Abstract
Genome-wide association studies have recently identified single nucleotide polymorphisms in proximity to the interleukin-28B (IL-28B) gene that can predict sustained virologic response (SVR) in patients with chronic hepatitis C virus (HCV) infection who are undergoing therapy with pegylated interferon (IFN) a and ribavirin. IL-28B encodes IFN-λ3, a type III IFN involved in host antiviral immunity. Favorable variants of the 2 most widely studied IL-28B polymorphisms, rs1 2979860 and rs8099917, are strong pretreatment predictors of early viral clearance and SVR in patients with genotype 1 HCV infection. Variations in the distribution of IL-28B alleles may partly explain differences in SVR rates among ethnic groups. Further investigations have implicated IL-28B in the development of chronic HCV infection versus spontaneous resolution of acute infection and suggest that IL-28B may be a key factor involved in host immunity against HCV. Clinical trials of IFN-λ as a therapeutic agent for chronic HCV infection are currently underway. The use of IL-28B polymorphisms as a predictive tool will have a major impact on treatment strategies for chronic HCV infection, particularly in the context of emerging therapies and direct-acting antiviral agents.
Keywords: Chronic hepatitis C virus, genetic, genome, IL-28B, interferon lambda, pegylared interferon, ribavirin, sustained virologic response
Hepatitis C virus (HCV) is the most common chronic blood-borne infection in the United States, affecting over 4 million people and accounting for the majority of newly diagnosed cases of chronic liver disease.1,2 Advanced liver disease resulting from chronic HCV infection is a major cause of liver-related mortality and is expected to increase in prevalence over the next decade.3,4 Pegylated interferon (pegIFN) α-2a or pegIFN α-2b in combination with ribavirin (RBV) was the stan-dard-of-care treatment for chronic hepatitis C—prior to the recent licensure of boceprevir (Victrelis, Merck) and telaprevir (Incivek, Vertex) in the United States—and sustained virologic response (SVR) occurred in only approximately half of patients.5–8
The ability to predict treatment outcomes is an important consideration in the management of chronic HCV infection, as the approach to treatment has become more individualized in order to achieve optimal toler-ability, duration of therapy, and virologic response. HCV genotype is well established as the strongest predictor of treatment response and is a primary determinant of treatment duration and RBV dose.7 Other host and viral factors have been identified as predictors of a poor response to therapy; these factors include high serum HCV RNA levels, advanced hepatic fibrosis, increased age, obesity, and black race.5–9
A major unresolved question is the extent to which host genetic factors may influence response to antiviral therapy in patients with chronic HCV infection and whether reliable genetic markers predictive of SVR can be identified. As knowledge of the human genome has expanded and our ability to detect genomic differences among individuals has become more advanced, recent studies have identified single nucleotide polymorphisms (SNPs) in proximity to the interleukin-28B (IL-28B) gene as important predictors of treatment outcome. These findings have not only provided a new window into host responsiveness to IFN-based therapy but have also yielded insights into genetic susceptibility to chronic HCV infection, the molecular biology of host immunity against HCV, and the potential for discovery of new therapeutic agents.
Discovery of IL-28B Polymorphisms
The recent development of genome-wide association studies (GWAS) has made possible the ability to efficiently identify genomic variations among individuals on a large scale.10 These studies involve techniques that allow investigators to survey the entire genome and identify SNPs that link groups of individuals with complex traits, such as response to antiviral therapy. Four recently published GWAS have investigated the genetic predictors associated with SVR in patients with genotype 1 HCV infection.11–14 These GWAS were performed in distinct populations, but all 4 studies identified SNPs located on chromosome 19 in the region of the IL-28B gene as the strongest predictors of virologic response.
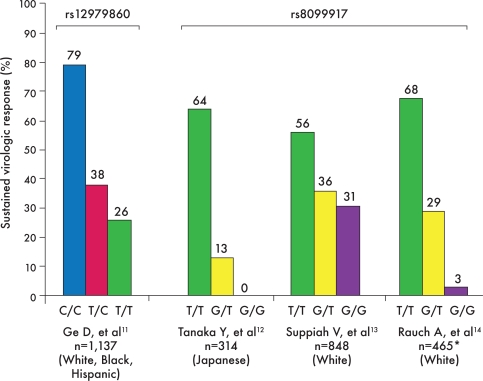
The first and largest of these studies arose from the IDEAL study, in which 1,137 patients were included in a genome-wide association study.11 The IDEAL study was a large, randomized, controlled trial of over 3,000 patients that investigated different dosing regimens of pegIFN a-2b and pegIFN α-2a in combination with RBV.8 A study of patients in the IDEAL trial who consented to undergo a genome-wide association study showed a significantly increased likelihood of SVR in association with the C/C genotype of the rs12979860 SNP, which is located 3 kb upstream of the IL-28B gene (Figure 1). Three smaller studies involving Japanese, Australian, and European cohorts all reported increased SVR rates in patients with the T/T genotype of another SNP, rs8099917, which is located 8.9 kb downstream from IL-28B in the intergenic region between the IL-28A and IL-28B genes.12–14
Figure 1.
This graph shows rates of sustained virologic response (SVR) based on interleukin-28B single nucleotide polymorphism genotypes as reported by 4 genome-wide association studies of patients who underwent antiviral therapy with combination pegylated interferon α and ribavirin. The C/C variant of the rsl2979860 polymorphism and the T/T variant of the rs8099917 polymorphism are associated with an increased likelihood of SVR.
*Approximately one half of patients from the study by Rauch and colleagues were infected with genotype 2 or 3 hepatitis C virus (HCV) infection, while the other studies included only patients with genotype 1 HCV infection.11–14
Subsequent studies investigating both the rs12979860 and rs8099917 SNPs have confirmed these findings. The IL-28A and IL-28B genes encode IFN-λ2 and IFN-λ3, respectively, both of which are cytokines in the IFN-λ family.15 The discovery of IL-28B in association with virologic response has many implications, particularly in relation to the role that IFN-λ plays in host immunity and clearance of HCV.16
Sustained Virologic Response
Genotype 1
The predictive value of genetic markers has the greatest potential impact in patients with genotype 1 infection who are undergoing antiviral therapy with pegIFN and RBV for chronic HCV infection, as this group has lower rates of SVR.5–8 As new and more effective antiviral therapy becomes available, a better understanding of an individual's potential for virologic response may influence the decision of whether to initiate antiviral therapy, which antiviral agents to choose, and how long to continue therapy.17–19 Given the challenges associated with optimizing treatment outcomes in patients with chronic genotype 1 HCV infection, much of the data on IL-28B as a predictor of SVR involves this population.
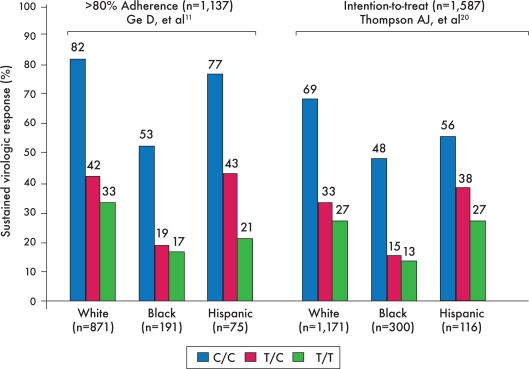
The first published genome-wide association study of IL-28B as a predictor of SVR, reported by Ge and colleagues, included only genotype 1-infected patients.11 Most of these patients were enrolled in the IDEAL study, but this study also included a small subset of patients from another prospective clinical trial.11 In addition to its large size, another major advantage of this study was its diversity: One quarter of these patients were either black or Hispanic. Overall, patients with the C/C genotype of the rs12979860 polymorphism had a 2-fold higher rate of SVR compared to patients with the T/T genotype (Figure 1). This relationship was consistent across different ethnic groups and was later confirmed through an intention-to-treat analysis that included all members of this cohort, regardless of adherence (Figure 2).20 In addition, the C/C genotype was found to be the strongest predictor of SVR, independent of serum HCV RNA level, fibrosis stage, or ethnicity.11,20 Subsequent studies in European and American cohorts demonstrated a stronger association between SVR rates and the C/C genotype of rs12979860, compared to variants that carry the T allele, such that the T allele was found to be an independent predictor of treatment failure.21,22
Figure 2.
Sustained virologic response (SVR) rates based on rs12979860 polymorphism genotypes differ across ethnic groups. This graph shows SVR rates for both the adherent and intention-to-treat cohorts of the genome-wide association study that was conducted among patients enrolled in the IDEAL study. The C/C variant was most strongly associated with SVR, independent of ethnicity or adherence.
Additional GWAS identified a second SNP, rs8099917, as a predictor of virologic response in patients with genotype 1 HCV infection. In a Japanese cohort, Tanaka and colleagues reported that rs8099917 had a strong association with response to therapy and that the presence of the G allele was the most significant independent predictor of null response.12 Likewise, the T/T genotype of the rs8099917 polymorphism was associated with a high SVR rate (Figure 1). Suppiah and colleagues also identified rs8099917 as being strongly associated with virologic response; this association was seen in an initial group of Australian patients and was then validated in a larger cohort that included Europeans.13 Similar to the Tanaka study, the Suppiah study reported the highest rate of SVR in patients with the T/T genotype of rs8099917, while the presence of the G allele was predictive of nonresponse.12,13 In both of these studies, the association between rs8099917 and SVR was independent of serum HCV RNA level, fibrosis stage, or gender.12,13 A more recent Swiss genome-wide association study also found the presence of the G allele to be strongly associated with treatment failure.14 Additional reports have validated these findings, including a Japanese study in which the T/T genotype of rs8099917 was found to be the strongest independent predictor of SVR, whereas the G allele was the strongest independent predictor of null virologic response in multivariate analyses.23
At this time, only limited preliminary data are available on IL-28B as a predictor of SVR in patients with chronic genotype 1 HCV infection who receive triple therapy consisting of pegIFN and RBV plus either tela-previr or boceprevir.24–26 In a small study from Japan, treat-ment-naïve patients and prior nonresponders were treated with a 12-week regimen of triple therapy that included telaprevir (with or without an additional 12 weeks of dual therapy with pegIFN and RBV), and increased SVR rates were noted in patients with favorable rs12979860 and rs8099917 genotypes.24 In a larger phase III study, the impact of rs12979860 genotypes on SVR rates was retrospectively assessed in 527 of 662 patients enrolled in a pivotal study of telaprevir administered in combination with pegIFN and RBV in patients with prior treatment failure.25 The highest proportion of rs12979860 T/T genotype was observed in prior null responders (28%), while the highest frequency of the C/C genotype occurred among relapsers (27%). In the overall population of prior relapsers, partial responders, and null responders who were treated with triple therapy, SVR rates were 79% in patients with the C/C genotype, 60% with the T/C genotype, and 61% with the T/T genotype. However, IL-28B genotype did not predict outcomes in the individual groups based on prior treatment response. In pivotal trials of triple therapy involving boceprevir plus pegIFN and RBV, rs12979860 genotype was a strong predictor of SVR in both treatment-naïve and nonre-sponder cohorts, although virologic response at Week 4 was a stronger predictor than IL-28B genotype or other baseline variables.26
Another challenging population in which virologic response to therapy is decreased includes patients with chronic hepatitis C who are co-infected with HIV. Data have emerged suggesting that rs12979860 and rs8099917 IL-28B polymorphisms may play an important role in predicting SVR in this population as well. Several studies involving European cohorts have reported the rs12979860 C/C and rs8099917 T/T variants to be independent predictors of SVR in patients with HCV/HIV co-infection, particularly in patients with genotype 1 HCV infection.27–29
Genotypes 2 and 3
In contrast to the data on genotype 1 HCV infection, the available data on IL-28B polymorphisms as predictors of SVR in patients with genotype 2 or 3 HCV infection are conflicting. However, IL-28B may play a role in some patients, particularly those who do not demonstrate a rapid virologic response (RVR), which is defined by undetectable levels of HCV RNA in serum at 4 weeks of therapy. Several studies, including 1 of the initial GWAS that identified rs8099917 as a predictor of SVR in patients with genotype 1 HCV infection, have not demonstrated any clear association between IL-28B polymorphisms and SVR in patients with genotype 2 or 3 HCV infection.14,21,30 Some reports have identified favorable variants of both rs8099917 and rs12979860 as being independently associated with RVR but not SVR, suggesting an increased rate of relapse in this subset of patients.31,32
In contrast, 2 European studies found an association between the C/C variant of rs12979860 and SVR in cohorts with genotype 2 or 3 HCV infection, although this relationship may be strongest in patients who do not achieve RVR.33,34 Ultimately, given that overall rates of SVR are significantly higher in patients with genotype 2 or 3 HCV infection compared to patients with genotype 1 HCV infection, the role of predictive factors such as IL-28B polymorphisms may not be as important in the former group.
Early Viral Clearance
The most important predictive factor during a course of antiviral therapy is the change in serum HCV RNA level, with clearance of HCV earlier during therapy being strongly predictive of SVR. RVR has a positive predictive value for SVR of up to 90% in patients with genotype 1 HCV infection, while failure to achieve an early virologic response (EVR; defined as a greater-than-2-log10 decrease in HCV RNA level at Week 12 of therapy) has a negative predictive value for SVR of up to 100%.6,35,36
Several reports in different populations have shown that IL-28B polymorphisms are also predictive of these parameters. Studies evaluating early viral kinetics during therapy have observed that patients with the rs12979860 C/C genotype have significantly faster first-phase declines in serum HCV RNA levels during the first 24 hours following initiation of therapy.21,37 Likewise, achievement of RVR has been strongly associated with the rs12979860 C/C genotype as well as the T/T genotype of the rs8099917 polymorphism.20,21,23 These IL-28B profiles are also associated with achievement of complete EVR, with undetectable levels of HCV RNA at Week 12, independent of ethnicity.20,23
Role of IL-28B Polymorphisms in Liver Transplantation
IL-28B polymorphisms may play an important role in patients with chronic HCV infection and end-stage liver disease who undergo liver transplantation. Recurrence of HCV infection occurs in all liver transplant recipients with detectable HCV RNA levels at the time of transplantation, resulting in chronic hepatitis, progressive fibrosis, and cirrhosis in up to 30% of patients.38 Recent data reveal that IL-28B polymorphisms may predict both recurrence of disease and response to antiviral therapy following liver transplantation.
A study assessing both recipient and donor IL-28B rs12979860 genotypes in consecutive HCV-infected patients undergoing liver transplantation found that the presence of the recipient C allele was an independent predictor of delayed recurrence of disease at 2 and 5 years.39 Both recipient and donor C/C genotypes appear to be strongly associated with SVR, although 1 recent study suggested that the donor genotype may be more important in this setting.39,40
Mechanisms and Immunology
Epidemiology
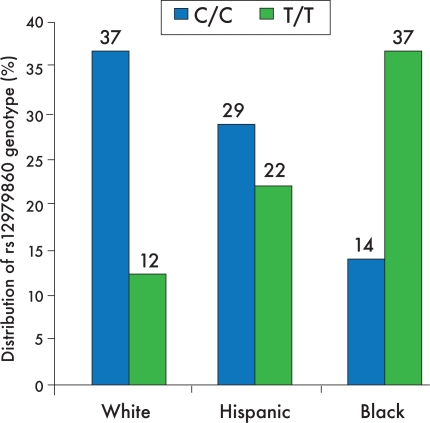
An important finding associated with the IL-28B rs12979860 polymorphism is the distribution of alleles across different ethnic groups. Ge and colleagues reported the highest frequency of the C allele in East Asians, followed by Americans of European ancestry and Hispanics; the lowest frequency of the C allele was found in blacks.11 In a further analysis of the intention-to-treat population, the C/C genotype was more prevalent in whites and Hispanics, while the T/T genotype was most common in blacks (Figure 3).20 Ge and colleagues proposed that possibly one half of the difference in SVR rates between whites and blacks could be accounted for by differences in C allele frequency.11
Figure 3.
The relative distribution of interleukin-28B rs12979860 polymorphism genotypes varies among different ethnic groups. This graph shows the distribution of alleles associated with the rs12979860 polymorphism across different ethnic backgrounds, with specific attention to the C/C and T/T genotypes. Data were obtained from a large multiethnic cohort with genotype 1 hepatitis C virus infection, based on an intention-to-treat analysis.
Adapted from Thompson AJ, et al.20
A large, population-based study of over 2,000 individuals from different areas of the world revealed that the C allele occurred with the highest frequency in areas of East Asia, followed by Europe and then Africa.41 The predictive value of the C/C genotype may also vary across different ethnic groups, as the presence of a C/C genotype appears to have a greater specificity and negative predictive value for SVR in blacks than in whites or Hispanics (Table 1).20
Table 1.
Performance Characteristics of Interleukin-28B Polymorphisms in the Prediction of Sustained Virologic Response
| Sensitivity | Specificity | PPV | NPV | |
|---|---|---|---|---|
| C/C (fs12979860) | ||||
| Stättermayer AF, et al21* | 0.50 | 0.86 | 0.81 | 0.59 |
| Thompson AJ, et al20† | ||||
| White | 0.56 | 0.79 | 0.69 | 0.68 |
| Black | 0.35 | 0.91 | 0.48 | 0.86 |
| Hispanic | 0.40 | 0.78 | 0.56 | 0.66 |
| McCarthy JJ, et al22* | 0.64 | 0.77 | — | — |
| T/T (rs8099917) | ||||
| Stättermayer AF, et al21* | 0.68 | 0.62 | 0.72 | 0.58 |
Included patients with both genotype 1 and non-genotype 1 hepatitis C virus infection.
Included only patients with genotype 1 hepatitis C virus infection.
- NPV
negative predictive value
- PPV
positive predictive value
Association with Spontaneous Viral Clearance
Findings from the initial genome-wide association study suggested that IL-28B may play a role in determining whether acute HCV infection leads to spontaneous clearance or chronic infection. Ge and colleagues first noted that the presence of the C allele (rs12979860) was lower in patients with chronic genotype 1 HCV infection compared to matched healthy controls, suggesting that the C allele may be associated with spontaneous res-olution.11 The same finding has been noted in additional studies, including a large study of over 1,000 individuals in which the prevalence of the rs12979860 genotype was investigated in patients who achieved spontaneous resolution and those who developed chronic HCV infection.30,41 This study found that the C allele occurred more frequently in patients who spontaneously cleared HCV. Individuals with the C/C genotype had a 3-fold greater likelihood of spontaneous HCV clearance compared to patients who had the T/C or T/T genotype. Additionally, the strong association between C/C genotype and spontaneous clearance was present in both whites and blacks.
A recent German cohort study reported similar findings: The C/C genotype was associated with the greatest likelihood of spontaneous clearance, followed by the T/C genotype at an intermediate rate, and then the T/T genotype with the lowest likelihood of clear-ance.42 Interestingly, patients with the C/C variant were more likely to have acute jaundice, although the positive association between jaundice and spontaneous clearance was more significant in those with non-C/C genotypes. Similar findings have been reported in association with the rs8099917 polymorphism, in which the favorable T/T genotype is associated with spontaneous resolution; this relationship was also found in HCV/HIV co-infected individuals.14,43
The distribution of IL-28B polymorphisms may also vary among HCV genotypes, suggesting further differences in the risk of developing a chronic infection. Studies have shown that the C/C variant occurs in a higher proportion of patients with genotype 2 or 3 HCV infection compared to patients with genotype 1 HCV infection.22,30 When compared with healthy controls, the frequency of the C/C genotype appears to be highest in controls, intermediate in patients with genotype 2 or 3 HCV infection, and lowest in patients with genotype 1 HCV infection.34 These findings suggest a potentially increased rate of spontaneous clearance in patients infected with genotype 2 or 3 HCV compared to genotype 1 HCV. However, further studies will be required to determine the clinical relevance of these observations and to ascertain whether these findings bear any relationship to differences in rates of response to therapy among HCV genotypes.
Interferon-λ and Hepatitis C Virus Infection
The IFN-λ cytokine family includes IFN-λ1, IFN-λ2, and IFN-λ3, which are encoded by the IL-29, IL-28A, and IL-28B genes, respectively.15 The IFN-λ cytokines are considered to be type III IFNs; similar to type I IFNs, they are involved in the host response to viral infection. The strong association between IL-28B polymorphisms and response to antiviral therapy in patients with chronic HCV infection has led to a greater understanding of the role that IFN-λ plays in immune-mediated and treatment-associated clearance of HCV. IL-28B appears to be uniquely important in response to HCV therapy, as GWAS have not reported any SNPs outside of the IL-28B haplotype block or IL-28A/B locus that demonstrate a significant association with virologic response.13,14 Although patients with nonresponder alleles have been reported to have decreased whole blood expression of IL-28A and IL-28B and decreased peripheral blood mononuclear cell IL-28 mRNA expression, the question remains how SNPs in the region of IL-28B may regulate IFN-λ cytokine activity or function.12,13,16
A series of intracellular events are critically important for limiting HCV replication and activating the cellular immunity involved in the clearance of infected hepato-cytes.44 In response to HCV infection, the activation of cellular transcription factors promotes secretion of type I IFNs such as IFN-a and IFN-ß. Binding of type I IFNs to hepatocyte receptors results in activation of the Janus kinase-signal transducer and activator of transcription (JAK-STAT) signal transduction pathway and subsequent production of IFN-stimulated genes (ISGs), inhibition of HCV replication, and priming of effector cells involved in HCV-specific host adaptive immunity.44 This adaptive immune response, which is characterized by CD4-positive helper T cells and CD8-positive cytotoxic T cells, plays a critical role in eradicating HCV in the setting of acute infection and during treatment of chronic infection.45–48
Emerging data reveal that IFN-λ3 (IL-28B) may inhibit HCV replication via the JAK-STAT pathway, leading to expression of ISGs in a manner similar to that seen with type I IFNs.49 Recent evidence also suggests that the intrahepatic expression of ISGs may vary based on IL-28B genotype.50 These findings suggest that IFN-k cytokines may play an important role in clearance of HCV both in the acute setting and during antiviral therapy. The use of IFN-λ as a therapeutic agent for chronic HCV infection is currently being studied, and phase I clinical studies are underway.51
IL-28B and Treatment Strategies
IL-28B polymorphisms have been reported to be the strongest pretreatment predictors of SVR independent of other host and viral factors, including ethnicity, presence of advanced fibrosis, gender, or HCV RNA level. IL-28B genotype may lack sufficient sensitivity and specificity as a predictor of SVR in some populations when used alone, but it will greatly contribute to assessing patient responsiveness to therapy when used in conjunction with other clinical predictors. Whether IL-28B will be instrumental in selecting individuals who may benefit from a shorter duration of therapy has yet to be determined.
As novel, more effective, direct-acting antiviral agents (DAAs) become available for the treatment of chronic HCV infection, IL-28B may play a role in selecting specific antiviral agents or deciding to treat patients with pegIFN and RBV alone. Overall, IL-28B will certainly remain a key factor in treatment algorithms and will be included as part of an individualized approach to HCV treatment. Ongoing clinical studies will further elucidate how genetic profiling with IL-28B will affect treatment strategies and decision-making, particularly in the context of emerging DAAs.
Summary
The discovery of IL-28B as a predictor of SVR in patients with genotype 1 HCV infection will have a lasting impact, particularly in the development of individualized treatment strategies, understanding of host molecular biology and determinants of immune-mediated viral clearance, and development of novel antiviral therapies. The strong associations between IL-28B polymorphism genotypes and SVR are consistent across different ethnic groups and populations. IL-28B will be utilized most in the setting of chronic genotype 1 HCV infection and will be particularly beneficial to patients with difficult-to-treat characteristics, in whom IL-28B genotype may guide decisions on whether to initiate therapy, how long to continue therapy, and which antiviral agents to use. How IL-28B genetic profiling will be used in the setting of DAA therapy has yet to be determined. Further investigations into the molecular biology of IFN-λ cytokines as they relate to HCV infection are ongoing, and the development of IFN-λ as a viable treatment alternative remains an exciting prospect.
References
- 1.Armstrong GL, Wasley A, Simard EP, et al. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med. 2006;144:705–714. doi: 10.7326/0003-4819-144-10-200605160-00004. [DOI] [PubMed] [Google Scholar]
- 2.Bell BP, Manos MM, Zaman A, et al. The epidemiology of newly diagnosed chronic liver disease in gastroenterology practices in the United States: results from population-based surveillance. Am J Gastroenterol. 2008;103:2727–2736. doi: 10.1111/j.1572-0241.2008.02071.x. [DOI] [PubMed] [Google Scholar]
- 3.Wise M, Bialek S, Finelli L, et al. Changing trends in hepatitis C-related mortality in the United States, 1995–2004. Hepatology. 2008;47:1128–1135. doi: 10.1002/hep.22165. [DOI] [PubMed] [Google Scholar]
- 4.Davis GL, Alter MJ, El-Serag H, et al. Aging of hepatitis C virus (HCV)-infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology. 2010;138:513–521. doi: 10.1053/j.gastro.2009.09.067. [DOI] [PubMed] [Google Scholar]
- 5.Manns MP, McHutchison JG, Gordon SC, et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet. 2001;358:958–965. doi: 10.1016/s0140-6736(01)06102-5. [DOI] [PubMed] [Google Scholar]
- 6.Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347:975–982. doi: 10.1056/NEJMoa020047. [DOI] [PubMed] [Google Scholar]
- 7.Hadziyannis SJ, Sette H, Jr, Morgan TR, et al. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med. 2004;140:346–355. doi: 10.7326/0003-4819-140-5-200403020-00010. [DOI] [PubMed] [Google Scholar]
- 8.McHutchison JG, Lawitz EJ, Shiffman ML, et al. Peginterferon alfa-2b or alfa-2a with ribavirin for treatment of hepatitis C infection. N Engl J Med. 2009;361:580–593. doi: 10.1056/NEJMoa0808010. [DOI] [PubMed] [Google Scholar]
- 9.Muir AJ, Bornstein JD, Killenberg PG. Peginterferon alfa-2b and ribavirin for the treatment of chronic hepatitis C in blacks and non-Hispanic whites. N Engl J Med. 2004;350:2265–2271. doi: 10.1056/NEJMoa032502. [DOI] [PubMed] [Google Scholar]
- 10.Manolio TA. Genomewide association studies and assessment of the risk of disease. N Engl J Med. 2010;363:166–176. doi: 10.1056/NEJMra0905980. [DOI] [PubMed] [Google Scholar]
- 11.Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461:399–401. doi: 10.1038/nature08309. [DOI] [PubMed] [Google Scholar]
- 12.Tanaka Y, Nishida N, Sugiyama M, et al. Genome-wide association of IL28B with response to pegylated interferon-alpha and ribavirin therapy for chronic hepatitis C. Nat Genet. 2009;41:1105–1109. doi: 10.1038/ng.449. [DOI] [PubMed] [Google Scholar]
- 13.Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet. 2009;41:1100–1104. doi: 10.1038/ng.447. [DOI] [PubMed] [Google Scholar]
- 14.Rauch A, Kutalik Z, Descombes P, et al. Genetic variation in IL28B is associated with chronic hepatitis C and treatment failure: a genome-wide association study. Gastroenterology. 2010;138:1338–1345. doi: 10.1053/j.gastro.2009.12.056. [DOI] [PubMed] [Google Scholar]
- 15.Balagopal A, Thomas DL, Thio CL. IL28B and the control of hepatitis C virus infection. Gastroenterology. 2010;139:1865–1876. doi: 10.1053/j.gastro.2010.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kelly C, Klenerman P, Barnes E. Interferon lambdas: the next cytokine storm. Gut. Epub ahead of print. [DOI] [PMC free article] [PubMed]
- 17.McHutchison JG, Everson GT, Gordon SC, et al. Telaprevir with pegin-terferon and ribavirin for chronic HCV genotype 1 infection. N Engl J Med. 2009;360:1827–1838. doi: 10.1056/NEJMoa0806104. [DOI] [PubMed] [Google Scholar]
- 18.Hezode C, Forestier N, Dusheiko G, et al. Telaprevir and peginterferon with or without ribavirin for chronic HCV infection. N Engl J Med. 2009;360:1839–1850. doi: 10.1056/NEJMoa0807650. [DOI] [PubMed] [Google Scholar]
- 19.Kwo PY, Lawitz EJ, McCone J, et al. Efficacy of boceprevir, an NS3 protease inhibitor, in combination with peginterferon alfa-2b and ribavirin in treatment-naive patients with genotype 1 hepatitis C infection (SPRINT-1): an open-label, randomised, multicentre phase 2 trial. Lancet. 2010;376:705–716. doi: 10.1016/S0140-6736(10)60934-8. [DOI] [PubMed] [Google Scholar]
- 20.Thompson AJ, Muir AJ, Sulkowski MS, et al. Interleukin-28B polymorphism improves viral kinetics and is the strongest pretreatment predictor of sustained viro-logic response in genotype 1 hepatitis C virus. Gastroenterology. 2010;139:120–129. doi: 10.1053/j.gastro.2010.04.013. [DOI] [PubMed] [Google Scholar]
- 21.Stättermayer AF, Stauber R, Hofer H, et al. Impact of IL28B genotype on the early and sustained virologic response in treatment-naive patients with chronic hepatitis C. Clin Gastroenterol Hepatol. 2011;9:344–350. doi: 10.1016/j.cgh.2010.07.019. [DOI] [PubMed] [Google Scholar]
- 22.McCarthy JJ, Li JH, Thompson A, et al. Replicated association between an IL28B gene variant and a sustained response to pegylated interferon and ribavirin. Gastroenterology. 2010;138:2307–2314. doi: 10.1053/j.gastro.2010.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kurosaki M, Tanaka Y, Nishida N, et al. Pre-treatment prediction of response to pegylated-interferon plus ribavirin for chronic hepatitis C using genetic polymorphism in IL28B and viral factors. J Hepatol. 2011;54:439–448. doi: 10.1016/j.jhep.2010.07.037. [DOI] [PubMed] [Google Scholar]
- 24.Akuta N, Suzuki F, Hirakawa M, et al. Amino acid substitution in hepatitis C virus core region and genetic variation near the interleukin 28B gene predict viral response to telaprevir with peginterferon and ribavirin. Hepatology. 2010;52:421–429. doi: 10.1002/hep.23690. [DOI] [PubMed] [Google Scholar]
- 25.Pol S, Aerssens J, Zeuzem S, et al. Similar SVR rates in IL28B CC, CT or TT prior relapser, partial- or null-responder patients treated with telaprevir/peginter-feron/ribavirin: retrospective analysis of the REALIZE study [abstract] J Hepatol. In press.
- 26.Poordad F, Bronowicki JP, Gordon SC, et al. IL28B polymorphism predicts virologic response in patients with hepatitis C genotype 1 treated with boceprevir (BOC) combination therapy [abstract] J Hepatol. In press.
- 27.Rallon NI, Naggie S, Benito JM, et al. Association of a single nucleotide polymorphism near the interleukin-28B gene with response to hepatitis C therapy in HIV/hepatitis C virus-coinfected patients. AIDS. 2010;24:F23–F29. doi: 10.1097/QAD.0b013e3283391d6d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pineda JA, Caruz A, Rivero A, et al. Prediction of response to pegylated interferon plus ribavirin by IL28B gene variation in patients coinfected with HIV and hepatitis C virus. Clin Infect Dis. 2010;51:788–795. doi: 10.1086/656235. [DOI] [PubMed] [Google Scholar]
- 29.Aparicio E, Parera M, Franco S, et al. IL28B SNP rs8099917 is strongly associated with pegylated interferon-alpha and ribavirin therapy treatment failure in HCV/HIV-1 coinfected patients. PLoS One. 2010;5:e13771. doi: 10.1371/journal.pone.0013771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Montes-Cano MA, Garcia-Lozano JR, Abad-Molina C, et al. Interleukin-28B genetic variants and hepatitis virus infection by different viral genotypes. Hepatol-ogy. 2010;52:33–37. doi: 10.1002/hep.23624. [DOI] [PubMed] [Google Scholar]
- 31.Moghaddam A, Melum E, Reinton N, et al. IL28B genetic variation and treatment response in patients with hepatitis C virus genotype 3 infection. Hepatology. 2011;53:746–754. doi: 10.1002/hep.24154. [DOI] [PubMed] [Google Scholar]
- 32.Yu ML, Huang CF, Huang JF, et al. Role of interleukin-28B polymorphisms in the treatment of hepatitis C virus genotype 2 infection in Asian patients. Hepatology. 2011;53:7–13. doi: 10.1002/hep.23976. [DOI] [PubMed] [Google Scholar]
- 33.Mangia A, Thompson AJ, Santoro R, et al. An IL28B polymorphism determines treatment response of hepatitis C virus genotype 2 or 3 patients who do not achieve a rapid virologic response. Gastroenterology. 2010;139:821–827. doi: 10.1053/j.gastro.2010.05.079. [DOI] [PubMed] [Google Scholar]
- 34.Sarrazin C, Susser S, Doehring A, et al. Importance of IL28B gene polymorphisms in hepatitis C virus genotype 2 and 3 infected patients. J Hepatol. 2011;54:415–421. doi: 10.1016/j.jhep.2010.07.041. [DOI] [PubMed] [Google Scholar]
- 35.Ferenci P, Fried MW, Shiffman ML, et al. Predicting sustained virological responses in chronic hepatitis C patients treated with peginterferon alfa-2a (40KD)/ribavirin. J Hepatol. 2005;43:425–433. doi: 10.1016/j.jhep.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 36.Davis GL, Wong JB, McHutchison JG, et al. Early virologic response to treatment with peginterferon alfa-2b plus ribavirin in patients with chronic hepatitis C. Hepatology. 2003;38:645–652. doi: 10.1053/jhep.2003.50364. [DOI] [PubMed] [Google Scholar]
- 37.Bochud PY, Bibert S, Negro F, et al. IL28B polymorphisms predict reduction of HCV RNA from the first day of therapy in chronic hepatitis C. J Hepatol. Epub ahead of print. [DOI] [PubMed]
- 38.Neumann UP, Berg T, Bahra M, et al. Fibrosis progression after liver transplantation in patients with recurrent hepatitis C. J Hepatol. 2004;41:830–836. doi: 10.1016/j.jhep.2004.06.029. [DOI] [PubMed] [Google Scholar]
- 39.Charlton MR, Thompson A, Veldt BJ, et al. Interleukin-28B polymorphisms are associated with histological recurrence and treatment response following liver transplantation in patients with hepatitis C virus infection. Hepatology. 2011;53:317–324. doi: 10.1002/hep.24074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lange CM, Moradpour D, Doehring A, et al. Impact of donor and recipient IL28B rs12979860 genotypes on hepatitis C virus liver graft reinfection. J Hepatol. Epub ahead of print. [DOI] [PubMed]
- 41.Thomas DL, Thio CL, Martin MP, et al. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nature. 2009;461:798–801. doi: 10.1038/nature08463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tillmann HL, Thompson AJ, Patel K, et al. A polymorphism near IL28B is associated with spontaneous clearance of acute hepatitis C virus and jaundice. Gastroenterology. 2010;139:1586–1592. doi: 10.1053/j.gastro.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 43.Grebely J, Petoumenos K, Hellard M, et al. Potential role for interleukin-28B genotype in treatment decision-making in recent hepatitis C virus infection. Hepatology. 2010;52:1216–1224. doi: 10.1002/hep.23850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gale M, Jr, Foy EM. Evasion of intracellular host defence by hepatitis C virus. Nature. 2005;436:939–945. doi: 10.1038/nature04078. [DOI] [PubMed] [Google Scholar]
- 45.Thimme R, Oldach D, Chang KM, et al. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J Exp Med. 2001;194:1395–1406. doi: 10.1084/jem.194.10.1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cramp ME, Rossol S, Chokshi S, et al. Hepatitis C virus-specific T-cell reactivity during interferon and ribavirin treatment in chronic hepatitis C. Gastroenterology. 2000;118:346–355. doi: 10.1016/s0016-5085(00)70217-4. [DOI] [PubMed] [Google Scholar]
- 47.Kamal SM, Fehr J, Roesler B, et al. Peginterferon alone or with ribavirin enhances HCV-specific CD4 T-helper 1 responses in patients with chronic hepatitis C. Gastroenterology. 2002;123:1070–1083. doi: 10.1053/gast.2002.36045. [DOI] [PubMed] [Google Scholar]
- 48.Barnes E, Harcourt G, Brown D, et al. The dynamics of T-lymphocyte responses during combination therapy for chronic hepatitis C virus infection. Hepatology. 2002;36:743–754. doi: 10.1053/jhep.2002.35344. [DOI] [PubMed] [Google Scholar]
- 49.Zhang L, Jilg N, Shao RX, et al. IL28B inhibits hepatitis C virus replication through the JAK-STAT pathway. J Hepatol. Epub ahead of print. [DOI] [PMC free article] [PubMed]
- 50.Urban TJ, Thompson AJ, Bradrick SS, et al. IL28B genotype is associated with differential expression of intrahepatic interferon-stimulated genes in patients with chronic hepatitis C. Hepatology. 2010;52:1888–1896. doi: 10.1002/hep.23912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ramos EL. Preclinical and clinical development of pegylated interferon-lambda 1 in chronic hepatitis C. J Interferon Cytokine Res. 2010;30:591–595. doi: 10.1089/jir.2010.0066. [DOI] [PubMed] [Google Scholar]