Abstract
The use of progestins as a component of hormone replacement therapy has been linked to an increase in breast cancer risk in postmenopausal women. We have previously shown that medroxyprogesterone acetate (MPA), a commonly administered synthetic progestin, increases production of the potent angiogenic factor vascular endothelial growth factor (VEGF) by tumor cells, leading to the development of new blood vessels and tumor growth. We sought to identify nontoxic chemicals that would inhibit progestin-induced tumorigenesis. We used a recently developed progestin-dependent mammary cancer model in which tumors are induced in Sprague-Dawley rats by 7,12-dimethylbenz(a)anthracene (DMBA) treatment. The flavonoid apigenin, which we previously found to inhibit progestin-dependent VEGF synthesis in human breast cancer cells in vitro, significantly delayed the development of, and decreased the incidence and multiplicity of, MPA-accelerated DMBA-induced mammary tumors in this animal model. Whereas apigenin decreased the occurrence of such tumors, it did not block MPA-induced intraductal and lobular epithelial cell hyperplasia in the mammary tissue. Apigenin blocked MPA-dependent increases in VEGF, and suppressed VEGF receptor-2 (VEGFR-2) but not VEGFR-1 in regions of hyperplasia. No differences were observed in estrogen or progesterone receptor levels, or the number of estrogen receptor-positive cells, within the mammary gland of MPA-treated animals administered apigenin, MPA-treated animals, and placebo treated animals. However, the number of progesterone receptor-positive cells was reduced in animals treated with MPA or MPA and apigenin compared with those treated with placebo. These findings suggest that apigenin has important chemopreventive properties for those breast cancers that develop in response to progestins.
Keywords: Apigenin, breast cancer, medroxyprogesterone acetate, chemoprevention
Introduction
Recent clinical trials and studies demonstrate an increased risk of breast cancer in postmenopausal women undergoing hormone replacement therapy (HRT) that consists of both estrogen and progestin (E+P) compared with women taking estrogen alone or placebo (1–3). Furthermore, women on a regimen of E+P are more at risk of metastasis and death than those taking estrogen alone (4). Progestins are prescribed in HRT regimens to counter the unopposed proliferative effects of estrogen on the uterus that could lead to uterine cancer; however, overwhelming evidence shows that progestins stimulate proliferation of normal and neoplastic breast cells in various species, including humans (5–8).
A number of mechanisms have been implicated in the progestin-induced increase in breast cancer risk observed in women undergoing HRT, including induction of angiogenic growth factors such as vascular endothelial growth factor (VEGF) (9, 10), which renders the microenvironment conducive to tumor development and progression. VEGF is considered one of the most important growth factors and its effects have been reported to be vital to the angiogenesis that is integral to the growth and metastasis of tumors (11), including those that arise in response to progestins (7). Recently, other mechanisms have also been proposed to explain progestin-dependent breast tumor development and progression. For example, in animal models, RANK-L has also been associated with progestin-dependent increased proliferation of breast cancer cells (12, 13), while it has also been reported that medroxyprogesterone acetate (MPA), a progestin commonly used in HRT, increased the stem-like cell subpopulation in mammary glands (14). Collectively, these observations suggest that progestins promote the growth of latent breast tumor cells by increasing their proliferative potential and/or promote tumor development through activation and transformation of dormant breast cancer stem cells into intermediate subpopulations, which then differentiate into breast cancer cells.
We have previously shown that progestins accelerate the development of 7,12-dimethylbenz(a)anthracene (DMBA)-induced mammary tumors in Sprague-Dawley rats (15–17). Whereas the molecular basis of this phenomenon has not been fully established, it is clear that progestins accelerate the tumor development process by increasing VEGF and subsequent angiogenesis in this model (15–17). Thus, it appears that once tumor initiation has occurred, progestins have the capacity to increase tumor cell proliferation, producing palpable tumors sooner. The progestin-accelerated DMBA-induced mammary tumor model is therefore a useful model in which to identify natural “neutraceuticals” that can prevent the development of progestin-accelerated breast cancers. Because HRT is used widely to alleviate the severe symptoms of menopause, the inclusion of neutraceuticals in a drug regimen could well prove beneficial, as these compounds would selectively limit the proliferative effects of progestins in the breast, protecting against the development of frank tumors.
Previous studies have shown that apigenin inhibits the growth of several human cancers (18). Apigenin is a low molecular weight flavonoid commonly found in fruits, vegetables, nuts, and plant-derived beverages (19). Apigenin exhibits low intrinsic toxicity and appears to have little effect on normal cells (20, 21), making it an attractive candidate for chemoprevention. While apigenin has been shown to be effective against several types of tumors (18), its ability to act as an antiprogestin as a possible mechanism by which it exerts its antitumor action has not been well-studied. However, we recently demonstrated that apigenin inhibits progestin-dependent VEGF induction in human breast cancer cells in vitro (22). These findings provided a strong rationale for us to extend our studies to an in vivo model and examine apigenin’s ability to inhibit progestin-accelerated DMBA-induced mammary tumors. Herein we provide evidence that apigenin significantly delays the appearance of and decreases the incidence and multiplicity of MPA-accelerated, DMBA-induced mammary tumors, though it is unable to prevent the formation of MPA-induced epithelial cell hyperplasia in the mammary gland. We also show that at least one of the mechanisms by which apigenin exerts its preventive effects is through suppression of progestin-induced VEGF and VEGFR-2 expression, both of which are vital components of the angiogenic pathway (11, 23).
Materials and Methods
In Vivo Studies
Intact 40- to 45-day-old female Sprague-Dawley rats (Harlan Breeders, Indianapolis, IN) were used in this study. The rats were maintained according to the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International under conditions of 12-hour light/dark cycles and ad libitum access to food and water. All surgical and experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Missouri (Columbia, MO) and were in accordance with procedures outlined in the “Guide for Care and Use of Laboratory Animals” (NIH publication 85-23). Each experiment was performed at least twice.
Apigenin
Apigenin (Indofine Chemical Company, Hillsborough, NJ) was dissolved in a dimethyl sulfoxide (DMSO)/PBS mixture (1:4) to produce the required concentration. Fresh apigenin was prepared every two days, with the remaining apigenin solution stored at −20°C for use the following day.
Experimental Design
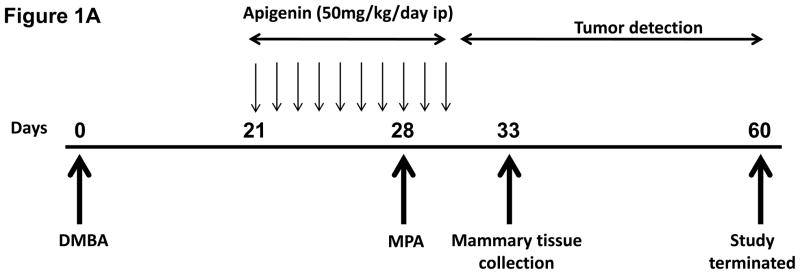
We followed a protocol previously established by our laboratory for progestin-acceleration of DMBA-induced mammary tumors in rats (15–17; Fig 1A). Briefly, 45- to 50-day-old Sprague-Dawley rats were given a single dose of 20 mg DMBA (Sigma, St. Louis, MO) in vegetable oil by gavage (on day 0). On day 28 post-DMBA administration, animals were anesthetized and implanted with a single 25 mg-60 day release MPA or placebo pellet (Innovative Research, Sarasota, FL) subcutaneously on the dorsal part of the neck. The dose of apigenin used initially (50 mg/kg) was based on a previous report (24), and was administered daily via intraperitoneal injection, beginning 21 days after the initial dose of DMBA and continuing for 10–13 days. Control animals from the MPA implanted group and those implanted with placebo pellets only were treated with DMSO/PBS vehicle on the same schedule. Following pellet implantation, animals were palpated every 2 days to detect tumors. The time preceding the appearance of the first tumor (latency) and tumor size, number, and location were recorded. Tumor length (L) and width (W) were measured with a micrometer caliper, and tumor size was calculated using the formula L/2 X W/2 X π (15). All animals were sacrificed and the experiment terminated on day 60 and the abdominal mammary tissue lacking tumors were collected.
Figure 1. Apigenin prevents progression of DMBA-induced progestin accelerated tumors in rats.
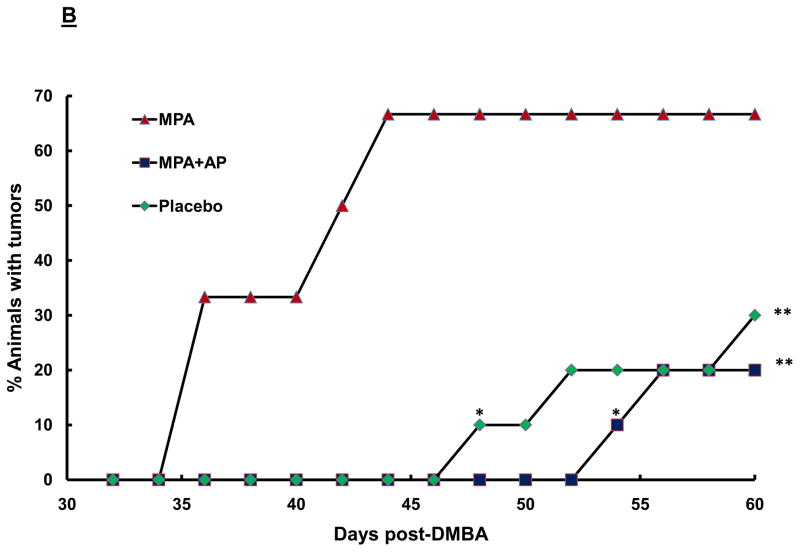
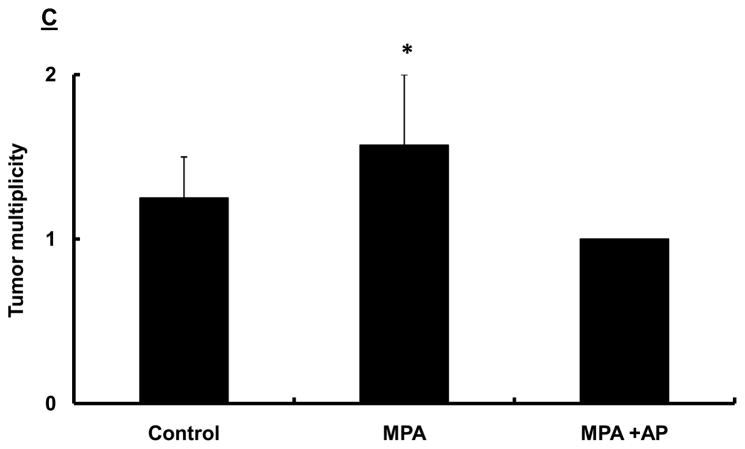
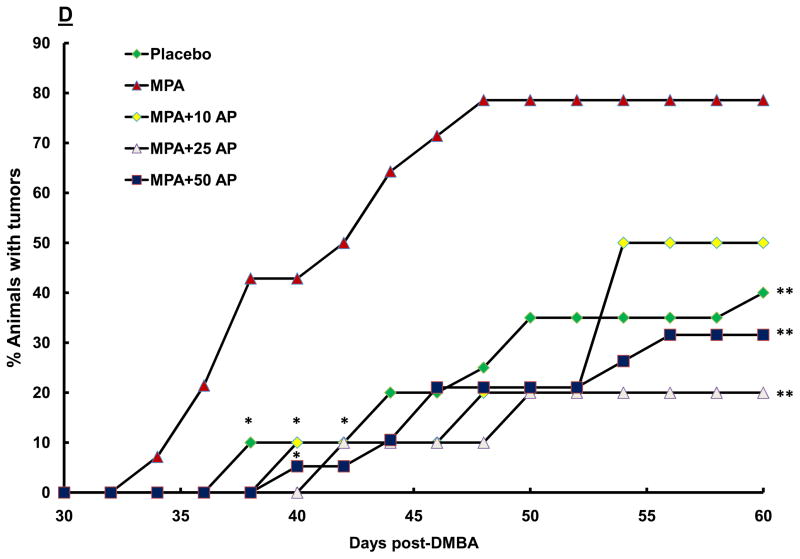
(A) Treatment Protocol. Sprague-Dawley rats were treated with DMBA and subsequently implanted with a 25 mg/60-day release MPA (or placebo) pellet on day 28 as described in the Materials and Methods. Apigenin (AP; 50 mg/kg) or vehicle injections were given once daily between days 21 and 31 following DMBA treatment. Following pellet implantation, animals were palpated for tumors every other day through day 60. (B) Preventive effect of apigenin on MPA-accelerated DMBA-induced mammary tumors. The percent of animals with tumors in each experimental group at each time point is presented (n=6–10 animals/group). Tumor latency was assessed using LIFETEST procedure in SAS as described in the Materials and Methods; * indicates p<0.05 compared with tumor latency in the MPA-treated group. Tumor incidence was determined using the General Linear Mixed Model procedure in SAS as described in the Materials and Methods; ** indicates P<0.05 compared with tumor incidence at day 60 in the MPA-treated group. (C) Effect of apigenin on tumor multiplicity. The mean number of tumors per tumor-bearing animal at the conclusion of the study in B (60 days after DMBA treatment) is presented. * indicates p<0.05 compared with the other two groups; error bars represent SEM. (D) Dose-response effects of apigenin on MPA-accelerated DMBA-induced mammary tumors. Experimental details are as described above except that varying amounts of apigenin were used as indicated in the figure. 10AP, 10 mg/kg dose of AP; 25AP, 25 mg/kg dose of AP, and 50AP, 50 mg/kg dose of AP. * indicates p<0.05 compared with MPA-treated group (latency) and ** indicates p<0.05 compared with MPA-treated group at day 60 (incidence). Combined results from two different experiments are shown (n=10–20 animals/group).
In order to determine potential mechanisms by which apigenin could exert anticancer effects against MPA-accelerated DMBA tumors, we hypothesized that differences in morphology and protein expression in mammary tissue between apigenin- and nonapigenin-treated animals would best be distinguished if tissues were collected immediately after the last apigenin injection. Therefore, during the course of our experiment as described above, we collected abdominal mammary tissues, devoid of tumors, from randomly selected animals on day 33, which was 5 days after implantation of MPA (or placebo) pellets and 6 hours after the last apigenin injection. We selected 5 days after implantation of MPA pellets as the tissue collection time point because we normally first detect tumors at around this time in this model, and therefore expected sufficient hyperplasia to be present within the mammary glands at this point. To further confirm the effects of MPA and apigenin, we monitored the remaining animals for tumor development as described above until the study was terminated 60 days after DMBA administration.
Histology and Immunohistochemical Analysis
Immunohistochemical staining of mammary tissue was carried out according to a previously described method (16, 17). The following polyclonal antibodies were used: anti-progesterone receptor (PR) antibody (1:50 dilution [A0098], DAKO, Carpinteria, CA), anti-estrogen receptor (ERα) (1:300 dilution [sc-542], Santa Cruz Biotechnology, Inc., Santa Cruz, CA), anti-ERβ (1:25 dilution [MCA1974S], AbD Serotec), anti-VEGF antibody (1:100 dilution [sc-152], Santa Cruz Biotechnology, Inc.), anti-VEGFR-1 antibody (1:50 dilution [Flt-1, H225, sc-9029], Santa Cruz Biotechnology, Inc.), anti-VEGFR-2 antibody (1:50 dilution [Flk-1, ab2349], Abcam, Inc., Cambridge, MA). Histologic staining was then quantified using morphometric software (Fovea Pro 3.0, Reindeer Graphics, Asheville, NC). Images were recorded at 20X magnification, and threshold image intensity was adjusted for measurement in pixels. Two sections were prepared for each animal (right and left abdominal mammary glands), and 4 different images per section were obtained to minimize errors due to differences in cellularity, for a total of 8 images per animal. Section images were collected from at least 3 animals in each treatment group. Staining area in regions of epithelial hyperplasia was recorded and the means determined. For further quantitation of PR and ER, PR-, ERβ-, and ERα– positive cells were counted and results expressed as a percentage of total cells. At least 150–200 cells were counted per section prepared as described above.
Statistical Analysis
Time-to-event data (latency) was analyzed using the LIFETEST procedure in SAS. We estimated survival functions for each group using the Kaplan-Meier method and made comparisons across groups using the Wilcoxon log-rank test. Those animals that had not developed a tumor by the end of the study were censored. Tumor incidence among groups was compared using a General Linear Mixed Model procedure in SAS (PROC GLIMMIX), where the link function was a logit and the distribution binomial and differences were determined using the LS means statement. Multiplicity and immunohistochemical staining data were analyzed using Kruskal-Wallis one-way analysis of variance (ANOVA) followed by Tukey’s procedure as a posthoc test. For all comparisons, p<0.05 was regarded as statistically significant. Values are reported as mean ± standard error of the mean (SEM).
Results
Apigenin inhibits MPA-accelerated DMBA-induced mammary tumors
Using our well-established rodent model in which MPA reduces the latency of DMBA-induced mammary tumors (15) we examined the ability of apigenin to prevent the MPA-induced acceleration of tumor development. In brief, 21 days after a single dose of DMBA was given, apigenin was administered daily at a dose of 50 mg/kg body weight as described previously (24) for one week. A single pellet containing MPA was then implanted, and treatment with apigenin was continued for an additional 3 days, after which animals were palpated for tumors for 60 days following treatment with DMBA. In those animals given DMBA + placebo, mean tumor latency was 58 ± 1.6 days and tumor incidence ranged from 20–44%, compared with DMBA-treated animals that were also administered MPA, where mean tumor latency was reduced significantly (41.0 ± 1.7 days) and tumor incidence elevated to 67–88%. However, when DMBA-treated animals were administered both MPA and apigenin, tumor incidence was reduced to 20–44% and mean latency period increased considerably, to 55.8 ± 0.3 days (p<0.05), demonstrating that apigenin significantly delays the development of MPA-induced mammary tumors. Furthermore, by the time the first tumor was detected in the MPA + apigenin group (day 54), tumor incidence in the group given MPA alone had already reached a peak of 66.7% (Fig. 1B). The apigenin dose used in these experiments (50 mg/kg/day for 10 days i.p.) was deemed nontoxic, as the animals maintained their body weight throughout the study (data not shown).
In terms of its effect on tumor multiplicity, apigenin significantly reduced the number of tumors per tumor-bearing animal (Fig 1C). In the placebo group, multiplicity was 1.25 ± 0.3; this increased to 1.57 ± 0.4 in animals receiving MPA alone, and was 1.0 ± in animals administered both MPA and apigenin.
Prevention of MPA-accelerated tumors by apigenin is dose-dependent
To determine whether apigenin suppresses MPA-accelerated tumor development in a dose-dependent manner, we treated animals with 10, 25, and 50 mg/kg apigenin daily as described above. Each dose significantly delayed the first appearance of progestin-accelerated tumors compared with animals treated with MPA alone (Fig. 1D). However, at the lowest dose of 10 mg/kg apigenin, tumor incidence at termination of the study was not statistically different from that seen in animals treated with only MPA, indicating that at a lower dose apigenin was less effective at preventing tumor formation.
Apigenin does not prevent MPA-induced morphological changes in rodent mammary gland
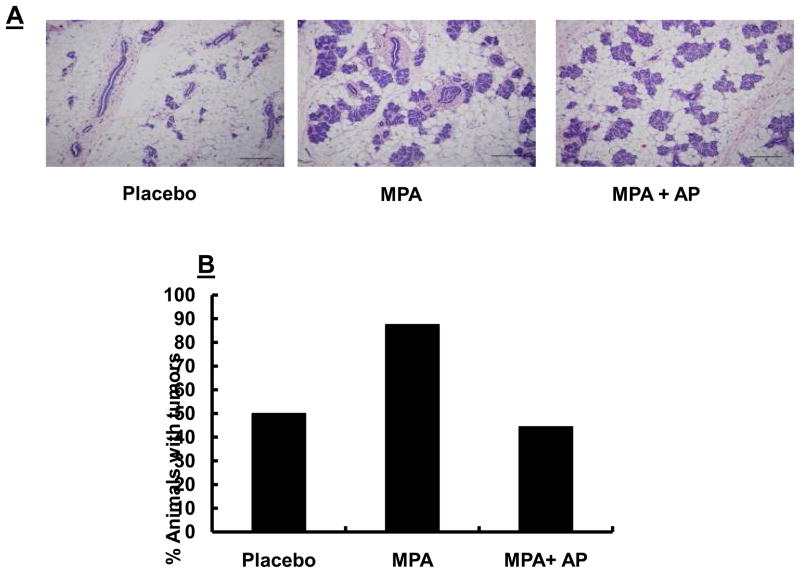
Previous studies have shown that mammary tissue from animals exposed to progestins exhibited extensive proliferation of the mammary epithelium, resulting in formation of hyperplastic alveolar nodules and intraductal hyperplasia (15–17). In order to determine whether apigenin prevents MPA-induced epithelial hyperplasia, we collected abdominal mammary tissue 5 days after implantation of MPA pellets and 6 hours after the last apigenin injection from DMBA-treated animals as described in the Materials and Methods. A second group of treated animals from the same experiment was maintained up to day 60 to ensure that the effects of MPA and apigenin were observed. Our observations concurred with previous reports in that extensive hyperplasia occurred within the mammary glands of animals treated with MPA for 3–5 days (15–17, 25), compared with hyperplasia observed at the same tine in animals given placebo pellet alone. Apigenin failed to block MPA-induced hyperplasia within the mammary gland (Fig. 2A). However, even though apigenin was unable to prevent hyperplasia in MPA-treated animals, by the end of the experiment it had reduced overall tumor incidence (Fig. 2B). This suggests that apigenin arrests the development of MPA-induced tumors at a stage beyond the formation of hyperplastic alveolar nodules and intraductal hyperplasia.
Figure 2. Apigenin does not block MPA-induced morphological changes in mammary gland tissues.
Sprague-Dawley rats were treated with DMBA and subsequently implanted with a 25 mg/60-day release MPA pellet on day 28 as described in the Materials and Methods. Apigenin (AP; 50 mg/kg) or vehicle injections were given once daily between days 21 and 33 following DMBA treatment. (A) Abdominal mammary tissues were collected from randomly selected animals sacrificed on day 33, 6 hours after the last apigenin injection and processed as described in the Materials and Methods. One representative H&E stained section is shown for each experimental group. Images were taken at 20X; scale bar, 100 μm. (B) Tumor incidence at termination of study (day 60; n=8–10 animals/group) to ensure expected MPA effects were obtained in the group of animals used for tissue dissection in (A).
Immunohistochemical analysis
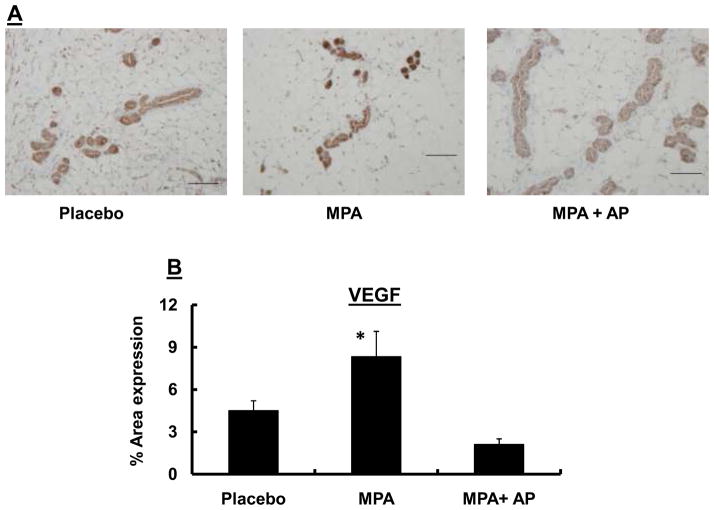
In previous studies, we showed that MPA-induced elevation in tumor cell VEGF was an important mechanism by which the progestin accelerated development of mammary tumors (15–17). In the present study, we sought to determine whether the antitumor effects of apigenin might arise as a consequence of its ability to suppress VEGF production within areas of MPA-induced epithelial hyperplasia in the mammary gland. We examined VEGF staining in sections of mammary tissue collected from animals treated with placebo, MPA or MPA + apigenin. We found that VEGF staining in regions of hyperplasia of mammary tissue from animals in the MPA + apigenin group (2.1 ± 0.5; staining/area of hyperplasia) was significantly lower than that observed in animals receiving MPA alone (8.3 ± 1.9) or in control animals receiving placebo (4.5 ± 0.7; p<0.05; Fig. 3A & B). These findings led us to propose that the ability of apigenin to prevent MPA-accelerated DMBA-induced tumor development is at least in part due to its effect on VEGF expression.
Figure 3. Apigenin suppresses expression of VEGF in DMBA-induced MPA-treated mammary glands.
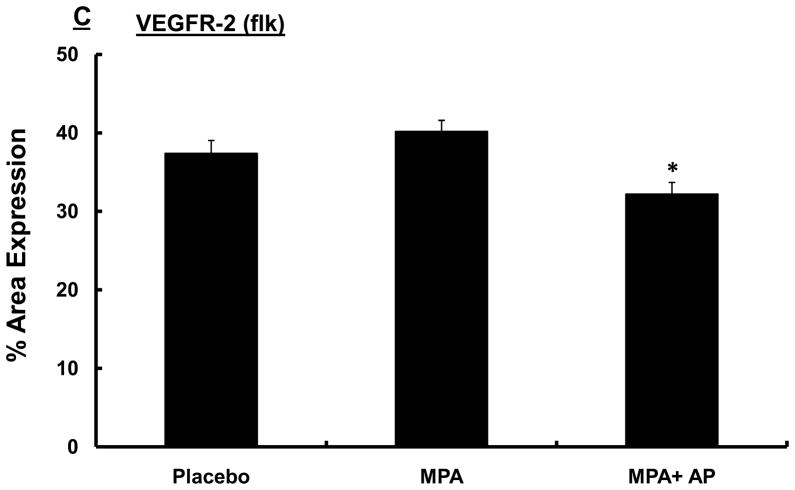
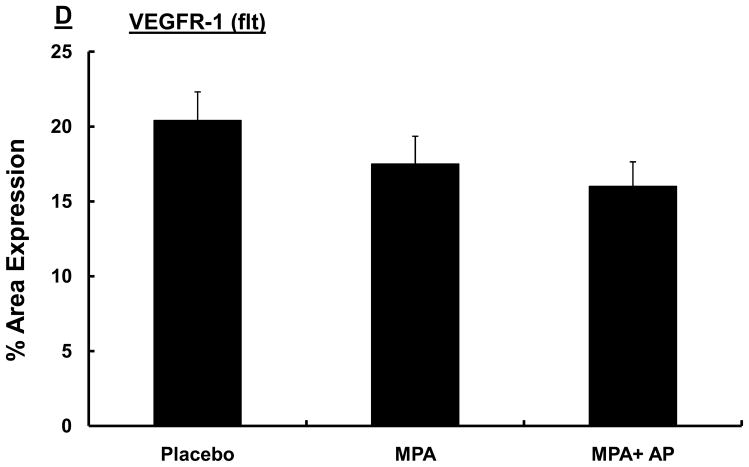
Sprague-Dawley rats were treated with DMBA and subsequently implanted with a 25 mg/60-day release MPA pellet on day 28 as described in the Materials and Methods. Apigenin (AP; 50 mg/kg) or vehicle injections were given once daily between days 21 and 33 following DMBA treatment. (A) Mammary glands were collected from randomly selected animals sacrificed on day 33, 6 hours after the last apigenin injection and processed and stained for VEGF as described in the Materials and Methods. One representative section is shown for each group. Images were taken at 20X; scale bar, 100 μm. (B–D) Quantitative analysis of VEGF (B), VEGFR-2 (flk) (C), and VEGFR-1 (flt) (D) expression in mammary tissues described in A using Fovea Pro software as described in the Materials and Methods. Data represent the mean percent area positive for staining; error bars represent SEM.* indicates p<0.05 compared with the rest of the groups by Kruskal-Wallis one-way ANOVA on ranks and all pair-wise multiple comparisons by Tukey’s procedure.
VEGF signaling is mediated largely by the VEGF receptors, VEGFR-1 (flt) and VEGFR-2 (flk). Consequently, we conducted studies to determine whether apigenin influences VEGFR-1 and VEGFR-2, which are essential mediators of the proliferative and survival effects of VEGF (26). In mammary tissue collected from animals in the experimental groups described above we found that VEGFR-2 (flk) expression in areas of hyperplasia was significantly reduced in the MPA + apigenin group (32.2 ± 1.5) compared with animals receiving MPA alone (40.2 ± 1.4) or placebo (37.4 ± 1.6; p<0.05; Fig. 3C). In contrast, apigenin had no effect on levels of VEGFR-1 expression within epithelial hyperplastic areas of mammary glands exposed to MPA, and no significant difference was seen among VEGFR-1 expression levels in MPA (17.5 ± 1.9), MPA + apigenin (16 + 1.7), and placebo-treated animals (20.4 ± 1.9; Fig. 3D).
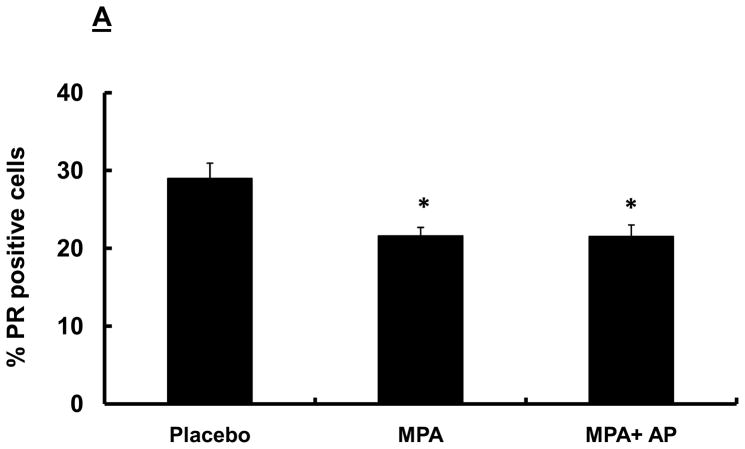
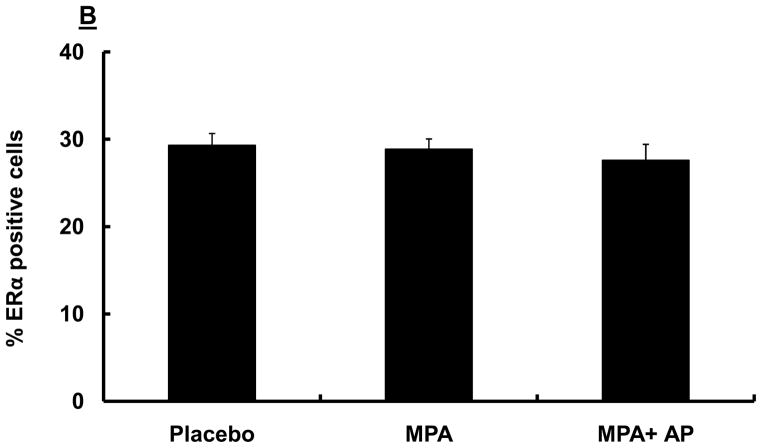
Previous studies have shown that signaling through ERs is critical for PR expression, and that PR activity is essential for VEGF induction (27, 28). We therefore carried out studies aimed at determining whether apigenin blocks MPA-dependent effects by suppressing expression of ER and/or PR. We found that, while levels of PR expression in mammary tissue were similar among all three treatment groups (placebo, MPA and MPA + apigenin; data not shown), the percentage of cells staining positive for PR was significantly lower in animals in the MPA (22 ± 1) and MPA+ apigenin (22 ± 2) groups compared with that in animals in the placebo group (29 ± 2; Fig. 4A). We attribute the decline in PR staining in proportion of total cells to the known effects of MPA in lowering the levels of PR, as previously reported (17, 29), and suggest that this is further evidence that apigenin does not block all MPA-dependent effects. Similarly, we found that expression of both ERα (Fig 4B) and ERβ (data not shown) was similar in the mammary tissues of MPA-treated animals, whether or not animals were administered apigenin. The proportion of cells in mammary tissue that stained positive for ERα was similar for animals in the MPA and MPA + apigenin groups, and was not significantly different from that observed in the placebo group (Fig. 4B), while almost all epithelial cells were ERβ positive in all the groups mentioned above (data not shown).
Figure 4. Immunohistochemical analysis of ER and PR in the mammary glands of DMBA-induced MPA accelerated tumors in rats with and without apigenin treatment.
Sprague-Dawley rats were treated with DMBA and subsequently implanted with a 25 mg/60-day release MPA pellet on day 28 as described in the Materials and Methods. Apigenin (AP; 50 mg/kg) or vehicle injections were given once daily between days 21 and 33 following DMBA treatment. Mammary glands were collected from randomly selected animals sacrificed on day 33, 6 hours after the last apigenin injection and processed and stained for PR and ER as described in the Materials and Methods. Images were taken at 40X, and at least 150 cells counted per section as described in the Materials and Methods. Data represent mean percentages of PR- (A) and ERα–positive (B) stained cells in DMBA-induced MPA-treated mammary glands of animals treated with or without apigenin; error bars represent SEM. * indicates p<0.05 compared with placebo group by Kruskal-Wallis one-way ANOVA on ranks and all pair-wise multiple comparisons by Tukey’s procedure.
Discussion
In this study, we provide evidence that apigenin significantly delays the appearance of progestin-accelerated breast tumors in a DMBA-induced tumor model, reduces tumor multiplicity, and reduces tumor incidence. While several mechanisms have been proposed for the anticancer effects of apigenin, to our knowledge this is the first report indicating that this naturally occurring flavonoid might inhibit in vivo tumor development by opposing the effects of progestins. Using cell culture, we previously showed that apigenin inhibits progestin-dependent induction of VEGF in human breast cancer cells (22). With this in mind, we conducted a series of in vivo studies aimed at determining whether apigenin might be used to prevent the emergence of mammary tumors, especially in women exposed to synthetic progestins as a component of E+P HRT. Our studies were focused on examining the effects of apigenin on MPA-accelerated mammary tumors in an animal model, because MPA is the most widely prescribed progestin and its use is associated with increased risk of breast cancer and mortality (1–4).
Following the administration of apigenin to DMBA-treated animals that were also given MPA, tumor latency was significantly increased. Furthermore, tumor incidence decreased at least 50% in these animals, compared with animals that were treated with vehicle alone. Importantly, apigenin also reduced tumor multiplicity to levels below those normally observed following progestin treatment. It was interesting to note that, even though apigenin suppressed tumor development, it did not prevent the MPA-induced morphological changes and hyperplasia in the mammary gland of DMBA-treated animals, a finding in direct contrast to the effects of curcumin (16), a natural plant derived curcuminoid with anti-cancer properties (30). Surprisingly though, with respect to its ability to increase tumor latency and reduce the incidence of MPA-accelerated tumors in this model, apigenin was a more effective preventive compound than curcumin.
Apigenin has been reported to exert its anticancer effects via a variety of mechanisms, including induction of cell cycle arrest and apoptosis, through both tumor necrosis factor- α induced NFκB-mediated and intrinsic apoptosis pathways (31) and attenuation of the phosphorylation of epidermal growth factor receptor and MAP kinase (18, 32). Compelling evidence from a variety of model systems suggests that VEGF is essential for tumor development and progression (11, 33), and it has been postulated that, unless tumors develop blood vessels, they will not grow beyond 2 mm3 (34). Our earlier in vitro studies using human breast cancer cell lines (22) have demonstrated that apigenin significantly reduces the expression of both VEGF and its receptor (VEGFR-2), an essential mediator of VEGF-dependent tumor cell proliferation (33). It is therefore likely that, by suppressing VEGF expression, apigenin renders the mammary tissue microenvironment less conducive to tumor development.
Accumulating evidence has shown that many types of cancer, including breast cancer, are initiated by a small population of cancer stem cells (35). Due to the rapid rate at which breast tumors develop in postmenopausal women taking E+P HRT, we and others have suggested that progestins most likely increase the proliferation of existing tumor cells in the breast (9, 23, 36). Recently, Horwitz and Sartorius (14) suggested that progestins may promote tumor development through activation and transformation of dormant breast cancer stem cells into intermediate subpopulations, which then differentiate into breast cancer cells. Because we found that apigenin did not prevent MPA-induced hyperplasia, but did inhibit the emergence of tumors and reduce the incidence and multiplicity of tumors normally associated with MPA treatment, apigenin may target a subset of cells essential for tumor development (cancer stem cells) in this model system. Recent studies showing that sulphorane, a component of broccoli, prevents tumor development by targeting and killing critical cancer stem cells (35) support this idea. Furthermore, a number of dietary compounds such as curcumin (30), quercetin, and epigallocatechin-gallate (37) have long been recognized as agents capable of suppressing cancer stem cell proliferation. Further studies are necessary to confirm whether apigenin might prevent the development of progestin-accelerated DMBA-induced mammary tumors via a mechanism that targets breast cancer stem cells.
Breinholt et al. (38) reported that apigenin reduced levels of endogenous ER in mouse uterus. With this in mind, we sought to determine whether apigenin might have a similar effect in mammary gland. ER plays a vital role in controlling mammary levels of PR (27), a fact which could explain the lack of MPA effects, because the presence of adequate levels of PR is essential if MPA is to exert effects such as induction of VEGF (9, 33). However, we did not detect any differences in the expression levels of either ER or PR in the mammary gland of MPA-, MPA +apigenin-, or vehicle-treated animals. Apigenin has been shown to function in both an ERα-dependent and independent manner (39) and thus it is possible that in our study apigenin mediates its effect in an ERα independent manner, although this remains to be tested. Interestingly, we observed that while the percentage of cells expressing ERα was similar in all treatment groups, the percentage of PR-positive cells was significantly lower in MPA-treated animals (both those treated with and without apigenin) compared with the placebo group. However, insufficient PR expression is unlikely to explain the antiprogestin effects of apigenin, and we cannot rule out the possibility that it blocks PR-mediated functions by modifying its activity, either through suppressing phosphorylation of the PR protein or through an unidentified mechanism. These scenarios remain to be examined.
Although data on the bioavailability of dietary apigenin in humans is extremely limited, Meyer et al. (40) reported that, following ingestion of parsley, which is rich in apigenin, plasma levels of the flavonoid increased to a concentration of 0.34 μmol/l. Consequently, it appears that concentrations of apigenin sufficient to be biologically effective can be obtained orally through the diet. Furthermore, it has been reported that apigenin’s slow pharmacokinetics allow it to remain in the circulation for a prolonged period (24), suggesting that it may accumulate within tissues at sufficient levels to exert chemopreventive effects. Consumption of apigenin-rich foods may be chemopreventive, particularly with respect to progestin-accelerated tumors. Furthermore, regular intake of foods rich in the flavonoid may decrease the cancer risk in postmenopausal women undergoing HRT with a progestin component, as well as in those who have already been exposed to combined HRT. Fortunately, apigenin appears to be nontoxic, since it appears to have different effects on normal versus cancerous cells (20, 21), suggesting that the intake of high doses will likely prove benign. Indeed, we administered apigenin repeatedly to animals at levels up to 50 mg/kg for 10–13 days and observed no signs of toxicity.
In this study, our main focus was to determine the effects of short-term apigenin administration (approx 10 days) on preventing the development of progestin-accelerated tumors in a model of DMBA-induced mammary carcinogenesis. In the future, we will perform long-term studies (30–60 days of apigenin administration) with a view to determining whether continuous treatment with apigenin can further prevent the appearance of tumors while remaining nontoxic to animals. Such studies will be valuable in helping to determine whether the reduction in tumor incidence (approximately 50%) seen following short-term treatment with apigenin (10 days) can be further improved upon in a long-term preventive strategy. The present study is also limited in that we have yet to determine the maximum tolerated dose that might be used to prevent the emergence of MPA-accelerated tumors in DMBA-treated animals. In the future, we will also look into whether administration of apigenin before the carcinogenic insult protects against the initial formation of DMBA-induced mammary tumors, as well as opposing the effects of MPA on tumor development. Additional preclinical and clinical studies are required to further investigate the efficacy, pharmacokinetics, and suitability of apigenin as a chemopreventive candidate for progestin-accelerated breast cancer and other hormone-dependent cancers in humans.
Acknowledgments
We thank Dr. M Burrow and L. Rhoades (University of Tulane, New Orleans) and Dr. Kenneth Nephew (Indiana School of Medicine) for consultation related to apigenin treatment. Our thanks to Ms Jill Gruenkemeyer and the histology laboratory at RADIL for help with the immunohistochemical studies. The experiments were supported by an NIH grant R56-CA-86916, COR grant from the College of Veterinary Medicine, University of Missouri, Columbia, and by research funds from RADIL, University of Missouri, Columbia.
References
- 1.Ross RK, Paganini-Hill A, Wan PC, Pike MC. Effect of hormone replacement therapy on breast cancer risk: estrogen versus estrogen plus progestin. J Natl Cancer Inst. 2000;92:328– 332. doi: 10.1093/jnci/92.4.328. [DOI] [PubMed] [Google Scholar]
- 2.Rossouew JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women‘s Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- 3.Chen WY, Hankinson SE, Schnitt SL, Rosner BA, Holmes MD, Colditz GA. Association of hormone replacement therapy to estrogen and progesterone receptor status in invasive breast carcinoma. Cancer. 2004;101:1490–1500. doi: 10.1002/cncr.20499. [DOI] [PubMed] [Google Scholar]
- 4.Chlebowski RT, Anderson GL, Gass M, et al. Writing group for Women’s Health Initiative Investigators. Estrogen plus progestin and breast cancer incidence and mortality in postmenopausal women. JAMA. 2010;304:1719–1720. doi: 10.1001/jama.2010.1500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Raafat AM, Hofseth LJ, Haklan SZ. Proliferative effects of combination estrogen and progesterone replacement therapy on the normal postmenopausal gland in a murine model. Am J Obstet Gynecol. 2001;183:1–14. doi: 10.1067/mob.2001.110447. [DOI] [PubMed] [Google Scholar]
- 6.Soderqvist G, Isaksson E, Von Schoultz B, Carlstrom K, Tani E, Skoog L. Proliferation of breast epithelial cells in healthy women during the menstrual cycle. Am J Obstet Gynecol. 1997;176:123–128. doi: 10.1016/s0002-9378(97)80024-5. [DOI] [PubMed] [Google Scholar]
- 7.Liang Y, Besch-Williford C, Brekken RA, Hyder SM. Progestin-dependent progression of human breast tumor xenografts: a novel model for evaluating antitumor therapeutics. Cancer Res. 2007;67:9929–9936. doi: 10.1158/0008-5472.CAN-07-1103. [DOI] [PubMed] [Google Scholar]
- 8.Liang Y, Benakanakere I, Besch-Williford C, Hyder RS, Ellersierck MR, Hyder SM. Synthetic progestins induce growth and metastasis of BT-474 human breast cancer xenografts in nude mice. Menopause. 2010;17:1040–1047. doi: 10.1097/gme.0b013e3181d3dd0c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hyder SM, Murphy L, Stancel GM. Progestin regulation of vascular endothelial growth factor in human breast cancer. Cancer Res. 1998;58:392–395. [PubMed] [Google Scholar]
- 10.Liang Y, Hyder SM. Proliferation of endothelial and tumor epithelial cells by progestin-induced VEGF from human breast cancer cells: paracrine and autocrine effects. Endocrinol. 2005;146:3632–3641. doi: 10.1210/en.2005-0103. [DOI] [PubMed] [Google Scholar]
- 11.Ferrara N. Role of vascular endothelial growth factor in physiologic and pathologic angiogenesis: therapeutic implications. Semin Oncol. 2002;29:10–14. doi: 10.1053/sonc.2002.37264. [DOI] [PubMed] [Google Scholar]
- 12.Gonzalez-Suarez E, Jacob AP, Jones J, et al. RANK ligand mediates progestin-induced mammary epithelial proliferation and carcinogenesis. Nature. 2010;468:103–107. doi: 10.1038/nature09495. [DOI] [PubMed] [Google Scholar]
- 13.Schramek D, Leibbrandt A, Sigl V, et al. Osteoclast differentiation factor RANKL controls development of progestin-driven mammary cancer. Nature. 2010;468:98–102. doi: 10.1038/nature09387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Horwitz KB, Sartorius CA. Progestins in hormone replacement therapies reactivate cancer stem cells in women with preexisting breast cancers: A hypothesis. J Clin Endocrinol Metab. 2008;93:3295–3298. doi: 10.1210/jc.2008-0938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Benakanakere I, Besch-Wiliford C, Schnell J, Brandt S, Molinolo A, Hyder SM. Natural and synthetic progestins accelerate 7, 12- dimethylbenz<a> anthracene (DMBA)-initiated mammary tumors and increase angiogenesis in Sprague-Dawley rats. Clin Cancer Res. 2006;12:4062–4071. doi: 10.1158/1078-0432.CCR-06-0427. [DOI] [PubMed] [Google Scholar]
- 16.Carrol CE, Benakanakere I, Besch-Williford C, Ellersierck MR, Hyder SM. Curcumin delays development of medroxyprogesterone acetate-accelerated 7,12-dimethybenz(a)anthracene-induced mammary tumors. Menopause. 2010;17:178–184. doi: 10.1097/gme.0b013e3181afcce5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Benakanakere I, Besch-Williford C, Carroll CE, Hyder SM. Synthetic progestins differentially promote or prevent 7,12-Dimethylbenz(a)anthracene-induced mammary tumors in Sprague-Dawley rats. Cancer Prev Res. 2010;3:1157 – 1167. doi: 10.1158/1940-6207.CAPR-10-0064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Patel D, Shukla S, Gupta S. Apigenin and cancer chemoprevention: Progress, potential and promise. Int J Oncol. 2007;30:233–245. [PubMed] [Google Scholar]
- 19.Birt DF, Hendrich S, Wang W. Dietary agents in cancer prevention: Flavonoids and isoflavonoid. Pharmacol Ther. 2001;90:157–177. doi: 10.1016/s0163-7258(01)00137-1. [DOI] [PubMed] [Google Scholar]
- 20.Gupta S, Afaq F, Mukhtar H. Selective growth-inhibitory, cell-cycle deregulatory and apoptotic response of apigenin in normal versus human prostate carcinoma cells. Biochem Biophys Res Commun. 2001;287 (4):914–920. doi: 10.1006/bbrc.2001.5672. [DOI] [PubMed] [Google Scholar]
- 21.Lien-Chai Chiang, Ng Lean Teik, I-Cheng Lin, Po-Lin Kuo, Chun-Ching Lin. Anti-proliferative effect of apigenin and its apoptotic induction in human Hep G2 cells. Cancer Lett. 2006;237:207–214. doi: 10.1016/j.canlet.2005.06.002. [DOI] [PubMed] [Google Scholar]
- 22.Mafuvadze B, Benakanakere I, Hyder SM. Apigenin blocks induction of VEGF mRNA and protein in progestin-treated human breast cancer cells. Menopause. 2010;17:1055–1063. doi: 10.1097/gme.0b013e3181dd052f. [DOI] [PubMed] [Google Scholar]
- 23.Hyder SM, Chiappetta C, Stancel GM. Pharmacological and endogenous progestins induce vascular endothelial growth factor expression in human breast cancer cells. Int J Cancer. 2001;92:469–473. doi: 10.1002/ijc.1236. [DOI] [PubMed] [Google Scholar]
- 24.Chen D, Landis-Piwowar KR, Chen MS, Dou QP. Inhibition of proteasome activity by the dietary flavonoid apigenin is associated with growth inhibition in cultured breast cancer cells and xenografts. Breast Cancer Res. 2007;9:R80. doi: 10.1186/bcr1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Aldaz CM, Liao QY, LaBate M, Johnston DA. Medroxyprogesterone acetate accelerates the development and increases the incidence of mouse mammary tumors induced by dimethylbenzanthracene. Carcinogenesis. 1996;17:2069–2072. doi: 10.1093/carcin/17.9.2069. [DOI] [PubMed] [Google Scholar]
- 26.Wu J, Brandt S, Hyder SM. Ligand- and cell-specific effects of signal transduction pathway inhibitors on progestin-induced VEGF levels in human breast cancer cells. Molecular Endocrinology. 2005;19:312–326. doi: 10.1210/me.2004-0252. [DOI] [PubMed] [Google Scholar]
- 27.Narduli AM, Greene GL, O’Malley BW, Katzenellenbogen BS. Regulation of progesterone receptor message ribonucleic acid and protein levels in MCF-7 cells by estradiol: analysis of estrogen’s effect on progesterone receptor synthesis and degradation. Endocrinol. 1988;122:935–944. doi: 10.1210/endo-122-3-935. [DOI] [PubMed] [Google Scholar]
- 28.Horwitz KB, Koseki Y, McGuire WL. Estrogen control of progesterone receptor in human breast cancer: Role of estradiol and antiestrogen. Endocrinol. 1978;103:1742–1751. doi: 10.1210/endo-103-5-1742. [DOI] [PubMed] [Google Scholar]
- 29.Dennis AP, Lonard DM, Nawaz Z, O’Malley BW. Inhibition of the 26S proteasome blocks progesterone receptor-dependent transcription through failed recruitment of RNA polymerase II. J Steroid Biochem Mol Biol. 2005;94:337–346. doi: 10.1016/j.jsbmb.2004.11.009. [DOI] [PubMed] [Google Scholar]
- 30.Jaiswal AS, Marlow BP, Gupta N, Narayan S. β-catenin-mediated transactivation and cell-cell adhesion pathways are important in curcumin (diferuylmethane)-induced growth arrest and apoptosis in colon cancer cells. Oncogene. 2002;21:8414–8427. doi: 10.1038/sj.onc.1205947. [DOI] [PubMed] [Google Scholar]
- 31.Horinaka M, Yoshida T, Nakata S, Wakada M, Sakai T. The dietary flavonoid apigenin sensitizes malignant tumor cells to tumor necrosis factor-related apoptosis inducing ligands. Molecular Cancer Therapeutics. 2006;5:945–951. doi: 10.1158/1535-7163.MCT-05-0431. [DOI] [PubMed] [Google Scholar]
- 32.Yin F, Giuliano AE, Van Herle AJ. Apigenin inhibits growth and induces G2/M arrest by modulating cyclin-CDK regulators and ERK MAP kinase activation in breast carcinoma cells. Anticancer Res. 2001;21:413–420. [PubMed] [Google Scholar]
- 33.Hyder SM. Sex-steroid regulation of vascular endothelial growth factor in breast cancer. End Related Cancer. 2006;13:667–687. doi: 10.1677/erc.1.00931. [DOI] [PubMed] [Google Scholar]
- 34.Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med. 1995;1:27–31. doi: 10.1038/nm0195-27. [DOI] [PubMed] [Google Scholar]
- 35.Li Y, Zhang T, Korkaya H, Liu S, Lee H-F, Newman B, Yu Y, Clotheir SG, Schwartz SJ, Wicha MS, Sun D. Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clin Cancer Res. 2010;16:2580–2590. doi: 10.1158/1078-0432.CCR-09-2937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Joshi PA, Jackson HW, Beristain AG, et al. Progesterone induces adult mammary stem cell expansion. Nature. 465:803–807. doi: 10.1038/nature09091. [DOI] [PubMed] [Google Scholar]
- 37.Pahlke G, Ngiewih Y, Kern M, Jakobs S, Marko D, Eisenbrand G. Impact of quercetin and EGCG on key elements of the Wnt pathway in human colon carcinoma cells. J Agric Food Chem. 2006;54:7075–7082. doi: 10.1021/jf0612530. [DOI] [PubMed] [Google Scholar]
- 38.Breinholt V, Hossaini A, Svendsen GW, Brouwer C, Nielsen SE. Estrogenic activity of flavonoids in mice. The importance of estrogen receptor distribution, metabolism and bioavailability. Food Chem Toxicol. 2000;38:555–564. doi: 10.1016/s0278-6915(00)00046-6. [DOI] [PubMed] [Google Scholar]
- 39.Long X, Fan M, Bigsby RM, Nephew KP. Apigenin inhibits antiestrogen-resistant breast cancer cell growth through estrogen receptor-alpha-dependent and estrogen receptor-alpha-independent mechanisms. Mol Cancer Ther. 2008;7:2096–2108. doi: 10.1158/1535-7163.MCT-07-2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Meyer H, Bolarinwa A, Wolfram G, Linseisen J. Bioavailability of apigenin from apiin-rich parsley in humans. Ann Nutr Metab. 2006;50:167–172. doi: 10.1159/000090736. [DOI] [PubMed] [Google Scholar]