Abstract
Two recently generated targeted mouse alleles of the neurogenic gene Ascl1 were used to characterize cerebellum circuit formation. First, genetic inducible fate mapping (GIFM) with an Ascl1CreER allele was found to specifically mark all glial and neuron cell types that arise from the ventricular zone (vz). Moreover, each cell type has a unique temporal profile of marking with Ascl1CreER GIFM. Of great utility, Purkinje cells (Pcs), an early cohort of Bergmann glia, and four classes of GABAergic interneurons can be genetically birth dated during embryogenesis using Ascl1CreER GIFM. Astrocytes and oligodendrocytes, in contrast, express Ascl1CreER throughout their proliferative phase in the white matter. Interestingly, the final position each neuron type acquires differs depending on when it expresses Ascl1. Interneurons (including candelabrum) attain a more outside position the later they express Ascl1, whereas Pcs have distinct settling patterns each day they express Ascl1. Second, using a conditional Ascl1 allele, we discovered that Ascl1 is differentially required for generation of most vz-derived cells. Mice lacking Ascl1 in the cerebellum have a major decrease in three types of interneurons with a tendency toward a loss of later-born interneurons, as well as an imbalance of oligodendrocytes and astrocytes. Double-mutant analysis indicates that a related helix-loop-helix protein, Ptf1a, functions with Ascl1 in generating interneurons and Pcs. By fate mapping vz-derived cells in Ascl1 mutants, we further discovered that Ascl1 plays a specific role during the time period when Pcs are generated in restricting vz progenitors from becoming rhombic lip progenitors.
Introduction
A fundamental question in neural development is how neurons and glia are generated in temporally and spatially restricted manners such that their ultimate settling patterns result in formation of functional circuits. The cerebellum, a primary center for motor coordination, is an ideal system to study this question because of its highly stereotyped cytoarchitecture and protracted period of development (Altman and Bayer, 1997; Sillitoe and Joyner, 2007). We have used the powerful technique of genetic inducible fate mapping (GIFM) (Joyner and Zervas, 2006; Legué and Joyner, 2010) combined with conditional ablation of the gene encoding the basic helix-loop-helix (bHLH) transcription factor Ascl1 to gain novel insights into development of the cerebellar microcircuitry.
The cerebellum consists of a uniform layered cytoarchitecture organized into a complex set of folia. Embedded in the white matter (wm) are three pairs of cerebellar nuclei (cn) that consist of glutamatergic projection neurons and GABA-expressing interneurons. The cerebellum has an internal granule layer (igl) containing glutamatergic granule cells (gcs) and scattered interneurons. The adjacent monolayer consists of the cell bodies of GABAergic Purkinje cells (Pcs) and Bergmann glia (Bg). The outer molecular layer (ml) contains three types of interneurons (candelabrum, basket, and stellate) and gc axons and Pc dendrites. A critical question is whether there is a relationship between the timing of production of each cell type and its final organization within the microcircuitry.
The cerebellum has two spatially distinct germinal zones: the ventricular zone (vz) and the upper rhombic lip (url). A GIFM study using a url-specific Atoh1-CreER transgene demonstrated that glutamatergic neurons of the cn are generated before gc precursors (gcps) (Machold and Fishell, 2005). Cumulative fate mapping of the vz using a Ptf1aCre allele identified Pcs, three of the interneuron types (Golgi, stellate, basket), and possibly astrocytes (Hoshino et al., 2005; Pascual et al., 2007) as arising from the vz. Recent bromodeoxyuridine (BrdU) birth-dating experiments in rodents indicated that GABAergic cerebellar interneurons are generated in an inside-to-outside progression (Leto et al., 2009). While unipolar brush cells in the igl are thought to arise from the url (Englund et al., 2006), the origin of Lugaro and candelabrum cells remains unknown. However, the full details of the temporal sequence of production and subsequent settling patterns of all vz-derived cells have not been determined.
We reasoned that the Ascl1 gene could provide a tool for genetically birth dating cerebellar vz cells and studying the genetic regulation of cerebellum development, since Ascl1 is expressed in many neural progenitors as they transition from proliferation to differentiation (Guillemot et al., 1993; Battiste et al., 2007; Kim et al., 2007). In addition, Ascl1 is required to generate a normal number of Pax2+ interneurons and oligodendrocyte precursors in the cerebellum, at least at birth when null mutants die (Grimaldi et al., 2009). By combining GIFM with an Ascl1CreER allele and cerebellum-specific conditional deletion of Ascl1, we discovered Ascl1 has diverse roles in generation of all vz-derived cell types, and propose a model for construction of cerebellar microcircuitry.
Materials and Methods
Mouse lines.
The following mouse lines were used and genotyped as described previously: Ptf1aCre (Kawaguchi et al., 2002), Ascl1CreER (Kim et al., 2011), Ascl1flx (Pacary et al., 2011), Rosa26loxP-STOP-lacZ (Soriano, 1999), Rosa26loxP-STOP-YFP (Srinivas et al., 2001), and En1Cre (Kimmel et al., 2000). Animals were maintained on a 12 h light/dark cycle and allowed ad libitum access to food and water. All animal studies were performed on an outbred SW background, under an approved Institutional Animal Care and Use Committee animal protocol according to the institutional guidelines at Memorial Sloan-Kettering Cancer Center. The day that a plug was detected was designated as embryonic day 0.5 (E0.5). The day of birth was designated as postnatal day 0 (P0). Adults were designated as P21 or older.
Fate mapping.
Double heterozygous males (Ascl1CreER; R26loxP-STOP-YFP or Ascl1CreER; R26loxP-STOP-lacZ) were bred with SW females (5–6 weeks of age; Taconic Farms) to generate double heterozygous embryos. The R26 alleles were genotyped as previously described (Soriano, 1999). Tamoxifen (Tm) (T-5648; Sigma-Aldrich) was dissolved in corn oil (Sigma-Aldrich; C-8267) at a final concentration of 20 mg/ml. The females were given Tm via gavage with animal feeding needles (Fine Science Tools) at noon on the designated days (2 mg per 40 g of body weight). Dissected brains were immersion fixed for 20 min in 4% paraformaldehyde (PFA) at 4°C, and then cryoprotected in 15 and 30% sucrose. The equilibrated samples were embedded in OCT (Tissue-Tek) using liquid N2-chilled 2-methyl butane (Sigma-Aldrich). Frozen sections were obtained at a thickness of 20 μm. For whole-mount β-galactosidase (βgal) analysis, the samples were washed twice for 10 min at room temperature in Xgal buffer (0.05% deoxycholate, 1 mm MgCl2, 0.02% NP40 in PBS, pH 7.4) and then placed in Xgal reaction buffer (1 mg/ml Xgal, 5 mm potassium ferrocyanide, 5 mm potassium ferricyanide) at 37°C overnight. The extent of the marked domain was the same for embryos and postnatal mice within a litter (n > 5) and between litters (n > 4), although the percentage of labeled cells varied a small amount between animals. Sections were fixed for 5 min in 4% PFA/PBS (room temp), washed twice in Xgal wash buffer for 10 min each, and then stained overnight in Xgal reaction buffer at 37°C. Sections were counterstained with 0.005% Nuclear Fast Red. Ptf1a-Ascl1 conditional knock-out (cko) and En1-Ascl1 cko animals were given an intraperitoneal injection of sodium pentobarbital and transcardially perfused with 20–30 ml of PBS followed by 60 ml of 4% paraformaldehyde. Brains were dissected and cryoprotected in 30% sucrose until sectioning. Tissue was sectioned on a cryostat at 40 μm and serial free-floating sections were collected in 96-well plates containing 0.05% sodium azide, and then stored at 4°C until use.
RNA in situ hybridization, histology, and immunofluorescent immunohistochemistry.
Section RNA in situ analysis was performed as described previously (Li and Joyner, 2001). Ascl1 (Guillemot and Joyner, 1993) and Cre (Machold and Fishell, 2005) probes were prepared as described previously. Immunohistochemistry using immunofluorescence was performed using standard staining procedures with the following antibodies: mouse anti-BrdU (BD Biosciences), mouse anti-calbindin (Swant), rabbit anti-GABA (Sigma-Aldrich), rat anti-GFP (Nacalai Tesque), rabbit anti-GFP (Invitrogen), rabbit anti-βgal (Biogenesis), chicken anti-βgal (Abcam), rabbit anti-Pax6 (Millipore Bioscience Research Reagents), rabbit anti-Pax2 (Abcam), mouse anti-Ki67 (BD Biosciences), guinea pig anti-Ascl1 (gift from J. Johnson, University of Texas Southwestern, Dallas, TX), mouse anti-parvalbumin (Abcam), rabbit anti-neurogranin (Abcam), mouse anti-GFAP (Abcam), sheep anti-CAII (Serotec), and mouse anti-S100β (Abcam). Sections were mounted in Vectashield with DAPI (Vector Laboratories) and examined with a fluorescent microscope (Axio Observer; Zeiss). Fluorescent images were captured as 1.5 μm optical sections using Zeiss Observer software for Apotome and processed using Adobe Photoshop. Orthogonal analysis was performed to confirm coexpression of two markers. For BrdU staining, sections were pretreated for antigen retrieval by steaming in sodium citrate for 15 min. For bright-field staining, species-specific, biotinylated secondary antibodies (Vector) were used at 1:400 dilution followed by incubation in ABC (avidin–biotin complex) (Vector). Visualization was achieved using diaminobenzidine (Vector) as the developing agent.
BrdU staining and quantification.
BrdU (Sigma-Aldrich; B9285) was dissolved in 0.007N NaOH and administered by intraperitoneal injection. To determine proliferative status of cells in the cerebellum, pregnant females were injected intraperitoneally with 100 μg of BrdU per gram of body weight 1 h before killing. To assay the birth date of cells, pregnant females were injected intraperitoneally with 100 μg of BrdU per gram of body weight 12 h before Tm administration and were killed at P21. For early postnatal stages, pups were injected with BrdU 12 h before Tm intracutaneous injection. To quantify the number of BrdU+ Ascl1CreER GIFM-marked cells at P21, the percentage of BrdU+ cells was calculated by counting the total number of fate-mapped cells (βgal+ cells) on at least six sections per cerebellum of at least three animals. Measurements of cells from multiple embryos/animals were pooled into datasets.
Quantification of anterior–posterior distribution of Pcs.
To count the percentage of Pcs per cardinal lobe, E18.5 Xgal-stained sections of Ascl1CreER GIFM animals that received Tm at E11.5 or E12.5 were analyzed at the midline, and 100 and 200 μm off the midline. For Figure 3G, we used one colored dot for every three to five Xgal+ Pcs to schematize the average distribution of marked Pcs.
Figure 3.
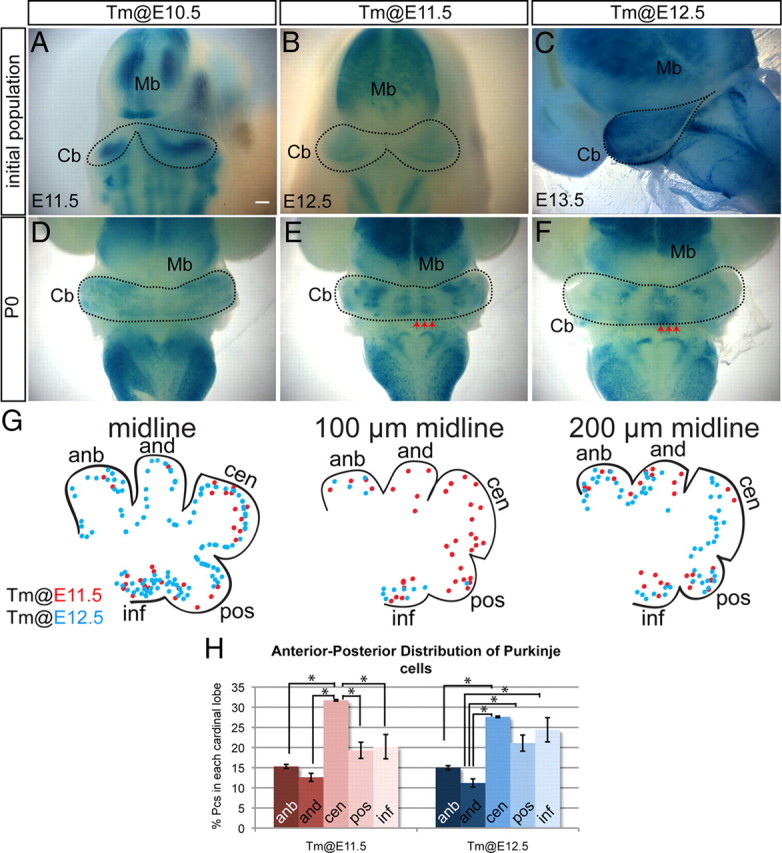
Purkinje cells marked with Ascl1CreER GIFM on consecutive days settle in distinct M-L and A-P patterns. A–C, Whole-mount Xgal staining of embryos 24 h after Tm administration. A, E11.5 whole-mount Xgal staining of Pcs marked at E10.5 reveals that Pcs were uniformly labeled in the lateral cerebellar primordium. B, At E12.5, whole-mount Xgal staining of Pcs marked at E11.5 shows labeling of Pcs in the cerebellar primordium excluding the midline. C, E13.5 whole-mount Xgal staining of Pcs marked at E12.5 reveals that Pcs were uniformly labeled throughout the M-L axis of the cerebellum. D, P0 whole-mount Xgal staining of Pcs marked at E10.5 shows that Pcs were found throughout the cerebellum except the midline. E, P0 whole-mount Xgal staining of Pcs marked at E11.5 depicts four pairs of distinct M-L Pc bands. F, P0 whole-mount Xgal staining of Pcs marked at E12.5 shows a single midline Pc band and two bilateral clusters in the paravermis. G, Summary schematic of A-P distribution pattern of Pcs marked at E11.5 (red) and E12.5 (blue). The arrows in E and F indicate positions analyzed in G. H, The percentage of marked Pcs residing in each of the cardinal lobes at E18.5 cerebellum. Error bars indicate SEM. Mb, Midbrain; Cb, cerebellum; anb, anterobasal; and, anterodorsal; cen, central; pos, posterior; inf, inferior. Scale bar: A–C, 200 μm; D–F, 350 μm. *p < 0.05.
Quantification of Ascl1CreER GIFM-marked cells.
To determine the temporal patterns of Ascl1CreER-marked cells, we analyzed GFP-neurogranin, GFP-parvalbumin, GFP-CAII, and GFP-GFAP double-immunostained P21 cerebella of Ascl1CreER/+;R26loxP-STOP-YFP/+ mice that received Tm at E13.5, E14.5, E17.5, P0, P1, P4, and P7. At least four to six serial sections per GIFM mouse were analyzed to determine the percentage of each cell type marked at each Tm administration time point. In addition to distinct cell type-specific markers, we also examined the morphology and layer position to determine the cell type.
Quantification of Ptf1a-Ascl1 cko and En1-Ascl1 cko phenotype.
For all different measurements we analyzed at least four to six sections from at least three animals per genotype. To quantify the number of Pcs, the number of Pc bodies was counted per length of Pc layer on sections immunostained for anti-calbindin. To assess the number of basket and stellate interneurons in the ml, the ml of hematoxylin and eosin-stained sections was divided along the middle to produce lower and upper sublayers and the number of positive cells was counted within each sublayer, respectively. To quantify the number of Golgi interneurons, oligodendrocytes, and astrocytes, cells double positive for GFP and neurogranin, CAII, or S100β, respectively, were counted on at least six serial sections from at least three animals per genotype. The average number of each cell type per square millimeter was determined and the mutant numbers were normalized to controls, which were set as 1. To quantify the cerebellum circumference, the ImageJ (http://rsb.info.nih.gov/ij/) tracing tool was used to outline the outer surface of the cerebellum on at least six sections evenly spaced along the medial–lateral (M-L) axis per animal and the length measured. The average length in millimeters was determined and the mutant numbers were normalized to controls, which were set as 1.
Quantification of fate-mapped gcps within the external granule layer of Ptf1aCre/+;Ascl1−/−.
Sections of wild-type and mutant littermate controls were processed and stained for Xgal simultaneously. The number of gcps was counted within a 100 μm length of the external granule layer (egl) at the top of the three cardinal lobes depicted in Figure 8, E and F.
Figure 8.
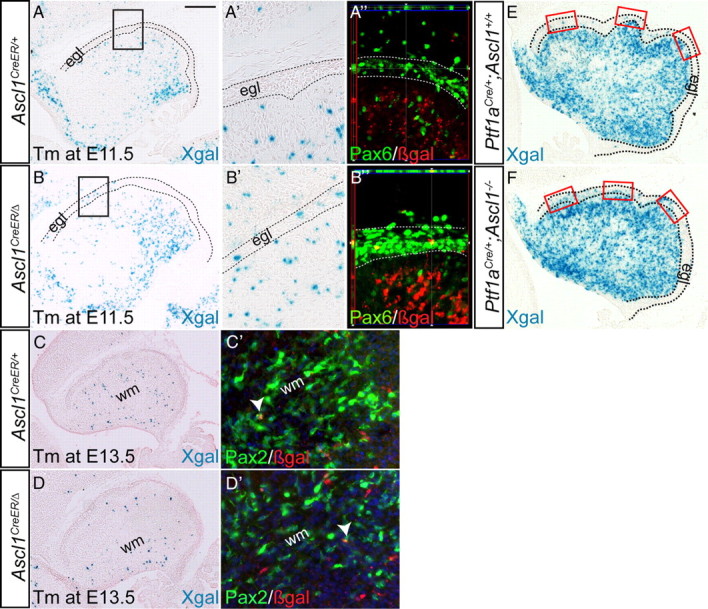
In the absence of Ascl1, granule cell precursors are marked with Ascl1CreER GIFM. A, A′, Xgal staining of cerebellar sections of E17.5 Ascl1CreER/+;R26lacZ/+ mice administered Tm at E11.5 showing marking of only Pcs, in contrast to Ascl1CreER/Δ;R26lacZ/+ mice in which gcps in the egl (dotted outline) are marked in addition to Pcs (B, B′). A″, B″, Double-labeling immunohistochemistry of Pax6 (green) and βgal (red) shows that Ascl1CreER/Δ GIFM-marked cells within the egl are gcps (Pax6+). Xgal staining of E17.5 cerebellar sections of Ascl1CreER/+;R26lacZ/+ (C, C′) and Ascl1CreER/Δ;R26lacZ/+ (D, D′) when Tm was administered at E13.5 reveals marking only of cells within the white matter, some of which are positive for Pax2 (C′, D′). Xgal staining of Ptf1aCre/+;Ascl1CreER/Δ;R26lacZ/+ shows more gcps marked within the egl (E, dotted lines) compared with littermate controls (F). Scale bar: A–F, 200 μm; A′, A″, B′, B″, C′, D″, 100 μm.
Results
Ascl1CreER expression is similar to endogenous Ascl1 in the cerebellum vz during embryogenesis
As a possible means to genetically mark and birth date cells as they leave the cerebellum vz, we engineered an Ascl1CreER knock-in allele using gene targeting to replace the coding sequences of Ascl1 with CreERT2 (Kim et al., 2011). In subsequent GIFM studies, temporal control of cell marking was obtained by administering Tm at specific time points during development to activate CreERT2. To highlight the morphology of marked cells, two reporter alleles, R26loxP-STOP-lacZ (Soriano, 1999) and R26loxP-STOP-YFP (Srinivas et al., 2001), were used. We refer to double heterozygous mice Ascl1CreER/+; R26loxP-STOP-lacZ/+ or Ascl1CreER/+;R26loxP-STOP-YFP/+ as Ascl1CreER GIFM mice. Many cells expressing Ascl1CreER should undergo Cre-mediated excision of the loxP flanked STOP cassette ∼6–36 h after administration of Tm, resulting in permanent and heritable βgal or YFP expression (Joyner and Zervas, 2006). By activating the CreERT2 protein at different embryonic and early postnatal stages, distinct cohorts of Ascl1-expressing cells were labeled and their subsequent fates determined during development or in the adult mouse cerebellum (see details below).
We verified that the Ascl1CreER knock-in allele is expressed like Ascl1 in the cerebellum by comparing CreER and Ascl1 mRNA expression on adjacent sections of Ascl1CreER/+ embryos and postnatal mice using RNA in situ hybridization analysis (Fig. 1A–J) (Zordan et al., 2008). Indeed, we observed a similar expression pattern of CreER and Ascl1 at all stages. At E10.5, Ascl1 and CreER were detected near the vz, but restricted to the lateral cerebellum primordium (Fig. 1A,F). By E11.5, both transcripts were found near the vz throughout the M-L axis, except near the midline (Fig. 1B,G). At E12.5–E14.5 (Fig. 1C,H) (data not shown), Ascl1 and CreER were detected in the vz region throughout the M-L axis. In contrast, at E17.5 only a few Ascl1- and CreER-expressing cells were found near the vz, and instead cells in the cerebellar wm expressed both transcripts (Fig. 1D,I, arrows). At P0 and later, Ascl1 was only detected in the wm (Fig. 1E,J) (data not shown). In conclusion, Ascl1CreER is expressed similar to Ascl1 from at least E10.5 to P5 and is initially restricted to cells near the vz and then becomes expressed in the wm where glial and interneuron progenitors reside.
Figure 1.
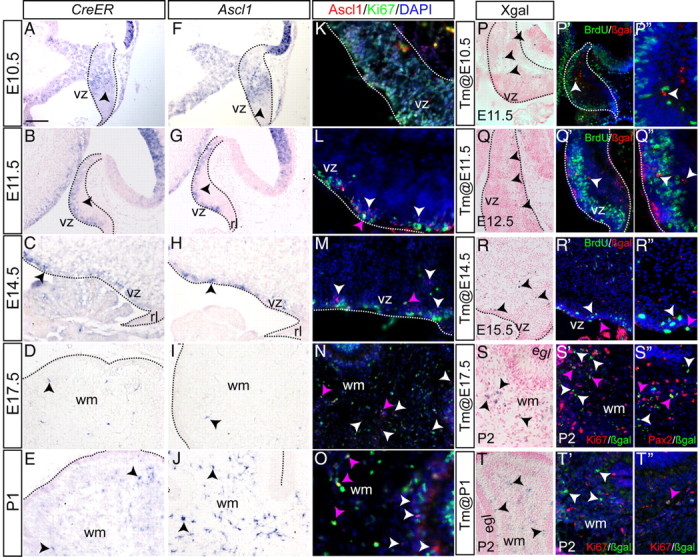
The Ascl1CreER allele is expressed similar to Ascl1 and primarily marks cells that rapidly stop dividing. CreER expression (A–E) closely resembles endogenous Ascl1 expression (F–J) in the cerebellum (outline) throughout embryogenesis. K–O, Ascl1-expressing cells have distinct proliferative characteristics at various time points. P–T″, Ascl1CreER/+;R262RlacZ/+ animals were given Tm at different time points and killed 24 h later. One hour BrdU pulse was given to Ascl1CreER/+;R262RlacZ/+ animals before killing. P, Tm was administered at E10.5, and sagittal sections of E11.5 embryos were analyzed by Xgal staining. Xgal+ cells were found away from the vz. P′, Double-labeling immunohistochemistry for BrdU and βgal shows BrdU (green) in the vz, whereas Ascl1CreER GIFM-marked cells were BrdU negative. P″, Higher magnification of P′. Same type of analysis was performed for Tm administration at E11.5 (Q–Q″) and E14.5 (R–R″). Few Ascl1CreER GIFM-marked cells incorporated BrdU (pink arrows) when Tm was administered at E14.5 (R′, R″). S′, When Tm was administered at E17.5 and the cerebellum analyzed at P2, few cells were Ki67+ (pink arrows). S″, The majority of βgal+ cells were Pax2+ (pink arrowheads). T′, T″, When Tm was administered at P1, very few of the fate-mapped cells were Ki67+ (pink arrow). rl, Rhombic lip. Scale bar: A–P′, Q, Q′, S–T″, 50 μm; P″, Q″, R″, 30 μm.
The earliest Ascl1CreER-marked cells are GABAergic interneurons of the cn and Pcs
In many regions of the embryonic CNS and PNS, studies have indicated that Ascl1 is a proneural gene. For example, overexpression of Ascl1 in the chick neural tube induces progenitors to rapidly exit the cell cycle and leave the vz (Nakada et al., 2004), whereas loss of Ascl1 results in a delay of neuronal differentiation in the spinal cord and telencephalon (Guillemot et al., 1993; Casarosa et al., 1999; Parras et al., 2004; Helms et al., 2005; Pattyn et al., 2006; Wildner et al., 2006). Furthermore, GIFM studies in the spinal cord using an Ascl1-CreER bacterial artificial chromosome (BAC) found that the initial population of marked cells was outside the mantle zone (Battiste et al., 2007). In the cerebellum, however, the transgene was found to mark only a limited number of vz-derived cell types, specifically Pcs only on the last of the three days when they are born (Tm administration at E12.5), cn interneurons at two stages (E10.5 and E14.5) rather than only the one day they are born (Altman and Bayer, 1997), and oligodendrocytes only at a late embryonic stage (E17.5) (Kim et al., 2008). Additionally, no GABAergic interneurons in the igl or ml were labeled using the Ascl1-CreER BAC transgenic line (Kim et al., 2008). Thus, either Ascl1 is not expressed in all cells as they leave the cerebellar vz, or the BAC transgene does not faithfully recapitulate Ascl1 expression.
Given the extensive expression of CreER in the vz and wm of Ascl1CreER mice, we used GIFM to test whether the Ascl1CreER allele can be used to mark all vz-derived cell types in the cerebellum, and whether marking occurs near the time when they exit the cell cycle. We began by administering Tm at embryonic stages when Pcs and cn interneurons are born (E11–E13). Consistent with traditional 3H thymidine birth dating experiments (Altman and Bayer, 1997), we found that when Tm was administered at E10.5 only Pcs and GABAergic cells of the cn were marked, as confirmed by double immunohistochemistry (IHC) for βgal and GABA or the Pc-specific marker calbindin (Fig. 2B,C). In addition, when Tm was administered at E11.5 or E12.5, only Pcs were marked with Ascl1CreER GIFM (Fig. 2D,E). Furthermore, no GABAergic interneurons were found in the igl or ml (cerebellar cortex) at all three time points. Thus, the first cell types to express Ascl1 and be generated by the vz are the GABAergic interneurons of the cn and Pcs in the cortex, both of which project to the excitatory neurons of the cn that are derived from the url around the same time.
Figure 2.
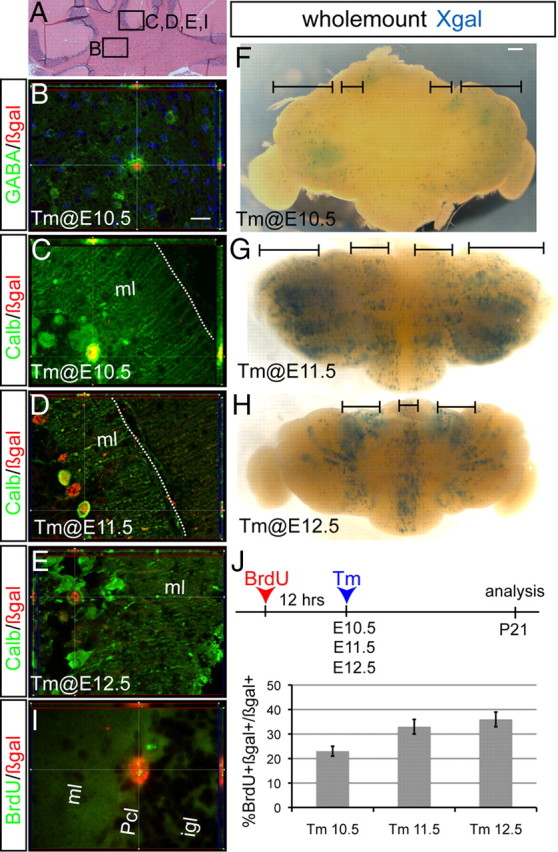
GABAergic cn interneurons and Purkinje cells are the first cell types marked by Ascl1CreER GIFM. A, Coronal section of the adult cerebellum highlighting where images in B–E and I were taken. B, Double-labeling immunohistochemistry for GABA (green) and βgal (red) shows that cn interneurons were marked when Tm was administered at E10.5. Anti-calbindin (green) and anti-βgal (red) double-labeling immunohistochemistry shows that fate-mapped cells are Purkinje cells when Tm was administered at E10.5 (C), E11.5 (D), and E12.5 (E). F–H, Dorsal views of whole-mount Xgal staining of P21 cerebella when Tm was administered at E10.5 (F), E11.5 (G), and E12.5 (H). I, BrdU was administered 12 h before Tm administration at E10.5, E11.5, or E12.5 and analysis performed at P21. Double-labeling immunohistochemistry for βgal (red) and BrdU (green) shows that Ascl1CreER GIFM-marked Pcs retained BrdU labeling. J, Quantification of the number of double-positive BrdU and βgal Pcs (mean ± SEM; n = 3; unpaired t test). Pcl, Purkinje cell layer. Scale bar: A, 100 μm; B–E, I, 20 μm; F–H, 600 μm.
During E10.5–E12.5, Ascl1CreER GIFM marks cerebellar ventricular zone-derived cells around the time they become postmitotic
Given the correlation between the cell types marked with Ascl1CreER GIFM between E10.5 and E12.5 and previous birth-dating experiments showing that these cells are born during this time period, we tested whether cells express Ascl1CreER around the time of their final cell division. IHC analysis of Ascl1 protein and Ki67 (proliferation marker) at E10.5 detected Ascl1 in many dividing cells throughout the cerebellar vz (Fig. 1K). In contrast, at E11.5 and E12.5 only rare Ascl1+ cells expressed Ki67 (Fig. 1L) (data not shown). We next analyzed the initial populations of cells marked with Ascl1CreER GIFM 24 h after Tm administration (Fig. 1P–T″). When Tm was administered on each day between E10.5 and E12.5, the initial populations of marked cells (βgal+) within the cerebellar primordium appeared to be beside the vz (Fig. 1P–P″,Q–Q″). To determine whether any of the initial population of Ascl1CreER GIFM-marked cells were mitotically active, a single dose of BrdU was administered 1 h before killing the mice 24 h after administering Tm. Strikingly, none of the βgal+ cells were BrdU+. Thus, cells marked using Ascl1CreER GIFM by administration of tamoxifen at E10.5, E11.5, or E12.5 continue to divide for <24 h (Fig. 1P′,P″,Q′,Q″). To investigate whether Ascl1CreER GIFM marks cells close to the time of their final cell division throughout E10.5–E12.5, we administered BrdU 12 h before administering Tm and colabeled marked cells for βgal and BrdU at P21. Indeed, we found that many fate-mapped cells marked using Ascl1CreER GIFM at all three stages retained BrdU labeling (∼22–35%) and thus underwent their final cell division around the time of genetic marking (Fig. 2I,J). Our finding that many cells labeled with BrdU at E10 and marked with Ascl1CreER GIFM by Tm administration at E10.5 are BrdU/βgal+ at P21 suggests that many of the BrdU+/Ascl1+ cells detected at E10.5 must be undergoing their final (or second-to-last) cell division. In summary, during at least E10.5–E12.5, Ascl1CreER GIFM can be used to construct a temporal birth date map of vz-derived cells in the cerebellum. Furthermore, the resolution provided by genetic marking allows for the characterization of the marked cells at a level unattainable with BrdU.
Purkinje cells born on different days settle in distinct positions along two axes
Previous studies suggested that cohorts of Pcs born on each day between E10.5 and E12.5 settle in distinct M-L or anterior–posterior (A-P) positions (Altman and Bayer, 1997; Hashimoto and Mikoshiba, 2003). Since Ascl1 is expressed homogenously in the lateral cerebellum vz and progressively expands into the midline, we were curious to determine whether Ascl1-expressing progenitors give rise to the predicted spatially distinct Pc populations on different days. Strikingly, whole-mount staining of the cerebella of adult Ascl1CreER GIFM mice with Xgal revealed a distinct M-L organization of the Pcs marked on each day (Fig. 2F–H). The earliest marked Pcs (Tm at E10.5) not only predominantly settled in the lateral cerebellum (hemispheres) but also in addition formed one pair of bilateral thin stripes at the junction between the vermis and hemispheres (paravermis) (Fig. 2F). In contrast, the Pcs marked with Tm at E11.5 settled in four bilateral pairs of broad stripes, with a mostly negative stripe in the midline (Fig. 2G). Surprisingly, the latest marked Pcs at E12.5 settled in a single stripe in the midline and in a pair of bilateral stripes in the paravermis, despite Ascl1CreER expression throughout the M-L axis (Fig. 2H). These findings provide further evidence that Ascl1CreER is expressed in all Pcs around the time they are born, since the stripe patterns are similar to those reported by marking Pcs with a replication-defective adenoviral vectors thought to infect cells in their last cell division on E10.5, E11.5, and E12.5 (Hashimoto and Mikoshiba, 2003).
Given the distinct M-L organization of Pcs observed in the adult cerebellum, we next tested whether Pcs are organized in the same M-L organization when they first leave the vz by analyzing the distribution of Pcs marked by Ascl1CreER GIFM 24 h after Tm administration in whole-mount brains (Fig. 3). Strikingly, unlike the pattern of labeling in the adult, we found that the initial marked populations of Pcs were not in stripes as previously reported (Hashimoto and Mikoshiba, 2003) but instead were distributed similar to the endogenous Ascl1 RNA expression. Specifically, Pcs marked with Ascl1CreER GIFM at E10.5 were observed uniformly across the lateral E11.5 cerebellum (Fig. 3A), whereas labeling at E11.5 resulted in uniform marking everywhere but the midline (Fig. 3B). Similarly, when Tm was administered at E12.5, the initial marked population of Pcs was found throughout the M-L axis, although very few vz-derived cells are present at the midline at E13.5 (Fig. 3C). Interestingly, when cerebella of Ascl1CreER GIFM mice were marked with Tm on each day between E10.5 and E12.5 and stained with Xgal in whole mount at P0, the Pcs were organized in distinct M-L stripes similar to those in the adult (Fig. 3D–F). Thus, Pcs are born uniformly within the vz, but then settle into parasaggital stripes by late embryogenesis.
We next examined the A-P distribution pattern of Pcs marked with Tm on E11.5 and E12.5 at E18.5, after the M-L stripes of Pcs born on different days are established. Since the vast majority of Pcs labeled with Tm administration on E10.5 settle laterally, we did not analyze such fate-mapped cells. When the sum of the distribution of Pcs in three sagittal sections (midline, ∼100 and ∼200 μm from the midline) was determined, we found no striking spatial pattern in settling of Pcs, although the central lobe, which is the largest lobe, contained a significantly higher percentage of total labeled Pcs compared with the other lobes (Fig. 3G,H). If the size of each lobe were taken into account, then the inferior lobe would have contained the highest proportion of marked cells. Interestingly, when we examined the A-P distribution of Pcs at each of the three separate M-L positions, we found distinct settling patterns for each position and day of marking. In the most midline sections, we found that the few of Pcs marked at E11.5 primarily occupied the central and inferior lobes, whereas Pcs marked a day later were spread throughout the A-P axis with the highest proportion in the inferior lobe. In sections ∼100 μm away from the midline, Pcs marked with Tm at E11.5 settled throughout the A-P axis, whereas Pcs marked a day later primarily occupied the most anterior lobes (anterobasal) and the inferior lobe. Last, in the area ∼200 μm away from the midline, Pcs marked at E11.5 preferentially settled in all lobes but the central lobe, whereas Pcs marked at E12.5 settled throughout the A-P axis. A [3H]thymidine labeling study of Pcs done in rat suggested that, in the medial vermis, the earliest born Pcs occupy the central lobe, and the next born Pcs settle in the posterior lobe followed by the inferior lobe, with the latest born Pcs occupying the anterodorsal then anterobasal lobe (Altman and Bayer, 1997). There are both similarities and differences if our Pc genetic birth-dating study is compared with the previous [3H]thymidine study. The most likely explanation for the differences is that we analyzed the A-P settling patterns in three different areas of the vermis, whereas the previous study described the A-P pattern only near the midline region.
Cerebellar inhibitory interneurons express Ascl1 in distinct temporal patterns starting at E13.5
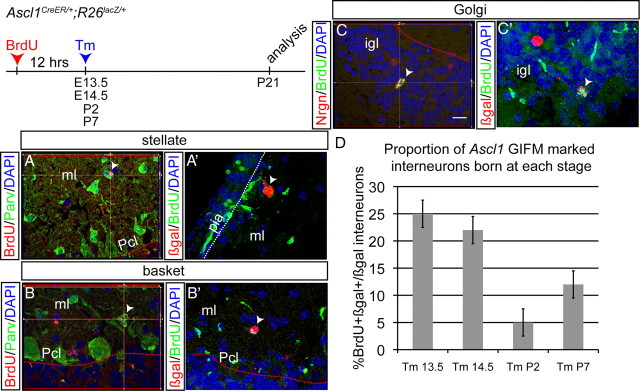
Given the exclusivity in marking of vz-derived Pcs and cn neurons with Ascl1CreER GIFM at E10.5–E12.5, we next asked whether other GABAergic neurons of the cerebellar cortex are marked with Ascl1CreER GIFM after E12.5. Each of the four GABAergic interneuron subtypes can be distinguished based on their morphology and their distinct position in the cortex (Altman and Bayer, 1997). Basket cells located in the inner molecular layer (Fig. 4A,A′), and stellate cells located in the outer molecular layer (Fig. 4B,B′), synapse with Pc soma and dendrites, respectively, and express parvalbumin (Celio, 1990; Bastianelli, 2003). Neurogranin-expressing Golgi cells have large soma within the glomeruli of the igl where they synapse with gcs, and receive inputs from both gc parallel fibers in the molecular layer and mossy fiber axon terminals (Fig. 4C,C′). Basket and stellate cells sharpen the response of Pcs to activity from the parallel fibers, whereas the Golgi cells limit the duration of gc activation by mossy fibers (Altman and Bayer, 1997; Barmack and Yakhnitsa, 2008). Much less is known about the recently identified candelabrum interneurons that have small cell bodies in the Pc layer and extensive chandelier-like processes in the molecular layer (Fig. 4D) (Lainé and Axelrad, 1994).
Figure 4.
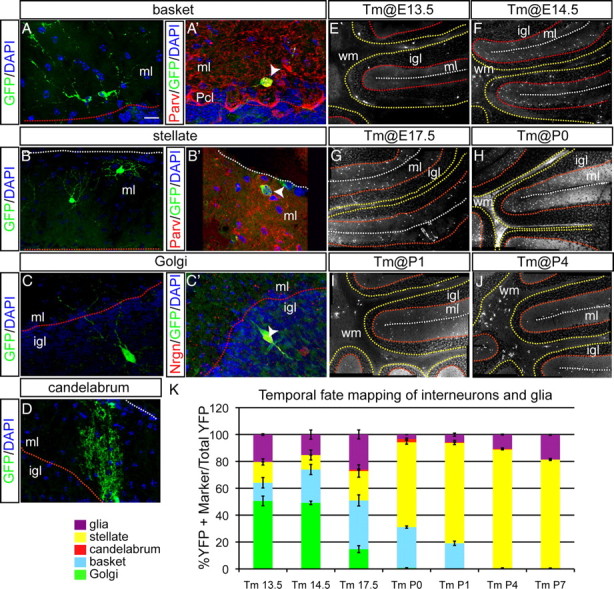
Temporal fate mapping of cerebellar interneurons reveals inside-to-outside settling in the cortex. All sections were analyzed at P21. A–C, An example of fate-mapped cell types when Tm was administered at E14.5. A, A′, Distinct morphology of basket cells, found in the ml close to Pcl, is shown. Double-labeling immunohistochemistry for parvalbumin (red) and YFP (green) identifies some fate-mapped cells in inner ml as basket cells. B, B′, Distinct morphology of stellate cells located in the outer ml is shown. Double-labeling immunohistochemistry for parvalbumin (red) and YFP (green) in the outer ml identifies some fate-mapped cells as stellate cells. C, C′, Large size of fate-mapped interneurons in the igl indicates Golgi cells. Double-labeling immunohistochemistry for neurogranin (red) and YFP (green) confirms Golgi cell identity. D, A chandelier-shaped candelabrum fate-mapped (YFP+) cell in the inner ml is shown. E–J, Mosaic images of YFP immunohistochemistry in Ascl1CreER GIFM animals when Tm was administered at E13.5 (E), E14.5 (F), E17.5 (G), P0 (H), P1 (I), and P4 (J). K, A comparison of the percentage of each cell type marked at different time points during development is shown (mean ± SEM; n = 3; unpaired t test). The white dotted line indicates pial surface, the red dotted line indicates border between Pcl and igl, and the yellow dotted line indicates border between igl and wm. Pcl, Purkinje cell layer. Scale bar: A–D, 20 μm; E–J, 100 μm.
We found that, when Tm was administered at E13.5, E14.5, E17.5, P0, P1, P4, or P7, the majority of fate-mapped cells in P21 Ascl1CreER GIFM mice were interneurons in the cerebellar cortex. The remaining marked cells were glia (see details below). Strikingly, no Pcs or cn interneurons were marked at these stages of Tm administration. Furthermore, the relative proportion of each interneuron cell type that was marked changed as development progressed (Fig. 4K). When Tm was administered at E13.5, ∼50% of all the marked cells were Golgi cells (igl interneurons), whereas only ∼13 and ∼16% were basket and stellate cells (molecular layer interneurons), respectively (Fig. 4E,K). When Tm was administered at E14.5, the majority of marked cells continued to be Golgi cells (∼49%), but ∼25% of the fate-mapped (YFP+) cells were now basket cells and only ∼10% were stellate cells (Fig. 4F,K). In contrast, when Tm was administered at E17.5, only ∼14% of marked cells were Golgi cells, the majority were basket cells (∼36% of all YFP+ cells), and ∼22% were stellate cells (Fig. 4G,K). Interestingly, ∼1% of YFP+ cells were candelabrum interneurons at this stage of marking (Fig. 4D,K). When Tm was administered at P0, only one labeled Golgi cell was observed in three mice examined (Fig. 4H,K) (n = 36 sections in 3 animals). The proportion of marked basket cells remained at ∼30%, whereas the majority of all marked cells were stellate interneurons (∼63%) with candelabrum cells at ∼2%. When Tm was administered at P1, the majority of all marked cells continued to be stellate cells (∼75%), basket cells were decreased to only ∼19%, and candelabrum cells to only ∼0.2% (Fig. 4I,K). Finally, at P4 and P7, the only interneurons labeled were stellate cells (∼89% of all YFP+ cells) (Fig. 4J,K) (data not shown). Our finding that the four GABAergic interneuron subtypes express Ascl1 in a temporal sequence that correlates with their final inside-to-outside position is reminiscent of the pattern observed when three of the interneuron types were previously birth dated in rodents at more limited time points (Altman and Bayer, 1997; Leto et al., 2009). By extrapolation, our results indicate that candelabrum interneurons likely follow this same rule.
Early-born cerebellar cortex inhibitory interneurons express Ascl1 as they leave the cell cycle and ventricular zone
We next tested whether cerebellum interneurons, like Pcs, are marked with Ascl1CreER GIFM when they are born. Interestingly, when BrdU was administered 12 h before administering Tm at E13.5, E14.5, P2, or P7, double IHC staining for BrdU and βgal at P21 revealed that some stellate (Fig. 5A,A′), basket (Fig. 5B,B′), and Golgi (Fig. 5C,C′) cells retained BrdU and thus were birth dated with Ascl1CreER GIFM. Quantification of cells labeled when Tm was administered at E13.5 or E14.5 demonstrated that ∼25 and ∼22%, respectively, of Ascl1CreER GIFM-marked cerebellar interneurons retained BrdU. Furthermore, ∼45% of the BrdU- and βgal-labeled interneurons were Golgi cells (located in the igl) and the remainder were basket and stellate cells (found in the ml). Therefore, all three interneuron types are born as early as E13, with an apparent preference for Golgi cells (Fig. 5D). In contrast to interneurons labeled at E13.5/E14.5, only ∼5 and ∼12% of interneurons labeled by Tm administration on P2 and P7 were double positive for BrdU and βgal, respectively. It is possible that the lower percentage of BrdU-labeled interneurons after birth compared with midembryonic stages is a result of the different Tm administration techniques. For embryonic stages, mothers were administered Tm by oral gavage, whereas postnatal pups received Tm via subcutaneous injection which results in faster (6–12 h) activation of Cre (Nguyen et al., 2009). The latter route of administration would therefore result in the active form of Tm reaching the cerebellum closer to the time of Tm administration, and thus closer to the time of BrdU incorporation (within 18–24 h rather than 24–30 h). If Ascl1 expression is initiated later than 18 h after DNA synthesis, then the active form of Tm would be available for only a subset of the time when Ascl1 is expressed.
Figure 5.
A population of cerebellar interneurons is born during embryonic development. BrdU was administered 12 h before Tm administration at E13.5, E14.5, P2, and P7. A, Double-labeling immunohistochemistry in the outer ml for BrdU (red) and parvalbumin (green) shows some stellate cells are born at E14.5. A′, Double-labeling immunohistochemistry for βgal (red) and BrdU (green) shows an Ascl1CreER GIFM fate-mapped stellate cell born embryonically. B, Double-labeling immunohistochemistry in the inner ml for BrdU (red) and parvalbumin (green) shows some basket cells are born at E14.5. B′, Double-labeling immunohistochemistry for βgal (red) and BrdU (green) shows an Ascl1CreER GIFM fate-mapped basket cell born embryonically. C, Double-labeling immunohistochemistry in the igl for BrdU (green) and neurogranin (red) shows some Golgi cells are born at E14.5. C′, Double-labeling immunohistochemistry for βgal (red) and BrdU (green) shows an Ascl1CreER GIFM fate-mapped Golgi cell born embryonically. D, Quantification of the number of BrdU and βgal double-positive interneurons marked at each time point with Ascl1CreER GIFM is shown (mean ± SEM; n = 3; unpaired t test). Pcl, Purkinje cell layer; pia, pial surface. Scale bar, 20 μm.
We next examined whether Ascl1 is expressed in proliferating precursors after E13.5. IHC showed that, at E14.5, E17.5, and P1, few Ascl1+ cells were Ki67+ (proliferating) (Fig. 1M–O, pink arrowheads). Using Ascl1CreER GIFM, we found that, when Tm was administered at E13.5 or E14.5, only a few marked cells incorporated BrdU 24 h after Tm administration (Fig. 1R–R″, pink arrowheads) (data not shown). When Tm was administered at E17.5 or P1, we also found that very few marked cells incorporated BrdU 24 h later or were positive for Ki67 (Fig. 1T–T″) (data not shown). When the proliferative state of fate-mapped cells was analyzed using Ki67 (which labels most of the cell cycle) 3 d after Tm was administered at E17.5, we also observed very few βgal+ cells that were Ki67+ (Fig. 1S′). Furthermore, we found that the majority of βgal+ cells expressed Pax2, a postmitotic interneuron progenitor marker (Fig. 1S″), although some expressed the oligodendrocyte progenitor marker Olig2 (data not shown) or the astrocyte progenitor marker Sox9 (data not shown).
In summary, our data show that each class of cerebellar interneuron precursor expresses Ascl1 in a distinct temporal pattern that correlates with the final inside-to-outside position the cells will attain. Additionally, our studies suggest that Golgi cells preferentially come from vz progenitors that express Ascl1, whereas stellate and basket cells are primarily, but not solely, derived from Ascl1-expressing progenitors in the wm. Ascl1CreER GIFM can also be used for genetically birth dating cerebellar interneurons at embryonic stages, and possibly postnatal stages.
Bergmann glia, astrocyte, and oligodendrocyte precursors express Ascl1 in unique temporal patterns
Glial cells play a critical role in development and functioning of the cerebellar microcircuitry. The Bg are a specialized type of astrocytes that provide a framework for Pc dendrite outgrowth and synapse formation (Yamada et al., 2000; Lordkipanidze and Dunaevsky, 2005), as well as the physical structure upon which gcs migrate to the igl (Rakic, 1971). Additionally, ablation of oligodendrocytes in the mouse cerebellum during the first postnatal week results in disorganization of the cortical cytoarchitecture, with abnormal Pc dendritic arborization and axonal fasciculation, as well as impaired migration of interneurons (Mathis et al., 2003). There is also growing evidence that astrocytes function in regulation of synaptic transmission in the CNS (Ullian et al., 2001; Doetsch, 2003; Newman, 2003; Lin and Bergles, 2004).
Fate-mapping studies using either postnatal infection with replication-defective retroviruses to mark cerebellar astrocyte and oligodendrocyte precursors, or Pft1aCre mice to mark all vz-derived cells, have shown that these glial cells arise from the vz and then migrate through the wm and continue to proliferate during early postnatal stages (Zhang and Goldman, 1996a,b; Milosevic and Goldman, 2002; Hoshino et al., 2005). A recent study, however, concluded that cerebellar oligodendrocytes arise from outside the cerebellum (Grimaldi et al., 2009). Finally, previous data using an Ascl1-CreER BAC transgene raised the question of whether Ascl1-expressing cerebellar progenitors give rise only to oligodendrocytes and not to astrocytes or Bg (Kim et al., 2008).
Since we observed Ascl1 expression in both the vz and wm at E17.5, cells derived from either region could account for our observed marking of glia with Ascl1CreER GIFM after E16.5. At earlier stages, Ascl1CreER must mark precursors as they leave the vz region. Since the three different types of cerebellar glia have distinct morphologies, we used the R26RYFP reporter line as one means to identify the three types of glia based on their morphology. Interestingly, we found that Bg were marked with Ascl1CreER GIFM only when Tm was administered at E13.5, whereas oligodendrocytes and astrocytes were labeled at all stages after E12.5. The identity of Bg was confirmed by their distinct morphology (small cell bodies and long radial fibers with glial endfeet at the pial surface), location within the Pc layer, and expression of GFAP (Fig. 6A,A′). Oligodendrocytes were identified by their location within the wm, their unique morphology (flat cell bodies with parallel oriented radial processes), and coexpression of CAII (Fig. 6B,B′), whereas astrocytes were identified by their small bodies and many processes, as well as expression of GFAP (Fig. 6C,C′).
Figure 6.
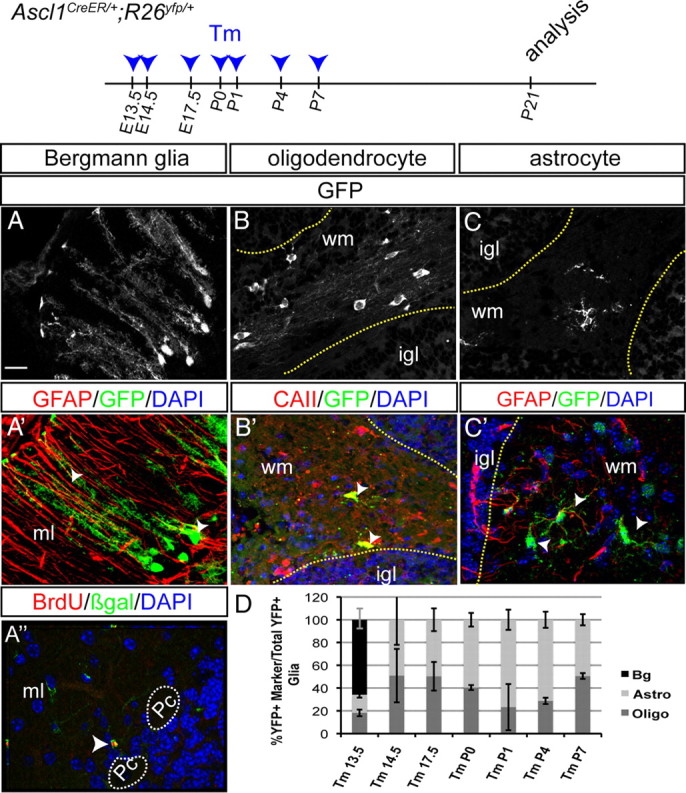
Temporal fate mapping of cerebellar glia with Ascl1CreER GIFM reveals marking of Bergmann glia only at one time point and other glia during their proliferative phase. Ascl1CreER GIFM marking of Bergmann glia (A–A″), oligodendrocytes (B, B′), and astrocytes (C, C′) is shown. A′, Double-labeling immunohistochemistry for GFAP (red) and YFP (green) confirms the identity of Bergmann glia. A″, BrdU was administered 12 h before Tm administration at E13.5. Double-labeling immunohistochemistry for BrdU (green) and βgal (red) shows Ascl1CreER GIFM and long-term BrdU-marked Bergmann glia. B′, The identity of oligodendrocytes is shown by double-labeling immunohistochemistry for CAII (red) and YFP (green). C′, Double-labeling immunohistochemistry for GFAP (red) and YFP (green) shows some Ascl1CreER GIFM-marked cells are astrocytes. D, The percentage of each type of cerebellar glia marked with Ascl1CreER GIFM at each time point during development is shown (mean ± SEM; n = 4; unpaired t test). Scale bar, 20 μm.
Next, we determined whether there is a temporal bias as to when cerebellum oligodendrocytes and astrocytes are marked with Ascl1CreER GIFM (Fig. 6D). We found that, when Tm was administered at E13.5, astrocytes and oligodendrocytes comprised only ∼15 and ∼18%, respectively, of the total YFP+ glial population and the rest of the marked glia were Bg. In Ascl1CreER GIFM mice, when Tm was administered at E14.5 or E17.5, astrocytes and oligodendrocytes were equally represented in the YFP+ glial population (Fig. 6D). At P0, ∼40% of all marked glia were oligodendrocytes and the rest were astrocytes. Tm administration at P1 and P4 resulted in a greater proportion of cerebellar astrocytes being marked (∼71–76% of all YFP+ glia) than oligodendrocytes (∼23–28%). Finally, at P7, astrocytes and oligodendrocytes were marked in similar numbers. These data indicate that the Ascl1-expressing cells in the vz and wm produce all cerebellar glial cell types in a unique temporal pattern during cerebellar development.
Since Bergmann glia were marked only when Tm was administered at E13.5, we tested whether Bergmann glia are marked with Ascl1CreER GIFM around the time of their last cell division. Strikingly, when BrdU was administered 12 h before administration of Tm to Ascl1CreER GIFM mice at E13.5, a large percentage of YFP+ Bg in P21 mice was found to be double positive for BrdU and βgal (∼30%) (Fig. 6A″). In contrast to Bg, Pcs, and interneurons, no YFP+ oligodendrocytes or astrocytes were found to retain BrdU at P21.
Conditional ablation of Ascl1 in the cerebellum results in a reduction in cerebellar interneurons and oligodendrocytes and increase in astrocytes
Given our finding that Ascl1 is transiently expressed in all vz-derived cell types, including interneuron and glial precursors in the wm after birth, it is possible that the phenotype reported for Ascl1-null mutants at birth does not reflect the entire requirement for Ascl1 during cerebellum development. Since Ascl1-null mutants die at birth (Guillemot et al., 1993), we used a recently generated Ascl1loxP conditional allele (Pacary et al., 2011) to remove Ascl1 specifically in the cerebellum. We reasoned that such mice should be viable and therefore allow us to identify all mature cerebellar cell types. Indeed, using the vz-specific Ptf1aCre line (Kawaguchi et al., 2002) to selectively delete Ascl1 in vz-derived cells of the cerebellum (and a subset of cells in the hindbrain and spinal cord), we found that Ptf1aCre/+; Ascl1loxP/loxP conditional knock-outs (Ptf1a-Ascl1 ckos) were viable. The conditional mutants also did not have any obvious locomotor defects. The deleted Ascl1loxP allele (Ascl1Δ) functions as a null allele, since similar to Ascl1-null mutants (Grimaldi et al., 2009), we found that Ascl1CreER/Δ E18.5 embryos had a decrease in postmitotic Pax2+ cells (cerebellar interneuron progenitors), and proliferating Olig2+ oligodendrocytes (data not shown).
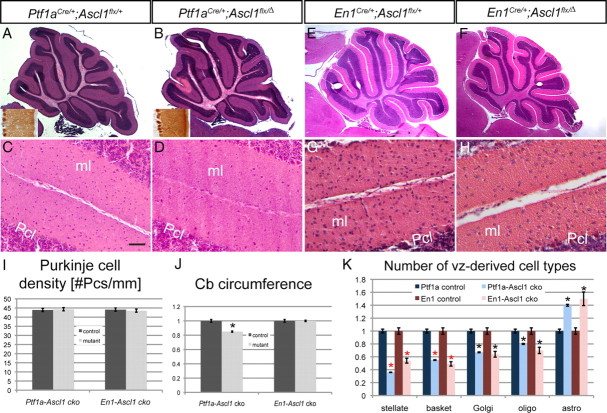
We first determined whether the overall size and foliation pattern was altered in mice lacking Ascl1. Hematoxylin and eosin-stained sagittal sections of P21 Ptf1a-Ascl1 ckos revealed a normal foliation pattern and basic cortical laminar organization (Fig. 7A,B). However, the overall size of the cerebellum appeared slightly reduced in Ptf1a-Ascl1 ckos compared with littermate controls, and there was a clear reduction in the number of interneurons in the ml (Fig. 7C,D). Quantification of the circumference of the cerebellum in a series of sections across the M-L axis revealed that the cerebellum of Ptf1a-Ascl1 cko mutants was indeed reduced by ∼15% (average length of 36.5 mm in controls compared with 31.3 mm in mutants; p < 0.01) (Fig. 7J). Quantification of Pc density and the overall thickness and density of cells in the igl revealed no difference between Ptf1a-Ascl1 ckos and controls (Fig. 7A,B,I). Since the length of the Pc layer is reduced but the density of Pcs is normal, the total number of Pcs must be slightly reduced in Ptf1a-Ascl1 ckos. The overall morphology of Pcs appeared normal based on IHC labeling of Pcs with a calbindin antibody in Ptf1a-Ascl1 ckos (Fig. 7A,B, inset).
Figure 7.
Conditional deletion of Ascl1 in the cerebellum differentially affects the number of interneurons and glia in the adult. Hematoxylin and eosin (H&E)-stained sections show smaller cerebellum in a Ptf1a-Ascl1 cko (B) compared with a littermate control (A). The insets in A and B are immunostaining for calbindin showing no difference in the overall morphology of Pcs. H&E-stained sections of En1-Ascl1 ckos (F) show a similar cellular phenotype to Ptf1a-Ascl1 ckos (A). Higher magnification images show severe reductions in the number of ml interneurons in both ckos (D, H) compared with littermate controls (C, G). I, Quantification of the number of Purkinje cells per millimeter in Ptf1a-Ascl1 and En1-Ascl1 ckos and littermate controls. J, Quantification of the average cerebellar circumference in Ptf1a-Ascl1 and En1-Ascl1 ckos and littermate controls with controls set as 1 shows significantly smaller cerebellum size in Ptf1a-Ascl1 ckos (p = 0.01) but not En1-Ascl1 ckos. K, Quantification of the GABAergic interneurons and glia in Ptf1a-Ascl1 and En1-Ascl1 ckos and littermate controls with average control numbers set as 1 (mean ± SEM; Ptf1a-Ascl1 cko, n = 3; Ptf1a-Ascl1 control, n = 3; En1-Ascl1 cko, n = 3; En1-Ascl1 control, n = 3; unpaired t test; the red asterisk depicts p < 0.003; the black asterisk, p < 0.05). Scale bar: A, B, E, F, 100 μm; C, D, G, H, 20 μm.
We next tested whether the three types of cortical interneurons derived from Pax2-expressing immature interneurons around birth are equally affected by loss of Ascl1 throughout cerebellum development. Interestingly, we found that, in Ptf1a-Ascl1 ckos, the interneurons located in the outer ml (stellate cells) were reduced to the greatest extent (by ∼64%), and the next most affected interneurons were the basket cells (reduced by ∼45%), with GABAergic Golgi interneurons in the igl being reduced the least (∼33%) (Fig. 7K). Thus, although Pax2+ cells are greatly diminished in Ascl1-null mutants at birth, before the time when the majority of stellate interneurons are generated, it is the stellate interneurons that are ultimately depleted the most in Ptf1a-Ascl1 ckos.
We next determined whether the balance of cerebellar oligodendrocytes and astrocytes was altered in Ptf1a-Ascl1 ckos, as null mutants were reported to have a significant decrease in Olig2+ oligodendrocyte progenitors and slight increase in Sox9+ astrocyte precursors at birth. Quantification of the number of oligodendrocytes in Ptf1a-Ascl1 ckos at P21 revealed a ∼25% reduction in Ptf1a-Ascl1 ckos compared with controls (Fig. 7K), and a ∼30% increase in the number of astrocytes in Ptf1a-Ascl1 cko mutants (Fig. 7K). In summary, Ascl1 expression in progenitors is necessary for the generation of the proper relative proportions of GABAergic interneurons and the two major types of glia. Furthermore, the interneurons that express Ascl1 latest in development are preferentially dependent on Ascl1, and Pcs appear to have only a minor requirement for Ascl1.
We reasoned that one possibility for the mild phenotype in early-born cerebellum neurons in Ptf1a-Ascl1 ckos could be that the Ptf1aCre/+ allele deletes Ascl1 after it begins to be expressed, and thus the phenotype of Ptf1a-Ascl1 ckos could represent a hypomorphic phenotype. Indeed, analysis of lacZ expression in E15.5 Ptf1aCre/+;RosaloxP-STOP-lacZ/+ embryos revealed that postmitotic cells were primarily marked, with only a few scattered marked cells in the vz (data not shown) (Pascual et al., 2007). We therefore generated another Ascl1 cko mutant using En1Cre, which deletes floxed genes in all cells of the cerebellum by E9 (Kimmel et al., 2000; Li et al., 2005). IHC analysis of the cerebellum primordium from E11 En1-Ascl1 ckos confirmed that Ascl1 protein was mostly absent at this stage (data not shown). Analysis of sagittal sections of P21 En1-Ascl1 ckos stained with hematoxylin and eosin, or using IHC with cell type-specific markers, however, revealed a similar cellular phenotype to Ptf1a-Ascl1 ckos (Fig. 7E–K) (data not shown). GABAergic basket and stellate interneurons were reduced by 51 and 46%, respectively, and Golgi interneurons were reduced by 30% (Fig. 7K). Quantification of glia in En1-Ascl1 ckos revealed that oligodendrocytes were reduced significantly (by 30%) and astrocytes increased by 34% compared with littermate controls (Fig. 7K). The quantification of the different cell types in the two Ascl1 ckos was similar except that interestingly stellate interneurons appeared to be more reduced in Ptf1a-Ascl1 ckos than in En1-Ascl1 ckos. Moreover, we found no difference in the circumference of the cerebellum of En1-Ascl1 ckos compared with littermate controls (27.4 and 26.5 mm, respectively). Our finding that Ascl1 ckos lacking one copy of the related bHLH gene Ptf1a have a stronger phenotype than mice lacking only Ascl1 in the cerebellum suggests there is overlap in the functions of the two genes in regulating generation of Pcs and stellate interneurons.
To address whether other bHLH genes could also contribute to the milder cerebellar phenotype in Ascl1 ckos compared with Ptf1a-null mutants that lack all GABAergic cell types (Hoshino et al., 2005; Pascual et al., 2007), we analyzed Neurogenin1 (Ngn1) and Ngn2 expression in Ascl1-null mutants at E12.5. As previously reported (Zordan et al., 2008), Ngn2 but not Ngn1 was expressed in the same region of the cerebellum as Ascl1 after E11.5. Furthermore, no obvious difference in expression of either gene was detected in Ascl1-null mutants (data not shown), suggesting only Ngn2 could compensate for Ascl1 loss. Additionally, we examined Delta1 and Delta3 expression at E12.5 and found that expression of both genes was only slightly decreased in some Ascl1-null mutants throughout M-L axis (data not shown). It is possible that Ngn2 prevents a major downregulation of Delta1/3 expression in the cerebellar vz of Ascl1-null mutants. Together, our two Ascl1 ckos demonstrate that Ascl1 is preferentially required in the cerebellum for production of late-born GABAergic interneurons and to produce the correct balance of oligodendrocytes and astrocytes.
In the absence of Ascl1, some cerebellum vz progenitors acquire a gcp phenotype
Curiously, fate mapping in Ptf1aCre/Cre;RosalacZ/+ mice revealed that a greater proportion of fate-mapped cells reside in the egl of homozygous Ptf1a-null mutants than in Ptf1aCre/+;RosalacZ/+ heterozygous controls (Pascual et al., 2007), suggesting a switch in fate of vz progenitors to url progenitors. One potential problem with interpreting the results of the Ptf1aCre/Cre fate-mapping study is that, in the homozygous mutants, the level of Cre protein should be double that in Ptf1aCre/+ heterozygotes, and thus the marking of more cells in the egl of Ptf1aCre/Cre mice could be due to more cells initially being marked (i.e., cells with a low level of Cre in heterozygotes might not be marked, whereas the same cells in homozygotes will express a sufficient level of Cre to induce recombination of the reporter allele). To determine whether the fate of Ascl1-expressing cells is altered in the absence of Ascl1 function, we performed a fate-mapping study in Ascl1CreER/Δ; RosalacZ/+ animals.
Strikingly, we found that when Tm was administered at E10.5, E11.5, or E12.5 to Ascl1CreER/Δ;RosalacZ/+ animals and the cerebellum analyzed at E17.5, not only were Pcs marked and cn interneurons (only at E10.5 marking), but also cells located within the egl were marked (Fig. 8A,B″). Consistent with GIFM using an Atoh1-CreER transgene to mark gcps at these time points of Tm administration (Machold and Fishell, 2005), we found that most of the marked gcps were located in the anterior half of the cerebellum at E17.5 (location of the earliest generated gcps) (Fig. 8B). Double IHC staining for βgal and Pax6, a marker for gcps, confirmed that the Ascl1CreER fate-mapped cells located in the egl were gcps (Fig. 8B″). Interestingly, we did not find any Tbr2+ cn projection neurons marked in Ascl1CreER/Δ;RosalacZ/+ mutants (data not shown). Furthermore, when Tm was administered at E13.5 or E14.5 to Ascl1CreER/Δ;RosalacZ/+ animals and the cerebellum analyzed at E17.5, no marked cells were detected in the egl, but as expected the population of Pax2+ marked cells was reduced (Fig. 8C,D″). Additionally, we used the Ptf1aCre allele to fate map vz-derived cells in Ascl1CreER/CreER-null animals in the absence of Tm administration. Compared with Ptf1aCre/+;RosalacZ/+ animals, nearly twice as many gcps were marked in Ptf1aCre/+;Ascl1CreER/CreER;RosalacZ/+ mutants (Fig. 8E,F) (for Ptf1aCre/+;RosalacZ/+ fate mapping, see also Pascual et al., 2007). Quantification of the number of Ptf1aCre fate-mapped gcps showed that ∼12 gcps were marked per 100 μm of egl in Ascl1+/+ E18.5 embryo cerebellum sections compared with ∼20 gcps per 100 μm of egl in Ascl1-null embryos. Finally, Atoh1 expression was used to mark the url and gcps at E12.5 in Ascl1CreER/Δ;RosalacZ/+ animals, but no major difference in expression was detected compared with controls (data not shown). It is possible that, since only a small number of gcps is labeled in Ascl1CreER/Δ;RosalacZ/+ embryos, any change in Atoh1 expression would be subtle and difficult to detect. Together, the results of our fate-mapping studies suggest that Ascl1 is required before E13 to ensure vz cells do not enter the url.
Discussion
Ascl1CreER GIFM is a powerful tool for characterizing all cell types derived from the cerebellar vz
Our Ascl1CreER GIFM study demonstrates that Ascl1 is transiently expressed by precursors of all cell types thought to be derived from the cerebellar vz. One significant difference between the recently described Ascl1-CreER BAC transgenic line (Kim et al., 2008) and the CreER knock-in allele is that Ascl1CreER marks all the GABAergic interneurons (Golgi, stellate, basket, and candelabrum), as well as Bg and astrocytes, in addition to Pcs and oligodendrocytes. A second significant difference is that, except for Bg, the Ascl1CreER allele marks progenitors throughout the period of their generation in the vz and wm. Furthermore, Pcs and embryonic interneurons are marked around the time of their last of cell division. Interestingly, Ascl1CreER also birth dates Bg, but only those in the vz leaving the cell cycle at ∼E14 and not those that later divide in the Pc layer (Shiga et al., 1983). The power of our Ascl1CreER GIFM approach is highlighted by comparison to previous fate mapping of the cerebellar vz with Ptf1aCre (Hoshino et al., 2005; Pascual et al., 2007) where the cumulative nature of cell marking obscured the location and relationship between temporal marking and final position of cells.
Purkinje cells with similar birth dates populate distinct A-P and M-L positions
The inherent Ascl1 expression in newly differentiating cells enabled us to generate the first genetic temporal birth-dating fate map of Pcs. We found that cohorts of Pcs born around the same time occupy distinct positions along the A-P and M-L axes of the adult cerebellum. The finding that Pcs successively settle in M-L parasagittal stripes and A-P zones raises the possibility that distinct functional properties of Pcs could be specified in progenitors as they leave the vz and express Ascl1. There is ample evidence for distinct functional domains within the cerebellum. The spinocerebellar tract projects only to vermis lobules I–V and VIII/IX. Afferents resolve into parasagittal bands after birth, which are mirrored by parasagittal Pc gene expression patterns (Larouche and Hawkes, 2006; Sillitoe and Joyner, 2007; Sillitoe et al., 2010). Information encoded in Pcs based on their birth date and transient expression of Ascl1 could therefore contribute to the complexity of Pc molecular identity and circuitry in the adult.
GABAergic cerebellar interneurons are sequentially generated from inside to outside
Our GIFM results provide the most detailed description of the temporal pattern of interneuron generation in the mouse cerebellum. Previous studies have suggested that cerebellar interneurons are generated mainly during early postnatal stages in rodents (Zhang and Goldman, 1996a). [3H]Thymidine birth-dating studies in the rat indicated sequential generation of cortical interneurons, Golgi (E14–P3), basket (P3–P9), and stellate (P5–P12) (Altman and Bayer, 1997), similar to the sequence seen in monkey (Rakic, 1973). BrdU birth-dating and retroviral labeling studies in mouse and rat suggested that the vast majority of interneurons come from proliferating progenitors in the wm thought to be derived from the vz (Zhang and Goldman, 1996a,b; Milosevic and Goldman, 2002, 2004; Leto et al., 2009). Additionally, it was proposed that the positional (inside-to-outside) and neurochemical identities of GABAergic interneurons are specified within the wm (Leto et al., 2009). In our study, we demonstrate that the deepest located interneurons of the cn are generated first from the vz (∼E11), followed primarily by the Golgi subpopulation of interneurons (∼E14–E18). At early postnatal stages (until ∼P7), the remaining ml interneurons are then generated in the wm (basket and candelabrum before stellate). Our results raise the question of whether the progenitors in the vz and wm constitute separate progenitor populations, or whether a common progenitor initially generates all three interneuron types in the vz and then moves to the wm where it preferentially generates ml interneurons. It is tempting to speculate that the small population of early-born ml interneurons lay down a skeleton of the ultimate interneuron circuit, and possibly provide cues for the later-born interneurons of the same type to differentiate in the correct location.
Ascl1 is preferentially required for generation of the late-born GABAergic cerebellar neurons
By generating two cerebella-specific Ascl1 cko mutants that survive to adulthood, we discovered that, unlike Ptf1a, Ascl1 is necessary to generate only a subset of vz-derived cell types. Ascl1 is required to generate a full complement of GABAergic interneurons, as well as the appropriate ratio of oligodendrocytes and astrocytes (En1Cre/+; Ascl1loxP/loxP mutants). Interestingly, the ml interneurons appear to be the most sensitive to loss of Ascl1, indicating that the later the birth of a neuron the greater its dependence on Ascl1. Furthermore, there appears to be an overlap in function between Ascl1 and Ptf1a as there is a greater reduction in stellate interneurons and a slight reduction in Pcs when one allele of Ptf1a is removed in Ascl1 ckos (Ptf1aCre/+; Ascl1loxP/loxP mutants). Our results are particularly important since an analysis of Ascl1-null mutants showed a large reduction in Pax2+ (postmitotic) cells at birth, which based on birth-dating studies should primarily be Golgi interneuron precursors generated from the vz. However, we uncovered that it is actually the interneurons born primarily after birth in the wm that appear to be preferentially reduced in the absence of Ascl1. Since the number of proliferating cerebellar cells was reduced in Ascl1-null mutants at E18.5 (Grimaldi et al., 2009), one role for Ascl1 might be to promote proliferation. Alternatively, since few proliferating cells express Ascl1 after E10.5, Ascl1 might inhibit differentiation. Therefore, in Ascl1 mutants, the pool of proliferating progenitors would be reduced prematurely.
Our phenotype analysis revealed a decrease in oligodendrocytes and an increase in astrocytes in adult Ascl1 ckos, indicating that Ascl1 has opposite effects on these two glia cell types. If a common progenitor gives rise to both glial cell types, then one possibility is that Ascl1 promotes an oligodendrocyte fate and inhibits an astrocyte fate. Consistent with this, previous studies in the cerebral cortex (Parras et al., 2004, 2007) and spinal cord (Sugimori et al., 2007) showed that Ascl1 is required to specify oligodendrocyte precursors and to repress the astrocytic fate. However, a recent study provided evidence that the majority of cerebellar oligodendrocytes are generated from outside the cerebellum (Grimaldi et al., 2009), arguing against a common progenitor. As we found that Ascl1 is expressed in proliferating oligodendrocyte (Olig2+) and astrocyte (Sox9+) progenitors, our mutant study indicates that Ascl1 promotes oligodendrocyte and inhibits astrocyte precursor expansion.
Finally, our fate-mapping studies in Ascl1-null mutants using Ptf1aCre and Ascl1CreER revealed that Ascl1 plays an additional role in ensuring proper segregation of cells to the vz rather than the url specifically during the time when Pcs are generated. In contrast to Ptf1a that plays an “all-or-nothing” role in generation of cerebellar GABAergic cell types, Ascl1 thus plays temporally dynamic and multifaceted roles in regulating the generation of the correct balance of all cerebellar inhibitory neurons and glia.
Model for constructing the cerebellar microcircuitry
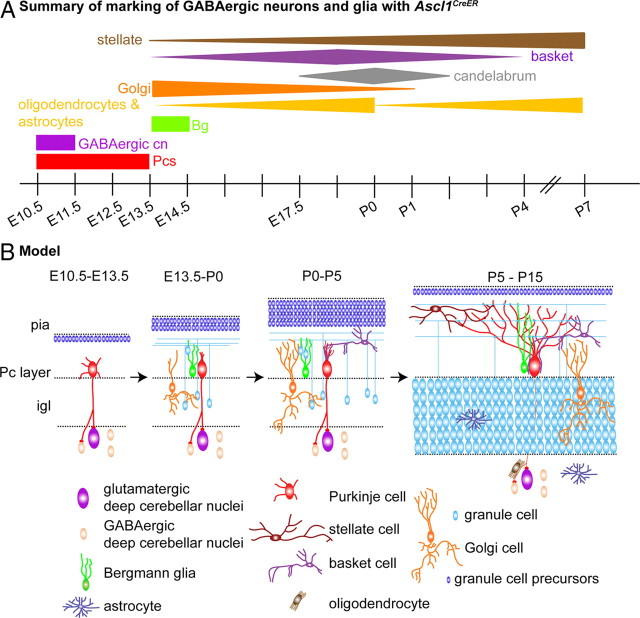
We propose a temporal model and rationale for building the local cerebellar circuitry (Fig. 9) that is based on our finding that all but the astrocytes and oligodendrocytes have distinct temporal origins as well as previous studies that delineated the temporal origins of url-derived neurons (Wang and Zoghbi, 2001; Machold and Fishell, 2005). The main efferent neurons are the first cells of the cerebellar circuitry to be established followed by the neurons that project to (GABAergic cells of the cn and Pcs between E10.5 and E12.5). Interestingly, the first Pcs to be born are those that settle in the lateral cerebellum where efferent Pcs project directly outside the cerebellum (to the vestibular nuclei). The next cell type generated is the Bergmann glia (∼E13.5), which are critical for gc migration (starting at ∼E17.5), Pc dendrite growth, and organization of ml interneuron axons (Rakic and Sidman, 1973; Hatten and Mason, 1990; Gao and Hatten, 1993; Lordkipanidze and Dunaevsky, 2005; Ango et al., 2008). Granule and Golgi cells, which form the glomeruli in the igl, are the next neurons generated. The basket interneurons, which synapse with Pc bodies and apical stem dendrites (Altman and Bayer, 1997), are then preferentially generated before the stellate interneurons that synapse with the later forming Pc distal dendrites (Altman and Bayer, 1997). While the exact role remains unknown of candelabrum interneurons in cerebellar circuitry, it is tempting to speculate that they contact Pc dendrites throughout the width of the ml, as their own projections run parallel with Pc dendrites and we found they express Ascl1CreER when the Pc dendrites begin to form. Finally, oligodendrocytes and astrocytes are primarily generated after birth, once their target cells are in place. Thus, there is a predictable sequence of generation of cells in the cerebellum, with target neurons being born before their regulatory cells. This is in contrast to mossy fiber neurons that are born long before their targets, the gcs, have formed the igl.
Figure 9.
Model for the temporal generation of the cells that form the cerebellar local circuitry. A, Summary of the time points at which vz-derived cells are preferentially marked using Ascl1CreER GIFM. The thickness of the line indicates the relative number of each cell type marked at different time points. B, During early embryogenesis (E10.5–E13.5), Pcs and neurons of the cn are born and begin to make contacts. During mid and late embryogenesis (E13.5–P0), a population of Bg and Golgi interneurons are born. Bg migrate to their final position within the Pc layer to guide newly differentiated gcs toward the igl. Golgi interneurons and gcs within the igl then form synaptic contacts within glomeruli. During early postnatal stages (P0–P5), basket interneurons are primarily generated and contact Pc apical dendrites. Finally, during late postnatal stages (P5–P15), stellate interneurons are mainly generated and occupy the outer region of the ml, and start making contacts with Pc dendrites. Astrocytes and oligodendrocytes are generated after birth and contact their target neurons. Thus, target neurons are generated before the neurons or glia that contact them are generated.
Footnotes
This work was supported by NIH Grant R01 MH085726 (A.L.J.). The Ascl1CreERT2 mice were created under a collaborative arrangement with the J. Johnson Laboratory (University of Texas Southwestern, Dallas, TX) and R. Reed Laboratory (Johns Hopkins University, Baltimore, MD). R. Reed and C. Leung provided the Ascl1 genomic DNA containing the homology arms, E.J.K. and J. Johnson generated the targeting construct, and A.L.J. and A.S. were responsible for targeting in ES cells and generating the targeted Ascl1CreERT2 mouse line. We are grateful to F. Berenshteyn and M. Bowers for help with mouse ES cell work, to D. Stephen for technical help, and to A. D. Garcia and E. Legue for insightful discussion and critical reading of this manuscript.
References
- Altman J, Bayer SA. Development of the cerebellar system in relation to its evolution, structure, and functions. New York: CRC; 1997. [Google Scholar]
- Ango F, Wu C, Van der Want JJ, Wu P, Schachner M, Huang ZJ. Bergmann glia and the recognition molecule CHL1 organize GABAergic axons and direct innervation of Purkinje cell dendrites. PLoS Biol. 2008;6:e103. doi: 10.1371/journal.pbio.0060103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barmack NH, Yakhnitsa V. Functions of interneurons in mouse cerebellum. J Neurosci. 2008;28:1140–1152. doi: 10.1523/JNEUROSCI.3942-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bastianelli E. Distribution of calcium-binding proteins in the cerebellum. Cerebellum. 2003;2:242–262. doi: 10.1080/14734220310022289. [DOI] [PubMed] [Google Scholar]
- Battiste J, Helms AW, Kim EJ, Savage TK, Lagace DC, Mandyam CD, Eisch AJ, Miyoshi G, Johnson JE. Ascl1 defines sequentially generated lineage-restricted neuronal and oligodendrocyte precursor cells in the spinal cord. Development. 2007;134:285–293. doi: 10.1242/dev.02727. [DOI] [PubMed] [Google Scholar]
- Casarosa S, Fode C, Guillemot F. Mash1 regulates neurogenesis in the ventral telencephalon. Development. 1999;126:525–534. doi: 10.1242/dev.126.3.525. [DOI] [PubMed] [Google Scholar]
- Celio MR. Calbindin D-28K and parvalbumin in the rat nervous system. Neuroscience. 1990;35:375–475. doi: 10.1016/0306-4522(90)90091-h. [DOI] [PubMed] [Google Scholar]
- Doetsch F. The glial identity of neural stem cells. Nat Neurosci. 2003;6:1127–1134. doi: 10.1038/nn1144. [DOI] [PubMed] [Google Scholar]
- Englund C, Kowalczyk T, Daza RA, Dagan A, Lau C, Rose MF, Hevner RF. Unipolar brush cells of the cerebellum are produced in the rhombic lip and migrate through developing white matter. J Neurosci. 2006;26:9184–9195. doi: 10.1523/JNEUROSCI.1610-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao WQ, Hatten ME. Neuronal differentiation rescued by implantation of Weaver granule cell precursors into wild-type cerebellar cortex. Science. 1993;260:367–369. doi: 10.1126/science.8469990. [DOI] [PubMed] [Google Scholar]
- Grimaldi P, Parras C, Guillemot F, Rossi F, Wassef M. Origins and control of the differentiation of inhibitory interneurons and glia in the cerebellum. Dev Biol. 2009;328:422–433. doi: 10.1016/j.ydbio.2009.02.008. [DOI] [PubMed] [Google Scholar]
- Guillemot F, Joyner AL. Dynamic expression of the murine Achaete-Scute homologue Mash-1 in the developing nervous system. Mech Dev. 1993;42:171–185. doi: 10.1016/0925-4773(93)90006-j. [DOI] [PubMed] [Google Scholar]
- Guillemot F, Lo LC, Johnson JE, Auerbach A, Anderson DJ, Joyner AL. Mammalian achaete-scute homolog 1 is required for the early development of olfactory and autonomic neurons. Cell. 1993;75:463–476. doi: 10.1016/0092-8674(93)90381-y. [DOI] [PubMed] [Google Scholar]
- Hashimoto M, Mikoshiba K. Mediolateral compartmentalization of the cerebellum is determined on the “birth date” of Purkinje cells. J Neurosci. 2003;23:11342–11351. doi: 10.1523/JNEUROSCI.23-36-11342.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatten ME, Mason CA. Mechanisms of glial-guided neuronal migration in vitro and in vivo. Experientia. 1990;46:907–916. doi: 10.1007/BF01939383. [DOI] [PubMed] [Google Scholar]
- Helms AW, Battiste J, Henke RM, Nakada Y, Simplicio N, Guillemot F, Johnson JE. Sequential roles for Mash1 and Ngn2 in the generation of dorsal spinal cord interneurons. Development. 2005;132:2709–2719. doi: 10.1242/dev.01859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoshino M, Nakamura S, Mori K, Kawauchi T, Terao M, Nishimura YV, Fukuda A, Fuse T, Matsuo N, Sone M, Watanabe M, Bito H, Terashima T, Wright CV, Kawaguchi Y, Nakao K, Nabeshima Y. Ptf1a, a bHLH transcriptional gene, defines GABAergic neuronal fates in cerebellum. Neuron. 2005;47:201–213. doi: 10.1016/j.neuron.2005.06.007. [DOI] [PubMed] [Google Scholar]
- Joyner AL, Zervas M. Genetic inducible fate mapping in mouse: establishing genetic lineages and defining genetic neuroanatomy in the nervous system. Dev Dyn. 2006;235:2376–2385. doi: 10.1002/dvdy.20884. [DOI] [PubMed] [Google Scholar]
- Kawaguchi Y, Cooper B, Gannon M, Ray M, MacDonald RJ, Wright CV. The role of the transcriptional regulator Ptf1a in converting intestinal to pancreatic progenitors. Nat Genet. 2002;32:128–134. doi: 10.1038/ng959. [DOI] [PubMed] [Google Scholar]
- Kim EJ, Leung CT, Reed RR, Johnson JE. In vivo analysis of Ascl1 defined progenitors reveals distinct developmental dynamics during adult neurogenesis and gliogenesis. J Neurosci. 2007;27:12764–12774. doi: 10.1523/JNEUROSCI.3178-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EJ, Battiste J, Nakagawa Y, Johnson JE. Ascl1 (Mash1) lineage cells contribute to discrete cell populations in CNS architecture. Mol Cell Neurosci. 2008;38:595–606. doi: 10.1016/j.mcn.2008.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EJ, Ables JL, Dickel LK, Eisch AJ, Johnson JE. Ascl1 (Mash1) defines cells with long-term neurogenic potential in subgranular and subventricular zones in adult mouse brain. PLoS One. 2011;6:e18472. doi: 10.1371/journal.pone.0018472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimmel RA, Turnbull DH, Blanquet V, Wurst W, Loomis CA, Joyner AL. Two lineage boundaries coordinate vertebrate apical ectodermal ridge formation. Genes Dev. 2000;14:1377–1389. [PMC free article] [PubMed] [Google Scholar]
- Lainé J, Axelrad H. The candelabrum cell: a new interneuron in the cerebellar cortex. J Comp Neurol. 1994;339:159–173. doi: 10.1002/cne.903390202. [DOI] [PubMed] [Google Scholar]
- Larouche M, Hawkes R. From clusters to stripes: the developmental origins of adult cerebellar compartmentation. Cerebellum. 2006;5:77–88. doi: 10.1080/14734220600804668. [DOI] [PubMed] [Google Scholar]
- Legué E, Joyner AL. Genetic fate mapping using site-specific recombinases. Methods Enzymol. 2010;477:153–181. doi: 10.1016/S0076-6879(10)77010-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leto K, Bartolini A, Yanagawa Y, Obata K, Magrassi L, Schilling K, Rossi F. Laminar fate and phenotype specification of cerebellar GABAergic interneurons. J Neurosci. 2009;29:7079–7091. doi: 10.1523/JNEUROSCI.0957-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li JY, Joyner AL. Otx2 and Gbx2 are required for refinement and not induction of mid-hindbrain gene expression. Development. 2001;128:4979–4991. doi: 10.1242/dev.128.24.4979. [DOI] [PubMed] [Google Scholar]
- Li JY, Lao Z, Joyner AL. New regulatory interaction and cellular responses in the isthmic organizer region revealed by altering Gbx2 expression. Development. 2005;132:1971–1981. doi: 10.1242/dev.01727. [DOI] [PubMed] [Google Scholar]
- Lin SC, Bergles DE. Synaptic signaling between neurons and glia. Glia. 2004;47:290–298. doi: 10.1002/glia.20060. [DOI] [PubMed] [Google Scholar]
- Lordkipanidze T, Dunaevsky A. Purkinje cell dendrites grow in alignment with Bergmann glia. Glia. 2005;51:229–234. doi: 10.1002/glia.20200. [DOI] [PubMed] [Google Scholar]
- Machold R, Fishell G. Math1 is expressed in temporally discrete pools of cerebellar rhombic-lip neural progenitors. Neuron. 2005;48:17–24. doi: 10.1016/j.neuron.2005.08.028. [DOI] [PubMed] [Google Scholar]
- Mathis C, Collin L, Borrelli E. Oligodendrocyte ablation impairs cerebellum development. Development. 2003;130:4709–4718. doi: 10.1242/dev.00675. [DOI] [PubMed] [Google Scholar]
- Milosevic A, Goldman JE. Progenitors in the postnatal cerebellar white matter are antigenically heterogeneous. J Comp Neurol. 2002;452:192–203. doi: 10.1002/cne.10384. [DOI] [PubMed] [Google Scholar]
- Milosevic A, Goldman JE. Potential of progenitors from postnatal cerebellar neuroepithelium and white matter: lineage specified vs. multipotent fate. Mol Cell Neurosci. 2004;26:342–353. doi: 10.1016/j.mcn.2004.02.008. [DOI] [PubMed] [Google Scholar]
- Nakada Y, Hunsaker TL, Henke RM, Johnson JE. Distinct domains within Mash1 and Math1 are required for function in neuronal differentiation versus neuronal cell-type specification. Development. 2004;131:1319–1330. doi: 10.1242/dev.01008. [DOI] [PubMed] [Google Scholar]
- Newman EA. New roles for astrocytes: regulation of synaptic transmission. Trends Neurosci. 2003;26:536–542. doi: 10.1016/S0166-2236(03)00237-6. [DOI] [PubMed] [Google Scholar]
- Nguyen MT, Zhu J, Nakamura E, Bao X, Mackem S. Tamoxifen-dependent, inducible Hoxb6CreERT recombinase function in lateral plate and limb mesoderm, CNS isthmic organizer, posterior trunk neural crest, hindgut, and tailbud. Dev Dyn. 2009;238:467–474. doi: 10.1002/dvdy.21846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pacary E, Heng J, Azzarelli R, Riou P, Castro D, Lebel-Potter M, Parras C, Bell DM, Ridley AJ, Parsons M, Guillemot F. Proneural transcription factors regulate different steps of cortical neuron migration through Rnd-mediated inhibition of RhoA signaling. Neuron. 2011;69:1069–1084. doi: 10.1016/j.neuron.2011.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parras CM, Galli R, Britz O, Soares S, Galichet C, Battiste J, Johnson JE, Nakafuku M, Vescovi A, Guillemot F. Mash1 specifies neurons and oligodendrocytes in the postnatal brain. EMBO J. 2004;23:4495–4505. doi: 10.1038/sj.emboj.7600447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parras CM, Hunt C, Sugimori M, Nakafuku M, Rowitch D, Guillemot F. The proneural gene Mash1 specifies an early population of telencephalic oligodendrocytes. J Neurosci. 2007;27:4233–4242. doi: 10.1523/JNEUROSCI.0126-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pascual M, Abasolo I, Mingorance-Le Meur A, Martínez A, Del Rio JA, Wright CV, Real FX, Soriano E. Cerebellar GABAergic progenitors adopt an external granule cell-like phenotype in the absence of Ptf1a transcription factor expression. Proc Natl Acad Sci U S A. 2007;104:5193–5198. doi: 10.1073/pnas.0605699104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattyn A, Guillemot F, Brunet JF. Delays in neuronal differentiation in Mash1/Ascl1 mutants. Dev Biol. 2006;295:67–75. doi: 10.1016/j.ydbio.2006.03.008. [DOI] [PubMed] [Google Scholar]
- Rakic P. Neuron-glia relationship during granule cell migration in developing cerebellar cortex. A Golgi and electronmicroscopic study in Macacus rhesus. J Comp Neurol. 1971;141:283–312. doi: 10.1002/cne.901410303. [DOI] [PubMed] [Google Scholar]
- Rakic P. Kinetics of proliferation and latency between final cell division and onset of differentiation of cerebellar stellate and basket neurons. J Comp Neurol. 1973;147:523–546. doi: 10.1002/cne.901470407. [DOI] [PubMed] [Google Scholar]
- Rakic P, Sidman RL. Sequence of developmental abnormalities leading to granule cell deficit in cerebellar cortex of weaver mutant mice. J Comp Neurol. 1973;152:103–132. doi: 10.1002/cne.901520202. [DOI] [PubMed] [Google Scholar]
- Shiga T, Ichikawa M, Hirata Y. Spatial and temporal pattern of postnatal proliferation of Bergmann glial cells in rat cerebellum: an autoradiographic study. Anat Embryol (Berl) 1983;167:203–211. doi: 10.1007/BF00298511. [DOI] [PubMed] [Google Scholar]
- Sillitoe RV, Joyner AL. Morphology, molecular codes and circuitry produce the three dimensional complexity of the cerebellum. Annu Rev Cell Dev Biol. 2007;23:549–577. doi: 10.1146/annurev.cellbio.23.090506.123237. [DOI] [PubMed] [Google Scholar]
- Sillitoe RV, Vogel MW, Joyner AL. Engrailed homeobox genes regulate establishment of the cerebellar afferent circuit map. J Neurosci. 2010;30:10015–10024. doi: 10.1523/JNEUROSCI.0653-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soriano P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet. 1999;21:70–71. doi: 10.1038/5007. [DOI] [PubMed] [Google Scholar]
- Srinivas S, Watanabe T, Lin CS, William CM, Tanabe Y, Jessell TM, Costantini F. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev Biol. 2001;1:4. doi: 10.1186/1471-213X-1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugimori M, Nagao M, Bertrand N, Parras CM, Guillemot F, Nakafuku M. Combinatorial actions of patterning and HLH transcription factors in the spatiotemporal control of neurogenesis and gliogenesis in the developing spinal cord. Development. 2007;134:1617–1629. doi: 10.1242/dev.001255. [DOI] [PubMed] [Google Scholar]
- Ullian EM, Sapperstein SK, Christopherson KS, Barres BA. Control of synapse number by glia. Science. 2001;291:657–661. doi: 10.1126/science.291.5504.657. [DOI] [PubMed] [Google Scholar]
- Wang VY, Zoghbi HY. Genetic regulation of cerebellar development. Nat Rev Neurosci. 2001;2:484–491. doi: 10.1038/35081558. [DOI] [PubMed] [Google Scholar]
- Wildner H, Müller T, Cho SH, Bröhl D, Cepko CL, Guillemot F, Birchmeier C. dILA neurons in the dorsal spinal cord are the product of terminal and non-terminal asymmetric progenitor cell divisions, and require Mash1 for their development. Development. 2006;133:2105–2113. doi: 10.1242/dev.02345. [DOI] [PubMed] [Google Scholar]
- Yamada K, Fukaya M, Shibata T, Kurihara H, Tanaka K, Inoue Y, Watanabe M. Dynamic transformation of Bergmann glal fibers proceeds in correlation with dendritic outgrowth and synapse formation of cerebellar Purkinje cells. J Comp Neurol. 2000;418:106–120. [PubMed] [Google Scholar]
- Zhang L, Goldman JE. Generation of cerebellar interneurons from dividing progenitors in white matter. Neuron. 1996a;16:47–54. doi: 10.1016/s0896-6273(00)80022-7. [DOI] [PubMed] [Google Scholar]
- Zhang L, Goldman JE. Developmental fates and migratory pathways of dividing progenitors in the postnatal rat cerebellum. J Comp Neurol. 1996b;370:536–550. doi: 10.1002/(SICI)1096-9861(19960708)370:4<536::AID-CNE9>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- Zordan P, Croci L, Hawkes R, Consalez GG. Comparative analysis of proneural gene expression in the embryonic cerebellum. Dev Dyn. 2008;237:1726–1735. doi: 10.1002/dvdy.21571. [DOI] [PubMed] [Google Scholar]