Abstract
Segmented flow in microfluidic devices involves the use of droplets that are generated either on- or off-chip. When used with off-chip sampling methods, segmented flow has been shown to prevent analyte dispersion and improve temporal resolution by periodically surrounding an aqueous flow stream with an immiscible carrier phase as it is transferred to the microchip. To analyze the droplets by methods such as electrochemistry or electrophoresis, a method to “desegment” the flow into separate aqueous and immiscible carrier phase streams is needed. In this paper, a simple and straightforward approach for this desegmentation process was developed by first creating an air/water junction in natively hydrophobic and perpendicular PDMS channels. The air-filled channel was treated with a corona discharge electrode to create a hydrophilic/hydrophobic interface. When a segmented flow stream encounters this interface, only the aqueous sample phase enters the hydrophilic channel, where it can be subsequently analyzed by electrochemistry or microchip-based electrophoresis with electrochemical detection. It is shown that the desegmentation process does not significantly degrade the temporal resolution of the system, with rise times as low as 12 s reported after droplets are recombined into a continuous flow stream. This approach demonstrates significant advantages over previous studies in that the treatment process takes only a few minutes, fabrication is relatively simple, and reversible sealing of the microchip is possible. This work should enable future studies where off-chip processes such as microdialysis can be integrated with segmented flow and electrochemical-based detection.
Introduction
Segmented flow is a well-established method for automated high-throughput sample processing1 and has recently been implemented in microfluidic devices, which is often termed droplet-based microfluidics.2 Segmented flow involves the use of a carrier phase such as oil or air to divide an aqueous flow stream into discrete droplets. Droplets can be generated in a microchip device through perpendicular channels3–5 or off-chip with a micro-mixing tee.6 On-chip generation of segmented flow has been used to promote reagent mixing for applications such as nanoscale PCR,7 studying reaction kinetics,5 and trapping single cells.2,8 Recently, there has also been an interest in using segmented flow to help improve temporal resolution when samples from off-chip processes are transferred to the microchip for subsequent analysis.3 One of the major applications of this approach is coupling microdialysis (MD) sampling to microfluidic devices.3,9 MD is a powerful in vivo sampling technique that has been used to continuously sample dynamic systems such as neurotransmitter release.10 In such complex systems, a high temporal resolution is desirable; however, it is often limited by low flow rates (<1 μL/min), long transfer tubing needed for in vivo studies, and tubing interconnects.
Because of the low flow rates used in off-chip sampling methods when maximal recovery is desired,11 low volume separation and detection techniques are preferred in order to decrease analysis time and minimize the amount of sample that must be collected. Microchip electrophoresis is an ideal separation technique for this application since on-line monitoring of a continuously pumping flow stream is possible,12 picoliter injection volumes are typical,13 and separations can be performed in seconds.14 Detection methods commonly used with microchip electrophoresis include fluorescence detection, electrochemical detection, and mass spectrometry, all of which are highly sensitive.15 Electrochemical detection is particularly advantageous for monitoring neurotransmitters, as no derivatization is required. Electrodes can be incorporated directly into the microchannel so that detection occurs immediately following the separation with minimal band broadening.16–17 Our group was the first to report a microchip device to integrate MD sampling with microchip electrophoresis and electrochemical detection.18 The valving-based microchip was used to monitor catecholamine release from stimulated PC 12 cells; however, the rise time of the device was 150 s. While this rise time is similar to values obtained by other groups using microchip-based electrophoresis with fluorescence detection to analyze MD samples,19–20 substantial improvements are needed to enable near-real time analysis of dynamic systems.
It has been shown by Kennedy’s group that the use of segmented flow helps to improve temporal resolution with MD sampling by minimizing the effect of dead volume and Taylor dispersion.3,6,9 The latter can cause the concentration distribution of the analytes to spread out as the sample is transported from the probe to the microchip, resulting in band broadening and degrading the temporal resolution of the entire system. When segmented flow is used, the immiscible carrier phase serves to prevent Taylor dispersion by confining the analyte stream into discrete droplets as soon as sampling occurs. The group reported rise times of 15 s if direct fluorescence detection is used for on-line analysis and as low as 2 s if the microdialysate sample is collected off-line in tubing and subsequently analyzed on-chip.3,21 The difference in rise time for a segmented versus non-segmented flow stream can be as dramatic as ~140 s when the detection point is located relatively far downstream from the sampling probe.3
The goal of this work is to couple segmented flow with microchip electrophoresis and electrochemical detection to achieve high temporal resolution using tubing and flow rates realistic for off-chip sampling methods. Unfortunately, using segmented flow with analysis techniques such as electrophoresis and electrochemical detection requires the removal of the non-conductive carrier phase (usually oil or a fluorinated organic) prior to the measurement. In this area, research has been focused on developing methods to recombine droplets into a continuous aqueous flow stream (“desegmentation”) before their contents are analyzed.9,22–24 Many of these techniques utilize all glass devices with selective chemical derivatization of individual channels to create hydrophobic/hydrophilic interfaces.9 Other desegmentation methods that have been described make use of pillar,23 comb,22 or membrane24 structures within the microfluidic network to act as filters to remove the oil or aqueous phase. However, many of these techniques involve complicated fabrication steps and in some cases minor fluctuations in pressure can cause solution to cross the interface between hydrophilic and hydrophobic regions.
Clearly, a need exists for a simple channel modification procedure that can result in reproducible desegmentation. In addition, it would be desirable that this approach be applicable to integration of microelectrodes for electrochemical detection. Here, we show that PDMS-based perpendicular microchannels can be selectively treated with a corona discharge electrode to become hydrophilic after an air/water boundary is created at the channel intersection. This process generates a hydrophobic/hydrophilic interface in just seconds and the microchip can be reversibly sealed over microelectrodes and reused. When a segmented flow stream encounters the hydrophobic/hydrophilic interface, only aqueous sample enters the hydrophilic channel where it can be quickly analyzed by electrochemistry or microchip-based electrophoresis with electrochemical detection. To our knowledge, this is the first published report of using a corona discharge electrode to create a hydrophilic/hydrophobic interface that can be used to desegment droplets in microfluidic devices. In addition, this is the first report of using microchip electrophoresis with electrochemical detection to analyze the contents of desegmented droplets.
Experimental
Microchip Fabrication
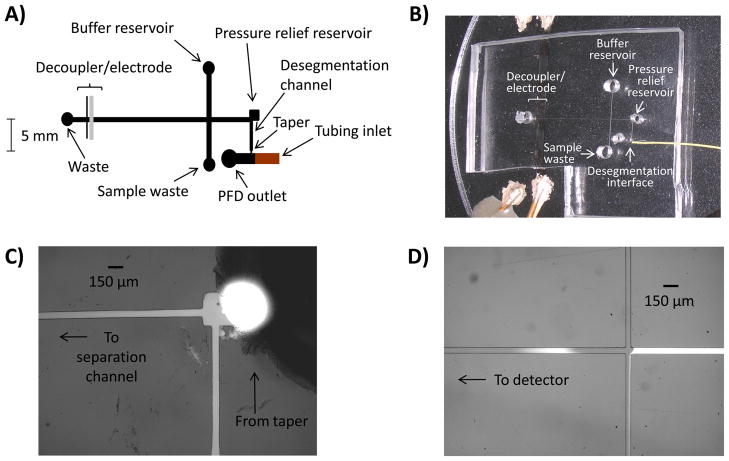
Information about the microchip fabrication can be found in the Supporting Information. Unless otherwise specified, microchannels used with both on- and off-chip droplets were 55 μm tall and segmentation and desegmentation channels were 150 μm and 80 μm wide, respectively. The desegmentation channel included a 20 μm wide taper designed to create flow resistance for immiscible phase to enter the channel (Figures 1 and 2). To facilitate transfer from off-chip tubing to the PDMS microchip, a short piece of 360 μm o.d. capillary was aligned over the raised master structure prior to pouring the PDMS. This created an impression in the PDMS into which the segmented flow tubing could be inserted (see segmented flow section).
Figure 1.
A) Microchip diagram for use with off-chip droplet generation. Droplets are formed in an off-chip tee and are transferred on-chip via the tubing inlet. Lengths are to scale. B) Micrograph displaying the air/water interface in a microchip for use with off-chip droplets. C) Photograph of corona treatment. D) Micrograph of desegmentation process. Droplets enter the chip through HPFA+ tubing and are immediately desegmented from the immiscible phase into the tapered desegmentation channel.
Figure 2.
A) Diagram of a microchip with channels for on-chip droplet generation and desegmentation. Lengths are to scale. B) Micrograph of the segmentation and desegmentation channels without corona treatment. The desegmentation channel contains both PFD (transparent) and aqueous sample (fluorescein). C) Micrograph showing the protection of an on-chip droplet segmentation channel with water. The desegmentation channel contains air which is ionized during corona treatment. D) Photograph of the on-chip corona treatment process. E) Micrograph showing the droplet desegmentation process after corona treatment. Only aqueous sample enters the desegmentation channel.
Corona Treatment
A corona discharge unit fitted with a fine tip electrode (Model ETP BD-20, Electro-technic Products, Inc., Chicago) was used to oxidize air-filled PDMS channels. For both off- and on-chip droplet studies, water was used to protect one channel from corona treatment so that the channel remained hydrophobic (Figures 1 and 2). For off-chip droplet experiments (Figure 1), the PDMS chip was reversibly sealed to a glass layer. The hydrophilic/hydrophobic interface was made by first pipetting water into the perfluorodecalin outlet reservoir and placing an aspirator at the capillary inlet, pulling the water past the taper. Since the aqueous outlet remained open to air, an air “pocket” was generated at the end of the tapered desegmentation channel (Figure 1B). The corona discharge electrode was placed in the aqueous reservoir (or pressure relief reservoir for electrophoresis experiments) at a ~70° angle from the microchip and actuated for ~1 s. Water in the segmentation channel was then seen to enter the desegmentation channel following treatment even while the aspirator was still on, indicating that a hydrophilic surface was successfully generated. As seen in Figure 1, the hydrophobic/hydrophilic interface created during corona treatment matched the air/water interface. Fluorescence detection could be done directly in the corona treated microchannel (Figure 3) or the chip could be removed from the glass layer and re-sealed over an electrode for direct electrochemical detection (Figure 4).
Figure 3.
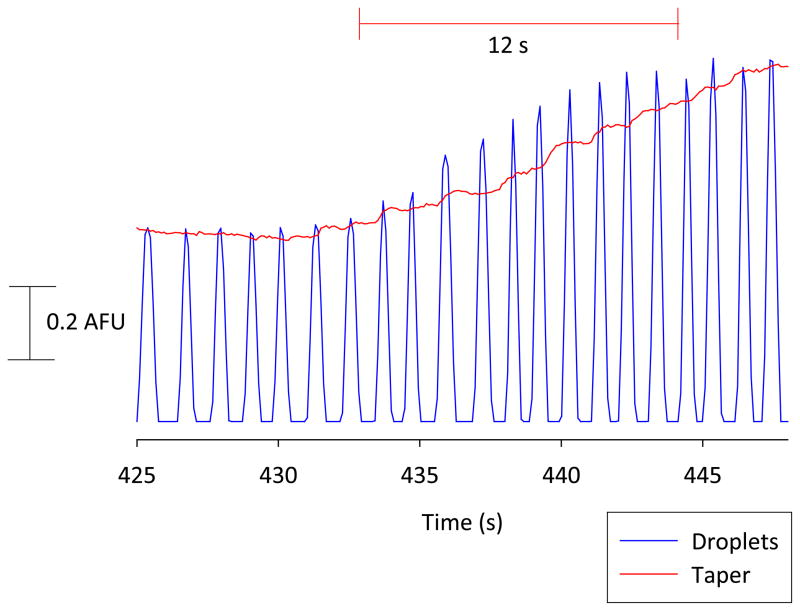
Off-chip concentration change experiment. Response obtained when fluorescein was changed from 100 μM to 200 μM via a 4-port injector located immediately before the segmentation tee (100 μm i.d. tee). Injection was performed at time = 0. Detection took place in the droplets at the end of the HPFA+ tubing and on-chip immediately following the taper after desegmentation. The plot shows rise times obtained in the droplets (9 s) and after desegmentation (12 s).
Figure 4.
A) Micrograph of desegmentation with HPFA+ tubing inserted into a microchip reversibly sealed over a thin layer platinum electrode. Corona treatment occurred before reversibly sealing over the electrode. B) Calibration curve showing the results from 3 separate injections of differing catechol concentrations using an off-chip 4-port injector and a 100 μm i.d. segmentation tee. Detection at the Pt electrode = +700 mV (vs. a platinum wire pseudo-reference).
The treatment process for on-chip droplets (described in Figure 2) was similar with the exception of the air/water interface. In this case, water was placed in the perfluorodecalin outlet reservoir and vacuum was applied to one of the inlet holes. When the vacuum was removed, the taper provided enough flow resistance to keep water out of the desegmentation channel (Figure 2C). As with the off-chip studies, after a 1 s corona treatment water immediately filled the desegmentation channel.
Segmented Flow
All chemicals were obtained from Sigma Aldrich (St. Louis, MO) unless otherwise specified. Perfluorodecalin (PFD) was used as the carrier (immiscible) phase in all experiments. Unless otherwise specified, all samples were prepared in 25 mM boric buffer, pH 9.2, with fluorescein added for droplet visualization. Solutions were pumped using either 11 Plus or PHD 2000 syringe pumps (Harvard Apparatus, Holliston, MA). To generate off-chip droplets, PFD was pumped through either high purity perfluoroalkoxy plus (HPFA+) or polyether ether ketone (PEEK) tubing (both 150 μm i.d., Idex, Oak Harbor, OR) that was connected to one arm of either a 100 or 150 μm i.d. T-junction (Valco, Houston, TX). Aqueous sample was pumped through 150 μm i.d. fused silica capillary (Polymicro Technologies, Phoenix, AZ) attached to the arm of the tee perpendicular to the HPFA+ or PEEK tubing. Unless otherwise specified, flow rates were 600 nL/min and 300 nL/min for carrier and aqueous phases, respectively. The tubing connecting the tee to the microchip was either PEEK or HPFA+ (both 150 μm i.d. and 28 cm in length, unless otherwise noted). In concentration change experiments, a 4-port injection valve (200 nL internal rotor, Valco, Houston, TX) was used to inject a different sample concentration into the droplet flow stream so that the rise time of the system could be determined. As in previous studies,3,18 the rise time is defined here as the time during which signals increase from 10% to 90% of the maximum intensity. In these experiments, the capillary from the aqueous syringe was first directed through the 4-port valve and a 3 cm long, 75 μm i.d. capillary was used to connect the 4-port to the mixing tee.
To generate droplets on-chip, gas-tight syringes connected to fluorinated ethylene propylene tubing (FEP, 76.2 μm i.d., Idex, Oak Harbor, OR) were used to pump aqueous fluorescein sample and PFD through perpendicular flow channels. Inlet holes were made with a circular razor (Technical Innovations, Angleton, TX). With the on-chip design, it was necessary to irreversibly seal the PDMS device to a glass layer with an oxygen plasma cleaner (PDC-32G, Harrick Plasma, Ithaca, NY).25
Electrophoresis
Desegmentation was coupled to a gated injection scheme with the chip design shown in Figure 5. The channel height in these studies was either 22 μm (for studies with fluorescence detection) or 18 μm (for studies with electrochemical detection), with the electrophoresis and desegmentation channels being 40 μm wide. The electrophoresis potentials were applied with a high-voltage power supply (Labsmith, Livermore, CA) via platinum wires (Alfa Aesar, Ward Hill, MA) placed in the reservoirs on the microchip. The electrophoresis buffer for all studies was 25 mM boric acid pH 9.2.
Figure 5.
A) Diagram of an electrophoresis microchip for use with off-chip droplet generation. The design includes a pressure relief reservoir to minimize pulsed flow from droplets entering the electrophoresis portion of the device. Lengths are to scale. B) Picture of a desegmentation/electrophoresis microchip reversibly sealed over the decoupler and detection electrode. C) Micrograph of a plug of fluorescein entering the region around the pressure relief reservoir. D) Micrograph of an injection performed in the gated portion of the microchip.
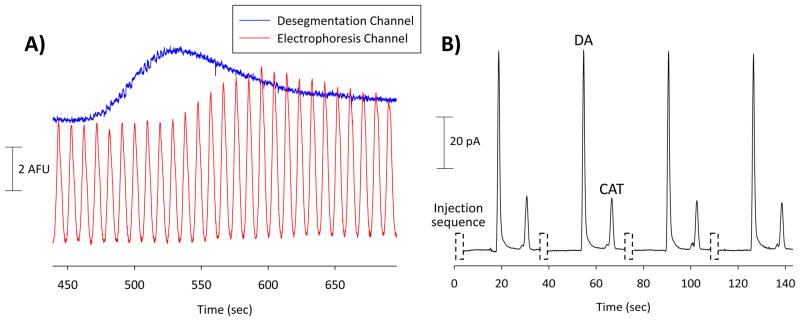
For electrophoresis with fluorescence detection (Figures 5D and 6A), +950 V was applied to the buffer reservoir and +550 V was applied to the pressure relief reservoir, with the waste reservoirs being held at ground (field strength = 241 V/cm). Injections were made by floating the voltage applied to the buffer reservoir for 1 s; injections were repeated every 5 s for a duration of 700 s. Typical injection volumes were 870 pL. For studies involving microchip electrophoresis with electrochemical detection (Figure 6B), +1350 V and +960 V were applied to the buffer and pressure relief reservoirs, respectively, and the sample waste reservoir was held at ground (field strength = 300 V/cm). A palladium decoupler grounded the separation, ensuring that the carbon ink microelectrode was in a field-free region of the microchannel.18,26 During injection, the buffer potential was floated for 0.8 s, with a typical injection volume being 540 pL. The separation channel was 2.4 cm from injection point to decoupler.
Figure 6.
A) Response obtained when the fluorescein concentration in off-chip droplets (100 μm i.d. segmentation tee) was changed from 100 μM to 400 μM. Detection took place in the desegmentation channel and at the end of the electrophoresis channel. B) Electropherogram demonstrating 4 different injection, separation, and detection events. The off-chip generated droplets (via a 150 μm i.d. segmentation tee) contained dopamine (DA) and catechol (CAT) (100 μM each) and the buffer was 25 mM boric acid (pH 9.2).
Detection
An 812 B potentiostat (CH Instruments, Austin, TX) was used for amperometric detection in all electrochemistry experiments. All potentials were versus a platinum wire pseudo-reference. For studies involving direct electrochemical detection (Figure 4), a thin-layer, 110 μm long platinum electrode (operated at +700 mV) was reversibly sealed in the desegmentation channel (with the electrode width being defined by this channel); the reference electrode was placed in the aqueous reservoir. For the electrophoresis studies (Figure 6B), a single carbon ink microelectrode was used for detection, with the electrode being patterned on a palladium connector such that the leading edge of the working electrode was 250 μm downstream from the palladium decoupler.17,26 The carbon ink electrode was operated at +900 mV. See the Supporting Information for information about the electrode fabrication. Fluorescence detection was performed on an inverted fluorescence microscope (IX71, Olympus America). More details on the fluorescence detection can be found in Supporting Information.
Results and Discussion
Desegmentation
The integration of segmented flow into microfluidic devices is now fairly standard, with fluorescence being widely utilized to analyze the droplets. The ability to use other detection techniques such as electrochemistry is desirable to increase the applicability of droplet-based microfluidics. Microchip electrophoresis and electrochemical detection are especially challenging to couple with segmented flow due to the need to remove the non-conductive immiscible fluid. Our group and others have shown that PDMS is a preferred material for integrating microelectrodes into fluidic channels, as it can be reversibly or irreversibly sealed over the electrodes without the need for high temperature bonding steps.17,27–32 The goal of this work was to develop a new approach of desegmenting perfluorodecalin droplets in PDMS devices in a manner where electrochemical detection (either direct or following an electrophoretic separation) is possible.
Corona discharge electrodes have been used to irreversibly seal PDMS devices33 as well as in closed microchannels to improve EOF.34 Both of these applications make use of the hydrophilic surface that is created by oxygen-rich silanol groups formed on the PDMS surface during corona treatment. The procedure that is presented herein uses a corona discharge electrode to selectively create a hydrophilic/hydrophobic interface in PDMS channels. As PDMS is natively hydrophobic, a method to prevent the corona from oxidizing the entire fluidic network was needed. As shown in Figure 1A, this was done by using a perpendicular channel design in which one reservoir was filled with water and the other left open to the atmosphere. By applying a vacuum to a common channel, an air/water interface was created at the channel intersection (Figure 1B). After a short 1 s corona treatment (Figure 1C), the air filled channel was oxidized and a small hydrophilic “pocket” was generated at the end of the taper (Figure 1D). When droplets encountered this region, the aqueous component fused with the desegmented flow stream while the carrier phase emptied into the waste outlet downstream from the desegmentation channel. It was found that this process was reproducible and could be done in all PDMS or hybrid PDMS/glass devices that have been either irreversibly or reversibly sealed. In the case of reversible sealing, the device could be disassembled after corona treatment and resealed onto a different substrate without affecting the desegmentation process shown in Figure 1D. As can be in Figure S-1 (see Supporting Information), it was also found that the desegmentation process works well for long-term experiments over the course of a day, with no difference in the hydrophilic/hydrophobic interface being seen 24 hrs after corona treatment. This is consistent with other studies that have shown that hydrophobic recovery of PDMS is greatly reduced when the PDMS is stored in water after plasma/corona treatment.35–37
While the primary purpose of this study was to use off-chip droplets to conserve concentration changes for off-chip sampling processes, a similar treatment procedure can be used for droplets created on-chip (Figure 2). This type of droplet-based microfluidics has been used for applications such as trapping single cells.2 The microchip design shown in Figure 2A was used for the on-chip droplet studies. As was the case with off-chip droplets, there was no desegmentation when the device was not treated with the corona discharge (Figure 2B). The approach described above for off-chip droplet generation utilized a continuous vacuum during the corona treatment. When a similar approach was used for the on-chip design in Figure 2A (by continuously applying vacuum at the inlet holes, placing water in the PFD outlet and leaving the aqueous outlet open to air), an air pocket (from air being pulled from the aqueous outlet) extended all the way down the segmentation channel. This resulted in a desegmentation channel that was ~ ½ hydrophilic and ~ ½ hydrophobic for a substantial distance. This caused the droplets to break apart far from the desegmentation channel intersection. As it has been shown that a hydrophobic environment is necessary to maintain droplet stability,3–4 a slightly different method to create the air/water interface was developed. This involved using vacuum to pull water from the outlet reservoir into the segmentation channel followed by removal of the vacuum. When vacuum was removed, the taper provided enough flow resistance to keep water out of the desegmentation channel (Figure 2C). The rest of the treatment process was the same as the off-chip desegmentation procedure, with the corona electrode being placed in the aqueous reservoir and actuated (Figure 2D). The corona treatment process was found to completely and reproducibly desegment the droplets, with the droplets remaining stable until the intersection was encountered (Figure 2E). Furthermore, it was found with both on- and off-chip treatment that if the microchip was allowed to recover its hydrophobicty with the channels being open to air (overnight at room temperature or in a 120 °C oven for 1 hour38), the same chip could be reused and retreated multiple times with the same desegmentation results.
To verify that the desegmentation procedure did not considerably degrade the temporal resolution that is achieved when segmented flow is coupled with off-chip sampling, rise times for an off-chip concentration change were determined before and after droplet desegmentation (using the device in Figure 1). For this experiment, droplets were created by pumping 100 μM aqueous fluorescein and PFD, both at 300 nL/min, through perpendicular arms of an off-chip micro-mixing tee. A 200 μM fluorescein sample was injected into the aqueous flow stream via a 4-port rotary injector and this concentration change was conserved within droplets as soon as the plug was transferred to the mixing tee. Fluorescence detection took place at the end of a 20 cm-long section of tubing before the droplets entered the microchip as well as immediately after the taper (following desegmentation). Figure 3 shows the results of this study. The injection was made at time = 0 s, giving a lag time for droplet transfer of approximately 430 s. The rise time was 9 s in the segmented flow stream (tubing) and 12 s after the droplets were transferred to the microchip and desegmented, with the latter measurement being made in the desegmentation channel after the taper (0.5 mm from the interface). These results are similar to rise times achieved by other groups3 using on-line fluorescence detection within a segmented flow stream (15 s) and confirm that both the droplet transfer process and our desegmentation procedure were highly efficient and had little effect on temporal resolution.
Integration of Electrochemical Detection
There have been few reports of using electrochemical detection to monitor the contents of droplets, as the immiscible phase is non-conductive and can foul the electrode surface. As described above, the corona treatment procedure developed here can be done in irreversibly or reversibly sealed devices. The ability to use reversibly sealed PDMS chips is particularly advantageous for electrochemical detection, as it was found that the high voltage output at the corona electrode can damage any detection electrode in its vicinity. In the electrochemistry experiment described here, the PDMS microchip was corona treated on a glass substrate followed by the microchip being removed and resealed over a glass plate containing a thin-layer platinum electrode (Figure 4A). Off-chip droplets and a concentration change experiment were performed using a 4-port injector, as described above. Injections of 10, 20, and 30 μM catechol were made to generate a calibration curve (Figure 4B). It is important to note that the signal oscillates due to the “pulsed” nature of the flow down the desegmentation channel; this is because the flow stream moves only when a new droplet enters the channel. A rise time of 14 s was obtained for the 30 μM peak, which is similar to the fluorescence results and are quite promising, especially considering the distance required for the analyte to reach the detection electrode (2 mm from the interface).
Electrophoresis Studies
In order to use microchip electrophoresis to analyze the contents of the desegmented flow stream, a method to discretely inject a small plug into an electrophoresis network was needed. This was addressed by using the chip design shown in Figure 5A. This design includes a pressure relief reservoir (Figures 5A and 5C) which served 3 main purposes: 1) to function as a corona treatment reservoir for the desegmentation procedure; 2) to vent the hydrodynamic pressure caused by droplets entering the desegmentation channel before the sample stream enters the electrophoresis network; and 3) to allow a potential to be applied to the sample flow stream so that a gated injection scheme can be used. The corona treatment procedure for this design was the same as that for off-chip droplets without electrophoresis, with the air/water interface being generated in the same manner as in Figure 1B. As shown in Figure 5A and B, electrophoresis voltages were applied to the buffer and pressure relief reservoirs with the sample waste reservoir and palladium decoupler being held at ground. The buffer potential was floated for a specified time to produce an injection (Figure 5D). To verify the ability of the pressure relief reservoir to minimize pulsed flow, injections (n=13) were performed while fluorescence was monitored in the separation channel, 0.9 mm from the injection tee (detection window = 13 × 80 μm). This injection sequence led to an average injection plug size of 870 pL, with the resulting average peak height being 67.05 ± 0.34 AFU (RSD = 0.51%, see Figure S-2). These results indicate that the pressure relief reservoir enables a consistent, non-pulsed flow to be maintained in the electrophoresis portion of the device, allowing reproducible injections into the separation channel.
One concern with using this design was the extra channel volume and its effect on the rise time of the system. Moreover, the electrophoresis buffer in the pressure-relief reservoir could possibly contribute to sample dilution, minimizing the gain in temporal resolution from the segmentation process. To study these issues, off-chip droplets were made with 25 mM boric buffer (pH 9.2) as the aqueous phase and a 4-port injector was used to inject 150 μM fluorescein into the segmented flow stream. The droplets were then transferred to a corona treated electrophoresis chip and fluorescence was monitored immediately before and after the pressure relief reservoir (approximately 0.4 mm on either side of the reservoir). It was found that the fluorescein plug moved through this distance in 2.7 s, with minimal loss in the fluorescence signal after the reservoir (Figure 5C). Thus, the pressure relief reservoir appears to be analogous to a flow splitter in that a good portion of the sample enters the pressure relief reservoir but there is minimal dilution of the fraction that enters the electrophoresis portion of the chip.
To demonstrate the performance of the device, a concentration change was generated in off-chip droplets. Initially the droplets contained 100 μM fluorescein and flow rates were 500 nL/min and 800 nL/min for the aqueous and carrier (PFD) phases, respectively. Fluorescence was monitored in the desegmentation channel (1 mm from the pressure relief reservoir) following injection of 400 μM fluorescein into the segmented flow stream (from the off-chip 4-port injector). The experiment was repeated with fluorescence detection at the end of the separation channel (1.8 cm from the injection tee), with gated injections being performed every 5 s. In both trials, the concentration change was made at time = 0 s. Figure 6A shows the superimposed data obtained in each trial. Rise times were 24 s when fluorescence was monitored in the desegmentation channel prior to the pressure relief reservoir and 36 s when fluorescence was monitored in the electrophoresis channel (following a gated injection). These times could be minimized by using shorter desegmentation and gated injection channels;39 however, the latter value is similar to that obtained in an all-glass device when using a different desegmentation procedure for integrating segmented flow with microchip electrophoresis for online analysis.9
The ultimate goal of this work was to integrate electrodes into the device to perform electrochemical detection following an electrophoretic separation. To our knowledge, there have been no reports of analyzing the contents of desegmented droplets with microchip electrophoresis and electrochemical detection. Our group has previously described the fabrication and use a palladium decoupler and carbon ink microelectrodes to integrate microchip electrophoresis with electrochemical detection.17,26 A similar integration was possible using the desegmentation/electrophoresis design, with the electrophoresis channel being reversibly sealed over a 2 mm palladium decoupler and the carbon ink electrode (Figure 5A). For this experiment, a sample containing 100 μM each dopamine and catechol was segmented and transferred onto the microchip. Figure 6B shows the electropherogram obtained following desegmentation, a discrete injection, and an electrophoretic separation. The number of theoretical plates for catechol was found to be 3,600 (150,000 plates/m), which is typical for PDMS devices.38,40 While this value is lower than all glass devices that have been used to analyze droplets with microchip electrophoresis,9,41 it should be noted with electrochemical detection an additional level of selectivity can be achieved by judicious control of the detection potential and/or working electrode material.42–43 As with the previously described electrochemistry experiment, the electrophoresis microchip could be corona treated on an all-glass substrate and then resealed over the decoupler and microelectrode.
Conclusions
In this paper, we have presented a simple and straightforward approach that uses a corona discharge to create a hydrophilic/hydrophobic interface in PDMS devices. This interface can be used to desegment droplets created off- or on-chip. An important feature of this approach is that it can be performed in reversibly sealed devices, which enables microelectrodes to be easily integrated into the fluidic network. An approach to couple the desegmented stream with microchip electrophoresis and electrochemical detection for droplet analysis was also presented. Rise times on the order of 12 s (for direct detection) and 36 s (when using microchip electrophoresis) were possible with these designs. This lays the foundation for future work which will focus on using this approach to analyze catecholamine release from both in vitro (PC 12 cells) and in vivo systems. These studies will require integration of MD sampling with segmented flow and microchip electrophoresis/electrochemical detection as well as improvements in the separation performance and limit of detection, with the latter issue possibly being addressed by the use of an electrode array or pillar electrodes.26,44
Supplementary Material
Acknowledgments
This project described was supported by Award Number 2R15GM084470-03 from the National Institute of General Medical Sciences as well as the President’s Research Fund at Saint Louis University.
Footnotes
Supporting Information. Additional information as noted in text. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Begg RD. Anal Chem. 1971;43:854–857. [Google Scholar]
- 2.Song H, Chen DL, Ismagilov RF. Angew Chem Int Ed. 2006;45:7336–7356. doi: 10.1002/anie.200601554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang M, Roman GT, Schultz K, Jennings C, Kennedy RT. Anal Chem. 2008;80:5607–5615. doi: 10.1021/ac800622s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Edgar JS, Pabbati CP, Lorenz RM, He M, Fiorini GS, Chiu DT. Anal Chem. 2006;78:6948–6954. doi: 10.1021/ac0613131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Song H, Ismagilov RF. J Am Chem Soc. 2003;125:14613–14619. doi: 10.1021/ja0354566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li Q, Pei J, Song P, Kennedy RT. Anal Chem. 2010;82:5260–5267. doi: 10.1021/ac100669z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Felbel J, Reichert A, Kielpinski M, Urban M, Häfner N, Dürst M, Köhler J, Weber J, Henkel T. Eng Life Sci. 2008;8:68–72. [Google Scholar]
- 8.He M, Edgar JS, Jeffries GDM, Lorenz RM, Shelby JP, Chiu DT. Anal Chem. 2005;77:1539–1544. doi: 10.1021/ac0480850. [DOI] [PubMed] [Google Scholar]
- 9.Wang M, Roman GT, Perry ML, Kennedy RT. Anal Chem. 2009;81:9072–9078. doi: 10.1021/ac901731v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ungerstedt U. Measurement of Neurotransmitter Release In Vivo. John Wiley & Sons Ltd; New York: 1984. [Google Scholar]
- 11.Watson CJ, Venton BJ, Kennedy RT. Anal Chem. 2006;78:1391–1399. doi: 10.1021/ac0693722. [DOI] [PubMed] [Google Scholar]
- 12.Li MW, Huynh BH, Hulvey MK, Lunte SM, Martin RS. Anal Chem. 2006;78:1042–1051. doi: 10.1021/ac051592c. [DOI] [PubMed] [Google Scholar]
- 13.Li MW, Bowen AL, Batz NG, Martin RS. In: Lab on a Chip Technology, Part II: Fluid Control and Manipulation. Herold KE, Rasooly A, editors. Caister Academic Press; Nofolk, UK: 2009. pp. 385–403. [Google Scholar]
- 14.Jacobson SC, Culbertson CT, Daler JE, Ramsey JM. Anal Chem. 1998;70:3476–3480. [Google Scholar]
- 15.Pasas SA, Fogarty BA, Huynh BH, Lacher NA, Carlson B, Martin RS, Vandeveer WR, IV, Lunte SM. In: Separation Methods in Microanalytical Systems. Kutter JP, Fintschenko Y, editors. CRC Press; Boca Raton: 2006. [Google Scholar]
- 16.Lacher NA, Lunte SM, Martin RS. Anal Chem. 2004;76:2482–2491. doi: 10.1021/ac030327t. [DOI] [PubMed] [Google Scholar]
- 17.Mecker LC, Martin RS. Electrophoresis. 2006;27:5032–5042. doi: 10.1002/elps.200600401. [DOI] [PubMed] [Google Scholar]
- 18.Mecker LC, Martin RS. Anal Chem. 2008;80:9257–9264. doi: 10.1021/ac801614r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huynh BH, Fogarty BA, Martin RS, Lunte SM. Anal Chem. 2004;76:6440–6447. doi: 10.1021/ac049365i. [DOI] [PubMed] [Google Scholar]
- 20.Sandlin ZD, Shou M, Shackman JG, Kennedy RT. Anal Chem. 2005;77:7702–7708. doi: 10.1021/ac051044z. [DOI] [PubMed] [Google Scholar]
- 21.Wang M, Slaney T, Mabrouk O, Kennedy RT. J Neurosci Methods. 2010;190:39–48. doi: 10.1016/j.jneumeth.2010.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Angelescu DE, Mercier B, Siess D, Schroeder R. Anal Chem. 2010;82:2412–2420. doi: 10.1021/ac902698m. [DOI] [PubMed] [Google Scholar]
- 23.Niu XZ, Zhang B, Marszalek RT, Ces O, Edel JB, Klug DR, deMello AJ. Chem Commun. 2009;41:6159–6161. doi: 10.1039/b918100h. [DOI] [PubMed] [Google Scholar]
- 24.Kralj JG, Sahoo HR, Jensen KF. Lab on a Chip. 2007;7:256–263. doi: 10.1039/b610888a. [DOI] [PubMed] [Google Scholar]
- 25.Duffy DC, McDonald JC, Schueller OJA, Whitesides GM. Anal Chem. 1998;70:4974–4984. doi: 10.1021/ac980656z. [DOI] [PubMed] [Google Scholar]
- 26.Mecker LC, Filla LA, Martin RS. Electroanalysis. 2010;22:2141–2146. doi: 10.1002/elan.201000118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vandaveer WR, Pasas-Farmer SA, Fischer DJ, Frankenfeld CN, Lunte SM. Electrophoresis. 2004;25:3528–3549. doi: 10.1002/elps.200406115. [DOI] [PubMed] [Google Scholar]
- 28.Hulvey MK, Frankenfeld CN, Lunte SM. Anal Chem. 2010;82:1608–1611. doi: 10.1021/ac902821v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Martin RS, Gawron AJ, Lunte SM, Henry CS. Anal Chem. 2000;72:3196–3202. doi: 10.1021/ac000160t. [DOI] [PubMed] [Google Scholar]
- 30.Bowen AL, Martin RS. Electrophoresis. 2009;30:3347–3354. doi: 10.1002/elps.200900234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Holcomb RE, Kraly JR, Henry CS. Analyst. 2009;134:486–492. doi: 10.1039/b816289a. [DOI] [PubMed] [Google Scholar]
- 32.Vickers JA, Henry CS. Electrophoresis. 2005;26:4641–4647. doi: 10.1002/elps.200500508. [DOI] [PubMed] [Google Scholar]
- 33.Eddings MA, Johnson MA, Gale BK. J Micromech Microeng. 2008;18:067001. [Google Scholar]
- 34.Thorslund S, Nikolajeff F. J Micromech Microeng. 2007;17:N16–N21. [Google Scholar]
- 35.Bausch GG, Stasser JL, Tonge JS, Owen MJ. Plasmas Polym. 1998;3:23–34. [Google Scholar]
- 36.Tan SH, Nguyen NT, Chua YC, Kang TG. Biomicrofluidics. 2010;4:032204. doi: 10.1063/1.3466882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Morraa M, Occhiello E, Marola R, Garbassi F, Humphrey P, Johnson D. J Colloid Interf Sci. 1990;137:11–24. [Google Scholar]
- 38.Vickers JA, Caulum MM, Henry CS. Anal Chem. 2006;78:7446–7452. doi: 10.1021/ac0609632. [DOI] [PubMed] [Google Scholar]
- 39.Rogers M, Leong C, Niu X, de Mello A, Parker KH, Boutelle MG. Phys Chem Chem Phys. 2011;13:5298–5303. doi: 10.1039/c0cp02810j. [DOI] [PubMed] [Google Scholar]
- 40.Lacher NA, de Rooij NF, Verpoorte E, Lunte SM. J Chromatogr A. 2003;1004:225–235. doi: 10.1016/s0021-9673(03)00722-2. [DOI] [PubMed] [Google Scholar]
- 41.Roman GT, Wang M, Shultz KN, Jennings C, Kennedy RT. Anal Chem. 2008;80:8231–8238. doi: 10.1021/ac801317t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lacher NA, Garrison KE, Martin RS, Lunte SM. Electrophoresis. 2001;22:2526–2536. doi: 10.1002/1522-2683(200107)22:12<2526::AID-ELPS2526>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 43.Lunte SM, Martin RS, Lunte CE. In: Electroanalytical Methods for Biological Materials. Brajter-Toth A, Chambers JQ, editors. Marcel Dekker; New York: 2002. pp. 461–490. [Google Scholar]
- 44.Selimovic A, Johnson AS, Kiss IZ, Martin RS. Electrophoresis. 2011;32:822–831. doi: 10.1002/elps.201000665. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.