Abstract
Advancing care has markedly improved survival after kidney transplantation, leaving patients susceptible to the effects of chronic transplant-associated morbidities. New-onset diabetes mellitus (NODM) is common in kidney recipients, threatening health and longevity by predisposing to microvascular and cardiovascular disease and by reducing graft survival. A strong rationale therefore exists for the aggressive treatment of NODM in kidney recipients to limit these complications. Screening for diabetes should be systematic and should span the pre- and posttransplantation periods. Once NODM is diagnosed in the kidney transplant patient, a comprehensive plan of therapy should be used to achieve treatment targets. As in the general population, treatment includes lifestyle modification and drug therapy as needed, but transplant-specific factors add complexity to the care of kidney recipients. Among these, minimizing immunosuppression-related toxicity without compromising graft outcomes is of paramount importance. Preexisting allograft functional impairment and the potential for significant interactions with immunosuppressive agents mandate that the expanding armamentarium of hypoglycemic agents be used with care. A team-oriented treatment approach that capitalizes on the collective expertise of transplant physicians, diabetologists, nurse-educators, and dieticians will optimize both glycemic control and the overall health of hyperglycemic kidney recipients.
Immunosuppression advances have dramatically improved kidney transplant outcomes (1). Chronic transplant-associated morbidities have consequently assumed greater importance in determining posttransplantation life expectancy and quality of life. We previously proposed the term “transplant-associated hyperglycemia” (TAH) to encompass abnormal glucose homeostasis developing for the first time after transplantation, including new-onset diabetes mellitus (NODM), impaired fasting glucose (IFG), and impaired glucose tolerance (IGT; Table 1). TAH is a common comorbidity that has been associated with graft failure and cardiovascular risk in kidney recipients (2,3). Furthermore, diabetes-related microvascular complications may develop within the lifetime of recipients who have NODM. Effective management of hyperglycemia is therefore fundamental to optimizing posttransplantation care. We and others (4,5) have reviewed the state of knowledge regarding the epidemiology, pathogenesis, and health implications of TAH. This review more specifically focuses on current approaches to hyperglycemia detection and the use of expanding therapeutic options for achieving NODM treatment goals in kidney recipients.
Table 1.
Definitions of hyperglycemia in kidney recipientsa
| Blood Glucose Measurement | Terminology |
|---|---|
| FPG (mg/dl) | Normal |
| <100 | IFG |
| 100 to 125 | NODM |
| >126 | |
| 2-h plasma glucose after OGTT (mg/dl) | |
| <140 | Normal |
| 140 to 199 | IGT |
| >200 | NODM |
Adapted from American Diabetes Association criteria. FPG, fasting plasma glucose; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; NODM, new-onset diabetes mellitus; OGTT, oral glucose tolerance test.
Rationale for Treating NODM
Epidemiologic (6,7) and general population analyses have established that diabetes promotes cardiovascular disease and microvascular complications, including retinopathy and neuropathy (8–10). In the absence of contradictory evidence, it is presumed that these risks are shared by kidney recipients with NODM. This is underscored by transplant recipient studies associating NODM with inferior graft and patient survival (3,11,12), largely attributable to cardiovascular and infectious complications (2,3). Although controlled intervention trials are lacking in kidney recipients, general population studies demonstrate that aggressive treatment of hyperglycemia reduces diabetic microvascular complications (8–10,13) and may decrease cardiovascular events (14). These data collectively validate the notion that treatment of NODM in kidney recipients is an important goal.
Pathogenesis of TAH
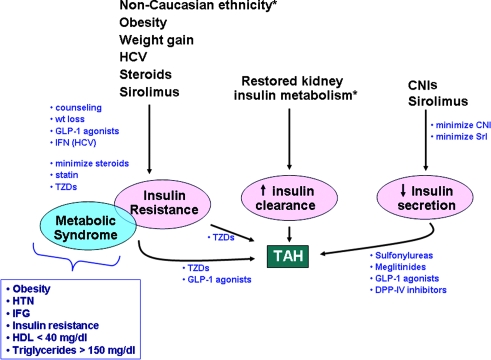
A detailed understanding of TAH pathogenesis underlies effective intervention and has been extensively reviewed (4,5). Briefly, hyperglycemia results from imbalance between insulin production and target tissue insulin demand (15). In the transplant setting, this disparity may occur as a result of insulin resistance, increased insulin metabolism, or diminished insulin secretion (Figure 1). Preexisting TAH risk factors that antedate the transplant may be nonmodifiable (e.g., advancing age, male gender, nonwhite ethnicity) or modifiable (e.g., obesity, hepatitis C virus [HCV] infection) (3,15,16). These characteristics reflect either inherited or acquired defects in insulin sensitivity and β cell function that contribute to hyperglycemia (Table 2). In addition, whereas normal kidneys contribute significantly to insulin degradation, decreased insulin requirements are observed as kidney disease progresses (17); restoration of insulin metabolism is unmasked by a functioning allograft and is likely an important, nonmodifiable factor that increases posttransplantation insulin requirements. TAH is most prevalent in the first few posttransplantation months (17), reflecting superimposition of transplant-specific factors (e.g., weight gain, diabetogenic immunosuppressive therapies) on the baseline metabolic milieu of predisposed individuals (3,18). These transplant-specific factors, as well as the modifiable preexisting risk factors, are potential targets for treatment or prevention of hyperglycemia (4), complementing pharmacologic approaches to increasing insulin delivery and decreasing demand.
Figure 1.
Illustration of the pathogenic mechanisms that lead to transplant-associated hyperglycemia (TAH) in kidney recipients. Bulleted blue text represents potential therapeutic interventions depicted at sites where their effects may be optimally mediated. *Nonmodifiable risk factors for TAH. CNIs, calcineurin inhibitors; DPP-IV, dipeptidyl peptidase-IV; GLP-1, glucagon-like peptide-1; HCV, hepatitis C virus; HTN, hypertension; IFG, impaired fasting glucose; Srl, sirolimus; TZDs, thiazolidinediones.
Table 2.
Risk factors for TAHa
| Preexisting |
Transplant Specific |
|---|---|
| Nonmodifiable | |
| age | restored graft insulin |
| gender | metabolism |
| race/ethnicity | |
| family history | |
| Potentially modifiable | ||
| obesity | weight gain | |
| physical inactivity | glucocorticoids | |
| hepatitis C | CNIs | |
| sirolimus | ||
CNIs, calcineurin inhibitors; TAH, transplant-associated hyperglycemia.
Metabolic Syndrome and TAH
The metabolic syndrome comprises a constellation of modifiable cardiovascular risk factors that cluster together sharing systemic insulin resistance as a root cause (19). Metabolic syndrome predisposes to systemic inflammation and cardiovascular disease (20,21), and its components (hyperglycemia, hypertension, dyslipidemia, and central obesity) are risk factors for progressive kidney disease (22–25).
After kidney transplantation, metabolic syndrome prevalence increases in conjunction with weight gain (26,27) and together with obesity has been associated with NODM, as well as inferior patient and graft outcomes (28,29). We therefore recommend that screening for diabetes and the metabolic syndrome be conducted in concert. Moreover, the pivotal pathogenic role of insulin resistance in metabolic syndrome should guide lifestyle intervention and promote investigation of pharmacotherapies that enhance insulin sensitivity and weight loss in transplant recipients.
Detection and Management of Diabetes in the Transplant Setting
Epidemiologic studies indicate that approximately 6% of candidates awaiting kidney transplantation develop diabetes annually (18). This prevalence increases two- to three-fold in the first posttransplantation year and returns to baseline rate thereafter (3,18). A recent prospective study (30) that investigated TAH in kidney recipients demonstrated that almost 30% of patients developed NODM or IFG within the first 6 mo after transplantation. This high risk for early posttransplantation hyperglycemia mandates vigilant screening during this period; however, cumulatively, the occurrence of diabetes is great even beyond this window. This knowledge obligates a methodical approach to diabetes screening and management that begins with a patient’s initial pretransplantation visit, continuing through the immediate postoperative period into the long term (Table 3).
Table 3.
Screening and treatment of TAHa
| Parameter | Before Transplantation | Perioperatively | After Discharge |
|---|---|---|---|
| Screening | 75 g OGTT every 1 to 2 yr | Multiple daily blood glucose assessments | Weekly FPG for first month after transplantation |
| Test for metabolic syndrome components every year | FPG every 4 wk thereafter until 6 mo after transplantation | ||
| HCV testing | FPG every 8 wk between 6 and 12 mo after transplantation | ||
| FPG at least annually after the first posttransplantation year | |||
| 75 g OGTT at 3 to 6 mo | |||
| Annual OGTT after first year | |||
| Treatmentb | Limit intravenous/oral glucose intake | Medical nutrition therapy | |
| Daily moderate exercise | |||
| Insulin | Weight loss | ||
| Consider meglitinides | Minimize CNI/steroids | ||
| Insulin/oral hypoglycemic agents as needed | |||
| TZDs | |||
| meglitinides | |||
| exenatide | |||
| Lipid management |
TZD, thiazolidinediones.
Glycemic goals are those of American Diabetes Association (35): FPG 90 to 130 mg/dl; 2-h postprandial glucose <180 mg/dl; glycosylated hemoglobin (HbA1c) < 7%.
Pretransplantation Screening
As detailed elsewhere (16), diabetes screening comprising risk factor assessment and measurement of fasting plasma glucose (FPG) and glucose tolerance should commence during pretransplantation evaluation. A thorough medical and family history to assess NODM risk is required and may influence decisions regarding when and how to perform screening tests. FPG should be tested at initial evaluation and regularly thereafter. We concur with recommendations that a 2-h oral glucose tolerance test (OGTT) be performed for patients who are on the waiting list and have normal FPG levels (16), for several reasons: (1) to detect individuals with isolated postprandial hyperglycemia, (2) to identify otherwise normoglycemic patients who have preexisting diabetes that is masked by diminished insulin metabolism associated with kidney dysfunction, and (3) higher 2-h glucose values are predictive of posttransplantation NODM (31) and may also reflect cardiovascular risk more accurately than FPG measurements (32). Although optimal timing and frequency of pretransplantation OGTT testing is undefined, the annual 6% diabetes incidence suggests that testing every 1 to 2 yr is acceptable. Positive screening tests should be confirmed, and patients with diabetes should be treated. IFG and IGT are prediabetic states, and their identification provides an opportunity to counsel patients and to initiate appropriate lifestyle changes (33).
Finally, screening and management of two additional risk factors for NODM in kidney recipients should be commenced during pretransplantation evaluation. First, because metabolic syndrome is common in patients with chronic kidney disease (34), screening for its components should be performed (19) to permit intervention as appropriate. Second, for HCV-infected candidates, the pretransplantation setting provides an opportunity to attempt viral clearance with interferon therapy. Successful elimination of HCV before transplantation may reduce posttransplantation NODM risk (35,36).
Immediate Posttransplantation Setting
Hyperglycemia is common immediately after transplantation, typically related to surgical stress and high glucocorticoid dosing (2). This frequently resolves as glucocorticoids are tapered, but persistent hyperglycemia 1 wk after transplantation is predictive of long-term glucose dysregulation (2). Handheld glucose meter monitoring should be performed multiple times daily to survey fasting and 2-h glucose concentrations until glucocorticoid dosages are <20 mg/d prednisone or equivalent and recipients have demonstrated euglycemia while eating a full diet. American Diabetes Association glycemic targets for hospitalized noncritical patients include premeal values of 90 to 130 mg/dl and postprandial values <180 mg/dl, based on association of hyperglycemia beyond this range with increased nosocomial infection rates (37).
The initial treatment approach (Table 3) involves ensuring that the diet is diabetic-appropriate. Avoidance of unnecessary infusion of glucose-containing fluids is recommended, although this has to be balanced against the risk of administering alternative solutions such as normal saline that may exacerbate hypertension and volume overload. A full, solid diet should be limited to 130 to 180 g of carbohydrate and 1800 to 2000 total kcal/d for most (38), and concentrated sweets should be eliminated. Despite the plethora of agents now available for treating hyperglycemia, exogenous insulin remains the treatment mainstay immediately after transplantation. If very high insulin requirements are apparent, then we have a low threshold for starting an insulin infusion because it may limit postoperative morbidity and because timely glycemic control can be difficult to achieve with subcutaneous insulin (37,39,40). Otherwise, an insulin “sliding scale” can be used whereby rapid-acting insulin is dosed every 6 h according to the glucose level. Total insulin requirements estimated from the cumulative 24-h insulin dosage required can be subsequently administered in a standing regimen divided between rapid-acting insulin given with meals and long-acting basal insulin. Typically, one third to one half of total is given as basal insulin, but decisions are individualized taking into account the dynamic changes in glucose homeostasis that occur early after transplantation. If 24-h insulin requirements are <15 to 20 U, then conversion to an insulin secretagogue (sulfonylurea or meglitinide) can be considered. Nonsecretagogue oral hypoglycemic agents are rarely started at this early time point. Education regarding diet, glucose monitoring, and insulin administration should be initiated as early as possible if postdischarge hyperglycemia seems likely. Because optimal posttransplantation glycemic control is often complex and long-term management frequently required, collaboration with the diabetology service is strongly advisable.
Long-Term Posttransplantation Detection and Management
Periodic diabetes screening is a routine facet of posttransplantation care for patients who are not hyperglycemic at discharge, although the intensity of surveillance declines with time (Table 3). International consensus guidelines have proposed screening patients weekly for the first month after transplantation, then every 3 mo thereafter (16). In light of the high risk for NODM within the first 3 to 6 mo after transplantation (2,3,18,30), we recommend more aggressive screening: FPG measurement weekly for the first posttransplantation month, at least monthly until month 6, at least bimonthly between months 6 to 12, and annually thereafter. We again favor routine use of the OGTT after transplantation. This recommendation is supported by a recent observational study in which OGTT was found to be more sensitive than FPG for diagnosing either NODM or IGT (41). Initial OGTT testing should be performed at 3 to 6 mo after transplantation and annually thereafter. If NODM is suggested and confirmed by repeat testing, then treatment should be started to target American Diabetes Association–defined goals for diabetes: FPG 90 to 130 mg/dl, 2-h postprandial glucose <180 mg/dl, and glycosylated hemoglobin (HbA1c) <7% (37). Long-term NODM management is multidimensional and may involve (1) lifestyle intervention, (2) immunosuppression modification, and (3) effective use of diabetes therapies.
Lifestyle Intervention.
Medical nutrition therapy describes the overall nutritional approach to preventing and managing diabetes and has been recently reviewed (38). Two medical nutrition therapy goals that contribute specifically to glycemic management are (1) to moderate overall carbohydrate intake, thereby lowering dietary glycemic burden, and (2) to increase insulin sensitivity by promoting moderate weight loss in overweight individuals (38). Studies have demonstrated a 1 to 2% decrease in HbA1c with medical nutrition therapy implementation (42,43).
The principle dietary carbohydrate modification objective is to reduce overall carbohydrate intake to 130 to 180 g/d. A secondary goal is the favoring of carbohydrates with a lower glycemic index that may further facilitate glucose control (44,45). Calorie reduction is recommended in overweight (body mass index 25 to ≤30 kg/m2) or obese (body mass index ≥30 kg/m2) patients to promote weight loss. Subsequent weight maintenance is challenging, and many patients return to baseline weight. Thirty minutes of daily low-intensity exercise, such as walking, facilitates weight loss and promotes insulin sensitivity and should be specifically prescribed as a concrete intervention (33,38). Medical nutrition therapy education is specialized and time consuming, and referral to an experienced dietician is recommended (38). We advocate medical nutrition therapy for all recipients who are at risk for NODM to promote benefit to glucose regulation, hyperlipidemia (46), and hypertension (47,48).
Immunosuppression Modification.
Several immunosuppressants, including glucocorticoids, calcineurin inhibitors (CNIs) cyclosporine and tacrolimus, and sirolimus are potentially diabetogenic (reviewed in reference [4]). Immunosuppression modification to prevent or mitigate NODM can take the form of minimization, withdrawal, or conversion from one drug or regimen to another, less diabetogenic one.
Glucocorticoids.
Glucocorticoids typically either are tapered rapidly to a dosage that is unlikely to affect glucose homeostasis (49) or are discontinued altogether. Emerging data suggest that although early steroid withdrawal may be safe from an immunosuppression standpoint in the short to midterm, it may not affect long-term glucose metabolism; however, final publication of clinical trials in this area is awaited.
CNIs.
CNIs, especially tacrolimus at present, form the backbone of most maintenance immunosuppressive regimens. Initial efficacy studies that compared cyclosporine and tacrolimus in kidney recipients demonstrated higher NODM risk with tacrolimus therapy (50,51). These findings were recently confirmed by the Diabetes Incidence after Renal Transplantation (DIRECT) trial, a randomized, prospective study in which the composite of NODM and IFG within the first 6 mo after transplantation was the primary safety end point analyzed (30). In that study, which used sequential OGTT, abnormal glucose homeostasis developed within 6 mo after transplant in 26% and 33.6% of cyclosporine- and tacrolimus-treated patients, respectively, with no difference in efficacy between the two drugs. NODM was more often transient among cyclosporine-treated recipients, with only 8.9% of patients requiring diabetes therapy at 6 mo after transplantation, compared with 16.8% among the tacrolimus-treated cohort. Study limitations include a lack of standardization of steroid dosage and open-label design. That the majority of the patients were white additionally limits applicability to other populations, from either an efficacy or adverse events stand point. The reduced diabetogenicity associated with cyclosporine does, however, raise the possibility that conversion to this CNI in tacrolimus-treated patients with NODM could successfully eliminate hyperglycemia. Although supported by small series and anecdotes, this approach is not yet validated in large trials. This practice has to be balanced further against the superior lipid, BP, and kidney function profile observed with tacrolimus as compared with cyclosporine (52).
Minimization and Withdrawal Regimens.
As far as minimization regimens are concerned, use of mycophenolate mofetil as an adjunctive agent spares tacrolimus exposure and may reduce the incidence of hyperglycemia (53–55). Efforts to spare CNI exposure with sirolimus, in contrast, have been associated with decreased insulin sensitivity and pancreatic β cell function, resulting in exacerbated glucose intolerance (56). Although avoidance of toxic therapies represents the holy grail of immunosuppression management, we do not advocate withdrawal of CNIs from maintenance regimens, on the basis of increased risk for graft dysfunction and immunologic injury. In summary, an optimal maintenance regimen should balance risk for rejection as well as long-term cardiovascular disease versus the potential for permanent hyperglycemia.
Antiviral Therapies.
Virus infections have been associated with diabetes in the general population (57). HCV infection is a well-established diabetes risk factor. Pretransplantation administration of interferon may successfully clear viremia and reduce the risk for development of NODM after transplantation (35,36). Similarly, a recent study suggested that asymptomatic cytomegalovirus infection may impair insulin release and provoke diabetes. The impact of either a prophylactic or preemptive antiviral approach remains to be ascertained (58).
Pharmacologic Therapies.
Drug treatment options for TAH are expanding, although safety and efficacy data for most agents are limited in transplant populations. Other than insulin, most have at least theoretical potential to interact with immunosuppressants through either shared metabolic pathways or effects on gastric motility. We therefore recommend careful monitoring of FPG and potential adverse effects in the first weeks after initiation of any noninsulin agents. Generally, combination therapy with agents from different classes is more effective than monotherapy. An exception is the sulfonylureas and meglitinides, which act through a related mechanism and are not used together (Table 4).
Table 4.
Noninsulin drug therapy for NODMa
| Class | Principle Mechanism of Action | Example | Effect on HbA1c | Adverse Effects | Metabolism/Elimination | Altered Dosing in CKD |
|---|---|---|---|---|---|---|
| Sulfonylureas | Insulin secretagogue | Glipizide Glyburide Glimepiride | −1.0 to −2.0% | Hypoglycemia | Major: Hepatic | No |
| Meglitinides | Insulin secretagogue | Repaglinide Nateglinide | −0.6% | Hypoglycemia | Major: CYP2C8 and 3A4 | CrCl >40 ml/min: No CrCl <40 ml/min: Gradual introduction |
| Biguanides | Decrease hepatic glucose production | Metformin | −1.0 to −1.7% | Nausea Lactic acidosis (extremely rare) | Major: Renal tubular secretion | Avoid with more severe levels of kidney dysfunction |
| TZD | Increase insulin sensitivity | Pioglitazone Rosiglitazone | −1.0 to −1.9% | Weight gain Fluid retention | Major: CYP2C8 Minor: CYP3A4 | No |
| α-Glucosidase inhibitors | Decrease intestinal glucose absorption | Acarbose | −0.3 to −0.6% | Nausea Flatulence | Major: Fecal Minor: Renal | Not recommended if creatinine >2 mg/dl |
| GLP-1 receptor agonists | Stimulates glucose mediated insulin secretion Inhibits glucagon Induces weight loss due to delayed gastric emptying and appetite suppression | Exenatide | −0.4 to −0.8% | Nausea, other gastrointestinal | Major: Renal | Not recommended if CrCl ≤30 ml/min |
| DPP-IV inhibitors | Increases GLP-1 | Sitagliptin | −0.5% | Minimal | Major: Renal Minor: CYP3A4, 2C8 | Dosage reduction |
CKD, chronic kidney disease; CrCl, creatinine clearance; DPP-IV, dipeptidyl peptidase-IV; GLP-1, glucagon-like peptide-1.
Insulin.
Modern insulin regimens use once-daily long-acting agents such as glargine or detemir to provide basal glycemic control and rapid-acting insulin analogues such as aspart, lispro, and glulisine to provide postprandial control (59). Newer insulins are favored over their older counterparts because of greater ease in achieving glycemic targets with less hypoglycemic potential. Adequacy of basal insulin dosing is demonstrated by FPG measurements, and prandial control is indicated by glucose excursion from just before eating to 90 to 120 min after a meal. Highly portable insulin pens facilitate administration, and some patients may be candidates for insulin pump therapy, providing even greater flexibility. Because the kidney plays an important role in insulin metabolism (60,61), perturbations in allograft function may necessitate changes in insulin dosage. Insulin is not known to interact significantly with currently used immunosuppression. Despite the complexity of its use, insulin should be initiated sooner rather than later if judgment suggests that other agents will be inadequate.
Sulfonylureas.
Sulfonylureas directly stimulate insulin secretion and lower both fasting and postprandial glucose concentrations. Second-generation sulfonylureas (e.g., glipizide, glyburide, glimepiride) have largely replaced the original agents. Glipizide is favored when kidney function is impaired, because it is almost entirely metabolized by the liver (62). Glipizide is a reasonable option for NODM if hyperglycemia is not severe and can be introduced at a low dosage and up-titrated every 3 to 4 wk as needed. Because the principal adverse effect of sulfonylureas is hypoglycemia, they should be used with caution, especially with deteriorating allograft function.
Meglitinides.
The meglitinides repaglinide and nateglinide are insulin secretagogues with a rapid onset and short duration of action. They can be taken immediately before meals and may have less hypoglycemic potential than sulfonylureas (63). Repaglinide lowers fasting and postprandial glucose and has been demonstrated to be safe and efficacious in kidney recipients with NODM (64). Our personal experience is that repaglinide is useful in treating modest postprandial hyperglycemia. Both repaglinide and nateglinide are hepatically metabolized and can be used with any level of kidney function (package inserts of Prandin [Novo Nordisk Inc., Princeton, NJ; 65] and Starlix [Novartis Pharmaceuticals, East Hanover, NJ; 66]).
Thiazolidinediones.
Thiazolidinediones (TZDs) promote peripheral insulin sensitivity and are widely used for type 2 diabetes treatment (67). TZDs may exert favorable glucose-independent effects on vascular health (68–70), and one, pioglitazone, has displayed modest efficacy in the prevention of cardiovascular events in a population with diabetes (71). The initial TZD marketed and subsequently withdrawn in the United States, troglitazone, raised concerns in the transplant population because it altered cyclosporine metabolism and rarely caused severe liver injury (72,73). The currently available TZDs rosiglitazone and pioglitazone are not associated with significant liver injury, have no apparent significant interaction with CNIs, and seem to be safe and effective for treating NODM in organ recipients (74–77).
TZDs promote fluid retention and are contraindicated in patients with heart failure or refractory edema. TZDs are metabolized via the liver, and dosing adjustment is not required for kidney allograft dysfunction (package inserts for Avandia [GlaxoSmithKline, Philadelphia, PA; 78] and Actos [Takeda Pharmaceuticals, Lincolnshire, IL; 79]). Although a recent meta-analysis suggested that rosiglitazone may promote cardiovascular events (80), inherent weaknesses in design make this study difficult to interpret (81). This finding is further not supported by a prospective clinical trial in a prediabetic population (82). For now, pioglitazone is preferred on the basis of once-daily dosing frequency, a more favorable lipid effect (83,84), and a large, published clinical trial suggesting modest potential cardiovascular benefits (71).
Agents that Promote Glucagon-Like Peptide-1 Receptor Activation.
Glucagon-like peptide-1 (GLP-1) is a natural hormone that facilitates postprandial nutrient disposal. GLP-1 stimulates pancreatic β cell insulin secretion in a glucose-dependent manner and has minimal hypoglycemic potential. GLP-1 also slows gastric emptying and suppresses appetite (85). GLP-1 is degraded by dipeptidyl peptidase-IV (DPP-IV) (86) and has a very short half-life; therefore, synthetic GLP-1 receptor agonists that are resistant to DPP-IV and small molecule DPP-IV inhibitors have been designed to promote GLP-1 receptor activation (85).
Exenatide is the only GLP-1 receptor agonist currently available for clinical use. It is used as a twice-daily premeal injection and has demonstrated the beneficial effects of native GLP-1 (87). It has been shown to blunt postprandial glucose excursions and durably lowers HbA1c in patients with diabetes (88–90). Another striking benefit of exenatide is substantial weight loss attributed to appetite suppression, averaging 4 to 5 kg during 18 mo of treatment in one trial (91,92). No reports describing use of this agent in transplant recipients has yet emerged. Anecdotally, several of our own patients have and continue to be successfully treated with it. Because exenatide is renally metabolized, it should not be used if creatinine clearance is <30 ml/min (Eli-Lilly package insert). Given the effect of exenatide on gastric emptying and weight loss, immunosuppressive dosing may require adjustment and should be carefully monitored. The most common adverse effect of exenatide is nausea, which is usually transient (88–90).
The DPP-IV inhibitor sitagliptin has recently been approved for clinical use in the United States. When taken as a once-daily pill, it effectively raises blood concentrations of endogenous active GLP-1 (93), lowers fasting and postprandial blood glucose concentrations, and decreases HbA1c by 0.6 to 0.8% when used as monotherapy (94) or when added to metformin or a TZD (Januvia package insert [Merck and Co. Inc., Whitehouse Station, NJ; 95]) (93). Advantages over exenatide include ease of administration and a milder adverse effect profile, although sitagliptin does not promote weight loss. Dosage reduction is required for kidney dysfunction, and few data are available concerning potential drug interactions. Because available clinical information is limited, we recommend careful monitoring of kidney recipients and their immunosuppression dosing requirements if sitagliptin is used. Vildagliptin, a second DPP-IV inhibitor with similar properties (96,97), may become available in the near future.
Biguanides.
Metformin is the only biguanide available in the United States. It reduces hepatic glucose production and is effective in the general type 2 diabetes population for both control of hyperglycemia and weight loss or stabilization (10,33). Metformin is largely cleared by renal tubular secretion, and exposure increases in the setting of renal insufficiency (Metformin package insert). Because metformin has extremely rarely been associated with lactic acidosis (98,99), we recommend avoiding its use in kidney recipients with more severe degrees of kidney dysfunction.
Conclusions
Optimizing glycemic control in kidney recipients with NODM is an important goal in long-term posttransplantation care. General population studies provide strong evidence that improved glucose regulation decreases the risk for microvascular disease and, potentially, cardiovascular disease. The demonstrated association between NODM and adverse recipient outcomes suggests that treatment of hyperglycemia may enhance patient and graft survival; therefore, screening for glucose dysregulation should be systematic and should be implemented in all phases of the transplant process, commencing at evaluation. Efforts should be made to diminish the impact of transplantation on glucose homeostasis by limiting posttransplantation weight gain and minimizing exposure to diabetogenic immunosuppression while simultaneously preserving efficacy. Lifestyle intervention should promote dietary carbohydrate moderation and exercise in all recipients and weight loss in those who are overweight. An expanding battery of pharmacologic agents can be used to reach glycemic targets. Many of these agents require dosage adjustment for kidney dysfunction, and several have potential to alter immunosuppressant pharmacokinetics, underscoring the need for caution. Finally, NODM is often present as a component of the metabolic syndrome, and metabolic syndrome screening and treatment are an integral part of NODM care. A multifaceted and team-oriented approach to treatment that capitalizes on the skills of physicians, nurse-educators, and nutritionists will have the greatest chance of successfully and safely diagnosing and treating NODM, thereby optimizing the health of affected recipients.
Disclosures
R.D.B. receives grant support from Novartis and Roche and has consulted for Novartis, Roche, Merck, and CSL Behring.
Acknowledgments
Preparation of this manuscript was supported by a National Institutes of Health K12 (RR017625-03) Career Development Award (to M.F.C.).
References
- 1.Cohen DJ, St Martin L, Christensen LL, Bloom RD, Sung RS: Kidney and pancreas transplantation in the United States, 1995–2004. Am J Transplant 6 :1153 –1169,2006 [DOI] [PubMed] [Google Scholar]
- 2.Cosio FG, Kudva Y, van der Velde M, Larson TS, Textor SC, Griffin MD, Stegall MD: New onset hyperglycemia and diabetes are associated with increased cardiovascular risk after kidney transplantation. Kidney Int 67 :2415 –2421,2005 [DOI] [PubMed] [Google Scholar]
- 3.Kasiske BL, Snyder JJ, Gilbertson D, Matas AJ: Diabetes mellitus after kidney transplantation in the United States. Am J Transplant 3 :178 –185,2003 [DOI] [PubMed] [Google Scholar]
- 4.Crutchlow MF, Bloom RD: Transplant-associated hyperglycemia: A new look at an old problem. Clin J Am Soc Nephrol 2 :343 –355,2007 [DOI] [PubMed] [Google Scholar]
- 5.Davidson J, Wilkinson A, Dantal J, Dotta F, Haller H, Hernández D, Kasiske BL, Kiberd B, Krentz A, Legendre C, Marchetti P, Markell M, van der Woude FJ, Wheeler DC, International Expert Panel: New-onset diabetes after transplantation: 2003 International consensus guidelines. Proceedings of an international expert panel meeting. Barcelona, Spain, 19 February 2003. Transplantation 75[Suppl] :SS3 –SS24,2003 [DOI] [PubMed] [Google Scholar]
- 6.Charles MA, Fontbonne A, Thibult N, Warnet JM, Rosselin GE, Eschwege E: Risk factors for NIDDM in white population. Paris prospective study. Diabetes 40 :796 –799,1991 [DOI] [PubMed] [Google Scholar]
- 7.McCance DR, Hanson RL, Charles MA, Jacobsson LT, Pettitt DJ, Bennett PH, Knowler WC: Comparison of tests for glycated haemoglobin and fasting and two hour plasma glucose concentrations as diagnostic methods for diabetes. BMJ 308 :1323 –1328,1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.The Diabetes Control and Complications Trial Research Group: The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329 :977 –986,1993 [DOI] [PubMed] [Google Scholar]
- 9.The UK Prospective Diabetes Study (UKPDS) Group: Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352 :837 –853,1998 [PubMed] [Google Scholar]
- 10.The UK Prospective Diabetes Study (UKPDS) Group: Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 352 :854 –865,1998 [PubMed] [Google Scholar]
- 11.Cosio FG, Pesavento TE, Kim S, Osei K, Henry M, Ferguson RM: Patient survival after renal transplantation: IV. Impact of post-transplant diabetes. Kidney Int 62 :1440 –1446,2002 [DOI] [PubMed] [Google Scholar]
- 12.Revanur VK, Jardine AG, Kingsmore DB, Jaques BC, Hamilton DH, Jindal RM: Influence of diabetes mellitus on patient and graft survival in recipients of kidney transplantation. Clin Transplant 15 :89 –94,2001 [DOI] [PubMed] [Google Scholar]
- 13.The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group: Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. N Engl J Med 342 :381 –389,2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, Raskin P, Zinman B; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group: Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 353 :2643 –2653,2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Porte D Jr: Clinical importance of insulin secretion and its interaction with insulin resistance in the treatment of type 2 diabetes mellitus and its complications. Diabetes Metab Res Rev 17 :181 –188,2001 [DOI] [PubMed] [Google Scholar]
- 16.Wilkinson A, Davidson J, Dotta F, Home PD, Keown P, Kiberd B, Jardine A, Levitt N, Marchetti P, Markell M, Naicker S, O’Connell P, Schnitzler M, Standl E, Torregosa JV, Uchida K, Valantine H, Villamil F, Vincenti F, Wissing M: Guidelines for the treatment and management of new-onset diabetes after transplantation. Clin Transplant 19 :291 –298,2005 [DOI] [PubMed] [Google Scholar]
- 17.Snyder RW, Berns JS: Use of insulin and oral hypoglycemic medications in patients with diabetes mellitus and advanced kidney disease. Semin Dial 17 :365 –370,2004 [DOI] [PubMed] [Google Scholar]
- 18.Woodward RS, Schnitzler MA, Baty J, Lowell JA, Lopez-Rocafort L, Haider S, Woodworth TG, Brennan DC: Incidence and cost of new onset diabetes mellitus among U.S. wait-listed and transplanted renal allograft recipients. Am J Transplant 3 :590 –598,2003 [DOI] [PubMed] [Google Scholar]
- 19.Executive Summary of the Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 285 :2486 –2497,2001 [DOI] [PubMed] [Google Scholar]
- 20.Lakka HM, Laaksonen DE, Lakka TA, Niskanen LK, Kumpusalo E, Tuomilehto J, Salonen JT: The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 288 :2709 –2716,2002 [DOI] [PubMed] [Google Scholar]
- 21.Isomaa B, Almgren P, Tuomi T, Forsén B, Lahti K, Nissén M, Taskinen MR, Groop L: Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 24 :683 –689,2001 [DOI] [PubMed] [Google Scholar]
- 22.Whelton PK, Perneger TV, He J, Klag MJ: The role of blood pressure as a risk factor for renal disease: A review of the epidemiologic evidence. J Hum Hypertens 10 :683 –689,1996 [PubMed] [Google Scholar]
- 23.Humphrey LL, Ballard DJ, Frohnert PP, Chu CP, O’Fallon WM, Palumbo PJ: Chronic renal failure in non-insulin-dependent diabetes mellitus: A population-based study in Rochester, Minnesota. Ann Intern Med 111 :788 –796,1989 [DOI] [PubMed] [Google Scholar]
- 24.Chen J, Muntner P, Hamm LL, Jones DW, Batuman V, Fonseca V, Whelton PK, He J: The metabolic syndrome and chronic kidney disease in U.S. adults. Ann Intern Med 140 :167 –174,2004 [DOI] [PubMed] [Google Scholar]
- 25.Palaniappan L, Carnethon M, Fortmann SP: Association between microalbuminuria and the metabolic syndrome: NHANES III. Am J Hypertens 16 :952 –958,2003 [DOI] [PubMed] [Google Scholar]
- 26.Ducloux D, Kazory A, Simula-Faivre D, Chalopin JM: One-year post-transplant weight gain is a risk factor for graft loss. Am J Transplant 5 :2922 –2928,2005 [DOI] [PubMed] [Google Scholar]
- 27.Armstrong KA, Campbell SB, Hawley CM, Nicol DL, Johnson DW, Isbel NM: Obesity is associated with worsening cardiovascular risk factor profiles and proteinuria progression in renal transplant recipients. Am J Transplant 5 :2710 –2718,2005 [DOI] [PubMed] [Google Scholar]
- 28.de Vries AP, Bakker SJ, van Son WJ, van der Heide JJ, Ploeg RJ, The HT, de Jong PE, Gans RO: Metabolic syndrome is associated with impaired long-term renal allograft function; not all component criteria contribute equally. Am J Transplant 4 :1675 –1683,2004 [DOI] [PubMed] [Google Scholar]
- 29.Porrini E, Delgado P, Bigo C, Alvarez A, Cobo M, Checa MD, Hortal L, Fernández A, García JJ, Velázquez S, Hernández D, Salido E, Torres A: Impact of metabolic syndrome on graft function and survival after cadaveric renal transplantation. Am J Kidney Dis 48 :134 –142,2006 [DOI] [PubMed] [Google Scholar]
- 30.Vincenti F, Friman S, Scheuermann E, Rostaing L, Jenssen T, Campistol JM, Uchida K, Pescovitz MD, Marchetti P, Tuncer M, Citterio F, Wiecek A, Chadban S, El-Shahawy M, Budde K, Goto N, DIRECT (Diabetes Incidence after Renal Transplantation: Neoral C Monitoring Versus Tacrolimus) Investigators: Results of an international, randomized trial comparing glucose metabolism disorders and outcome with cyclosporine versus tacrolimus. Am J Transplant 7 :1506 –1514,2007 [DOI] [PubMed] [Google Scholar]
- 31.Nam JH, Mun JI, Kim SI, Kang SW, Choi KH, Park K, Ahn CW, Cha BS, Song YD, Lim SK, Kim KR, Lee HC, Huh KB: Beta-cell dysfunction rather than insulin resistance is the main contributing factor for the development of postrenal transplantation diabetes mellitus. Transplantation 71 :1417 –1423,2001 [DOI] [PubMed] [Google Scholar]
- 32.The DECODE study group: Glucose tolerance and mortality: Comparison of WHO and American Diabetes Association diagnostic criteria. Diabetes Epidemiology: Collaborative analysis Of Diagnostic criteria in Europe. Lancet 354 :617 –621,1999 [PubMed] [Google Scholar]
- 33.American Diabetes Association: Standards of medical care in diabetes—2007. Diabetes Care 30[Suppl 1] :S4 –S41,2007 [DOI] [PubMed] [Google Scholar]
- 34.Tsangalis G, Papaconstantinou S, Kosmadakis G, Valis D, Zerefos N: Prevalence of the metabolic syndrome in hemodialysis. Int J Artif Organs 30 :118 –123,2007 [DOI] [PubMed] [Google Scholar]
- 35.Gursoy M, Köksal R, Karavelioğlu D, Colak T, Gür G, Ozdemir N, Boyacioğlu S, Bilgin N: Pretransplantation alpha-interferon therapy and the effect of hepatitis C virus infection on kidney allograft recipients. Transplant Proc 32 :580 –582,2000 [DOI] [PubMed] [Google Scholar]
- 36.Kamar N, Toupance O, Buchler M, Sandres-Saune K, Izopet J, Durand D, Rostaing L: Evidence that clearance of hepatitis C virus RNA after alpha-interferon therapy in dialysis patients is sustained after renal transplantation. J Am Soc Nephrol 14 :2092 –2098,2003 [DOI] [PubMed] [Google Scholar]
- 37.American Diabetes Association: Standards of medical care in diabetes—2006. Diabetes Care 29[Suppl 1] :S4 –S42,2006 [PubMed] [Google Scholar]
- 38.Bantle JP, Wylie-Rosett J, Albright AL, Apovian CM, Clark NG, Franz MJ, Hoogwerf BJ, Lichtenstein AH, Mayer-Davis E, Mooradian AD, Wheeler ML: Nutrition recommendations and interventions for diabetes—2006: A position statement of the American Diabetes Association. Diabetes Care 29 :2140 –2157,2006 [DOI] [PubMed] [Google Scholar]
- 39.Van den Berghe G, Wilmer A, Milants I, Wouters PJ, Bouckaert B, Bruyninckx F, Bouillon R, Schetz M: Intensive insulin therapy in mixed medical/surgical intensive care units: Benefit versus harm. Diabetes 55 :3151 –3159,2006 [DOI] [PubMed] [Google Scholar]
- 40.van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R: Intensive insulin therapy in the critically ill patients. N Engl J Med 345 :1359 –1367,2001 [DOI] [PubMed] [Google Scholar]
- 41.Armstrong KA, Prins JB, Beller EM, Campbell SB, Hawley CM, Johnson DW, Isbel NM: Should an oral glucose tolerance test be performed routinely in all renal transplant recipients? Clin J Am Soc Nephrol 1 :100 –108,2006 [DOI] [PubMed] [Google Scholar]
- 42.Pastors JG, Franz MJ, Warshaw H, Daly A, Arnold MS: How effective is medical nutrition therapy in diabetes care? J Am Diet Assoc 103 :827 –831,2003 [DOI] [PubMed] [Google Scholar]
- 43.Pastors JG, Warshaw H, Daly A, Franz M, Kulkarni K: The evidence for the effectiveness of medical nutrition therapy in diabetes management. Diabetes Care 25 :608 –613,2002 [DOI] [PubMed] [Google Scholar]
- 44.Brand-Miller J, Hayne S, Petocz P, Colagiuri S: Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 26 :2261 –2267,2003 [DOI] [PubMed] [Google Scholar]
- 45.Sheard NF, Clark NG, Brand-Miller JC, Franz MJ, Pi-Sunyer FX, Mayer-Davis E, Kulkarni K, Geil P: Dietary carbohydrate (amount and type) in the prevention and management of diabetes: A statement by the American Diabetes Association. Diabetes Care 27 :2266 –2271,2004 [DOI] [PubMed] [Google Scholar]
- 46.Yu-Poth S, Zhao G, Etherton T, Naglak M, Jonnalagadda S, Kris-Etherton PM: Effects of the National Cholesterol Education Program’s Step I and Step II dietary intervention programs on cardiovascular disease risk factors: A meta-analysis. Am J Clin Nutr 69 :632 –646,1999 [DOI] [PubMed] [Google Scholar]
- 47.Whitworth JA, Chalmers J: World Health Organisation-International Society Of Hypertension (WHO/ISH) hypertension guidelines. Clin Exp Hypertens 26 :747 –752,2004 [DOI] [PubMed] [Google Scholar]
- 48.Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, Jones DW, Materson BJ, Oparil S, Wright JT Jr, Roccella EJ, National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; National High Blood Pressure Education Program Coordinating Committee: The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: The JNC 7 report. JAMA 289 :2560 –2572,2003 [DOI] [PubMed] [Google Scholar]
- 49.Midtvedt K, Hjelmesaeth J, Hartmann A, Lund K, Paulsen D, Egeland T, Jenssen T: Insulin resistance after renal transplantation: The effect of steroid dose reduction and withdrawal. J Am Soc Nephrol 15 :3233 –3239,2004 [DOI] [PubMed] [Google Scholar]
- 50.Mayer AD, Dmitrewski J, Squifflet JP, Besse T, Grabensee B, Klein B, Eigler FW, Heemann U, Pichlmayr R, Behrend M, Vanrenterghem Y, Donck J, van Hooff J, Christiaans M, Morales JM, Andres A, Johnson RW, Short C, Buchholz B, Rehmert N, Land W, Schleibner S, Forsythe JL, Talbot D, Pohanka E, et al.: Multicenter randomized trial comparing tacrolimus (FK506) and cyclosporine in the prevention of renal allograft rejection: A report of the European Tacrolimus Multicenter Renal Study Group. Transplantation 64 :436 –443,1997 [DOI] [PubMed] [Google Scholar]
- 51.Pirsch JD, Miller J, Deierhoi MH, Vincenti F, Filo RS: A comparison of tacrolimus (FK506) and cyclosporine for immunosuppression after cadaveric renal transplantation. FK506 Kidney Transplant Study Group. Transplantation 63 :977 –983,1997 [DOI] [PubMed] [Google Scholar]
- 52.Artz MA, Boots JM, Ligtenberg G, Roodnat JI, Christiaans MH, Vos PF, Blom HJ, Sweep FC, Demacker PN, Hilbrands LB: Improved cardiovascular risk profile and renal function in renal transplant patients after randomized conversion from cyclosporine to tacrolimus. J Am Soc Nephrol 14 :1880 –1888,2003 [DOI] [PubMed] [Google Scholar]
- 53.Bloom RD, Rao V, Weng F, Grossman RA, Cohen D, Mange KC: Association of hepatitis C with posttransplant diabetes in renal transplant patients on tacrolimus. J Am Soc Nephrol 13 :1374 –1380,2002 [DOI] [PubMed] [Google Scholar]
- 54.Miller J, Mendez R, Pirsch JD, Jensik SC: Safety and efficacy of tacrolimus in combination with mycophenolate mofetil (MMF) in cadaveric renal transplant recipients. FK506/MMF Dose-Ranging Kidney Transplant Study Group. Transplantation 69 :875 –880,2000 [DOI] [PubMed] [Google Scholar]
- 55.Johnson C, Ahsan N, Gonwa T, Halloran P, Stegall M, Hardy M, Metzger R, Shield C 3rd, Rocher L, Scandling J, Sorensen J, Mulloy L, Light J, Corwin C, Danovitch G, Wachs M, van Veldhuisen P, Salm K, Tolzman D, Fitzsimmons WE: Randomized trial of tacrolimus (Prograf) in combination with azathioprine or mycophenolate mofetil versus cyclosporine (Neoral) with mycophenolate mofetil after cadaveric kidney transplantation. Transplantation 69 :834 –841,2000 [DOI] [PubMed] [Google Scholar]
- 56.Teutonico A, Schena PF, Di Paolo S: Glucose metabolism in renal transplant recipients: Effect of calcineurin inhibitor withdrawal and conversion to sirolimus. J Am Soc Nephrol 16 :3128 –3135,2005 [DOI] [PubMed] [Google Scholar]
- 57.Jaeckel E, Manns M, Von Herrath M: Viruses and diabetes. Ann N Y Acad Sci 958 :7 –25,2002 [DOI] [PubMed] [Google Scholar]
- 58.Hjelmesaeth J, Sagedal S, Hartmann A, Rollag H, Egeland T, Hagen M, Nordal KP, Jenssen T: Asymptomatic cytomegalovirus infection is associated with increased risk of new-onset diabetes mellitus and impaired insulin release after renal transplantation. Diabetologia 47 :1550 –1556,2004 [DOI] [PubMed] [Google Scholar]
- 59.Hirsch IB: Insulin analogues. N Engl J Med 352 :174 –183,2005 [DOI] [PubMed] [Google Scholar]
- 60.Rubenstein AH, Mako ME, Horwitz DL: Insulin and the kidney. Nephron 15 :306 –326,1975 [DOI] [PubMed] [Google Scholar]
- 61.Rabkin R, Ryan MP, Duckworth WC: The renal metabolism of insulin. Diabetologia 27 :351 –357,1984 [DOI] [PubMed] [Google Scholar]
- 62.Harrower AD: Pharmacokinetics of oral antihyperglycaemic agents in patients with renal insufficiency. Clin Pharmacokinet 31 :111 –119,1996 [DOI] [PubMed] [Google Scholar]
- 63.Holstein A, Egberts EH: Risk of hypoglycaemia with oral antidiabetic agents in patients with type 2 diabetes. Exp Clin Endocrinol Diabetes 111 :405 –414,2003 [DOI] [PubMed] [Google Scholar]
- 64.Turk T, Pietruck F, Dolff S, Kribben A, Janssen OE, Mann K, Philipp T, Heemann U, Witzke O: Repaglinide in the management of new-onset diabetes mellitus after renal transplantation. Am J Transplant 6 :842 –846,2006 [DOI] [PubMed] [Google Scholar]
- 65.Prandin [package insert], Princeton, NJ, Novo Nordisk Pharmaceuticals Inc.,2002
- 66.Starlix [package insert], East Hanover, NJ, Novartis Pharmaceuticals,2004
- 67.Lehrke M, Lazar MA: The many faces of PPARgamma. Cell 123 :993 –999,2005 [DOI] [PubMed] [Google Scholar]
- 68.Paradisi G, Steinberg HO, Shepard MK, Hook G, Baron AD: Troglitazone therapy improves endothelial function to near normal levels in women with polycystic ovary syndrome. J Clin Endocrinol Metab 88 :576 –580,2003 [DOI] [PubMed] [Google Scholar]
- 69.Qayyum R, Schulman P: Cardiovascular effects of the thiazolidinediones. Diabetes Metab Res Rev 22 :88 –97,2006 [DOI] [PubMed] [Google Scholar]
- 70.Watanabe I, Tani S, Anazawa T, Kushiro T, Kanmatsuse K: Effect of pioglitazone on arteriosclerosis in comparison with that of glibenclamide. Diabetes Res Clin Pract 68 :104 –110,2005 [DOI] [PubMed] [Google Scholar]
- 71.Dormandy JA, Charbonnel B, Eckland DJ, Erdmann E, Massi-Benedetti M, Moules IK, Skene AM, Tan MH, Lefèbvre PJ, Murray GD, Standl E, Wilcox RG, Wilhelmsen L, Betteridge J, Birkeland K, Golay A, Heine RJ, Korányi L, Laakso M, Mokán M, Norkus A, Pirags V, Podar T, Scheen A, Scherbaum W, Schernthaner G, Schmitz O, Skrha J, Smith U, Taton J, PROactive investigators: Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): A randomised controlled trial. Lancet 366 :1279 –1289,2005. 16214598 [Google Scholar]
- 72.Graham DJ, Drinkard CR, Shatin D: Incidence of idiopathic acute liver failure and hospitalized liver injury in patients treated with troglitazone. Am J Gastroenterol 98 :175 –179,2003 [DOI] [PubMed] [Google Scholar]
- 73.Kaplan B, Friedman G, Jacobs M, Viscuso R, Lyman N, DeFranco P, Bonomini L, Mulgaonkar SP: Potential interaction of troglitazone and cyclosporine. Transplantation 65 :1399 –1400,1998 [DOI] [PubMed] [Google Scholar]
- 74.Villanueva G, Baldwin D: Rosiglitazone therapy of posttransplant diabetes mellitus. Transplantation 80 :1402 –1405,2005 [DOI] [PubMed] [Google Scholar]
- 75.Luther P, Baldwin D Jr: Pioglitazone in the management of diabetes mellitus after transplantation. Am J Transplant 4 :2135 –2138,2004 [DOI] [PubMed] [Google Scholar]
- 76.Baldwin D Jr, Duffin KE: Rosiglitazone treatment of diabetes mellitus after solid organ transplantation. Transplantation 77 :1009 –1014,2004 [DOI] [PubMed] [Google Scholar]
- 77.Pietruck F, Kribben A, Van TN, Patschan D, Herget-Rosenthal S, Janssen O, Mann K, Philipp T, Witzke O: Rosiglitazone is a safe and effective treatment option of new-onset diabetes mellitus after renal transplantation. Transpl Int 18 :483 –486,2005 [DOI] [PubMed] [Google Scholar]
- 78.Avandia [package insert], Philadelphia, PA, GlaxoSmith- Kline Inc.,1999
- 79.Actos [package insert], Lincolnshire, IL, Takeda Pharmaceuticals Inc.,1999
- 80.Nissen SE, Wolski K: Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 356 :2457 –2471,2007 [DOI] [PubMed] [Google Scholar]
- 81.Rosiglitazone: Seeking a balanced perspective. Lancet 369 :1834.2007 [DOI] [PubMed] [Google Scholar]
- 82.DREAM (Diabetes REduction Assessment with ramipril and rosiglitazone Medication) Trial Investigators, Gerstein HC, Yusuf S, Bosch J, Pogue J, Sheridan P, Dinccag N, Hanefeld M, Hoogwerf B, Laakso M, Mohan V, Shaw J, Zinman B, Holman RR: Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: A randomised controlled trial. Lancet 368 :1096 –1105,2006 [DOI] [PubMed] [Google Scholar]
- 83.Raskin P, Rendell M, Riddle MC, Dole JF, Freed MI, Rosenstock J: A randomized trial of rosiglitazone therapy in patients with inadequately controlled insulin-treated type 2 diabetes. Diabetes Care 24 :1226 –1232,2001 [DOI] [PubMed] [Google Scholar]
- 84.Rosenblatt S, Miskin B, Glazer NB, Prince MJ, Robertson KE: The impact of pioglitazone on glycemic control and atherogenic dyslipidemia in patients with type 2 diabetes mellitus. Coron Artery Dis 12 :413 –423,2001 [DOI] [PubMed] [Google Scholar]
- 85.De Leon DD, Crutchlow MF, Ham JY, Stoffers DA: Role of glucagon-like peptide-1 in the pathogenesis and treatment of diabetes mellitus. Int J Biochem Cell Biol 38 :845 –859,2006 [DOI] [PubMed] [Google Scholar]
- 86.Kieffer TJ, McIntosh CH, Pederson RA: Degradation of glucose-dependent insulinotropic polypeptide and truncated glucagon-like peptide 1 in vitro and in vivo by dipeptidyl peptidase IV. Endocrinology 136 :3585 –3596,1995 [DOI] [PubMed] [Google Scholar]
- 87.Kolterman OG, Kim DD, Shen L, Ruggles JA, Nielsen LL, Fineman MS, Baron AD: Pharmacokinetics, pharmacodynamics, and safety of exenatide in patients with type 2 diabetes mellitus. Am J Health Syst Pharm 62 :173 –181,2005 [DOI] [PubMed] [Google Scholar]
- 88.Buse JB, Henry RR, Han J, Kim DD, Fineman MS, Baron AD: Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 27 :2628 –2635,2004 [DOI] [PubMed] [Google Scholar]
- 89.DeFronzo RA, Ratner RE, Han J, Kim DD, Fineman MS, Baron AD: Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 28 :1092 –1100,2005 [DOI] [PubMed] [Google Scholar]
- 90.Kendall DM, Riddle MC, Rosenstock J, Zhuang D, Kim DD, Fineman MS, Baron AD: Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 28 :1083 –1091,2005 [DOI] [PubMed] [Google Scholar]
- 91.Riddle MC, Henry RR, Poon TH, Zhang B, Mac SM, Holcombe JH, Kim DD, Maggs DG: Exenatide elicits sustained glycaemic control and progressive reduction of body weight in patients with type 2 diabetes inadequately controlled by sulphonylureas with or without metformin. Diabetes Metab Res Rev 22 :483 –491,2006 [DOI] [PubMed] [Google Scholar]
- 92.Ratner RE, Maggs D, Nielsen LL, Stonehouse AH, Poon T, Zhang B, Bicsak TA, Brodows RG, Kim DD: Long-term effects of exenatide therapy over 82 weeks on glycaemic control and weight in over-weight metformin-treated patients with type 2 diabetes mellitus. Diabetes Obes Metab 8 :419 –428,2006 [DOI] [PubMed] [Google Scholar]
- 93.Miller S, St Onge EL: Sitagliptin: A dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. Ann Pharmacother 40 :1336 –1343,2006 [DOI] [PubMed] [Google Scholar]
- 94.Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H, Sitagliptin Study 023 Group: Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 49 :2564 –2571,2006 [DOI] [PubMed] [Google Scholar]
- 95.Januvia [package insert], Whitehouse Station, NJ, Merck and Co.,2006
- 96.Pratley RE, Jauffret-Kamel S, Galbreath E, Holmes D: Twelve-week monotherapy with the DPP-4 inhibitor vildagliptin improves glycemic control in subjects with type 2 diabetes. Horm Metab Res 38 :423 –428,2006 [DOI] [PubMed] [Google Scholar]
- 97.Ahren B, Gomis R, Standl E, Mills D, Schweizer A: Twelve- and 52-week efficacy of the dipeptidyl peptidase IV inhibitor LAF237 in metformin-treated patients with type 2 diabetes. Diabetes Care 27 :2874 –2880,2004 [DOI] [PubMed] [Google Scholar]
- 98.Stang M, Wysowski DK, Butler-Jones D: Incidence of lactic acidosis in metformin users. Diabetes Care 22 :925 –927,1999 [DOI] [PubMed] [Google Scholar]
- 99.Salpeter S, Greyber E, Pasternak G, Salpeter E: Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev CD002967,2006 [DOI] [PubMed]