Abstract
CD40, a member of the TNF receptor (TNFR) superfamily, is expressed on antigen presenting cells (APCs) and is essential for immune activation. While agonistic CD40 antibodies have been developed for immunotherapy, their clinical efficacy has been limited. We have found that co-engagement of the Fc domain of agonistic CD40 monoclonal antibodies (mAbs) with the inhibitory Fcγ receptor FcγRIIB is required for immune activation. Direct comparison of anti-CD40 mAbs enhanced for activating FcγR binding hence capable of cytotoxicity, or FcγRIIB binding revealed that enhancing FcγRIIB binding conferred immunostimulatory activity and considerably greater anti-tumor responses. This unexpected requirement for FcγRIIB in enhancing CD40 mediated immune activation has direct implications for the design of agonistic, anti-TNFR antibodies as therapeutics.
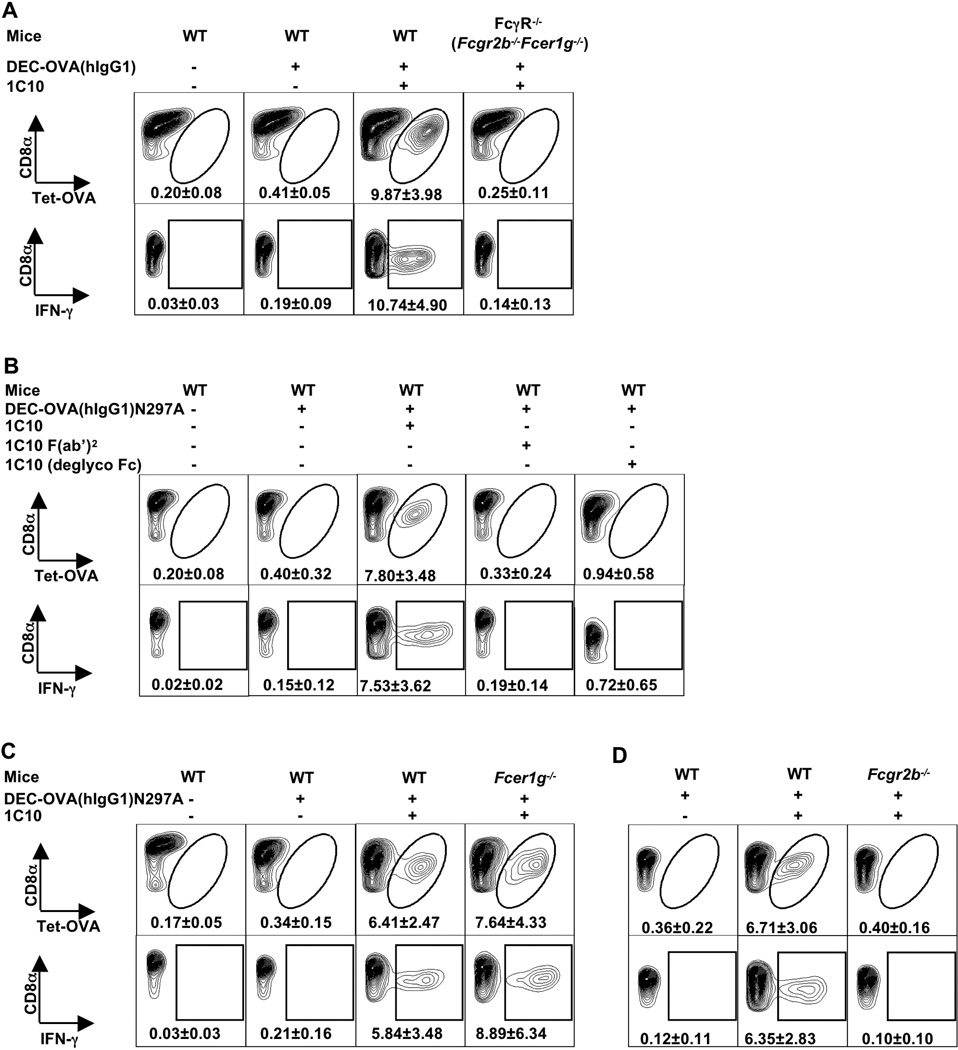
Recent clinical success in the antigen-independent activation of cytotoxic T cells has highlighted the potential of immune activation as a therapeutic strategy for the treatment of neoplastic diseases (1). However, such non-specific activation can result in considerable toxicity as a consequence of cytokine release from activated T cells (2). Ideally, effective immunotherapy would capture the specificity of antigen-driven T cell responses through the T cell receptor (TCR) by activating the APCs responsible for restricted T cell responses directed at a tumor target. Antigen delivery to APCs will result in antigen processing and presentation of peptides on MHC molecules to T cells expressing the cognate TCR. However, activation of those T cells requires that an additional signal be delivered concurrently. These additional signals can result from the enhanced expression of co-stimulatory molecules including CD80 and CD86 on APCs triggered by adjuvants, such as microbial products or CD40 ligation, among others. To determine the requirements for CD40 ligation that result in optimal T cell activation, we used a dendritic cell targeting strategy to deliver OVA to DEC205 positive cells (3) together with an agonistic CD40 antibody, 1C10 (4). A chimeric DEC205 antibody with human IgG1 Fc was constructed, to which OVA was fused at the carboxy terminus of the Fc. This antibody (DEC-OVA(hIgG1), ref. (5)) was injected into wild-type (WT) mice along with 1C10. As observed previously (3), this targeting approach efficiently generated OVA-specific CD8+ T cells that could be activated to express interferon γ (IFN-γ) (Fig. 1). In mice lacking all FcγRs (FcγR−/−), however, T cell expansion and activation were not observed, implicating a role for the Fc region of the targeting (DEC-OVA(hIgG1)) or adjuvant (anti-CD40) antibody (Fig. 1A). Mutation of DEC-OVA(hIgG1) to eliminate FcγR binding (DEC-OVA(hIgG1)N297A, ref. (5)), had no effect on the ability to generate OVA-specific T cells (fig. S1). In contrast, elimination of FcγR binding activity from 1C10, either by generating F(ab’)2 or de-glycosylated Fc (fig. S2), eliminated the ability to generate OVA-specific T cells (Fig. 1B). Mice express four IgG Fc receptors, activating FcγRI, III, IV, and inhibitory FcγRIIB (6). FcγRI, III and IV require the common γ chain (FcRγ) for assembly and surface expression and, upon crosslinking, result in cellular activation. In contrast, FcγRIIB mediates an inhibitory signal. Mice lacking the common γ chain (encoded by Fcer1g) and thus deficient in FcγRI, III and IV were able to generate OVA-specific T cells upon OVA targeting and 1C10 stimulation (Fig. 1C). In contrast, Fcgr2b−/− (the gene that encodes FcγRIIB) mice targeted by DEC-OVA(hIgG1)N297A showed no OVA-specific T cells expansion or activation with 1C10 or with either of two agonistic CD40 mAbs, clone 3/23 and FGK45 (Fig. 1D and fig. S3). Similar results were obtained when wild-type mice were administered DEC-OVA(hIgG1)N297A and 1C10 in conjunction with 2.4G2 antibody blockade of FcγRIIB and FcγRIII (fig. S4). Fcgr2b−/− mice were, however, able to generate OVA-specific T cells when OVA targeting was combined with another adjuvant, poly I:C (fig. S5), indicating that the lack of T cell activation was specific to the requirement for agonistic CD40 mAbs to engage FcγRIIB.
Fig. 1.
FcγRIIB-engagement is required for the adjuvant activity of agonistic CD40 antibodies. (A) WT and FcγR−/− (Fcgr2b−/−Fcer1g−/−, deficient in all FcγRs) mice were injected i.p. with DEC-OVA(hIgG1) in the presence or absence of 1C10. Spleen cells were harvested 7 days later and analyzed by flow cytometry for OVA-specific CD8+ T cells, detected by OVA tetramer staining or IFN-γ expression upon in vitro OVA peptide stimulation as described in “Materials of methods” (5). (B–D) WT, Fcer1g−/− (FcRγ-deficient), and Fcgr2b−/− (FcγRIIB-deficient) mice were immunized with DEC-OVA(hIgG1)N297A (the N297A variant of DEC-OVA(hIgG1), null for FcγR binding) in the presence or absence of 1C10, or deglycosylated 1C10, or 1C10 F(ab’)2 fragment, and analyzed as in (A). Shown are representative contour plots gated on CD4−CD8α+ cells, with gate and percentage (mean ± S.D) of tet-OVA+ or IFN-γ+ cells, of two or more independent experiments with 4–5 mice per group with similar results.
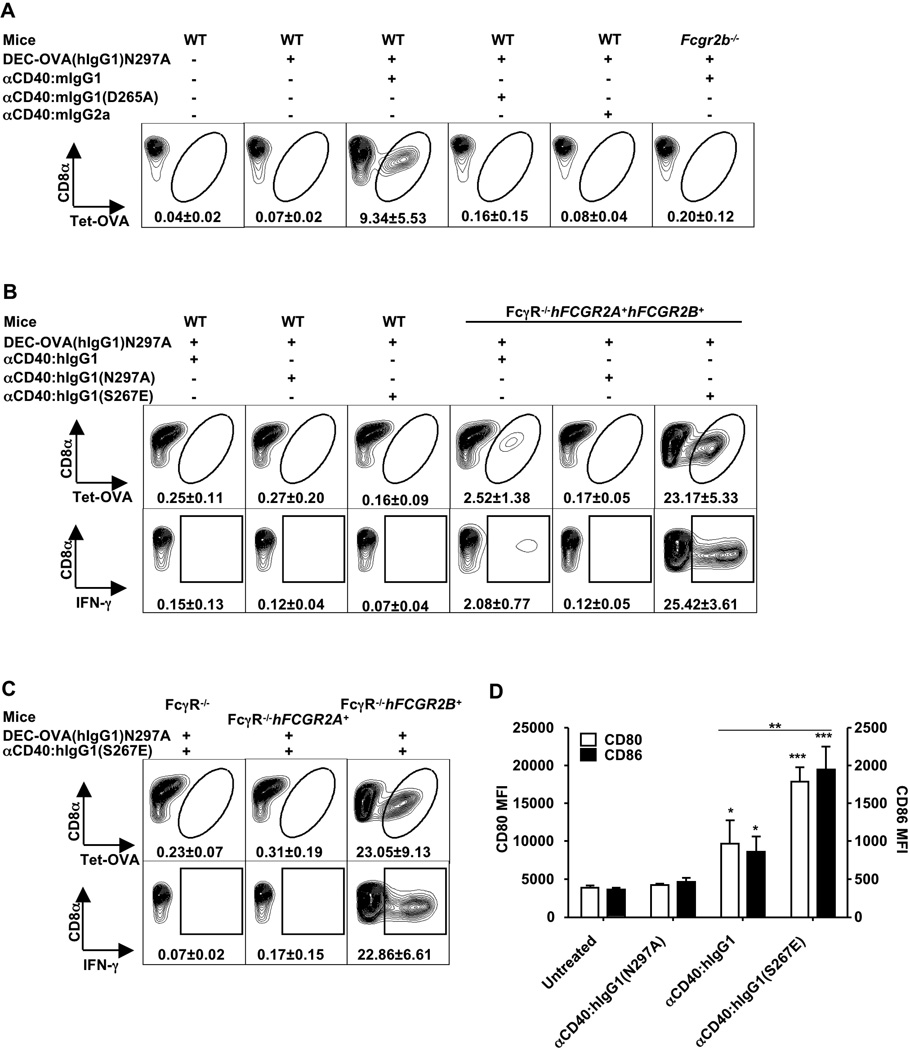
The absolute requirement for FcγRIIB raised the possibility that the adjuvant effect of anti-CD40 could be modulated by altering the binding affinity of the anti-CD40 Fc to FcγRIIB. We generated both mouse and human Fc variants of 1C10 with different binding affinities to their respective FcγRs (fig. S6) and confirmed that the CD40 binding specificity and affinity were unchanged from 1C10, the parental rat IgG2a mAb (fig. S7). Mouse IgG1 has a tenfold higher affinity for mouse FcγRIIB as compared to mouse IgG2a (7–8). Introducing these two variants of 1C10 (αCD40:mIgG1 and αCD40:mIgG2a, respectively) or a D265A mutant of the mouse IgG1 variant that lacks FcγR binding affinity (αCD40:mIgG1(D265A), ref. (8–9)) with DEC-OVA(hIgG1)N297A into wild-type mice resulted in expansion and activation of OVA-specific CD8+ T cells only when αCD40:mIgG1 was used (Fig. 2A). Similarly, a chimeric anti-CD40 antibody with either an unmutated human IgG1 Fc (αCD40:hIgG1), its S267E variant with 30-fold higher binding affinity for human FcγRIIB (αCD40:hIgG1(S267E), ref. (10)), or a human Fc null variant for FcγR binding (αCD40:hIgG1(N297A), ref. (8, 11)) was introduced into mice deleted for murine FcγRs and expressing human FcγRIIA and IIB (encoded by hFCGR2A and hFCGR2B, respectively) in a cell appropriate pattern (fig. S8 and ref. (12–13)) together with DEC-OVA(hIgG1)N297A. αCD40:hIgG1(S267E) showed approximately a tenfold enhancement of OVA-specific CD8+ T cells over αCD40:hIgG1, as measured by both tetramer staining and intracellular IFN-γ expression (Fig. 2B). Dose dependent enhancement of OVA-specific T cell responses was observed in Fcgr2b−/− mice expressing a human FCGR2B transgene (fig. S9). This enhanced T cell response was dependent upon the expression of human FcγRIIB in these mice; human FcγRIIA expression could not compensate for this requirement (Fig. 2C and fig. S9). CD40 co-engagement with FcγRIIB resulted in increased DC maturation as measured by enhanced CD80 and CD86 expression (Fig. 2D), supporting its role in inducing T cell activation through APCs.
Fig. 2.
The adjuvant effect of agonistic CD40 antibodies can be modulated by manipulating their binding affinities to FcγRIIB. (A–C) WT, Fcgr2b−/−, FcγR−/−, and FcγR−/− mice with human FCGR2A and/or FCGR2B transgenes (FcγR−/−hFCGR2A+, FcγR−/−hFCGR2B+, or FcγR−/−hFCGR2A+hFCGR2B+) were immunized with DEC-OVA(hIgG1)N297A in the presence or absence of CD40 antibodies with the indicated Fc’s, and analyzed for OVA-specific CD8+ T cells in peripheral blood (A) or spleen (B–C) as in Fig. 1. Shown are representative contour plots gated on CD4−CD8α+ cells, with gate and percentage (mean ± S.D) of tet-OVA+ or IFN-γ+ cells, of two or more independent experiments with 4~5 mice per group with similar results. (D) Increased in vivo DC maturation induced by anti-CD40 with enhanced hFcγRIIB-binding affinity. FcγR−/−hFCGR2B+ mice (3 per group) were either untreated or injected i.p. with 100µg of the indicated CD40 antibodies. Three days later, CD80 and CD86 expression were analyzed on splenic dendritic cells by flow cytometry. Presented is a bar-graph showing mean fluorescence intensity (MFI) values with S.D. * p < 0.05; ** p < 0.01; *** p < 0.001. One-way ANOVA with Dunnett post hoc test was used to compare: all groups to the untreated control group; and the αCD40:hIgG1 and αCD40:hIgG1(S267E) treated groups.
Mouse IgG1 preferentially binds to FcγRIIB and displays minimal antibody-dependent cellular cytotoxicity (ADCC) activity in vivo by virtue of the inhibitory effect of FcγRIIB binding, whereas mouse IgG2a efficiently engages FcγRIV and is the most potent mouse IgG subclass in mediating ADCC (7–8). The lack of the adjuvant effects by αCD40:mIgG2a could be the result of depletion of CD40+ APCs. To test this possibility, αCD40:mIgG2a was injected with DEC-OVA(hIgG1)N297A into NK cell depleted wild-type mice or FcγRIV-deficient (Fcgr4−/−) mice. As shown in fig. S10, neither NK cell depletion nor FcγRIV-deficiency restored T cell responses. In addition, no attenuation of T cell activation was seen when the combination of αCD40:mIgG1 and αCD40:mIgG2a was injected with DEC-OVA(hIgG1)N297A, supporting a dominant role for the FcγRIIB pathway in the adjuvant effect of agonistic CD40 mAbs.
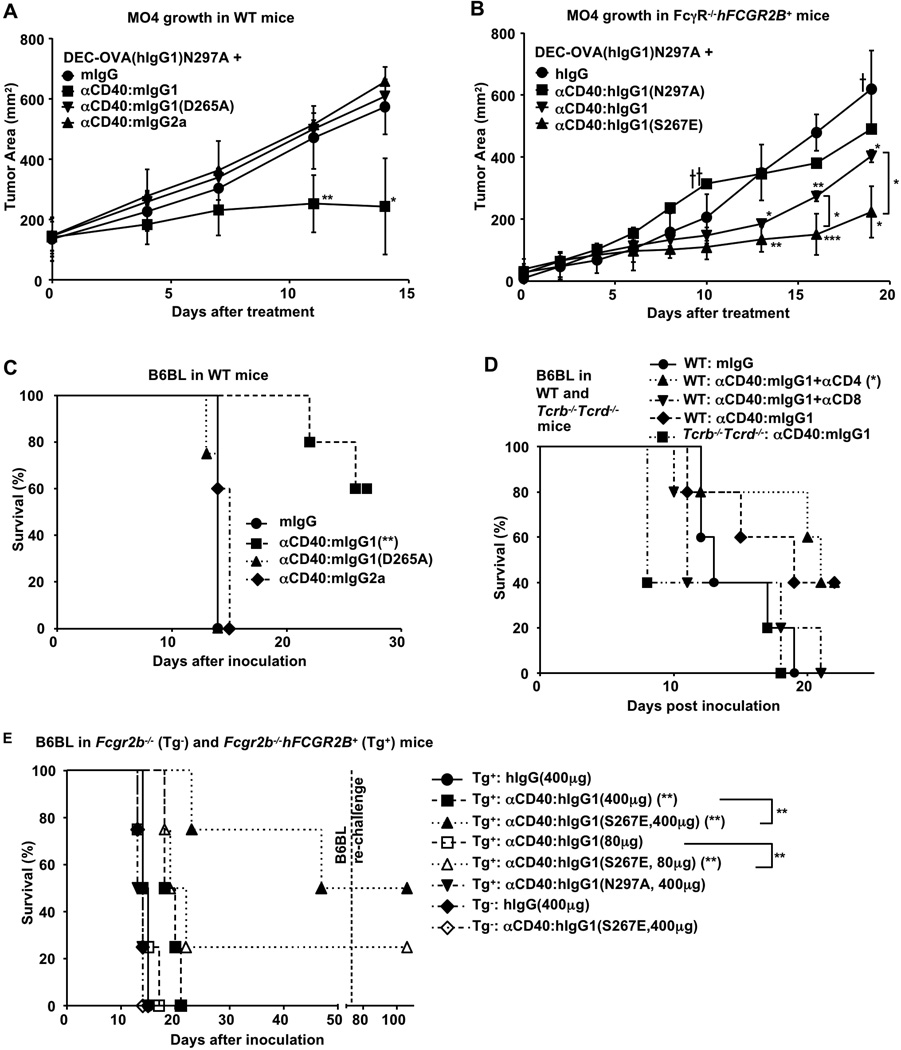
Next, the ability of DEC-OVA(hIgG1)N297A targeted DCs stimulated by anti-CD40 enhanced for FcγRIIB binding to generate OVA restricted CD8+ cells was tested for their ability to translate into an anti-tumor response against MO4, a CD40-negative melanoma tumor line engineered to express OVA (3). MO4 tumor cells were implanted subcutaneously in wild-type or human FcγRIIB transgenic mice (FcγR−/−hFCGR2B+) and then treated with DEC-OVA(hIgG1)N297A and either anti-CD40 engineered to express Fc’s that preferentially engaged mouse (αCD40:mIgG1) or human (αCD40:hIgG1(S267E)) FcγRIIB, or anti-CD40 engineered to express mouse IgG2a Fc (αCD40:mIgG2a) or human IgG1 Fc (αCD40:hIgG1) thus preferentially engaging activating FcγR, or anti-CD40 with Fc’s unable to engage FcgRs such as αCD40:mIgG1(D265A) and αCD40:hIgG1(N297A). Anti-tumor activity was significantly greater for the DEC-OVA(hIgG1)N297A targeted mice when treated with CD40 antibodies engineered to selectively engage FcγRIIB (Fig. 3, A and B). The anti-tumor activity of these antibodies required FcγRIIB as Fcgr2b−/− and FcγR−/− mice showed no anti-tumor activity with these treatments (fig. S11). Anti-CD40 engineered with either a mouse IgG2a Fc or human IgG1 Fc showed no (αCD40:mIgG2a) or reduced (αCD40:hIgG1) anti-tumor activity (Fig. 3A, B). The FcγR null binding variants αCD40:mIgG1(D265A) or αCD40:hIgG1(N297A) had no anti-tumor effects in these studies (Fig. 3A, B). These data, taken together with previous studies (3), indicates that the enhancement of tumor killing we observe likely results from enhanced T cell dependent mechanisms.
Fig. 3.
Anti-tumor activities of CD40 antibodies correlate with their adjuvant effects in CD40− tumor models. (A–B) WT and FcγR−/−hFCGR2B+ mice were inoculated with MO4 tumor cells subcutaneously, and treated with DEC-OVA(hIgG1)N297A plus low doses (30µg) of control IgG or the indicated CD40 antibodies with either mouse (A) or human (B) IgG Fc’s after tumor establishment. Tumor growth curves (3–5 mice per group) are shown. (C) WT mice were inoculated intravenously with B6BL tumor cells and treated with high doses of control IgG or the indicated CD40 antibodies (d3: 200µg; d4; 200µg). (D) WT and T cell deficient mice (Tcrb−/− Tcrd−/−) were inoculated with B6BL as in (C) and treated with control IgG or the indicated CD40 antibodies (d3: 200µg) with or without αCD4 or αCD8 depleting antibodies; (E) Fcgr2b−/−(Tg−) and Fcgr2b −/−hFCGR2B+ (Tg+) mice were inoculated with B6BL as in (C) and treated with high doses (d3: 200µg; d4: 200µg) or lower doses (d3: 40µg; d4: 40µg) of the indicated CD40 antibodies or control IgG. Long-term survivors were re-challenged at 10 weeks with B6BL tumor cells. (C–E) Survival curves of 4–5 mice are shown. Error bars are S.D. *p<0.05, **p<0.01, ***p<0.001. One-way ANOVA with Dunnett post hoc test (A–B) or Logrank test (C–E) was used to compare: all groups to the control groups (“mIgG” in (A), “hIgG” in (B), “mIgG” in (C), “WT:mIgG” in (D), or Tg+:hIgG(400µg) in (E), respectively); or “αCD40:hIgG1” to “αCD40:hIgG1(S267E)” in (B); or between Tg+ mice treated with same doses of αCD40:hIgG1 or αCD40:hIgG1(S267E) in (E).
Because previous studies have shown that high doses of agonistic CD40 antibodies alone can induce cytotoxic T cells against lymphoma cells (14), we further tested whether agonistic CD40 antibodies engineered to co-engage FcγRIIB have enhanced anti-tumor activity in the absence of immunization with a tumor expressed antigen. A CD40-negative B cell lymphoma line (B6BL, fig. S12, ref. (5, 15)) was studied with our engineered CD40 antibodies. The enhanced adjuvant effect of co-engagement of CD40 antibodies to FcγRIIB was sufficient to mediate anti-tumor activity against B6BL tumor (Figure 3C, D, E). The lack of anti-tumor activity of αCD40:hIgG1(N297A) in FcγRIIB-humanized mice (Fcgr2b−/−hFCGR2B+) and αCD40:hIgG1(S267E) in Fcgr2b−/− mice, suggest that FcγRIIB-engagement is necessary and sufficient for anti-tumor activity (Fig. 3E). This anti-tumor effect was mediated by CD8+ T cells as T cell deficiency (Tcrb−/−Tcrd−/− mice, ref. (5)) or CD8+ cell depletion (αCD8) abrogated the anti-tumor effect of αCD40:mIgG1 (Figure 3D). Re-challenge of the B6BL-surviving animals at 10 weeks with B6BL tumor cells resulted in resistance, indicating the presence of a memory response.
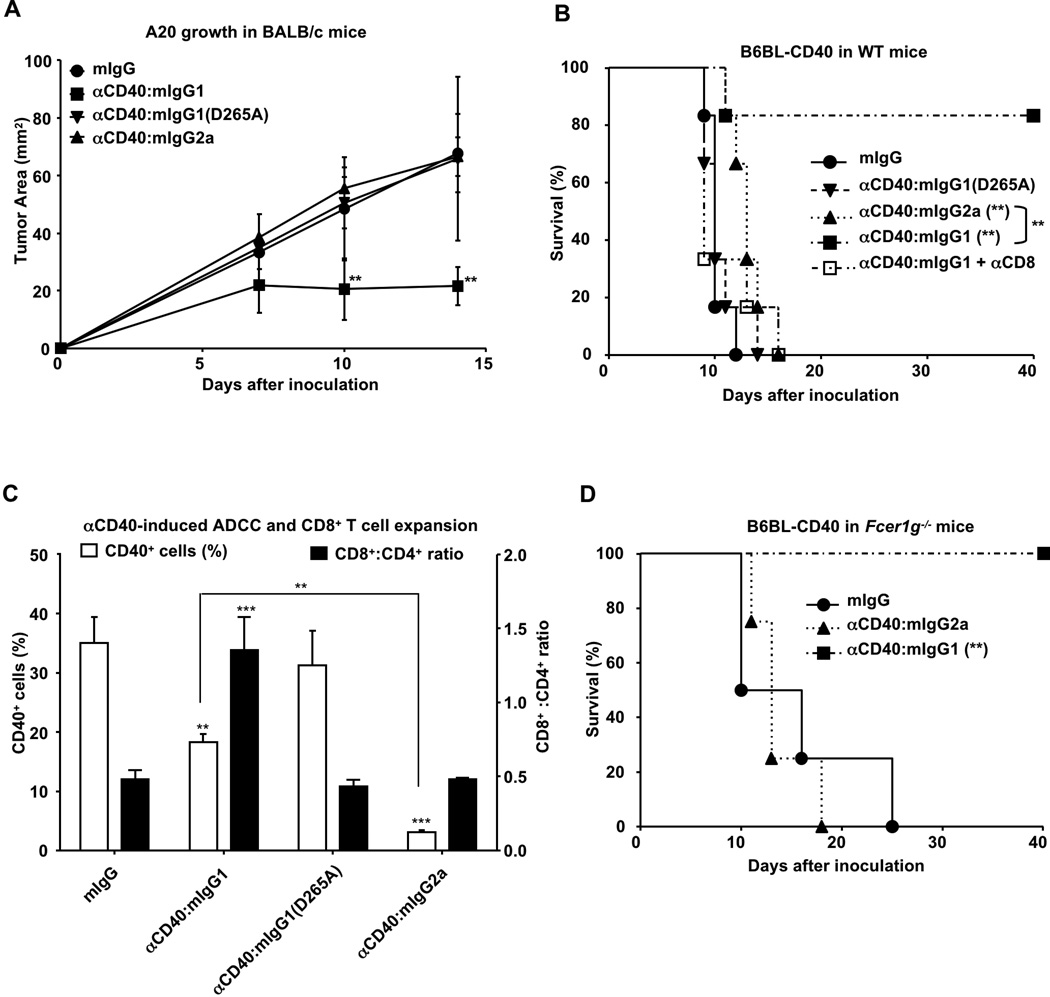
Previous studies have documented the importance of ADCC, mediated by activating FcγRs, as an anti-tumor mechanism (6–7, 9). Correlation of ADCC activity to patient survival has been observed for the therapeutic antibodies anti-CD20 for non-Hodgkin lymphoma (NHL) or anti-Her2/Neu for metastatic breast carcinoma (16–18). In contrast, the anti-tumor activities we have observed for agonistic CD40 antibodies require the co-engagement of the inhibitory FcγRIIB, leading to the maturation of dendritic cells promoting the expansion and activation of cytotoxic CD8+ T cells. We therefore set out to compare the relative potencies of antibodies enhanced for ADCC with those enhanced for adjuvancy in two different CD40+ tumor models, the BALB/c-derived A20 lymphoma and B6-derived B6BL engineered to express CD40 (B6BL-CD40, fig. S12, (5)). Animals were challenged with either A20 or B6BL-CD40 tumors and treated with agonistic CD40 mAbs enhanced for either ADCC (αCD40:mIgG2a) or FcγRIIB-binding (αCD40:mIgG1) (Fig. 4A and B). αCD40:mIgG2a treatment showed no effect on A20 growth and a small, but significant improvement in survival in B6BL-CD40 challenged mice at the dose indicated, while treatment at the same dose of αCD40:mIgG1 resulted in arrest of tumor growth for A20 (Fig. 4A) or long-term survival for B6BL-CD40 challenged mice (Fig. 4B). The lack of anti-tumor activity for αCD40:mIgG2a was not due to defects in ADCC activity since the antibody displayed robust depletion of peripheral CD40+ cells in treated mice (Fig. 4C). In contrast, αCD40:mIgG1 treated mice displayed marked expansion of CD8+ cells in the periphery (Fig. 4C). Thus, the adjuvant effects of agonistic CD40 mAbs (activation of cytotoxic T cells through CD40 mediated stimulation of APCs) results in a more potent anti-tumor effect than cytotoxicity triggered through effector cell activation through FcγR crosslinking. Indeed, the anti-tumor effect of αCD40:mIgG1 in prolonging survival of B6BL-CD40 challenged mice was not affected by deficiency in FcRγ chain (required for all activating FcγRs) (Fig. 4D), supporting an ADCC-independent mechanism for this anti-tumor effect. In addition, depleting CD8+ cells abrogated the anti-tumor effect of αCD40:mIgG1, confirming that CD8+ T cells were required for this response (Fig. 4B).
Fig. 4.
Anti-CD40 enhanced for FcγRIIB-binding has greater anti-tumor activities than anti-CD40 enhanced for activating FcγR-binding in CD40+ tumor models. (A) BALB/c mice were treated with the indicated CD40 antibodies or control IgG (200µg) 1h before the subcutaneous inoculation of A20 tumor cells. Tumor growth curves of 5 mice per group are shown. WT (B) and Fcer1g−/− (D) mice were inoculated with B6BL-CD40 tumor cells and treated with the indicated CD40 antibodies or control IgG (d3: 200µg; d6: 200µg) with or without αCD8 depleting antibodies. Survival curves of 4–6 mice per group are shown in (B, D). (C) WT mice (3 per group) were treated with 200µg of the indicated CD40 antibodies or control IgG. Six days later, the percentage of CD40+ cells and the ratio of CD8+ to CD4+ T cells were analyzed in peripheral blood and presented in the bar-graph. Error bars are S.D. **p<0.01, ***p<0.001. One-way ANOVA with Dunnett post hoc test (A, C) or Logrank test (B, D) was used to compare: all groups to the “mIgG” control groups; “αCD40:mIgG1” to “αCD40:mIgG2a” in (C).
The FcγRIIB pathway required for agonistic CD40 antibody activities may be general to other TNFR family members. For example, Fas mediated toxicity, triggered by agonistic Fas antibodies, requires FcγRIIB (19). Similarly, DR4, DR5, and CD30 agonistic antibodies show greater anti-tumor activity in vivo when their Fc’s are capable of FcγRIIB engagement (20–22). Finally, the recent results showing that an agonistic CD40 antibody (clone FGK45) has anti-tumor activity in a mouse model of pancreatic ductal adenocarcinoma and can enhance the characteristic APC indicators such as MHC class II, CD80 and CD86 expression of stromal macrophages support an immune stimulatory component in its anti-tumor activity (23). Thus, the results presented here establish a new paradigm for immune activation of agonistic TNFR antibodies through FcγRIIB co-engagement that will inform the rational design of novel therapeutic antibodies.
Supplementary Material
Acknowledgments
We thank R. Anthony, and A. Pincetic, S. Bournazos for helpful discussion and comments on the manuscript; M. Nussenzweig, D. Robbiani, S. Deroubaix for kindly providing B6BL cells; P. Smith, B. Wang, L. Bozzacco, A. Kamphorst, K. Horiuchi, R. Peraza M. Kibe, I. Londono J. Carroll, J. Brown, T. Shabaneh for excellent technical support. This work was performed with support from NIH grants to J.V.R. F. L. is supported in part by Grant# 2757 from Paralyzed Veterans of America research foundation.
Footnotes
References and notes
- 1.Hodi FS, et al. N Engl J Med. 2010 Aug 19;363:711. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Suntharalingam G, et al. N Engl J Med. 2006 Sep 7;355:1018. doi: 10.1056/NEJMoa063842. [DOI] [PubMed] [Google Scholar]
- 3.Bonifaz LC, et al. J Exp Med. 2004 Mar 15;199:815. doi: 10.1084/jem.20032220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Heath AW, Wu WW, Howard MC. Eur J Immunol. 1994 Aug;24:1828. doi: 10.1002/eji.1830240816. [DOI] [PubMed] [Google Scholar]
- 5.Materials ans methods are available as supporting material on Science online.
- 6.Nimmerjahn F, Ravetch JV. Nat Rev Immunol. 2008 Jan;8:34. doi: 10.1038/nri2206. [DOI] [PubMed] [Google Scholar]
- 7.Nimmerjahn F, Ravetch JV. Science. 2005 Dec 2;310:1510. doi: 10.1126/science.1118948. [DOI] [PubMed] [Google Scholar]
- 8.Nimmerjahn F, Bruhns P, Horiuchi K, Ravetch JV. Immunity. 2005 Jul;23:41. doi: 10.1016/j.immuni.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 9.Clynes RA, Towers TL, Presta LG, Ravetch JV. Nat Med. 2000 Apr;6:443. doi: 10.1038/74704. [DOI] [PubMed] [Google Scholar]
- 10.Chu SY, et al. Mol Immunol. 2008 Sep;45:3926. doi: 10.1016/j.molimm.2008.06.027. [DOI] [PubMed] [Google Scholar]
- 11.Shields RL, et al. J Biol Chem. 2001 Mar 2;276:6591. doi: 10.1074/jbc.M009483200. [DOI] [PubMed] [Google Scholar]
- 12.McKenzie SE, et al. J Immunol. 1999 Apr 1;162:4311. [PubMed] [Google Scholar]
- 13.Horton HM, et al. J Immunol. 2011 Apr 1;186:4223. doi: 10.4049/jimmunol.1003412. [DOI] [PubMed] [Google Scholar]
- 14.French RR, Chan HT, Tutt AL, Glennie MJ. Nat Med. 1999 May;5:548. doi: 10.1038/8426. [DOI] [PubMed] [Google Scholar]
- 15.Robbiani DF, et al. Mol Cell. 2009 Nov 25;36:631. doi: 10.1016/j.molcel.2009.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cartron G, et al. Blood. 2002 Feb 1;99:754. doi: 10.1182/blood.v99.3.754. [DOI] [PubMed] [Google Scholar]
- 17.Musolino A, et al. J Clin Oncol. 2008 Apr 10;26:1789. doi: 10.1200/JCO.2007.14.8957. [DOI] [PubMed] [Google Scholar]
- 18.Tamura K, et al. Ann Oncol. 2010 Nov 25; [Google Scholar]
- 19.Xu Y, et al. J Immunol. 2003 Jul 15;171:562. doi: 10.4049/jimmunol.171.2.562. [DOI] [PubMed] [Google Scholar]
- 20.Zhang M, et al. Blood. 2006 Jul 15;108:705. doi: 10.1182/blood-2005-11-4607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chuntharapai A, et al. J Immunol. 2001 Apr 15;166:4891. doi: 10.4049/jimmunol.166.8.4891. [DOI] [PubMed] [Google Scholar]
- 22.Wilson NS, et al. Cancer Cell. 2011 Jan 18;19:101. doi: 10.1016/j.ccr.2010.11.012. [DOI] [PubMed] [Google Scholar]
- 23.Beatty GL, et al. Science. 2011 Mar 25;331:1612. doi: 10.1126/science.1198443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Takai T, Li M, Sylvestre D, Clynes R, Ravetch JV. Cell. 1994 Feb 11;76:519. doi: 10.1016/0092-8674(94)90115-5. [DOI] [PubMed] [Google Scholar]
- 25.Takai T, Ono M, Hikida M, Ohmori H, Ravetch JV. Nature. 1996 Jan 25;379:346. doi: 10.1038/379346a0. [DOI] [PubMed] [Google Scholar]
- 26.Boscardin SB, et al. J Exp Med. 2006 Mar 20;203:599. doi: 10.1084/jem.20051639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sazinsky SL, et al. Proc Natl Acad Sci U S A. 2008 Dec 23;105:20167. doi: 10.1073/pnas.0809257105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Heckman KL, Pease LR. Nat Protoc. 2007;2:924. doi: 10.1038/nprot.2007.132. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.