Abstract
To explore the pathophysiological role of leptin in obesity-related hypertension, we examined cardiovascular phenotypes of transgenic skinny mice whose elevated plasma leptin concentrations are comparable to those seen in obese subjects. We also studied genetically obese KKAy mice with hyperleptinemia, in which hypothalamic melanocortin system is antagonized by ectopic expression of the agouti protein. Systolic blood pressure (BP) and urinary catecholamine excretion are elevated in transgenic skinny mice relative to nontransgenic littermates. The BP elevation in transgenic skinny mice is abolished by α1-adrenergic, β-adrenergic, or ganglionic blockers at doses that do not affect BP in nontransgenic littermates. Central administration of an α-melanocyte–stimulating hormone antagonist causes a marked increase in cumulative food intake but no significant changes in BP. The obese KKAy mice develop BP elevation with increased urinary catecholamine excretion relative to control KK mice. After a 2-week caloric restriction, BP elevation is reversed in nontransgenic littermates with the Ay allele, in parallel with a reduction in plasma leptin concentrations, but is sustained in transgenic mice overexpressing leptin with the Ay allele, which remain hyperleptinemic. This study demonstrates BP elevation in transgenic skinny mice and obese KKAy mice that are both hyperleptinemic, thereby suggesting the pathophysiological role of leptin in some forms of obesity-related hypertension.
Introduction
Obesity is defined as increased mass of adipose tissue and confers a higher risk of arterial blood pressure (BP) elevation or hypertension (1–4). On the other hand, weight reduction lowers BP in obese hypertensive subjects (5–8), suggesting an important association between energy homeostasis and BP. However, the mechanism for that association is poorly understood. Obesity-related hypertension may be secondary to insulin resistance and/or hyperinsulinemia (2, 3, 8). Several lines of evidence have also suggested that increased sympathetic nerve activity may contribute to the development of obesity-related hypertension (2, 6, 7).
The adipose tissue participates in the regulation of a variety of homeostatic processes as an endocrine organ that secretes many biologically active molecules such as FFA, adipsin, angiotensinogen, and TNF-α (9). Leptin is such an adipocyte-derived hormone that is involved in the regulation of food intake and body weight (10). It also increases the overall sympathetic nerve activity, which leads to a significant increase in energy expenditure (11–15). The biologic actions of leptin are thought to be mediated through the activation of leptin receptor that is expressed in the hypothalamus (16–19). We and others demonstrated that the hypothalamic arcuate nucleus is a primary site of the satiety effect of leptin (20, 21) and that its satiety effect is mediated at least partly by hypothalamic melanocortin system (22, 23).
Numerous studies have demonstrated that plasma leptin concentrations are elevated significantly in several models of rodent obesity and in human obesity in proportion to the degree of adiposity (24–26), suggesting the state of “leptin resistance” in obesity. Nevertheless, because of the potent pleiotropic actions of leptin, it is conceivable, though paradoxically, that hyperleptinemia may be involved in the pathogenesis of obesity and its related disorders. In this regard, a recent study reported a significant correlation between BP and plasma leptin concentrations in patients with essential hypertension (27), suggesting that leptin may play roles in the pathogenesis of obesity-related hypertension.
We have recently produced transgenic skinny mice overexpressing leptin under the control of the liver-specific promoter and demonstrated that chronic hyperleptinemia results in complete disappearance of adipose tissue for a long period (28). These mice also exhibit increased glucose metabolism and insulin sensitivity, accompanied by an activation of insulin signaling in the skeletal muscle and liver (28, 29). Accordingly, transgenic skinny mice will serve as the unique experimental model system with which to assess the long-term effects of leptin in vivo.
In this study, to explore the pathophysiological role of leptin in obesity-related hypertension, we examined the cardiovascular phenotypes of transgenic skinny mice overexpressing leptin. We also studied genetically obese KKAy mice with hyperleptinemia, which develop a maturity-onset obesity and diabetes syndrome and resistance to the satiety effect of leptin (30–32) as a consequence of the antagonism of hypothalamic melanocortin system by ectopic expression of the agouti protein (33, 34).
Methods
Animals.
Generation of transgenic skinny mice overexpressing leptin under the control of the human serum amyloid P component (SAP) promoter on the C57BL/6J background was reported elsewhere (28). Male transgenic skinny mice carrying 30 copies of the transgene were used in this study. Ten-week-old male leptin-deficient C57BL/6J ob/ob mice and their control C57BL/6J mice (10), and male KKAy mice and their control KK mice (35, 36), were purchased from Japan CLEA (Tokyo, Japan). We also crossed transgenic skinny mice with KKAy mice to obtain F1 animals with different genotypes on the C57BL/6J and KK hybrid background (29); they were nontransgenic mice without the Ay allele (+/+ mice), transgenic mice without the Ay allele (Tg/+ mice), nontransgenic mice with the Ay allele (Ay/+ mice), and transgenic mice with the Ay allele (Tg/+:Ay/+ mice). Animals were maintained in standard rat chow (CE-2, 352 kcal/100 g; Japan CLEA) on a 12-hour light/dark cycle. All experimental procedures were approved by the Kyoto University Graduate School of Medicine Committee on Animal Research.
Systolic BP and heart rate measurements.
Systolic BP and heart rate were measured in 12-week-old transgenic skinny mice and ob/ob mice, and KKAy mice and their age-matched control mice, by the tail-cuff method (PS-200; Riken-Kaihatsu, Tokyo, Japan) (37). At least 6 recordings were taken for each measurement. The systolic BP levels measured by the tail-cuff method correlated well with those by the direct measurement through carotid artery catheter as reported (37). Systolic BP and heart rate were measured in 24-week-old transgenic and nontransgenic littermates before and 2, 4, 6, 8, and 10 hours after an intraperitoneal injection of an α1-adrenergic blocker, bunazosin (15 μg/g body weight; Eizai Pharmaceuticals Inc., Tokyo, Japan), or a nonselective β-adrenergic blocker, propranolol (6 μg/g body weight; Sigma Chemical Co., St. Louis, Missouri, USA), and before and 15, 30, and 60 minutes after a single intraperitoneal injection of a ganglionic blocker, hexamethonium (3 μg/g body weight; Sigma Chemical Co.). We performed a single intracerebroventricular injection of SHU9119, a potent antagonist of α-melanocyte–stimulating hormone (α-MSH) (0.05 μg/5 μL per mouse) (Phoenix Pharmaceuticals, Mountain View, California, USA) (22, 23, 34) or artificial cerebrospinal fluid (aCSF) (15) through a 27-gauge stainless steel infusion cannula inserted into the lateral ventricle in mice under slight diethylether anesthesia, using the following coordinates: 4 mm anterior to the lamboidal suture, 2 mm lateral to the midline, and 2.5 mm vertically. Only the animals that showed increased food intake after the intracerebroventricular injection of SHU9119 were included in this study. The BP and heart rate were measured before and 3, 6, 9 hours after the injection at the onset of light phase.
Body weight and food intake measurements.
Body weights of transgenic skinny mice, ob/ob mice, KKAy mice, and their control mice at 12 weeks of age were measured. Cumulative food intake was measured simultaneously in 12-week-old transgenic and nontransgenic littermates 8 hours after a single intracerebroventricular injection of SHU9119 or aCSF, when fed ad libitum.
Blood and urinary parameter measurements.
Transgenic and nontransgenic littermates, KKAy and KK mice at 12 weeks of age were placed in metabolic cages that provide free access to tap water and food. Blood was sampled from the retro-orbital sinus of mice at 0900 hours. Plasma leptin concentrations in 12-week-old transgenic skinny mice, ob/ob mice, and KKAy mice and their control mice were determined using the RIA for mouse leptin (Linco Research Immunoassay, St. Charles, Missouri, USA). Serum osmolarity and hematocrit; serum concentrations of Na+, K+, and Cl+; serum creatinine; blood urea nitrogen; daily urine volume; urinary Na+, K+, and Cl+ excretion; and water intake were measured according to the standard methods. Daily urinary catecholamine excretion was determined by reverse-phase HPLC (15).
Caloric restriction experiments.
We observed that during a caloric restriction, hyperleptinemia are sustained in Tg/+:Ay/+ mice (29), indicating that Tg/+:Ay/+ mice provide the unique experimental model system to investigate the effect of leptin during fasting or food restriction. Based on the daily food intake of 12-week-old +/+, Tg/+, Ay/+, and Tg/+:Ay/+ mice, they were provided with 60% of the amount of food consumed regularly as described elsewhere (29). During a 2-week food restriction, body weight, plasma leptin concentrations, and systolic BP were measured.
Leptin injection experiments.
We examined the effect of continuous leptin administration on BP in ob/ob mice. On the first day of the experiment, recombinant mouse leptin (PeproTech EC, London, United Kingdom) (2.4 μg/g per day) or vehicle (saline) started to be infused into 12-week-old male ob/ob mice and wild-type control littermates using osmotic minipump (Alzet model 1003D; Alza Corp, Palo Alto, California, USA) delivering 1 μL/h (100-μL pump). Mice were anesthetized with pentobarbital (Nembtal, 50 mg/kg intraperitoneally), and an osmotic minipump was implanted subcutaneously in the midscapular region. During the 3-day infusion experiment, body weights, food intake, plasma leptin concentrations, and the tail-cuff systolic BP were measured.
We also examined the effect of chronic leptin administration on BPs in Ay/+ mice during the caloric restriction. Based on the daily food intake of 12-week-old Ay/+ mice, they were provided with 60% of the amount of food consumed regularly. On the first day of food restriction, recombinant mouse leptin (1.2 μg/g per day) or vehicle were infused into 12-week-old Ay/+ mice using an osmotic minipump (Alzet model 2002) implanted subcutaneously, delivering 0.5 μL/h (200-μL pump). During the 10-day food restriction, body weights, plasma leptin concentrations, and the tail-cuff systolic BPs were measured.
Statistical analysis.
Statistical significance was assessed by ANOVA with repeated measures analysis (Statview 4.01; Abacus Concepts Inc., Berkeley, California, USA), ANOVA with Bonferroni Dunn test, and Student’s t test, when applicable. All values were expressed as the mean ± SEM.
Results
Systolic BP and heart rate in transgenic skinny mice and ob/ob mice.
At 12 weeks of age, body weight of transgenic skinny mice was decreased significantly relative to nontransgenic littermates (20.3 ± 1.1 vs. 26.1 ± 0.9 g; n = 6; P < 0.01). On the other hand, leptin-deficient ob/ob mice developed marked obesity relative to nontransgenic wild-type mice (47.9 ± 1.1 vs. 26.1 ± 0.9 g; n = 6; P < 0.001). Plasma leptin concentrations were 60.6 ± 14.4 ng/mL in transgenic skinny mice and were approximately 10-fold higher than those in nontransgenic littermates (4.8 ± 0.6 ng/mL) (n = 10; P < 0.001). No significant amount of leptin was detected in ob/ob mice. These data are consistent with previous reports (24, 25, 28, 29).
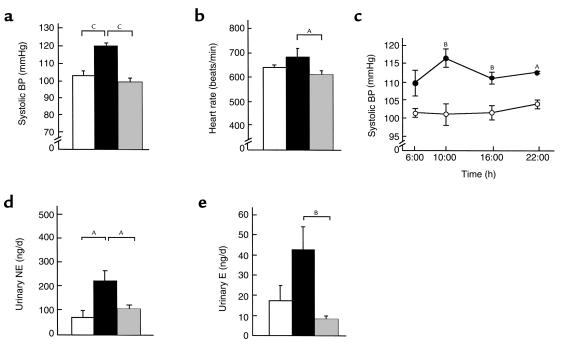
To elucidate the cardiovascular consequences of chronic hyperleptinemia, we measured systolic BP and heart rate in 12-week-old transgenic and nontransgenic littermates. At 0900 hours, the tail-cuff systolic BP was elevated significantly in transgenic skinny mice compared with nontransgenic littermates (120.0 ± 1.6 vs. 102.7 ± 2.7 mmHg; n = 12; P < 0.001) (Figure 1a). Systolic BP elevation in transgenic skinny mice was observed sustained throughout the day (n = 3) (Figure 1c). No significant difference in heart rate was noted between transgenic and nontransgenic littermates (684 ± 35 vs. 642 ± 10 beats per minute; n = 12) (Figure 1b). Heart rate tended to be decreased in ob/ob mice (610 ± 17 beats per minute) compared with their control mice.
Figure 1.
Profiles of transgenic skinny mice, ob/ob mice, and their control mice. Systolic BP (a) and heart rate (b) in 12-week-old transgenic skinny mice (black bars), ob/ob mice (shaded bars), and their control mice (open bars). (c) Daily profile of systolic BP in 12-week-old transgenic skinny mice (filled circles) and nontransgenic littermates (open circles). Urinary NE (c) and E (d) excretion levels in 12-week-old transgenic skinny mice (filled bars), ob/ob mice (shaded bars), and their control mice (open bars). AP < 0.05, BP < 0.01, and CP < 0.001 compared with control mice assessed by 1-way ANOVA with Bonferroni Dunn test (a, b, d, e), and by ANOVA with repeated measures analysis with Student’s t test at a given time (c).
Daily urinary catecholamine excretion in transgenic skinny mice and ob/ob mice.
There was no significant difference between transgenic and nontransgenic mice in daily urine volume (data not shown). Daily urinary norepinephrine (NE) excretion was increased significantly in transgenic skinny mice compared with nontransgenic littermates (224.0 ± 43.3 vs. 74.4 ± 28.7 ng/d; n = 6; P < 0.05) (Figure 1d). Daily urinary epinephrine (E) excretion also tended to be increased in transgenic skinny mice relative to nontransgenic littermates (42.7 ± 11.5 vs. 17.6 ± 7.2 ng/d; n = 6) (Figure 1e).
Daily urine volume of ob/ob mice tended to be increased relative to their control mice (1.7 ± 0.2 vs. 1.2 ± 0.1 mL/d; n = 8). Daily urinary E excretion tended to be decreased in ob/ob mice relative to their control mice (8.7 ± 1.1 vs. 17.6 ± 7.2 ng/d; n = 6), whereas no significant difference in daily urinary NE excretion was noted between genotypes (107.3 ± 15.2 vs. 74.4 ± 28.7 ng/d; n = 6) (Figure 1, d and e). These results are consistent with a previous report (38).
Effects of sympathetic blockade on BP and heart rate in transgenic skinny mice.
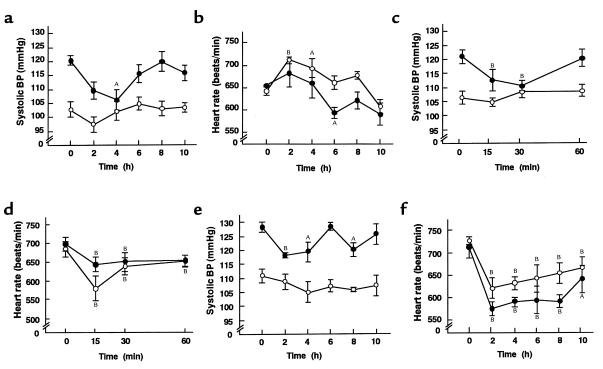
To explore the involvement of sympathetic activation in BP elevation in transgenic skinny mice, we examined the effect of sympathetic blockade on BP and heart rate in these animals. On examination of the dose-related effects of bunazosin, propranolol, and hexamethonium, we found that the maximal doses that do not affect BP in nontransgenic littermates are 15 μg/g body weight, 6 μg/g body weight, and 3 μg/g body weight, respectively. In transgenic skinny mice, systolic BP elevation was normalized 4 hours after the treatment with bunazosin at such a dose (15 μg/g body weight) that does not affect BP in nontransgenic littermates (Figure 2a). No significant change in heart rate was noted between genotypes treated with bunazosin (Figure 2b). Hexamethonium at a dose of 3 μg/g body weight also abolished significantly systolic BP elevation in transgenic skinny mice 15 and 30 minutes after the treatment, whereas no significant changes were noted in nontransgenic littermates (Figure 2, c and d). Furthermore, systolic BP elevation was attenuated significantly in transgenic skinny mice that received a single intraperitoneal injection of propranolol at a dose of 6 μg/g body weight, when heart rates were reduced significantly in both genotypes (Figure 2, e and f).
Figure 2.
Effects of sympathetic blockades on systolic BP and heart rate in 12-week-old transgenic skinny mice and nontransgenic littermates. Effect of bunazosin on systolic BP (a) and heart rate (b). Effect of hexamethonium on systolic BP (c) and heart rate (d). Effect of propranolol on systolic BP (e) and heart rate (f). AP < 0.05 and BP < 0.01 compared to the initial values.
Blood and urinary parameters and heart and kidney weights in transgenic skinny mice.
To explore the effect of chronic hyperleptinemia on body fluid homeostasis and electrolyte balance, we measured blood and urinary parameters in transgenic skinny mice. No significant differences were noted between transgenic and nontransgenic littermates in daily urine volume and water intake; serum osmolarity and hematocrit; serum concentrations of Na+, K+, and Cl+; serum creatinine; blood urea nitrogen; and urinary Na+, K+, and Cl+ excretion (data not shown). There were no appreciable differences in the weights of heart and kidney between transgenic and nontransgenic littermates (data not shown).
Effects of hypothalamic melanocortin system blockade on BP elevation in transgenic skinny mice.
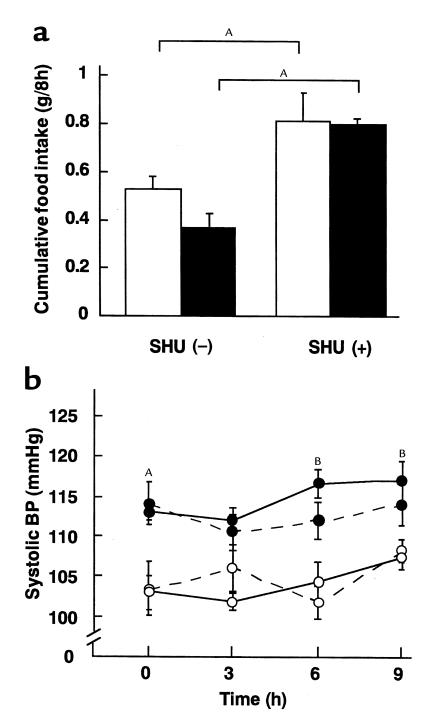
To explore the role of hypothalamic melanocortin system in BP elevation in transgenic skinny mice, we examined the effect of a single intracerebroventricular injection of SHU9119 on systolic BP in these animals. When treated with aCSF, cumulative food intake of transgenic skinny mice was smaller than that of nontransgenic littermates as reported (28) (0.37 ± 0.06 vs. 0.51 ± 0.06 g/8 h; n = 4) (Figure 3a). On the other hand, a single intracerebroventricular injection of SHU9119 at a dose of 0.05 μg per mouse caused a significant increase in cumulative food intake in both transgenic and nontransgenic littermates during 8 hours at the onset of the light phase (0.81 ± 0.02 vs. 0.81 ± 0.17 g/8 h; n = 4) (about 2-fold increase in both genotypes relative to aCSF-treated groups) (P < 0.05) (Figure 3a). Nevertheless, no significant changes in systolic BP were noted in transgenic and nontransgenic littermates up to 8 hours after the SHU9119 treatment (Figure 3b).
Figure 3.
Effects of SHU9119 on cumulative food intake and systolic BP in 12-week-old transgenic skinny mice and nontransgenic littermates. (a) Effect of SHU9119 on cumulative food intake in transgenic skinny mice (filled bars) and nontransgenic littermates (open bars). AP < 0.05 compared with vehicle-treated groups. (b) Effect of SHU9119 on systolic BP in transgenic skinny mice (filled circles) and nontransgenic littermates (open circles). Mice treated with and without SHU9119 are depicted by solid and dotted lines, respectively. AP < 0.05 and BP < 0.01 compared with SHU9119-treated nontransgenic littermates.
Body weight, plasma leptin concentrations, systolic BP, and urinary catecholamine excretion in KK and KKAy mice.
To explore further the possible role of leptin in obesity-related hypertension, we examined the cardiovascular phenotypes of genetically obese KKAy mice. At 12 weeks of age, KKAy mice developed obesity relative to age-matched control KK mice (39.8 ± 1.2 vs. 31.3 ± 0.3 g; n = 6; P < 0.001). Plasma leptin concentrations in KKAy mice were 27.8 ± 1.1 ng/mL and were approximately 4.5-fold higher than those in KK mice (6.3 ± 1.0 ng/mL) (n = 10; P < 0.001).
At 0900 hours, systolic BP was elevated significantly in KKAy mice compared with KK mice (116.4 ± 1.4 vs. 109.5 ± 1.2 mmHg; n = 10; P < 0.01). Systolic BP elevation in KKAy mice was also sustained throughout the day (data not shown). No significant difference in heart rate was noted between KK and KKAy mice (479.5 ± 14.6 vs. 489.1 ± 19.7 beats per minute; n = 10). Daily urine volume of KKAy mice was significantly larger than that of KK mice (14.6 ± 1.2 vs. 2.6 ± 0.7 mL/d; n = 4; P < 0.001). Daily urinary NE and E excretion was increased significantly in KKAy mice relative to KK mice (NE, 434.9 ± 42.0 vs. 221.2 ± 69.9 ng/d [n = 4; P < 0.05]; E, 37.3 ± 4.6 vs. 7.7 ± 1.5 ng/d [n = 4; P < 0.001]).
Body weight change, plasma leptin concentrations, and systolic BP in +/+, Tg/+, Ay/+, and Tg/+:Ay/+ mice during caloric restriction.
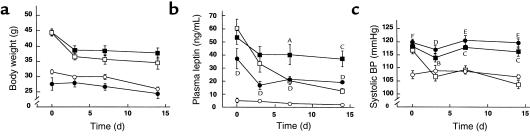
We reported that at younger ages (i.e., at 4 weeks of age), Tg/+:Ay/+ mice exhibit skinny phenotype similar to that of Tg/+ mice but that at older ages (i.e. at 12 week of age), they develop obesity similar to that of Ay/+ mice (29). To assess the involvement of hyperleptinemia in BP elevation in obese KKAy mice and to explore the role of leptin in BP decrease during weight loss (5–8), we examined the cardiovascular phenotypes of 12-week-old +/+, Tg/+, Ay/+, and Tg/+:Ay/+ mice during a caloric restriction. Body weights of Tg/+:Ay/+ and Ay/+ mice (44.7 ± 1.2 and 44.5 ± 0.8 g, respectively) were significantly greater than those of +/+ and Tg/+ mice (31.8 ± 0.7 and 27.8 ± 2.1 g, respectively) (n = 7) (Figure 4a). No significant difference in body weight was noted between Tg/+:Ay/+ and Ay/+ mice. After a 2-week caloric restriction, body weights of Tg/+ and +/+ mice were 16.1 ± 2.4 and 13.4 ± 2.0% less, respectively, than those before the experiment (Figure 4a). Body weights of Tg/+:Ay/+ and Ay/+ mice after the caloric restriction were also decreased significantly (19.4 ± 2.7 and 23.9 ± 1.7% less, respectively) relative to those before the caloric restriction (Figure 4a). After the caloric restriction, no significant difference in body weight was noted among +/+, Ay/+, and Tg/+:Ay/+ mice.
Figure 4.
Effects of a 2-week caloric restriction on body weights, plasma leptin concentrations, and systolic BPs in 12-week-old F1 animals (+/+, Tg/+, Ay/+, and Tg/+:Ay/+ mice). (a) Time course of body weights of +/+ (open circles), Tg/+ (filled circles), Ay/+ (open boxes), and Tg/+:Ay/+ (filled boxes) mice during the caloric restriction. (b) Time course of plasma leptin concentrations in +/+ (open circles), Tg/+ (filled circles), Ay/+ (open boxes), and Tg/+:Ay/+ (filled boxes) mice. (c) Time course of systolic BPs in +/+ (open circles), Tg/+ (filled circles), Ay/+ (open boxes), and Tg/+:Ay/+ (filled boxes) mice. DP < 0.05, EP < 0.01, and FP < 0.001 (Tg/+ mice compared with +/+ mice), and AP < 0.05, BP < 0.01, and CP < 0.001 (Tg/+:Ay/+ mice compared with Ay/+ mice) assessed by ANOVA with repeated measures analysis with Student’s t test at a given time.
Plasma leptin concentrations in 12-week-old Tg/+, Ay/+, and Tg/+:Ay/+ mice were 37.3 ± 7.9, 60.4 ± 7.0, and 53.4 ± 5.2 ng/mL, respectively, and were significantly higher than those in +/+ mice (5.3 ± 1.9 ng/mL) (P < 0.05, n = 7) (Figure 4b). No significant difference in plasma leptin concentration was noted between Tg/+:Ay/+ and Ay/+ mice. Plasma leptin concentrations in +/+ and Ay/+ mice after a 2-week caloric restriction (2.1 ± 0.4 and 12.7 ± 2.0 ng/mL, respectively) were approximately 60% and 80% lower, respectively, than those before the caloric restriction (Figure 4b). On the other hand, after the caloric restriction, plasma leptin concentrations remained high in Tg/+ and Tg/+:Ay/+ mice (19.3 ± 0.1 and 37.3 ± 5.9 ng/mL, respectively); it is because of hepatic overproduction of leptin in Tg/+ and Tg/+:Ay/+ mice.
Before the caloric restriction, systolic BP was elevated significantly in Tg/+, Ay/+, and Tg/+:Ay/+ mice (119.8 ± 1.3, 116.8 ± 1.4, and 118.2 ± 1.1 mmHg, respectively) relative to +/+ mice (107.4 ± 1.5 mmHg) (n = 7) (Figure 4c). No significant difference in systolic BP was noted among Tg/+, Ay/+, and Tg/+:Ay/+ mice. After the caloric restriction, systolic BP elevation was reversed in Ay/+ mice (103.7 ± 1.5 mmHg), which is roughly comparable to that in +/+ mice (106.7 ± 0.7 mmHg). On the other hand, systolic BP elevation was sustained in Tg/+:Ay/+ mice after the caloric restriction (116.3 ± 1.7 mmHg) (Figure 4c).
Leptin injection experiments.
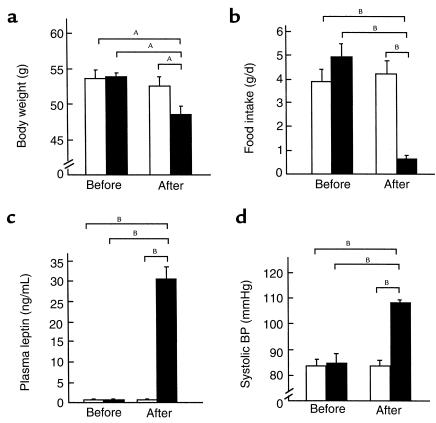
To assess whether hyperleptinemia during adulthood plays a major role in the development of BP elevation in transgenic skinny mice, we examined the effect of continuous leptin administration on BP in ob/ob mice (n = 6). After a 3-day infusion, body weights of ob/ob mice treated with leptin decreased significantly relative to vehicle-treated animals (48.6 ± 1.2 vs. 52.6 ± 1.3 g, P < 0.05) (Figure 5a). Food intake of leptin-treated ob/ob mice was also decreased significantly compared with vehicle-treated groups at the end of the experiment (0.6 ± 0.2 vs. 4.2 ± 0.6 g/d; P < 0.01) (Figure 5b). Plasma leptin concentrations were elevated in leptin-treated ob/ob mice 3 days after the treatment (Figure 5c). Three days after the treatment, plasma leptin concentrations were 30.6 ± 2.9 ng/mL in leptin-treated ob/ob mice, whereas no significant amount of leptin was detected in vehicle-treated groups. During this experiment, systolic BP was unchanged in vehicle-treated ob/ob mice. By contrast, leptin administration increased BP by as much as 25 mmHg in ob/ob mice (Figure 5d). These observations demonstrate that leptin can reverse the otherwise lowered BP in genetically obese leptin-deficient ob/ob mice.
Figure 5.
Effects of a 3-day leptin administration on body weights, plasma leptin concentrations, and systolic BPs in 12-week-old male ob/ob mice and control littermates. (a) Body weights of ob/ob mice before and 3 days after the treatment. (b) Food intake of ob/ob mice before and 3 days after the treatment. (c) Plasma leptin concentrations in ob/ob mice before and 3 days after the treatment. (d) Systolic BPs of ob/ob mice before and 3 days after the treatment. The ob/ob mice treated with leptin or vehicle are indicated by filled and open bars, respectively. AP < 0.05, BP < 0.01 versus vehicle-treated groups assessed by ANOVA with repeated measures analysis with Student’s t test.
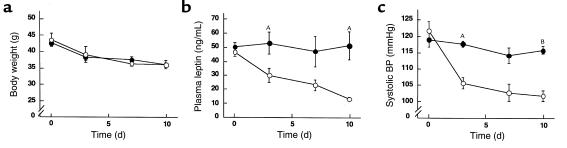
To see whether weight loss does not lower BP in calorically restricted Ay/+ mice when plasma leptin concentrations are elevated, we studied the effect of chronic leptin administration on BP in Ay/+ mice during the caloric restriction (n = 6). During the 10-day food restriction, body weights of Ay/+ mice treated with leptin decreased comparably to those of vehicle-treated animals (Figure 6a). Plasma leptin concentrations were reduced in vehicle-treated Ay/+ mice during the caloric restriction, whereas they remained high in leptin-treated Ay/+ mice (Figure 6b). Ten days after the caloric restriction, plasma leptin concentrations were 51.3 ± 10.1 and 13.6 ± 1.2 ng/mL in leptin- and vehicle-treated groups, respectively (P < 0.05). During the experiment, systolic BP was decreased in vehicle-treated Ay/+ mice, whereas no significant change in BP was noted in leptin-treated Ay/+ mice (Figure 6c). The systolic BPs in leptin- and vehicle-treated Ay/+ mice were 115.8 ± 1.2 and 101.9 ± 1.6 mmHg, respectively (P < 0.001). These observations demonstrate that with chronic hyperleptinemia, BP remains high in Ay/+ mice even during the caloric restriction, thereby supporting the notion that maintenance of hyperleptinemia in the adult mice is sufficient to explain BP elevation in Ay/+ mice.
Figure 6.
Effects of chronic leptin administration on BPs in 12-week-old male Ay/+ mice during a 10-day caloric restriction. (a) Time course of body weights of Ay/+ mice treated with leptin or vehicle. (b) Time course of plasma leptin concentrations in Ay/+ mice treated with leptin or vehicle. (c) Time course of systolic BPs in Ay/+ mice treated with leptin or vehicle. The Ay/+ mice treated with leptin or vehicle are indicated by filled and open circles, respectively. AP < 0.05, BP < 0.001 versus vehicle-treated groups assessed by ANOVA with repeated measures analysis with Student’s t test at a given time.
Discussion
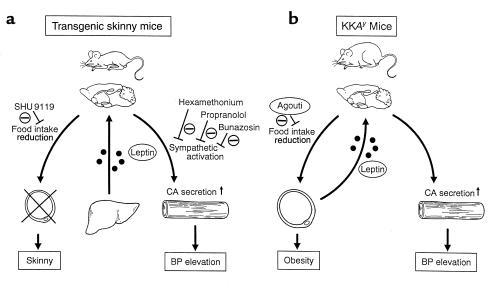
In this study, we have demonstrated that BP is elevated significantly in transgenic skinny mice relative to nontransgenic littermates. These findings indicate that chronic hyperleptinemia can lead to a significant BP elevation in conscious mice. This is also consistent with the data that chronic as well as acute treatment of leptin can increase BP in conscious rats and in ob/ob mice (13, 39–41; Figure 5). On the other hand, there is a report that acute leptin treatment has no effect on BP in anesthetized rats (12). Transgenic skinny mice exhibit a significant increase in urinary catecholamine excretion, suggesting chronic sympathetic activation. Furthermore, BP elevation is abolished in transgenic skinny mice by treatment with the α1-adrenergic, sympathetic ganglionic, and nonselective β-adrenergic blockers at such doses that do not affect BP in nontransgenic littermates. These findings suggest that BP elevation in transgenic skinny mice is mediated through the sympathetic activation (Figure 7a).
Figure 7.
Possible mechanisms for the leptin-induced BP elevation in transgenic skinny mice (a) and KKAy mice (b). In transgenic skinny mice (a), leptin is oversecreted ectopically from the liver into the circulation, which causes the skinny phenotype with decreased food intake and a significant BP elevation with increased catecholamine production. They are hypoinsulinemic with hypersensitivity to insulin (28). Decrease in food intake is reversed by SHU9119, whereas BP elevation is not abolished by SHU9119 but by bunazosin, propranolol, and hexamethonium. In KKAy mice (b), leptin is oversecreted from the adipose tissue into the circulation, which causes significant BP elevation with increased catecholamine production, although they are hyperphagic owing to the antagonism of hypothalamic melanocortin system by the agouti protein. They are hyperinsulinemic with marked resistance to insulin (30, 31). CA, catecholamine.
Several studies have demonstrated that central administration of leptin can increase uncoupling protein-1 mRNA expression in the brown adipose tissue (BAT) (14, 23) and catecholamine secretion (15), suggesting that sympathetic activation of leptin is mediated primarily via the central nervous system. We postulate that BP elevation in transgenic skinny mice is also due primarily to the central effect of leptin. This notion is consistent with previous reports that a single intracerebrovascular injection of leptin can increase BP in rats (40, 41). The discussion, however, does not rule out the possibility that leptin can increase BP via peripheral mechanisms, because leptin receptor is expressed in peripheral tissues including vascular endothelial cells and adrenal medulla (42–44).
We and others demonstrated that a single intracerebrovascular injection of SHU9119 abolishes the leptin-induced decrease in food intake and body weight in rats and postulated that hypothalamic melanocortin system is involved in the satiety effect of leptin (22, 23). In this study, treatment with SHU9119 increases significantly cumulative food intake in transgenic and nontransgenic littermates relative to vehicle-treated groups, suggesting that a marked decrease in food intake in transgenic skinny mice is also mediated at least partly via hypothalamic melanocortin system. On the other hand, no significant changes in BP are noted in both genotypes after the SHU9119 treatment. These findings suggest that decreased food intake and BP elevation in transgenic skinny mice are mediated via different mechanisms, i.e., leptin may exert its satiety effect via hypothalamic melanocortin system, whereas it can induce a significant BP elevation independently (Figure 7a). In this regard, it has been recently suggested that the acutely increased mean arterial pressure in response to a single intracerebrovascular injection of leptin may be mediated by proopiomelanocortin products (45). It is conceivable that leptin can acutely increase BP via hypothalamic melanocortin system; however chronic hyperleptinemia can increase BP independently. We previously demonstrated that a single intracerebrovascular injection of SHU9119 abolishes the leptin-induced increase in UCP-1 mRNA expression in the BAT in rats (23). On the other hand, Haynes et al. have reported that leptin can increase sympathetic nerve activity through the activation of hypothalamic melanocortin system, whereas it may increase sympathetic nerve traffic to the BAT independently (46). These findings suggest that some but not all of the leptin-induced sympathetic activation are mediated via hypothalamic melanocortin system. Because central administration of neuropeptide Y (NPY) reduces BP in rats and dogs (47, 48), decreased hypothalamic NPY production by exogenous administration of leptin (49) and in transgenic skinny mice (Y. Ogawa et al, unpublished data) might be involved in hypertensive effect of leptin.
The KKAy mice develop a maturity-onset obesity and diabetes syndrome because of the antagonism of hypothalamic melanocortin system by the agouti protein (30, 31), thereby conferring resistance to the satiety effect of leptin (32). This study demonstrated significant increases in BP and urinary catecholamine excretion in obese KKAy mice with hyperleptinemia (Figure 7b). Furthermore, after the caloric restriction, BP elevation is reversed in Ay/+ mice in parallel with a significant reduction in plasma leptin concentrations, whereas no such changes are noted in Tg/+:Ay/+ mice with sustained hyperleptinemia. These findings, taken together, suggest that chronic hyperleptinemia may be involved in BP elevation in obese KKAy mice. Thus, obese KKAy mice may provide the unique opportunities to investigate the pathophysiological role of leptin in obesity-related hypertension; they develop obesity owing to their resistance to the satiety effect of leptin and develop BP elevation owing possibly to their sensitivity to hypertensive effect of leptin (Figure 7b). This is also consistent with the hypothesis described earlier here that in transgenic skinny mice, leptin can exert its satiety effect via hypothalamic melanocortin system but induce BP elevation independently. There are a variety of phenotypic differences between yellow agouti mice such as KKAy mice and leptin signaling-deficient ob/ob and db/db mice (10, 50–52). In contrast with ob/ob and db/db mice, yellow agouti mice show a maturity-onset obesity and diabetes syndrome, normal tone of hypothalamic-pituitary-adrenal axis, increased linear growth, normal thermoregulation against cold stress, and normal locomotor activity (30, 31, 50–52), indicating that some of leptin actions are independent of hypothalamic melanocortin system. Our data herein suggest that hypertensive effect of leptin might be one of such actions.
The mechanism of obesity-related hypertension is not clear at present. It has been suggested that obesity-related hypertension is secondary to insulin resistance and/or hyperinsulinemia (2, 3, 7). Lansberg and Krieger have proposed that hyperinsulinemia causes sodium retention in the kidney as well as increases vascular resistance, thereby leading to an significant BP elevation in obese subjects (3). On the other hand, transgenic skinny mice exhibit increased glucose metabolism and insulin sensitivity with a significant reduction in plasma insulin concentrations compared with nontransgenic littermates (28). Taken together, the data of this study indicate that chronic hyperleptinemia alone can cause a significant BP elevation in mice without obesity, insulin resistance, and hyperinsulinemia (Figure 7a). Several studies revealed that there is a significant correlation between plasma insulin and leptin concentrations (53) and that insulin increases synthesis and secretion of leptin in the adipose tissue in vivo (54). We hypothesize that obesity- and/or hyperinsulinemia-induced hyperleptinemia may increase sympathetic nerve activity, thus contributing to a significant BP elevation in obese subjects.
Weight reduction is an effective and nonpharmacologic method to lower BP in obese hypertensive subjects; however, the mechanism responsible for it has remained to be elucidated. Several studies suggested that the BP decrease after weight loss is related to the suppression of sympathetic nerve activity in rats and humans (6–8). In this study, after the caloric restriction, BP elevation is reversed in Ay/+ mice in parallel with a reduction in plasma leptin concentrations, whereas no significant changes are noted in Tg/+:Ay/+ mice. We also examined the effect of chronic leptin administration on BP in Ay/+ mice during a caloric restriction and observed that with continuous hyperleptinemia, weight loss does not lower BP in calorically restricted Ay/+ mice. These observations suggest that hyperleptinemia alone can increase BP even during a caloric restriction. It is also tempting to speculate that like starvation-induced suppression of neuroendocrine and immune functions (55, 56), the BP decrease after weight loss is due partly to leptin deficiency.
The clinical features of obesity-related hypertension also include sodium retention with a concomitant increase in blood volume and cardiac output (3). It seems, however, that transgenic skinny mice and obese KKAy mice develop no such manifestations. Thus, chronic hyperleptinemia is not sufficient to recapitulate all signs and symptoms of obesity-related hypertension. Previously, we identified a nonsense mutation of leptin receptor in obese spontaneously hypertensive Koletsky (fak/fak) rats (57). The rats have been maintained on the SHR background, which explains their severe hypertension. Furthermore, obese Zucker fatty rats (fa/fa rats) with leptin receptor mutations (17, 58) exhibit marked BP elevation (59). In Zucker fatty rats, increased BP appears to be associated with the obese genotype (59). These observations suggest that the genetic factors might affect BP in obesity independently of leptin signaling. Thus, hyperleptinemia in combination with other factors should contribute to the overall profile of obesity-related hypertension.
Our data suggest that leptin treatment may cause BP elevation in humans. In this regard, Farooqi et al. have reported the effects of recombinant human leptin in a child with congenital leptin deficiency and found no significant BP differences before and after the leptin treatment (60). Furthermore, no significant BP change was reported in obese and lean adults treated with leptin (61). Thus, there might be species difference in the leptin-induced BP increase.
In conclusion, this study demonstrates BP elevation in transgenic skinny mice and KKAy mice, both of which are hyperleptinemic, thereby raising the possibility that leptin may play a role in the pathogenesis of some forms of obesity-related hypertension.
Acknowledgments
We thank Y. Yamori, G. Katsuura, S. Yura, and J. Hiraoka for discussions. The authors also acknowledge Y. Isa, Y. Nakajima, and K. Hiramatsu for secretarial assistance. This work is supported in part by research grants from the Japanese Ministry of Education, Science, Sports, and Culture; the Japanese Ministry of Health and Welfare; the Yamanouchi Foundation for Research on Metabolic Disorders; the Foundation for Total Health Promotion 1998; The Naito Foundation; The Mitsubishi Foundation; The Uehara Memorial Foundation; and Research for the Future of Japanese Society for the Promotion of Science (JSPS-RFTF 96100204 and 98L00801).
References
- 1.Kannel WB, Brand M, Skinner JJ, Jr, Sawber TR, McNamara PM. The relation of adiposity to blood pressure and development of hypertension: the Framingham study. Ann Intern Med. 1967;67:48–59. doi: 10.7326/0003-4819-67-1-48. [DOI] [PubMed] [Google Scholar]
- 2.Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37:1595–1607. doi: 10.2337/diab.37.12.1595. [DOI] [PubMed] [Google Scholar]
- 3.Landsberg L, Krieger DR. Obesity, metabolism, and the sympathetic nervous system. Am J Hypertension. 1989;2:125S–132S. doi: 10.1093/ajh/2.3.125s. [DOI] [PubMed] [Google Scholar]
- 4.Mark AL, Correia M, Morgan DA, Shaffer RA, Haynes WG. Obesity-Induced Hypertension : New Concepts From the Emerging Biology of Obesity. Hypertension. 1999;33:537–541. doi: 10.1161/01.hyp.33.1.537. [DOI] [PubMed] [Google Scholar]
- 5.Rocchini AP, et al. The effect of weight loss on the sensitivity of blood pressure to sodium in obese adolescents. N Engl J Med. 1989;31:580–585. doi: 10.1056/NEJM198908313210905. [DOI] [PubMed] [Google Scholar]
- 6.Overton JM, VanNess JM, Casto RM. Food restriction reduces sympathetic support of blood pressure in spontaneously hypertensive rats. J Nutr. 1996;127:655–660. doi: 10.1093/jn/127.4.655. [DOI] [PubMed] [Google Scholar]
- 7.Kushiro T, et al. Role of sympathetic activity in blood pressure reduction with low calorie regimen. Hypertension. 1991;17:965–968. doi: 10.1161/01.hyp.17.6.965. [DOI] [PubMed] [Google Scholar]
- 8.Ikeda T, Gomi T, Hirawa N, Sakurai J, Yoshikawa N. Improvement of insulin sensitivity contributes to blood pressure reduction after weight loss in hypertensive subjects with obesity. Hypertension. 1996;27:1180–1186. doi: 10.1161/01.hyp.27.5.1180. [DOI] [PubMed] [Google Scholar]
- 9.Spiegelman BM, Flier JS. Adipogenesis and obesity: rounding out the big picture. Cell. 1996;87:377–389. doi: 10.1016/s0092-8674(00)81359-8. [DOI] [PubMed] [Google Scholar]
- 10.Zhang Y, et al. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425–432. doi: 10.1038/372425a0. [DOI] [PubMed] [Google Scholar]
- 11.Collins S, et al. Role of leptin in fat regulation. Nature. 1996;380:677. doi: 10.1038/380677a0. [DOI] [PubMed] [Google Scholar]
- 12.Haynes WG, Morgan DA, Walsh SA, Mark AL, Sivitz WI. Receptor-mediated regional sympathetic nerve activation by leptin. J Clin Invest. 1997;100:270–278. doi: 10.1172/JCI119532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dunbar JC, Hu Y, Lu H. Intracerebroventricular leptin increases lumbar and renal sympathetic nerve activity and blood pressure in normal rats. Diabetes. 1997;46:2040–2043. doi: 10.2337/diab.46.12.2040. [DOI] [PubMed] [Google Scholar]
- 14.Scarpace PJ, Matheny M, Pollock BH, Tümer N. Leptin increases uncoupling protein expression and energy expenditure. Am J Physiol. 1997;273:E226–E230. doi: 10.1152/ajpendo.1997.273.1.E226. [DOI] [PubMed] [Google Scholar]
- 15.Satoh N, et al. Sympathetic activation of leptin via the ventromedial hypothalamus: leptin-induced increase in catecholamine secretion. Diabetes. 1999;48:1787–1793. doi: 10.2337/diabetes.48.9.1787. [DOI] [PubMed] [Google Scholar]
- 16.Tartaglia LA, et al. Identification and expression cloning of a leptin receptor, OB-R. Cell. 1995;83:1263–1271. doi: 10.1016/0092-8674(95)90151-5. [DOI] [PubMed] [Google Scholar]
- 17.Takaya K, et al. Molecular cloning of rat leptin receptor isoform complementary DNAs: identification of a missense mutation in Zucker fatty (fa/fa) rats. Biochem Biophys Res Commun. 1996;225:75–83. doi: 10.1006/bbrc.1996.1133. [DOI] [PubMed] [Google Scholar]
- 18.Schwartz MW, Seeley RJ, Campfield LA, Burn P, Baskin DG. Identification of target of leptin action in rat hypothalamus. J Clin Invest. 1996;98:1101–1106. doi: 10.1172/JCI118891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fei H, et al. Anatomic localization of alternatively spliced leptin receptors (Ob-R) in mouse brain and other tissues. Proc Natl Acad Sci USA. 1997;94:7001–7005. doi: 10.1073/pnas.94.13.7001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Satoh N, et al. The arcuate nucleus is a primary site of satiety effect of leptin in rats. Neurosci Lett. 1997;224:149–152. doi: 10.1016/S0304-3940(97)00163-8. [DOI] [PubMed] [Google Scholar]
- 21.Elmquist JK, Maratos-Flier E, Saper CB, Flier JS. Unraveling the central nervous system pathway underlying responses to leptin. Nat Neurosci. 1998;1:445–450. doi: 10.1038/2164. [DOI] [PubMed] [Google Scholar]
- 22.Seeley, R.J., et al. 1997. Melanocortin receptors in leptin effects.Nature. 390–349. [DOI] [PubMed]
- 23.Satoh N, et al. Satiety effect and sympathetic activation of leptin are mediated by hypothalamic melanocortin system. Neurosci Lett. 1998;249:107–110. doi: 10.1016/s0304-3940(98)00401-7. [DOI] [PubMed] [Google Scholar]
- 24.Frederich RC, et al. Leptin levels reflect body lipid content in mice: Evidence for diet-induced resistance to leptin action. Nat Med. 1995;1:1311–1314. doi: 10.1038/nm1295-1311. [DOI] [PubMed] [Google Scholar]
- 25.Maffei M, et al. Leptin levels in human and rodent: measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat Med. 1995;1:1155–1161. doi: 10.1038/nm1195-1155. [DOI] [PubMed] [Google Scholar]
- 26.Considine RV, et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med. 1996;334:292–295. doi: 10.1056/NEJM199602013340503. [DOI] [PubMed] [Google Scholar]
- 27.Agata J, et al. High plasma immunoreactive leptin level in essential hypertension. Am J Hypertension. 1997;10:1171–1174. doi: 10.1016/s0895-7061(97)00310-5. [DOI] [PubMed] [Google Scholar]
- 28.Ogawa Y, et al. Increased glucose metabolism and insulin sensitivity in transgenic skinny mice overexpressing leptin. Diabetes. 1999;48:1822–1829. doi: 10.2337/diabetes.48.9.1822. [DOI] [PubMed] [Google Scholar]
- 29.Masuzaki H, et al. Glucose metabolism and insulin sensitivity in transgenic mice overexpressing leptin with lethal yellow agouti mutation: usefulness of leptin for the treatment of obesity-associated diabetes. Diabetes. 1999;48:1615–1622. doi: 10.2337/diabetes.48.8.1615. [DOI] [PubMed] [Google Scholar]
- 30.Bultman SJ, Michaud EJ, Woychik RP. Molecular characterization of the mouse agouti locus. Cell. 1992;71:1195–1204. doi: 10.1016/s0092-8674(05)80067-4. [DOI] [PubMed] [Google Scholar]
- 31.Miller MW, et al. Cloning of the mouse agouti gene predicts a secreted protein ubiquitously expressed in mice carrying the lethal yellow mutation. Genes Dev. 1993;7:454–467. doi: 10.1101/gad.7.3.454. [DOI] [PubMed] [Google Scholar]
- 32.Halaas JL, et al. Physiological response to long-term peripheral and central leptin infusion in lean and obese mice. Proc Natl Acad Sci USA. 1997;94:8878–8883. doi: 10.1073/pnas.94.16.8878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lu D, et al. Agouti protein is an antagonist of the melanocyte-stimulating hormone receptor. Nature. 1994;371:799–802. doi: 10.1038/371799a0. [DOI] [PubMed] [Google Scholar]
- 34.Fan W, Boston BA, Kesterson RA, Hruby VJ, Cone RD. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature. 1997;385:165–168. doi: 10.1038/385165a0. [DOI] [PubMed] [Google Scholar]
- 35.Iwatsuka H, Shino A, Suzuoki Z. General survey of diabetic features of yellow KK mice. Endocrinol Jpn. 1970;17:23–35. doi: 10.1507/endocrj1954.17.23. [DOI] [PubMed] [Google Scholar]
- 36.Kondo K, Nozawa K, Tomita T, Ezaki K. Inbred strains resulting from Japanese mice. Bull Exp Anim. 1957;6:107–116. [Google Scholar]
- 37.Ogawa Y, et al. Molecular cloning of the complementary DNA and gene that encode mouse brain natriuretic peptide and generation of transgenic mice that overexpress the brain natriuretic peptide gene. J Clin Invest. 1994;93:1911–1921. doi: 10.1172/JCI117182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Leigh FSM, Kaufman LN, Young JB. Diminished epinephrine excretion in genetically obese (ob/ob) mice and monosodium glutamate-treated rats. Int J Obes. 1992;16:597–604. [PubMed] [Google Scholar]
- 39.Shek EW, Brands MW, Hall JE. Chronic leptin infusion increases arterial pressure. Hypertension. 1998;31:409–414. doi: 10.1161/01.hyp.31.1.409. [DOI] [PubMed] [Google Scholar]
- 40.Casto RM, VanNess JM, Overton JM. Effects of central leptin administration on blood pressure in normotensive rats. Neurosci Lett. 1998;246:29–32. doi: 10.1016/s0304-3940(98)00223-7. [DOI] [PubMed] [Google Scholar]
- 41.Lu H, et al. Obesity due to high fat diet decreases the sympathetic nervous and cardiovascular responses to intracerebroventricular leptin in rats. Brain Res Bull. 1998;47:331–335. doi: 10.1016/s0361-9230(98)00086-0. [DOI] [PubMed] [Google Scholar]
- 42.Sierra-Honigmann MR, et al. Biological action of leptin as an angiogenic factor. Science. 1998;281:1683–1686. doi: 10.1126/science.281.5383.1683. [DOI] [PubMed] [Google Scholar]
- 43.Bouloumie A, Drexler HCA, Lafontan M, Busse R. Leptin, the product of ob gene, promotes angiogenesis. Circ Res. 1998;83:1059–1066. doi: 10.1161/01.res.83.10.1059. [DOI] [PubMed] [Google Scholar]
- 44.Cao GY, Considine RV, Lynn RB. Leptin receptor in the adrenal medulla of the rat. Am J Physiol. 1997;273:E448–E452. doi: 10.1152/ajpendo.1997.273.2.E448. [DOI] [PubMed] [Google Scholar]
- 45.Dunbar JC, Lu H. Leptin-induced increase in sympathetic nervous and cardiovascular tone is mediated by proopiomelanocortin (POMC) products. Brain Res Bull. 1999;50:215–221. doi: 10.1016/s0361-9230(99)00197-5. [DOI] [PubMed] [Google Scholar]
- 46.Haynes WG, Morgan DA, Djalali A, Sivitz WI, Mark AL. Interactions between the melanocortin system and leptin in control of sympathetic nerve traffic. Hypertension. 1999;33:542–547. doi: 10.1161/01.hyp.33.1.542. [DOI] [PubMed] [Google Scholar]
- 47.VanNess JM, DeMaria JE, Overton JM. Increased NPY activity in the PVN contributes to food-restriction induced reductions in blood pressure in aortic coarctation hypertensive rats. Brain Res. 1999;821:263–269. doi: 10.1016/s0006-8993(99)01058-6. [DOI] [PubMed] [Google Scholar]
- 48.Morton KDR, McCloskey MJD, Potter EK. Cardiorespiratory responses to intracerebroventricular injection of neuropeptide Y in anaesthetised dogs. Regul Peptides. 1999;81:81–88. doi: 10.1016/s0167-0115(99)00021-x. [DOI] [PubMed] [Google Scholar]
- 49.Stephens TW, et al. The role of neuripeptide Y in the antiobesity action of the obese gene product. Nature. 1995;377:530–532. doi: 10.1038/377530a0. [DOI] [PubMed] [Google Scholar]
- 50.Herberg L, Coleman DL. Laboratory animals exhibiting obesity and diabetes syndromes. Metabolism. 1977;26:59–87. doi: 10.1016/0026-0495(77)90128-7. [DOI] [PubMed] [Google Scholar]
- 51.Leibel RL, Chung WK, Chua SC. The molecular genetics of rodent single gene obesities. J Biol Chem. 1997;272:31937–31940. doi: 10.1074/jbc.272.51.31937. [DOI] [PubMed] [Google Scholar]
- 52.Huszar D, et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell. 1997;88:131–141. doi: 10.1016/s0092-8674(00)81865-6. [DOI] [PubMed] [Google Scholar]
- 53.Segal KR, Landt M, Klein S. Relationship between insulin sensitivity and plasma leptin concentrations in lean and obese men. Diabetes. 1996;45:988–991. doi: 10.2337/diab.45.7.988. [DOI] [PubMed] [Google Scholar]
- 54.Saladin R, et al. Transient increase in obese gene expression after food intake or insulin administration. Nature. 1995;377:527–529. doi: 10.1038/377527a0. [DOI] [PubMed] [Google Scholar]
- 55.Ahima RS, et al. Role of leptin in the neuroendocrine response to fasting. Nature. 1996;382:250–252. doi: 10.1038/382250a0. [DOI] [PubMed] [Google Scholar]
- 56.Lord GM, et al. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature. 1998;394:897–901. doi: 10.1038/29795. [DOI] [PubMed] [Google Scholar]
- 57.Takaya K, et al. Nonsense mutation of leptin receptor in the obese spontaneously hypertensive Koletsky rat. Nat Genet. 1996;14:130–131. doi: 10.1038/ng1096-130. [DOI] [PubMed] [Google Scholar]
- 58.Phillips MS, et al. Leptin receptor missense mutation in the fatty Zucker rat. Nat Genet. 1996;13:18–19. doi: 10.1038/ng0596-18. [DOI] [PubMed] [Google Scholar]
- 59.Kurtz TW, Morris RC, Pershadsingh HA. The Zucker fatty rat as a genetic model of obesity and hypertension. Hypertension. 1989;13:896–901. doi: 10.1161/01.hyp.13.6.896. [DOI] [PubMed] [Google Scholar]
- 60.Farooqi IS, et al. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N Engl J Med. 1999;341:879–884. doi: 10.1056/NEJM199909163411204. [DOI] [PubMed] [Google Scholar]
- 61.Heymsfield SB, et al. Recombinant leptin for weight loss in obese and lean adults: a randomized, controlled, dose-escalation trial. JAMA. 1999;282:1568–1575. doi: 10.1001/jama.282.16.1568. [DOI] [PubMed] [Google Scholar]