Abstract
Aim
To determine the prevalence, number, and location of multiple (≥2) T2-hyperintensities on brain magnetic resonance imaging (MRI) in children with neurofibromatosis type 1 (NF1) and their correlation with age, and to establish their sensitivity, specificity, and accuracy for the diagnosis of NF1 in children, especially in the early age (2-7 years).
Methods
We performed a cross-sectional study of 162 patients with NF1 from Croatian Neurofibromatosis Association Database and 163 control children between the ages of 2 and 18 years who underwent brain MRI between 1989 and 2009.
Results
Multiple T2-hyperintensities were present in 74% of NF1 patients and 1.8% of controls. They were mainly located in the basal ganglia, brainstem, and cerebellum and were significantly decreased in prevalence and number in the older age. T2-hyperintensities had excellent diagnostic accuracy with the area under the receiver operating characteristic (ROC) curve of 0.849 and 95% confidence interval (CI) of 0.805-0.886. The diagnostic sensitivity, specificity, and accuracy rate of T2-hyperintensities for NF1 were highest in the youngest age (2-7 years): 81% (95% CI 71%-89.1%), 99% (95% CI 92.3%-100%), and 85.8 (95% CI 83.3-93.8), respectively.
Conclusion
This study strongly suggests the inclusion of T2-hyperintensities on brain MRI on the list of diagnostic criteria for NF1, especially in children of early age, when the clinical penetration of the NF1 gene has not yet been completely finished.
Neurofibromatosis type 1 (NF1) or von Recklinghausen's disease is one of the most common autosomal dominant inherited diseases in humans with an estimated birth incidence of 1/2500 and a disease prevalence of 1/3000-4000 (1). NF1 gene is located on the pericentromeric region of chromosome 17q11.2 (2). It is a megagene, spaning 350 kb of genomic DNA and consisting of 60 exons encoding an 11-13 kb GTPase activating protein – neurofibromin of 2818 amino acids (3,4). The mutation rate for NF1 gene is high. A half of all NF1 cases are familial, while the other half is caused by a new mutation (5). Genetic molecular testing confirms the disease’s existence but has no predictive value for its severity and course.
In the past thirty years, the diagnosis of NF1 was made using a set of clinical criteria developed by the National Institutes of Health Consensus Conference, so called NIH diagnostic criteria (6). The diagnosis of NF1 is based on the presence of two or more of the following: 1) six or more café au lait macules, the greatest diameter of which is more than 0.5 cm in prepubertal patients and more than 1.5 cm in postpubertal patients; 2) two or more neurofibromas of any type, or one plexiform neurofibroma; 3) freckling in the axillary or inguinal region; 4) optic glioma; 5) two or more Lisch nodules; 6) a distinctive osseous lesion such as sphenoid dysplasia or pseudarthrosis; 7) a first-degree relative with NF1 according to the preceding criteria. However, the diagnosis cannot always be made in all children using the above mentioned criteria, especially in early childhood when the penetration of NF1 gene in usually not complete (7,8).
The cranial magnetic resonance imaging (MRI) is the best method for showing many features of NF1, including optic pathway gliomas, brain tumors of various locations, brain stem tumors, and orbital neurofibromas. Most frequent brain changes in children with NF1 are areas of increased T2-weighted signal intensity – T2-hyperintensities or “unidentified bright objects,” as they usually cannot be visualized using T1-weighted imaging. T2-hyperintensities are age-related findings on MRI and have been observed in 43%-93% of children who suffer from NF1 (9-11). These lesions do not exert mass effect, contrast enhancement, or surrounding edema. They are most commonly found in the basal ganglia, thalamus, cerebellum, and brainstem (12). Former research on multiple T2-hyperintensities on brain MRI in children as diagnostic criterion for NF1 was contradictory and scarce (13-17).
The aim of this cross-sectional study was to determine the prevalence, number, and location of multiple (≥2) brain T2-hyperintensities on MRI, and their correlation with age in children with NF1. A further aim was to determine the indicators of diagnostic accuracy of T2-hyperintensities in children of different ages, especially in the early age (from 2 to 7 years) when the NF1 gene penetration is still not completed.
Patients and methods
Study design, setting, data collection, and patients
This cross-sectional study with prospective data collection was performed at Sabol Outpatient Clinic for Sick Children in Zagreb, Croatia, from March to November 2010. It was approved by the Ethics Committee of Zagreb University School of Medicine.
The study included 325 patients of both sexes between 2 and 18 years old, divided into two groups. The NF1 patient group consisted of 162 children who were diagnosed with NF1 by the NIH diagnostic criteria (6) and underwent cranial MRI examinations. They were selected from the Croatian Neurofibromatosis Association Database, created by the main investigator on the basis of routine clinical follow-up of NF1 patients between January 1989 and December 2009 at the Department of Pediatrics, University Hospital Zagreb, Clinical Medical Center, and Sabol Outpatient Clinic for Sick Children in Zagreb. For each patient, data were available about NIH diagnostic criteria, presence or absence of T2-hyperintensities on cranial MRI examination, their number and location, as well as data on other changes and complications of the disease. Although some of the patients had more than one MRI scanning, cross-sectional analysis included only the data about clinical NIH diagnostic manifestations of NF1 and MRI changes at the time of the first MRI.
The control group included 163 patients selected by accidental sampling from patient database of the above mentioned institutions (3542 patients) during the same period as the patient group who performed the brain MRI examination for other complaints not related to NF1. The exclusion criteria for control group were diseases in which T2-hyperintensities were expected: hypoxic-ischemic encephalopathy, intracranial hemorrhage, infection, changes caused by radio- and/or chemotherapy, neurometabolic diseases, brain heredodegenerative diseases, tumors, multiple sclerosis, and acute disseminated encephalomyelitis. NF1 and other neurocutaneous diseases were excluded by careful clinical examination. All patients of both groups were clinically examined and prospectively evaluated by the head investigator.
Brain MRI
The examinations of participants of both groups were done with the MRI scanners operated at different magnetic field strength in different medical institutions in Croatia. The brain MR images were performed with a low- and medium-field systems (0.2-1.0 Tesla) on 90 children from NF1 patient group and 79 children from the control group, and with high-field MRI systems (1.5 -3 Tesla) on 72 children from the NF1 patient group and 84 children from the control group. Standard spin-echo T1- and T2-weighted sequences in the axial and sagittal planes, respectively, and axial fluid-attenuated inversion recovery T1- and T2-weighted sequences were obtained in all patients. Additional T1- or T2-weighted coronal images were obtained in most patients. A contrast agent (gadopentetate dimeglumine) was administered at 0.1 mmol/kg to 91 of 162 (56.2%) children from NF1 group and to 2 of 163 (1.2%) children from the control group. No adverse events were noticed in either of the groups.
All MR scans were examined independently by two neuroradiologists, who used visual inspection and were blinded for each group of patients. T2-hyperintensities on MRI were defined operationally as areas of confluent hyperintensity (signal intensity higher than that of cortical gray matter) on the proton density and the T2-weighted images without associated mass effect. A specific attention was paid to the number of lesions, location, signal characteristics on T1- and T2-weighted images, and presence or absence of contrast enhancement. In case of incompatible findings, the consensus between two neurologists was made.
Reference standard
The basic reference standard – NIH diagnostic criteria for NF1 (6) were used to determine or exclude a definitive clinical diagnosis of NF1. Presence of at least two of seven NIH diagnostic criteria confirmed clinical diagnosis of NF1 in children.
Statistical analysis
For continuous variables, such as the number of T2-hyperintensities or age at presentation, the differences between groups were compared using t test. Categorical variables such as sex, type of NF1 (sporadic or familial), and brain location of T2-hyperintensities were estimated in terms of relative frequencies. For these variables, the comparison between two groups was made using contingency tables and χ2 test. For the analysis of correlation of number of T2-hyperintensities and the age of patients, the Pearson test of correlation was performed. For the analysis of association of prevalence of T2-hyperintensities and the age of patients, children were grouped according to age. For presence/absence of T2-hyperintensities on brain MRI, we constructed a 2 × 2 contingency table that consisted of true-positive, true-negative, false-positive, false-negative, and true-negative results in accordance with the reference standard used in each case. The sensitivity and specificity of T2-hyperintensities as diagnostic criterion for NF1, as well as the negative and positive predictive value, odds ratio (OR), and accuracy rate were calculated by using standard formula. Indicators of diagnostic accuracy were evaluated in different age groups: 2-7 years, 8-12 years, and 13-18 years. The accuracy of T2-hyperintensities in diagnosis of NF1 in children was verified by receiver operating characteristic (ROC) curve. P value <0.05 was indicated as significant difference.
Results
The clinical and epidemiological characteristics of patients and controls
The patient and control group were comparable according to age and sex. The median age of 162 children in the NF1 patient group (91 boys and 71 girls) at the time when the definite clinical diagnosis of NF1 was made was 5.6 years (range 0.3-17.9). The median age of children with NF1 and of 163 control group children (82 boys and 81 girls) at the time when brain MRI examinations were made was 7.7 years (range 1.9-18.3) and 8.2 years (range 2.0-17.9), respectively (P = 0.196). Of the children with NF1, 87 (53.7%) were familial and 73 (45.1%) were sporadic cases. For two adopted children (1.2%), family history was unknown. Ten NF1 patients, all sporadic cases with multiple café au lait spots as the only diagnostic sign of NF1, underwent brain MRI examination prior to meeting the NIH diagnostic criteria. At the age of 13, they all met the NIH diagnostic criteria for NF1.
Prevalence and location of T2-hyperintensities on brain MRI
Multiple (≥2) T2-hyperintensities on brain MRI were found in 119 of 162 children with NF1 (73.5%) and only 7 of 163 children in the control group (4.3%) (P < 0.001). Four children in the control group had a single lesion and other 3 had two or more lesions. The actual prevalence of multiple T2-hyperintensities in the control group was 1.8% (3/163 patients).
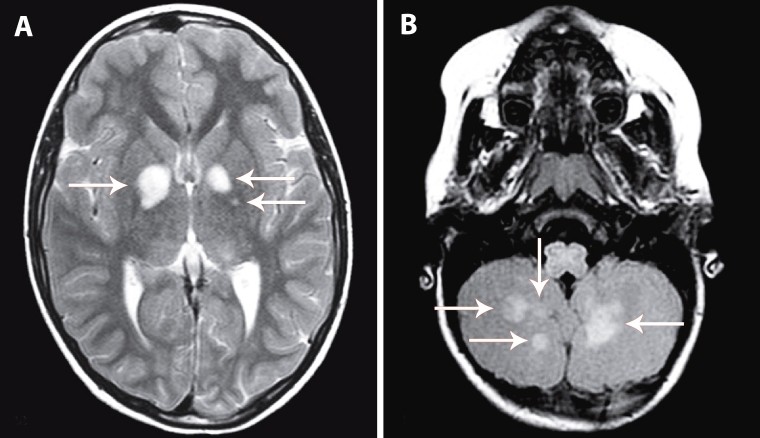
In the NF1 group, there were 636 T2-hyperintesities (median 3, range 0-16) and in the control group there were only 11 (median 0, range 0-3) (P < 0.001). T2-hyperintensities in the patient group were mainly located in the basal ganglia (81%), most frequently in the globus pallidus (78%), brainstem (38%), and cerebellum (36%) (Figure 1A and 1B). In the control group, they were located in the subcortical structures of the cerebrum (3/7 patients) and parasagittal watershed regions (3/7 patients). Only one patient in the control group had two T2-hyperintensities in the basal ganglia (putamen) (Table 1). The overall agreement in identification of multiple (≥2) T2-hyperintensities between the two radiologists was 99.4%. The agreement for localization and total number of lesions was 91.4% and 77%, respectively.
Figure 1.
Multiple hyperintensities on T2-weighted cranial magnetic resonance imaging in children with neurofibromatosis type 1. Axial image shows multiple oval discrete lesions with increased intensity (arrows) in the globus pallidus (A); axial fluid attenuated inversion recovery image shows multiple diffuse T2-hyperintensities (arrows) in the cerebellum (B).
Table 1.
Prevalence, total number, and locations of T2-hyperintensities on brain magnetic resonance imaging in children with neurofibromatosis type 1 (NF1) (n = 162) and children without NF1 (n = 163)
| Children |
T2-hyperintensities in children |
|||||||
|---|---|---|---|---|---|---|---|---|
| with NF1 |
without NF1 |
with NF1 |
without NF1 |
|||||
| T2-hyperintensities | No. | % | No | % | No. | median (range) | No. | median (range) |
| Prevalence |
119/162* |
73.5 |
7/163* |
4.3 |
636† |
3 (0-16) |
11† |
0 (0-3) |
| Location of lesions: | ||||||||
| basal ganglia |
96/119 |
80.7 |
1/7 |
14.3 |
1 (0-9) |
0 (0-2) |
||
| globus pallidus |
93/119 |
78.2 |
1 (0-7) |
|||||
| nucleus caudatus |
19/119 |
16.0 |
0 (0-3) |
|||||
| putamen |
17/119 |
14.3 |
1/7 |
14.4 |
0 (0-3) |
0 (0-2) |
||
| brainstem |
45/119 |
37.8 |
0 (0-6) |
|||||
| cerebellum |
43/119 |
36.1 |
0 (0-7) |
|||||
| cerebrum |
31/119 |
26.0 |
3/7 |
42.9 |
0 (0-5) |
0 (0-1) |
||
| periventricular | 0 | 0 | 3/7 | 42.9 | - | 0 (0-3) | ||
*P < 0.001 vs prevalence of T2-hyperintensities in children with NF1 and children without NF1 (t test).
†P < 0.001 vs total number of T2-hyperintensities in children with NF1 and children without NF1(t test).
There were no significant differences between the groups of patients who were examined with low-, medium-, and high-field MRI systems in the prevalence (72% and 75%, P = 0.363) and the number of T2-hyperintensities (median 4, range 1-16 and median 5, range 1-14, P = 0.076). Also, there were no significant differences in locations of T2-hyperintensities between two groups: the basal ganglia (P = 0.160), brainstem (P = 0.554), and cerebellum (P = 0.738) (Table 2).
Table 2.
Prevalence, total number, and locations of T2-hyperintensities on brain magnetic resonance imaging (MRI) in children with neurofibromatosis type 1 (NF1) assessed by low- and medium field MRI (n = 90) and high-field MRI (n = 72).
| T2-hyperintensities | No. (%) of children with NF1 assessed by |
Median (range) of the number of T2-hyperintensities in children with NF1 assessed by |
|||
|---|---|---|---|---|---|
| low- and medium-field MRI | high-field MRI | low- and medium-field MRI | high-field MRI | P | |
| Prevalence |
65 (72.2) |
54 (75.0) |
0.363* |
||
| Total number of lesions |
322 |
314 |
4 (1-16) |
5 (1-14) |
0.076* |
| Location of lesions: |
|||||
| basal ganglia |
51 (56.7) |
45 (62.5) |
2 (0-9) |
2 (0-8) |
0.160† |
| brainstem |
27 (30.0) |
18 (25.0) |
0 (0-6) |
0 (0-4) |
0.554† |
| cerebellum | 19 (21.1) | 24 (33.3) | 0 (0-7) | 0 (0-6) | 0.738† |
*t test.
†χ2 test.
The association of T2-hyperintensities and age
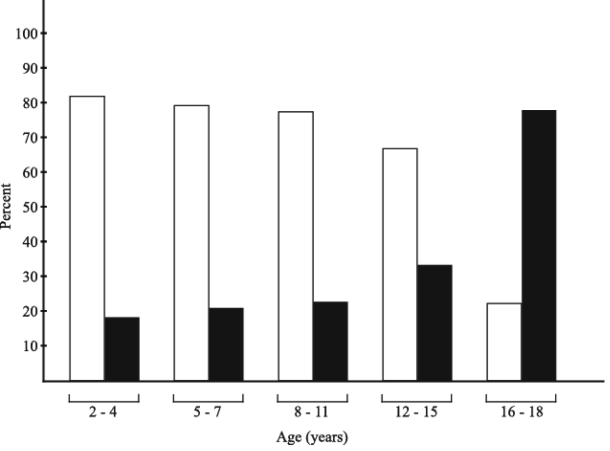
There was a significant decrease in the frequency of T2-hyperintensities with age, from 82% at 2-4 years and 79% at 5-7 years to 22% at 16-18 years (P = 0.002) (Figure 2). The T2-hyperintensities presence/absence ratio until the end of puberty (age 15) was in favor of the presence. However, in adolescence the presence of T2 hyperintensities significantly decreased and the ratio was in favor of the absence (Figure 2). There was a significant decrease in number of lesions from the earliest age, 2-7 years (total number 368, median 6, range 0-16) until puberty/adolescence, 13-18 years (total number 40, median 3, range 0-8, P = 0.011) (Figure 3).
Figure 2.
Prevalence of T2-hyperintensities on brain magnetic resonance imaging (MRI) in different age groups of children with neurofibromatosis type 1. The frequency of T2-hyperintensities significantly decreased from age 2-4 years to age 16-18 years (χ2 = 12.03, P = 0.002). Open bars – T2-hyperintensities present; closed bars – T2-hyperintensities absent.
Figure 3.
The correlation of the number of T2-hyperintensities and age in 119 children with neurofibromatosis type 1. Pearson correlation showed a significant correlation between the number T2-hyperintensities and age of patients with NF1 (r2 linear = 0.100, P = 0.011).
Clinical diagnostic sensitivity and specificity of presence of T2-hyperintensivities on brain MRI
Clinical diagnostic sensitivity and specificity of the presence of T2-hyperintensities in the group of children with NF1 aged 2-18 years was 74% (95% CI 66%-80.1%) and 98% (95% CI 94.7%-99.6%), respectively, with positive predictive value, negative predictive value, and accuracy rate of 98% (95% CI 93%-99.5%), 79% (95% CI 72.6%-84.2%), and 86% (95% CI 82%-89.5%), respectively. Positive likelihood ratio, negative likelihood ratio, and odds ratio were 40 (95% CI 13-123), 3.7 (95% CI 2.9-4,8), and 1.5 (95% CI 0.4-4.9), respectively.
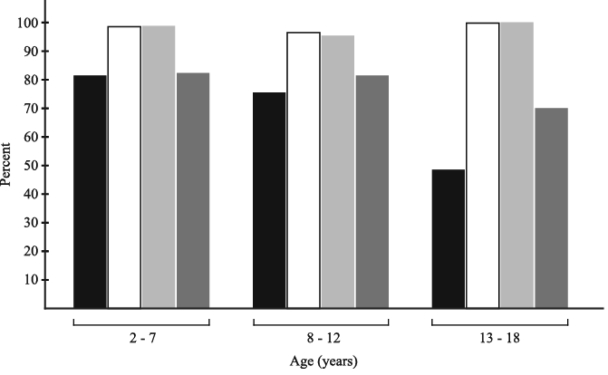
Sensitivity of T2-hyperintensities (81%, 95% CI 71%-89.1%), positive predictive value (99%, 95% CI 91.8%-99.9%), negative predictive value (82%, 95% CI 72.3%-89.7%), positive likelihood ratio (56.9, 95% CI 8-399), negative likelihood ratio (5.3, 95% CI 3-8), odds ratio (3, 95% CI 0.4-23.3), and the accuracy rate (89%) were highest in the youngest patient group, 2-7 years, and decreased as patients got older. In the age groups 8-12 years and 13-18 years, sensitivity of T2-hyperintensities was 76% (95% CI 62%-86%) and 48% (CI 30%-68%), respectively. Specificity of T2-hyperintensities was high in all age groups, ranging from 97% (95% CI 88%-100%) to 100% (Figure 4).
Figure 4.
Clinical diagnostic sensitivity, specificity, and positive and negative predictive value of T2-hyperintensities in patients with neurofibromatosis type 1 of different age. Closed bars – sensitivity; open bars – specificity; light gray – positive predictive value; dark gray – negative predictive value.
ROC analysis
ROC analysis for the evaluation of diagnostic accuracy in the total group of children (N = 325) showed that multiple T2-hyperintensities on brain MRI can have excellent diagnostic accuracy, with the area under the ROC curve of 0.849 (95% CI 0.805-0.886; P < 0.001) (Figure 5).
Figure 5.
Receiver operating characteristic (ROC) curve of T2-hyperintensities on brain magnetic resonance imaging. ROC curve (full line) and confidence interval (dashed line) for T2-hyperintensities. Gray line defines the area of 0.5.
Discussion
Our study showed very high specificity of the presence of T2-hyperintensities on brain MRI in diagnosis of NF1 in children – 98% (from 97% to 100% depending on age). The sensitivity was also high – 74% for all age groups, but was the highest in children of early age, until 7 years – 81%. None of the previous studies examined the indicators of diagnostic accuracy of T2-hyperintensities, particularly in the early age. ROC analysis showed an excellent diagnostic accuracy of T2-hyperintensities on brain MRI in children and suggested their clinical usefulness as a diagnostic criterion for NF1, especially in the early age when the definitive clinical diagnosis cannot be established using the NIH diagnostic criteria in all patients. T2-hyperintensities can be used to establish the definitive clinical diagnosis of NF1 as a second, crucial criterion, as it was shown in our 10 patients with the sporadic form of the disease. The finding of multiple T2-hyperintensities in the basal ganglia, brain stem, and cerebellum should raise the suspicion to NF1 and necessitate clinical search for other NF1 features of diagnostic value.
The diagnostic specificity and sensitivity of T2-hyperintensities were examined in only two studies so far (15,17), with results similar to ours. The retrospective study by DeBella et al (15) reported the specificity of T2-hyperintensities of 84% and 74%, respectively, assessed independently by two neuroradiologists in 19 affected children and 19 age-matched controls, aged 4-10. The sensitivity of T2-hyperintensities in the same study was 95% and 100%, respectively. The prospective study by Lopes Ferraz Filho et al (17) showed the specificity of 100% and the sensitivity of 70% in 40 children with NF1 and 48 children from the control group aged 2-18.
The data collection in this study reflects our routine clinical practice. We also recommend routine application of brain MRI in patients with NF1 in the basic clinical evaluation of the severity of disease because of numerous and, sometimes, severe organic and functional changes of central and peripheral nervous system in NF1 (7).
Application of MRI should not be limited only to symptomatic cases with NF1 as it has been recommended so far by some authors (14). We recommend the evaluation by brain MRI in children with NF1 during early childhood, after the second year, before the eighth year, or at the time of establishing the definitive clinical diagnosis if it is established after the previously mentioned age. Brain MRI can confirm the presence of T2-hyperintensities that, apart from their diagnostic value, can contribute to NF1 cognitive impairments through thalamo-cortical dysfunction (18). The recent studies have suggested that T2-hyperintesities may represent pathological foci of hyperplastic or dysplastic glial proliferation with vacuolar or spongiotic changes, with fluid-filled, coalescted, or conflated vacuoles (19). MRI reveals also other specific changes as optic glioma and other brain tumors, which develop in NF1 patients with higher frequency than in general population (20). Other NIH diagnostic changes, as sphenoid dysplasia, also demand neuroradiologic evaluation. Early detection of optic glioma and other neoplasms by MRI in NF1, which can be asymptomatic for longer period, can assure systematic and careful prospective ophthalmologic and neurologic follow-up and prompt intervention in the case of the appearance of symptoms.
The results of our study showed that the MRI systems of different field strength had no significant influence on the detection of brain T2-hyperintensities. Namely, the differences in prevalence, total number of the brain lesions, and their frequency in various brain regions were not significant between the group of patients examined with low/medium-field systems and those examined with high-field systems.
The total prevalence of multiple (≥2) T2-hyperintensities of 74% on the brain MRI in children with NF1 in our study is concordant to the majority of previous studies done in patients of the same age – from 50% to 93% (13,17,21-27). In our control group, only several patients (4.3%) had T2-hyperintensities and only 3 patients (1.8%) had multiple (≥2) lesions. Although the mode of sample selection for our control group cannot present the ideal reflection of healthy pediatric population, the prevalence of T2-hyperintensities in our study is the same to that of the accidental findings of T2-hyperintensities in healthy children up until the age of 18 established by Kim and al (2.2%) (28). In other studies, the presence of T2-hyperintensities was determined in 8 out of 1000 (0.8%) healthy individuals, 3-83 years old (29).
The multiple T2-hyperintensities in our patients were most frequently localized in the basal ganglia (predominantly in the globus pallidus), brainstem, or cerebellum. These results are concordant to the results of practically every former study (10-13,15-17,24). The locations of multiple T2-hyperintensities in our NF1 patients were different from those in healthy individuals. The majority of lesions in the control group were distributed outside the above mentioned three regions, ie, in the cerebrum, which is concordant to the findings of previous studies (15,29).
The agreement in identification of the number and location of T2-hyperintensities between the two neuroradiologists was 77% and 91.4%, respectively. This inconsistency is caused by the subjectivity of visual interpretation and difficulty in defining the exact number of lesions located in the two neighboring brain regions. Namely, T2-hyperintensities in NF1 can present themselves as discrete or diffuse lesions (10). Discrete lesions have well defined margins that are distinct from normal tissue, while diffuse lesions have poorly defined margins and confluent appearance.
In spite of the time limitation of the cross-sectional study design, a relatively large sample of respondents gives us a possibility to conclude that T2-hyperintensities on brain MRI in pediatric patients with NF1 tend to develop in toddlerhood and at preschool age. This is earlier than most NIH diagnostic features, with the exception of multiple café au lait spots. The latter feature is in the most cases the first suspected sign with predictive value in the diagnosis of NF1 (30).
This study found a significant association between T2- hyperintensities and age. Both the prevalence and the number of lesions decreased with age, concordant to the findings of other researchers (10,12). Other prospective studies showed that most T2-hyperintensities on brain MRI completely disappeared during late childhood and seemed to be benign (21,24,31,32). On the other hand, cross-sectional study by Szudek and Friedman (16), designed similarly as ours, showed that the frequency of T2-hyperintensities did not change with age.
Our clinical experience and data from this study also show that the clinical diagnosis of NF1 cannot be made using the NIH diagnostic criteria in all children, especially in patients of early age and in sporadic cases. In today’s practice, there are still many cases of late clinical diagnosis, despite of clearly defined clinical diagnostic criteria for NF1 (6). About 50% of children and 33% of adults had been treated for complications associated with NF1 before the clinical diagnosis of the disease was established. Moreover, 35% of children at the age of 5 were not diagnosed. Parents of children suspected of having NF1 want to have the diagnosis confirmed as early as possible (33). A serious diagnostic problem occurs in young children who already develop complications of the disease before showing any of the NIH diagnostic criteria (34). Ninety-seven percent of our patients were diagnosed by using a set of NIH diagnostic criteria until the age of 8, when the NF1 gene penetration should be complete. The remaining 3% were diagnosed later in life – until the age of 13. The results are similar to the results of previous investigations (8,9).
As far as 15 years ago, several authors (21-26) suggested, due to the impossibility to establish the timely diagnosis of NF1 using NIH diagnostic criteria in some children, especially in their early age, that T2-hyperintensities on the brain MRI were specific and characteristic sign of NF1 and can be an additional diagnostic criterion for the disease. However, the expert consensus did not support the use of T2- hyperintensities because data on their specificity were scarce (14). On the other hand, more recent research confirms the value of T2-hyperintensities as a diagnostic sign for NF1 in children (15,17).
In conclusion, our results of high prevalence, characteristic location, and high specificity and sensitivity of T2-hyperintensities on brain MRI strongly suggest their inclusion as diagnostic criterion for NF1 in children. As a diagnostic sign, they show very high levels of accuracy, the highest until 8 years, when the clinical penetration of the NF1 gene is not yet finished.
Acknowledgments
The authors thank colleague pediatricians at the Pediatric University Clinics in Croatia who participated in clinical examinations of NF1 patients; primary care pediatricians and family physicians who recognized the disease and have been referring the NF1 patients to our clinic for the last 20 years; Ana-Maria Šimundić, PhD, and Mario Štefanović, PhD, for their assistance in the statistical analysis; and patients and their families.
Funding None.
Ethical approval Received from the Ethics Committee of Zagreb University School of Medicine.
Declaration of authorship ZS selected the patients, created the database, and entered the data; contributed to the conception and design as well as analysis and interpretation; wrote the draft of the manuscript and approved the final version; and cooperated with co-authors while writing the discussion and conclusion.
BR interpreted the data, revised the work intellectually, and selected and approved the final version for publishing. RGJ interpreted the data, revised the work intellectually, selected the tables and figures, contributed to the next revision, and approved the final version for publishing. FS interpreted the data, contributed to the text, figures, tables, and the revision, and approved the final version for publishing. MKŠ interpreted the data, revised the text, figures, and tables, and approved the final version for publishing. KO in part contributed to the conception, design, and analysis, MRI interpretation; approved the final version, and cooperated with co-authors while writing the discussion and conclusion. DO contributed intellectually to the manuscript, interpreted the MRI findings and prepared figures for publishing; revised the manuscript and approved the final version; and collaborated with other co-authors in writing the manuscript. DŠG contributed intellectually to the manuscript, revised the text, tables, and figures, and approved the final version for publishing.
Competing interests All authors have completed the Unified Competing Interest form at www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author) and declare: no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work.
References
- 1.Huson SM, Harper PS, Compston DAS. Von Recklinghausen neurofibromatosis. A clinical and population study in South East Wales. Brain. 1988;111:1355–81. doi: 10.1093/brain/111.6.1355. [DOI] [PubMed] [Google Scholar]
- 2.Barker D, Wright E, Nguyen K, Cannon L, Fain P, Goldgar D, et al. Gene for von Recklinghausen neurofibromatosis is in pericentromeric region of chromosome 17. Science. 1987;236:1100–2. doi: 10.1126/science.3107130. [DOI] [PubMed] [Google Scholar]
- 3.Viskochil D, Cawthon R, O'Connel P, Xu GF, Stevens J, Culver M, et al. The gene encoding the oligodendrocyte-myelin glycoprotein is embedded within the neurofibromatosis type 1 gene. Mol Cell Biol. 1991;11:906–12. doi: 10.1128/mcb.11.2.906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marchuk DA, Saulino AM, Tavakkol RL, Swaroop M, Wallace MR, Andersen LB, et al. cDNA cloning of the type 1 neurofibromatosis gene: complete sequence of the NF1 gene product. Genomics. 1991;11:931–40. doi: 10.1016/0888-7543(91)90017-9. [DOI] [PubMed] [Google Scholar]
- 5.Crowe FW, Schull WJ, Neel JV. A clinical, pathological, and genetic study of multiple neurofibromatosis. Springfield, (IL): Charles C. Thomas; 1956. [Google Scholar]
- 6.National Institutes of Health Consensus Development Conference. Neurofibromatosis Conference Statement. Arch Neurol. 1988;45:575–8. [PubMed] [Google Scholar]
- 7.Riccardi VM. Neurofibromatosis: phenotype, natural history, and pathogenesis. 2nd ed. Baltimore (MD): Johns Hopkins University Press; 1992. [Google Scholar]
- 8.Obringer AC, Meadows AT, Zackai EH. The diagnosis of neurofibromatosis–1 in the child under age of 6 years. Am J Dis Child. 1989;143:717–9. doi: 10.1001/archpedi.1989.02150180099028. [DOI] [PubMed] [Google Scholar]
- 9.DeBella K, Szudek J, Friedman JM. Use of the National Institutes of Health criteria for diagnosis of neurofibromatosis 1 in children. Pediatrics. 2000;105:608–14. doi: 10.1542/peds.105.3.608. [DOI] [PubMed] [Google Scholar]
- 10.Gill DS, Hyman SL, Steinberg A, North KN. Age-related findings on MRI in neurofibromatosis type 1. Pediatr Radiol. 2006;36:1048–56. doi: 10.1007/s00247-006-0267-2. [DOI] [PubMed] [Google Scholar]
- 11.Bognanno JR, Edwards MK, Lee TA, Dunn DW, Roos KL, Klatte EC. Cranial MR imaging in neurofibromatosis. AJR Am J Roentgenol. 1988;151:381–8. doi: 10.2214/ajr.151.2.381. [DOI] [PubMed] [Google Scholar]
- 12.Sevick RJ, Barkovich AJ, Edwards MS, Koch T, Berg B, Lempert T. Evolution of white matter lesions in neurofibromatosis type 1: MR findings. AJR Am J Roentgenol. 1992;159:171–5. doi: 10.2214/ajr.159.1.1609692. [DOI] [PubMed] [Google Scholar]
- 13.Duffner PK, Cohen ME, Seidel FG, Shucard DW. The significance of MRI abnormalities in children with neurofibromatosis. Neurology. 1989;39:373–8. doi: 10.1212/wnl.39.3.373. [DOI] [PubMed] [Google Scholar]
- 14.Gutmann DH, Aylsworth A, Carey JC, Korf B, Marks J, Pyeritz RE, et al. The diagnostic evaluation and multidisciplinary management of neurofibromatosis 1 and neurofibromatosis 2. JAMA. 1997;278:51–7. doi: 10.1001/jama.278.1.51. [DOI] [PubMed] [Google Scholar]
- 15.DeBella K, Poskitt K, Szudek BS, Friedman JM. Use of «unidentified bright objects» on MRI for diagnosis of neurofibromatosis 1 in children. Neurology. 2000;54:1646–51. doi: 10.1212/wnl.54.8.1646. [DOI] [PubMed] [Google Scholar]
- 16.Szudek J, Friedman JM. Unidentified bright objects associated with features of neurofibromatosis 1. Pediatr Neurol. 2002;27:123–7. doi: 10.1016/S0887-8994(02)00403-4. [DOI] [PubMed] [Google Scholar]
- 17.Lopes Ferraz Filho JR, Munis MP, Soares Souza A, Sanches RA, Goloni-Bertollo EM, Pavarino-Bertelli EC. Unidentified bright objects on brain MRI in children as a diagnostic criterion for neurofibromatosis type 1. Pediatr Radiol. 2008;38:305–10. doi: 10.1007/s00247-007-0712-x. [DOI] [PubMed] [Google Scholar]
- 18.Chabernaud C, Sirinelli D, Barbier C, Cottier JP, Sembely C, Giraudeau B, et al. Thalamo-striatal T2-weighted hyperintensities (unidentified bright objects) correlate with cognitive impairments in neurofibromatosis type 1 during childhood. Dev Neuropsychol. 2009;34:736–48. doi: 10.1080/87565640903265137. [DOI] [PubMed] [Google Scholar]
- 19.DiPaolo DP, Zimmerman RA, Rorke LB, Zackai EH, Bilaniuk LT, Yachnis AT. Neurofibromatosis type 1: pathologic substrate of high-signal-intensity foci in the brain. Radiology. 1995;195:721–4. doi: 10.1148/radiology.195.3.7754001. [DOI] [PubMed] [Google Scholar]
- 20.Rosser T, Packer RJ. Intracranial neoplasms in children with neurofibromatosis 1. J Child Neurol. 2002;17:630–7. doi: 10.1177/088307380201700815. [DOI] [PubMed] [Google Scholar]
- 21.Van Es S, North KN, McHugh K, De Silva M. MRI findings in children with neurofibromatosis type 1: a prospective study. Pediatr Radiol. 1996;26:478–87. doi: 10.1007/BF01377205. [DOI] [PubMed] [Google Scholar]
- 22.Menor F, Marti-Bonmati L, Arana E, Poyatos C, Cortina H. Neurofibromatosis type 1 in children: MR imaging and follow-up studies of central nervous system findings. Eur J Radiol. 1998;26:121–31. doi: 10.1016/S0720-048X(97)00088-0. [DOI] [PubMed] [Google Scholar]
- 23.Bonawitz C, Castillo M, Chin CT, Mukherji SK, Barkovich J. Usefulness of contrast material in MR of patients with neurofibromatosis type 1. AJNR Am J Neuroradiol. 1998;19:541–6. [PMC free article] [PubMed] [Google Scholar]
- 24.Griffiths PD, Blaser S, Mukonoweshuro W, Armstrong D, Milo-Mason G, Cheung S. Neurofibromatosis bright objects in children with neurofibromatosis type 1: a proliferative potential? Pediatrics. 1999;104:e49. doi: 10.1542/peds.104.4.e49. [DOI] [PubMed] [Google Scholar]
- 25.Rosenbaum T, Engelbrecht V, Krölls W, van Dorsten FA, Hoehn-Berlage M, Lenard HG. MRI abnormalities in neurofibromatosis type 1 (NF1): a study of men and mice. Brain Dev. 1999;21:268–73. doi: 10.1016/S0387-7604(99)00024-8. [DOI] [PubMed] [Google Scholar]
- 26.Goldstein SM, Curless RG, Donovan Post MJ, Quencer RM. A new sign of neurofibromatosis on magnetic resonance imaging of children. Arch Neurol. 1989;46:1222–4. doi: 10.1001/archneur.1989.00520470086032. [DOI] [PubMed] [Google Scholar]
- 27.Hashimoto T, Tayama M, Miyazaki M, Kuroda Y, Hiura K, Hujino K, et al. Cranial MR imaging in patients with von Recklinghausen's disease (neurofibromatosis type 1). Neuropediatrics. 1990;21:193–8. doi: 10.1055/s-2008-1071494. [DOI] [PubMed] [Google Scholar]
- 28.Kim BS, Illes J, Kaplan RT, Reiss A, Atlas SW. Incidental findings on pediatric MR images of the brain. AJNR Am J Neuroradiol. 2002;23:1674–7. [PMC free article] [PubMed] [Google Scholar]
- 29.Katzman GL, Dagher AP, Patronas NJ. Incidental findings on brain magnetic resonance imaging from 1000 asymptomatic volunteers. JAMA. 1999;282:36–9. doi: 10.1001/jama.282.1.36. [DOI] [PubMed] [Google Scholar]
- 30.Nunley KS, Gao F, Albers AC, Bayliss SJ, Gutman DH. Predictive value of café au lait macules at initial consultation in the diagnosis of neurofibromatosis type 1. Arch Dermatol. 2009;145:883–7. doi: 10.1001/archdermatol.2009.169. [DOI] [PubMed] [Google Scholar]
- 31.Balestri P, Calistri L, Vivarelli R, Bartalini G, Mancini L, Berardi A, et al. Central nervous system imaging in reevaluation of patients with neurofibromatosis type 1. Childs Nerv Syst. 1993;9:448–51. doi: 10.1007/BF00393546. [DOI] [PubMed] [Google Scholar]
- 32.Kraut MA, Gerring JP, Cooper KL, Thompson RE, Denckla MB, Kaufmann WE. Longitudinal evolution of unidentified bright objects in children with neurofibromatosis-1. Am J Med Genet. 2004;129A:113–9. doi: 10.1002/ajmg.a.20656. [DOI] [PubMed] [Google Scholar]
- 33.Cnossen MH, Smit FJ, de Goede-Bolder A, Frets PG, Duivenvoorden HJ, Niermeijer MF. Diagnostic delay in neurofibromatosis type 1. Eur J Pediatr. 1997;156:482–7. doi: 10.1007/s004310050644. [DOI] [PubMed] [Google Scholar]
- 34.Stoll C. Difficulties in the diagnosis of neurofibromatosis-1 in children. Am J Med Genet. 2002;112:422–6. doi: 10.1002/ajmg.10655. [DOI] [PubMed] [Google Scholar]