Abstract
Lung function predicts mortality; whether it is associated with functional status in the general population remains unclear. This study examined the association of lung function with multiple measures of functioning in early old age. Data are drawn from the Whitehall II study; data on lung function (forced expiratory volume in 1 s, height FEV1), walking speed (2.44 m), cognitive function (memory and reasoning) and self-reported physical and mental functioning (SF-36) were available on 4,443 individuals, aged 50–74 years. In models adjusted for age, 1 standard deviation (SD) higher height-adjusted FEV1 was associated with greater walking speed (beta = 0.16, 95% CI: 0.13, 0.19), memory (beta = 0.09, 95% CI: 0.06, 0.12), reasoning (beta = 0.16, 95% CI: 0.13, 0.19) and self-reported physical functioning (beta = 0.13, 95% CI: 0.10, 0.16). Socio-demographic measures, health behaviours (smoking, alcohol, physical activity, fruit/vegetable consumption), body mass index (BMI) and chronic conditions explained two-thirds of the association with walking speed and self-assessed physical functioning and over 80% of the association with cognitive function. Our results suggest that lung function is a good ‘summary’ measure of overall functioning in early old age.
Keywords: Ageing, Lung function, Cognitive function, Physical function
Introduction
Lung function is known to increase up to the mid-twenties and then diminish with age (Knudson et al. 1976; Sharma and Goodwin 2006) as static recoil pressure of the lungs decreases leading to lowered forced expiratory flow and vital capacity. There is evidence, from at least two strands of research, to show that poor lung function is associated with poor functional status. One research suggests that lung function predicts poor cognitive outcomes (Cook et al. 1995; Albert et al. 1995; Chyou et al. 1996; Richards et al. 2005; Sachdev et al. 2006) and dementia (Schaub et al. 2000), perhaps due to changes in the central nervous system through processes such as subclinical vascular disease, resulting from inflammation, oxidative stress or cardiovascular risk factors (Liao et al. 1999), or hypoxia-induced changes in neurotransmitter metabolism (Gibson et al. 1981; Grant et al. 1982). Two, chronic obstructive pulmonary disease (COPD) is associated with complex chronic comorbidities (Fabbri and Rabe 2007), even in middle-aged patients (Eisner et al. 2008a; Thorpe et al. 2009). There is also some evidence to support the hypothesis that pulmonary function is associated with physical function in the general population (Thorpe et al. 2009; Myint et al. 2005; Simpson et al. 2005).
Lung function (Knudson et al. 1976; Kauffmann and Frette 1993; Pride 2005; Cohn and Donoso 1963; Janssens et al. 1999), like cognitive (Brayne 2007) and physical function (Rowe and Kahn 1987), declines with age. Whether lung function is a causal risk factor for cognitive and physical functioning is difficult to establish using observational data, as is the case in most of the studies in this domain. Nevertheless, the interrelationship between different aspects of functioning is important as the decline in lung function might be a forerunner of decline in physical and cognitive function. If this is the case, then determinants of lung function and decline might provide important targets of intervention. In this study we examine the relationship of lung function with cognitive and physical in early old age. As the data come from an observational study and the analysis is cross-sectional, we cannot infer causality. However, by examining the association of lung function with both cognitive and physical functioning, we hope to gain better understanding of the importance of lung function for ageing outcomes.
Methods
Data are drawn from Phase 7 (2002–2004) of the Whitehall II study, set up in 1985–1988 on 10,308 (67% men) individuals, aged 35–55 years (Marmot and Brunner 2005). All participants gave consent to participate and the University College London ethics committee (UCLH Committee Alpha, #85/0938) approved this study.
Lung function
Participants were allowed to opt out of spirometry testing if they were coughing up blood or had a pneumothorax, severe angina, heart attack, stroke, pulmonary embolism, aneurysm, a perforated ear drum or hernia, recent surgery (ear, eye, stomach, chest) or blood pressure >180/96 mmHg on the day; in total, 25.5% of participants were excluded.
Lung function was measured using a portable flow spirometer (MicroPlus Spirometer, Micro Medical Ltd, Kent, UK), administered by a trained nurse. We assessed forced vital capacity (FVC) and forced expiratory volume in 1 s (FEV1) based on standardised methods (Miller et al. 2005). The largest FVC and FEV1 values from the three manoeuvres were used in the analysis. As lung volumes are related to body size and standing height is the most important correlating variable, the lung function measure was corrected for height by dividing by the square of the subject’s standing height and multiplying by the square of the sample mean height, 1.77 m in men and 1.63 m in women (Xu et al. 1995). This standard correction procedure ensures that the observed variation in lung function is due to factors other than body size.
Functioning
Walking speed Walking speed was measured over a clearly marked 8-ft (2.44 m) walking course using a standardised protocol (Guralnik et al. 1994). Participants wore either low-heeled close-fitting footwear or walked barefoot with instructions to ‘walk to the other end of the course at your usual walking pace, just as if you were walking down the street to go the shops. Walk all the way past the other end of the tape before you stop’. Three tests were conducted and the fastest walk (2.44 m/min) was used in the analysis.
Cognitive function Two tests were used. The first was a test of short-term verbal memory, assessed with a 20-word free recall test. Participants were presented a list of 20 one- or two-syllable words at 2-s intervals and were then asked to recall in writing as many of the words in any order and had 2 min to do so. The second was a test of reasoning, the Alice Heim 4-I (AH4-I) test, composed of a series of 65 verbal and mathematical reasoning items of increasing difficulty (Heim 1970). It tests inductive reasoning and with only 10 min allocated to the test it is also a measure of processing speed.
Self-assessed health functioning Self-assessed health functioning was assessed using the Short Form 36 (SF-36) General Health Survey Scales (Ware et al. 1993). The SF-36 is a 36-item questionnaire on general health status that can be summarised into physical and mental components scores (PCS and MCS) to assess physical and mental functioning (Ware et al. 1995). Physical functioning declines with age but mental functioning has been shown to improve with age (Chandola et al. 2007).
Covariates
The covariates were age, sex, ethnicity (white or non-white), education (lower primary or lower, lower secondary, high school, first university degree or higher), occupational position, health behaviours, body mass index (BMI) and chronic conditions. Occupational position, classified as high, (administrative grades), intermediate (professional or executive grades) and low (clerical or support grades) position) is a comprehensive marker of socioeconomic circumstances and is related to salary, social status and level of responsibility at work. As of August 1992 the salary range among high-grade employees was £25,330–87,620 and among low-grade employees £7,387–11,917 (Marmot and Brunner 2005).
Smoking status was self-reported (never, ex, or current smoker). Alcohol consumption was assessed as number of alcoholic drinks (‘measures’ of spirits, ‘glasses’ of wine and ‘pints’ of beer) consumed in the last week and converted to number of alcohol units (1 unit=8 g alcohol) consumed per week (Britton et al. 2004). This measure was highly skewed and was log transformed for the analysis. Diet was assessed via a question on the frequency of fruit and vegetable consumption (8-point scale, ranging from ‘seldom or never’ to ‘two or more times a day’), converted to frequency of consumptions per week (Stringhini et al. 2010). Physical activity was assessed using 20 items on frequency and duration of participation in different physical activities (e.g. walking, cycling, sports) that were used to compute hours per week of moderate and vigorous activity. BMI was calculated as weight in kg/height in metres squared. Weight was measured in underwear to the nearest 0.1 kg on Soehnle electronic scales. Height was measured in bare feet to the nearest 1 mm using a stadiometer with the participant standing erect with head in the Frankfort plane.
Chronic conditions included as covariates were coronary heart disease (CHD), diabetes, stroke and respiratory illness. CHD events included non-fatal myocardial infarction (MI) and ‘definite’ angina. MI was determined using data from electrocardiograms (ECGs), cardiac enzymes and physician records following MONICA criteria (Tunstall-Pedoe et al. 1994). Angina was assessed based on the participant’s reports of symptoms (Rose et al. 1977), with corroboration in medical records for nitrate medication or ECG abnormalities. Diabetes assessment was based on fasting glucose (≥7.0 mmol/l) or 2-h postload glucose (≥11.1 mmol/l) or previous use of anti-diabetic medication or reported doctor diagnosed diabetes. Both stroke and respiratory illness (chronic bronchitis, emphysema, asthma, allergy resulting in lung or breathing problems, sinusitis) were assessed using self-reports at Phases 1, 3, 5 and 7.
Statistical analysis
For the descriptive analysis height-corrected FEV1 values were divided into tertiles separately for men and women. Its association with covariates was examined using chi-square or a one way analysis of variance. Subsequently, age and all measures of functioning were standardised to z scores (mean = 0 and standard deviation (SD) = 1) separately in men and women. As all predictors and outcomes in the regression analyses that follow were continuous measures, we used standardised z scores (mean = 0, SD = 1) in the analysis. Standardised regression coefficients represent the change in the outcome variable, expressed as a fraction of the SD, per 1 SD change in the predictor variable. Standardised regression estimates allow comparison of the associations of the predictor with different outcome measures (Newman and Browner 1991) (in our case, first the association of age with different measures of functioning and then that of lung function with measures of physical, cognitive and mental functioning). We first examined the association between an SD greater age and standardised measures of lung, cognitive, physical and mental functioning using linear regression. These analyses were successively adjusted for ethnicity, sex, education, occupational position, health behaviours (tobacco and alcohol consumption, diet, physical activity), BMI and chronic conditions.
The next set of analysis examined the association between FEV1 and functioning using linear regression. We used five blocks of covariates to explain this association using the following formula  (Judd and Kenny 1981). These blocks were age, ethnicity and sex, education and occupation, health behaviours and BMI, and the final block was chronic conditions. As a next step, all these covariates were entered together in order to estimate the attenuation in the association between lung function and measures of cognitive, physical and mental functioning.
(Judd and Kenny 1981). These blocks were age, ethnicity and sex, education and occupation, health behaviours and BMI, and the final block was chronic conditions. As a next step, all these covariates were entered together in order to estimate the attenuation in the association between lung function and measures of cognitive, physical and mental functioning.
Results
A total of 6,483 participants came to the medical examination, and 4,829 of these undertook the lung function tests, our analysis is based on 4,443 (3,111 men and 1,332 women) participants with complete data; those not in the analysis were older (62.1 vs. 60.7 years, p < 0.0001). The age of those included in the analysis ranged from 50.5 to 73.6 years. Table 1 shows all covariates to be associated with FEV1 (all p ≤ 0.03). The analysis in this study used height-corrected FEV1, but replacing it with height-corrected FVC did not much change the results (available from the first author).
Table 1.
Sample characteristics as a function of tertiles of forced expiratory volume (FEV1) in 3,111 men and 1,332 women
| Lowest tertile N = 1,481 69% men | Mid tertile N = 1481 71% men | Highest tertile N = 1481 70% men | p | |
|---|---|---|---|---|
| FEVa1 men (l), M (SD) | 2.55 (0.43) | 3.28 (0.14) | 3.84 (0.26) | <0.001 |
| FEVa1 women (l), M (SD) | 1.60 (0.31) | 2.18 (0.11) | 2.65 (0.22) | <0.001 |
| Age, M (SD) | 63.45 (5.89) | 60.74 (5.64) | 58.02 (4.86) | <0.001 |
| University degree, N (%) | 395 (26.7%) | 438 (29.6%) | 557 (37.6%) | <0.001 |
| High grade, N (%) | 611 (41.3%) | 701 (47.3%) | 782 (52.8%) | <0.001 |
| Non-white, N (%) | 224 (15.1%) | 62 (4.2%) | 14 (0.9%) | <0.001 |
| Current smokers, N (%) | 148 (10.0%) | 116 (7.8%) | 73 (4.9%) | <0.001 |
| Alcohol (units/week), GM(SDL) | 6.55 (3.35) | 7.85 (3.13) | 8.94 (2.87) | <0.001 |
| Consumption of fruit and vegetable (/week), M (SD) | 8.69 (4.27) | 9.29 (4.34) | 9.56 (4.31) | <0.001 |
| Physical activity (hours/week),bM (SD) | 3.61 (3.34) | 3.91 (3.22) | 3.85 (3.41) | 0.03 |
| Body mass index, M (SD) | 26.80 (4.57) | 26.59 (4.19) | 26.15 (4.01) | <0.001 |
| Self-reported respiratory illness, N (%) | 255 (17.2%) | 180 (12.2%) | 149 (10.1%) | <0.001 |
| Self-reported stroke, N (%) | 27 (1.8%) | 26 (1.8%) | 11 (0.7%) | 0.02 |
| Diabetes, N (%) | 198 (13.4%) | 112 (7.6%) | 83 (5.6%) | <0.001 |
| Coronary heart disease, N (%) | 157 (10.6%) | 84 (5.7%) | 48 (3.2%) | <0.001 |
| Memory (range 0–20), M (SD) | 6.33 (2.41) | 6.90 (2.41) | 7.32 (2.34) | <0.001 |
| Reasoning, AH4-I (range 0–65), M (SD) | 41.39 (11.67) | 44.58 (10.51) | 47.01 (9.45) | <0.001 |
| Walking speed (m/min), M (SD) | 1.27 (0.30) | 1.36 (0.29) | 1.39 (0.28) | <0.001 |
| Physical functioning (SF 36), M (SD) | 47.80 (9.04) | 49.93 (7.94) | 50.89 (7.76) | <0.001 |
| Mental functioning (SF 36), M (SD) | 52.84 (8.64) | 52.80 (8.55) | 51.06 (9.15) | <0.001 |
M mean, SD standard deviation, GM geometric mean, SDL standard deviation of logged values
aFEV1 values corrected for height by dividing by own height (Sharma and Goodwin 2006) and multiply by mean height of 1.77 m in men and 1.63 m in women
bModerate and vigorous physical activity
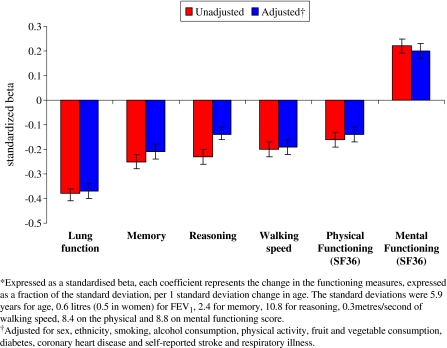
Figure 1 presents the cross-sectional associations, modelled to show effects of an SD increment in age on functioning, standardised to allow comparability. The interaction terms showed no sex differences in the association between age and functioning (all p ≥ 0.09) allowing us to combine men and women in the analysis although lung function was standardised separately in men and women due to differences in lung volume. The standardised beta represents the change in the functioning measures, expressed as a fraction of the SD, per 1 SD change in age. The SDs were 5.9 years for age, 0.6 l (0.5 in women) for FEV1, 2.4 for memory, 10.8 for reasoning, 0.3 m/min of walking speed, 8.4 on the physical and 8.8 on mental functioning score. These SDs allow the reader to convert the standardised results back to regression coefficients. For example, 1 SD increase in age (corresponds to 5.9 years) was associated with lower scores on lung function (beta = −0.38, 95% CI: −0.41, −0.36) (Fig. 1). In men, this corresponds to 0.23 l (0.38 multiplied by the SD, here 0.6 l), and in women 0.19 l lower FEV1.
Fig. 1.
Association* of a standard deviation increase in age with lung, cognitive, physical and mental functioning
One SD increase in age was also associated (Fig. 1) with lower memory (beta = −0.25, 95% CI: −0.28, −0.22), reasoning (beta = −0.23, 95% CI: −0.26, −0.20), walking speed (beta = −0.20, 95% CI: −0.23, −0.17), physical functioning (beta = −0.16, 95% CI: −0.19, −0.13), but higher scores on mental functioning (beta = 0.22, 95% CI: 0.19, 0.25), implying that the older participants had better mental functioning. The association with age was strongest for lung function, adjustment for multiple covariates did not much change this association (beta = −0.37, 95% CI: −0.40, −0.34) (see Fig. 1). The association between age and all functioning measures was robust to adjustment for the covariates (results in tabular form available upon request).
Table 2 presents the association of lung function with walking speed, cognitive function and self-reported physical and mental functioning, again standardised to z scores. The non-significant interaction term between sex and FEV1 (all p ≥ 0.10) allowed men and women to be combined in the analysis. One standard deviation higher FEV1 was associated with greater memory (beta = 0.17 (95% CI: 0.15, 0. 20)), reasoning (beta = 0.23 (95% CI: 0.20, 0. 26)), walking speed (beta = 0.21 (95% CI: 0.18, 0.24)) and physical functioning (beta = 0.17 (95% CI: 0.14, 0.20)), but lower mental functioning (beta = −0.07 (95% CI: −0.10, −0.04)). Age explained the largest part of this association, 47% for memory, 30% for reasoning, 24% for walking speed, 24% for physical functioning and all of it for mental functioning (129%). All covariates taken together explained less of the association of lung function with the measures of physical functioning, walking speed (62%) and self-assessed physical functioning (65%), compared to the measures of cognitive function, memory (82%) and reasoning (91%).
Table 2.
Explaining the association between lung function and physical, mental and cognitive function
| Memory | Reasoning AH4-I | Walking speed | Physical functioning (PCS, SF-36) | Mental functioning (MCS, SF-36) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| betaa (95% CI) | %∆b | betaa (95% CI) | %∆b | betaa (95% CI) | %∆b | betaa (95% CI) | %∆b | betaa (95% CI) | %∆b | |
| Height-corrected FEV1 | 0.17 (0.15, 0.20) | 0.23 (0.20, 0.26) | 0.21 (0.18, 0.24) | 0.17 (0.14, 0.20) | −0.07 (−0.10, −0.04) | |||||
| + Age | 0.09 (0.06, 0.12) | 47% | 0.16 (0.13, 0.19) | 30% | 0.16 (0.13, 0.19) | 24% | 0.13 (0.10, 0.16) | 24% | 0.02 (−0.01, 0.05) | 129% |
| + Ethnicity, sex | 0.15 (0.12, 0.18) | 12% | 0.15 (0.12, 0.18) | 35% | 0.18 (0.15, 0.21) | 14% | 0.16 (0.13, 0.19) | 6% | −0.07 (−0.10, −0.04) | 0% |
| + Education and occupation | 0.14 (0.11, 0.17) | 18% | 0.16 (0.13, 0.18) | 13% | 0.19 (0.16, 0.22) | 10% | 0.16 (0.13, 0.19) | 6% | −0.06 (−0.09, −0.003) | 14% |
| + Health behavioursc and BMI | 0.15 (0.12, 0.18) | 12% | 0.18 (0.15, 0.21) | 30% | 0.17 (0.15, 0.20) | 19% | 0.15 (0.12, 0.18) | 12% | −0.08 (−0.10, −0.05) | −6% |
| + Chronic conditionsd | 0.17 (0.14, 0.20) | 0% | 0.22 (0.19, 0.25) | 4% | 0.20 (0.17, 0.23) | 5% | 0.14 (0.11, 0.17) | 18% | −0.07 (−0.10, −0.04) | 0% |
| + All | 0.03 (−0.01, 0.06) | 82% | 0.02 (−0.01, 0.05) | 91% | 0.08 (0.05, 0.11) | 62% | 0.06 (0.03, 0.09) | 65% | −0.001 (−0.03, 0.03) | 99% |
BMI body mass index, PCS physical component score, MCS mental component score
aFrom standardised values, separately in men and women
b100*(beta unadjusted– beta controlling for the covariate)/beta unadjusted
cSmoking, alcohol consumption, physical activity, fruit and vegetable consumption
dDiabetes, coronary heart disease and self-reported stroke and respiratory illness
Discussion
This study, based on a large non-patient sample of adults aged 50–74 years, shows lung function to be associated with both cognitive and physical functioning, associations only partly explained by age. Our results also show lung function to have a pervasive association with a range of socioeconomic, behavioural and health measures. These results extend previous knowledge on the value of lung function as a good ‘summary’ measure of health and functional status.
Life expectancy is increasing at the rate of 5 or more hours per day in the developed world (Kirkwood 2008). The challenge posed by population ageing is to ensure that the extra years of life will be of good quality and free from high-cost dependency. Thus, functioning—physical, mental and cognitive—is increasingly examined as an outcome in the ageing literature. Positive health trajectories are related to higher quality of life, longer independence and considerably lower medical and social care costs. Chronological age is a good indicator of ageing but it does not capture the variability in exposure and response to environmental insults that could well be better captured in lung function tests, even in populations not composed of COPD patients.
Previous research has shown poor lung function to be associated with poor cognitive outcomes (Cook et al. 1995; Albert et al. 1995; Chyou et al. 1996; Richards et al. 2005; Sachdev et al. 2006) and dementia (Schaub et al. 2000). Three mechanisms have been proposed to explain this association: (1) lung function as a risk factor, (2) poor lung function as a consequence of dementia or impaired cognition and (3) the common cause hypothesis (Schaub et al. 2000). Many studies have examined the first explanation (Cook et al. 1995; Albert et al. 1995; Chyou et al. 1996; Richards et al. 2005; Sachdev et al. 2006; Schaub et al. 2000; Cerhan et al. 1998); inferring causality due to the analytic method (Cook et al. 1995) or longitudinal design of the study (Albert et al. 1995; Chyou et al. 1996). It is clear that the association between lung and cognitive function is not restricted to old age or to sick populations. Studies show this association in mid (Richards et al. 2005) and late midlife (Sachdev et al. 2006). Spirometry tests in children aged 7 years are associated with cognition in analysis adjusted for multiple covariates (Suglia et al. 2008), perhaps reflecting shared neural and endocrine regulatory processes as well as common response to environmental exposures.
There is evidence of the impact of COPD on the non-pulmonary system, even in non-elderly populations were it was shown to be associated with lower extremity functioning, exercise performance, skeletal muscle strength and self-reported limitation in basic physical actions (Eisner et al. 2008b). Research on COPD increasingly views it not simply as a disease of the lungs but as a chronic inflammatory syndrome accompanied by complex chronic comorbidities (Fabbri and Rabe 2007; Vogelmeier and Bals 2007). Our results show lung function to be associated with both an objective (walking speed) and a subjective (self-reported) measure of physical function was robust and all the covariates taken together explain only two thirds of this association. This is a new finding and it is possible that the association stems from the shared pathophysiological processes that lead to declines in pulmonary and physical function.
There are a number of caveats to the results reported here. The Whitehall II study is based on a white collar cohort and thus is not representative of the general population. Furthermore, a quarter of those who came to the medical examination did not do the spirometry. Finally, it is clear that cross-sectional studies are not ideal, they under-estimate the effect of age due to healthy survivor effect (Fowler 1985) and over-estimate it due to cohort effects such as changes in environmental exposures, nutritional factors and childhood infections as younger individuals have been shown to have better lung function compared with 50 years ago (Sharma and Goodwin 2006).
Conclusions and implications
Our study, like most other reports on lung function and functioning, cannot answer questions on causality but provides evidence to suggest that lung function in early old age—because it reflects the impact of environmental insults over the life course—might be a good indicator of ‘ageing’. In our data, adjustment for demographic, socioeconomic, behavioural and health measures fully attenuated the association between lung function and cognition, measured via a test of memory and reasoning, but the association with objective and subjective physical functioning remained robust to these adjustments. The finding that physical activity is associated with a slower decline in pulmonary function offer new ways of thinking about prevention of age related decline in functioning (Pelkonen et al. 2003; Garcia-Aymerich et al. 2007). Further research using longitudinal data is required to understand the mechanisms linking decline in lung function to health functioning in the general population.
Acknowledgments
Funding ASM is supported by a European Young Investigator Award from the European Science Foundation, and MK by the BUPA Foundation and the Academy of Finland. The Whitehall II study has been supported by grants from the British Medical Research Council (MRC); the British Heart Foundation; the British Health and Safety Executive; the British Department of Health; the National Heart, Lung, and Blood Institute, NIH (R01HL036310); the National Institute on Aging, NIH (R01AG013196, R01AG034454).We thank all of the participating civil service departments and their welfare, personnel, and establishment officers; the British Occupational Health and Safety Agency; the British Council of Civil Service Unions; all participating civil servants in the Whitehall II study; and all members of the Whitehall II study team. The Whitehall II Study team comprises research scientists, statisticians, study coordinators, nurses, data managers, administrative assistants and data entry staff, who make the study possible.
References
- Albert MS, Jones K, Savage CR, et al. Predictors of cognitive change in older persons: MacArthur studies of successful aging. Psychol Aging. 1995;10:578–589. doi: 10.1037/0882-7974.10.4.578. [DOI] [PubMed] [Google Scholar]
- Brayne C. The elephant in the room—healthy brains in later life, epidemiology and public health. Nat Rev Neurosci. 2007;8:233–239. doi: 10.1038/nrn2091. [DOI] [PubMed] [Google Scholar]
- Britton A, Singh-Manoux A, Marmot M. Alcohol consumption and cognitive function in the Whitehall II Study. Am J Epidemiol. 2004;160:240–247. doi: 10.1093/aje/kwh206. [DOI] [PubMed] [Google Scholar]
- Cerhan JR, Folsom AR, Mortimer JA, et al. Correlates of cognitive function in middle-aged adults. Atherosclerosis Risk in Communities (ARIC) Study Investigators. Gerontology. 1998;44:95–105. doi: 10.1159/000021991. [DOI] [PubMed] [Google Scholar]
- Chandola T, Ferrie J, Sacker A, Marmot M. Social inequalities in self reported health in early old age: follow-up of prospective cohort study. BMJ. 2007;334:990. doi: 10.1136/bmj.39167.439792.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chyou PH, White LR, Yano K, et al. Pulmonary function measures as predictors and correlates of cognitive functioning in later life. Am J Epidemiol. 1996;143:750–756. doi: 10.1093/oxfordjournals.aje.a008812. [DOI] [PubMed] [Google Scholar]
- Cohn JE, Donoso HD. Mechanical properties of lung in normal men over 60 years old. J Clin Invest. 1963;42:1406–1410. doi: 10.1172/JCI104825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook NR, Albert MS, Berkman LF, Blazer D, Taylor JO, Hennekens CH. Interrelationships of peak expiratory flow rate with physical and cognitive function in the elderly: MacArthur Foundation studies of aging. J Gerontol A Biol Sci Med Sci. 1995;50:M317–M323. doi: 10.1093/gerona/50A.6.M317. [DOI] [PubMed] [Google Scholar]
- Eisner MD, Iribarren C, Yelin EH, et al. Pulmonary function and the risk of functional limitation in chronic obstructive pulmonary disease. Am J Epidemiol. 2008;167:1090–1101. doi: 10.1093/aje/kwn025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner MD, Blanc PD, Yelin EH, et al. COPD as a systemic disease: impact on physical functional limitations. Am J Med. 2008;121:789–796. doi: 10.1016/j.amjmed.2008.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabbri LM, Rabe KF. From COPD to chronic systemic inflammatory syndrome? Lancet. 2007;370:797–799. doi: 10.1016/S0140-6736(07)61383-X. [DOI] [PubMed] [Google Scholar]
- Fowler RW. Ageing and lung function. Age Ageing. 1985;14:209–215. doi: 10.1093/ageing/14.4.209. [DOI] [PubMed] [Google Scholar]
- Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Anto JM. Regular physical activity modifies smoking-related lung function decline and reduces risk of chronic obstructive pulmonary disease: a population-based cohort study. Am J Respir Crit Care Med. 2007;175:458–463. doi: 10.1164/rccm.200607-896OC. [DOI] [PubMed] [Google Scholar]
- Gibson GE, Pulsinelli W, Blass JP, Duffy TE. Brain dysfunction in mild to moderate hypoxia. Am J Med. 1981;70:1247–1254. doi: 10.1016/0002-9343(81)90834-2. [DOI] [PubMed] [Google Scholar]
- Grant I, Heaton RK, McSweeny AJ, Adams KM, Timms RM. Neuropsychologic findings in hypoxemic chronic obstructive pulmonary disease. Arch Intern Med. 1982;142:1470–1476. doi: 10.1001/archinte.142.8.1470. [DOI] [PubMed] [Google Scholar]
- Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85–M94. doi: 10.1093/geronj/49.2.m85. [DOI] [PubMed] [Google Scholar]
- Heim AW. AH 4 group test of general intelligence. Windsor: NFER-Nelson Publishing Company Ltd; 1970. [Google Scholar]
- Janssens JP, Pache JC, Nicod LP. Physiological changes in respiratory function associated with ageing. Eur Respir J. 1999;13:197–205. doi: 10.1183/09031936.99.14614549. [DOI] [PubMed] [Google Scholar]
- Judd CM, Kenny DA. Process analysis: estimating mediation in treatment evaluations. Eval Rev. 1981;5:602–619. doi: 10.1177/0193841X8100500502. [DOI] [Google Scholar]
- Kauffmann F, Frette C. The aging lung: an epidemiological perspective. Respir Med. 1993;87:5–7. doi: 10.1016/S0954-6111(05)80306-3. [DOI] [PubMed] [Google Scholar]
- Kirkwood TB. A systematic look at an old problem. Nature. 2008;451:644–647. doi: 10.1038/451644a. [DOI] [PubMed] [Google Scholar]
- Knudson RJ, Slatin RC, Lebowitz MD, Burrows B. The maximal expiratory flow-volume curve. Normal standards, variability, and effects of age. Am Rev Respir Dis. 1976;113:587–600. doi: 10.1164/arrd.1976.113.5.587. [DOI] [PubMed] [Google Scholar]
- Liao D, Higgins M, Bryan NR, et al. Lower pulmonary function and cerebral subclinical abnormalities detected by MRI: the Atherosclerosis Risk in Communities study. Chest. 1999;116:150–156. doi: 10.1378/chest.116.1.150. [DOI] [PubMed] [Google Scholar]
- Marmot M, Brunner E. Cohort profile: the Whitehall II study. Int J Epidemiol. 2005;34:251–256. doi: 10.1093/ije/dyh372. [DOI] [PubMed] [Google Scholar]
- Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26:319–338. doi: 10.1183/09031936.05.00034805. [DOI] [PubMed] [Google Scholar]
- Myint PK, Luben RN, Surtees PG, et al. Respiratory function and self-reported functional health: EPIC-Norfolk population study. Eur Respir J. 2005;26:494–502. doi: 10.1183/09031936.05.00023605. [DOI] [PubMed] [Google Scholar]
- Newman TB, Browner WS. In defense of standardized regression coefficients. Epidemiology. 1991;2:383–386. doi: 10.1097/00001648-199109000-00014. [DOI] [PubMed] [Google Scholar]
- Pelkonen M, Notkola IL, Lakka T, Tukiainen HO, Kivinen P, Nissinen A. Delaying decline in pulmonary function with physical activity: a 25-year follow-up. Am J Respir Crit Care Med. 2003;168:494–499. doi: 10.1164/rccm.200208-954OC. [DOI] [PubMed] [Google Scholar]
- Pride NB. Ageing and changes in lung mechanics. Eur Respir J. 2005;26:563–565. doi: 10.1183/09031936.05.00079805. [DOI] [PubMed] [Google Scholar]
- Richards M, Strachan D, Hardy R, Kuh D, Wadsworth M. Lung function and cognitive ability in a longitudinal birth cohort study. Psychosom Med. 2005;67:602–608. doi: 10.1097/01.psy.0000170337.51848.68. [DOI] [PubMed] [Google Scholar]
- Rose G, Hamilton PS, Keen H, Reid DD, McCartney P, Jarrett RJ. Myocardial ischaemia, risk factors and death from coronary heart-disease. Lancet. 1977;1:105–109. doi: 10.1016/S0140-6736(77)91701-9. [DOI] [PubMed] [Google Scholar]
- Rowe JW, Kahn RL. Human aging: usual and successful. Science. 1987;237:143–149. doi: 10.1126/science.3299702. [DOI] [PubMed] [Google Scholar]
- Sachdev PS, Anstey KJ, Parslow RA, et al. Pulmonary function, cognitive impairment and brain atrophy in a middle-aged community sample. Dement Geriatr Cogn Disord. 2006;21:300–308. doi: 10.1159/000091438. [DOI] [PubMed] [Google Scholar]
- Schaub RT, Munzberg H, Borchelt M, et al. Ventilatory capacity and risk for dementia. J Gerontol A Biol Sci Med Sci. 2000;55:M677–M683. doi: 10.1093/gerona/55.11.M677. [DOI] [PubMed] [Google Scholar]
- Sharma G, Goodwin J. Effect of aging on respiratory system physiology and immunology. Clin Interv Aging. 2006;1:253–260. doi: 10.2147/ciia.2006.1.3.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson CF, Punjabi NM, Wolfenden L, Shardell M, Shade DM, Fried LP. Relationship between lung function and physical performance in disabled older women. J Gerontol A Biol Sci Med Sci. 2005;60:350–354. doi: 10.1093/gerona/60.3.350. [DOI] [PubMed] [Google Scholar]
- Stringhini S, Sabia S, Shipley M, et al. Association of socioeconomic position with health behaviors and mortality. JAMA. 2010;303:1159–1166. doi: 10.1001/jama.2010.297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suglia SF, Wright RO, Schwartz J, Wright RJ. Association between lung function and cognition among children in a prospective birth cohort study. Psychosom Med. 2008;70:356–362. doi: 10.1097/PSY.0b013e3181656a5a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorpe RJ, Jr, Szanton SL, Whitfield K. Association between lung function and disability in African-Americans. J Epidemiol Community Health. 2009;63:541–545. doi: 10.1136/jech.2008.084418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tunstall-Pedoe H, Kuulasmaa K, Amouyel P, Arveiler D, Rajakangas AM, Pajak A. Myocardial infarction and coronary deaths in the World Health Organization MONICA Project. Registration procedures, event rates, and case-fatality rates in 38 populations from 21 countries in four continents. Circulation. 1994;90:583–612. doi: 10.1161/01.cir.90.1.583. [DOI] [PubMed] [Google Scholar]
- Vogelmeier C, Bals R. Chronic obstructive pulmonary disease and premature aging. Am J Respir Crit Care Med. 2007;175:1217–1218. doi: 10.1164/rccm.200703-513ED. [DOI] [PubMed] [Google Scholar]
- Ware JE, Snow KK, Kosinski M. SF-36 health Survey: manual and interpretation guide. Boston: The Health Institute, New England Medical Centre; 1993. [Google Scholar]
- Ware JE, Kosinski M, Bayliss MS, McHorney CA, Rogers WH, Raczek A. Comparison of methods for the scoring and statistical analysis of SF-36 health profile and summary measures: summary of results from the Medical Outcomes Study. Med Care. 1995;33:AS264–AS279. doi: 10.1097/00005650-199501001-00005. [DOI] [PubMed] [Google Scholar]
- Xu X, Laird N, Dockery DW, Schouten JP, Rijcken B, Weiss ST. Age, period, and cohort effects on pulmonary function in a 24-year longitudinal study. Am J Epidemiol. 1995;141:554–566. doi: 10.1093/oxfordjournals.aje.a117471. [DOI] [PubMed] [Google Scholar]