ABSTRACT
We report on a 3 years old boy with severe mental retardation, hypotonia, deafness, cerebral dysmyelination, low levels of immunoglobulin A and dysmorphic features, bearing a distal deletion of 18q: 18q21-qter. Karyotype analysis by GTG banding was performed, revealing a deletion of the distal fragment of chromosome 18q, ranging from band q21 to qter, later confirmed by Fluorescence In Situ Hybridization (FISH). Whole chromosome painting for chromosome 18 ruled out any cryptic rearrangements involving other chromosomes. An attempted therapy with growth hormone did not bring any improvement and was discontinued due to an unexplained febrile episode and overall regress of the patient.
Keywords: 18q deletion, hypotonia, GH therapy
INTRODUCTION
The 18q-syndrome represents a contiguous gene deletion syndrome; it is one of the commonest of the human segmental aneusomies, with an estimated prevalence of about 1 in 40,000 live births (1). Like Wolf-Hirschhorn syndrome, Cri-du-Chat syndrome, and Miller-Dieker syndrome, it is a terminal deficiency or macrodeletion syndrome characterized by mental retardation and congenital malformations. The phenotype is highly variable, generally characterized by mental retardation, short stature, hypotonia, hearing impairment, dysmorphic features, and foot deformities. Low levels of immunoglobulin A and growth hormone deficiency were, also, reported. Common features include short stature, microcephaly, palatal defects, short frenulum, carp-like mouth, short palpebral fissures, and external ear anomalies. Cardiac anomalies are observed in 24% of cases, including atrial and ventricular septal defects, and pulmonary stenosis. Skeletal defects include scoliosis, and tapering fingers. Cognitive function ranges from normal intelligence to severe mental retardation. Other neurologic features include nystagmus, seizures, clumsiness, hearing loss, and diffuse white matter abnormalities on MRI. Most cases are sporadic, but an autosomal dominant transmission was also reported (2, 3). Feenstra et al.(4) refined genotype/phenotype correlations for several critical regions, including microcephaly (18q21.33), short stature (18q12.1-q12.3, 18q21.1-q21.33, and 18q22.3-q23), white matter disorders and delayed myelination (18q22.3-q23), growth hormone insufficiency (18q22.3-q23), and congenital aural atresia (18q22.3). The overall level of mental retardation appears to be mild in patients with deletions distal to 18q21.33 and severe in patients with deletions proximal to 18q21.31. The critical region for the typical 18q- phenotype is a 4.3 Mb region within 18q22.3-q23. The maximum estimated size of the deletion ranges from 7.7 to 29.4 Mb. ❑
CASE REPORT
The patient is a 3 years 6 months old boy, admitted in our department for psychomotor retardation. He is the 3rd child of healthy, non-consanguineous parents (following 2 healthy elder daughters), born after an uneventful pregnancy and delivery, with a birth weight of 3500 gr. and an Apgar score of 9. From the first year of life, muscular hypotonia was observed; subsequently, a global developmental delay was noted: the boy had head control at 9 months, sat at 2 years, and has not achieved yet the ability to walk or to speak. Because he did not react at noises, at the age of 6 months he underwent auditory evoked potentials, and a bilateral deafness was diagnosed. Since the age of one year, the child presented frequent infections and long febrile episodes, for which he was evaluated in the Department of Pediatrics; a low level of immunoglobulin A was discovered. At the age of 2, because of poor weight and height gain, the boy was evaluated in the Department of Endocrinology, and a growth hormone (GH) deficiency was discovered.
A cerebral MRI was done, showing a delayed myelination and enlarged ventricles.
At the admittance in our department, the clinical examination of the child showed: weight of 10 kg (less than 2 SD), height of 85 cm (less than 2 SD), occipito-frontal perimeter of 48 cm (25 percentile); dysmorphic features: large forehead; flat midface; micrognathia; malformed earlobes; hypertelorism; flat nasal bridge; thin lips; short neck; clinodactyly of the 5th finger; planovalgus. Neurological examination showed severe mental retardation (a mental age of 6 months); motor delay (inability to walk or crawl); axial and limb hypotonia; slight dysmetria; bilateral deafness; lack of expressive and receptive language. Table 1 summarizes which of the typical features of the 18q– syndrome were found in this child. All the above characteristics suggested an 18q- syndrome.
Table 1.
Clinical features associated with 18q deletion and their occurrence in our patient
| Feature | Occurrence |
|---|---|
| Decreased growth | + |
| Decreased head circumference | - |
| Face | |
| Midface hypoplasia | + |
| Up/downward slanting | - |
| palpebral fissures | + |
| Dysplastic ears | - |
| Flat philtrum | - |
| Downturned corners of mouth | - |
| Prognathism | - |
| High or cleft lip/palate | + |
| Flat nasal bridge | |
| Limbs | |
| Tapered fingers | - |
| Proximal thumbs | - |
| Increased whorls on fingers | + |
| Abnormal toes | - |
| Club feet | + |
| Genitourinary | |
| Hypoplasia of labia/scrotum | - |
| Central nervous system | |
| Hypotonia | + |
| Seizures | - |
| Hearing loss | + |
| Strabismus | - |
| Poor coordination | + |
| Nystagmus | - |
| Developmental delay | + |
| Mental retardation | + |
| Delayed myelination | + |
| Enlarged ventricles | + |
| Respiratory | |
| Recurrent respiratory infections | + |
| Endocrine features | |
| Growth hormone deficiency | + |
| Endocrine features | |
| Low levels of immunoglobulin A | + |
+ present, -absent
In order to confirm the 18q- syndrome by cytogenetic investigations, phytohemagglutinin-stimulated lymphocyte cultures were initiated from peripheral blood, for the patient, his sisters and parents.
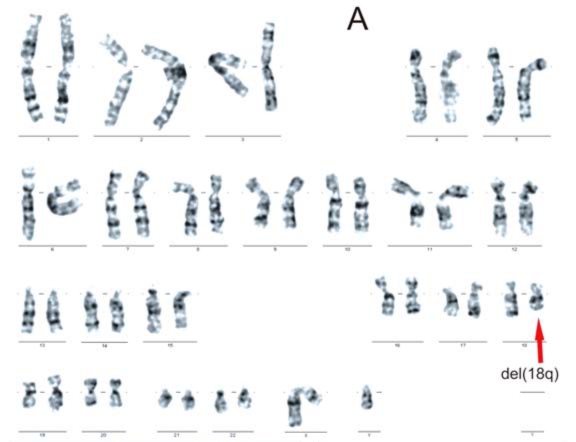
Classical cytogenetic analysis of the karyotype revealed a deletion in the distal fragment of chromosome 18q, ranging from band q21 to qter: del(18)(q21) (FIGURE 1A). The cytogenetic investigations of the parents and sisters showed no abnormalities.
Figure 1. Cytogenetic results for the patient.
A.
A. GTG-banded karyotype illustrating del(18q) [red arrow];
B.

B. Painting FISH for chromosome 18 showing no cryptic arrangements involving other chromosomes;
C.

C. FISH with BAC probes 18q23 (red signal) and 18p11.32 (green signals) showing the terminal deletion of 18q.
Fluorescence In Situ Hybridization (FISH) was carried out with bacterial artificial chromosomes (BAC) clones, including: whole chromosome 18-specific paint BAC probe (CP18-Alu); human locus-specific probes for chromosome 18: [RP11-14P20 for 18p11.32, RP11-248 M19 for 18q23]; counterstaining was done with DAPI. Whole chromosome painting was performed to confirm that the deleted fragment of 18q was not translocated elsewhere in the genome – as it was indeed the case (FIGURE 1B). Therefore, any cryptic rearrangements were ruled out. The distal extension of 18q deletion was confirmed using BAC-FISH probe RP11-248M19, with a physical location on band 18q23 (75660081-75829893) (FIGURE 1C). ❑
EVOLUTION
After the identification of the 18q deletion syndrome, the child received Somatropine 0.45 mg per day. After 3 weeks, he presented high fever (40 Celsius degrees), upper eyelid swelling, and psychomotor regression (inability to sit, weakness, hyporeactivity). One week later, because of the persistence of fever (despite the treatment with antibiotics and antipyretics) and psychomotor regression, the parents required the GH treatment to be discontinued. The child started an intensive physical therapy and cognitive stimulation, and after one month he begun to recover. Currently, he is in good health, without fever, and with the same neurological aspect as at the first evaluation. ❑
DISCUSSIONS
The 18q deletion syndrome is a well-described disorder resulting from a partial deletion of the long arm of chromosome 18. The breakpoints vary greatly between different reports. The clinical features of this syndrome include short stature, hypotonia, hearing impairment, dysmorphic features, foot deformities, low levels of immunoglobulin A and growth hormone deficiency. The incidence of these clinical symptoms in different patients with 18q syndrome is variable.
The patient reported here has a severe form of the 18q syndrome, presenting almost all characteristic features of this condition. This severe clinical picture is explained by the cytogenetic findings, namely a deletion proximal to 18q21.
Some studies showed that GH therapy in individuals with 18 q deletions can increase linear growth, improve nonverbal IQ and cause a change in the T1 relaxation time in specific areas of the brain (5). In our case, not only that GH therapy did not bring any improvement, but patient's health declined. To our knowledge, our patient reaction was not reported before and we were unable to find any explanation. Intensive physical therapy and cognitive stimulation of the child were the only measures which proved to be efficient in the recovery of the patient. ❑
CONCLUSION
The 18q deletion syndrome is a very heterogeneous condition with a highly variable phenotype, generally characterized by mental retardation, short stature, hypotonia, hearing impairment, dysmorphic features, low levels of immunoglobulin A, and growth hormone deficiency. Although GH therapy in individuals with 18 q deletions was used with good results, in our case this therapy failed to bring any improvement and even a psychomotor decline was noted. Motor and cognitive stimulation was very efficient in our patient. ❑
ACKNOLEDGENENTS
The authors thank Prof. Jean-Michel Dupont and Mrs. Dominique Blancho (Laboratoire de Cytogénétique, Groupe Hospitalier COCHIN, Hotel Dieu, Paris, France) for kindly providing BAC-FISH probes. This paper was financially supported by CNCSIS Project No. 1203
References
- 1.Andler W, Heuveldop A, Polichronidou T. Endokrinologische storungen bei Deletionen des chromosomes 18. Mschr Kinderheilk. 1992;140:303–306. [PubMed] [Google Scholar]
- 2.Fryns JP, Logghe N, van Eygen M, et al. 18q- syndrome in mother and daughter. Europ J Pediat. 1979;130:189–192. doi: 10.1007/BF00455265. [DOI] [PubMed] [Google Scholar]
- 3.Chen CP, Lin SP, Chern SR, et al. Direct transmission of the 18q- syndrome from mother to daughter. Genet Counsel. 2006;17:185–189. [PubMed] [Google Scholar]
- 4.Feenstra I, Vissers LELM, Orsel M, et al. Genotype-phenotype mapping of chromosome 18q deletions by high-resolution array CGH: an update of the phenotypic map. Am J Med Genet. 2007;143:1858–1867. doi: 10.1002/ajmg.a.31850. [DOI] [PubMed] [Google Scholar]
- 5.Cody JD, Hale DE, Brkanac Z, et al. Growth hormone insufficiency associated with haploinsufficiency at 18q23. Am J Med Genet. 1997;71:420–425. [PubMed] [Google Scholar]