Abstract
Oxidative stress in the bronchopulmonary airways can occur through a variety of inflammatory mechanisms and also following the inhalation of environmental pollutants. Oxidative stress causes cellular dysfunction and thus mammals (including humans) have developed mechanisms for detecting oxidative stress, such that defensive behavior and defensive biological mechanisms can be induced to lessen its potential damage. Vagal sensory nerves innervating the airways play a critical role in the detection of the microenvironment in the airways. Oxidative stress and associated compounds activate unmyelinated bronchopulmonary C-fibers, initiating action potentials in these nerves that conduct centrally to evoke unpleasant sensations (e.g. urge to cough, dyspnea, chest-tightness) and to stimulate/modulate reflexes (e.g. cough, bronchoconstriction, respiratory rate, inspiratory drive). This review will summarize the published evidence regarding the mechanisms by which oxidative stress, reactive oxygen species, environmental pollutants and lipid products of peroxidation activate bronchopulmonary C-fibers. Evidence suggests a key role for transient receptor potential ankyrin 1 (TRPA1), although transient receptor potential vanilloid 1 (TRPV1) and purinergic P2X channels may also play a role. Knowledge of these pathways greatly aids our understanding of the role of oxidative stress in health and disease and represents novel therapeutic targets for diseases of the airways.
1. Introduction
Oxidation of phospholipids, proteins and nucleic acids by reactive oxygen species (ROS) causes cellular dysfunction and thus oxidative stress can be considered a threat to the organism (Karihtala and Soini, 2007). Multicellular organisms have developed defensive systems that can specifically detect noxious stimuli and enact defensive cellular responses and behavior. Sensory (afferent) nerves are the initial part of this defense mechanism. Afferent nerves project nerve terminals into peripheral tissue where, through the activation of specific nerve terminal proteins (ion channels), they detect specific stimuli and relay such information to the central nervous system (CNS) in the form of action potentials. In the case of the large airways and lungs, the afferent innervation comes largely from the vagus nerve, and their activation can cause cough, dyspnea, changes in breathing control, decreased inspiratory capacity, bronchospasm and hypersecretion. In this review, we will summarize the mechanisms by which oxidative stress is detected by vagal airway sensory nerves.
The phrase ‘oxidative stress’ defines a state in which there is a misbalance in the redox environment. This can be induced in two ways: by increasing the levels of pro-oxidants (e.g. ROS) or by decreasing the levels of anti-oxidant defenses (e.g. cytosolic glutathione) (Karihtala and Soini, 2007). In general terms electrophilic pro-oxidants accept electrons from nucleophilic biological compounds (e.g. molecules with -OH or -SH groups). Examples of ROS include superoxide radical (O2−), hydroxyl radical, nitric oxide radical, hydrogen peroxide (H2O2) and ozone (O3). The redox potential of ROS varies between chemical species and in biological tissue this tends to negatively correlate with half-life: e.g. the hydroxyl radical is considered the most reactive radical in biology and has a half-life of approximately 1ns, whereas H2O2 is far less reactive and (unless broken down by catalase) has a half-life that is measured in hours/days. In addition to their direct oxidant properties, ROS can cause the production (via a self-propagating lipid peroxidation reaction) of a host of compounds with electrophilic reactive alpha-beta unsaturated carbonyl groups that have many of the same chemical properties (Blair, 2006).
The airways are subjected to oxidative stress derived from two separate sources: inhalation of exogenous irritants and endogenous inflammation. Oxidative stress can be induced directly by inhaled irritants (e.g. electrophilic aldehydes in cigarette smoke, diisocyanates from the manufacture of polyurathane), downstream of ROS production (metal ions and quinones in particulate matter induces O2− production), or downstream of ROS propagation (e.g. O3 induces O2− and H2O2 production and lipid peroxidation). The initial step in the majority of endogenous ROS production is the synthesis of O2−, largely through the actions of NADPH oxidase, xanthine oxidase and through inherent (although mild in basal conditions) inefficiencies in mitochondrial electron transfer. Inflammation in the airways causes substantial ROS production and this is largely due to NADPH oxidase activity in activated granulocytes, although evidence suggests mitochondria in airway cells also contribute subsequent to their inflammation-induced dysfunction (Mabalirajan et al., 2010; Mabalirajan et al., 2009; Mabalirajan et al., 2008). ROS and biomarkers for ROS production (e.g. H2O2, isoprostanes, nitrotyrosine residues) are found in lung samples from individuals with asthma and chronic obstructive pulmonary disease (COPD) (Kharitonov and Barnes, 2004; Kirkham and Rahman, 2006; Nadeem et al., 2008) or individuals following exposure to inhalation irritants such as O3, chlorine, cigarette smoke, particulate matter, diisocyanates, isothiocyanates; and these findings concur with animal models of airway disease (Brown and Burkert, 2002; Comandini et al., 2009; Hazbun et al., 1993; Lin and Thomas, 2010; Rouse et al., 2008; White and Martin, 2010). Precisely how stress contributes to the more complex disease states of inflammation, asthma and COPD is unresolved. Through the oxidation of lipid, protein and nucleic acids, exogenously applied ROS induce a variety of inflammatory pathways (Gloire and Piette, 2009; Valko et al., 2007) which causes in vivo epithelial damage, remodeling, bronchospasm and airway hyperreactivity in various models (Comhair and Erzurum, 2010; Hollingsworth et al., 2007; Kinnula et al., 1995; Li et al., 2008b; North et al., 2011; Uysal and Schapira, 2003). The effect of antioxidants in reducing airway inflammation has been less than impressive, particularly in humans (Nadeem et al., 2008). Further evidence for the contribution of oxidative stress to airway disease can be found in functional polymorphisms of antioxidant defense systems. Polymorphisms that reduce the reserve capacity of glutathione correlate with severity of airway disease and exacerbations (Islam et al., 2009; Polimanti et al., 2010; Reddy et al., 2010; Schroer et al., 2009; Shaheen et al., 2010).
2. Lower Airways innervation
Sensory nerves throughout the mammalian body possess similar cellular structure: a cell body (soma) that contains the nucleus and the vast majority of gene transcription and translation machinery/organelles, a peripheral axonal projection from the soma to its target tissue that ends in the peripheral terminal, and a central axonal projection from the soma to synapses with second order neurons within the CNS. Bronchopulmonary vagal sensory nerves are no exception to this rule: with cell bodies residing in the vagal ganglia; peripheral projections to the larynx, trachea, main bronchi or lungs; and central projections to second order neurons in the brain stem, in particular neurons in the nucleus tractus solitarius (nTS) (Carr and Undem, 2003).
Vagal sensory neurons are not homogenous and vary with respect to myelination, conduction velocity, ion channel expression, neurotransmitter expression, peripheral targets and central targets – and considerable effort has been made to elucidate the role of various vagal sensory nerve subtypes in health and disease (Carr and Undem, 2003; Coleridge and Coleridge, 1984; Lee, 2009; Taylor-Clark and Undem, 2006). Axonal myelination determines the conduction velocity of action potentials along the nerve, and this correlates with the expression of certain ion channels that determine nerve functionality. As with all sensory systems, airway unmyelinated (C fibers, conduction velocity of 0.3–1.2 m/s) and partially myelinated (Aδ fibers, 2–8m/s) nerves conduct action potentials initiated by a wide range of noxious stimuli, whereas highly myelinated rapidly conducting fibers (Aβ fibers, 12–50m/s) are exquisitely mechano-sensitive but are not directly activated by noxious stimuli. The ability to discern noxious stimuli comes from the selective expression of ion channels on nociceptive sensory nerves, for example inhalation of capsaicin (the pungent ingredient is chili peppers) activates nociceptive airway sensory nerves via the direct activation of Transient Receptor Potential Vanilloid 1 (TRPV1) (Kollarik and Undem, 2004). Aβ fibers do not express TRPV1 and so are insensitive to capsaicin treatment.
Regardless of the stimuli, sensory nerve activation at the peripheral terminal occurs following the opening of specific ion channels, which causes ion fluxes at the nerve membrane leading to a graded depolarization of the membrane referred to as a “generator potential”. If the generator potential is great enough to surpass the voltage threshold of voltage-gated sodium channels this will cause the initiation of action potentials that then travel in an all-or-nothing fashion along the nerve fiber. The gating of peripheral terminal ion channels that is required for afferent nerve activation can occur through ionotropic and metabotropic mechanisms (Taylor-Clark and Undem, 2006). The inotropic mechanism refers to an ion channel that has a self-contained activation/binding site for a specific stimulus (e.g. TRPV1 is directly gated by capsaicin). The metabotropic mechanism refers to the gating of certain ion channels downstream of second messenger systems (metabotropic), typically following the activation of G-protein-coupled receptors (GPCRs): for example bradykinin, via the Gq-coupled B2 receptor, can activate TRPV1 channels, inducing nerve depolarization and action potential discharge (Carr et al., 2003; Chuang et al., 2001; Shin et al., 2002).
In guinea pigs and larger mammals (including humans) vagal sensory nerve cell bodies reside in one of two anatomically distinct ganglia: the nodose and the jugular (supranodose) ganglia. The embryological source of the neurons in these two ganglia is different (the epibrachial placodes and the neural crest, respectively) and consequently the neurons express different growth factor receptors (Baker and Bronner-Fraser, 2000). This differential development of jugular and nodose neurons is thought to underlie differences in nerve function and protein expression observed between these two sets of nerves (Kwong et al., 2008; Undem et al., 2004). The gross anatomy of mouse vagal nerves often reveal a single sensory ganglion. Despite this, recent evidence indicates that the single ganglion is actually a fusion of neurons derived from the epibrachial placode and the neural crest; in effect the mouse vagal ganglion is a nodose/jugular fused ganglion, and there are substantial differences between mouse nodose and mouse jugular neurons (as there is in larger mammals) (Nassenstein et al., 2010). For example, airway nodose nerves express purinergic P2X2/3 ion channels and are activated by ATP and P2X2/3 selective agonists. Jugular neurons do not express P2X2/3 channels and ATP fails to cause action potential discharge from airway jugular neurons. Conversely, most jugular C-fibers express neuropeptides (e.g. substance P) whereas very few nodose neurons do. Neuropeptides are released from activated nociceptor nerve terminals and, peripherally, these substances can contribute to bronchoconstriction, vasodilation and leukocyte infiltration (neurogenic inflammation). The release of neuropeptides at central synapses is thought to augment synaptic excitability in a process called ‘central sensitization’ (Mazzone and Canning, 2002a).
The outcomes of afferent nerve stimulation depend on the afferent subtype in question. In general terms, non-nociceptive fibers relay information to the CNS on mechanical changes in the airways in order to fine-tune eupnic breathing, whereas nociceptor activation induces defensive reflexes and behaviors (Carr and Undem, 2003; Coleridge and Coleridge, 1984; Fuller et al., 1985; Lee, 2009). In particular bronchopulmonary C-fiber activation can evoke peripheral neurogenic inflammation, reflex-bronchoconstriction, reflex-hypersecretion, decreased inspiratory capacity and cough and can cause sensations of chest-tightness, urge to cough and dyspnea. With the exception of the neurogenic inflammation, all these signs and symptoms are dependent on excitatory afferent input into the CNS.
Sensory nociceptors are polymodal sensors of the peripheral nerve terminal environment and due to the expression of specific receptors and ion channels are able to directly discriminate changes in temperature, pH and osmolarity; mechanical force (either stretch or punctate); and the presence of irritants and inflammatory mediators (Taylor-Clark and Undem, 2006). However, it should be noted that some stimuli can also activate airway afferents indirectly following changes in mechanical forces within the airways, such as bronchoconstriction and edema (Roberts et al., 1986). For example, some non-nociceptive bronchopulmonary A fibers (which are very sensitive to stretch) are activated by histamine infusion into the airways, in a manner that is dependent on histamine-induced bronchial smooth muscle contraction (Bergren, 1997). Airway inflammation and inhalation of noxious pro-oxidant irritants can cause epithelial damage, edema and bronchoconstriction, and thus has the potential to indirectly modulate mechano-sensitive airway afferent firing. However, in the interests of brevity we have decided to limit the scope of this review to the mechanisms by which pro-oxidants directly modulate airway afferent activity.
3. Activation of airway afferents by oxidative stress
Inhalation of pro-oxidants acutely evoke sensations and changes in lung function, consistent with the activation of airway afferent nerves. Inhalation of H2O2 causes a decrease in respiratory rate in rats and mice (Bessac et al., 2008; Ruan et al., 2003). Inhalation of O3 causes cough, dyspnea, decreased forced expiratory volume in 1 second (FEV1) and decreased inspiratory capacity in humans (Hazucha et al., 1989; Kerr et al., 1975; McDonnell et al., 1999); and O3 modulates respiratory rates and airway afferent excitability in other mammals including dogs and rodents (Coleridge et al., 1993; Ho and Lee, 1998; Lee et al., 1979; Schelegle et al., 1993). Inhalation of diisocyanates causes cough, dyspnea and chest tightness in man (Brugsch and Elkins, 1963; Butcher et al., 1976) and decreased respiratory rate in mice (Taylor-Clark et al., 2009). Inhalation of acrolein causes cough, airway irritation, neurogenic inflammation and dyspnea with decreased respiratory rate in humans and other mammals (Faroon et al., 2008; Morris et al., 1999; Morris et al., 2003). Inhalation of chlorine causes irritation and increased airway resistance and evokes cough, wheezing and dyspnea in humans and decreased respiratory rate in mice (Bessac et al., 2008; Morris et al., 2005; Shusterman et al., 1998; White and Martin, 2010). Cough, dyspnea and irritation are sensory nerve mediated phenomenon. Changes in lung function (e.g. decrease in FEV1) can occur through sensory nerve-mediated pathways (e.g. reflex increase in the release of acetylcholine from parasympathetic nerves in the airway), but this is rarely confirmed in clinical reports of pro-oxidant inhalation.
The acute symptoms of pro-oxidant inhalation suggest the involvement of nociceptive airway sensory nerves: airway nociceptors are selectively activated by the TRPV1 agonist capsaicin, and capsaicin inhalation causes increased bronchial resistance (which is sensitive to ipratropium), cough and chest-tightness in humans (Fuller et al., 1985) and cough, reflex-bronchospasm and decreased in respiratory rate in rodents (Chou et al., 2008; Forsberg et al., 1988; Mazzone and Canning, 2002b). Electrophysiological recordings of airway sensory nerves in rodents have confirmed this association – O3, H2O2 and alpha-beta unsaturated carbonyl groups evoke action potential discharge in vagal bronchopulmonary C-fibers (Lin et al., 2010; Ruan et al., 2003; Taylor-Clark et al., 2008a; Taylor-Clark and Undem, 2010). Evidence suggests these ROS have no effect on Aβ fibers. However O3 has been associated with the activation of A fibers, an effect likely to be secondary to changes in lung compliance (Coleridge et al., 1993). O3, unlike the other ROS, has also been shown to increase the excitability of vagal bronchopulmonary C-fibers, such that other stimuli evoke augmented responses (Ho and Lee, 1998; Taylor-Clark and Undem, 2010). Vagal sensory nerve recordings in vivo or ex vivo do not prove a direct interaction between ROS and nociceptive sensory nerves. Nevertheless direct interactions have been subsequently shown in dissociated sensory nerves studies using Ca2+ imaging or patch clamp analysis. Capsaicin-sensitive (i.e. nociceptive) neurons are activated by H2O2, hypochlorite, O3, 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, formaldehyde, acrolein, isocyanates and particulate matter (Agopyan et al., 2003b; Andersson et al., 2008; Andre et al., 2008; Bautista et al., 2006; Bessac et al., 2009; Bessac et al., 2008; Macpherson et al., 2007b; McNamara et al., 2007; Taylor-Clark et al., 2009; Taylor-Clark et al., 2008a; Taylor-Clark and Undem, 2010; Trevisani et al., 2007).
A number of specific mechanisms have been proposed for the activation of nociceptive bronchopulmonary nerves. We will concentrate specifically on airway sensory nerve studies and review the available evidence for the roles of transient receptor potential vanilloid 1 (TRPV1), transient receptor potential ankyrin 1 (TRPA1) and purinergic P2X receptors.
3.1 Transient Receptor Potential A1
Although first cloned from a fibroblast cell line, expression of TRPA1 is largely restricted to a subset of nociceptive sensory nerves, including vagal sensory nerves (Bandell et al., 2004; Jordt et al., 2004; Story et al., 2003). Single cell RT-PCR of airway-specific vagal sensory neurons demonstrate the selective expression of TRPA1 in those neurons that also express TRPV1 (a marker for nociceptive function) (Nassenstein et al., 2008). Selective TRPA1 ligands (allyl isothiocyanate and cinnamaldehyde) activate dissociated vagal neurons and bronchopulmonary C-fibers in a TRPA1-dependent manner (Nassenstein et al., 2008; Taylor-Clark et al., 2008a). Exposing the airways to allyl isothiocyanate evokes decreased respiratory rates in wild-type mice but not TRPA1 knockout (TRPA1−/−) mice (Taylor-Clark et al., 2009). The TRPA1 agonist cinnamaldehyde causes cough in human volunteers and several TRPA1 agonists cause cough in guinea pigs by a mechanism sensitive to TRPA1 antagonists (Andre et al., 2009; Birrell et al., 2009).
TRPA1 is a polymodal sensor which in heterologous systems can be activated by cold temperatures, increases in cytosolic Ca2+ and irritants such as menthol, icillin and Δ9-tetrahydrocannabinol (Bessac and Jordt, 2008; Patapoutian et al., 2009). However, its activation via covalent modification is likely more relevant to bronchopulmonary detection of airway oxidative stress. TRPA1 ion channels are activated by cysteine-modifying agents and this seems to be largely independent of the specific structure of the ligands. Hence iodoacetamide, allyl isothiocyanate, cinnamaldehyde and deoxy Δ12,14 prostaglandin J2 all activate TRPA1 due to their reactivity (Hinman et al., 2006; Macpherson et al., 2007a; Takahashi et al., 2008; Taylor-Clark et al., 2008c). Point mutation studies have identified a few key cysteines on the intracellular N-terminus of human and mouse TRPA1 that are responsible for this covalent modification-induced TRPA1 activation, although a lone lysine residue has also been implicated. ROS are strong electrophiles capable of cysteine-modification – this chemical reaction has been exploited by cellular anti-oxidant mechanisms, such that the cysteine-bearing tripeptide glutathione (often termed GSH) binds covalently to a huge host of ROS including H2O2, 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, acrolein, formaldehyde and isocyanates (Blair, 2006). Therefore the basic channel pharmacology of TRPA1 predicted that it would be relevant to ROS activation of airway nociceptive nerves. In recent years this hypothesis has received considerable experimental support (Table 1, Figure 1).
Table 1.
Published effects of various oxidants on TRPA1, TRPV1 and P2X2 channels. Data comes from ion channel overexpression in cell lines (e.g. HEK293 and CHO cells). N.D., not determined. See references for more details.
| TRPA1 | TRPV1 | P2X2 | |
|---|---|---|---|
| Superoxide (O2−) | EC50 333μM (Sawada et al., 2008) | N.D. | N.D. |
| Hydrogen Peroxide (H2O2) | EC50 23μM (Takahashi et al., 2008) EC50 200μM (Sawada et al., 2008) EC50 230μM (Andersson et al., 2008) |
No effect < 10mM (Andersson et al., 2008) Activation with 10mM (Susankova et al., 2006) Activation with 1mM (Chuang and Lin, 2009) |
Potentiation with 300μM (Coddou et al., 2009) Inhibition IC50730 μM (Mason et al., 2004) |
| Ozone (O3) | EC50 3μM (Taylor-Clark and Undem, 2010) | No effect < 30 μM (Taylor-Clark and Undem, 2010) | N.D. |
| Hypochlorite (OCl−) | EC50 11ppm (Bessac et al., 2008) | N.D. | N.D. |
| Chloramine-T | EC50 11μM (Bessac et al., 2008) | Activation with 1mM (Susankova et al., 2006) | N.D. |
| Toluene diisocyanate | EC50 10μM (Taylor-Clark et al., 2009) | No effect < 100μM (Taylor-Clark et al., 2009) | N.D. |
| Methyl isocyanate | EC50 25μM (Bessac et al., 2009) | No effect < 100μM (Bessac et al., 2009) | N.D. |
| Hexamethylene diisocyanate | EC50 2.6μM (Bessac et al., 2009) | No effect < 100μM (Bessac et al., 2009) | N.D. |
| Acrolein | EC50 5μM (Bautista et al., 2006) EC50 0.8μM (Andre et al., 2008) |
No effect < 100μM (Bautista et al., 2006) No effect < 1mM (Dinis et al., 2004) |
N.D. |
| 4-hydroxy-2- nonenal | EC50 27μM (Trevisani et al., 2007) EC50 13μM (Macpherson et al., 2007b) EC50 20μM (Andersson et al., 2008) EC50 10μM (Taylor-Clark et al., 2008a) |
No effect < 500μM (Macpherson et al., 2007b) No effect < 100uM (Andersson et al., 2008) |
N.D. |
| 4-oxo-2-nonenal | EC50 1.9μM (Andersson et al., 2008) EC50 1.6μM (Taylor-Clark et al., 2008a) |
Activation with 100μM (Taylor-Clark et al., 2008a) | N.D. |
| Crotoaldehyde | EC50 23μM (Andre et al., 2008) | N.D. | N.D. |
| Formaldehyde | EC50 200μM (McNamara et al., 2007), EC 50 357μM (Macpherson et al., 2007b) |
No effect seen < 1mM (Macpherson et al., 2007b) No effect seen < 1.25mM (McNamara et al., 2007) |
N.D. |
| Particulate Matter (PC10) | N.D. | Activation with 1 particle (Agopyan et al., 2003a) | N.D. |
| Particulate Matter (PC2) | N.D. | Activation with 4 particles (Agopyan et al., 2003a) | N.D. |
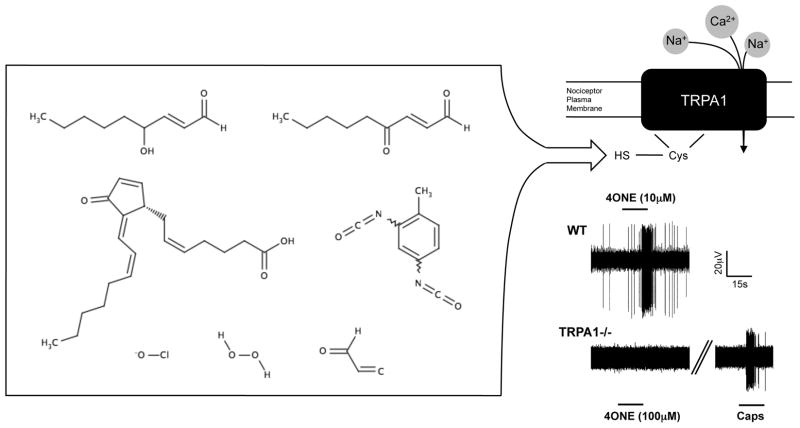
Figure 1.
A wide variety of exogenous and endogenous ROS activate the ion channel TRPA1. Left, (clockwise from top left) 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, toluene diisocyanate, acrolein, hydrogen peroxide, hypochlorite, deoxy Δ12,14 prostaglandin J2. Top right, The free sulphydryl group on specific TRPA1 cysteine residues is the main functional target of electrophilic ROS. Bottom right, Representative traces of bronchopulmonary C-fiber action potential discharge to 4-oxo-2-nonenal (4ONE) in wild-type and TRPA1−/− mice. All C-fibers responded to capsaicin (300nM) at the end of the experiment. Taken from (Taylor-Clark et al., 2008a).
Studies of H2O2 demonstrate that 3–5mM H2O2 selectively activates dissociated neurons that also express TRPA1 and this response is abolished with ruthenium red (non-selective TRP channel blocker) or in TRPA1−/− neurons (Andersson et al., 2008; Bessac et al., 2008; Takahashi et al., 2008). In vivo inhalation of 3% H2O2 evoked a decrease in respiratory rate and an increase in end expiratory pause that was reduced in TRPA1−/− mice by approximately 50% and 80%, respectively. In heterologous expression systems, H2O2 (which is membrane-permeable) activates TRPA1 via cysteine modification (Bessac et al., 2008). Direct association of H2O2 and the TRPA1 channel has not be proved and so it is not clear if H2O2 activates TRPA1 directly or downstream of lipid peroxidation. Nevertheless TRPA1 activation by H2O2 is reversed by dithiothrietol (Andersson et al., 2008; Takahashi et al., 2008), a reducing agent that is unable to break C-S bonds that would be expected to be formed by cysteine covalent modification by downstream products of lipid peroxidation (e.g. 4-hydroxy-2-nonenal).
Alpha-beta unsaturated carbonyl groups (e.g. 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, crotoaldehyde and acrolein) activate TRPA1 in heterologous expression systems (Andersson et al., 2008; Andre et al., 2008; Bautista et al., 2006; Macpherson et al., 2007b; Taylor-Clark et al., 2008a; Trevisani et al., 2007) and consistent with their cysteine-modifying reactivity (Blair, 2006) 4-hydroxy-2-nonenal activation of TRPA1 is reduced following point mutations of specific N-terminal cysteine residues (Trevisani et al., 2007). 1–30μM 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, crotonaldehyde and acrolein selectively activate dissociated neurons that also express TRPA1 and this response is substantially reduced with ruthenium red or in TRPA1−/− neurons (Andersson et al., 2008; Andre et al., 2008; Bautista et al., 2006; Macpherson et al., 2007b; Taylor-Clark et al., 2008a; Trevisani et al., 2007). 4-hydroxy-2-nonenal, 4-oxo-2-nonenal, crotonaldehyde and acrolein all cause tachykinin-dependent contraction of isolated guinea pig bronchus (Andre et al., 2008; Taylor-Clark et al., 2008a). These responses were inhibited by nociceptor tachykinin depletion, ruthenium red and the selective TRPA1 antagonist HC-030031. 10μM 4-oxo-2-nonenal evoked robust action potential discharge from cinnamaldehyde-sensitive mouse bronchopulmonary C-fibers, which was reduced by ruthenium red and abolished in nerves derived from TRPA1−/− mice (Taylor-Clark et al., 2008a). In this study, however, 4-hydroxy-2-nonenal failed to evoke bronchopulmonary C-fiber activation even at 100uM. 4-hydroxy-2-nonenal evokes noicefensive behavior after intraplanatar injections of 3–6mM (in a TRPA1-dependent manner) (Trevisani et al., 2007). It is consistent with the substantial nucleophilic anti-oxidant defense mechanisms (e.g. glutathione) that are present in complex tissues that these electrophilic TRPA1 agonists may have dramatically different EC50 in single cell vs. whole tissue experiments. In inhalation studies in guinea pigs crotonaldehyde and acrolein evoke cough in awake animals that was reduced by HC-030031 (Andre et al., 2009; Birrell et al., 2009).
Toluene diisocyanate, methyl isocyanate and hexamethylene diisocyanate are industrial pollutants that activate TRPA1 channels in heterologous expression systems (Bessac et al., 2009; Taylor-Clark et al., 2009). 10–100μM isocyanate selectively activates dissociated neurons that also express TRPA1 and this response is substantially reduced in TRPA1−/− neurons. 1% toluene diisocyanate evokes defensive reflexes in mice after inhalation consistent with nociceptive nerve activation such as decreased respiratory rate and an increased time of brake (Alarie, 1973). These airway reflexes are abolished in TRPA1−/− mice (Taylor-Clark et al., 2009).
Hypochlorite (OCl−), the oxidizing mediator of chlorine, and chloramine-T, a chlorine donor, activate heterologously expressed TRPA1 (Bessac et al., 2008). Hypochlorite selectively activates dissociated neurons that also express TRPA1 and this response is substantially reduced with ruthenium red treatment and abolished in TRPA1−/− neurons (Bessac et al., 2008). In vivo inhalation of 6ppm NaOCl evoked a decrease in respiratory rate and an increase in end expiratory pause that was abolished in TRPA1−/− mice (Bessac et al., 2008).
Solubilized O3 activates heterologously expressed TRPA1 (Taylor-Clark and Undem, 2010). O3 selectively activates dissociated neurons that also express TRPA1 and this response is abolished in TRPA1−/− neurons. O3 evoked robust action potential discharge from cinnamaldehyde-sensitive mouse bronchopulmonary C-fibers, which was reduced by approximately 80% by ruthenium red (Taylor-Clark and Undem, 2010). O3 also causes airway nociceptor hyperexcitability (Ho and Lee, 1998; Taylor-Clark and Undem, 2010), although it is unclear if this is due to TRPA1 function. It should be noted that due to O3 high reactivity it would seem unlikely that inhaled O3 would be able to directly access the intracellular N-terminal of TRPA1 on nociceptive nerves (Pryor, 1992), thus O3 may stimulate TRPA1 indirectly downstream of ROS production and lipid peroxidation.
The role of TRPA1 in airway afferent signaling has been studied in more complex conditions: cigarette smoke and inflammation. Both cigarette smoke (Andre et al., 2008; Andre et al., 2009; Lin et al., 2010) and inflammation (Joseph et al., 2008; Kamboj et al., 2010; Kuhad and Chopra, 2009; Tall et al., 2004) have the potential to modulate afferent excitability downstream of ROS production, although it is well established that cigarette smoke contains nicotine and inflammation induces bradykinin, lipoxygenase products and cyclooxygenase products – all of which can modulate airway nociceptor function independent of oxidative stress (Carr et al., 2003; Kwong and Lee, 2005; Lee et al., 2007; Lee et al., 1989; McAlexander et al., 1998; Taylor-Clark et al., 2008b). Cigarette smoke activated guinea pig vagal neurons and evoked a tachykinin-dependent contraction of isolated guinea pig bronchus in a manner that was reduced by over 80% by either the TRPA1 channel blocker HC-030031 or glutathione (Andre et al., 2008). In nerve fiber recordings of nociceptive airway afferents in rats, cigarette smoke evoked substantial action potential discharge that was reduced by 90% by N-acetyl-cysteine and by 60% by HC-030031 (Lin et al., 2010). In addition, inhibiting TRPA1 with HC-030031 reduced cigarette-evoked cough in awake guinea pigs by 50% (Andre et al., 2009).
An important role for TRPA1 has also emerged in a mouse model of allergic asthma (Caceres et al., 2009). Allergen-induced eosinophilia, mucin production and airway hyperreactivity were reduced by HC-030031 in wild-type mice by 35%, 90% and 90%, respectively. These markers of allergen-induced airway inflammation were also reduced in TRPA1−/− mice. More work is needed to determine if ROS was involved in the pathway to TRPA1 activation in this model.
3.2 Transient Receptor Potential Vanilloid 1 (TRPV1)
TRPV1 is a cation channel selectively expressed on nociceptive nerves. TRPV1 is a polymodal sensor of noxious stimuli, including capsaicin, heat, acid, endocannabinoids and lipoxygenase products (Bessac and Jordt, 2008; Caterina et al., 1997; Patapoutian et al., 2009). TRPV1 is expressed on bronchopulmonary C-fibers in all mammals studied, and capsaicin-induced bronchopulmonary C-fiber activation is abolished by the selective TRPV1 inhibitors (iodoresiniferatoxin (IRTX) and capsazepine) and in TRPV1 knockout (TRPV1−/−) mice (Kollarik and Undem, 2004; Lee and Lundberg, 1994; Nassenstein et al., 2008; Ruan et al., 2005; Undem and Kollarik, 2002). There is evidence to support a role of TRPV1 in oxidative-stress induced airway nociceptor activation. In heterologous expression systems TRPV1 has been shown to be activated by the reactive compounds allicin, 2-aminoethyl methanethiosulfonate and 4-oxo-2-nonenal (Macpherson et al., 2005; Salazar et al., 2008; Taylor-Clark et al., 2008a), which is dependent on an individual cysteine residue on TRPV1. Heterologously expressed TRPV1 has also been shown to be sensitized by chloramine-T (Susankova et al., 2006) and H2O2, in a dithiothrietol-sensitive mechanism (Chuang and Lin, 2009). It should be noted that several reactive compounds that are known to stimulate TRPA1 have no effect on TRPV1 including acrolein, formaldehyde, iodoacetamide, 4-hydroxy-2-nonenal, toluene diisocyanate and O3 (Bautista et al., 2006; Dinis et al., 2004; Macpherson et al., 2007a; McNamara et al., 2007; Taylor-Clark et al., 2009; Taylor-Clark and Undem, 2010; Trevisani et al., 2007).
Allicin, 2-aminoethyl methanethiosulfonate and 4-oxo-2-nonenal are also TRPA1 agonists and TRPA1 and TRPV1 are co-expressed on many nociceptive nerves including bronchopulmonary C-fibers (Nassenstein et al., 2008). Although these 3 reactive compounds activate dissociated nociceptive neurons in a TRPV1-dependent mechanism, evidence is unclear as to whether this effect can be observed without inhibiting TRPA1 first (Salazar et al., 2008; Taylor-Clark et al., 2008a). Of the 3 reactive TRPV1 ligands, only the actions of 4-oxo-2-nonenal has been studied at the nerve terminal of bronchopulmonary afferents, and no evidence was found for a role of TRPV1. When 100 μM 4-oxo-2-nonenal (a concentration that activates heterologously expressed TRPV1) was directly applied to the airway, action potential discharge is observed in wild-type, but not in TRPA1−/− mice (Taylor-Clark et al., 2008a). It may be possible that higher concentrations may activate the bronchopulmonary C-fibers via TRPV1, but a recent paper studying nocifensive behaviour induced by 200–600uM 4-oxo-2-nonenal intraplantar injections demonstrated no role for TRPV1 (even after TRPA1 inhibition) (Graepel et al., 2011). 4-oxo-2-nonenal has yet to be shown to induce action potential discharge from nociceptor nerve terminals via TRPV1. As mentioned above, H2O2 can induce TRPV1 hyperexcitability and a study of rat bronchopulmonary C-fibers suggest that capsazepine (a TRPV1 antagonist) can reduce H2O2-induced action potential discharge from nociceptive nerve terminals by 40% (Ruan et al., 2005). Furthermore, inhalation of H2O2 induces reflex bradypneic responses that are reduced by 50% by capsazepine (Ruan et al., 2006).
Particulate matter (both PC10 and smaller PC2 particles) activates heterologously expressed TRPV1, in a non-desensitizing manner (Agopyan et al., 2003a). Dissociated trigeminal nociceptive neurons are also activated by particulate matter which is abolished by capsazepine in wild-type neurons and absent in TRPV1−/− neurons (Agopyan et al., 2004; Agopyan et al., 2003b). To date there are no published studies of airway nerve activation by particulate matter, although reflex changes in cardiac function following inhalation of particulate matter was reduced by capsazepine (Ghelfi et al., 2008). Particulate matter is capable of substantial ROS production, via the actions of metal ions and quinones in particles (Li et al., 2008b); although whether ROS mediate airway nociceptor TRPV1-mediated activation by particulate matter is yet to be confirmed.
Oxidative stress is associated with cigarette smoke toxicity, however there is little evidence to suggest a role for TRPV1. Capsazepine has no effect on neurogenic inflammation induced by cigarette smoke extract (Andre et al., 2008; Geppetti et al., 1993), although it does reduce cough by approximately 30% (Andre et al., 2009).
3.3 P2X receptors
P2X receptors are cation channels that are primarily activated by ATP and its analogs. Airway sensory nerves express mRNA for both P2X2 and P2X3. P2X3 protein is expressed in virtually all airway nociceptive vagal neurons, whereas P2X2 protein is only expressed in airway nociceptive vagal neurons whose cell bodies reside in the nodose ganglia (Kwong et al., 2008; Nassenstein et al., 2010). Either protein can assemble into functional homomeric channels, but when co-expressed they form P2X2/3 heteromeric channels that possess properties distinct from the homomeric channels (Liu et al., 2001). As such airway jugular nociceptors express mainly rapidly inactivating homomeric P2X3 channels whereas airway nodose nociceptors express more slowly inactivating P2X2/3 channels. Although both P2X3 and P2X2/3 channels are activated by ATP and α,β methylene ATP, at the level of the nerve terminals in airways, nodose but not jugular C-fibers respond with action potential discharge (Undem et al., 2004).
H2O2 has been shown to activate rat bronchopulmonary C-fibers and this response was inhibited by 50% by iso-PPADS (non-specific P2X inhibitor) (Ruan et al., 2005). In addition inhalation of H2O2 induces reflex bradypneic responses that are reduced by 50% by iso-PPADS (Ruan et al., 2006). The bradypneic responses were also reduced by 50% by scavenging ATP, suggesting that H2O2 may activate P2X receptors indirectly following ATP release. The direct effect of ROS and H2O2 in particular on P2X channels is unclear. H2O2 has been shown to directly modulate P2X2 channels, both potentiating ATP-induced responses (Coddou et al., 2009) and attenuating ATP-induced responses (although only with homomeric P2X2 and not heteromeric P2X2/3 channels) (Mason et al., 2004). In both studies P2X3 channels were unaffected. Reactive carbonyl groups have no effect on P2X2 channels (Li et al., 2008a). Lastly, cigarette smoke-induced activation of rat nociceptive airway nerves was reduced by 50% by either iso-PPADS or ATP scavenging (Lin et al., 2010).
4. Conclusions
There is a growing literature demonstrating the selective activation of nociceptive airway afferents with various ROS and productions of oxidative stress. Evidence supporting a role of TRPA1 in these responses is very strong. Results from a plethora of studies now indicate that ROS, regardless of structure, categorically stimulate TRPA1. Furthermore, inhibition of TRPA1 dramatically reduces ROS-induced afferent nerve activation. Whether other ion channels (TRPV1 or P2X) also contribute is more questionable and may depend more on the specific ROS. Whereas it is likely that ROS and products of lipid peroxidation directly bind and activate TRPA1, evidence suggests that purinergic receptor and TRPV1 may be activated downstream of other ROS-induced biological processes (cellular destruction, lipid metabolism, ATP release). TRPA1 channels represent a potential target for respiratory symptoms following inhalation of pro-oxidants. Inhibition of TRPA1 may also be useful in inflammatory diseases associated with oxidative stress, namely asthma and COPD.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributor Information
Thomas E. Taylor-Clark, Email: ttaylorc@health.usf.edu.
Bradley J. Undem, Email: bundem@jhmi.edu.
References
- Agopyan N, Bhatti T, Yu S, Simon SA. Vanilloid receptor activation by 2- and 10-microm particles induces responses leading to apoptosis in human airway epithelial cells. Toxicol Appl Pharmacol. 2003a;192:21–35. doi: 10.1016/s0041-008x(03)00259-x. [DOI] [PubMed] [Google Scholar]
- Agopyan N, Head J, Yu S, Simon SA. TRPV1 receptors mediate particulate matter-induced apoptosis. Am J Physiol Lung Cell Mol Physiol. 2004;286:L563–572. doi: 10.1152/ajplung.00299.2003. [DOI] [PubMed] [Google Scholar]
- Agopyan N, Li L, Yu S, Simon SA. Negatively charged 2- and 10-microm particles activate vanilloid receptors, increase cAMP, and induce cytokine release. Toxicol Appl Pharmacol. 2003b;186:63–76. doi: 10.1016/s0041-008x(02)00013-3. [DOI] [PubMed] [Google Scholar]
- Alarie Y. Sensory irritation of the upper airways by airborne chemicals. Toxicol Appl Pharmacol. 1973;24:279–297. doi: 10.1016/0041-008x(73)90148-8. [DOI] [PubMed] [Google Scholar]
- Andersson DA, Gentry C, Moss S, Bevan S. Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J Neurosci. 2008;28:2485–2494. doi: 10.1523/JNEUROSCI.5369-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andre E, Campi B, Materazzi S, Trevisani M, Amadesi S, Massi D, Creminon C, Vaksman N, Nassini R, Civelli M, Baraldi PG, Poole DP, Bunnett NW, Geppetti P, Patacchini R. Cigarette smoke-induced neurogenic inflammation is mediated by alpha, beta-unsaturated aldehydes and the TRPA1 receptor in rodents. J Clin Invest. 2008;118:2574–2582. doi: 10.1172/JCI34886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andre E, Gatti R, Trevisani M, Preti D, Baraldi PG, Patacchini R, Geppetti P. Transient receptor potential ankyrin receptor 1 is a novel target for pro-tussive agents. Br J Pharmacol. 2009;158:1621–1628. doi: 10.1111/j.1476-5381.2009.00438.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker CV, Bronner-Fraser M. Establishing neuronal identity in vertebrate neurogenic placodes. Development. 2000;127:3045–3056. doi: 10.1242/dev.127.14.3045. [DOI] [PubMed] [Google Scholar]
- Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, Earley TJ, Patapoutian A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron. 2004;41:849–857. doi: 10.1016/s0896-6273(04)00150-3. [DOI] [PubMed] [Google Scholar]
- Bautista DM, Jordt SE, Nikai T, Tsuruda PR, Read AJ, Poblete J, Yamoah EN, Basbaum AI, Julius D. TRPA1 Mediates the Inflammatory Actions of Environmental Irritants and Proalgesic Agents. Cell. 2006;124:1269–1282. doi: 10.1016/j.cell.2006.02.023. [DOI] [PubMed] [Google Scholar]
- Bergren DR. Sensory receptor activation by mediators of defense reflexes in guinea-pig lungs. Respir Physiol. 1997;108:195–204. doi: 10.1016/s0034-5687(97)00030-3. [DOI] [PubMed] [Google Scholar]
- Bessac BF, Jordt SE. Breathtaking TRP channels: TRPA1 and TRPV1 in airway chemosensation and reflex control. Physiology (Bethesda) 2008;23:360–370. doi: 10.1152/physiol.00026.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessac BF, Sivula M, von Hehn CA, Caceres AI, Escalera J, Jordt SE. Transient receptor potential ankyrin 1 antagonists block the noxious effects of toxic industrial isocyanates and tear gases. Faseb J. 2009;23:1102–1114. doi: 10.1096/fj.08-117812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessac BF, Sivula M, von Hehn CA, Escalera J, Cohn L, Jordt SE. TRPA1 is a major oxidant sensor in murine airway sensory neurons. J Clin Invest. 2008;118:1899–1910. doi: 10.1172/JCI34192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birrell MA, Belvisi MG, Grace M, Sadofsky L, Faruqi S, Hele DJ, Maher SA, Freund-Michel V, Morice AH. TRPA1 Agonists Evoke Coughing in Guinea-pig and Human Volunteers. Am J Respir Crit Care Med. 2009;180:1042–1047. doi: 10.1164/rccm.200905-0665OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair IA. Endogenous glutathione adducts. Curr Drug Metab. 2006;7:853–872. doi: 10.2174/138920006779010601. [DOI] [PubMed] [Google Scholar]
- Brown WE, Burkert AL. Biomarkers of toluene diisocyanate exposure. Appl Occup Environ Hyg. 2002;17:840–845. doi: 10.1080/10473220290107039. [DOI] [PubMed] [Google Scholar]
- Brugsch HG, Elkins HB. Toluene di-isocyanate (TDI) toxicity. N Engl J Med. 1963;268:353–357. doi: 10.1056/NEJM196302142680705. [DOI] [PubMed] [Google Scholar]
- Butcher BT, Salvaggio JE, Weill H, Ziskind MM. Toluene diisocyanate (TDI) pulmonary disease: immunologic and inhalation challenge studies. J Allergy Clin Immunol. 1976;58:89–100. doi: 10.1016/0091-6749(76)90110-x. [DOI] [PubMed] [Google Scholar]
- Caceres AI, Brackmann M, Elia MD, Bessac BF, del Camino D, D’Amours M, Witek JS, Fanger CM, Chong JA, Hayward NJ, Homer RJ, Cohn L, Huang X, Moran MM, Jordt SE. A sensory neuronal ion channel essential for airway inflammation and hyperreactivity in asthma. Proc Natl Acad Sci U S A. 2009;106:9099–9104. doi: 10.1073/pnas.0900591106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr MJ, Kollarik M, Meeker SN, Undem BJ. A role for TRPV1 in bradykinin-induced excitation of vagal airway afferent nerve terminals. J Pharmacol Exp Ther. 2003;304:1275–1279. doi: 10.1124/jpet.102.043422. [DOI] [PubMed] [Google Scholar]
- Carr MJ, Undem BJ. Bronchopulmonary afferent nerves. Respirology. 2003;8:291–301. doi: 10.1046/j.1440-1843.2003.00473.x. [DOI] [PubMed] [Google Scholar]
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389:816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- Chou YL, Scarupa MD, Mori N, Canning BJ. Differential effects of airway afferent nerve subtypes on cough and respiration in anesthetized guinea pigs. Am J Physiol Regul Integr Comp Physiol. 2008;295:R1572–1584. doi: 10.1152/ajpregu.90382.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang HH, Lin S. Oxidative challenges sensitize the capsaicin receptor by covalent cysteine modification. Proc Natl Acad Sci U S A. 2009;106:20097–20102. doi: 10.1073/pnas.0902675106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang HH, Prescott ED, Kong H, Shields S, Jordt SE, Basbaum AI, Chao MV, Julius D. Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P2-mediated inhibition. Nature. 2001;411:957–962. doi: 10.1038/35082088. [DOI] [PubMed] [Google Scholar]
- Coddou C, Codocedo JF, Li S, Lillo JG, Acuna-Castillo C, Bull P, Stojilkovic SS, Huidobro-Toro JP. Reactive oxygen species potentiate the P2X2 receptor activity through intracellular Cys430. J Neurosci. 2009;29:12284–12291. doi: 10.1523/JNEUROSCI.2096-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleridge JC, Coleridge HM. Afferent vagal C fibre innervation of the lungs and airways and its functional significance. Rev Physiol Biochem Pharmacol. 1984;99:1–110. doi: 10.1007/BFb0027715. [DOI] [PubMed] [Google Scholar]
- Coleridge JC, Coleridge HM, Schelegle ES, Green JF. Acute inhalation of ozone stimulates bronchial C-fibers and rapidly adapting receptors in dogs. J Appl Physiol. 1993;74:2345–2352. doi: 10.1152/jappl.1993.74.5.2345. [DOI] [PubMed] [Google Scholar]
- Comandini A, Rogliani P, Nunziata A, Cazzola M, Curradi G, Saltini C. Biomarkers of lung damage associated with tobacco smoke in induced sputum. Respir Med. 2009;103:1592–1613. doi: 10.1016/j.rmed.2009.06.002. [DOI] [PubMed] [Google Scholar]
- Comhair SA, Erzurum SC. Redox control of asthma: molecular mechanisms and therapeutic opportunities. Antioxid Redox Signal. 2010;12:93–124. doi: 10.1089/ars.2008.2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinis P, Charrua A, Avelino A, Yaqoob M, Bevan S, Nagy I, Cruz F. Anandamide-evoked activation of vanilloid receptor 1 contributes to the development of bladder hyperreflexia and nociceptive transmission to spinal dorsal horn neurons in cystitis. J Neurosci. 2004;24:11253–11263. doi: 10.1523/JNEUROSCI.2657-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faroon O, Roney N, Taylor J, Ashizawa A, Lumpkin MH, Plewak DJ. Acrolein health effects. Toxicol Ind Health. 2008;24:447–490. doi: 10.1177/0748233708094188. [DOI] [PubMed] [Google Scholar]
- Forsberg K, Karlsson JA, Theodorsson E, Lundberg JM, Persson CG. Cough and bronchoconstriction mediated by capsaicin-sensitive sensory neurons in the guinea-pig. Pulm Pharmacol. 1988;1:33–39. doi: 10.1016/0952-0600(88)90008-7. [DOI] [PubMed] [Google Scholar]
- Fuller RW, Dixon CM, Barnes PJ. Bronchoconstrictor response to inhaled capsaicin in humans. J Appl Physiol. 1985;58:1080–1084. doi: 10.1152/jappl.1985.58.4.1080. [DOI] [PubMed] [Google Scholar]
- Geppetti P, Bertrand C, Baker J, Yamawaki I, Piedimonte G, Nadel JA. Ruthenium red, but not capsazepine reduces plasma extravasation by cigarette smoke in rat airways. Br J Pharmacol. 1993;108:646–650. doi: 10.1111/j.1476-5381.1993.tb12855.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghelfi E, Rhoden CR, Wellenius GA, Lawrence J, Gonzalez-Flecha B. Cardiac oxidative stress and electrophysiological changes in rats exposed to concentrated ambient particles are mediated by TRP-dependent pulmonary reflexes. Toxicol Sci. 2008;102:328–336. doi: 10.1093/toxsci/kfn005. [DOI] [PubMed] [Google Scholar]
- Gloire G, Piette J. Redox regulation of nuclear post-translational modifications during NF-kappaB activation. Antioxid Redox Signal. 2009;11:2209–2222. doi: 10.1089/ars.2009.2463. [DOI] [PubMed] [Google Scholar]
- Graepel R, Fernandes ES, Aubdool AA, Andersson D, Bevan S, Brain SD. 4-oxo-2-nonenal (4-ONE): Evidence of TRPA1-dependent and - independent nociceptive and vasoactive responses in vivo. J Pharmacol Exp Ther. 2011 doi: 10.1124/jpet.110.172403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hazbun ME, Hamilton R, Holian A, Eschenbacher WL. Ozone-induced increases in substance P and 8-epi-prostaglandin F2 alpha in the airways of human subjects. Am J Respir Cell Mol Biol. 1993;9:568–572. doi: 10.1165/ajrcmb/9.5.568. [DOI] [PubMed] [Google Scholar]
- Hazucha MJ, Bates DV, Bromberg PA. Mechanism of action of ozone on the human lung. J Appl Physiol. 1989;67:1535–1541. doi: 10.1152/jappl.1989.67.4.1535. [DOI] [PubMed] [Google Scholar]
- Hinman A, Chuang HH, Bautista DM, Julius D. TRP channel activation by reversible covalent modification. Proc Natl Acad Sci U S A. 2006;103:19564–19568. doi: 10.1073/pnas.0609598103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho CY, Lee LY. Ozone enhances excitabilities of pulmonary C fibers to chemical and mechanical stimuli in anesthetized rats. J Appl Physiol. 1998;85:1509–1515. doi: 10.1152/jappl.1998.85.4.1509. [DOI] [PubMed] [Google Scholar]
- Hollingsworth JW, Kleeberger SR, Foster WM. Ozone and pulmonary innate immunity. Proc Am Thorac Soc. 2007;4:240–246. doi: 10.1513/pats.200701-023AW. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Islam T, Berhane K, McConnell R, Gauderman WJ, Avol E, Peters JM, Gilliland FD. Glutathione-S-transferase (GST) P1, GSTM1, exercise, ozone and asthma incidence in school children. Thorax. 2009;64:197–202. doi: 10.1136/thx.2008.099366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordt SE, Bautista DM, Chuang HH, McKemy DD, Zygmunt PM, Hogestatt ED, Meng ID, Julius D. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature. 2004;427:260–265. doi: 10.1038/nature02282. [DOI] [PubMed] [Google Scholar]
- Joseph EK, Chen X, Bogen O, Levine JD. Oxaliplatin acts on IB4-positive nociceptors to induce an oxidative stress-dependent acute painful peripheral neuropathy. J Pain. 2008;9:463–472. doi: 10.1016/j.jpain.2008.01.335. [DOI] [PubMed] [Google Scholar]
- Kamboj SS, Vasishta RK, Sandhir R. N-acetylcysteine inhibits hyperglycemia-induced oxidative stress and apoptosis markers in diabetic neuropathy. J Neurochem. 2010;112:77–91. doi: 10.1111/j.1471-4159.2009.06435.x. [DOI] [PubMed] [Google Scholar]
- Karihtala P, Soini Y. Reactive oxygen species and antioxidant mechanisms in human tissues and their relation to malignancies. Apmis. 2007;115:81–103. doi: 10.1111/j.1600-0463.2007.apm_514.x. [DOI] [PubMed] [Google Scholar]
- Kerr HD, Kulle TJ, McIlhany ML, Swidersky P. Effects of ozone on pulmonary function in normal subjects. An environmental-chamber study. Am Rev Respir Dis. 1975;111:763–773. doi: 10.1164/arrd.1975.111.6.763. [DOI] [PubMed] [Google Scholar]
- Kharitonov SA, Barnes PJ. Effects of corticosteroids on noninvasive biomarkers of inflammation in asthma and chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2004;1:191–199. doi: 10.1513/pats.200402-011MS. [DOI] [PubMed] [Google Scholar]
- Kinnula VL, Pietarinen P, Aalto K, Virtanen I, Raivio KO. Mitochondrial superoxide dismutase induction does not protect epithelial cells during oxidant exposure in vitro. Am J Physiol. 1995;268:L71–77. doi: 10.1152/ajplung.1995.268.1.L71. [DOI] [PubMed] [Google Scholar]
- Kirkham P, Rahman I. Oxidative stress in asthma and COPD: antioxidants as a therapeutic strategy. Pharmacol Ther. 2006;111:476–494. doi: 10.1016/j.pharmthera.2005.10.015. [DOI] [PubMed] [Google Scholar]
- Kollarik M, Undem BJ. Activation of bronchopulmonary vagal afferent nerves with bradykinin, acid and vanilloid receptor agonists in wild-type and TRPV1−/− mice. J Physiol. 2004;555:115–123. doi: 10.1113/jphysiol.2003.054890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhad A, Chopra K. Tocotrienol attenuates oxidative-nitrosative stress and inflammatory cascade in experimental model of diabetic neuropathy. Neuropharmacology. 2009;57:456–462. doi: 10.1016/j.neuropharm.2009.06.013. [DOI] [PubMed] [Google Scholar]
- Kwong K, Kollarik M, Nassenstein C, Ru F, Undem BJ. P2X2 Receptors Differentiate Placodal vs Neural Crest C-fiber Phenotypes Innervating Guinea Pig Lungs and Esophagus. Am J Physiol Lung Cell Mol Physiol. 2008;295:L858–865. doi: 10.1152/ajplung.90360.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwong K, Lee LY. Prostaglandin E2 potentiates a TTX-resistant sodium current in rat capsaicin-sensitive vagal pulmonary sensory neurones. J Physiol. 2005;564:437–450. doi: 10.1113/jphysiol.2004.078725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee LY. Respiratory sensations evoked by activation of bronchopulmonary C-fibers. Respir Physiol Neurobiol. 2009;167:26–35. doi: 10.1016/j.resp.2008.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee LY, Burki NK, Gerhardstein DC, Gu Q, Kou YR, Xu J. Airway irritation and cough evoked by inhaled cigarette smoke: role of neuronal nicotinic acetylcholine receptors. Pulm Pharmacol Ther. 2007;20:355–364. doi: 10.1016/j.pupt.2006.10.007. [DOI] [PubMed] [Google Scholar]
- Lee LY, Dumont C, Djokic TD, Menzel TE, Nadel JA. Mechanism of rapid, shallow breathing after ozone exposure in conscious dogs. J Appl Physiol. 1979;46:1108–1114. doi: 10.1152/jappl.1979.46.6.1108. [DOI] [PubMed] [Google Scholar]
- Lee LY, Kou YR, Frazier DT, Beck ER, Pisarri TE, Coleridge HM, Coleridge JC. Stimulation of vagal pulmonary C-fibers by a single breath of cigarette smoke in dogs. J Appl Physiol. 1989;66:2032–2038. doi: 10.1152/jappl.1989.66.5.2032. [DOI] [PubMed] [Google Scholar]
- Lee LY, Lundberg JM. Capsazepine abolishes pulmonary chemoreflexes induced by capsaicin in anesthetized rats. J Appl Physiol. 1994;76:1848–1855. doi: 10.1152/jappl.1994.76.5.1848. [DOI] [PubMed] [Google Scholar]
- Li M, Chang TH, Silberberg SD, Swartz KJ. Gating the pore of P2X receptor channels. Nat Neurosci. 2008a;11:883–887. doi: 10.1038/nn.2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li N, Xia T, Nel AE. The role of oxidative stress in ambient particulate matter-induced lung diseases and its implications in the toxicity of engineered nanoparticles. Free Radic Biol Med. 2008b;44:1689–1699. doi: 10.1016/j.freeradbiomed.2008.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin JL, Thomas PS. Current perspectives of oxidative stress and its measurement in chronic obstructive pulmonary disease. COPD. 2010;7:291–306. doi: 10.3109/15412555.2010.496818. [DOI] [PubMed] [Google Scholar]
- Lin YS, Hsu CC, Bien MY, Hsu HC, Weng HT, Kou YR. Activations of TRPA1 and P2X receptors are important in ROS-mediated stimulation of capsaicin-sensitive lung vagal afferents by cigarette smoke in rats. J Appl Physiol. 2010;108:1293–1303. doi: 10.1152/japplphysiol.01048.2009. [DOI] [PubMed] [Google Scholar]
- Liu M, King BF, Dunn PM, Rong W, Townsend-Nicholson A, Burnstock G. Coexpression of P2X(3) and P2X(2) receptor subunits in varying amounts generates heterogeneous populations of P2X receptors that evoke a spectrum of agonist responses comparable to that seen in sensory neurons. J Pharmacol Exp Ther. 2001;296:1043–1050. [PubMed] [Google Scholar]
- Mabalirajan U, Ahmad T, Leishangthem GD, Dinda AK, Agrawal A, Ghosh B. L-arginine reduces mitochondrial dysfunction and airway injury in murine allergic airway inflammation. Int Immunopharmacol. 2010;10:1514–1519. doi: 10.1016/j.intimp.2010.08.025. [DOI] [PubMed] [Google Scholar]
- Mabalirajan U, Aich J, Leishangthem GD, Sharma SK, Dinda AK, Ghosh B. Effects of vitamin E on mitochondrial dysfunction and asthma features in an experimental allergic murine model. J Appl Physiol. 2009;107:1285–1292. doi: 10.1152/japplphysiol.00459.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mabalirajan U, Dinda AK, Kumar S, Roshan R, Gupta P, Sharma SK, Ghosh B. Mitochondrial structural changes and dysfunction are associated with experimental allergic asthma. J Immunol. 2008;181:3540–3548. doi: 10.4049/jimmunol.181.5.3540. [DOI] [PubMed] [Google Scholar]
- Macpherson LJ, Dubin AE, Evans MJ, Marr F, Schultz PG, Cravatt BF, Patapoutian A. Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature. 2007a;445:541–545. doi: 10.1038/nature05544. [DOI] [PubMed] [Google Scholar]
- Macpherson LJ, Geierstanger BH, Viswanath V, Bandell M, Eid SR, Hwang S, Patapoutian A. The pungency of garlic: activation of TRPA1 and TRPV1 in response to allicin. Curr Biol. 2005;15:929–934. doi: 10.1016/j.cub.2005.04.018. [DOI] [PubMed] [Google Scholar]
- Macpherson LJ, Xiao B, Kwan KY, Petrus MJ, Dubin AE, Hwang S, Cravatt B, Corey DP, Patapoutian A. An ion channel essential for sensing chemical damage. J Neurosci. 2007b;27:11412–11415. doi: 10.1523/JNEUROSCI.3600-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason HS, Bourke S, Kemp PJ. Selective modulation of ligand-gated P2X purinoceptor channels by acute hypoxia is mediated by reactive oxygen species. Mol Pharmacol. 2004;66:1525–1535. doi: 10.1124/mol.104.000851. [DOI] [PubMed] [Google Scholar]
- Mazzone SB, Canning BJ. Central nervous system control of the airways: pharmacological implications. Curr Opin Pharmacol. 2002a;2:220–228. doi: 10.1016/s1471-4892(02)00151-0. [DOI] [PubMed] [Google Scholar]
- Mazzone SB, Canning BJ. Synergistic interactions between airway afferent nerve subtypes mediating reflex bronchospasm in guinea pigs. Am J Physiol Regul Integr Comp Physiol. 2002b;283:R86–98. doi: 10.1152/ajpregu.00007.2002. [DOI] [PubMed] [Google Scholar]
- McAlexander MA, Myers AC, Undem BJ. Inhibition of 5-lipoxygenase diminishes neurally evoked tachykinergic contraction of guinea pig isolated airway. J Pharmacol Exp Ther. 1998;285:602–607. [PubMed] [Google Scholar]
- McDonnell WF, Stewart PW, Smith MV, Pan WK, Pan J. Ozone-induced respiratory symptoms: exposure-response models and association with lung function. Eur Respir J. 1999;14:845–853. doi: 10.1034/j.1399-3003.1999.14d21.x. [DOI] [PubMed] [Google Scholar]
- McNamara CR, Mandel-Brehm J, Bautista DM, Siemens J, Deranian KL, Zhao M, Hayward NJ, Chong JA, Julius D, Moran MM, Fanger CM. TRPA1 mediates formalin-induced pain. Proc Natl Acad Sci U S A. 2007;104:13525–13530. doi: 10.1073/pnas.0705924104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris JB, Stanek J, Gianutsos G. Sensory nerve-mediated immediate nasal responses to inspired acrolein. J Appl Physiol. 1999;87:1877–1886. doi: 10.1152/jappl.1999.87.5.1877. [DOI] [PubMed] [Google Scholar]
- Morris JB, Symanowicz PT, Olsen JE, Thrall RS, Cloutier MM, Hubbard AK. Immediate sensory nerve-mediated respiratory responses to irritants in healthy and allergic airway-diseased mice. J Appl Physiol. 2003;94:1563–1571. doi: 10.1152/japplphysiol.00572.2002. [DOI] [PubMed] [Google Scholar]
- Morris JB, Wilkie WS, Shusterman DJ. Acute respiratory responses of the mouse to chlorine. Toxicol Sci. 2005;83:380–387. doi: 10.1093/toxsci/kfi038. [DOI] [PubMed] [Google Scholar]
- Nadeem A, Masood A, Siddiqui N. Oxidant--antioxidant imbalance in asthma: scientific evidence, epidemiological data and possible therapeutic options. Ther Adv Respir Dis. 2008;2:215–235. doi: 10.1177/1753465808094971. [DOI] [PubMed] [Google Scholar]
- Nassenstein C, Kwong K, Taylor-Clark T, Kollarik M, Macglashan DM, Braun A, Undem BJ. Expression and function of the ion channel TRPA1 in vagal afferent nerves innervating mouse lungs. J Physiol. 2008;586:1595–1604. doi: 10.1113/jphysiol.2007.148379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nassenstein C, Taylor-Clark TE, Myers AC, Ru F, Nandigama R, Bettner W, Undem BJ. Phenotypic distinctions between neural crest and placodal derived vagal C-fibres in mouse lungs. J Physiol. 2010;588:4769–4783. doi: 10.1113/jphysiol.2010.195339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- North ML, Amatullah H, Khanna N, Urch B, Grasemann H, Silverman F, Scott JA. Augmentation of arginase 1 expression by exposure to air pollution exacerbates the airways hyperresponsiveness in murine models of asthma. Respir Res. 2011;12:19. doi: 10.1186/1465-9921-12-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patapoutian A, Tate S, Woolf CJ. Transient receptor potential channels: targeting pain at the source. Nat Rev Drug Discov. 2009;8:55–68. doi: 10.1038/nrd2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polimanti R, Piacentini S, Moscatelli B, Pellicciotti L, Manfellotto D, Fuciarelli M. GSTA1, GSTO1 and GSTO2 gene polymorphisms in Italian asthma patients. Clin Exp Pharmacol Physiol. 2010;37:870–872. doi: 10.1111/j.1440-1681.2010.05385.x. [DOI] [PubMed] [Google Scholar]
- Pryor WA. How far does ozone penetrate into the pulmonary air/tissue boundary before it reacts? Free Radic Biol Med. 1992;12:83–88. doi: 10.1016/0891-5849(92)90060-t. [DOI] [PubMed] [Google Scholar]
- Reddy P, Naidoo RN, Robins TG, Mentz G, London SJ, Li H, Naidoo R. GSTM1, GSTP1, and NQO1 polymorphisms and susceptibility to atopy and airway hyperresponsiveness among South African school children. Lung. 2010;188:409–414. doi: 10.1007/s00408-010-9246-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts AM, Bhattacharya J, Schultz HD, Coleridge HM, Coleridge JC. Stimulation of pulmonary vagal afferent C-fibers by lung edema in dogs. Circ Res. 1986;58:512–522. doi: 10.1161/01.res.58.4.512. [DOI] [PubMed] [Google Scholar]
- Rouse RL, Murphy G, Boudreaux MJ, Paulsen DB, Penn AL. Soot nanoparticles promote biotransformation, oxidative stress, and inflammation in murine lungs. Am J Respir Cell Mol Biol. 2008;39:198–207. doi: 10.1165/rcmb.2008-0057OC. [DOI] [PubMed] [Google Scholar]
- Ruan T, Ho CY, Kou YR. Afferent vagal pathways mediating respiratory reflexes evoked by ROS in the lungs of anesthetized rats. J Appl Physiol. 2003;94:1987–1998. doi: 10.1152/japplphysiol.01047.2002. [DOI] [PubMed] [Google Scholar]
- Ruan T, Lin YS, Lin KS, Kou YR. Sensory transduction of pulmonary reactive oxygen species by capsaicin-sensitive vagal lung afferent fibres in rats. J Physiol. 2005;565:563–578. doi: 10.1113/jphysiol.2005.086181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan T, Lin YS, Lin KS, Kou YR. Mediator mechanisms involved in TRPV1 and P2X receptor-mediated, ROS-evoked bradypneic reflex in anesthetized rats. J Appl Physiol. 2006;101:644–654. doi: 10.1152/japplphysiol.00192.2006. [DOI] [PubMed] [Google Scholar]
- Salazar H, Llorente I, Jara-Oseguera A, Garcia-Villegas R, Munari M, Gordon SE, Islas LD, Rosenbaum T. A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. Nat Neurosci. 2008;11:255–261. doi: 10.1038/nn2056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawada Y, Hosokawa H, Matsumura K, Kobayashi S. Activation of transient receptor potential ankyrin 1 by hydrogen peroxide. Eur J Neurosci. 2008;27:1131–1142. doi: 10.1111/j.1460-9568.2008.06093.x. [DOI] [PubMed] [Google Scholar]
- Schelegle ES, Carl ML, Coleridge HM, Coleridge JC, Green JF. Contribution of vagal afferents to respiratory reflexes evoked by acute inhalation of ozone in dogs. J Appl Physiol. 1993;74:2338–2344. doi: 10.1152/jappl.1993.74.5.2338. [DOI] [PubMed] [Google Scholar]
- Schroer KT, Biagini Myers JM, Ryan PH, LeMasters GK, Bernstein DI, Villareal M, Lockey JE, Reponen T, Grinshpun S, Khurana Hershey GK. Associations between multiple environmental exposures and Glutathione S-Transferase P1 on persistent wheezing in a birth cohort. J Pediatr. 2009;154:401–408. 408, e401. doi: 10.1016/j.jpeds.2008.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaheen SO, Newson RB, Ring SM, Rose-Zerilli MJ, Holloway JW, Henderson AJ. Prenatal and infant acetaminophen exposure, antioxidant gene polymorphisms, and childhood asthma. J Allergy Clin Immunol. 2010;126:1141–1148. e1147. doi: 10.1016/j.jaci.2010.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin J, Cho H, Hwang SW, Jung J, Shin CY, Lee SY, Kim SH, Lee MG, Choi YH, Kim J, Haber NA, Reichling DB, Khasar S, Levine JD, Oh U. Bradykinin-12-lipoxygenase-VR1 signaling pathway for inflammatory hyperalgesia. Proc Natl Acad Sci U S A. 2002;99:10150–10155. doi: 10.1073/pnas.152002699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shusterman DJ, Murphy MA, Balmes JR. Subjects with seasonal allergic rhinitis and nonrhinitic subjects react differentially to nasal provocation with chlorine gas. J Allergy Clin Immunol. 1998;101:732–740. doi: 10.1016/S0091-6749(98)70302-1. [DOI] [PubMed] [Google Scholar]
- Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, Earley TJ, Hergarden AC, Andersson DA, Hwang SW, McIntyre P, Jegla T, Bevan S, Patapoutian A. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell. 2003;112:819–829. doi: 10.1016/s0092-8674(03)00158-2. [DOI] [PubMed] [Google Scholar]
- Susankova K, Tousova K, Vyklicky L, Teisinger J, Vlachova V. Reducing and oxidizing agents sensitize heat-activated vanilloid receptor (TRPV1) current. Mol Pharmacol. 2006;70:383–394. doi: 10.1124/mol.106.023069. [DOI] [PubMed] [Google Scholar]
- Takahashi N, Mizuno Y, Kozai D, Yamamoto S, Kiyonaka S, Shibata T, Uchida K, Mori Y. Molecular characterization of TRPA1 channel activation by cysteine-reactive inflammatory mediators. Channels (Austin) 2008;2:287–298. doi: 10.4161/chan.2.4.6745. [DOI] [PubMed] [Google Scholar]
- Tall JM, Seeram NP, Zhao C, Nair MG, Meyer RA, Raja SN. Tart cherry anthocyanins suppress inflammation-induced pain behavior in rat. Behav Brain Res. 2004;153:181–188. doi: 10.1016/j.bbr.2003.11.011. [DOI] [PubMed] [Google Scholar]
- Taylor-Clark T, Undem BJ. Transduction mechanisms in airway sensory nerves. J Appl Physiol. 2006;101:950–959. doi: 10.1152/japplphysiol.00222.2006. [DOI] [PubMed] [Google Scholar]
- Taylor-Clark TE, Kiros F, Carr MJ, McAlexander MA. Transient receptor potential ankyrin 1 mediates toluene diisocyanate-evoked respiratory irritation. Am J Respir Cell Mol Biol. 2009;40:756–762. doi: 10.1165/rcmb.2008-0292OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Clark TE, McAlexander MA, Nassenstein C, Sheardown SA, Wilson S, Thornton J, Carr MJ, Undem BJ. Relative contributions of TRPA1 and TRPV1 channels in the activation of vagal bronchopulmonary C-fibres by the endogenous autacoid 4-oxononenal. J Physiol. 2008a;586:3447–3459. doi: 10.1113/jphysiol.2008.153585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Clark TE, Nassenstein C, Undem BJ. Leukotriene D(4) increases the excitability of capsaicin-sensitive nasal sensory nerves to electrical and chemical stimuli. Br J Pharmacol. 2008b;154:1359–1368. doi: 10.1038/bjp.2008.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Clark TE, Undem BJ. Ozone activates airway nerves via the selective stimulation of TRPA1 ion channels. J Physiol. 2010;588:423–433. doi: 10.1113/jphysiol.2009.183301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Clark TE, Undem BJ, Macglashan DW, Jr, Ghatta S, Carr MJ, McAlexander MA. Prostaglandin-Induced Activation of Nociceptive Neurons via Direct Interaction with Transient Receptor Potential A1 (TRPA1) Mol Pharmacol. 2008c;73:274–281. doi: 10.1124/mol.107.040832. [DOI] [PubMed] [Google Scholar]
- Trevisani M, Siemens J, Materazzi S, Bautista DM, Nassini R, Campi B, Imamachi N, Andre E, Patacchini R, Cottrell GS, Gatti R, Basbaum AI, Bunnett NW, Julius D, Geppetti P. 4-Hydroxynonenal, an endogenous aldehyde, causes pain and neurogenic inflammation through activation of the irritant receptor TRPA1. Proc Natl Acad Sci U S A. 2007;104:13519–13524. doi: 10.1073/pnas.0705923104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Undem BJ, Chuaychoo B, Lee MG, Weinreich D, Myers AC, Kollarik M. Subtypes of vagal afferent C-fibres in guinea-pig lungs. J Physiol. 2004;556:905–917. doi: 10.1113/jphysiol.2003.060079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Undem BJ, Kollarik M. Characterization of the vanilloid receptor 1 antagonist iodo-resiniferatoxin on the afferent and efferent function of vagal sensory C-fibers. J Pharmacol Exp Ther. 2002;303:716–722. doi: 10.1124/jpet.102.039727. [DOI] [PubMed] [Google Scholar]
- Uysal N, Schapira RM. Effects of ozone on lung function and lung diseases. Curr Opin Pulm Med. 2003;9:144–150. doi: 10.1097/00063198-200303000-00009. [DOI] [PubMed] [Google Scholar]
- Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44–84. doi: 10.1016/j.biocel.2006.07.001. [DOI] [PubMed] [Google Scholar]
- White CW, Martin JG. Chlorine gas inhalation: human clinical evidence of toxicity and experience in animal models. Proc Am Thorac Soc. 2010;7:257–263. doi: 10.1513/pats.201001-008SM. [DOI] [PMC free article] [PubMed] [Google Scholar]