Abstract
Partitioning ectoderm precisely into neurogenic and non-neurogenic regions is an essential step for neurogenesis of almost all bilaterian embryos. Although it is widely accepted that antagonism between BMP and its inhibitors primarily sets up the border between these two types of ectoderm, it is unclear how such extracellular, diffusible molecules create a sharp and precise border at the single-cell level. Here, we show that Fez, a zinc finger protein, functions as an intracellular factor attenuating BMP signaling specifically within the neurogenic region at the anterior end of sea urchin embryos, termed the animal plate. When Fez function is blocked, the size of this neurogenic ectoderm becomes smaller than normal. However, this reduction is rescued in Fez morphants simply by blocking BMP2/4 translation, indicating that Fez maintains the size of the animal plate by attenuating BMP2/4 function. Consistent with this, the gradient of BMP activity along the aboral side of the animal plate, as measured by pSmad1/5/8 levels, drops significantly in cells expressing Fez and this steep decline requires Fez function. Our data reveal that this neurogenic ectoderm produces an intrinsic system that attenuates BMP signaling to ensure the establishment of a stable, well-defined neural territory, the animal plate.
Keywords: BMP, Cell fate specification, Body axis, Neurogenesis
INTRODUCTION
Nervous system development in bilaterians is composed of a number of developmental processes, such as partitioning of the ectoderm into neuroectoderm and non-neuroectoderm, patterning of the neuroectoderm, and differentiation of neural cells including neural network formation. Among these, partitioning of ectoderm is one of the earliest and the most crucial events because all of the following processes of neurogenesis depend on it. In vertebrates, the prospective neuroectoderm is formed at the future dorsal side by the function of the Spemann organizer, which can induce an ectopic neuroectoderm when grafted to the ventral region of the embryo (Spemann and Mangold, 1924). From a number of previous studies analyzing the inductive function of the organizer, we now know that the molecular entities mediating this induction are diffusible extracellular molecules, such as Chordin, Noggin and Follistatin (De Robertis and Kuroda, 2004). These molecules antagonize the function of bone morphogenetic proteins (BMPs), members of TGF-β superfamily, on the dorsal side thereby protecting this region from BMP-mediated induction of non-neuroectoderm (Khokha et al., 2005). When one or two of them are knocked down, the neuroectoderm is normally partitioned and the morphants have normal neural cell differentiation. However, when the expression of all three is knocked down, the embryo contains no neuroectoderm because of enhanced and expanded BMP signaling. Conversely, when the translation of three BMP members (BMP2, BMP4 and BMP7) is blocked, the embryo has expanded neuroectoderm (Reversade et al., 2005). Therefore, it is widely accepted that protection against BMP activity is the mechanism that specifies the dorsal region as neuroectoderm during early development in vertebrates (De Robertis and Kuroda, 2004). However, BMPs are diffusible extracellular molecules that form an activity gradient that is reinforced by positive feedback (Shen et al., 2007), such that a small initial change can have a large influence on the effective signaling range (Gardner et al., 2000). Therefore, it has been unclear how BMP activity can be precisely controlled to partition ectoderm fates at a single-cell level. Because the size of the neuroectoderm is identical in individual embryos of the same species at the same developmental stage, there must be additional intrinsic mechanism(s) that downregulate BMP signaling and establish precise domains of neural and non-neural ectoderm.
The neurogenic ectoderm of the embryo of the sea urchin, a basal deuterostome, consists of two major parts. One is the ectoderm at the animal pole, which is highly related to the vertebrate anterior neuroectoderm, judged from the temporal and spatial expression patterns of genes encoding regulatory factors, and from the fact that canonical Wnt signaling is involved in positioning the region anteriorly during cell-fate specification during early embryogenesis (Wikramanayake et al., 1998; Kiecker and Niehrs, 2001; Wei et al., 2009). The second forms within the ciliary band located at the border between oral and aboral signaling territories (Duboc et al., 2004; Yaguchi et al., 2006; Wei et al., 2009; Lapraz et al., 2009; Yaguchi et al., 2010a; Saudemont et al., 2010). The anterior neurogenic region, called the animal plate, is the focus of the present study. FoxQ2 is among several genes that are the earliest to be expressed specifically in neurogenic ectoderm. FoxQ2 transcripts appear in the entire animal half at the 32-cell stage and are progressively restricted to the animal pole region in pre-hatching blastulae (Tu et al., 2006; Yaguchi et al., 2008). This process restricts at least several genes required for animal plate development and depends on the Wnt/β-catenin pathway, which functions in the vegetal hemisphere (Logan et al., 1999; Yaguchi et al., 2008). After the restriction of the FoxQ2-positive region is completed at the blastula stage, the prospective neuroectoderm starts to express additional genes that are specific for nervous system development (Wei et al., 2009). However, at around this stage, nodal and bmp2/4, which are common anti-neural factors during early development, are transcribed on the oral side and then BMP2/4 diffuses to the aboral ectoderm. Both Nodal and BMP2/4 induce non-neurogenic ectoderm fates (Angerer et al., 2000; Duboc et al., 2004; Bradham et al., 2009; Lapraz et al., 2009; Saudemont et al., 2010). The prospective animal plate, as well as ciliary band, neuroectoderm must have robust mechanisms that antagonize these signals and maintain their precise sizes during these stages. Here, we report that, in the animal plate, FoxQ2 induces production of an intracellular transcription factor, Fez (forebrain embryonic zinc finger), at the blastula stage that specifically attenuates BMP signaling, thereby maintaining and stabilizing the border of the anterior neuroectoderm in the sea urchin embryo.
MATERIALS AND METHODS
Animals and embryo culture
Adult sea urchins, Strongylocentrotus purpuratus, were obtained from The Cultured Abalone (Goleta, CA, USA) and the embryos were used only for the microarray experiment. For all other experiments, embryos of Hemicentrotus pulcherrimus collected around the Shimoda Marine Research Center (University of Tsukuba, Shizuoka, Japan) and around the Marine and Coastal Research Center (Ochanomizu University, Chiba, Japan) were used. The gametes were collected by intrablastocoelar injection of 0.5 M KCl and the embryos were cultured with filtered natural seawater (FSW) at 15°C. For some experiments, γ-secretase inhibitor (DAPT; Sigma-Aldrich, St Louis, MO, USA) was added at the hatching blastula stage to a final concentration of 20 γM.
Microarray
Sample preparation, microarray and data processing were described previously (Wei et al., 2006). The double-strand cDNAs synthesized from control embryos and SpFoxQ2 morphants were labeled, hybridized and scanned by Roche Nimblegen microarray services. The microarray was prepared based on S. purpuratus genome sequence information (Wei et al., 2006; Sea Urchin Genome Sequencing Consortium, 2006) and hybridized with amplified cDNAs isolated from S. purpuratus mesenchyme blastulae either containing or lacking the SpFoxQ2-MO1 previously described (Yaguchi et al., 2008) (see below). FoxQ2-dependent genes identified in the microarray were confirmed by in situ hybridization in H. pulcherrimus embryos, as described below and by quantitative polymerase chain reaction (QPCR) as previously described (Yaguchi et al., 2010b).
Microinjection of morpholino antisense oligonucleotides (MO) and synthetic mRNAs
Microinjection was performed as described previously (Yaguchi et al., 2010b). We used the following morpholinos (Gene Tools, Philomath, OR, USA) at the indicated concentrations in 24% glycerol in injection needles: SpFoxQ2-MO1 (800 γM) (Yaguchi et al., 2008), FoxQ2-MO (200 γM) (Yaguchi et al., 2010b), Fez-MO1 (1.9 mM), Fez-MO2 (1.6 mM), BMP2/4-MO (400 γM) and Smad1/5/8-MO (1.0 mM). All morpholinos were designed against H. pulcherrimus genes except for the SpFoxQ2-MO1 that blocks S. purpuratus FoxQ2 translation. The morphant phenotypes were the same as those previously published (Duboc et al., 2004; Yaguchi et al., 2008). The morpholino sequences were the following: Fez-MO1: 5′-GAATGCTTTTTTCATGCACTAAAGA-3′; Fez-MO2: 5′-GCGTTCAAATCTACTTAAGGAGTGT-3′; BMP2/4-MO: 5′-GACCCCAATGTGAGGTGGTAACCAT-3′; and Smad1/5/8-MO: 5′-GCCATCGACATAGTGGAGCAAGCTT-3′.
mRNAs were synthesized from linearized plasmids using the mMessage mMachine kit (Life Technologies, Carlsbad, CA, USA) and injected at the indicated concentrations in 24% glycerol in injection needles: Δ-cadherin (0.3-0.6 γg/γl) (Logan et al., 1999), Fez-mRNA (2.0 γg/γl), BMP2/4-mRNA (2.5 γg/γl) and actSmad2/3-mRNA (2.5 γg/γl) (Yaguchi et al., 2007).
Whole-mount in situ hybridization and immunohistochemistry
Whole-mount in situ hybridization was performed as described previously (Minokawa et al., 2004; Yaguchi et al., 2010b). The foxQ2-positive cell number was determined by counting DAPI signals in the foxQ2 region through serial optical sections. Immunohistochemistry for detecting serotonin, synaptotagminB (synB) and Nk2.1 was performed as described previously (Yaguchi et al., 2006). To detect FoxQ2 and pSmad1/5/8 immunochemically, embryos were fixed with 4% paraformaldehyde in FSW for 10 minutes. After washing with phosphate-buffered saline containing 0.5% Tween-20 (PBS-T) five times, the embryos were blocked with 1% skimmed milk (Difco; BD, Franklin Lakes, NJ, USA) in PBS-T and incubated with primary antibodies diluted as follows with Can Get Signal Immunostain Solution B (TOYOBO, Tokyo, Japan): mouse anti-FoxQ2 IgG at 1:100, rabbit anti-pSmad1/5/8 (9511; Cell Signaling Technology, Danvers, MA, USA) at 1:1000. The primary antibodies were detected with secondary antibodies conjugated with Alexa-568 for FoxQ2 and Alexa-488 for pSmad1/5/8 (Life Technologies). The specimens were observed with a Zeiss Axio Imager.Z1 equipped with Apotome system, and optical sections were stacked and analyzed using ImageJ and Adobe Photoshop. Panels and drawings for figures were made with CANVAS software.
Anti-FoxQ2 antibody preparation
cDNA encoding full-length FoxQ2 was cloned into the pET vector (Novagen, Darmstadt, Germany) and histidine-tagged FoxQ2 was induced in bacteria with IPTG. The bacteria were lysed with 6 M urea, and the fusion protein was purified by Ni-column chromatography and used to immunize three mice. Antisera were screened by whole-mount immunohistochemistry, and IgG was purified with Melon gel (Thermo Fisher Scientific, Waltham, MA, USA).
RESULTS
Fez expression depends strongly on FoxQ2 during blastula stages
FoxQ2 is expressed very early in the prospective animal plate of sea urchin embryos and is required for the development of neurons in this region of the embryonic nervous system. We searched for regulatory genes downstream of FoxQ2 that are required for formation of the animal plate using a microarray approach to identify genes strongly downregulated in FoxQ2 morphants at the blastula stage of Strongylocentrotus purpuratus. Previous studies have shown this approach to be successful in identifying genes whose expression depends on upstream regulatory proteins (Wei et al., 2009; Yaguchi et al., 2010b). One candidate FoxQ2-dependent regulatory gene encoded an apparent ortholog of Fez, forebrain embryonic zinc finger, (SPU_027491; Sea Urchin Genome Sequencing Consortium, 2006). We recovered this gene from the genome of the Japanese sea urchin, Hemicentrotus pulcherrimus, and found that it contains sequences encoding an engrailed homology repressor motif 1 (EH1) at the N-terminal region and six zinc finger domains at the C-terminus (accession number: AB610478), as previously reported for vertebrate Fez or Fez-like proteins (e.g. mice; AK014242 and AB042399) (Hirata et al., 2006b). Because of the presence of EH1, Fez is thought to function as a transcriptional repressor (Hirata et al., 2006a). After confirming the microarray result that, during blastula stages, H. pulcherrimus FoxQ2 morphants have significantly decreased fez mRNA levels by in situ hybridization and QPCR (Fig. 1M-O), we focused on the function of Fez in neurogenesis.
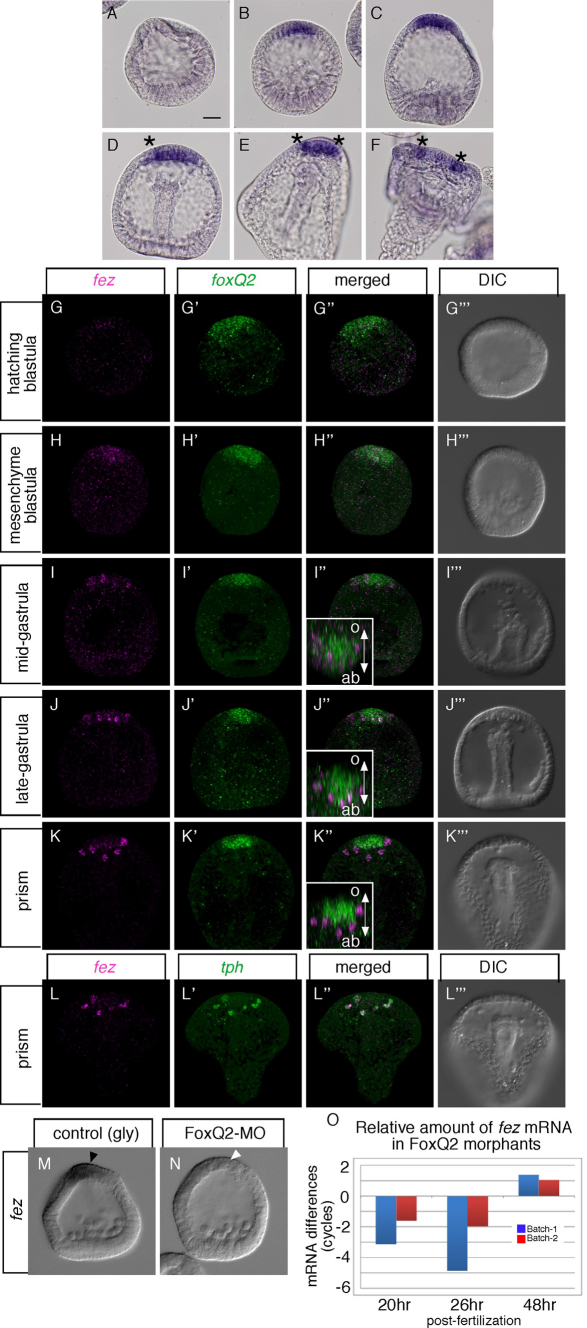
Fig. 1.
fez is expressed throughout the animal plate during the hatched and mesenchyme blastula stages and later in the serotonergic neurons. (A-F) Expression of fez in Hemicentrotus pulcherrimus at the unhatched blastula (A), mesenchyme blastula (B), early gastrula (C), mid-late gastrula (D), prism larva (E) and pluteus (F) stages. Asterisks in D-F show the individually stained cells. (G-H′′) fez is co-expressed in the animal plate with foxQ2 at blastula stages. (I-K′′) By contrast, at gastrula and prism stages, fez is expressed in individual cells that do not express foxQ2. Insets in I′, J′ and K′ show animal pole views of each image digitally rotated to show the asymmetry of foxQ2 and fez patterns along the secondary axis (o and ab show oral and aboral side, respectively). (L-L′′) The pattern of later expression of fez is identical to that of tph, which marks serotonergic neurons. (M-O) fez expression depends on FoxQ2. (M) fez is expressed in the animal plate region of control (gly; glycerol-injected) mesenchyme blastula-stage embryos (black arrowhead). (N) FoxQ2 morphant (FoxQ2-MO) lacks fez expression (white arrowhead). (O) Quantitative PCR (QPCR) shows that fez expression depends on FoxQ2 at early stages (20 hours, hatching-mesenchyme blastula; 26 hours, gastrula), but not at a later stage (48 hours; pluteus). x-axis shows time after fertilization. y-axis shows ΔΔCt, the cycle differences after normalization for load between experimental and control embryos (one negative cycle difference means that the mRNA concentration is reduced twofold). QPCR was performed by following the method described previously (Yaguchi et al., 2010b). Scale bar: 20 γm.
Expression of fez during development
To examine the gene expression pattern of fez, we performed in situ hybridization at developmental stages from egg to pluteus larva. No signals are detected before hatching (Fig. 1A), consistent with the temporal profiling data conducted by Wei et al. (Wei et al., 2006) using microarrays. After hatching, fez transcripts accumulate throughout the entire animal plate (Fig, 1B) until the early gastrula stage (Fig. 1C). After the embryo reaches the mid-gastrula stage, the uniform expression fades and is progressively replaced by stronger signals in a few individual cells within the animal plate (Fig. 1D-F, asterisks). To compare fez and foxQ2 expression patterns in detail, we employed fluorescent in situ hybridization. fez is expressed at low levels in the animal plate at the hatching blastula stage (Fig. 1G) but is expressed at much higher levels at the mesenchyme blastula stage and in a pattern that is almost identical to that of foxQ2 (Fig. 1H). As shown using chromogenic detection (Fig. 1D), fez transcripts accumulate in individual cells at the gastrula stage, whereas foxQ2 expression remains uniform throughout the animal plate (Fig. 1I,J). The expression domain of foxQ2 is restricted to the oral side of the animal plate at later stages (Yaguchi et al., 2008) whereas three to five individual fez-positive cells are located on the opposite (aboral) side at that time (Fig. 1K; insets in Fig. 1I′,J′,K′). To examine which cell type(s) express fez mRNA, we compared the distributions of tryptophan 5-hydroxylase (tph) and fez mRNAs by double fluorescent in situ hybridization. Because TPH is the rate-limiting enzyme in serotonin synthesis, it is a specific marker for serotonergic neurons in sea urchin embryos (Yaguchi and Katow, 2003). At the prism stage, fez is clearly expressed in the serotonergic neurons, although each mRNA localizes to a slightly different position in the same cell (Fig. 1L). Because the expressions of fez and foxQ2 do not overlap at later stages, fez expression does not depend on FoxQ2 at that time (Fig. 1O). In summary, fez is expressed throughout the animal plate between the hatching blastula and gastrula stages, and in serotonergic neural precursors and neurons after gastrulation.
Fez controls the size of the animal plate
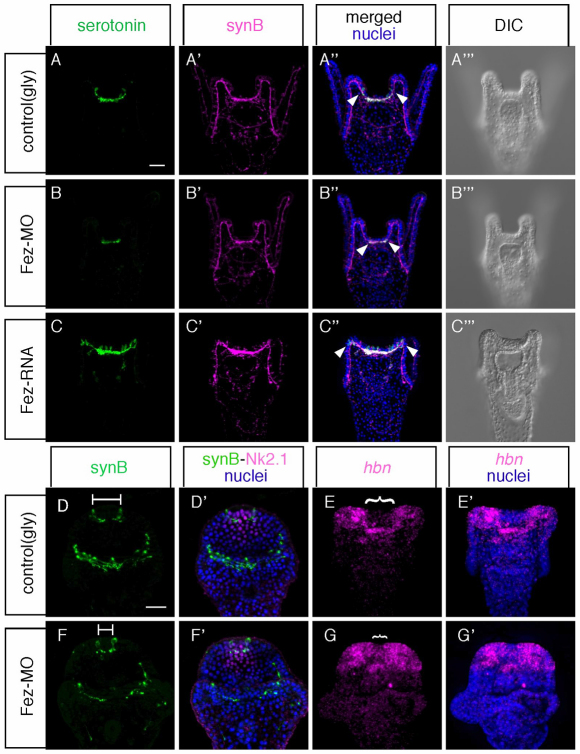
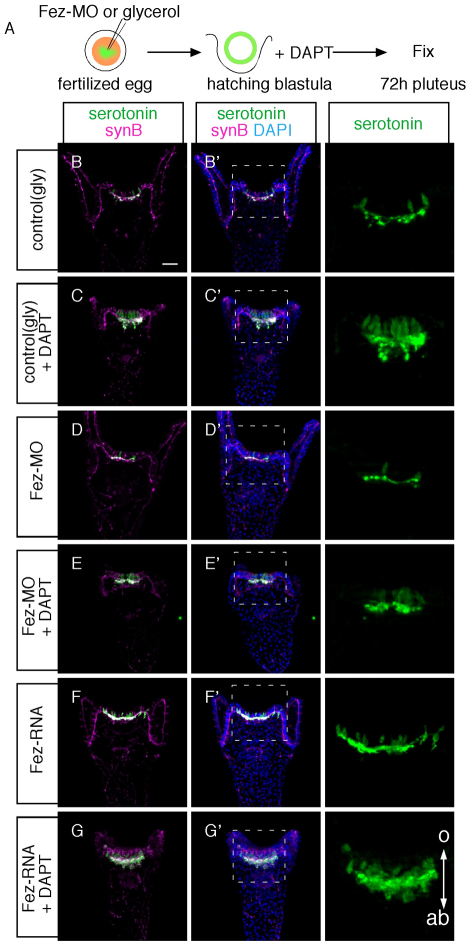
To examine Fez function in sea urchin development and neurogenesis, we performed knockdown and mis-expression experiments by injecting morpholino anti-sense oligonucleotides (MOs) and mRNA encoding full-length Fez protein, respectively. In Fez morphants, the timing of cleavage and invagination is slightly delayed but otherwise no morphological defects are detectable (data not shown). However, Fez morphants have fewer serotonergic neurons than do normal embryos and the region in which they differentiate is smaller, being restricted to an area between the bases of anterolateral arms at the 72-hour pluteus stage (Fig. 2B). By contrast, in normal embryos, the left and right borders of the serotonergic neural complex extend up to the middle of those arms (Fig. 2A). This phenotype is specific because it was also obtained with a second morpholino targeted to a non-overlapping sequence in the mRNA (see Fig. S1 in the supplementary material). Because the cell bodies and axon network of synaptotagminB-positive, non-serotonergic neurons beneath the ciliary band in Fez morphants are almost identical to those of normal embryos (compare Fig. 2B′ with 2A′) (Nakajima et al., 2004; Burke et al., 2006), the loss of Fez function probably affects only the serotonergic neurons on the aboral side of the animal plate. There are two possibilities for the decreased number of serotonergic neurons in Fez morphants. One is that the animal plate itself becomes smaller than that of normal embryos, and another is that the size of animal plate is the same but the differentiation of serotonergic neurons is perturbed at the periphery of this region. To test this, we focused on the position of the first pair of serotonergic neurons that develop near the aboral-lateral margins of the animal plate, which are marked with Nk2.1 expression (Fig. 2D,F) (Yaguchi et al., 2000; Yaguchi et al., 2006). Although Fez morphants have fewer serotonergic neurons than do control embryos at later stages (Fig. 2A,B), the timing of the differentiation of the first pair of serotonergic neurons is the same. In Fez morphants, these serotonergic neurons are closer to each other than those in normal embryo are, and there are fewer Nk2.1-positive cells between them (Fig. 2D′,F′). Here, the serotonergic neurons are stained with anti-SynB antibody because at this stage all SynB neurons are serotonergic. In addition, the expression of homeobrain (hbn) suggests that the animal plate is smaller. hbn expression is cleared from the animal plate and surrounds it at the gastrula and pluteus stages (Wei et al., 2009). However, the central region of clearance is smaller in Fez morphants than in controls (compare Fig. 2G with 2E). Moreover, treatment of embryos with the γ-secretase inhibitor DAPT, which suppresses Notch signaling, supports these results. In sea urchin embryos, delta-Notch signaling suppresses differentiation of serotonergic neurons through lateral inhibition (Wei et al., 2011). Thus, if Notch signaling is blocked, additional neural precursors can differentiate and contribute to a cluster of serotonergic neurons. If the primary function of Fez, the early expression of which is in all cells of the animal plate, is to support the differentiation of each cell as a neuron, then the additional neurons that develop in DAPT-treated embryos (Fig. 3C) would not appear in Fez morphants. However, this is not the case, because the number of neurons increases and they form a cluster in DAPT-treated embryos, regardless of whether Fez is present or absent (Fig. 3A-E). This indicates that Fez is not required just for the differentiation of serotonergic neurons, but rather for determining the size of the neurogenic ectoderm. Furthermore, mis-expression of Fez can significantly increase the size of the animal plate and the number of serotonergic neurons, which extend all the way to the tip of anterolateral arms (Fig. 2C, Fig. 3F,G). In addition, because the size of the neural cluster along the oral-aboral axis in DAPT-treated fez mRNA-injected embryos is wider than that in control embryos, Fez mis-expression can expand the animal plate along both the ciliary band and the aboral ectoderm.
Fig. 2.
The animal plate size is smaller in Fez morphants (Fez-MO). (A-C′′) Serotonergic neurons and synaptotagminB (synB)-positive neurons in 72-hour-old control embryos (A-A′′), 72-hour-old Fez morphants (B-B′′) and 72-hour-old Fez mRNA-injected embryos (C-C′′). (D,D′,F,F′) SynB-neurons and Nk2.1, prism stage in control (D,D′) and in Fez morphant (Fez-MO; F,F′). All of the syn-B positive neurons at the animal plate are serotonergic at this stage. Lines in D and F show the distance between two neurons. (E,E′,G,G′) hbn expression pattern in control (E) and in Fez morphant (G). Brackets in E and G show the size of neurogenic ectoderm. Scale bars: in A, 20 γm for A-C′′; in D, 20 γm for D-G′.
Fig. 3.
Fez is not involved in the differentiation of serotonergic neurons. (A) Experimental flow to investigate whether Fez functions in neural differentiation. Orange and green show the intact egg cytoplasm and the reagent that we injected, respectively. (B-G′) Serotonergic neurons and synaptotagminB (synB)-positive neurons in pluteus larvae subjected to the following treatments: glycerol alone (B), DAPT (C), Fez morpholino (D), DAPT and Fez morpholino (E), Fez mRNA injection (F), and DAPT and Fez mRNA injection (G). The anterior regions of embryos indicated by the boxed area in the middle panels are magnified in the right-hand panels. A double arrow shows the direction of oral (o)-aboral (ab) axis. Scale bar: 20 γm.
Fez maintains the neurogenic ectoderm during blastula stages
The loss-of-function experiments suggest that Fez is required to maintain the size of the neurogenic animal plate in the sea urchin embryo. To investigate when Fez executes this function, we examined the expression patterns of foxQ2 and its downstream gene, nk2.1. fez begins to be expressed between the hatching and mesenchyme blastula stages in control embryos (Fig. 1), and the size of the animal plate, marked by foxQ2, is almost identical at both stages in control embryos (Fig. 4A,B). The FoxQ2-dependent gene nk2.1 begins to be expressed at the mesenchyme blastula stage in a similarly sized region of the neurogenic ectoderm (Fig. 4C). By contrast, in Fez morphants, the size of the foxQ2-expressing domain at the mesenchyme blastula stage becomes smaller than that observed at the hatching blastula stage (Fig. 4D,E), and significantly smaller than that observed in control embryos at the mesenchyme blastula stage (compare Fig. 4E with 4B). As well, the size of the nk2.1-expressing region decreases (Fig. 4F). Taken together, these data indicate that Fez is required to maintain the precise size of the neurogenic animal plate as soon as it is expressed between the hatching and mesenchyme blastula stages.
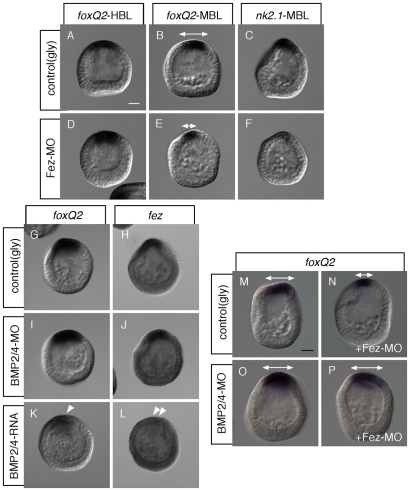
Fig. 4.
Fez attenuates BMP signaling, which antagonizes neurogenic ectoderm development. (A-F) Fez is required to maintain the size of the region expressing foxQ2 and nk2.1. (A) foxQ2 mRNA expression in control hatching blastula (HBL). (B) foxQ2 mRNA expression in control mesenchyme blastula (MBL). (C) nk2.1 mRNA expression in control MBL. (D) foxQ2 mRNA expression in Fez morphant HBL. (E) foxQ2 mRNA expression in Fez morphant MBL. (F) nk2.1 mRNA expression in Fez morphant MBL. (G-J) BMP2/4 does not suppress foxQ2 or fez expression outside the animal plate of MBL. (G) foxQ2 mRNA expression in control. (H) fez mRNA expression in control. (I) foxQ2 mRNA expression in BMP2/4 morphant. (J) fez mRNA expression in BMP2/4 morphant. (K,L) Mis-expressed BMP2/4 can suppress foxQ2 (K) and fez (L) expression. Single and double arrowheads show that in embryos over-expressing BMP2/4, both foxQ2 and fez expression is strongly reduced. (M-P) The foxQ2 expression domain is reduced in Fez morphants (N) versus controls (M), but not in double Fez/BMP2/4 morphant MBL (P) versus BMP2/4 morphant MBL (O). Double-headed arrows in B, E and M-P show foxQ2-positive animal plate. Scale bars: in A, 20 γm for A-F,I-N; in O, 20 γm for O-R.
BMP2/4 can restrict the neurogenic ectoderm
Fez could regulate the size of the animal plate by supporting positive regulators of this domain or by inhibiting negative regulators such as BMP signaling, which suppresses development of other neuroectoderm territories in the sea urchin embryo (Lapraz et al., 2009; Saudemont et al., 2010; Yaguchi et al., 2010a). To investigate the first alternative, we employed Δcadherin (Δcad) mRNA-injected embryos that lack Wnt/β-catenin and TGF-β signals, and develop a greatly expanded animal plate and flanking region (Logan et al., 1999; Duboc et al., 2004; Yaguchi et al., 2006; Wei et al., 2009). In these embryos, fez is expressed throughout the expanded animal plate (see Fig. S2 in the supplementary material). When Fez translation is blocked in Δcad mRNA-injected embryos with a Fez morpholino, foxQ2 expression still covers half or more of the embryo, and its distribution is not distinguishable from that of Δcad only-injected embryos (see Fig. S2 in the supplementary material). This indicates that Fez does not function as an essential feedback inducer of foxQ2, an upstream regulator for animal plate development, and suggests the that it functions instead as an inhibitor of animal plate-repressing signal(s).
To test whether BMP signaling might be the postulated signal that suppresses animal plate development and that is suppressed by Fez, we examined the effects on the expression of foxQ2 and fez of knocking down or mis-expressing Fez throughout the embryo. As shown in Fig. 4, loss of BMP2/4 does not expand the animal plate or significantly alter the expression of these two genes (Fig. 4G-J), but mis-expression strongly suppresses the expression of both (Fig. 4K,L, arrowhead and double arrowheads). We then asked whether the reduction of the animal plate in Fez morphants, as marked by foxQ2 expression, requires BMP2/4. To test this, we injected Fez and BMP2/4 morpholinos simultaneously. In Fez morphants in which BMP signaling is normal, the foxQ2 domain is reduced compared with control embryos (Fig. 4M,N), but this is not the case in a double Fez-BMP2/4 morphant (Fig. 4O,P), indicating that in the absence of Fez, BMP signaling changes the size of the animal plate.
Fez protects neurogenic ectoderm against BMP signaling
Although the data strongly suggest that it is BMP2/4 signaling that reduces the animal plate in the absence of Fez, they do not rigorously rule out the possibility that, in the context of Fez-BMP2/4 double morphants, ectopic Nodal, which diffuses to the aboral side of the animal plate (Yaguchi et al., 2010a) might affect its properties or interfere with some other signal that antagonizes the animal plate. To eliminate ectopic Nodal signaling in double morphants from regions where BMP2/4 normally signals, we used an experimental embryo described previously (Yaguchi et al., 2007), in which Nodal signaling is restricted to one side of the embryo.
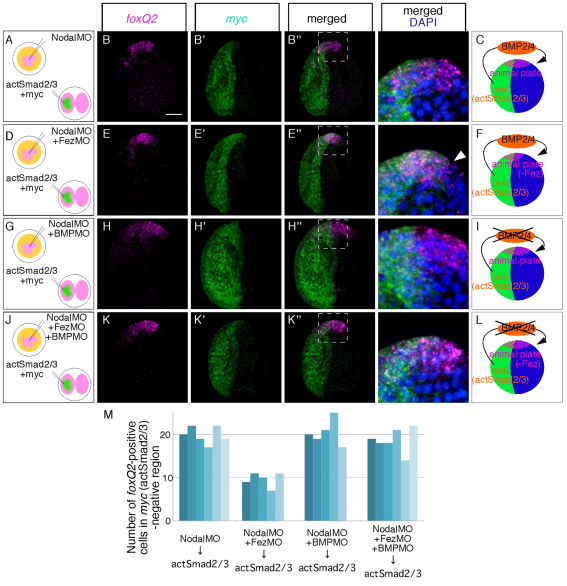
Endogenous Nodal is eliminated with a morpholino injection at the one-cell stage, and Nodal signaling is supplied on only one side of the embryo by injecting into one blastomere at the two-cell stage mRNA encoding constitutively active Smad2/3 (actSmad2/3) along with mRNA encoding a myc marker. This treatment forces oral and aboral fates on the injected and non-injected sides of the embryo, respectively, because actSmad2/3 activates the oral program that produces BMP2/4 (see Fig. S3 in the supplementary material), which diffuses to and signals to cells on the aboral side and activates the aboral ectoderm program (Yaguchi et al., 2007) (Fig. 5A-C). In this embryo, the number of foxQ2-positive cells in the aboral half is about 20 (Fig. 5A-C,M; see Materials and methods), but this number is cut approximately in half in Fez morphants (Fig. 5D-F,M), showing a similar difference to that observed between normal and Fez morphant embryos. However, this reduction did not occur in the experimental embryo (doubly injected with Nodal morpholino and actSmad2/3) in which BMP2/4 is also knocked down. The size of neurogenic ectoderm is unchanged in those embryos, whether or not Fez protein is present or absent (Fig. 5G-M). These data indicate that the signal that Fez blocks in the animal plate is BMP2/4. Thus, FoxQ2 causes production of Fez, which attenuates BMP2/4 signaling in the aboral animal plate ectoderm.
Fig. 5.
BMP2/4, not Nodal, reduces the neurogenic ectoderm size in the absence of Fez in mesenchyme blastulae (MBL). (A) Protocol producing control experimental embryos in which Nodal signaling is restricted to one half of the embryo by injection of activated Smad2/3 mRNA into one of the two-cell embryo blastomeres. Orange, magenta and green show the intact egg cytoplasm, the reagent that we injected into the fertilized egg and the reagent that we injected into one blastomere of a two-cell stage egg, respectively. (B-B′) foxQ2 and myc mRNA expression in embryos produced using the method shown in A. Merged and magnified image of boxed region in B′ is shown in the right-hand panel with DAPI. (C) Drawing showing the ectodermal partitioning of an experimental control embryo. Green, blue and magenta show oral, aboral and animal plate ectoderm, respectively. bmp2/4, transcribed in oral ectoderm in response to activated Smad2/3, produces BMP2/4 which diffuses to the aboral side. (D) Protocol producing experimental embryos lacking Fez function. (E-E′) foxQ2 and myc expression in embryos produced using the method shown in D. Merged and magnified image of boxed region in E′ is shown in the right-hand panel with DAPI. Arrowhead shows the site where foxQ2 expression is lost. (F) Drawing showing the ectodermal partitioning of an experimental embryo in which Fez translation is blocked, which causes reduction of the animal plate on the side of the embryo where BMP2/4 signals. (G) Protocol producing experimental embryos lacking BMP2/4 function. (H-H′) foxQ2 and myc expression in embryos produced using the method shown in G. Merged and magnified image of boxed region in H′ is shown in the right-hand panel with DAPI. (I) Drawing showing the ectodermal partitioning of an experimental embryo, in which BMP2/4 translation is blocked. (J) Protocol producing experimental embryos lacking BMP2/4 and Fez function. (K-K′) foxQ2 and myc expression in embryos produced using the method shown in J. Merged and magnified image of boxed region in K′ is shown in the right-hand panel with DAPI. (L) Drawing showing the ectodermal partitioning of an experimental embryo, in which BMP2/4 and Fez translation is blocked. (M) Histogram showing the number of foxQ2-positive cells in the myc- and Nodal signaling-negative side of experimental embryos. Counts were made from specimens from two independent batches. Scale bar: 20 γm.
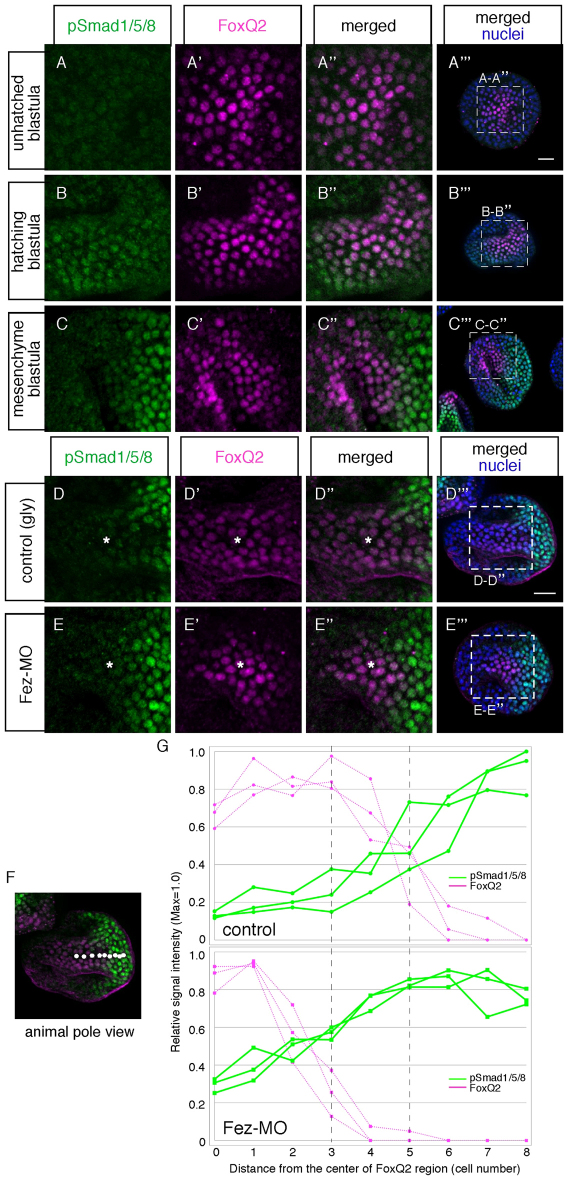
To determine if and when BMP2/4 signals in the animal plate, we monitored the presence of phospho-Smad1/5/8 (pSmad1/5/8) (Lapraz et al., 2009) and FoxQ2 protein by immunostaining between the unhatched blastula and mesenchyme blastula stages. The specificity of each of these antibodies in H. pulcherrimus was confirmed because all signals were absent in embryos injected with the corresponding morpholinos (see Fig. S4 in the supplementary material). Furthermore, all pSmad1/5/8 signals at this stage are attributable to BMP2/4 activity, because there are no signals in BMP2/4 morphants (see Fig. S4 in the supplementary material). Before the hatching blastula stage, no BMP signaling is detected, but when the embryo hatches, a clear signal appears in the nuclei of cells in the animal half of the embryo, including the animal plate marked by FoxQ2 expression (Fig. 6A,B). However, a few hours later, at mesenchyme blastula stage, BMP2/4 signaling increases dramatically in the future aboral ectoderm and establishes a decreasing gradient from aboral to oral through animal plate (Fig. 6C) (Lapraz et al., 2009). Based on the immunohistochemical data, the relatively strong BMP signaling area is closer to the animal pole in Fez morphants, compared with that in control embryos (Fig. 6D,E). To analyze this further, we measured pixel values on images and used ImageJ software to quantitate relative immunofluorescent signal intensities that reflect the relative levels of FoxQ2 and pSmad1/5/8 proteins in nuclei in cells in a line from the animal pole to the center of the aboral side (Fig. 6F, white dots). The data were normalized in each embryo, then averaged among individual batches and shown as relative intensity. The measurements were performed in 10-14 embryos in three independent batches to check reproducibility. In normal embryos, the concentration of FoxQ2 drops from maximal levels to background in the area three to six cells from the center of the animal plate and within that same area an inverse relationship exists for pSmad1/5/8 (Fig. 6G, upper panel). A similar inverse relationship exists in Fez morphants, but the corresponding drop in FoxQ2 occurs several cells closer to the center of the animal plate (Fig. 6G, lower panel). In both normal and Fez morphants, maximal levels of FoxQ2 are achieved when pSmad1/5/8 levels are 30-40% of maximal. The clear difference in Fez morphants is that, within the critical region of the animal plate that is defined by half maximal levels of FoxQ2 in control and Fez morphant embryo (vertical dotted dashed lines), the pSmad1/5/8 levels are ∼20-40% of maximum in controls, but 50-80% of maximum in Fez morphants. Because maximum levels of FoxQ2 and pSmad1/5/8 are the same in control and Fez morphants, and because Fez is not required for FoxQ2 expression, these data indicate that, in this outer region of the animal plate, maximal FoxQ2 expression occurs if Fez suppresses BMP signaling below a threshold level ∼30-40% of maximal.
Fig. 6.
BMP2/4 diffuses over the animal plate region and builds an activity gradient at the mesenchyme blastula stage. Accumulation of pSmad1/5/8 in the ectoderm during blastula stages. (A-C′′) Phospho-Smad1/5/8 (pSmad1/5/8) and FoxQ2 expression at unhatched blastula (A), hatching blastula (B) and mesenchyme blastula (C) stages. Boxed areas in A′′, B′′ and C′′ indicate magnified regions shown in A-A′, B-B′ and C-C′, respectively. DAPI nuclear stain is also shown in A′′, B′′ and C′′. (D-E′′) The accumulation of pSmad1/5/8 in the animal plate region is altered in Fez morphants at the mesenchyme blastula stage. FoxQ2 (D′,E′) marks the animal plate. Boxed areas in D′′ and E′′ indicate magnified regions shown in D-D′ and E-E′, respectively. DAPI nuclear stain is also shown in D′′ and E′′. Asterisks indicate the center of the FoxQ2-expressing region. (F) The intensities of fluorescent signals in nuclei for pSmad1/5/8 and FoxQ2 were measured with ImageJ for each cell along a line extending outward from the center of the animal plate to the center of the aboral side as illustrated (white dots). Some dark regions unlabeled by either FoxQ2 or pSmad1/5/8 antibodies are ectoderm folds that occur as a result of embryo processing procedures. We selected embryos in which there are no folds observed along the line to avoid inaccurate measurements. (G) Relative fluorescent signal intensities of pSmad1/5/8 and FoxQ2 in the animal plate/aboral ectoderm region in controls (upper panel) and Fez morphants (lower panel). The data were normalized in each individual, and then averaged for three to four embryos per mating pair. Data collected from three independent batches are shown by individual lines, and pSmad1/5/8 and FoxQ2 signals are shown by solid green and dotted magenta lines, respectively. Scale bars: in A′′ 20 γm for A-C; in D′′, 20 γm for D,E.
DISCUSSION
The data presented here establish that Fez functions as soon as it is produced downstream of FoxQ2 to attenuate BMP signaling on one side of the animal plate, thereby maintaining the size of this neuroectodermal domain in the sea urchin embryo. This mechanism is superimposed on the well-known pathway that produces a gradient of extracellular BMP activity through the activities of its antagonists or their activating proteases (Lamb et al., 1993; Sasai et al., 1994; Piccolo et al., 1997; Lee et al., 2006; Plouhinec and De Robertis, 2009). Fez produces a significant decline of BMP2/4 activity in the outer regions of the animal plate of the early embryo along which the serotonergic neurons will subsequently develop.
The model for the mechanism and timing of Fez function is described in Fig. 7. bmp2/4 begins to be expressed in prospective oral ectoderm of an unhatched blastula as a result of Nodal signaling, prior to the appearance of fez mRNA in the animal plate (Fig. 7A) (Duboc et al., 2004; Materna et al., 2010). Although no pSmad1/5/8 signals were observed in this embryo, it is likely that BMP2/4 protein starts to be secreted there and to signal at low levels because only a few hours later at hatching blastula stage low levels of pSmad1/5/8 can be detected in both aboral and anterior neurogenic ectoderm (Fig. 6B). At this time, FoxQ2 and perhaps other factors induce fez transcription in animal plate cells (Fig. 7A). During mesenchyme blastula stages, BMP2/4 moves towards the aboral side, away from the site of bmp2/4 transcription, possibly aided by Chordin, and establishes a gradient of activity between aboral and oral sides of the embryo (Fig. 6C, Fig. 7A) (Ben-Zvi et al., 2008; Lapraz et al., 2009). The BMP activity gradient in the neurogenic ectoderm is shallow and continuous in the absence of Fez function (Fig. 6G) and probably reflects the normal morphogen gradient pattern in the extracellular space. However, in the normal embryo, the continuous gradient is perturbed at the border between neurogenic and non-neurogenic ectoderm by Fez (Fig. 6G, Fig. 7B,C), sharpening the border between the animal plate and the aboral ectoderm.
Fig. 7.
Model for the timing and location of Fez suppression of BMP signaling in the sea urchin embryo. (A) BMP2/4 protein theoretically synthesized in the future oral ectoderm of unhatched blastula diffuses throughout the ectoderm including the animal plate. The highest activity of BMP2/4 is observed on the aboral side at mesenchyme blastula stage. Fez begins to be expressed in the neurogenic animal plate at the hatching blastula stage. Green and yellow show the regions where pSmad1/5/8 signal is present and absent, respectively. (B) FoxQ2 appears first in the animal plate before BMP signaling. Fez is induced by FoxQ2 and reaches levels sufficient to attenuate BMP at the outer edges of the animal plate so that BMP cannot inhibit FoxQ2 there. (C) The relationship between pSmad1/5/8 activity (green) and the size of the animal plate (magenta) with/without Fez function. In normal embryos with Fez function (upper panel), the activity of pSmad1/5/8 is dramatically decreased at the border between Fez-negative and -positive regions to a level that preserves FoxQ2 expression and the animal plate. In Fez morphants (lower panel), the gradient of pSmad1/5/8 is continuous from the aboral ectoderm to the animal pole and the abnormally high levels of pSmad1/5/8 in the periphery of the animal plate prevent expression of FoxQ2 and differentiation of the animal plate.
The abnormal shift of the animal plate border in Fez morphants occurs on the side where BMP signals, which reduces the size of the animal plate size in these embryos. This conclusion is supported by the observation that foxQ2 expression is reduced where BMP2/4 signaling occurs, but Nodal signaling is prevented. The consequence is that Fez functions to stabilize the border on the aboral side along which the serotonergic neurons develop; Fez is later also expressed specifically in these neurons, although it is not required to confer neuronal identity to them. Instead it might continue to protect them from BMP signaling and/or it might be involved in promoting normal axon guidance because elongation of axons in these neurons is poorly developed in Fez morphants (Fig. 2B), as has been observed in the olfactory bulb of Fez mutant mice (Hirata et al., 2006b).
Although the data presented here indicate that Fez is involved in attenuating BMP signaling along the outer regions of the aboral animal plate ectoderm, it is still not clear how the central part of this domain is protected against BMP2/4 signaling in Fez morphants (Fig. 6). Although pSmad1/5/8 activity is not as low as in normal embryos in this region of the animal plate, it might be below the threshold required to induce aboral, non-neurogenic ectoderm there. Alternatively, this central region might be protected by a Fez-independent mechanism(s). The latter possibility is more likely because neurons differentiate in this region of animal pole ectoderm even when mis-expressed BMP2/4 generates dramatic downregulation of foxQ2 and fez expression in these cells at blastula stage (Fig. 4) (Yaguchi et al., 2006).
Downregulation of BMP signaling along the aboral side of the animal plate by Fez function is probably cell autonomous, although the possibility that Fez induces a short-range signal cannot be strictly ruled out. So far, because the sequence of Fez suggests that it acts as a transcriptional repressor (Hirata et al., 2006a) rather than a Smad-interacting protein, such as SIP1 (Verschueren et al., 1999; van Grunsven et al., 2003; van Grunsven et al., 2007), it is unlikely that Fez interferes directly with pSmad1/5/8 activity in intracellular regions. Thus, it is possible that Fez regulates a gene(s) that is involved in pathways to maintain pSmad1/5/8 levels, such as degradation, dephosphorylation, and/or nucleocytoplasmic shuttling of Smad (Inman et al., 2002; Chen et al., 2006). Target genes repressed by Fez have not yet been identified in sea urchin, but, for instance, could include gene(s) encoding protein(s) that promote BMP signaling or regulatory gene(s) specifying aboral ectoderm downstream of BMP2/4 signaling. It might also support expression of several intracellular BMP signaling attenuators that have recently been identified, including SIP1 (Nitta et al., 2004), Smurf1 (Alexandrova and Thomsen, 2006; Sapkota et al., 2007), Ski and SnoN (Deheuninck and Luo, 2009). However, it is thought that the function of these intracellular factors is to pattern the neuroectoderm, not to establish it as a domain, which is what we focus on in this study. Whether they are also involved as mediators of Fez function to establish the precise border between neurogenic and non-neurogenic regions will require further investigation.
Based on previous studies, the Fez family functions at several developmental steps in the central nervous systems in other animals (Shimizu and Hibi, 2009), such as defining a subregion of anterior neuroectoderm (Hirata et al., 2006a) and controlling individual neural fate (Shimizu et al., 2010). With respect to molecular mechanisms, more is understood about its role in neural fate specification. In vertebrates, Fezf1 and Fezf2 control neural differentiation by suppressing Hes5 (Shimizu et al., 2010), and in flies, Erm, an ortholog of Fez in Drosophila melanogaster, maintains the restricted developmental potential of intermediate neural progenitors (Weng et al., 2010). Although it has been reported that Fezf1 and Fezf2 suppress development of the caudal diencephalon in mice, and expression of Irx1 and Wnt3a, the molecular mechanisms controlling these steps are unclear. It is also not known whether attenuation of BMP signaling is involved, as shown here in the sea urchin embryo.
A combination of extrinsic and intrinsic mechanisms controls the size of the animal plate. After restriction of the animal plate to the animal pole by canonical Wnt-dependent processes, further signaling through BMP signaling can affect its size. Here, we show that extracellular control of BMP signaling through antagonists is supplemented by intracellular control within the plate itself. Similarly, Nodal signaling on the other side of the plate suppresses serotonergic neuron development and it is blocked from the animal plate by the extracellular antagonist Lefty and the intracellular factor FoxQ2. What emerges from this and previous work (Yaguchi et al., 2008) is that a fascinating regulatory cassette, FoxQ2 and its downstream gene Fez, are involved in attenuating expression of Nodal and its downstream gene BMP2/4 sequentially to protect the neurogenic animal plate on its oral and aboral sides, respectively. These parallel mechanisms ensure that the edges of the field where neurons develop in the animal pole ectoderm are stably defined.
Supplementary Material
Acknowledgments
We thank Robert Angerer and Angerer laboratory members for fruitful comments on this work; Robert Burke, Yoko Nakajima, Yasunori Sasakura and David McClay for essential reagents and devices; and Julius Barsi and Eric Davidson for sharing unpublished data. We thank Masato Kiyomoto and Mamoru Yamaguchi for providing the adult sea urchins and Mrs Y. Tsuchiya, T. Sato, H. Shinagawa and Y. Yamada from the Shimoda Marine Research Center for collecting and keeping the adult sea urchins.
Footnotes
Funding
This work was supported, in part, by Special Coordination Funds for Promoting Science and Technology of the Ministry of Education, Culture, Sports, Science and Technology of the Japanese Government (MEXT), by Grant-in Aid for Young Scientists (Start-up No. 20870006 and B No. 21770227) and Takeda Science Foundation to S.Y., in part by the Intramural Program of the National Institutes of Health, NIDCR to L.M.A., and in part by MEXT (No. 22370023) to K.I. Deposited in PMC for release after 12 months.
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.069856/-/DC1
References
- Alexandrova E. M., Thomsen G. H. (2006). Smurf1 regulates neural patterning and folding in Xenopus embryos by antagonizing the BMP/Smad1 pathway. Dev. Biol. 299, 398-410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angerer L. M., Oleksyn D. W., Logan C. Y., McClay D. R., Dale L., Angerer R. C. (2000). A BMP pathway regulates cell fate allocation along the sea urchin animal-vegetal embryonic axis. Development 127, 1105-1114 [DOI] [PubMed] [Google Scholar]
- Ben-Zvi D., Shilo B. Z., Fainsod A., Barkai N. (2008). Scaling of the BMP activation gradient in Xenopus embryos. Nature 453, 1205-1211 [DOI] [PubMed] [Google Scholar]
- Bradham C. A., Oikonomou C., Kuhn A., Core A. B., Modell J. W., McClay D. R., Poustka A. J. (2009). Chordin is required for neural but not axial development in sea urchin embryos. Dev. Biol. 328, 221-233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke R. D., Osborne L., Wang D., Murabe N., Yaguchi S., Nakajima Y. (2006). Neuron-specific expression of a synaptotagmin gene in the sea urchin Strongylocentrotus purpuratus. J. Comp. Neurol. 496, 244-251 [DOI] [PubMed] [Google Scholar]
- Chen H. B., Shen J., Ip Y. T., Xu L. (2006). Identification of phosphatases for Smad in the BMP/DPP pathway. Gene Dev. 20, 648-653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Robertis E. M., Kuroda H. (2004). Dorsal-ventral patterning and neural induction in Xenopus embryos. Annu. Rev. Cell Dev. Biol. 20, 285-308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deheuninck J., Luo K. (2009). Ski and SnoN, potent negative regulators of TGF-beta signaling. Cell Res. 19, 47-57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duboc V., Rottinger E., Besnardeau L., Lepage T. (2004). Nodal and BMP2/4 signaling organizes the oral-aboral axis of the sea urchin embryo. Dev. Cell 6, 397-410 [DOI] [PubMed] [Google Scholar]
- Gardner T. S., Cantor C. R., Collins J. J. (2000). Construction of a genetic toggle switch in Escherichia coli. Nature 403, 339-342 [DOI] [PubMed] [Google Scholar]
- Hirata T., Nakazawa M., Muraoka O., Nakayama R., Suda Y., Hibi M. (2006a). Zinc-finger genes Fez and Fez-like function in the establishment of diencephalon subdivisions. Development 133, 3993-4004 [DOI] [PubMed] [Google Scholar]
- Hirata T., Nakazawa M., Yoshihara S., Miyachi H., Kitamura K., Yoshihara Y., Hibi M. (2006b). Zinc-finger gene Fez in the olfactory sensory neurons regulates development of the olfactory bulb non-cell-autonomously. Development 133, 1433-1443 [DOI] [PubMed] [Google Scholar]
- Inman G. J., Nicolás F. J., Hill C. S. (2002). Nucleocytoplasmic shuttling of Smads 2, 3, and 4 permits sensing of TGF-β receptor activity. Mol. Cell 10, 283-294 [DOI] [PubMed] [Google Scholar]
- Khokha M. K., Yeh J., Grammer T. C., Harland R. M. (2005). Depletion of three BMP antagonists from Spemann's organizer leads to a catastrophic loss of dorsal structures. Dev. Cell 8, 401-411 [DOI] [PubMed] [Google Scholar]
- Kiecker C., Niehrs C. (2001). A morphogen gradient of Wnt/beta-catenin signaling regulates anteroposterior neural patterning in Xenopus. Development 128, 4189-4201 [DOI] [PubMed] [Google Scholar]
- Lamb T. M., Knecht A. K., Smith W. C., Stachel S. E., Economides A. N., Stahl N., Yancopolous G. D., Harland R. M. (1993). Neural induction by the secreted polypeptide noggin. Science 262, 713-718 [DOI] [PubMed] [Google Scholar]
- Lapraz F., Besnardeau L., Lepage T. (2009). Patterning of the dorsal-ventral axis in echinoderms: insights into the evolution of the BMP-chordin signaling network. PLoS Biol. 7, e1000248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H. X., Ambrosio A. L., Reversade B., De Robertis E. M. (2006). Embryonic dorsal-ventral signaling: secreted Frizzled-related proteins as inhibitors of tolloid proteinases. Cell 124, 147-159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan C. Y., Miller J. R., Ferkowicz M. J., McClay D. R. (1999). Nuclear beta-catenin is required to specify vegetal cell fates in the sea urchin embryo. Development 126, 345-357 [DOI] [PubMed] [Google Scholar]
- Materna S. C., Nam J., Davidson E. H. (2010). High accuracy, high-resolution prevalence measurement for the majority of locally expressed regulatory genes in early sea urchin development. Gene Expr. Patterns 10, 177-184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minokawa T., Rast J. P., Arenas-Mena C., Franco C. B., Davidson E. H. (2004). Expression patterns of four different regulatory genes that function during sea urchin development. Gene Expr. Patterns 4, 449-456 [DOI] [PubMed] [Google Scholar]
- Nakajima Y., Kaneko H., Murray G., Burke R. D. (2004). Divergent patterns of neural development in larval echinoids and asteroids. Evol. Dev. 6, 95-104 [DOI] [PubMed] [Google Scholar]
- Nitta K. R., Tanegashima K., Takahashi S., Asashima M. (2004). XSIP1 is essential for early neural gene expression and neural differentiation by suppression of BMP signaling. Dev. Biol. 275, 258-267 [DOI] [PubMed] [Google Scholar]
- Piccolo S., Agius E., Lu B., Goodman S., Dale L., De Robertis E. M. (1997). Cleavage of Chordin by Xolloid metalloprotease suggests a role for proteolytic processing in the regulation of Spemann organizer activity. Cell 91, 407-416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plouhinec J. L., De Robertis E. M. (2009). Systems biology of the self-regulating morphogenetic gradient of the Xenopus gastrula. Cold Spring Harb. Perspect. Biol. 1, a001701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reversade B., Kuroda H., Lee H., Mays A., De Robertis E. M. (2005). Depletion of Bmp2, Bmp4, Bmp7 and Spemann organizer signals induces massive brain formation in Xenopus embryos. Development 132, 3381-3392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapkota G., Alarcon C., Spagnoli F. M., Brivanlou A. H., Massague J. (2007). Balancing BMP signaling through integrated inputs into the Smad1 linker. Mol. Cell 25, 441-454 [DOI] [PubMed] [Google Scholar]
- Sasai Y., Lu B., Steinbeisser H., Geissert D., Gont L. K., De Robertis E. M. (1994). Xenopus chordin: a novel dorsalizing factor activated by organizer-specific homeobox genes. Cell 79, 779-790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saudemont A., Haillot E., Mekpoh F., Bessodes N., Quirin M., Lapraz F., Duboc V., Rottinger E., Range R., Oisel A., et al. (2010). Ancestral regulatory circuits governing ectoderm patterning downstream of Nodal and BMP2/4 revealed by gene regulatory network analysis in an echinoderm. PLoS Genet. 6, e1001259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sea Urchin Genome Sequencing Consortium: Sodergren E., Weinstock G. M., Davidson E. H., Cameron R. A., Gibbs R. A., Angerer R. C., Angerer L. M., Arnone M. I., Burgess D. R., Burke R. D., et al. (2006). The genome of the sea urchin Strongylocentrotus purpuratus. Science 314, 941-952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen Z. J., Kim S. K., Jun D. Y., Park W., Kim Y. H., Malter J. S., Moon B. J. (2007). Antisense targeting of TGF-beta1 augments BMP-induced upregulation of osteopontin, type I collagen and Cbfa1 in human Saos-2 cells. Exp. Cell Res. 313, 1415-1425 [DOI] [PubMed] [Google Scholar]
- Shimizu T., Hibi M. (2009). Formation and patterning of the forebrain and olfactory system by zinc-finger genes Fezf1 and Fezf2. Dev. Growth Differ. 51, 221-231 [DOI] [PubMed] [Google Scholar]
- Shimizu T., Nakazawa M., Kani S., Bae T.-K., Shimizu T., Kageyama R., Hibi M. (2010). Zinc finger genes Fezf1 and Fezf2 control neuronal differentiation by repressing Hes5 expression in the forebrain. Development 137, 1875-1885 [DOI] [PubMed] [Google Scholar]
- Spemann H., Mangold H. (1924). The induction of embryonic predispositions by implantation of organizers foreign to the species. Arch. Mikrosk. Anat. Entwicklungsmech. 100, 599-638 [Google Scholar]
- Tu Q., Brown C. T., Davidson E. H., Oliveri P. (2006). Sea urchin Forkhead gene family: phylogeny and embryonic expression. Dev. Biol. 300, 49-62 [DOI] [PubMed] [Google Scholar]
- van Grunsven L. A., Michiels C., Van de Putte T., Nelles L., Wuytens G., Verschueren K., Huylebroeck D. (2003). Interaction between Smad-interacting protein-1 and the corepressor C-terminal binding protein is dispensable for transcriptional repression of E-cadherin. J. Biol. Chem. 278, 26135-26145 [DOI] [PubMed] [Google Scholar]
- van Grunsven L. A., Taelman V., Michiels C., Verstappen G., Souopgui J., Nichane M., Moens E., Opdecamp K., Vanhomwegen J., Kricha S., et al. (2007). XSip1 neuralizing activity involves the co-repressor CtBP and occurs through BMP dependent and independent mechanisms. Dev. Biol. 306, 34-49 [DOI] [PubMed] [Google Scholar]
- Verschueren K., Remacle J. E., Collart C., Hraft H., Baker B. S., Tylzanowski P., Nelles L., Wuytens G., Su M.-T., Bodmer R., et al. (1999). SIP1, a novel zinc finger/homeodomain repressor, interacts with Smad proteins and binds to 5′-CACCT sequences in candidate target genes. J. Biol. Chem. 274, 20489-20498 [DOI] [PubMed] [Google Scholar]
- Wei Z., Angerer R. C., Angerer L. M. (2006). A database of mRNA expression patterns for the sea urchin embryo. Dev. Biol. 300, 476-484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Z., Yaguchi J., Yaguchi S., Angerer R. C., Angerer L. M. (2009). The sea urchin animal pole domain is a Six3-dependent neurogenic patterning center. Development 136, 1179-1189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Z., Angerer R. C., Angerer L. M. (2011). Direct development of neurons within foregut endoderm of sea urchin embryos. Proc. Natl. Acad. Sci. USA 108, 9143-9147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weng M., Golden K. L., Lee C.-Y. (2010). dFezf/Earmuff maintains the restricted developmental potential of intermediate neural progenitors in Drosophila. Dev. Cell 18, 126-135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wikramanayake A. H., Huang L., Klein W. H. (1998). β-catenin is essential for patterning the maternally specified anima-vegetal axis in the sea urchin embryo. Proc. Natl. Acad. Sci. USA 95, 9343-9348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaguchi S., Katow H. (2003). Expression of tryptophan 5-hydroxylase gene during sea urchin neurogenesis and role of serotonergic nervous system in larval behavior. J. Comp. Neurol. 466, 219-229 [DOI] [PubMed] [Google Scholar]
- Yaguchi S., Kanoh K., Amemiya S., Katow H. (2000). Initial analysis of immunochemical cell surface properties, location and formation of the serotonergic apical ganglion in sea urchin embryos. Dev. Growth Differ. 42, 479-488 [DOI] [PubMed] [Google Scholar]
- Yaguchi S., Yaguchi J., Burke R. D. (2006). Specification of ectoderm restricts the size of the animal plate and patterns neurogenesis in sea urchin embryos. Development 133, 2337-2346 [DOI] [PubMed] [Google Scholar]
- Yaguchi S., Yaguchi J., Burke R. D. (2007). Sp-Smad2/3 mediates patterning of neurogenic ectoderm by nodal in the sea urchin embryo. Dev. Biol. 302, 494-503 [DOI] [PubMed] [Google Scholar]
- Yaguchi S., Yaguchi J., Angerer R. C., Angerer L. M. (2008). A Wnt-FoxQ2-nodal pathway links primary and secondary axis specification in sea urchin embryos. Dev. Cell 14, 97-107 [DOI] [PubMed] [Google Scholar]
- Yaguchi S., Yaguchi J., Angerer R. C., Angerer L. M., Burke R. D. (2010a). TGFbeta signaling positions the ciliary band and patterns neurons in the sea urchin embryo. Dev. Biol. 347, 71-81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaguchi S., Yaguchi J., Wei Z., Shiba K., Angerer L. M., Inaba K. (2010b). ankAT-1 is a novel gene mediating the apical tuft formation in the sea urchin embryo. Dev. Biol. 348, 67-75 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.