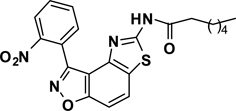
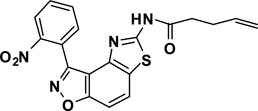
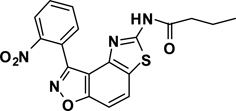
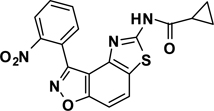
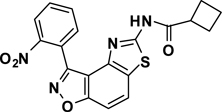
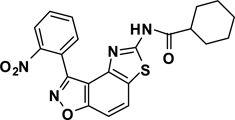
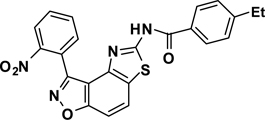
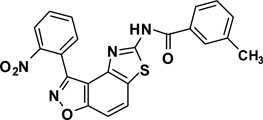
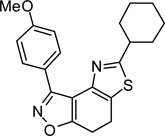
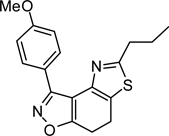
Abstract
The first examples of thiazolo[4,5-e]benzoisoxazoles are obtained from bromination of dihydrobenzo[d]isoxazol-4(5H)-one followed by cyclocondensation with thiourea, in the presence of DDQ. Analogously, cyclocondensation with thioamides provided 7,8-dihydrothiazolo[4,5-e]benzoisoxazoles. A 55-member library of these heterocycles is reported.
Introduction
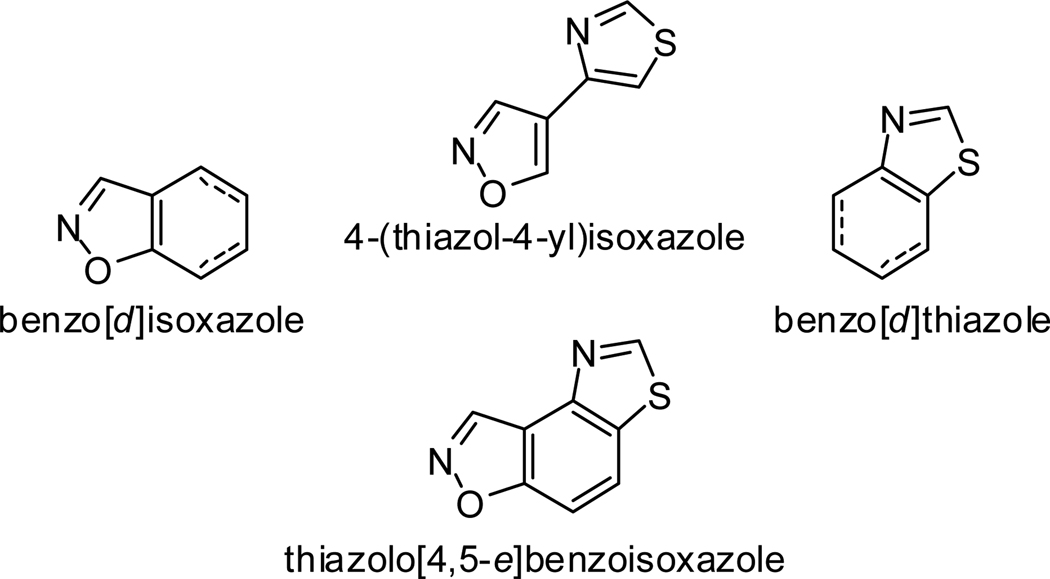
Benzoisoxazoles exhibit a wide variety of biological activities. For example, 3-aminobenzoisoxazole N,N′-diphenyl urea derivatives show inhibitory activity toward receptor tyrosine kinases (RTKs).1 Furthermore, benzo[d]isoxazole-containing compounds are therapeutic in hereditary angioedema,2 show HIV-1 non-nucleoside reverse transcriptase inhibitor activity,3 and dopamine D2 receptor activity.4 4-(Thiazol-4-yl)isoxazole-containing derivatives inhibit p38 kinase,5 have C. elegans toxicity,6 and express antimicrobial activity.7 Benzo[d]thiazole-containing compounds inhibit DNA synthesis,8 have antibacterial activity as FabK inhibitors,9 and serve as imaging agents in Alzheimer’s diagnosis.10
In the chemical literature, there are reports regarding the synthesis of imidazobenzothiazole,11 diaminobenzobis-thiazoles,12,13 and oxazolobenzoisoxazol,14 however, no literature coverage on the synthesis of thiazolo[4,5-e]benzoisoxazoles is known. In view of the pharmacological activities of benzo[d]isoxazole-, 4-(thiazol-4-yl)isoxazole-, and benzo[d]thiazole-containing compounds, we set out to develop a general method for the synthesis of thiazolo[4,5-e]benzoisoxazole, in which thiazole and isoxazole ring systems are fused to a benzene ring (Figure 1). A 55 compound collection of these heterocycles has been added to the National Institutes of Health molecular repository.
Figure 1.
Components and structure of thiazolo[4,5-e]benzoisoxazole scaffold.
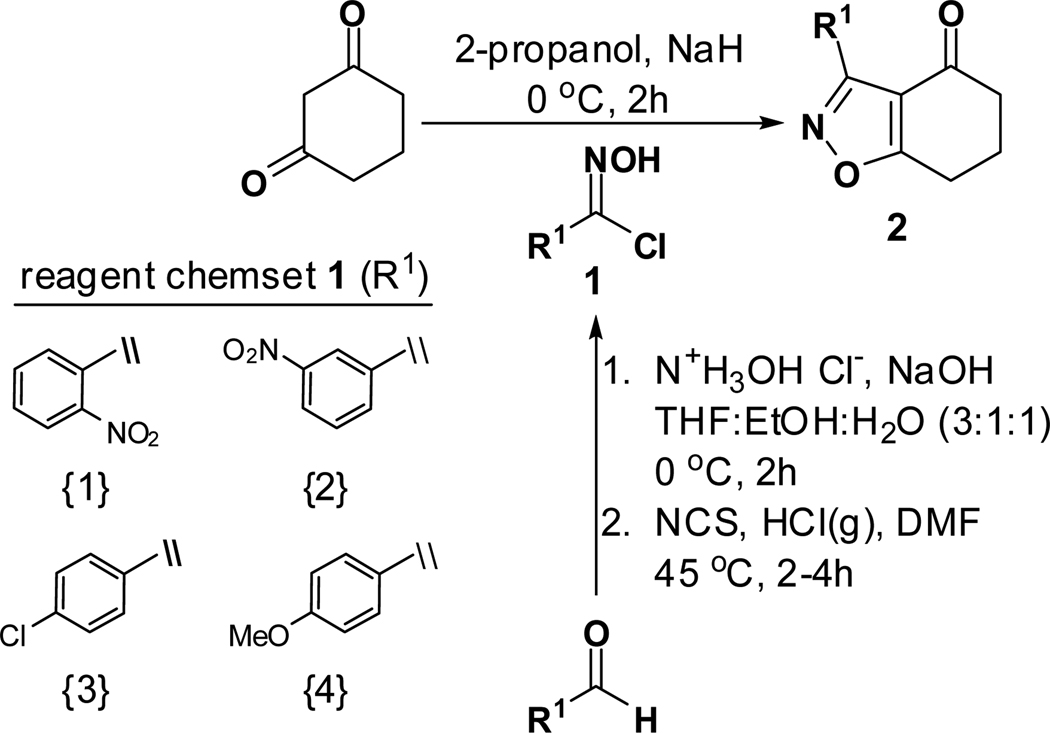
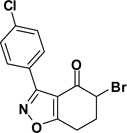
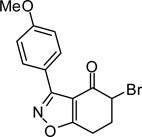
Formation of 6,7-dihydrobenzo[d]isoxazol-4(5H)-one 2 was carried out using Suzuki’s base-promoted cyclocondensation of a C-chlorooxime with cyclohexane-1,3-diones (Scheme 1).15 In the event, treatment of cyclohexane-1,3-dione with C-chlorooxime 1 in 2-propanol plus sodium hydride delivered tetrahydrobenzo[d]isoxazole 2 in 76–78% yield. C-Chlorooxime 1 was readily prepared in two steps from the corresponding aldehyde by condensation with hydroxylamine followed by C-chlorination with N-chlorosuccinimide (NCS); >95% overall yield.16 In contrast, attempts to form 5,6-dihydro-4H-cyclopenta[d]isoxazol-4-one by this method from cyclopentane-1,3-dione and 1 were not successful.17
Scheme 1.
Suzuki’s base-promoted cyclocondensation.
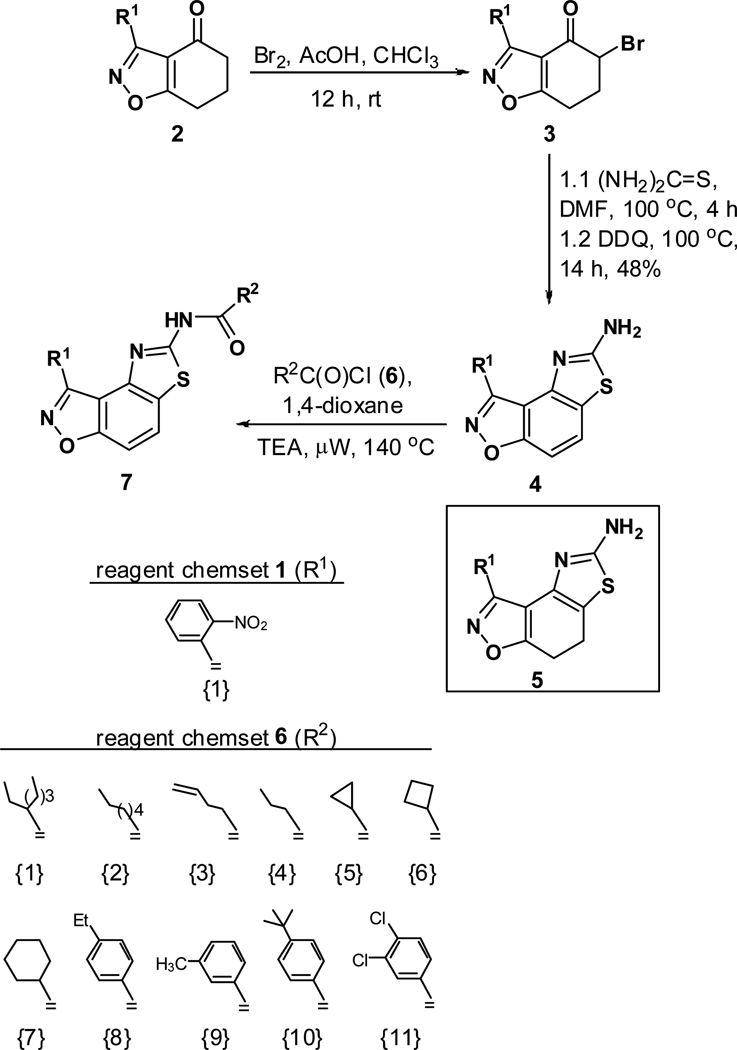
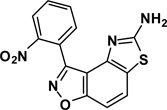
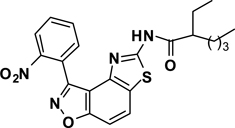
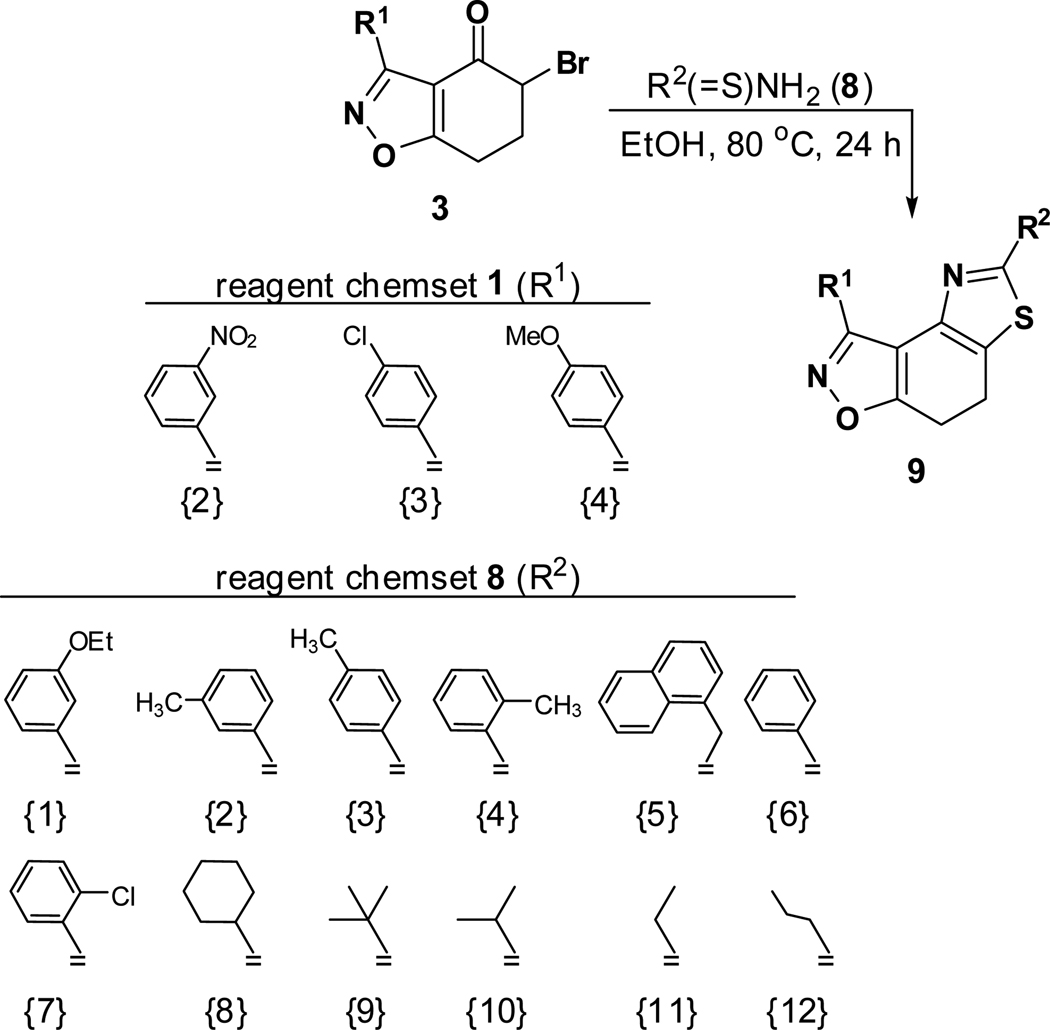
The second key transformation involves a ketone → α-bromoketone → thiazole sequence. While dihydrobenzo[d]isoxazol-4-ones of general structure 2 have been employed in ketoaldehyde benzoin cyclizations,18 reductive aminations,19 oxime formation,20 Schmidt reactions,21 their Cα-bromination is little studied.22 A survey of various keto α-bromination protocols (Amberlyst A-26 Br3−, CHCl3, rt; NBS, CHCl3, reflux; C5H5N+H Br3−, 30% HBr, HOAc) led to Br2 + HOAc in chloroform23 as the preferred method for the conversion of 2 to α-bromoketone 3 (78–80%; Scheme 2). Subsequent thiazole formation24 with concomitant aromatization of the resulting cyclohexadiene ring was accomplished by reaction of 3 with thiourea in either DMF (100 °C) or ethanol (refluxing) for 4 h followed by addition of a sub-stoichiometric amount (0.4 equiv.) of 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) (Scheme 2).25 In the absence of DDQ, a mixture of 4 and 5 (25:75, respectively) was obtained. A common characteristic of 4 in the subsequent N-acylation reaction is the low nucleophilicity of the thiazol-2-amine functionality, which dictates the use of high temperature and an excess of acylating agent. In the end, acylation of the amino functionality of 4 with different acid chlorides (6) to give amides 7 was performed best under microwave-assisted conditions.26
Scheme 2.
Thiazolo[4,5-e]benzoisoxazole formation.
In these reactions, 1,4-dioxane was used as the solvent and 2–4 equiv. of the acid chloride (6) was employed for maximum yield. The reaction of 4 with acyclic aliphatic acid chlorides resulted in formation of amides 7 in moderate to good yields (58–78%). Cycloalkanoyl chlorides provided the corresponding amides (7) in good yields (70%), but a low yield (40%) was obtained in the case of cyclobutanecarbonyl chloride. Acylation with aromatic acid chlorides delivered the subsequent amides in low to moderate yields (7; 41–56%). Attempts to improve the yields of the acylation reactions of aromatic acid chlorides using higher equivalents of 6 (6–8 equiv.) and/or longer reaction times were not successful.
To the best of our knowledge, only one example of the 7,8-dihydrothiazolo[4,5-e]benzoisoxazole scaffold (9) is reported in the chemical literature.27 In this work, we present a general method for the synthesis of this heterocycle by the reaction of α-bromoketone 3 with thioamide 828 in refluxing ethanol. Employing this method, a collection of heterocycles 9 was obtained in 83–94% yield, with no aromatisation of the cyclohexadiene ring.
In conclusion, the synthesis of thiazolo[4,5-e]benzoisoxazoles has been successfully achieved. Acylation of the amine functionality of 2-aminothiazolo[4,5-e]benzoisoxazole with various acid chlorides delivers the corresponding amides in moderate to good yields with aliphatic acid chlorides and in low to moderate yields with aromatic acid chlorides. In addition, a new method for the synthesis of the 7,8-dihydrothiazolo[4,5-e]benzoisoxazole scaffold is described.
Experimental section
General Experimental
All reagents were used without further purification. Glassware was oven-dried before use. To visualize reaction progress on TLC plates, a short-wave UV lamp was used. Flash column chromatography (FCC) was conducted using flash silica Gel (100–200 mesh) and the product was eluted using isocratic or stepwise solvent polarity gradient. All infrared spectra were determined on a Genesis II Mattson FT-IR. 1H and 13C NMR were measured in either DMSO-d6 or CDCl3 at 600 (or 400) and 150 (or 100) MHz, respectively.
3-(2-Nitrophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{1})
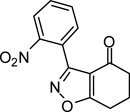
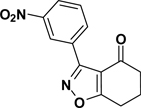
To a stirred solution of dry isopropanol (100 mL) at 0 °C was added NaH (0.46 g, 19.0 mmol), cyclohexane-1,3-dione (2.2 g, 20.0 mmol), and 2-nitrobenzaldoximoyl chloride (1{1}; 2.0 g, 10 mmol). The reaction temperature was raised gradually to room temperature with stirring over 2 h. Brine (50 mL) was added and the product was extracted with EtOAc (3 × 100 mL), dried over Na2SO4, filtered, and concentrated to give the titled compound as a crude material that was recrystalyzed from hexane. A small portion of the product was further purified, for analysis purposes, using column chromatography; 100% dichloromethane. Yield = 76%; colorless crystals; mp = 128 °C. IR (thin film, selected peaks) ν 3103, 1681, 1575, 1599, 1528, 1464, 1349, 726 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H10N2O4 259.0719, found 259.0720. 1H NMR (CDCl3, 600 MHz) δ 8.23 (dd, 1 H, J = 7.8, 1.2 Hz), 7.72-7.65 (m, 2 H), 7.60 (dd, 1 H, J = 7.8, 1.2 Hz), 3.08 (t, 2 H, J = 6.6), 2.48 (t, 2 H, J = 6.6 Hz), 2.25 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 192.3, 181.0, 158.0, 148.6, 133.6, 132.3, 131.3, 125.2, 123.5, 114.6, 37.7, 23.2, 22.3.
3-(3-Nitrophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{2})
The procedure given for 2{1} was followed with these modifications: 3-nitrobenzaldoximoyl chloride (1{2}; 2.0 g, 10 mmol). Yield = 76%; colorless crystals; mp = 139 °C. IR (thin film, selected peaks) ν 3103, 1689, 1568, 1525, 1464, 1435, 726 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H10N2O4 259.0719, found 259.0725. 1H NMR (CDCl3, 600 MHz) δ 9.03 (s, 1 H), 8.45 (dd, 1 H, J = 7.8, 1.2 Hz), 8.34 (m, 1 H), 7.66 (t, 1 H, J = 8.4 Hz), 3.12 (t, 2 H, J = 6.6), 2.65 (t, 2 H, J = 6.9 Hz), 2.30 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 192.2, 182.9, 158.3, 148.4, 135.2, 129.6, 129.3, 125.3, 124.5, 114.2, 38.7, 23.6, 22.1.
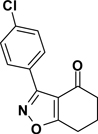
3-(4-Chlorophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{3})
The procedure given for 2{1} was followed with these modifications: 4-chlorobenzaldoximoyl chloride (1{3}; 2.0 g, 10.5 mmol). Yield = 78%; colorless crystals; mp = 93–95 °C. IR (thin film, selected peaks) ν 2964, 1685, 1583, 1503, 1454, 1427, 752 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H10ClNO2 248.0478, found 248.0472. 1H NMR (CDCl3, 600 MHz) δ 8.05 (d, 2 H, J = 9.0 Hz), 7.43 (d, 2 H, J = 9.0 Hz), 3.08 (t, 2 H, J = 6.6 Hz), 2.61 (t, 2 H, J = 6.4 Hz), 2.27 (p, 2 H, J = 6.6 Hz). 13C NMR (CDCl3, 150 MHz) δ 192.2, 182.5, 159.2, 136.9, 130.7, 128.9, 125.9, 114.2, 38.9, 23.6, 22.1.
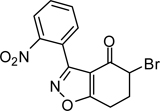
5-Bromo-3-(2-nitrophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (3{1})
To a stirred solution of 2{1} (0.2 g, 0.78 mmol) in CHCl3 (3 mL) at room temperature was added acetic acid (25 µL. 0.4 mmol) followed by the slow addition of Br2 (50 µL, 1.0 mmol). After 4–12 h the reaction mixture was washed with a saturated solution of sodium bisulfite (3 mL) and water (2 × 3 mL). The organic layer was dried over Na2SO4, filtered, and concentrated to give crude 3{1}, which was recrystallized using 30% EtOAc in hexane. A small portion of the product was further purified, for analysis purposes, using column chromatography; 100% dichloromethane. Yield = 78%; colorless crystals; mp = 131–132 °C. IR (thin film, selected peaks) ν 3083, 1694, 1598, 1524, 1461, 1347, 786, 730 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H9BrN2O4 336.9824, found 336.9824. 1H NMR (CDCl3, 600 MHz) δ 8.23 (dd, 1 H, J = 7.8, 1.8 Hz), 7.74-7.65 (m, 2 H), 7.61 (dd, 1 H, J = 7.8, 1.8 Hz), 4.48 (t, 1 H, J = 3.6 Hz), 3.29-3.24 (m, 1 H), 3.14-3.10 (m, 1 H), 2.62-2.52 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 185.1, 179.4, 158.9, 148.3, 133.8, 132.3, 131.6, 125.1, 122.8, 112.1, 47.8, 30.8, 20.6.
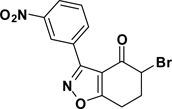
5-Bromo-3-(3-nitrophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (3{2})
The procedure given for 3{1} was followed with these modifications: 3-(3-nitrophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{2}; 0.2 g, 0.78 mmol). Yield = 78%; colorless crystals; mp = 157–159 °C. IR (thin film, selected peaks) ν 3083, 1682, 1565, 1521, 1471, 1432, 723 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H9BrN2O4 336.9824, found 336.9820. 1H NMR (CDCl3, 600 MHz) δ 8.98 (s, 1 H), 8.41 (d, 2 H, J = 7.8 Hz), 8.34 (m, 1 H), 7.67 (t, 1 H, J = 7.8 Hz), 4.66 (t, 1 H, J = 3.3), 3.37-3.31 (m, 1 H), 3.20-3.16 (m, 1 H), 2.63 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 185.3, 181.4, 159.2, 148.5, 135.2, 129.9, 128.9, 125.6, 124.5, 111.7, 48.8, 30.6, 20.8.
5-Bromo-3-(4-chlorophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (3{3})
The procedure given for 3{1} was followed with these modifications: 3-(4-chlorophenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{3}; 0.2 g, 0.81 mmol). Yield = 80%; colorless crystals; mp = 141 °C. IR (thin film, selected peaks) ν 2969, 1692, 1583, 1502, 1458, 1440, 758 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C13H9BrClNO2 325.9583, found 325.9579. 1H NMR (CDCl3, 600 MHz) δ 8.00 (d, 2 H, J = 9.0 Hz), 7.45 (d, 2 H, J = 9.0 Hz), 4.60 (t, 1 H, J = 3.6 Hz), 3.32 (m, 1 H), 3.14 (m, 1 H), 2.61-2.58 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 185.2, 180.8, 159.9, 137.2, 130.7, 128.9, 125.5, 111.6, 49.0, 30.6, 20.8.
3-(4-Methoxy)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (3{4})
The procedure given for 3{1} was followed with these modifications: 3-(4-methoxyphenyl)-6,7-dihydrobenzo[d]isoxazol-4(5H)-one (2{4}; 0.2 g, 0.82 mmol). Yield = 78%; colorless crystals; mp = 113 °C. IR (thin film, selected peaks) ν 2941, 1686, 1608, 1523, 1433, 1297, 1249, 1178, 1026, 832, 725 cm−1. HRMS (ESI) m/z [M+H]+ calcd for C14H12BrNO3 322.0079, found 322.0079. 1H NMR (CDCl3, 600 MHz) δ 8.00 (d, 2 H, J = 9.0 Hz), 6.97 (d, 2 H, J = 9.0 Hz), 4.60 (t, 1 H, J = 3.6 Hz), 3.41 (s, 3 H), 3.31-3.24 (m, 1 H), 3.11-3.05 (m, 1 H), 2.56-2.54 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 185.2, 180.5, 161.7, 160.4, 130.9, 119.3, 114.0, 111.5, 55.5, 49.3, 30.6, 20.8.
3-(2-Nitrophenyl)-5-aminothiazolo[4,5-e]benzoisoxazole (4{1})
To a stirred solution of 3{1} (5.6 g, 16.6 mmol) in DMF (80 mL) was added thiourea (1.36 g, 18.4 mmol), and the reaction mixture was heated to 100 °C for 4 h to yield a mixture of 4 and 5 (~25:75, respectively). DDQ (1.36 g, 6.8 mmol) was added and the reaction was allowed to proceed at the same temperature for additional 12 h, after which, TLC (40% ethyl acetate in hexane) showed complete conversion of 3{1} to aminothiazole 4{1}. Ice water (40 mL) was added to the reaction mixture and the product was extracted with ethyl acetate (2 × 80 mL). The organic layer was washed with a saturated solution of sodium bicarbonate (2 × 40 mL), water (30 mL), dried over Na2SO4, and stripped of solvents to give crude material. Column chromatography (neutral alumina, 40-0% hexane in ethyl acetate) gave 4{1} as a yellow solid, 48% yield, mp = 229–231 °C. IR (thin film, selected peaks) ν 3438, 3144, 1625, 1514, 1342, 1223, 907, 783, 743 cm−1. HRMS (ESI) m/z [M-H]+ calcd for C14H10N4O3S 313.0395, found 313.0390. 1H NMR (DMSO-d6, 600 MHz) δ 8.35 (d, 1 H, J= 8.4 Hz), 7.97-7.94 (m, 2 H), 7.90-7.88 (m, 2 H), 7.71 (2, 2 H), 7.45 (d, 1 H, J= 8.4 Hz); 13C NMR (DMSO-d6, 150 MHz) δ 170.6, 162.9, 155.3, 148.2, 145.9, 134.0, 132.9, 131.5, 125.1, 124.7, 123.8, 123.2, 111.0, 101.9.
A typical experimental procedure for the acylation reaction is the formation of 3-(2-Nitrophenyl)-5-(2-ethylhexanamide)-thiazolo[4,5-e]benzoisoxazole (7{1,1})
To a stirred solution of 4{1} (100 mg, 0.32 mmol) in 1,4-dioxane (3 mL) at RT was added TEA (220 µL, 1.6 mmol) followed by 2-ethylhexanoyl chloride (221 µL, 1.28 mmol) and the reaction vessel was sealed and it was placed in a microwave reactor at 140 °C for 80 min. Upon completion the reaction mixture was diluted with DCM, washed with H2O/brine, NaHCO3, dried over Na2SO4 and concentrated in vacuo to give crude material that was subjected to column chromatography (silica gel, 30% ethyl acetate in hexane) to deliver 7{1,1} in 66% yield. A yellow solid, mp = 180–181 °C. IR (thin film, selected peaks) ν 2954, 2927, 1676, 1521, 1345, 1268, 1166, 1070, 747 cm−1. LRMS (ESI) m/z: 439.11 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.80 (s, 1 H), 8.34 (dd, 1 H, J= 8.4, 1.2 Hz), 7.96 (d, 1 H, J= 8.4 Hz), 7.87 (dd, 1 H, J= 7.8, 1.2 Hz), 7.80 (dt, 1 H, J= 7.8, 1.2 Hz), 7.75 (dt, 1 H, J= 7.8, 1.2 Hz), 7.63 (d, 1 H, J= 8.4 Hz), 2.29 (m, 1 H), 1.77-1.69 (m, 2 H), 1.66-1.53 (m, 2 H), 1.32-1.26 (m, 4 H), 0.93 (t, 3 H, J= 7.2 Hz), 0.88 (t, 3 H, J= 7.2 Hz); 13C NMR (CDCl3, 150 MHz) δ 174.4, 164.2, 160.2, 155.7, 148.8, 141.9, 133.9, 133.4, 131.1, 127.4, 125.4, 125.0, 123.3, 113.3, 106.7, 49.6, 32.1, 29.7, 25.9, 22.8, 14.0, 12.0.
3-(2-Nitrophenyl)-5-(heptanamide)thiazolo[4,5-e]benzoisoxazole (7{1,2})
A yellow solid, 58% yield, mp = 151–152 °C. IR (thin film, selected peaks) ν 2927, 1682, 1521, 1342, 1276, 1220, 1158, 1070, 744 cm−1. LRMS (ESI) m/z: 425.13 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.03 (s, 1 H), 8.30 (dd, 1 H, J= 7.8, 1.2 Hz), 7.97 (d, 1 H, J= 9.0 Hz), 7.88 (dd, 1 H, J= 7.8, 1.2 Hz), 7.77 (dt, 1 H, J= 7.8, 1.2 Hz), 7.72 (dt, 1 H, J= 7.8, 1.2 Hz), 7.62 (d, 1 H, J= 9.0 Hz), 2.40 (t, 2 H, J= 7.2 Hz), 1.68 (m, 2 H), 1.26 (m, 6 H), 0.86 (t, 3 H, J= 6.6 Hz); 13C NMR (CDCl3, 150 MHz) δ 171.2, 164.2, 160.5, 155.6, 148.8, 141.9, 133.9, 133.5, 131.1, 127.4, 125.3, 124.9, 123.3, 113.3, 106.7, 36.6, 31.5, 28.9, 24.9, 22.6, 14.1.
3-(2-Nitrophenyl)-5-(4-pentenamide)thiazolo[4,5-e]benzoisoxazole (7{1,3})
A yellow solid, 69% yield, mp = 205–206 °C. IR (thin film, selected peaks) ν 2975, 1687, 1524, 1345, 1278, 1070, 910, 852, 747 cm−1. LRMS (ESI) m/z: 395.09 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.06 (s, 1 H), 8.31 (dd, 1 H, J= 7.8, 1.2 Hz), 7.96 (d, 1 H, J= 8.4 Hz), 7.88 (dd, 1 H, J= 7.8, 1.2 Hz), 7.79 (dt, 1 H, J= 7.8, 1.2 Hz), 7.74 (dt, 1 H, J= 7.8, 1.2 Hz), 7.62 (d, 1 H, J= 8.4 Hz), 5.82 (m, 1 H), 5.13 (dd, 1 H, J= 16.8, 1.2 Hz), 5.06 (dd, 1 H, J= 10.8, 1.2 Hz), 2.55 (t, 2 H, J= 7.6 Hz), 2.48 (t, 2 H, J= 7.6 Hz); 13C NMR (CDCl3, 150 MHz) δ 170.4, 164.2, 160.3, 155.6, 148.8, 141.9, 136.1, 133.9, 133.5, 131.1, 127.4, 125.3, 124.9, 123.3, 116.8, 113.3, 106.7, 35.6, 28.8.
3-(2-Nitrophenyl)-5-(4-butanamide)thiazolo[4,5-e]benzoisoxazole (7{1,4})
A yellow solid, 68% yield, mp = 222–224 °C. IR (thin film, selected peaks) ν 2965, 1684, 1580, 1524, 1348, 1276, 1169, 1067, 747 cm−1. LRMS (ESI) m/z: 383.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.13 (s, 1 H), 8.28 (dd, 1 H, J= 7.8, 1.2 Hz), 7.97 (d, 1 H, J= 9.0 Hz), 7.87 (dd, 1 H, J= 7.8, 1.2 Hz), 7.77 (dt, 1 H, J= 7.8, 1.2 Hz), 7.71 (dt, 1 H, J= 7.8, 1.2 Hz), 7.64 (d, 1 H, J= 9.0 Hz), 3.39 (t, 2 H, J= 7.8 Hz), 1.74 (m, 2 H), 0.96 (t, 3 H, J= 7.8 Hz), 13C NMR (CDCl3, 150 MHz) δ 171.1, 164.2, 160.5, 155.5, 148.7, 141.9, 133.9, 133.5, 131.1, 127.4, 125.3, 124.9, 123.3, 113.3, 106.7, 38.4, 18.5, 13.7.
3-(2-Nitrophenyl)-5-(cyclopropanamide)thiazolo[4,5-e]benzoisoxazole (7{1,5})
A yellow solid, 70% yield, mp = 252–253 °C. IR (thin film, selected peaks) ν 2956, 1671, 1527, 1388, 1350, 1270, 1150, 1073, 961, 747 cm−1. LRMS (ESI) m/z: 381.05 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.60 (s, 1 H), 8.20 (d, 1 H, J= 7.8 Hz), 7.97 (dd, 1 H, J= 9.0, 1.2 Hz), 7.89 (d, 1 H, J= 7.8 Hz), 7.72-7.63 (m, 3 H), 1.40 (m, 1 H), 1.13 (m, 2 H), 0.91(m, 2 H); 13C NMR (CDCl3, 150 MHz) δ 172.0, 164.3, 161.1, 155.5, 148.8, 141.8, 133.8, 133.5, 131.1, 127.3, 125.2, 124.9, 123.3, 113.4, 106.7, 15.2, 9.9.
3-(2-Nitrophenyl)-5-(cyclobutanamide)thiazolo[4,5-e]benzoisoxazole (7{1,6})
A yellow oil, 40% yield. IR (thin film, selected peaks) ν 3265, 2941, 1668, 1522, 1352, 1269, 1163, 1070, 776, 748 cm−1. LRMS (ESI) m/z: 395.07 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.76 (s, 1 H), 8.30 (d, 1 H, J= 7.8 Hz), 7.95 (d, 1 H, J= 8.4 Hz), 7.86 (d, 1 H, J= 7.2 Hz), 7.76 (t, 1 H, J= 7.2 Hz), 7.71 (t, 1 H, J= 7.8 Hz), 7.61 (d, 1 H, J= 8.4 Hz), 3.21 (t, 1 H, J= 8.4 Hz), 2.39 (m, 2 H), 2.25 (m, 2 H), 2.05-1.95 (m, 2 H); 13C NMR (CDCl3, 150) δ 172.9, 164.2, 160.5, 155.6, 148.9, 142.1, 133.8, 133.4, 131.1, 127.5, 125.3, 125.1, 123.3, 113.5, 106.6, 39.8, 25.2, 18.3.
3-(2-Nitrophenyl)-5-(cyclohexanamide)thiazolo[4,5-e]benzoisoxazole (7{1,7})
A yellow solid, 69% yield, mp = 221 °C. IR (thin film, selected peaks) ν 2926, 2855, 1678, 1522, 1446, 1347, 1264, 1160, 1072, 748 cm−1. LRMS (ESI) m/z: 423.11 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.66 (s, 1 H), 8.36 (dd, 1 H, J= 7.8, 1.2 Hz), 7.95 (d, 1 H, J= 9.0 Hz), 7.87 (dd, 1 H, J= 7.8, 1.2 Hz), 7.81 (dt, 1 H, J= 7.8, 1.2 Hz), 7.75 (dt, 1 H, J= 7.8, 1.2 Hz), 7.62 (d, 1 H, J= 9.0 Hz), 2.36 (m, 1 H), 1.97 (d, 2 H, J= 13.2 Hz), 1.85 (m, 2 H), 1.71 (m, 1 H), 1.58-1.52 (m, 2 H), 1.35-1.25 (m, 3 H); 13C NMR (CDCl3, 150) δ 173.9, 164.2, 160.4, 155.7, 148.9, 142.1, 133.8, 133.5, 131.0, 127.6, 125.4, 125.2, 123.2, 113.5, 106.6, 45.3, 29.3, 25.6, 25.5.
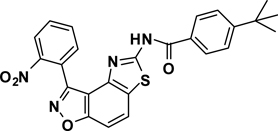
3-(2-Nitrophenyl)-5-(4-ethylbenzamide)thiazolo[4,5-e]-benzoisoxazole (7{1,8})
A yellow oil, 56% yield, LRMS (ESI) m/z: 445.09 [M+H+]. 1H NMR (DMSO-d6, 600 MHz) δ 12.67 (s, 1 H), 8.40 (d, 1 H, J= 7.8 Hz), 8.36 (d, 1 H, J= 9.0 Hz), 8.01-7.98 (m, 4 H), 7.97-7.94 (m, 1 H), 7.84 (d, 1 H, J= 8.4 Hz), 7.39 (d, 2 H, J= 8.4 Hz), 2.69 (q, 2 H, J= 7.8 Hz), 1.22 (t, 3 H, J= 7.8 Hz), 13C NMR (DMSO-d6, 150 MHz) δ 166.8, 164.2, 162.7, 155.7, 150.0, 149.1, 142.6, 134.9, 133.7, 132.4, 129.8, 129.4, 129.3, 128.6, 128.5, 127.7, 126.0, 125.0, 124.2, 113.2, 106.2, 28.8, 15.8.
3-(2-Nitrophenyl)-5-(3-methylbenzamide)thiazolo[4,5-e]-benzoisoxazole (7{1,9})
A yellow oil, 54% yield, LRMS (ESI) m/z: 430.08 [M+H+]. 1H NMR (DMSO-d6, 600 MHz) δ 12.78 (s, 1 H), 8.39 (m, 2 H), 7.99 (m, 2 H), 7.95 (m, 1), 7.89 (s, 1 H), 7.86 (dd, 2 H, J= 9.0, 1.2 Hz), 7.48-7.43 (m, 2 H), 2.39 (s, 3 H); 13C NMR (DMSO-d6, 150 MHz) δ 166.4, 163.5, 162.0, 155.1, 148.2, 141.9, 137.9, 134.3, 133.5, 132.9, 131.8, 131.6, 128.9, 128.4, 126.9, 125.7, 125.4, 124.4, 123.4, 112.5, 106.0, 20.9.
3-(2-Nitrophenyl)-5-(4-tert-butylbenzamide)thiazolo-[4,5-e]benzoisoxazole (7{1,10})
A yellow oil, 48% yield, LRMS (ESI) m/z: 473.11 [M+H+]. 1H NMR (DMSO-d6, 600 MHz) δ 12.63 (s, 1 H), 8.33 (d, 1 H, J= 7.8 Hz), 8.36 (d, 1 H, J= 7.8 Hz), 8.01-7.91 (m, 5 H), 7.80 (d, 1 H, J= 7.8 Hz), 7.52 (d, 2 H, J= 7.2 Hz), 1.24 (s, 9 H); 13C NMR (DMSO-d6, 150) δ 165.9, 163.4, 161.9, 155.9, 154.9, 148.2, 141.8, 134.1, 132.9, 131.6, 128.9, 128.4, 128.3, 126.9, 125.3, 125.2, 124.2, 123.4, 112.4, 105.9, 34.7, 30.7.
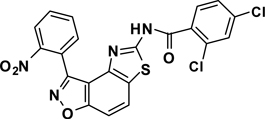
3-(2-Nitrophenyl)-5-(2,4-dichlorobenzamide)thiazolo-[4,5-e]benzoisoxazole(7{1,11})
A yellow solid, 41% yield, mp = 267–269 °C. LRMS (ESI) m/z: 485.01 [M+H+]. 1H NMR (DMSO-d6, 600 MHz) δ 12.99 (s, 1 H), 8.39 (m, 2 H), 7.99-7.90 (m, 3 H), 7.88 (d, 1 H, J= 8.4 Hz), 7.80 (d, 1 H, J= 1.8 Hz), 7.71 (d, 1 H, J= 8.4 Hz), 7.56 (dd, 1 H, J= 8.4, 1.8 Hz); 13C NMR (DMSO-d6, 150 MHz) δ 165.0, 163.4, 160.9, 155.2, 148.1, 141.7, 135.9, 134.3, 133.0, 132.9, 131.8, 131.5, 130.9, 129.3, 127.4, 126.9, 125.4, 124.5, 123.4, 112.6, 106.4.
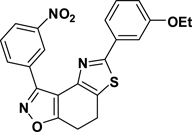
A typical experimental procedure for the synthesis of 7,8-dihydrothiazolo[4,5-e]benzoisoxazoles 9: 3-(3-Nitrophenyl)-5-(3-ethoxyphenyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,1})
To a solution of α-bromoketone 3{2} (150 mg, 0.45 mmol) in ethanol (10 mL) was added 3-ethoxybenzothioamide (98 mg, 0.54 mmol) and the reaction mixture was stirred under reflux for 24 h. Upon cooling to room temperature, water was added and the resulting precipitate was collected by filtration and washed with water. Further purification of the crude product was accomplished with column chromatography (SiO2, 10–20% ethyl acetate in hexane). Yield = 88%; yellow crystals; mp = 134–137 °C. IR (thin film, selected peaks) ν 2977, 1596, 1579, 1523, 1442, 720 cm−1. LRMS (ESI) m/z: 419.09 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.57 (t, 1 H, J= 2.4 Hz), 8.67 (dd, 1 H, J= 7.2, 1.2 Hz), 8.33 (m, 1 H), 7.65 (t, 1 H, J= 7.8 Hz), 7.55 (d, 1 H, J= 7.8 Hz), 7.41 (t, 1 H, J= 2.4 Hz), 7.34 (t, 1 H, J= 8.4 Hz), 6.94 (dd, 1 H, J= 7.8, 2.4 Hz), 4.06 (q, 2 H, J= 7.2 Hz), 3.32-3.23 (m, 4 H), 1.45 (t, 3 H, J= 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 171.5, 166.4, 159.5, 156.1, 148.5, 144.7, 134.8, 134.6, 130.9, 130.2, 129.3, 126.9, 125.0, 124.7, 118.9, 116.8, 111.8, 110.5, 63.8, 22.3, 22.1, 14.9.
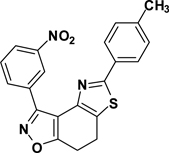
3-(3-Nitrophenyl)-5-(4-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,3})
Yield = 88%; yellow crystals; mp = 171–173 °C. IR (thin film, selected peaks) ν 2962, 1603, 1525, 1458, 1427, 723 cm−1. LRMS (ESI) m/z: 390.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.75 (t, 1 H, J= 1.8 Hz), 8.66 (d, 1 H, J= 8.4 Hz), 8.34 (m, 1 H), 7.89 (d, 2 H, J= 8.4 Hz), 7.66 (t, 1 H, J= 7.8 Hz), 7.26 (d, 2 H, J= 7.8 Hz), 3.32-3.29 (m, 2 H), 3.27-3.24 (m, 2 H), 2.39 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 171.5, 166.7, 156.1, 148.6, 144.6, 140.6, 134.7, 130.9, 130.8, 129.9, 129.3, 126.4, 126.3, 125.2, 124.8, 110.6, 22.3, 22.1, 21.6.
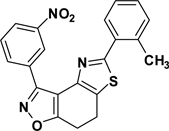
3-(3-Nitrophenyl)-5-(2-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,4})
Yield = 92%; yellow crystals; mp = 152 °C. IR (thin film, selected peaks) ν 2964, 1603, 1525, 1461, 1432, 719 cm−1. LRMS (ESI) m/z: 390.15 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.31 (t, 1 H, J= 2.4 Hz), 8.72 (m, 1 H), 8.31 (m, 1 H), 7.81 (m, 1 H), 7.64 (t, 1 H, J= 7.8 Hz), 7.30 (m, 3 H), 3.36 (m, 2 H), 3.28 (m, 2 H), 2.57 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 171.5, 166.1, 156.3, 148.5, 144.3, 136.2, 135.3, 132.6, 131.8, 130.9, 129.9, 129.7, 129.4, 127.4, 126.4, 124.6, 124.5, 110.7, 22.3, 22.1, 21.9.
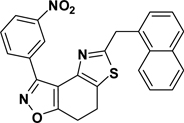
3-(3-Nitrophenyl)-5-(1-methylnaphthyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,5})
Yield = 94%; yellow crystals; mp = 149–151 °C. IR (thin film, selected peaks) ν 1603, 1525, 1432, 1349, 718 cm−1. LRMS (ESI) m/z: 439.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.73 (s, 1 H), 8.62 (dd, 1 H, J = 7.8, 1.2 Hz), 8.34-8.32 (m, 1 H), 8.15 (m, 1 H), 7.87 (t, 1 H, J = 6.0 Hz), 7.84 (d, 1 H, J = 7.8 Hz), 7.57 (t, 1 H, J = 7.2 Hz), 7.54 (d, 1 H, J = 7.2 Hz), 7.48 (t, 3 H, J = 6.0 Hz), 4.80 (s, 2 H), 3.18-3.09 (m, 4 H). 13C NMR (CDCl3, 150 MHz) δ 171.3, 169.1, 156.0, 148.5, 143.2, 134.6, 134.1, 133.9, 131.8, 130.8, 129.3, 128.9, 128.4, 127.8, 126.9, 126.6, 126.0, 125.7, 125.2, 124.5, 124.2, 110.6, 37.7, 22.3, 22.2.
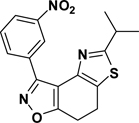
3-(3-Nitrophenyl)-5-isopropy-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,10})
Yield = 86%; yellow crystals; mp = 102–105 °C. IR (thin film, selected peaks) ν 2966, 2870, 1727, 1578, 1527, 723 cm−1. LRMS (ESI) m/z: 342.11 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.66 (t, 1 H, J= 2.4 Hz), 8.66 (d, 1 H, J= 9.0 Hz), 8.31-8.30 (m, 1 H), 7.33 (t, 1 H, J= 7.8 Hz), 3.31 (sp, 1 H, J= 7.2 Hz), 3.27-3.20 (m, 4 H), 1.43 (d, 6 H, J = 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 176.3, 171.3, 156.1, 148.5, 143.1, 134.6, 130.9, 129.3, 125.3, 125.2, 124.5, 110.7, 33.5, 23.1, 22.4, 22.1.
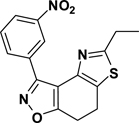
3-(3-Nitrophenyl)-5-ethyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,11})
Yield = 94%; yellow crystals; mp = 85–86 °C. IR (thin film, selected peaks) ν 2969, 2922, 1604, 1577, 1524, 1455, 1432, 717 cm−1. LRMS (ESI) m/z: 328.07 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.70 (t, 1 H, J= 1.8 Hz), 8.64 (d, 1 H, J= 9.6 Hz), 8.31-8.29 (m, 1 H), 7.62 (t, 1 H, J= 7.8 Hz), 3.23 (m, 4 H), 3.04 (q, 2 H, J= 7.8 Hz), 1.42 (t, 3 H, J= 7.8 Hz). 13C NMR (CDCl3, 150 MHz) δ 171.3, 171.1, 156.0, 148.5, 143.2, 134.5, 130.9, 129.3, 125.6, 125.3, 124.5, 110.7, 27.0, 22.4, 22.1, 14.1.
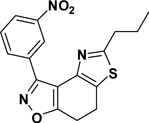
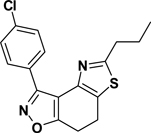
3-(3-Nitrophenyl)-5-propyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{2,12})
Yield = 89%; yellow crystals; mp = 105–108 °C. IR (thin film, selected peaks) ν 2969, 2927, 2876, 1608, 1574, 1526, 1433, 719 cm−1. LRMS (ESI) m/z: 342.18 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 9.69 (t, 1 H, J= 1.8 Hz), 8.63 (d, 1 H, J= 7.8 Hz), 8.3 (m, 1 H), 7.62 (t, 1 H, J= 8.4 Hz), 3.23 (m, 4 H), 2.98 (t, 2 H, J= 7.2 Hz), 1.85 (sextet, 2 H, J= 7.2 Hz), 1.04 (t, 3 H, J= 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 171.3, 169.6, 156.0, 148.5, 143.2, 134.5, 130.9, 129.3, 125.6, 125.3, 124.5, 110.7, 35.6, 23.3, 22.4, 22.0, 13.8.
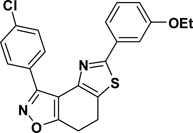
3-(4-Chlorophenyl)-5-(3-ethoxyphenyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,1})
Yield = 84%; yellow crystals; mp = 91–93 °C. IR (thin film, selected peaks) ν 2974, 2922, 1598, 1504, 1471, 1437, 727 cm−1. LRMS (ESI) m/z: 409.04 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.35 (d, 2 H, J = 8.4 Hz), 7.43 (m, 3 H), 7.39 (d, 1 H, J = 7.2 Hz), 7.30 (t, 1 H, J = 8.4 Hz), 6.93 (dd, 1 H, J = 8.4, 2.4 Hz), 4.08 (q, 2 H, J = 7.2 Hz), 3.22-3.25 (m, 2 H), 3.16-3.19(m, 2 H), 1.47 (t, 3 H, J = 6.6 Hz). 13C NMR (CDCl3, 150 MHz) δ 170.8, 165.6, 159.4, 157.0, 144.9, 135.9, 134.7, 130.1, 128.4, 127.4, 126.5, 118.4, 116.8, 111.2, 110.3, 63.6, 22.2, 22.0, 14.9.
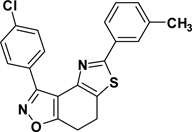
3-(4-Chlorophenyl)-5-(3-toluyl)-7,8-dihydrothiazolo[4,5-e]-benzoisoxazole (9{3,2})
Yield = 94%; yellow crystals; mp = 147–150 °C. IR (thin film, selected peaks) ν 2922, 2850, 1670, 1500, 1429, 731 cm−1. LRMS (ESI) m/z: 379.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.42 (d, 2 H, J = 8.4 Hz), 7.67 (d, 2 H, J = 6.6 Hz), 7.47 (d, 2 H, J = 8.4 Hz), 7.33(t, 1 H, J =7.2 Hz), 7.22 (d, 1 H, J = 7.2 Hz), 3.27-3.17 (m, 4 H), 2.43 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 170.8, 165.9, 157.0, 145.0, 138.8, 135.9, 133.5, 130.9, 130.8, 129.0, 128.5, 127.4, 126.7, 126.3, 123.2, 110.3, 22.2, 22.1, 21.6.
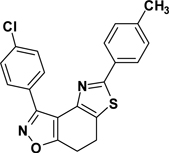
3-(4-Chlorophenyl)-5-(4-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,3})
Yield = 86%; colorless crystals; mp = 174–177 °C. IR (thin film, selected peaks) ν 2922, 2845, 1686, 1568, 1502, 1428, 734 cm−1. LRMS (ESI) m/z: 379.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.40 (d, 2 H, J = 8.4 Hz), 7.78 (d, 2 H, J = 7.8 Hz), 7.48 (d, 2 H, J = 8.4 Hz), 7.25 (d, 2 H, J = 7.8 Hz), 3.30-3.27 (m, 2 H), 3.24-3.21 (m, 2 H), 2.4 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 170.9, 166.1, 157.2, 145.1, 140.4, 136.0, 131.1, 130.9, 129.9, 128.6, 127.5, 126.1, 126.0, 110.5, 22.4, 22.2, 21.6.
3-(4-Chlorophenyl)-5-(2-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,4})
Yield = 89%; colorless crystals; mp = 148–146 °C. IR (thin film, selected peaks) ν 2958, 2917, 2850, 1602, 1571, 1499, 1470, 1426, 753 cm−1. LRMS (ESI) m/z: 379.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.35 (d, 2 H, J = 9.0 Hz), 7.70 (d, 1 H, J = 7.8 Hz), 7.43, (d, 2 H, J = 9.0 Hz), 7.34-7.26 (m, 3 H), 3.30 (m, 2 H), 3.23(m, 2 H), 2.65 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 170.9, 165.9, 157.2, 144.9, 136.5, 135.9, 132.7, 131.9, 130.8, 129.7, 129.5, 128.6, 127.5, 126.9, 126.4, 110.5, 22.3, 22.2, 22.0.
3-(4-Chlorophenyl)-5-(1-methylnaphthyl)-7,8-dihydrothiazolo-[4,5-e]benozisoxazole (9{3,5})
Yield = 91%; yellow crystals; mp = 112–115 °C. IR (thin film, selected peaks) ν 2917, 2850, 1602, 1568, 1502, 1434, 726 cm−1. LRMS (ESI) m/z: 429.08 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.26 (d, 2 H, J= 9.0 Hz), 8.13-8.12 (m, 1 H), 7.92-7.90 (m, 1 H), 7.86 (dd, 1 H, J= 7.2, 1.8 Hz), 7.53-7.48 (m, 4 H), 7.38 (d, 2 H, J= 9.0 Hz), 4.74 (s, 2 H), 3.08 (m, 4 H). 13C NMR (CDCl3, 150 MHz) δ 170.7, 168.0, 157.0, 143.8, 135.8, 134.1, 134.0, 131.8, 130.7, 128.9, 128.6, 128.4, 127.7, 127.4, 126.5, 126.3, 126.0, 125.7, 124.2, 110.4, 37.5, 22.2, 21.9.
3-(4-Chlorophenyl)-5-phenyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,6})
Yield = 91%; yellow crystals; mp = 188–191 °C. IR (thin film, selected peaks) ν 2922, 2855, 1602, 1570, 1500, 1458, 1436, 733 cm−1. LRMS (ESI) m/z: 364.98 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.40 (d, 2 H, J = 9.0 Hz), 7.88 (d, 2 H, J = 8.4 Hz), 7.49 (d, 2 H, J = 8.4 Hz), 7.47-7.42 (m, 3 H), 3.28 (m, 2 H), 3.22 (m, 2 H). 13C NMR (CDCl3, 150 MHz) δ 170.9, 165.8, 157.2, 145.3, 136.0, 133.7, 130.9, 130.1, 129.2, 128.6, 127.4, 126.5, 126.1, 110.4, 22.3, 22.2.
3-(4-Chlorophenyl)-5-(2-Chlorophenyl)-7,8-dihydrothiazolo[4,5-e]-benzoisoxazole (9{3,7})
Yield = 83%; yellow solid; mp = 152–155 °C. IR (thin film, selected peaks) ν 1603, 1569, 1499, 734 cm−1. LRMS (ESI) m/z: 298.98 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.39 (d, 2 H, J = 9.0 Hz), 8.16 (dd, 1 H, J = 7.8, 1.2 Hz), 7.49 (dd, 1 H, J = 7.8, 1.2 Hz), 7.46 (d, 2 H, J = 9.0 Hz), 7.38 (m, 1 H), 7.33 (m, 1 H), 3.38 (t, 2 H, J = 7.8 Hz), 3.24 (t, 2 H, J = 7.8 Hz). 13C NMR (CDCl3, 150 MHz) δ 171.1, 161.0, 157.2, 144.3, 136.0, 131.8, 131.6, 131.0, 130.8, 130.4, 130.3, 128.64, 128.63, 127.4, 127.3, 110.2, 22.3, 22.0.
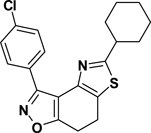
3-(4-Chlorophenyl)-5-cyclohexyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,8})
Yield = 92%; colorless crystals; mp = 139–142 °C. IR (thin film, selected peaks) ν 2927, 2850, 1600, 1568, 1445, 1432, 734 cm−1. LRMS (ESI) m/z: 371.12 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.39 (d, 2 H, J = 8.4 Hz), 7.43 (d, 2 H, J = 8.4 Hz), 3.17 (m, 4 H), 2.95 (m, 1 H), 2.15 (d, 2 H, J = 10.8 Hz), 1.87 (t, 1 H, J = 3.0 Hz), 1.85 (t, 1 H, J= 3.0 Hz), 1.74 (m, 1 H), 1.54 (m, 2 H), 1.44 (m, 2 H), 1.30 (m, 1 H). 13C NMR (CDCl3, 150 MHz) δ 174.6, 170.7, 156.9, 143.4, 135.8, 130.8, 128.5, 127.5, 124.8, 110.6, 42.4, 33.6, 26.03, 26.0, 22.4, 22.0.
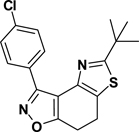
3-(4-Chlorophenyl)-5-tert-butyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,9})
Yield = 86%; colorless crystals; mp = 134–136 °C. IR (thin film, selected peaks) ν 2969, 1604, 1502, 1432, 726cm−1. LRMS (ESI) m/z: 345.15 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.47 (d, 2 H, J = 8.4 Hz), 7.43 (d, 2 H, J = 8.4 Hz), 3.17 (m, 4 H), 1.44 (s, 9 H); 13C NMR (CDCl3, 150 MHz) δ 178.9, 170.7, 156.9, 143.2, 135.8, 130.9, 128.4, 127.6, 125.1, 110.6, 37.9, 30.9, 22.4, 22.0.
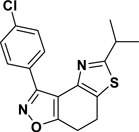
3-(4-Chlorophenyl)-5-isopropyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,10})
Yield = 92%; colorless crystals; mp = 115–119 °C. IR (thin film, selected peaks) ν 2964, 2922, 2865, 1600, 1568, 1500, 1478, 1430, 734 cm−1. LRMS (ESI) m/z: 331.19 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.40 (d, 2 H, J = 8.4 Hz), 7.43 (d, 2 H, J = 8.4 Hz), 3.26 (sp, 1 H, J = 6.6 Hz), 3.18 (m, 4 H), 1.42 (d, 6 H, J = 6.6 Hz). 13C NMR (CDCl3, 150 MHz) δ 175.6, 170.7, 157.0, 143.5, 135.9, 130.8, 128.5, 127.5, 125.0, 110.6, 33.4, 23.0, 22.4, 22.1.
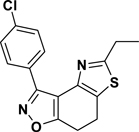
3-(4-Chlorophenyl)-5-ethyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,11})
Yield = 87%; colorless crystals; mp = 119–122 °C. IR (thin film, selected peaks) ν 2974, 1602, 1568, 1499, 1431, 726 cm−1. LRMS (ESI) m/z: 317.09 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.33 (d, 2 H, J = 8.4 Hz), 7.43 (d, 2 H, J = 8.4 Hz), 3.18 (m, 4 H), 3.00 (q, 2 H, J = 7.2 Hz), 1.40 (t, 3 H, J = 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 170.8, 170.5, 157.0, 143.6, 135.9, 130.8, 128.6, 127.5, 125.2, 110.5, 27.1, 22.4, 22.0, 13.9.
3-(4-Chlorophenyl)-5-propyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{3,12})
Yield = 93%; colorless crystals; mp = 117–120 °C. IR (thin film, selected peaks) ν 2958, 2933, 2876, 1603, 1568, 1502, 1431, 736 cm−1. LRMS (ESI) m/z: 331.12 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.33(d, 2 H, J= 8.4 Hz), 7.43(d, 2H, J= 8.4 Hz), 3.18 (m, 4 H), 2.94 (t, 2 H, J= 7.2 Hz), 1.84 (m, 2 H), 1.05(t, 3 H, J= 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 170.8, 168.9, 157.0, 143.7, 135.9, 130.7, 128.6, 127.5, 125.2, 110.5, 35.5, 23.1, 22.4, 22.0, 13.8.
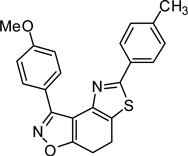
3-(4-Methoxy)-5-(3-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{4,2})
Yield = 87%; yellow crystals; mp = 119–121 °C. IR (thin film, selected peaks) ν 2926, 1608, 1513, 1428, 1249, 1175, 1069, 834, 728 cm−1. LRMS (ESI) m/z: 375.15 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.40 (d, 2 H, J= 8.4 Hz), 7.71 (m, 2 H), 7.32 (m, 1 H), 7.21 (d, 1 H, J= 7.8 Hz), 7.04 (d, 2 H, J= 8.4 Hz), 3.89 (s, 3 H), 3.29-3.19 (m, 4 H), 2.42 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 170.5, 165.7, 161.1, 157.8, 145.6, 138.7, 133.7, 131.0, 130.8, 129.0, 126.9, 126.1, 123.3, 121.4, 113.7, 110.3, 55.5, 22.4, 22.2, 21.6.
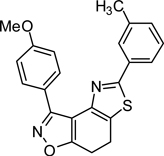
3-(4-Methoxy)-5-(4-toluyl)-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{4,3})
Yield = 88%; yellow crystals; mp = 151–153 °C. IR (thin film, selected peaks) ν 2916, 1608, 1514, 1428, 1246, 1178, 1031, 832, 811 cm−1. LRMS (ESI) m/z: 375.15 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.40 (d, 2 H, J= 9.0 Hz), 7.81 (d, 2 H, J= 8.4 Hz), 7.26 (m, 2 H), 7.05 (d, 2 H, J= 9.0 Hz), 3.91 (s, 3 H), 3.31-3.20 (m, 4 H), 2.41 (s, 3 H). 13C NMR (CDCl3, 150 MHz) δ 170.5, 165.8, 161.0, 157.8, 145.6, 140.2, 131.2, 131.0, 129.7, 126.1, 125.7, 121.5, 113.7, 110.3, 55.5, 22.4, 22.2, 21.6.
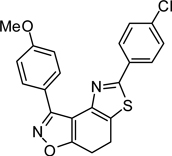
3-(4-Methoxy)-5-(4-chlorophenyl)-7,8-dihydrothiazolo[4,5-e]-benzoisoxazole (9{4,7})
Yield = 89%; yellow crystals; mp = 174 °C. IR (thin film, selected peaks) ν 2936, 1608, 1512, 1413, 1244, 1178, 1067, 1031, 826, 728 cm−1. LRMS (ESI) m/z: 395.09 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.33 (d, 2 H, J= 8.4 Hz), 7.81 (d, 2 H, J= 8.4 Hz), 7.39 (d, 2 H, J= 8.4 Hz), 7.03 (d, 2 H, J= 8.4 Hz), 3.90 (s, 3 H), 3.27-3.19 (m, 4 H). 13C NMR (CDCl3, 150 MHz) δ 170.6, 164.3, 161.2, 157.9, 146.0, 135.8, 132.4, 131.0, 129.4, 127.4, 126.7, 121.4, 113.8, 110.3, 55.6, 22.4, 22.3.
3-(4-Methoxy)-5-cyclohexyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{4,8})
Yield = 88%; yellow crystals; mp = 137 °C. IR (thin film, selected peaks) ν 2916, 1610, 1514, 1428, 1254, 1175, 1031, 832 cm−1. LRMS (ESI) m/z: 367.17 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.36 (d, 2 H, J= 9.0 Hz), 6.98 (d, 2 H, J= 9.0 Hz), 3.91 (s, 3 H), 3.21-3.13 (m, 4 H), 2.96 (m, 1 H), 2.17 (m, 2 H), 1.87-1.84 (m, 2 H), 1.75-1.71 (m, 1 H), 1.64-1.53 (m, 2 H), 1.46-1.39 (m, 2 H), 1.32-1.28 (m, 1 H). 13C NMR (CDCl3, 150 MHz) δ 174.5, 170.4, 161.0, 157.8, 143.9, 131.1, 124.5, 121.6, 113.8, 110.6, 55.5, 42.6, 33.7, 26.1, 22.6, 22.2.
3-(4-Methoxy)-5-isopropyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{4,10})
Yield = 86%; yellow crystals; mp = 108–110 °C. IR (thin film, selected peaks) ν 2962, 1608, 1517, 1428, 1246, 1175, 1031, 832 cm−1. LRMS (ESI) m/z: 327.16 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.36 (d, 2 H, J= 9.0 Hz), 6.98 (d, 2 H, J= 9.0 Hz), 3.90 (s, 3 H), 3.27 (m, 1 H), 3.21-3.14 (m, 4 H), 1.41 (d, 6 H, J= 6.6 Hz). 13C NMR (CDCl3, 150 MHz) δ 175.3, 170.3, 161.0, 157.7, 143.9, 131.0, 124.6, 121.5, 113.7, 110.5, 55.4, 33.4, 23.1, 22.5, 22.1.
3-(4-Methoxy)-5-propyl-7,8-dihydrothiazolo[4,5-e]benzoisoxazole (9{4,12})
Yield = 85%; yellow crystals; mp = 91 °C. IR (thin film, selected peaks) ν 2951, 1608, 1517, 1431, 1249, 1178, 1031, 832 cm−1. LRMS (ESI) m/z: 327.16 [M+H+]. 1H NMR (CDCl3, 600 MHz) δ 8.32 (d, 2 H, J= 9.0 Hz), 6.98 (d, 2 H, J= 9.0 Hz), 3.88 (s, 3 H), 3.20-3.16 (m, 4 H), 2.96 (t, 2 H, J= 7.2 Hz), 1.84 (m, 2 H), 1.05 (t, 3 H, J= 7.2 Hz). 13C NMR (CDCl3, 150 MHz) δ 170.4, 168.8, 161.1, 157.8, 144.3, 131.0, 124.9, 121.6, 113.9, 110.5, 55.5, 35.6, 23.3, 22.6, 22.2, 13.9.
Supplementary Material
Scheme 3.
7,8-Dihydrothiazolo[4,5-e]benzoisoxazole formation.
Acknowledgements
This work was supported by the National Science Foundation (CHE-0614756) and the National Institute for General Medical Sciences (GM076151). NMR spectrometers used in this work were funded in part by the National Science Foundation (CHE-0443516 and CHE-9808183).
Footnotes
Supporting Information Available. 1H- and 13C-NMR spectra are presented. This information is available free of charge via the Internet at http://pubs.acs.org.
REFERENCES
- 1.Ji Z, Ahmed AA, Albert DH, Bouska JJ, Bousquet PF, Cunha GA, Diaz G, Glaser KB, Guo J, Harris CM, Li J, Marcotte PA, Moskey MD, Oie T, Pease L, Soni NB, Stewart KD, Davidsen SK, Michaelides MR. J. Med. Chem. 2008;51:1231–1241. doi: 10.1021/jm701096v. [DOI] [PubMed] [Google Scholar]
- 2.Thon V, Harle P, Scholmerich J, Kuklinek P, Lokaj J, Straub RH. Allergy. Vol. 62. Oxford: United Kingdom; 2007. pp. 1320–1325. [DOI] [PubMed] [Google Scholar]
- 3.Cullen MD, Deng B-L, Hartman TL, Watson KM, Buckheit RW, Jr, Pannecouque C, De Clercq E, Cushman M. J. Med. Chem. 2007;50:4854–4867. doi: 10.1021/jm070382k. [DOI] [PubMed] [Google Scholar]
- 4.Barcelo M, Ravina E, Masaguer CF, Dominguez E, Areias FM, Brea J, Loza MI. Bioorg. Med. Chem. Lett. 2007;17:4873–4877. doi: 10.1016/j.bmcl.2007.06.045. [DOI] [PubMed] [Google Scholar]
- 5.Severance DL, Gardiner EMM, Noble SA, Lou B, Borchardt AJ, Kahraman M, Roppe JR, Siegel DL, Scranton SA. WO 2006116355 A1. 2006 [Google Scholar]
- 6.Donohue BA, Michelotti EL, Reader JC, Reader V, Stirling M, Tice CM. J. Comb. Chem. 2002;4:23–32. doi: 10.1021/cc010035a. [DOI] [PubMed] [Google Scholar]
- 7.Forood B. WO 2000045635 A1. 2000 [Google Scholar]
- 8.Kijjoa A, Wattanadilok R, Campos N, Nascimento MSJ, Pinto M, Herz W. Marine Drugs. 2007;5:6–22. doi: 10.3390/md502006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kitagawa H, Ozawa T, Takahata S, Iida M. Bioorg. Med. Chem. Lett. 2007;17:4982–4986. doi: 10.1016/j.bmcl.2007.06.040. [DOI] [PubMed] [Google Scholar]
- 10.Bacskai BJ, Hickey GA, Skoch J, Kajdasz ST, Wang Y, Huang G-F, Mathis CA, Klunk WE, Hyman BT. Proc. Natl. Acad. Sci. U. S. A. 2003;100:12462–12467. doi: 10.1073/pnas.2034101100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rao VS, Rao SSP, Reddy AP, Veeranagaiah V. Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem. 1993;32B:322–327. [Google Scholar]
- 12.Landquist JK. J. Chem. Soc., Sect. C: Org. 1967;21:2212–2220. [Google Scholar]
- 13.Cullen E, Becker R, Freter K, LeClerq T, Possanza G, Wong HC. J. Med. Chem. 1992;35:350–361. doi: 10.1021/jm00080a022. [DOI] [PubMed] [Google Scholar]
- 14.Rao EB, Kumari SS, Rao KM. Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem. 1987;26B:366–367. [Google Scholar]
- 15.(a) Takikawa H, Hikita K, Suzuki K. Synlett. 2007:2252–2256. [Google Scholar]; (b) Matsuura T, Bode JW, Hachisu Y, Suzuki K. Synlett. 2003:1746–1748. [Google Scholar]; (c) Bode JW, Suzuki K. Tetrahedron Lett. 2003;44:3559–3563. [Google Scholar]; (d) Bode JW, Hachisu Y, Matsuura T, Suzuki K. Tetrahedron Lett. 2003;44:3555–3558. [Google Scholar]; (e) Bode JW, Hachisu Y, Matsuura T, Suzuki K. Org. Lett. 2003;5:391–394. doi: 10.1021/ol027283f. [DOI] [PubMed] [Google Scholar]
- 16.Liu K-C, Shelton BR, Howe RK. J. Org. Chem. 1980;45:3916–3918. [Google Scholar]
- 17.(a) Attempts to prepare 5,6-dihydro-4H-cyclopenta[d]isoxazol-4-one employing cyclopentane-1,3-dione failed. (b) There are numerous literature examples of this cyclocondensation reaction employing various 1,3-diones; for representative examples, see 17c–e. Matsuura T, Bode JW, Hachisu Y, Suzuki K. Synlett. 2003:1746–1748. Umesha KB, Kumar K Ajay, Rai K M Lokanatha. Synth. Commun. 2002;32:1841–1846. Giannella M, Gualtieri F, Pigini M. Farmaco Ed. Sci. 1967;22:333–341.
- 18.(a) Takikawa H, Hachisu Y, Bode JW, Suzuki K. Angew. Chem., Int. Ed. 2006;45:3492–3494. doi: 10.1002/anie.200600268. [DOI] [PubMed] [Google Scholar]; (b) Hachisu Y, Bode JW, Suzuki K. J. Am. Chem. Soc. 2003;125:8432–8433. doi: 10.1021/ja035308f. [DOI] [PubMed] [Google Scholar]
- 19.Clausen RP, Moltzen EK, Perregaard J, Lenz SM, Sanchez C, Falch E, Frolund B, Bolvig T, Sarup A, Larsson OM, Schousboe A, Krogsgaard-Larsen P. Bioorg. Med. Chem. 2005;13:895–908. doi: 10.1016/j.bmc.2004.10.029. [DOI] [PubMed] [Google Scholar]
- 20.Falch E, Perregaard J, Frolund B, Sokilde B, Buur A, Hansen LM, Frydenvang K, Brehm L, Bolvig T, Larsson OM, Sanchez C, White HS, Schousboe A, Krogsgaard-Larsen P. J. Med. Chem. 1999;42:5402–5414. doi: 10.1021/jm9904452. [DOI] [PubMed] [Google Scholar]
- 21.Shutske GM. J. Heterocycl. Chem. 1990;27:1617–1621. [Google Scholar]
- 22.Lakhvich FA, Petrusevich II, Buravskaya TN. Vestsi Nats. Akad. Navuk Belarusi, Ser. Khim. Navuk. 1989:64–68. [Google Scholar]
- 23.Chiarino D, Grancini G, Frigeni V, Biasini I, Carenzi A. J. Med. Chem. 1991;34:600–605. doi: 10.1021/jm00106a020. [DOI] [PubMed] [Google Scholar]
- 24.For examples of heterocycle—thiazole formation, see: Lu J-Y, Arndt H-D. J. Org. Chem. 2007;72:4205–4212. doi: 10.1021/jo0703505. Mashraqui SH, Mistry H, Sundaram S. J. Heterocycl. Chem. 2006;43:917–923. Clough JM, Dube H, Martin BJ, Pattenden G, Reddy KS, Waldron IR. Org. Biomol. Chem. 2006;4:2906–2911. doi: 10.1039/b603433k. Hearn BR, Zhang D, Li Y, Myles DC. Org. Lett. 2006;8:3057–3059. doi: 10.1021/ol061087h.
- 25.a) Loska R, Maükosza M. J. Org. Chem. 2007;72:1354–1365. doi: 10.1021/jo062254u. [DOI] [PubMed] [Google Scholar]; b) Sissouma D, Maingot L, Collet S, Guingant A. J. Org. Chem. 2006;71:8384–8389. doi: 10.1021/jo061270o. [DOI] [PubMed] [Google Scholar]
- 26.Goeblyoes A, Santiago SN, Pietra D, Mulder-Krieger T, von Frijtag Drabbe Kuenzel J, Brussee J, IJzerman AP. Bioorg. Med. Chem. 2005;13:2079–2087. doi: 10.1016/j.bmc.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 27.Fravolini A, Grandolini G, Martani A. Gazz. Chim. Ital. 1973;103:755–769. [Google Scholar]
- 28.Curphey TJ. J. Org. Chem. 2002;67:6461–6473. doi: 10.1021/jo0256742. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.