Abstract
Humans are continually exposed to ionizing radiation from terrestrial sources. The two major contributors to radiation exposure of the U.S. population are ubiquitous background radiation and medical exposure of patients. From the early 1980s to 2006, the average dose per individual in the United States for all sources of radiation increased by a factor of 1.7–6.2 mSv, with this increase due to the growth of medical imaging procedures. Radiation can place individuals at an increased risk of developing cardiovascular disease. Excess risk of cardiovascular disease occurs a long time after exposure to lower doses of radiation as demonstrated in Japanese atomic bomb survivors. This review examines sources of radiation (atomic bombs, radiation accidents, radiological terrorism, cancer treatment, space exploration, radiosurgery for cardiac arrhythmia, and computed tomography) and the risk for developing cardiovascular disease. The evidence presented suggests an association between cardiovascular disease and exposure to low-to-moderate levels of radiation, as well as the well-known association at high doses. Studies are needed to define the extent that diagnostic and therapeutic radiation results in increased risk factors for cardiovascular disease, to understand the mechanisms involved, and to develop strategies to mitigate or treat radiation-induced cardiovascular disease. Antioxid. Redox Signal. 15, 1945–1956.
Introduction
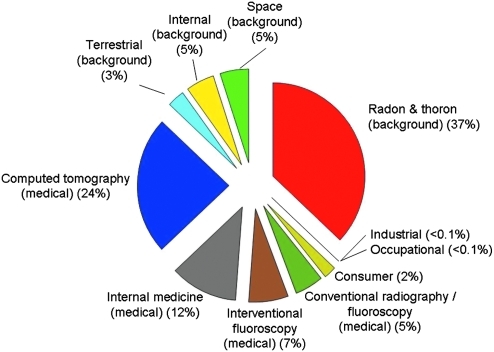
Ionizing radiation is any electromagnetic wave or particle that can remove an electron from an atom or molecule of the medium through, which it is propagating. The basic unit used to measure absorbed dose from ionizing radiation is the gray (Gy), defined as 1 joule of initial energy per kilogram of tissue (91a). The biological effects per unit of absorbed dose (Gy) differ with the type of radiation, so that a weighted quantity called the “effective dose” is used, termed the sievert (Sv) (91a). For X-rays and γ-rays, 1 Sv equals 1 Gy, but for other types of radiation this is not true (e.g., 1 Gy of high-energy protons equals 10 Sv and 1 Gy of alpha particles equals 20 Sv) (91a). All forms of life are constantly exposed to ionizing radiation from both terrestrial and cosmic sources. The major contributors to the exposure of the U.S. population from ionizing radiation are from ubiquitous background radiation and medical exposure of patients (Fig. 1). From the early 1980s to 2006, the average annual effective dose per individual in the U.S. population from all sources of radiation increased by a factor of 1.7–6.2 mSv, with the increase being almost entirely due to the growth of relatively high-dose medical imaging procedures using X-rays and radionuclides. This review discusses evidence that exposure to ionizing radiation increases the risk of cardiovascular disease.
FIG. 1.
Percent contribution of various sources of exposure to the total effective dose per individual in the U.S. population (6.2 mSv) for 2006. Reprinted with permission from the National Council on Radiation Protection and Measurements, http://NCRPonline.ord.
Sources of Cardiac Radiation Injury
Atomic bomb
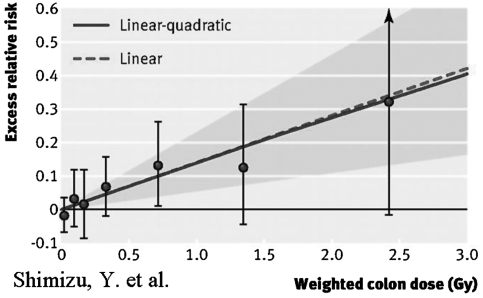
The radiation from the Hiroshima (235U) and the Nagasaki bombs (239Pu) was comprised of neutrons and γ-rays, resulting in an absorbed dose of up to 4 Gy in survivors. Neutrons account for only 1%–2% of the total radiation dose received by the survivors of the Hiroshima atomic bomb (although after accounting for their greater biological effectiveness relative to γ-rays, the proportion of the total dose becomes 10%–20%) (80a, 86). The risk of developing cardiovascular disease after whole-body exposure to radiation has been demonstrated in the Japanese atomic bomb survivors. Mortality from myocardial infarction >40 years after radiation exposure was significantly increased in people who had received an acute single dose of 1–2 Gy (70). The risk of death from heart disease increased by 17% per Gy after whole-body irradiation doses in the range 0–4 Gy, mostly from γ-rays. Approximately 16% of the 117 myocardial infarctions seen in exposed persons could be attributed to radiation exposure from the atomic bomb, most of which could be attributed to doses >1 Gy (99). A recent report indicates that radiation increases the rates of heart disease in the atomic bomb survivors at moderate dose levels (mainly 0.5–2 Gy) (Fig. 2) (79); however, the degree of risk associated with the lower doses of radiation is unclear. Stroke and heart disease combined now account for about one-third the number of radiation-associated excess deaths in atomic bomb survivors compared with cancer (79).
FIG. 2.
Radiation dose–response relation (excess relative risk) for death from heart disease, showing linear and linear-quadratic functions. Shaded area is 95% confidence region for fitted linear line. Vertical lines are 95% confidence intervals for specific dose category risks. Point estimates of risk for each dose category are indicated by circles. Reproduced from Shimizu et al. (79) with permission from BMJ Publishing Group Ltd.
Radiation accidents
The accident at the Chernobyl nuclear power plant in 1986 was the most severe accident in the history of the civilian nuclear power industry. The initial radiation exposure originated from >40 different radionuclides, notably in the first 10 days after the accident. The most significant of these were iodine (131I), caesium (137Cs), and strontium (particularly 90Sr). 131I produces β- and γ-ray emissions, 137Cs produces γ-rays, and 90Sr produces β-emissions. In 1986, the average dose to >300,000 recovery workers, “liquidators,” was nearly 150 mSv, and >350,000 other individuals received doses >10 mSv (71a). Increased risk of death from cardiovascular disease in highly exposed individuals is possible, and long-term follow-up studies are needed to determine the extent that cardiovascular disease is manifest in this population. Injury to the cardiovascular system is present early after accidental total body irradiation (TBI). In a study of 110 highly exposed individuals, 1–10 days after radiation exposure with injury to the hematopoietic system and many with severe skin lesions, 20 patients (18%) exhibited injury to the cardiovascular system, and mostly as heart failure (38).
Radiological terrorism
There is an urgent need to understand the extent of injury likely to develop to vital organs such as the heart after radiation exposure from a terrorist attack or a nuclear power plant accident, to define the mechanisms underlying the injury, and to devise treatment strategies using currently available pharmaceuticals and nutriceuticals. The International Atomic Energy Agency (IAEA) reports that there have been 175 cases of trafficking in nuclear materials, including 18 cases involving highly enriched uranium or plutonium, as well as 201 cases of trafficking in other radioactive material. According to an article in The Lancet (44), “a terrorist organization could explode a crude weapon made of stolen plutonium with a yield of 0.1–20 kilotons”; in comparison, the bomb used on Hiroshima had a yield of ∼15 kilotons. A study published in the British Medical Journal (41) has estimated that a small 12.5-kiloton nuclear bomb detonated in New York City would cause 50,000 deaths immediately with an additional several hundred thousand cases of “radiation sickness.” Another potential nuclear threat is that of an attack or accident at a nuclear power plant or waste storage facility. An equal concern is the vulnerability of spent fuel pools that can contain 20–30 times more radioactive material than the reactor core and are in buildings not nearly as strong as those that house the reactors.
The extent to which exposure to 10 Gy TBI, a potentially survivable dose in a radiation accident or radiological terrorism event (22), could result in injury to the cardiovascular system is unknown. In this situation, the entire body rather than a single organ such as the heart would be exposed to a single, possibly heterogeneous dose of radiation. Evidence that TBI injury may be a cardiovascular risk factor comes from longitudinal studies of Japanese atomic bomb survivors. In this population, mortality from cardiovascular disease is significantly increased after >40 years after single dose exposure of 1–2 Gy to the whole body (70, 99). Radiation therapy used in the treatment of benign and malignant disease has also been linked to the development of cardiovascular disease. For example, radiation therapy for the treatment of peptic ulcer disease is correlated with an increased mortality from coronary heart disease (17). Radiotherapy treatment for breast cancer is associated with an increased risk of cardiovascular disease (45), and for Hodgkin's lymphoma it is associated with an increased risk of myocardial infarction (87). In these clinical studies, nonthoracic organs were the specific target of the treatment, and yet cardiovascular disease was a significant cause of long-term morbidity or mortality (59).
In a radiological terrorism or nuclear accident event, children would account for a significant portion of the population affected and thus studies are needed to determine whether a single exposure to 10 Gy TBI in the child might increase the risk factor for development of cardiovascular disease and damage to heart morphology and ventricular function. Evidence from the clinical use of radiation to treat children with cancer supports the existence of an increased risk of cardiac injury. Exposure to 10 Gy TBI in association with bone marrow transplantation in the treatment of children for leukemia results in immediate and delayed cardiac abnormalities manifest as decreased left ventricular ejection fraction and abnormal electrocardiogram (32, 68), supporting the notion that radiation exposure in children can lead to cardiac dysfunction during development into adulthood. However, these patients with leukemia also receive chemotherapy, which may contribute to the cardiotoxicity of the overall treatment. Further studies are needed to understand the molecular events involved in radiation-induced cardiovascular disease.
Space exploration
Human space exploration missions to a Lunar and Martian environment are actively being planned. These missions will require humans to live for prolonged periods outside the protection provided by the Earth's atmosphere and geomagnetic field. The terrestrial environment effectively shields humans against exposure to deep space radiation. On the Earth most human radiation exposures (medical and natural background) are from low-linear-energy transfer photons (low LET X- and γ-rays), low-energy α-particles from radon, and γ-rays and neutrons in the survivors of atomic bomb explosions. Throughout any Lunar and Martian mission, radiation exposure will be a major hazard for humans. Understanding the long-term effects of radiation on human health is essential before undertaking space exploration for extended periods. Space radiation is very distinct from the radiation exposure on the Earth. During exploratory missions to the Lunar and Martian environments, astronauts will be exposed to heavy ions and energetic protons in galactic cosmic rays (GCR) and lower-energy protons in solar particle events (SPE), plus secondary protons, neutrons, and heavy ions produced in the spacecraft shielding particle type. Based on estimated fluence rates (number of atomic nuclei per unit area), protons are by far the most abundant. However, heavy ions have much greater ionization potential and hence are major contributors to total equivalent physical dose, with heavy iron ions being the most important contributor (31). Providing shielding in spacecraft has excessive costs and will not eliminate GCRs. The risk of developing degenerative diseases such as coronary heart disease from radiation exposure during and after deep space exploration from GCR and SPE needs to be defined before attempting exploratory missions to the Lunar and Martian surfaces. As degenerative cardiovascular disease takes extended periods to develop, the risk to the astronaut remains after the astronauts return to the Earth. Radiation dose limits need to be established that will limit the risk of degenerative cardiovascular disease from occupational radiation exposure in space to an acceptable level.
A 1000-day exploratory mission to the Martian surface may require 400 days in deep space and 600 days on the surface of Mars (23). For men aged 40 at the time of the mission, the corresponding excess risk of death from radiation exposure is estimated to be between ∼1.3% and 13% (25). During the transit to Mars, the nucleus of every cell in the body may be traversed by a high-energy proton once every 3 days, and a helium ion once every 30 days (24, 26). On the assumption that the human body contains ∼1014 cells, at least 3 × 1012 cell nuclei will be traversed by an ion of iron during the course of the mission (74). The resulting physical radiation dose incurred is estimated to be ∼0.4 Gy, corresponding to ∼1.1 Sv (74). Exposure of mice to total physical doses of 0.2 Gy of 20Ne, 1.6 Gy 40Ar, and 3.2 Gy of 12C resulted in structural injury to the coronary vessels 15 months after irradiation manifest as smooth muscle cell degeneration, fibrosis, and extracellular deposition (101). A single physical dose of 0.1 or 0.2 Gy of 56Fe ions to the orbital region of the mouse results in degenerative changes in the coronary arteries 15 months after irradiation (100). These findings suggest that exposure to ions of iron at fluence rates relevant to those that would be encountered in space exploration results in late cardiovascular injury. In addition, the observation that radiation of the brain may be responsible for causing remote injury in the heart supports recent findings that injury to the heart caused by 10 Gy TBI is indirect and likely caused by dysfunction of other organs, resulting in the export of factors that contribute to coronary sclerosis and cardiac ventricular dysfunction (6). Based on these estimates, it is likely that exposure to protons, helium, and iron ions during an exploratory mission to Mars could subsequently result in degenerative cardiovascular disease. Further studies are needed to define the risk for late cardiovascular disease after exposure to high atomic number, high-energy (HZE) ions, and protons at low fluence rates.
The physical dose needed to produce a biological effect from irradiation with a higher LET is lower when compared with γ-rays. Ions of iron and helium have a higher relative biological effectiveness than γ-rays. For humans, there is no information available as to the relative biological effectiveness of these particles in the heart. In studies on other organs, the relative biological effectiveness of high-energy heavy ions in mice and rats for carcinogenesis in skin and Harderian or mammary glands can be as high as 25–40 after doses <1 Gy (5, 16, 30). Thus, by extrapolation, the radiation dose from ions of iron, helium, and possibly protons needed to increase the risk factors for cardiovascular disease is likely to be considerably less than for γ-rays. Nontargeted, bystander effects may also increase the risk for degenerative cardiovascular disease at low doses. Studies are needed to determine the relative biological effectiveness of ions of iron, helium, and protons for increasing risk factors for cardiovascular disease at the low doses associated with space exploration.
Cancer treatment
There has been general recognition since the mid-1960s that high doses of radiation, administered as a consequence of the treatment of patients with cancer, can cause damage to the heart and pericardium (21). In the treatment of localized malignant disease, for example, Hodgkin's disease and carcinoma of the breast, the dose distribution to the heart is highly nonuniform. For example, in 50 consecutive patients treated for left-sided breast cancer using tangential fields in a single center in the United Kingdom in 2006, 44% (22/50) received a dose of >20 Gy to up to 5% of the total heart volume (88). The average maximum dose in this series was 30.7 Gy and the minimum ∼2 Gy. An average mean heart dose of 2.3 Gy was quoted, but the biological significance of an average dose when the dose variation is so large is difficult to judge. Average maximum and minimum doses to the heart for patients treated with the same technique for cancer of the right breast were of the order of 2 Gy. All of these doses represent total accumulated dose from fractionated irradiation, in this instance, a standard 15 fractions, given as 5 fractions/week over 3 weeks. Based on the application of the linear quadratic model of cell survival [a model in which biological effect E is a linear-quadratic function based on the number of dose fractions (N) and the physical dose/fraction (d), where E = (d + αβd2)Nd; the α/β ratio is a tissue-specific constant for which a value of 2 Gy has been obtained from experimental studies of heart damage (75)], the maximum average dose quoted above would translate to a equivalent single dose of ∼10 Gy. The minimum dose of ∼2 Gy (left-sided breast cancer) and about 2 Gy (right-sided breast cancer) would remain unchanged because after the small doses/fraction associated with such a total physical dose, the linear component in the model predominates. Average heart doses are not applicable to such an analysis.
A difference in the risk of mortality from cardiovascular disease for patients treated with radiotherapy for left-sided versus right-sided breast cancer have been used as evidence of radiation-induced heart disease. A recent analysis of the extensive data from the U.S. Surveillance Epidemiology and End Result (SEER) cancer registries (28) has shown that for patients treated with radiotherapy, the mortality ratio (left sided vs. right sided) was 1.16 (95% confidence interval [CI] 1.08–1.24; two-sided p-value [2p] = 0.00004) and that, furthermore, the increase in cardiac risk increased with time from diagnosis from 1.04 (0.93–1.15) for <5 years, to 1.1 (0.97–1.25) for 5–10 years, to 1.37 (1.14–1.64) 10–15 years, and 1.53 (1.25–1.86) for 15 years or more. The risk ratio for patients not treated with radiotherapy was not significantly different from 1.0. A similar trend has also been observed in an analysis of the cases from the trials included in the follow-up of the Early Breast Trialists' Collaborative Group (EBCTCG), where an excess mortality of heart disease of 1.27 (standard error 0.07, 2p = 0.0001) was quoted (19). In both of these studies, the variation in doses to the heart will be much more variable than those quoted earlier. This is because of the wide variation in the treatment methods used both in terms of the dose fractionation schedule and the orientation of the treatment fields. An indication of the likely variability was obtained from a reconstruction of heart doses for treatment schedules between 1950s and 1990s (89). Other breast cancer studies have also indicated an increased risk ratio of cardiovascular mortality after treatment for left-sided breast cancer of 1.1 (1.03–1.180) for all vascular disease and 1.13 (1.03–1.25) for Swedish women (27) and 2.1 (1.11–3.95) in a small population based study in Canada (67).
Evidence of cardiac disease developing in patients treated with mediastinal radiotherapy for Hodgkin's disease has come from a number of publications (83). It has been reported to account for 25% of all deaths that were not associated with Hodgkin's disease. Estimates of relative risk are, in general, in the range 2.2–3.1, although one study estimated it to be as high as 7.2 (4). The dose distribution, like that for patients treated for breast cancer, was highly nonuniform.
One additional study quoted as evidence that radiotherapy causes an increased risk of cardiovascular disease involved a study of 1859 patients treated for a benign condition, peptic ulcer, between 1936 and 1965, with a similar number of patients suffering from the same disease treated by other means serving as control (17). The relative risk of mortality from cardiovascular disease was 1.24 (1.04–1.47) after >10 years and only 1.02 for <10 years. The relative risk also appeared to increase with the average total cardiac dose from unity after average dose of 0.1–1.9 Gy to 1.51 after 3.1–7.6 Gy. However, the mean peak dose in the beam assumed that 5% of the total heart volume also increased with the average dose from 7.6 to 18.4 Gy, although as with the average dose there was considerable variation in the total dose in each of 4 dose ranges used for the analysis. These represent cumulative doses from fractionated irradiation using various machines, although typically 250 kVp X-rays. Doses were given as 1.5 Gy daily fractions during one or two 6–14-day treatment courses. The objective was to give a total dose of 16–17 Gy to the stomach and with about 5% of the heart (the apex) in the irradiation field during treatment. These authors extensively discussed the relative importance of the high doses to a small volume as opposed to the much lower average, very homogeneous dose to the whole volume as the cause of the increased risk. This debate holds true for all the reported studies demonstrating an increased risk of mortality from heart disease after local radiotherapy for tumors adjacent to the heart. Perhaps only after the risk from relatively standardized total doses, fractionation schedules and treatment setups have been assessed will this debate finally be resolved. The observation that average doses, normally based on the cumulative dose from fractionated irradiation, are low and in the range estimated for the Japanese atomic bomb survivors may give a false sense of security, since unlike the dose in radiotherapy patients, the dose in these survivors is more likely to be much more uniform and associated with irradiation of the whole body.
Therapeutic whole-body irradiation results in a significantly more homogeneous dose to the heart. A safe, single, and effective dose is 10 Gy given at a low dose rate. This has proven to be the critical issue since too high a dose rate will result in fatal acute pulmonary complications. Total whole-body irradiation dose can also be increased to 12–15 Gy, given as six equal fractions twice/day over 3 days. The interval between the two fractions should be at least 6 h, and dose rate is less important (69). Despite the clinical use of TBI for the treatment of a number of conditions, and in particular leukemia in childhood, there have been no systematic reports relating this treatment to delayed cardiac toxicity. Indeed in one report (58), toxicity related to TBI is implied because of the cardiac toxicity associated with the use of mediastinal irradiation used in the treatment of Hodgkin's disease in childhood. In a more recent systematic review (93) on the risk of morbidity and mortality from cardiovascular disease after radiotherapy for childhood cancer, no data sets related to the use of TBI are included. This lack of data may simply reflect the poor prognosis of patients treated using this particular modality or the lack of clarity in ascribing effects to radiation alone. Since bone marrow transplantation follows TBI, graft versus host disease and the use of chemotherapy are confounding factors. Two very recent reports deal specifically with the cardiovascular risk factors associated with the retreatment of childhood and adolescent cancers (61, 66), and some of the patients included received TBI. The hazard ratios associated with congestive heart failure were significantly increased but only after average heart dose of ≥15 Gy, which is very unlikely to include TBI cases. On the other hand, the odds ratio (OR) associated with cardiovascular risk factors for adult survivors of childhood cancer was found to be higher (OR 5.5; 95% CI 1.5–15.8) for those treated with TBI (61) compared with just chest and abdominal irradiation (OR 2.3; 95% CI 1.2–2.4). Although not significant, this provides evidence for a trend toward a greater effect of TBI. The OR values for radiation exposure were greater than those for anthracycline use in treatment, but it should be noted that this study (61) only applied to patients who had been treated between 1970 and 1986 and who had survived for at least 5 years after diagnosis, and thus represents a long-term study of the type reported earlier for breast cancer patients. The cardiovascular risk factors for this study group were also clearly linked to the current age of patients, the OR being 1.0 for <30 year olds, rising to 8.2 for the +40-year-old group.
As mentioned above, it is frequently assumed but as yet not totally proven, that the heart of children is more sensitive to radiation exposure than adults. However, there have been only a few studies published where both the follow-up of young and older patients have been compared (51, 57). In the earlier study, a total of 326 patients who had received mantle irradiation survived for over 3 years. Of these, 5.5% had a morbid cardiac event related to coronary artery disease. The mean interval from radiotherapy to the event was 13.1 years (range 4.4–27 years). The mean age at the time of irradiation was 26.3 years (range 10–53 years). The observed to expected ratio of cardiac artery disease was 38.2 in patients treated under the age of 21 compared with only 2.8 in the group as a whole. The increased risk of cardiovascular events in patients treated with radiotherapy at the age of <18 years was 44.0 in the study by Lee et al. (57), compared with the overall risk in all patients of 7.0. Difference in the age structure of the two populations and the treatment techniques used may explain the differences between the two studies, but the overall trend is self-evident.
Radiosurgery for cardiac arrhythmia
Atrial fibrillation is the most common sustained cardiac arrhythmia and is associated with impaired quality of life and an increased risk of stroke and mortality. Radiofrequency ablation of arrhythmogenic cardiac lesions can injure normal tissue beyond the intended pathological territory. Recently, X-rays have been used to target a local area of the heart associated with the genesis of atrial arrhythmias (78). A local dose of 25 Gy or larger created a lesion that altered the electrophysiology of the heart. Fibrosis and inflammatory infiltration, secondary to radiation, was present in the target area. Targeted lesion volumes were not reported. The concept of a noninvasive procedure to ablate cardiac arrhythmias is appealing. However, long-term consequences of using X-ray radiation in this setting are not well understood. Radiation-induced injury can take many years to become manifest, and the effects on surrounding tissues need to be defined. Further studies are needed to define the risk from local irradiation on developing cardiovascular disease.
Diagnostic radiology
The contribution of medical imaging to the exposure of the U.S. population is almost the same as background radiation (3.0 mSv or 48% of that from all sources) (Fig. 1). This is due to major developments in the diagnosis and treatment of cancer and heart disease over the past 20 years, predominantly in the fields of computed tomography (CT), interventional radiology, and nuclear medicine. CT is a medical imaging method used to generate three-dimensional images of the inside of an object including the heart. Multiple X-ray scans are used to generate images. CT scanning involves larger radiation doses than conventional X-ray imaging procedures (12) and accounts for 17% of hospital departmental work load, but 70%–75% of the collective dose from medical radiation. During a CT session the heart may be subjected to two to three scans. In the last 20 years, technical improvements have lowered radiation dose in CT scanning. The relatively high dose of cardiac CT (10–15 mSv) as initially implemented using retrospective gating and intended wide application in the medical community caused controversy. Cardiac CT is now performed with prospective gating resulting in a dose reduction of 80% and an average patient radiation dose of 2–4 mSv (63). This compares to a cardiac nuclear medicine stress test with a dose approximating 7.5 mSv. Given improvements in scanner performance and, in particular, image reconstruction techniques, an average radiation dose of 1–2 mSv for cardiac CT scanning may be expected. This reduction in dose would be expected to decrease the risk for developing late cardiovascular disease. However, the long-term consequences of CT interventional radiology and nuclear medicine to the development of cardiovascular disease remain unknown.
Radiation as the Cause of Late Cardiovascular Disease in Animals
Further evidence that radiation causes cardiovascular disease comes from animal studies. Radiation either directly to the thorax or specifically to the heart (18, 37, 55, 56, 103, 104) will result in the development of injury after doses of 15 Gy and higher. A single exposure to 15–60 Gy exerts an adverse long-term effect on cardiovascular function in the rat, resulting in morphological degeneration (53), mechanical dysfunction (98), damage to the endothelium (9), and increased mortality (56). It has been suggested that radiation-induced cardiac injury is mediated by micro-vascular injury caused by inflammation and oxidative stress (6, 75, 81), but further studies are needed.
The extent to which radiation affects the cardiovascular system is poorly understood. The effect of 10 Gy TBI on cardiac risk factors, coronary vasculature, and cardiac function in the WAG/RijCmcr rat has recently been determined.
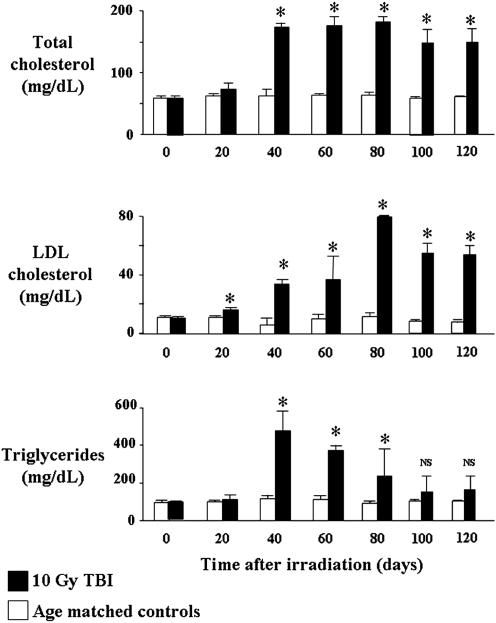
From 20 days after TBI, a progressive increase in total serum cholesterol was seen. Low-density lipoprotein cholesterol progressively increased to a peak value of 82 ± 8 mg/dl at 80 days compared with 13 ± 3 mg/dl in unirradiated rats. There was also a transient increase in triglyceride levels 40 days after TBI, which then declined to values present in unirradiated rats by 100 days (Fig. 3).
FIG. 3.
Time-related changes in total cholesterol, low-density lipoprotein (LDL) cholesterol, and triglycerides after total body irradiation (TBI). Data shown as mean ± SD, n = 6/group. *p < 0.05, 10 gray (Gy) versus unirradiated controls. Reproduced from Baker et al. (6) with permission from Taylor & Francis, Ltd. (www.tandf.co.uk/journals).
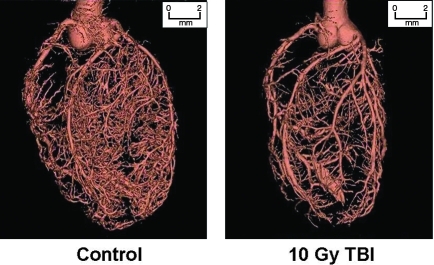
Micro-CT showed a striking reduction in the density of the smaller diameter coronary vessels (<50 μm diameter) at 120 days after TBI (Fig. 4). The diameter of the epicardial arteries were unaffected by TBI. Three-dimensional reconstruction of the coronary network revealed a decrease in the smaller diameter penetrating vessels in 32% of the irradiated heart fields examined at random compared with the unirradiated controls.
FIG. 4.
Computed tomography reconstruction of coronary arteries at 120 days after TBI, compared to age-matched control. TBI reduced the number of vessels of smaller diameter (<50 μm diameter) but not the epicardial vessels. Reproduced from Baker et al. (6) with permission from Taylor & Francis, Ltd. (www.tandf.co.uk/journals).
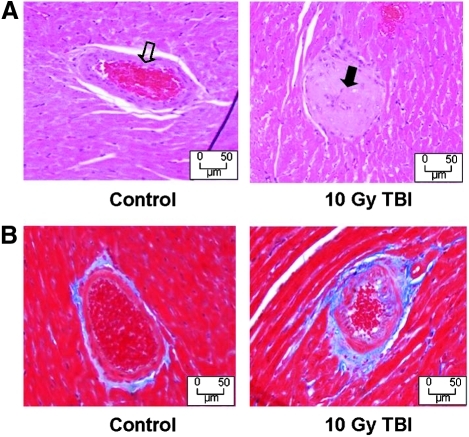
Histological studies confirmed the CT findings seen in hearts 120 days after TBI showing sclerosis of intramural coronary vessels. Affected vessels had partial to complete luminal sclerosis due to concentric laminar thickening of the vessel walls with accumulation of matrix material between layers of hyperplastic and vacuolated smooth muscle cells. The cardiomyocytes from TBI-treated hearts remained normal in appearance. Trichrome staining revealed peri-arterial fibrosis and irregular collagen deposition around the penetrating coronary vessels of irradiated hearts. Control hearts had symmetrical penetrating vessels with less collagen and fibrosis (Fig. 5).
FIG. 5.
Morphological changes to the coronary vasculature at 120 days after 10 Gy TBI. (A) Heart sections, stained with H&E, show vessel lumen completely blocked (→) as a result of myointimal proliferation 120 days after 10 Gy TBI. The lumen of a comparable vessel in an age-matched unirradiated heart is patent and contains red blood cells (⇒). (B) Heart section, stained with Trichrome, showing increased peri-arterial fibrosis in small caliber coronary vessel 120 days after 10 Gy TBI compared with a comparable vessel in an age-matched control. Fibrosis appears as blue using trichrome staining. Reproduced from Baker et al. (6) with permission from Taylor & Francis, Ltd. (www.tandf.co.uk/journals).
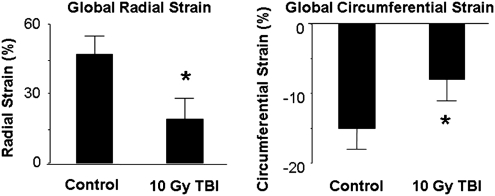
Also after TBI, rats had significantly reduced global radial (Fig. 6) and circumferential strain compared to unirradiated controls as assessed by echocardiography. These changes represent significant biological injury and are an early sign of clinical heart failure. Thus, a single exposure to 10 Gy TBI was associated with the development of risk factors for cardiovascular disease, cardiac tissue degeneration, and cardiac dysfunction.
FIG. 6.
Change in ventricular function at 120 days after 10 Gy TBI compared with age-matched controls. *p < 0.05 for 10 Gy versus unirradiated age-matched control. Reproduced from Baker et al. (6) with permission from Taylor & Francis, Ltd. (www.tandf.co.uk/journals).
Individual Radiation Sensitivity: Genetic and Gender Effects
It is likely that in exposure to space radiation, there will also be genetic and gender differences in the patterns of injury. Genetic diversity and gender differences need to be assessed as risk factors for degenerative cardiovascular disease after simulated space irradiation.
Gender differences in risk factors for cardiovascular disease in astronauts exposed to deep space radiation are incompletely understood. Female mice have been reported to be more sensitive than male mice to the effects of 56Fe ion irradiation (2–3 Gy) to the brain or hippocampal function (95). Female monkeys are more sensitive to the mortality effects of proton irradiation than males, with a dose effect occurring in the 0.25–1.13 Gy range, compared with a dose effect in the range of 3.6–4.0 Gy in males (105). Gender differences also exist in susceptibility and progression of cardiovascular disease (7, 85). As the astronaut corps at NASA is comprised of males and females, there will be a need to determine the influence of gender on risk for degenerative cardiovascular disease resulting from space radiation.
Subjects with disease of genetic repair such as ataxia telangiectasia have greatly enhanced sensitivity to irradiation, and a greater sensitivity is also reported for subjects with scleroderma (20). It is likely that in astronauts exposed to space radiation, there will be genetic differences in the patterns of injury. In support of the notion of genetics as a factor influencing cardiovascular disease, one of us (John Hopewell) has shown that after local heart irradiation, cardiac output declined in Sprague Dawley rats and increased in Wistar rats (104). Selection of astronauts based upon the composition of an individual's genome may be a useful strategy to decrease risk factors for developing degenerative cardiovascular disease from radiation exposure associated with deep space exploration. Investigations are urgently needed to determine the mechanistic basis for these genetic and gender differences.
Potential for Medical Countermeasures to Prevent, Mitigate, and Treat Radiation-Induced Injury
There is an immediate need for a practical therapy to prevent, mitigate, and/or treat radiation-induced injury to the heart after irradiation (22). The U.S. National Institutes of Health have recommended terminology for such therapies that is consistent with other areas of medicine: prophylactic agents/protectors are agents that must be given before radiation exposure; mitigators are agents that are given after exposure, but before the appearance of overt evidence of injury; and treatment refers to agents that are given after overt symptoms develop (84). Agents with evidence for cardiac efficacy include free radical scavengers, antioxidants, statins, steroids, natural products, and other agents (8, 10, 36, 80, 90).
In the present discussion, this will be limited to compounds that are potentially available to act as pharmaceutical and nutraceutical medical countermeasures against radiation-induced injury to the heart. The compounds discussed are not approved for use as mitigators of radiation-induced injury by the Food and Drug Administration (FDA). Design and development of novel pharmaceutical and nutraceuticals countermeasures against radiation-induced injury are outside the scope of this review.
Vascular endothelium
The vascular endothelium is believed to be a target for radiation-induced injury. The endothelium is the sentry against cardiovascular disease. It plays a pivotal role in vascular tone and remodeling as well as regulating thrombosis and inflammation. The thrombotic and inflammatory pathways are regulated by nitric oxide produced from endothelial nitric oxide synthase, thrombin, and the thrombin receptor (PAR1), and fibrinogen among other factors. Nitric oxide, through its anti-inflammatory and antithrombotic effects, is able to diminish leukocyte adhesion and arterial thrombosis. Nitric oxide and thrombosis have an intricate relationship. Nitric oxide decreases thrombosis by inhibiting the expression of the prothrombotic protein plasminogen activator inhibitor-1 and decreasing platelet aggregation (60), whereas thrombin enhances nitric oxide production through the thrombin receptor (43). Moreover, long-term exposure of endothelial cells (ECs) to thrombin results in a downregulation of endothelial nitric oxide synthase (35). Thrombin and endothelial PAR1 also contribute to the expression of inflammatory cytokines and adhesion molecules, and after leukocyte adhesion, their migration into tissue (50, 64, 71). Fibrinogen, a protein secreted by the liver, is a key component of the coagulation cascade and functions at the level of the endothelium not only to promote thrombosis, but also to upregulate the inflammatory response. Elevated fibrinogen is correlated with an inflammatory state and the progression of atherosclerosis (73). EC damage and/or cell loss after radiation is likely to disrupt the anti-inflammatory and antithrombotic mechanisms, thereby promoting tissue damage and fibrosis through inflammatory and thrombotic mechanisms. Nitric oxide, thrombin, and PAR1 are central in this role. Thus, pharmaceutical and nutriceutical countermeasures directed toward maintaining endothelial function, especially by normalizing nitric oxide production and PAR1 expression, have potential to both mitigate and treat radiation-induced injury to the heart.
Angiotensin-converting enzyme inhibitors
The angiotensin-converting enzyme (ACE) inhibitor captopril, given after radiation exposure, mediates against structural changes to the heart after radiation (20 Gy), but is unable to prevent the decline in cardiac function (102). However, the effect of captopril with the lower levels of radiation associated with either a terrorist exploding a radiological device or as a nuclear accident on subsequent injury to the heart is unknown. Captopril also mediates radiation-induced renal injury (65). These ACE inhibitors are strong candidates for further evaluation in rats exposed to lower doses of irradiation, for example, 10 Gy.
Statins
The vascular endothelium is a major effector compartment for the pleiotropic effects of statins, many of which appear to be related to the ability of statins to prevent the development of EC dysfunction during various disease states. Maintaining an anticoagulant cell surface is a critical aspect of endothelial function, and ensures not only thrombo-hemorrhagic homeostasis, but also the appropriate regulation of inflammatory and fibro-proliferative responses. Considerable research has focused on the mechanisms by which statins enhance endothelial anticoagulant and fibrinolytic properties. These studies show that statins increase the expression and enhance the activity of endothelial nitric oxide synthase (54), upregulate prostacyclin (76) and tissue-type plasminogen activator (34), decrease oxidative stress (72), and downregulate thrombin receptor expression (49).
Simvastatin is a prodrug of a specific inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase (29). Simvastatin is able to decrease lipid levels in blood. Moreover, simvastatin has recently been shown to be capable of decreasing radiation-induced injury to rats. Administration of dietary simvastatin for 1 year significantly decreased the incidence (36%) of radiation-induced mammary tumors compared with that of the control rats (88%) (48). Also, the development of adenocarcinoma in the simvastatin-fed rats was significantly reduced compared to that in the control rats. Simvastatin is also effective in reducing radiation-induced brain injury (S. Brown, pers. comm.). There have been no studies on the ability of simvastatin to ameliorate radiation-induced injury to the heart.
Vascular injury is believed to be a key factor in the pathogenesis of radiation-induced sclerosis, fibrosis, and thrombosis, but only recently have studies proposed a link between the persistence of endothelial dysfunction and radiation pathologies (33, 62, 82, 96). Radiation induces an early and sustained activation of vascular ECs (39, 40, 92), resulting in maintenance of the functional pro-inflammatory and thrombogenic properties of these cells (40, 94). The slow loss of ECs after irradiation can invoke a change in EC function including the increased expression of adhesion molecules. Because it has been proposed that there may be indications for statins in conditions with endothelial dysfunction (11, 97), statins may improve endothelial function after radiation exposure. Pravastatin inhibits radiation-induced increases in the production of IL6, IL8, and MCP1 and the expression of ICAM1 by ECs, thus providing additional evidence for the anti-inflammatory properties of statins on EC. There have been no studies on the ability of pravastatin to treat radiation-induced injury to the heart.
Curcumin
Curcumin is a natural tropical plant polyphenol compound derived from the Curcuma longa Linn, a root related to Ginger. It is commonly found as a food additive and in the spice turmeric. Curcumin is a deep yellow pigment, and is responsible for the color found in South Asian curries as well as American yellow mustard, and has been an ingredient used in cooking since 600 B.C. Powdered turmeric is widely available in supermarkets throughout the United States as a household spice. The major pigment found in turmeric is curcumin (diferuloymethane), which is now known to possess pharmacologic anti-inflammatory and antioxidant properties. The Curcumin content of turmeric spice may range from 189 to 250 mg/g (i.e., 18%–25% of turmeric by weight) (77). Curcumin has been used for hundreds of years as a component of Ayurvedic medicines, as a dietary supplement known to help gastrointestinal ailments, inflammatory lesions of the skin, and as an exfoliant. Western medical interest in the compound has increased dramatically after the discovery of the potent effect of curcumin as an inhibitor of NFκB and of the mitogen-activated protein kinases. There are over 20 clinical trials of curcumin currently registered at clinicaltrials.gov (Web site reference www.clinicaltrials.gov). Mammary and pituitary tumors in rats induced by radiation can be inhibited by curcimin administered after radiation exposure (47). However, there have been no studies on the ability of curcumin to ameliorate radiation-induced injury to the heart.
Nitric oxide
The reduction in nitric oxide generation as a result of dysfunctional vascular endothelium contributes to cardiovascular disease. Continuous generation of nitric oxide is essential to the survival and function of the heart, and decreased production of nitric oxide is central to the development of cardiovascular diseases (42, 46). Nitrite is a major storage form of nitric oxide (•NO) in blood and tissues (14). Oral nitrite reverses NG-nitro-L-arginine methyl ester-induced hypertension and serves as an alternate source of •NO in vivo (91). Further, plasma nitrite levels progressively decrease with increasing cardiovascular risk (52). Nitrite is currently undergoing clinical trials to increase nitric oxide in patients with coronary artery disease (ClinicalTrials.gov identifier NCT00069654). Because a substantial portion of steady-state nitrite concentrations in blood and tissue are derived from dietary sources (15), modulation of nitrite intake may provide a first-line defense against radiation-induced injury. However, there is no experimental evidence indicating the consequences of dietary nitrite supplementation on risk factors for cardiovascular disease after radiation exposure.
Conclusion
Humans are continually exposed to radiation from terrestrial sources. On the Earth most human radiation exposures (medical and natural background) are from low LET photons (low LET X- and γ-rays), low energy α-particles from radon, and γ-rays and neutrons in the survivors of atomic bomb explosions. Diagnostic and therapeutic applications of radiation are widespread, and threats from radiological terrorism and nuclear power plants persist. These sources of radiation may increase risk for developing cardiovascular disease. Health risks associated with exposure to radiation exposure from deep space radiation will persist after astronauts return to the Earth as cardiovascular changes develop at long periods after exposure. For example, cardiovascular injury was observed in Japanese atomic bomb survivors at doses as low as 0.5–2.0 Gy to the entire body, doses lower than those normally associated with cardiac injury if the exposure dose is localized to the thorax or heart. The risk of developing cardiovascular disease in patients exposed to radiation for diagnostic and therapeutic purposes, and from occupational radiation exposure remains a health concern. Further studies are needed to define the mechanisms underlying injury to the cardiovascular system from exposure to terrestrial and cosmic radiation, and to develop strategies for mitigation and treatment of radiation-induced injury.
Abbreviations Used
- 2p
two sided p-value
- ACE
angiotensin-converting enzyme
- CI
confidence interval
- CT
computed tomography
- EBCTCG
Early Breast Trialists' Collaborative Group
- EC
endothelial cells
- FDA
Food and Drug Administration
- GCR
galactic cosmic rays
- Gy
gray
- HMG-CoA
3-hydroxy-3-methylglutaryl-coenzyme A
- HZE
high energy
- IAEA
International Atomic Energy Agency
- LET
linear energy transfer
- •NO
nitric oxide
- OR
odds ratio
- Sv
sievert
- SPE
solar particle events
- TBI
total body irradiation
- U.S. SEER
U.S. Surveillance Epidemiology and End Result
Acknowledgment
This work was supported in part by cooperative agreement AI067734 and grants HL54075 and AI080363 from the National Institutes of Health.
References
- 1–3. These references have been deleted.
- 4.Adams MJ. Lipshultz SE. Schwartz C. Fajardo LF. Coen V. Constine LS. Radiation-associated cardiovascular disease: manifestations and management. Semin Radiat Oncol. 2003;13:346–356. doi: 10.1016/S1053-4296(03)00026-2. [DOI] [PubMed] [Google Scholar]
- 5.Alpen EL. Powers-Risius P. Curtis SB. DeGuzman R. Tumorigenic potential of high-Z, high-LET charged-particle radiations. Radiat Res. 1993;136:382–391. [PubMed] [Google Scholar]
- 6.Baker JE. Fish B. Su J. Haworth S. Strande J. Komorowski R. Migrino R. Doppalapudi A. Harmann L. Li X. Hopewell J. Moulder J. 10 Gy Total body irradiation increases risk of coronary sclerosis, degeneration of heart structure and function in a rat model. Int J Radiat Biol. 2009;85:1089–1100. doi: 10.3109/09553000903264473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barrett-Connor E. Sex differences in coronary heart disease. Why are women so superior? The 1995 Ancel Keys Lecture. Circulation. 1997;95:252–264. doi: 10.1161/01.cir.95.1.252. [DOI] [PubMed] [Google Scholar]
- 8.Beller CJ. Radovits T. Seres L. Kosse J. Krempien R. Gross ML. Penzel R. Berger I. Huber PE. Hagl S. Szabo C. Szabo G. Poly(ADP-ribose) polymerase inhibition reverses vascular dysfunction after gamma-irradiation. Int J Radiat Oncol Biol Phys. 2006;65:1528–1535. doi: 10.1016/j.ijrobp.2006.03.058. [DOI] [PubMed] [Google Scholar]
- 9.Boerma M. Kruse JJ. van Loenen M. Klein HR. Bart CI. Zurcher C. Wondergem J. Increased deposition of von Willebrand factor in the rat heart after local ionizing irradiation. Strahlenther Onkol. 2004;180:109–116. doi: 10.1007/s00066-004-1138-0. [DOI] [PubMed] [Google Scholar]
- 10.Boerma M. Roberto KA. Hauer-Jensen M. Prevention and treatment of functional and structural radiation injury in the rat heart by pentoxifylline and alpha-tocopherol. Int J Radiat Oncol Biol Phys. 2008;71:170–177. doi: 10.1016/j.ijrobp.2008.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bonetti PO. Lerman LO. Napoli C. Lerman A. Statin effects beyond lipid lowering—are they clinically relevant? Eur Heart J. 2003;24:225–248. doi: 10.1016/s0195-668x(02)00419-0. [DOI] [PubMed] [Google Scholar]
- 12.Brenner DJ. Hall EJ. Computed tomography—an increasing source of radiation exposure. N Engl J Med. 2007;357:2277–2284. doi: 10.1056/NEJMra072149. [DOI] [PubMed] [Google Scholar]
- 13. This reference has been deleted.
- 14.Bryan NS. Nitrite in nitric oxide biology: cause or consequence? A systems-based review. Free Radic Biol Med. 2006;41:691–701. doi: 10.1016/j.freeradbiomed.2006.05.019. [DOI] [PubMed] [Google Scholar]
- 15.Bryan NS. Fernandez BO. Bauer SM. Garcia-Saura MF. Milsom AB. Rassaf T. Maloney RE. Bharti A. Rodriguez J. Feelisch M. Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat Chem Biol. 2005;1:290–297. doi: 10.1038/nchembio734. [DOI] [PubMed] [Google Scholar]
- 16.Burns FJ. Jin Y. Koenig KL. Hosselet S. The low carcinogenicity of electron radiation relative to argon ions in rat skin. Radiat Res. 1993;135:178–188. [PubMed] [Google Scholar]
- 17.Carr ZA. Land CE. Kleinerman RA. Weinstock RW. Stovall M. Griem ML. Mabuchi K. Coronary heart disease after radiotherapy for peptic ulcer disease. Int J Radiat Oncol Biol Phys. 2005;61:842–850. doi: 10.1016/j.ijrobp.2004.07.708. [DOI] [PubMed] [Google Scholar]
- 18.Cilliers GD. Harper IS. Lochner A. Radiation-induced changes in the ultrastructure and mechanical function of the rat heart. Radiother Oncol. 1989;16:311–326. doi: 10.1016/0167-8140(89)90044-3. [DOI] [PubMed] [Google Scholar]
- 19.Clarke M. Collins R. Darby S. Davies C. Elphinstone P. Evans E. Godwin J. Gray R. Hicks C. James S. MacKinnon E. McGale P. McHugh T. Peto R. Taylor C. Wang Y. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;366:2087–2106. doi: 10.1016/S0140-6736(05)67887-7. [DOI] [PubMed] [Google Scholar]
- 20.Cohen EP. Radiation nephropathy after bone marrow transplantation. Kidney Int. 2000;58:903–918. doi: 10.1046/j.1523-1755.2000.00241.x. [DOI] [PubMed] [Google Scholar]
- 21.Cohn KE. Stewart JR. Fajardo LF. Hancock EW. Heart disease following radiation. Medicine (Baltimore) 1967;46:281–298. doi: 10.1097/00005792-196705000-00003. [DOI] [PubMed] [Google Scholar]
- 22.Coleman CN. Blakely WF. Fike JR. MacVittie TJ. Metting NF. Mitchell JB. Moulder JE. Preston RJ. Seed TM. Stone HB. Tofilon PJ. Wong RS. Molecular and cellular biology of moderate-dose (1–10 Gy) radiation and potential mechanisms of radiation protection: report of a workshop at Bethesda, Maryland, December 17–18, 2001. Radiat Res. 2003;159:812–834. doi: 10.1667/rr3021. [DOI] [PubMed] [Google Scholar]
- 23.Cucinotta F. Kim M. Ren L. Managing Luncar and Mars Mission Radiation Risks, Part I: Cancer Risks, Uncertainties, and Shielding Effectiveness, edited by NASA/TP-2005-213164. Washington, DC: NASA; 2005. p. 18. [Google Scholar]
- 24.Cucinotta FA. Nikjoo H. Goodhead DT. The effects of delta rays on the number of particle-track traversals per cell in laboratory and space exposures. Radiat Res. 1998;150:115–119. [PubMed] [Google Scholar]
- 25.Cucinotta FA. Schimmerling W. Wilson JW. Peterson LE. Badhwar GD. Saganti PB. Dicello JF. Space radiation cancer risks and uncertainties for Mars missions. Radiat Res. 2001;156:682–688. doi: 10.1667/0033-7587(2001)156[0682:srcrau]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 26.Curtis SB. Vazquez M. Wilson JW. Atwell W. Kim M. Capala J. Cosmic hit frequencies in critical sites in the central nervous system. Adv Space Res. 1998;12:197–207. doi: 10.1016/s0273-1177(98)80011-2. [DOI] [PubMed] [Google Scholar]
- 27.Darby S. McGale P. Peto R. Granath F. Hall P. Ekbom A. Mortality from cardiovascular disease more than 10 years after radiotherapy for breast cancer: nationwide cohort study of 90 000 Swedish women. BMJ. 2003;326:256–257. doi: 10.1136/bmj.326.7383.256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Darby SC. McGale P. Taylor CW. Peto R. Long-term mortality from heart disease and lung cancer after radiotherapy for early breast cancer: prospective cohort study of about 300,000 women in US SEER cancer registries. Lancet Oncol. 2005;6:557–565. doi: 10.1016/S1470-2045(05)70251-5. [DOI] [PubMed] [Google Scholar]
- 29.Del Puppo M. Rauli S. Galli Kienle M. Inhibition of cholesterol synthesis and hepatic 3-hydroxy-3-methylglutaryl—CoA reductase in rats by simvastatin and pravastatin. Lipids. 1995;30:1057–1061. doi: 10.1007/BF02536292. [DOI] [PubMed] [Google Scholar]
- 30.Dicello JF. Christian A. Cucinotta FA. Gridley DS. Kathirithamby R. Mann J. Markham AR. Moyers MF. Novak GR. Piantadosi S. Ricart-Arbona R. Simonson DM. Strandberg JD. Vazquez M. Williams JR. Zhang Y. Zhou H. Huso D. In vivo mammary tumourigenesis in the Sprague-Dawley rat and microdosimetric correlates. Phys Med Biol. 2004;49:3817–3830. doi: 10.1088/0031-9155/49/16/024. [DOI] [PubMed] [Google Scholar]
- 31.Durante M. Kronenberg A. Ground-based research with heavy ions for space radiation protection. Adv Space Res. 2005;35:180–184. doi: 10.1016/j.asr.2004.12.034. [DOI] [PubMed] [Google Scholar]
- 32.Eames GM. Crosson J. Steinberger J. Steinbuch M. Krabill K. Bass J. Ramsay NK. Neglia JP. Cardiovascular function in children following bone marrow transplant: a cross-sectional study. Bone Marrow Transplant. 1997;19:61–66. doi: 10.1038/sj.bmt.1700600. [DOI] [PubMed] [Google Scholar]
- 33.Epperly MW. Sikora CA. DeFilippi SJ. Gretton JE. Bar-Sagi D. Archer H. Carlos T. Guo H. Greenberger JS. Pulmonary irradiation-induced expression of VCAM-I and ICAM-I is decreased by manganese superoxide dismutase-plasmid/liposome (MnSOD-PL) gene therapy. Biol Blood Marrow Transplant. 2002;8:175–187. doi: 10.1053/bbmt.2002.v8.pm12014807. [DOI] [PubMed] [Google Scholar]
- 34.Essig M. Nguyen G. Prie D. Escoubet B. Sraer JD. Friedlander G. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors increase fibrinolytic activity in rat aortic endothelial cells. Role of geranylgeranylation and Rho proteins. Circ Res. 1998;83:683–690. doi: 10.1161/01.res.83.7.683. [DOI] [PubMed] [Google Scholar]
- 35.Eto M. Barandier C. Rathgeb L. Kozai T. Joch H. Yang Z. Luscher TF. Thrombin suppresses endothelial nitric oxide synthase and upregulates endothelin-converting enzyme-1 expression by distinct pathways: role of Rho/ROCK and mitogen-activated protein kinase. Circ Res. 2001;89:583–590. doi: 10.1161/hh1901.097084. [DOI] [PubMed] [Google Scholar]
- 36.Evans ML. Graham MM. Mahler PA. Rasey JS. Use of steroids to suppress vascular response to radiation. Int J Radiat Oncol Biol Phys. 1987;13:563–567. doi: 10.1016/0360-3016(87)90072-1. [DOI] [PubMed] [Google Scholar]
- 37.Fajardo LF. Stewart JR. Experimental radiation-induced heart disease. I. Light microscopic studies. Am J Pathol. 1970;59:299–316. [PMC free article] [PubMed] [Google Scholar]
- 38.Fliedner TM. D Dorr H. Meineke V. Multi-organ involvement as a pathogenetic principle of the radiation syndromes: a study involving 110 case histories documented in SEARCH and classified as the bases of haematopoietic indicators of effect. BJR Suppl. 2005;27:1–8. [Google Scholar]
- 39.Gaugler MH. Squiban C. Mouthon MA. Gourmelon P. van der Meeren A. Irradiation enhances the support of haemopoietic cell transmigration, proliferation and differentiation by endothelial cells. Br J Haematol. 2001;113:940–950. doi: 10.1046/j.1365-2141.2001.02852.x. [DOI] [PubMed] [Google Scholar]
- 40.Gaugler MH. Squiban C. van der Meeren A. Bertho JM. Vandamme M. Mouthon MA. Late and persistent up-regulation of intercellular adhesion molecule-1 (ICAM-1) expression by ionizing radiation in human endothelial cells in vitro. Int J Radiat Biol. 1997;72:201–209. doi: 10.1080/095530097143428. [DOI] [PubMed] [Google Scholar]
- 41.Helfand I. Forrow L. Tiwari J. Nuclear terrorism. BMJ. 2002;324:356–359. doi: 10.1136/bmj.324.7333.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Herman AG. Moncada S. Therapeutic potential of nitric oxide donors in the prevention and treatment of atherosclerosis. Eur Heart J. 2005;26:1945–1955. doi: 10.1093/eurheartj/ehi333. [DOI] [PubMed] [Google Scholar]
- 43.Hirano K. Nomoto N. Hirano M. Momota F. Hanada A. Kanaide H. Distinct Ca2+ Requirement for NO production between proteinase-activated receptor 1 and 4 (PAR1 and PAR4) in vascular endothelial cells. J Pharmacol Exp Ther. 2007;322:668–677. doi: 10.1124/jpet.107.121038. [DOI] [PubMed] [Google Scholar]
- 44.Holdstock D. Waterston L. Nuclear weapons, a continuing threat to health. Lancet. 2000;355:1544–1547. doi: 10.1016/S0140-6736(00)02176-0. [DOI] [PubMed] [Google Scholar]
- 45.Hooning MJ. Botma A. Aleman BM. Baaijens MH. Bartelink H. Klijn JG. Taylor CW. van Leeuwen FE. Long-term risk of cardiovascular disease in 10-year survivors of breast cancer. J Natl Cancer Inst. 2007;99:365–375. doi: 10.1093/jnci/djk064. [DOI] [PubMed] [Google Scholar]
- 46.Ignarro LJ. Nitric oxide as a unique signaling molecule in the vascular system: a historical overview. J Physiol Pharmacol. 2002;53:503–514. [PubMed] [Google Scholar]
- 47.Inano H. Onoda M. Radioprotective action of curcumin extracted from Curcuma longa LINN: inhibitory effect on formation of urinary 8-hydroxy-2′-deoxyguanosine, tumorigenesis, but not mortality, induced by gamma-ray irradiation. Int J Radiat Oncol Biol Phys. 2002;53:735–743. doi: 10.1016/s0360-3016(02)02794-3. [DOI] [PubMed] [Google Scholar]
- 48.Inano H. Suzuki K. Onoda M. Wakabayashi K. Anti-carcinogenic activity of simvastatin during the promotion phase of radiation-induced mammary tumorigenesis of rats. Carcinogenesis. 1997;18:1723–1727. doi: 10.1093/carcin/18.9.1723. [DOI] [PubMed] [Google Scholar]
- 49.Jacobson JR. Dudek SM. Birukov KG. Ye SQ. Grigoryev DN. Girgis RE. Garcia JGN. Cytoskeletal activation and altered gene expression in endothelial barrier regulation by simvastatin. Am J Respir Cell Mol Biol. 2004;30:662–670. doi: 10.1165/rcmb.2003-0267OC. [DOI] [PubMed] [Google Scholar]
- 50.Johnson K. Choi Y. DeGroot E. Samuels I. Creasey A. Aarden L. Potential mechanisms for a proinflammatory vascular cytokine response to coagulation activation. J Immunol. 1998;160:5130–5135. [PubMed] [Google Scholar]
- 51.King V. Constine LS. Clark D. Schwartz RG. Muhs AG. Henzler M. Hutson A. Rubin P. Symptomatic coronary artery disease after mantle irradiation for Hodgkin's disease. Int J Radiat Oncol Biol Phys. 1996;36:881–889. doi: 10.1016/s0360-3016(96)00295-7. [DOI] [PubMed] [Google Scholar]
- 52.Kleinbongard P. Dejam A. Lauer T. Jax T. Kerber S. Gharini P. Balzer J. Zotz RB. Scharf RE. Willers R. Schechter AN. Feelisch M. Kelm M. Plasma nitrite concentrations reflect the degree of endothelial dysfunction in humans. Free Radic Biol Med. 2006;40:295–302. doi: 10.1016/j.freeradbiomed.2005.08.025. [DOI] [PubMed] [Google Scholar]
- 53.Kruse JJ. Zurcher C. Strootman EG. Bart CI. Schlagwein N. Leer JW. Wondergem J. Structural changes in the auricles of the rat heart after local ionizing irradiation. Radiother Oncol. 2001;58:303–311. doi: 10.1016/s0167-8140(00)00327-3. [DOI] [PubMed] [Google Scholar]
- 54.Laufs U. La Fata V. Plutzky J. Liao JK. Upregulation of endothelial nitric oxide synthase by HMG CoA reductase inhibitors. Circulation. 1998;97:1129–1135. doi: 10.1161/01.cir.97.12.1129. [DOI] [PubMed] [Google Scholar]
- 55.Lauk S. Strain differences in the radiation response of the rat heart. Radiother Oncol. 1986;5:333–335. doi: 10.1016/s0167-8140(86)80182-7. [DOI] [PubMed] [Google Scholar]
- 56.Lauk S. Kiszel Z. Buschmann J. Trott KR. Radiation-induced heart disease in rats. Int J Radiat Oncol Biol Phys. 1985;11:801–808. doi: 10.1016/0360-3016(85)90314-1. [DOI] [PubMed] [Google Scholar]
- 57.Lee CK. Aeppli D. Nierengarten ME. The need for long-term surveillance for patients treated with curative radiotherapy for Hodgkin's disease: University of Minnesota experience. Int J Radiat Oncol Biol Phys. 2000;48:169–179. doi: 10.1016/s0360-3016(00)00647-7. [DOI] [PubMed] [Google Scholar]
- 58.Leiper AD. Late effects of total body irradiation. Arch Dis Child. 1995;72:382–385. doi: 10.1136/adc.72.5.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Little MP. Tawn EJ. Tzoulaki I. Wakeford R. Hildebrandt G. Paris F. Tapio S. Elliott P. A systematic review of epidemiological associations between low and moderate doses of ionizing radiation and late cardiovascular effects, and their possible mechanisms. Radiat Res. 2008;169:99–109. doi: 10.1667/RR1070.1. [DOI] [PubMed] [Google Scholar]
- 60.Loscalzo J. Nitric oxide insufficiency, platelet activation, and arterial thrombosis. Circ Res. 2001;88:756–762. doi: 10.1161/hh0801.089861. [DOI] [PubMed] [Google Scholar]
- 61.Meacham L. Chow E. Ness K. Kamdar K. Chen Y. Yasui Y. Oeffinger K. Sklar C. Robison L. Mertens A. Cardiovascular risk factors in adult survivors of pediatric cancer-a report from the childhood cancer survivor study. Cancer Epidemiol Biomarkers Prev. 2010;19:170–181. doi: 10.1158/1055-9965.EPI-09-0555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Molla M. Gironella M. Miquel R. Tovar V. Engel P. Biete A. Pique JM. Panes J. Relative roles of ICAM-1 and VCAM-1 in the pathogenesis of experimental radiation-induced intestinal inflammation. Int J Radiat Oncol Biol Phys. 2003;57:264–273. doi: 10.1016/s0360-3016(03)00523-6. [DOI] [PubMed] [Google Scholar]
- 63.Morin RL. Gerber TC. McCollough CH. Radiation dose in computed tomography of the heart. Circulation. 2003;107:917–922. doi: 10.1161/01.cir.0000048965.56529.c2. [DOI] [PubMed] [Google Scholar]
- 64.Moser R. Groscurth P. Fehr J. Promotion of transendothelial neutrophil passage by human thrombin. J Cell Sci. 1990;96(Pt 4):737–744. doi: 10.1242/jcs.96.4.737. [DOI] [PubMed] [Google Scholar]
- 65.Moulder JE. Fish BL. Cohen EP. Treatment of radiation nephropathy with ACE inhibitors. Int J Radiat Oncol Biol Phys. 1993;27:93–99. doi: 10.1016/0360-3016(93)90425-u. [DOI] [PubMed] [Google Scholar]
- 66.Mulrooney D. Yeazel M. Kawashima T. Mertens A. Mitby P. Stovall M. Donaldson S. Green D. Sklar C. Robison L. Lesenring W. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: retrospective analysis of the childhood cancer survivor study cohort. BMJ. 2010;340:34–44. doi: 10.1136/bmj.b4606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Paszat LF. Mackillop WJ. Groome PA. Schulze K. Holowaty E. Mortality from myocardial infarction following postlumpectomy radiotherapy for breast cancer: a population-based study in Ontario, Canada. Int J Radiat Oncol Biol Phys. 1999;43:755–762. doi: 10.1016/s0360-3016(98)00412-x. [DOI] [PubMed] [Google Scholar]
- 68.Pihkala J. Saarinen UM. Lundstrom U. Salmo M. Virkola K. Virtanen K. Siimes MA. Pesonen E. Effects of bone marrow transplantation on myocardial function in children. Bone Marrow Transplant. 1994;13:149–155. [PubMed] [Google Scholar]
- 69.Plowman N. A review of total body irradiation. Br J Radiol. 1987;22:135–145. [PubMed] [Google Scholar]
- 70.Preston DL. Shimizu Y. Pierce DA. Suyama A. Mabuchi K. Studies of mortality of atomic bomb survivors. Report 13: Solid cancer and noncancer disease mortality: 1950–1997. Radiat Res. 2003;160:381–407. doi: 10.1667/rr3049. [DOI] [PubMed] [Google Scholar]
- 71.Rahman A. Anwar KN. True AL. Malik AB. Thrombin-induced p65 homodimer binding to downstream NF-kappa B site of the promoter mediates endothelial ICAM-1 expression and neutrophil adhesion. J Immunol. 1999;162:5466–5476. [PubMed] [Google Scholar]
- 71a.Report of the United Nations Scientific Committee on the Effects of Atomic Radiation. 2008. ISSN 0255-1372.
- 72.Rikitake Y. Kawashima S. Takeshita S. Yamashita T. Azumi H. Yasuhara M. Nishi H. Inoue N. Yokoyama M. Anti-oxidative properties of fluvastatin, an HMG-CoA reductase inhibitor, contribute to prevention of atherosclerosis in cholesterol-fed rabbits. Atherosclerosis. 2001;154:87–96. doi: 10.1016/s0021-9150(00)00468-8. [DOI] [PubMed] [Google Scholar]
- 73.Sabeti S. Exner M. Mlekusch W. Amighi J. Quehenberger P. Rumpold H. Maurer G. Minar E. Wagner O. Schillinger M. Prognostic impact of fibrinogen in carotid atherosclerosis: nonspecific indicator of inflammation or independent predictor of disease progression? Stroke. 2005;36:1400–1404. doi: 10.1161/01.STR.0000169931.96670.fc. [DOI] [PubMed] [Google Scholar]
- 74.Schimmerling W. Cucinotta FA. Dose and dose rate effectiveness of space radiation. Radiat Prot Dosimetry. 2006;122:349–353. doi: 10.1093/rpd/ncl464. [DOI] [PubMed] [Google Scholar]
- 75.Schultz-Hector S. Trott KR. Radiation-induced cardiovascular diseases: is the epidemiologic evidence compatible with the radiobiologic data? Int J Radiat Oncol Biol Phys. 2007;67:10–18. doi: 10.1016/j.ijrobp.2006.08.071. [DOI] [PubMed] [Google Scholar]
- 76.Seeger H. Mueck AO. Lippert TH. Fluvastatin increases prostacyclin and decreases endothelin production by human umbilical vein endothelial cells. Int J Clin Pharmacol Ther. 2000;38:270–272. doi: 10.5414/cpp38270. [DOI] [PubMed] [Google Scholar]
- 77.Shankariah V. Fernando L. Guren I. Curcumin content and heat effect on curcumin in turmeric samples. International Food Technologies Annual Meeting; New Orleans, LA. 2001. [Google Scholar]
- 78.Sharma A. Wong D. Weidlich G. Fogarty T. Jack A. Sumanaweera T. Maguire P. Noninvasive stereotactic radiosurgery (CyberHeart) for creation of ablation lesions in the atrium. Heart Rhythm. 2010;7:802–810. doi: 10.1016/j.hrthm.2010.02.010. [DOI] [PubMed] [Google Scholar]
- 79.Shimizu Y. Kodama K. Nishi N. Kasagi F. Suyama A. Soda M. Grant EJ. Sugiyama H. Sakata R. Moriwaki H. Hayashi M. Konda M. Shore RE. Radiation exposure and circulatory disease risk: Hiroshima and Nagasaki atomic bomb survivor data, 1950–2003. BMJ. 2010;340:b5349. doi: 10.1136/bmj.b5349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Soucy KG. Lim HK. Attarzadeh DO. Santhanam L. Kim JH. Bhunia AK. Sevinc B. Ryoo S. Vazquez ME. Nyhan D. Shoukas AA. Berkowitz DE. Dietary inhibition of xanthine oxidase attenuates radiation-induced endothelial dysfunction in rat aorta. J Appl Physiol. 2010;108:1250–1258. doi: 10.1152/japplphysiol.00946.2009. [DOI] [PubMed] [Google Scholar]
- 80a.Sources and Effects of Ionizing Radiation. UNSCEAR 2000 Reports. I and II. New York: UNSCEAR; 2000. [Google Scholar]
- 81.Stewart FA. Hoving S. Russell NS. Vascular damage as an underlying mechanism of cardiac and cerebral toxicity in irradiated cancer patients. Radiat Res. 2010;174:865–869. doi: 10.1667/RR1862.1. [DOI] [PubMed] [Google Scholar]
- 82.Stewart FA. Te Poele JA. Van der Wal AF. Oussoren YG. Van Kleef EM. Kuin A. Verheij M. Dewit LG. Radiation nephropathy—the link between functional damage and vascular mediated inflammatory and thrombotic changes. Acta Oncol. 2001;40:952–957. doi: 10.1080/02841860152708233. [DOI] [PubMed] [Google Scholar]
- 83.Stewart JR. Fajardo LF. Radiation-induced heart disease: an update. Prog Cardiovasc Dis. 1984;27:173–194. doi: 10.1016/0033-0620(84)90003-3. [DOI] [PubMed] [Google Scholar]
- 84.Stone HB. Moulder JE. Coleman CN. Ang KK. Anscher MS. Barcellos-Hoff MH. Dynan WS. Fike JR. Grdina DJ. Greenberger JS. Hauer-Jensen M. Hill RP. Kolesnick RN. Macvittie TJ. Marks C. McBride WH. Metting N. Pellmar T. Purucker M. Robbins ME. Schiestl RH. Seed TM. Tomaszewski JE. Travis EL. Wallner PE. Wolpert M. Zaharevitz D. Models for evaluating agents intended for the prophylaxis, mitigation and treatment of radiation injuries. Report of an NCI workshop, December 3–4, 2003. Radiat Res. 2004;162:711–728. doi: 10.1667/rr3276. [DOI] [PubMed] [Google Scholar]
- 85.Stoney CM. Davis MC. Matthews KA. Sex differences in physiological responses to stress and in coronary heart disease: a causal link? Psychophysiology. 1987;24:127–131. doi: 10.1111/j.1469-8986.1987.tb00264.x. [DOI] [PubMed] [Google Scholar]
- 86.Straume T. Rugel G. Marchetti AA. Ruhm W. Korschinek G. McAninch JE. Carroll K. Egbert S. Faestermann T. Knie K. Martinelli R. Wallner A. Wallner C. Measuring fast neutrons in Hiroshima at distances relevant to atomic-bomb survivors. Nature. 2003;424:539–542. doi: 10.1038/nature01815. [DOI] [PubMed] [Google Scholar]
- 87.Swerdlow AJ. Higgins CD. Smith P. Cunningham D. Hancock BW. Horwich A. Hoskin PJ. Lister A. Radford JA. Rohatiner AZ. Linch DC. Myocardial infarction mortality risk after treatment for Hodgkin disease: a collaborative British cohort study. J Natl Cancer Inst. 2007;99:206–214. doi: 10.1093/jnci/djk029. [DOI] [PubMed] [Google Scholar]
- 88.Taylor CW. McGale P. Povall JM. Thomas E. Kumar S. Dodwell D. Darby SC. Estimating cardiac exposure from breast cancer radiotherapy in clinical practice. Int J Radiat Oncol Biol Phys. 2009;73:1061–1068. doi: 10.1016/j.ijrobp.2008.05.066. [DOI] [PubMed] [Google Scholar]
- 89.Taylor CW. Nisbet A. McGale P. Darby SC. Cardiac exposures in breast cancer radiotherapy: 1950s-1990s. Int J Radiat Oncol Biol Phys. 2007;69:1484–1495. doi: 10.1016/j.ijrobp.2007.05.034. [DOI] [PubMed] [Google Scholar]
- 90.Tokatli F. Uzal C. Doganay L. Kocak Z. Kaya M. Ture M. Kurum T. Alkaya F. Karadag H. Kutlu K. The potential cardioprotective effects of amifostine in irradiated rats. Int J Radiat Oncol Biol Phys. 2004;58:1228–1234. doi: 10.1016/j.ijrobp.2003.09.071. [DOI] [PubMed] [Google Scholar]
- 91.Tsuchiya K. Kanematsu Y. Yoshizumi M. Ohnishi H. Kirima K. Izawa Y. Shikishima M. Ishida T. Kondo S. Kagami S. Takiguchi Y. Tamaki T. Nitrite is an alternative source of NO in vivo. Am J Physiol Heart Circ Physiol. 2005;288:H2163–H2170. doi: 10.1152/ajpheart.00525.2004. [DOI] [PubMed] [Google Scholar]
- 91a.Sources and Effects of Ionizing Radiation. UNSCEAR 2000 Report to the General Assembly, with Scientific Annexes. Volume I: Sources. New York: United Nations; 2000. United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) [Google Scholar]
- 92.Van der Meeren A. Vandamme M. Squiban C. Gaugler MH. Mouthon MA. Inflammatory reaction and changes in expression of coagulation proteins on lung endothelial cells after total-body irradiation in mice. Radiat Res. 2003;160:637–646. doi: 10.1667/rr3087. [DOI] [PubMed] [Google Scholar]
- 93.van der Pal HJ. van Dalen EC. Kremer LC. Bakker PJ. van Leeuwen FE. Risk of morbidity and mortality from cardiovascular disease following radiotherapy for childhood cancer: a systematic review. Cancer Treat Rev. 2005;31:173–185. doi: 10.1016/j.ctrv.2005.03.008. [DOI] [PubMed] [Google Scholar]
- 94.Vereycken-Holler V. Aigueperse J. Gaugler MH. Radiation effects on circulating and endothelial cell interactions studied by real-time videomicroscopy. Int J Radiat Biol. 2002;78:923–930. doi: 10.1080/09553000210152980. [DOI] [PubMed] [Google Scholar]
- 95.Villasana L. Rosenberg J. Raber J. Sex-dependent effects of 56Fe irradiation on contextual fear conditioning in C57BL/6J mice. Hippocampus. 2010;20:19–23. doi: 10.1002/hipo.20659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wang J. Zheng H. Ou X. Fink LM. Hauer-Jensen M. Deficiency of microvascular thrombomodulin and up-regulation of protease-activated receptor-1 in irradiated rat intestine: possible link between endothelial dysfunction and chronic radiation fibrosis. Am J Pathol. 2002;160:2063–2072. doi: 10.1016/S0002-9440(10)61156-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wolfrum S. Jensen KS. Liao JK. Endothelium-dependent effects of statins. Arterioscler Thromb Vasc Biol. 2003;23:729–736. doi: 10.1161/01.ATV.0000063385.12476.A7. [DOI] [PubMed] [Google Scholar]
- 98.Wondergem J. van der Laarse A. van Ravels FJ. van Wermeskerken AM. Verhoeve HR. de Graaf BW. Leer JW. In vitro assessment of cardiac performance after irradiation using an isolated working rat heart preparation. Int J Radiat Biol. 1991;59:1053–1068. doi: 10.1080/09553009114550931. [DOI] [PubMed] [Google Scholar]
- 99.Yamada M. Wong FL. Fujiwara S. Akahoshi M. Suzuki G. Noncancer disease incidence in atomic bomb survivors, 1958–1998. Radiat Res. 2004;161:622–632. doi: 10.1667/rr3183. [DOI] [PubMed] [Google Scholar]
- 100.Yang VC. Late structural changes in mouse coronary arteries after iron-particle irradiation of the orbital region. Radiat Res. 1993;134:390–393. [PubMed] [Google Scholar]
- 101.Yang VV. Ainsworth EJ. Late effects of heavy charged particles on the fine structure of the mouse coronary artery. Radiat Res. 1982;91:135–144. [PubMed] [Google Scholar]
- 102.Yarom R. Harper IS. Wynchank S. van Schalkwyk D. Madhoo J. Williams K. Salie R. Genade S. Lochner A. Effect of captopril on changes in rats' hearts induced by long-term irradiation. Radiat Res. 1993;133:187–197. [PubMed] [Google Scholar]
- 103.Yeung TK. Hopewell JW. Effects of single doses of radiation on cardiac function in the rat. Radiother Oncol. 1985;3:339–345. doi: 10.1016/s0167-8140(85)80047-5. [DOI] [PubMed] [Google Scholar]
- 104.Yeung TK. Lauk S. Simmonds RH. Hopewell JW. Trott KR. Morphological and functional changes in the rat heart after X irradiation: strain differences. Radiat Res. 1989;119:489–499. [PubMed] [Google Scholar]
- 105.Yochmowitz MG. Wood DH. Salmon YL. Seventeen-year mortality experience of proton radiation in Macaca mulatta. Radiat Res. 1985;102:14–34. [PubMed] [Google Scholar]