Abstract
Amikacin efficacy is based on peak concentrations and the possibility of reaching therapeutic levels at the infection site. This study aimed to describe amikacin concentrations in the epithelial lining fluid (ELF) through bronchoalveolar lavage (BAL) in newborns. BAL fluid was collected in ventilated neonates treated with intravenous (i.v.) amikacin. Clinical characteristics, amikacin therapeutic drug monitoring serum concentrations, and the concentrations of urea in plasma were extracted from the individual patient files. Amikacin and urea BAL fluid concentrations were determined using liquid chromatography with pulsed electrochemical detection (LC-PED) and capillary electrophoresis with capacitively coupled contactless conductivity detection (CE-C4D), respectively. ELF amikacin concentrations were converted from BAL fluid concentrations through quantification of dilution (urea in plasma/urea in BAL fluid) during the BAL procedure. Twenty-two observations in 17 neonates (postmenstrual age, 31.9 [range, 25.1 to 41] weeks; postnatal age, 3.5 [range, 2 to 37] days) were collected. Median trough and peak amikacin serum concentrations were 2.1 (range, 1 to 7.1) mg/liter and 39.1 (range, 24.1 to 73.2) mg/liter; the median urea plasma concentration was 30 (8 to 90) mg/dl. The median amikacin concentration in ELF was 6.5 mg/liter, the minimum measured concentration was 1.5 mg/liter, and the maximum (peak) was 23 mg/liter. The highest measured ELF concentration was reached between 6 and 14.5 h after i.v. amikacin administration, and an estimated terminal elimination half-life was 8 to 10 h. The median and highest (peak) ELF amikacin concentrations observed in our study population were, respectively, 6.5 and 23 mg/liter. Despite the frequent use of amikacin in neonatal (pulmonary) infections, this is the first report of amikacin quantification in ELF in newborns.
INTRODUCTION
Aminoglycosides like amikacin are frequently administered in the treatment of suspected or proven Gram-negative infections in neonates, often in combination with penicillins. Due to a concentration-dependent killing combined with a postantibiotic effect, the bactericidal efficacy of amikacin relates to its peak serum concentration. Consequently, therapeutic peak levels at the infection site will define the effectiveness of antibiotic therapy. Renal side effects and ototoxicity relate to the trough serum amikacin concentration, based on the saturation of renal and cochlear cell-binding sites. The pharmacokinetics (PK) of amikacin displays extensive interindividual variability, which makes it difficult to achieve an effective and safe administration in the individual neonate (1).
Based on maturational differences in body composition and renal immaturity in early life, differences in both distribution volume (Vd, in liters/kg) and clearance (CLt, in ml/kg/min) of this hydrophilic drug have been observed. Because of the higher water content in preterm infants, and thus a higher distribution volume for hydrophilic drugs, a relatively higher amikacin dose is necessary in this population (3, 25).
Since pulmonary infections can be a major cause of neonatal morbidity and mortality, antibiotic levels in bronchial secretions and bronchial and alveolar epithelial lining fluid (ELF) are of specific interest. Measuring antibiotic concentrations in the lung is not easy and is usually represented by ELF concentrations. Keeping the anatomy of the blood-bronchial barrier in mind, one can imagine that to reach ELF, the antibiotic must pass through the epithelial lining cells linked by tight junctions (14). Consequently, biochemical characteristics like the degree of protein binding and the lipophilicity and diffusibility of the antibiotic will influence antibiotic concentrations in interstitial fluid and in ELF. In adults, we are aware of studies to assess the blood-alveolar barrier after parenteral administration of amikacin. Dull et al. showed that, after intramuscular (i.m.) administration, the highest amikacin serum concentration correlated significantly with the highest bronchial secretion concentration of that individual and that elimination of amikacin from serum and bronchial secretions occurred at approximately the same rate, with a peak concentration that is blunted in the alveolar compared to the blood compartment (12). Since data in neonates are lacking, the aim of this study is to describe amikacin concentration in the bronchial ELF of newborns.
MATERIALS AND METHODS
Clinical characteristics and drug administration.
From March 2009 to June 2009, bronchoalveolar lavage fluid (BALF) samples were prospectively collected in ventilated neonates to whom amikacin was administered for clinical indications. All the patients were enrolled in the Neonatal Intensive Care Unit (NICU) of the University Hospitals of Leuven, Belgium, following approval by the Medical Ethical Committee of the hospital (B32220084581, S51291) and signed parental consent. The latter was specifically obtained to collect and analyze the BAL fluid samples and to integrate the results with individual clinical characteristics (postmenstrual age [PMA], postnatal age [PNA], birth weight, amikacin therapeutic drug monitoring [TDM], and urea plasma measurements) and treatment (amikacin dosing regimen and duration between intravenous [i.v.] administration and BAL procedure).
Bronchoalveolar lavage aspirates were collected using an endotracheal suctioning method as described earlier in the literature (4). Samples were collected when bronchial suctioning was performed based on perceived clinical need to perform bronchial suctioning.
The amikacin dosing regimen used in the study was implemented in 2002 based on the suggestions of Langhendries et al. (1, 16). In this regimen, dosing depends on PMA as follows: PMA of <28 weeks, 20 mg/kg/42 h; PMA of 28 to 30 weeks, 20 mg/kg/36 h; PMA of 31 to 33 weeks, 18.5 mg/kg/30 h; PMA of 34 to 37 weeks, 17 mg/kg/24 h; and PMA of >37 weeks, 15.5 mg/kg/24 h, with an additional dosing interval increase of 6 h if ibuprofen was coadministered or if neonates had suffered asphyxia or hypoxia. Amikacin (Amukin; 50-mg/ml pediatric vial; Bristol Myers Squibb Belgium) was given as an intravenous infusion over 20 min via syringe driver (SIMS; Graseby, Watford, United Kingdom).
Amikacin assay in serum and BAL fluid. (i) Serum.
Blood samples for TDM were collected by arterial line or venous puncture just before (“trough”) and 1 h after the initiation of administration (“near peak”) of the second dose of amikacin, approximately 40 min after the 20-min intravenous infusion (1, 16). Amikacin serum concentration was determined using a fluorescence polarization immunoassay (TDxFLx; Abbott Laboratories, Diagnostics Division, Abbott Park, IL.) following sample collection, and results are expressed in mg/liter (1). Drug recovery from extraction was 100% (standard deviation [SD], 2.6%) over a tested expected concentration range of 3 to 35 mg/liter. The precision was assessed at 5, 15, and 30 mg/liter. These concentrations yielded, respectively, a within-run coefficient of variation (CV) of 2.1, 1.4, and 1.8%, a between-day CV of 0, 1.5, and 1.7%, and a total CV of 3.2, 2.6, and 2.5%. The minimal quantifiable concentration was 0.8 mg/liter as defined by a CV of <20% (information from Abbott Laboratories). The CV was typically <5%, based on an internal quality assessment covering a concentration range of up to 50 mg/liter (2). For quantification of concentrations between 50 to 200 mg/liter, a manual dilution (4-fold dilution with dilution buffer TDX) is needed.
(ii) BAL fluid.
Measurements of concentrations in deep bronchial secretions were performed on the supernatant after it was processed as described by Santré et al. (23). Following collection, samples were frozen (−20°C) until completion of the study. After thawing, secretions were diluted in an equivalent sterile water volume and then centrifuged after incubation at 37°C for 18 h to provide better viscosity.
Supernatant was collected to determine the amikacin concentration. Quantification was performed by liquid chromatography with pulsed electrochemical detection (LC-PED) based on the method described by Brajanoski et al. for determination of amikacin in cerebrospinal fluid (CSF) (6). A few modifications were done: as column, a reversed-phase C18 Hypersil BDS (100-mm by 2.1-mm; 3-μm particle size) was used, and the flow rate was 0.3 ml/min. The quantification limit for amikacin base was found to be 0.06 mg/liter. Good linearity was obtained for amikacin base in the concentration range from 0.06 mg/liter to 4.0 mg/liter, with a correlation coefficient greater than 0.995. The precision (relative standard deviation [RSD], n = 3) on the peak area of a 1.0-mg/liter amikacin base reference solution was 0.1%. The recovery was found to be 99.1% and 100.9% for BAL fluid spiked with amikacin base at concentrations of 0.2 mg/liter and 2.0 mg/liter, respectively.
Since BAL fluid only in part reflects bronchial epithelial lining concentrations, we used urea concentration in plasma and in the BAL fluid to correct the bronchial epithelial lining amikacin concentrations for the BAL procedure-related dilution. When the urea concentration in plasma and the urea quantity in a lavage sample are known, VELF (ELF volume) can be calculated, using following equation: VELF = (volumeBALF × [ureaBALF]/[ureaplasma], where [ureaBALF] is the urea concentration in BALF and [ureaplasma] is the urea concentration in plasma. As urea is a free low-molecular-weight substance that diffuses readily through the alveolar capillary membrane barrier, it may be assumed that [ureaplasma] = [ureaELF]. To calculate amikacin concentration in ELF, [amikacinELF], the following equation can be used: [amikacinELF] = [amikacinBALF] × ([ureaplasma]/[ureaBALF]) (14, 22).
Urea determination in plasma and BAL fluid. (i) Plasma.
Urea determination in plasma was performed by a modular urea/blood urea nitrogen (BUN) Cobas system (Roche/Hitachi, IN). The measuring range in plasma is 5 to 400 mg/dl urea with 5 mg/dl as a detection limit. Precision was determined using human samples and controls in an internal protocol. Repeatibility (n = 21) showed a CV of 0.8% at a concentration of 198 mg/dl. Intermediate precision yielded a CV of 3.4% at a concentration of 31 mg/dl.
(ii) BAL fluid.
Samples were collected as mentioned above and kept in the freezer until analysis by capillary electrophoresis with capacitively coupled contactless conductivity detection (CE-C4D) was performed. Since coelution of urea and sodium was observed in the CE-C4D system, urease was added to the sample to hydrolyze urea and quantify it as ammonium (24). BAL fluid was centrifuged for 2 min at a speed of 14,500 rpm using a Mini-Spin Plus (Eppendorf, Hamburg, Germany). A volume of 100 μl of supernatant of the BAL fluid sample was pipetted into a 1.5-ml Eppendorf vial. A total of 100 μl of 0.1 mg/ml lithium hydroxide as an internal standard and 100 μl of 0.2 mg/ml urease (type IX from jack beans; 50,000 to 100,000 units/g of solid) were added and completed to 1,000 μl with water. The mixture was incubated for 20 min at 37°C by using a Thermomixer comfort system (Eppendorf, Hamburg, Germany). Next, the solution was centrifuged for 10 min at room temperature at a speed of 14,500 rpm. The supernatant (100 μl) was transferred into a microsample vial and directly injected in the CE-C4D instrument for quantification. The background electrolyte (BGE) for analysis of ammonium contained 30 mM malic acid, adjusted to pH 4.1 by l-arginine, and 10 mM 18 Crown-6 ether. The 10 mM 18 Crown-6 ether was added to the BGE to enhance the separation of ammonium from potassium ions, two cations of similar size, by partial complexation. A voltage of 30 kV was applied on a fused silica capillary with a 75-μm internal diameter and total length of 65 cm (41 cm to C4D). The experiments were performed on a P/ACE MDQ capillary electrophoresis instrument (Beckman Coulter, Inc., Fullerton, CA), coupled with an eDAQ C4D system (eDAQ, Denistone East, Australia). The eDAQ C4D detector was employed at a peak-to-peak amplitude of 100 V, and the frequency was 1,200 kHz. This method provided conditions without interference of matrix components and the recovery was found to be 97.9% and 99.3% for BAL fluid spiked with urea at concentrations of 1.8 mg/liter and 7.2 mg/liter, respectively. For the CE-C4D method, the intraday precision is 0.7% (n = 6), and the interday precision is 1.4% (n = 18, representing 3 days with six analyses/day).
RESULTS
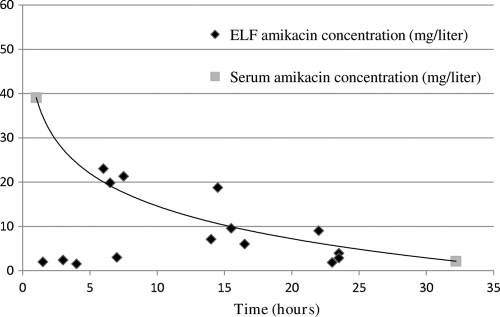
Twenty-two observations were collected in 17 neonates: 19 in male and 3 in female neonates. Median postmenstrual age (PMA) at inclusion was 31.9 (range, 25.1 to 41) weeks, median postnatal age (PNA) was 3.5 (range, 2 to 37) days, and median birth weight was 1,715 (range, 550 to 3,540) grams. All observations were collected in ventilated neonates, with 19 collected during conventional mechanical ventilation and 3 collected during high-frequency oscillation. The median oxygenation index was 3.6 (range, 1.2 to 7.9). Median creatinemia and serum urea were, respectively, 0.6 (range, 0.4 to 1) mg/dl and 26.5 (range, 8 to 90) mg/dl. The median trough and peak amikacin concentrations in serum were 2.1 (range, 1 to 7.1) mg/liter and 39.1 (range, 24.1 to 73.2) mg/liter, respectively, (Fig. 1). Median time of bronchoalveolar lavage was 13.5 (range, 1.5 to 23.5) h after amikacin administration. Median amikacin concentration in ELF was 6.5 mg/liter, the minimum measured concentration was 1.5 mg/liter, and the maximum was 23 mg/liter. The highest measured concentration in our study was reached between 6 and 14.5 h after i.v. amikacin administration, and a subsequent estimated elimination half-life was 8 to 10 h (Fig. 1). Figure 1 illustrates the amikacin ELF concentrations for different time points. During analysis, one observation with an ELF amikacin concentration of 89 mg/liter was excluded since this was an extreme outlier. However, BAL fluid sampling occurred in this patient at 18 h after amikacin administration.
Fig. 1.
Amikacin disposition in ELF and serum. The x axis shows the time (hours) after the start of intravenous amikacin administration; the y axis shows the amikacin concentration (mg/liter). For ELF, each symbol represents a patient. For serum, median peak and trough concentrations of the study population are represented, with an accompanying trendline assuming a one-compartment model with instantaneous input and first-order output.
DISCUSSION
To prevent antibiotic resistance, antibiotics need to be administered based on pharmacokinetics (PK) and pharmacodynamics (PD). Therefore, it is useful to measure the concentrations of antibiotics at infection sites because the distribution of antibiotics may be different among a variety of tissues, in part depending on disease characteristics, maturational changes, or tissue characteristics (14). Amikacin is a commonly administered aminoglycoside to treat neonatal bacterial infections, but data about its concentrations in neonatal bronchial secretions are not yet described. In this study we showed that maximum (peak) and median concentrations of, respectively, 23 and 6 mg/liter can be reached in the epithelial lining fluid after intravenous administration of amikacin in neonates (Fig. 1).
To compare our data with already published evidence, we performed a systematic literature search on aminoglycoside (amikacin [8, 12, 17, 20, 23], tobramycin [5, 7, 9, 17, 19, 20], gentamicin [21, 22], and netilmicin [26]) quantification in BAL fluid after parenteral (i.v. or i.m.) administration of the antibiotic drug. Twelve studies were selected, and they were all conducted in adults (11/12 studies) or children (1/12 studies), not in neonates. Five of 12 studies used amikacin, either in a once-daily (n = 1), twice-daily (n = 1), or three-times-daily dosing regimen (n = 2). One study (n = 1) compared once- versus twice-daily dosage. Even within the adult population, variability in sample material (sputum, bronchial secretions, and BAL fluid) and antibiotic dosage regimen was found. The anatomic site of sample collection is of importance since earlier reports mentioned that aminoglycoside concentrations in whole-lung tissue, sputum, and bronchial secretions approximate, respectively, 50%, 20 to 60%, and 20% of serum levels (22). It is also known that bronchial secretions cannot be used to predict ELF concentrations of aminoglycosides (26). The introduction of BAL as a sampling procedure resulted in more standardized measurements. When the results of the different dosage schedules are examined, higher maximum serum and bronchial amikacin concentrations can be detected with a once-daily i.v. drug administration regimen than with a twice-daily or continuous-infusion regimen (20). Based on the data retrieved, Valcke et al. registered the highest peak aminoglycoside concentration in ELF (14.7 mg/liter) after i.v. administration of 450 mg of netilmicin once daily in adults with pneumonia (26). Our study was performed only in neonates after an extended antibiotic interval and with the BAL procedure to collect samples.
To reach (higher) therapeutic aminoglycoside concentrations in bronchial secretions, one can assume that aerosol-delivered drug administration directly into the bronchial tree is preferable to intravenous administration. No data can be found on neonates. Nebulized amikacin in mechanically ventilated adults with Gram-negative ventilator-associated pneumonia resulted in amikacin ELF concentrations more than 10-fold the MIC90 for microorganisms responsible for nosocomial pneumonia (18). Specific characteristics of nebulized antibiotic administration are the heterogeneous drug disposition within different anatomical parts of the lung and major interindividual variability in achieved drug concentrations. Aerosol-delivered drug administration also results in low systemic drug absorption. This is important in neonates since amikacin is frequently used to treat (suspected) systemic infections.
When we compare amikacin disposition in other deep, extravascular compartments in neonates, only one report can be found concerning amikacin quantification in cerebrospinal fluid (CSF) with a median value of 1.1 mg/liter. Besides serum, EFL, and CSF, further research on amikacin disposition in different body compartments is necessary for optimal treatment of neonatal infections.
Although the endotracheal suctioning method used to collect BAL samples in neonates is part of the routine medical care in ventilated neonates, the quantification of amikacin in ELF is technically more complex. We showed that amikacin concentrations in ELF are relatively low but are detectable. As mentioned above, we used the urea concentration in plasma and in the BAL fluid to correct the ELF amikacin concentrations for the BAL procedure-related dilution. Urea, as an endogenous marker, is small and relatively nonpolar, and its concentration is considered the same in ELF as in serum, implying complete distribution. Significant diffusion of urea across the epithelium during the BAL procedure does not occur (12, 13). In literature, albumin and the secretory component of immunoglobulin A (scIgA) are also presented as denominators for BAL fluid constituents, but they are less appropriate markers (10, 11, 15, 27).
In addition to the already mentioned factors (protein binding, lipophilicity, and diffusibility of the drug) influencing the penetration capacity of antibiotic drugs through the blood-bronchial barrier, it has been postulated that inflammation, infection, and disease severity also influence this passage. Canis et al. investigated the pharmacokinetics of once-daily i.v. amikacin (35 mg/kg/day) administration during the first treatment day in children (mean age, 9.8 years) with cystic fibrosis. Amikacin sputum concentrations ranged from 5.1 to 19.9 mg/liter at 1 h. The highest concentrations were obtained at 2 h (mean, 10.9 ± 7.5 mg/liter) (8). Our study aimed to describe population-specific amikacin kinetics in ELF but was not powered to elaborate on the potential impact of covariates on amikacin bronchial disposition. Based on the reported range in PK serum estimates within the neonatal population, we assume that further exploration of these covariates within the neonatal population will be extremely difficult and of limited add-on value.
We conclude that amikacin concentrations in neonatal epithelial lining fluid after intravenous administration can be quantified. During analysis, urea has to be used to correct concentrations for the BAL procedure dilution. The median and highest (peak) ELF amikacin concentrations found in our study population were, respectively, 6.5 and 23 mg/liter. Despite the frequent use of amikacin in neonatal (pulmonary) infections, this is the first report of amikacin quantification in ELF in newborns.
ACKNOWLEDGMENTS
The clinical research of C. Tayman was supported by The Scientific and Technological Research Council of Turkey (TUBITAK). M. N. El-Attug thanks the Libyan General Peoples Committee (Ministry) for Education and Scientific Research for a scholarship. The clinical research of K. Allegaert is supported by the Fund for Scientific Research, Flanders (FWO Vlaanderen), by a Fundamental Clinical Investigatorship (1800209 N).
Footnotes
Published ahead of print on 27 June 2011.
REFERENCES
- 1. Allegaert K., et al. 2004. Effects of co-administration of ibuprofen-lysine on the pharmacokinetics of amikacin in preterm infants during the first days of life. Biol. Neonate 86:207–211 [DOI] [PubMed] [Google Scholar]
- 2. Allegaert K., et al. 2008. Cerebrospinal fluid compartmental pharmacokinetics of amikacin in neonates. Antimicrob. Agents Chemother. 52:1934–1939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Allegaert K., Scheers I., Cossey V., Anderson B. J. 2008. Covariates of amikacin clearance in neonates: the impact of postnatal age on predictability. Drug Metab. Lett. 2:286–289 [DOI] [PubMed] [Google Scholar]
- 4. Been J. V., et al. 2010. Early alterations of growth factor patterns in bronchoalveolar lavage fluid from preterm infants developing bronchopulmonary dysplasia. Pediatr. Res. 67:83–89 [DOI] [PubMed] [Google Scholar]
- 5. Boselli E., et al. 2007. Reliability of mini-bronchoalveolar lavage for the measurement of epithelial ling fluid concentrations of tobramycin in critically ill patients. Intensive Care Med. 33:1519–1523 [DOI] [PubMed] [Google Scholar]
- 6. Brajanoski G., Hoogmartens J., Allegaert K., Adams E. 2008. Determination of amikacin in cerebrospinal fluid by high-performance liquid chromatography with pulsed electrochemical detection. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 867:149–152 [DOI] [PubMed] [Google Scholar]
- 7. Braude A. C., Hornstein A., Klein M., Vas S., Rebuck A. S. 1983. Pulmonary disposition of tobramycin. Am. Rev. Respir. Dis. 127:563–565 [DOI] [PubMed] [Google Scholar]
- 8. Canis F., et al. 1997. Pharmacokinetics and bronchial diffusion of single daily dose amikacin in cystic fibrosis patients. J. Antimicrob. Chemother. 39:431–433 [DOI] [PubMed] [Google Scholar]
- 9. Carcas J., Garcia-Satue J. L., Zapater P., Frias-Iniesta J. 1999. Tobramycin penetration into epithelial ling fluid of patients with pneumonia. Clin. Pharmacol. Ther. 65:245–250 [DOI] [PubMed] [Google Scholar]
- 10. Dargaville P. A., South M., Vervaart P., McDougall P. N. 1999. Validity of markers of dilution in small volume lung lavage. Am. J. Respir. Crit. Care Med. 160:778–784 [DOI] [PubMed] [Google Scholar]
- 11. de Blic J., et al. 2000. Bronchoalveolar lavage in children. Eur. Respir. J. 15:217–231 [DOI] [PubMed] [Google Scholar]
- 12. Dull W. L., Alexander M. R., Kasik J. E. 1979. Bronchial secretion levels of amikacin. Antimicrob. Agents Chemother. 16:767–771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Grigg J., Amon S., Silverman M. 1992. Fractional processing of sequential bronchoalveolar lavage fluid from intubated babies. Eur. Respir. J. 5:727–732 [PubMed] [Google Scholar]
- 14. Kiem S., Schentag J. J. 2008. Interpretation of antibiotic concentration ratios measured in epithelial lining fluid. Antimicrob. Agents Chemother. 52:24–36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kotecha S. 1999. Bronchoalveolar lavage of newborn infants. Pediatr. Pulmonol. Suppl. 18:122–124 [PubMed] [Google Scholar]
- 16. Langhendries J. P., et al. 1998. Adaptation in neonatology of the once-daily concept of aminoglycoside administration: evaluation of a dosing chart for amikacin in an intensive care unit. Biol. Neonate 74:351–362 [DOI] [PubMed] [Google Scholar]
- 17. Levy J., Baran D., Klastersky J. 1982. Comparative study of the antibacterial activity of amikacin and tobramycin during Pseudomonas pulmonary infection in patients with cystic fibrosis. J. Antimicrob. Chemother. 10:227–234 [DOI] [PubMed] [Google Scholar]
- 18. Luyt C.-E., et al. 2009. Pharmacokinetics and lung delivery of PDDS-aerosolized amikacin (NKTR-061) in intubated and mechanically ventilated patients with nosocomial pneumonia. Crit. Care 13:R200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Mazzei T., Novelli A., De Lalla F., Mini E., Periti P. 1995. Tissue penetration and pulmonary disposition of tobramycin. J. Chemother. 7:363–370 [DOI] [PubMed] [Google Scholar]
- 20. Mombelli G., Coppens L., Thys F. P., Klastersky J. 1981. Anti-Pseudomonas activity in bronchial secretions of patients receiving amikacin or tobramycin as a continuous infusion. Antimicrob. Agents Chemother. 19:72–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Odio W., Van Laer E., Klastersky J. 1975. Concentrations of Gentamicin in bronchial secretions after intramuscular and endotracheal administration. J. Clin. Pharmacol. 15:518–524 [DOI] [PubMed] [Google Scholar]
- 22. Panidis D., Markantonis S. L., Boutzouka E., Karatzas S., Baltopoulos G. 2005. Penetration of gentamicin into the alveolar lining fluid of critically ill patients with ventilator-associated pneumonia. Chest 128:545–552 [DOI] [PubMed] [Google Scholar]
- 23. Santré C., et al. 1995. Amikacin levels in bronchial secretions of 10 pneumonia patients with respiratory support treated once daily versus twice daily. Antimicrob. Agents Chemother. 39:264–267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schuchert-Shi A., Hauser P. C. 2008. Monitoring the enzymatic conversion of urea to ammonium by conventional or microchip capillary electrophoresis with contactless conductivity detection. Anal. Biochem. 376:262–267 [DOI] [PubMed] [Google Scholar]
- 25. Sherwin C. M. T., et al. 2009. Individualised dosing of amikacin in neonates: a pharmacokinetic / pharmacodynamic analysis. Eur. J. Clin. Pharmacol. 65:705–713 [DOI] [PubMed] [Google Scholar]
- 26. Valcke Y. J., Vogelaers D. P., Colardyn F. A., Pauwels A. 1992. Penetration of netilmicin in the lower respiratory tract after once-daily dosing. Chest 101:1028–1032 [DOI] [PubMed] [Google Scholar]
- 27. Watts C. L., Bruce M. C. 1995. Comparison of secretory component for immunoglobulin A with albumin as reference proteins in tracheal aspirate from preterm infants. J. Pediatr. 127:113–122 [DOI] [PubMed] [Google Scholar]