Abstract
A strategy is described and demonstrated for the formation of reagent anions via electrospray ionization (ESI) for electron transfer dissociation (ETD). To circumvent difficulties associated with formation of high mass-to-charge ratio (m/z) reagent anions, it is desirable to form ETD reagents via means other than those that require reagent molecule vaporization. ESI is a candidate method but anions that are generally generated efficiently by ESI tend to react with multiply protonated polypeptides via proton transfer. The strategy described herein involves the use of a precursor reagent molecule that ionizes efficiently via electrospray ionization and that can subsequently be converted to an ETD reagent via gas-phase dissociation. The approach is demonstrated with arene carboxylic acids that yield strong signals associated with the deprotonated molecule and that subsequently undergo collision-induced dissociation (CID) by loss of CO2. In the present work, triply protonated KGAILKGAILR served as a test substrate for the CID product ions to give rise to ETD. Several precursor molecules were shown to be capable of generating ETD reagents via ESI followed by CID. These included 9-anthracenecarboxylic acid, 2-fluoro-5-iodobenzoic acid, and 2-(fluoranthene-8-carbonyl)-benzoic acid. The latter molecule has the most attractive set of characteristics as a precursor for a relatively high m/z ratio ETD reagent.
Keywords: electron transfer dissociation, nanoelectrospray ionization, ion/ion reaction
INTRODUCTION
Electron transfer dissociation (ETD) of multiply-charged polypeptide cations1 is a gas-phase ion/ion reaction2 phenomenon that provides useful primary structure information. The structural information forthcoming from ETD often complements or supercedes that derived from conventional collision-induced dissociation (CID). ETD appears to be analogous to electron capture dissociation (ECD)3,4 in terms of the structural information provided. ECD is typically performed using Fourier transform ion cyclotron resonance (FTICR) instrumentation or with specialized approaches using electrodynamic ion traps.5,6,7 The simultaneous storage of electrons and cations is not straightforward in electrodynamic ions traps, whereas the mutual storage of cations and anions is much simpler due to the smaller difference in mass-to-charge ratio (m/z) between the oppositely charged entities. In order to effect ETD in an ion trap, the apparatus must be capable of forming and admitting ions of opposite polarity into the reaction volume. In this sense, “specialized” instrumentation is required. However, the motivation for adapting instrumentation for ion/ion reactions goes beyond ETD as there are numerous additional applications for ion/ion reactions.2
To date, ion sources used to form reagent anions for ETD have relied upon the vaporization of a neutral precursor molecule. These approaches include negative ion chemical ionization using a filament based ion source,8 chemical ionization using glow discharge,9,10 and atmospheric pressure chemical ionization.11 Such ion sources provide a high degree of flexibility and are capable of forming anions in high abundances from molecules that can be readily vaporized. However, it is desirable, in general, to expand the range of ionization techniques that can generate anions capable of transferring electrons to polypeptide cations in order to expand options for implementing ETD experiments. For example, the m/z range over which ions can be stored efficiently and simultaneously in an electrodynamic ion trap is limited, which makes it desirable to generate reagent ions of relatively high m/z ratio to react with analyte ions of relatively high m/z ratio.12 The m/z range for mutual ion storage is determined by the ion trap geometry and operating conditions, such as the frequency of the radio-frequency trapping voltage, and therefore depends upon the selection of the key ion trap parameters. Ion sources that rely on the thermal vaporization of precursor molecules are inherently limited in the ion masses that they can generate due to the general inverse relationship between mass and vapor pressure. The use of perfluorocarbons in vapor-based ion sources, such as perfluoro-1,3-dimethyl cyclohexane and perfluoromethyldecalin, which allow for generation of reagent anions in the range of m/z 350–500, has enabled the study of ion/ion reactions of protein ions with m/z ratios in the tens of thousands.13 Perfluorocarbon anions, however, tend to react exclusively via proton transfer with multiply protonated peptides and proteins. The key characteristics of an anion that shows significant electron transfer are relatively low electron affinity and favorable Franck-Condon overlap between the anionic and neutral states of the reagent anion.10 Perfluorocarbon anions are typically characterized by high electron affinities.
Electrospray ionization (ESI) can, in principle, address limitations in reagent ion mass due to the fact that ionization yields for high mass species are largely independent of precursor ion volatility. Dual electrospray ionization sources have already been demonstrated that employ a common vacuum/atmosphere interface and ion path to enable ion/ion reaction studies in hybrid tandem mass spectrometers comprised of linear ion traps.11,14 It is therefore desirable to identify precursor molecules for formation of reagent anions that can serve as ETD reagents using electrospray as the ionization source. However, the types of anions typically formed via ESI are good Brønsted acids. Such species tend to have high electron affinities and therefore react exclusively via proton transfer. Any anionic species that survive the electrospray process, which involves a desolvation process and many gas-phase ion/molecule collisions, are likely to have relatively high electron affinities, which minimizes the likelihood that they can serve as efficient ETD reagents. These factors limit the range of species that might give rise to useful high mass ETD reagents directly via ESI.
The present studies were motivated by the possibility to use molecules that readily form ions directly via ESI that may not react by electron transfer but that can be subsequently converted to ions that can serve as ETD reagents. The general strategy evaluated here is to form anions from carboxylic acids via ESI and to subsequently decarboxylate the anions via ion trap collision-induced dissociation (CID). The gas phase decarboxylation reaction of carboxylate anions was reported by Cooks and coworkers nearly 30 years ago15,16 and has been subsequently exploited by Squires17,18 and others19,20,21 as a way of “synthesizing” carbanions22 in the gas phase for subsequent reactivity studies via ion-molecule reactions. These decarboxylation reactions (represented by equation 1)
| (1) |
occur under conditions of collisional activation in a range of mass spectrometers (e.g. sector instruments,15 ion cyclotron resonance instruments,17 triple quadrupoles18 and ion traps21) for carboxylate anions formed via both chemical ionization and electrospray ionization. Given the low electron affinities of many species represented by R that can be generated via the process of equation 1,23 it seemed worth exploring these decarboxylation reactions as means of generating new electron transfer reagents for use in ion/ion reactions. This report demonstrates that the strategy can be effective and can yield ETD reagents of higher m/z ratio than most thus far reported for vapor-based ionization methods.
EXPERIMENTAL SECTION
Materials
Methanol, acetonitrile, ammonium hydroxide, and glacial acetic acid were purchased from Mallinckrodt (Phillipsburg, NJ). KGAILKGAILR was synthesized by SynPep (Dublin, CA), and was used without further purification. Peptide ions were produced from a ~100 μM solution of equal parts of water and methanol with ~1% acetic acid added for positive nano-electrospray ionization. 9-Anthracenecarboxylic acid, 2-fluoro-5-iodobenzoic acid, 2-(fluoranthene-8-carbonyl)-benzoic acid, and 1,2-methano (60) fullerene-61-carboxylic acid were obtained from Sigma-Aldrich (St. Louis, MO). Negative ions of the former three compounds were produced from a ~1 mM solution of 50/50 (v/v) water/acetonitrile with ~1.5% ammonium hydroxide added for negative nano-electrospray ionization, while negative ions of 1,2-methano(60) fullerene-61-carboxylic acid were produced from a ~0.1 mM solution of 20/80 (v/v) water/methanol with ~1.5% ammonium hydroxide added for negative nano-electrospray ionization.
Apparatus and Procedures
All experiments were performed using a modified Finnigan ion trap mass spectrometer (ITMS) that consists of four ion sources (three electrospray ionization sources and one atmospheric sampling glow discharge ionization sources) as described in detail elsewhere.24 The three electrospray ion sources sequentially inject ions through a DC turning quadrupole, whose potentials are under ITMS software control, into the ion trap endcap electrode. A fourth ion source (an atmosphering sampling glow discharge ion source), also under ITMS software control, injects ions through a hole in the ion trap ring electrode. In this study, only two of the three front-end sources were used as nano-electrospray ionization sources to independently generate peptide cations and arene carboxylate anions. The samples were loaded into nano-electrospray emitters pulled from borosilicate capillaries (1.5 mm o.d., 0.86 mm i.d.) using a P-87 Flaming/Brown micropipet puller (Sutter Instruments, Novato, CA). A stainless steel wire was inserted into the back of the capillary, and a potential of 1.1–2 kV was applied to the wire for ionization.25
The ETD reagent anions were produced from the reagent precursor molecules by accumulation and isolation of the deprotonated species, [RCO2]−, followed by collision-induced dissociation to produce the ETD reagent anions R−. The ion/ion reaction sequence consisted of R− ion isolation, peptide cation accumulation and partial isolation, and mutual storage of oppositely charged ions to induce electron-transfer dissociation. Ion isolation steps were performed using rf ion isolation ramps tuned to eject ions from selected ranges of m/z ratio.26 Peptide cations were only partially isolated because complete isolation of the peptide cations using the rf ramps would have ejected the reagent anions. Mass analysis was achieved by axial modulation.27
RESULTS AND DISCUSSION
The strategy examined here requires three major criteria to be met for the reagent precursor molecule. First, it should yield strong negative ion signal concentrated in the deprotonated molecule. Second, collision-induced dissociation should yield efficient conversion of the [RCO2]− ion to the R− species. And third, the R− species should engage in electron transfer with multiply charged polypeptide cations. The higher the tendency for electron transfer, relative to proton transfer, the more useful the anion is as an ETD reagent. The percent ETD, %ETD, defined as
| (2) |
is a figure of merit that has been used to characterize a reagent anion for the fraction of dissociation products due to electron transfer that result from an ion/ion reaction.10,28 It is the percentage of ion/ion product signal that can be attributed to ETD. The denominator includes the total post ion/ion reaction signal but does not include signal due to any residual unreacted polypeptide ion or signal from b- and y-ions that appear in pre-ion/ion spectrum because this signal is dependent upon reaction time and anion number density and is therefore highly variable. The use of the relationship of equation (2) takes into account only the peptide ions that undergo an electron transfer ion/ion reaction. Singly charged anions derived from relatively rigid highly conjugated systems have been noted to react with multiply protonated polypeptides via electron transfer8,9,10 thereby yielding relatively large values of %ETD (e.g., >10% for triply protonated peptides with up to 20 residues). Species with carboxylic acid functionalities tend to yield strong ESI signals but tend to react with polypeptide cations via proton transfer. Hence, arene carboxylate anions were chosen for evaluation of the strategy based on the process of equation 1.
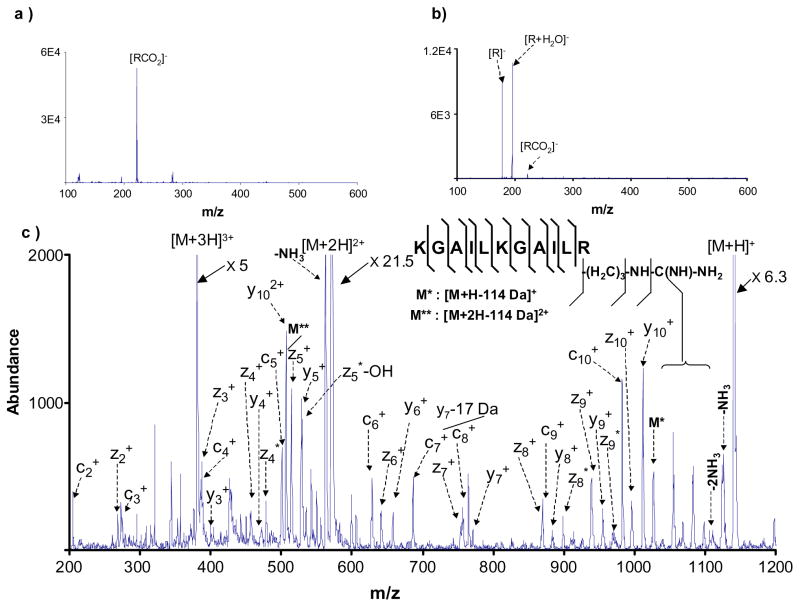
Deprotonated anthracene, formed via negative ion chemical ionization, has already been shown to give rise to ETD.8 Anthracene does not yield strong [R]− signals directly via electrospray. However, ESI of 9-anthracenecarboxylic acid gives rise to a strong signal at m/z 221, which corresponds to the deprotonated molecule, [RCO2]−. This anion reacted with triply protonated KGAILKGAILR via proton transfer (data not shown), which is consistent with the reactivity observed for other carboxylate anions. Ion trap collisional activation of deprotonated 9-anthracenecarboxylic acid leads predominantly to a loss of 44 amu, presumably from loss of CO2, to yield the equivalent of deprotonated anthracene, [R]−. While it is not necessarily justified to conclude that the site(s) of deprotonation associated with the m/z 177 ion formed via CID of the [RCO2]− ion of 9-anthracenecarboxylic acid is the same as that of the deprotonated anthracene ions reported in the negative chemical ionization study, the two ions show similar ion/molecule reactivity. That is, storage of the ion at m/z 177 resulted in the formation of an ion at m/z 195 (labeled as [R+H2O]− in Figure 1), which has been interpreted to be due to an ion/molecule reaction with water present in the vacuum system.8 The m/z 195 ion reacts via proton transfer.8 For this reason, during the mutual ion storage period used to test the m/z 177 anion for electron transfer reactivity, a supplementary frequency tuned to eject m/z 195 ions as they were formed was applied to the end-cap electrodes to minimize proton transfer contributions from this secondary product. The results obtained for 9-anthracenecarboxylic acid are summarized in Figure 1.
Figure 1.
a) Negative ion nano-electrospray ionization mass spectrum of 9-anthracenecarboxylic acid. b) Ion trap collision-induced dissociation of deprotonated 9- anthracenecarboxylic acid. The ion labeled [R+H2O]− is an ion molecule reaction product formed during the collisional activation period and prior to ejection of the reagent anion, [R]−. c) ETD spectrum derived from the reaction of the reagent anion, [R]−, of 9-anthracenecarboxylic acid with triply protonated KGAILKGAILR.
The spectrum resulting from the ETD reaction between triply protonated KGAILKGAILR and anthracene anion (Figure 1c) is similar to ETD spectra collected on this instrument and other instruments using more common ETD reagents10,11. Figure 1c shows a series of c-ions (c2- c10) and z-ions (z2-z10) except for c1 and z1 ions, which may not have been observed because the low mass cut-off of the ion trap instrument was too high to store the low m/z c1 and z1 ions. Several (z+32 amu) ions (z-adducts with molecular oxygen O2), denoted as zn* as well as (zn*-HO·) ions (z-adducts–HO·) were also present in the post ion/ion reaction spectrum.29 The observed neutral losses are predominantly associated with neutral losses from the arginine side chain and are shown in the figure by the formula –(H2C)3-NH-C(NH)-NH2. The peaks labeled M* and M** are singly and doubly charged ions that represent a neutral loss of 114 amu from the parent ion. A neutral loss of 114 amu corresponds to a loss of 42 amu (NH-C-NH) from the arginine side chain plus the loss of 72 amu (-(CH2)4-NH2) from the lysine side chain. In addition, the M** ion appeared in the pre ion/ion spectrum and was subsequently charge reduced during the ion/ion reaction to M*. Along with the c- and z-ions associated with the ETD reaction, several y-ions appear in the ETD spectrum. These y-ions are also present in the pre-ion/ion spectrum predominantly as doubly charged y-ions, which are charge reduced to singly charged y-ions in the course of the ion/ion reaction. None of the y-ions or any other ions that appeared in the pre ion/ion spectrum were used in the %ETD calculations.
The experiment summarized in Figure 1 clearly shows that the overall strategy for forming an ETD reagent anion via electrospray ionization and subsequent CID can be effective. In this particular experiment, a %ETD of 17% was obtained. Deprotonated anthracene, formed either directly via gas-phase ionization or indirectly via electrospray and CID, however, suffers from the complication of a competing ion/molecule reaction and, at m/z 177, does not represent a particularly high mass reagent.
Several potential reagents of mass higher than that of 9-anthracenecarboxylic acid were also evaluated, and some gave rise to anions that show a significant degree of electron transfer reactivity. These included, for example, 2-fluoro-5-iodobenzoic acid (M.W. = 266.01 Da) and 2-(fluoranthene-8-carbonyl)-benzoic acid (M.W. = 350.37 Da). While the former compound gave rise to an abundant reagent anion, [R]−, of m/z 221 upon ion trap CID that reacted via electron transfer (data not shown), roughly 35% of the CID product ion signal was partitioned into I−, which reacts with protonated polypeptides either via proton transfer or attachment.30 A reagent anion of m/z 221 is more desirable for ETD of high m/z ratio cations than the analogous reagent anion of m/z 177 from 9-anthracenecarboxylic acid but the competing CID channel leads to a complication analogous to the ion/molecule reactivity noted above.
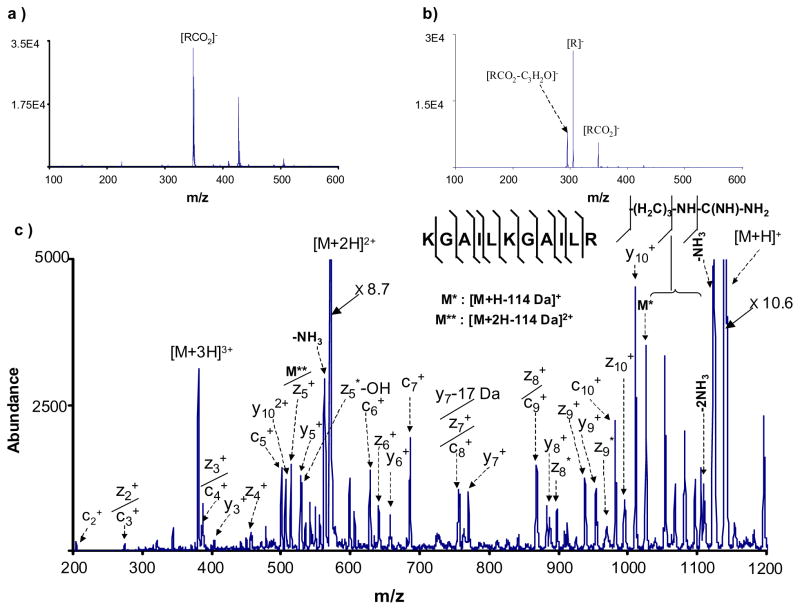
The 2-(fluoranthene-8-carbonyl)-benzoic acid reagent provided a more desirable set of characteristics as a precursor for an ETD reagent than either of the other molecules described above. In this case, electrospray readily yielded a strong anion signal corresponding to the deprotonated molecule, [RCO2]−, and loss of CO2 was clearly the dominant CID process. A competing product ion channel involved a loss of 54 amu from the deprotonated molecule, which is consistent with the loss of C3H2O, and labeled as [RCO2-C3H2O]− in Figure 2b. This product was not studied further. The most abundant product ion, the [M-H-CO2]− anion (m/z 305), showed no ion/molecule reactivity and reacted with the test peptide via electron transfer (%ETD = 26%). The results for this compound are summarized in Figure 2. The ETD reaction spectrum (Figure 2c) shows the same ETD product ions as shown in Figure 1c, including the series of c-ions (c2-c10) and z-ions (z2-z10). This species, therefore, constitutes a relatively high m/z ratio reagent anion for ETD that can be formed indirectly via electrospray ionization.
Figure 2.
a) Negative ion nano-electrospray ionization mass spectrum of 2-(fluoranthene-8-carbonyl)-benzoic acid. Ions of m/z greater than the [RCO2]− arise from sample impurities. b) Ion trap collision-induced dissociation of deprotonated 2-(fluoranthene-8-carbonyl)-benzoic acid. c) ETD spectrum derived from the reaction of the reagent anion [R]− of 2-(fluoranthene-8-carbonyl)-benzoic acid with triply protonated KGAILKGAILR.
In an attempt to identify an ETD reagent of significantly higher m/z ratio, 1,2-methano(60) fullerene-61-carboxylic acid (M.W.= 778.68 Da) was evaluated. Previous negative ESI-FTICR studies of 1,6-methano(60) fullerene-61,61-dicarboxylic acid showed formation of the [C60C(COOH)COO]− ion and losses of one and two molecules of CO2 upon CID of the deprotonated molecule.31 We found that it was possible to form the [C60CH]− ion directly via electrospray by use of relatively high voltages in the vacuum/atmosphere interface used in these studies for anion injection. The resulting anion, however, reacted predominantly via proton transfer, presumably due to the relatively high electron affinity expected for fullerene related anions. For example, an electron affinity for the [C60CH]− ion formed via CO2 loss was calculated to be 3.84 eV.30 Hence, while the precursor molecule has several desirable characteristics associated with the formation of ETD reagents, it fails probably due to the very high electron affinity of the CID product. Interestingly, small signals due to ETD products were noted in the ion/ion reaction experiment with triply protonated KGAILKGAILR when anions over a wide range of m/z were allowed to react (i.e., in the absence of any anion isolation step). The negative ion spectrum showed only one dominant anion, corresponding to [C60CH]−, but there were low levels of chemical noise across much of the spectrum. Apparently, at least some of the low signal level anions that comprised the chemical noise gave rise to ETD. However, due to the complexity of the background and the low signal levels for the background species, no attempt was made to identify which species might be responsible for ETD.
CONCLUSIONS
An effective strategy for the generation of ETD reagents for dissociation of multiply protonated polypeptides using electrospray ionization has been demonstrated. It is based on the use of suitable reagent species with a carboxylate functionality that facilitates ionization in the negative electrospray mode. Subsequent collisional activation of the resulting anion that leads to efficient CO2 loss can give rise to products that react via electron transfer. The types of species that are expected to be useful as precursors for electron transfer reagents are those that, after loss of CO2, have relatively low electron affinities and favorable geometries for vertical electron detachment. Three precursor reagent molecules were identified herein as being able to lead to ETD reagents via electrospray ionization and CID. The strategy, therefore, shows a degree of generality. Of the three described, 2-(fluoranthene-8-carbonyl)-benzoic acid has the most attractive set of characteristics for an ETD reagent precursor insofar as the reagent anion, [R]−, has the highest m/z ratio, provides moderately high %ETD, and shows no competing CID or ion/molecule reaction channels.
Acknowledgments
This research was sponsored by the National Institutes of Health, Institute of General Medical Sciences under Grant GM 45372. RAJO thanks the Faculty of Science at the University of Melbourne for teaching release and financial support for a sabbatical through the Special Studies Program (Long).
References
- 1.Syka JEP, Coon JJ, Schroeder MJ, Shabanowitz J, Hunt DF. Proc Natl Acad Sci USA. 2004;101:9528–9533. doi: 10.1073/pnas.0402700101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pitteri SJ, McLuckey SA. Mass Spectrom Rev. 2005;24:931–958. doi: 10.1002/mas.20048. [DOI] [PubMed] [Google Scholar]
- 3.Zubarev RA, Kelleher NL, McLafferty FW. J Am Chem Soc. 1998;120:3265–3266. [Google Scholar]
- 4.Zubarev RA. Mass Spectrom Rev. 2003;22:57–77. doi: 10.1002/mas.10042. [DOI] [PubMed] [Google Scholar]
- 5.Baba T, Hashimoto Y, Hasegawa H, Hirabayashi A, Waki I. Anal Chem. 2004;76:4263–4266. doi: 10.1021/ac049309h. [DOI] [PubMed] [Google Scholar]
- 6.Silivra OA, Kjeldsen F, Ivonin IA, Zubarev RA. J Am Soc Mass Spectrom. 2006;16:22–27. doi: 10.1016/j.jasms.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 7.Ding L, Brancia FL. Anal Chem. 2006;78:1995–2000. doi: 10.1021/ac0519007. [DOI] [PubMed] [Google Scholar]
- 8.Coon JJ, Syka JEP, Schwartz JC, Shabanowitz J, Hunt DF. Int J Mass Spectrom. 2004;236:33–42. [Google Scholar]
- 9.Pitteri SJ, Chrisman PA, Hogan JM, McLuckey SA. Anal Chem. 2005;77:1831–1839. doi: 10.1021/ac0483872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gunawardena HP, He M, Chrisman PA, Pitteri SJ, Hogan JM, Hodges BDM, McLuckey SA. J Am Chem Soc. 2005;127:12627–12639. doi: 10.1021/ja0526057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liang X, Xia Y, McLuckey SA. Anal Chem. 2006;78:3208–3212. doi: 10.1021/ac052288m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McLuckey SA, Stephenson JL., Jr Mass Spectrom Rev. 1998;17:369–407. doi: 10.1002/(SICI)1098-2787(1998)17:6<369::AID-MAS1>3.0.CO;2-J. [DOI] [PubMed] [Google Scholar]
- 13.Reid GE, Wells JM, Badman ER, McLuckey SA. Int J Mass Spectrom. 2003;222:243–258. [Google Scholar]
- 14.Xia Y, Chrisman PA, Erickson DE, Liu J, Liang X, Londry FA, Yang MJ, McLuckey SA. Anal Chem. 2006;78:4146–4154. doi: 10.1021/ac0606296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kondrat RW, McClusky GA, Cooks RG. Anal Chem. 1978;50:1222–1223. [Google Scholar]
- 16.McClusky GA, Kondrat RW, Cooks RG. J Am Chem Soc. 1978;100:6045–6051. [Google Scholar]
- 17.Froelicher SW, Freiser BS, Squires RR. J Am Chem Soc. 1986;108:2853–2862. [Google Scholar]
- 18.Squires RR. Acc Chem Res. 1992;25:461–467. [Google Scholar]
- 19.Bienkowski T, Danikiewicz W. Rapid Commun Mass Spectrom. 2003;17:697–705. doi: 10.1002/rcm.969. [DOI] [PubMed] [Google Scholar]
- 20.Kumar MK, Sateesh B, Prabhakar S, Sastry GN, Vairamani M. Rapid Commun Mass Spectrom. 2006;20:987–993. doi: 10.1002/rcm.2400. [DOI] [PubMed] [Google Scholar]
- 21.Gronert S, Feng WY, Chew F, Wu W. Int J Mass Spectrom. 2000;195/196:251–258. [Google Scholar]
- 22.Bowie JH. Mass Spectrom Rev. 1990;9:349–379. [Google Scholar]
- 23.Rienstra-Kiracofe JC, Tschumper GS, Schaefer HF, Nandi S, Ellison GB. Chem Rev. 2002;102:231–282. doi: 10.1021/cr990044u. [DOI] [PubMed] [Google Scholar]
- 24.Badman ER, Chrisman PA, McLuckey SA. Anal Chem. 2002;74:6237–6243. doi: 10.1021/ac026020w. [DOI] [PubMed] [Google Scholar]
- 25.Van Berkel GJ, Asano KG, Schnier PD. J Am Soc Mass Spectrom. 2001;12:853–862. doi: 10.1016/S1044-0305(01)00264-1. [DOI] [PubMed] [Google Scholar]
- 26.McLuckey SA, Goeringer DE, Glish GL. J Am Soc Mass Spectrom. 1991;2:11–21. doi: 10.1016/1044-0305(91)80056-D. [DOI] [PubMed] [Google Scholar]
- 27.Kaiser RE, Cooks RG, Stafford GC, Syka JEP, Hemberger PH. Int J Mass Spectrom Ion Processes. 1991;106:79–115. [Google Scholar]
- 28.Pitteri SJ, Chrisman PA, McLuckey SA. Anal Chem. 2005;77:5662–5669. doi: 10.1021/ac050666h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xia Y, Chrisman PA, Pitteri SJ, Erickson DE, McLuckey SA. J Am Chem Soc. 2006;128:11792–11798. doi: 10.1021/ja063248i. [DOI] [PubMed] [Google Scholar]
- 30.Stephenson JL, Jr, McLuckey SA. J Am Chem Soc. 1997;119:1688–1696. [Google Scholar]
- 31.Zhang R, Fisher KJ, Smith DR, Willett GD, Peel JB, Gan L, Shi Y, Gao Z. Eur J Mass Spectrom. 2000;6:161–168. [Google Scholar]