Abstract
In patients with prostate cancer, a positive surgical margin is associated with an increased risk of cancer recurrence and poorer outcome, yet, margin status cannot be determined during the surgery. An in vivo optical imaging probe that could identify the tumor margins, during surgery, could result in improved outcomes. The design of such a probe focuses on a highly specific targeting moiety and a near infrared (NIR) fluorophore that is activated only when bound to the tumor. In this study, we successfully synthesized an activatable monoclonal antibody-fluorophore conjugate consisting of a humanized anti-prostate specific membrane antigen (PSMA) antibody (J591) linked to an indocyanine green ICG-derivative. Prior to binding to PSMA and cellular internalization, the conjugate yielded little light, however after binding an 18-fold activation was observed permitting the specific detection of PSMA+ tumors up to 10 days after injection of a low dose (0.25 mg/kg) of the reagent. This agent demonstrates promise as a method to image the extent of prostate cancer in vivo and could assist with real time resection of extracapsular extension of tumor.
Keywords: prostate cancer, activatable fluorescence imaging, prostate specific membrane antigen, humanized monoclonal antibody, optical surgery
INTRODUCTION
Prostate cancer is the 2nd leading cause of cancer mortality in men. Surgical resection is associated with a high cure rate in men with low risk tumors, however, the risk of recurrent disease rises with the Gleason score and clinical stage of the tumor. Nevertheless, especially in patients with locally invasive prostate cancer, long term cancer control can be achieved if adequate margins are obtained at surgery. Unfortunately, despite improving clinical imaging modalities 1-3 , the process of achieving negative margins is retrospective and is determined post hoc by histopathology. A method to aid the surgeon in obtaining negative margins during the procedure would be welcomed and could be anticipated to result in better patient outcomes. In order to achieve this goal, a highly specific optical imaging probe must be developed that fulfills at least two criteria. The agent must be highly targeted to prostate cancer with few false positives and the fluorescent probe to which the targeting ligand is attached, must be activatable only at sites of tumor in order to achieve adequate target to background ratios (TBR) to display the tumor in real time to the surgeon. Additionally, the fluorescent probe should emit light in the near infrared to permit deeper tissue penetration than is possible with visible light.
Prostate specific membrane antigen (PSMA) is a type 2 integral membrane glycoprotein, also known as glutamate carboxypeptidase II (GCPII) 4 and folate hydrolase 1, found initially in prostate cancer cell lines (LNCaP) 5 and a few other tissues including kidney, intestine and central nervous system 6 . It is considered the most well-established cell membrane marker of prostate cancer and therefore a plausible target for an optical probe. PSMA is expressed on tumor cells as a noncovalent homodimer 7 . PSMA has been used in imaging as a single photon emission computed tomography (SPECT) scan in which a PSMA antibody (Capromab Pentitide, Prostascint) is labeled with radioactive indium-111 8 . Experimentally, small PSMA receptor binding ligands have also been tested 9 for imaging prostate cancer. These methods have met with variable success, in part owing to the specificity of the capromab antibody used: it recognizes an intracellular epitope that is only accessible to circulating antibody when the cell membrane is disrupted. Furthermore, while SPECT scans are potentially useful in the pre-operative or post-operative setting, they are of little value for surgical guidance or on-site tissue diagnosis because they offer low spatial and temporal resolution. Optical probes, however, could provide higher resolution and near real time imaging without the risks to the patient and surgeon, associated with exposure to ionizing radiation.
Herein, we describe our initial experience with an optical antibody-flourophore conjugate consisting of an anti-PSMA humanized antibody (J591) coupled to an activatable near infrared (NIR) fluorophore, indocyanine green (ICG) 10 . We tested this agent in PSMA expressing and non-expressing prostate cancer xenografts in mice and compared it to a comparable conjugate, trastuzumab-ICG (Tra-ICG) which targets HER2 expressing tumors.
EXPERIMENTAL PROCEDURES
Reagents
Two humanized monoclonal antibodies were used in this study; J591, a humanized PSMA-specific monoclonal antibody, was developed at Weill Cornell Medical College by one of the authors (NHB), trastuzumab (Tra), a humanized anti-HER-2 antibody, was purchased from Genentech Inc. (South San Francisco, CA). For activatable labeling of antibodies, ICG-sulfo-OSu was purchased from Dojindo Molecular Technologies (Gaithersburg, MD), IR700 was purchased from LI-COR Biosciences (Lincoln, NE). All other chemicals used were of reagent grade.
Synthesis of fluorophore conjugated antibodies
Both humanized monoclonal antibodies; J591 and Tra (1 mg, 6.2 nmol) were incubated with ICG-sulfo-OSu (50 nmol, 1:8) or IR700 (31 nmol, 1:5) in 0.1M Na2HPO4 (pH 8.5) at room temperature for 30 min. Then, the mixture was purified with a Sephadex™ G50 column (PD-10; GE Healthcare, Piscataway, NJ). The elution fraction of 1.8 mL obtained from 2.6 ml to 4.4 mL of the elutant, was collected. The concentration of ICG or IR700 was measured by absorption with a UV-Vis system (8453 Value UV-Visible Value System; Agilent Technologies, Santa Clara, CA) to confirm the number of fluorophore molecules conjugated to each antibody molecule. The number of ICG per each antibody measured was ~1 for ICG at the quenching state or ~3 for IR700. As a quality control of the J591-ICG conjugate, we performed SDS-PAGE and a binding assay with modified Lindmo’s method 11 and Scatchard plot using 125I-labeled J591 and J591-ICG, and PC3-PSMA+ cells, as previously described 12 .
Validation of quenching ability IN VITRO
For validation of the quenching and dequenching ability of bound ICG, both J591-ICG and Tra-ICG conjugates were treated with 1% SDS to diminish hydrophobic k-k interactions and separate IgG chains. The change in fluorescence intensity for each conjugate was measured with an in vivo imaging system, (Pearl Imager,LI-COR Biosciences, Lincoln, NE) using 785 nm laser excitation and 820 nm emission.
Cell culture
A PSMA transfected PC3 cell line, PC3-PSMA+ (PC3pip) and a control blank-vector transfected PC3 cell line, PC3-PSMA- (PC3flu) 13 were used for PSMA targeting studies with activatable J591-ICG conjugates. Both cells were established at the Cleveland Clinic Foundation. For HER2 targeting studies by trastuzumab conjugates, the HER2 transfected NIH3T3 (3T3/HER2) cell line and a control Balb/3T3 cell line were used 14 .
All cell lines were grown in RPMI 1640 medium (Life Technologies, Gaithersburg, MD) containing 10% fetal bovine serum (FBS) (Life Technologies) and 1% Pen-Strep (Biofluids, Camarillo, CA). All cell cultures were maintained in 5% carbon dioxide (CO2), at 37.0°C in a humidified incubator.
Flow cytometry
Fluorescence signal from PC3 cells after incubation with ICG- or IR700-conjugated J591 agents was measured using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA) and CellQuest software (BD Biosciences). Briefly, for the binding experiment, 1 × 105 PC3-PSMA+ or PC3-PSMA- cells were incubated with J591-ICG or J591-IR700 at 37.0°C for 8 hours. To validate the specific binding of the antibody, excess J591 antibody (50 μg) was used to block 0.5 μg of J591-IR700 conjugates.
Fluorescence microscopy studies
PC3-PSMA+ cells (1 × 104) were plated on a covered glass-bottomed culture well and incubated for 16 h. Then, J591-ICG was added to the medium (5 μg /mL) and the cells were incubated for either 1 or 8 h. Cells were washed once with PBS, and fluorescence microscopy was performed using an Olympus BX61 microscope (Olympus America, Inc.) equipped with the following filters: excitation wavelength 672.5 to 747.5 nm, emission wavelength 765 to 855 nm. Transmitted light differential interference contrast (DIC) images were also acquired. As a control, the conjugates were also incubated with PC3-flu (PSMA negative) cells.
Animal tumor model
All procedures were carried out in compliance with the Guide for the Care and Use of Laboratory Animal Resources (1996), National Research Council, and approved by the local Animal Care and Use Committee. Seven- to 8-week old, athymic nu/nu mice were used for all experiments. Both receptor positive and negative tumor cell lines were implanted in the same mice. For PSMA targeting studies, 2 × 106 PC3-PSMA+ cells suspended in PBS were injected subcutaneously in the left dorsum of the mice, and the same number of PC3-PSMA- cells were injected subcutaneously into the right dorsum in each mouse.
For HER2 targeting studies, 1 × 106 3T3/HER2 cells suspended in PBS were injected subcutaneously in the left dorsum of the mice, 2 days prior to injecting with 1 ×106 Balb/3T3 cells in PBS into the right dorsum. The imaging experiments were performed at 5-7 days after cell injection, when tumor size is approximately 0.5 cm in diameter.
IN VIVO PSMA targeted imaging studies
J591-ICG was diluted in 200μL PBS at the dose of 50 μg, 25 μg, 12.5 μg, or 6.25 μg was then injected via the tail vein into mice bearing both PC3-PSMA+ and PC3-PSMA- tumors. All mice were anesthetized with 2% isoflurane throughout all procedures. Then, fluorescence images were obtained using the Pearl Imager (LI-COR Biosciences) at 800 nm at the following time points: 6 hours, 1, 2, 3, 4, 6 and 10 d after injection.
Regions of interest were placed on ICG spectrum images with reference to white light to measure fluorescence intensities of PC3-PSMA+ and PC3-PSMA- tumors and body (background) at every time point up to day 6.. The software, Pearl Cam Software (LI-COR Biosciences) was used for calculating ROI signal data of each point. After acquiring images at day 10, mice were sacrificed with carbon dioxide. Ex vivo images of resected tumors were obtained.
Comparison of images between PSMA+/− and HER2+/− tumors
To compare the signal obtained with J591-ICG from PC3-PSMA+ tumor with HER2 targeting images injected with Tra-ICG, both PSMA targeted and HER2 targeted images were serially acquired in side by side views 6 hrs, 1, 2, 3, and 4 days after injection of J591-ICG or Tra-ICG antibodies (12.5 μg in 200 μL PBS) into PC3-PSMA+/- or 3T3-HER2+/− tumor bearing mice, respectively. The fluorescence intensities of tumors and body (background) were processed with the same method as above.
RESULTS
Antibody-fluorophore conjugates show high quenching capacity
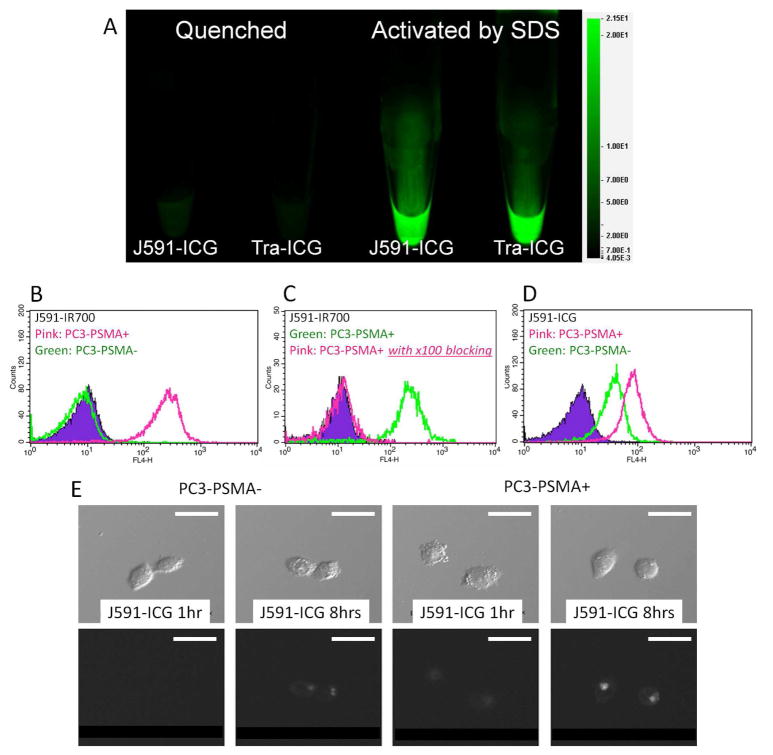
Fluorescence intensity was low for both J591-ICG and Tra-ICG in their native state. After exposure to 1% SDS, a 18-fold and 15-fold increase in fluorescence signals was detected for J591-ICG and Tra-ICG, respectively (Fig. 1A).
Figure 1.
A. Quenched (left) and chemically dequenched (right) J591-ICG and Tra-ICG. Dequenched conjugates show increased fluorescence signal shown in green. In vitro cell experiment with flow cytometry (B-D) and microscopy (E) showing specific binding of J591 to PSMA and activation of fluorescence signal after binding. Scale bar indicates 20 μm.
As defined by SDS-PAGE, 70% of ICG on the J591-ICG conjugate was covalently conjugated to the antibody. The specific immunoreactivity fraction of 125I-labeled J591-ICG (69%) was only 7% less than the 125I-labeled J591 without ICG (76%). The dissociation constant of 125I-labeled J591 or J591-ICG as measured by Scatchard plot analysis was 2.8 nM, similar to published values 15, 16 . In addition, the copy number of PSMA antigen on a single PC3-PSMA+ cell was calculated to be 2.0 million.
IN VITRO cell analysis
J591-IR700, a non-activatable control conjugate, demonstrated intense signal arising from PC3-PSMA+ cells and minimal signal from PC3-PSMA- cells (Fig. 1B). Blocking experiments using 100-fold levels of unlabeled J591 showed almost complete competitive blocking of J591-IR700 uptake indicating highly specific binding of J591-IR700 to PSMA (Fig. 1C). Flow cytometry analysis using J591-ICG showed strong fluorescence from PC3- PSMA+ cells and weak signal from PC3-PSMA- cells (Fig. 1D). Since this weak binding could not be blocked with a 100-fold excess of unlabeled J591 antibody, this signal represents low level, non-specific activation of quenched ICG, which was seen only in vitro. In vitro microscopy demonstrated results similar to flow cytometry experiments namely, specific uptake by PSMA expressing cells that could be blocked with unlabeled J591 (Fig. 1E).
IN VIVO imaging studies showed specific target visualization with J591-ICG even at low doses
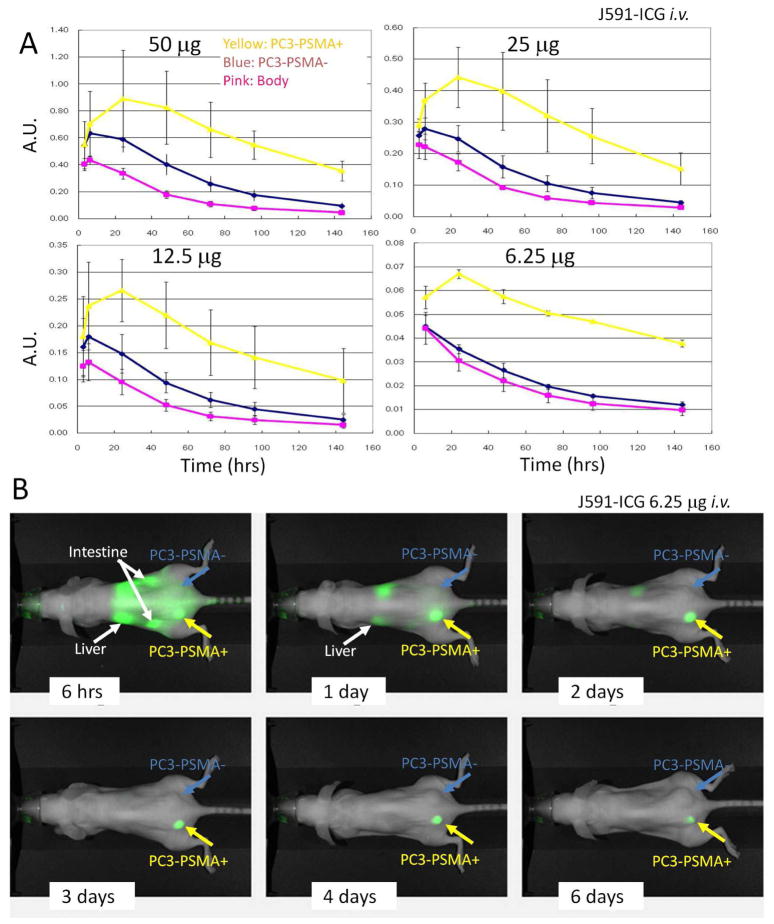
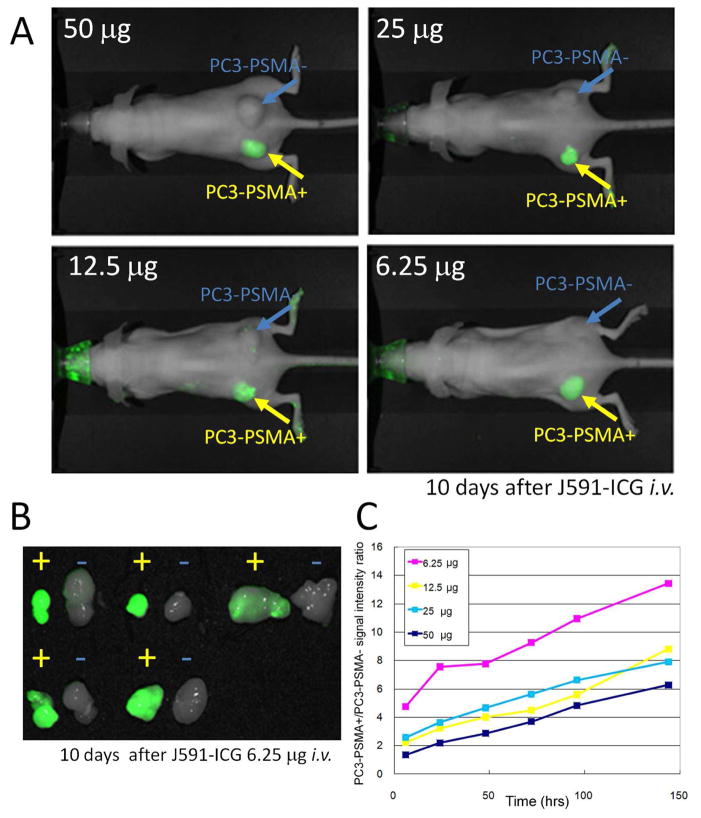
The time course of fluorescence intensity changes in PC3-PSMA+ and PC3-PSMA- tumors and of background signal after injection of various doses of J591-ICG are summarized in Fig. 2A. PC3-PSMA+ tumors yielded significantly higher fluorescent signal at all injected doses =1 day after injection. PC3-PSMA+ tumors were clearly depicted at all dose levels (50μg, 25μg, 12.5μg and 6.25μg) (Fig. 2B) up to 10 days after injection (Fig. 3A), although fluorescence signal from PC3-PSMA+ tumors increased with dose. All PC3-PSMA+ tumor specimen, which obtained 10 days after injection of 6.25μg J591-ICG, were clearly identified with ICG-specific fluorescence signal, although all PC3-PSMA- tumor specimen did not produce detectable signal (Fig. 3B). Signal intensity ratios between PC3-PSMA+ and PC3-PSMA- tumors increased with time reaching 14 at 96 hr after injection (Fig. 3C). Since fluorescence from PC3-PSMA- tumors was practically equivalent to the background, ICG signals from scattered PC3-PSMA-tumors were often measured as negative numbers and thus, were eliminated from the calculation. Therefore, the ratio of PC3-PSMA+ tumor-to- PC3-PSMA- tumor as shown in Fig. 3C is an underestimate.
Figure 2.
A. In vivo serial fluorescence signals of PC3-PSMA+ and PC3-PSMA- tumor as well as background in the body of mice injected with 50, 25, 12.5, and 6.25 μg of J591-ICG. B. In vivo serial fluorescence images of a PC3-PSMA+ and PC3-PSMA- tumor bearing mouse injected with 6.25 μg of J591-ICG. Only PC3-PSMA+ yields ICG fluorescence signal 3 days after injection or later.
Figure 3.
A. In vivo fluorescence images of a PC3-PSMA+ and PC3-PSMA- tumor bearing mice 10 days after injection of 50, 25, 12.5, or 6.25 μg of J591-ICG are shown. Although ICG signal from PC3-PSMA+ tumors is stronger in mice injected with a larger dose, all PC3-PSMA+ tumors show ICG fluorescence signal. B. Ex vivo fluorescence images of five PC3-PSMA+ and PC3-PSMA- tumors obtained 10 days after injection of 6.25 μg J591-ICG are shown. PC3-PSMA+ tumors clearly show specific fluorescence signal of ICG. C. PC3-PSMA+ tumor-to-PC3-PSMA- tumor signal intensity ratio is plotted. All doses show a similar trend of changes on PC3-PSMA+ tumor-to-PC3-PSMA- tumor signal intensity ratio. Scattered PC3-PSMA- tumors, which showed signal below background, are eliminated from the calculation.
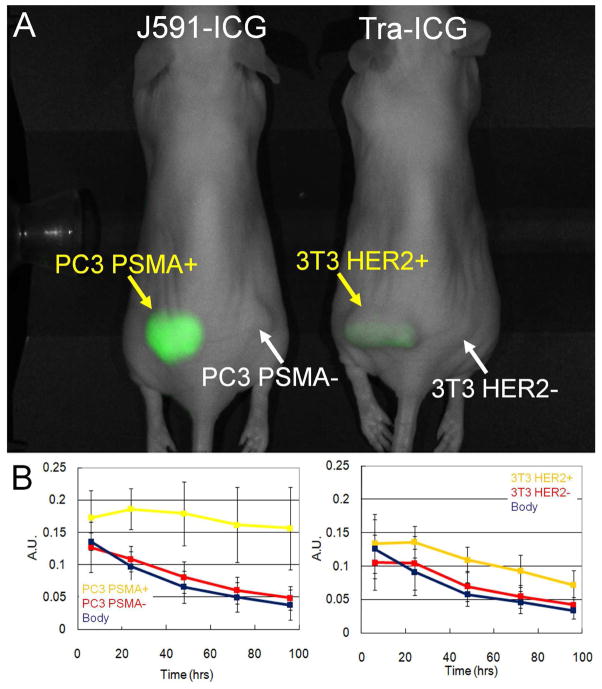
J591-ICG yielded brighter fluorescence signal from PC3-PSMA+ tumor than Tra-ICG yielded from 3T3/HER2+ tumor
At the same dose injection (12.5μg), the fluorescence signal arising from PSMA+ tumors after J591-ICG was higher than that from HER2+ tumors using the same dose of Tra-ICG (Fig. 4).
Figure 4.
Side-by-side comparison of images (A) and serial fluorescence intensity curves (B) for PC3-PSMA+ and PC3-PSMA- tumor bearing mouse injected with 12.5 μg of J591-ICG versus 3T3HER2+ and 3T3HER2- tumor bearing mouse injected with 12.5 μg of Tra-ICG. Images (A) are obtained 3 days after injection of antibodies. The anti-PSMA conjugate yields higher signal than the anti-HER2 conjugate suggesting better delivery and/or more efficient internalization.
DISCUSSION
This agent demonstrated several desirable features of a prostate cancer imaging optical probe. It appeared to be highly specific for PSMA expressing tumors, producing very bright targets within 24 hours of administration. It appeared to have a favorable efficiency in comparison to a similar HER2 targeted Tra-ICG compound 10 . Its ability to be activated only at the PSMA+ cells led to very high TBRs. This is related to a unique property of ICG, whereby it loses fluorescence when bound to the antibody but regains it, once it has been chemically separated from the antibody as occurs after internalization and degradation of the conjugate in the lysosome. One drawback of this agent is that it requires 2 days to be sufficiently taken up by the tumor, internalized and activated. However, once this has occurred the activation appears to last for at least 10 days. This suggests that there is a wide “window of opportunity” wherein the agent could be administered prior to a surgical procedure and still retain the desired benefits. Potential applications include detecting extracapsular extension, identifying lymph node metastasis, detecting remnant tumor after resection, and validating at the point of care that tissue obtained via optical biopsy or surgery contains cancer.
Optical imaging is one of the few types of imaging that can be “activated” 17, 18 . Ideally, in the unbound state a targeted compound would yield no signal but after specific binding would become fluorescent. In its injected state the probe is quenched, emitting no light. After binding and internalization the ICG is separated from the antibody, allowing it to dequench and leading to high TBR. In this study, quenched J591-ICG was efficiently activated in PC3-PSMA+ tumors. This phenomenon occurred at all doses of PSMA and suggests that the injected dose for clinical applications could be minimized to 0.25 mg/kg that is comparable to the antibody doses used in radioimmunoscintigraphy. The flow cytometry results suggest that although the numbers of PSMA and HER2 antigens on respective PC3-PSMA+ cells and 3T3Her2+ cells were similar at 2.0 and 2.2 million copies per cell, respectively, the anti-PSMA conjugate was considerably more effective as an imaging agent than the anti-HER2 conjugate. The difference in fluorescence signal could derive from differences in the delivery of the antibody and/or the internalization/activation ratio 17 . In any case, the low dose of antibody required for effectiveness is encouraging for clinical translation. .
Although safety was not specifically studied, there is reason for optimism regarding toxicity. Both major components of J591-ICG have been safely administered to patients. In the case of J591 this testing has been limited to several hundred patients in clinical trials at considerably higher doeses 19, 20 but in the case of ICG there is more than 50 years of clinical experience, primarily in ophthalmology 21 . Of course, formal safety studies will be required prior to approval for human use but the low dose needed for both components bodes well for a favorable toxicity profile.
In order to utilize a conjugate such as J591-ICG it will be necessary to have NIR cameras in the operating room 22 . Such cameras could be incorporated easily into existing endoscopic cameras providing both white light and NIR imaging during the procedure. Such endoscopic cameras have already been developed experimentally 23 .
In conclusion, we successfully synthesized an activatable anti-PSMA humanized monoclonal antibody, J591, conjugated with a linkable ICG derivative. The activatable antibody was able to depict PSMA+ tumors with high contrast compared to PSMA- tumors up to 10 days after injection of low dose of reagent (0.25mg/Kg). Activatable J591-ICG is a promising molecular imaging agent for detecting both primary and metastatic PSMA-positive prostate cancer based on the expression of PSMA and could be especially important in assisting surgeons performing biopsies and prostatectomies for cancer.
Acknowledgments
This research was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, Center for Cancer Research.
References
- 1.Hricak H, Choyke PL, Eberhardt SC, Leibel SA, Scardino PT. Imaging prostate cancer: a multidisciplinary perspective. Radiology. 2007;243:28–53. doi: 10.1148/radiol.2431030580. [DOI] [PubMed] [Google Scholar]
- 2.Mease RC. Radionuclide based imaging of prostate cancer. Curr Top Med Chem. 2010;10:1600–16. doi: 10.2174/156802610793176774. [DOI] [PubMed] [Google Scholar]
- 3.Turkbey B, Pinto PA, Choyke PL. Imaging techniques for prostate cancer: implications for focal therapy. Nat Rev Urol. 2009;6:191–203. doi: 10.1038/nrurol.2009.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.O'Keefe DS, Su SL, Bacich DJ, Horiguchi Y, Luo Y, Powell CT, Zandvliet D, Russell PJ, Molloy PL, Nowak NJ, Shows TB, Mullins C, Vonder Haar RA, Fair WR, Heston WD. Mapping, genomic organization and promoter analysis of the human prostate-specific membrane antigen gene. Biochim Biophys Acta. 1998;1443:113–27. doi: 10.1016/s0167-4781(98)00200-0. [DOI] [PubMed] [Google Scholar]
- 5.Israeli RS, Powell CT, Fair WR, Heston WD. Molecular cloning of a complementary DNA encoding a prostate-specific membrane antigen. Cancer Res. 1993;53:227–30. [PubMed] [Google Scholar]
- 6.Barinka C, Sacha P, Sklenar J, Man P, Bezouska K, Slusher BS, Konvalinka J. Identification of the N-glycosylation sites on glutamate carboxypeptidase II necessary for proteolytic activity. Protein Sci. 2004;13:1627–35. doi: 10.1110/ps.04622104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schulke N, Varlamova OA, Donovan GP, Ma D, Gardner JP, Morrissey DM, Arrigale RR, Zhan C, Chodera AJ, Surowitz KG, Maddon PJ, Heston WD, Olson WC. The homodimer of prostate-specific membrane antigen is a functional target for cancer therapy. Proc Natl Acad Sci U S A. 2003;100:12590–5. doi: 10.1073/pnas.1735443100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gregorakis AK, Holmes EH, Murphy GP. Prostate-specific membrane antigen: current and future utility. Semin Urol Oncol. 1998;16:2–12. [PubMed] [Google Scholar]
- 9.Foss CA, Mease RC, Fan H, Wang Y, Ravert HT, Dannals RF, Olszewski RT, Heston WD, Kozikowski AP, Pomper MG. Radiolabeled small-molecule ligands for prostate-specific membrane antigen: in vivo imaging in experimental models of prostate cancer. Clin Cancer Res. 2005;11:4022–8. doi: 10.1158/1078-0432.CCR-04-2690. [DOI] [PubMed] [Google Scholar]
- 10.Ogawa M, Kosaka N, Choyke PL, Kobayashi H. In vivo molecular imaging of cancer with a quenching near-infrared fluorescent probe using conjugates of monoclonal antibodies and indocyanine green. Cancer Res. 2009;69:1268–72. doi: 10.1158/0008-5472.CAN-08-3116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lindmo T, Boven E, Cuttitta F, Fedorko J, Bunn PA., Jr Determination of the immunoreactive fraction of radiolabeled monoclonal antibodies by linear extrapolation to binding at infinite antigen excess. J Immunol Methods. 1984;72:77–89. doi: 10.1016/0022-1759(84)90435-6. [DOI] [PubMed] [Google Scholar]
- 12.Barrett T, Koyama Y, Hama Y, Ravizzini G, Shin IS, Jang BS, Paik CH, Urano Y, Choyke PL, Kobayashi H. In vivo diagnosis of epidermal growth factor receptor expression using molecular imaging with a cocktail of optically labeled monoclonal antibodies. Clin Cancer Res. 2007;13:6639–48. doi: 10.1158/1078-0432.CCR-07-1119. [DOI] [PubMed] [Google Scholar]
- 13.Chang SS, Reuter VE, Heston WD, Bander NH, Grauer LS, Gaudin PB. Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res. 1999;59:3192–8. [PubMed] [Google Scholar]
- 14.Koyama Y, Hama Y, Urano Y, Nguyen DM, Choyke PL, Kobayashi H. Spectral fluorescence molecular imaging of lung metastases targeting HER2/neu. Clin Cancer Res. 2007;13:2936–45. doi: 10.1158/1078-0432.CCR-06-2240. [DOI] [PubMed] [Google Scholar]
- 15.McDevitt MR, Barendswaard E, Ma D, Lai L, Curcio MJ, Sgouros G, Ballangrud AM, Yang WH, Finn RD, Pellegrini V, Geerlings MW, Jr, Lee M, Brechbiel MW, Bander NH, Cordon-Cardo C, Scheinberg DA. An alpha-particle emitting antibody ([213Bi]J591) for radioimmunotherapy of prostate cancer. Cancer Res. 2000;60:6095–100. [PubMed] [Google Scholar]
- 16.Smith-Jones PM, Vallabahajosula S, Goldsmith SJ, Navarro V, Hunter CJ, Bastidas D, Bander NH. In vitro characterization of radiolabeled monoclonal antibodies specific for the extracellular domain of prostate-specific membrane antigen. Cancer Res. 2000;60:5237–43. [PubMed] [Google Scholar]
- 17.Kobayashi H, Choyke PL. Target-cancer-cell-specific activatable fluorescence imaging probes: rational design and in vivo applications. Acc Chem Res. 2011;44:83–90. doi: 10.1021/ar1000633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kobayashi H, Ogawa M, Alford R, Choyke PL, Urano Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem Rev. 2010;110:2620–40. doi: 10.1021/cr900263j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bander NH, Milowsky MI, Nanus DM, Kostakoglu L, Vallabhajosula S, Goldsmith SJ. Phase I trial of 177lutetium-labeled J591, a monoclonal antibody to prostate-specific membrane antigen, in patients with androgen-independent prostate cancer. J Clin Oncol. 2005;23:4591–601. doi: 10.1200/JCO.2005.05.160. [DOI] [PubMed] [Google Scholar]
- 20.Bander NH, Trabulsi EJ, Kostakoglu L, Yao D, Vallabhajosula S, Smith-Jones P, Joyce MA, Milowsky M, Nanus DM, Goldsmith SJ. Targeting metastatic prostate cancer with radiolabeled monoclonal antibody J591 to the extracellular domain of prostate specific membrane antigen. J Urol. 2003;170:1717–21. doi: 10.1097/01.ju.0000091655.77601.0c. [DOI] [PubMed] [Google Scholar]
- 21.Alford R, Simpson HM, Duberman J, Hill GC, Ogawa M, Regino C, Kobayashi H, Choyke PL. Toxicity of organic fluorophores used in molecular imaging: literature review. Mol Imaging. 2009;8:341–54. [PubMed] [Google Scholar]
- 22.Gioux S, Choi HS, Frangioni JV. Image-guided surgery using invisible near-infrared light: fundamentals of clinical translation. Mol Imaging. 2010;9:237–55. [PMC free article] [PubMed] [Google Scholar]
- 23.Mitsunaga M, Kosaka N, Kines RC, Roberts JN, Lowy DR, Schiller JT, Ishihara Y, Hasegawa A, Choyke PL, Kobayashi H. In vivo longitudinal imaging of experimental human papillomavirus infection in mice with a multicolor fluorescence mini-endoscopy system. Cancer Prev Res (Phila) 2011;4:767–73. doi: 10.1158/1940-6207.CAPR-10-0334. [DOI] [PMC free article] [PubMed] [Google Scholar]