Abstract
Hepatocellular carcinoma (HCC) is among the most common cancer diseases worldwide. Arterial hypervascularisation is an essential step for HCC tumorigenesis and can be targeted by transarterial chemoembolization (TACE). This interventional method is the standard treatment for patients with intermediate stage HCC, but is also applied as “bridging” therapy for patients awaiting liver transplantation in many centers worldwide. Usually the devascularization effect induced by TACE is transient, consequently resulting in repeated cycles of TACE every 4-8 wk. Despite documented survival benefits, TACE can also induce the up-regulation of proangiogenic and growth factors, which might contribute to accelerated progression in patients with incomplete response. In 2007, sorafenib, a multi-tyrosine kinase and angiogenesis inhibitor, was approved as the first systemic treatment for advanced stage HCC. Other active targeted compounds, either inhibitors of angiogenesis and/or growth factors, are currently being investigated in numerous clinical trials. To overcome revascularisation or tumor progression under TACE treatment it seems therefore attractive to combine TACE with systemic targeted agents, which might theoretically block the effects of proangiogenic and growth factors. Over the last 12 mo, several retrospective or prospective cohort studies combining TACE and sorafenib have been published. Nevertheless, robust results of the efficacy and tolerability of such combination strategies as proven by randomized, controlled trials are awaited in the next two years.
Keywords: Hepatocellular carcinoma, Sorafenib, Anti-angiogenesis, Transarterial chemoembolization
INTRODUCTION
The incidence of hepatocellular carcinoma (HCC) is rising with a world-wide annual incidence above 600 000[1]. Treatment of HCC is challenging because HCC mainly occurs within liver cirrhosis[1], and therapy options and prognosis are determined by tumor biology as well as impaired liver function. Several clinical staging systems have been proposed[2]. However, the most commonly used in Western countries is the Barcelona Clinic Liver Cancer (BCLC) system[3,4]. According to this algorithm, treatment is stratified according to tumor stage, liver function, and performance status. Intermediate stage HCC (BCLC stage B) without options for surgical treatment or ablation is treated by transarterial chemoembolization (TACE). TACE has been shown to expand median survival from 16 to 19-20 mo[5,6]. In patients with advanced (BCLC stage C) and especially end-stage HCC (BCLC stage D), survival depends not only on progression of tumor disease but depends incremental on accompanying liver dysfunction, also. Without intervention, survival of patients with advanced HCC rarely exceeds 6 mo, and median survival in patients with end-stage HCC (BCLC stage D, Okuda stage III, performance status 3-4) is commonly below 3-4 mo[4,7-9]. According to the modified BCLC system, the dual kinase inhibitor sorafenib is considered the standard of care for patients with advanced HCC[10]. However, the survival benefit is limited to approximately 3 mo, whereas disease stabilization can be achieved in 27%-78% as shown in prospective trials[11-14].
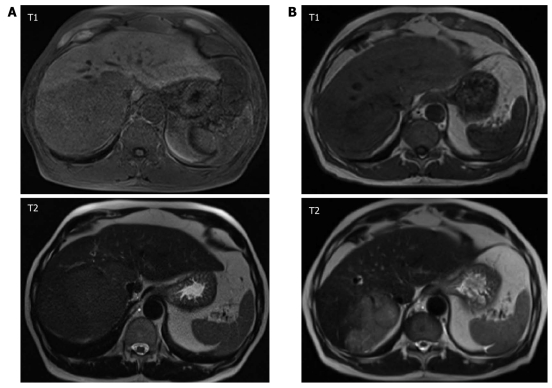
Typically, HCC is a hypervascularized tumor with characteristic early arterial enhancement during dynamic imaging, which is the rational for TACE. By TACE, however, mainly central vessels of a tumor nodule are occluded, while progression may occur via neovascularization in the tumor periphery. In theory, this might be prevented or at least attenuated by concomitant systemic treatment with anti-angiogenic agents (Figure 1).
Figure 1.
Dynamic gadolinium-enhanced magnetic resonance imaging (MRI; T1, T2 weighting), in a 67 year old patient with hepatocellular carcinoma evolved from liver cirrhosis due to hemochromatosis (A) before initiation of anti-angiogenic therapy and (B) after 70 d or three cycles of transarterial chemoembolization and continuous administration of sorafenib, respectively. Patient showed partial response according to RECIST criteria. Serum alfa-fetoprotein level decreased from 276 to 115 ng/mL.
ANGIOGENESIS IN PATHOGENESIS OF HEPATOCELLULAR CARCINOMA
Chronic hepatitis and hepatic fibrogenesis are closely connected to angiogenesis[15]. Different cytokines, growth factors, and metalloproteinases are involved in these processes. Vascular endothelial growth factor (VEGF) was shown to be crucially involved in angiogenesis as well as fibrogenesis[15]. Despite other factors, hepatic tissue hypoxia seems to be a relevant trigger for angiogenesis in necroinflammatory liver disease, especially by induction of VEGF, resulting in increasing arterial contribution to hepatic perfusion[16,17]. At this stage, the majority of neo-vessels originate from the portal vein, supporting short-circuits between the portal vein system and the hepatic veins[16,18]. Despite the predominant occurrence of HCC in liver cirrhosis rather than in non-cirrhotic liver disease[1], it is still unknown whether HCC arises from hepatic stem cells or from hepatocytes via malignant transformation. The latter concept is supported by the observation that development of HCC from dysplastic nodules has been described[19,20]. Arterial hypervascularization seems to be pathognomonic for established HCC, and HCC nodules larger than 2 cm regularly show arterial enhancement[21,22]. Therefore, neovascularization seems to be crucial for HCC tumorigenesis.
Consistently, increased expression of angiopoietin-1/-2 mRNA in tumor tissue was reported, suggesting a critical role of neo-vascularisation for HCC pathogenesis[23]. Moreover, augmented expression of VEGF was found in HCC, and higher serum VEGF levels were associated with poor prognosis of patients with HCC[24-29]. In contrast, a recent study showed that neither VEGF-A nor VEGFR were up-regulated in HCC tissue, and angiotensin-1/-2 expression were only modestly changed[30]. Of note, sinusoidal capillarization suggesting vascular remodeling was observed within the same study[30]. These inconsistent data further highlight that tumor angiogenesis is a complex process and most likely heterogeneous. The angiopoietin/VEGF system seems to play an important role in angiogenesis of HCC, but other, yet incompletely understood pathways may also be involved.
THERAPEUTIC INHIBITION OF ANGIOGENESIS IN HEPATOCELLULAR CARCINOMA
Inhibition of angiogenesis is an established and successful treatment strategy in a variety of malignant diseases. The liver is predominantly supplied by the portal venous system, whereas HCC nodules are characterized by typical arterial hypervascularization. This accounts for the rationale for use of hypervascularization as a diagnostic criterion as well as development of angiogenesis inhibition treatment strategies. In the absence of targeted agents, embolization of arterial tumor vessels was established in the 1980s. Currently, TACE is commonly used in patients with HCC BCLC stage 0/A as bridging therapy until liver transplantation and as non-curative therapy in patients with HCC BCLC stage B and C[6].
Indeed, TACE may lead to reduction of tumor vascularization and viable tumor volume[6]. Recently, this has also been confirmed for a modified TACE technique using doxorubicin eluting beads (DEB)[31]. Furthermore, VEGF levels as a surrogate marker for angiogenesis were shown to correlate with therapeutic outcome after TACE. Pretreatment VEGF levels were significantly higher in patients not responding to TACE compared to patients with disease stabilization. Moreover, pretreatment VEGF serum levels > 240 pg/mL were an independent prognostic factor for survival[32].
It has been suggested that tumor progression after TACE may be caused by activation of angiogenesis due to TACE-induced hypoxemia[33]. Plasma VEGF levels were shown to increase shortly after TACE, reaching a peak value one day after TACE[34-37]. Additionally, increase of plasma VEGF levels after TACE was correlated with the development of metastasis and a reduced progression free survival[35,37]. Unfortunately, reliable biomarkers predicting response to TACE are missing. Nevertheless, a median survival of 35 mo has been reported in patients with complete tumor response[38]. In this study low VEGF levels were associated with a longer survival, while higher VEGF levels were detectable in patients without tumor response. Of note, prior TACE was reported to induce angiogenesis in surgical specimens, whereas patients who underwent surgery without prior TACE had no induction of angiogenesis[39]. Whether the use of DEB-TACE, which can induce higher rates of tumor response, also leads to upregulation of proangiogenic factors is under debate[40,41].
Sorafenib, the first systemically agent approved for HCC, is a multikinase inhibitor with activity against VEGFR2, PDGFR, c-Kit receptors, b-RAF, and p38[42], signal transduction pathways which seem to be involved in pathogenesis of HCC[43]. However, there are limitations on the therapy with sorafenib, founded on restricted efficacy and potential side effects, mainly fatigue, diarrhea and hand-food syndrome. In comparison to TACE valid predictive biomarkers are missing, also[11].
STRATEGIES FOR COMBINATION OF TACE AND TARGETED AGENTS IN HCC
Combination of local and systemic inhibition of angiogenesis seems to be a consequential step to improve outcome in intermediate and advanced stage HCC[44]. Tolerability of combination therapy with sorafenib and conventional TACE as well as DEB-TACE was shown within different trials[45-49]. Currently, the combination of conventional TACE and sorafenib as well as combination of sorafenib and DEB-TACE (SPACE trial) is being evaluated in phase II and III trials[50]. Moreover, sorafenib was combined with selective internal radiation therapy within a multicenter phase II study showing good efficacy in patients with advanced HCC but without extra-hepatic metastasis[45]. So far, no increased toxicity has been reported. The combination of brivanib, a dual VEGFR and fibroblast growth factor inhibitor[51], and TACE is currently evaluated within the multicenter phase III BRISK TA Study.
Another interesting approach could be the inhibition of VEGF driven angiogenesis by targeting VEGF with siRNA as shown in a proof-of-concept study recently[52]. Furthermore, promising results were reported for other agents alone or in combination with TACE, e.g. tegafur/uracil, the multi-tyrosine kinase inhibitor TSU-68, sunitinib, erlotinib, and the VEGF antibody bevacizumab[53-57]. However, none of these agents is approved for HCC. Of these, bevacizumab is the currently most commonly clinical used VEGF inhibitor in a variety of malignant entities. However, despite encouraging results in earlier trials, even as single agent treatment, bleeding complications were reported in up to 11% of patients treated with bevacizumab[58]. For the combination of bevacizumab with TACE, severe bleeding and septic complications have been reported in 25% of patients, and the AVATACE-1 trial investigating TACE in combination with bevacizumab has been terminated due to safety concerns in the treatment arm, which does not justify a further clinical development of bevacizumab in this indication. This highlights that large phase III trials are required for new agents in HCC, which seems challenging given the increasing number of phase I and II studies addressing HCC in the last years (Tables 1, 2, 3).
Table 1.
Efficacy of systemic targeted monotherapy in hepatocellular carcinoma according to current phase I-III studies
| Author | Year | Phase | Investigational drug | n | RR | DS | PFS/TTP | PFS-6m | OS |
| O'Neil et al[59]1 | 2009 | II | AZD 6244 | 16 | 0 | 37.5 | NR | NR | NR |
| Malka et al[60] | 2007 | II | Bevacizumab | 30 | 12.5 | 54 | 3.5/NR | 17 | NR |
| Schwartz et al[61] | 2006 | II | Bevacizumab | 30 | 6.7 | 57 | NR/6.4 | NR | NR |
| Siegel et al[58] | 2008 | II | Bevacizumab | 46 | 13 | NR | 6.9/NR | NR | 12.4 |
| Raoul et al[62] | 2009 | II | Brivanib | 55 | 11 | 10 | NR/2.8 | NR | 10 |
| Gruenwald et al[63] | 2007 | II | Cetuximab | 27 | 0 | 44 | 2.0/1.9 | 22.2 | NR |
| Zhu et al[64] | 2007 | II | Cetuximab | 30 | 0 | 17 | 1.4/NR | NR | 9.6 |
| Philip et al[65] | 2005 | II | Erlotinib | 38 | 9 | 50 | 3.2/NR | 32 | 13 |
| Thomas et al[66] | 2007 | II | Erlotinib | 40 | 0 | 43 | 3.1/NR | NR | 6.25 (10.75)2 |
| Blaszkowsky et al[67] | 2010 | III | Everolimus | 25 | 4 | 44 | 3.8/3.9 | 8% | 8.4 |
| O’Dwyer et al[68] | 2006 | II | Gefitinib | 31 | 3 | 22.5 | 2.8/NR | NR | 6.5 |
| Lin et al[69] | 2008 | II | Imatinib | 15 | 0 | 13.3 | NR/NR | NR | NR |
| Ramanathan et al[70] | 2006 | II | Lapatinib | 37 | 5 | 35 | 2.3/NR | 2.3 | 6.2 |
| Rizell et al[71] | 2008 | II | Sirolimus | 21 | 4.8 | 23.8 | NR/NR | NR | 6.5 |
| Abou-Alfa et al[12] | 2006 | II | Sorafenib | 137 | 2.2 | 33.6 | NR/4.2 | NR | 9.2 |
| Cheng et al[72] | 2009 | III | Sorafenib | 226 (150 treated) | 3.3 | 54 | NR/2.8 | NR | 6.5 |
| Furuse et al[13] | 2008 | I | Sorafenib | 27 | 4 | 83 | NR/4.9 | 46.2 | 15.6 |
| Llovet et al[11] | 2008 | III | Sorafenib | 602 (299 treated) | 2 | 71 | NR/5.5 | NR | 10.7 |
| Yau et al[14] | 2009 | II | Sorafenib | 51 | 8 | 18 | 3.0/NR | NR | 5 |
| Zhu et al[73] | 2009 | II | Sunitinib | 34 | 2.9 | 47 | 3.9/4.1 | NR | 9.8 |
| Faivre et al[74] | 2009 | II | Sunitinib | 37 | 2.7 | 35 | 3.7/5.3 | NR | 8 |
| Hoda et al[75] | 2008 | II | Sunitinib | 23 | 6 | 35 | NR/NR | NR | NR |
| Koeberle et al[54] | 2010 | II | Sunitinib | 45 | 2 | 40 | 2.8/2.8 | NR | 9.3 |
| Kanai et al[57] | 2010 | I/II | TSU-68 | 35 | 8.6 | 42.8 | NR/2.1 | NR | 13.1 |
1Trial stopped;
Recorded from therapy start (recorded from diagnosis). DS: Disease stabilization (%); NR: Not reported; OS: Overall survival (mo); PFS/TTP: Progression free survival/time to progression (mo); PFS-6m: Progression free survival rate at 6 mo (%); RR: Response rate [complete + partial response (%)].
Table 2.
Efficacy of combination therapy with systemic acting agents and targeted therapy in hepatocellular carcinoma according to current phase I-II studies
| Author | Year | Phase | Investigational drug | n | RR | DS | PFS/TTP | PFS-6m (%) | OS |
| Sun et al[76] | 2007 | II | Bevacizumab/CapOx | 30 | 11 | 78 | 4.5/NR | 40 | NR |
| Thomas et al[55] | 2009 | II | Bevacizumab/erlotinib | 40 | 25 | 42.5 | 9.0/NR | NR | 15.7 |
| Hsu et al[77] | 2008 | II | Bevacizumab/capecitabine | 45 | 9 | 42 | 4.1/NR | NR | 10.7 |
| Zhu et al[78] | 2006 | II | Bevacizumab/GemOX | 33 | 20 | 27 | 5.3/NR | NR | 9.6 |
| Berlin et al[79] | 2008 | II | Bortezomib/doxorubicin | 39 | 2.3 | 25.6 | 2.4/NR | NR | 5.7 |
| Asnacios et al[80]1 | 2008 | II | Cetuximab/GemOx | 45 | 20 | 40 | 4.7/NR | NR | 9.5 |
| Louafi et al[81]1 | 2007 | II | Cetuximab/GemOx | 35 | 24 | 4.5 | NR/NR | 40 | 9.2 |
| Knox et al[82]2 | 2008 | II | G3139/doxorubicin | 17 | 0 | 35 | NR/1.8 | 17.2 | 5.4 |
| Abou-Alfa et al[83]3 | 2010 | II | Sorafenib/doxorubicin | 96 | 4 | 77 | 6.9/8.6 | 2.7 | 13.7 |
| Richly et al[84] | 2009 | I | Sorafenib/doxorubicin | 18 | 6.3 | 69 | 4.04/NR | NR | NR |
1Overlap of patient cohorts cannot be excluded from information provided in the abstracts; 2Trial stopped due to lack of efficacy; 3Trial stopped due to superiority of sorafenib;
Calculated from a median duration of disease control rate (combined endpoint for complete and partial response as well as stable disease) of 17.4 wk. CapOx: Capecitabine and oxaliplatin; DS: Disease stabilization (%); GemOx: Gemcitabine and oxaliplatin; NR: Not reported; OS: Overall survival (mo); PFS/TTP: Progression free survival/time to progression (mo); PFS-6m: Progression free survival rate at 6 mo (%); RR: Response rate [complete + partial response (%)].
Table 3.
Efficacy of sorafenib and transarterial chemoembolization in hepatocellular carcinoma (sequential therapy not included) according to current phase I-II studies
| Author | Year | Phase | Investigational drug | n | RR | DS | PFS/TTP | OS |
| Chow et al[45] | 2010 | II | Sorafenib + SIRT | 35 | 31.4 | 77.1 | NR/NR | 10.8 |
| Chung et al[47]1 | 2010 | II | Sorafenib + TACE | 50 | NR2 | 96 | NR/NR | NR |
| Dufour et al[48] | 2010 | I | Sorafenib + TACE | 14 | NR3 | NR3 | NR3 | NR3 |
| Erhardt et al[46]1 | 2009 | II | Sorafenib + TACE | 44 | NR4 | 63.6 | 8.0/16.1 | 11.7 |
| Reyes et al[49]1 | 2009 | II | Sorafenib + DEB-TACE | 50 | NR5 | NR | NR/NR | NR |
1Interim analysis;
20/50 patients received 2 cycles of transarterial chemoembolization (TACE), only, and 18 of these 20 patients achieved complete response compared to 2 patients with progressive disease. 30/50 patients received more than 2 cycles of TACE and achieved partial response or stable disease;
Primary objective of this prospective trial was evaluation of safety and tolerability of a continuous regimen of sorafenib combined with TACE;
According to 31 patients who received at least 1 cycle of TACE, 2/31 (6.5%) showed complete response, 15/31 (48.4%) showed partial response, and 11/31 (35.5%) showed stable disease. PFS, TTP, and OS are given for all 44 patients enrolled at time point of interim analysis;
Patients who completed DEB-TACE showed 100% objective tumor response and 100% partial response or stable disease according to EASL or RECIST criteria, respectively. DEB-TACE: (Drug eluting beads)-transarterial chemoembolization; NR: Not reported; DS: Disease stabilization (%); OS: Overall survival (mo); PFS/TTP: Progression free survival/time to progression (mo); PFS-6m: Progression free survival rate at 6 mo (%); RR: Response rate [complete + partial response (%)]; SIRT: Selective internal radio therapy.
In summary, inhibition of angiogenesis in HCC seems a very promising approach for future treatment of HCC. Multimodal approaches with combination of local and systemic therapy may further improve survival in intermediate and advanced stage HCC.
Footnotes
Peer reviewer: Dr. Paolo Del Poggio, Department of Internal Medicine, Hepatology Unit, Treviglio Hospital, Treviglio, 24047, Italy
S- Editor Tian L L- Editor O’Neill M E- Editor Ma WH
References
- 1.El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557–2576. doi: 10.1053/j.gastro.2007.04.061. [DOI] [PubMed] [Google Scholar]
- 2.Meier V, Ramadori G. Clinical staging of hepatocellular carcinoma. Dig Dis. 2009;27:131–141. doi: 10.1159/000218345. [DOI] [PubMed] [Google Scholar]
- 3.Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362:1907–1917. doi: 10.1016/S0140-6736(03)14964-1. [DOI] [PubMed] [Google Scholar]
- 4.Llovet JM, Brú C, Bruix J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis. 1999;19:329–338. doi: 10.1055/s-2007-1007122. [DOI] [PubMed] [Google Scholar]
- 5.Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429–442. doi: 10.1053/jhep.2003.50047. [DOI] [PubMed] [Google Scholar]
- 6.Vogl TJ, Naguib NN, Nour-Eldin NE, Rao P, Emami AH, Zangos S, Nabil M, Abdelkader A. Review on transarterial chemoembolization in hepatocellular carcinoma: palliative, combined, neoadjuvant, bridging, and symptomatic indications. Eur J Radiol. 2009;72:505–516. doi: 10.1016/j.ejrad.2008.08.007. [DOI] [PubMed] [Google Scholar]
- 7.Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]
- 8.Llovet JM, Bruix J. Novel advancements in the management of hepatocellular carcinoma in 2008. J Hepatol. 2008;48 Suppl 1:S20–S37. doi: 10.1016/j.jhep.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 9.Okuda K, Ohtsuki T, Obata H, Tomimatsu M, Okazaki N, Hasegawa H, Nakajima Y, Ohnishi K. Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer. 1985;56:918–928. doi: 10.1002/1097-0142(19850815)56:4<918::aid-cncr2820560437>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 10.Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M, Talwalkar J, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100:698–711. doi: 10.1093/jnci/djn134. [DOI] [PubMed] [Google Scholar]
- 11.Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 12.Abou-Alfa GK, Schwartz L, Ricci S, Amadori D, Santoro A, Figer A, De Greve J, Douillard JY, Lathia C, Schwartz B, et al. Phase II study of sorafenib in patients with advanced hepatocellular carcinoma. J Clin Oncol. 2006;24:4293–4300. doi: 10.1200/JCO.2005.01.3441. [DOI] [PubMed] [Google Scholar]
- 13.Furuse J, Ishii H, Nakachi K, Suzuki E, Shimizu S, Nakajima K. Phase I study of sorafenib in Japanese patients with hepatocellular carcinoma. Cancer Sci. 2008;99:159–165. doi: 10.1111/j.1349-7006.2007.00648.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yau T, Chan P, Ng KK, Chok SH, Cheung TT, Fan ST, Poon RT. Phase 2 open-label study of single-agent sorafenib in treating advanced hepatocellular carcinoma in a hepatitis B-endemic Asian population: presence of lung metastasis predicts poor response. Cancer. 2009;115:428–436. doi: 10.1002/cncr.24029. [DOI] [PubMed] [Google Scholar]
- 15.Fernández M, Semela D, Bruix J, Colle I, Pinzani M, Bosch J. Angiogenesis in liver disease. J Hepatol. 2009;50:604–620. doi: 10.1016/j.jhep.2008.12.011. [DOI] [PubMed] [Google Scholar]
- 16.Hoofring A, Boitnott J, Torbenson M. Three-dimensional reconstruction of hepatic bridging fibrosis in chronic hepatitis C viral infection. J Hepatol. 2003;39:738–741. doi: 10.1016/s0168-8278(03)00413-6. [DOI] [PubMed] [Google Scholar]
- 17.Ross MA, Sander CM, Kleeb TB, Watkins SC, Stolz DB. Spatiotemporal expression of angiogenesis growth factor receptors during the revascularization of regenerating rat liver. Hepatology. 2001;34:1135–1148. doi: 10.1053/jhep.2001.29624. [DOI] [PubMed] [Google Scholar]
- 18.Onori P, Morini S, Franchitto A, Sferra R, Alvaro D, Gaudio E. Hepatic microvascular features in experimental cirrhosis: a structural and morphometrical study in CCl4-treated rats. J Hepatol. 2000;33:555–563. doi: 10.1034/j.1600-0641.2000.033004555.x. [DOI] [PubMed] [Google Scholar]
- 19.Takayama T, Makuuchi M, Hirohashi S, Sakamoto M, Okazaki N, Takayasu K, Kosuge T, Motoo Y, Yamazaki S, Hasegawa H. Malignant transformation of adenomatous hyperplasia to hepatocellular carcinoma. Lancet. 1990;336:1150–1153. doi: 10.1016/0140-6736(90)92768-d. [DOI] [PubMed] [Google Scholar]
- 20.Borzio M, Fargion S, Borzio F, Fracanzani AL, Croce AM, Stroffolini T, Oldani S, Cotichini R, Roncalli M. Impact of large regenerative, low grade and high grade dysplastic nodules in hepatocellular carcinoma development. J Hepatol. 2003;39:208–214. doi: 10.1016/s0168-8278(03)00190-9. [DOI] [PubMed] [Google Scholar]
- 21.Forner A, Vilana R, Ayuso C, Bianchi L, Solé M, Ayuso JR, Boix L, Sala M, Varela M, Llovet JM, et al. Diagnosis of hepatic nodules 20 mm or smaller in cirrhosis: Prospective validation of the noninvasive diagnostic criteria for hepatocellular carcinoma. Hepatology. 2008;47:97–104. doi: 10.1002/hep.21966. [DOI] [PubMed] [Google Scholar]
- 22.Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236. doi: 10.1002/hep.20933. [DOI] [PubMed] [Google Scholar]
- 23.Torimura T, Ueno T, Kin M, Harada R, Taniguchi E, Nakamura T, Sakata R, Hashimoto O, Sakamoto M, Kumashiro R, et al. Overexpression of angiopoietin-1 and angiopoietin-2 in hepatocellular carcinoma. J Hepatol. 2004;40:799–807. doi: 10.1016/j.jhep.2004.01.027. [DOI] [PubMed] [Google Scholar]
- 24.Miura H, Miyazaki T, Kuroda M, Oka T, Machinami R, Kodama T, Shibuya M, Makuuchi M, Yazaki Y, Ohnishi S. Increased expression of vascular endothelial growth factor in human hepatocellular carcinoma. J Hepatol. 1997;27:854–861. doi: 10.1016/s0168-8278(97)80323-6. [DOI] [PubMed] [Google Scholar]
- 25.Moon WS, Rhyu KH, Kang MJ, Lee DG, Yu HC, Yeum JH, Koh GY, Tarnawski AS. Overexpression of VEGF and angiopoietin 2: a key to high vascularity of hepatocellular carcinoma? Mod Pathol. 2003;16:552–557. doi: 10.1097/01.MP.0000071841.17900.69. [DOI] [PubMed] [Google Scholar]
- 26.Yamaguchi R, Yano H, Iemura A, Ogasawara S, Haramaki M, Kojiro M. Expression of vascular endothelial growth factor in human hepatocellular carcinoma. Hepatology. 1998;28:68–77. doi: 10.1002/hep.510280111. [DOI] [PubMed] [Google Scholar]
- 27.Chao Y, Li CP, Chau GY, Chen CP, King KL, Lui WY, Yen SH, Chang FY, Chan WK, Lee SD. Prognostic significance of vascular endothelial growth factor, basic fibroblast growth factor, and angiogenin in patients with resectable hepatocellular carcinoma after surgery. Ann Surg Oncol. 2003;10:355–362. doi: 10.1245/aso.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 28.Poon RT, Lau CP, Ho JW, Yu WC, Fan ST, Wong J. Tissue factor expression correlates with tumor angiogenesis and invasiveness in human hepatocellular carcinoma. Clin Cancer Res. 2003;9:5339–5345. [PubMed] [Google Scholar]
- 29.Poon RT, Ho JW, Tong CS, Lau C, Ng IO, Fan ST. Prognostic significance of serum vascular endothelial growth factor and endostatin in patients with hepatocellular carcinoma. Br J Surg. 2004;91:1354–1360. doi: 10.1002/bjs.4594. [DOI] [PubMed] [Google Scholar]
- 30.Zeng W, Gouw AS, van den Heuvel MC, Zwiers PJ, Zondervan PE, Poppema S, Zhang N, Platteel I, de Jong KP, Molema G. The angiogenic makeup of human hepatocellular carcinoma does not favor vascular endothelial growth factor/angiopoietin-driven sprouting neovascularization. Hepatology. 2008;48:1517–1527. doi: 10.1002/hep.22490. [DOI] [PubMed] [Google Scholar]
- 31.Sadick M, Haas S, Loehr M, Elshwi M, Singer MV, Brade J, Schoenberg SO, Diehl SJ. Application of DC beads in hepatocellular carcinoma: clinical and radiological results of a drug delivery device for transcatheter superselective arterial embolization. Onkologie. 2010;33:31–37. doi: 10.1159/000264620. [DOI] [PubMed] [Google Scholar]
- 32.Poon RT, Lau C, Yu WC, Fan ST, Wong J. High serum levels of vascular endothelial growth factor predict poor response to transarterial chemoembolization in hepatocellular carcinoma: a prospective study. Oncol Rep. 2004;11:1077–1084. [PubMed] [Google Scholar]
- 33.Seki T, Tamai T, Ikeda K, Imamura M, Nishimura A, Yamashiki N, Nakagawa T, Inoue K. Rapid progression of hepatocellular carcinoma after transcatheter arterial chemoembolization and percutaneous radiofrequency ablation in the primary tumour region. Eur J Gastroenterol Hepatol. 2001;13:291–294. doi: 10.1097/00042737-200103000-00014. [DOI] [PubMed] [Google Scholar]
- 34.Li X, Feng GS, Zheng CS, Zhuo CK, Liu X. Expression of plasma vascular endothelial growth factor in patients with hepatocellular carcinoma and effect of transcatheter arterial chemoembolization therapy on plasma vascular endothelial growth factor level. World J Gastroenterol. 2004;10:2878–2882. doi: 10.3748/wjg.v10.i19.2878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shim JH, Park JW, Kim JH, An M, Kong SY, Nam BH, Choi JI, Kim HB, Lee WJ, Kim CM. Association between increment of serum VEGF level and prognosis after transcatheter arterial chemoembolization in hepatocellular carcinoma patients. Cancer Sci. 2008;99:2037–2044. doi: 10.1111/j.1349-7006.2008.00909.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Leelawat K, Laisupasin P, Kiatdilokrut A, Pongtongpool T, Narong S, Samkhumphim N, Ket-Horm S. The effect of doxorubicin on the changes of serum vascular endothelial growth factor (VEGF) in patients with hepatocellular carcinoma after transcatheter arterial chemoembolization (TACE) J Med Assoc Thai. 2008;91:1539–1543. [PubMed] [Google Scholar]
- 37.Xiong ZP, Yang SR, Liang ZY, Xiao EH, Yu XP, Zhou SK, Zhang ZS. Association between vascular endothelial growth factor and metastasis after transcatheter arterial chemoembolization in patients with hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2004;3:386–390. [PubMed] [Google Scholar]
- 38.Sergio A, Cristofori C, Cardin R, Pivetta G, Ragazzi R, Baldan A, Girardi L, Cillo U, Burra P, Giacomin A, et al. Transcatheter arterial chemoembolization (TACE) in hepatocellular carcinoma (HCC): the role of angiogenesis and invasiveness. Am J Gastroenterol. 2008;103:914–921. doi: 10.1111/j.1572-0241.2007.01712.x. [DOI] [PubMed] [Google Scholar]
- 39.Xiao EH, Guo D, Bian DJ. Effect of preoperative transcatheter arterial chemoembolization on angiogenesis of hepatocellular carcinoma cells. World J Gastroenterol. 2009;15:4582–4586. doi: 10.3748/wjg.15.4582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dhanasekaran R, Kooby DA, Staley CA, Kauh JS, Kim HS. Drug eluting beads versus conventional TACE for unresectable hepatocellular carcinoma: Survival benefits and safety. J Clin Oncol. 2009;27(suppl 15s):A4524. [Google Scholar]
- 41.Farris AB, Dhanasekaran R, Dursun N, Coban EB, McIntosh EB, Adsay V, Kim HS. Tumoral and angiogenesis factors in hepatocellular carcinoma (HCC) after drug eluting bead (DEB) transarterial chemoembolization (TACE) with doxorubicin. J Clin Oncol. 2010;28(suppl 15s):A4162. [Google Scholar]
- 42.Wilhelm S, Carter C, Lynch M, Lowinger T, Dumas J, Smith RA, Schwartz B, Simantov R, Kelley S. Discovery and development of sorafenib: a multikinase inhibitor for treating cancer. Nat Rev Drug Discov. 2006;5:835–844. doi: 10.1038/nrd2130. [DOI] [PubMed] [Google Scholar]
- 43.Avila MA, Berasain C, Sangro B, Prieto J. New therapies for hepatocellular carcinoma. Oncogene. 2006;25:3866–3884. doi: 10.1038/sj.onc.1209550. [DOI] [PubMed] [Google Scholar]
- 44.Strebel BM, Dufour JF. Combined approach to hepatocellular carcinoma: a new treatment concept for nonresectable disease. Expert Rev Anticancer Ther. 2008;8:1743–1749. doi: 10.1586/14737140.8.11.1743. [DOI] [PubMed] [Google Scholar]
- 45.Chow PK, Poon D, Win KM, Singh H, Han HS, Goh A, Choo S, Lo RH, Tan SB, Soo KC. Multicenter phase II study of SIR-sphere plus sorafenib as first-line treatment in patients with nonresectable hepatocellular carcinoma: The Asia-Pacific Hepatocellular Carcinoma Trials Group Protocol 05 (AHCC05) J Clin Oncol. 2010;28(suppl 15s):A4072. [Google Scholar]
- 46.Erhardt A, Kolligs FT, Dollinger M, Schott E, Lohse A, Bitzer M, Gog C, Rädle J, Schuchmann M, Walter C, et al. First-in-men demonstration of sorafenib plus TACE for the treatment of advanced hepatocellular carcinoma - interim analysis of the SOCRATES trial. Hepatology. 2009;50[S4](suppl 1080A):A1675. [Google Scholar]
- 47.Chung Y, Kim B, Chen C, Wang J, Chu H, Yoon J, Seetzalarom K, Bae S, Chao Y, Li C, et al. Study in Asia of the combination of transcatheter arterial chemoembolozation (TACE) with sorafenib in patients with hepatocellular carcinoma (HCC) trial (START): Second interim safety and efficay analysis. J Clin Oncol. 2010;28(suppl 15S):A4026. [Google Scholar]
- 48.Dufour JF, Hoppe H, Heim MH, Helbling B, Maurhofer O, Szucs-Farkas Z, Kickuth R, Borner M, Candinas D, Saar B. Continuous administration of sorafenib in combination with transarterial chemoembolization in patients with hepatocellular carcinoma: results of a phase I study. Oncologist. 2010;15:1198–1204. doi: 10.1634/theoncologist.2010-0180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Reyes DK, Azad N, Kamel IR, Koteish AA, Hamilton JP, Pawlik TM, Choti MA, Geschwind JFH. Phase II Trial of Sorafenib Combined with Doxorubicin Eluting Bead-Transarterial Chemoembolization (Deb-Tace) for Patients with Hepatocellular Carcinoma (Hcc): Interim Safety and Efficacy Analysis. Hepatology. 2009;50:6A–7A. [Google Scholar]
- 50.Hoffmann K, Glimm H, Radeleff B, Richter G, Heining C, Schenkel I, Zahlten-Hinguranage A, Schirrmacher P, Schmidt J, Büchler MW, et al. Prospective, randomized, double-blind, multi-center, Phase III clinical study on transarterial chemoembolization (TACE) combined with Sorafenib versus TACE plus placebo in patients with hepatocellular cancer before liver transplantation - HeiLivCa [ISRCTN24081794] BMC Cancer. 2008;8:349. doi: 10.1186/1471-2407-8-349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dempke WC, Zippel R. Brivanib, a novel dual VEGF-R2/bFGF-R inhibitor. Anticancer Res. 2010;30:4477–4483. [PubMed] [Google Scholar]
- 52.Raskopf E, Vogt A, Sauerbruch T, Schmitz V. siRNA targeting VEGF inhibits hepatocellular carcinoma growth and tumor angiogenesis in vivo. J Hepatol. 2008;49:977–984. doi: 10.1016/j.jhep.2008.07.022. [DOI] [PubMed] [Google Scholar]
- 53.Ueda H, Tanaka H, Kida Y, Fukuchi H, Ichinose M. Adjuvant chemotherapy with tegafur/uracil administration after transcatheter arterial chemoembolization for advanced hepatocellular carcinoma. Oncol Rep. 2008;19:1355–1361. [PubMed] [Google Scholar]
- 54.Koeberle D, Montemurro M, Samaras P, Majno P, Simcock M, Limacher A, Lerch S, Kovàcs K, Inauen R, Hess V, et al. Continuous Sunitinib treatment in patients with advanced hepatocellular carcinoma: a Swiss Group for Clinical Cancer Research (SAKK) and Swiss Association for the Study of the Liver (SASL) multicenter phase II trial (SAKK 77/06) Oncologist. 2010;15:285–292. doi: 10.1634/theoncologist.2009-0316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Thomas MB, Morris JS, Chadha R, Iwasaki M, Kaur H, Lin E, Kaseb A, Glover K, Davila M, Abbruzzese J. Phase II trial of the combination of bevacizumab and erlotinib in patients who have advanced hepatocellular carcinoma. J Clin Oncol. 2009;27:843–850. doi: 10.1200/JCO.2008.18.3301. [DOI] [PubMed] [Google Scholar]
- 56.Arai Y, Inaba Y, Yamamoto T, Kanai F, Aramaki T, Tanaka T, Yamakado K, Kudo M, Kanedo S, Imanaka K. A randomized phase II study of TSU-68 in patients with hepatocellular carcinoma (HCC) treated by transarterial chemoembolization (TACE) J Clin Oncol. 2010;28(suppl 15s):A4030. doi: 10.1016/j.ejca.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 57.Kanai F, Yoshida H, Tateishi R, Sato S, Kawabe T, Obi S, Kondo Y, Taniguchi M, Tagawa K, Ikeda M, et al. A phase I/II trial of the oral antiangiogenic agent TSU-68 in patients with advanced hepatocellular carcinoma. Cancer Chemother Pharmacol. 2011;67:315–324. doi: 10.1007/s00280-010-1320-2. [DOI] [PubMed] [Google Scholar]
- 58.Siegel AB, Cohen EI, Ocean A, Lehrer D, Goldenberg A, Knox JJ, Chen H, Clark-Garvey S, Weinberg A, Mandeli J, et al. Phase II trial evaluating the clinical and biologic effects of bevacizumab in unresectable hepatocellular carcinoma. J Clin Oncol. 2008;26:2992–2998. doi: 10.1200/JCO.2007.15.9947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.O'Neil BH, Williams-Goff LW, Kauh J, Bekaii-Saab T, Strosberg JR, Lee R, Deal AM, Sullivan D, Sebti SM. A phase II study of AZD6244 in advanced or metastatic hepatocellular carcinoma. J Clin Oncol. 2009;27(suppl):Ae15574. [Google Scholar]
- 60.Malka D, Dromain C, Farace F, Horn S, Pignon J, Ducreux M, Boige V. Bevacizumab in patients (pts) with advanced hepatocellular carcinoma (HCC): Preliminary results of a phase II study with circulating endothelial cell (CEC) monitoring. J Clin Oncol. 2007;25 [18S]:4570. [Google Scholar]
- 61.Schwartz JD, Schwartz M, Sung M, Lehrer D, Cohen E, Kinkhabwala M., Holloway SB, Siegel A, Ocean A, Wadler S. Bevacizumab in unresectable hepatocellular carcinoma (HCC) for patients without metastasis and without invasion of the portal vein. Gastrointestinal Cancers Symposium. 2006:A210. [Google Scholar]
- 62.Raoul JL, Finn RS, Kang YK, Park JW, Harris R, Coric V, Donica M, Walters I. An open-label phase II study of first- and second-line treatment with brivanib in patients with hepatocellular carcinoma (HCC) J Clin Oncol. 2009;27(suppl 15S):A4577. [Google Scholar]
- 63.Gruenwald V, Wilkens LGM, Greten TF, Kubicka S, Ganser A, Manns MP, Malek NP. A phase II open-label study of cetuximab in unresectable hepatocellular carcinoma: Final results. J Clin Oncol. 2007;(suppl 15S):25 [18S], 4598. [Google Scholar]
- 64.Zhu AX, Stuart K, Blaszkowsky LS, Muzikansky A, Reitberg DP, Clark JW, Enzinger PC, Bhargava P, Meyerhardt JA, Horgan K, et al. Phase 2 study of cetuximab in patients with advanced hepatocellular carcinoma. Cancer. 2007;110:581–589. doi: 10.1002/cncr.22829. [DOI] [PubMed] [Google Scholar]
- 65.Philip PA, Mahoney MR, Allmer C, Thomas J, Pitot HC, Kim G, Donehower RC, Fitch T, Picus J, Erlichman C. Phase II study of Erlotinib (OSI-774) in patients with advanced hepatocellular cancer. J Clin Oncol. 2005;23:6657–6663. doi: 10.1200/JCO.2005.14.696. [DOI] [PubMed] [Google Scholar]
- 66.Thomas MB, Chadha R, Glover K, Wang X, Morris J, Brown T, Rashid A, Dancey J, Abbruzzese JL. Phase 2 study of erlotinib in patients with unresectable hepatocellular carcinoma. Cancer. 2007;110:1059–1067. doi: 10.1002/cncr.22886. [DOI] [PubMed] [Google Scholar]
- 67.Blaszkowsky LS, Abrams TA, Miksad RA, Zheng H, Meyerhardt JA, Schrag D, Kwak EL, Fuchs C, Ryan DP, Zhu AX. Phase I/II study of everolimus in patients with advanced hepatocellular carcinoma (HCC) J Clin Oncol. 2010;28(suppl 15S):Ae14542. [Google Scholar]
- 68.O'Dwyer PJ, Giantonio BJ, Levy DE, Fitzgerald DB, Benson AB. Gefitinib in advanced unresectable hepatocellular carcinoma: Results from the Eastern Cooperative Oncology Group's Study E1203. J Clin Oncol. 2006;24 (suppl 18S):4143. [Google Scholar]
- 69.Lin AY, Fisher GA, So S, Tang C, Levitt L. Phase II study of imatinib in unresectable hepatocellular carcinoma. Am J Clin Oncol. 2008;31:84–88. doi: 10.1097/COC.0b013e3181131db9. [DOI] [PubMed] [Google Scholar]
- 70.Ramanathan RK, Belani CP, Singh DA, Tanaka M, Lenz HJ, Yen Y, Kindler HL, Iqbal S, Longmate J, Gandara DR. Phase II study of lapatinib, a dual inhibitor of epidermal growth factor receptor (EGFR) tyrosine kinase 1 and 2 (Her2/Neu) in patients (pts) with advanced biliary tree cancer (BTC) or hepatocellular cancer (HCC). A California Consortium (CCC-P) Trial. J Clin Oncol. 2006;24(suppl 18S):A4010. [Google Scholar]
- 71.Rizell M, Andersson M, Cahlin C, Hafström L, Olausson M, Lindnér P. Effects of the mTOR inhibitor sirolimus in patients with hepatocellular and cholangiocellular cancer. Int J Clin Oncol. 2008;13:66–70. doi: 10.1007/s10147-007-0733-3. [DOI] [PubMed] [Google Scholar]
- 72.Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34. doi: 10.1016/S1470-2045(08)70285-7. [DOI] [PubMed] [Google Scholar]
- 73.Zhu AX, Sahani DV, Duda DG, di Tomaso E, Ancukiewicz M, Catalano OA, Sindhwani V, Blaszkowsky LS, Yoon SS, Lahdenranta J, et al. Efficacy, safety, and potential biomarkers of sunitinib monotherapy in advanced hepatocellular carcinoma: a phase II study. J Clin Oncol. 2009;27:3027–3035. doi: 10.1200/JCO.2008.20.9908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Faivre S, Raymond E, Boucher E, Douillard J, Lim HY, Kim JS, Zappa M, Lanzalone S, Lin X, Deprimo S, et al. Safety and efficacy of sunitinib in patients with advanced hepatocellular carcinoma: an open-label, multicentre, phase II study. Lancet Oncol. 2009;10:794–800. doi: 10.1016/S1470-2045(09)70171-8. [DOI] [PubMed] [Google Scholar]
- 75.Hoda D, Catherine C, Strosberg J, Valone T, Jump H, Campos T, Halina G, Wood G, Hoffe S, Garrett CR. Phase II study of sunitinib malate in adult pts (pts) with metastatic or surgically unresectable hepatocellular carcinoma (HCC). 2008 Gastrointestinal Cancers Symposium, A267 [Google Scholar]
- 76.Sun W, Haller DG, Mykulowycz K, Rosen M, Soulen M, Capparo M, Faust T, Giantonia B, Olthoff K. Combination of capecitabine, oxaliplatin with bevacizumab in treatment of advanced hepatocellular carcinoma: a phase II study. J Clin Oncol. 2007;25(suppl 18S):A4574. [Google Scholar]
- 77.Hsu C, Yang T, Hsu C, Toh H, Epstein R, Hsiao L, Cheng A. Phase II study of bevacizumab (A) plus capecitabine (X) in patients (pts) with advanced/metastatic hepatocellular carcinoma (HCC): Final report. J Clin Oncol. 2008;26(suppl 15S):A4603. [Google Scholar]
- 78.Zhu AX, Blaszkowsky LS, Ryan DP, Clark JW, Muzikansky A, Horgan K, Sheehan S, Hale KE, Enzinger PC, Bhargava P, et al. Phase II study of gemcitabine and oxaliplatin in combination with bevacizumab in patients with advanced hepatocellular carcinoma. J Clin Oncol. 2006;24:1898–1903. doi: 10.1200/JCO.2005.04.9130. [DOI] [PubMed] [Google Scholar]
- 79.Berlin JD, Powell ME, Su Y, Horton L, Short S, Richmond A, Kauth JS, Staley CA, Mulchay M, Benson AB. Bortezomib (B) and doxorubicin (dox) in patients (pts) with hepatocellular cancer (HCC): A phase II trial of the Eastern Cooperative Oncology Group (ECOG 6202) with laboratory correlates. J Clin Oncol. 2008;26(suppl 20S):A4592. [Google Scholar]
- 80.Asnacios A, Fartoux L, Romano O, Tesmoingt C, Louafi S S, Mansoubakht T, Artru P, Poynard T, Rosmorduc O, Hebbar M, et al. Gemcitabine plus oxaliplatin (GEMOX) combined with cetuximab in patients with progressive advanced stage hepatocellular carcinoma: results of a multicenter phase 2 study. Cancer. 2008;112:2733–2739. doi: 10.1002/cncr.23489. [DOI] [PubMed] [Google Scholar]
- 81.Louafi S, Boige V, Ducreux M, Bonyhay L, Mansourbakht T, de Baere T, Asnacios A, Hannoun L, Poynard T, Taïeb J. Gemcitabine plus oxaliplatin (GEMOX) in patients with advanced hepatocellular carcinoma (HCC): results of a phase II study. Cancer. 2007;109:1384–1390. doi: 10.1002/cncr.22532. [DOI] [PubMed] [Google Scholar]
- 82.Knox JJ, Chen XE, Feld R, Nematollahi M, Cheiken R, Pond G, Zwiebel JA, Gill S, Moore M. A phase I-II study of oblimersen sodium (G3139, Genasense) in combination with doxorubicin in advanced hepatocellular carcinoma (NCI # 5798) Invest New Drugs. 2008;26:193–194. doi: 10.1007/s10637-007-9104-1. [DOI] [PubMed] [Google Scholar]
- 83.Abou-Alfa GK, Johnson P, Knox JJ, Capanu M, Davidenko I, Lacava J, Leung T, Gansukh B, Saltz LB. Doxorubicin plus sorafenib vs doxorubicin alone in patients with advanced hepatocellular carcinoma: a randomized trial. JAMA. 2010;304:2154–2160. doi: 10.1001/jama.2010.1672. [DOI] [PubMed] [Google Scholar]
- 84.Richly H, Schultheis B, Adamietz IA, Kupsch P, Grubert M, Hilger RA, Ludwig M, Brendel E, Christensen O, Strumberg D. Combination of sorafenib and doxorubicin in patients with advanced hepatocellular carcinoma: results from a phase I extension trial. Eur J Cancer. 2009;45:579–587. doi: 10.1016/j.ejca.2008.10.039. [DOI] [PubMed] [Google Scholar]