Abstract
While the role of neurocognitive impairment in predicting functional outcome in chronic schizophrenia is now widely accepted, the results that have examined this relationship in the early phase of psychosis are surprisingly rather mixed. The predictive role of cognitive impairment early in the illness is of particular interest because interventions during this initial period may help to prevent the development of chronic disability. In a University of California, Los Angeles (UCLA) longitudinal study, we assessed schizophrenia patients with a recent first episode of psychosis using a neurocognitive battery at an initial clinically stabilized outpatient point and then followed them during continuous treatment over the next 9 months. Three orthogonal cognitive factors were derived through principal components analysis: working memory, attention and early perceptual processing, and verbal memory and processing speed. All patients were provided a combination of maintenance antipsychotic medication, case management, group skills training, and family education in a UCLA research clinic. A modified version of the Social Adjustment Scale was used to assess work outcome. Multiple regression analyses indicate that the combination of the 3 neurocognitive factors predicts 52% of the variance in return to work or school by 9 months after outpatient clinical stabilization. These data strongly support the critical role of neurocognitive factors in recovery of work functioning after an onset of schizophrenia. Cognitive remediation and other interventions targeting these early cognitive deficits are of major importance to attempts to prevent chronic disability.
Keywords: first-episode schizophrenia, cognition, functional outcome, recovery, disability, cognitive remediation
Introduction
Cognitive deficits have become increasingly accepted in recent years as a core component of schizophrenia. This view is the result of a confluence of multiple sources of evidence. First, cognitive deficits have been detected very early in the course of schizophrenia and remain present across psychotic and remitted clinical states1–4 and across time.5–7 Second, similar but less severe cognitive deficits have been identified in populations at genetic risk for schizophrenia8 and those considered to be at risk due to presumably prodromal symptoms.9,10
A third reason for the recent prominence of cognitive deficits in schizophrenia is that it has become increasingly recognized that these factors are linked to everyday functioning level and thus have practical implications for treatment of the disorder. Influential reviews and a meta-analysis by Green and colleagues11–13 have demonstrated that, at least for chronic schizophrenia, several key individual cognitive deficits have small to medium correlations with functional outcome in schizophrenia, including immediate and secondary verbal memory, card sorting ability, and attention/vigilance. A recent meta-analysis concluded that the association between various neurocognitive domains and functional outcome in schizophrenia cuts across many cognitive domains, with estimated average correlations ranging from 0.16 to 0.39.14 In chronic schizophrenia, composite global cognitive variables and multivariate combinations of different cognitive variables sometimes account for as much as 40–50% of the variance in functional outcome.13,15,16
In contrast to this strong evidence for a connection between cognitive deficits and functional outcome in chronic schizophrenia, Allott and colleagues17 found in a recent systematic review that the evidence is mixed regarding whether cognition is predictive of functional outcome in the early phase of schizophrenia. Allott et al located 22 studies that examined the relationship between baseline cognitive performance and functional outcome at least 6 months later in the early phase of psychosis (less than 2 years after onset of psychosis). While 16 of 22 found at least 1 cognitive domain that significantly predicted functional outcome, each of the cognitive domains actually yielded more null relationships than significant relationships when tallied across studies.17 Furthermore, the studies varied greatly in their methodological sophistication. Thus, while the evidence for a predictive relationship between cognition and functional outcome is strong is chronic schizophrenia, surprisingly, it is not well established in first-episode or recent-onset schizophrenia.
The current article focuses on the relationship between neurocognitive performance and work outcome in a longitudinal study of recent-onset schizophrenia patients who were treated at the University of California, Los Angeles (UCLA) Aftercare Research Program. All patients entered the study within 2 years of the onset of their first psychotic episode and are typical of first-episode or recent-onset schizophrenia samples in prior studies.17 All participated in the same psychosocial treatments and started their outpatient treatment period on 1 of 2 common antipsychotic medications, allowing treatment to be relatively standardized across patients. A battery of neurocognitive tests covering several domains was administered at an outpatient stabilized baseline point, thereby limiting the influence of any acute symptoms on cognitive performance. Patients were then treated throughout the follow-through period while being assessed for clinical symptoms and functional outcome. We focus here on predictors of work outcome because our analyses have indicated that typical outpatient treatment of recent-onset schizophrenia tends to yield a high rate of remission of psychotic symptoms and a reasonably high rate of social functioning recovery, but work recovery is much more limited in the absence of specific interventions that target it.18,19 Thus, we sought to understand better the factors that may be limiting work recovery after an initial psychotic episode.
Methods
Participants
The participants were 47 schizophrenia patients with a recent first psychotic episode who were recruited from local inpatient and outpatient facilities in the Los Angeles area or were directly referred to the UCLA Aftercare Research Program, a research clinic for first-episode psychosis patients. The patients were participating in a longitudinal study called “Developmental Processes in the Early Course of Illness” in the 1995–2001 period, which was Sample 2 within the Developmental Processes in Schizophrenic Disorders project. All participants were presented with oral and written information about the research procedures involved in the study and provided informed consent using Institutional Review Board (IRB) approved forms. Inclusion criteria were (1) onset of a first psychotic episode within the last 2 years, (2) a diagnosis by Research Diagnostic Criteria20 of schizophrenia (n = 36) or schizoaffective disorder, depressed, mainly schizophrenic (n = 11), (3) age of 18–45 years, and (4) sufficient fluency in the English language to avoid invalidating research measures of thought and language processes. Using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) with the Present State Exam psychosis section as well as information from family members, hospital staff, and medical records, we determined that all participants also met criteria from the DSM-IV21 for schizophrenia (n = 33), schizoaffective disorder depressed type (n = 6), schizophreniform (n = 7), or psychotic disorder, not otherwise specified (n = 1). Exclusion criteria were (1) evidence of a neurological disorder, (2) evidence of significant and habitual drug abuse or alcoholism in the 6 months prior to hospitalization or of substance use that triggered the psychotic episode, or (3) premorbid mental retardation.
The demographic characteristics of the sample are presented in table 1. Our sample is quite typical of first-episode or recent-onset schizophrenia patients, with a mean age in the mid-20s and a mean educational level of about 1 year past high school. Our sample included substantial racial and ethnic diversity, which is typical of Los Angeles.
Table 1.
Demographic and Clinical Characteristics of Individuals with Recent-Onset Schizophrenia
| Mean | SD | Range | |
| Age | 24.9 | 5.3 | 18–40 |
| Education level (years) | 13.5 | 2.1 | 8–19 |
| Highest parental education | 14.5 | 3.9 | 0–22 |
| Duration of psychosis (months) | 7.3 | 6.9 | 0.5–23.5 |
| Brief Psychiatric Rating Scale at baselinea | |||
| Thinking disturbance | 1.51 | 0.71 | 1.00–3.67 |
| Withdrawal–retardation | 2.01 | 0.87 | 1.00–4.33 |
| Hostile–suspiciousness | 1.33 | 0.46 | 1.00–2.67 |
| Anxious depression | 1.90 | 0.82 | 1.00–4.00 |
| Frequencies | |||
| Gender | 34 male | 13 female | |
| Race | 25 White | 6 Asian | |
| 11 African-American | 5 Mixed | ||
| Ethnicity | 37 Non-Latino | 10 Latino | |
BPRS factor scores are means of items based on 4-factor solution of Overall et al50 Individual items were rated on a scale from 1 to 7.
Procedures
Participants typically entered the Aftercare Research Program shortly after hospital discharge, although a few had not required hospitalization and entered from an outpatient facility. The participants were treated with either oral risperidone (n = 41) or fluphenazine decanoate (n = 6), psychoeducation, individual case management, and group skills training at the UCLA Aftercare Research Program. The psychosocial treatments focused on clinical symptoms and interpersonal skills and did not involve specialized vocational rehabilitation procedures (eg, supported employment).
Participants were stabilized on their maintenance medication dosage over the first 2–3 months and then assessed at this stabilized baseline with a comprehensive battery of cognitive, psychophysiological, symptomatic, and everyday functioning measures. The mean daily dosage of risperidone at the time of baseline assessments was 4.3 mg/day (SD = 2.2). If antiparkinsonian medication was prescribed, it was discontinued, in all instances judged clinically feasible, 48 hours before major test batteries to avoid anticholinergic effects on cognitive measures.
Measures
Diagnosis and Psychiatric History.
The Structured Clinical Interview for DSM-IV with the Present State Examination psychosis section was completed at the initial contact and used with informant supplemental information to establish diagnosis for study entry. Our diagnosticians were rigorously trained to high interrater reliability standards and participated in an ongoing quality assurance program.22 A Psychiatric and Social History Schedule was also completed to provide information on demographics, symptom history, and premorbid adjustment.
Cognitive Measures.
A battery of cognitive measures was administered at baseline to assess several domains in which cognitive deficits are prominent in schizophrenia.
Accuracy of early perceptual processing was examined with a visual backward masking paradigm that involved identification of target letters followed by a mask at various interstimulus intervals.23 Accuracy at the 40-millisecond interval between target and mask was used as the individual differences variable because it yielded a reasonable range of scores within this sample.
Sustained attention during a perceptual load task was measured by a computerized version of the Degraded Stimulus Continuous Performance Test (CPT),24,25 a vigilance task in which participants are asked to detect each occurrence of a single blurred digit (“0”) in a quasirandom sequence of single blurred digits presented for 29 milliseconds each at a rate of 1/second for 8 minutes. The overall signal/noise discrimination index, d’, was used as the primary variable.
Sustained attention in a working memory context was measured by a computerized version of a memory-load CPT version, the 3-7 CPT.26,27 Subjects detected, within a series of single digits presented at 29 milliseconds each at a rate of 1/second for almost 11 minutes, occasions on which a “3” was followed by a “7.” The overall d’ level was the primary score.
Another measure of perceptual processes, the forced-choice span of apprehension (Span), emphasized speed of encoding and readout from initial covert scanning of a tachistoscopic visual array.28 The accuracy of detecting the “T” or “F” in a 12-letter array presented at 71 milliseconds in a computerized version29 was the primary variable.
The Trail Making Test, Part A (Trails A), was used to measure processing speed in a simple psychomotor task.30 Trail Making Test, Part B (Trails B), was included to index its additional demands for working memory for an alternating sequence of letters and digits. Completion time was the primary score.
Additional measures of working memory were drawn from the digit-span distractibility task.31 To index maintenance of auditory working memory, we used the proportion of digits correctly recalled in sequence in the short-digit series and in the long-digit series.
Finally, the California Verbal Learning Test (CVLT) was used to assess verbal learning and memory.32 Its measures of immediate recall (T score for total recall for trials 1–5) and recognition (z score) were included as primary variables.
Work Outcome.
The work section of the Social Adjustment Scale,33 as modified by our UCLA group for use with an interview format,34 was used to measure real-world work outcome. Information from patients and their relatives was used to complete these ratings. We completed sections on employment and education every 3 months during a 9-month follow-through period after baseline assessments, during ongoing treatment in the UCLA Aftercare Research Program. Our primary outcome was whether a participant returned to paid work or school during the 9-month follow-up period. We chose this dichotomous variable as our primary outcome due to its practical importance and its ease of measurement. Continuous variables such as the amount of earnings and the number of hours per week do not apply in a comparable way to paid work and schooling. Any number of paid work hours or classroom hours was considered a return to work or school.
Data Analyses
Preliminary analyses examined the distributions for each of the cognitive variables. Several distributions showed a substantial skew, so log transformations were used to create more normally distributed scores. Specifically, we used ln (number of letters identified in backward masking), ln (Trails A completion time), ln (Trails B completion time), ln (5 − (d’ for 3-7 CPT)), ln ((1/(number of digit span items recalled in short series)) + 1), and ln (1 − (CVLT recognition d’ standard score)).
To reduce the number of cognitive variables for prediction of work outcome, we completed principal component analyses with the 10 primary scores for the 47 recent-onset schizophrenia patients with these baseline measures. Three components with eigenvalues greater than or equal to 1.0 were submitted to orthogonal rotation. Factor scores were then derived for use in multivariate prediction analyses.
Our primary multivariate prediction analysis employed multivariate regression with the 3 cognitive factor scores as predictors and the return to work/school within 9 months after baseline as the criterion because this analysis allows derivation of the amount of variance accounted for by the predictors (R2). Because the criterion variable was dichotomous, we also cross-checked the result using logistic regression.
Results
Cognitive Performance
The cognitive performance of this recent-onset schizophrenia sample on the 10 primary scores is summarized in table 2. Principal components analysis (SAS FACTOR procedure) to reduce the number of predictor variables resulted in 3 factors with eigenvalues of 4.23, 1.32, and 1.00, which together accounted for 65% of the variance. Orthogonal and oblique (promax) rotations produced factor patterns that were very similar, so the orthogonal solution was used for computing factor scores for ease of interpretation of the later regression analyses.
Table 2.
Cognitive Performance of Recent-Onset Schizophrenia Participants at Outpatient Baseline Assessment (N = 47)
| Cognitive Variable | Mean | SD |
| Backward masking (number correctly identified at 40 msec ISI) | 4.53 | 1.92 |
| Degraded Stimulus CPT (d’) | 2.29 | 0.94 |
| 3-7 CPT (d’) | 3.76 | 0.59 |
| Span of apprehension (number correct for 12-letter arrays) | 52.5 | 5.6 |
| Trail Making Test, Part A (completion time in seconds) | 27.5 | 9.9 |
| Trail Making Test, Part B (completion time in seconds) | 76.8 | 58.8 |
| Digit span (proportion recalled short series) | 0.84 | 0.17 |
| Digit span (proportion recalled long series) | 0.74 | 0.19 |
| California Verbal Learning Test recall | ||
| T score for total recall, trials 1–5 | 28.8 | 18.3 |
| California Verbal Learning Test recognition | ||
| z score for recognition memory | −0.57 | 0.85 |
ISI, interstimulus interval.
The loadings of the 10 cognitive scores on the 3 factors are shown in table 3, with loadings >.50 italicized. High loadings on Factor 1 were from ability to recall longer digit strings, complete the alternating number/letter sequence of the Trails B more quickly, detect target sequences accurately in the memory-load CPT, and covertly scan and readout from initial temporary buffer memory more accurately (Span). Thus, we named this factor working memory to reflect the primary shared processes of these tasks. Factor 2 received high loadings from accurate identification of target letters at the short 40 milliseconds interstimulus interval (ISI) in backward masking, accurate tachistoscopic detection of blurred digits in the Degraded Stimulus CPT, and accurate target detection in the memory-load CPT. We labeled this factor attention and early perceptual processing to capture its core cognitive processes. Factor 3 obtained substantial loadings from the 2 verbal memory scores (recall and recognition) of the CVLT as well as from the psychomotor processing speed index from Trails A. The name verbal memory and processing speed is used to acknowledge that it contains elements of both cognitive components, which have sometimes been separated in studies that included a larger number of indices of each of these cognitive abilities.35 Factor scores for each participant were computed using the factor loadings for all 10 primary cognitive variables.
Table 3.
Loadings on Cognitive Factors Derived by Principal Components Analysis with Orthogonal Rotation
| Cognitive Variable | Factor 1: Working Memory | Factor 2: Attention and Early Perceptual Processing | Factor 3: Verbal Memory and Processing Speed |
| Digit span recall (short series) | 0.83 | 0.14 | 0.09 |
| Trails B completion time | 0.74 | −0.14 | 0.25 |
| Digit span recall (long series) | 0.60 | 0.42 | 0.21 |
| 3-7 CPT (d’) | 0.60 | 0.51 | 0.05 |
| Span of apprehension (12-letter) | 0.54 | 0.36 | 0.24 |
| Backward masking (40 msec ISI) | 0.03 | 0.78 | 0.16 |
| Degraded stimulus CPT (d’) | 0.26 | 0.75 | 0.22 |
| CVLT recognition | −0.10 | 0.34 | 0.83 |
| CVLT recall (trials 1–5) | 0.17 | 0.37 | 0.74 |
| Trails A completion time | 0.46 | −0.15 | 0.65 |
Note: For this table, all loadings are represented such that positive loadings indicate better scores on the item. Loadings >.50 are italicized for emphasis. ISI, interstimulus interval.
Prediction of Work Outcome
Thirty-five of 47 recent-onset schizophrenia patients remained at the UCLA Aftercare Research Program through at least 9 months of treatment after the baseline assessment and had work outcome data available. The most common reason for dropping out of treatment before that point was a lack of awareness that one had an illness that required ongoing treatment. Of the 35 with work outcome data, 23 had returned to some level of paid employment or schooling within the 9 months, while 12 had not.
Multivariate regression (SAS REG procedure) using the 3 cognitive factor scores as predictors and return to work/school within 9 months as the criterion yielded a highly significant model (F3, 31 = 11.06, P < .0001). The 3 cognitive factors at baseline predicted 52% of the variance in work outcome (R = .719), indicating that the level of these cognitive deficits at a stabilized outpatient baseline point was a very strong indicator of likelihood of return to work or school in this period. All 3 factors contributed to the overall prediction of work outcome, with working memory (t = 4.47, P < .0001) and verbal memory and processing speed (t = 3.11, P = .004) making significant contributions and Attention and Perceptual Processing showing a nonsignificant tendency to add predicted variance (t = 1.70, P = .099).
We cross-checked this predictive model using logistic regression (SAS LOGISTIC procedure), which is more robust for dichotomous variables. Again, the 3 cognitive factors predicted return to work or school within 9 months to a highly significant degree (likelihood ratio chi square [3 df] = 23.18, P < .0001).
Because many readers may wonder about the magnitude of correlations between individual cognitive scores and 9-month work outcome, we also examined these correlations as an additional exploratory analysis. Several individual cognitive variables were significantly related to whether these 35 recent-onset schizophrenia patients returned to work or school within 9 months, including shorter completion time for Trails A (r = −.64, P < .001), higher d’ for the memory-load CPT (3-7 CPT, r = .58, P < .001), more correct detections in the 12-letter Span (r = .57, P < .001), higher digit span recall proportion for the long series (r = .52, P < .001) and the short series (r = .47, P < .004), better CVLT total recall for trials 1–5 (r = .42, P < .02), shorter Trails B completion time (r = −.42, P < .02), better CVLT recognition (r = .41, P < .02), and higher d’ for the Degraded Stimulus CPT (r = .41, P < .02). The number of correct target identifications in backward masking was in the right direction but not significant (r = .22, P = .21). Thus, while the combination of the 3 factor scores predicted return to work/school better than any individual cognitive score, several cognitive scores were moderately to strongly predictive of this work outcome.
Discussion
Three cognitive factors measured at a stabilized baseline point predicted very well whether our participants with a recent first episode of schizophrenia returned to paid work or schooling within 9 months, accounting for 52% of the variance. These 3 factors reflect working memory, verbal memory and processing speed, and attention and early perceptual processing abilities, all of which typically show prominent deficits in the early course of schizophrenia.2,36 In addition to this very strong multivariate prediction of work outcome, several individual cognitive performance scores at baseline predicted work outcome with medium to large effect sizes, supporting the view that several cognitive deficits are good predictors of return to work or school following an initial psychotic episode.
The strength of the relationship between cognitive performance and functional outcome in this study is at the high end of those reported in some prior studies of patients with chronic schizophrenia.12,14–16 Thus, it appears that the mixed results of prior studies examining relationships between individual cognitive components and functional outcome in recent-onset schizophrenia17 might be attributable to particular features of individual studies rather than the early phase of schizophrenia per se. Features of the current study that may have aided detection of a strong relationship may include the relatively standardized treatment package offered to the participants, a reasonably wide range of cognitive predictors, and focus on an aspect of functional outcome (work/school functioning) that may particularly require neurocognitive abilities.
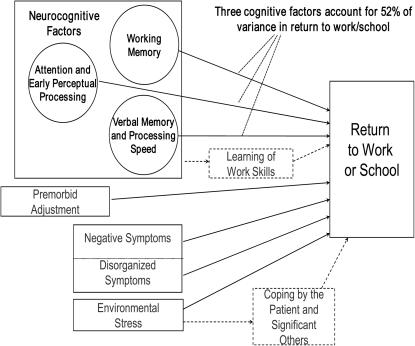
The current report focuses on cognitive factors in predicting work outcome in recent-onset schizophrenia, but the larger study from which these data are drawn examined several additional factors that are part of our conceptual model of influences on work functioning in recent-onset schizophrenia, as shown in figure 1. We readily acknowledge that non-cognitive factors also influence work functioning in schizophrenia and that neurocognitive factors may operate through various mediators, as has been explored in some recent studies of chronic schizophrenia.37,38 In addition, we did not include measures of social cognition in this study, which have been shown to have, if anything, stronger relationships to functional outcome than do neurocognitive measures14 and may partially mediate the relationships between neurocognition and functional outcome.39 Nevertheless, it is striking that neurocognitive measures predict return to work or school in the early phase of schizophrenia as well as shown here.
Fig. 1.
Conceptual Model Showing Possible Factors Influencing Work Functioning in Recent-Onset Schizophrenic Disorders.
Limitations of the current study include the relatively small sample size, the lack of a standardized cognitive battery widely used to assess cognition in schizophrenia, and the absence of social cognition and functional capacity measures. We reduced the number of primary cognitive predictor variables to 3 to avoid chance findings, but a larger sample would allow greater statistical power to examine a wider range of cognitive predictors. We agree with the conclusion of Allott et al17 that inclusion of a widely used standardized cognitive battery intended for schizophrenia, such as the MATRICS Consensus Cognitive Battery,40 would allow improved comparison of results across such studies, but these data were collected before that battery had been developed. Subsequent studies would also benefit by examining possible mediators such as social cognition and functional capacity to clarify pathways of effects between neurocognition and work recovery in early psychosis. Thus, it would be very useful to determine the extent to which better neurocognitive functioning in the early course of illness directly leads to return to work or whether it contributes to other skills (eg, social competence, adaptive competence) that are in turn needed for successful transition into real-world work functioning, as recent models of predictive relationships within chronic schizophrenia and bipolar disorder have suggested.37,39
The present results certainly support the importance of finding effective ways to improve cognitive deficits in schizophrenia, a treatment target that has been increasingly recognized.41–43 Cognitive deficits appear to be strong rate-limiting factors in recovery of everyday functioning in the early phase of schizophrenia as well as in chronic schizophrenia. Many approaches to cognitive training have focused on different components of neurocognition and offer promise for significantly improving these core deficits of schizophrenia.44,45 Meta-analyses and a recent randomized trial with chronic schizophrenia patients suggest that cognitive training has stronger effects on work functioning if combined with active vocational rehabilitation approaches, such as supported employment.44–46 Supported employment and supported education approaches have recently started to be successfully applied in the initial period of schizophrenia to substantially increase rates of return to work or school,47–49 so the combination of cognitive training and supported employment/education seems like a very promising next early intervention step. Longitudinal predictive relationships such as those demonstrated in this study clearly support the view that improvements in neurocognition should increase functional capacity and functional outcome. Application of cognitive training and other cognition-enhancing interventions during the early phase of schizophrenia may be particularly promising, particularly if combined with supported employment/education because it may allow work recovery to occur before chronic disability develops.
Funding
National Institute of Mental Health research grant (MH037705) and Center grants (MH030911, MH0 66286). Medication was provided by Bristol-Myers Squibb and Ortho-McNeil Janssen Scientific Affairs, LLC.
Acknowledgments
We are grateful to the patients and staff of the UCLA Aftercare Research Program for their support of this study.
References
- 1.Asarnow RF, MacCrimmon DJ. Residual performance deficit in clinically remitted schizophrenics: a marker of schizophrenia? J Abnorm Psychol. 1978;87:597–608. doi: 10.1037//0021-843x.87.6.597. [DOI] [PubMed] [Google Scholar]
- 2.Nuechterlein KH, Dawson ME. Information processing and attentional functioning in the developmental course of schizophrenic disorders. Schizophr Bull. 1984;10:160–203. doi: 10.1093/schbul/10.2.160. [DOI] [PubMed] [Google Scholar]
- 3.Nuechterlein KH, Dawson ME, Gitlin M, et al. Developmental processes in schizophrenic disorders: longitudinal studies of vulnerability and stress. Schizophr Bull. 1992;18:387–425. doi: 10.1093/schbul/18.3.387. [DOI] [PubMed] [Google Scholar]
- 4.Addington J, Addington D. Cognitive functioning in first-episode schizophrenia. J Psychiatry Neurosci. 2002;27:188–192. [PMC free article] [PubMed] [Google Scholar]
- 5.Censits DM, Ragland JD, Gur RC, Gur RE. Neuropsychological evidence supporting a neurodevelopmental model of schizophrenia: a longitudinal study. Schizophr Res. 1997;24:289–298. doi: 10.1016/s0920-9964(96)00091-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoff AL, Sakuma M, Wieneke M, Horon R, Kushner M, DeLisi LE. Longitudinal neuropsychological follow-up study of patients with first-episode schizophrenia. Am J Psychiatry. 1999;156:1336–1341. doi: 10.1176/ajp.156.9.1336. [DOI] [PubMed] [Google Scholar]
- 7.Nopoulos P, Flashman L, Flaum M, Arndt S, Andreasen N. Stability of cognitive functioning early in the course of schizophrenia. Schizophr Res. 1994;14:29–37. doi: 10.1016/0920-9964(94)90006-x. [DOI] [PubMed] [Google Scholar]
- 8.Snitz BE, MacDonald AW, III, Carter CS. Cognitive deficits in unaffected first-degree relatives of schizophrenia patients: a meta-analytic review of putative endophenotypes. Schizophr Bull. 2006;32:179–194. doi: 10.1093/schbul/sbi048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Niendam TA, Bearden CE, Johnson JK, et al. Neurocognitive performance and functional disability in the psychosis prodrome. Schizophr Res. 2006;84:100–111. doi: 10.1016/j.schres.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 10.Seidman LJ, Giuliano AJ, Meyer EC, et al. Neuropsychology of the prodrome to psychosis in the NAPLS consortium: relationship to family history and conversion to psychosis. Arch Gen Psychiatry. 2010;67:578–588. doi: 10.1001/archgenpsychiatry.2010.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Green MF. What are the functional consequences of neurocognitive deficits in schizophrenia? Am J Psychiatry. 1996;153:321–330. doi: 10.1176/ajp.153.3.321. [DOI] [PubMed] [Google Scholar]
- 12.Green MF, Kern RS, Braff DL, Mintz J. Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the “right stuff”? Schizophr Bull. 2000;26:119–136. doi: 10.1093/oxfordjournals.schbul.a033430. [DOI] [PubMed] [Google Scholar]
- 13.Green MF, Kern RS, Heaton RK. Longitudinal studies of cognition and functional outcome in schizophrenia: implications for MATRICS. Schizophr Res. 2004;72:41–51. doi: 10.1016/j.schres.2004.09.009. [DOI] [PubMed] [Google Scholar]
- 14.Fett AK, Viechtbauer W, Dominguez MD, Penn DL, van Os J, Krabbendam L. The relationship between neurocognition and social cognition with functional outcomes in schizophrenia: a meta-analysis. Neurosci Biobehav Rev. 2011;35:573–588. doi: 10.1016/j.neubiorev.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 15.Velligan DI, Mahurin RK, Diamond PL, Hazleton BC, Eckert SL, Miller AL. The functional significance of symptomatology and cognitive function in schizophrenia. Schizophr Res. 1997;25:21–31. doi: 10.1016/S0920-9964(97)00010-8. [DOI] [PubMed] [Google Scholar]
- 16.Harvey PD, Howanitz E, Parrella M, et al. Symptoms, cognitive functioning, and adaptive skills in geriatric patients with lifelong schizophrenia: a comparison across treatment sites. Am J Psychiatry. 1998;155:1080–1086. doi: 10.1176/ajp.155.8.1080. [DOI] [PubMed] [Google Scholar]
- 17.Allott K, Liu P, Proffitt TM, Killackey E. Cognition at illness onset as a predictor of later functional outcome in early psychosis: systematic review and methodological critique. Schizophr Res. 2011;125:221–235. doi: 10.1016/j.schres.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 18.Nuechterlein KH, Miklowitz DJ, Ventura J, Gitlin MJ, Stoddard M, Lukoff D. Classifying episodes in schizophrenia and bipolar disorder: criteria for relapse and remission applied to recent-onset samples. Psychiatry Res. 2006;144:153–166. doi: 10.1016/j.psychres.2004.04.018. [DOI] [PubMed] [Google Scholar]
- 19.Ventura J, Subotnik KL, Guzik LH, Hellemann GS, Gitlin MJ, Nuechterlein KH. Remission and recovery during the first outpatient year of the early course of schizophrenia. doi: 10.1016/j.schres.2011.06.025. Schizophr Res. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Spitzer RL, Endicott J, Robins E. Research diagnostic criteria: rationale and reliability. Arch Gen Psychiatry. 1978;35:773–782. doi: 10.1001/archpsyc.1978.01770300115013. [DOI] [PubMed] [Google Scholar]
- 21.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) 4th ed. Washington, DC: American Psychiatric Press; 1994. [Google Scholar]
- 22.Ventura J, Liberman RP, Green MF, Shaner A, Mintz J. Training and quality assurance with the Structured Clinical Interview for DSM-IV(SCID-I/P) Psychiatry Res. 1998;79:163–173. doi: 10.1016/s0165-1781(98)00038-9. [DOI] [PubMed] [Google Scholar]
- 23.Green MF, Nuechterlein KH, Mintz J. Backward masking in schizophrenia and mania, II: specifying the visual channels. Arch Gen Psychiatry. 1994;51:945–951. doi: 10.1001/archpsyc.1994.03950120017004. [DOI] [PubMed] [Google Scholar]
- 24.Nuechterlein, KH, Asarnow, RF. Degraded Stimulus Continuous Performance Test Program for PCs [computer program]. Version 7. Los Angeles, CA: University of California, Los Angeles; 1994. [Google Scholar]
- 25.Nuechterlein KH, Parasuraman R, Jiang Q. Visual sustained attention: image degradation produces rapid sensitivity decrement over time. Science. 1983;220:327–329. doi: 10.1126/science.6836276. [DOI] [PubMed] [Google Scholar]
- 26.Nuechterlein KH, Edell WS, Norris M, Dawson ME. Attentional vulnerability indicators, thought disorder, and negative symptoms. Schizophr Bull. 1986;12:408–426. doi: 10.1093/schbul/12.3.408. [DOI] [PubMed] [Google Scholar]
- 27.Nuechterlein, KH, Asarnow, RF. 3–7 Continuous Performance Test Program for PCs [computer program]. Version 2. Los Angeles, CA: University of California, Los Angeles; 1993. [Google Scholar]
- 28.Asarnow RF, Granholm EL, Sherman T. Span of apprehension in schizophrenia. In: Steinhauer SR, Gruzelier JH, Zubin J, editors. Neuropsychology, Psychophysiology and Information Processing. Vol. 5. Amsterdam, the Netherlands: Elsevier Science Publishers; 1991. pp. 335–370. [Google Scholar]
- 29.Asarnow, RF, Nuechterlein, KH. Forced-Choice Span of Apprehension Program for PCs [computer program]. Version 4.0. Los Angeles, CA: University of California, Los Angeles; 1994. [Google Scholar]
- 30.Reitan RM. Validity of the trail making test as an indicator of organic brain damage. Percept. Mot. Skills. 1958;8:271–276. [Google Scholar]
- 31.Oltmanns TF, Neale JM. Schizophrenic performance when distractors are present: attentional deficit or differential task difficulty? J Abnorm Psychol. 1975;84:205–209. doi: 10.1037/h0076721. [DOI] [PubMed] [Google Scholar]
- 32.Delis DC, Kramer JH, Kaplan E, Ober BA. California Verbal Learning Test. Research ed. Cleveland, OH: The Psychological Corporation; 1987. [Google Scholar]
- 33.Weissman MM, Bothwell S. Assessment of social adjustment by patient self-report. Arch Gen Psychiatry. 1976;33:1111–1115. doi: 10.1001/archpsyc.1976.01770090101010. [DOI] [PubMed] [Google Scholar]
- 34.Subotnik KL, Nuechterlein KH, Kelly KA, Kupic AL. Modified Version of Social Adjustment Scale-Work Outcome (by M. Weisman) Westwood, CA: University of California Los Angeles; 1997. [Google Scholar]
- 35.Nuechterlein KH, Barch DM, Gold JM, Goldberg TE, Green MF, Heaton RK. Identification of separable cognitive factors in schizophrenia. Schizophr Res. 2004;72:29–39. doi: 10.1016/j.schres.2004.09.007. [DOI] [PubMed] [Google Scholar]
- 36.Mesholam-Gately RI, Giuliano AJ, Goff KP, Faraone SV, Seidman LJ. Neurocognition in first-episode schizophrenia: a meta-analytic review. Neuropsychology. 2009;23:315–336. doi: 10.1037/a0014708. [DOI] [PubMed] [Google Scholar]
- 37.Bowie CR, Depp C, McGrath JA, et al. Prediction of real-world functional disability in chronic mental disorders: a comparison of schizophrenia and bipolar disorder. Am J Psychiatry. 2010;167:1116–1124. doi: 10.1176/appi.ajp.2010.09101406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ventura J, Hellemann GS, Thames AD, Koellner V, Nuechterlein KH. Symptoms as mediators of the relationship between neurocognition and functional outcome in schizophrenia: a meta-analysis. Schizophr Res. 2009;113:189--199 doi: 10.1016/j.schres.2009.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sergi MJ, Rassovsky Y, Nuechterlein KH, Green MF. Social perception as a mediator of the influence of early visual processing on functional status in schizophrenia. Am J Psychiatry. 2006;163:448–454. doi: 10.1176/appi.ajp.163.3.448. [DOI] [PubMed] [Google Scholar]
- 40.Nuechterlein KH, Green MF, Kern RS, et al. The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry. 2008;165:203–213. doi: 10.1176/appi.ajp.2007.07010042. [DOI] [PubMed] [Google Scholar]
- 41.Marder SR, Fenton W. Measurement and treatment research to improve cognition in schizophrenia: NIMH MATRICS initiative to support the development of agents for improving cognition in schizophrenia. Schizophr Res. 2004;72:5–9. doi: 10.1016/j.schres.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 42.Green MF, Nuechterlein KH, Gold JM, et al. Approaching a consensus cognitive battery for clinical trials in schizophrenia: the NIMH-MATRICS conference to select cognitive domains and test criteria. Biol Psychiatry. 2004;56:301–307. doi: 10.1016/j.biopsych.2004.06.023. [DOI] [PubMed] [Google Scholar]
- 43.Fenton WS, Stover EL, Insel TR. Breaking the log-jam in treatment development for cognition in schizophrenia: NIMH perspective. Psychopharmacology (Berl) 2003;169:365–366. doi: 10.1007/s00213-003-1564-1. [DOI] [PubMed] [Google Scholar]
- 44.McGurk SR, Twamley EW, Sitzer DI, McHugo GJ, Mueser KT. A meta-analysis of cognitive remediation in schizophrenia. Am J Psychiatry. 2007;164:1791–1802. doi: 10.1176/appi.ajp.2007.07060906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wykes T, Huddy V, Cellard C, McGurk SR, Czobor P. A meta-analysis of cognitive remediation for schizophrenia: methodology and effect sizes. Am J Psychiatry. 2011;168:472–485. doi: 10.1176/appi.ajp.2010.10060855. [DOI] [PubMed] [Google Scholar]
- 46.McGurk SR, Mueser KT, Derosa TJ, Wolfe R. Work, recovery, and comorbidity in schizophrenia: a randomized controlled trial of cognitive remediation. Schizophr Bull. 2009;35:319–335. doi: 10.1093/schbul/sbn182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rinaldi M, McNeil K, Firn M, Koletsi M, Perkins R, Singh SP. What are the benefits of evidence-based supported employment for patients with first-episode psychosis? Psychiatr Bull. 2004;28:281–284. [Google Scholar]
- 48.Killackey EJ, Jackson HJ, McGorry PD. Vocational intervention in first-episode psychosis: individual placement and support versus treatment as usual. Br J Psychiatry. 2008;193:114–120. doi: 10.1192/bjp.bp.107.043109. [DOI] [PubMed] [Google Scholar]
- 49.Nuechterlein KH, Subotnik KL, Turner LR, Ventura J, Becker DR, Drake RE. Individual placement and support for individuals with recent-onset schizophrenia: integrating supported education and supported employment. Psychiatr Rehabil J. 2008;31:340–349. doi: 10.2975/31.4.2008.340.349. [DOI] [PubMed] [Google Scholar]
- 50.Overall JE, Hollister LE, Pichot P. Major psychiatric disorder: a four-dimensional model. Arch Gen Psychiatry. 1967;16:146–151. doi: 10.1001/archpsyc.1967.01730200014003. [DOI] [PubMed] [Google Scholar]