular leak syndrome. J. Immunol. 161:3077–3086 [PubMed] [Google Scholar]
- 29. Rafi-Janajreh A. Q., et al. 1999. Evidence for the involvement of CD44 in endothelial cell injury and induction of vascular leak syndrome by IL-2. J. Immunol. 163:1619–1627 [PubMed] [Google Scholar]
- 30. Ragin M. J., Sahu N., August A. 2006. Differential regulation of cytokine production by CD1d-restricted NKT cells in response to superantigen staphylococcal enterotoxin B exposure. Infect. Immun. 74:282–288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Singh A. K., et al. 2001. Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis. J. Exp. Med. 194:1801–1811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Strandberg K. L., et al. 2010. Staphylococcal superantigens cause lethal pulmonary disease in rabbits. J. Infect. Dis. 202:1690–1697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Taniguchi M., Seino K., Nakayama T. 2003. The NKT cell system: bridging innate and acquired immunity. Nat. Immunol. 4:1164–1165 [DOI] [PubMed] [Google Scholar]
- 34. Tumurkhuu G., et al. 2008. The mechanism of development of acute lung injury in lethal endotoxic shock using alpha-galactosylceramide sensitization. Clin. Exp. Immunol. 152:182–191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. van der Vliet H. J., et al. 2004. The immunoregulatory role of CD1d-restricted natural killer T cells in disease. Clin. Immunol. 112:8–23 [DOI] [PubMed] [Google Scholar]
- 36. Van Kaer L. 2005. alpha-Galactosylceramide therapy for autoimmune diseases: prospects and obstacles. Nat. Rev. Immunol. 5:31–42 [DOI] [PubMed] [Google Scholar]
- 37. Van Kaer L. 2007. NKT cells: T lymphocytes with innate effector functions. Curr. Opin. Immunol. 19:354–364 [DOI] [PubMed] [Google Scholar]