Abstract
The proepicardium (PE) migrates over the heart and forms the epicardium. A subset of these PE-derived cells undergoes epithelial-mesenchymal transformation (EMT) and gives rise to cardiac fibroblasts and components of the coronary vasculature. We report that transforming growth factor-β (TGFβ) 1 and TGFβ2 increase EMT in PE explants as measured by invasion into a collagen gel, loss of cytokeratin expression, and redistribution of ZO1. The Type I TGFβ receptors ALK2 and ALK5 are both expressed in the PE. However, only constitutively active (ca) ALK2 stimulates PE-derived epithelial cell activation, the first step in transformation, while caALK5 stimulates neither activation nor transformation in PE explants. Overexpression of Smad6, an inhibitor of ALK2 signaling, inhibits epithelial cell activation, while BMP7, a known ligand for ALK2, has no effect. These data demonstrate that TGFβ stimulates transformation in the PE and suggest that ALK2 partially mediates this effect.
Keywords: chick, embryo, coronary vessel, transforming growth factor-β
INTRODUCTION
In higher vertebrates, coronary arteries supply blood to the thick muscular component of the heart where metabolic demand cannot be met by diffusion of oxygen from blood flowing through the interior of the heart. Although it was long believed that coronary vessels formed by angiogenesis as an outgrowth of the proximal aorta, it is now recognized that the coronary vessels initially arise by vasculogenesis (Mikawa and Fischman, 1992; Olivey et al., 2004). Thus vessels are well-formed before contacting and eventually penetrating the proximal aorta. Precursor cells that form the coronary vascular system are contained within the proepicardium (PE), a cluster of somatopleural cells that arise anterior of the liver primordium adjacent to the sinus venosus (Ho and Shimada, 1978; Viragh and Challice, 1981).
In the chick, the PE forms at stage 14 (Hamburger and Hamilton, 1951) and contacts the heart at stage 16 in the region of the atrioventricular (AV) sulcus. Cells derived from the PE migrate over the myocardium as an epithelial sheet, covering the AV groove first and the truncus arteriosus last to form the epicardium (Viragh et al., 1993). Although migration is not complete until stage 24, epicardial cells initiate epithelial-mesenchymal transformation (EMT) at stage 17, immediately after migration over the heart has begun (Mikawa and Fischman, 1992). EMT can be divided into three steps: activation, invasion, and migration (Ramsdell and Markwald, 1997 and references therein). During activation, cells lose contact with adjacent cells and elongate. Cells enter the underlying matrix in a step termed invasion. Finally, cells migrate through the extracellular matrix. Epicardially derived mesenchymal cells invade the myocardium and give rise to cardiac fibroblasts and vascular smooth muscle cells (Poelmann et al., 1993).
Reports from our laboratory and others have demonstrated a role for transforming growth factor-β (TGFβ)k signaling during EMT in the developing heart (Potts and Runyan, 1989; Potts et al., 1992; Brown et al., 1996; Brown et al., 1999; Desgrosellier et al., 2005). We chose therefore to examine the role of TGFβ signaling during EMT in the PE. TGFβ ligands bind to the type II TGFβ bbreceptor (TBRII), allowing TBRII to phosphorylate the type I receptor, Activin receptor-like kinase (ALK) 5. ALK5 then phosphorylates intracellular signaling molecules including members of the Smad family of transcription factors (Attisano and Wrana, 1998; Mulder, 2000). ALK5 activation signals TGFβ-dependent cell cycle arrest and induction of plasminogen activator inhibitor 1 (Bassing et al., 1994a) (Bassing et al., 1994b). TBRII can also interact with another type I receptor, ALK2 (Ebner et al., 1993). ALK2 has been implicated in mediating TGFβ̃-stimulated EMT in AV cushion endocardial cells (Lai et al., 2000; Desgrosellier et al., 2005) and cultured NMuMG breast cancer epithelial cells (Miettinen et al., 1994). These data suggest that TGFβ may signal by the activation of ALK5 or ALK2.
Here we demonstrate that addition of either TGFβ1 or TGFβ2 to PE explants increases the number of cells that enter a collagen matrix, a direct measure of EMT. Incubation of PE explants with TGFβ1 or TGFβ2 is also associated with decreased cytokeratin expression and a redistribution of the adherens junction protein ZO1, consistent with a loss of epithelial character. Both ALK5 and ALK2 are expressed in the PE prior to and after contact with the myocardium. Constitutively active (ca) ALK2 increases epithelial cell activation, while expression of caALK5 is without effect. Further we show that Smad6, an inhibitor of ALK2 signaling, is expressed in the PE and overexpression of Smad6 decreases activation of PE explant epithelial cells. Neither fibroblast growth factor (FGF) 1, FGF7 nor bone morphogenetic protein (BMP) 7 affected transformation in PE explants, suggesting that the effects seen with TGFβ were specific. These data demonstrate that TGFβ stimulates EMT in the PE and suggest that this effect may be partially mediated through ALK2 in a Smad-dependent manner.
RESULTS
To assess EMT in response to TGFβ we directly measured invasion of cells into a three dimensional collagen matrix (Hay, 1995; Markwald et al., 1996). This culture system has been most widely used to study endocardial cell EMT, and was originally shown to mimic closely the morphology and behavior of transformed mesenchymal cells in situ during endocardial cushion development (Bernanke and Markwald, 1982). Other investigators have successfully adapted this collagen culture system to examine EMT in PE explants (Mikawa and Gourdie, 1996; Dettman et al., 1998). Although the collagen matrix does not mimic the complex extracellular matrix environments found in either the endocardial cushions or the subepicardial matrix, this system does facilitate the study of the cellular behaviors that occur during EMT and has been effectively used to identify important signals that regulate EMT (Barnett and Desgrosellier, 2003; Olivey et al., 2004).
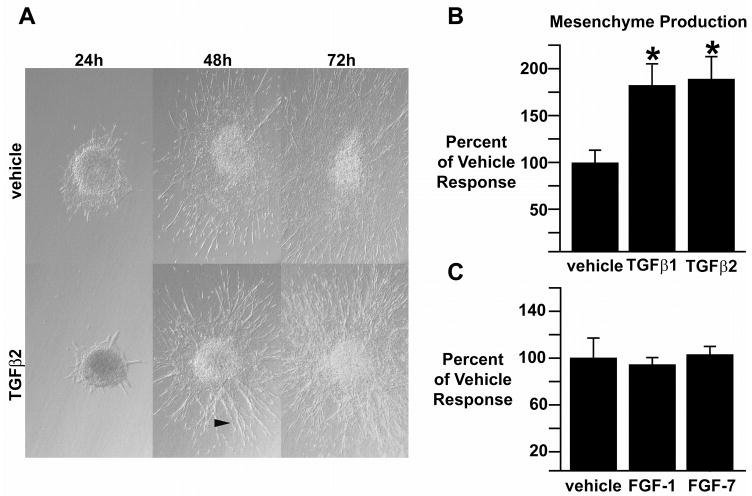
PE explants incubated on three-dimensional collagen gels with 200 pM TGFβ1 or TGFβ2 display a distinct phenotype by 48 hours in culture when compared to explants incubated with vehicle (Fig. 1A). At 48 hours, explants incubated with vehicle have an expansive epithelial sheet with elongate cells visible at the edges. In contrast, explants incubated with TGFβ1 or TGFβ2 had relatively small epithelial sheets, with an abundance of elongate, widely separated cells radiating from the explant. This difference between TGFβ1- or TGFβ2-incubated explants versus vehicle-incubated explants was still evident at 72 hours. The number of transformed cells, that is, cells found in the collagen gel, was determined for both TGFβ- and vehicle-incubated explants at 72 hours. Both TGFβ1- and TGFβ2-incubated explants exhibited a significant increase in transformed cells when compared to explants incubated with vehicle (TGFβ1: 188.8±10.1% of vehicle response, mean±sem; TGFβ2: 195.5±12.3%; n=21 TGFβ1 incubated explants, average 124 transformed cells per explant; n=19 TGFβ2 incubated explants, average 129 transformed cells per explant; n=22 vehicle incubated explants, average 66 transformed cells per explant; p<0.05, Student’s t-test, Fig. 1B).
Fig. 1.
Effect of TGFβ and FGF on PE explants. A, Representative TGFβ2- and vehicle-incubated PE explants photographed at 24, 48 and 72 hours after harvest. Explants incubated with 200 pM TGFβ2 contain more elongate cells radiating from the explant (arrowhead) as compared to vehicle incubated explants. B, The number of transformed cells was increased significantly in explants incubated with 200 pM of either TGFβ1 or TGFβ2, *=p<0.05. C, EMT in PE explants incubated with 10 ng/ml FGF1 or FGF7 did not differ from vehicle incubated explants. All photomicrographs at 100x.
Because FGF has been demonstrated to stimulate EMT in PE-derived epicardium (Morabito et al., 2001), we tested FGF1 and FGF7 and observed that neither stimulates cell transformation in PE explants, even when cultured for 96 hours in the presence of growth factor (FGF1: 94.0±6.2% of vehicle response, mean±sem; n=39 FGF1 incubated explants, n=38 vehicle incubated explants; FGF7: 103±7.2% of vehicle response, n=37 FGF7 incubated explants, n=41 vehicle incubated explants; p>0.05, Student’s t-test, Fig. 1C). The number of transformed cells in TGFβ2-incubated explants remained elevated at 96 hours when compared to vehicle-incubated explants (data not shown). Taken together, these data demonstrate a difference in growth factor-stimulated cell transformation between TGFβ and FGF.
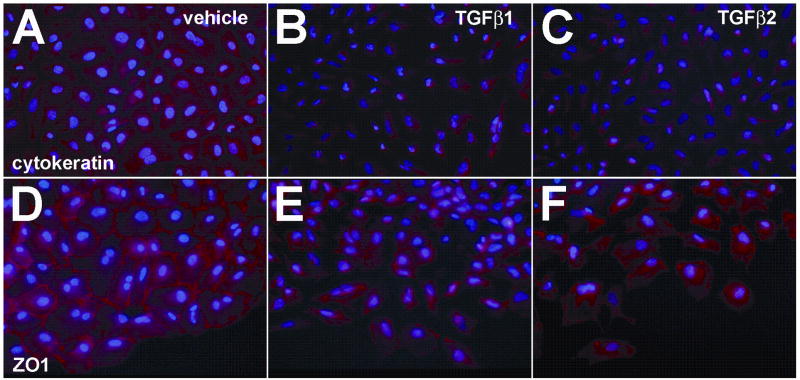
To support our morphological observations, we examined the expression and subcellular distribution of the epithelial markers cytokeratin and ZO1 in PE cells incubated on collagen-coated chamber slides. Others have reported that PE explants undergo the initial steps of EMT in two-dimensional culture systems (Lu et al., 2001). Consistent with these results, we observed that PE explants incubated with vehicle on collagen-coated slides form an expansive epithelial sheet composed of cells that express cytokeratin abundantly. These cells also express ZO1 at their periphery, demarcating cell-cell contact points between adjacent epithelial cells (Fig. 2A,D). In PEs incubated on collagen-coated chamber slides with either TGFβ1 or TGFβ2 we observed a decrease in cytokeratin staining (Fig. 2B,C) consistent with these cells undergoing the initial steps of EMT. The addition of either TGFβ1 or TGFβ2 altered the distribution of ZO1 in cells such that ZO1 immunoreactivity was less prominent at the cell periphery, consistent with a loss of cell-cell contacts and the transition from an epithelial to a mesenchymal cell phenotype (Fig. 2E, F).
Fig. 2.
TGFβ alters cytokeratin and ZO1 expression in PE cells. PEs were incubated with vehicle, TGFβ1 or TGFβ2 for 24 hours. After fixation, explants were processed to detect either cytokeratin or ZO1 expression by immunofluorescence. A, Epithelial cells in PEs incubated with vehicle express cytokeratin abundantly. B,C, Cells from explants incubated with 200 pM TGFβ1 (B) or TGFβ2 (C) separate from the epithelial sheet, become spindle shaped, and have decreased expression of cytokeratin. D, ZO1 immunoreactivity in vehicle incubated explants is located at the periphery of cells demarcating cell-cell contact points between adjacent epithelial cells. E,F, Cells from explants incubated with 200 pM TGFβ1 (E) or TGFβ2 (F) separated from the epithelial sheet and changed shape. ZO1 immunoreactivity was less prominent at the cell periphery, consistent with a loss of cell-cell contacts and the transition from an epithelial to a mesenchymal cell phenotype. All panels, 400x.
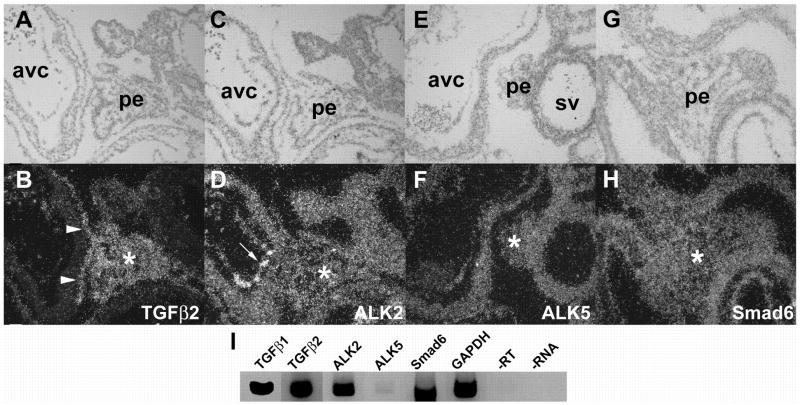
Having observed that TGFβ increases the number of transformed cells in PE explants, we hypothesized that this effect is mediated by activation of a Type I receptor. We performed in situ hybridization and RT-PCR to examine expression of ALK5 and ALK2 in the PE. As TGFβ2 expression has been documented in the mouse PE and epicardium (Molin et al., 2003), we used a TGFβ2 probe as a positive control for in situ hybridization experiments. Consistent with observations in the mouse, we observed abundant TGFβ2 expression in the chick PE from stage 14 until stage 18, demonstrating expression both prior to and after contact with the myocardium (Fig. 3A, B). ALK2 (Fig. 3C, D) and ALK5 (Fig. 3E, F) are uniformly expressed at low levels throughout the PE. We also noted that Smad6, a known regulator of ALK2 signaling, is expressed in the PE (Fig. 3G, H). RT-PCR analysis confirmed the presence of each of these transcripts in the PE (Fig. 3I). While Molin and colleagues did not observe TGFβ1 mRNA in the mouse PE using in situ hybridization (Molin et al., 2003), we did detect TGFβ1 in the chick PE via RT-PCR. This may reflect species differences in the expression pattern of this ligand, or may be due to the ability of RT-PCR to detect smaller quantities of transcript.
Fig. 3.
Expression of TGFβ2, ALK2, ALK5, and Smad6 in PE. Representative sections are depicted that reflect the pattern of expression from stages 14–20. A,B, At stage 18, TGFβ2 mRNA is seen in cells within the body of the PE (asterisks) and in cells forming the primitive epicardium (arrowheads). C, D, ALK2 mRNA in the PE (asterisk) and endocardium (arrow) at stage 18. E, F, ALK5 and G, H, Smad6 are both expressed in the PE (asterisks), shown here at stages 18 and 19, respectively. I, RT-PCR was performed as described in “Experimental Procedures” to amplify specific regions of the mRNAs encoding TGFβ1, TGFβ2, ALK2, ALK5, Smad6 and GAPDH from PEs. GAPDH was not appreciably amplified when the reaction was performed without reverse transcriptase enzyme (-RT) or template (-RNA). All photomicrographs at 200x. avc, atrioventricular cushion; pe, proepicardium; sv, sinus venosus
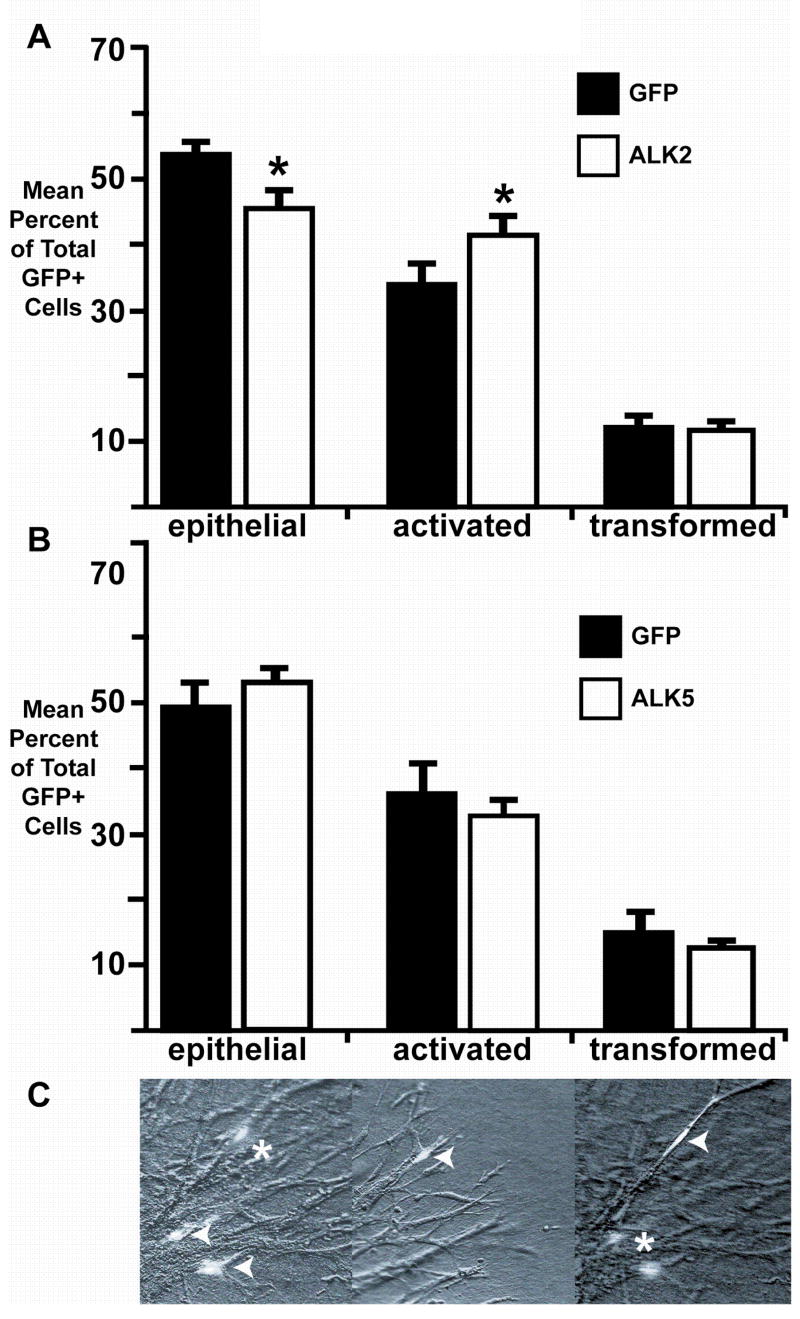
To determine whether ALK2 or ALK5 could mimic the effects of TGFβ, we infected PEs with an adenovirus encoding GFP and caALK5, GFP and caALK2 (Wieser et al., 1995; Ward et al., 2002), or GFP alone. After 72 hours in culture, GFP-expressing cells were classified by morphology and position in or on the collagen gel as epithelial, activated, or transformed (Fig. 4C). We defined epithelial cells as rounded cells having more than one direct contact with the epithelial sheet. Activated cells were defined as elongate cells on the surface of the collagen gel having no more than one contact with the epithelial sheet. Transformed cells were defined as elongate cells in the collagen gel that lack contact with the epithelial sheet. The number of GFP-positive cells in each group –epithelial, activated, and transformed – was divided by the total number of GFP-positive cells to determine the percentage of each.
Fig. 4.
ALK2, but not ALK5, promotes epithelial cell activation in PE explants. Explants were infected with adenovirus coding for GFP alone or GFP and constitutively active ALK2 (caALK2) or ALK5 (caALK5). A, Cells expressing caALK2 were less likely to be epithelial and more likely to be activated than cells expressing only GFP (*=p<0.05). B, The distribution of GFP-positive cells in PEs infected with caALK5 virus was similar to the distribution of GFP-positive cells in explants infected with virus encoding only GFP. C, GFP-positive cells were scored as epithelial (arrowheads, left panel), activated (arrowhead, middle panel), or transformed (arrowhead, right panel) as defined in “Results.” Asterisks indicate transformed (left panel) or epithelial (right panel) cells in different focal planes of the same field.
Cells infected with caALK2 adenovirus were significantly more likely to be activated (41.7±2.4% versus 34.0±2.6%, mean±sem; n=1097 caALK2/GFP infected cells in 34 explants examined, n=1039 GFP infected cells in 31 explants examined; p<0.05, Student’s t-test; Fig. 4A) and less likely to be epithelial (46.3±2.6% versus 54.0±1.7%; n=1341 caALK2/GFP infected cells in 34 explants examined, n=1112 GFP infected cells in 31 explants examined; p<0.05, Student’s t-test) than cells infected with adenovirus encoding GFP alone. We did not observe a difference in the percentage of transformed cells between explants infected with caALK2 adenovirus versus GFP adenovirus (12.1±0.7% versus 11.9±1.4%; n=334 caALK2/GFP infected cells in 34 explants examined, n=233 GFP infected cells in 31 explants examined; p>0.05, Student’s t-test). Infecting PE explants with caALK5 adenovirus did not alter the distribution of GFP-expressing epithelial, activated and transformed cells when compared to cells infected with adenovirus expressing only GFP (epithelial: 53.9±1.3% versus 49.6±3.3%, mean±sem; n=1560 caALK5/GFP infected cells, n=3585 GFP infected cells; activated: 33.0±1.8% versus 36.1±4.1%; n=939 caALK5/GFP infected cells, n=2879 GFP infected cells; transformed: 13.0±0.7% versus 14.4±2.6%, n=378 caALK5/GFP infected cells, n=1005 GFP infected cells; 29 GFP/ALK5 infected explants examined, 37 GFP infected explants examined; p>0.05 for all groups; Fig. 4B). These experiments demonstrate that ALK2 can mediate epithelial cell activation, the initial step in transformation, in PE explants, while ALK5 does not effect transformation in these cells.
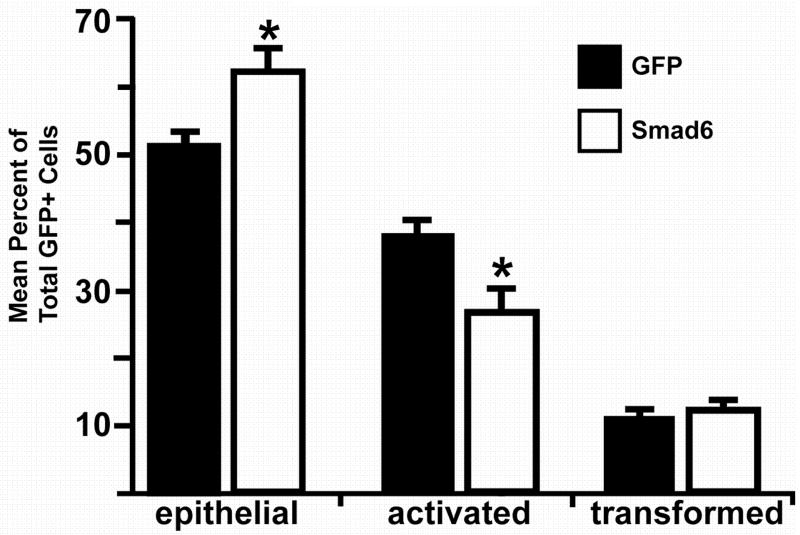
As a second approach to support our implication of ALK2 in mediating epithelial cell activation, we overexpressed Smad6, a molecule demonstrated to inhibit signaling downstream of ALK2, and scored for cell activation in PE explants. Smad6 inhibits ALK2 signaling through Smad1, but has no effect on signaling through Smads 2 and 3 that are downstream of ALK5 (Macias-Silva et al., 1996; Imamura et al., 1997; Liu et al., 1997; Hata et al., 1998; Macias-Silva et al., 1998). Explants were infected with adenovirus coding for chicken Smad6 (Yamada et al., 1999) and GFP or adenovirus coding for GFP alone. PE explant cells overexpressing Smad6 were more likely to be epithelial (61.7±2.6% versus 51.0±1.7%, mean±sem; n=2069 Smad6/GFP infected cells in 30 explants examined, n=5163 GFP infected cells in 32 explants examined; p<0.05, Student’s t-test; Fig. 5) and less likely to be activated (26.3±2.9% versus 37.8±1.3%; n=832 Smad6/GFP infected cells in 30 explants examined, n=3905 GFP infected cells in 32 explants examined; p<0.05, Student’s t-test) than were cells expressing only GFP, indicating that Smad6 inhibited epithelial cell activation. However, the percentage of transformed cells in explants infected with Smad6 adenovirus was similar to explants infected with GFP adenovirus (12.0±0.4% versus 11.2±0.9%, n=403 Smad6/GFP infected cells in 30 explants examined, n=1171 GFP infected cells in 32 explants examined; p>0.05, Student’s t-test). Inhibition of epithelial cell activation by Smad6 is confirmatory of our data that ALK2 increases epithelial cell activation.
Fig. 5.
Smad6 inhibits epithelial cell activation in PE explants. Explants were infected with adenovirus coding for GFP alone or GFP and Smad6. GFP-positive cells were scored as epithelial, activated, or transformed as described in “Results.” Cells overexpressing Smad6 were more likely to be epithelial and less likely to be activated than cells expressing GFP alone (*=p<0.05).
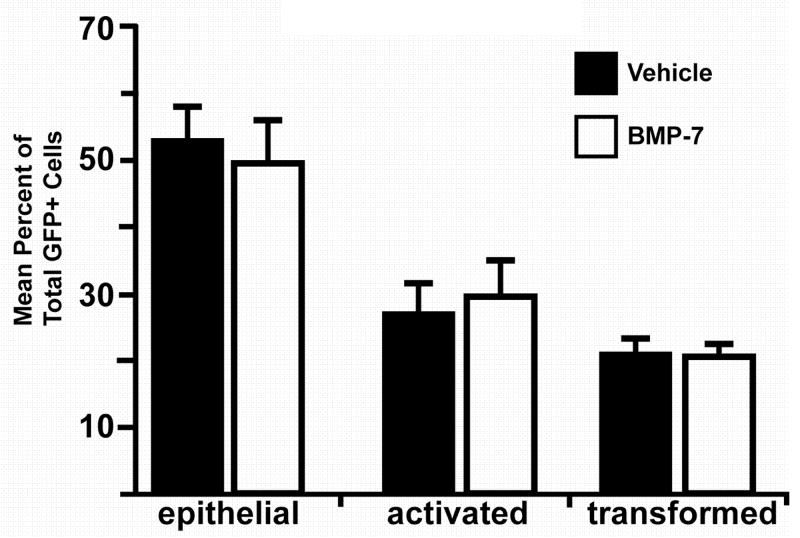
ALK2 has been reported to signal downstream of BMP7 (ten Dijke et al., 1994; Macias-Silva et al., 1998). We thus asked if addition of BMP7 to PE explants could stimulate epithelial cell activation or transformation. PE explants were first infected with adenovirus encoding GFP to facilitate the analysis of cell phenotype as epithelial, activated, or transformed. Explants were incubated with BMP7 or vehicle for 72 hours. Unlike results obtained in explants infected with virus encoding caALK2, the distribution of epithelial, activated, and transformed cells in explants incubated with BMP7 was not significantly different than the distribution in explants incubated with vehicle (epithelial: 49.6±6.2% versus 52.7±4.9%, n=477 BMP7-incubated cells, n=847 vehicle-incubated cells; activated: 29.6±5.6% versus 26.8±4.8%, n=365 BMP7-incubated cells, n=490 vehicle-incubated cells; transformed: 20.8±1.7% versus 20.5±2.9%, n=227 BMP7-incubated cells, n=358 vehicle-incubated cells; 22 BMP7 incubated explants and 24 vehicle incubated explants examined; p>0.05, Student’s t test; Fig. 6). These data suggest that ALK2-mediated epithelial cell activation is not downstream of BMP7 in the PE.
Fig. 6.
BMP7 does not increase epithelial cell activation. Explants were infected with adenovirus coding for GFP alone and incubated with 15 nM BMP7 or vehicle. After 72 hours in culture, GFP-positive cells were scored as epithelial, activated, or transformed as described in “Results.” The distribution of GFP-positive cells in PE explants incubated with BMP7 was similar to the distribution of GFP-positive cells in vehicle-incubated explants.
DISCUSSION
We report that TGFβ1 and TGFβ2, but neither FGF1 nor FGF7, stimulate EMT in PE explants. PEs incubated with either TGFβ1 or TGFβ2 display altered expression or distribution of both cytokeratin and ZO1, consistent with cells undergoing EMT. Constitutively active ALK2 promotes epithelial cell activation, the initial step in EMT, while caALK5 has no effect on either activation or transformation. Further, epithelial cell activation in explants is decreased by overexpressing Smad6, an inhibitor of Smad signaling downstream of ALK2. These data demonstrate that TGFβ stimulates PE cell EMT and that ALK2 mediates epithelial cell activation. BMP7, a known ligand for ALK2, does not affect EMT. Taken together, these data suggest that ALK2 signaling downstream of TGFβ may initiate EMT (Fig. 7).
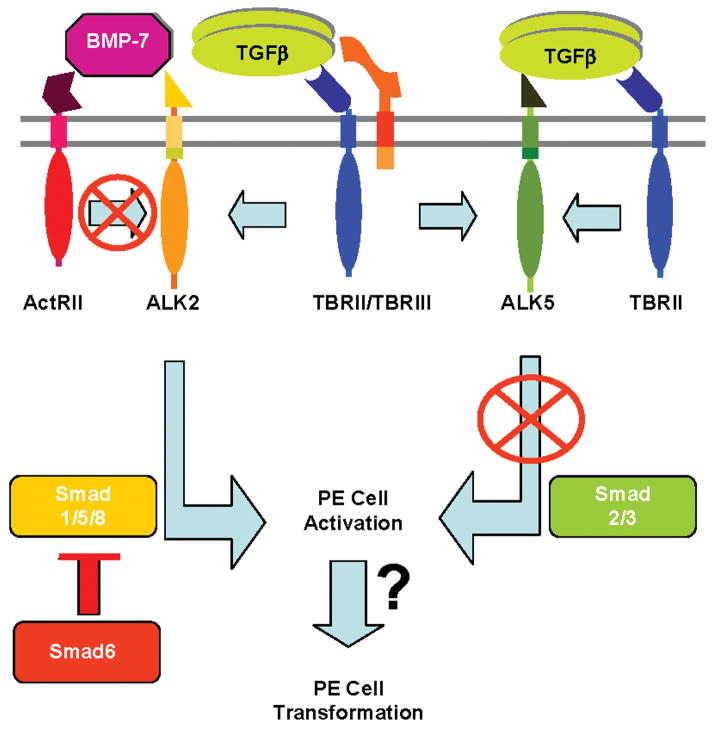
Fig. 7.
Model of TGFβ signaling during PE cell EMT. While TGFβ stimulates PE explant cells to undergo EMT, caALK5 is insufficient to recapitulate this response. caALK2 drives PE cells to undergo activation, the first step in EMT, while Smad6 inhibits epithelial cell activation. BMP-7 does not stimulate epithelial cell activation, suggesting ALK2-mediated activation is independent of BMP-7 signaling via the Type II activin receptor (ActRII). These data are consistent with a model where TGFβ signals via ALK2, and possibly a second unidentified pathway, to elicit EMT in PE explants. Data from the endocardial cushion model of EMT suggest that ALK2 signaling may also require TBRIII.
Our finding that TGFβ stimulates EMT in the PE is consistent with the well described role of TGFβ in stimulating EMT during embryonic development and tumorigenesis. Surprisingly, we found that caALK5, the canonical Type I TGFβ receptor, did not mimic the effects of TGFβ in PE cells. However, caALK2, a Type I receptor that can interact with the Type II TGFβ receptor, initiates cell activation, the first step in EMT. ALK2 is reported to play a similar role in the TGFβ-stimulated EMT of endothelial cells in the heart during early valvulogenesis. Experiments using explants of a valve-forming region of the heart, the AV cushion, demonstrated that neutralizing antisera to ALK2, but not ALK5, blocked EMT (Lai et al., 2000). Further, caALK2 introduced into normally nontransforming ventricular endocardial cells stimulated these cells to undergo EMT while caALK5 did not (Desgrosellier et al., 2005) Therefore, our experiments using PE explants are a second example of a TGFβ-stimulated EMT in the developing heart that is not mimicked by ALK5 signaling alone.
Additionally, we have implicated Smad6, an inhibitor of ALK2 signaling, in regulating EMT in PE explants. The finding that Smad6 specifically inhibits cells from undergoing activation complements and supports our data that caALK2 causes PE cell activation. Our observations that Smad6 is expressed in the PE and inhibits epithelial cell activation in PE explants is especially significant given the report that Smad6 null mice display abnormal coronary vessels (Galvin et al., 2000). In these animals, subepicardial vessels lack sufficient smooth muscle cells to maintain proper vascular wall integrity. It is unclear if this defect is caused by improperly regulated EMT or by deficient recruitment or differentiation of coronary vascular smooth muscle cell precursors. Our observation that Smad6 is expressed at the earliest stages of PE development suggests that loss of Smad6 may affect coronary vessel development at any stage, including formation of the PE, PE migration over the heart, EMT, or vessel assembly. Together, ALK2 and Smad6 may represent components of an important regulatory system that controls the number of PE-derived cells that can undergo activation and, ultimately, transformation, to supply the precursors for proper coronary vessel assembly.
Our data suggest that TGFβ, and not BMP7, may activate ALK2 in PE cells. In 14 days in ovo chick atrial myocytes TGFβ signals via ALK2 to decrease Gαi2 expression (Ward et al., 2002), whereas TGFβ signals primarily via ALK5 and decreases Gαi2 in 5 days in ovo cardiac myocytes. This dual regulation of cell responsiveness by a single growth factor, TGFβ, may be relevant to the report by Morabito and colleagues that TGFβ inhibits EMT in the PE-derived epicardium (Morabito et al., 2001), an effect completely opposite to what we observed in the cells of the PE itself. Our experiments were performed on PEs cultured prior to contact with the myocardium and formation of the epicardium, whereas Morabito and colleagues examined the effects of TGFβ in cultured epicardium. As was observed in the regulation of Gαi2 expression in 5 days in ovo versus 14 days in ovo cardiac myocytes, it is possible that TGFβ elicits different cellular effects in the PE versus the epicardium by activating different ALKs.
The failure of caALK2 to mimic fully the effects of TGFβ in mediating EMT may reflect a requirement for additional downstream signaling components. In endothelial cells, TGFβ requires both ALK5 and ALK1 signaling to regulate endothelial cell proliferation and migration. At lower concentrations, TGFβ stimulates endothelial cell proliferation and migration via ALK1 in an ALK5-dependent manner. As the TGFβ concentration is increased, TGFβ activates only ALK5-mediated pathways to inhibit endothelial cell proliferation and migration. Therefore, by activating ALK1 and ALK5, or ALK5 alone, TGFβ can both stimulate and inhibit endothelial cell proliferation and migration to balance angiogenesis (Goumans et al., 2002; Goumans et al., 2003).
In our studies, not all epithelial cells undergo activation in response to caALK2, nor are all cells inhibited from undergoing activation by Smad6 overexpression. This heterogeneous cell response could be explained by a number of different mechanisms. First, the PE is composed of precursors for at least three different cell types – epicardial cells, vascular smooth muscle cells, and cardiac fibroblasts. Since most proepicardially derived cells contribute to the epicardium and remain epithelial, it may be that only cells not yet committed to an epicardial fate are competent to initiate EMT. Thus cells in PE explants committed to become epicardium may be refractory to the manipulations described in our study. Furthermore, while both vascular cell precursors and cardiac fibroblast precursors undergo EMT, they may undergo EMT in response to different signals. Thus misexpression of caALK2 or Smad6 would reasonably only affect a subset of PE cells.
In summary, we demonstrate a role for TGFβ in promoting EMT of PE cells. We have implicated ALK2 and Smad6, but not ALK5, in cell activation, the first step in EMT. These data suggest that ALK2 may be a component of TGFβ signaling pathways that regulate EMT during organogenesis and tumorigenesis.
EXPERIMENTAL PROCEDURES
Immunohistochemistry, in situ hybridization, and RT-PCR
For immunofluorescence experiments, PEs were cultured in BioCoat Collagen I-coated 4-well chamber slides (BD Biosciences, Bedford, MA). For cytokeratin (Dako, Carpinteria, CA) staining, explants were fixed with 2% PFA for 30 min at room temperature and permeabilized with PBS plus 0.1% Triton X-100 for 5 min. For Zona Occludens-1 (ZO-1; Zymed, San Francisco, CA) staining, explants were fixed with ice-cold 70% methanol for 10 min. All explants were blocked in 2% BSA, PBS for 1 hour and incubated with dilute primary antibody (cytokeratin-1:100; ZO-1–2 μg/ml) overnight at 4ºC. Primary antibody was detected with goat anti-rabbit cy3 secondary antibody (1:800, Jackson ImmunoResearch, West Grove, PA). Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma, St. Louis, MO).
In situ hybridization was performed as described previously, with the substitution of [α-33P]UTP for [α-35S]UTP (Barnett et al., 1994). Riboprobes against chicken TGFβ2 were made as described (Barnett et al., 1994). ALK5 nucleotides (nt) 184 to 468, ALK2 nt 247 to 467 (Lai et al., 2000) and Smad6 nt 886 to 1233 (Yamada et al., 1999) were subcloned into pGEM-3Z (Invitrogen, Carlsbad, CA) such that generation of both antisense and sense riboprobes was under control of the T7 RNA polymerase promoter. Following hybridization, slides were coated with emulsion (Kodak NTB-2, Eastman Kodak Co., Rochester, NY) and exposed for two weeks. Sections were developed using Kodak Developer and Fixer and counterstained with hematoxylin. Light micrographs were captured on an Olympus Provis AX microscope using an Optronics digital camera.
Reverse transcriptase (RT-) PCR reactions were carried out on RNA isolated from PEs harvested from chicks between Hamburger and Hamilton stages 15 and 18. Tissue was flash-frozen before total RNA was harvested using the NucleoSpin RNA II kit (Clontech, Palo Alto, CA). Primers for the RT-PCR reaction were designed to amplify the following genes and regions: TGFβ1, nt 214–526 (5’-ATGGACCCGATGAGTATTGG-3’ and 5’-GACGATGAGTGGCTCTCCTT-3’) (Jakowlew et al., 1988); TGFβ2, nt 1151-1286 (5’-TCCAGTGGTGATGTGAAAGC-3’ and 5’-CTAGAGCACGCTTCTTCCGC-3’) (Jakowlew et al., 1990); ALK5, nt 601–1057 (5’-TACAATTTCCATGTGAAGAT-3’ and 5’-TCTGAAGGAACTACTTTGAA-3’) (Lai et al., 2000); ALK2, nt 181-635 (5’-AATGAATGTGTGTGTGAAGG-3’ and 5’-CTCGAGAATTGAGTCTCTCCATGT-3’) (Lai et al., 2000); Smad6, nt 886-1383 (5’-TGCTGCAATCCGTACCACTTCAGC and 5’-AAAGTCCTACAGTTGGGGAT-3’) (Yamada et al., 1999); GAPDH, nt 234-579 (5’-GGGCACGCCATCACTATCTTCC-3’ and 5’-GAGGGGCCATCCACCGTCTT-3’) (Dugaiczyk et al., 1983). RT-PCR amplifications were performed using the Titan One Tube RT-PCR System (Roche, Indianapolis, IN). The identity of each amplification product was verified by mobility on a 4% agarose gel and by restriction endonuclease digestion.
PE Explants incubated with TGFβ or FGF
Proepicardia were harvested from 3 day in ovo white leghorn chicken embryos (Charles River/SPAFAS, N. Franklin, CT) and placed onto 0.12% collagen gels as previously described (Brown et al., 1996). Explants were incubated with M199 (Cambrex Bio Science, Walkersville, MD) containing 1:400 antibiotic/antimycotic (Invitrogen, Carlsbad, CA) supplemented with 10% heat-inactivated fetal calf serum (Sigma, St. Louis, MO) and either 200 pM recombinant human (rh) TGFβ1, 200 pM rhTGFβ2, 10 ng/ml rhFGF1, 10 ng/ml rhFGF7 (R&D Systems, Minneapolis, MN) or vehicle (0.1% BSA in 4mM HCl for TGFβ, 0.1% BSA in PBS for FGF). Explants were cultured at 37°C in a 5% CO2 atmosphere for 72–96 hours and examined daily. At the end of the culture period, explants were fixed for 5 minutes with 2% paraformaldehyde in PBS at room temperature and washed extensively with PBS. Transformation was assayed directly by quantitating the number of cells that invaded the collagen matrix beneath each explant as previously described (Dettman et al., 1998). Briefly, each individual explant was optically sectioned using Hoffman optics by a naïve observer to determine unequivocally whether individual cells were on the surface of the collagen gel or beneath it. All elongate cells clearly beneath the surface of the collagen gel under each explant were scored as transformed. A subset of explants were overread by a second naïve observer to ensure consistency. The mean number of invasive cells per explant was determined and compared between TGFβ1- and vehicle-, TGFβ2-and vehicle-, FGF1- and vehicle-, or FGF7- and vehicle-incubated groups using Student’s t-test. Results from either three or four consecutive, independent experiments are shown for each treatment group.
PE Explants infected with adenovirus
Adenoviral constructs coding for GFP alone or GFP and caALK2, caALK5 (Ward et al., 2002) or Smad6 (Yamada et al., 1999) were made according to the method of He et al. (1998). Constitutive activity of proteins produced by each virus was tested using luciferase reporter plasmids that are activated by either ALK5 (3TP-lux) (Wrana et al., 1992) or ALK2 (pVent-luc) (Onichtchouk et al., 1999). Activity of Smad6 protein produced by viral infection was determined by its ability to decrease Smad1-dependent alkaline phosphatase activity in C3H10T1/2 cells (McDonnell et al., 2001). Data regarding activity of viral constructs have been reported (Desgrosellier et al., 2005).
Proepicardia were harvested as described above. Following harvest, explants were incubated in 50 μl of culture medium (M199 containing 10% FCS and 1:400 antibiotic/antimycotic) and infected with 2x106 to 2x107 pfu adenovirus coding for either GFP alone or both GFP and caALK2, caALK5, or Smad6. Following infection, explants were cultured as described above for 72 hours. At the end of the culture period, explants were fixed in PBS containing 0.05% glutaraldehyde and 0.8% formaldehyde for 5 minutes at room temperature and washed extensively in PBS.
Infected cells, identified by GFP expression, were counted using Hoffman optics on a Nikon Eclipse TE2000-E inverted fluorescent microscope and photographed with a QImaging digital camera. Cells were identified as epithelial, activated, or transformed based on morphology and position in the gel. Epithelial cells were defined as rounded cells having more than one direct contact with the epithelial sheet. Activated cells were defined as elongate cells on the surface with one or zero contacts with the epithelial sheet. Transformed cells were defined as elongate cells in the collagen gel that lack contact with the epithelial sheet. The percentage of GFP-expressing cells that were epithelial, activated or transformed was determined for each group (GFP alone, GFP-caALK2, GFP-caALK5, GFP-Smad6). Mean percentages from four consecutive, independent experiments were determined for each group and compared to GFP alone using Student’s t-test.
PE Explants Infected with GFP Adenovirus and Incubated with BMP7
Explants were infected with GFP adenovirus and subsequently incubated with 15 nM rhBMP7 (R&D Systems) or vehicle (0.1% BSA in 4mM HCl) for 72 hours. Explants were fixed and GFP-positive cells were scored as epithelial, activated or transformed as described. The distribution of epithelial, activated, and transformed cells was compared between vehicle- and BMP7-incubated explants and analyzed by the chi squared method. Results from four consecutive, independent experiments are shown.
Acknowledgments
We thank Dr. Charles Lin, Dr. Chris Brown and the members of the Barnett laboratory for their critical reading of the manuscript. We thank Jay Desgrosellier, Leigh Compton, Tiffany Davis, Dru Potash, Brian Culbreath, and Todd Townsend for reagents and assistance. Histological sections were generated by the Vanderbilt Vascular Histology Imaging Core facility. J.V.B. acknowledges the support of the Vanderbilt-Ingram Cancer Center.
Grant sponsor: NIH; Grant numbers: HL067105, HL52922, GM007628, HL076133.
Grant sponsor: March of Dimes. Grant numbers: FY98-446, FY99–563.
References
- Attisano L, Wrana JL. Mads and Smads in TGF beta signalling. Curr Opin Cell Biol. 1998;10:188–194. doi: 10.1016/s0955-0674(98)80141-5. [DOI] [PubMed] [Google Scholar]
- Barnett JV, Desgrosellier JS. Early events in valvulogenesis: a signaling perspective. Birth Defects Res C Embryo Today. 2003;69:58–72. doi: 10.1002/bdrc.10006. [DOI] [PubMed] [Google Scholar]
- Barnett JV, Moustakas A, Lin W, Wang XF, Lin HY, Galper JB, Maas RL. Cloning and developmental expression of the chick type II and type III TGF beta receptors. Dev Dyn. 1994;199:12–27. doi: 10.1002/aja.1001990103. [DOI] [PubMed] [Google Scholar]
- Bassing CH, Howe DJ, Segarini PR, Donahoe PK, Wang XF. A single heteromeric receptor complex is sufficient to mediate biological effects of transforming growth factor-beta ligands. J Biol Chem. 1994a;269:14861–14864. [PubMed] [Google Scholar]
- Bassing CH, Yingling JM, Howe DJ, Wang T, He WW, Gustafson ML, Shah P, Donahoe PK, Wang XF. A transforming growth factor beta type I receptor that signals to activate gene expression. Science. 1994b;263:87–89. doi: 10.1126/science.8272871. [DOI] [PubMed] [Google Scholar]
- Bernanke DH, Markwald RR. Migratory behavior of cardiac cushion tissue cells in a collagen-lattice culture system. Dev Biol. 1982;91:235–245. doi: 10.1016/0012-1606(82)90030-6. [DOI] [PubMed] [Google Scholar]
- Brown CB, Boyer AS, Runyan RB, Barnett JV. Antibodies to the Type II TGFbeta receptor block cell activation and migration during atrioventricular cushion transformation in the heart. Dev Biol. 1996;174:248–257. doi: 10.1006/dbio.1996.0070. [DOI] [PubMed] [Google Scholar]
- Brown CB, Boyer AS, Runyan RB, Barnett JV. Requirement of type III TGF-beta receptor for endocardial cell transformation in the heart. Science. 1999;283:2080–2082. doi: 10.1126/science.283.5410.2080. [DOI] [PubMed] [Google Scholar]
- Desgrosellier JS, Mundell NA, McDonnell MA, Moses HL, Barnett JV. Activin receptor-like kinase 2 and Smad6 regulate epithelial-mesenchymal transformation during cardiac valve formation. Dev Biol. 2005;280:201–210. doi: 10.1016/j.ydbio.2004.12.037. [DOI] [PubMed] [Google Scholar]
- Dettman RW, Denetclaw W, Jr, Ordahl CP, Bristow J. Common epicardial origin of coronary vascular smooth muscle, perivascular fibroblasts, and intermyocardial fibroblasts in the avian heart. Dev Biol. 1998;193:169–181. doi: 10.1006/dbio.1997.8801. [DOI] [PubMed] [Google Scholar]
- Dugaiczyk A, Haron JA, Stone EM, Dennison OE, Rothblum KN, Schwartz RJ. Cloning and sequencing of a deoxyribonucleic acid copy of glyceraldehyde-3-phosphate dehydrogenase messenger ribonucleic acid isolated from chicken muscle. Biochemistry. 1983;22:1605–1613. doi: 10.1021/bi00276a013. [DOI] [PubMed] [Google Scholar]
- Ebner R, Chen RH, Shum L, Lawler S, Zioncheck TF, Lee A, Lopez AR, Derynck R. Cloning of a type I TGF-beta receptor and its effect on TGF-beta binding to the type II receptor. Science. 1993;260:1344–1348. doi: 10.1126/science.8388127. [DOI] [PubMed] [Google Scholar]
- Galvin KM, Donovan MJ, Lynch CA, Meyer RI, Paul RJ, Lorenz JN, Fairchild-Huntress V, Dixon KL, Dunmore JH, Gimbrone MA, Jr, Falb D, Huszar D. A role for smad6 in development and homeostasis of the cardiovascular system. Nat Genet. 2000;24:171–174. doi: 10.1038/72835. [DOI] [PubMed] [Google Scholar]
- Goumans MJ, Valdimarsdottir G, Itoh S, Lebrin F, Larsson J, Mummery C, Karlsson S, ten Dijke P. Activin receptor-like kinase (ALK)1 is an antagonistic mediator of lateral TGFbeta/ALK5 signaling. Mol Cell. 2003;12:817–828. doi: 10.1016/s1097-2765(03)00386-1. [DOI] [PubMed] [Google Scholar]
- Goumans MJ, Valdimarsdottir G, Itoh S, Rosendahl A, Sideras P, ten Dijke P. Balancing the activation state of the endothelium via two distinct TGF-beta type I receptors. Embo J. 2002;21:1743–1753. doi: 10.1093/emboj/21.7.1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamburger V, Hamilton HL. A series of normal stages in the development of the chick embryo. J Morphol. 1951;88:49–92. [PubMed] [Google Scholar]
- Hata A, Lagna G, Massague J, Hemmati-Brivanlou A. Smad6 inhibits BMP/Smad1 signaling by specifically competing with the Smad4 tumor suppressor. Genes Dev. 1998;12:186–197. doi: 10.1101/gad.12.2.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay ED. An overview of epithelio-mesenchymal transformation. Acta Anat (Basel) 1995;154:8–20. doi: 10.1159/000147748. [DOI] [PubMed] [Google Scholar]
- He T-C, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci U S A. 1998;95:2509–2514. doi: 10.1073/pnas.95.5.2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho E, Shimada Y. Formation of the epicardium studied with the scanning electron microscope. Dev Biol. 1978;66:579–585. doi: 10.1016/0012-1606(78)90263-4. [DOI] [PubMed] [Google Scholar]
- Imamura T, Takase M, Nishihara A, Oeda E, Hanai J, Kawabata M, Miyazono K. Smad6 inhibits signalling by the TGF-beta superfamily. Nature. 1997;389:622–626. doi: 10.1038/39355. [DOI] [PubMed] [Google Scholar]
- Jakowlew SB, Dillard PJ, Sporn MB, Roberts AB. Complementary deoxyribonucleic acid cloning of a messenger ribonucleic acid encoding transforming growth factor beta 4 from chicken embryo chondrocytes. Mol Endocrinol. 1988;2:1186–1195. doi: 10.1210/mend-2-12-1186. [DOI] [PubMed] [Google Scholar]
- Jakowlew SB, Dillard PJ, Sporn MB, Roberts AB. Complementary deoxyribonucleic acid cloning of an mRNA encoding transforming growth factor-beta 2 from chicken embryo chondrocytes. Growth Factors. 1990;2:123–133. doi: 10.3109/08977199009071499. [DOI] [PubMed] [Google Scholar]
- Lai YT, Beason KB, Brames GP, Desgrosellier JS, Cleggett MC, Shaw MV, Brown CB, Barnett JV. Activin receptor-like kinase 2 can mediate atrioventricular cushion transformation. Dev Biol. 2000;222:1–11. doi: 10.1006/dbio.2000.9698. [DOI] [PubMed] [Google Scholar]
- Liu X, Sun Y, Constantinescu SN, Karam E, Weinberg RA, Lodish HF. Transforming growth factor beta-induced phosphorylation of Smad3 is required for growth inhibition and transcriptional induction in epithelial cells. Proc Natl Acad Sci U S A. 1997;94:10669–10674. doi: 10.1073/pnas.94.20.10669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu J, Landerholm TE, Wei JS, Dong XR, Wu SP, Liu X, Nagata K, Inagaki M, Majesky MW. Coronary smooth muscle differentiation from proepicardial cells requires rhoA-mediated actin reorganization and p160 rho-kinase activity. Dev Biol. 2001;240:404–418. doi: 10.1006/dbio.2001.0403. [DOI] [PubMed] [Google Scholar]
- Macias-Silva M, Abdollah S, Hoodless PA, Pirone R, Attisano L, Wrana JL. MADR2 is a substrate of the TGFbeta receptor and its phosphorylation is required for nuclear accumulation and signaling. Cell. 1996;87:1215–1224. doi: 10.1016/s0092-8674(00)81817-6. [DOI] [PubMed] [Google Scholar]
- Macias-Silva M, Hoodless PA, Tang SJ, Buchwald M, Wrana JL. Specific activation of Smad1 signaling pathways by the BMP7 type I receptor, ALK2. J Biol Chem. 1998;273:25628–25636. doi: 10.1074/jbc.273.40.25628. [DOI] [PubMed] [Google Scholar]
- Markwald R, Eisenberg C, Eisenberg L, Trusk T, Sugi Y. Epithelial-mesenchymal transformations in early avian heart development. Acta Anat (Basel) 1996;156:173–186. doi: 10.1159/000147845. [DOI] [PubMed] [Google Scholar]
- McDonnell MA, Law BK, Serra R, Moses HL. Antagonistic Effects of TGF[beta]1 and BMP-6 on Skin Keratinocyte Differentiation. Experimental Cell Research. 2001;263:265–273. doi: 10.1006/excr.2000.5117. [DOI] [PubMed] [Google Scholar]
- Miettinen PJ, Ebner R, Lopez AR, Derynck R. TGF-beta induced transdifferentiation of mammary epithelial cells to mesenchymal cells: involvement of type I receptors. J Cell Biol. 1994;127:2021–2036. doi: 10.1083/jcb.127.6.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikawa T, Fischman DA. Retroviral analysis of cardiac morphogenesis: discontinuous formation of coronary vessels. Proc Natl Acad Sci U S A. 1992;89:9504–9508. doi: 10.1073/pnas.89.20.9504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikawa T, Gourdie RG. Pericardial mesoderm generates a population of coronary smooth muscle cells migrating into the heart along with ingrowth of the epicardial organ. Dev Biol. 1996;174:221–232. doi: 10.1006/dbio.1996.0068. [DOI] [PubMed] [Google Scholar]
- Molin DG, Bartram U, Van der Heiden K, Van Iperen L, Speer CP, Hierck BP, Poelmann RE, Gittenberger-de-Groot AC. Expression patterns of Tgfbeta1-3 associate with myocardialisation of the outflow tract and the development of the epicardium and the fibrous heart skeleton. Dev Dyn. 2003;227:431–444. doi: 10.1002/dvdy.10314. [DOI] [PubMed] [Google Scholar]
- Morabito CJ, Dettman RW, Kattan J, Collier JM, Bristow J. Positive and negative regulation of epicardial-mesenchymal transformation during avian heart development. Dev Biol. 2001;234:204–215. doi: 10.1006/dbio.2001.0254. [DOI] [PubMed] [Google Scholar]
- Mulder KM. Role of Ras and Mapks in TGFbeta signaling. Cytokine Growth Factor Rev. 2000;11:23–35. doi: 10.1016/s1359-6101(99)00026-x. [DOI] [PubMed] [Google Scholar]
- Olivey HE, Compton LA, Barnett JV. Coronary vessel development: The epicardium delivers. Trends Cardiovasc Med. 2004;14:247–251. doi: 10.1016/j.tcm.2004.07.001. [DOI] [PubMed] [Google Scholar]
- Onichtchouk D, Chen YG, Dosch R, Gawantka V, Delius H, Massague J, Niehrs C. Silencing of TGF-beta signalling by the pseudoreceptor BAMBI. Nature. 1999;401:480–485. doi: 10.1038/46794. [DOI] [PubMed] [Google Scholar]
- Poelmann RE, Gittenberger-de Groot AC, Mentink MM, Bokenkamp R, Hogers B. Development of the cardiac coronary vascular endothelium, studied with antiendothelial antibodies, in chicken-quail chimeras. Circ Res. 1993;73:559–568. doi: 10.1161/01.res.73.3.559. [DOI] [PubMed] [Google Scholar]
- Potts JD, Runyan RB. Epithelial-mesenchymal cell transformation in the embryonic heart can be mediated, in part, by transforming growth factor beta. Dev Biol. 1989;134:392–401. doi: 10.1016/0012-1606(89)90111-5. [DOI] [PubMed] [Google Scholar]
- Potts JD, Vincent EB, Runyan RB, Weeks DL. Sense and antisense TGF beta 3 mRNA levels correlate with cardiac valve induction. Dev Dyn. 1992;193:340–345. doi: 10.1002/aja.1001930407. [DOI] [PubMed] [Google Scholar]
- Ramsdell AF, Markwald RR. Induction of endocardial cushion tissue in the avian heart is regulated, in part, by TGFbeta-3-mediated autocrine signaling. Dev Biol. 1997;188:64–74. doi: 10.1006/dbio.1997.8637. [DOI] [PubMed] [Google Scholar]
- ten Dijke P, Yamashita H, Sampath TK, Reddi AH, Estevez M, Riddle DL, Ichijo H, Heldin CH, Miyazono K. Identification of type I receptors for osteogenic protein-1 and bone morphogenetic protein-4. J Biol Chem. 1994;269:16985–16988. [PubMed] [Google Scholar]
- Viragh S, Challice CE. The origin of the epicardium and the embryonic myocardial circulation in the mouse. Anat Rec. 1981;201:157–168. doi: 10.1002/ar.1092010117. [DOI] [PubMed] [Google Scholar]
- Viragh S, Gittenberger-de Groot AC, Poelmann RE, Kalman F. Early development of quail heart epicardium and associated vascular and glandular structures. Anat Embryol (Berl) 1993;188:381–393. doi: 10.1007/BF00185947. [DOI] [PubMed] [Google Scholar]
- Ward SM, Desgrosellier JS, Zhuang X, Barnett JV, Galper JB. Transforming growth factor beta (TGFbeta ) signaling via differential activation of activin receptor-like kinases 2 and 5 during cardiac development. Role in regulating parasympathetic responsiveness. J Biol Chem. 2002;277:50183–50189. doi: 10.1074/jbc.M209668200. [DOI] [PubMed] [Google Scholar]
- Wieser R, Wrana JL, Massague J. GS domain mutations that constitutively activate T beta R-I, the downstream signaling component in the TGF-beta receptor complex. Embo J. 1995;14:2199–2208. doi: 10.1002/j.1460-2075.1995.tb07214.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrana JL, Attisano L, Carcamo J, Zentella A, Doody J, Laiho M, Wang XF, Massague J. TGF beta signals through a heteromeric protein kinase receptor complex. Cell. 1992;71:1003–1014. doi: 10.1016/0092-8674(92)90395-s. [DOI] [PubMed] [Google Scholar]
- Yamada M, Szendro PI, Prokscha A, Schwartz RJ, Eichele G. Evidence for a role of Smad6 in chick cardiac development. Dev Biol. 1999;215:48–61. doi: 10.1006/dbio.1999.9419. [DOI] [PubMed] [Google Scholar]