Abstract
Background
Contact tracing (CT) has rarely been used to improve HIV case-finding in sub-Saharan Africa because of concerns regarding privacy protection and possibly high costs.
Methods
We estimate the relative cost of identifying an undiagnosed HIV infection through CT compared to client-initiated voluntary counseling and testing (VCT) and provider-initiated door-to-door provider-initiated testing (PIT). We used data from the Likoma Network Study (LNS), a study of sexual networks and HIV infection conducted on the small island of Likoma in northern Malawi, to inform these calculations.
Results
In Likoma, the probability that partners of HIV index cases could be traced ranged from 61.2% to 97.6% and the likelihood that they would consent to HIV testing and counseling ranged from 68.9% to 83.3%. HIV prevalence ranged from 48.1% to 66.7% among the partners who were tested. In populations where 80% of HIV-infected individuals are aware of their status, CT is rarely a cheaper case-finding approach than VCT. When only 20% know their status, CT would be cheaper than VCT if CT costs are less than 2 to 4.5 times that of an instance of VCT. Compared to door-to-door PIT, CT is preferred when the population prevalence is below 10% and CT costs are less than 2 to 5 times higher than that of door-to-door PIT. When HIV prevalence is > 10%, providing CT for current spouses of index cases remains an attractive approach to HIV case-finding.
Conclusion
Contact tracing could complement client-initiated VCT or door-to-door PIT in a large number of sub-Saharan populations affected by generalized epidemics of varying magnitudes.
I. Introduction
Despite a significant scale-up of HIV testing and treatment services [1–5], the majority of HIV-infected individuals in sub-Saharan Africa (SSA) remain unaware of their infection [6]. Current approaches to increasing the capacity to detect undiagnosed HIV infections consist of expanding access to client-initiated voluntary counseling and testing (VCT, [7, 8]), as well as provider-initiated approaches such as routine testing in clinical settings [9–11] and door-to-door HIV testing [12–15]. All these strategies involve encouraging all members of a target population to get tested, even in the absence of signs of the disease. Contact tracing (CT, also known as provider-initiated partner notification) is another approach to increasing HIV case-finding. During CT, a health provider first elicits the number and location of the sexual partners of HIV index cases (ICs), and then seeks to inform locatable partners about their possible exposure to infection and the need for HIV testing and counseling, prevention and possibly treatment. In developed settings with concentrated HIV epidemics, CT is frequently conducted by health services [16, 17], but it has generally not been practiced in SSA.
This has been the case because of a) concerns about privacy and potential for harm for CT participants, b) lack of proven interventions to reduce HIV transmission among couples reached through CT [18] and c) perceptions of CT as possibly expensive in settings of intense migration, where partners may be difficult to trace due to lack of specific contact information (e.g., no street names). The former two obstacles to the use of CT are increasingly being eroded in SSA. Recent work in ethics has placed emphasis on the responsibilities of sexual partners to inform each other about potential exposures to lethal diseases [19–21]. Effective approaches to preventing HIV transmission have also recently become available. In particular, HIV transmission is considerably reduced (if not interrupted) in serodiscordant couples when ICs are put on antiretroviral treatment [22]. Two recent pioneering trials of CT conducted in Malawi [23] and Cameroon [24] further indicate that obstacle c) may also have been exaggerated. Both trials found that CT was feasible, acceptable, generated few adverse events and increased the number of HIV diagnoses.
It remains however unknown whether the resources (human and material) needed to conduct CT in SSA populations are commensurate to the benefits of CT [25]. CT likely requires investments in time and training of health personnel and upgrading of health information system to guarantee confidentiality of patient information. By identifying additional undiagnosed HIV infections, it also leads to increased treatment costs. On the other hand, CT could also generate large savings by averting new HIV infections. Whereas estimates of the cost of existing testing strategies are widely available for sub-Saharan countries [13], at present, there are no data available on the costs of a CT program for HIV in SSA [25]. As a result, a full assessment of the cost-effectiveness of CT is not feasible.
In this paper, we do not consider treatment costs and potential benefits due to infections averted through CT. Instead, we focus on the costs incurred by health services to detect a single previously undiagnosed HIV infection (case-finding). CT could be a cheaper case-finding approach in SSA than existing strategies, if its costs are low or its “yield”, i.e., the proportion of newly diagnosed HIV cases among the partners of an IC sought by a health provider, is high. The yield of CT is a function of the probability that a sexual partner is infected and has yet to be diagnosed. It also depends on the ease of locating that partner and the provider’s ability to gain consent for testing from him/her. Unfortunately, outside of the two recent trials of CT, the data on such parameters is lacking for sub-Saharan populations, particularly in more remote, rural areas.
We use simple calculations to investigate the relative costs of CT vs. other testing approaches in detecting undiagnosed HIV cases in SSA populations, under a broad range of epidemiological and service provision conditions.
II. Methods
1. Cost calculations
a. General approach
We examine the relative costs of CT compared to 1) client-initiated testing (i.e., VCT) and 2) provider-initiated testing (PIT) in identifying an undiagnosed HIV infection. Among PIT strategies, we only consider the case of door-to-door testing, in which health providers attempt to visit every household in a population to offer HTC [12–14, 26]. We conduct cost calculations from a provider’s perspective. We define the yield of CT (noted q) as the proportion of initiated partners who become newly diagnosed HIV cases [27]. The yield is a function of the probability of successfully locating (i.e., tracing) a partner (“tracing probability” thereafter, noted t), the probability that the traced partner will accept to be tested (“testing probability” thereafter, noted h), the prevalence of HIV among tested partners (“partner prevalence” thereafter, noted i) and the proportion of traced partners already aware of their possible infection, noted aCT. As a result,
| (1) |
We call C the unit cost of tracing and providing HTC to a sexual partner. Thus the cost of finding an undiagnosed HIV infection through CT is C/q.
In a population with prevalence p, the prevalence of undiagnosed HIV infection among users of other testing services (e.g., PIT or VCT) is
| (2) |
where aT is the proportion of infected users already aware of their infection and δ corrects for the selective use of testing services in the population (see below). The average cost of finding an undiagnosed infected person through other testing strategies is S/p′, where S is the cost of providing testing services to one client.
As mentioned above, S and C do not include treatment costs as well as savings generated from prevented infections. We estimate t, h and i from the LNS data (see below). In the LNS, a is unknown and we treat a as a varying parameter. Identifying an undiagnosed infection through CT is cheaper than doing so through other testing strategies when C/q <S/p′. Thus at the break-even point, the relative cost of contact tracing, C/S equals q/p′. We conduct these calculations for each type of sexual partner: current spouses, former spouses and non-marital partners. We plot the break-even relative cost of contact tracing, C/S, as a function of q and p. Below the curve, CT is cheaper, and above, it is more expensive.
b. CT vs. client-initiated testing
In comparing CT to client-initiated VCT, we assume that previously diagnosed HIV cases do not seek VCT again after diagnosis, i.e., aT = 0(we ignore possible confirmatory testing of a previous diagnosis). On the other hand, aCT ≠ 0. We thus consider two scenarios: a high awareness scenario in which 80% of HIV-infected individuals are aware of their infection (aCT = 0.8) and a “low awareness scenario” in which 20% of HIV-infected individuals are aware of their infection (aCT = 0.2). Individuals in SSA tend to seek VCT once they have shown signs of the disease or after they have taken particular risks. As a result, the prevalence of HIV among users of testing services can be significantly larger than in the general population. We use data from Menzies et al. [13] to estimate δ(p). In this study, the odds of HIV infection among VCT clients were 4.4 times larger than among the general population. Thus we define δ(p) so that δ(p)/(1−δ(p)) = 4.4·p/(1−p).
c. CT vs. provider-initiated testing
In comparing CT to door-to-door PIT, we assume for simplicity that δ(p)=p, i.e., that the prevalence of HIV among those who test reflects the underlying population prevalence. In door-to-door PIT, health workers will also contact clients who are already aware of their infection. We thus assume that aCT = aT, i.e., the proportion of HIV-positive partners/clients contacted during CT or PIT who are aware of their infection is the same. As a result, the relative costs of CT vs. PIT in detecting undiagnosed infections do not depend on the proportion of infected population members who are aware of their status.
2. Contact tracing parameters in a remote sub-Saharan setting
a. Data source
We use data collected on Likoma – a remote island of Lake Malawi - [28, 29] to estimate some of the parameters in (1). The data analyzed in this paper were collected in 2007/08. They were not collected in a public health setting, but the LNS data collection emulates the steps of the CT process with some notable differences (see below). We first constructed a roster of the adult population of Likoma during a census. Inhabitants aged 18–49 were then asked to provide the names and characteristics of up to 5 of their most recent sexual partners (starting with the most recent) during audio computer-assisted interviews (ACASI). Based on this information, we attempted to locate each nominated partner in the pre-established rosters of the island population. Home-based HIV testing and counseling was offered to island inhabitants aged 18–49 years old.
b. Parameter estimates
We refer to all HIV+ respondents identified during the course of the LNS as “index cases” (ICs). Some nominated sexual partners of ICs were not “initiated” [27, 30]--i.e., we did not attempt to locate them in the island roster because the IC reported that their partner was dead, was outside of the 18–49 years old range, or resided outside of the LNS study area. All other partners were initiated. The partner of an IC was “traced” when s/he was identified in the island roster.
Based on this information, we measure the tracing probability (t) as the number of traced partners divided by the total number of initiated partners. We then estimate the testing probability (h) as the proportion of traced partners consenting to HTC during the LNS. Finally, we estimate partner prevalence (i) as the proportion of partners that tested positive among all partners tested during the LNS.
The LNS data differ from data on CT conducted in public health settings in several key ways. First, some partners elicited during the LNS would not be elicited during CT (e.g., deceased partners). Second, in the LNS, sexual partners are not contacted directly by health workers on the basis of information provided by ICs. Instead, they are approached by the study team solely because they reside in study villages [29]. We obtain linked partner data only indirectly, by matching nominations made by ICs to the island population roster.
c. Covariates
The LNS survey measured several characteristics of respondents, partners and their relationships. Respondent characteristics included age (less than 30 years old, 30–39, 40 years old and older), marital status (never married, currently married, widowed, divorced/separated), self-rated health (measured as a score ranging from 1 = excellent to 5 = poor), alcohol consumption (1 = consumes alcohol more than once a week, 0 = consumes alcohol only once a week or less), and self-reported ARV treatment. Sexual partners were classified into current spouses, former spouses and non-marital partners on the basis of reports from the IC. Relationship characteristics analyzed included date of last sex with a given partner (within last month, > 1 month but< 1 year ago, and > 1 year ago), reported partnership concurrency (whether the IC or the partner had other partner(s) at the time of the relationship) and co-residence of partners in the same household. Partnership concurrency was assessed directly [31, 32], by asking ICs whether themselves or their partner had other partners at any time during the course of their relationship.
III. Results
1. Descriptive statistics
We interviewed 194 HIV cases during the LNS in 2007/08 (Table 1). Most HIV ICs were women, and first tested positive during the LNS in 2007/08. The average age of female ICs was lower than that of male ICs. Male ICs were more likely to report having a current marital partner, whereas female ICs were more likely to report having former spouses (due to divorce or widowhood). Polygamy was uncommon among male ICs (2/56, 3.6%). Finally, males were more likely to report non-marital partners. Female ICs were less likely to report frequent alcohol consumption. Between 10 and 15% of ICs reported being on ARV treatment.
Table 1.
Characteristics and behaviors of HIV-infected respondents reporting at least one sexual partner
| Women (N=138) | Men (N=56) | |
|---|---|---|
| Background characteristics | ||
| First tested positive by the LNS1 in | ||
| 2005/06 | 33 (23.9%) | 6 (10.7%) |
| 2007/08 | 105 (76.1%) | 50 (89.3%) |
| Age | ||
| < 30 years old | 54 (39.1%) | 13 (23.2%) |
| 30 – 39 years old | 59 (42.8%) | 20(35.7%) |
| ≥40 years old | 25 (18.1%) | 23 (41.1%) |
| Marital Status | ||
| Never married | 21 (15.1%) | 8 (14.3%) |
| Currently married | 69 (49.6%) | 43 (76.8%) |
| Divorced/separated | 25(18.0%) | 2 (3.6%) |
| Widowed | 22(15.8%) | 3 (5.3%) |
| Number of reported sexual partners | ||
| Number of reported current spouses | ||
| 0 | 84 (60.9%) | 22 (39.3%) |
| 1 | 54 (39.1%) | 32 (57.1%) |
| 2 | -- | 2 (3.6%) |
| Number of reported former spouses | ||
| 0 | 79 (57.3%) | 41 (73.2%) |
| 1 | 54 (39.1%) | 13 (23.2%) |
| 2 | 5 (3.6%) | 1 (1.8%) |
| 3 | -- | 1 (1.8%) |
| Number of reported non-marital sex partners | ||
| 0 | 62 (44.9%) | 23 (41.1%) |
| 1 | 44 (31.9%) | 15 (26.8%) |
| 2 | 19 (13.8%) | 10 (17.9%) |
| 3 | 9 (6.5%) | 6 (10.7%) |
| 4 | 4 (2.9%) | 2 (3.6%) |
| Health and risk behaviors | ||
| General Health | ||
| Average score (1 = excellent, 5= poor) | 2.41 (1.38) | 2.24 (1.28) |
| Alcohol consumption | ||
| Several times a week | 15 (10.8%) | 23 (41.1%) |
| ARV treatment | ||
| Reports being on ARV treatment | 15 (10.8%) | 8 (14.3%) |
Note:
first HIV diagnosis could have taken place earlier. Source, Likoma Network Study (2007–2008)
ICs reported 357 relationships, with 343 unique partners. Ninety-one relationships were with current spouses (25.5%), 82 were with former spouses (23.0%), and 184 were non-marital relations (51.5%). More ICs had sexual intercourse recently with their current spouses than with other types of partners. Partnership concurrency was prevalent in all types of partnerships, with 20.2% of ICs and close to 30% of partners reported to have been engaged in concurrency at the time of the relationship. ICs resided with 73% of their current spouses, whereas only 3.6% of divorced spouses and 2.4% of non-marital partners lived in the IC’s household.
2. Contact tracing parameters
Twenty-seven (32.9%) former spouses and 14 (7.6%) non-marital partners were not initiated because they were reported as deceased (lower panels of table 2). Virtually no current spouses resided outside of the island, but this was the case for 18.3% and 34.2% of former spouses and non-marital partners, respectively. Between 7 and 12% of partners were not initiated because they were not eligible for HIV testing due to age restrictions. In total, 90.1% (82/91) of current spouses were initiated vs. 46.3% (38/82) and 53.3% (98/184) of former spouses and non-marital partners, respectively. Among those initiated, the tracing probability was highest for current spouses of ICs, and was lowest for non-marital partners: only 2.4% (2/82) of current spouses initiated by the study team could not be traced compared to 38.8% (37/98) of non-marital partners. The testing probability was high among all traced partners, with 68.9% to 83.3% of partners consenting to HTC when offered by the study team.
Table 2.
Characteristics of relationships reported by HIV infected respondents.
| All partners (N=357) | Current spouses (N=91) | Former spouses (N=82) | Non-marital relations (N=184) | |
|---|---|---|---|---|
| Characteristics of the relationship | ||||
| Date of last sexual intercourse | ||||
| Within last month | 114 (32.0%) | 71 (78.9%) | 12 (14.6%) | 31 (17.0%) |
| Within last year | 63 (17.7%) | 9 (10.0%) | 21 (25.6%) | 33 (18.1%) |
| More than a year ago | 177 (49.7%) | 10 (11.1%) | 49 (59.8%) | 118 (64.8%) |
| Partnership concurrency | ||||
| Index case (IC) | 72 (20.2%) | 23 (25.3%) | 13 (15.8%) | 36 (19.6%) |
| Partner | 105 (29.6%) | 30 (33.0%) | 15 (18.3%) | 60 (33.0%) |
| Residence of the partner | ||||
| Same household as respondent | 73 (23.0%) | 67 (73.6%) | 2(3.6%) | 4 (2.4%) |
| Contact tracing outcomes | ||||
| Not initiated because deceased | 41 (11.5%) | -- | 27(32.9%) | 14 (7.6%) |
| Not initiated because does not reside on the island | 79 (22.1%) | 1 (1.1%) | 15(18.3%) | 63(34.2%) |
| Not initiated because not eligible for LNS | 19 (5.3%) | 8(8.8%) | 2(2.4%) | 9 (4.9%) |
| Total initiated | 218 (61.1%) | 82 (90.1%) | 38 (46.3%) | 98 (53.3%) |
| Total traced (Tracing probability1, t) | 172 (78.9%) | 80 (97.6%) | 31 (81.6%) | 61 (62.2%) |
| HTC outcome of the partner2 | ||||
| Tested and counseled (Testing probability2, h) | 127(78.9%) | 60 (75.0%) | 25 (83.3%) | 42 (68.9%) |
| Counseled but refused test | 3(1.7%) | -- | -- | 3 (4.9%) |
| Refused HTC | 19 (11.0%) | 10 (12.5%) | 1 (3.2%) | 8 (13.1%) |
| Absent at time of visit(s) | 23 (13.4%) | 10 (12.5%) | 5 (16.1%) | 8 (13.1%) |
Notes:
Among initiated partners,
among traced partners. For some variables, missing data implies that the counts do not sum up to the total in the column header.
Source, Likoma Network Study (2007–2008)
Partner prevalence was also high in all types of relationships (Figure 1): 42 of the 63 current spouses of HIV infected respondents (66.7%), 13 of the 27 former spouses (48.1%) and 24 of the 44 non-marital partners (54.5%) were themselves infected with HIV. The prevalence of HIV among tested partners did not vary by gender of the IC except in non-marital relations: 16 out of 22 tested non-marital partners of male ICs vs. 8 out of 23 non-marital partners of female ICs were infected with HIV (p=0.02 according to Fisher’s exact test).
Figure 1.
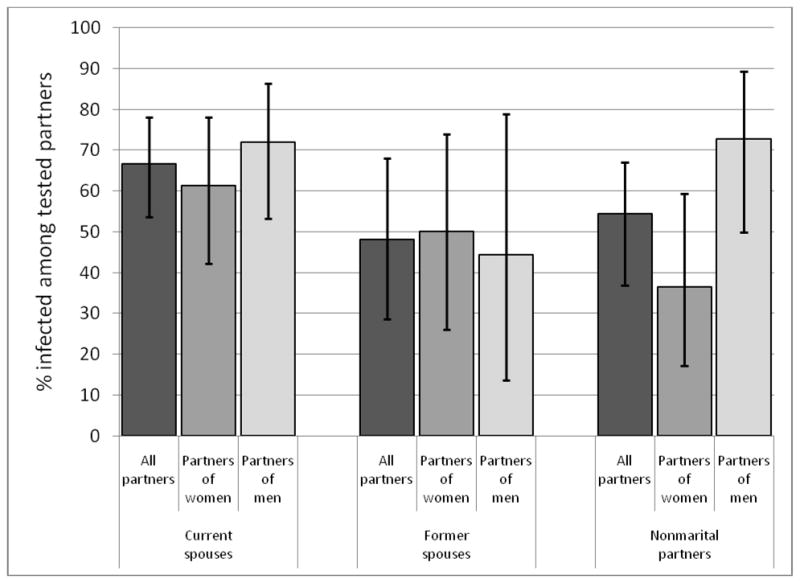
HIV prevalence among traced partners of HIV-seropositive respondents, Source: Likoma Network Study, 2007–2008. Error bars show 95% CI.
3. Cost calculations
In all the scenarios and comparisons we consider (figure 2), the attractiveness of CT as a case-finding approach is highest for current spouses, followed by former spouses and non-marital partners. It also decreases when the prevalence of HIV in the population increases. Compared to VCT (solid lines), providing CT is rarely the cheapest case-finding approach in populations with high HIV status awareness, at any level of HIV prevalence. In populations with low HIV status awareness, on the other hand, the relative costs of CT vs. VCT increase sharply when the prevalence of HIV declines below 5%. In a population with 2% HIV prevalence and low HIV status awareness, for example, CT would be the cheaper case-finding approach if its cost was up to 2 to 4.5 times higher than the costs of providing VCT, depending on the type of partner to be traced.
Figure 2.

Relative cost of providing contact tracing for different types of sexual partners. Below the curve, CT is cheaper than VCT or PIT; above the curve, CT is more expensive.
Notes: The dotted curves represent provider-initiated testing (e.g., door-to-door testing); the thick solid curve represent low awareness scenarios of VCT; and the thing solid curves represent high awareness scenarios of VCT. CS, FS, and NMP refer to current spouses, former spouses, and non-married partners respectively.
Compared to door-to-door PIT, CT is much more commonly an attractive option for HIV case-finding. Below 5% prevalence in the population, CT is almost invariably preferred to PIC unless its costs are more than 4 to 9 times higher than the costs of PIT. In a population with HIV prevalence ≈10%, CT would be a cheaper case-finding approach than door-to-door PIC if CT costs were up to 2 to 5 times larger than the costs of PIC. In populations with prevalence above 10%, providing CT remains an attractive approach for current spouses, even at high levels of relative costs (>2). Providing CT for non-marital partners and former spouses would be a cheaper case-finding approach than PIT only if the relative costs of CT vs. door-to-door PIC are below 2.
IV. Discussion
Using parameter estimates derived from the Likoma Network Study, in conjunction with simple calculations, we showed that CT could be a cheaper approach to detecting an undiagnosed HIV infection than both client-initiated and provider-initiated testing strategies in a large number of rural sub-Saharan populations affected by generalized epidemics. This was particularly true in populations with low to moderate HIV prevalence (i.e., countries of western, central and eastern Africa). In these settings, CT would be cheaper than provider-initiated testing unless the costs of CT are prohibitively high. CT would also be cheaper than VCT in settings where awareness of HIV status is low. In the most afflicted populations (e.g., southern Africa), CT is unlikely to be a cheaper case-finding approach than VCT unless CT costs are comparable to (or even lower than) the costs of VCT. CT remains a cheaper case-finding approach than provider-initiated strategies, even at high costs levels, when it is offered for current spouses of HIV ICs. CT could thus complement existing strategies used by health services for HIV case-finding.
Our results suffer from important limitations. First, our analysis does not constitute an assessment of the cost-effectiveness of CT. It does not take into account treatment costs associated with increased case finding or potential savings stemming from averted infections. Even if partners are traced only after they have already been infected by the index case, CT can lead to identifying undiagnosed HIV cases earlier than waiting for infected persons to spontaneously present for testing. This aspect is not considered in our calculations. Second, our estimates of CT parameters were obtained using research data rather real-life data obtained in a public health setting. In the LNS, partners of HIV ICs were not directly approached by health workers, but rather their location and HIV status were indirectly obtained by data linking procedures [29]. The effectiveness of CT in public health settings could be significantly lower if i) health workers have less information on the members of their catchment population than was available to LNS researchers; ii) health workers elicit systematically lower numbers of partners from their patients because they cannot use ACASI techniques or do not have time to establish trust and rapport with them during the provision of care; or iii) health workers have less time to devote to CT than LNS study team members. However, the effectiveness of CT in a public health setting could also be higher if health workers can employ more aggressive tracing strategies based on local inquiries with neighbors and relatives, use of mobile phones etc. In the LNS, if a nominated partner could not be identified in the pre-established island roster using information reported by respondents, no further inquiries were made [29].
Third, we did not consider that the potential effectiveness of CT in identifying undiagnosed infections could be lower if ICs preferentially refer to testing their partners who have already been tested elsewhere and are aware of their HIV status. There are unfortunately no data on this parameter in the LNS or in Brown et al’s study [23]. Fourth, because the study was conducted in a remote island setting, it may also have overestimated the proportion of partners who are actually traceable. In more accessible mainland settings, individual sexual networks may extend well-beyond neighboring villages and a smaller proportion of one’s partners may thus be locatable. Because the estimated relative costs of CT vs. both client-initiated and provider-initiated testing were often highly favorable to CT, it is however likely that our findings translate to a large number of mainland populations, particularly where the prevalence of HIV is moderate.
Fifth, the cost calculations we conducted relied on strong assumptions. In particular, we assumed that the prevalence of HIV among partners of ICs is independent of the population-level prevalence; in doing so, we ignored the possibility that the partner of an IC was infected by someone other than the IC. This implies that we slightly underestimate the break-even cost of CT vs. other testing strategies when the population prevalence is high and overestimates it when the prevalence is low. More detailed models of CT can relax this assumption [33, 34]. Sixth, our analysis only considered relative costs per newly diagnosed infection. It did not address other important outcomes such as the total number of new infections diagnosed through CT vs. client or provider-initiated testing strategies. This outcome depends on the uptake of CT services among HIV ICs and their partners (i.e., whether 10 or 90% of ICs choose to have health workers assist them in notifying their partners of HIV exposure). Unfortunately, there are no available data on the uptake of CT in SSA to inform such calculations.
Our results nonetheless have important implications for strategies of HIV control, particularly those relying on “test and treat” approaches to HIV prevention [35–37]. In seeking to increase HIV case finding, current strategies employed in sub-Saharan countries mobilize patients and health providers independently (Table 3). Strategies mobilizing patients have aimed to encourage partner disclosure but have systematically failed to raise the rate of partner notification and referral to HIV services [38–43]. On the other hand, strategies mobilizing health providers may not always significantly increase case finding rates in the general population [44], may come at high costs [13] or may threaten the functioning of weak health systems [35]. Because it mobilizes patients and providers simultaneously, CT could help achieve better case-finding outcomes with more limited resources. In addition, CT is a highly transferable capacity, which can be usefully mobilized in epidemiological investigations of outbreaks of other diseases besides HIV (e.g., other sexually transmitted infections such as syphilis, or diseases transmitted by non-sexual contacts such as TB or meningitis) or in improving patient retention in chronic care programs (e.g., ARV treatment). We thus strongly support the call for further operational studies of the use of CT in sub-Saharan settings [25].
Table 3.
Strategies available to improve HIV case finding
| In seeking undiagnosed HIV patients… | Health providers are passive | Health providers are active |
|---|---|---|
| HIV index cases are passive | Stand-alone VCT (spontaneous presentation) | Door-to-door Testing Routine “opt-out” testing |
| HIV index cases are active | Partner disclosure Couples’ counseling and testing | Contact tracing (Provider-initiated partner notification) |
Acknowledgments
Funding: This research received support through the National Institute of Child Health and Development (grant nos. RO1 HD044228 and RO1 HD/MH41713), National Institute on Aging (grant no. P30 AG12836), the Boettner Center for Pensions and Retirement Security at the University of Pennsylvania, and the National Institute of Child Health and Development Population Research Infrastructure Program (grant no. R24 HD-044964), all at the University of Pennsylvania
Role of study sponsors:
The study sponsors did not play any role in the design, implementation and analysis of the study.
Footnotes
Conflict of interest statement:
All authors declare that the answer to the questions on your competing interest form are all No and therefore have nothing to declare.
Details of contributors:
Designed the study: SH, HPK; Analyzed the data: BA, SH; Collected data for the study: SH, JM; Designed and conducted mathematical modeling: BA; Supervised HIV testing procedures: JM; Produced first draft of the paper: BA, SH; Reviewed, edited and approved manuscript: HPK, JM, LKP; Guarantor: SH.
Ethical statement:
The Likoma Network Study was approved by the ethical review boards of the University of Malawi College of Medicine (COMREC) and the University of Pennsylvania. All study participants provided informed consent prior to participation.
Contributor Information
B. Armbruster, Northwestern University, Evanston USA.
S. Helleringer, Columbia University Mailman School of Public Health, New York USA.
H.-P. Kohler, University of Pennsylvania, Philadelphia USA.
J. Mkandawire, University of Malawi College of Medicine, Blantyre Malawi.
L. Kalilani-Phiri, Division of Community Health, University of Malawi College of Medicine, Blantyre Malawi.
References
- 1.Access to HIV treatment improves. AIDS Patient Care STDS. 2008;22:535–536. doi: 10.1089/apc.2008.9952. [DOI] [PubMed] [Google Scholar]
- 2.De Cock K, Grubb I. Towards universal access: WHO’s role in HIV prevention, treatment and care. Bull World Health Organ. 2006;84:506. doi: 10.2471/blt.06.0339104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.El-Sadr WM, Abrams EJ. Scale-up of HIV care and treatment: can it transform healthcare services in resource-limited settings? AIDS. 2007;21 (Suppl 5):S65–70. doi: 10.1097/01.aids.0000298105.79484.62. [DOI] [PubMed] [Google Scholar]
- 4.Loubiere S, Boyer S, Protopopescu C, Bonono CR, Abega SC, Spire B, Moatti JP. Decentralization of HIV care in Cameroon: increased access to antiretroviral treatment and associated persistent barriers. Health Policy. 2009;92:165–173. doi: 10.1016/j.healthpol.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 5.Reid A, Scano F, Getahun H, Williams B, Dye C, Nunn P, et al. Towards universal access to HIV prevention, treatment, care, and support: the role of tuberculosis/HIV collaboration. Lancet Infect Dis. 2006;6:483–495. doi: 10.1016/S1473-3099(06)70549-7. [DOI] [PubMed] [Google Scholar]
- 6.Anonymous. Organization WH. Towards universal access: Scaling up priority HIV/AIDS interventions in the health sector. Geneva: World Health Organization; 2010. [Google Scholar]
- 7.Sweat M, Morin S, Celentano D, Mulawa M, Singh B, Mbwambo J, et al. Community-based intervention to increase HIV testing and case detection in people aged 16–32 years in Tanzania, Zimbabwe, and Thailand (NIMH Project Accept, HPTN 043): a randomised study. Lancet Infect Dis. 2011 doi: 10.1016/S1473-3099(11)70060-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mbopi-Keou FX, Ongolo-Zogo P, Angwafo F, 3rd, Ndumbe PM, Belec L. High impact of mobile units for mass HIV testing in Africa. AIDS. 2007;21:1994–1996. doi: 10.1097/QAD.0b013e3282f006c3. [DOI] [PubMed] [Google Scholar]
- 9.Introduction of routine HIV testing in prenatal care--Botswana, 2004. MMWR Morb Mortal Wkly Rep. 2004;53:1083–1086. [PubMed] [Google Scholar]
- 10.Cockcroft A, Andersson N, Milne D, Mokoena T, Masisi M. Community views about routine HIV testing and antiretroviral treatment in Botswana: signs of progress from a cross sectional study. BMC Int Health Hum Rights. 2007;7:5. doi: 10.1186/1472-698X-7-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Topp SM, Chipukuma JM, Chiko MM, Wamulume CS, Bolton-Moore C, Reid SE. Opt-out provider-initiated HIV testing and counselling in primary care outpatient clinics in Zambia. Bull World Health Organ. 2011;89:328–335A. doi: 10.2471/BLT.10.084442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Were W, Mermin J, Bunnell R, Ekwaru JP, Kaharuza F. Home-based model for HIV voluntary counselling and testing. Lancet. 2003;361:1569. doi: 10.1016/S0140-6736(03)13212-6. [DOI] [PubMed] [Google Scholar]
- 13.Menzies N, Abang B, Wanyenze R, Nuwaha F, Mugisha B, Coutinho A, et al. The costs and effectiveness of four HIV counseling and testing strategies in Uganda. AIDS. 2009;23:395–401. doi: 10.1097/QAD.0b013e328321e40b. [DOI] [PubMed] [Google Scholar]
- 14.Helleringer S, Kohler HP, Frimpong JA, Mkandawire J. Increasing uptake of HIV testing and counseling among the poorest in sub-Saharan countries through home-based service provision. J Acquir Immune Defic Syndr. 2009;51:185–193. doi: 10.1097/QAI.0b013e31819c1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Negin J, Wariero J, Mutuo P, Jan S, Pronyk P. Feasibility, acceptability and cost of home-based HIV testing in rural Kenya. Trop Med Int Health. 2009;14:849–855. doi: 10.1111/j.1365-3156.2009.02304.x. [DOI] [PubMed] [Google Scholar]
- 16.Recommendations for partner services programs for HIV infection, syphilis, gonorrhea, and chlamydial infection. MMWR Recomm Rep. 2008;57:1–83. quiz CE81–84. [PubMed] [Google Scholar]
- 17.Mackellar DA, Hou SI, Behel S, Boyett B, Miller D, Sey E, et al. Exposure to HIV Partner Counseling and Referral Services and Notification of Sexual Partners among Persons Recently Diagnosed with HIV. Sex Transm Dis. 2008 doi: 10.1097/OLQ.0b013e31818d6500. [DOI] [PubMed] [Google Scholar]
- 18.Cassell MM, Surdo A. Testing the limits of case finding for HIV prevention. Lancet Infect Dis. 2007;7:491–495. doi: 10.1016/S1473-3099(07)70114-7. [DOI] [PubMed] [Google Scholar]
- 19.Dixon-Mueller R. The sexual ethics of HIV testing and the rights and responsibilities of partners. Stud Fam Plann. 2007;38:284–296. doi: 10.1111/j.1728-4465.2007.00141.x. [DOI] [PubMed] [Google Scholar]
- 20.Dixon-Mueller R, Germain A. HIV testing: the mutual rights and responsibilities of partners. Lancet. 2007;370:1808–1809. doi: 10.1016/S0140-6736(07)61754-1. [DOI] [PubMed] [Google Scholar]
- 21.Dixon-Mueller R, Germain A, Fredrick B, Bourne K. Towards a sexual ethics of rights and responsibilities. Reprod Health Matters. 2009;17:111–119. doi: 10.1016/S0968-8080(09)33435-7. [DOI] [PubMed] [Google Scholar]
- 22.Donnell D, Baeten JM, Kiarie J, Thomas KK, Stevens W, Cohen CR, et al. Heterosexual HIV-1 transmission after initiation of antiretroviral therapy: a prospective cohort analysis. Lancet. 375:2092–2098. doi: 10.1016/S0140-6736(10)60705-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brown L, Miller W, Kamanga G, Nyirenda N, Mmodzi P, Pettifor A, et al. HIV partner notification is effective and feasible in sub-Saharan Africa: opportunities for HIV treatment and prevention. Journal of Acquired Immune Deficiency Syndromes. 2011;56:437–442. doi: 10.1097/qai.0b013e318202bf7d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tih PM, Forgwei G, Welty T, Welty S, Harrington C. Integrated HIV contact tracing and partner notification in Cameroon: a feasible HIV infection risk reduction intervention for resource-poor settings. IAS Conference on HIV Pathogenesis, Treatment and Prevention; Cape Town. 2009. [Google Scholar]
- 25.Potterat JJ. Puzzling observations in a trial of HIV partner notification in Sub-Saharan Africa. Journal of Acquired Immune Deficiency Syndromes. 2011;56:384–392. doi: 10.1097/QAI.0b013e318211b451. [DOI] [PubMed] [Google Scholar]
- 26.Bunnell R, Cherutich P. Universal HIV testing and counselling in Africa. Lancet. 2008;371:2148–2150. doi: 10.1016/S0140-6736(08)60929-0. [DOI] [PubMed] [Google Scholar]
- 27.Brewer DD. Case-finding effectiveness of partner notification and cluster investigation for sexually transmitted diseases/HIV. Sex Transm Dis. 2005;32:78–83. doi: 10.1097/01.olq.0000153574.38764.0e. [DOI] [PubMed] [Google Scholar]
- 28.Helleringer S, Kohler HP. Sexual network structure and the spread of HIV in Africa: evidence from Likoma Island, Malawi. AIDS. 2007;21:2323–2332. doi: 10.1097/QAD.0b013e328285df98. [DOI] [PubMed] [Google Scholar]
- 29.Helleringer S, Kohler HP, Chimbiri A, Chatonda P, Mkandawire J. The Likoma Network Study: Context, data collection, and initial results. Demogr Res. 2009;21:427–468. doi: 10.4054/DemRes.2009.21.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Golden MR, Gift TL, Brewer DD, Fleming M, Hogben M, St Lawrence JS, et al. Peer referral for HIV case-finding among men who have sex with men. AIDS. 2006;20:1961–1968. doi: 10.1097/01.aids.0000247118.74208.6a. [DOI] [PubMed] [Google Scholar]
- 31.Nelson SJ, Manhart LE, Gorbach PM, Martin DH, Stoner BP, Aral SO, Holmes KK. Measuring sex partner concurrency: it’s what’s missing that counts. Sex Transm Dis. 2007;34:801–807. doi: 10.1097/OLQ.0b013e318063c734. [DOI] [PubMed] [Google Scholar]
- 32.Maughan-Brown BG, Venkataramani AS. Measuring concurrent partnerships: potential for underestimation in UNAIDS recommended methods. AIDS. 2011 doi: 10.1097/QAD.0b013e32834905c4. [DOI] [PubMed] [Google Scholar]
- 33.Armbruster B, Brandeau ML. Contact tracing to control infectious disease: when enough is enough. Health Care Manag Sci. 2007;10:341–355. doi: 10.1007/s10729-007-9027-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Armbruster B, Brandeau ML. Who Do You Know? A Simulation Study of Infectious Disease Control Through Contact Tracing. International Conference on Health Sciences Simulation; San Diego. 2007. [Google Scholar]
- 35.Garnett GP, Baggaley RF. Treating our way out of the HIV pandemic: could we, would we, should we? Lancet. 2009;373:9–11. doi: 10.1016/S0140-6736(08)61698-0. [DOI] [PubMed] [Google Scholar]
- 36.Granich RM, Gilks CF, Dye C, De Cock KM, Williams BG. Universal voluntary HIV testing with immediate antiretroviral therapy as a strategy for elimination of HIV transmission: a mathematical model. Lancet. 2009;373:48–57. doi: 10.1016/S0140-6736(08)61697-9. [DOI] [PubMed] [Google Scholar]
- 37.De Cock KM, Crowley SP, Lo YR, Granich RM, Williams BG. Preventing HIV transmission with antiretrovirals. Bull World Health Organ. 2009;87:488–488A. doi: 10.2471/BLT.09.067330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Brou H, Djohan G, Becquet R, Allou G, Ekouevi DK, Viho I, et al. When do HIV-infected women disclose their HIV status to their male partner and why? A study in a PMTCT programme, Abidjan. PLoS Med. 2007;4:e342. doi: 10.1371/journal.pmed.0040342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Makin JD, Forsyth BW, Visser MJ, Sikkema KJ, Neufeld S, Jeffery B. Factors affecting disclosure in South African HIV-positive pregnant women. AIDS Patient Care STDS. 2008;22:907–916. doi: 10.1089/apc.2007.0194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Medley A, Garcia-Moreno C, McGill S, Maman S. Rates, barriers and outcomes of HIV serostatus disclosure among women in developing countries: implications for prevention of mother-to-child transmission programmes. Bull World Health Organ. 2004;82:299–307. [PMC free article] [PubMed] [Google Scholar]
- 41.Ndiaye C, Boileau C, Zunzunegui MV, Koala S, Aboubacrine SA, Niamba P, et al. Gender-related factors influencing HIV serostatus disclosure in patients receiving HAART in West Africa. World Health Popul. 2008;10:43–54. [PubMed] [Google Scholar]
- 42.Varga CA, Sherman GG, Jones SA. HIV-disclosure in the context of vertical transmission: HIV-positive mothers in Johannesburg, South Africa. AIDS Care. 2006;18:952–960. doi: 10.1080/09540120500356906. [DOI] [PubMed] [Google Scholar]
- 43.Wong LH, Rooyen HV, Modiba P, Richter L, Gray G, McIntyre JA, et al. Test and tell: correlates and consequences of testing and disclosure of HIV status in South Africa (HPTN 043 Project Accept) J Acquir Immune Defic Syndr. 2009;50:215–222. doi: 10.1097/QAI.0b013e3181900172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pope DS, Deluca AN, Kali P, Hausler H, Sheard C, Hoosain E, et al. A cluster-randomized trial of provider-initiated (opt-out) HIV counseling and testing of tuberculosis patients in South Africa. J Acquir Immune Defic Syndr. 2008;48:190–195. doi: 10.1097/QAI.0b013e3181775926. [DOI] [PMC free article] [PubMed] [Google Scholar]


