Abstract
Polyploidy has been linked to tumorigenicity mainly due to the chromosomal aberrations. Elevated reactive oxygen species (ROS) generation, on the other hand, has also been associated with oncogenic transformation in most cancer cells. However, a possible link between ploidy and ROS is largely unexplored. Here we have exemined the role of ROS in the tumorigenicity of polyploid cells. We show that polyploid prostate and mammary epithelial cells contain higher levels of ROS due to their higher mitochondrial contents. ROS levels and mitochondrial mass are also higher in dihydrocytochalasin B (DCB)-induced polyploid cells, suggesting that higher levels of ROS observed in polyploid cell can occur due to cytokinesis failure. Interestingly, polyploid cells were more sensitive to the inhibitory effect of the antioxidant, N-Acetyl-L-cysteine (NAC), than control diploid cells. Treatment of polyploid/diploid cells with NAC led to the selective elimination of polyploid cells over time and abrogated the tumorigenicity of polyploid cells. This effect was partially mediated via the Akt signaling pathway. We next explored a possible role for ROS in promoting chromosomal instability by analyzing the effects of ROS on the mitotic stage of the cell cycle. Enhancing ROS levels by treating cells with hydrogen peroxide delayed not only entry into and but also exit from mitosis. Furthermore, increasing ROS levels significantly increased taxol resistance. Our results indicated that increased ROS in polyploid cells can contribute to tumorigenicity and highlight the therapeutic potential of antioxidants by selectively targeting the tumorigenic polyploid cells and by reversing taxol resistance.
Keywords: ROS, polyploidy, antioxidant, tumorigenicity, cell cycle
Introduction
Aneuploidy has been proposed to contribute to tumorigenicity for a long time (Boveri, 2008; Storchova and Pellman, 2004). Aneuploidy is believed to arise via polyploidy/tetraploidy followed by further chromosomal abnormalities due to an increase in chromosomal mass and number of centrosomes. Direct experimental support for a causative role for polyploidy in tumorigenesis was provided by several recent studies, including through the use of p53-null tetraploid mouse epithelial cells (Fujiwara et al., 2005) and Pim1-expressing human prostate and mammary epithelial cells (Roh et al., 2008). Genetically engineered mice with mutations in oncogenes/tumor suppressors (such as Aurora A, Mad 2, Eg5, Apc) show evidence of polyploidy, chromosomal aberrations and tumor development (Caldwell et al., 2007; Castillo et al., 2007; Sotillo et al., 2007; Wang et al., 2006).
Apart from inducing chromosomal abnormalities, polyploidy may also affect ROS levels as suggested by some literatures (Kraniak et al., 2006; Limoli et al., 2003; van de Wetering et al., 2008). Whether these ROS have any significant functional consequence on the polyploid cell and especially on its tumorigenic potential are not known at all. The effects of ROS on the cell may depend on their concentrations and on the cell context (Martin and Barrett, 2002). Under normal conditions, the deleterious effects of ROS are kept in check by the cellular antioxidant system. Some evidence suggests a possible relationship between ploidy and ROS levels. Antioxidant agents can inhibit aneuploidy progression (Kraniak et al., 2006), and overexpression of the antioxidant enzyme, manganese superoxide dismutase, inhibits chromosomal instability (van de Wetering et al., 2008). Oxidative damage to the liver is associated with an increase in the polyploid cell population (Gorla et al., 2001) and overexpression of antioxidant enzymes in mice decreases cellular ploidy during liver regeneration (Nakatani et al., 1997).
In this study, we have used a model of spontaneous polyploidy induced by the oncogenic kinase Pim-1 (Roh et al., 2008; Roh et al., 2003; Roh et al., 2005) to examine the potential role of ROS in the tumorigenic potential of polyploid human prostate and mammary epithelial cells. Our results indicate a significant role for ROS in the tumorigenicity of these cells. ROS may also further fuel chromosomal instability and affect sensitivity to mitotic poisons such as taxol by interfering with the mitotic stage of the cell cycle. Our study also highlights a possible therapeutic role for antioxidants in selectively targeting tumorigenic polyploid cells.
Materials and Methods
Cell lines and reagents
Non-malignant prostate (RWPE1) and mammary (hTERT-HME) epithelial cells were obtained from ATCC. Establishment of matched diploid and polyploid RWPE1 and hTERT-HME cells by Pim1 expression and cell sorting has been described (Roh et al., 2008; Roh et al., 2003; Roh et al., 2005). Dichlorodihydrofluorescein diacetate (DCF-DA; cat # D6883), N-Acetyl-L-cysteine (NAC; cat # A 9165), nocodazole (cat # M1404), paclitaxel (cat # T1912), hydrogen peroxide (cat # H009) and 4′, 6-diamidino-2-phenylindole (DAPI; cat # D-9642) were purchased from Sigma. Dihydroethidium (DHE; cat # D-1168), oxidation insensitive dye DCF-DA (cat # C-369), oxidation sensitive dye DCF-DA (cat # C-400), rhodamine 123 (cat # R-302), nonyl acridine orange (NAO; cat # A-1372), and MitoSox ™ Red (cat # M36008) were purchased from Molecular Probe/Invitrogen. Akt inhibitor, triciribine, was purchased form Calbiochem (Akt inhibitor V, cat # 124012). Rotenone (cat # EPA-1168), malonate (cat #102238), antimycin A (cat # ALX-380-075-M005) and DPI (diphenyleneiodonium, cat # 50-809-729) were purchased from Fisher Scientific. Antibodies against phospho-Akt (Ser 473, cat # 4060), total Akt (cat # 9272), and cleaved caspase 3 (Asp 175, Cat # 9661) were purchased from Cell Signaling. Antibodies against BrdU (5G15, cat# sc-70443), p27 (C-19, cat# sc-528), cyclin B1 (cat# sc-752), Cdc25C (cat# sc-327), Cdc25A (cat# sc-7389), and actin (cat# sc-1615) were purchased from Santa Cruz. Phospho-histone H3 antibody (cat # 06-570) was purchased from Upstate.
Determination of cellular ROS
Cellular ROS contents were measured by incubating cells with 20 μM DCF-DA or 10 μM DHE for 30 min followed by flow cytometry analysis.
Mitochondrial content and function
To determine the content of mitochondria, cells were incubated with NAO that provides a fluorescent signal proportional to mitochondrial content. Functionality of mitochondria was analyzed using rhodamine 123. Briefly, cells were incubated with each dye at 10 μM for 30 min before FACS analysis. MitoSOX ™ Red mitochondrial superoxide dye was used to measure mitochondrial superoxide levels after mitochondrial electron transport complex inhibitor treatment. Briefly, RWPE1-polyploid cells were treated with 1.5-5 μM rotenone, 1-5 mM malonate, 20 μM antimycin A for 30 min (Panee et al., 2007) (McLennan and Degli Esposti, 2000) and fluorescence signals of MitoSOX™ Red dye were measured by FACS analysis. For DPI, 1∼20 μM concentration was used.
Electron Microscopy
RWPE1-diploid as well as RWPE1-polyploid cells were fixed in glutaraldehyde and processed for electron microscopy analysis through the use of the Vanderbilt University Medical Center Cell Imaging Shared Resource.
Soft agar assay
For soft-agar colony formation assay, 0.8 × 105 cells were suspended in complete 2× medium containing 0.3% agarose and overlaid onto a bottom layer of solidified 0.5% agar in a complete medium as described in previous study (Roh et al., 2008). NAC was added in medium. The colonies were counted after 2-3 weeks. At least two independent experiments were performed.
Apoptosis and 5-Bromo-2′-deoxyuridine (BrdU) incorporation
Cells were seeded on cover slides, treated with 5 mM NAC for 2 or 4 days, and were processed for immunofluorescence using either active caspase 3 or anti-BrdU antibodies. After counterstaining with DAPI, images were captured with a Zeiss Axioskop 40 microscope. For BrdU incorporation, cells were first treated with 5 mM NAC for 2 days or 4 days, incubated with 10 μM BrdU (3 μg/ml) for 4 hr, and processed for immunofluorescence. Briefly, cells were washed with PBS, fixed in cold methanol, permeabilized with 0.25 % triton X-100 in PBS for 10 min, and then denatured with 1.5 N HCl for 10 minute followed by primary and secondary antibody incubation.
Cell viability and cell cycle analysis
Cell viability was measured using CellTiter-Glo Luminescence Cell Viability Assay kit (Promega, G 7570). Cell cycle profile was analyzed as described before (Roh et al., 2003). Briefly, cells were harvested, washed with PBS, fixed with cold 70 % ethanol, and stained with propidium iodide (0.05 mg/ml) in a solution containing 0.1 % Triton X-100 and RNase A (0.005 mg/ml) before analyzed for DNA content with a flow cytometer. For G1/S and mitotic synchronization, double thymidine and nocodazole were used respectively as described before (Roh et al., 2005). Briefly, cells were treated with 2 mM thymidine for 22 hr, released into normal medium for 10 hr, treated with thymidine for another 18 hr, and then released in the absence or presence of 400 μM H2O2 for indicated time points. For mitotic synchronization, cells were treated with 40 ng/ml nocodazole for 22 hr, released in the absence or presence of 400 μM H2O2. Samples were either fixed in cold 70 % ethanol and processed for cell cycle analysis by FACS, or were processed for immunoblotting.
Immunoblot analysis
Extracts of total protein as well as western blotting were performed as described before (Roh et al., 2008; Roh et al., 2003; Roh et al., 2005)
Statistical Analysis
All values are expressed as means ± SD of the indicated number of independent experiments. In some cases, each experiment was performed at multiple plates or dishes. The two-tailed, unpaired Student t test was used to compare two different groups and a P-value of <0.05 was considered statistically significant.
Results
Polyploid cells display higher ROS level than diploid cells
To investigate the possible contributions of ROS in tumorigenicity of cancer cells, we first measured ROS levels in polyploid prostate (RWPE1) and mammary (hTERT-HME) epithelial cells. These cells were established by stable expression of the Pim1 oncogene followed by FACS-sorting based on DNA content to isolate isogenic diploid and polyploid cells (Roh et al., 2003; Roh et al., 2005). The two cell populations expressed equal levels of Pim1, however only polyploid cells are tumorigenic (Roh et al., 2008). Using dichlorodihydrofluorescein diacetate (DCF-DA) fluorescent dye which measures peroxide levels, we found that elevated ROS levels in polyploid cells compared to diploid cells both in RWPE1 and hTERT-HME cells (Fig. 1A and B). To examine for a possible differences in dye uptake in diploid versus polyploid cells, we used an oxidation insensitive DCF-DA dye as a control. The results (Supplementary Fig.1) indicate that the polyploid cells showed a lower signal when the oxidation insensitive dye was used, suggesting that polyploid cells might have slower dye absorbance rate. Thus elevated levels of ROS observed in polyploid cells might in fact be an underestimate. Polyploid cells also showed increased level of superoxide as measured by the fluorescence signal of an oxidized form of dihydroethidium (Fig. 1C and D).
FIGURE 1.
Polyploid cells display higher ROS level than diploid cells. ROS levels of Pim1-overexpressing diploid and polyploid cells were measured by FACS using dichlorodihydrofluorescein diacetate (DCF-DA) or dihydroethidium (DHE) (bottom) fluorescence dye. A, C: RWPE1 cells. B, D: hTERT-HME cells. In both cell lines, ROS levels were elevated in polyploid cells. Data are represented as means ± SD of three independent experiments and significance of pair-wise comparisons is determined by Student's t test. Representative FACS profiles are also shown. The asterisk (*) indicates a significant increase in polyploid cells compared to diploid cells (**, P < 0.05, ***, P < 0.005).
Polyploid cells have higher mitochondrial content and mitochondria are the major source of ROS
Since mitochondria are a major source of ROS in animal cells, we assessed whether mitochondria are responsible for different ROS levels between diploid and polyploid cells. For this, cells were analyzed for mitochondrial content and function using nonyl acridine orange (NAO) and rhodamine 123, respectively, by FACS. NAO provides a fluorescence signal proportional to mitochondrial content, whereas rhodamine 123 accumulates in intact mitochondria, and a decrease of fluorescence indicates a loss of transmembrane potential, reflecting mitochondrial dysfunction. Our data showed that polyploid cells displayed higher levels of both NAO and rhodamine 123 than diploid cells (Fig. 2A and B). These results suggest that mitochondria of polyploid cells are intact and polyploid cells have higher number of mitochondria than diploid cells. The FACS results were further supported by electron microscopy (EM) analysis. EM data confirmed that polyploid cells contain higher numbers of mitochondria than diploid cells (Fig. 2C). However, the individual sizes of mitochondria themselves were similar in two cell populations (diploid, 491 ± 77 nm × 218 ± 63 nm, n=7; polyploid, 450 ± 115 nm × 233 ± 62 nm, n=16).
FIGURE 2.
Polyploid cells have higher mitochondrial content and mitochondria are the major source of ROS. A & B, relative signals of rhodamine and NAO fluorescence dyes. RWPE1-polyploid (A) or hTERT-HME- polyploid (B) cells as well as their control diploid cells were incubated with 10 μM of either rhodamine 123 or NAO for 30 min before FACS analysis. Rhodamine 123 and NAO measure mitochondrial function and contents respectively. In hTERT-HME cells, Neo empty vector control cells were also used as an additional control. (C). Representative EM image of RWPE1-polyploid cells demonstrating the higher number of mitochondria. Mitochondria were marked as *. Notice multiple mitochondria in the polyploid cell. (D). Relative DCF-DA signals after inhibitor treatment. RWPE1-polyploid cells were treated with rotenone, malonate, antimycin or DPI for 30 min, followed by DCF-DA addition and DCF-DA signals were measured. (E). Representative MitoSOX fluorescent images are shown. RWPE1-diploid and polyploid cells were stained with 5 μM MitoSOX for 10 min followed by counterstaining with 5 μg/ml Hoechst 33342 for 20 min and images were captured using ZEISS 17 microscopy. × 63. (F). Relative fluorescence signals of MitoSOX ™ Red mitochondrial superoxide dye after mitochondria electron transport chain complex inhibitors treatment. RWPE1-polyploid cells were used. (G) Relative DCF-DA signals of DCB treated-cells along with cell cycle profile after DCB treatment. RWPE1 cells were treated with 10 μM DCB for 24 hr and stained with DCF-DA along with Hoechst 33345. Diploid cells in G1 phase (2N) and tetraploid cells in G2/M phase (8N) were used to measure DCF-DA level. (H) Relative signals of DCF-DA, rhodamine 123 and NAO fluorescence dyes after DCB treatment. RWPE-Neo cells were used. (I) MitoSOX ™ Red signal after DCB treatment. Tetraploid cells in G2/M phase (8N) contain higher mitochondrial superoxide than diploid cells in G1 phase (2N). A, B, D, F, G, H, I: Data are represented as means ± SD of two to three independent experiments. **, P < 0.05, ***, P < 0.005.
To identify the source of the ROS, we employed mitochondria electron transport chain (ETC) complex inhibitors (rotenone; complex I and cytosolic ATP inhibitor, malonate; complex II inhibitor and antimycin A; complex III inhibitor) as well as NADH/NADPH oxidase inhibitor, diphenyliodonium (DPI). ETC inhibitors are known to increase the rate of ROS production from mitochondria (Boveris and Chance, 1973; Sugioka et al., 1988; Turrens and Boveris, 1980), whereas DPI decrease ROS production (Hancock and Jones, 1987). Results show that rotenone and antimycin A increased ROS levels as measured by DCF-DA signal, however DPI treatment had no effect on ROS levels (Fig. 2D and Supplementary Fig.2), indicating mitochondria is the major source of ROS in polyploid cells. Diploid cells treated with ETC complex inhibitors showed similar results like polyploid cells, suggesting that mitochondria is also the source ROS in diploid cells (Supplementary Fig.2). We also used MitoSOX™ Red mitochondrial superoxide indicator to specifically measure mitochondrial superoxide. As expected, MitoSOX™ Red stained-diploid and polyploid cells showed mitochondria staining pattern as shown in Fig. 2E. Consistent with DCF-DA data, MitoSOX™ Red levels were not changed in malonate treated cells compared to untreated cells, however they were significantly increased in rotenone and antimycin A treated cells (3.7-3.9 fold increase) (Fig. 2F), suggesting that complex I and III are the major player for ROS generation in polyploid cells. Our data also confirmed that polyploid cells have intact mitochondria as complex I and complex III have been reported to be the major ROS-generating sites in ETC in normal cells (Turrens and Boveris, 1980).
Mitochondrial mass is also high in DCB-induced polyploid cells
When cells have defect in cytokinesis, it is likely that they have more mitochondrial number and this can lead to higher ROS levels. Since polyploid cells are reported to have defect in cytokinesis (Roh et al., 2003), we hypothesized that cyokinesis defect can lead to higher number of mitochondria and this can lead to higher ROS production. To test this possibility, we employed dihydrocytochalasin B (DCB), a compound that prevents remodeling of the actin cytoskeleton. DCB has been known to induce tetraploidy due to cytokinesis failure (Fujiwara et al., 2005). When RWPE1-Neo or RWPE1-Diploid cells were treated with 2 μM DCB for 24 hrs, 34∼52 % cells were progressed into tetraploid/polyploid cells. Diploid cells in G1 phase (2N) and tetraploid cells in G2/M phase (8N) were used to measure ROS and mitochondrial dye signal (Fig. 2G). ROS levels measured by DCF-DA were higher in tetraploid cells than diploid cells (Fig. 2G). DCB-induced tetraploid cells showed higher level of mitochondrial mass than that of diploid cells as measured by rhodamine123 and NAO (Fig. 2H). Importantly, mitochondrial ROS levels measured by MitoSOX™ Red signal were also higher in DCB-induced tetraploid cells (Fig. 2I). Our results indicated that higher levels of ROS and mitochondrial mass observed in polyploid cell is possibly due to cytokinesis failure.
The antioxidant NAC abrogates tumorigenicity of polyploid cells
To assess the biological role of ROS in polyploid cells, we employed antioxidant reagent, N-Acetyl-L-cysteine (NAC). NAC was added once to the media to reconstitute the agar. Treatment of hTERT-HME polyploid cells with NAC for 2 weeks inhibited their tumorigenic potential in a dose-dependent manner as shown by soft agar assay (Fig. 3A and B). At 2 mM NAC concentration, the tumorigenicity of the cells was completely abrogated. 2 week NAC treatment had a similar effect on the tumorigenicity of RWPE1-polyploid cells as well (Fig. 3C). To rule out the possibility that this is simply due to a cytotoxic effect of NAC, we measured cell viability after different doses of NAC treatment using CellTiter-Glo Luminescence Cell Viability Assay. The results demonstrated that at a dose where tumorigenicity is severely impaired (1 mM), cell viability is only slightly affected (Supplementary Fig. 3A). RWPE1-polyploid cells treated with 1 mM NAC showed a slight reduction in cell viability (8.1% reduction) compared to non-treated cells (Supplementary Fig. 3A). However, the same dose of NAC treatment led to the dramatic reduction (85.5 % reduction) of colony forming ability of polyploid cells compared to non-treated cells (Fig. 3B). ROS levels measured by DCF-DA confirmed that NAC indeed reduced ROS levels (Supplementary Fig. 3B). To prove that this effect is truly due to the reduced ROS levels, we employed another antioxidant, vitamin E. When RWPE1-polyploid cells were treated with vitamin E, they also lost their tumorigenicity as shown by soft agar assay (Supplementary Fig. 4A). Vitamin E did not affect cell viability (Supplementary Fig. 4B).
FIGURE 3.
The antioxidant NAC abrogates tumorigenicity of polyploid cells and selective removal of polyploid cells by NAC in mixed polyploid/diploid cell cultures. (A). 5 mM NAC treatment for 2 weeks completely blocked colony formation of hTERT-HME-polyploid cells. (B). Colony counting for hTERT-HME-polyploid cells. The number of colonies was decreased in a NAC dose-dependent manner. Data are represented as means ± SD of three independent experiments (five plate each). The asterisk (*) indicates a significant decrease verse untreated group (P< 0.05). (C). Size of colonies was decreased with increasing dose of NAC in RWPE1-polyploid cells. Cells were treated with NAC for 3 weeks. (D). Cell cycle profile of RWPE1-polyploid cells after NAC treatment for 6 weeks. RWPE1-polyploid cells were treated with different doses of NAC for 6 weeks and analyzed for cell cycle profile by FACS. 5 mM NAC treatment preferentially eliminated polyploid cells, thus the minority diploid cells in the culture became a major cell population. (E). Graph showing dose-dependent decrease of polyploid cell population upon NAC treatment. (F). Representative soft agar colony images of NAC-treated polyploid cells as well as untreated polyploid cells. Post-NAC: polyploid cells treated for 6 weeks with 5 mM NAC. Pre-NAC: untreated control polyploid cells. (G). Cell viability of the same cells used for soft agar in E was measured after different dose of NAC treatment. Data are represented as means ± SD of three independent experiments. **, P < 0.05, ns, non significant.
Selective removal of polyploid cells by NAC in mixed polyploid/diploid cell cultures
FACS analysis for DNA content in our polyploid RWPE1 cells (see Figure 3d, 0 mM NAC) indicates the presence of a small fraction of cells with a 2N DNA profile (diploid cells). This provided us the opportunity to examine the effect of NAC treatment on the relative fraction of diploid/polyploid cells in the same culture and whether loss of tumorigenicity due to NAC treatment is linked to a change in the diploid/polyploid fraction. NAC treatment for 6 weeks led to a dose-dependent decrease in the proportion of polyploid cells with a concomitant increase in the proportion of diploid cells (Fig. 3D and E). At the highest dose of 5 mM NAC, the FACS profile has reverted from a largely polyploid to a largely diploid pattern (Fig. 3D). This profile is stable after ending the treatment, and the pre-NAC treated (original) and post-NAC treated (revertant) cells grew similarly even in the presence of NAC in vitro (Fig. 3G). However, the revertant cells have completely lost their tumorigenicity in soft agar assay (Fig. 3F), suggesting that the loss of tumorigenicity is not due to loss of cell viability. These results further solidify the link between polyploidy and tumorigenic potential and implicate ROS in maintaining the polyploid, tumorigenic population in this model.
Polyploid cells are more sensitive to the NAC inhibition of the Akt signaling pathway
Selective removal of polyploid cells after long-term treatment of mixed polyploid/diploid cell cultures with NAC may be due to an increase in cell death or a reduction in cell proliferation. Despite the increase of percentage of active caspase 3 positive cells after NAC treatment, there were no significant differences in apoptosis between diploid and polyploid cells treated with 5 mM NAC as determined by staining for the cleaved, active form of caspase 3 (Fig. 4A and Supplementary Fig. 5A). Active caspase 3 immunoblot confirmed that overall there is an increase in apoptosis after NAC treatment; however there was no significant difference in apoptosis in both cells (Supplementary Fig. 5C). By contrast, examination of DNA synthesis by BrdU incorporation showed a significant reduction in polyploid cells following NAC treatment (Fig. 4B and Supplementary Fig. 5B). Thus although DNA synthesis is inhibited by NAC in both diploid and polyploid cells, the polyploid cells are more sensitive to this inhibition.
FIGURE 4.
Polyploid cells are more sensitive to the NAC inhibition of the Akt signaling pathway. (A). Apoptotic rate was measured by active caspase 3. NAC treatment increased apoptosis in both cells; however there is no significant difference between diploid and polyploid cells. (B). DNA synthesis rate was measured by BrDU incorporation in RWPE1-diploid and RWPE1-polyploid cells after NAC treatment. % of BrdU positive cells in untreated cells was similar, however 5 mM NAC treatment decreased % of BrdU positive cells to 29.6 % (2 day), 20.8 % (4 day) (diploid cells), and to 18.8 % (2 day), 10.2 % (4 day) (polyploid cells). A, B: Data are represented as means ± SD of two to three independent experiments. **, P < 0.05, ns, not significant. (C). Reduced Akt activity upon NAC treatment. Polyploid as well as diploid cells were treated with 5 mM NAC in different time point, and Akt activity was measured by phospho-Akt immunoblotting. (D). Akt activity is NAC dose-dependent and polyploid cells are more sensitive to NAC treatment. Different dose of NAC was treated to cells for 24 hr and Akt activity was measured by phospho-Akt immunoblotting. (E). Graph showing quantitation of D. Phospho-Akt level normalized by total Akt level after NAC treatment. Notice dramatically reduced phospho-Akt level in polyploid cells after NAC treatment. (F). Effects of Akt inhibitor on its target molecule. Inhibition of Akt led to the increased level of its target molecule p27kip1. (G). Effects of Akt inhibitor, triciribine, on polyploid cell cycle profile. Polyploid cell fraction was decreased over time upon Akt inhibitor treatment, partially mimicking NAC's effect (see Fig. 3E).
ROS can affect several signaling pathways to mediate effects on cell proliferation and tumorigenicity, including the Akt signaling pathway (Ma et al., 2009; Wang et al., 2000). NAC treatment attenuated Akt activity as shown by reduced phospho-Akt levels in both diploid and polyploid cells (Fig. 4C), consistent with previously published results (Ma et al., 2009; Wang et al., 2000). However, quantitation of phospho-Akt levels following treatment with different doses of NAC indicated that polyploid cells are much more sensitive to inhibitory effect of NAC on phospho-Akt than diploid cells (Fig. 4D and E). At the low dose of 0.5 mM NAC, Akt activity was unaffected in diploid cells, while polyploid cells show a dramatic reduction in Akt activity (down to 28.7%).
We further examined the role of the Akt signaling pathway in mediating the effects of NAC on polyploid cells through the use of the Akt inhibitor, triciribine (Yang et al., 2004). In addition to phospho-Akt, we also examined the level of p27kip1, a cell cycle inhibitor that is a downstream target of the Akt signaling pathway (Rassidakis et al., 2005). Treatment of diploid and polyploid cells with triciribine inhibited Akt phosphorylation and led to the accumulation of p27kip1 protein (Fig. 4F). The effect of triciribine is more apparent in polyploid than diploid cells. Total p27kip1 levels were significantly increased even after 3 hr treatment of triciribine in polyploid cells, whereas there was no significant change in diploid cells at the same time point (Fig. 4F, lane 2 & 8). Accordingly, long term treatment of mixed polyploid/diploid cultures with triciribine led to a gradual decrease in the proportion of polyploid cells with a concomitant increase in diploid cells (Fig. 4G). Together these results suggest that selective removal of polyploid cells by NAC treatment is at least in part mediated through effects on the Akt signaling pathway.
Increasing ROS levels using hydrogen peroxide (H2O2) delays mitotic entry and exit
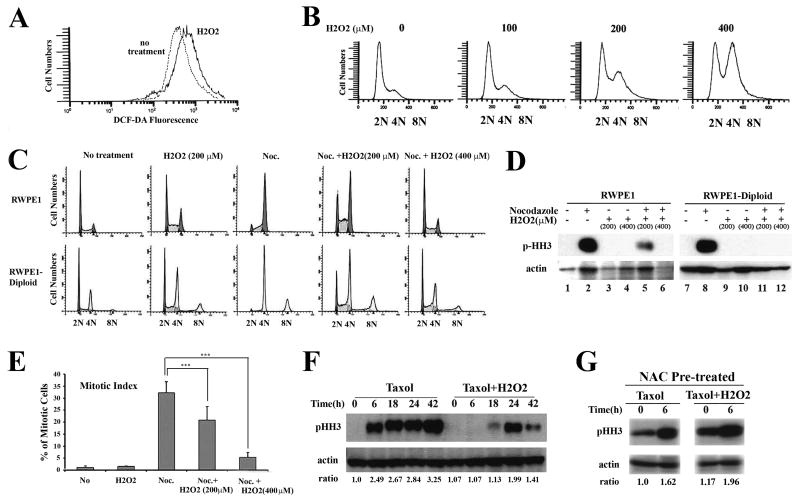
Previous studies have implicated ROS in chromosomal instability by affecting the mitotic checkpoint and chromosome segregation (Chae et al., 2005; D'Angiolella et al., 2007; Zhang et al., 2003). Increased ROS levels in polyploid cells can thus lead to further chromosomal abnormalities by affecting proper mitotic function. We examined this by treating RWPE1 cells with H2O2 to induce oxidative stress, which is confirmed by higher DCF-DA signal in the treated cells (Fig. 5A). Cell cycle analysis revealed a dose-dependent G2/M cell cycle arrest in the treated cells as expected (Fig. 5B). Furthermore, treating RWPE1 and RWPE1-Pim1-diploid cells with 40 ng/ml nocodazole in the presence of H2O2 for 18 hrs attenuated the accumulation of cells in the G2/M phase of the cell cycle, phosphorylation of histone H3 (marker of mitosis), and mitotic index determined by DAPI staining (Fig. 5C-E). Similarly, treatment of RWPE1 cells with taxol in the presence of H2O2 attenuated the accumulation of cells in M phase as shown by a delayed and reduced phosphorylation of histone H3 (Fig. 5F). These results are consistent with either H2O2 delaying entry of the cells into mitosis with an attendant arrest in G2 and/or impairing exit from mitosis.
FIGURE 5.
Increasing ROS using hydrogen peroxide impairs mitotic progression. (A). ROS levels measured by DCF-DA were increased after H2O2 treatment. RWPE1 cells were treated with 200 μM H2O2 for 24 hr and DCF-DA signal intensity was measured. (B). H2O2 treatment induces G2/M cell cycle arrest in a H2O2 dose-dependent manner. RWPE1 cells were used. Data are representative FACS profiles of three independent experiments (C). Nocodazole-induced mitotic cell cycle arrest was abrogated by H2O2 treatment. RWPE1 and RWPE1-diploid cells were treated with 40 nM nocodazole along with H2O2 for 22 hr and cell cycles were analyzed by FACS. Nocodazole treatment induced robust mitotic cell cycle arrest as shown by increased G2/M cell population, however H2O2 treatment abrogated nocodazole-induced mitotic cell cycle arrest as shown by reduced fraction of G2/M cell population. Data are representative FACS profiles of three independent experiments (D). Mitotic checkpoint activation induced by nocodazole is weakened by H2O2. Phospho-histone H3 level was significantly lower or absent in cells treated with nocodazole and H2O2 than in nocodazole only treated-cells (compare lane 2 with lane 5, 6 and lane 8 with 11, 12). (E). Reduced mitotic index after H2O2 and nocodazole treatment. Mitotic index was calculated by counting mitotic cells of DAPI stained cells after H2O2 treatment along with nocodazole. Data are represented as means ± SD of three independent experiments. ***, P < 0.005. (F). Mitotic checkpoint of RWPE1 cells was examined by immunoblotting using phospho-histone H3 after 10 nM taxol treatment along with H2O2 treatment at a different time point. H2O2 treatment not only delayed mitotic checkpoint activation but also failed to sustain it. (G) NAC-pretreated RWPE1 cells can induce phospho-histone H3 after 6 hr taxol treatment even in the presence of H2O2, suggesting that NAC can abrogate effect elicited by H2O2.
To distinguish between these possibilities, we used cell synchronization. For analysis of mitotic entry, we synchronized RWPE1 cells in G1/S phase through a double thymidine block, then released them in the presence or absence of H2O2 for 24 hr. Cells released in the absence of H2O2 showed progressive increase of G2/M cell fraction, while cells released in the presence of H2O2 did not progress to G2/M up to 12 hrs later (Fig. 6A and B). This indicates delayed progression to G2/M by H2O2. To examine this further, we examined levels of the mitotic cell marker, phospho-histone H3 in the same samples. Phospho-histone H3 begin to appear from 8 hr and reached its maximum level at 12 hr in cells released without H2O2, however appearance of phospho-histone H3 was delayed until 12 hr in cells released with H2O2 (Fig. 6C), indicating delay in mitotic entry (G2 arrest). For analysis of mitotic exit, we synchronized cells in M phase by nocodazole treatment, and then released them into media supplemented or not supplemented with H2O2. Releasing cells into media containing H2O2 led to a significant delay in exit from mitosis as shown by higher percentage of G2/M determined by FACS (Fig. 6D and E). This was confirmed by measuring the levels of cyclin B1 by immunoblotting in the same samples, as degradation of cyclin B1 is known to be required to exit from mitosis (Murray, 1995; Murray et al., 1989). Release of cells in the presence of H2O2 led to a delay in the loss of cyclin B1 (Fig. 6F).
FIGURE 6.
Increasing ROS using hydrogen peroxide delays both mitotic entry and exit. RWPE1 cells were used. A-C: Thymidine-synchronization. H2O2 delayed entry into mitosis and induced G2 cell cycle arrest. (A). Cell cycle analysis of thymidine-synchronized RWPE1 cells released with and without H2O2. (B). Graph showing G2/M cell fraction after release from thymidine-synchronization with and without H2O2. Cells released in the presence of H2O2 show delayed entry into mitosis compared to cells released in the absence of H2O2. (C). H2O2 delayed entry into mitosis and G2 cell cycle arrest as shown by delayed appearance of phospho-histone H3 in cells released in the presence of H2O2. Phospho-histone H3 levels were examined using the samples prepared like FACS analysis. Phospho-histone H3 begin to appear from 8 hr and reached maximum level at 12 hr time point in cells released without H2O2 (lane 4-6), however it did not appear until 12 hr (lane 12) in cells released in the presence of H2O2, indicating delayed entry into mitosis. D-F: Nocodazole synchronization. H2O2 blocked mitotic cell cycle exit. RWPE1 cells were synchronized in mitosis by nocodazole, released in the presence of H2O2, and cyclin B1 level was measured by immunoblotting (D). Cell cycle analysis of nocodazole-synchronized RWPE1 cells released in the absence or presence of H2O2. Notice almost complete block of mitotic cell cycle exit in cells released in the presence of H2O2. (E). Graph showing G2/M cell fraction after release from nocodazole-synchronization in the absence or presence of H2O2. G2/M cell fraction decreased over time in cells released in the absence of H2O2 due to cell cycle progression to G1 phase, however, G2/M cell fraction almost unchanged in cells released in the presence of H2O2, indicating block of mitotic cell cycle exit. (F). Inhibition of cyclin B1 degradation in cells released in the presence of H2O2 after nocodazole synchronization. Cyclin B1 level was reduced in cells released in the absence of H2O2, indicating mitotic exit, however H2O2 treatment inhibited cyclin B1 degradation. B, E: Data are represented as mean ± SD of three independent experiments. The asterisk (*) indicates a significant difference between each group (P< 0.05).
Enhanced ROS level renders paclitaxel (taxol) resistance
The ability of H2O2 to interfere with mitotic progression suggests that it may affect the potency of chemotherapeutic agents that function in mitosis (mitotic poisons), such as paclitaxel (taxol). Interestingly, recent studies indicated that overexpression of Pim1 might confer resistance to chemotherapeutic drugs (Mumenthaler et al., 2009; Xie et al., 2006). Consistent with these, we observed that polyploid RWPE-Pim1 cells are taxol resistant compared to their control RWPE1-Neo counterparts (Fig. 7A). RWPE1-Neo cells contain lower levels of ROS (data not shown) and are taxol sensitive, whereas, polyploid cells contain higher levels of ROS and they are taxol resistant, suggesting that ROS level might affect taxol response. Indeed, two day taxol treatment reduced cell viability to 44.6-52.7 % compared to that of untreated cells, but H2O2 treatment largely reversed this effect (Fig. 7B). H2O2 treatment alone under these conditions had only a modest effect on cell viability in a dose-dependent manner (81.6-86.2 %). These results suggest that elevated ROS levels, as seen in polyploid cells, can confer taxol resistance. To rule out the possibility that these effects are due to an interaction between taxol and H2O2, we treated the cells separately, first with H2O2 and then with taxol. For this, we first tested whether 1 day H2O2 treatment can sustain ROS level and G2/M cell cycle arrest even after release from H2O2. Our results showed that cells treated with 200 μM H2O2 for 1 day and released into normal medium for 2 days were able to maintain ROS levels and G2/M cell cycle arrest (Fig. 7C). Next, we treated cells with 200 or 400 μM H2O2 for 1 day, followed by 5 or 10 nM taxol for two days subsequently. Our results showed that separate treatment also significantly increased cell viability compared to taxol only treatment (Fig. 7D), indicating that increasing ROS level can promote taxol resistance. Interestingly, when the same cells were treated with NAC and taxol, either simultaneous or separately, cells became sensitized to taxol (Fig. 7E and supplementary Fig.6).
FIGURE 7.
Enhanced ROS level renders paclitaxel (taxol) resistance. (A). Pim1-overexpressing RWPE1 cells are taxol resistant as shown by significantly lower percentage of sub G1 cell population than control Neo cells after taxol treatment. (B). Combined treatment of RWPE1 cells with H2O2 along with taxol significantly decreased taxol-induced cytotoxicity (increased taxol resistance). Compare taxol only with H2O2 and taxol treatment. (C). H2O2 treatment for one day followed by two day release can maintain ROS level as well as G2/M cell cycle arrest in RWPE1 cells. (D). Separate treatment of H2O2 for one day followed by two day taxol treatment also increased taxol resistance significantly compared to taxol only treatment. (E). RWPE1 cells treated with NAC for one day followed by 2 day taxol treatment significantly decreased taxol resistance compared to taxol only treatment. (F). Model summarizing our data. Polyploidy is believed to induce chromosomal instability (CIN) and this can lead to tumor by inducing further chromosomal instabilities, for example, gain of oncogenes or loss of tumor suppressor genes (TSG). ROS, on the other hand, can also contribute to tumorigenicity by affecting mitotic checkpoint, cell cycle progression and signaling pathway, for example Akt pathway. Increased ROS is demonstrated to cause delay in cell cycle progression, both entry into and exit from mitosis, and this can lead to CIN and tumorigenicity. Interestingly, enhanced ROS also caused taxol resistance. A, B, D, E: Data are represented as mean ± SD of three independent experiments (each 2∼98 different plates or well). **, P < 0.05, ***, P < 0.005.
Discussion
Polyploidy is thought to represent a pro-tumorigenic condition mainly through its ability to lead to chromosomal instability and therefore gains or losses of cancer genes that can confer a selective growth advantage to the emerging tumor cell (Fujiwara et al., 2005; Storchova and Pellman, 2004). The mechanisms that give rise to polyploidy, including failure of cell division and cell fusion can also result in a higher mitochondrial content. Mitochondria are a major source of ROS in the cells as by products of cellular metabolism and ROS can play diverse roles in the cell including promoting tumorigenicity (Naziroglu, 2009) (Droge, 2002; Suh et al., 1999; Szatrowski and Nathan, 1991). Our study suggests that the increased number of mitochondria in polyploid cells produce high levels of ROS that contribute to the tumorigenicity of these cells by affecting mitosis and cells signaling pathways such as Akt (Fig. 7E). Thus in addition to having increased number of chromosomes, which can fuel chromosomal instability, the increased mitochondrial content of polyploid cells may further promote tumorigenicity via increased production of ROS.
In this study, we have demonstrated that tumorigenic polyploid cells induced by Pim-1 overexpression display higher levels of ROS compared to non-tumorigenic diploid cells. High levels of ROS in polyploid cells seem to be due to the increased number of mitochondria in polyploid cells rather than mitochondrial dysfunction since polyploid cells showed higher levels of both nonyl acridine orange (NAO) and rhodamine 123 dye than those in diploid cells. EM data also confirmed FACS results. When cells were treated with mitochondria electron transport chain complex inhibitors, rotenone and antimycin A, ROS levels were significantly reduced; however NADH/NADPH oxidase (NOX) inhibitor, diphenyliodonium, did not affect ROS level, indicating that mitochondria, not NOX, is the major source of ROS in our system. Other possible reason for the increased ROS in polyploid cells is that the ROS scavenging capacity of polyploid cells is also reduced. To address this possibility, we examined expression levels of some of antioxidant enzymes by RT PCR. However, we did not find any significant difference in expression levels of ROS scavenging enzymes such as manganese superoxide dismutase (MnSOD), glutathione peroxidase 2 (Gpx 2), Gpx 3, and Quiescin Q6 (Qscn6) between diploid and polyploid cells (data not shown). Overall our data suggest that polyploid cells have intact but increased number of mitochondria, however possible contribution of mitochondrial dysfunction to tumorigenicity in polyploid cells cannot be completely excluded. In fact, mitochondrial dysfunction is reported to contribute to genomic instability by increasing ROS level after exposure to ionizing radiation (Dayal et al., 2009). Mitochondrial DNA mutation and/or altered metabolism in polyploid cells might also contribute to the high levels of ROS. It is also possible that ROS promoted genetic instability via DNA damage. In support of this, chronic oxidative stress by H2O2 is reported to induce mutator phenotype (Dayal, 2008 #149).
A significant finding from our study is to show that polyploid cells are much more sensitive to the inhibitory effects of the well-known antioxidant, NAC. Not only did NAC treatment abrogate the tumorigenicity of polyploid cells, it also preferentially inhibited their proliferation. Remarkably, 6 week NAC treatment of cells containing both polyploid and diploid fractions led to the “curing” of these cells with the almost complete selective removal of the polyploid cells. The inhibition of polyploid cell tumorigenicity by antioxidants such as NAC and vitamin E supports a causal link between oxidative stress and tumorigenesis. The increased sensitivity of polyploid cells to NAC treatment is also reflected in the effects on the Akt signaling pathway. ROS can activate Akt signaling pathway via inactivation of PTEN, a negative regulator of Akt, by oxidative modification (Leslie et al., 2003). Interestingly, Akt is also reported to increase ROS levels (Nogueira et al., 2008), thus there seem to be a positive feedback loop between ROS and Akt pathway. In our study, we found that not only can NAC inhibit Akt activity, but polyploid cells are more sensitive to this inhibitory of NAC on Akt activity as measured by Akt phosphorylation. Polyploid cells were also more sensitive to the Akt inhibitor triciribine than diploid cells as shown by Akt phosphorylation and accumulation of the Akt target p27kip1. Our observations support the notion that the NAC effects observed on polyploid cells are at least partially mediated via the Akt signaling pathway. Akt inhibition by triciribine however did not fully recapitulate the effects of NAC, indicating that other pathways affected by ROS also might play a role. These results imply that antioxidants may have a role as chemo-preventive agents to selectively eliminate polyploid cells with the potential to develop into tumors. For example, mutation of the Adenomatous Polyposis Coli (APC) tumor suppressor has been shown to lead to cytokinesis failure and tetraploidy prior to the development of adenomas (Caldwell et al., 2007; Dikovskaya et al., 2007). Therefore, strategies to selectively eliminate tetraploid cells using antioxidants could be useful in this setting.
We explored how ROS may promote polyploidy/aneuploidy by analyzing the effects of ROS on the mitotic stage of the cell cycle in our model. We found that ROS interferes with mitosis by delaying both entry into and exit from mitosis. Consistent with our findings, oxidative stress has been shown to cause G2 arrest and to delay exit from mitosis (Barnouin et al., 2002; Sarsour et al., 2008). ROS may affect mitotic entry and exit by affecting molecules regulating these processes, including the Cdc25 phosphatase and cyclin B1. In fact, ROS have been considered to regulate the cell cycle by modifying the redox state of cell cycle regulatory molecules (Uguz et al., 2009; Menon and Goswami, 2007). Interestingly, Cdc25 was reported to be rapidly inactivated by oxidation, for example by H2O2 (Seth and Rudolph, 2006; Sohn and Rudolph, 2003). Activation of Cdc25 is required for mitotic cell entry. The anaphase-promoting complex 11 (APC 11) is also reported as a target of H2O2 (Chang et al., 2004; Rhee et al., 2005). APC regulates the onset of sister-chromatid separation and exit from mitosis by regulating ubiquitination and degradation of securin and cyclin B (Nilsson et al., 2008). Cyclin B1 degradation is necessary for mitotic exit. Our data showing delayed mitotic entry and exit by H2O2 could be due to inactivation of Cdc25C and APC complex by H2O2. Our data showing lack of activation of phospho-histione H3 after taxol or nocodazole treatment in the presence of H2O2 also suggested that ROS affect mitotic checkpoint. The intact mitotic checkpoint ensures that a proper genomic content is passed on to both daughter cells by delaying the onset of anaphase until all chromosomes are properly oriented on the microtubule spindle. Defects in the mitotic checkpoint can lead to chromosome mis-segregation, aneuploidy, cell death, and even early embryonic lethality (Fernandez-Capetillo and Nussenzweig, 2004; Kops et al., 2005; Holland and Cleveland, 2009). Together, our data strongly supports the idea that ROS contribute to tumorigenesis by affecting cell cycle progression and mitotic checkpoint.
Another important finding of our study is the observation that ROS level could modulate taxol sensitivity. Since ROS play a critical role in tumorigenicity, treating cancer with antioxidant has been considered an attractive therapy. However, combined use of antioxidant with chemotherapeutic agents during cancer therapy has produced inconclusive results (Conklin, 2000; Seifried et al., 2003; Wang and Yi, 2008). Ironically, ROS producing-agents have also been exploited to kill cancer cells (Pelicano et al., 2004; Trachootham et al., 2006; Yi et al., 2004), emphasizing the differential effects of ROS on cells based on the cell context and amount of ROS. In our study, we found that enhancing ROS with H2O2 can increase taxol resistance. This effect is probably due to the fact that H2O2 treatment delays entry into mitosis, the cell cycle stage in which mitotic poisons like taxol acts.
Supplementary Material
Acknowledgments
We thank Dr. Chisu Song (Infectious disease, Department of Medicine, Vanderbilt University) for helpful discussions and critical reading, Dr. Douglas R. Spitz, Dr. Nukhet Aykin-Burns (Department of Radiation Oncology, Iowa University) and Dr. David Gius (Department of Radiation Oncology, Vanderbilt University) for helpful discussions, Dr. William D. Dupont for statistical advice, Cathy Alford (VA Hospital, Flow Cytometry Laboratory) and the Vanderbilt Electron Microscopy Core facility for technical assistance.
Grant support: NIH grant R01 CA123484 (S.A.A.)
Literature Cited
- Barnouin K, Dubuisson ML, Child ES, Fernandez de Mattos S, Glassford J, Medema RH, Mann DJ, Lam EW. H2O2 induces a transient multi-phase cell cycle arrest in mouse fibroblasts through modulating cyclin D and p21Cip1 expression. J Biol Chem. 2002;277(16):13761–13770. doi: 10.1074/jbc.M111123200. [DOI] [PubMed] [Google Scholar]
- Boveri T. Concerning the origin of malignant tumours by Theodor Boveri. Translated and annotated by Henry Harris. J Cell Sci. 2008;121(1):1–84. doi: 10.1242/jcs.025742. [DOI] [PubMed] [Google Scholar]
- Boveris A, Chance B. The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J. 1973;134(3):707–716. doi: 10.1042/bj1340707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell CM, Green RA, Kaplan KB. APC mutations lead to cytokinetic failures in vitro and tetraploid genotypes in Min mice. J Cell Biol. 2007;178(7):1109–1120. doi: 10.1083/jcb.200703186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castillo A, Morse HC, 3rd, Godfrey VL, Naeem R, Justice MJ. Overexpression of Eg5 causes genomic instability and tumor formation in mice. Cancer Res. 2007;67(21):10138–10147. doi: 10.1158/0008-5472.CAN-07-0326. [DOI] [PubMed] [Google Scholar]
- Chae S, Yun C, Um H, Lee JH, Cho H. Centrosome amplification and multinuclear phenotypes are Induced by hydrogen peroxide. Exp Mol Med. 2005;37(5):482–487. doi: 10.1038/emm.2005.59. [DOI] [PubMed] [Google Scholar]
- Chang TS, Jeong W, Lee DY, Cho CS, Rhee SG. The RING-H2-finger protein APC11 as a target of hydrogen peroxide. Free Radic Biol Med. 2004;37(4):521–530. doi: 10.1016/j.freeradbiomed.2004.05.006. [DOI] [PubMed] [Google Scholar]
- Conklin KA. Dietary antioxidants during cancer chemotherapy: impact on chemotherapeutic effectiveness and development of side effects. Nutr Cancer. 2000;37(1):1–18. doi: 10.1207/S15327914NC3701_1. [DOI] [PubMed] [Google Scholar]
- D'Angiolella V, Santarpia C, Grieco D. Oxidative stress overrides the spindle checkpoint. Cell Cycle. 2007;6(5):576–579. doi: 10.4161/cc.6.5.3934. [DOI] [PubMed] [Google Scholar]
- Dayal D, Martin SM, Owens KM, Aykin-Burns N, Zhu Y, Boominathan A, Pain D, Limoli CL, Goswami PC, Domann FE, Spitz DR. Mitochondrial complex II dysfunction can contribute significantly to genomic instability after exposure to ionizing radiation. Radiat Res. 2009;172(6):737–745. doi: 10.1667/RR1617.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dikovskaya D, Schiffmann D, Newton IP, Oakley A, Kroboth K, Sansom O, Jamieson TJ, Meniel V, Clarke A, Nathke IS. Loss of APC induces polyploidy as a result of a combination of defects in mitosis and apoptosis. J Cell Biol. 2007;176(2):183–195. doi: 10.1083/jcb.200610099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Droge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002;82(1):47–95. doi: 10.1152/physrev.00018.2001. [DOI] [PubMed] [Google Scholar]
- Fernandez-Capetillo O, Nussenzweig A. Aging counts on chromosomes. Nat Genet. 2004;36(7):672–674. doi: 10.1038/ng0704-672. [DOI] [PubMed] [Google Scholar]
- Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature. 2005;437(7061):1043–1047. doi: 10.1038/nature04217. [DOI] [PubMed] [Google Scholar]
- Gorla GR, Malhi H, Gupta S. Polyploidy associated with oxidative injury attenuates proliferative potential of cells. J Cell Sci. 2001;114(Pt 16):2943–2951. doi: 10.1242/jcs.114.16.2943. [DOI] [PubMed] [Google Scholar]
- Hancock JT, Jones OT. The inhibition by diphenyleneiodonium and its analogues of superoxide generation by macrophages. Biochem J. 1987;242(1):103–107. doi: 10.1042/bj2420103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland AJ, Cleveland DW. Boveri revisited: chromosomal instability, aneuploidy and tumorigenesis. Nat Rev Mol Cell Biol. 2009;10(7):478–487. doi: 10.1038/nrm2718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kops GJ, Weaver BA, Cleveland DW. On the road to cancer: aneuploidy and the mitotic checkpoint. Nat Rev Cancer. 2005;5(10):773–785. doi: 10.1038/nrc1714. [DOI] [PubMed] [Google Scholar]
- Kraniak JM, Abrams J, Nowak JE, Tainsky MA. Antioxidant agents transiently inhibit aneuploidy progression in Li-Fraumeni cell strains. Mol Carcinog. 2006;45(3):141–156. doi: 10.1002/mc.20145. [DOI] [PubMed] [Google Scholar]
- Leslie NR, Bennett D, Lindsay YE, Stewart H, Gray A, Downes CP. Redox regulation of PI 3-kinase signalling via inactivation of PTEN. Embo J. 2003;22(20):5501–5510. doi: 10.1093/emboj/cdg513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Limoli CL, Giedzinski E, Morgan WF, Swarts SG, Jones GD, Hyun W. Persistent oxidative stress in chromosomally unstable cells. Cancer Res. 2003;63(12):3107–3111. [PubMed] [Google Scholar]
- Ma Q, Cavallin LE, Yan B, Zhu S, Duran EM, Wang H, Hale LP, Dong C, Cesarman E, Mesri EA, Goldschmidt-Clermont PJ. Antitumorigenesis of antioxidants in a transgenic Rac1 model of Kaposi's sarcoma. Proc Natl Acad Sci U S A. 2009;106(21):8683–8688. doi: 10.1073/pnas.0812688106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin KR, Barrett JC. Reactive oxygen species as double-edged swords in cellular processes: low-dose cell signaling versus high-dose toxicity. Hum Exp Toxicol. 2002;21(2):71–75. doi: 10.1191/0960327102ht213oa. [DOI] [PubMed] [Google Scholar]
- McLennan HR, Degli Esposti M. The contribution of mitochondrial respiratory complexes to the production of reactive oxygen species. J Bioenerg Biomembr. 2000;32(2):153–162. doi: 10.1023/a:1005507913372. [DOI] [PubMed] [Google Scholar]
- Menon SG, Goswami PC. A redox cycle within the cell cycle: ring in the old with the new. Oncogene. 2007;26(8):1101–1109. doi: 10.1038/sj.onc.1209895. [DOI] [PubMed] [Google Scholar]
- Mumenthaler SM, Ng PY, Hodge A, Bearss D, Berk G, Kanekal S, Redkar S, Taverna P, Agus DB, Jain A. Pharmacologic inhibition of Pim kinases alters prostate cancer cell growth and resensitizes chemoresistant cells to taxanes. Mol Cancer Ther. 2009;8(10):2882–2893. doi: 10.1158/1535-7163.MCT-09-0293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray A. Cyclin ubiquitination: the destructive end of mitosis. Cell. 1995;81(2):149–152. doi: 10.1016/0092-8674(95)90322-4. [DOI] [PubMed] [Google Scholar]
- Murray AW, Solomon MJ, Kirschner MW. The role of cyclin synthesis and degradation in the control of maturation promoting factor activity. Nature. 1989;339(6222):280–286. doi: 10.1038/339280a0. [DOI] [PubMed] [Google Scholar]
- Nakatani T, Inouye M, Mirochnitchenko O. Overexpression of antioxidant enzymes in transgenic mice decreases cellular ploidy during liver regeneration. Exp Cell Res. 1997;236(1):137–146. doi: 10.1006/excr.1997.3715. [DOI] [PubMed] [Google Scholar]
- Naziroglu M. Role of Selenium on Calcium Signaling and Oxidative Stress-induced Molecular Pathways in Epilepsy. Neurochem Res. 2009 doi: 10.1007/s11064-009-0015-8. [DOI] [PubMed] [Google Scholar]
- Nilsson J, Yekezare M, Minshull J, Pines J. The APC/C maintains the spindle assembly checkpoint by targeting Cdc20 for destruction. Nat Cell Biol. 2008;10(12):1411–1420. doi: 10.1038/ncb1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nogueira V, Park Y, Chen CC, Xu PZ, Chen ML, Tonic I, Unterman T, Hay N. Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell. 2008;14(6):458–470. doi: 10.1016/j.ccr.2008.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panee J, Liu W, Nakamura K, Berry MJ. The responses of HT22 cells to the blockade of mitochondrial complexes and potential protective effect of selenium supplementation. Int J Biol Sci. 2007;3(5):335–341. doi: 10.7150/ijbs.3.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelicano H, Carney D, Huang P. ROS stress in cancer cells and therapeutic implications. Drug Resist Updat. 2004;7(2):97–110. doi: 10.1016/j.drup.2004.01.004. [DOI] [PubMed] [Google Scholar]
- Rassidakis GZ, Feretzaki M, Atwell C, Grammatikakis I, Lin Q, Lai R, Claret FX, Medeiros LJ, Amin HM. Inhibition of Akt increases p27Kip1 levels and induces cell cycle arrest in anaplastic large cell lymphoma. Blood. 2005;105(2):827–829. doi: 10.1182/blood-2004-06-2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhee SG, Yang KS, Kang SW, Woo HA, Chang TS. Controlled elimination of intracellular H(2)O(2): regulation of peroxiredoxin, catalase, and glutathione peroxidase via post-translational modification. Antioxid Redox Signal. 2005;7(5-6):619–626. doi: 10.1089/ars.2005.7.619. [DOI] [PubMed] [Google Scholar]
- Roh M, Franco OE, Hayward SW, van der Meer R, Abdulkadir SA. A role for polyploidy in the tumorigenicity of Pim-1-expressing human prostate and mammary epithelial cells. PLoS One. 2008;3(7):e2572. doi: 10.1371/journal.pone.0002572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roh M, Gary B, Song C, Said-Al-Naief N, Tousson A, Kraft A, Eltoum IE, Abdulkadir SA. Overexpression of the oncogenic kinase Pim-1 leads to genomic instability. Cancer Res. 2003;63(23):8079–8084. [PubMed] [Google Scholar]
- Roh M, Song C, Kim J, Abdulkadir SA. Chromosomal instability induced by Pim-1 is passage-dependent and associated with dysregulation of cyclin B1. J Biol Chem. 2005;280(49):40568–40577. doi: 10.1074/jbc.M509369200. [DOI] [PubMed] [Google Scholar]
- Sarsour EH, Venkataraman S, Kalen AL, Oberley LW, Goswami PC. Manganese superoxide dismutase activity regulates transitions between quiescent and proliferative growth. Aging Cell. 2008;7(3):405–417. doi: 10.1111/j.1474-9726.2008.00384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seifried HE, McDonald SS, Anderson DE, Greenwald P, Milner JA. The antioxidant conundrum in cancer. Cancer Res. 2003;63(15):4295–4298. [PubMed] [Google Scholar]
- Seth D, Rudolph J. Redox control of cell cycle progression via Cdc25 phosphatase (Mih1p) in S. cerevisiae. Cell Cycle. 2006;5(18):2172–2173. doi: 10.4161/cc.5.18.3252. [DOI] [PubMed] [Google Scholar]
- Sohn J, Rudolph J. Catalytic and chemical competence of regulation of cdc25 phosphatase by oxidation/reduction. Biochemistry. 2003;42(34):10060–10070. doi: 10.1021/bi0345081. [DOI] [PubMed] [Google Scholar]
- Sotillo R, Hernando E, Diaz-Rodriguez E, Teruya-Feldstein J, Cordon-Cardo C, Lowe SW, Benezra R. Mad2 overexpression promotes aneuploidy and tumorigenesis in mice. Cancer Cell. 2007;11(1):9–23. doi: 10.1016/j.ccr.2006.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Storchova Z, Pellman D. From polyploidy to aneuploidy, genome instability and cancer. Nat Rev Mol Cell Biol. 2004;5(1):45–54. doi: 10.1038/nrm1276. [DOI] [PubMed] [Google Scholar]
- Sugioka K, Nakano M, Totsune-Nakano H, Minakami H, Tero-Kubota S, Ikegami Y. Mechanism of O2- generation in reduction and oxidation cycle of ubiquinones in a model of mitochondrial electron transport systems. Biochim Biophys Acta. 1988;936(3):377–385. doi: 10.1016/0005-2728(88)90014-x. [DOI] [PubMed] [Google Scholar]
- Suh YA, Arnold RS, Lassegue B, Shi J, Xu X, Sorescu D, Chung AB, Griendling KK, Lambeth JD. Cell transformation by the superoxide-generating oxidase Mox1. Nature. 1999;401(6748):79–82. doi: 10.1038/43459. [DOI] [PubMed] [Google Scholar]
- Szatrowski TP, Nathan CF. Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Res. 1991;51(3):794–798. [PubMed] [Google Scholar]
- Trachootham D, Zhou Y, Zhang H, Demizu Y, Chen Z, Pelicano H, Chiao PJ, Achanta G, Arlinghaus RB, Liu J, Huang P. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer Cell. 2006;10(3):241–252. doi: 10.1016/j.ccr.2006.08.009. [DOI] [PubMed] [Google Scholar]
- Turrens JF, Boveris A. Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem J. 1980;191(2):421–427. doi: 10.1042/bj1910421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uguz AC, Naziroglu M, Espino J, Bejarano I, Gonzalez D, Rodriguez AB, Pariente JA. Selenium modulates oxidative stress-induced cell apoptosis in human myeloid HL-60 cells through regulation of calcium release and caspase-3 and -9 activities. J Membr Biol. 2009;232(1-3):15–23. doi: 10.1007/s00232-009-9212-2. [DOI] [PubMed] [Google Scholar]
- van de Wetering CI, Coleman MC, Spitz DR, Smith BJ, Knudson CM. Manganese superoxide dismutase gene dosage affects chromosomal instability and tumor onset in a mouse model of T cell lymphoma. Free Radic Biol Med. 2008;44(8):1677–1686. doi: 10.1016/j.freeradbiomed.2008.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Yi J. Cancer cell killing via ROS: to increase or decrease, that is the question. Cancer Biol Ther. 2008;7(12):1875–1884. doi: 10.4161/cbt.7.12.7067. [DOI] [PubMed] [Google Scholar]
- Wang X, McCullough KD, Franke TF, Holbrook NJ. Epidermal growth factor receptor-dependent Akt activation by oxidative stress enhances cell survival. J Biol Chem. 2000;275(19):14624–14631. doi: 10.1074/jbc.275.19.14624. [DOI] [PubMed] [Google Scholar]
- Wang X, Zhou YX, Qiao W, Tominaga Y, Ouchi M, Ouchi T, Deng CX. Overexpression of aurora kinase A in mouse mammary epithelium induces genetic instability preceding mammary tumor formation. Oncogene. 2006;25(54):7148–7158. doi: 10.1038/sj.onc.1209707. [DOI] [PubMed] [Google Scholar]
- Xie Y, Xu K, Dai B, Guo Z, Jiang T, Chen H, Qiu Y. The 44 kDa Pim-1 kinase directly interacts with tyrosine kinase Etk/BMX and protects human prostate cancer cells from apoptosis induced by chemotherapeutic drugs. Oncogene. 2006;25(1):70–78. doi: 10.1038/sj.onc.1209058. [DOI] [PubMed] [Google Scholar]
- Yang L, Dan HC, Sun M, Liu Q, Sun XM, Feldman RI, Hamilton AD, Polokoff M, Nicosia SV, Herlyn M, Sebti SM, Cheng JQ. Akt/protein kinase B signaling inhibitor-2, a selective small molecule inhibitor of Akt signaling with antitumor activity in cancer cells overexpressing Akt. Cancer Res. 2004;64(13):4394–4399. doi: 10.1158/0008-5472.CAN-04-0343. [DOI] [PubMed] [Google Scholar]
- Yi J, Yang J, He R, Gao F, Sang H, Tang X, Ye RD. Emodin enhances arsenic trioxide-induced apoptosis via generation of reactive oxygen species and inhibition of survival signaling. Cancer Res. 2004;64(1):108–116. doi: 10.1158/0008-5472.can-2820-2. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Leonard SS, Huang C, Vallyathan V, Castranova V, Shi X. Role of reactive oxygen species and MAPKs in vanadate-induced G(2)/M phase arrest. Free Radic Biol Med. 2003;34(10):1333–1342. doi: 10.1016/s0891-5849(03)00145-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.