Abstract
NK-cell leukemia is a clonal expansion of NK cells. The illness can occur in an aggressive or chronic form. We studied cell lines from human and rat NK-cell leukemias (aggressive NK-cell leukemia) as well as samples from patients with chronic NK-cell leukemia to investigate pathogenic mechanisms. Here we report that Mcl-1 was overexpressed in leukemic NK cells and that knockdown of Mcl-1 induced apoptosis in these leukemic cells. In vitro treatment of human and rat NK leukemia cells with FTY720 led to caspase-dependent apoptosis and decreased Mcl-1 expression in a time- and-dose-dependent manner. These biologic effects could be inhibited by blockade of reactive oxygen species generation and the lysosomal degradation pathway. Lipidomic analyses after FTY720 treatment demonstrated elevated levels of sphingosine, which mediated apoptosis of leukemic NK cells in vitro. Importantly, systemic administration of FTY720 induced complete remission in the syngeneic Fischer rat model of NK-cell leukemia. Therapeutic efficacy was associated with decreased expression of Mcl-1 in vivo. These data demonstrate that therapeutic benefit of FTY720 may result from both altered sphingolipid metabolism as well as enhanced degradation of a key component of survival signaling.
Introduction
Large granular lymphocyte (LGL) leukemia is a clonal disease representing a spectrum of biologically distinct lymphoproliferative diseases originating either from mature T cells (CD3+) or NK cells (CD3−). Both CD3+ and CD3− LGLs function as cytotoxic lymphocytes. The 2008 World Health classification of mature T- and NK-cell neoplasms continues to distinguish T-cell LGL leukemia (T-LGL leukemia) from aggressive NK-cell leukemia based on their unique molecular and clinical features. Furthermore, a new provisional entity of chronic lymphoproliferative disorder of NK cells (also known as chronic NK-cell lymphocytosis or chronic NK-LGL leukemia) was created to distinguish it from much more aggressive NK-cell leukemia.1 Both aggressive NK-cell leukemia and chronic NK-cell leukemia display CD3−CD56+ immunophenotype.1 Aggressive NK-cell leukemia is a fatal illness which pursues a very malignant clinical course characterized by refractoriness to conventional chemotherapy, and a short median survival time of 2 months.2–4 There are no effective therapies for this disorder and pathogenetic mechanisms remain undefined. FTY720 has recently shown preclinical activity in hematologic malignancies. At low nanomolar concentrations, FTY720 becomes phosphorylated in vivo and acts as a potent agonist for sphingosine 1 phosphate receptors. In this capacity, as an immunomodulatory drug, FTY720 was recently approved by the Food and Drug Administration for treatment of multiple sclerosis, based on phase 3 efficacy data.5 In contrast, at high micromolar dosages, FTY720 has shown antitumor activity, inducing apoptosis of leukemic cells through a PP2A-dependent mechanism.6,7
Here we report biologic activity of FTY720 in chronic and aggressive NK-cell leukemia. FTY720 induced apoptosis of leukemic NK cells by degrading Mcl-1 through a lysosomal rather than a proteosomal pathway. Lipidomic experiments demonstrated increased levels of proapoptosis sphingosine in leukemic cells after FTY720 treatment. Previously, we used the Fischer rat NK-cell leukemia model to demonstrate efficacy of nanoliposomal ceramide. Using this animal model, we show here that FTY720 induced complete remission of aggressive NK-cell leukemia.
Methods
Reagents and cell culture
FTY720 was synthesized and supplied in powder form by the Organic Synthesis Core facility in the Penn State Hershey Cancer Institute following methods as previously described.8 GAPDH Ab and cathepsin B Ab were purchased from Cell Signaling Technology Inc. Mcl-1 Ab (S-19) was purchased from Santa Cruz Biotechnology. For Western blotting, 12% precasted Nupage electrophoresis gels were obtained from Invitrogen, and ECL reagent was purchased from Amersham Biosciences Inc. Human NKL cells (kindly provided by Dr Howard Young, National Cancer Institute) were grown in RPMI 1640 medium supplemented with 20% FBS plus 100 IU/mL IL-2. RNK-16 cells (kindly provided by Dr Craig Reynolds, National Cancer Institute) were cultured in RPMI 1640 supplemented with 10% FBS.
Patient characteristics and preparation of PBMCs
All patients met the clinical criteria of NK-cell leukemia with increased numbers (> 80%) of CD3−CD56+ NK cells in the peripheral blood. Nine patients had chronic NK-cell leukemia while one patient had aggressive NK-cell leukemia. Patients received no treatment at the time of sample acquisition. Peripheral blood specimens from these patients were obtained and informed consents signed for sample collection according to a protocol approved by the Institutional Review Board of Penn State Hershey Cancer Institute. Buffy coats from 8 age- and sex-matched normal donors were also obtained from the blood bank of Milton S. Hershey Medical Center (College of Medicine, Penn State University). PBMCs were isolated by Ficoll-Hypaque gradient separation, as described previously.9 Cell viability was determined by trypan blue exclusion assay with > 95% viability in all the samples. NK cells from additional 11 age- and sex-matched healthy donors were isolated by a negative selection process (StemCell Technologies), as described previously.10 The purity of freshly isolated CD3−CD56+ cells (2 × 105/sample in triplicate) in each of the samples was determined by flow cytometric assay by detecting positive staining of the CD56 marker for NK cells. The purity for normal purified NK cells was between 85% and 90%.
In vitro apoptosis and cell viability assay
Apoptosis was determined by 2-color flow cytometry with annexin V (5 μL/sample; BD Pharmingen) and 7-amino-actinomycin D (7-AAD; 10 μL/sample) staining using 5 × 105 cells per sample. The percentage-specific apoptosis was calculated using the following: apoptosis (%) = % annexin V–FITC-positive in assay well. Cell viability was performed using the CellTiter 96 Aqueous One Solution assay kit (Promega).10 The relative viable cell number was determined by reading the plates at 490-nm wavelength in a Synergy HT Multi-Detection microplate reader (Bio-Tek).
Mcl-1 expression: Western blot
Purified NK cells from 5 chronic NK-cell leukemia patients as well as 10 pooled normal NK cells from age- and sex-matched healthy donors were isolated by a negative selection process (StemCell Technologies). RNK-16 cells (5 × 106) or patients' PBMCs (CD3−CD56+ > 80%) treated with different doses of FTY720 at different times were lysed in buffer containing: 150mM NaCl, 1% Nonidet P-40, 0.5% deoxycholic acid, 0.1% SDS, 50mM Tris, and protease inhibitor cocktail (Sigma-Aldrich). Lysate was then used for Western blotting with Mcl-1 Ab.
Mcl-1 shRNA-mediated protein knockdown
The pLKO.1-based lentiviral shRNA targeting human Mcl-1, TRCN0000005517, and pLKO.1-based scrambled control shRNA vector were purchased from Open Biosystems. The plasmids were prepared with the plasmid maxi kit and transfected in HEK293FT cells (Invitrogen) with the transfection reagent (Invitrogen) to produce lentiviral particles. NKL, a human NK-cell leukemia cell line, was infected with the concentrated lentiviral stocks in the presence of 8 μg/mL polybrene for 24 hours and then changed to RPMI 1640 medium for an additional 48 hours. Protein lysates were collected for Western blot analysis of Mcl-1 expression and apoptosis assays were performed.
Cathepsin B release assay: isolation of cytosol
Lysosomal integrity was assessed by estimation of the amount of cathepsin B translocated to the cytosol during lysosomal stress. Cytosolic extraction was performed using a Subcellular Protein Fractionation Kit (Thermo Scientific). The cytosolic fraction lysate was then used for Western blotting with a cathepsin B Ab.
Confocal studies
To verify cell accumulation of FTY720 into RNK16 cells, cells were plated at 2.0 × 104 per well in 8-well chamber slides and allowed to grow overnight. RNK-16 cells were treated with 5μM FTY720 for 2 hours. Cellular nuclei were counterstained with Hoechst 33258; Lysotracker and MitoTracker Deep Red 633 (Molecular Probes) were used as markers for lysosome and mitochondria, respectively, following the manufacturer's instructions. FTY720 delivery and accumulation was evaluated by confocal microscopy at ×63 magnification for Lysotracker costaining or at ×40 magnification for MitoTracker costaining (Leica Microsystems).
Lipid quantification by mass spectroscopy
RNK-16 cells were treated with DMSO or 10μM FTY720 for 6, 16, and 24 hours. Sphingolipids from these cells were then analyzed by electrospray ionization–tandem mass spectrometry (ESI-MS/MS) based on the method described previously.11 Briefly, 5 × 106 of cells were homogenized by sonication in a 10mM Tris buffer (pH = 7.2). These samples then were subjected to lipid extraction by the use of chloroform/methanol/water as originally described by Bligh and Dyer12 after internal standards had been added (C17-sphingosine-1-phosphate, C17-sphingosine, C17-sphinganine, C17-sphinganine-1-phosphate, C12-ceramide). Sphingoid bases and 1-phosphates were separated by HPLC (Agilent 1100) with a C18-reverse phase column, and ceramide was separated on a LC-NH2-amino column before detection via multiple reaction monitoring (MRM) and quantification with an inline ABI 4000 mass spectrometer (a hybrid quadrupole-ion trap mass spectrometer; Applied Biosystems). The peak areas for the different sphingolipid subspecies were quantified according to the internal standards and normalized to cell numbers.
Animal studies
Animal experimentation was performed according to protocols approved by the Institutional Animal Care and Use Committee at the Pennsylvania State College of Medicine. Male F344 rats of approximately 6 weeks of age were obtained from Charles River Laboratories. One million RNK-16 cells, an in vivo NK-cell leukemia cell line (provided by Dr Craig Reynolds, National Cancer Institute), were IP transplanted into each of F344 rats. Five weeks after inoculation, leukemic rats were then injected intraperitoneally with 4.5 mg/kg FTY720 or PBS every day over a 4-week treatment period. Rats losing 20% body weight were most likely affected by NK-cell leukemia and were euthanized. At necropsy, spleens were dissected, weighed, and fixed in 4% paraformaldehyde and embedded in paraffin, and sections (4 microns) were generated for histologic analysis and immunohistochemical staining. Mononuclear cells were isolated from blood, BM, and spleen using Ficoll-Hypaque density gradient centrifugation,13 and used for quantification of CD3−CD8a+ NK cells by flow cytometry. PerCP-conjugated mouse anti–rat CD8a and FITC-conjugated mouse anti–rat CD3 were purchased from BD Pharmingen.
Statistical analysis
Differences among 2 treatment groups were statistically analyzed using a 2-tailed Student t test for statistical analyses. The Kaplan-Meier survival curves were plotted to evaluate the survival of leukemic rats; the log-rank (Mantel-Cox) test was used to analyze the statistical difference between the FTY720 and PBS-treated leukemic or normal rats. A statistically significant difference was reported with P indicated where applicable. Data are reported as the mean ± SE from at least 3 separate experiments.
Results
FTY720 triggers caspase-dependent apoptotic cell death in leukemic but not normal NK cells
FTY720 exerts apoptotic effects in some human leukemic cells in vitro.6,7 Therefore, we wanted to determine whether it can induce apoptosis in leukemic NK cells. Initial experiments demonstrated that FTY720 produced dose-dependent apoptotic cell death in NKL, an aggressive human NK-cell leukemia cell line (Figure 1A) and RNK-16, an aggressive rat NK leukemia cell line (Figure 1B). We thought it important to validate the apoptotic effect of FTY720 observed in cell lines in patient samples. Indeed, we found that FTY720 induced apoptosis in PBMCs from one aggressive NK-cell leukemia patient and 3 chronic NK-cell leukemia patients (CD3−CD56+ > 80%, Figure 1C). Because limited material was available from patients with aggressive NK-cell leukemia, we used aggressive NK-cell leukemia cell lines throughout the remainder of the manuscript for further mechanistic studies. Such experiments showed that FTY720 resulted in time-dependent apoptosis of NKL (supplemental Figure 1A, available on the Blood Web site; see the Supplemental Materials link at the top of the online article), RNK-16 (supplemental Figure 1B), as well as PBMCs from 3 chronic NK-cell leukemia patients (CD3−CD56+ > 80%, supplemental Figure 1C). In contrast, treatment with 10μM FTY720 had no effect on viability of normal PBMCs (supplemental Figure 2A), and did not induce apoptosis in normal NK cells (supplemental Figure 2B). Moreover, we demonstrated that the FTY720-induced apoptosis in patients' PBMCs occurred via cleavage of caspase 3 and PARP, the hallmarks of caspase-dependent apoptosis (supplemental Figure 3A). Furthermore, inhibition of caspase activity via z-VAD-fmk could block FTY720-induced cell death in leukemic NK cells, suggesting that the apoptosis by FTY720 was caspase-dependent (supplemental Figure 3B).
Figure 1.
FTY720 induces apoptotic cell death in leukemic NK cells in a dose-dependent manner. (A) Human NKL cell line was treated with 5, 10, and 15μM FTY720 or DMSO for 24 hours, then cells were assayed for apoptosis by flow cytometry. (B) Rat RNK-16 cell line was treated with 2.5, 5, and 10μM FTY720 or DMSO for 24 hours then cells were assayed for apoptosis by flow cytometry. (C) PBMCs from one acute NK-cell leukemia patient (no. 1) and 3 chronic NK-cell leukemia patients (nos. 2, 3, 4) were treated with 2.5, 5, and 10μM FTY720 or DMSO for 18 hours, then cells were assayed for apoptosis by flow cytometry. *P < .05, **P < .005, and ***P < .0005 indicate significant differences of FTY720-treated cells versus DMSO-treated cells (Student t test).
FTY720-induced apoptosis in leukemic NK cells is dependent on degradation of Mcl-1 and not activation of PP2A
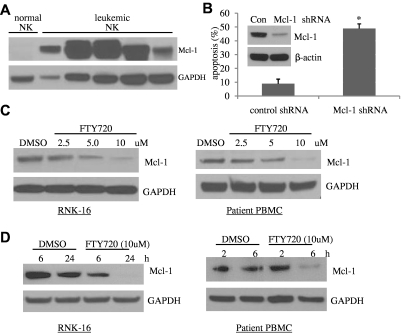
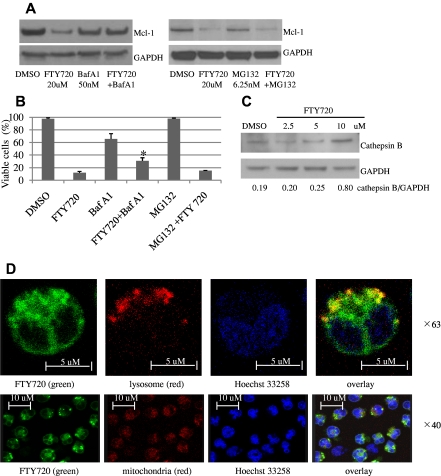
To further investigate the mechanism of FTY720-induced apoptosis, we focused our attention on Mcl-1, which plays an important role in apoptosis resistance in leukemic T LGL cells.13 Leukemic NK cells expressed high levels of Mcl-1 whereas normal NK cells did not (Figure 2A). Furthermore, efficient shRNA-mediated knockdown (80%) of Mcl-1 (Figure 2B inset) resulted in a 5.5-fold increase in apoptosis in the human NKL cell line (Figure 2B). We found that FTY720 treatment of leukemic NK cells led to significant reduction in Mcl-1 protein levels in a dose- and time-dependent fashion (Figure 2C-D). Down-regulation of Mcl-1 by FTY720 did not occur at the transcriptional level (supplemental Figure 4). Therefore, we investigated protein turnover pathways in leukemic NK cells. Notably, bafilomycin A1, a lysosomal inhibitor, but not MG132, a proteosomal inhibitor, restored Mcl-1 expression in FTY720-treated cells (Figure 3A), and rescued FTY720 induced cell death (Figure 3B). These data lend support to the hypothesis that FTY720 might down-regulate Mcl-1 via the lysosomal degradation pathway. Indeed, we demonstrated in a dose-dependent manner release of lysosomal enzyme cathepsin B, the most abundant cysteine protease in the lysosome, to cytosol in RNK-16 cells following FTY720 treatment (Figure 3C). Furthermore, colocalization of NBD-FTY720 and Lysotracker Red in RNK-16 cells confirmed that FTY720 accumulation occurs in cellular lysosomes but not in mitochondria at 2 hours after treatment, implicating FTY720 as a direct cause of lysosomal membrane permeabilization (LMP) in leukemic NK cells (Figure 3D). In addition, NAC, a reactive oxygen species (ROS) scavenger, can also restore Mcl-1 expression (supplemental Figure 5A) and rescue FTY720-induced apoptosis (supplemental Figure 5B) in RNK-16 cells, supporting the notion that FTY720 decreased expression of Mcl-1 via ROS production.
Figure 2.
Mcl-1 plays an essential role in survival of leukemic NK cells and is degraded by FTY720. (A) Increased expression of Mcl-1 in NK-cell leukemia. Proteins were isolated from PBMCs of 5 individual patients with chronic NK-cell leukemia (CD3−CD56+ cells > 80%), or pooled enriched NK cells (CD3−CD56+ 80%-95%) from 10 normal human donors, then resolved in the SDS-PAGE gel loading buffer in a boiling water bath for 5 minutes. Western blot analysis was performed for detection of Mcl-1. GAPDH was used as a loading control. (B) Knockdown of Mcl-1 induces apoptosis in leukemic NK cells. Human NKL cells were infected with the concentrated PLKO.1 Mcl-1 shRNA or scramble shRNA lentiviral stocks for 48 hours, and then apoptosis assay was performed. *P < .05 indicates significant difference in apoptosis of Mcl-1 shRNA-infected cells compared with control shRNA-infected cells (Student t test). (Inset) Western blot analysis was performed for Mcl-1 in the control shRNA or Mcl-1 shRNA-infected NKL cells 48 hours after infection. The equal loading of protein was confirmed by probing with GAPDH Ab. (C) Dose-dependent reduced expression of Mcl-1 after FTY720 treatment. Western blot analysis was performed for Mcl-1 after 24-hour treatment of RNK-16 cells or PBMC cells from an NK-cell leukemia patient, with 2.5, 5, and 10μM FTY720. Data are representative of 3 independent experiments on RNK-16 cells or PBMCs from 3 chronic NK-cell leukemia patients. (D) Time-dependent decreased expression of Mcl-1 after FTY720 treatment. Western blot analysis was performed for Mcl-1 after treatment of RNK-16 cells with 10μM FTY720 for 6 and 24 hours, or PBMC cells from an NK-cell leukemia patient with 5μM FTY720 for 2 and 6 hours. Data are representative of 3 independent experiments on RNK-16 cells or PBMCs from 3 chronic NK-cell leukemia patients.
Figure 3.
FTY720 down-regulates Mcl-1 expression via lysosomal degradation. RNK-16 cells were pretreated with 50nM bafilomycin A1 or 6.25nM MG132 for 30 minutes then exposed to 20μM FTY720 for 3 hours, then Western blot was performed to examine Mcl-1 expression (A) or cells were assayed for cell viability by MTT (B). (C) Dose-dependent increased expression of cathepsin B after FTY720 treatment. Western blot analysis was performed for cathepsin B in cytosolic fraction of RNK-16 cells after 24-hour treatment with 2.5, 5, and 10μM FTY720. ImageJ software was used to analyze the ratio of densitometry of cathepsin B to GAPDH as labeled in the bottom of Western blot images. (D) Confocal microscopic image of RNK-16 cells after 2 hours of treatment, showing that yellow staining indicating NBD-FTY720 (green) colocalized with cellular lysosome (top panel, red, magnification: ×63) but not mitochondria (bottom panel, red, magnification: ×40). Cellular nucleus was stained with (Hoechst 33258 blue).
Previous studies had determined that FTY720-induced apoptosis in leukemic cells was PP2A-dependent.6,7 In contrast, pretreatment with okadaic acid, a specific PP2A inhibitor, did not result in inhibition of FTY720-induced cell death in either patient leukemic cells (supplemental Figure 6A) or rat RNK-16 cells (supplemental Figure 6B); furthermore, direct measurement of PP2A phosphatase activity showed no change in human NKL cells on FTY720 treatment (supplemental Figure 6C). These data show that FTY720 induced apoptosis in leukemic NK cells is not dependent on activation of PP2A.
Lipidomic analyses demonstrate increased levels of proapoptotic sphingosine after FTY720 treatment of leukemic NK cells
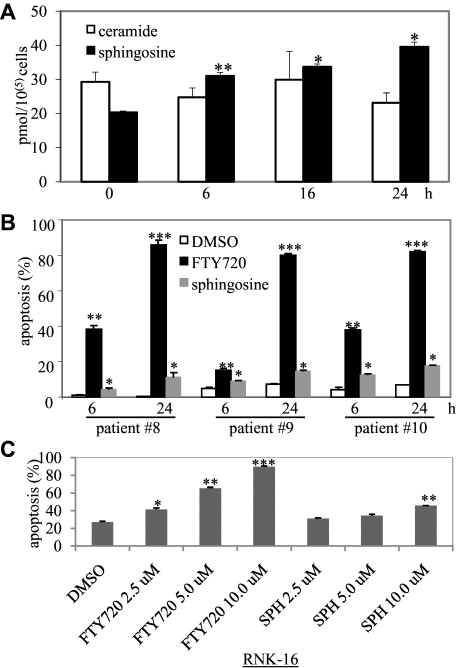
Lipidomic analyses to determine the effects of FTY720 on sphingolipid metabolites are limited to one study. That report demonstrated increased levels of proapoptotic lipid sphingosine in endothelial cells after FTY720 treatment.14 Therefore, to investigate an additional mechanism for FTY720-induced apoptosis in leukemic NK cells, we were interested in evaluating the sphingolipid metabolites in leukemic NK cells after FTY720 treatment. Importantly, we found that levels of the proapoptotic sphingolipid, sphingosine, but not ceramide, were increased in FTY720-treated RNK-16 cells in a time-dependent manner (Figure 4A). Prosurvival lipid sphingosine 1-phosphate (S1P) levels were not significantly altered (data not shown). Moreover, administration of sphingosine led to an increase in apoptosis in PBMCs from 3 chronic NK-cell leukemia patients (CD3−CD56+ > 80%, Figure 4B) or RNK-16 cells (Figure 4C); although to a lesser extent than treatment with FTY720 (Figure 4B-C). Of interest, treatment of leukemic NK cells with sphingosine did not alter Mcl-1 expression (data not shown). These data indicate that induction of apoptosis by FTY720 in leukemic NK cells may be partially mediated by the accumulation of endogenous sphingosine.
Figure 4.
FTY720 increased production of apoptosis-inducing lipid sphingosine. (A) Five million RNK-16 cells were treated with 10μM FTY720 for 6, 16, and 24 hours then were extracted to obtain lipids; extracts were then identified and quantitated by tandem mass spectrometry. Each sample had 4 replicates. *P < .05 and **P < .005 indicate significant differences versus control (Student t test). (B) PBMCs from 3 individual chronic NK-cell leukemia patients (nos. 8, 9, 10) were treated with 5μM FTY720, 5μM sphingosine, or DMSO for 6 and 24 hours then cells were assayed for apoptosis by flow cytometry. (C) RNK-16 cells were treated with 2.5, 5.0, 10.0μM FTY720 or sphinginosine (SPH) for 48 hours, then cells were assayed for apoptosis by flow cytometry. *P < .05, **P < .005, and ***P < .0005 indicate significant differences versus control (Student t test).
Therapeutic efficacy of FTY720 in a rat model of NK-cell leukemia
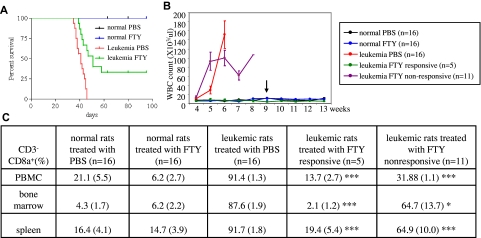
We further tested FTY720 in a Fischer F344 rat NK-cell leukemia model, which has been established as an important experimental model for the study of NK-cell leukemia progression and for evaluation of therapeutics.15–17 This animal model closely resembles human aggressive NK-cell leukemia based on morphologic, functional, and clinical criteria.16 One million RNK-16 cells from in vivo NK-cell leukemic cell line were IP transplanted into each of 6-week-old male Fischer rats. At 4 weeks after transplantation, the animals displayed early signs of leukemia, including weight loss, rough hair coat and increased level of neutrophils as reported previously.15 By week 5, circulating blasts, anemia, thrombocytopenia, and splenomegaly were observed. These leukemic rats were then injected IP with 4.5 mg/kg FTY720 or PBS every day over a 4-week treatment period. Animals died within the next 1 to 2 weeks if treated with PBS (Figure 5A). The median survival in PBS-treated group was 41 days compared with 51 days in FTY720-treated group (Mantel-Cox test, P < .0001; Figure 5A). Importantly, 5 of 16 leukemic rats treated with FTY720 had maintenance of normal blood counts without circulating blasts suggesting achievement of complete clinical remission (Figure 5B). The remaining 11 leukemic rats treated with FTY720 had transient improvement, as evidenced by reduction of white blood cell counts and maintenance of platelet counts 2 weeks after treatment (data not shown). Subsequently, however, blast counts rose and animals died within the following week. To further determine remission status, the 5 responsive animals were euthanized 1, 18, 18, 18, and 39 days after cessation of the treatment. At necropsy, we found these rats had normal levels of CD3−CD8a+ NK cells in the blood, marrow, and spleen, thereby documenting complete remission (Figure 5C). Of interest, even in the 11 leukemic rats not achieving remission, FTY720 treatment resulted in a significant reduction of NK cells in these tissues, although to a lesser extent (Figure 5C). Examination of spleen sections from rats responsive to FTY720 showed normal splenic histology (supplemental Figure 7A). In contrast, leukemic rats not achieving remission showed leukemic NK-cell infiltration of the red pulp and depletion of the white pulp (supplemental Figure 7A). Moreover, we found that Mcl-1 was significantly ablated in the spleens of responsive rats, indicating that it may be a unique target of FTY720 in NK-cell leukemia (supplemental Figure 7B). These data suggest that in vivo therapeutic efficacy of FTY720 may be a consequence of modulation of Mcl-1 signaling which led to resolution of leukemic cell infiltration. Previously, we demonstrated that nanoliposomal ceramide induced complete remission in this rat model by targeting survivin.17 FTY720 treatment of NKL or RNK-16 cells in vitro had no effect on level of survivin protein (data not shown). Given that FTY720 and C6-ceramide have distinct cellular targets in leukemic NK cells, we were interested in examining whether combining these 2 drugs would enhance therapeutic efficacy in leukemic NK cells in vitro. Indeed, we found that treatment with the combination of FTY720 and nanoliposomal C6-ceramide induced apoptosis with a synergistic effect in NKL cell line and with an additive effect in RNK-16 cells (supplemental Figure 8). These data provide a rationale for further investigating such combined treatment in vivo, using the preclinical Fischer rat model of NK-cell leukemia.
Figure 5.
FTY720 induces complete remission in NK-cell leukemic rats. (A) The Kaplan-Meier survival curves for normal rats after treatment with 4.5 mg/kg of FTY720 (n=16) or PBS (n=16) or leukemic rats after treatment with either 4.5 mg/kg (n=16) or PBS (n=16), were plotted. (B) Maintenance of normal white blood counts, hemoglobin values, and platelet counts in responding leukemic and normal rats treated with FTY720. Blood (200 μL) from leukemic rats treated with FTY720 (n=16) and PBS (n=16) and normal rats treated with FTY720 (n=16) and PBS (n=16) was collected every week from tail veins of the animals and placed in EDTA K2-coated tubes, then CBC (complete blood count) analysis was performed. Arrow indicates the cessation of the treatment. (C) Flow cytometry was used to identify rat leukemic NK cells, which are CD3−CD8a+. Comparison of CD3−CD8a+ NK cells isolated from multiple tissues among normal rats treated with PBS (n=16) or FTY720 (n=16), leukemic rats treated with PBS (n=16) or leukemic rats responsive to FTY720 treatment (n=5) or unresponsive to FTY720 (n=11). *P < .05, **P < .005, ***P < .005 indicates significance between leukemic rats responsive to FTY720 or unresponsive to FTY20 and leukemic rats treated with PBS (unpaired t test).
Discussion
Our studies show that FTY720 led to apoptosis in leukemic NK cells from patients with chronic NK-cell leukemia and from NK-cell leukemia cell lines in vitro as well as complete remission in a rat model of aggressive NK-cell leukemia. There is an urgent unmet need for development of new therapeutics for both NK-cell leukemia. This is particularly important for aggressive disease. Combination chemotherapy is usually not successful in achieving remission and median survival is only a few months for this fatal illness. We found that FTY720 induced apoptosis in NK-cell leukemia by 2 mechanisms: degradation of prosurvival protein Mcl-1 and altered sphingolipid metabolism resulting in accumulation of proapoptotic sphingosine. We focused first on the effect of FTY720 on expression of Mcl-1, which has been reported to play an essential survival role in T-LGL leukemia13 Interestingly, we found that Mcl-1 is also highly expressed in human leukemic NK cells while it is undetectable in normal NK cells (Figure 2A). Moreover, we also showed that efficient knockdown of Mcl-1 led to 5.5-fold increase of apoptosis in leukemic NK cells, demonstrating the important survival role of Mcl-1 in leukemic NK cells.
Because FTY720 did not affect transcription of Mcl-1, we examined whether Mcl-1 was degraded by FTY720 in leukemic NK cells. Having demonstrated this to be the case, we next examined mechanisms of degradation. A widely accepted mechanism for regulation of Mcl-1 expression after death stimulus activation involves proteosomal degradation.18 However, a proteosomal inhibitor, MG132, had no effect on the reduced levels of expression of Mcl-1 protein and apoptotic cell death produced by FTY720 treatment, suggesting that degradation of Mcl-1 by FTY720 was not through proteosomal pathway. Interestingly, we found that FTY720 was delivered to lysosome of leukemic NK cells only 2 hours after treatment. In addition, a lysosomal inhibitor, bafilomycin A1, was able to block both Mcl-1 reduction and cell death induced by FTY720, suggesting that the lysosome is the organelle tightly linked with FTY720's induction of apoptosis in leukemic NK cells. Lysosomes are acidic organelles containing a wide spectrum of hydrolytic enzymes that have a major role in intracellular protein recycling. Cathepsins are the most abundant lysosomal enzymes. Lysosomes and lysosomal cathepsins are believed to be involved in terminal steps of necrotic and autophagic cell death,19 and more recently also in apoptosis.20 A key step in this pathway is LMP, which is followed by the release of lysosomal cathepsins into the cytosol.20 Once cathepsin is released from the lysosomes into the cytoplasm, it may cleave some target proteins to enhance cell death.21 Antiapoptotic proteins including Mcl-1 are substrates for cathepsin.22 Therefore, we hypothesize that release of lysosomal cathepsins on LMP might be responsible for Mcl-1 degradation by FTY720 in leukemic NK cells. Indeed, we showed that cathepsin B was released to cytosol on FTY720 treatment (Figure 3C). Alternatively, Mcl-1 plays a role in the stabilization of the mitochondrial membrane via its binding and sequestering of proapoptotic Bcl-2 family members.23,24 Thus, the induced lysosomal destabilization by FTY720 in leukemic NK cells might result in cleavage of the proapoptotic Bcl-2 protein Bid,25 followed by a decrease in the antiapoptotic protein Mcl-1 and mitochondrial membrane permeabilization.
To further investigate how FTY720 triggers lysosomal degradation pathway, we examined the influence of FTY720 on oxygen species (ROS) because of the fact that lysosomes are particularly sensitive toward oxidative stress.26 Production of ROS after FTY720 treatment had been shown previously in hepatocellular carcinoma.27 Indeed, we showed that a free radical scavenger NAC restored Mcl-1 expression and rescued FTY720-induced apoptosis in leukemic NK cells. These data suggest that FTY720 regulation of Mcl-1 expression occurs through a novel mechanism involving ROS production.
We were also interested in examining sphingolipid metabolism after FTY720 treatment of leukemic NK cells. Sphingolipids are key components of the lipid bilayer of membranes and also are vital second messengers involved in cell survival, proliferation, and apoptosis within a cell.28 The balance between sphingolipid metabolites S1P and ceramide/sphingosine (CER/SPH), the upstream precursors of S1P, is believed to provide a rheostat mechanism that determines whether a cell proliferates or undergoes apoptosis.29 In contrast to the growth-inhibitory and proapoptotic effects of ceramide and sphingosine, S1P, which is produced by phosphorylation of sphingosine in a reaction catalyzed by sphingosine kinase, promotes cell growth and survival.30,31 Previously, it had been shown that high micromolar doses of FTY720 can inhibit sphingosine kinase 1 activity, leading to accumulation of sphingosine.14,32 Depending on the cell lines and experimental procedures used, sphingosine has been shown to mediate apoptosis as a putative second messenger via activation of JNK and p38 and inhibition of ERK/MAPK, PKC, and Akt.33–37 Importantly, we found increased levels of sphingosine but not ceramide, in leukemic NK cells treated with FTY720. Sphingosine led to increase in apoptosis in leukemic NK cells but to a much lesser extent than treatment with the same dose of FTY720 (Figure 4B-C). These data suggest that efficacy of FTY720 in NK-cell leukemia might also be attributed in part to altered sphingolipid metabolism.
We have previously shown that targeting survivin by nanoliposomal ceramide induced complete remission in F344 rat NK-cell leukemia model.17 Mitochondrial survivin was a critical ceramide target responsible for the efficacy of liposomal ceramide therapeutics.17 Interestingly, FTY720 targeted Mcl-1 but not survivin expression, indicating a unique mechanism causing apoptosis of leukemic NK cells. Furthermore, sphingolipidomics profiling (Figure 4A) showed that ceramide levels were not altered by FTY720 treatment, suggesting FTY720 therapeutics in NK-cell leukemia is independent of ceramide/survivin signaling. Because these therapeutics target different components of survival signaling and because we found that treatment with both FTY720 and C6-ceramide nanoliposome had additive/synergistic effects on leukemic NK cells, it will be interesting to investigate such combined therapies in this animal model of NK-cell leukemia.
Supplementary Material
Acknowledgments
The authors thank Della Reynolds (National Cancer Institute) for the advice on the culture of RNK-16 cells; Nate Sheaffer and David Stanford (Flow Cytometry Core Facility), Robert Brucklacher (Functional Genomics Core Facility), and personnel of the Organic Synthesis Core Facility and Biostatistics Core Facility (Penn State Hershey Cancer Institute/Milton S. Hershey Medical Center) for their technical assistance; and Jenny Dunkinson for help during the preparation of the manuscript.
This work was supported by National Institutes of Health Grants CA098472 and CA133525 (T.P.L).
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: A.L. performed the majority of the in vitro and in vivo research and analyzed the data; K.B. assisted with PP2A and lipidomics experiments; T.F. performed lipidomics experiments; K.B., L.Q.Z., and Y.L. performed the lentiviral Mcl-1 shRNA knockdown experiment; S.-F.T., R.W., J.Y., C.A., and X.L. assisted with the animal studies; L.R., A.D., A.R., K.L., and L.H. assisted with Western blot experiments; R.W., M.V.S., and L.Q.Z. assisted with apoptosis assays and MTT studies; S.-F.T. assisted with immunohistochemical studies; K.B., N.R.J., and K.T.B. assisted with isolation of PBMCs or NK cells from patients or normal donors; J.L. performed the statistics studies; D.D. synthesized FTY720 and reviewed drafts of the manuscript; H.-G.W., M.K., and S.A. assisted in planning these studies and reviewed drafts of the manuscripts; and T.P.L. and X.L. designed and organized the experiments and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Xin Liu, Penn State Hershey Cancer Institute, Experimental Therapeutics-CH74, Rm 4401, 500 University Dr, PO Box 850, Hershey, PA 17033-0850; e-mail: Xliu2@hmc.psu.edu.
References
- 1.Lim MS, de Leval L, Quintanilla-Martinez L. Commentary on the 2008 WHO classification of mature T- and NK-cell neoplasms. J Hematop. 2009;2:75–76. doi: 10.1007/s12308-009-0034-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lamy T, Loughran TP. Large granular lymphocyte leukemia. Cancer Control. 1998;5(1):25–33. doi: 10.1177/107327489800500103. [DOI] [PubMed] [Google Scholar]
- 3.Sokol L, Loughran TP., Jr Large granular lymphocyte leukemia. Oncologist. 2006;11(3):263–273. doi: 10.1634/theoncologist.11-3-263. [DOI] [PubMed] [Google Scholar]
- 4.Cheung MM, Chan JK, Wong KF. Natural killer cell neoplasms: a distinctive group of highly aggressive lymphomas/leukemias. Semin Hematol. 2003;40(3):221–232. doi: 10.1016/s0037-1963(03)00136-7. [DOI] [PubMed] [Google Scholar]
- 5.Gold R. Oral therapies for multiple sclerosis: a review of agents in phase III development or recently approved. CNS Drugs. 2011;25(1):37–52. doi: 10.2165/11539820-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 6.Neviani P, Santhanam R, Oaks JJ, et al. FTY720, a new alternative for treating blast crisis chronic myelogenous leukemia and Philadelphia chromosome-positive acute lymphocytic leukemia. J Clin Invest. 2007;117(9):2408–2421. doi: 10.1172/JCI31095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Q, Zhao X, Frissora F, et al. FTY720 demonstrates promising preclinical activity for chronic lymphocytic leukemia and lymphoblastic leukemia/lymphoma. Blood. 2008;111(1):275–284. doi: 10.1182/blood-2006-10-053884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hinterding K, Albert R, Cottens S. First asymmetric synthesis of chiral analogues of the novel immunosuppressant FTY720. Tetrahedron Letters. 2002;43:8095–8097. [Google Scholar]
- 9.Lamy T, Liu JH, Landowski TH, Dalton WS, Loughran TP., Jr Dysregulation of CD95/CD95 ligand-apoptotic pathway in CD3(+) large granular lymphocyte leukemia. Blood. 1998;92(12):4771–4777. [PubMed] [Google Scholar]
- 10.Epling-Burnette PK, Bai F, Wei S, et al. ERK couples chronic survival of NK cells to constitutively activated Ras in lymphoproliferative disease of granular lymphocytes (LDGL). Oncogene. 2004;23(57):9220–9229. doi: 10.1038/sj.onc.1208122. [DOI] [PubMed] [Google Scholar]
- 11.Fox TE, Bewley MC, Unrath KA, et al. Circulating sphingolipid biomarkers in models of type 1 diabetes. J Lipid Res. 2011;52(3):509–517. doi: 10.1194/jlr.M010595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37(8):911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- 13.Epling-Burnette PK, Liu JH, Catlett-Falcone R, et al. Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest. 2001;107(3):351–362. doi: 10.1172/JCI9940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee WJ, Yoo HS, Suh PG, Oh S, Lim JS, Lee YM. Sphingosine mediates FTY720-induced apoptosis in LLC-PK1 cells. Exp Mol Med. 2004;36(5):420–427. doi: 10.1038/emm.2004.54. [DOI] [PubMed] [Google Scholar]
- 15.Reynolds CW, Bere EW, Jr, Ward JM. Natural killer activity in the rat. III. Characterization of transplantable large granular lymphocyte (LGL) leukemias in the F344 rat. J Immunol. 1984;132(1):534–540. [PubMed] [Google Scholar]
- 16.Thomas J, Haseman JK, Goodman JI, Ward JM, Loughran TP, Jr, Spencer PJ. A review of large granular lymphocytic leukemia in Fischer 344 rats as an initial step toward evaluating the implication of the endpoint to human cancer risk assessment. Toxicol Sci. 2007;99(1):3–19. doi: 10.1093/toxsci/kfm098. [DOI] [PubMed] [Google Scholar]
- 17.Liu X, Ryland L, Yang J, et al. Targeting of survivin by nanoliposomal ceramide induces complete remission in a rat model of NK-LGL leukemia. Blood. 2010;116(20):4192–4201. doi: 10.1182/blood-2010-02-271080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nijhawan D, Fang M, Traer E, et al. Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev. 2003;17(12):1475–1486. doi: 10.1101/gad.1093903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290(5497):1717–1721. doi: 10.1126/science.290.5497.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guicciardi ME, Deussing J, Miyoshi H, et al. Cathepsin B contributes to TNF-alpha-mediated hepatocyte apoptosis by promoting mitochondrial release of cytochrome c. J Clin Invest. 2000;106(9):1127–1137. doi: 10.1172/JCI9914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang H, Zhong C, Shi L, Guo Y, Fan Z. Granulysin induces cathepsin B release from lysosomes of target tumor cells to attack mitochondria through processing of bid leading to necroptosis. J Immunol. 2009;182(11):6993–7000. doi: 10.4049/jimmunol.0802502. [DOI] [PubMed] [Google Scholar]
- 22.Droga-Mazovec G, Bojic L, Petelin A, et al. Cysteine cathepsins trigger caspase-dependent cell death through cleavage of bid and antiapoptotic Bcl-2 homologues. J Biol Chem. 2008;283(27):19140–19150. doi: 10.1074/jbc.M802513200. [DOI] [PubMed] [Google Scholar]
- 23.Leu JI, Dumont P, Hafey M, Murphy ME, George DL. Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol. 2004;6(5):443–450. doi: 10.1038/ncb1123. [DOI] [PubMed] [Google Scholar]
- 24.Adams JM, Cory S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene. 2007;26(9):1324–1337. doi: 10.1038/sj.onc.1210220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blomgran R, Zheng L, Stendahl O. Cathepsin-cleaved Bid promotes apoptosis in human neutrophils via oxidative stress-induced lysosomal membrane permeabilization. J Leukoc Biol. 2007;81(5):1213–1223. doi: 10.1189/jlb.0506359. [DOI] [PubMed] [Google Scholar]
- 26.Brunk UT, Dalen H, Roberg K, Hellquist HB. Photo-oxidative disruption of lysosomal membranes causes apoptosis of cultured human fibroblasts. Free Radic Biol Med. 1997;23(4):616–626. doi: 10.1016/s0891-5849(97)00007-5. [DOI] [PubMed] [Google Scholar]
- 27.Hung JH, Lu YS, Wang YC, et al. FTY720 induces apoptosis in hepatocellular carcinoma cells through activation of protein kinase C delta signaling. Cancer Res. 2008;68(4):1204–1212. doi: 10.1158/0008-5472.CAN-07-2621. [DOI] [PubMed] [Google Scholar]
- 28.Sabourdy F, Kedjouar B, Sorli SC, et al. Functions of sphingolipid metabolism in mammals–lessons from genetic defects. Biochim Biophys Acta. 2008;1781(4):145–183. doi: 10.1016/j.bbalip.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 29.Ogretmen B, Hannun YA. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat Rev Cancer. 2004;4(8):604–616. doi: 10.1038/nrc1411. [DOI] [PubMed] [Google Scholar]
- 30.Pi X, Tan SY, Hayes M, et al. Sphingosine kinase 1-mediated inhibition of Fas death signaling in rheumatoid arthritis B lymphoblastoid cells. Arthritis Rheum. 2006;54(3):754–764. doi: 10.1002/art.21635. [DOI] [PubMed] [Google Scholar]
- 31.Bektas M, Jolly PS, Muller C, Eberle J, Spiegel S, Geilen CC. Sphingosine kinase activity counteracts ceramide-mediated cell death in human melanoma cells: role of Bcl-2 expression. Oncogene. 2005;24(1):178–187. doi: 10.1038/sj.onc.1208019. [DOI] [PubMed] [Google Scholar]
- 32.Tonelli F, Lim KG, Loveridge C, et al. FTY720 and (S)-FTY720 vinylphosphonate inhibit sphingosine kinase 1 and promote its proteasomal degradation in human pulmonary artery smooth muscle, breast cancer and androgen-independent prostate cancer cells. Cell Signal. 2010;22(10):1536–1542. doi: 10.1016/j.cellsig.2010.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hung WC, Chang HC, Chuang LY. Activation of caspase-3-like proteases in apoptosis induced by sphingosine and other long-chain bases in Hep3B hepatoma cells. Biochem J. 1999;338(Pt 1):161–166. [PMC free article] [PubMed] [Google Scholar]
- 34.Jarvis WD, Fornari FA, Jr, Auer KL, et al. Coordinate regulation of stress- and mitogen-activated protein kinases in the apoptotic actions of ceramide and sphingosine. Mol Pharmacol. 1997;52(6):935–947. doi: 10.1124/mol.52.6.935. [DOI] [PubMed] [Google Scholar]
- 35.Frasch SC, Nick JA, Fadok VA, Bratton DL, Worthen GS, Henson PM. p38 mitogen-activated protein kinase-dependent and -independent intracellular signal transduction pathways leading to apoptosis in human neutrophils. J Biol Chem. 1998;273(14):8389–8397. doi: 10.1074/jbc.273.14.8389. [DOI] [PubMed] [Google Scholar]
- 36.Hannun YA, Loomis CR, Merrill AH, Jr, Bell RM. Sphingosine inhibition of protein kinase C activity and of phorbol dibutyrate binding in vitro and in human platelets. J Biol Chem. 1986;261(27):12604–12609. [PubMed] [Google Scholar]
- 37.Chang HC, Tsai LH, Chuang LY, Hung WC. Role of AKT kinase in sphingosine-induced apoptosis in human hepatoma cells. J Cell Physiol. 2001;188(2):188–193. doi: 10.1002/jcp.1108. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.