Abstract
The heart exhibits remarkable adaptive responses to a wide array of genetic and extrinsic factors to maintain contractile function. When compensatory responses are not sustainable, cardiac dysfunction occurs, leading to cardiomyopathy. The many forms of cardiomyopathy exhibit a set of overlapping phenotypes reflecting the limited range of compensatory responses that the heart can use. These include cardiac hypertrophy, induction of genes normally expressed during development, fibrotic deposits that replace necrotic and apoptotic cardiomyocytes, and metabolic disturbances. The compensatory responses are mediated by signaling pathways that initially serve to maintain normal contractility; however, persistent activation of these pathways leads to cardiac dysfunction. Current research focuses on ways to target these specific pathways therapeutically.
Introduction
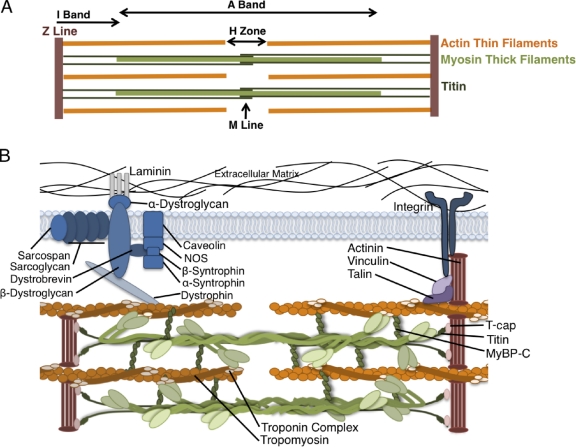
The heart relies on a complex network of cells to maintain appropriate function. Cardiomyocytes, the contracting cells in the heart, exist in a three-dimensional network of endothelial cells, vascular smooth muscle, and an abundance of fibroblasts as well as transient populations of immune cells. Gap junctions electrochemically coordinate the contraction of individual cardiomyocytes, and their connection to the extracellular matrix (ECM) transduces force and coordinates the overall contraction of the heart. Intracellularly, repeating units of actin and myosin form the backbone of sarcomere structure, the basic functional unit of the cardiomyocyte (Fig. 1). The sarcomere itself consists of ∼20 proteins; however, more than 20 other proteins form connections between the myocytes and the ECM and regulate muscle contraction (Fig. 1 B). Given the complexity of the coordinated efforts of the many proteins that exist in multimeric complexes, dysfunction occurs when these interactions are disrupted.
Figure 1.
Anatomy of the cardiac sarcomere. (A) Diagram of the basic organization of the sarcomere. The sarcomere forms the basic contractile unit in the cardiomyocytes of the heart. Thin filaments composed of actin are anchored at the Z line and form transient sliding interactions with thick filaments composed of myosin molecules. The M Line, I Band, and A Band are anatomical features defined by their components (actin, myosin, and cytoskeletal proteins) and appearance in polarized light. Titin connects the Z line with the M line and contributes to the elastic properties and force production of the sarcomere through its extensible region in the I Band. Coordinated shortening of the sarcomere creates contraction of the cardiomyocyte. (B) Representation of the major proteins of the cardiac sarcomere. Attachment to the ECM is mediated by costameres composed of the dystroglycan–glycoprotein complex and the integrin complex. Force transduction and intracellular signaling are coordinated through the costamere. The unique roles of each of these proteins are critical to appropriate function of the heart. T-cap, titin cap; MyBP-C, myosin-binding protein C; NOS, nitric oxide synthase.
Although the heart may functionally tolerate a variety of pathological insults, adaptive responses that aim to maintain function eventually fail, resulting in a wide range of functional deficits or cardiomyopathy. Although a multitude of intrinsic and extrinsic stimuli promote cardiomyopathies, the best described causes are the >900 mutations in genes expressed in the cardiomyocyte (Fig. 1 B; Wang et al., 2010). Mutations in most of these genes cause a diverse range of cardiomyopathies, many with overlapping clinical phenotypes. Mutations in sarcomeric genes are usually inherited in an autosomal-dominant manner and are missense mutations that are incorporated into sarcomeres (Seidman and Seidman, 2001). Thus far >400 mutations in 13 sarcomeric proteins including β-myosin heavy chain (β-MyHC), α-cardiac actin, tropomyosin, and troponin have been associated with cardiomyopathy (www.cardiogenomics.med.harvard.edu). Table I summarizes these mutated proteins.
Table I.
Cardiomyopathies, genetic causes, and cellular pathology
| Disease | Mutated gene | Pathology |
| Hypertrophic cardiomyopathy | Myosin binding protein C | Increased heart mass |
| β-Myosin heavy chain | Left ventricular wall thickening | |
| α-Myosin heavy chain | Increased cardiomyocyte size | |
| α-Tropomyosin | Myofibrillar disarray | |
| Troponin T | Decreased volume of left ventricular chamber | |
| α-Cardiac actin | Interstitial fibrosis | |
| Troponin I | Enlarged nuclei | |
| Titin | Ventricular wall stiffness | |
| Myosin light chains | Inflammation | |
| Troponin C | ||
| Vinculin | ||
| Muscle LIM protein | ||
| Telothonin | ||
| Dilated cardiomyopathy | β-Myosin heavy chain | Ventricular wall thinning |
| Desmin | Ventricular chamber enlargement | |
| N-cadherin | Cardiomyocyte apoptosis | |
| α-Cardiac actin | Interstitial fibrosis | |
| Skeletal muscle myopathies | Enlarged nuclei | |
| α-Tropomyosin 1 | Ventricular wall stiffness | |
| Muscle LIM protein δ-sarcoglycan | Inflammation | |
| Lamin A/C | ||
| TAZ (G4.5) | ||
| Titin | ||
| Phospholamban | ||
| Vinculin | ||
| Troponin I | ||
| Troponin T2 | ||
| SCN5A | ||
| Presenilin 1 & 2 | ||
| Troponin C | ||
| α-Cardiac actinin | ||
| Plakoglobin | ||
| Restrictive cardiomyopathy | Troponin I | Ventricular wall stiffness |
| Desmin | Apoptosis | |
| α-Cardiac actin | Fibrosis | |
| Skeletal muscle myopathies | Myofibrillar disarray | |
| Troponin T | ||
| β-Myosin heavy chain | ||
| Left ventricular noncompaction | TAZ (G4.5) | Trabeculated ventricular walls |
| α-Dystrobrevin | Noncompaction of ventricular endocardium | |
| FKBP12 | Necrotic myocytes | |
| β-Myosin heavy chain | Fibrosis | |
| α-Cardiac actin | Myofibrillar disarray | |
| Cardiac troponin | ||
| Arrhythmogenic right ventricular cardiomyopathy | Plakophilin-2 | Right ventricular dilation |
| Junctional plakoglobin | Right ventricular fibrosis | |
| Desmocollin-2 | Fibrofatty infiltration | |
| Desmoglein-2 | Apoptosis | |
| Desmoplakin | ||
| Muscular dystrophy–associated cardiomyopathy | Dystrophin | Left ventricular hypertrophy or dilation |
| Sarcoglycan | Fibrosis | |
| Dystroglycan | Apoptosis | |
| Dystrobrevin |
Bold: mutations that are common to multiple forms of cardiomyopathy.
There are several examples in which mutations in the same gene can cause different disease phenotypes (indicated in bold in Table I), suggesting that the position of the mutation within the gene influences the cardiac phenotype. Clinical phenotypes are also likely to be modified by sex hormones, diet, and polymorphisms in many genes including those involved in angiotensin signaling, thus complicating attempts to make predictive correlations between specific mutations with disease prognosis (Maron et al., 1995; Deinum et al., 2001; Stauffer et al., 2006). In fact, a modifier gene was identified in one strain of mice that blocked the pathology induced by a mutated β-MyHC (R403Q) in another strain of mice (Semsarian et al., 2001). Characterization and mechanistic study of cardiomyopathies are therefore challenging owing to the complex protein structures, the multitude of disease-causing mutations, and modulators of pathology.
A wide range of inherited and acquired cardiomyopathies has been described by the World Health Organization: hypertrophic cardiomyopathy, dilated cardiomyopathy, restrictive cardiomyopathy, and arrhythmogenic right ventricular cardiomyopathy as well as a large group of unclassified cardiomyopathies. Acquired cardiomyopathies induced by stress, diabetes, some chemotherapeutic agents, pregnancy, and alcohol intake, for example, represent a diverse class of cardiac disease (Bybee et al., 2004; Ro and Frishman, 2006; Dolci et al., 2008; Dobrin and Lebeche, 2010; Iacovoni et al., 2010). In this review, we focus on inherited cardiomyopathies with known mutations in proteins expressed in the heart, describing their prevalence, pathology, and the cellular mechanisms of disease.
Inherited cardiomyopathies
Hypertrophic cardiomyopathy (HCM).
HCM, which is inherited in an autosomal-dominant pattern in more than 50% of patients, occurs in ∼1 in 500 individuals with men affected nearly twice as much as women and is the leading cause of sudden cardiac death among young people in North America (Maron et al., 1984, 1995; Maron and Pelliccia, 2006).
Mutations in 13 sarcomeric proteins have been associated with HCM; those in β-MyHC were the first to be identified in HCM and cause 30% of all cases (Kamisago et al., 2000; Peddy et al., 2006). Clinical phenotypes associated with these mutations range from mild to severe, likely due to the locations of mutations in different functional domains as well as genetic modifiers and environmental stimuli. Most mutations are in the motor domain of myosin; however, disease-causing mutations in the rod region have also been identified (Buvoli et al., 2008). It is not yet clear whether clinical predictions can be made based solely on which functional domains carry mutations.
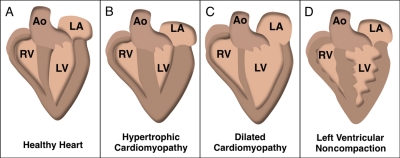
Cardiac dysfunction in HCM is attributable to initial increases in heart mass and asymmetric interventricular septal thickening (Fig. 2, A and B; Maron et al., 1995; Marian, 2000; Wang et al., 2010). Histological hallmarks include an increase in cardiomyocyte size, disorganized arrangement of myocytes (myofibrillar disarray), and both perivascular and interstitial fibrosis, a common phenotype in many cardiomyopathies. Persistent cardiac architectural changes in HCM can result in thinning of the ventricular walls, dilation of the ventricular chamber, and decreases in cardiac output that can lead to heart failure.
Figure 2.
Gross pathology associated with cardiomyopathies. Illustrations depicting (A) a healthy heart with normal wall thicknesses and chamber volumes and (B) hypertrophic cardiomyopathy. Asymmetric hypertrophy of the interventricular septum as well as the left ventricular posterior wall and apex is present. (C) Dilated cardiomyopathy. Thinning of the ventricular walls enlarges the interior dimensions of the ventricular chamber. The left and right atria are also enlarged due to impaired ventricular relaxation. (D) Left ventricular noncompaction. Trabeculated ventricular walls create a spongy appearance, prominent in the left ventricular wall. Ao, aorta; LA, left atrium; LV, left ventricle; RV, right ventricle.
Dilated cardiomyopathy (DCM).
DCM occurs in 1 in 2,500 individuals in the United States and is considered the most prevalent form of cardiomyopathy because it is a common clinical outcome of many prolonged cardiac insults (Cregler, 1989; Vikstrom et al., 1996). Enlargement of the ventricular chamber and thinning of the ventricular walls are the predominant phenotypes in this disease (Fig. 2 C). Loss of heart mass is highly correlated with heart failure and mortality; indeed, 50% of those diagnosed with DCM die within five years of becoming symptomatic (Michels et al., 1992; Grogan et al., 1995).
Although DCM can occur secondary to systemic pathology, approximately one half of cases are not associated with a systemic cause (idiopathic DCM). Among these cases, about one third of patients have a family history of DCM (familial DCM) that is transmitted autosomally (recessive and dominant), through maternal mitochondrial DNA, or as an X-linked mutation (Berko and Swift, 1987; Keeling et al., 1995; Mestroni et al., 1999). In contrast to HCM, which is almost exclusively associated with mutations in sarcomeric proteins, familial DCM is primarily associated with mutations in cytoskeletal proteins (Table I). These and other proteins that connect the sarcomere to the ECM form the mechanotransduction apparatus that allows cardiomyocytes to sense and respond to changes in contractility. Disruption of components of this apparatus therefore prevents the heart from productively responding to both extrinsic and intrinsic stressors.
Restrictive cardiomyopathy (RCM).
RCM is the least common but most lethal form of cardiomyopathy (Davis et al., 2007). Causes of RCM are extremely variable and include induced and genetic initiators as well as idiopathic etiologies. Idiopathic RCM occurs in the absence of other pathology, although it can be associated with skeletal muscle myopathies (Katritsis et al., 1991). Injury to the heart caused by radiation, scarring after heart surgery, infection, or diseases such as amyloidosis can also cause RCM. Recently, mutations in the sarcomeric protein troponin I, which participates in the regulation of calcium-mediated actin–myosin interactions and contractility, were associated with a familial form of the disease, and it is now recognized as the primary disease-causing mutation in familial RCM (Mogensen et al., 2003). Mutations in other sarcomeric proteins such as cardiac desmin, α-cardiac actin, and β-MyHC as well as mutations that have been implicated in HCM have been described in RCM patients (Karam et al., 2008; Parvatiyar et al., 2010).
Unlike in HCM, alterations in tissue flexibility occur in RCM in the absence of changes in ventricular myofibrillar arrangement and gross abnormalities (Huang and Du, 2004). RCM is, therefore, characterized by increased stiffness of the myocardium that causes increased pressure in one or both ventricles. Ventricular relaxation is impaired, similar to the phenotype observed in HCM, and the atria then enlarge in response to the increased pressure in some patients (Parvatiyar et al., 2010). The restrictive phenotype is common to RCM and some cases of HCM, emphasizing the need for more precise diagnostic criteria.
Arrhythmogenic right ventricular cardiomyopathy (ARVC).
ARVC occurs in ∼1 in 5,000 individuals and primarily affects adults (Basso et al., 2009). However, ARVC is also an important cause of death among young people, accounting for ∼20% of deaths occurring in individuals under 30 years old (Shen et al., 1995). ARVC is inherited in an autosomal-dominant or recessive pattern in 50% of cases often with incomplete penetrance, so unknown genetic or environmental modifiers are predicted (Basso et al., 2009).
Mutations in proteins that comprise the cardiac desmosome, a junctional complex that mechanically couples neighboring cardiomyocytes to coordinate contractile activity, account for most inherited cases of ARVC (Yang et al., 2006). Disruptions in desmosome stability cause structural and functional alterations and are associated with cardiomyocyte apoptosis (Yamaji et al., 2005). Adipose and fibrotic tissue that replace cardiomyocytes are a hallmark of ARVC, primarily in the right ventricle; however, left ventricular involvement is common in later stages of the disease (Corrado et al., 1997). The right ventricle enlarges as myocyte loss progresses, causing a reduction in blood volume pumped from the heart and arrhythmias.
Left ventricular noncompaction cardiomyopathy (LVNC).
LVNC, a recently delineated form of cardiomyopathy, is defined anatomically by deep trabeculations that create a spongiform appearance of the ventricular walls (Fig. 2 D; Chin et al., 1990). Clinically, patients present with depressed systolic and diastolic dysfunction, left and right ventricular hypertrophy, arrhythmias, and, in some cases, conduction block. Familial forms with autosomal-dominant or X-linked transmission account for up to 50% of all LVNC cases (Chin et al., 1990; Sasse-Klaassen et al., 2003). The mechanisms responsible for pathology are poorly defined, but indirect evidence suggests that malformation of the myocardium contributes to functional abnormalities in the adult (Yousef et al., 2009). During development, the loose network of fibers that form the myocardium becomes compacted between the fifth and eighth prenatal weeks. In patients with LVNC, a normal pattern of compaction occurs in the outer band of the myocardium, but an abnormal noncompacted region within the inner (endocardial) layer localized mainly in the apical and midventricular regions of the heart is also observed (Fig. 2 D; Chin et al., 1990; Ritter et al., 1997; Freedom et al., 2005) . Similarities among LVNC, HCM, and DCM, likely caused by mutations in the same gene (β-MyHC, for example), motivate differential diagnosis criteria aimed at better defining this syndrome (Weiford et al., 2004; Kaneda et al., 2008).
Cardiomyopathies associated with muscular dystrophies.
Muscular dystrophies, caused by autosomal-recessive mutations in proteins that anchor the sarcomere to the ECM, are a heterogeneous group of skeletal muscle disorders. Incidence of cardiac involvement among patients with muscular dystrophies varies and is dependent upon the disease-causing mutations. Although Duchenne and Becker muscular dystrophies are X-linked, up to 18% of female carriers that do not exhibit skeletal muscle disease exhibit heart failure, illustrating that even subtle disruption of the interactions between the sarcomere and ECM can impart cardiac dysfunction (Hoogerwaard et al., 1999).
Identification of disease-causing mutations in the heart has contributed to the delineation of many phenotypically similar cardiomyopathies. However, much work remains in elucidating the specific cellular mechanisms of disease. In the past decade, resources available to study cardiomyopathies at the cellular level have evolved to allow the study of mechanisms of pathogenesis using a variety of experimental approaches including in vitro biochemistry, cell biology, and animal models (Flavigny et al., 1999; Haq et al., 2000; Maass and Buvoli, 2007). These studies have revealed the roles of a variety of cell types in the development of cardiomyopathy. In addition to cardiomyocyte-mediated effects, nonmyocytes induce functional changes in cardiomyocytes and in the structural properties of the ECM. Deposition of collagen by fibroblasts, for example, creates fibrotic scars that stiffen the heart tissue and impede normal cardiomyocyte contractility. In the sections that follow, we present an overview of the cellular pathology mediated by cardiac cells with an emphasis on the cardiomyocyte, whose responses to intrinsic and extrinsic insults most profoundly affect cardiac function.
Altered cardiomyocyte cell biology in cardiomyopathy
Different classifications of cardiomyopathy exhibit a subset of overlapping symptoms, emphasizing the limited range of cellular responses in the cardiomyocyte (Table I). Alterations in gene expression patterns, changes in cellular morphology, and metabolic shifts in cardiomyocytes initially improve or maintain function in the early stages of many forms of cardiomyopathy. An exception to this is DCM, which does not appear to progress through this initial series of compensatory responses. Patients with DCM exhibit a dilated phenotype that decompensates rapidly when patients become symptomatic, similar to end-stage heart failure HCM.
Cardiomyocyte hypertrophy.
In response to altered contractility or physiological stress, ventricular cardiomyocytes increase in size to keep pace with increased functional demand, thus increasing the thickness of the walls but reducing interior dimensions of the ventricular chambers. Regardless of the initial stimulus, cardiac hypertrophy is thought to be compensatory as it normalizes increased stress in the ventricular wall and augments the contractile capacity of the heart. The development of hypertrophy therefore implies an anatomical adaptation that does not explicitly signify a disease state.
Cardiac hypertrophy is attributable to structural alterations within individual cardiomyocytes. The addition of sarcomeres in parallel is an adaptive response to increase force production in stressed cells. Increased myocyte width (concentric hypertrophy; Fig. 3, A and B) correlates with compensatory ventricular hypertrophy, a hallmark of HCM. In HCM, however, concentric hypertrophy causes a distortion of the cardiomyocyte alignment, which may affect contractility in the heart (Seidman and Seidman, 2001). There remains no clear consensus on whether cardiomyocyte hypertrophy is truly compensatory or merely a component in the pathway to heart failure. However, when hypertrophic growth in a myosin model of HCM was blocked by constitutive activation of glycogen synthase kinase 3β (GSK3β), all male animals died, suggesting that the initial phases of hypertrophy are indeed compensatory (Luckey et al., 2009). Persistent hypertrophy with disease is positively associated with the development of heart failure. After an initial phase of compensatory hypertrophy, addition of sarcomeres to ventricular cardiomyocytes increases length-to-width ratios, which leads to decreased force production. Increased myocyte length produced by end-to-end addition of sarcomeres (eccentric hypertrophy; Fig. 3, C and D) is associated with ventricular dilation in DCM and with advanced HCM.
Figure 3.
Isolated cardiomyocytes exhibiting hypertrophy and heart failure. (A) Untreated adult ventricular cardiomyocyte demonstrating normal brick-like morphology. (B) An adult ventricular cardiomyocyte treated with endothelin-1 precursor protein exhibiting significant concentric hypertrophy. The cardiomyocyte is decreased in length and increased in width, consistent with the phenotype observed in human cardiomyocytes with concentric hypertrophy. A and B are reproduced from Gupta et al. (2005) with permission from Elsevier. (C) Cardiomyocyte isolated from a healthy human heart. (D) Cardiomyocyte isolated from the heart of a patient in heart failure. Increased length and decreased width are characteristic of eccentric hypertrophy. C and D are reproduced from Ohler et al. (2009) with permission from Brian O’Rourke.
Fetal gene program.
In cardiomyopathy, induction of genes normally expressed during embryogenesis occurs including natriuretic factors, β-MyHC, and α-skeletal actin (Kuwahara et al., 2003). Natriuretic proteins secreted by cells in the atria and ventricles, atrial natriuretic factor (ANF) and brain natriuretic peptide (BNP), respectively, induce diuresis and vasodilation, which are capable of reducing blood volume and pressure in response to increased wall stress. These factors are up-regulated in response to transient increases in blood pressure. However, in cardiomyopathy, transcription and secretion are chronic.
ANF expression is increased in atrial myocytes when hearts exhibit diastolic dysfunction, which increases tension on the walls of the atria, and is released by ventricular myocytes when there is increased pressure on the ventricular walls in cardiomyopathy (Rodeheffer et al., 1986; Edwards et al., 1988). Ventricular hypertrophy causes excessive stress in the ventricular wall during contraction, which also serves as a stimulus for release of BNP (Kinnunen et al., 1993). Induced expression of these genes, therefore, implies a cardioprotective mechanism that indirectly attempts to normalize pathological changes that affect the heart. Some evidence suggests that these genes may be anti-hypertrophic to prevent overgrowth of the organ in response to stress (Silberbach et al., 1999).
Myosin isoform shift.
In the face of a wide variety of pathological insults, up-regulation of the β-MyHC isoform occurs. Myosin exists as two heavy chains and two pairs of light chains. Cardiac muscle contains two isoforms of MyHC, α-MyHC and β-MyHC, which are expressed in species-specific proportions in the heart. β-MyHC accounts for more than 90% of the MyHC in the adult human ventricle. The proportion of these isoforms in the heart is tightly regulated, such that up-regulation of β-MyHC expression with a concomitant decrease in α-MyHC in the hearts of humans and rodents is a sensitive marker of early cardiomyopathy (Lowes et al., 1997; Miyata et al., 2000). This response may be adaptive, as β-MyHC exhibits lower ATPase activity resulting in improved ATP use, and may be a requirement in the presence of mutations that reduce efficiency of contractility. However, features of β-MyHC, which exhibits slower filament sliding velocity, may contribute to pathology (Schwartz et al., 1981).
Contractility of the heart is inversely proportional to the amount of expression of β-MyHC (Palmiter et al., 1999; Herron and McDonald, 2002). Titration of β-MyHC expression in adult ventricular cardiomyocytes results in a dose-dependent reduction in sarcomere shortening and velocity, independent of Ca2+ handling (Herron et al., 2007). The shift toward β-MyHC in the pathological heart may contribute to the decreased myocyte shortening and prolonged relaxation of cardiomyocytes observed during the transition from compensatory hypertrophy to heart failure (Nagata et al., 1998).
Fibrosis.
The development of interstitial and perivascular fibrosis is a hallmark of pathology in the heart. Focal fibrosis, composed primarily of collagen types I and III, occurs in the early stages of cardiomyopathy, but over time fibrosis increases and directly compromises the function of cardiomyocytes. The presence of collagen-rich regions in the myocardium causes disruption of excitation–contraction coupling between cardiomyocytes and increased stiffness of the myocardium, and leads to decreased contractility in the heart (Menon et al., 2009). Fibrosis is primarily produced by resident fibroblasts in the heart; however, there is evidence for collagen production by cardiomyocytes (Schram et al., 2010).
Recent studies support a link between sarcomere mutations and gene expression changes in nonmyocytes that promotes production of fibrosis. Increased transcription and release of TGF-β in the hearts of mice expressing HCM-causing mutations in α-MyHC (R403Q, R719W) activate nonmyocytes and promote increased focal and interstitial fibrosis (Teekakirikul et al., 2010). Interference with the development of excessive fibrosis in the hearts of patients with cardiomyopathy could represent a promising therapy to reduce the functional deficits that result from disease-causing mutations in cardiomyocytes.
Alteration in metabolic substrate utilization.
In the healthy heart, phosphocreatine is the main reserve source of ATP during acute stress. As pathogenesis progresses in cardiomyopathy, the demand for ATP increases, and the levels of phosphocreatine are progressively depleted (Ingwall and Weiss, 2004; Smith et al., 2006). This reduction in energy-rich resources forces the heart to favor alternative energy substrates during prolonged stress mediated by exogenous cardiotoxic stimuli or contractile dysfunction caused by mutated sarcomeric proteins. During the initial compensatory stages of cardiomyopathy, substrate utilization favors free fatty acids, which yield about three times more ATP than glucose but require more oxygen to metabolize, making it a more lucrative but less efficient means to produce energy (Ashrafian and Frenneaux, 2007; Neubauer, 2007).
In the failing heart, as in DCM and end-stage HCM, energy substrate utilization in the heart shifts from fatty acid oxidation to glucose utilization, a less efficient means of producing energy (Stanley et al., 1997; Kalsi et al., 1999; Dávila-Román et al., 2002). A proposed mechanism for this transition from compensatory to decompensatory cardiac hypertrophy is excessive cardiac growth without adequate coronary angiogenesis. A mildly hypoxic state up-regulates genes that facilitate utilization of glucose instead of fatty acids (Huang et al., 2004). This is supported by studies in the developing heart in which exposure to the oxygen-rich postnatal environment at birth correlates with a dramatic shift in metabolic enzyme isoforms to use fatty acids as an energy substrate (Lopaschuk et al., 1991). Indeed, hypertrophy induced by volume overload in newborns delays this shift (Kantor et al., 1999). Expression and activity of enzymes that mediate fatty acid use, such as medium-chain acyl-CoA dehydrogenase and peroxisome proliferator-activated receptor-α, are down-regulated in the failing adult heart to resemble expression in the fetal heart (Sack et al., 1996). Therefore, the heart progresses through several stages of metabolic changes as it enters heart failure: the increased use of fatty acids during compensation and a shift to glucose utilization as the heart decompensates.
Sensing and responding to cellular stress
The cellular responses observed in cardiomyopathies arise from the detection of functional changes mediated primarily by ECM–sarcomere connections and collectively aim to maintain sufficient contractile force and prevent progression to heart failure. Signaling pathways initiated by mechanotransduction in the cardiomyocyte initially serve to reduce blood volume, add sarcomeres to increase contractile capacity, and shift metabolic processes to those that favor the utilization of substrates that yield more energy (lipids).
Mechanotransduction and signaling in the cardiomyocyte.
The responses of cardiomyocytes to systemic stress or genetic abnormalities are modulated by mechanosensitive mechanisms within the cardiomyocyte (Molkentin and Dorn, 2001; Seidman and Seidman, 2001; Frey and Olson, 2003). A complex network of proteins that connects the sarcomere to the ECM forms the basis of the mechanotransduction apparatus. For example, components of the costamere complex, which form the connection between the sarcomere and the ECM via integrins, initiate intracellular signaling and subsequently alter contractile properties and transcriptional regulation in response to membrane distortion. Mechanosensitive ion channels are also implicated in signal initiation in response to systemic stress (Le Guennec et al., 1990; Zhang et al., 2000; de Jonge et al., 2002). These channels are likely responsible for acute changes that might initiate other longer-term responses in the heart but are nonetheless important to consider when examining possible transducers of systemic and tissue alterations to the cardiomyocyte.
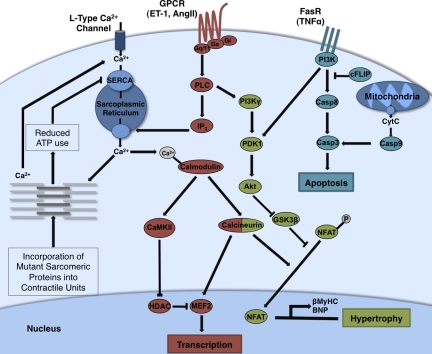
Changes in wall stress induce signaling pathways that are associated with the development of cardiac pathology. The many intracellular signaling pathways that mediate responses to increased demand on the heart have been extensively reviewed elsewhere (Force et al., 1999; Molkentin and Dorn, 2001; Heineke and Molkentin, 2006). Here, we focus on pathways that are intimately involved in pathogenesis (Fig. 4). Although their effects in compensatory responses early in pathology initially increase function by promoting growth and contractility, persistent responses eventually compromise function.
Figure 4.
Signaling pathways associated with cardiac hypertrophy. Although many pathways are associated with cardiomyopathy, up-regulation of transcription and induction of apoptosis are major mediators of pathogenic responses in the heart. The GPCR-associated pathway (dark red) can be activated by ET-1 and AngII, which are released in response to reduced contractility, and mediates contractile adaptation through increased calcium release from the sarcoplasmic reticulum. Increased intracellular calcium activates calmodulin and induces activation of the transcription factor MEF2. Incorporation into the sarcomere of mutant proteins that exhibit reduced ATP efficiency inhibits the sequestration of calcium from the cytosol and further enhances increases in intracellular calcium concentration. GPCR signaling is also associated with activation of the Akt signaling pathway (light green) that induces fetal gene expression and the cardiac hypertrophic response through inhibition of GSK3β. Apoptotic pathways (light blue) are induced by cytochrome c (CytC) release from mitochondria and activation of death receptors (like FasR) by cytokines such as TNF. Calcium overload and myocyte loss significantly contribute to reduced contractility in many forms of cardiomyopathy. ET-1, endothelin-1; HDAC, histone deacetylase; NFAT, nuclear factor of activated T cells; MEF-2, myocyte enhancer factor 2; SERCA, sarco/endoplasmic reticulum calcium-ATPase; cFLIP, cellular FLICE-inhibitory protein; AngII, angiotensin II; FasR, Fas receptor.
Akt signaling.
Akt, the major effector of phosphoinositide 3-kinase (PI3K) signaling, mediates many processes important to cardiac adaptation including protein synthesis, inhibition of apoptosis, and metabolism. Despite clear beneficial effects of acute Akt activation, chronic activation of the PI3K/Akt pathway causes cardiomyopathy. The divergent effects of activated Akt are dependent on the length of time Akt is activated and the initiator of the pathway. Mice expressing constitutively activated (myristoylated) Akt have hearts and cardiac myocytes that are approximately twofold larger than littermate controls and are protected from ischemia reperfusion injury. However, other founder mice expressing the transgene die with cardiac enlargement and presumed heart failure (Matsui et al., 2002). Also, a conditional mutant with activated Akt1 exhibits reversible hypertrophy with two weeks of transgene expression but irreversible hypertrophy with fibrosis and reduced angiogenesis when expressed for six weeks (Shiojima et al., 2005). Therefore, activation of Akt signaling increases cell size and contractile efficiency in the face of acute intensification of functional demand; however, the temporal activation of this pathway determines whether the adaptive effects are maintained productively (Matsui et al., 2002; O’Neill and Abel, 2005).
Downstream of Akt is the kinase GSK-3β, which negatively regulates cardiomyocyte size. GSK-3β is largely unphosphorylated and constitutively active in the healthy heart; phosphorylation of GSK-3β by Akt inactivates it. Overexpression of activated GSK-3β causes reduced heart size and decreased hypertrophy in response to pathological stimuli (Haq et al., 2000; Michael et al., 2004). In genetic models of HCM with mutations in sarcomeric proteins, GSK-3β is inactivated, consistent with hypertrophy in these models and demonstrating downstream alterations in the Akt signaling cascade (Stauffer et al., 2006; Luckey et al., 2009).
G protein signaling.
Pathological stimuli that induce increased pressure or mechanical stretch in the heart promote release of several paracrine and autocrine factors including adrenergic factors, angiotensin II, and endothelin (ET1), which each activate G protein–coupled receptor (GPCR) signaling. G protein subunits serve as effectors for downstream signaling cascades and modulate cardiac adaptation, namely those processes intended to increase force production like cardiomyocyte hypertrophy. Mechanical stretch of adult ventricular cardiomyocytes and administration of Gαq activating agents (norepinephrine, angiotensin II) in neonatal rat ventricular myocytes specifically induce hypertrophy (Sadoshima and Izumo, 1993; Kojima et al., 1994). Each of the agents that activate Gαq in neonatal rat ventricular myocytes also induces activation of phospholipase C and hypertrophy (Simpson et al., 1982; Knowlton et al., 1993; Adams et al., 1996; Sakata et al., 1998). In agreement with these data, Gαq attenuation decreases hypertrophy, cardiac remodeling, and functional deficits in response to pressure overload in mice (Esposito et al., 2001). Transgenic mice overexpressing Gαq exhibit cardiac growth similar to that induced by pressure overload, independent of this stimulus (D’Angelo et al., 1997; Sakata et al., 1998). The hearts of these mice also exhibit some qualities of pathological hypertrophy including left ventricular dysfunction and induction of the fetal gene program indicative of cardiomyopathy (D’Angelo et al., 1997; Sakata et al., 1998).
Ca2+ regulation and calcineurin signaling.
Ca2+ concentrations inside the cardiomyocyte are critically important to actin–myosin interactions. Ca2+ is sequestered within the sarcoplasmic reticulum and the sarcomere itself, which serves as an intracellular reserve that is released in response to electrical stimulation of the cardiomyocyte. After contraction, sarco/endoplasmic reticulum Ca2+-ATPase sequesters the Ca2+ back into the sarcoplasmic reticulum to restore Ca2+ balance. There is a clear correlation between force production and perturbation of Ca2+ regulation, alterations of which might directly induce pathological, anatomical, and functional alterations that lead to heart failure via activation of GPCRs (Minamisawa et al., 1999).
Ca2+ in the cytosol can be increased to modulate sarcomere contractility by signaling through Gαq recruitment and activation of PLCβ. Ca2+ released from the sarcoplasmic reticulum activates calmodulin, which phosphorylates calcineurin, a serine/threonine phosphatase. Upon activation, calcineurin interacts with and dephosphorylates nuclear factor of activated T cells (NFAT), which then translocates into the nucleus. Calcineurin activation exacerbates hypertrophic signals and expedites the transition to a decompensatory state. Indeed, cardiac-specific overexpression of calcineurin or NFAT leads to significant cardiac hypertrophy that progresses rapidly to heart failure (Molkentin et al., 1998). Administration of antagonists of calcineurin attenuates the hypertrophic response of neonatal rat ventricular myocytes to stimuli such as phenylephrine (PE) and angiotensin II (Taigen et al., 2000).
Apoptotic pathways.
Apoptosis has recently been shown to play a major role in cardiac disease (Gill et al., 2002). Adult cardiomyocytes are terminally differentiated, so cell loss is detrimental to cardiac function. Additionally, when cardiomyocytes are lost, deposition of collagen occurs. Ultimately, this decreases heart compliance, increases cardiomyocyte wall stress, and impairs ventricular relaxation.
Apoptosis has been demonstrated in HCM and DCM as evidenced by activated apoptotic pathway components and caspase activity (Narula et al., 1996; Olivetti et al., 1997; Akyürek et al., 2001). Many of the signaling pathways activated with increased cardiomyocyte stretch are implicated in regulation of apoptosis, including Akt and adrenergic stimulation. Several groups have demonstrated that hyperactivation of β-adrenergic signaling pathways and cytochrome c release from mitochondria are associated with increased apoptosis and fibrosis (Communal et al., 1998; Geng et al., 1999; Iwai-Kanai et al., 1999; Narula et al., 1999). Additionally, evidence for increased signaling through Fas receptors by up-regulation of TNF is common in heart failure, and the degradation of inhibitors of Fas receptor signaling such as cFlip is another mechanism by which apoptosis may occur (Krown et al., 1996; Torre-Amione et al., 1996). Studies from human heart biopsies reveal that greater cell loss is observed in the early stages of cardiomyopathy, suggesting that anti-apoptotic pathways are up-regulated after cell loss has been initiated (Akyürek et al., 2001). It follows that tight regulation of apoptosis is required for proper cardiac adaptation and that disruption of these pathways can have irreversible consequences in the heart.
Summary
Advances in molecular techniques have vastly improved the understanding of mechanisms responsible for cardiac dysfunction in cardiomyopathies. It is clear that mutant proteins in cardiomyocytes can perturb cardiac function whether they occur in the contractile apparatus or in the mechanosensitive complexes. Alterations in cardiomyocyte function initiate cascades of cellular responses that attempt to compensate for these insults by normalizing functional parameters in the cells. However, persistent responses at the cellular level lead to organ-wide alterations that are highly correlated with sudden cardiac death and heart failure. Much remains to be learned about precise mechanisms that modulate the transition between compensatory responses in the heart and the development of heart failure.
Despite these challenges, basic studies of the processes that mediate cellular responses to cardiac stress have allowed the development of therapeutics for some cardiomyopathies that include drugs that decrease blood pressure and heart rate (angiotensin II receptor antagonists, Ca2+ channel blockers) to reduce the strain on the ventricular walls. Implantable devices that regulate the synchronization of contraction also reduce the progression of cardiomyopathies (Rose et al., 2001). Although there exist no treatments to directly address the causes of inherited cardiomyopathies, cell transplantation, gene therapy, small interfering RNAs, and microRNAs remain on the horizon of innovative treatments that address the causes rather than reduction of symptoms of cardiomyopathies. Utilization of animal and cellular models to further probe the mechanisms of cardiomyopathy and demonstrate efficacy of drugs that specifically target disease-causing pathways hold promise that further reduction in the mortality associated with cardiomyopathies can be achieved.
Acknowledgments
We gratefully acknowledge Dr. Massimo Buvoli for useful discussion related to the content of this manuscript and Dr. Kristen Barthel, Dr. Matthew Bell, and Christopher Haines for critical review. We also thank Dr. Brian O’Rourke for providing images used in Fig. 3, C and D.
This work was supported by National Institutes of Health grant HL-50560 (L.A. Leinwand) and National Institutes of Health training grant T32 HL-07822 (P.A. Harvey).
Footnotes
Abbreviations used in this paper:
- DCM
- dilated cardiomyopathy
- HCM
- hypertrophic cardiomyopathy
- MyHC
- myosin heavy chain
- RCM
- restrictive cardiomyopathy
References
- Adams J.W., Migita D.S., Yu M.K., Young R., Hellickson M.S., Castro-Vargas F.E., Domingo J.D., Lee P.H., Bui J.S., Henderson S.A. 1996. Prostaglandin F2 alpha stimulates hypertrophic growth of cultured neonatal rat ventricular myocytes. J. Biol. Chem. 271:1179–1186 10.1074/jbc.271.2.1179 [DOI] [PubMed] [Google Scholar]
- Akyürek O., Akyürek N., Sayin T., Dinçer I., Berkalp B., Akyol G., Ozenci M., Oral D. 2001. Association between the severity of heart failure and the susceptibility of myocytes to apoptosis in patients with idiopathic dilated cardiomyopathy. Int. J. Cardiol. 80:29–36 10.1016/S0167-5273(01)00451-X [DOI] [PubMed] [Google Scholar]
- Ashrafian H., Frenneaux M.P. 2007. Metabolic modulation in heart failure: the coming of age. Cardiovasc. Drugs Ther. 21:5–7 10.1007/s10557-007-6000-z [DOI] [PubMed] [Google Scholar]
- Basso C., Corrado D., Marcus F.I., Nava A., Thiene G. 2009. Arrhythmogenic right ventricular cardiomyopathy. Lancet. 373:1289–1300 10.1016/S0140-6736(09)60256-7 [DOI] [PubMed] [Google Scholar]
- Berko B.A., Swift M. 1987. X-linked dilated cardiomyopathy. N. Engl. J. Med. 316:1186–1191 10.1056/NEJM198705073161904 [DOI] [PubMed] [Google Scholar]
- Buvoli M., Hamady M., Leinwand L.A., Knight R. 2008. Bioinformatics assessment of beta-myosin mutations reveals myosin’s high sensitivity to mutations. Trends Cardiovasc. Med. 18:141–149 10.1016/j.tcm.2008.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bybee K.A., Kara T., Prasad A., Lerman A., Barsness G.W., Wright R.S., Rihal C.S. 2004. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann. Intern. Med. 141:858–865 [DOI] [PubMed] [Google Scholar]
- Chin T.K., Perloff J.K., Williams R.G., Jue K., Mohrmann R. 1990. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation. 82:507–513 10.1161/01.CIR.82.2.507 [DOI] [PubMed] [Google Scholar]
- Communal C., Singh K., Pimentel D.R., Colucci W.S. 1998. Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the beta-adrenergic pathway. Circulation. 98:1329–1334 [DOI] [PubMed] [Google Scholar]
- Corrado D., Basso C., Thiene G., McKenna W.J., Davies M.J., Fontaliran F., Nava A., Silvestri F., Blomstrom-Lundqvist C., Wlodarska E.K., et al. 1997. Spectrum of clinicopathologic manifestations of arrhythmogenic right ventricular cardiomyopathy/dysplasia: a multicenter study. J. Am. Coll. Cardiol. 30:1512–1520 10.1016/S0735-1097(97)00332-X [DOI] [PubMed] [Google Scholar]
- Cregler L.L. 1989. Progression from hypertrophic cardiomyopathy to dilated cardiomyopathy. J. Natl. Med. Assoc. 81:820: 824–826 [PMC free article] [PubMed] [Google Scholar]
- D’Angelo D.D., Sakata Y., Lorenz J.N., Boivin G.P., Walsh R.A., Liggett S.B., Dorn G.W., II 1997. Transgenic Galphaq overexpression induces cardiac contractile failure in mice. Proc. Natl. Acad. Sci. USA. 94:8121–8126 10.1073/pnas.94.15.8121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dávila-Román V.G., Vedala G., Herrero P., de las Fuentes L., Rogers J.G., Kelly D.P., Gropler R.J. 2002. Altered myocardial fatty acid and glucose metabolism in idiopathic dilated cardiomyopathy. J. Am. Coll. Cardiol. 40:271–277 10.1016/S0735-1097(02)01967-8 [DOI] [PubMed] [Google Scholar]
- Davis J., Wen H., Edwards T., Metzger J.M. 2007. Thin filament disinhibition by restrictive cardiomyopathy mutant R193H troponin I induces Ca2+-independent mechanical tone and acute myocyte remodeling. Circ. Res. 100:1494–1502 10.1161/01.RES.0000268412.34364.50 [DOI] [PubMed] [Google Scholar]
- de Jonge H.W., Dekkers D.H., Tilly B.C., Lamers J.M. 2002. Cyclic stretch and endothelin-1 mediated activation of chloride channels in cultured neonatal rat ventricular myocytes. Clin. Sci. 103(Suppl 48):148S–151S [DOI] [PubMed] [Google Scholar]
- Deinum J., van Gool J.M., Kofflard M.J., ten Cate F.J., Danser A.H. 2001. Angiotensin II type 2 receptors and cardiac hypertrophy in women with hypertrophic cardiomyopathy. Hypertension. 38:1278–1281 10.1161/hy1101.096114 [DOI] [PubMed] [Google Scholar]
- Dobrin J.S., Lebeche D. 2010. Diabetic cardiomyopathy: signaling defects and therapeutic approaches. Expert Rev. Cardiovasc. Ther. 8:373–391 10.1586/erc.10.17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolci A., Dominici R., Cardinale D., Sandri M.T., Panteghini M. 2008. Biochemical markers for prediction of chemotherapy-induced cardiotoxicity: systematic review of the literature and recommendations for use. Am. J. Clin. Pathol. 130:688–695 10.1309/AJCPB66LRIIVMQDR [DOI] [PubMed] [Google Scholar]
- Edwards B.S., Zimmerman R.S., Schwab T.R., Heublein D.M., Burnett J.C., Jr 1988. Atrial stretch, not pressure, is the principal determinant controlling the acute release of atrial natriuretic factor. Circ. Res. 62:191–195 [DOI] [PubMed] [Google Scholar]
- Esposito G., Prasad S.V., Rapacciuolo A., Mao L., Koch W.J., Rockman H.A. 2001. Cardiac overexpression of a G(q) inhibitor blocks induction of extracellular signal-regulated kinase and c-Jun NH(2)-terminal kinase activity in in vivo pressure overload. Circulation. 103:1453–1458 [DOI] [PubMed] [Google Scholar]
- Flavigny J., Souchet M., Sébillon P., Berrebi-Bertrand I., Hainque B., Mallet A., Bril A., Schwartz K., Carrier L. 1999. COOH-terminal truncated cardiac myosin-binding protein C mutants resulting from familial hypertrophic cardiomyopathy mutations exhibit altered expression and/or incorporation in fetal rat cardiomyocytes. J. Mol. Biol. 294:443–456 10.1006/jmbi.1999.3276 [DOI] [PubMed] [Google Scholar]
- Force T., Hajjar R., Del Monte F., Rosenzweig A., Choukroun G. 1999. Signaling pathways mediating the response to hypertrophic stress in the heart. Gene Expr. 7:337–348 [PMC free article] [PubMed] [Google Scholar]
- Freedom R.M., Yoo S.J., Perrin D., Taylor G., Petersen S., Anderson R.H. 2005. The morphological spectrum of ventricular noncompaction. Cardiol. Young. 15:345–364 10.1017/S1047951105000752 [DOI] [PubMed] [Google Scholar]
- Frey N., Olson E.N. 2003. Cardiac hypertrophy: the good, the bad, and the ugly. Annu. Rev. Physiol. 65:45–79 10.1146/annurev.physiol.65.092101.142243 [DOI] [PubMed] [Google Scholar]
- Geng Y.J., Ishikawa Y., Vatner D.E., Wagner T.E., Bishop S.P., Vatner S.F., Homcy C.J. 1999. Apoptosis of cardiac myocytes in Gsalpha transgenic mice. Circ. Res. 84:34–42 [DOI] [PubMed] [Google Scholar]
- Gill C., Mestril R., Samali A. 2002. Losing heart: the role of apoptosis in heart disease—a novel therapeutic target? FASEB J. 16:135–146 10.1096/fj.01-0629com [DOI] [PubMed] [Google Scholar]
- Grogan M., Redfield M.M., Bailey K.R., Reeder G.S., Gersh B.J., Edwards W.D., Rodeheffer R.J. 1995. Long-term outcome of patients with biopsy-proved myocarditis: comparison with idiopathic dilated cardiomyopathy. J. Am. Coll. Cardiol. 26:80–84 10.1016/0735-1097(95)00148-S [DOI] [PubMed] [Google Scholar]
- Gupta A., Aberle N.S., II, Ren J., Sharma A.C. 2005. Endothelin-converting enzyme-1-mediated signaling in adult rat ventricular myocyte contractility and apoptosis during sepsis. J. Mol. Cell. Cardiol. 38:527–537 10.1016/j.yjmcc.2005.01.002 [DOI] [PubMed] [Google Scholar]
- Haq S., Choukroun G., Kang Z.B., Ranu H., Matsui T., Rosenzweig A., Molkentin J.D., Alessandrini A., Woodgett J., Hajjar R., et al. 2000. Glycogen synthase kinase-3beta is a negative regulator of cardiomyocyte hypertrophy. J. Cell Biol. 151:117–130 10.1083/jcb.151.1.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heineke J., Molkentin J.D. 2006. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat. Rev. Mol. Cell Biol. 7:589–600 10.1038/nrm1983 [DOI] [PubMed] [Google Scholar]
- Herron T.J., McDonald K.S. 2002. Small amounts of alpha-myosin heavy chain isoform expression significantly increase power output of rat cardiac myocyte fragments. Circ. Res. 90:1150–1152 10.1161/01.RES.0000022879.57270.11 [DOI] [PubMed] [Google Scholar]
- Herron T.J., Vandenboom R., Fomicheva E., Mundada L., Edwards T., Metzger J.M. 2007. Calcium-independent negative inotropy by beta-myosin heavy chain gene transfer in cardiac myocytes. Circ. Res. 100:1182–1190 10.1161/01.RES.0000264102.00706.4e [DOI] [PubMed] [Google Scholar]
- Hoogerwaard E.M., van der Wouw P.A., Wilde A.A., Bakker E., Ippel P.F., Oosterwijk J.C., Majoor-Krakauer D.F., van Essen A.J., Leschot N.J., de Visser M. 1999. Cardiac involvement in carriers of Duchenne and Becker muscular dystrophy. Neuromuscul. Disord. 9:347–351 10.1016/S0960-8966(99)00018-8 [DOI] [PubMed] [Google Scholar]
- Huang X.P., Du J.F. 2004. Troponin I, cardiac diastolic dysfunction and restrictive cardiomyopathy. Acta Pharmacol. Sin. 25:1569–1575 [PubMed] [Google Scholar]
- Huang Y., Hickey R.P., Yeh J.L., Liu D., Dadak A., Young L.H., Johnson R.S., Giordano F.J. 2004. Cardiac myocyte-specific HIF-1alpha deletion alters vascularization, energy availability, calcium flux, and contractility in the normoxic heart. FASEB J. 18:1138–1140 10.1096/fj.03-1377com [DOI] [PubMed] [Google Scholar]
- Iacovoni A., De Maria R., Gavazzi A. 2010. Alcoholic cardiomyopathy. J. Cardiovasc. Med. (Hagerstown). 11:884–892 10.2459/JCM.0b013e32833833a3 [DOI] [PubMed] [Google Scholar]
- Ingwall J.S., Weiss R.G. 2004. Is the failing heart energy starved? On using chemical energy to support cardiac function. Circ. Res. 95:135–145 10.1161/01.RES.0000137170.41939.d9 [DOI] [PubMed] [Google Scholar]
- Iwai-Kanai E., Hasegawa K., Araki M., Kakita T., Morimoto T., Sasayama S. 1999. alpha- and beta-adrenergic pathways differentially regulate cell type-specific apoptosis in rat cardiac myocytes. Circulation. 100:305–311 [DOI] [PubMed] [Google Scholar]
- Kalsi K.K., Smolenski R.T., Pritchard R.D., Khaghani A., Seymour A.M., Yacoub M.H. 1999. Energetics and function of the failing human heart with dilated or hypertrophic cardiomyopathy. Eur. J. Clin. Invest. 29:469–477 10.1046/j.1365-2362.1999.00468.x [DOI] [PubMed] [Google Scholar]
- Kamisago M., Sharma S.D., DePalma S.R., Solomon S., Sharma P., McDonough B., Smoot L., Mullen M.P., Woolf P.K., Wigle E.D., et al. 2000. Mutations in sarcomere protein genes as a cause of dilated cardiomyopathy. N. Engl. J. Med. 343:1688–1696 10.1056/NEJM200012073432304 [DOI] [PubMed] [Google Scholar]
- Kaneda T., Naruse C., Kawashima A., Fujino N., Oshima T., Namura M., Nunoda S., Mori S., Konno T., Ino H., et al. 2008. A novel beta-myosin heavy chain gene mutation, p.Met531Arg, identified in isolated left ventricular non-compaction in humans, results in left ventricular hypertrophy that progresses to dilation in a mouse model. Clin. Sci. 114:431–440 10.1042/CS20070179 [DOI] [PubMed] [Google Scholar]
- Kantor P.F., Robertson M.A., Coe J.Y., Lopaschuk G.D. 1999. Volume overload hypertrophy of the newborn heart slows the maturation of enzymes involved in the regulation of fatty acid metabolism. J. Am. Coll. Cardiol. 33:1724–1734 10.1016/S0735-1097(99)00063-7 [DOI] [PubMed] [Google Scholar]
- Karam S., Raboisson M.J., Ducreux C., Chalabreysse L., Millat G., Bozio A., Bouvagnet P. 2008. A de novo mutation of the beta cardiac myosin heavy chain gene in an infantile restrictive cardiomyopathy. Congenit. Heart Dis. 3:138–143 10.1111/j.1747-0803.2008.00165.x [DOI] [PubMed] [Google Scholar]
- Katritsis D., Wilmshurst P.T., Wendon J.A., Davies M.J., Webb-Peploe M.M. 1991. Primary restrictive cardiomyopathy: clinical and pathologic characteristics. J. Am. Coll. Cardiol. 18:1230–1235 10.1016/0735-1097(91)90540-P [DOI] [PubMed] [Google Scholar]
- Keeling P.J., Gang Y., Smith G., Seo H., Bent S.E., Murday V., Caforio A.L., McKenna W.J. 1995. Familial dilated cardiomyopathy in the United Kingdom. Br. Heart J. 73:417–421 10.1136/hrt.73.5.417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinnunen P., Vuolteenaho O., Ruskoaho H. 1993. Mechanisms of atrial and brain natriuretic peptide release from rat ventricular myocardium: effect of stretching. Endocrinology. 132:1961–1970 10.1210/en.132.5.1961 [DOI] [PubMed] [Google Scholar]
- Knowlton K.U., Michel M.C., Itani M., Shubeita H.E., Ishihara K., Brown J.H., Chien K.R. 1993. The alpha 1A-adrenergic receptor subtype mediates biochemical, molecular, and morphologic features of cultured myocardial cell hypertrophy. J. Biol. Chem. 268:15374–15380 [PubMed] [Google Scholar]
- Kojima M., Shiojima I., Yamazaki T., Komuro I., Zou Z., Wang Y., Mizuno T., Ueki K., Tobe K., Kadowaki T., et al. 1994. Angiotensin II receptor antagonist TCV-116 induces regression of hypertensive left ventricular hypertrophy in vivo and inhibits the intracellular signaling pathway of stretch-mediated cardiomyocyte hypertrophy in vitro. Circulation. 89:2204–2211 [DOI] [PubMed] [Google Scholar]
- Krown K.A., Page M.T., Nguyen C., Zechner D., Gutierrez V., Comstock K.L., Glembotski C.C., Quintana P.J., Sabbadini R.A. 1996. Tumor necrosis factor alpha-induced apoptosis in cardiac myocytes. Involvement of the sphingolipid signaling cascade in cardiac cell death. J. Clin. Invest. 98:2854–2865 10.1172/JCI119114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwahara K., Saito Y., Takano M., Arai Y., Yasuno S., Nakagawa Y., Takahashi N., Adachi Y., Takemura G., Horie M., et al. 2003. NRSF regulates the fetal cardiac gene program and maintains normal cardiac structure and function. EMBO J. 22:6310–6321 10.1093/emboj/cdg601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Guennec J.Y., Peineau N., Argibay J.A., Mongo K.G., Garnier D. 1990. A new method of attachment of isolated mammalian ventricular myocytes for tension recording: length dependence of passive and active tension. J. Mol. Cell. Cardiol. 22:1083–1093 10.1016/0022-2828(90)90072-A [DOI] [PubMed] [Google Scholar]
- Lopaschuk G.D., Spafford M.A., Marsh D.R. 1991. Glycolysis is predominant source of myocardial ATP production immediately after birth. Am. J. Physiol. 261:H1698–H1705 [DOI] [PubMed] [Google Scholar]
- Lowes B.D., Minobe W., Abraham W.T., Rizeq M.N., Bohlmeyer T.J., Quaife R.A., Roden R.L., Dutcher D.L., Robertson A.D., Voelkel N.F., et al. 1997. Changes in gene expression in the intact human heart. Downregulation of alpha-myosin heavy chain in hypertrophied, failing ventricular myocardium. J. Clin. Invest. 100:2315–2324 10.1172/JCI119770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luckey S.W., Walker L.A., Smyth T., Mansoori J., Messmer-Kratzsch A., Rosenzweig A., Olson E.N., Leinwand L.A. 2009. The role of Akt/GSK-3beta signaling in familial hypertrophic cardiomyopathy. J. Mol. Cell. Cardiol. 46:739–747 10.1016/j.yjmcc.2009.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maass A.H., Buvoli M. 2007. Cardiomyocyte preparation, culture, and gene transfer. Methods Mol. Biol. 366:321–330 10.1007/978-1-59745-030-0_18 [DOI] [PubMed] [Google Scholar]
- Marian A.J. 2000. Pathogenesis of diverse clinical and pathological phenotypes in hypertrophic cardiomyopathy. Lancet. 355:58–60 10.1016/S0140-6736(99)06187-5 [DOI] [PubMed] [Google Scholar]
- Maron B.J., Pelliccia A. 2006. The heart of trained athletes: cardiac remodeling and the risks of sports, including sudden death. Circulation. 114:1633–1644 10.1161/CIRCULATIONAHA.106.613562 [DOI] [PubMed] [Google Scholar]
- Maron B.J., Nichols P.F., III, Pickle L.W., Wesley Y.E., Mulvihill J.J. 1984. Patterns of inheritance in hypertrophic cardiomyopathy: assessment by M-mode and two-dimensional echocardiography. Am. J. Cardiol. 53:1087–1094 10.1016/0002-9149(84)90643-X [DOI] [PubMed] [Google Scholar]
- Maron B.J., Gardin J.M., Flack J.M., Gidding S.S., Kurosaki T.T., Bild D.E. 1995. Prevalence of hypertrophic cardiomyopathy in a general population of young adults. Echocardiographic analysis of 4111 subjects in the CARDIA Study. Coronary Artery Risk Development in (Young) Adults. Circulation. 92:785–789 [DOI] [PubMed] [Google Scholar]
- Matsui T., Li L., Wu J.C., Cook S.A., Nagoshi T., Picard M.H., Liao R., Rosenzweig A. 2002. Phenotypic spectrum caused by transgenic overexpression of activated Akt in the heart. J. Biol. Chem. 277:22896–22901 10.1074/jbc.M200347200 [DOI] [PubMed] [Google Scholar]
- Menon S.C., Eidem B.W., Dearani J.A., Ommen S.R., Ackerman M.J., Miller D. 2009. Diastolic dysfunction and its histopathological correlation in obstructive hypertrophic cardiomyopathy in children and adolescents. J. Am. Soc. Echocardiogr. 22:1327–1334 10.1016/j.echo.2009.08.014 [DOI] [PubMed] [Google Scholar]
- Mestroni L., Rocco C., Gregori D., Sinagra G., Di Lenarda A., Miocic S., Vatta M., Pinamonti B., Muntoni F., Caforio A.L., et al. ; Heart Muscle Disease Study Group 1999. Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. J. Am. Coll. Cardiol. 34:181–190 10.1016/S0735-1097(99)00172-2 [DOI] [PubMed] [Google Scholar]
- Michael A., Haq S., Chen X., Hsich E., Cui L., Walters B., Shao Z., Bhattacharya K., Kilter H., Huggins G., et al. 2004. Glycogen synthase kinase-3beta regulates growth, calcium homeostasis, and diastolic function in the heart. J. Biol. Chem. 279:21383–21393 10.1074/jbc.M401413200 [DOI] [PubMed] [Google Scholar]
- Michels V.V., Moll P.P., Miller F.A., Tajik A.J., Chu J.S., Driscoll D.J., Burnett J.C., Rodeheffer R.J., Chesebro J.H., Tazelaar H.D. 1992. The frequency of familial dilated cardiomyopathy in a series of patients with idiopathic dilated cardiomyopathy. N. Engl. J. Med. 326:77–82 10.1056/NEJM199201093260201 [DOI] [PubMed] [Google Scholar]
- Minamisawa S., Hoshijima M., Chu G., Ward C.A., Frank K., Gu Y., Martone M.E., Wang Y., Ross J., Jr, Kranias E.G., et al. 1999. Chronic phospholamban-sarcoplasmic reticulum calcium ATPase interaction is the critical calcium cycling defect in dilated cardiomyopathy. Cell. 99:313–322 10.1016/S0092-8674(00)81662-1 [DOI] [PubMed] [Google Scholar]
- Miyata S., Minobe W., Bristow M.R., Leinwand L.A. 2000. Myosin heavy chain isoform expression in the failing and nonfailing human heart. Circ. Res. 86:386–390 [DOI] [PubMed] [Google Scholar]
- Mogensen J., Kubo T., Duque M., Uribe W., Shaw A., Murphy R., Gimeno J.R., Elliott P., McKenna W.J. 2003. Idiopathic restrictive cardiomyopathy is part of the clinical expression of cardiac troponin I mutations. J. Clin. Invest. 111:209–216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molkentin J.D., Dorn G.W., II 2001. Cytoplasmic signaling pathways that regulate cardiac hypertrophy. Annu. Rev. Physiol. 63:391–426 10.1146/annurev.physiol.63.1.391 [DOI] [PubMed] [Google Scholar]
- Molkentin J.D., Lu J.R., Antos C.L., Markham B., Richardson J., Robbins J., Grant S.R., Olson E.N. 1998. A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell. 93:215–228 10.1016/S0092-8674(00)81573-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K., Liao R., Eberli F.R., Satoh N., Chevalier B., Apstein C.S., Suter T.M. 1998. Early changes in excitation-contraction coupling: transition from compensated hypertrophy to failure in Dahl salt-sensitive rat myocytes. Cardiovasc. Res. 37:467–477 10.1016/S0008-6363(97)00278-2 [DOI] [PubMed] [Google Scholar]
- Narula J., Haider N., Virmani R., DiSalvo T.G., Kolodgie F.D., Hajjar R.J., Schmidt U., Semigran M.J., Dec G.W., Khaw B.A. 1996. Apoptosis in myocytes in end-stage heart failure. N. Engl. J. Med. 335:1182–1189 10.1056/NEJM199610173351603 [DOI] [PubMed] [Google Scholar]
- Narula J., Pandey P., Arbustini E., Haider N., Narula N., Kolodgie F.D., Dal Bello B., Semigran M.J., Bielsa-Masdeu A., Dec G.W., et al. 1999. Apoptosis in heart failure: release of cytochrome c from mitochondria and activation of caspase-3 in human cardiomyopathy. Proc. Natl. Acad. Sci. USA. 96:8144–8149 10.1073/pnas.96.14.8144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neubauer S. 2007. The failing heart—an engine out of fuel. N. Engl. J. Med. 356:1140–1151 10.1056/NEJMra063052 [DOI] [PubMed] [Google Scholar]
- O’Neill B.T., Abel E.D. 2005. Akt1 in the cardiovascular system: friend or foe? J. Clin. Invest. 115:2059–2064 10.1172/JCI25900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohler A., Weisser-Thomas J., Piacentino V., Houser S.R., Tomaselli G.F., O’Rourke B. 2009. Two-photon laser scanning microscopy of the transverse-axial tubule system in ventricular cardiomyocytes from failing and non-failing human hearts. Cardiol. Res. Pract. 2009:802373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olivetti G., Abbi R., Quaini F., Kajstura J., Cheng W., Nitahara J.A., Quaini E., Di Loreto C., Beltrami C.A., Krajewski S., et al. 1997. Apoptosis in the failing human heart. N. Engl. J. Med. 336:1131–1141 10.1056/NEJM199704173361603 [DOI] [PubMed] [Google Scholar]
- Palmiter K.A., Tyska M.J., Dupuis D.E., Alpert N.R., Warshaw D.M. 1999. Kinetic differences at the single molecule level account for the functional diversity of rabbit cardiac myosin isoforms. J. Physiol. 519:669–678 10.1111/j.1469-7793.1999.0669n.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parvatiyar M.S., Pinto J.R., Dweck D., Potter J.D. 2010. Cardiac troponin mutations and restrictive cardiomyopathy. J. Biomed. Biotechnol. 2010:350706 10.1155/2010/350706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peddy S.B., Vricella L.A., Crosson J.E., Oswald G.L., Cohn R.D., Cameron D.E., Valle D., Loeys B.L. 2006. Infantile restrictive cardiomyopathy resulting from a mutation in the cardiac troponin T gene. Pediatrics. 117:1830–1833 10.1542/peds.2005-2301 [DOI] [PubMed] [Google Scholar]
- Ritter M., Oechslin E., Sütsch G., Attenhofer C., Schneider J., Jenni R. 1997. Isolated noncompaction of the myocardium in adults. Mayo Clin. Proc. 72:26–31 10.4065/72.1.26 [DOI] [PubMed] [Google Scholar]
- Ro A., Frishman W.H. 2006. Peripartum cardiomyopathy. Cardiol. Rev. 14:35–42 10.1097/01.crd.0000174805.68081.f7 [DOI] [PubMed] [Google Scholar]
- Rodeheffer R.J., Tanaka I., Imada T., Hollister A.S., Robertson D., Inagami T. 1986. Atrial pressure and secretion of atrial natriuretic factor into the human central circulation. J. Am. Coll. Cardiol. 8:18–26 10.1016/S0735-1097(86)80086-9 [DOI] [PubMed] [Google Scholar]
- Rose E.A., Gelijns A.C., Moskowitz A.J., Heitjan D.F., Stevenson L.W., Dembitsky W., Long J.W., Ascheim D.D., Tierney A.R., Levitan R.G., et al. ; Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure (REMATCH) Study Group 2001. Long-term use of a left ventricular assist device for end-stage heart failure. N. Engl. J. Med. 345:1435–1443 10.1056/NEJMoa012175 [DOI] [PubMed] [Google Scholar]
- Sack M.N., Rader T.A., Park S., Bastin J., McCune S.A., Kelly D.P. 1996. Fatty acid oxidation enzyme gene expression is downregulated in the failing heart. Circulation. 94:2837–2842 [DOI] [PubMed] [Google Scholar]
- Sadoshima J., Izumo S. 1993. Mechanical stretch rapidly activates multiple signal transduction pathways in cardiac myocytes: potential involvement of an autocrine/paracrine mechanism. EMBO J. 12:1681–1692 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakata Y., Hoit B.D., Liggett S.B., Walsh R.A., Dorn G.W., II 1998. Decompensation of pressure-overload hypertrophy in G alpha q-overexpressing mice. Circulation. 97:1488–1495 [DOI] [PubMed] [Google Scholar]
- Sasse-Klaassen S., Gerull B., Oechslin E., Jenni R., Thierfelder L. 2003. Isolated noncompaction of the left ventricular myocardium in the adult is an autosomal dominant disorder in the majority of patients. Am. J. Med. Genet. A. 119A:162–167 10.1002/ajmg.a.20075 [DOI] [PubMed] [Google Scholar]
- Schram K., De Girolamo S., Madani S., Munoz D., Thong F., Sweeney G. 2010. Leptin regulates MMP-2, TIMP-1 and collagen synthesis via p38 MAPK in HL-1 murine cardiomyocytes. Cell. Mol. Biol. Lett. 15:551–563 10.2478/s11658-010-0027-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz K., Lecarpentier Y., Martin J.L., Lompré A.M., Mercadier J.J., Swynghedauw B. 1981. Myosin isoenzymic distribution correlates with speed of myocardial contraction. J. Mol. Cell. Cardiol. 13:1071–1075 10.1016/0022-2828(81)90297-2 [DOI] [PubMed] [Google Scholar]
- Seidman J.G., Seidman C. 2001. The genetic basis for cardiomyopathy: from mutation identification to mechanistic paradigms. Cell. 104:557–567 10.1016/S0092-8674(01)00242-2 [DOI] [PubMed] [Google Scholar]
- Semsarian C., Healey M.J., Fatkin D., Giewat M., Duffy C., Seidman C.E., Seidman J.G. 2001. A polymorphic modifier gene alters the hypertrophic response in a murine model of familial hypertrophic cardiomyopathy. J. Mol. Cell. Cardiol. 33:2055–2060 10.1006/jmcc.2001.1466 [DOI] [PubMed] [Google Scholar]
- Shen W.K., Edwards W.D., Hammill S.C., Bailey K.R., Ballard D.J., Gersh B.J. 1995. Sudden unexpected nontraumatic death in 54 young adults: a 30-year population-based study. Am. J. Cardiol. 76:148–152 10.1016/S0002-9149(99)80047-2 [DOI] [PubMed] [Google Scholar]
- Shiojima I., Sato K., Izumiya Y., Schiekofer S., Ito M., Liao R., Colucci W.S., Walsh K. 2005. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J. Clin. Invest. 115:2108–2118 10.1172/JCI24682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silberbach M., Gorenc T., Hershberger R.E., Stork P.J., Steyger P.S., Roberts C.T., Jr 1999. Extracellular signal-regulated protein kinase activation is required for the anti-hypertrophic effect of atrial natriuretic factor in neonatal rat ventricular myocytes. J. Biol. Chem. 274:24858–24864 10.1074/jbc.274.35.24858 [DOI] [PubMed] [Google Scholar]
- Simpson P., McGrath A., Savion S. 1982. Myocyte hypertrophy in neonatal rat heart cultures and its regulation by serum and by catecholamines. Circ. Res. 51:787–801 [DOI] [PubMed] [Google Scholar]
- Smith C.S., Bottomley P.A., Schulman S.P., Gerstenblith G., Weiss R.G. 2006. Altered creatine kinase adenosine triphosphate kinetics in failing hypertrophied human myocardium. Circulation. 114:1151–1158 10.1161/CIRCULATIONAHA.106.613646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley W.C., Lopaschuk G.D., McCormack J.G. 1997. Regulation of energy substrate metabolism in the diabetic heart. Cardiovasc. Res. 34:25–33 10.1016/S0008-6363(97)00047-3 [DOI] [PubMed] [Google Scholar]
- Stauffer B.L., Konhilas J.P., Luczak E.D., Leinwand L.A. 2006. Soy diet worsens heart disease in mice. J. Clin. Invest. 116:209–216 10.1172/JCI24676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taigen T., De Windt L.J., Lim H.W., Molkentin J.D. 2000. Targeted inhibition of calcineurin prevents agonist-induced cardiomyocyte hypertrophy. Proc. Natl. Acad. Sci. USA. 97:1196–1201 10.1073/pnas.97.3.1196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teekakirikul P., Eminaga S., Toka O., Alcalai R., Wang L., Wakimoto H., Nayor M., Konno T., Gorham J.M., Wolf C.M., et al. 2010. Cardiac fibrosis in mice with hypertrophic cardiomyopathy is mediated by non-myocyte proliferation and requires Tgf-β. J. Clin. Invest. 120:3520–3529 10.1172/JCI42028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torre-Amione G., Kapadia S., Lee J., Durand J.B., Bies R.D., Young J.B., Mann D.L. 1996. Tumor necrosis factor-alpha and tumor necrosis factor receptors in the failing human heart. Circulation. 93:704–711 [DOI] [PubMed] [Google Scholar]
- Vikstrom K.L., Factor S.M., Leinwand L.A. 1996. Mice expressing mutant myosin heavy chains are a model for familial hypertrophic cardiomyopathy. Mol. Med. 2:556–567 [PMC free article] [PubMed] [Google Scholar]
- Wang L., Seidman J.G., Seidman C.E. 2010. Narrative review: harnessing molecular genetics for the diagnosis and management of hypertrophic cardiomyopathy. Ann. Intern. Med. 152:513–520: W181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiford B.C., Subbarao V.D., Mulhern K.M. 2004. Noncompaction of the ventricular myocardium. Circulation. 109:2965–2971 10.1161/01.CIR.0000132478.60674.D0 [DOI] [PubMed] [Google Scholar]
- Yamaji K., Fujimoto S., Ikeda Y., Masuda K., Nakamura S., Saito Y., Yutani C. 2005. Apoptotic myocardial cell death in the setting of arrhythmogenic right ventricular cardiomyopathy. Acta Cardiol. 60:465–470 10.2143/AC.60.5.2004965 [DOI] [PubMed] [Google Scholar]
- Yang Z., Bowles N.E., Scherer S.E., Taylor M.D., Kearney D.L., Ge S., Nadvoretskiy V.V., DeFreitas G., Carabello B., Brandon L.I., et al. 2006. Desmosomal dysfunction due to mutations in desmoplakin causes arrhythmogenic right ventricular dysplasia/cardiomyopathy. Circ. Res. 99:646–655 10.1161/01.RES.0000241482.19382.c6 [DOI] [PubMed] [Google Scholar]
- Yousef Z.R., Foley P.W., Khadjooi K., Chalil S., Sandman H., Mohammed N.U., Leyva F. 2009. Left ventricular non-compaction: clinical features and cardiovascular magnetic resonance imaging. BMC Cardiovasc. Disord. 9:37 10.1186/1471-2261-9-37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y.H., Youm J.B., Sung H.K., Lee S.H., Ryu S.Y., Ho W.K., Earm Y.E. 2000. Stretch-activated and background non-selective cation channels in rat atrial myocytes. J. Physiol. 523:607–619 10.1111/j.1469-7793.2000.00607.x [DOI] [PMC free article] [PubMed] [Google Scholar]