Abstract
Non-invasive in vivo imaging of lymphatic vessels and lymphatic nodes is expected to fulfill the purpose of analyzing lymphatic vessels and their function, understanding molecular mechanisms of lymphangiogenesis and lymphatic spread of tumors, and utilizing lymphatic molecular markers as a prognostic or diagnostic indicator. In this review, we provide a comprehensive summary of in vivo imaging modalities for detecting lymphatic vessels, lymphatic drainage, lymphatic nodes, which include conventional lymphatic imaging techniques such as dyes and radionuclide scintigraphy as well as novel techniques for lymphatic imaging such as optical imaging, computed tomography (CT), magnetic resonance imaging (MRI), ultrasound, positron emission tomography (PET) using lymphatic biomarkers, photoacoustic imaging and combinations of multiple modalities. The field of lymphatic imaging is ever evolving, and technological advances, combined with the development of new contrast agents, continue to improve the research of lymphatic vascular system in health and disease states as well as to improve the accuracy of diagnosis in the relevant diseases.
Keywords: Lymphatic imaging, lymphangiography, contrast agent, molecular imaging
Introduction
The lymphatic system is a complex network of lymphatic vessels, lymph nodes and other lymphatic organs, which is essential for the maintenance of fluid homeostasis and immunocompetence. Lymphatic vessels are found in nearly every organ and blood vessel-containing interstitial tissue and serve as a drainage system for excess fluid and large molecules or cells that cannot easily find their way back into venules [1,2]. Although lymphatic vessels were first described back in the 17th century, the first growth factors and molecular markers specific for these vessels were not discovered until a decade ago [3,4]. Much progress has been made in recent years including the discovery of key molecular pathways regulating lymphangiogenesis, the identification of lymphangiogenesis markers, the isolation of lymphatic endothelial cells and the development of genetically modified animal models to study lymphangiogenesis [3–12].
The lymphatic vessels contribute to a wide range of human pathologies, such as cancer metastases [3,13,14], inflammation [15–17], diabetes [18], and obesity [19]. Especially in cancer metastasis, the lymphatic system transports fluid and cells from the periphery to the circulation. This system is increasingly appreciated as a conduit for the metastasis of a variety of human cancers such as breast, melanoma, and head and neck cancers [3]. Thus, investigation of the lymphatic contribution to metastasis may lead to diagnostic or therapeutic strategies to identify or prevent tumor dissemination. At present, some new in vivo models rapidly provide an abundance of information on the mechanisms underlying lymphatic development and the progression of diseases associated with lymphatic dysfunction [9]. Yet, compared with in vivo hemovascular imaging, lymphatics imaging has been paid much less attention [20]. Although technically challenging, the ability to image lymph flow, lymph nodes, lymphatic function and markers of lymphangiogenesis could have a profound impact on our understanding of these diseases.
Imaging of the hemovascular system often requires intravenous administration of contrast agents, whilst the routes of introducing contrast agents into the lymphatics are through interstitial (intradermal or subcutaneous) administration, direct administration into lymphatic vessel, or intravenous injection. Small nanoparticles can pool into the lymph nodes via blood circulation, but can also diffuse into lymphatic vessels via the gap junctions between the endothelial cells under a hydrostatic pressure gradient. Particles between 10–100 nm in diameter can extravasate into the interstitial space, where they are phagocytosed by macrophages and are then transported to lymph nodes. Particles larger than 100 nm typically remain trapped in the interstitium [20,21]. In the following sections, we will review various imaging modalities and contrast agents used for lymphatic imaging with focus on preclinical studies that have translational potential.
Conventional lymphatic imaging
Conventional lymphatic imaging using dyes and radionuclide scintigraphy has a long history. These methods delineate not only lymphatic vessels and drainage but also sentinel lymph nodes. Blue dye staining has been the principal method of determining blood volume in humans and animals for almost a century. The commonly used dyes are isosulfan blue, patent blue V, Evans blue (EB) or fluorescent dyes (see optical image section), which provide a visual signal of the draining lymphatic vessels and lymph nodes [22]. Since these dyes color living tissues or cells, they are also called vital dyes [23]. Typically, dye molecules are injected intradermally or subcutaneously into the interstitial tissue of animals or human beings, to obtain indirect micro lymphangiographies of superficial lymphatic vessels. After injection of blue dyes, cutaneous lymphatic vessels and lymphatic drainage from the skin can be macroscopically visualized [19,22,24]. Kajiya et al. [25] observed prominent enlargement, functionally impaired and hyperpermeable lymphatic vessels of the ultraviolet B-induced cutaneous damage as well as systemic blockade of VEGF-A inhibited enlargement of lymphatics by injection of Evans blue dye into the rim of mouse ears. Harvey et al. [19] showed that multiple hypoplastic and tortuous Evans blue dye–positive lymphatic vessels in the thoracic cavity in Prox1+/− mice. This phenomenon may reflect functional inactivation of a single allele of the homeobox gene Prox1 led to adult-onset obesity due to abnormal lymph leakage from mis-patterned and ruptured lymphatic vessels.
Lymphoscintigraphy is a special type of radionuclide imaging which requires injection of gamma-emitting radionuclides labeled macromolecules or larger colloidal particles into a local region of tissue in order to evaluate lymph drainage function by measurement of the removal rate [26]. Lymphoscintigraphy is the most widely used method for lymphedema quantification. Pereira et al. [27] provided an easy and rapid lymphatic drainage examination of mammary glands by intramammary administration of 99mTc-dextran to female mongrel dogs. Anthony et al. [28] used 99mTc-sulfur colloid to study lymphatic regeneration in rat unilateral hindlimb autotransplantation models. By gathering quantitative data, they found rapid regeneration of the subcutaneous lymphatics following replantation which can handle the lymphatic drainage of the limb adequately. This result may explain why there is no clinical lymphedema in human limb and other tissue replants.
Sentinel lymph node (SLN) is the hypothetical first lymph node or group of nodes reached by metastasizing cancer cells from a primary tumor. Thus, lymph node imaging can be applied to evaluate the metastatic status of a tumor. The basis for SLN imaging is that the contrast agent injected near the primary tumor will be taken up by the adjacent lymphatic system and transported to the SLN. Currently, blue dye and radionuclide-labeled sulfur colloid are most common imaging agents for SLN imaging. Alex and Krag [29] reported the first gamma-probe localization of SLNs utilizing 99mTc-sulfur colloid in an animal model. Their study demonstrated that gamma-probe guided localization could be used to identify and then surgically remove the first draining lymph nodes in 16 inguinal lymphatic basins of eight cats and this method was found to be comparable to an invasive method using a blue dye. Currently, lymphoscintigraphy has been employed widely in the detection of SLNs. For example, in patients with breast cancer, preoperative lymphoscintigraphy identified SLNs as hot spots 87% of the time, with 20% of the cases showing draining nodes to other basins in addition to or independent of the axilla [30]. Recently, a dual mapping procedure using isotope and dye injections was performed in 60 patients with clinically node-negative breast cancer. SLNs were identified in 59 of 60 patients (98.3%), with a node-positive rate of 41.7% and a false-negative rate of 1.7% [31].
The characteristics and the properties of the agents employed are the major factors governing the efficiency of lymphoscintigraphy. Although 99mTc-sulfur colloid has higher specificity and lower background radioactivity for lymphatics as compared with 99mTc-dextran, it has lower solubility in the lymph and requires longer time to obtain satisfactory lymphatic images. Pereira et al. [27] injected 18.5 MBq 99mTc-dextran into the mammary parenchyma around the mammary papilla in adult female mongrel dogs, and their result showed that 99mTc-dextran mammary lymphoscintigraphy provided easy and rapid lymphatic drainage examination of mammary glands. In addition to diagnostic imaging, a number of studies have investigated the biodistribution of drug delivery nanoparticles and drug transport to lymph nodes through 99mTc labeling in order to provide evidence for delivering intralymphatic therapy in the future [32].
Cyclosporine A (CyA) is an important immunosuppressive agent used to prevent allograft rejection in organ and tissue transplantation. Because of its low solubility, bioavailability and harmful reversible side effects, the clinical treatment is restricted to oral administration. Lymphatic transport seems to be an alternative to improve the therapeutic efficacy of CyA and to exclude its side effects [33,34]. Kim et al. [35] used CyA loaded 99mTc-labeled dextran acetate (DxA) and investigated the biodistribution of particles by lymphatic delivery in rats in order to evaluate lymphatic transport effect of CyA. Results indicated that the CyA-loaded 99mTc-DxA can be well distributed through the lymph nodes. Solid lipid nanoparticles have been proposed as carriers for anti-cancer drugs [36]. Videira and co-workers [37] investigated the biodistribution of 99mTc-labeled solid lipid nanoparticles by inhalation for pulmonary delivery to the lymphatic drainage in lung cancer. The data showed significant uptake of the radiolabeled nanoparticles into the lymphatics and high rate of distribution in lymph nodes after inhalation.
Because of their quantitative sensitivity, inherent biological safety and relative ease of use, it is agreed that vital dyes and lymphoscintigraphy hold enormous potential for preclinical and clinical applications. However, these methods have a number of drawbacks. For instance, SLNs need to be dissected to observe blue dye staining and lymphoscintigraphy has radiation exposure and relatively low resolution. Furthermore, SLN imaging is not applicable to all tumors as not all tumors drain to a single node hence this technique is not universally applicable. Some novel techniques such as computed tomography (CT), positron emission tomography (PET), ultrasound (US), magnetic resonance imaging (MRI), photoacoustic imaging and multi-modality imaging hold promise in overcoming these drawbacks.
Optical fluorescence imaging
Relatively high resolution, no radiation exposure and ability to image at molecular level has made optical imaging an advancing branch of medical imaging [1]. Optical imaging of the lymphatic system allows for mapping of lymphatic drainage, localization of SLNs and visualization of multiple nodes [38]. For example, indocyanine green (ICG) is an unconjugated free emissive fluorophore which can be excited at 780 nm with a significant Stoke’s shift, enabling fluorescence measurement at 830 nm and greater. ICG is the earliest near-infrared (NIR) dye utilized for the noninvasive detection of tumors in both animals and patients, and has been approved in humans for assessing cardiac and hepatic function as well as lymphatic flow and SLN imaging [39–41]. Kwon et al. [42] evaluated propulsive lymph function using ICG in a rodent model. By fixing regions of interest (ROIs) in fluorescent lymph channels and calculating the fluorescence intensity within each ROI, they computed that lymph flow velocity ranged from 0.28 to 1.35 mm/s. Sharma and co-workers [43] also studied the dynamic lymph flow using ICG in a swine model. Following intradermal administration of 3–6 nmol of ICG, lymph flow immediately progressed from the lymphatic plexus and the lymph vessels and nodes were stained. Lymph flow propelling frequencies were found to be between 0.5 and 3.3 pulses/min and velocities between 0.23 and 0.75 cm/s. ICG has been successfully used to guide SLN biopsy in breast cancer and melanoma by using NIR camera [44–46]. Hirche et al. [44] compared SLN detection by ICG optical imaging and by 99mTc-sulfur radiocolloid scintigraphy in combination with blue dye. Although the sensitivity and specificity of ICG for SLN detection is comparable to conventional methods, the main advantage of this technique is that it allows transcutaneous visualization of intraoperative lymph node detection without radioisotope.
Other NIR organic fluorophores such as Alexa 705, IRDye780, Cy7, and Cy5.5 can also be used to detect lymphatic drainage. These organic fluorophores can also be conjugated with bioactive molecules such as antibodies, proteins, and peptides to effectively enter and remain in the lymphatic system [47]. Hama and co-workers [48] used Cy5.5 and Cy7 conjugated IgG to map the drainage patterns of lymphatic basins and to locate SLNs. In their study, Cy5.5-IgG and Cy7-IgG were injected interstitially into the mammary fat pad, extremity and ear in a mouse model. One min after injection, the SLNs of mice were visualized with a depth penetration of approximately 5 mm. The mapping demonstrated that axillary nodes received input of lymph flow from the breast and upper extremity and cervical nodes received input from the ear and upper extremity.
Most of the organic NIR fluorophores often lack the ideal properties for generating strong fluorescent signals through deep tissue sections. The design of high emission dipole strength fluorophores is thus necessary for both animal research and clinical imaging applications [47]. The recent advance of nanotechnology has led to the development of quantum dots (Qdots) for use in imaging, detection and targeting [49–51]. Qdot are nano-sized semiconductor crystals ranging between 5 and 20 nm. The size and shape of Qdots can be fine-tuned to control the emission wavelengths of the Qdots, including ones that emit light in the NIR range [50]. Furthermore, their extreme brightness provides more depth penetration than organic fluorophores. Harrell and co-workers [52] used non-targeted Qdots to demonstrate that increased lymph flow precedes tumor metastasis in a melanoma xenograft model. In their studies, Qtracker 705 particles were injected to the dorsal toe of each hind foot and a real-time imaging assay found that tumors can affect lymph flow through the draining lymphatic system. Meanwhile, by measuring the fluorescent signal in a ROI over each popliteal LN, it was found that lymph flow is specifically increased through tumor-draining LNs. Ballou et al. [53] also demonstrated that passage from the tumor through lymphatics to adjacent nodes could be visualized after Qdot injection. Parungo et al. [54,55] also applied Qdots to illustrate the lymph drainage patterns of the peritoneum by continued flow to mediastinal lymph nodes. As Qdots can be used to perform multicolor images with high fluorescence intensity, Hama et al. [48] used two NIR Qdots with different emission spectra to simultaneously visualize two separate lymphatic flow drainings and variations in the drainage patterns. Kosaka et al. [56] used five visually well-distinguishable carboxyl-Qdots (Qdot 545, 565, 585, 605, and 655) injected subdermally into mice at five different sites. In vivo lymphatic images successfully distinguished all five lymphatic basins with different colors in real-time (Figure 1). Kim et al. [57] used QDots to visualize SLNs in mouse and pig models. In their study, localization of the SLNs required only 3–4 min and average detective depth of the SLN in the pig was approximately 1 cm from the skin surface. Knapp et al. [58] used NIR Qdots to detect SLNs in invasive urinary bladder cancer in animal models. Near infrared optical imaging shows promise for imaging superficial lymph drainage. However, poor tissue penetration and photon scattering limited optical imaging to detect deep lymphatic drainage basins [59]. Despite of the limited depth penetration, optical imaging maybe a good complementary technique for intraoperative detection of nodes that have been characterized using more conventional imaging test if dual imaging probes can be developed. For example magneto-optical probes could allow MRI characterization pre-surgically; and fluorescence based imaging will lead to confirmation intra-operatively akin to sentinel node imaging. More details about multimodality imaging will be elaborated later.
Figure 1.
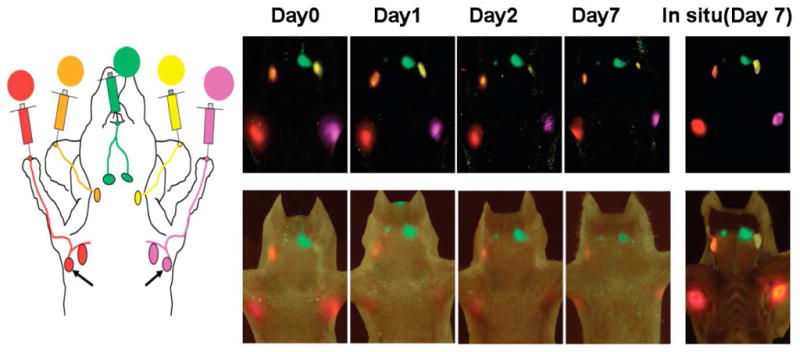
A five-color optical lymphatic image of lymphatic drainages from the upper extremity (red), the ear (yellow) and the chin (green) obtained using 5 NIR Qdots (Qdots 655,585,545,565, and 605) from day 1 to day 7. There are less marked reductions in signal, except in the left deep neck lymph node (Qdot 565) and a spectral fluorescence imaging technique is shown together with a schematic illustration. (Reproduced from Kosaka et al. [56] with permission)
CT imaging of lymph nodes
As previously mentioned, lymphoscintigraphy and vital blue dye are the two most common methods to map SLNs. One of the major concerns of these methods is their low resolution. CT with appropriate contrast agents, on the other hand, can easily discern enlarged lymph nodes. Wu et al. [60] showed in a VX2 carcinoma model that CT-LG was able to identify 96%–100% contrast enhanced SLNs. Suga et al. [61] chose a commercially available, water-soluble agent, iopamidol, to visualize breast lymphatic drainage and map breast SLNs in normal dogs and healthy human volunteers. They injected iopamidol into the periareolar skin areas overlying the mammary gland in animals and volunteers, and the connection of SLNs and lymphatic vessels draining from the injection sites can be clearly visualized. Hundred percent SLNs and 87.5% distant nodes resected had a good correlation with the locations and sizes measured by the CT images. In a follow-up study, the same group used CT-LG to evaluate SLNs in patients with operable breast cancer, and the localization of SLNs in all patients could be visualized [62]. Under 3D multidetector-row CT-LG (MDCT-LG) navigation, the sensitivity, false-negative rate, and accuracy of SLN biopsy were 92%, 7%, and 98%, respectively [63]. Margel et al. [64,65] demonstrated that intravenous injection of synthesized iodinated homopolymeric radiopaque nanoparticles allowed enhanced visibility of reticuloendothelial systems such as lymph nodes, liver, kidneys and spleen. Rabin et al. synthesized a polymer-coated Bi2S3 nanoparticle (BPNP) as an injectable CT imaging agent. Following intravenous administration of BPNP, strong vascular enhancement was observed with CT imaging and the enhancement resulted in clear delineation of the cardiac ventricles and all major arterial and venous structures. In addition, regional lymph nodes were clearly contrasted in mice injected subcutaneously with 50 μl BPNPs [66].
Although the current CT lymphography (CT-LG) is unable to detect occult metastases, it may serve as a viable alternative to lymphoscintigraphy and vital blue dye for SLN identification and guiding SLN biopsies. CT lymphography provides detailed lymphatic anatomy, which may eliminate the potential pitfalls of the lymphoscintigraphy and vital blue dye methods and increase sensitivity and accuracy for SLN biopsy. However, it is difficult for CT to provide information on the flow of lymph owing to the spatial resolution needed and radiation concerns of CT.
MR imaging of lymphatic system
Dynamic contrast-enhanced MR imaging (DCE-MRI) is a technique that acquires serial images following the intravenous injection of a contrast agent and then wash-in and wash-out curves can be derived from designated ROIs for direct comparison. This technique has been widely used to analyze angiogenesis [67,68]. Ruddell et al. [69] developed a DCE-MRI assay to measure changes of lymph flow and lymphatic dysfunction by dynamic mapping of lymphatic drainage in mice models. In their study, B16-F10 melanomas cells were injected into the left hind leg footpad in mice. MR lymphography was scanned with low–molecular weight gadolinium contrast agent, Gd-DTPA, which was injected subcutaneously into the dorsal toe of the rear foot to monitor lymph flow dynamically within 3 weeks after implantation. In order to obtain a time course of contrast flow, the kinetics of lymph drainage were examined by taking scans rapidly and repeatedly after Gd-DTPA injection. Results demonstrated that tumor growth in the footpad not only enhanced lymph flow through draining LNs, but also enhanced lymph delivery into the heart and vascular circulation. This finding was consistent with the previous hypothesis that tumor-induced lymph flow actively drives metastasis into and through draining LNs [52]. Subcutaneous injection of the conventional extracellular gadolinium-based agents also allows visualization of draining lymph vessels and nodes [70,71] (Figure 2). Compared with intravenous injection, the main advantage of interstitial injection is that both lymphatic vessels and lymph nodes can be seen at a lower dose [72]. However, the main problem of these small molecular contrast media is that only small amount can reach the lymphatic system and others diffuse into the blood system.
Figure 2.
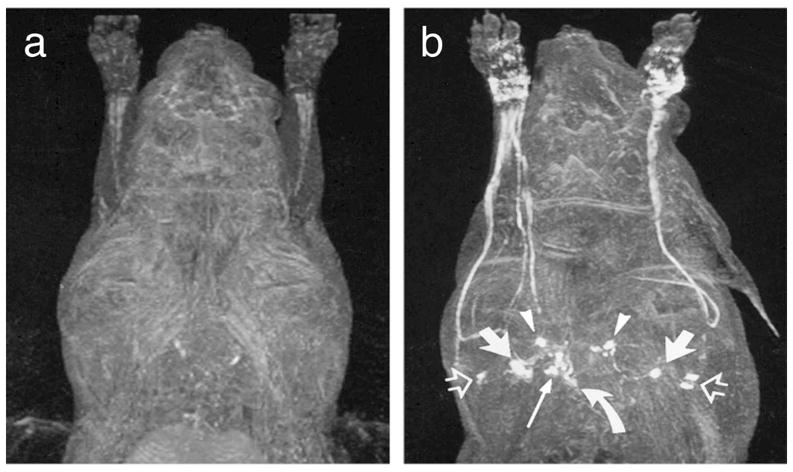
MRI lymphography demonstrates before (a) and 15 min after (b) subcutaneous administration of gadoterate meglumine bilaterally in the dorsal aspect of the forepaw. The following lymph node groups are depicted in axillary node, anterior thoracic lymph node (arrowheads), parasternal lymph node (curved arrow), and mediastinal lymph node (long solid arrow). (Reproduced from Ruehm et al. [70] with permission)
Lymphotrophic nanoparticle enhanced MRI offers a new method for accurate lymph node staging [73]. This technique employs a new class of MR contrast agents with unique biochemical properties including superparamagnetic iron oxide particles (SPIO), liposomes and micelle-forming compounds, polymers and protein-binding compounds [70,71,74]. Ultrasmall superparamagnetic iron oxide (USPIO) contrast may depict salient features of normal and cancer-positive lymph nodes. These nanoparticles are 30 to 50 nm in hydrodynamic size and are composed of an active iron oxide crystalline core and a covering material made of low-molecular-weight dextran or starch derivatives, which can shorten proton relaxation times, giving a signal distinct from tissue without particles [20,75]. The small size of the iron oxide nanoparticles enable them to cross capillary walls and localize into lymph nodes either though direct transcapillary passage of the nanoparticles from the venules to the medullary sinuses of lymph nodes or extravasation across permeable capillary beds by a nonselective endothelial transcytosis. Once delivered to the lymph node, the nanoparticles are phagocytosed by macrophages of the reticuloendothelial system (RES), thus causing nanoparticle accumulation within the node [76,77]. Imaging lymph nodes using USPIO is an indirect way to evaluate macrophage function within the lymph nodes. Accumulation of nanoparticles in benign nodes causes a decrease in signal intensity on T2-weighted and T2*-weighted MRI scans [78]. The RES in malignant lymph nodes lacking normal macrophages cannot phagocytose USPIO and thus retain the bright signals in MRI scans, which was called “negative enhancement” [79]. One disadvantage of USPIO for imaging lymph nodes is the slow transport to the lymphatic system so post-contrast imaging 24–36 h after contrast media injection is required [80]. In addition, the rather extended pharmacologic half-life of the iron oxide nanoparticles also limited the clinical use [81]. Other disadvantages include the unpredictability of iron-induced susceptibility artifacts, and the heterogeneous enhancement profile in normal lymph nodes [82].
Several limitations of these paramagnetic particles might be overcome with T1-shortening contrast agents. Misselwitz et al. [83] detected lymph node metastasis using Gd-carrying liposomes in a guinea pig mode, taking advantage of that Liposomes can be phagocytosed by lymph node macrophages after interstitial injection. Trubetskoy et al. [84] also showed that after subcutaneous injection of surface-modified liposomes containing Gd-DTPA phosphatidylethanolamine into the paws of rabbits, axillary and subscapular lymph nodes can be delineated within minutes. Other polymer based contrast agents have also been tested to image lymph node metastases. Kobayashi et al. used different dendrimer-based MRI contrast agents to visualize the anatomy and physiology of deep lymphatic vessels and lymph nodes in mouse models. Among them, PAMAM-G8 was the best for visualizing lymphatic vessels, whereas DAB-G5 was better for visualizing lymph nodes [85–87]. In addition, this group used PAMAM-G6 as a contrast agent and revealed the absence of filling in the metastatic foci of affected lymph nodes. Another dendrimeric agent such as Gadomer-17 was also used for MR imaging of the lymphatic system in different experimental models [88,89]. Herborn et al. [74] used a new blood-pool contrast agent, MS-325 to image regional lymph nodes. MS-325 is albumin-binding Gd-based contrast agent which protein-binding properties may make this agent large enough to be phagocytosed and lymphotropically cleared. After interstitial injection, lymphatic vessels and tumor-bearing lymph nodes can be detected clearly.
It is of note that the route of administration can be an issue for clinical translation. Interstitial and subcutaneous injections are viable techniques to study lymphatic pathways but are not suited to image and stage nodes which are better served with intravenous agent injection and delivery. This is important when staging needs characterization of nodes in regional distribution.
Ultrasound imaging of lymphatic system
Since the first description of contrast-enhanced ultrasound imaging (CEUS) by Gramiak and Shah [90], it has been widely used in both preclinical experiments and clinical diagnosis. In general, there are several advantages of CEUS such as low cost, high spatial resolution (comparable to MRI or CT and greater than PET or SPECT), and high contrast sensitivity and signal-to-noise ratio (comparable to PET and SPECT). These advantages make CEUS a promising imaging modality for detecting SLNs.
Microbubbles are the most widely used ultrasound contrast agents. The microbubble shell can be made with lipids, denatured albumin, or polymers incorporating polyethylene glycol in order to reduce gas diffusion into the blood, to reduce recognition by the immune system and to enhance stability [91,92]. Based on the specific acoustic properties, microbubbles can be sensitively imaged with regular ultrasound methods. For CEUS imaging of SLNs, microbubbles were injected directly into tissues of interest. In animal studies, ultrasound contrast agents have been shown to accumulate in sentinel lymph nodes but not second-order lymph nodes. The mechanisms of this passive targeting are poorly understood but probably related to the avidity of the shell material for macrophages [93]. Lurie et al. [94] injected perfluoropropane-filled microbubble contrast medium into peritumoral tissues in 10 dogs with spontaneous head or neck tumors, and compared the results with lymphoscintigraphy in which dogs were injected peritumorally with 99mTc-sulfur colloid. CEUS could identify SLNs and associated lymphatics and the results were consistent with lymphoscintigraphy findings, indicating that CEUS might be an effective adjunct or alternative to conventional techniques for SLN detection and localization.
Nielsen and co-workers investigated whether the distance from the contrast agent injection site to SLN could affect the detectability of CEUS in a swine model. The result showed that the distance did not interfere with SLN detection [95]. In a pilot clinical trial, before surgery, patients with breast cancer received a periareolar intradermal injection of microbubbles, lymphatic channels were visualized immediately by ultrasonography and followed to identify putative axillary SLNs. The sensitivity of SLN detection in this study was 89% [96].
Besides SLN detection, microbubbles have also been used to differentiate benign and malignant lymph nodes. The mechanism is probably that microbubbles are phagocytosed by macrophages or reticuloendothelial cells and appear bright on an ultrasonogram in benign lymph nodes and malignant lymph nodes that lack reticuloendothelial cells cannot take up the microbubbles would remain hypoechoic. Goldberg et al. demonstrated that the accuracy of SLN detection by CEUS and lymphoscintigraphy was 90% and 81%, respectively in a swine model with melanoma. Compared with histological examination, the sensitivity, specificity and accuracy for correctly depicting metastases in swine SLNs by lymphatic CEUS were 95%, 63% and 86%, respectively [97].
The main disadvantages of US are its poor spatial resolution, the large size of the microbubble and hence its slower rate of uptake, its limited use in the thorax and deep retroperitoneum, and its dependence on operator experience [98]. Indeed, microbubble-CEUS of the lymph nodes is unlikely to reflect either the extravasation of bubbles, with re-uptake by the lymphatic system, or uptake by macrophages and tracking to lymph nodes, due to the size of the contrast agents, and the timing of scanning, respectively [1].
Photoacoustic imaging of lymphatic system
Photoacoustic imaging (PAI), as a hybrid biomedical imaging modality, is developed based on the photoacoustic effect. In PAI, non-ionizing laser pulses are delivered into biological tissues. Some of the delivered energy will be absorbed and converted into heat, leading to transient thermoelastic expansion and thus ultrasonic emission. The generated ultrasonic waves are then detected by ultrasonic transducers to form images. The advantage of PAI is that it retains high optical contrast while overcoming the poor spatial resolution of pure optical imaging and provides good spatial resolution by detecting ultrasound, which has relatively low scattering [99,100].
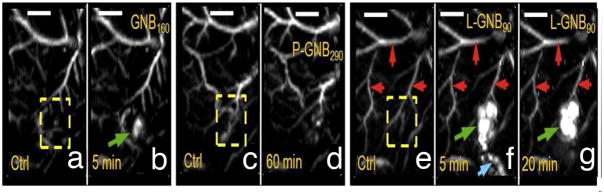
PAI in combination with different contrast agents is promising in improved detection of metastases. Song et al. [101] tried methylene blue as a PAI contrast agent to image SLNs in rats and were able to detect lesions about 2–3 cm in depth. Other groups also used a series of nanoparticle-based contrast agents such as carbon nanotubes, gold nanocages, gold nanorods, and gold nanobeacons to improve resolution and allow deep tissue imaging [102–104]. The single-walled carbon nanotubes (SWNTs) were first applied as a PA contrast agent by the Stanford group for imaging tumor integrin expression [102]. Later on, Pramanik et al. [105] used carbon nanotube enhanced photoacoustic identification of SLNs in a rat model. Recently, Pan et al. [104] demonstrated near infrared detection of SLN with gold nanobeacons (GNBs) providing the photoacoustic contrast in a rodent model. They found that smaller GNBs were noted trafficking through the lymphatic system and accumulating more efficiently in the lymph nodes in comparison to the bigger nanoagents. Within 1 h post-injection, the contrast ratio of the lymph nodes with the surrounding blood vessels was 9:1 [104] (Figure 3).
Figure 3.
In vivo noninvasive photoacoustic imaging of sentinel lymph nodes in rat. Lymph node is not visible in a 60 min post-injection image of nanobeacons P-GNB160 (d). But SLNs are clearly visible after L-GNB90 injection (10 nm) (g). (Reproduced from Pan et al. [104] with permission)
McCormack et al. [106] used photoacoustic method to detect SLN micrometastasis in a melanoma model nodes. In their study, HS 936 human melanoma cells were separated and injected into the canine lymph nodes. Because melanoma cells contain melanin which can absorb the light and produce photoacoustic pulses under laser and this photoacoustic pulses can be detected by special sensors. Photoacoustic responses from a lymph node with as few as 500 melanoma cells can be detected, while normal lymph nodes showed no response. In addition, recent study on photoacoustic detection and photothermal ablation of individual light-absorbing nanoparticles labeled tumor cells in lymph nodes in vivo will encourage further explorations of light-absorbing nanoparticles in biomedical applications, in particular non-invasive and target-specific lymphatic diagnosis and therapy [103].
PET imaging of lymphatic system
So far, PET is the most sensitive and specific technique for imaging molecular pathways in vivo in humans [107]. PET radiotracers are physiologically and pharmacologically relevant compounds labeled with positron-emitting radioisotopes (such as fluoride-18 or carbon-11). After internalization by injection or inhalation, the tracer reaches the target and the location and the quantity is then detected with a PET scanner. The inherent sensitivity and specificity of PET is the major strength of this technique. Isotopes can be detected down to the 100 picomolar level in the target tissues. At this low level the compounds often have little or no physiological effect on the patient or the test animal, which permits studying the mechanism of action or biodistribution independent of any physiological consequences [108,109]. The spatial resolution of PET down to the millimeter level permits applications not only to humans for diagnosis and drug development, but also to animals for preclinical studies. The ability of PET to translate studies from animals to humans adds to its appeal. 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG) is now the most commonly used PET tracer worldwide. 18F-FDG PET has been used to image metastases and detect metastatic spread of malignant cells to regional lymph nodes in a wide variety of tumor types [110]. 18F-FDG-PET is able to detect increased glycolytic rate in cancer cells compared to normal cells, therefore, PET can functionally assess lymph nodes and potentially detect metastases in normal sized nodes. When PET is combined with a CT scan (PET–CT), the fused image can demonstrate increased metabolism with its anatomical location. Besides FDG, other molecules specifically metabolized in tumor cells have also been tested to evaluate LN status. Choline is a molecule that is metabolized into phosphorylcholine and is incorporated into cellular membranes [111]. It can be labeled with 18F and 11C. 11C-choline sensitivity and specificity for LN metastasis in prostate cancer has been studied, which could detect metastasis with a sensitivity of 80%, a specificity of 96%, and an accuracy of 93% [112]. However, one limitation with the use of 11C-choline as a tracer is its short half-life. In a recent clinical study recruiting 132 patients with prostate cancer, the sensitivity, specificity, and positive and negative predictive values of 18F-fluorocholine PET/CT in the detection of malignant LNs were 45%, 96%, 82%, and 83%, respectively. For LN metastases greater than or equal to 5 mm in diameter, sensitivity, specificity, and positive and negative predictive values were 66%, 96%, 82%, and 92%, respectively [113]. 11C-acetate or 18F-acetate reflects another aspect of lipid metabolism. Fricke et al. [114] demonstrated that 11C-acetate is better than 18F-FDG in detecting local recurrences and regional lymph node metastases of prostate cancer. In addition to the above mentioned PET tracers, other positron-emitting molecules such as anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid (anti-18F-FACBC), 16β-18F-fluoro-5α-dihydrotestosterone (18F-FDHT) and 11C-methionine have also been used to detect LN metastasis [115]. However, the anatomical resolution of PET (approximately 4–8 mm3 in clinical and 1–2 mm3 in small animal imaging systems) is noticeably poorer than that achieved by CT or MRI [116]. Due to the relatively lower resolution of PET as compared to other anatomical imaging techniques, PET allows for detecting malignancy in normal sized nodes but is insufficient for detecting small nodes, thus preventing its application in staging tumors like breast, rectum, etc. Indeed, in breast cancer patients, the sensitivity, specificity, PPV, NPV and accuracy of FDG PET/CT was 58, 92, 82, 77 and 79%, indicating that FDG PET/CT cannot replace invasive approaches for axillary staging but may extend the indication for SLNB [117]. Low sensitivity of PET/CT also has been reported in patients with primary malignant melanoma [118].
Multi-modal imaging
Among the above mentioned imaging techniques, no single modality is perfect and sufficient to gain all the necessary information [119,120]. Therefore, the combination of multiple imaging techniques can offer synergistic advantages [121]. Multi-modal imaging can be achieved either through the combination of imaging hardware such as PET/CT and SPECT/CT [122], through the combination of different contrast agents [123], or through co-registration of images acquired with different modalities. With regard to lymphatic imaging, this leads to improved accuracy and sensitivity of lymph nodes detection as well as lymphatic drainage visualization.
The combination of PET and CT has been matured into an important clinical diagnostic tool. The underlying principle of PET/CT systems is the acquisition of two dedicated imaging procedures, CT and PET, in a single step followed by image co-registration which are able to provide anatomical and functional data sets in a single session with accurate image co-registration [124]. In this way, The CT component of the hybrid system is used to improve anatomical definition of the ROIs for analysis and to create radiation-attenuation maps to correct for non-uniform attenuation [125]. Clinical studies have demonstrated the advantages of PET/CT over separately performed PET and CT, particularly in the field of oncology [126]. A number of clinical studies compared FDG–PET/CT with CT alone for evaluating lymph nodes for metastatic spread. The results demonstrated that sensitivity, specificity, positive predictive value, negative predictive value and accuracy of lymph node staging were all significantly improved with FDG–PET/CT [127,128]. SPECT/CT is based on the similar principles of PET/CT. Hybrid SPECT/CT lymphoscintigraphic imaging facilitates the localization of SLNs by showing the relationships between SLNs and important anatomic structures, which enables precise SLN localization and helps to ensure minimal dissection in clinical application [129].
With the development of microPET or microPET/CT scanners dedicated to small animal imaging studies, it can provide a similar in vivo imaging capability in mice, rats, monkeys, and humans so one can readily transfer knowledge and molecular measurements between species [130,131]. Along with the progress made in several key techniques including identifying scintillators with adequate magnetic properties, in developing suitable PET detectors which use optical fibres to guide the scintillation light away from the MR magnetic fields or that replace the PMTs by magnetic field-insensitive avalanche photodiodes (APD) [132], several research groups have successfully developed small PET/MRI prototypes for small-animal studies [133,134] and even a human-sized prototype design for neurology applications [135]. With the PET/MRI system, three functional imaging techniques will be united including PET, fMRI and spectroscopy with morphological MRI, offering far more potential than just tracing biomarkers and displaying anatomy [134].
Most of the contrast agents for lymphatic imaging generally have one function and can only be imaged in monochrome. When radiocolloid and dye are used in tandem, there is a lower false-negative rate for sentinel node localization when compared with using either agent alone [136,137]. Combination of both radioactive and color signals into a single dose has also been investigated such as 99mTc-dextran-dyes [27], 99mTc-/111In-phthalocyanine tetrasulfonate [138], 125I-methylene blue [139], and 99mTc-EB [140]. Tsopelas et al. [140] confirmed that both the soluble 99mTc-EB and 99mTc-antimony trisulfide colloid (ATC) discriminated the SLNs up to 50 min after administration. However, 99mTc-EB had the advantage of providing radioactive (gamma probe) and color signals simultaneously during the operative exposure.
Kobayashi and co-workers [121] synthesized 111In-labeled radionuclide/five-color NIR optical dualmodal imaging probes using a polyamidoamine dendrimer (generation-6 PAMAM dendrimer) with an ethylenediamine core as the platform component. Radionuclide imaging of this dualmodal imaging probe allows increased depth penetration and absolute quantification whereas multi-color NIR optical imaging offers excellent real time spatial resolution and the ability to distinguish multiple lymphatic drainages. Both of these methods have a matched high sensitivity that allows for a minimization of the required injection dose [121]. Koyama and co-workers [141] synthesized MR/NIR optical hybrid contrast agent GdG6-Cy5.5 to perform anatomic MRL and NIR imaging with one injection. A number of nanoparticle based PET/MRI dual modality or optical/PET/MIR triple modality probes [142–144] showed promising results in tumor imaging. These probes may also have the potential to be applied for multimodality imaging of the lymphatic system. By combining strong optical contrast and high ultrasonic resolution in a single modality, photoacoustic tomography of lymph node demonstrated the potential to be used clinically for accurate, noninvasive imaging of SLNs for axillary lymph node staging [145]. With a modified clinical US imaging system, photoacoustic images of rat SLNs clearly help visualize methylene blue accumulation, whereas coregistered photoacoustic/US images depict lymph node positions relative to surrounding anatomy [146].
Active targeting biomarkers based imaging of lymphatics
Tumor blood vessels express molecular markers that distinguish them from normal blood vessels [147]. Recent data indicate that lymphatic endothelial cells are structurally different from blood endothelial cells and also express lymphatic system specific marker molecules such as podoplainin, Prox-1, LYVE-1, and VEGFR-3 [148–150]. Recent technical developments in optical imaging and other imaging modalities, in conjunction with these molecular markers, help to better understand the mechanisms underlying lymphangiogenesis and lymphatic metastases of tumor.
McElroy and co-workers [151] used AlexaFluor-labeled anti-mouse lymphatic endothelial hyaluronan receptor-1 (LYVE-1) monoclonal antibody to image mouse lymphatics and real-time track the movement of tumor cells engineered to express red-fluorescent protein (RFP) within the green fluorescently-labeled lymphatic vessels in a mouse model. In their study, they created a ventral skin flap to expose the inguinal, axillary lymph nodes and the interconnecting lymphatics of the anterior abdominal wall, and then injected AlexaFluor-labeled monoclonal anti-mouse LYVE-1 into the tissues around the exposed inguinal lymph node. The draining inguinal lymphatics and the afferent lymphatics of the receiving axillary lymph nodes were clearly shown after antibody delivery. After in vivo staining of mouse lymphatic endothelium, human pancreatic tumor cells expressing RFP were injected into the inguinal lymph node and surrounding tissues. Tumor cell trafficked along green labeled lymphatics could be reliably seen (Figure 4). This study apparently improved the understanding on interactions of primary tumors with lymphatic system and lymphatic dissemination of cancer cells.
Figure 4.
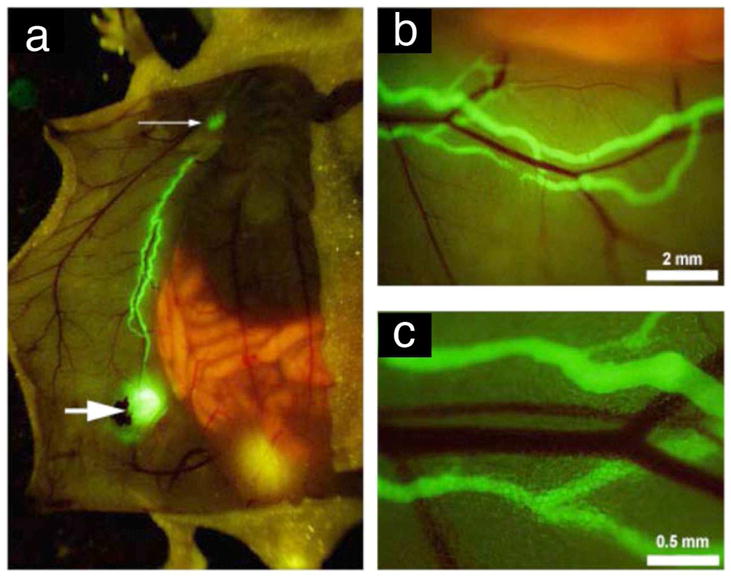
Fluorescence imaging of in vivo of the inguinal lymph nodes (larger arrow), axillary (small arrow) and the connecting lymphatics of the anterior abdominal wall after delivery of AlexaFluor-conjugated monoclonal anti-mouse LYVE-1 antibody (a–c). Neighboring blood vessels did not stain (b,c). (Reproduced from McElroy et al. [151] with permission)
Using in vivo phage display technology, Laakkonen et al. [150,152] identified a panel of peptides that home to lymphatic blood endothelial cells. Among them, a 9-amino-acid cyclic peptide (CGNKRTRGC) named as LyP-1 indeed had tumor homing specificity. In their study, intravenous injection of FITC-LyP-1 led to prominent accumulation in the tumor tissue 16–20 h after intravenous injection [153]. The extraordinary homing efficiency and ability to recognize metastatic lesions suggest the potential diagnostic uses for LyP-1.
The active targeting strategies can also be applied with ultrasound after attaching antibodies, peptides or other molecules to microbubble shell [92]. Examples include lipid shell microbubbles attached anti-ICAM-1 monoclonal antibody for imaging acute cardiac allograft transplant rejection in rats, microbubbles targeted to vascular endothelial growth factor receptor type 2 (VEGFR2) for imaging tumor angiogenesis in two murine tumor models, and microbubbles bear a covalently bound RGD peptide for targeting thrombosis and angiogenesis [154–157]. Li et al. [158] synthesized LyP-1 ultrasonic microbubbles and demonstrated the potential of this bubble conjugate for imaging tumor lymphatic vessels and as a targeted drug-delivery vehicle to treat lymph node metastasis. Hauff and co-workers [159] used L-selectin ligand-specific air-filled microparticles to target peripheral lymph nodes in normal mice and dogs. In their study, an L-selectin ligand, MECA-79 antigen, was selected for lymph node targeting. After intravenous administration of MECA-79 antibody conjugated polymer shell microbubbles, all the cervical, inguinal, axillary, popliteal, mesenteric lymph nodes in mice and the popliteal lymph nodes in dogs showed stimulated acoustic emission (SAE) signals.
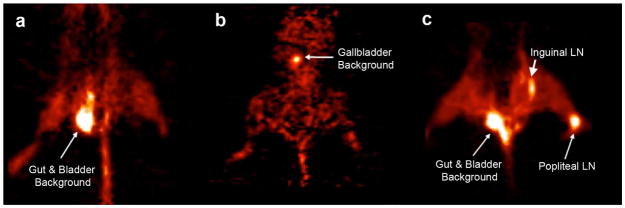
Currently, ongoing PET studies are attempting to better detect micrometastases in lymph nodes using molecular reporter genes. One of these promising reporters is the modified form of the herpes simplex type 1 thymidine kinase gene (HSV1-tk): HSV1-sr39tk. The protein sr39tk is a mutated form of the HSV-tk gene that can phosphorylate more than ten different substrates including 18F-FHBG and 18F-FEAU [160]. When these positron-emitting molecules are phosphorylated by sr39tk protein, the product is trapped in the cells and can be detected by PET–CT through its 18F positron-emitting isotope [161]. Brader et al. [162] demonstrated the feasibility of using recombinant human herpes simplex virus (HSV) vectors to detect nodal metastases in a human melanoma model by intratumoral injections of HSV NV1023 and intravenous injection of [18F]FEAU (Figure 5). In addition to functioning as a PET reporter gene, the gene encoding the sr39tk reporter also acts as a suicide gene. Coupling detection and suicide-gene therapy would enable not only the accurate assessment of lymph node status, but also the prospect of preventing metastatic outgrowth and systemic spread. Gambhir group has developed a two-step transcriptional amplification (TSTA) system [163] to increase the accuracy of imaging and the precise location of nodes. Based on this strategy, they developed several prostate-specific adenoviral vectors that express imaging reporter genes under the control of a highly amplified prostate-specific antigen (PSA) promoter system (TSTA) [164]. Burton et al. injected PSA–TSTA adenovirus in conjunction with its tracer 18F-FHBG into mouse forepaws in a LAPC9 prostate tumor xenograft model. Once in the node, the PSA–TSTA adenovirus can infect prostate tumor cells present there and activate the prostate restricted expression of an imaging reporter gene. This genetic biomarker–based PET was able to decisively seek out lymph node metastasis in a mouse bearing 2.5-mm lymph node lesions [164].
Figure 5.
[18F] FEAU-PET imaging of mice. (a) A control tumor bearing mice with saline injection and (b) mice without tumor injected with NV1023 had [18F] FEAU signal levels similar to background. (c) Positive PET signal was detected in mice with positive popliteal and inguinal lymph node metastases injected with NV1023. (Reproduced from Brader et al. [162] with permission).
Summary
The lymphatic vessel system imaging is an emerging field of research. It has long been overshadowed by research on angiogenesis because of the lack of imaging modalities to visualize and quantitatively assess lymphatic vasculature in vitro and in vivo. These difficulties were overcome partially during the past few years based on the discovery of lymphatic specific molecular markers and improvement of resolution and sensitivity of imaging modalities. Especially these new imaging modalities contributed to the rapid expansion of our knowledge on the mechanisms underlying lymphatic development and the diseases associated with lymphatic dysfunction. However, efforts are still needed, especially on several critical aspects including (i) optimization of multi-modality probes to take full synergistic advantage of each single imaging modality and minimize drawback; (ii) combination of imaging probes and therapeutic agents to realize theranostic effect for lymphatic targeted therapies; (iii) identification of novel lymphangiogenic markers and development of imaging probes with high affinity and specificity; (iv) development of imaging strategies to improve the ability of visualizing micro-metastases within lymph nodes.
Acknowledgments
F. Zhang and G. Lu are supported by the National Natural Science Foundation of China (NSFC grant No. 30930028) and 973 grants (2006, CB705707, China). G. Niu is an Imaging Sciences Training Program (ISTP) Fellow jointly supported by the Radiology and Imaging Sciences Department, NIH Clinical Center and the Intramural Research Program, NIBIB, NIH. This research was supported by the Intramural Research Program of the NIBIB, NIH. We acknowledge Dr. Henry S. Eden for critically reading this manuscript.
Footnotes
Conflict of Interest Disclosure
The authors declare that they have no conflict of interest.
References
- 1.Barrett T, Choyke PL, Kobayashi H. Imaging of the lymphatic system: new horizons. Contrast Media Mol Imaging. 2006;1:230–245. doi: 10.1002/cmmi.116. [DOI] [PubMed] [Google Scholar]
- 2.Zawieja D. Lymphatic biology and the microcirculation: past, present and future. Microcirculation. 2005;12:141–150. doi: 10.1080/10739680590900003. [DOI] [PubMed] [Google Scholar]
- 3.Mumprecht V, Detmar M. Lymphangiogenesis and cancer metastasis. J Cell Mol Med. 2009;13:1405–1416. doi: 10.1111/j.1582-4934.2009.00834.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alitalo K, Tammela T, Petrova TV. Lymphangiogenesis in development and human disease. Nature. 2005;438:946–953. doi: 10.1038/nature04480. [DOI] [PubMed] [Google Scholar]
- 5.Mandriota SJ, Jussila L, Jeltsch M, et al. Vascular endothelial growth factor-C-mediated lymphangiogenesis promotes tumour metastasis. EMBO J. 2001;20:672–682. doi: 10.1093/emboj/20.4.672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Skobe M, Hawighorst T, Jackson DG, et al. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat Med. 2001;7:192–198. doi: 10.1038/84643. [DOI] [PubMed] [Google Scholar]
- 7.Swartz MA, Skobe M. Lymphatic function, lymphangiogenesis, and cancer metastasis. Microsc Res Tech. 2001;55:92–99. doi: 10.1002/jemt.1160. [DOI] [PubMed] [Google Scholar]
- 8.Hirakawa S, Brown LF, Kodama S, et al. VEGF-C-induced lymphangiogenesis in sentinel lymph nodes promotes tumor metastasis to distant sites. Blood. 2007;109:1010–1017. doi: 10.1182/blood-2006-05-021758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bruyere F, Noel A. Lymphangiogenesis: in vitro and in vivo models. FASEB J. 24:8–21. doi: 10.1096/fj.09-132852. [DOI] [PubMed] [Google Scholar]
- 10.He Y, Rajantie I, Pajusola K, et al. Vascular endothelial cell growth factor receptor 3-mediated activation of lymphatic endothelium is crucial for tumor cell entry and spread via lymphatic vessels. Cancer Res. 2005;65:4739–4746. doi: 10.1158/0008-5472.CAN-04-4576. [DOI] [PubMed] [Google Scholar]
- 11.Tammela T, Alitalo K. Lymphangiogenesis: Molecular mechanisms and future promise. Cell. 140:460–476. doi: 10.1016/j.cell.2010.01.045. [DOI] [PubMed] [Google Scholar]
- 12.Nakamura K, Rockson SG. Biomarkers of lymphatic function and disease: state of the art and future directions. Mol Diagn Ther. 2007;11:227–238. doi: 10.1007/BF03256244. [DOI] [PubMed] [Google Scholar]
- 13.Rinderknecht M, Detmar M. Tumor lymphangiogenesis and melanoma metastasis. J Cell Physiol. 2008;216:347–354. doi: 10.1002/jcp.21494. [DOI] [PubMed] [Google Scholar]
- 14.Pepper MS, Tille JC, Nisato R, Skobe M. Lymphangiogenesis and tumor metastasis. Cell Tissue Res. 2003;314:167–177. doi: 10.1007/s00441-003-0748-7. [DOI] [PubMed] [Google Scholar]
- 15.Ristimaki A, Narko K, Enholm B, Joukov V, Alitalo K. Proinflammatory cytokines regulate expression of the lymphatic endothelial mitogen vascular endothelial growth factor-C. J Biol Chem. 1998;273:8413–8418. doi: 10.1074/jbc.273.14.8413. [DOI] [PubMed] [Google Scholar]
- 16.Saban MR, Memet S, Jackson DG, et al. Visualization of lymphatic vessels through NF-kappaB activity. Blood. 2004;104:3228–3230. doi: 10.1182/blood-2004-04-1428. [DOI] [PubMed] [Google Scholar]
- 17.Wilting J, Becker J, Buttler K, Weich HA. Lymphatics and inflammation. Curr Med Chem. 2009;16:4581–4592. doi: 10.2174/092986709789760751. [DOI] [PubMed] [Google Scholar]
- 18.Moriguchi P, Sannomiya P, Lara PF, et al. Lymphatic system changes in diabetes mellitus: role of insulin and hyperglycemia. Diabetes Metab Res Rev. 2005;21:150–157. doi: 10.1002/dmrr.500. [DOI] [PubMed] [Google Scholar]
- 19.Harvey NL, Srinivasan RS, Dillard ME, et al. Lymphatic vascular defects promoted by Prox1 haploinsufficiency cause adult-onset obesity. Nat Genet. 2005;37:1072–1081. doi: 10.1038/ng1642. [DOI] [PubMed] [Google Scholar]
- 20.Sharma R, Wendt JA, Rasmussen JC, et al. New horizons for imaging lymphatic function. Ann N Y Acad Sci. 2008;1131:13–36. doi: 10.1196/annals.1413.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moghimi SM, Rajabi-Siahboomi R. Advanced colloid-based systems for efficient delivery of drugs and diagnostic agents to the lymphatic tissues. Prog Biophys Mol Biol. 1996;65:221–249. doi: 10.1016/s0079-6107(96)00012-0. [DOI] [PubMed] [Google Scholar]
- 22.Petrova TV, Karpanen T, Norrmen C, et al. Defective valves and abnormal mural cell recruitment underlie lymphatic vascular failure in lymphedema distichiasis. Nat Med. 2004;10:974–981. doi: 10.1038/nm1094. [DOI] [PubMed] [Google Scholar]
- 23.Rodrigues EB, Costa EF, Penha FM, et al. The use of vital dyes in ocular surgery. Surv Ophthalmol. 2009;54:576–617. doi: 10.1016/j.survophthal.2009.04.011. [DOI] [PubMed] [Google Scholar]
- 24.Heath TJ, Brandon RA, Norman ST. Drainage of lymph from the foreleg to the superficial cervical lymph node in sheep. Res Vet Sci. 1984;37:66–71. [PubMed] [Google Scholar]
- 25.Kajiya K, Hirakawa S, Detmar M. Vascular endothelial growth factor-A mediates ultraviolet B-induced impairment of lymphatic vessel function. Am J Pathol. 2006;169:1496–1503. doi: 10.2353/ajpath.2006.060197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Modi S, Stanton AW, Mortimer PS, Levick JR. Clinical assessment of human lymph flow using removal rate constants of interstitial macromolecules: a critical review of lymphoscintigraphy. Lymphat Res Biol. 2007;5:183–202. doi: 10.1089/lrb.2007.5306. [DOI] [PubMed] [Google Scholar]
- 27.Pereira CT, Luiz Navarro Marques F, Williams J, Wlademir De Martin B, Primo Bombonato P. 99mTc-labeled dextran for mammary lymphoscintigraphy in dogs. Vet Radiol Ultrasound. 2008;49:487–491. doi: 10.1111/j.1740-8261.2008.00414.x. [DOI] [PubMed] [Google Scholar]
- 28.Anthony JP, Foster RD, Price DC, Mahdavian M, Inoue Y. Lymphatic regeneration following microvascular limb replantation: a qualitative and quantitative animal study. J Reconstr Microsurg. 1997;13:327–330. doi: 10.1055/s-2007-1006411. [DOI] [PubMed] [Google Scholar]
- 29.Alex JC, Krag DN. Gamma-probe guided localization of lymph nodes. Surg Oncol. 1993;2:137–143. doi: 10.1016/0960-7404(93)90001-f. [DOI] [PubMed] [Google Scholar]
- 30.Czerniecki BJ, Bedrosian I, Faries M, Alavi A. Revolutionary impact of lymphoscintigraphy and intraoperative sentinel node mapping in the clinical practice of oncology. Semin Nucl Med. 2001;31:158–164. doi: 10.1053/snuc.2001.21270. [DOI] [PubMed] [Google Scholar]
- 31.Nakashima K, Kurebayashi J, Sonoo H, et al. Preoperative dynamic lymphoscintigraphy predicts sentinel lymph node metastasis in patients with early breast cancer. Breast Cancer. 2010;17:17–21. doi: 10.1007/s12282-009-0123-y. [DOI] [PubMed] [Google Scholar]
- 32.Harivardhan Reddy L, Sharma RK, Chuttani K, Mishra AK, Murthy RS. Influence of administration route on tumor uptake and biodistribution of etoposide loaded solid lipid nanoparticles in Dalton’s lymphoma tumor bearing mice. J Control Release. 2005;105:185–198. doi: 10.1016/j.jconrel.2005.02.028. [DOI] [PubMed] [Google Scholar]
- 33.Boinpally RR, Zhou SL, Devraj G, et al. Iontophoresis of lecithin vesicles of cyclosporin A. Int J Pharm. 2004;274:185–190. doi: 10.1016/j.ijpharm.2004.01.016. [DOI] [PubMed] [Google Scholar]
- 34.Holm R, Porter CJ, Edwards GA, et al. Examination of oral absorption and lymphatic transport of halofantrine in a triple-cannulated canine model after administration in self-microemulsifying drug delivery systems (SMEDDS) containing structured triglycerides. Eur J Pharm Sci. 2003;20:91–97. doi: 10.1016/s0928-0987(03)00174-x. [DOI] [PubMed] [Google Scholar]
- 35.Kim J, Chung KH, Lee CM, et al. Lymphatic delivery of 99mTc-labeled dextran acetate particles including cyclosporine A. J Microbiol Biotechnol. 2008;18:1599–1605. [PubMed] [Google Scholar]
- 36.Mehnert W, Mader K. Solid lipid nanoparticles: production, characterization and applications. Adv Drug Deliv Rev. 2001;47:165–196. doi: 10.1016/s0169-409x(01)00105-3. [DOI] [PubMed] [Google Scholar]
- 37.Videira MA, Botelho MF, Santos AC, et al. Lymphatic uptake of pulmonary delivered radiolabelled solid lipid nanoparticles. J Drug Target. 2002;10:607–613. doi: 10.1080/1061186021000054933. [DOI] [PubMed] [Google Scholar]
- 38.Lucarelli RT, Ogawa M, Kosaka N, et al. New approaches to lymphatic imaging. Lymphat Res Biol. 2009;7:205–214. doi: 10.1089/lrb.2009.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Delaey E, van Laar F, De Vos D, et al. A comparative study of the photosensitizing characteristics of some cyanine dyes. J Photochem Photobiol B. 2000;55:27–36. doi: 10.1016/s1011-1344(00)00021-x. [DOI] [PubMed] [Google Scholar]
- 40.Landsman ML, Kwant G, Mook GA, Zijlstra WG. Light-absorbing properties, stability, and spectral stabilization of indocyanine green. J Appl Physiol. 1976;40:575–583. doi: 10.1152/jappl.1976.40.4.575. [DOI] [PubMed] [Google Scholar]
- 41.Branch RA, James JA, Read AE. The clearance of antipyrine and indocyanine green in normal subjects and in patients with chronic lever disease. Clin Pharmacol Ther. 1976;20:81–89. doi: 10.1002/cpt197620181. [DOI] [PubMed] [Google Scholar]
- 42.Kwon S, Sevick-Muraca EM. Noninvasive quantitative imaging of lymph function in mice. Lymphat Res Biol. 2007;5:219–231. doi: 10.1089/lrb.2007.1013. [DOI] [PubMed] [Google Scholar]
- 43.Sharma R, Wang W, Rasmussen JC, et al. Quantitative imaging of lymph function. Am J Physiol Heart Circ Physiol. 2007;292:H3109–3118. doi: 10.1152/ajpheart.01223.2006. [DOI] [PubMed] [Google Scholar]
- 44.Hirche C, Murawa D, Mohr Z, Kneif S, Hunerbein M. ICG fluorescence-guided sentinel node biopsy for axillary nodal staging in breast cancer. Breast Cancer Res Treat. doi: 10.1007/s10549-010-0760-z. [DOI] [PubMed] [Google Scholar]
- 45.Murawa D, Hirche C, Dresel S, Hunerbein M. Sentinel lymph node biopsy in breast cancer guided by indocyanine green fluorescence. Br J Surg. 2009;96:1289–1294. doi: 10.1002/bjs.6721. [DOI] [PubMed] [Google Scholar]
- 46.Fujiwara M, Mizukami T, Suzuki A, Fukamizu H. Sentinel lymph node detection in skin cancer patients using real-time fluorescence navigation with indocyanine green: preliminary experience. J Plast Reconstr Aesthet Surg. 2009;62:e373–378. doi: 10.1016/j.bjps.2007.12.074. [DOI] [PubMed] [Google Scholar]
- 47.Ghoroghchian PP, Therien MJ, Hammer DA. In vivo fluorescence imaging: a personal perspective. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1:156–167. doi: 10.1002/wnan.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hama Y, Koyama Y, Urano Y, Choyke PL, Kobayashi H. Two-color lymphatic mapping using Ig-conjugated near infrared optical probes. J Invest Dermatol. 2007;127:2351–2356. doi: 10.1038/sj.jid.5700892. [DOI] [PubMed] [Google Scholar]
- 49.Dubertret B, Skourides P, Norris DJ, et al. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science. 2002;298:1759–1762. doi: 10.1126/science.1077194. [DOI] [PubMed] [Google Scholar]
- 50.Gao X, Cui Y, Levenson RM, Chung LW, Nie S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol. 2004;22:969–976. doi: 10.1038/nbt994. [DOI] [PubMed] [Google Scholar]
- 51.Michalet X, Pinaud FF, Bentolila LA, et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science. 2005;307:538–544. doi: 10.1126/science.1104274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Harrell MI, Iritani BM, Ruddell A. Tumor-induced sentinel lymph node lymphangiogenesis and increased lymph flow precede melanoma metastasis. Am J Pathol. 2007;170:774–786. doi: 10.2353/ajpath.2007.060761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ballou B, Ernst LA, Andreko S, et al. Sentinel lymph node imaging using quantum dots in mouse tumor models. Bioconjug Chem. 2007;18:389–396. doi: 10.1021/bc060261j. [DOI] [PubMed] [Google Scholar]
- 54.Parungo CP, Colson YL, Kim SW, et al. Sentinel lymph node mapping of the pleural space. Chest. 2005;127:1799–1804. doi: 10.1378/chest.127.5.1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Parungo CP, Soybel DI, Colson YL, et al. Lymphatic drainage of the peritoneal space: a pattern dependent on bowel lymphatics. Ann Surg Oncol. 2007;14:286–298. doi: 10.1245/s10434-006-9044-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kosaka N, Ogawa M, Sato N, Choyke PL, Kobayashi H. In vivo real-time, multicolor, quantum dot lymphatic imaging. J Invest Dermatol. 2009;129:2818–2822. doi: 10.1038/jid.2009.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kim S, Lim YT, Soltesz EG, et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat Biotechnol. 2004;22:93–97. doi: 10.1038/nbt920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Knapp DW, Adams LG, Degrand AM, et al. Sentinel lymph node mapping of invasive urinary bladder cancer in animal models using invisible light. Eur Urol. 2007;52:1700–1708. doi: 10.1016/j.eururo.2007.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Harrell MI, Iritani BM, Ruddell A. Lymph node mapping in the mouse. J Immunol Methods. 2008;332:170–174. doi: 10.1016/j.jim.2007.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wu H, Xu X, Ying H, et al. Preliminary study of indirect CT lymphography-guided sentinel lymph node biopsy in a tongue VX2 carcinoma model. Int J Oral Maxillofac Surg. 2009;38:1268–1272. doi: 10.1016/j.ijom.2009.07.009. [DOI] [PubMed] [Google Scholar]
- 61.Suga K, Ogasawara N, Okada M, Matsunaga N. Interstitial CT lymphography-guided localization of breast sentinel lymph node: preliminary results. Surgery. 2003;133:170–179. doi: 10.1067/msy.2003.17. [DOI] [PubMed] [Google Scholar]
- 62.Suga K, Yuan Y, Okada M, et al. Breast sentinel lymph node mapping at CT lymphography with iopamidol: preliminary experience. Radiology. 2004;230:543–552. doi: 10.1148/radiol.2302021380. [DOI] [PubMed] [Google Scholar]
- 63.Suga K, Yamamoto S, Tangoku A, et al. Breast sentinel lymph node navigation with three-dimensional interstitial multidetector-row computed tomographic lymphography. Invest Radiol. 2005;40:336–342. doi: 10.1097/01.rli.0000164153.41638.32. [DOI] [PubMed] [Google Scholar]
- 64.Aviv H, Bartling S, Kieslling F, Margel S. Radiopaque iodinated copolymeric nanoparticles for X-ray imaging applications. Biomaterials. 2009;30:5610–5616. doi: 10.1016/j.biomaterials.2009.06.038. [DOI] [PubMed] [Google Scholar]
- 65.Galperin A, Margel D, Baniel J, et al. Radiopaque iodinated polymeric nanoparticles for X-ray imaging applications. Biomaterials. 2007;28:4461–4468. doi: 10.1016/j.biomaterials.2007.06.032. [DOI] [PubMed] [Google Scholar]
- 66.Rabin O, Manuel Perez J, Grimm J, Wojtkiewicz G, Weissleder R. An X-ray computed tomography imaging agent based on long-circulating bismuth sulphide nanoparticles. Nat Mater. 2006;5:118–122. doi: 10.1038/nmat1571. [DOI] [PubMed] [Google Scholar]
- 67.Sun MR, Ngo L, Genega EM, et al. Renal cell carcinoma: dynamic contrast-enhanced MR imaging for differentiation of tumor subtypes--correlation with pathologic findings. Radiology. 2009;250:793–802. doi: 10.1148/radiol.2503080995. [DOI] [PubMed] [Google Scholar]
- 68.Kobayashi H, Shirakawa K, Kawamoto S, et al. Rapid accumulation and internalization of radiolabeled herceptin in an inflammatory breast cancer xenograft with vasculogenic mimicry predicted by the contrast-enhanced dynamic MRI with the macromolecular contrast agent G6-(1B4M-Gd)(256) Cancer Res. 2002;62:860–866. [PubMed] [Google Scholar]
- 69.Ruddell A, Harrell MI, Minoshima S, et al. Dynamic contrast-enhanced magnetic resonance imaging of tumor-induced lymph flow. Neoplasia. 2008;10:706–713. 701. doi: 10.1593/neo.08342. following 713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ruehm SG, Corot C, Debatin JF. Interstitial MR lymphography with a conventional extracellular gadolinium-based agent: assessment in rabbits. Radiology. 2001;218:664–669. doi: 10.1148/radiology.218.3.r01fe21664. [DOI] [PubMed] [Google Scholar]
- 71.Ruehm SG, Schroeder T, Debatin JF. Interstitial MR lymphography with gadoterate meglumine: initial experience in humans. Radiology. 2001;220:816–821. doi: 10.1148/radiol.2203010090. [DOI] [PubMed] [Google Scholar]
- 72.Misselwitz B. MR contrast agents in lymph node imaging. Eur J Radiol. 2006;58:375–382. doi: 10.1016/j.ejrad.2005.12.044. [DOI] [PubMed] [Google Scholar]
- 73.Kobayashi H, Kawamoto S, Brechbiel MW, et al. Detection of lymph node involvement in hematologic malignancies using micromagnetic resonance lymphangiography with a gadolinum-labeled dendrimer nanoparticle. Neoplasia. 2005;7:984–991. doi: 10.1593/neo.05454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Herborn CU, Lauenstein TC, Vogt FM, et al. Interstitial MR lymphography with MS-325: characterization of normal and tumor-invaded lymph nodes in a rabbit model. AJR Am J Roentgenol. 2002;179:1567–1572. doi: 10.2214/ajr.179.6.1791567. [DOI] [PubMed] [Google Scholar]
- 75.Mouli SK, Zhao LC, Omary RA, Thaxton CS. Lymphotropic nanoparticle enhanced MRI for the staging of genitourinary tumors. Nat Rev Urol. 7:84–93. doi: 10.1038/nrurol.2009.254. [DOI] [PubMed] [Google Scholar]
- 76.Feldman AS, McDougal WS, Harisinghani MG. The potential of nanoparticle-enhanced imaging. Urol Oncol. 2008;26:65–73. doi: 10.1016/j.urolonc.2007.03.016. [DOI] [PubMed] [Google Scholar]
- 77.Saokar A, Braschi M, Harisinghani MG. Lymphotrophic nanoparticle enhanced MR imaging (LNMRI) for lymph node imaging. Abdom Imaging. 2006 doi: 10.1007/s00261-006-9006-2. [DOI] [PubMed] [Google Scholar]
- 78.Weissleder R, Elizondo G, Wittenberg J, et al. Ultrasmall superparamagnetic iron oxide: an intravenous contrast agent for assessing lymph nodes with MR imaging. Radiology. 1990;175:494–498. doi: 10.1148/radiology.175.2.2326475. [DOI] [PubMed] [Google Scholar]
- 79.Bellin MF, Beigelman C, Precetti-Morel S. Iron oxide-enhanced MR lymphography: initial experience. Eur J Radiol. 2000;34:257–264. doi: 10.1016/s0720-048x(00)00204-7. [DOI] [PubMed] [Google Scholar]
- 80.Harisinghani MG, Dixon WT, Saksena MA, et al. MR lymphangiography: imaging strategies to optimize the imaging of lymph nodes with ferumoxtran-10. Radiographics. 2004;24:867–878. doi: 10.1148/rg.243035190. [DOI] [PubMed] [Google Scholar]
- 81.Weissleder R, Stark DD, Engelstad BL, et al. Superparamagnetic iron oxide: pharmacokinetics and toxicity. AJR Am J Roentgenol. 1989;152:167–173. doi: 10.2214/ajr.152.1.167. [DOI] [PubMed] [Google Scholar]
- 82.Taupitz M, Wagner S, Hamm B, Binder A, Pfefferer D. Interstitial MR lymphography with iron oxide particles: results in tumor-free and VX2 tumor-bearing rabbits. AJR Am J Roentgenol. 1993;161:193–200. doi: 10.2214/ajr.161.1.8517301. [DOI] [PubMed] [Google Scholar]
- 83.Misselwitz B, Sachse A. Interstitial MR lymphography using Gd-carrying liposomes. Acta Radiol Suppl. 1997;412:51–55. [PubMed] [Google Scholar]
- 84.Trubetskoy VS, Cannillo JA, Milshtein A, Wolf GL, Torchilin VP. Controlled delivery of Gd-containing liposomes to lymph nodes: surface modification may enhance MRI contrast properties. Magn Reson Imaging. 1995;13:31–37. doi: 10.1016/0730-725x(94)00083-f. [DOI] [PubMed] [Google Scholar]
- 85.Kobayashi H, Kawamoto S, Choyke PL, et al. Comparison of dendrimer-based macromolecular contrast agents for dynamic micro-magnetic resonance lymphangiography. Magn Reson Med. 2003;50:758–766. doi: 10.1002/mrm.10583. [DOI] [PubMed] [Google Scholar]
- 86.Kobayashi H, Kawamoto S, Star RA, et al. Micro-magnetic resonance lymphangiography in mice using a novel dendrimer-based magnetic resonance imaging contrast agent. Cancer Res. 2003;63:271–276. [PubMed] [Google Scholar]
- 87.Kobayashi H, Kawamoto S, Sakai Y, et al. Lymphatic drainage imaging of breast cancer in mice by micro-magnetic resonance lymphangiography using a nano-size paramagnetic contrast agent. J Natl Cancer Inst. 2004;96:703–708. doi: 10.1093/jnci/djh124. [DOI] [PubMed] [Google Scholar]
- 88.Staatz G, Spuntrup E, Klosterhalfen B, et al. High-resolution T1-weighted MR-lymphography of inguinal lymph nodes after interstitial application of Gadomer-17 in animal experiments. Rofo. 2005;177:968–974. doi: 10.1055/s-2005-858239. [DOI] [PubMed] [Google Scholar]
- 89.Torchia MG, Misselwitz B. Combined MR lymphangiography and MR imaging-guided needle localization of sentinel lymph nodes using Gadomer-17. AJR Am J Roentgenol. 2002;179:1561–1565. doi: 10.2214/ajr.179.6.1791561. [DOI] [PubMed] [Google Scholar]
- 90.Gramiak R, Shah PM, Kramer DH. Ultrasound cardiography: contrast studies in anatomy and function. Radiology. 1969;92:939–948. doi: 10.1148/92.5.939. [DOI] [PubMed] [Google Scholar]
- 91.Voigt JU. Ultrasound molecular imaging. Methods. 2009;48:92–97. doi: 10.1016/j.ymeth.2009.03.011. [DOI] [PubMed] [Google Scholar]
- 92.Dayton PA, Rychak JJ. Molecular ultrasound imaging using microbubble contrast agents. Front Biosci. 2007;12:5124–5142. doi: 10.2741/2553. [DOI] [PubMed] [Google Scholar]
- 93.Lindner JR, Coggins MP, Kaul S, et al. Microbubble persistence in the microcirculation during ischemia/reperfusion and inflammation is caused by integrin- and complement-mediated adherence to activated leukocytes. Circulation. 2000;101:668–675. doi: 10.1161/01.cir.101.6.668. [DOI] [PubMed] [Google Scholar]
- 94.Lurie DM, Seguin B, Schneider PD, Verstraete FJ, Wisner ER. Contrast-assisted ultrasound for sentinel lymph node detection in spontaneously arising canine head and neck tumors. Invest Radiol. 2006;41:415–421. doi: 10.1097/01.rli.0000201230.29925.95. [DOI] [PubMed] [Google Scholar]
- 95.Nielsen KR, Grossjohann HS, Hansen CP, Nielsen MB. Use of contrast-enhanced ultrasound imaging to detect the first draining lymph node (FDLN) in a swine model: correlation of imaging findings with the distance from the injection site to the FDLN. J Ultrasound Med. 2008;27:1203–1209. doi: 10.7863/jum.2008.27.8.1203. [DOI] [PubMed] [Google Scholar]
- 96.Sever A, Jones S, Cox K, et al. Preoperative localization of sentinel lymph nodes using intradermal microbubbles and contrast-enhanced ultrasonography in patients with breast cancer. Br J Surg. 2009;96:1295–1299. doi: 10.1002/bjs.6725. [DOI] [PubMed] [Google Scholar]
- 97.Goldberg BB, Merton DA, Liu JB, et al. Sentinel lymph nodes in a swine model with melanoma: contrast-enhanced lymphatic US. Radiology. 2004;230:727–734. doi: 10.1148/radiol.2303021440. [DOI] [PubMed] [Google Scholar]
- 98.Ravizzini G, Turkbey B, Barrett T, Kobayashi H, Choyke PL. Nanoparticles in sentinel lymph node mapping. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1:610–623. doi: 10.1002/wnan.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Song KH, Stoica G, Wang LV. In vivo three-dimensional photoacoustic tomography of a whole mouse head. Opt Lett. 2006;31:2453–2455. doi: 10.1364/ol.31.002453. [DOI] [PubMed] [Google Scholar]
- 100.Wang X, Pang Y, Ku G, et al. Noninvasive laser-induced photoacoustic tomography for structural and functional in vivo imaging of the brain. Nat Biotechnol. 2003;21:803–806. doi: 10.1038/nbt839. [DOI] [PubMed] [Google Scholar]
- 101.Song KH, Stein EW, Margenthaler JA, Wang LV. Noninvasive photoacoustic identification of sentinel lymph nodes containing methylene blue in vivo in a rat model. J Biomed Opt. 2008;13:054033. doi: 10.1117/1.2976427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.De la Zerda A, Zavaleta C, Keren S, et al. Carbon nanotubes as photoacoustic molecular imaging agents in living mice. Nat Nanotechnol. 2008;3:557–562. doi: 10.1038/nnano.2008.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kim JW, Galanzha EI, Shashkov EV, Moon HM, Zharov VP. Golden carbon nanotubes as multimodal photoacoustic and photothermal high-contrast molecular agents. Nat Nanotechnol. 2009;4:688–694. doi: 10.1038/nnano.2009.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Pan D, Pramanik M, Senpan A, et al. Near infrared photoacoustic detection of sentinel lymph nodes with gold nanobeacons. Biomaterials. 2010;31:4088–4093. doi: 10.1016/j.biomaterials.2010.01.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pramanik M, Song KH, Swierczewska M, et al. In vivo carbon nanotube-enhanced non-invasive photoacoustic mapping of the sentinel lymph node. Phys Med Biol. 2009;54:3291–3301. doi: 10.1088/0031-9155/54/11/001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.McCormack D, Al-Shaer M, Goldschmidt BS, et al. Photoacoustic detection of melanoma micrometastasis in sentinel lymph nodes. J Biomech Eng. 2009;131:074519. doi: 10.1115/1.3169247. [DOI] [PubMed] [Google Scholar]
- 107.Jones T. The imaging science of positron emission tomography. Eur J Nucl Med. 1996;23:807–813. doi: 10.1007/BF00843711. [DOI] [PubMed] [Google Scholar]
- 108.Phelps ME, Hoffman EJ, Mullani NA, Ter-Pogossian MM. Application of annihilation coincidence detection to transaxial reconstruction tomography. J Nucl Med. 1975;16:210–224. [PubMed] [Google Scholar]
- 109.Phelps ME. PET: the merging of biology and imaging into molecular imaging. J Nucl Med. 2000;41:661–681. [PubMed] [Google Scholar]
- 110.Schoder H, Glass EC, Pecking AP, et al. Molecular targeting of the lymphovascular system for imaging and therapy. Cancer Metastasis Rev. 2006;25:185–201. doi: 10.1007/s10555-006-8498-0. [DOI] [PubMed] [Google Scholar]
- 111.Machtens S, Serth J, Meyer A, et al. Positron emission tomography (PET) in the urooncological evaluation of the small pelvis. World J Urol. 2007;25:341–349. doi: 10.1007/s00345-007-0194-3. [DOI] [PubMed] [Google Scholar]
- 112.de Jong IJ, Pruim J, Elsinga PH, Vaalburg W, Mensink HJ. Preoperative staging of pelvic lymph nodes in prostate cancer by 11C-choline PET. J Nucl Med. 2003;44:331–335. [PubMed] [Google Scholar]
- 113.Beheshti M, Imamovic L, Broinger G, et al. 18F choline PET/CT in the preoperative staging of prostate cancer in patients with intermediate or high risk of extracapsular disease: a prospective study of 130 patients. Radiology. 2010;254:925–933. doi: 10.1148/radiol.09090413. [DOI] [PubMed] [Google Scholar]
- 114.Fricke E, Machtens S, Hofmann M, et al. Positron emission tomography with 11C-acetate and 18F-FDG in prostate cancer patients. Eur J Nucl Med Mol Imaging. 2003;30:607–611. doi: 10.1007/s00259-002-1104-y. [DOI] [PubMed] [Google Scholar]
- 115.Larson SM, Schoder H. New PET tracers for evaluation of solid tumor response to therapy. Q J Nucl Med Mol Imaging. 2009;53:158–166. [PubMed] [Google Scholar]
- 116.Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS. Molecular imaging in drug development. Nat Rev Drug Discov. 2008;7:591–607. doi: 10.1038/nrd2290. [DOI] [PubMed] [Google Scholar]
- 117.Heusner TA, Kuemmel S, Hahn S, et al. Diagnostic value of full-dose FDG PET/CT for axillary lymph node staging in breast cancer patients. Eur J Nucl Med Mol Imaging. 2009;36:1543–1550. doi: 10.1007/s00259-009-1145-6. [DOI] [PubMed] [Google Scholar]
- 118.Klode J, Dissemond J, Grabbe S, et al. Sentinel lymph node excision and PET-CT in the initial stage of malignant melanoma: a retrospective analysis of 61 patients with malignant melanoma in American Joint Committee on Cancer stages I and II. Dermatol Surg. 2010;36:439–445. doi: 10.1111/j.1524-4725.2010.01479.x. [DOI] [PubMed] [Google Scholar]
- 119.Niu G, Cai W, Chen X. Molecular imaging of human epidermal growth factor receptor 2 (HER-2) expression. Front Biosci. 2008;13:790–805. doi: 10.2741/2720. [DOI] [PubMed] [Google Scholar]
- 120.Cai W, Niu G, Chen X. Multimodality imaging of the HER-kinase axis in cancer. Eur J Nucl Med Mol Imaging. 2008;35:186–208. doi: 10.1007/s00259-007-0560-9. [DOI] [PubMed] [Google Scholar]
- 121.Kobayashi H, Koyama Y, Barrett T, et al. Multimodal nanoprobes for radionuclide and five-color near-infrared optical lymphatic imaging. ACS Nano. 2007;1:258–264. doi: 10.1021/nn700062z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cai W, Chen X. Multimodality molecular imaging of tumor angiogenesis. J Nucl Med. 2008;49 (Suppl 2):113S–128S. doi: 10.2967/jnumed.107.045922. [DOI] [PubMed] [Google Scholar]
- 123.Lee S, Chen X. Dual-modality probes for in vivo molecular imaging. Mol Imaging. 2009;8:87–100. [PubMed] [Google Scholar]
- 124.Veit P, Ruehm S, Kuehl H, et al. Lymph node staging with dual-modality PET/CT: enhancing the diagnostic accuracy in oncology. Eur J Radiol. 2006;58:383–389. doi: 10.1016/j.ejrad.2005.12.042. [DOI] [PubMed] [Google Scholar]
- 125.Kamel E, Hany TF, Burger C, et al. CT vs 68Ge attenuation correction in a combined PET/CT system: evaluation of the effect of lowering the CT tube current. Eur J Nucl Med Mol Imaging. 2002;29:346–350. doi: 10.1007/s00259-001-0698-9. [DOI] [PubMed] [Google Scholar]
- 126.Townsend DW. Dual-modality imaging: combining anatomy and function. J Nucl Med. 2008;49:938–955. doi: 10.2967/jnumed.108.051276. [DOI] [PubMed] [Google Scholar]
- 127.Antoch G, Stattaus J, Nemat AT, et al. Non-small cell lung cancer: dual-modality PET/CT in preoperative staging. Radiology. 2003;229:526–533. doi: 10.1148/radiol.2292021598. [DOI] [PubMed] [Google Scholar]
- 128.Lardinois D, Weder W, Hany TF, et al. Staging of non-small-cell lung cancer with integrated positron-emission tomography and computed tomography. N Engl J Med. 2003;348:2500–2507. doi: 10.1056/NEJMoa022136. [DOI] [PubMed] [Google Scholar]
- 129.Mar MV, Miller SA, Kim EE, Macapinlac HA. Evaluation and localization of lymphatic drainage and sentinel lymph nodes in patients with head and neck melanomas by hybrid SPECT/CT lymphoscintigraphic imaging. J Nucl Med Technol. 2007;35:10–16. quiz 17–20. [PubMed] [Google Scholar]
- 130.Cherry SR, Shao Y, Silverman RW, et al. MicroPET: a high resolution PET scanner for imaging small animals. IEEE Trans Nucl Sci. 1997;44:1161–1166. [Google Scholar]
- 131.Chatziioannou AF, Cherry SR, Shao Y, et al. Performance evaluation of microPET: a high-resolution lutetium oxyorthosilicate PET scanner for animal imaging. J Nucl Med. 1999;40:1164–1175. [PMC free article] [PubMed] [Google Scholar]
- 132.Delso G, Ziegler S. PET/MRI system design. Eur J Nucl Med Mol Imaging. 2009;36 (Suppl 1):S86–92. doi: 10.1007/s00259-008-1008-6. [DOI] [PubMed] [Google Scholar]
- 133.Catana C, Procissi D, Wu Y, et al. Simultaneous in vivo positron emission tomography and magnetic resonance imaging. Proc Natl Acad Sci U S A. 2008;105:3705–3710. doi: 10.1073/pnas.0711622105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Judenhofer MS, Wehrl HF, Newport DF, et al. Simultaneous PET-MRI: a new approach for functional and morphological imaging. Nat Med. 2008;14:459–465. doi: 10.1038/nm1700. [DOI] [PubMed] [Google Scholar]
- 135.Schlemmer HP, Pichler BJ, Schmand M, et al. Simultaneous MR/PET imaging of the human brain: feasibility study. Radiology. 2008;248:1028–1035. doi: 10.1148/radiol.2483071927. [DOI] [PubMed] [Google Scholar]
- 136.Kim T, Giuliano AE, Lyman GH. Lymphatic mapping and sentinel lymph node biopsy in early-stage breast carcinoma: a metaanalysis. Cancer. 2006;106:4–16. doi: 10.1002/cncr.21568. [DOI] [PubMed] [Google Scholar]
- 137.Wong SL, Edwards MJ, Chao C, et al. Sentinel lymph node biopsy for breast cancer: impact of the number of sentinel nodes removed on the false-negative rate. J Am Coll Surg. 2001;192:684–689. doi: 10.1016/s1072-7515(01)00858-4. discussion 689-691. [DOI] [PubMed] [Google Scholar]
- 138.El-Tamer M, Saouaf R, Wang T, Fawwaz R. A new agent, blue and radioactive, for sentinel node detection. Ann Surg Oncol. 2003;10:323–329. doi: 10.1245/aso.2003.03.087. [DOI] [PubMed] [Google Scholar]
- 139.Stafford SJ, Wright JL, Schwimer J, et al. Development of 125I-methylene blue for sentinel lymph node biopsy. J Surg Oncol. 2006;94:293–297. doi: 10.1002/jso.20444. [DOI] [PubMed] [Google Scholar]
- 140.Tsopelas C, Bellon M, Bevington E, et al. Lymphatic mapping with 99mTc-Evans Blue dye in sheep. Ann Nucl Med. 2008;22:777–785. doi: 10.1007/s12149-008-0171-y. [DOI] [PubMed] [Google Scholar]
- 141.Koyama Y, Talanov VS, Bernardo M, et al. A dendrimer-based nanosized contrast agent dual-labeled for magnetic resonance and optical fluorescence imaging to localize the sentinel lymph node in mice. J Magn Reson Imaging. 2007;25:866–871. doi: 10.1002/jmri.20852. [DOI] [PubMed] [Google Scholar]
- 142.Chen K, Xie J, Xu H, et al. Triblock copolymer coated iron oxide nanoparticle conjugate for tumor integrin targeting. Biomaterials. 2009;30:6912–6919. doi: 10.1016/j.biomaterials.2009.08.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Lee HY, Li Z, Chen K, et al. PET/MRI dual-modality tumor imaging using arginine-glycine-aspartic (RGD)-conjugated radiolabeled iron oxide nanoparticles. J Nucl Med. 2008;49:1371–1379. doi: 10.2967/jnumed.108.051243. [DOI] [PubMed] [Google Scholar]
- 144.Xie J, Chen K, Huang J, et al. PET/NIRF/MRI triple functional iron oxide nanoparticles. Biomaterials. 2010;31:3016–3022. doi: 10.1016/j.biomaterials.2010.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wang LV. Prospects of photoacoustic tomography. Med Phys. 2008;35:5758–5767. doi: 10.1118/1.3013698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Erpelding TN, Kim C, Pramanik M, et al. Sentinel lymph nodes in the rat: noninvasive photoacoustic and US imaging with a clinical US system. Radiology. 2010;256:102–110. doi: 10.1148/radiol.10091772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Niu G, Chen X. PET Imaging of Angiogenesis. PET Clin. 2009;4:17–38. doi: 10.1016/j.cpet.2009.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Banerji S, Ni J, Wang SX, et al. LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J Cell Biol. 1999;144:789–801. doi: 10.1083/jcb.144.4.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Breiteneder-Geleff S, Soleiman A, Horvat R, et al. Podoplanin--a specific marker for lymphatic endothelium expressed in angiosarcoma. Verh Dtsch Ges Pathol. 1999;83:270–275. [PubMed] [Google Scholar]
- 150.Laakkonen P, Porkka K, Hoffman JA, Ruoslahti E. A tumor-homing peptide with a targeting specificity related to lymphatic vessels. Nat Med. 2002;8:751–755. doi: 10.1038/nm720. [DOI] [PubMed] [Google Scholar]
- 151.McElroy M, Hayashi K, Garmy-Susini B, et al. Fluorescent LYVE-1 antibody to image dynamically lymphatic trafficking of cancer cells in vivo. J Surg Res. 2009;151:68–73. doi: 10.1016/j.jss.2007.12.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Laakkonen P, Zhang L, Ruoslahti E. Peptide targeting of tumor lymph vessels. Ann N Y Acad Sci. 2008;1131:37–43. doi: 10.1196/annals.1413.003. [DOI] [PubMed] [Google Scholar]
- 153.Laakkonen P, Akerman ME, Biliran H, et al. Antitumor activity of a homing peptide that targets tumor lymphatics and tumor cells. Proc Natl Acad Sci U S A. 2004;101:9381–9386. doi: 10.1073/pnas.0403317101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Weller GE, Lu E, Csikari MM, et al. Ultrasound imaging of acute cardiac transplant rejection with microbubbles targeted to intercellular adhesion molecule-1. Circulation. 2003;108:218–224. doi: 10.1161/01.CIR.0000080287.74762.60. [DOI] [PubMed] [Google Scholar]
- 155.Korpanty G, Carbon JG, Grayburn PA, Fleming JB, Brekken RA. Monitoring response to anticancer therapy by targeting microbubbles to tumor vasculature. Clin Cancer Res. 2007;13:323–330. doi: 10.1158/1078-0432.CCR-06-1313. [DOI] [PubMed] [Google Scholar]
- 156.Wright WH, Jr, McCreery TP, Krupinski EA, et al. Evaluation of new thrombus-specific ultrasound contrast agent. Acad Radiol. 1998;5 (Suppl 1):S240–242. doi: 10.1016/s1076-6332(98)80117-7. [DOI] [PubMed] [Google Scholar]
- 157.Willmann JK, Paulmurugan R, Chen K, et al. US imaging of tumor angiogenesis with microbubbles targeted to vascular endothelial growth factor receptor type 2 in mice. Radiology. 2008;246:508–518. doi: 10.1148/radiol.2462070536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Li X, Jin Q, Chen T, et al. LyP-1 ultrasonic microbubbles targeting to cancer cell as tumor bio-acoustics markers or drug carriers: targeting efficiency evaluation in, microfluidic channels. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:463–466. doi: 10.1109/IEMBS.2009.5334473. [DOI] [PubMed] [Google Scholar]
- 159.Hauff P, Reinhardt M, Briel A, Debus N, Schirner M. Molecular targeting of lymph nodes with L-selectin ligand-specific US contrast agent: a feasibility study in mice and dogs. Radiology. 2004;231:667–673. doi: 10.1148/radiol.2313030425. [DOI] [PubMed] [Google Scholar]
- 160.Yaghoubi SS, Gambhir SS. PET imaging of herpes simplex virus type 1 thymidine kinase (HSV1-tk) or mutant HSV1-sr39tk reporter gene expression in mice and humans using [18F]FHBG. Nat Protoc. 2006;1:3069–3075. doi: 10.1038/nprot.2006.459. [DOI] [PubMed] [Google Scholar]
- 161.Pouliot F, Johnson M, Wu L. Non-invasive molecular imaging of prostate cancer lymph node metastasis. Trends Mol Med. 2009;15:254–262. doi: 10.1016/j.molmed.2009.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Brader P, Kelly K, Gang S, et al. Imaging of lymph node micrometastases using an oncolytic herpes virus and [F]FEAU PET. PLoS One. 2009;4:e4789. doi: 10.1371/journal.pone.0004789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Iyer M, Wu L, Carey M, et al. Two-step transcriptional amplification as a method for imaging reporter gene expression using weak promoters. Proc Natl Acad Sci U S A. 2001;98:14595–14600. doi: 10.1073/pnas.251551098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Burton JB, Johnson M, Sato M, et al. Adenovirus-mediated gene expression imaging to directly detect sentinel lymph node metastasis of prostate cancer. Nat Med. 2008;14:882–888. doi: 10.1038/nm.1727. [DOI] [PMC free article] [PubMed] [Google Scholar]