Abstract
To insure its survival, the human immunodeficiency virus (HIV), the causative agent of acquired immunodeficiency syndrome (AIDS), utilises more than one gate to enter a target cell. This strategy makes it more difficult for the immune system to defeat the virus. This has also made it difficult for scientists working in the HIV research to understand how exactly HIV infects a susceptible human cell. Such understanding will no doubt open many gates for scientists to vanquish the virus by designing agents that can block HIV entry into target cells, thus rendering the virus vulnerable to immune destruction. This will also help in understanding the replication of the virus and the pathogenesis of the disease. The present article briefly describes the past and current thinking of how HIV-1 infects a susceptible human cell, and the dilemma scientists are facing when studying the infectivity of this virus and applying current knowledge to design agents that can block HIVs entering target cells. In the context of our own experience with HIV infectivity in vitro, the present article will also address the behaviour of different HIV isolates, both laboratory-adopted and clinical (primary) isolates in culture and the difficulties in performing HIV infectivity testing. Moreover, our experience of peripheral blood mononuclear cells’ (PBMCs) susceptibility to HIVs infection in vitro is discussed.
Keywords: HIV, AIDS, infection, immune, virus, susceptible cell
Acquired immunodeficiency syndrome (AIDS) was first described in 1981 as a group of clinical signs and symptoms caused by the impairment of the immune system, mainly the cellular immune response.1,2 The conditions defining AIDS were recognised by the Centre for Disease Control (CDC), in Atlanta, Georgia (USA), before the identification of the human immunodeficiency viruses (HIVs) as the causative agents for the syndrome. The CDC classification for AIDS, list numerous opportunistic infections and neoplasms (cancers) which in the presence of HIV infection constitute an AIDS diagnosis.3 In addition, a CD4+ T-cell count below 200 per mm3 in the presence of HIV infection constitutes an AIDS diagnosis. People with AIDS often suffer infections of the lungs, brain, eyes and other organs, and frequently suffer debilitating weight loss, diarrhoea and are vulnerable to a type of cancer known as Kaposi’s sarcoma. Although there is no cure for AIDS at present, multiple drug therapy dramatically improves the quality of life for most people with AIDS.4 The HIVs are well accepted as the causative agents of AIDS.5–8
To be able to defeat the HIVs and design the proper prevention tools, basic scientific knowledge on how exactly these viruses infect human susceptible cells is essential. Although the literature is immense on this topic, in this short article I will briefly review our current understanding of how HIVs infect human susceptible cells and the application of this knowledge in designing drugs that can inhibit HIVs entering susceptible human cells.
THE HUMAN IMMUNODEFICIENCY VIRUSES (HIVS)
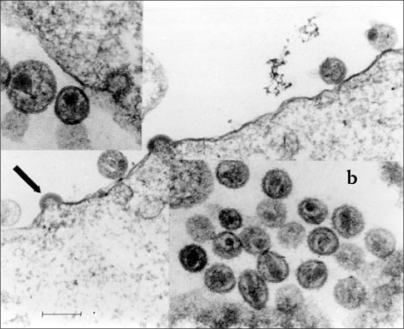
The HIVs are members of the lentivirus subgroup of the Retroviridae family. They are RNA viruses that posses the enzyme reverse transcriptase (RT). This enzyme allows the virus to reverse-transcribe its own RNA, and the resultant complementary DNA (cDNA) is incorporated into the cellular DNA of the host. Two main types of HIV exist: HIV-1 and HIV-2, which differ in genomic structure and antigenicity as well as in their latent period as pathogenicity. HIV-1 was identified, as the causative agent of AIDS, by three different groups in the early eighties. These were Montagnier’s at the Pasteur Institute in Paris (France), Gallo’s at the National Institute of Health (NIH) USA, and Levy’s at the University of California San Francisco (UCSF), USA. The HIVs were initially called LAV (lymphadenopathy-associated virus), HTLV-III-human T lymphotropic virus and ARV (AIDS associated retroviruses).5,6,8 In 1986 a second virus (HIV-2) was identified,9 and the original was renamed HIV-1 to distinguish them. The diameter of the HIV virion is approximately 100 nm and electron microscopy studies have revealed a characteristic cylindrical core in HIV particles surrounded by an outer envelope [Figure 1].
Figure 1.
Electron micrograph of HIV-1 and HIV-2 (MN and ROD).
HIV-1 (MN) virions are shown budding (arrow). HIV-2 (ROD) virions are shown in (b) (above). Magnification is 100,000 x. Scale bar =100nm.
This electron microscopy was kindly performed by Dr. Robert Dourmashkin.
HIV-1
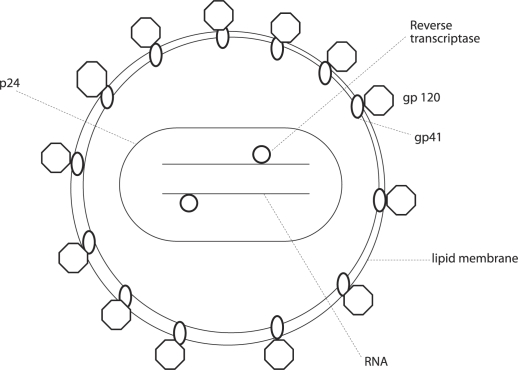
The inner core of HIV-1 consists of p24 protein (capsid). Inside this capsid are two copies of single stranded genomic RNA and the enzymes required for the initial stages of HIV-1 replication. These include the reverse transcriptase (p51/66), integrase (p32) and protease (p22). The inner core also contains p7 nucleic acid binding protein and p9 gag binding protein. The inner core is surrounded by a p17 myristylated gag matrix protein, thought to be essential for maintaining the structure of the virus or stabilising the exterior and interior components of the virion. The p17 protein is bounded by a lipid bilayer membrane (the outer envelope). This outer envelope contains spikes made up of gp120 and gp41. The gp120 contains the receptor binding region involved in infectivity as well as in syncytia formation (a major cytopathic effect of HIVs in vitro) and the gp41 is a transmembrane segment which is important for virus fusion.10,11,12 The HIV-1 lipid bilayer also contains various host cell proteins including HLA class I and II that are acquired during virus budding (Figure 1).
HIV-2
Although the general structure of HIV-2 is similar to that of HIV-1, differences within the genomic structure exist and genetic regulation of HIV-2 also differs from that of HIV-1.9,11,12 This article will be referring mainly to HIV-1.
Subtypes of HIVs
Now it is well established that there exist many subtypes of HIVs. The HIV-1-M (main) group subtypes are phylogenetically associated groups or clades of HIV-1 sequences, and are labelled A1, A2, B, C, D, F1, F2, G, H, J and K. The sequences within any one subtype or sub-subtype are more similar to each other than to sequences from other subtypes throughout their genomes. “O” is the “outlier” group, and group N is also a very distinctive form of the virus that is Non-M, Non-O (also sometimes referred to as the “new” group). For HIV-2, there exist A, B, C, F or G clades (formerly known as “subtypes”, now referred to as “groups”). The nomenclature of HIVs is becoming even more difficult with the introduction of Recombinants and Circulating Recombinant Forms as a result of inter-subtype recombinant genomes of different HIV clades. The existence of many subtypes of HIVs make it extremely difficult for vaccinologists to design the proper vaccine against HIVs, since certain subtypes circulate more than others in different populations. For example subtype B of HIV-1 is more common in western countries than subtypes C and D. HIVs are also named according to how they behave in culture producing cytopathic effects: sysncytium inducing isolates (SI) and none syncytium inducing isolates (NSI). Moreover, HIVs can also be called macrophage tropic (M-trpic), T cell line tropic (T-tropic), or dual tropic, when they are able to infect macrophages, T cells and both macrophage and T cells, respectively.
Cell tropism in HIVs
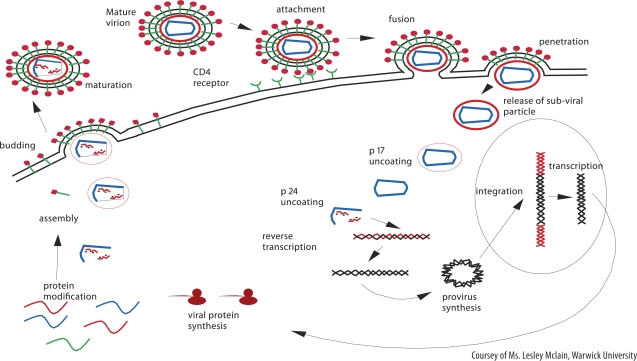
HIVs are termed retroviruses because they “reverse” the normal flow of the genetic information, i.e. RNA is reverse-transcribed to DNA. Until the mid nineties, it was considered that HIV-1 enters its host cell through the attachment of the envelope glycoproteins (gp120) to the CD4 molecules (receptors), found on T-helper cells, monocytes and macrophages.10 HIVs can also enter cells by other means, for example, through the Fc receptors of antibodies and complement receptors CR2 and CR3.11,12 After gp120 binds to a CD4 molecule, a conformational change is induced in the gp120, leading to its dissociation from the viral membrane. This results in the exposure of the amino terminal hydrophobic domain of the gp41which initiates the fusion process. The HIV, then, enters its host cell via membrane fusion, a pH independent process.13,14,15 The virus sheds its protein coat after entry into the cell, exposing the RNA core [Figure 2]. The enzyme reverse transcriptase is activated and transcribes the RNA into complementary DNA. The RNA is removed by ribonuclease H activity, and a second DNA strand is synthesised complementary to the first. This double-stranded DNA is integrated into the genome of the host cell by the viral integrase, with the potential for viral production at any time. The integrated DNA is termed a provirus and this provirus DNA can be transcribed into mRNAs, which direct the synthesis of viral proteins.12
Figure 2.
The life cycle of HIVs
V3 loop of the glycoprotein (gp) 120
Before HIV is able to gain entry into a target susceptible cell, the third hypervariable region (V3) loop of the surface glycoprotein gp120 molecule has been implicated as an important factor for cell tropism and for cell fusion. The determinants of HIV tropism can be mapped to the viral envelope gene,16–22 thus indicating that tropism is restricted at the level of virus entry. A significant finding was that a major determinant of HIV-1 tropism is the V3 region of the surface gp120.16–18 The HIV-1 envelope gene is approximately 2.5 Kb in length and gp120 has a 1.6 Kb nucleotide sequence. The gp120 is divided into six constant (C1–C6) and five hyper-variable regions (V1–V5).19,20 Studies have already shown that mutations (substitutions) in the V3 loop are essential in cellular tropism and in the antigenicity of gp120.21 The V3 loop, known as the principal neutralising domain (PND), is considered to be the major immunodominant region of the gp120 for both HIV-122,23 and HIV-2.24 This domain induces neutralising antibodies and is the target for cytotoxic T lymphocyte killing.25 Neutralising antibodies are able to block infection in vitro by blocking fusion of the virus.23 The acquisition of the syncytium inducing (SI) phenotype is linked to substitutions of acidic or neutral amino acids by more basic amino acids in the V3 loop.26
The V3 loop does not seem to play an important role in CD4 binding, since V3 specific antibodies do not affect the attachment of gp120 to CD4. However, the V3 loop is essential for virus infection, since V3 loop specific antibodies can block fusion and entry of HIV to susceptible cells.27
The V3 loop typically begins at amino acids number 300 and consists of between 33 to 35 amino acids bounded by disulphide linkage. At the tip of the loop is the sequence Gly-Pro-Gly-Arg (GPGR), known to be the most conserved region among HIV-1 clade B isolates.28 While the GPGR crown of the V3 loop of HIV-1 is conserved within all clade B isolates, the equivalent in HIV-2, FSHQ, seem to be conserved among HIV-2 isolates. A single amino acid substitution in the HIV-1 V3 loop has already been shown to change an isolate from non syncytium inducing (NSI) to a syncytium inducing (SI) phenotype.21
Chemokines are the co-receptors for HIVs entry
Nowadays it is well understood that the CD4 molecule alone is not sufficient to render a target cell permissive for fusion with virions (see below). Although the main receptors for HIV-1 entry into target cells is the CD4 molecules, the seven trans-membrane spanning G protein chemokine receptor family are now accepted as important co-receptors for virus entry.14,29,30,31,32
Many chemokine co-receptors were identified, including CCR-5 and fusin, and known to act as secondary receptors for virus entry.33,34,35 Fusin is expressed on a wide range of human cell lines; those which do not express fusin are not infected by T cell-tropic HIV-1. Importantly, cells expressing both CD4 and fusin remained refractory to macrophage (M)-tropic HIV envelope mediated fusion, consistent with the earlier prediction that fusion cofactors for T- and M-tropic HIV-1 strains would be distinct moieties. Furthermore, a CXC chemokine (SDF-1), has subsequently been identified as a ligand which both binds fusin and specifically blocks infection by T-tropic but not M-tropic HIV-1 strains.36,37 Based on the identification of its chemokine ligand, fusin was renamed CXCR-4.
The discovery that CXCR-4—whose closest known homologous is the IL-8 receptor (CXCR-2)—constitutes a functional co-receptor for T-tropic HIV-1 strains, added significance to the earlier finding that the C-C chemokines MIP-1-alpha, MIP-1-beta and RANTES are able to block infection of CD4+ human T-cells by M-tropic but not T-tropic HIV-1.38 Thus, since certain chemokines are known to bind several of the seven transmembrane receptors, it became likely that the discovery of a chemokine receptor that is recognised by all three of these chemokines would also result in the identification of an M-tropic HIV-1 specific co-receptor.
Role of chemokine co-receptors
The interaction of HIVs with a target cell is initially with CD4, thereby inducing a conformational change in gp120, which facilitates subsequent binding of envelope to the co-receptor.33,34 It is likely that the virus binding site or sites on the co-receptor involve multiple extra-cellular domains, and that within each of these domains multiple amino acids contribute to co-receptor function. While this complexity has largely frustrated attempts to define precise virus binding sites, studies of chimeric and mutant co-receptors have, to some extent, illuminated our understanding of how HIV interacts with co-receptors and suggested that several, functionally redundant, interactions between co-receptor and virus envelope are likely to occur. This has implications for the feasibility of using co-receptor targeted inhibitors of virus entry as therapies.39
Co-receptor recognition
Culturing virus in the laboratory imposes different selective pressures on viral populations that are likely to impact on the nature of their interaction with receptors. In addition, there are examples of very minor envelope sequence changes drastically influencing co-receptor recognition,39,40,41,42 thus different results are likely to be obtained with cloned versus uncloned forms of the same virus isolate. The relative expression levels of co-receptors on transfected cells, as compared to the natural targets of virus infection, remain largely unexplored and clearly affect the efficiency of infection. It was noted that over-expression of some chemokine receptors render cells permissive for viral envelope induced cell fusion, while cells expressing lower levels of the same co-receptor remain resistant to infection by cell-free virus.43 It is clear that there is considerable plasticity in co-receptor usage, both in terms of the viruses ability to use one or more of a number of “optional” co-receptors and the ability of strains to use a given co-receptor in different ways.40,42
Cell lines and in-vitro infection with HIVs
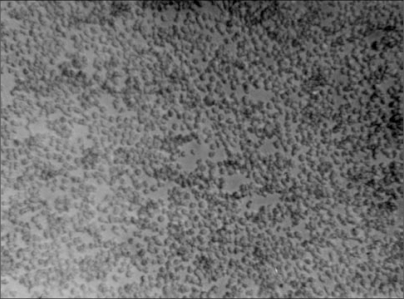
Our experience with T cell lines, namely, C8166, H9 and CEM which we tested and compared in relation to infectivity by four different HIVs, showed variation in both the time at which the HIV cytopathic effects (syncytia formation) were first observed, and the number of syncytia formed.44,45,46 Moreover, the replication capacity of each virus in each of the cell lines differed widely. The CEM cell line was shown to be the least infectable, whereas C8166 was very susceptible to infection by the three laboratory-adopted HIVs, MN, CBL-20, ROD and one fresh clinical HIV-1 isolate (H995). Although other factors may be involved in the susceptibility of the C8166 cells to HIV infection, the characteristic tendency of these cells to clump in culture may facilitate the infectivity process. The C8166 cell line was shown to be the most susceptible to infection by the laboratory adopted HIV strains MN, ROD and CBL-20 with high rates of replication. The C8166 cells showed large syncytia (balloon shaped) early in culture usually by day two post infection compared with other cell lines whilst the number of syncytia formed depended directly on the concentration of the virus [Figure 3].
Figure 3a.
Normal healthy H9 cells.
The H9 cell line was also infectable by all four mentioned HIVs. However, it was not as susceptible as C8166 in terms of the number of syncytia observed and the levels of viral replication. The CEM cell line was the least infectable showing very few syncytia at the highest concentration of the MN strain (TCID50 of approximately 105/ml). All these experiments, performed on cell lines, were reproducible confirming that the syncytium inducing ability of each HIV strain was a stable property for that particular virus. This is important in studies when infectivity results are compared between different experiments. Due to the differences in the behaviour of these cell lines to infection by each virus, and because these cell lines differ in their human leukocyte antigens (HLA) type, it is possible that HLA may have a role to play in HIV infectivity. The HLA-A1 allele, expressed on the surface of the C8166 cells, is believed to be associated with progression of AIDS from in vivo studies. In contrast, the CEM cell line which expresses none (or very low levels) of HLA class II DR antigens when activated12 showed very small syncytia which took longer to develop compared to the other cell lines (C8166 and H9). Thus, these results on cell lines support the possibility of a role for the HLA in the HIV infectivity process. Furthermore, we have shown that HLA do correlate, although weakly, with in vitro high or low HIV virus replication.44,45,46 Our in vitro results showed clearly that certain HLA specificities were significantly correlated in vitro with infectivity by both HIV-1s and HIV-2s and that each isolate had its own correlation with HIV infection.44,45 This may partly explain the differences in the in vivo results on the association of HLA with certain features of AIDS and the difficulty in confirming such correlation. However, more studies in vitro and in vivo on a large scale and on groups who are at high risk or low risk need to be addressed with the identification of the HIV isolate that is predominant in each individual patient. These studies will characterise better those HLA specificities that are important for infectivity by HIV-1 and HIV-2. The existence of a significant HLA association with all the known HIV-1 and all HIV-2 isolates infecting human populations may lead to the identification of important immunodominant epitopes that can be useful in vaccine development against the different clades (subtypes) of HIVs. There is a possibility that HLA are not the actual genes responsible for susceptibility or resistance to HIV infection, rather other, as yet unidentified genes closely linked to HLA, or masked by the HLA, could be the real genes determining susceptibility or resistance to HIV. This can be revealed only by intensive future research work and by better knowledge and understanding of the genes that are linked to the HLA molecules.45
While many immortalised human T-cell lines can be infected by laboratory adapted HIV-1 and HIV-2 strains, other CD4 positive cell lines are sometimes selectively permissive only for specific virus types.32 From our laboratory experience, when dealing with HIV-1 and 2, laboratory-adopted strains compared to fresh clinical (primary) isolates, heterogeneity in tropism is evident within a given virus type. For HIV-1, virtually all strains replicate efficiently in CD4+ peripheral blood mononuclear cells (PBMCs). However, many freshly isolated or primary strains are also able to propagate efficiently in primary macrophages but not in immortalised T-cell lines and are thus termed M-tropic. Conversely, other strains, particularly those which have been adapted to growth in immortalised T-cell lines (T-tropic), generally do not efficiently infect primary macrophages.47,48,49,50 Other groups of HIV isolates (termed dual-tropic) are able to replicate well in both T-cell lines and primary macrophages in addition to PBMCs.51,52,53 Dual-tropic and T-tropic strains induce cell fusion in primary and immortalised T-cells and are also referred to as syncytium-inducing (SI). The V3 loop is an excellent candidate for being a component of a co-receptor binding site. The V3 loop is not the sole determinant of co-receptor recognition. In some cases, alternative tropisms can be mapped to non-V3 sequences.20,21,22
HIVs affect on receptor usage
To speculate as to why HIVs have evolved plasticity in their interactions with co-receptors is important. The ability to use multiple functionally redundant contacts with co-receptors could conceivably facilitate immunological escape.54–60 Thus, in the face of a neutralising antibody response (which, in significant part, is directed against V3 sequences,61–65 a major modulator of co-receptor interaction), the selection of variants with altered envelope sequences would be permitted without compromising the ability of the virus to use a given co-receptor.39 Furthermore, changes in envelope sequence, that enable the virus to use additional co-receptors while retaining the ability to interact with CCR-5, could be tolerated. It appears that dual-tropic strains that use both CCR-5 and CXCR-4 are less tolerant of perturbations in CCR-5 sequence than are M-tropic strains,66,67 suggesting that acquisition of the ability to utilise CXCR-4 might involve the sacrifice of a degree of functional redundancy and/or affinity in the envelope/CCR-5 interaction.39
During the early, asymptomatic phase of HIV-1 infection M-tropic strains predominate. It is quite possible that this is the simple consequence of a ‘founder effect’: It is likely that the major route of transmission of HIV-1 is via infection using CCR-5 as a co-receptor, given the observation that CCR5 delta32 homozygous individuals are much less likely to be HIV infected than those with other genotypes.39 However, why do strains that also use CXCR-4 tend to arise only late in the course of infection, when in many cases they retain the ability to use CCR-5? While it has often been noted that SI viruses are associated with poor prognosis it is important to emphasise that the occurrence of SI viruses that can use CXCR-4 (and usually CCR-5) is only a correlate of disease progression.64,65 Although it is possible that a presumptive expansion in tropism might accelerate the course of HIV-1 disease, particularly since CXCR-4 is expressed on the naive T-cells subset from which CCR-5 is largely absent,68 it is equally possible that CXCR-4 utilising viruses arise as a consequence of, rather than being the cause of immunosuppression. Viruses isolated from a laboratory worker who was accidentally infected with the exclusively T-tropic, IIIB strain became M-tropic with time.69 It is known that the selective pressures on co-receptor utilisation are different in vivo as opposed to in vitro. At least two scenarios could be envisaged that provide CCR-5 utilising viruses with a selective advantage: Firstly, it is conceivable that major selective influences are the result of envelope directed neutralising immune responses, which could impose a restriction on the nature of co-receptor utilisation. Indeed, neutralisation phenotype and viral tropism (and therefore, presumably, co-receptor selection) are not independent properties of the viral envelope.70 A general observation is that primary HIV-1 isolates (which tend to use CCR-5 but not CXCR-4) are somewhat more resistant to neutralisation by naturally occurring HIV-1 antibodies than are laboratory adapted strains.71 Thus, it is possible that immunological constraints are placed on envelope sequence and conformation that could negatively influence the ability of the virus to use CXCR-4 as a co-receptor.39 Therefore, the occurrence of CXCR-4 utilising variants late in the course of disease might be a reflection of compromised ability of the infected individuals to mount immune responses to new variants capable of utilising CXCR-4 that would have been readily neutralised earlier during the infection. Conversely, a selective advantage for HIVs that use CCR-5 may be immune response-independence. An equally plausible hypothesis is that CXCR-4 utilising strains become prevalent only late in disease as a result of the selective depletion of CD4+ memory T-cells (the subset that preferentially expresses CCR-5). These are replaced at a high rate by naive counterparts which (at least in uninfected individuals), express higher levels of CXCR-4 than of CCR-5,68 thus providing T- or dual-tropic strains with a selective advantage. Regardless of the selective pressures on co-receptor usage, the occurrence of SI viruses in only about 50% of patients with advanced disease indicates that the ability of the virus to use CXCR-4 is by no means necessary for the onset of immune suppression.72,73,74,75
Glycoprotein-120 co-receptor interactions
It is known that gp120 CD4 interaction induces conformational changes in envelope, which expose previously concealed epitopes.76 These changes are not sufficient to induce membrane fusion; rather they are likely to facilitate co-receptor binding. Presumably co-receptor binding results in additional conformational modification, ultimately resulting in exposure of the gp41 N-terminal fusion peptide. The physiological function of seven trans-membrane receptors is to transduce signals via coupling to G-proteins. In several cases where post signalling events have been studied (using predominantly the beta-2-adrenergic receptor as a prototype) signalling is rapidly followed by receptor phosphorylation (mediated by specific G-protein coupled receptor kinases). Phosphorylation results in desensitisation of the receptor and recognition by arrestins that are necessary for the subsequent internalisation and recycling of the phosphorylated receptor.77 Other scientists have investigated whether any of these processes are coupled to the function of CCR-5 as an HIV-1 co-receptor.39,78,79,80 It may be possible that gp120/CCR-5 interaction might result in signalling, even though this may not be a necessary event for HIV-1 entry to occur.
Clearly much remains to be learned about the nature of viral envelope–co-receptor interactions. The functional sequences of the first wave of co-receptors are only partially characterised, and more data relating to those of the newly identified co-receptors is awaited. Moreover, the extent to which non-V3 sequences influence recognition of the expanding array of co-receptors is yet to be determined. It is also unclear to what extent co-receptors other that CCR-5 and CXCR-4 are truly utilised in vivo. For example, do viruses present in CCR-5 delta32 homozygotes utilise exclusively CXCR-4? Is the frequency of viruses using alternative receptors more frequent in individuals who express low levels of CCR-5? Virtually nothing is known about how the expression of co-receptor genes is regulated, and whether or not this might be a potential target for therapeutic intervention. Given the many unanswered questions, there can be no doubt that the already substantial number of publications on the subject of HIVs co-receptors will continue to grow for the foreseeable future.39
Inhibition of HIVs entry
The entry of HIVs is currently understood to be essentially a three-step process consisting of attachment, chemokine co-receptor interaction, and fusion. Therefore, specific areas of interest to inhibit HIVs entry include blocking gp120 binding to CD4 cell receptors (attachment inhibitors), blocking the binding sites of co-receptors such as CCR5 and CXCR4 (chemokine co-receptor inhibitors), and disrupting the fusion process (fusion inhibitors).
The fusion inhibitor, enfuvirtide (formerly called T-20), a 36-amino acid peptide being developed jointly by Trimeris and Roche, is in the most advanced stage of clinical development. HIV fusion with CD4 cells is a complex process, and not very well understood, and involves a conformational change in the HIV envelope gp120/gp41, leading to an interaction between gp41 that lead to intimate proximity between the HIV envelope and the cell membrane, allowing fusion to occur. Enfuvirtide is active against both CCR-5- and CXCR4-using viruses and are synergistic with CCR5 and CXCR4 antagonists.60
Many companies including Glaxo SmithKline are now investigating inhibitors of CCR5 and CXCR4. The lead Pfizer compound, UK-427, 857, is a non-competitive inhibitor of CCR5 that has an IC50 of 0.7 nmol in PBMCs, making it a more potent inhibitor of the co-receptor than RANTES, one of CCR-5’s natural ligands. The agent is active across a range of HIV-1 subtypes. Another candidate, the Takeda compound, TAK-220, has a mean IC50 of 4.37–10.5 nmol in PBMCs with no toxic effects observed in cell cultures at concentrations up to 100 nmol. The Ono compound, known as AK602, is active against HIV-1 with an IC50 of 0.2–0.6 nmol, and appears to bind non-competitively to the receptor.60
The identification of co-receptors that mediate HIVs entry might well have major significance for attempts to treat HIV infection. The chemokines themselves are inhibitors of HIV entry,36,37,38 and modified forms of RANTES, which bind CCR-5 yet fail to induce signal transduction, have similar properties.54,55 Beta chemokine homologues encoded by Kaposi’s sarcoma associated herpes virus have also been shown to block HIV-1 infection.56, 57 The lack of an overt phenotype associated with CCR-5 delta32, even among homozygotes,58,59 suggests that antagonists targeted specifically to this co-receptor should be relatively free of undesirable side effects. It might be possible to develop small molecule inhibitors that block viral interactions with CXCR-4 without compromising SDF-1 signal transduction, peptide and bicyclam compounds targeted to CXCR-4 that prevent T-tropic HIV infection have been described.61, 62, 63 However, all of these compounds also block signalling through CXCR-4. A potentially serious obstacle in the development of co-receptor targeted therapeutics is the plasticity of envelope co-receptor interactions.39 It is quite conceivable that co-receptor targeted compounds would simply select for strains that use an alternative co-receptor, or different regions of the same co-receptor. Furthermore, since AIDS has been documented in CCR-5 delta32 homozygotes, it is unlikely that even total ablation of the CCR-5 entry pathway will achieve much more than retardation of disease progression, once infection has been established. There is a possibility that CCR-5 targeted therapy might accelerate the course of disease by selection of viral strains that use alternative co-receptors (most notably CXCR-4, but also including CCR-2b and CCR-3) whose occurrence is associated with disease progression.64 It is possible that combinations of agents that ablate the co-receptor function of both CCR-5 and CXCR-4 would have a significant impact on the ability of HIV to propagate in vivo.
Cytopathic effects produced by in vitro infection with HIVs
The molecular mechanism by which HIV induces cytopathology in susceptible cells is not well understood. In vitro HIV infected cells formed multinucleated giant cells (syncytia) [Figure 3] that can produce large amounts of virus before they die, within a period of a few days, normally within 48 hours.81 Syncytia formation in culture results from fusion of infected cells with uninfected CD4+ cells82 and involves the CD4 molecule and HIV gp120 and gp41 proteins.83,84 Although the precise mechanism is still not known, syncytia formation is linked to the V3 loop of gp120 envelope region.85,86 Thus, the differences in the amino acids sequences of the V3 loop determine SI versus NSI isolates and whether an HIV isolate will induce syncytia formation or not. T cell tropic (SI) viruses generally have a basic amino acid at one or more of the positions 11, 24, 25 and 32 of the V3 loop, whereas macrophage tropic (NSI) viruses have either an acidic amino acid or alanine at position 25.
Generally syncytia formation is the most common event preceding cell death in vitro. There is some evidence that syncytia can occur in vivo and syncytia have been observed in the lymph nodes of homosexual men with lymphadenopathy, in lymph nodes and lungs of children with AIDS and in brain tissues of some adult patients who died with AIDS and is therefore possibly important in the pathogenesis of AIDS.87
Although syncytia formation may be an indirect indication of how virulent an HIV isolate is, syncytia formation itself is a direct effect of viral replication. Since the replication of HIVs depends on cellular activation and because HLA can cause activation of T cells through a mimicry mechanism,44,45,46 syncytia formation may indirectly depend on the HLA type of the PBMCs, which will determine the degree of cellular activation and hence the degree of viral replication.
As explained earlier, antigenic variation of the envelope protein of HIV is believed to be essential for the virus to escape specific humoral and cell-mediated immune responses that otherwise would suppress the replication of the virus. Moreover genetic variation can also lead to the emergence of more virulent syncytium-inducing (SI) isolates of HIV that may accelerate the progression of disease in infected individuals.88 Millions of genetic variants of HIVs can exist within one host and this diversity may be essential for the survival of the virus within its host.
Susceptibility of PBMCs to infection with HIVs
PBMCs include monocytes, lymphocytes and circulating dendritic cells. Monocytes can be infected by HIVs, and the susceptibility to infection can be observed at all stages of maturation.89 However, the kinetics of virus replication depends on the stage of cellular differentiation at the time of virus infection. CD4 (T helper) cells, the main target for HIVs, express the CD4 receptors and their destruction is associated with the deterioration of the immune system. T-lymphocytes do not support a productive infection with HIVs in vitro, unless they are first stimulated with agents such as mitogens or antigens.90 Activated T lymphocytes are easier to infect than non-activated ones.
Our experience of the differences of HIV infection observed among PBMCs from different individuals was not due to the differences in cell types (T cells, B cells and monocytes). Yamada et al,91 had found that differences between cultures still exist even if monocytes were removed and the remaining cells were only T cells. Williams and Cloyd92 also reported differences between CD4+ cell clones to infection by HIV indicating that the differences observed between different PBMCs to infection were most likely due to cellular factors. Moreover, these differences were not related to differences in CD4 antigen positive lymphocytes or to differences in cell growth.91
In addition, our finding that cells from some individuals produced more virus than others may indicate that this was due to the differences in lymphocytes proliferating in culture.44,45,46 However, considering that the concentration of PHA (and also IL2) used for lymphocyte stimulation and activation was the same in all cultures used in our tests, and that the cell concentration was also the same, then cell proliferation due to the presence of PHA may not be the answer for the differences observed among individual PBMCs. Therefore, factors that can cause cellular activation (which are different between different batches of PBMCs—e.g. HLA system)44,45,46 may provide an explanation for the differences in viral replication. Knowing the factors that determine susceptibility of PBMCs of an individual may also be important for in vitro initial HIV drug screening using PBMCs.44,46
Resistance to HIV Infection
The poorly understood phenomenon associated with the HIV-1 epidemic is the existence of individuals who have been repeatedly exposed to the virus but remain uninfected. It has been suggested that HIV-1 resistant individuals may have a non functioning co-receptor (such as CCR-5) preventing the virus from entering cells.59,93 The CCR-5 receptor was demonstrated as one of the main co-receptors for NSI (macrophage tropic) strains of HIV-1.34 Samson et al.,59 reported that a 32 bp deletion within the coding region for the CCR-5 generating a non-functional receptor that did not support infection by NSI strains of HIV-1. Moreover, white blood cells from individuals homozygous for the mutant CCR-5 were found to be highly resistant to infection by NSI viruses.59,93 Population studies indicate that the homozygous defect is found in only 1% of Caucasians of western European ancestry whereas the heterozygous defect is present in approximately 20% of this population.58 These results indicate that variants of the CCR-5 receptor could be responsible for the relative resistance to HIV-1 infection exhibited by some individuals and also for the variability of the course of the disease in infected patients.
In vitro results reported by many studies and our own44–46,91,58,94 showed no single PBMC was completely resistant to infection by different HIVs. The possibility that certain individuals are completely resistant to either, HIV-1, HIV-2, or both, still exists. Studies of larger groups may reveal whether certain individuals are completely resistant to HIV infection.
In some of these cases, a substantial in vitro infection resistance (specifically to CCR-5 utilising, M-tropic strains) of PBMCs and macrophages is evident.95,96 In addition, it is well known that there are large variations in the rate of progress of the disease among individuals who do become infected. Analysis of CCR-5 genes has revealed the existence of a defective CCR-5 allele that may contribute to each of these observations.58,59,97 Moreover, individuals who are heterozygous for the two major CCR-5 alleles do not manifest a high degree of infection resistance.59, 97 However, once infected, the progression of disease in heterozygotes appears to be somewhat retarded. This phenotype is associated with a measurably reduced virus load post seroconversion, and a decrease in frequency of symptomatic primary infection.98 Taken together, these observations strongly imply that the major route of HIV transmission both between individuals and between cells within an individual (at least during the early stages of infection) is mediated by the CCR-5 co-receptor.
Other receptors such as CCR-3 is only expressed in a restricted sub-population of T-cells99 and is, therefore, unlikely to play a major role in HIV infection of these cells. However, it is relatively abundant on microglial cells, the major targets of HIV-1 in the brain.100 Eotaxin, the physiological ligand for CCR-3, and a CCR-3 reactive monoclonal antibody have both been reported to possess infection inhibiting properties in primary brain cultures. M-tropic strains that are unable to use CCR-3 can also infect brain cultures, suggesting that both CCR-3 and CCR-5 play a role in infection of these cells. An interesting, but as yet unexplained, observation is the association of a CCR-2b polymorphism with retarded disease progression.101 This is unexpected given that the great majority of HIV-1 strains are not able to use CCR-2b as a co-receptor. The mutant CCR-2b allele, which encodes a receptor with a single valine to isoleucine change, is invariably associated with an intact CCR-5 allele. Since the two genes are very closely linked, it is quite likely that the mutant CCR-2b gene is associated with some, as yet unidentified, defect in CCR-5 expression, although this has not yet been thoroughly investigated. The wide variation of CCR-5 expression levels among individuals homozygous for intact reading frames has been documented, as has a measurably lower expression level in CCR-5 delta32 heterozygotes.102 It might well be that co-receptor expression levels contribute to the very large variation in rates of disease progression, particularly since the amount of CCR-5 expressed on PBMCs from different donors correlates with their ability to support the replication of M-tropic strains in vitro.102
CONCLUSION
This review has briefly described how HIVs use different molecules, including CD4 receptors and chemokine co-receptors, to gain entry into a human susceptible cell causing infection. Knowing exactly how HIVs infect peripheral blood mononuclear cells, and the factors that control the susceptibility or resistance of these cells to infection, is of importance for the understanding of the HIVs’ infectivity process. The chemokines are believed to be the most important molecules so far discovered which act as co-receptors for HIV entry. I believe that in the near future we will be seeing more details on the already discovered chemokine co-receptors, and more new co-receptors being discovered. This should aid in designing more potent weapons against the HIVs.
Figure 1b.
Simple diagram illustrating the main components of the basic structure of HIV-1.
Figure 3b.
Cytopathic effects of HIV-1 infection in vitro, using H9 cells (syncytia formation)
REFERENCES
- 1.Gottlieb MS, Schroff R, Schanker HM, Weisman JD, Fan PT, Wolf RA, et al. Pneumocystis carinii pneumonia and mucosal candidiasis in previously healthy homosexual men: evidence of a new acquired cellular immunodeficiency. N Engl J Med. 1981;305:1425–1431. doi: 10.1056/NEJM198112103052401. [DOI] [PubMed] [Google Scholar]
- 2.Masur H, Michelis MA, Greene JB, Onorata I, Vande Stouwe RA, Holzman RS, et al. An outbreak of community-acquired Pneumocystis carinii pneumonia. N Engl J Med. 1981;305:1431–1438. doi: 10.1056/NEJM198112103052402. [DOI] [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention. Revised classification system for HIV infection and expanded surveillance of definition for AIDS among adolescents and adults. MMWR. 1993;41:17. [PubMed] [Google Scholar]
- 4.Dybul M, Fauci AS, Bartlett JG, Kaplan JE, Pau AK. Guidelines for using antiretroviral agents among HIV-infected adults and adolescents. Ann Intern Med. 2002;137:381–433. doi: 10.7326/0003-4819-137-5_part_2-200209031-00001. [DOI] [PubMed] [Google Scholar]
- 5.Barre-Sinoussi F, Chermann JC, Rey R, Nugeyre MT, Chamaret S, Gruest J, et al. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS) Science. 1983;220:868–871. doi: 10.1126/science.6189183. [DOI] [PubMed] [Google Scholar]
- 6.Popovic M, Sarngadharan MG, Read E, Gallo RC. Detection, isolation, and continuous production of cytopathic retroviruses (HTLV-III) from patients with AIDS and pre AIDS. Science. 1984;244:497–500. doi: 10.1126/science.6200935. [DOI] [PubMed] [Google Scholar]
- 7.Sarngadharan MG, Popovic M, Bruch L, Schupbach J, Gallo RC. Antibodies reactive with Human T lymphotropic retroviruses (HTLV-III) in the serum of patients with AIDS. Science. 1984;224:506–508. doi: 10.1126/science.6324345. [DOI] [PubMed] [Google Scholar]
- 8.Levy JA, Hoffman AD, Kramer SM, Landis JA, Shimabukuro JM, Shiro LS. Isolation of lymphocytopathic retroviruses from San Francisco patients with AIDS. Science. 1984;225:840–842. doi: 10.1126/science.6206563. [DOI] [PubMed] [Google Scholar]
- 9.Clavel F, Guetard D, Brun-Vezinet F, Chamaret S, Rey MA, Santos-Ferreira MO, et al. Isolation of a new human retrovirus from West African patients with AIDS. Science. 1986;233:343–346. doi: 10.1126/science.2425430. [DOI] [PubMed] [Google Scholar]
- 10.Dalgleish AG, Beverley PC, Clapham PR, Crawford DH, Greaves MF, Weiss RA. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984;312:763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- 11.Cullen BR. Human immunodeficiency virus as a prototypic retrovirus. J Virol. 1991;65:1053–1056. doi: 10.1128/jvi.65.3.1053-1056.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Levy JA. Pathogenesis of HIV infection. Microbiol Rev. 1993;57:183–289. doi: 10.1128/mr.57.1.183-289.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stein BS, Gowda SD, Lifson JD, Penhallow RC, Bensch KG, Engleman EG. pH-independent HIV entry into CD4-positive T cells via virus envelope fusion to the plasma membrane. Cell. 1987;49:659–668. doi: 10.1016/0092-8674(87)90542-3. [DOI] [PubMed] [Google Scholar]
- 14.Berger EA, Murphy PM, Farber JM. Chemokine receptors as HIV-1 coreceptors: roles in viral entry, tropism, and disease. Annu Rev Immunol. 1999;17:657–700. doi: 10.1146/annurev.immunol.17.1.657. [DOI] [PubMed] [Google Scholar]
- 15.Murakami T, Yamamoto N. Roles of chemokines and chemokine receptors in HIV-1 infection. Int J Hematol 2000. 72:412–417. [PubMed] [Google Scholar]
- 16.Hwang SS, Boyle TJ, Lyerly HK, Cullen BR. Identification of the envelope V3 loop as the primary determinant of cell tropism in HIV-1. Science. 1991;253:71–74. doi: 10.1126/science.1905842. [DOI] [PubMed] [Google Scholar]
- 17.Shioda T, Levy JA, Cheng-Mayer C. Macrophage and T cell-line tropisms of HIV-1 are determined by specific regions of the envelope gp120 gene. Nature. 1991;349:167–169. doi: 10.1038/349167a0. [DOI] [PubMed] [Google Scholar]
- 18.O’Brien WA, Koyanagi Y, Namazie A, Zhao J-Q, Diagne A, Idler K, et al. HIV-1 tropism for mononuclear phagocytes can be determined by regions of gp120 outside the CD4-binding domain. Nature. 1990;348:69–73. doi: 10.1038/348069a0. [DOI] [PubMed] [Google Scholar]
- 19.Willey RL, Rutledge RA, Dias S, Folks T, Theodore T, Buckler CE, et al. Identification of conserved and divergent domains within the envelope gene of the acquired immunodeficiency syndrome retrovirus. Proc Natl Acad Sci USA. 1986;83:5038–5042. doi: 10.1073/pnas.83.14.5038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Simmonds P, Balfe P, Peutherer JF, Ludlam CA, Bishop JO, Brown AJL. Human immunodeficiency virus-infected individuals contain provirus in small numbers of peripheral mononuclear cells and at low copy numbers. J Virol. 1990;64:864–872. doi: 10.1128/jvi.64.2.864-872.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Willey RL, Theodore TS, Martin MA. Amino acid substitutions in the human immunodeficiency virus type 1 gp120 V3 loop that change viral tropism also alter physical and functional properties of the virion envelope. J Virol. 1994;68:4409–4419. doi: 10.1128/jvi.68.7.4409-4419.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goudsmit J, Boucher CA, Meloen RH, Epstein LG, Smit L, van der Hoek L, et al. Human antibody response to a strain-specific HIV-1 gp120 epitope associated with cell fusion inhibition. AIDS. 1988;2:157–164. [PubMed] [Google Scholar]
- 23.Kenealy WR, Matthews TJ, Ganfield MC, Langolis AJ, Waselefsky DM, Petteway RJ. Antibodies from human immunodeficiency virus-infected individuals bind to a short amino acid sequence that elicits neutralising antibodies in animals. AIDS Res Hum Retroviruses. 1989;5:173–182. doi: 10.1089/aid.1989.5.173. [DOI] [PubMed] [Google Scholar]
- 24.de Wolf F, Meloen RH, Bakker M, Barin F, Goudsmit J. Characterisation of human antibody-binding sites on the external envelope of human immunodeficiency virus type 2. J Gen Virol. 1991;72:1261–1267. doi: 10.1099/0022-1317-72-6-1261. [DOI] [PubMed] [Google Scholar]
- 25.Takahashi H, Merli S, Putney SD, Houghten R, Moss B, Germain RN, et al. A single amino acid interchange yields reciprocal CTL specificities for HIV-1 gp160. Science. 1989;246:118–121. doi: 10.1126/science.2789433. [DOI] [PubMed] [Google Scholar]
- 26.Fouchier RAM, Groenink M, Koostra NA, Tersmette M, Huisman G, Miedema F, et al. Phenotype-associated sequence variation in the third variable domain of the human immunodeficiency virus type 1 gp120 molecule. J Virol. 1992;66:3183–3187. doi: 10.1128/jvi.66.5.3183-3187.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ivanoff LA, Dubay JW, Morris JF, Roberts SJ, Gutshall L, Sternberg EJ, et al. V3 loop region of the HIV-1 gp120 envelope protein is essential for virus infectivity. Virology. 1992;187:423–432. doi: 10.1016/0042-6822(92)90444-t. [DOI] [PubMed] [Google Scholar]
- 28.Foley B, Korber B. Global variation in the HIV-1 V3 region. In: Myers G, Korber B, Hahn BH, Jeang K-T, Mellors JW, McCutchan FE, Henderson LE, Pavlakis GN, editors. Human Retroviruses and AIDS. Los Alamos, National Laboratory; Los Alamos, New Mexico, USA: 1995. pp. IIIpp. 77–108. [Google Scholar]
- 29.Agrawal L, Vanhom-Ali Z, Alkhatib G. Multiple determinants are involved in HIV coreceptor use as demonstrated by CCR4/CCL22 interaction in peripheral blood mononuclear cells (PBMCs) J Leukoc Biol. 2002;72:1063–1074. [PubMed] [Google Scholar]
- 30.Planelles V, Li Q-X, Chen ISY. The biological and molecular basis for cell tropism in HIV. In: Cullen BR, editor. Human Retroviruses. Oxford University Press; Oxford, UK: 1993. pp. 7–48. [Google Scholar]
- 31.James W, Weiss RA, Simon JH. The receptor for HIV: Dissection of CD4 and studies on putative accessory factors. Curr Top Microbiol Immunol. 1996;205:137–158. doi: 10.1007/978-3-642-79798-9_7. [DOI] [PubMed] [Google Scholar]
- 32.Clapham PR, Blanc D, Weiss RA. Specific cell surface requirements for the infection of CD4-positive cells by human immunodeficiency virus types 1 and 2 and by simian immunodeficiency virus. Virology. 1991;181:703–715. doi: 10.1016/0042-6822(91)90904-P. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Feng Y, Border CC, Kennedy PA, Berger EA. HIV-1 entry cofactor: Functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science. 1996;272:872–876. doi: 10.1126/science.272.5263.872. [DOI] [PubMed] [Google Scholar]
- 34.Deng H, Liu R, Ellmeier W, Choe S, Unutmaz D, Burkhart M, et al. Identification of a major co-receptor for primary isolates of HIV-1. Nature. 1996;381:661–666. doi: 10.1038/381661a0. [DOI] [PubMed] [Google Scholar]
- 35.Dragic T, Litwin V, Allaway GP, Martin SR, Huang Y, Nagashima KA, et al. HIV-1 entry into CD4+ cells is mediated by the chemokine receptor CC-CKR-5. Nature. 1996;381:667–673. doi: 10.1038/381667a0. [DOI] [PubMed] [Google Scholar]
- 36.Oberlin E, Amara A, Bachelerie F, Bessia C, Virelizier M, Arenzana-Seisdedos F, et al. The CXC chemokine SDF-1 is the ligand for LESTR/fusin and prevents infection by T-cell-line-adapted HIV-1. Nature. 1996;382:833–835. doi: 10.1038/382833a0. [DOI] [PubMed] [Google Scholar]
- 37.Bleul CC, Farzan M, Choe H, Parolin C, Clark-Lewis I, Sodroski J, et al. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature. 1996;382:829–833. doi: 10.1038/382829a0. [DOI] [PubMed] [Google Scholar]
- 38.Cocchi F, DeVico AL, Garzino-Demo A, Arya SK, Gallo RC, Lusso P. Identification of RANTES, MIP-1 alpha, and MIP-1 beta as the major HIV-suppressive factors produced by CD8+ T Cells. Science. 1995;270:1811–1815. doi: 10.1126/science.270.5243.1811. [DOI] [PubMed] [Google Scholar]
- 39.Bieniasz PD, Cullen BR. Chemokine receptors and human immunodeficiency virus infection. Frontiers in Bioscience. 1998;3:d44–58. doi: 10.2741/a265. [DOI] [PubMed] [Google Scholar]
- 40.Atchison RE, Gosling J, Monteclaro FS, Franci C, Digilio L, Charo IF, et al. Multiple extracellular elements of CCR5 and HIV-1 entry: Dissociation from response to chemokines. Science. 1996;274:1924–1926. doi: 10.1126/science.274.5294.1924. [DOI] [PubMed] [Google Scholar]
- 41.Bieniasz PD, Fridell RA, Aramori I, Ferguson SSG, Caron MG, Cullen BR. HIV-1-induced cell fusion is mediated by multiple regions within both the viral envelope and the CCR-5 co-receptor. EMBO J. 1997;16:2599–2609. doi: 10.1093/emboj/16.10.2599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Speck RF, Wehrly K, Platt EJ, Atchison RE, Charo IF, Kabat D, et al. Selective employment of chemokine receptors as human immunodeficiency virus type 1 co-receptors determined by individual amino acids within the envelope V3 loop. J Virol. 1997;71:7136–7139. doi: 10.1128/jvi.71.9.7136-7139.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bron R, Klasse PJ, Wilkinson D, Clapham PR, Pelchen-Matthews A, Power C, et al. Promiscuous use of CC and CXC chemokine receptors in cell-to-cell fusion mediated by a human immunodeficiency virus type 2 envelope protein. J Virol. 1997;71:8405–8415. doi: 10.1128/jvi.71.11.8405-8415.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Al-Jabri AA, Mccloskey D, Addawee M, Bottazzo FG, Sachs J, Oxford JS. In Vitro Correlation Between Human Leukocyte Antigen Class I and II Phenotype and HIV Infectivity of Activated Peripheral Blood Mononuclear Cell Cultures. AIDS. 1998;12:217–218. [PubMed] [Google Scholar]
- 45.Al-Jabri AA. HLA and in vitro susceptibility to HIV infection. Molecular Immunology. 2002;38:959–967. doi: 10.1016/s0161-5890(02)00023-8. [DOI] [PubMed] [Google Scholar]
- 46.Al-Jabri AA, Bottazo GF, Mccloskey D, Oxford JS. Evidence for Resistance Among PBMCS from Healthy Bangladeshis to Infection by Certain Isolates of HIV-1 & 2. Abstract P5/1, The First European Meeting of Virology; Wurzburg, Germany. September, 1995; pp. 10–13. [Google Scholar]
- 47.Gartner S, Markovits P, Markovitz D, Kaplan M, Gallo R, Popovic M. The role of mononuclear phagocytes in HTLV-III/LAV infection. Science. 1986;233:215–219. doi: 10.1126/science.3014648. [DOI] [PubMed] [Google Scholar]
- 48.Cheng-Mayer C, Seto D, Tateno M, Levy JA. Biological features of HIV that correlate with virulence in the host. Science. 1988;240:80–82. doi: 10.1126/science.2832945. [DOI] [PubMed] [Google Scholar]
- 49.Fenyo E, Morfeldt-Mason L, Chiodi F, Lind B, VonGegerfelt A, Albert J, Asjo B. Distinctive replicative and cytopathic characteristics of human immunodeficiency virus isolates. J Virol. 1988;62:4414–4419. doi: 10.1128/jvi.62.11.4414-4419.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schuitemaker H, Kootstra N, de Goede R, de Wolf F, Miedema F, Tersmette M. Monocytotropic human immunodeficiency virus type 1 (HIV-1) variants detectable at all stages of HIV-1 infection lack T-cell line tropism and syncytium-inducing ability in primary T-cell culture. J Virol. 1991;65:356–363. doi: 10.1128/jvi.65.1.356-363.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Collman R, Balliet JW, Gregory SA, Friedman H, Kolson DL, Nathanson N, Srinivasan A. An infectious molecular clone of an unusual macrophage-tropic and highly cytopathic strain of human immunodeficiency virus type 1. J Virol. 1992;66:7517–7521. doi: 10.1128/jvi.66.12.7517-7521.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Simmons G, Wilkinson D, Reeves JD, Dittmar MT, Beddows S, Weber J, et al. Primary, syncytium-inducing human immunodeficiency virus type 1 isolates are dual-tropic and most can use either Lestr or CCR5 as co-receptors for virus entry. J Virol. 1996;70:8355–8360. doi: 10.1128/jvi.70.12.8355-8360.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang L, Huang T, He Y, Cao Y, Ho DD. HIV-1 subtype and second-receptor use. Nature. 1996;383:768. doi: 10.1038/383768a0. [DOI] [PubMed] [Google Scholar]
- 54.Arenzanaseisdos F, Virelizier JL, Rousset D, Clark-Lewis I, Loetscher P, Moser B, Baggiolini M. HIV blocked by a chemokine antagonist. Nature. 1996;383:400. doi: 10.1038/383400a0. [DOI] [PubMed] [Google Scholar]
- 55.Simmons G, Clapham PR, Picard L, Offord RE, Rosenkilde MM, Schwartz TW, et al. Potent inhibition of HIV infectivity in macrophages and lymphocytes by a novel CCR-5 antagonist. Science. 1997;276:276–279. doi: 10.1126/science.276.5310.276. [DOI] [PubMed] [Google Scholar]
- 56.Kledal TN, Rosenkilde MM, Coulin F, Simmons G, Johnsen AH, Alouani S, et al. A broad spectrum chemokine antagonist encoded by Kaposi’s sarcoma-associated hepesvirus. Science. 1997;277:1656–1659. doi: 10.1126/science.277.5332.1656. [DOI] [PubMed] [Google Scholar]
- 57.Boschoff C, Endo Y, Collins PD, Takeuchi Y, Reeves JD, Schweickart VL, et al. Angiogenic and HIV-inhibitory functions of KSHV encoded chemokines. Science. 1997;278:290–294. doi: 10.1126/science.278.5336.290. [DOI] [PubMed] [Google Scholar]
- 58.Liu R, Paxton WA, Choe S, Ceradini D, Martin SR, Horuk R, et al. Homozygous defect in HIV-1 co-receptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell. 1996;86:367–377. doi: 10.1016/s0092-8674(00)80110-5. [DOI] [PubMed] [Google Scholar]
- 59.Samson M, Libert F, Doranz BJ, Rucker J, Liesnard C, Farber C-M, et al. Resistance to HIV-1 infection in caucasian individuals bearing mutant alleles of the CCR-5 chemokine receptor gene. Nature. 1996;382:722–725. doi: 10.1038/382722a0. [DOI] [PubMed] [Google Scholar]
- 60.Cooley LA, Lewin SR. HIV-1 cell entry and advances in viral entry inhibitor therapy. J Clin Virol. 2003;26:121–32. doi: 10.1016/s1386-6532(02)00111-7. [DOI] [PubMed] [Google Scholar]
- 61.Schols D, Struyf S, Van Damme J, Esté JA, Henson G, De Clercq E. Inhibition of T-tropic HIV strains by selective antagonization of the chemokine receptor CXCR4. J Exp Med. 1997;186:1383–1388. doi: 10.1084/jem.186.8.1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Murakami T, Nakajima T, Koyanagi Y, Tachibana K, Fujii N, Tamamura H, et al. A small molecule CXCR4 inhibitor that blocks T cell- line tropic HIV-1 infection. J Exp Med. 1997;186:1389–1393. doi: 10.1084/jem.186.8.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Doranz BJ, Grovit-Ferbasi K, Sharron MP, Mao SH, Goetz MB, Daar ES, et al. A small-molecule inhibitor directed against the chemokine receptor CXCR4 prevents its use as an HIV-1 co-receptor. J Exp Med. 1997;186:1395–1400. doi: 10.1084/jem.186.8.1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Connor RI, Sheridan KE, Ceradini D, Choe S, Landau NR. Change in co-receptor use correlates with disease progression in HIV-1-infected individuals. J Exp Med. 1997;185:621–628. doi: 10.1084/jem.185.4.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Connor RI, Mohri H, Cao Y, Ho DD. Increased viral burden and cytopathicity correlate temporally with CD4+ T-lymphocyte decline and clinical progression in human immunodeficiency virus type 1-infected individuals. J Virol. 1993;67:1772–1777. doi: 10.1128/jvi.67.4.1772-1777.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bieniasz PD, Fridell RA, Anthony K, Cullen BR. Murine CXCR-4 is a functional co-receptor for T-cell-tropic and dual-tropic strains of human immunodeficiency virus type 1. J Virol 1997. 71:7097–7100. doi: 10.1128/jvi.71.9.7097-7100.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tachibana K, Nakajima T, Sato A, Igarashi K, Shida H, Iizasa H, et al. CXCR4/fusin is not a species-specific barrier in murine cells for HIV-1 entry. J Exp Med. 1997;185:1865–1870. doi: 10.1084/jem.185.10.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bleul CC, Wu L, Hoxie JA, Springer TA, Mackay CR. The HIV co-receptors CXCR4 and CCR5 are differentially expressed and regulated on human T lymphocytes. Proc Natl Acad Sci USA. 1997;94:1925–1930. doi: 10.1073/pnas.94.5.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Malykh A, Reitz MS, Louie A, Hall L, Lori F. Multiple determinants for growth of human immunodeficiency virus in monocyte-macrophages. Virology. 1994;206:646–650. doi: 10.1016/s0042-6822(95)80082-4. [DOI] [PubMed] [Google Scholar]
- 70.McKnight A, Weiss RA, Chotton C, Takeuchi Y, Hoshino H, Clapham PR. Change in tropism upon immune escape by human immunodeficiency virus. J. Virol. 1995;69:3167–3170. doi: 10.1128/jvi.69.5.3167-3170.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Moore JP, Cao Y, Qing L, Sattentau QJ, Pyati J, Koduri R, et al. Primary isolates of human immunodeficiency virus type 1 are relatively resistant to neutralization by monoclonal antibodies to gp120, and their neutralization is not predicted by studies with monomeric gp120. J Virol. 1995;69:101–109. doi: 10.1128/jvi.69.1.101-109.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Biti R, French R, Young J, Bennetts B, Stewart G. HIV-1 infection in an individual homozygous for the CCR-5 deletion mutant. Nature Med. 1997;3:252–253. doi: 10.1038/nm0397-252. [DOI] [PubMed] [Google Scholar]
- 73.O’Brien TR, Winkler C, Dean M, Nelson JAE, Carrington M, Michael NL, White GC. HIV-1 infection in a man homozygous for CCR-5 delta32. Lancet. 1997;349:1219. doi: 10.1016/s0140-6736(97)24017-1. [DOI] [PubMed] [Google Scholar]
- 74.Theodorou I, Meyer l, Magierowska M, Katlana C, Rouzioux C. HIV infection in an individual homozygous for CCR-5 delta32. Lancet. 1997;349:1219–1220. [PubMed] [Google Scholar]
- 75.Balotta C, Bagnarelli P, Violin M, Ridolfo AL, Zho D, Berlusconi A, et al. Homozygous delta32 deletion of the CCR-5 chemokine receptor gene in an HIV-1-infected patient. AIDS. 1997;11:67–71. doi: 10.1097/00002030-199710000-00001. [DOI] [PubMed] [Google Scholar]
- 76.Moore J, Jameson B, Weiss R, Sattentau Q. In: Viral Fusion Mechanisms. Bentz J, editor. CRC Press; Boca Raton, FL: 1993. pp. 233–289. [Google Scholar]
- 77.Ferguson SSG, Zhang J, Barak LS, Caron MG. G-protein-coupled receptor kinases and arrestins: regulators of G-protein-coupled receptor sequestration. Biochem Soc Trans 1996. 24:953–959. doi: 10.1042/bst0240953. [DOI] [PubMed] [Google Scholar]
- 78.Gosling J, Monteclaro FS, Atchison RE, Arai H, Tsou C-L, Goldsmith MA, et al. Molecular uncoupling of C-C chemokine receptor 5-induced chemotaxis and signal transduction from HIV-1 co-receptor activity. Proc Natl Acad Sci, USA. 1997;94:5061–5066. doi: 10.1073/pnas.94.10.5061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Farzan M, Choe H, Martin KA, Sun Y, Sidelko M, Mackay CR, et al. HIV-1 entry and macrophage inflammatory protein-1 beta-mediated signaling are independent functions of the chemokine receptor CCR5. J Biol Chem. 1997;272:6854–6857. doi: 10.1074/jbc.272.11.6854. [DOI] [PubMed] [Google Scholar]
- 80.Aramori I, Zhang J, Ferguson SSG, Bieniasz PD, Cullen BR, Caron M G. Molecular mechanism of desensitization of the chemokine receptor CCR-5: Receptor signalling and internalization are dissociable from its role as an HIV-1 co-receptor. EMBO J. 1997;16:4606–4616. doi: 10.1093/emboj/16.15.4606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wigdahl B, Guyton RA, Sarin PS. Human immunodeficiency virus infection of the developing human nervous system. Virology. 1987;159:440–445. doi: 10.1016/0042-6822(87)90483-1. [DOI] [PubMed] [Google Scholar]
- 82.Lifson JD, Reyes GR, McGrath MS, Stein BS, Engleman EG. AIDS retrovirus induced cytopathology: Giant cell formation and involvement of CD4 antigen. Science. 1986;232:1123–1127. doi: 10.1126/science.3010463. [DOI] [PubMed] [Google Scholar]
- 83.Matthews TJ, Weinhold KJ, Lyerly HK, Langlois AJ, Wigzell H, Bolognesi DP. Interaction between the human T-cell lymphotropic virus type IIIB envelope glycoprotein gp120 and the surface antigen CD4: role of carbohydrate in binding and cell fusion. Proc Natl Acad Sci, USA. 1987;84:5424–5428. doi: 10.1073/pnas.84.15.5424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Andeweg AC, Leeflang P, Osterhaus ADME, Bosch ML. Both the V2 and V3 regions of the human immunodeficiency virus type 1 surface glycoprotein functionally interact with other envelope regions in syncytium formation. J Virol. 1993;67:3232–3239. doi: 10.1128/jvi.67.6.3232-3239.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Travis BM, Dykers TI, Hewgill D, Ledbetter J, Tsu TT, Hu SL, et al. Functional roles of the V3 hypervariable region of HIV-1 gp160 in the processing of gp160 and in the formation of syncytia in CD4+ cells. Virology. 1992;186:313–317. doi: 10.1016/0042-6822(92)90088-7. [DOI] [PubMed] [Google Scholar]
- 86.Trujillo JR, Goletiani NV, Bosch I, Kendrick C, Rogers RA, Trujillo EB, et al. T-tropic sequence of the V3 loop is critical for HIV-1 infection of CXCR4-positive colonic HT-29 epithelial cells. J Acquir Immune Defic Syndr. 2000;25:1–10. doi: 10.1097/00042560-200009010-00001. [DOI] [PubMed] [Google Scholar]
- 87.Sharer LR. Pathology of HIV-1 infection of the central nervous system. J Neuropathol Exp Neurol. 1992;51:3–11. doi: 10.1097/00005072-199201000-00002. [DOI] [PubMed] [Google Scholar]
- 88.Cornelissen M, Mulder-Kampinga G, Veenstra J, Zorgdrager F, Kuiken C, Hartman S, et al. Syncytium-inducing (SI) phenotype suppression at seroconversion after intramuscular inoculation of a non-syncytium-inducing/SI phenotypically mixed human immunodeficiency virus population. J Virol. 1995;69:1810–1818. doi: 10.1128/jvi.69.3.1810-1818.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Valentin A, Matsuda S, Asjo B. Characterisation of the in vitro maturation of monocytes and the susceptibility to HIV infection. AIDS Res Hum Retroviruses. 1990;6:977–978. doi: 10.1089/aid.1990.6.977. [DOI] [PubMed] [Google Scholar]
- 90.Wainberg MA, Blain N, Fitz-Gibbon L. Differential susceptibility of human lymphocyte cultures to infection by HIV. Clin Exp Immunol. 1987;70:136–142. [PMC free article] [PubMed] [Google Scholar]
- 91.Yamada O, Matsumoto T, Sasoaka R, Kurimura T. Variations in growth capacity of HIV in peripheral blood mononuclear cell preparations from different individuals. AIDS. 1990;4:35–40. doi: 10.1097/00002030-199001000-00005. [DOI] [PubMed] [Google Scholar]
- 92.Williams LM, Cloyd MW. Polymorphic human gene(s) determines differential susceptibility of CD4 lymphocytes to infection by certain HIV-1 isolates. Virology. 1991;184:723–728. doi: 10.1016/0042-6822(91)90442-e. [DOI] [PubMed] [Google Scholar]
- 93.Philpott S, Weiser B, Tarwater P, Vermund SH, Kleeberger CA, Gange SJ, et al. CC Chemokine Receptor 5 Genotype and Susceptibility to Transmission of Human Immunodeficiency Virus Type 1 in Women. J Infect Dis. 2003;187:569–575. doi: 10.1086/367995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jennes W, Sawadogo S, Koblavi-Deme S, Vuylsteke B, Maurice C, Roels TH, et al. Cellular Human Immunodeficiency Virus (HIV)-Protective Factors: A Comparison of HIV-Exposed Seronegative Female Sex Workers and Female Blood Donors in Abidjan, Cote d’Ivoire. J Infect Dis. 2003;187:206–14. doi: 10.1086/346049. [DOI] [PubMed] [Google Scholar]
- 95.Paxton WA, Martin SR, Tse D, O’Brien TR, Skurnick J, Vandevanter NL, et al. Relative resistance to HIV infection of CD4 lymphocytes from persons who remain uninfected despite multiple high risk sexual exposures. Nature Med. 1996;2:412–417. doi: 10.1038/nm0496-412. [DOI] [PubMed] [Google Scholar]
- 96.Connor RI, Paxton WA, Sheridan KE, Koup RA. Macrophages and CD4+ T lymphocytes from two multiply exposed uninfected individuals resist infection with primary non-syncytium inducing isolates of human immunodeficiency virus type 1. J Virol. 1996;70:8757–8764. doi: 10.1128/jvi.70.12.8758-8764.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Dean M, Carrington M, Winkler C, Huttley GA, Smith MW, Allikmets R, et al. Genetic Restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Science. 1996;273:1856–1862. doi: 10.1126/science.273.5283.1856. [DOI] [PubMed] [Google Scholar]
- 98.Meyer L, Magierowska M, Hubert J-B, Rouzioux C, Deveau C, Sanson F, et al. Early protective effect of CCR-5 delta32 heterozygosity on HIV-1 disease progression: Relationship with viral load. AIDS. 1997;11:73–78. doi: 10.1097/00002030-199711000-00001. [DOI] [PubMed] [Google Scholar]
- 99.Sallusto F, Mackay CR, Lanzavecchia A. Selective expression of of the Eotaxin receptor CCR-3 by human T helper 2 cells. Science. 1997;277:2005–2007. doi: 10.1126/science.277.5334.2005. [DOI] [PubMed] [Google Scholar]
- 100.He J, Chen Y, Farzan M, Choe H, Ohagen A, Gartner S, et al. CCR3 and CCR5 are co-receptors for HIV-1 infection of microglia. Nature. 1997;385:645–649. doi: 10.1038/385645a0. [DOI] [PubMed] [Google Scholar]
- 101.Smith MW, Dean M, Carrington M, Winkler C, Huttley GA, Lomb DA, et al. Contrasting genetic influence of CCR2 and CCR5 variants on HIV-1 infection and disease progression. Science. 1997;277:959–965. doi: 10.1126/science.277.5328.959. [DOI] [PubMed] [Google Scholar]
- 102.Wu BL, Paxton WA, Kassam N, Ruffing N, Rottman JB, Sullivan N, et al. CCR5 levels and expression pattern correlate with infectability by macrophage-tropic HIV-1 in vitro. J Exp Med. 1997;185:1681–1691. doi: 10.1084/jem.185.9.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]