Abstract
Upper airway muscles play an important role in regulating airway lumen and in increasing the ability of the pharynx to remain patent in the face of subatmospheric intraluminal pressures produced during inspiration. Due to the considerable technical challenges associated with recording from muscles of the upper airway, much of the experimental work conducted in human subjects has centered on recording respiratory-related activities of the extrinsic tongue protudor muscle, the genioglossus (GG). The GG is one of eight muscles that invest the human tongue (Abd-El-Malek, 1939). All eight muscles are innervated by the hypoglossal nerve (cranial nerve XII) the cell bodies of which are located in the hypoglossal motor nucleus (HMN) of the caudal medulla. Much of the earlier work on the respiratory-related activity of XII motoneurons was based on recordings obtained from single motor axons dissected from the whole XII nerve or from whole muscle GG EMG recordings. Detailed information regarding respiratory-related GG motor unit activities was lacking until as recently as 2006. This paper examines key findings that have emerged from the last decade of work conducted in human subjects. Wherever appropriate, these results are compared with results obtained from in vitro and in vivo studies conducted in non-human mammals. The review is written with the objective of facilitating some discussion and some new thoughts regarding future research directions. The material is framed around four topics: a) Motor unit type, b) Rate coding and recruitment, c) Motor unit activity patterns, and d) A compartment based view of pharyngeal airway control.
1. Introduction
Upper airway muscles play an important role in regulating airway lumen and in increasing the ability of the pharynx to remain patent in the face of subatmospheric intraluminal pressures produced during inspiration (Bartlett, 1986; Horner and Guz, 1991; Kuna and Smickley, 1988; Remmers et al., 1978; Sauerland and Harper, 1976; Wiegand et al., 1989). At least four muscle groups are involved in the maintenance of pharyngeal airway patency: a) the hyoid muscles (mylohyoid and geniohyoid) that move the hyoid anteriorly, b) muscles of the soft palate (tensor palatini), c) pharyngeal constrictor muscles and d) intrinsic (inferior and superior longitudinalis, transversus, verticalis) and extrinsic (genioglossus, hyoglossus, and styloglossus) muscles of the tongue that contribute to dilation and/or narrowing of the pharynx (Bailey and Fregosi, 2004; Bailey et al., 2006; Fuller et al., 1999; Kuna and Smickley, 1997; Kuna et al., 1997; Kuna and Vanoye, 1999; Mateika et al., 1999; Van Lunteren and Strohl, 1988; Wheatley et al., 1993). Due to the considerable technical challenges associated with recording from muscles of the upper airway, much of the experimental work carried out in human subjects has centered on recording respiratory-related activities of the more easily accessible extrinsic tongue protudor muscle genioglossus (GG) which forms much of the anterior pharyngeal wall.
2. Motor unit recording
The GG is one of eight muscles that invests the human tongue (Abd-El-Malek, 1939). All eight muscles are innervated by the hypoglossal nerve (cranial nerve XII) the cell bodies of which are located in the hypoglossal motor nucleus (HMN) of the caudal medulla. Much of the earlier work on the respiratory-related activity of XII motoneurons (Haxhiu et al., 1986; Hwang et al., 1983; Mitra and Cherniack, 1983; Mitra et al., 1986), was based on recordings obtained from single motor axons dissected from the whole XII nerve, from one of its major branches or by impaling cells in the XII motor nucleus (Kubin et al., 1996; Mifflin, 1990; Withington-Wray et al., 1988). However, because the hypoglossal nerve is a functionally mixed motor nerve i.e., XII motor axons project to any one of eight tongue muscles, it has not been possible to develop a clear understanding of the control of pharyngeal airway patency using these data alone (van Lunteren and Dick, 1989). In this regard, single motor unit recordings represent an opportunity to explore the contribution of identified motoneurons to the neuromuscular control of pharyngeal airway patency in human subjects.
A motor unit (MU) consists of a single motoneuron and the muscle fibers it supplies (Liddell and Sherrington, 1925), and is the fundamental entity by which activation of striated muscle is controlled (Buchthal and Schmalbruch, 1980). An action potential recorded from the muscle fiber of a single motor unit bears a one to one correspondance to the action potential recorded from the XII MN itself. Thus, in recording single motor unit activities we are provided a rare window onto the activity of an identified brainstem MN population (or subpopulation) in a behaving human subject.
Optimal single motor unit recordings necessitate intramuscular recording locations. Such recordings may be obtained using fine wires as originally described by Basmajian and Dutta, (Basmajian and Dutta, 1961) or tungsten (micro) electrodes (Hubel, 1957). The two electrode types have distinct advantages and limitations. Although fine-wire electrodes yield more stable recordings and allow for greater subject movement which is desirable for sleep or exercise studies, only modest adjustments in electrode position are possible after electrode insertion. By comparison, tungsten microelectrodes can be manufactured to specific impedances (i.e., presently up to 10 MΩ) and therefore, offer the opportunity for highly selective recordings. Whereas the rigid structure of tungsten electrodes makes it possible to relocate the electrode after insertion and thus to record from numbers of single MUs consecutively, the same feature necessarily restricts subject movement and precludes their use in any set-up that may require unrestricted subject movement i.e., overnight sleep studies.
3. Muscle and motor unit activities
Sauerland and Mitchell (Sauerland and Mitchell, 1970) obtained some of the earliest recordings of whole muscle and single motor unit GG EMG activity. Using fine wire electrodes inserted into the floor of mouth per-orally and per-cutaneously (see Figure 1), they described rhythmic, inspiratory activation of the GG in normal subjects in upright posture (Sauerland and Mitchell, 1975) and noted that in supine, the muscle displayed inspiratory augmenting or “phasic” activity and continuous or “tonic” activity that persisted throughout inspiration and expiration (Sauerland and Harper, 1976). Both the phasic and tonic components evident in waking were robustly maintained in stable NREM sleep, a finding that has since been confirmed by others (Bailey et al., 2007a; Tangel et al., 1992; Wiegand et al., 1989) (see Figure 2). However, in REM sleep the activities were strikingly different, characterized instead by profound EMG hypotonia (Sauerland and Harper, 1976).
Figure 1.
Parasagittal section through portion of the head approximately 3 mm from midline. The tongue is actively reflected. Per oral and per cutaneous needle insertion into the substance of the geniolgossus muscle. Abbreviations: Gh, geniohyoid; H, hyoid bone; W, insulated wire of bipolar recording electrode; Ps, lica sublingualis with underlying submandibular duct. (From Sauerland et al. 1981, Electromyography and Clinical Neurophysiology).
Figure 2.
Raw data from 1 subject demonstrating decrement from wakefulness to non-rapid-eye-movement (NREM) sleep in tonic tensor palatini moving time average (MTA) electromyogram (EMG) and masseter MTA EMG. Peak phasic and tonic genioglossus and diaphragm MTA EMG are well maintained. EEG, electroencephalogram. (From Tangel et al., 1993 J. App. Physiol., used with permission of the American Physiologic Society).
These early recordings proved to be of considerable clinical interest because they revealed a role for GG activity in the regulation of pharyngeal aperture and suggested that GG dysfunction may contribute to the events associated with obstructive sleep apnea. This clinical interest culminated in the landmark study by Remmers and colleagues (Remmers et al., 1978) in which it was hypothesized that the GG modulates oropharyngeal aperture and counterbalances the collapsing force exerted on the upper airway by contraction of inspiratory pump muscles. In more recent years, this hypothesis has been reformulated and under the current view, pharyngeal airway defense is attributed to the co-activation of multiple pharyngeal airway muscles (Fregosi and Fuller, 1997; Fuller et al., 1998).
In general, a variety of factors are considered to predispose an individual to upper airway narrowing and closure during sleep and the relative contribution of each factor will vary from individual to individual (Horner, 2008). Although the anatomy of the upper airway has long been viewed as critical in the pathogenesis of airway closure in OSA (Schellenberg et al., 2000) a recent study suggests that anatomy may account for as little as one third of the variability in severity of sleep apnea leaving a potentially substantial role for disturbances in neuromuscular control in the genesis of the disorder (Younes, 2003). If this assessment is correct, then our focus must shift toward identification of the mechanisms that modulate the activities of the pharyngeal airway musculature.
As outlined above, considerable research and clinical interest has centered on the GG and more specifically, on the “phasic inspiratory” activities of this muscle (Berry et al., 1997; Hudgel and Harasick, 1990; Hudgel et al., 1984; Onal et al., 1982; Pillar et al., 2001). By the same token, studies conducted by White and colleagues drew attention to the “tonic” component and the potential for this “background” activity that persists through inspiration and expiration to contribute to airway patency in the presence of an anatomically compromised airway (Fogel et al., 2001; Fogel et al., 2005; McGinley et al., 2008; Mezzanotte et al., 1992; Tangel et al., 1992; Tangel et al., 1991; Wiegand et al., 1991). Over the last decade, our quest to better understand the modulation of pharyngeal airway patency and pharyngeal airway muscle activities has fueled a renewed interest in and expansion of the tools applied to the problem. Accordingly, studies have incorporated magnetic resonance imaging (Cheng et al., 2008; Napadow et al., 1999a; Napadow et al., 1999b), pharmacologic manipulations (Saboisky et al., 2009), and application of increasingly refined muscle and nerve stimulation protocols (Decker et al., 1993; Eisele et al., 1995; Eisele et al., 2003; Eisele et al., 1997; Kezirian et al., 2010; Oliven et al., 2007b; Schwartz et al., 2001; Schwartz et al., 1996; Smith et al., 1996). Each of these approaches has afforded much needed new insight into the neuromuscular control of the pharyngeal airway over and above that which can be provided by traditional EMG recordings alone.
In addition to the aforementioned technologies, single motor unit recording has enjoyed a resurgence of interest and support with the realization that such recordings can yield information on firing thresholds, the time course of membrane potentials, the participation of specific motoneuron pools in particular movements and/or conditions and the nature and distribution of supraspinal connections. Although such data were obtained previously in human phrenic and intercostals MN pools, comparable information obtained from XII MUs was lacking until as recently as 2006 (Saboisky et al., 2006). This work is ongoing.
What follows below then is a review of some of the key findings that have emerged from this most recent decade of work conducted in human subjects. Wherever possible I draw on recent data obtained from in vitro and in vivo studies conducted in non-human mammals that can inform our interpretation of findings obtained in human subjects. The reader should be advised however, that the findings presented here do not represent an exhaustive review of the literature. Indeed, there are many significant findings and studies of XII MNs, the majority conducted in non-human mammal, that are not considered herein. Notwithstanding these omissions, it is my hope that the content of this review will serve as a source of material and perhaps to stimulate new thoughts/perspective about current and future work. I have framed the discussion around four topics as follows: a) Motor unit type, b) Rate coding and recruitment, c) Motor unit activity patterns, and d) Toward a compartment based view of pharyngeal airway control.
a) Motor unit type
More than fifty years ago studies conducted in human motor units gave rise to the notion that a motoneuron pool may be subdivided into classes or types of cells that are specialized for different functional requirements. In the early 1950’sTokizane et al. examined the discharge characteristics of 46 muscles with respiratory-related activity patterns in humans (Tokizane et al., 1952) and noted a dichotomous distribution of instantaneous firing rates which they interpreted as representing two “neuromuscular unit” types they designated phasic (i.e., activity during inspiration) and tonic (i.e., activity with greater firing frequency during inspiration) (Tokizane, 1955; Tokizane et al., 1952). These classifications of phasic and tonic respiratory MU types persist today.
A parallel schema adopted by muscle physiologists classifies motor units into different types based on their specific properties i.e., contractile speed and sensitivity to fatigue (i.e., Type Slow, Fast or Fast Fatigable) (Burke et al., 1973; Reinking et al., 1975). Similarly, muscle fibers were typed on the basis of their histochemical properties (i.e., Types I, IIa and IIb) (MacIntosh et al., 2006). For example, physiological studies in human upper airway muscles indicate the GG is composed of Type I (33%) and Type II fibers (67%) (Series et al., 1996). Yet, because the spatial distribution of the various muscle fiber types and motor unit types is not known, it’s not clear what if any, correspondence exists between muscle fiber type and contractile properties on the one hand and respiratory-related phasic and tonic MU types on the other (Sauerland and Mitchell, 1975; Tokizane et al., 1952; Tsuiki et al., 2000).
XII motoneurons themselves have been classified on the basis of their intrinsic properties (van Lunteren and Dick, 1992)(Berger et al., 1995). Parameters such as input resistance, resting membrane potential, firing rate in response to injected current, slope of the frequency–current relation, action potential amplitude, and spike voltage threshold are commonly used to describe and classify neurons in a wide variety of preparations (Berger, 2000; van Lunteren and Dick, 1992), perhaps in the belief that these properties are in some way predictive of the cell’s activity within a circuit (Grashow et al., 2010).
The utility of sorting motoneurons, muscle fibers or motor units into groups necessarily depends upon the stable expression of some set of characteristics that are distinguishable from those expressed by the other types. However, there are numerous examples wherein the particular set of characteristics used in sorting are not stable properties of the cell or MU and thus, serve as unreliable indicators of neuronal identity. For example, in regard to firing pattern there is good evidence that with modulation of chemical drive (John et al., 2005; Onal et al., 1981), vestibular input (Tsuiki et al., 2000), state (Bailey et al., 2007a) or load (Puckree et al., 1998), that phasically discharging MUs can begin to discharge tonically, suggestive of dynamic modulation. Similarly, extracellular recordings obtained from XII motoneurons describe modifications in firing pattern in response to hypoxia (Hwang et al., 1983; Lee and Fuller, 2010) or laryngeal mechanoreceptor stimulation (Withington-Wray et al., 1988). Thus, a motoneuron designated inspiratory at recruitment, shifts to an expiratory-inspiratory discharge pattern (Lee and Fuller, 2010), or a neuron that discharges primarily in inspiration begins to discharge tonically (Hwang et al., 1983) when respiratory drive increases (See Figures 3 and 4). These findings cast doubt on the reliability of “typing” as a means of distinguishing between MUs and suggest instead that the firing pattern of XII MUs – at least in healthy adults – is an inherently flexible and modifiable feature of MU activity (and see Activity patterns below). Although the significance of firing pattern flexibility is still unclear, the background levels of activation may be key in determining not only MU firing pattern but also whether a MN pool favors rate coding or recruitment, as discussed below.
Figure 3.
Alterations in discharge patterns on a hypoglossal fiber from inspiratory-expiratory to tonic by hypoxia. Phr, phrenic activity; Int Phr, integral of phrenic activity. FETCO2 and FETO2, end-tidal fractional concentrations of CO2 and O2. (From Hwang et al., 1983 J. App. Physiol., used with permission of the American Physiologic Society).
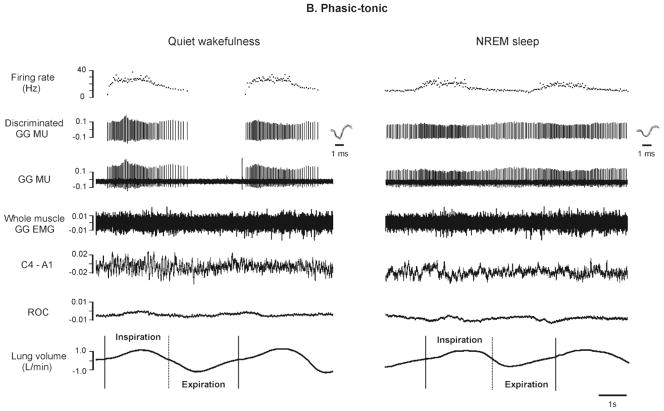
Figure 4.
Representative raw recordings of a MU with a phasic–tonic discharge pattern. In wakefulness, these MUs discharged maximally in inspiration falling silent in late expiration. In NREM sleep discharge persisted throughout the respiratory cycle. Top trace: instantaneous firing rate (dots). Middle trace: discriminated motor unit potentials,and insets show overlay of 10 consecutive potentials of the identified unit from each trial. Bottom traces: intramuscular whole muscle GG electromyogram (EMG), central electroencephalogram (EEG) recording site (C4/A1), right outer canthus (ROC) electrooculogram (EOG), and lung volume (sum of ribcage and abdomen volumes) signals. (From Bailey et al., 2007 J. Neurophysiology, used with permission of the American Physiologic Society).
b) Rate coding and recruitment
Previous studies have shown that for healthy human subjects, when the drive to breathe and thus the force required to defend the airway against more negative inspiratory pressures increases, the amplitude of whole muscle GG EMG increases (Shi et al., 1998; Williams et al., 2000). In general, increases in the intensity of muscle contraction are attributed to an increased number of active motor units (i.e., recruitment) and/or increased firing rates of already active motor units (i.e., rate coding). Traditionally, rate coding has been considered important for precise gradation of force, e.g., in small muscles of the hand (De Luca et al., 1982) whereas recruitment is thought to play a more significant role in modulating force during powerful but less precise contractions e.g., in muscles of the leg (Adrian and Bronk, 1929; Kukulka and Clamann, 1981).
Within the framework of respiration, single motor unit recordings in human subjects indicate that increases and decreases in the magnitude of the GG whole muscle EMG activity may depend almost exclusively on MU recruitment and derecruitment, respectively. Thus, increases in the drive to breathe secondary to steady state or progressive hypercapnia, evoke little if any change in the firing rates of GG MUs that typically remain stable throughout i.e., ~ 19.0 ± 2.0 Hz, despite significant increases in the magnitude of both the tonic and phasic components of the whole muscle EMG (Nicholas et al., 2010; Richardson and Bailey, 2010; Saboisky et al., 2010). If GG activity is assessed within the framework of volitional movement however, the results are markedly different. For example, when subjects perform simple planar protrusions, the average firing rate at recruitment is ~13.0 Hz and increases steeply to between 24.0–33.0 Hz as the magnitude of the displacement increases (Bailey et al., 2007b; Pittman and Bailey, 2009). How can we account for this disparity in GG MU firing rates between volitional and respiratory conditions?
There are several factors that play a role in determining the relative contributions of rate coding or recruitment including the initial firing rate of the unit and the magnitude of the neural drive. If the firing rate of the motor unit at recruitment is already quite high the unit has a limited potential to increase firing rate (Bigland and Lippold, 1954) as it is already at or close to the end of its dynamic range. A second and related factor is the magnitude of the drive impinging onto the MN (Person and Kudina, 1972). And here we can turn to studies conducted in non-human mammals that show dramatic increases in XII MN firing rate (e.g., from 50 to 80Hz) as a function of stepwise increments in drive (John et al., 2005) and studies conducted in the XII MN of the neonatal brainstem slice that show significant firing rate modulation (e.g., from 10 to 48Hz) as a function of the current injected into the cell (Berger et al., 1995; Pilarski et al., 2010). Thus, given a sufficient enhancement in excitatory input, XII MN/MUs clearly demonstrate a potential for firing rate modulation.
The absolute proportion of the motoneuron pool that is active can determine whether rate coding or recruitment is favored. For example, recruitment will be favored at low levels of force when drive onto the pool is modest and the total number of MUs active at baseline is small relative to the total number of MUs in the pool (Milner-Brown et al., 1973b). However, if drive is high proportionally more of the MU pool is active then firing rate modulation might reasonably become the principle means of achieving even greater force (Fuglevand et al., 1993; Milner-Brown et al., 1973a). Importantly, because the human hypoglossal nerve is almost exclusively a motor nerve comprising many thousands of motoneurons (i.e., current estimates based on the total number of myelinated nerve fibers (Atsumi and Miyatake, 1987) and motoneuron counts (O’Kusky and Norman, 1995) indicate that the XII MN pool comprises between 8000–9000 motoneurons per side), it may be, given physiologic levels of drive onto the pool, that the modulation of force in this particular MN pool is biased in favor of recruitment.
The potential to elicit rate coding within a respiratory framework remains an intriguing possibility. Interestingly, several previous studies indicate that the amplitude of whole muscle GG EMG attained in heavy exercise (Shi et al., 1998) may be significantly greater than in rebreathing or steady state hypercapnia (Richardson and Bailey, 2010). If this is the case, for a given increase in ventilation, input from higher central nervous system i.e., the so-called “central command” (Eldridge et al., 1985) or input arising in Group III and IV (i.e., trigeminal or glossopharyngeal afferents) (Amann et al., 2010) may prove more effective in driving the XII MN pool than chemoreceptor stimulation alone. For this reason, interventions that entail some combination of central drive and/or modulation of afferent input such as heavy exercise or respiratory muscle strength training (Davenport and Kifle, 2001; Davenport and Vovk, 2009; Huang et al., 2003; Martin et al., 2002) may provide the necessary framework within which to reassess the relative contributions of GG MU rate coding and recruitment (and see also Hill and Eastwood 2011, this issue).
c) Activity patterns
The cells of the respiratory system may be defined in several ways, including the respiratory phase in which the cell is active. The respiration cycle is typically broken down into three phases: inspiration, post inspiration (i.e., Stage 1 expiration, period of declining phrenic discharge after the peak inspiratory activity) and expiration (Richter, 1982). This three-phase organization is thought to offer explanation for a variety of respiratory control functions and disturbances in respiratory rhythm. However, within the three phases the range of behaviors of different cells is extraordinarily broad. For example, in a given state/condition an “inspiratory” cell may exhibit increasing activity as inspiration proceeds; some show decrementing activity and others exhibit activity that is consistent throughout.
The output from respiratory muscle is correspondingly complex and shows variations in instantaneous discharge frequency and patterns of action potentials of single motor units. In the case of GG MU activity, there is considerable variety in the number of impulses and instantaneous firing rates for inspiratory versus expiratory phases of the respiratory cycle as well as differences in MU firing onset and offset relative to each phase of the cycle. Consistent with convention, such activity patterns have been distinguished qualitatively on the basis of the duration and timing of the activity with respect to the respiratory cycle (Saboisky et al., 2006; Tsuiki et al., 2000; Wilkinson et al., 2008; Wilkinson et al., 2010) and quantified in terms of the strength of respiratory modulation i.e., the temporal relationship between spike train and the respiration cycle (Bailey et al., 2007a; Orem and Dick, 1983; Orem et al., 1985).
As many as six GG MU activity patterns have been identified in healthy human subjects in quiet wakefulness (Saboisky et al., 2006; Wilkinson et al., 2008) and which persist in NREM sleep (Bailey et al., 2007a) and after arousal from sleep (Wilkinson et al., 2010). The patterns of activity evident in healthy subjects appear well preserved in individuals diagnosed with OSA (Saboisky et al., 2007a). The latter finding is somewhat surprising in light of evidence of NREM sleep-related declines in inspiratory-related GG EMG activities in adult patients with OSA consistent with a diminution of neural drive to the upper airway in this population (Mezzanotte et al., 1996).
But whereas the full range of activity patterns may be evident in a given individual or in a given condition, I would argue that assessment of the summed output may be what matters most. For example, while the breadth of discharge patterns is preserved in NREM sleep, many motor units cease firing, others are recruited into activity and many more MUs switch pattern altogether i.e., a tonic pattern in wakefulness converts to episodic bursting in NREM sleep (Bailey et al., 2007b). Comparable examples of reconfiguration of activity occur when the drive to breathe increases secondary to chemoreceptor stimulation (Nicholas et al., 2010). In this case some number of MUs is recruited into activity and some number cease firing and others begin to fire more rapidly during expiration than inspiration, presumably to augment expiratory airflow by reducing expiratory resistance.
If we set aside qualitative assessments based on type and consider instead the nature of the summed output, it is somewhat remarkable that in a healthy individual the changes in activity appear to balance out. Whereas the activities of many GG MUs with strong respiratory modulation are extinguished at sleep onset, the activities of the majority of MUs with weak and moderate respiratory modulation persist and in some cases, the magnitude of the respiratory modulation of MUs increases. Thus, despite more than ~50% of MUs changing their activity pattern, the output from the whole muscle is robustly preserved with only slight changes in the overall strength of respiratory modulation (See Figure 5, Bailey et al., 2007a). This suggests that, in healthy subjects, the strength of respiratory modulation although not a stable feature of the individual GG MU activity, may be a stable feature of the population (Bailey et al., 2007a).
Figure 5.
Frequency histograms of the population of GG motor units (MUs) as a function of the consistency and strength of their respiratory activity as estimated by η2, in wakefulness (n-81) and NREM sleep (n=64). (From Bailey et al., 2007 J. Neurophysiology, used with permission of the American Physiologic Society).
d) A compartment based view of pharyngeal airway control
Inputs to XII MNs arise from multiple sources, including the primary motor cortex and several brainstem central pattern generators related to participation in critical functions such as speaking, chewing, swallowing and respiration (Ezure and Tanaka, 2006; Peever et al., 2001; Tarras-Wahlberg and Rekling, 2009) and reviewed in (Sawczuk and Mosier, 2001). From previous studies conducted in non human mammals it seems that only a portion of the XII MN pool receives central respiratory drive and that the cells that do are located in the ventromedial division of the XII nucleus (Sumi, 1969; Withington-Wray et al., 1988). This is consistent with the location of those motoneurons that innervate the GG (Aldes, 1995; Sokoloff and Deacon, 1992; Uemura et al., 1979). However, because these recordings were obtained from the whole XII nerve it is not possible to exclude the possibility that the neurons projected to tongue muscles other than the GG.
In recent years, single MU studies conducted in human subjects have documented that respiratory-related GG activity indeed appears localizable to “pockets” of activity in the posterior tongue – at least in conditions of rest breathing (Bailey et al., 2007a; Eastwood et al., 2003). A regional rather than distributed view of respiratory-related GG activity also is supported by magnetic resonance imaging (Cheng et al., 2008; Cheng et al., 2011; Napadow et al., 1999b; Stone et al., 2004) and intramuscular stimulation studies that reveal the mechanical action of the deeper, transversely orientated GG fibers and their contribution to pharyngeal airway opening (Cheng et al., 2008; Oliven et al., 2007a).
These apparently simple observations are in fact remarkable when considered from the perspective of motor system organization. To the best of my knowledge, previous models of lingual motor control have viewed muscles or groups of muscles as the output elements of the tongue motor system (Abd-El-Malek, 1938; Smith and Kier, 1989). At this level of organization, specific functions are ascribed to muscles based on their morphology (i.e., intrinsic vs extrinsic) or function (i.e., protrudor vs retractor)(Sokoloff, 2004). The results detailed above arising from MU recordings suggest however that muscles may not be the building blocks of pharyngeal movement and that pharyngeal airway lumen and stiffness may be controlled at the level of muscle compartments -- at least in rest breathing. In such a muscular compartment view, motion and stiffness are not modulated by activation of select muscle/s i.e., genioglossus vs. hyoglossus but rather depend on activation of single MUs that are brought into activity based on their compartmental location and mechanical effect (Sokoloff, 2004; Stone et al., 2004). Importantly, because the XII nerve is almost entirely a motor nerve such coordination likely entails inputs arising in the oral cavity or posterior pharynx and transduced by other cranial (i.e., lingual (CN V), superior laryngeal and/or glossopharyngeal) nerve afferents. If we expand on this idea further still and consider the anatomical evidence of extensive interdigitation of tongue muscle fibers throughout the tongue body (Abd-El-Malek, 1938), the distribution of respiratory drive potentials to multiple XII motoneuron (sub)populations (Koizumi et al., 2008) might underpin regional co-activation of multiple peripharyngeal MUs (Bailey and Fregosi, 2004; Stone et al., 2004).
If, as suggested (Freund, 1983; Person and Kudina, 1972), the output from a MU in a behaving subject is a complex interplay between synaptic strength and intrinsic properties it seems reasonable that we might begin to use respiratory-related stimuli, pharmacological and other therapies to modulate MN excitability. Importantly, if activities are, as suggested above, modulated in accordance with mechanical effect rather than distributed to an entire muscle, then perhaps MU activities may be considered as a functional assay for assessing the efficacy of pharmacological and/or behavioral therapies on network behavior.
4. Future directions
The information presented here underscores the utility and importance of single motor unit recordings. There can be no doubt that single motor unit recordings can provide a wealth of novel information regarding the control of the pharyngeal airway and in particular about how respiratory-related inputs are assembled and modulated in behaving human subjects (Bailey et al., 2007b; Butler et al., 1999; Gandevia et al., 1999; John et al., 2005; Laine and Bailey, 2011; Luschei et al., 1999; Nicholas et al., ; Nordstrom et al., 1990; Richardson and Bailey, 2010; Saboisky et al., 2006; Saboisky et al., 2007b; Saboisky et al., 2010; Sussman and MacNeilage, 1978; Sussman et al., 1977; Tsuiki et al., 2000; van Lunteren and Dick, 1989; van Lunteren and Dick, 2001; Wilkinson et al., 2008).
There are two additional points that merit consideration. First, the mammalian tongue is a remarkably complex structure. In selecting for study the GG, the hope is that the activities of this one muscle will yield insights into pharyngeal airway muscle activities more generally. Clearly, information garnered from a single muscle/MN pool is a poor substitute for activities derived from two or more subpopulations within a pool i.e., intrinsic vs. extrinsic (Bailey and Fregosi, 2006; Pittman and Bailey, 2009; Sauerland and Mitchell, 1975), protrudor vs. retractor (Fuller et al., 1998; Fuller and Fregosi, 2000; Fuller et al., 1999; Mateika et al., 1999), between adjacent cranial MN pools i.e., palatal (CN V) and pharyngeal (CN IX) (Fogel et al., 2005; Mezzanotte et al., 1996; Sauerland et al., 1981; Tangel et al., 1992; Wiegand et al., 1991) or for sampling system wide activity i.e., upper airway versus respiratory pump MU activity (Rice et al., 2011; Saboisky et al., 2007b). The results of each of the aforementioned studies serve to remind us that changes in activity pattern in one airway muscle typically will coincide with changes in the activation of adjacent muscles/MUs. Such coordinated “co-activation” may or may not enlarge the pharynx and improve pharyngeal airway patency depending on the particular muscles (Fregosi and Fuller, 1997; Fuller et al., 1999) and muscle fibers (i.e., transversely vs. vertically orientated GG fibers) activated (Oliven et al., 2007a).
Second, whether one subscribes to a compartment-based or muscle-based view of motor system organization it is clear that to understand pharyngeal airway motor control will require the simultaneous recording of single motor unit and whole muscle EMG activities. Whereas single motor unit recordings yield information about MU recruitment, firing rate modulation and timing, whole muscle EMG provides us the context within which those activities occur. In a behaving human subject and in a hydrostat, the normalization of whole muscle EMG activity to some maximum (e.g., in the case of GG, a tongue protrusion) may be the only reasonable means of determining the magnitude of the neural drive impinging on the MN pool in a particular task or condition. When studied in this manner, whole muscle GG EMG output provides us a means of comparing the magnitude of drive impinging on the pool across tasks or conditions and thus assists us in determining why rate coding or recruitment may be preferred means of modulating force.
If pharyngeal airway stability and cross sectional area are considered the target outcomes, then single motor unit recordings have shown us that those outcomes may be achieved in different ways. Thus, as described above, as conditions change there is evidence of MU recruitment, de-recruitment and alterations in MU activity patterns. On this basis, the whole muscle GG output is shaped both by the intrinsic properties of the MNs (which appear heterogeneous) and by the extrinsic drives that impinge upon the pool. Such shaping of activity is critical for maintaining airway stability and cross sectional area across a host of conditions, both normal and pathological – and in this regard may be considered compensatory. Indeed, it’s possible that the change in motor unit activity we detect in healthy individuals in response to an acute perturbation applied during the course of an experiment can provide a read-out of the types of changes we could expect in response to a chronic perturbation or illness. In this regard, short term change may yield insights into mechanisms that underpin long-term biological compensation (Grashow et al., 2010).
Acknowledgments
The author acknowledges Hilary Wakefield and Drs. Andrew Fuglevand, Ralph Fregosi, Jason Pilarski and Douglas Stuart for helpful discussions and editing of the manuscript. Dr Bailey’s work is supported by Grant Number 009587 from the NIH/NIDCD. The content is solely the responsibility of the author and does not necessarily represent the official views of the NIDCD or the National Institutes of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abd-El-Malek S. A contribution to the study of the movements of the tongue in animals, with special reference to the cat. Journal of Anatomy. 1938;73:5–30. [PMC free article] [PubMed] [Google Scholar]
- Abd-El-Malek S. Observations on the morphology of the human tongue. J Anat. 1939;73:201–21s. [PMC free article] [PubMed] [Google Scholar]
- Adrian ED, Bronk DW. The discharge of impulses in motor nerve fibres: Part II. The frequency of discharge in reflex and voluntary contractions. J Physiol. 1929;67:120–151. [PMC free article] [PubMed] [Google Scholar]
- Aldes LD. Subcompartmental organization of the ventral (protrusor) compartment in the hypoglossal nucleus of the rat. J Comp Neurol. 1995;353:89–108. doi: 10.1002/cne.903530109. [DOI] [PubMed] [Google Scholar]
- Amann M, et al. Group III and IV muscle afferents contribute to ventilatory and cardiovascular response to rhythmic exercise in humans. J Appl Physiol. 2010;109:966–76. doi: 10.1152/japplphysiol.00462.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atsumi T, Miyatake T. Morphometry of the degenerative process in the hypoglossal nerves in amyotrophic lateral sclerosis. Acta Neuropathol. 1987;73:25–31. doi: 10.1007/BF00695498. [DOI] [PubMed] [Google Scholar]
- Bailey EF, Fregosi RF. Coordination of intrinsic and extrinsic tongue muscles during spontaneous breathing in the rat. J Appl Physiol. 2004;96:440–9. doi: 10.1152/japplphysiol.00733.2003. [DOI] [PubMed] [Google Scholar]
- Bailey EF, Fregosi RF. Modulation of upper airway muscle activities by bronchopulmonary afferents. Journal of Applied Physiology. 2006;101:609–617. doi: 10.1152/japplphysiol.00204.2006. [DOI] [PubMed] [Google Scholar]
- Bailey EF, et al. Sleep/wake firing patterns of human genioglossus motor units. J Neurophysiol. 2007a;98:3284–91. doi: 10.1152/jn.00865.2007. [DOI] [PubMed] [Google Scholar]
- Bailey EF, et al. Anatomic consequences of intrinsic tongue muscle activation. J Appl Physiol. 2006;101:1377–85. doi: 10.1152/japplphysiol.00379.2006. [DOI] [PubMed] [Google Scholar]
- Bailey EF, et al. Firing patterns of human genioglossus motor units during voluntary tongue movement. Journal of Neurophysiology. 2007b;97:933–936. doi: 10.1152/jn.00737.2006. [DOI] [PubMed] [Google Scholar]
- Bartlett D., Jr . Upper airway motor systems. In: Cherniack NS, Widdicombe JG, editors. Handbook of Physiology. Section 3: The respiratory system. American Physiological Society; Washington DC: 1986. pp. 223–245. [Google Scholar]
- Basmajian JV, Dutta CR. Electromyography of the pharyngeal constrictors and levator palati in man. Anat Rec. 1961;139:561–3. doi: 10.1002/ar.1091390414. [DOI] [PubMed] [Google Scholar]
- Berger AJ. Determinants of respiratory motoneuron output. Respir Physiol. 2000;122:259–69. doi: 10.1016/s0034-5687(00)00164-x. [DOI] [PubMed] [Google Scholar]
- Berger AJ, et al. Postnatal development of hypoglossal motoneuron intrinsic properties. Adv Exp Med Biol. 1995;381:63–71. doi: 10.1007/978-1-4615-1895-2_7. [DOI] [PubMed] [Google Scholar]
- Berry RB, et al. Upper airway anesthesia reduces phasic genioglossus activity during sleep apnea. Am J Respir Crit Care Med. 1997;156:127–32. doi: 10.1164/ajrccm.156.1.9608037. [DOI] [PubMed] [Google Scholar]
- Bigland B, Lippold OC. Motor unit activity in the voluntary contraction of human muscle. J Physiol. 1954;125:322–35. doi: 10.1113/jphysiol.1954.sp005161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchthal F, Schmalbruch H. Motor unit of mammalian muscle. Physiol Rev. 1980;60:90–142. doi: 10.1152/physrev.1980.60.1.90. [DOI] [PubMed] [Google Scholar]
- Burke RE, et al. Physiological types and histochemical profiles in motor units of the cat gastrocnemius. J Physiol. 1973;234:723–48. doi: 10.1113/jphysiol.1973.sp010369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler JE, et al. Discharge properties and recruitment of human diaphragmatic motor units during voluntary inspiratory tasks. J Physiol. 1999;518(Pt 3):907–20. doi: 10.1111/j.1469-7793.1999.0907p.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng S, et al. Movement of the tongue during normal breathing in awake healthy humans. J Physiol. 2008;586:4283–94. doi: 10.1113/jphysiol.2008.156430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng S, et al. Movement of the human upper airway during inspiration with and without inspiratory resistive loading. J Appl Physiol. 2011;110:69–75. doi: 10.1152/japplphysiol.00413.2010. [DOI] [PubMed] [Google Scholar]
- Davenport PW, Kifle Y. Inspiratory resistive load detection in children with life-threatening asthma. Pediatr Pulmonol. 2001;32:44–8. doi: 10.1002/ppul.1087. [DOI] [PubMed] [Google Scholar]
- Davenport PW, Vovk A. Cortical and subcortical central neural pathways in respiratory sensations. Respir Physiol Neurobiol. 2009;167:72–86. doi: 10.1016/j.resp.2008.10.001. [DOI] [PubMed] [Google Scholar]
- De Luca CJ, et al. Behaviour of human motor units in different muscles during linearly varying contractions. J Physiol. 1982;329:113–28. doi: 10.1113/jphysiol.1982.sp014293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decker MJ, et al. Functional electrical stimulation and respiration during sleep. J Appl Physiol. 1993;75:1053–61. doi: 10.1152/jappl.1993.75.3.1053. [DOI] [PubMed] [Google Scholar]
- Eastwood PR, et al. Heterogeneous activity of the human genioglossus muscle assessed by multiple bipolar fine-wire electrodes. J Appl Physiol. 2003;94:1849–58. doi: 10.1152/japplphysiol.01017.2002. [DOI] [PubMed] [Google Scholar]
- Eisele DW, et al. The effects of selective nerve stimulation on upper airway airflow mechanics. Arch Otolaryngol Head Neck Surg. 1995;121:1361–4. doi: 10.1001/archotol.1995.01890120021004. [DOI] [PubMed] [Google Scholar]
- Eisele DW, et al. Tongue neuromuscular and direct hypoglossal nerve stimulation for obstructive sleep apnea. Otolaryngol Clin North Am. 2003;36:501–10. doi: 10.1016/s0030-6665(02)00178-0. [DOI] [PubMed] [Google Scholar]
- Eisele DW, et al. Direct hypoglossal nerve stimulation in obstructive sleep apnea. Arch Otolaryngol Head Neck Surg. 1997;123:57–61. doi: 10.1001/archotol.1997.01900010067009. [DOI] [PubMed] [Google Scholar]
- Eldridge FL, et al. Stimulation by central command of locomotion, respiration and circulation during exercise. Respir Physiol. 1985;59:313–37. doi: 10.1016/0034-5687(85)90136-7. [DOI] [PubMed] [Google Scholar]
- Ezure K, Tanaka I. Distribution and medullary projection of respiratory neurons in the dorsolateral pons of the rat. Neuroscience. 2006;141:1011–23. doi: 10.1016/j.neuroscience.2006.04.020. [DOI] [PubMed] [Google Scholar]
- Fogel RB, et al. Genioglossal activation in patients with obstructive sleep apnea versus control subjects. Mechanisms of muscle control. Am J Respir Crit Care Med. 2001;164:2025–30. doi: 10.1164/ajrccm.164.11.2102048. [DOI] [PubMed] [Google Scholar]
- Fogel RB, et al. The effect of sleep onset on upper airway muscle activity in patients with sleep apnoea versus controls. J Physiol. 2005;564:549–62. doi: 10.1113/jphysiol.2005.083659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fregosi RF, Fuller DD. Respiratory-related control of extrinsic tongue muscle activity. Respir Physiol. 1997;110:295–306. doi: 10.1016/s0034-5687(97)00095-9. [DOI] [PubMed] [Google Scholar]
- Freund HJ. Motor unit and muscle activity in voluntary motor control. Physiol Rev. 1983;63:387–436. doi: 10.1152/physrev.1983.63.2.387. [DOI] [PubMed] [Google Scholar]
- Fuglevand AJ, et al. Models of recruitment and rate coding organization in motor-unit pools. J Neurophysiol. 1993;70:2470–88. doi: 10.1152/jn.1993.70.6.2470. [DOI] [PubMed] [Google Scholar]
- Fuller D, et al. Co-activation of tongue protrudor and retractor muscles during chemoreceptor stimulation in the rat. J Physiol. 1998;507(Pt 1):265–76. doi: 10.1111/j.1469-7793.1998.265bu.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller DD, Fregosi RF. Fatiguing contractions of tongue protrudor and retractor muscles:influence of systemic hypoxia. Journal of Applied Physiology. 2000;88:2123–2130. doi: 10.1152/jappl.2000.88.6.2123. [DOI] [PubMed] [Google Scholar]
- Fuller DD, et al. Effect of co-activation of tongue protrudor and retractor muscles on tongue movements and pharyngeal airflow mechanics in the rat. J Physiol. 1999;519(Pt 2):601–13. doi: 10.1111/j.1469-7793.1999.0601m.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandevia SC, et al. Effects of increased ventilatory drive on motor unit firing rates in human inspiratory muscles. Am J Respir Crit Care Med. 1999;160:1598–603. doi: 10.1164/ajrccm.160.5.9904023. [DOI] [PubMed] [Google Scholar]
- Grashow R, et al. Compensation for variable intrinsic neuronal excitability by circuit-synaptic interactions. J Neurosci. 2010;30:9145–56. doi: 10.1523/JNEUROSCI.0980-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haxhiu MA, et al. Responses of hypoglossal and phrenic nerves to decreased respiratory drive in cats. Respiration. 1986;50:130–8. doi: 10.1159/000194919. [DOI] [PubMed] [Google Scholar]
- Horner RL. Pathophysiology of obstructive sleep apnea. J Cardiopulm Rehabil Prev. 2008;28:289–98. doi: 10.1097/01.HCR.0000336138.71569.a2. [DOI] [PubMed] [Google Scholar]
- Horner RL, Guz A. Some factors affecting the maintenance of upper airway patency in man. Respir Med. 1991;85(Suppl A):27–30. doi: 10.1016/s0954-6111(06)80250-7. [DOI] [PubMed] [Google Scholar]
- Huang CH, et al. Effect of inspiratory muscle strength training on inspiratory motor drive and RREP early peak components. J Appl Physiol. 2003;94:462–8. doi: 10.1152/japplphysiol.00364.2002. [DOI] [PubMed] [Google Scholar]
- Hubel DH. Tungsten Microelectrode for Recording from Single Units. Science. 1957;125:549–50. doi: 10.1126/science.125.3247.549. [DOI] [PubMed] [Google Scholar]
- Hudgel DW, Harasick T. Fluctuation in timing of upper airway and chest wall inspiratory muscle activity in obstructive sleep apnea. J Appl Physiol. 1990;69:443–50. doi: 10.1152/jappl.1990.69.2.443. [DOI] [PubMed] [Google Scholar]
- Hudgel DW, et al. Mechanics of the respiratory system and breathing pattern during sleep in normal humans. J Appl Physiol. 1984;56:133–7. doi: 10.1152/jappl.1984.56.1.133. [DOI] [PubMed] [Google Scholar]
- Hwang JC, et al. Characterization of respiratory-modulated activities of hypoglossal motoneurons. J Appl Physiol. 1983;55:793–8. doi: 10.1152/jappl.1983.55.3.793. [DOI] [PubMed] [Google Scholar]
- John J, et al. Respiratory-related discharge of genioglossus muscle motor units. Am J Respir Crit Care Med. 2005;172:1331–7. doi: 10.1164/rccm.200505-790OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kezirian EJ, et al. Electrical stimulation of the hypoglossal nerve in the treatment of obstructive sleep apnea. Sleep Med Rev. 2010;14:299–305. doi: 10.1016/j.smrv.2009.10.009. [DOI] [PubMed] [Google Scholar]
- Koizumi H, et al. Functional imaging, spatial reconstruction, and biophysical analysis of a respiratory motor circuit isolated in vitro. J Neurosci. 2008;28:2353–65. doi: 10.1523/JNEUROSCI.3553-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubin L, et al. Interaction of serotonergic excitatory drive to hypoglossal motoneurons with carbachol-induced, REM sleep-like atonia. Sleep. 1996;19:187–95. [PubMed] [Google Scholar]
- Kukulka CG, Clamann HP. Comparison of the recruitment and discharge properties of motor units in human brachial biceps and adductor pollicis during isometric contractions. Brain Res. 1981;219:45–55. doi: 10.1016/0006-8993(81)90266-3. [DOI] [PubMed] [Google Scholar]
- Kuna ST, Smickley J. Response of genioglossus muscle activity to nasal airway occlusion in normal sleeping adults. J Appl Physiol. 1988;64:347–53. doi: 10.1152/jappl.1988.64.1.347. [DOI] [PubMed] [Google Scholar]
- Kuna ST, Smickley JS. Superior pharyngeal constrictor activation in obstructive sleep apnea. Am J Respir Crit Care Med. 1997;156:874–80. doi: 10.1164/ajrccm.156.3.9702053. [DOI] [PubMed] [Google Scholar]
- Kuna ST, et al. Respiratory-related pharyngeal constrictor muscle activity in normal human adults. Am J Respir Crit Care Med. 1997;155:1991–9. doi: 10.1164/ajrccm.155.6.9196107. [DOI] [PubMed] [Google Scholar]
- Kuna ST, Vanoye CR. Mechanical effects of pharyngeal constrictor activation on pharyngeal airway function. J Appl Physiol. 1999;86:411–7. doi: 10.1152/jappl.1999.86.1.411. [DOI] [PubMed] [Google Scholar]
- Laine CM, Bailey EF. Common synaptic input to the human hypoglossal motor nucleus. J Neurophysiol. 2011;105:380–7. doi: 10.1152/jn.00766.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee KZ, Fuller DD. Preinspiratory and inspiratory hypoglossal motor output during hypoxia-induced plasticity in the rat. J Appl Physiol. 2010;108:1187–98. doi: 10.1152/japplphysiol.01285.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liddell EGT, Sherrington CS. Recruitment and some other factors of reflex inhibition. Proceedings of the Royal Society of London. 1925;B97:488–518. [Google Scholar]
- Luschei ES, et al. Discharge characteristics of laryngeal single motor units during phonation in young and older adults and in persons with parkinson disease. J Neurophysiol. 1999;81:2131–9. doi: 10.1152/jn.1999.81.5.2131. [DOI] [PubMed] [Google Scholar]
- MacIntosh BR, et al. Skeletal muscle: Form and function. Human Kinetics; Champaign, IL: 2006. [Google Scholar]
- Martin AD, et al. Use of inspiratory muscle strength training to facilitate ventilator weaning: a series of 10 consecutive patients. Chest. 2002;122:192–6. doi: 10.1378/chest.122.1.192. [DOI] [PubMed] [Google Scholar]
- Mateika JH, et al. Response of human tongue protrudor and retractors to hypoxia and hypercapnia. Am J Respir Crit Care Med. 1999;160:1976–82. doi: 10.1164/ajrccm.160.6.9903001. [DOI] [PubMed] [Google Scholar]
- McGinley BM, et al. Upper airway neuromuscular compensation during sleep is defective in obstructive sleep apnea. J Appl Physiol. 2008;105:197–205. doi: 10.1152/japplphysiol.01214.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mezzanotte WS, et al. Waking genioglossal electromyogram in sleep apnea patients versus normal controls (a neuromuscular compensatory mechanism) J Clin Invest. 1992;89:1571–9. doi: 10.1172/JCI115751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mezzanotte WS, et al. Influence of sleep onset on upper-airway muscle activity in apnea patients versus normal controls. Am J Respir Crit Care Med. 1996;153:1880–7. doi: 10.1164/ajrccm.153.6.8665050. [DOI] [PubMed] [Google Scholar]
- Mifflin SW. Arterial chemoreceptor input to respiratory hypoglossal motoneurons. J Appl Physiol. 1990;69:700–9. doi: 10.1152/jappl.1990.69.2.700. [DOI] [PubMed] [Google Scholar]
- Milner-Brown HS, et al. Changes in firing rate of human motor units during linearly changing voluntary contractions. J Physiol. 1973a;230:371–90. doi: 10.1113/jphysiol.1973.sp010193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner-Brown HS, et al. The orderly recruitment of human motor units during voluntary isometric contractions. J Physiol. 1973b;230:359–70. doi: 10.1113/jphysiol.1973.sp010192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitra J, Cherniack NS. The effects of hypercapnia and hypoxia on single hypoglossal nerve fiber activity. Respir Physiol. 1983;54:55–66. doi: 10.1016/0034-5687(83)90113-5. [DOI] [PubMed] [Google Scholar]
- Mitra J, et al. Comparison of the effects of hypercapnia on phrenic and hypoglossal activity in anesthetized decerebrate and decorticate animals. Brain Res Bull. 1986;17:181–7. doi: 10.1016/0361-9230(86)90114-0. [DOI] [PubMed] [Google Scholar]
- Napadow VJ, et al. Biomechanical basis for lingual muscular deformation during swallowing. Am J Physiol. 1999a;277:G695–701. doi: 10.1152/ajpgi.1999.277.3.G695. [DOI] [PubMed] [Google Scholar]
- Napadow VJ, et al. Intramural mechanics of the human tongue in association with physiological deformations. J Biomech. 1999b;32:1–12. doi: 10.1016/s0021-9290(98)00109-2. [DOI] [PubMed] [Google Scholar]
- Nicholas CL, et al. Motor unit recruitment in human genioglossus muscle in response to hypercapnia. Sleep. 2010;33:1529–38. [PMC free article] [PubMed] [Google Scholar]
- Nordstrom MA, et al. Synchronization of motor units in human masseter during a prolonged isometric contraction. J Physiol. 1990;426:409–21. doi: 10.1113/jphysiol.1990.sp018146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Kusky JR, Norman MG. Sudden infant death syndrome: increased number of synapses in the hypoglossal nucleus. J Neuropathol Exp Neurol. 1995;54:627–34. doi: 10.1097/00005072-199509000-00003. [DOI] [PubMed] [Google Scholar]
- Oliven A, et al. Effect of coactivation of tongue protrusor and retractor muscles on pharyngeal lumen and airflow in sleep apnea patients. J Appl Physiol. 2007a;103:1662–8. doi: 10.1152/japplphysiol.00620.2007. [DOI] [PubMed] [Google Scholar]
- Oliven A, et al. Effect of genioglossus contraction on pharyngeal lumen and airflow in sleep apnoea patients. Eur Respir J. 2007b;30:748–58. doi: 10.1183/09031936.00131106. [DOI] [PubMed] [Google Scholar]
- Onal E, et al. Pathogenesis of apneas in hypersomnia-sleep apnea syndrome. Am Rev Respir Dis. 1982;125:167–74. doi: 10.1164/arrd.1982.125.2.167. [DOI] [PubMed] [Google Scholar]
- Onal E, et al. Diaphragmatic and genioglossal electromyogram responses to CO2 rebreathing in humans. J Appl Physiol. 1981;50:1052–5. doi: 10.1152/jappl.1981.50.5.1052. [DOI] [PubMed] [Google Scholar]
- Orem J, Dick T. Consistency and signal strength of respiratory neuronal activity. J Neurophysiol. 1983;50:1098–107. doi: 10.1152/jn.1983.50.5.1098. [DOI] [PubMed] [Google Scholar]
- Orem J, et al. Activity of respiratory neurons during NREM sleep. J Neurophysiol. 1985;54:1144–56. doi: 10.1152/jn.1985.54.5.1144. [DOI] [PubMed] [Google Scholar]
- Peever JH, et al. Respiratory control of hypoglossal motoneurones in the rat. Pflugers Arch. 2001;442:78–86. doi: 10.1007/s004240000502. [DOI] [PubMed] [Google Scholar]
- Person RS, Kudina LP. Discharge frequency and discharge pattern of human motor units during voluntary contraction of muscle. Electroencephalogr Clin Neurophysiol. 1972;32:471–483. doi: 10.1016/0013-4694(72)90058-2. [DOI] [PubMed] [Google Scholar]
- Pilarski JQ, et al. Developmental nicotine exposure alters neurotransmission and excitability in hypoglossal motoneurons. J Neurophysiol. 2010;105:423–33. doi: 10.1152/jn.00876.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pillar G, et al. Genioglossal inspiratory activation: central respiratory vs mechanoreceptive influences. Respir Physiol. 2001;127:23–38. doi: 10.1016/s0034-5687(01)00230-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pittman LJ, Bailey EF. Genioglossus and intrinsic electromyographic activities in impeded and unimpeded protrusion tasks. J Neurophysiol. 2009;101:276–82. doi: 10.1152/jn.91065.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puckree T, et al. Abdominal motor unit activity during respiratory and nonrespiratory tasks. J Appl Physiol. 1998;84:1707–15. doi: 10.1152/jappl.1998.84.5.1707. [DOI] [PubMed] [Google Scholar]
- Reinking RM, et al. The motor units of cat medial gastrocnemius: problem of their categorisation on the basis of mechanical properties. Exp Brain Res. 1975;23:301–13. doi: 10.1007/BF00239742. [DOI] [PubMed] [Google Scholar]
- Remmers JE, et al. Pathogenesis of upper airway occlusion during sleep. J Appl Physiol. 1978;44:931–8. doi: 10.1152/jappl.1978.44.6.931. [DOI] [PubMed] [Google Scholar]
- Rice AD, et al. Synchronization of presynaptic input to motor units of tongue, inspiratory intercostal and diaphragm muscles. J Neurophysiol. 2011 doi: 10.1152/jn.01078.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson PA, Bailey EF. Tonically Discharging Genioglossus Motor Units Show No Evidence of Rate Coding with Hypercapnia. J Neurophysiol. 2010;103:1315–21. doi: 10.1152/jn.00686.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richter DW. Generation and maintenance of the respiratory rhythm. J Exp Biol. 1982;100:93–107. doi: 10.1242/jeb.100.1.93. [DOI] [PubMed] [Google Scholar]
- Saboisky JP, et al. Tonic and phasic respiratory drives to human genioglossus motoneurons during breathing. J Neurophysiol. 2006;95:2213–21. doi: 10.1152/jn.00940.2005. [DOI] [PubMed] [Google Scholar]
- Saboisky JP, et al. Neural drive to human genioglossus in obstructive sleep apnoea. J Physiol. 2007a;585:135–46. doi: 10.1113/jphysiol.2007.139584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saboisky JP, et al. Potential therapeutic targets in obstructive sleep apnoea. Expert Opin Ther Targets. 2009;13:795–809. doi: 10.1517/14728220903005608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saboisky JP, et al. Differential activation among five human inspiratory motoneuron pools during tidal breathing. J Appl Physiol. 2007b;102:772–80. doi: 10.1152/japplphysiol.00683.2006. [DOI] [PubMed] [Google Scholar]
- Saboisky JP, et al. Recruitment and rate-coding strategies of the human genioglossus muscle. J Appl Physiol. 2010;109:1939–49. doi: 10.1152/japplphysiol.00812.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauerland EK, Harper RM. The human tongue during sleep: electromyographic activity of the genioglossus muscle. Exp Neurol. 1976;51:160–70. doi: 10.1016/0014-4886(76)90061-3. [DOI] [PubMed] [Google Scholar]
- Sauerland EK, Mitchell SP. Electromyographic activity of the human Genioglossus muscle in response to respiration and to positional changes of the head. Bull Los Angeles Neurol Soc. 1970;35:69–73. [PubMed] [Google Scholar]
- Sauerland EK, Mitchell SP. Electromyographic activity of intrinsic and extrinsic muscles of the human tongue. Tex Rep Biol Med. 1975;33:444–55. [PubMed] [Google Scholar]
- Sauerland EK, et al. EMG patterns of oropharyngeal muscles during respiration in wakefulness and sleep. Electromyogr Clin Neurophysiol. 1981;21:307–316. [PubMed] [Google Scholar]
- Sawczuk A, Mosier KM. Neural control of tongue movement with respect to respiration and swallowing. Crit Rev Oral Biol Med. 2001;12:18–37. doi: 10.1177/10454411010120010101. [DOI] [PubMed] [Google Scholar]
- Schellenberg JB, et al. Physical findings and the risk for obstructive sleep apnea. The importance of oropharyngeal structures. Am J Respir Crit Care Med. 2000;162:740–8. doi: 10.1164/ajrccm.162.2.9908123. [DOI] [PubMed] [Google Scholar]
- Schwartz AR, et al. Therapeutic electrical stimulation of the hypoglossal nerve in obstructive sleep apnea. Arch Otolaryngol Head Neck Surg. 2001;127:1216–23. doi: 10.1001/archotol.127.10.1216. [DOI] [PubMed] [Google Scholar]
- Schwartz AR, et al. Electrical stimulation of the lingual musculature in obstructive sleep apnea. J Appl Physiol. 1996;81:643–52. doi: 10.1152/jappl.1996.81.2.643. [DOI] [PubMed] [Google Scholar]
- Series FJ, et al. Characteristics of the genioglossus and musculus uvulae in sleep apnea hypopnea syndrome and in snorers. Am J Respir Crit Care Med. 1996;153:1870–4. doi: 10.1164/ajrccm.153.6.8665048. [DOI] [PubMed] [Google Scholar]
- Shi YX, et al. Breathing route dependence of upper airway muscle activity during hyperpnea. J Appl Physiol. 1998;84:1701–6. doi: 10.1152/jappl.1998.84.5.1701. [DOI] [PubMed] [Google Scholar]
- Smith KK, Kier WM. Trunks, tongues and tentacles:moving with skeletons of muscle. American Scientist. 1989;77:22–35. [Google Scholar]
- Smith PL, et al. Electrical stimulation of upper airway musculature. Sleep. 1996;19:S284–7. [PubMed] [Google Scholar]
- Sokoloff AJ. Activity of tongue muscles during respiration: it takes a village? J Appl Physiol. 2004;96:438–9. doi: 10.1152/japplphysiol.01079.2003. [DOI] [PubMed] [Google Scholar]
- Sokoloff AJ, Deacon TW. Musculotopic organization of the hypoglossal nucleus in the cynomolgus monkey, Macaca fascicularis. J Comp Neurol. 1992;324:81–93. doi: 10.1002/cne.903240107. [DOI] [PubMed] [Google Scholar]
- Stone M, et al. Functional segments in tongue movement. Clin Linguist Phon. 2004;18:507–21. doi: 10.1080/02699200410003583. [DOI] [PubMed] [Google Scholar]
- Sumi T. Functional differentiation of hypoglossal neurons in cats. Jpn J Physiol. 1969;19:55–67. doi: 10.2170/jjphysiol.19.55. [DOI] [PubMed] [Google Scholar]
- Sussman HM, MacNeilage PF. Motor unit correlates of stress: preliminary observations. J Acoust Soc Am. 1978;64:338–40. doi: 10.1121/1.381953. [DOI] [PubMed] [Google Scholar]
- Sussman HM, et al. Recruitment and discharge patterns of single motor units during speech production. J Speech Hear Res. 1977;20:613–30. doi: 10.1044/jshr.2004.613. [DOI] [PubMed] [Google Scholar]
- Tangel DJ, et al. Influences of NREM sleep on the activity of tonic vs. inspiratory phasic muscles in normal men. J Appl Physiol. 1992;73:1058–66. doi: 10.1152/jappl.1992.73.3.1058. [DOI] [PubMed] [Google Scholar]
- Tangel DJ, et al. Influence of sleep on tensor palatini EMG and upper airway resistance in normal men. J Appl Physiol. 1991;70:2574–81. doi: 10.1152/jappl.1991.70.6.2574. [DOI] [PubMed] [Google Scholar]
- Tarras-Wahlberg S, Rekling JC. Hypoglossal motoneurons in newborn mice receive respiratory drive from both sides of the medulla. Neuroscience. 2009;161:259–68. doi: 10.1016/j.neuroscience.2009.02.064. [DOI] [PubMed] [Google Scholar]
- Tokizane T. Functional differentiation of human skeletal muscle. Kagaku. 1955;25:229–233. 291–297. [Google Scholar]
- Tokizane T, et al. Electromyographic studies on the human respiratory muscles; studies on the activity pattern of neuromuscular units. Jpn J Physiol. 1952;2:232–47. doi: 10.2170/jjphysiol.2.232. [DOI] [PubMed] [Google Scholar]
- Tsuiki S, et al. Functional divergence of human genioglossus motor units with respiratory-related activity. Eur Respir J. 2000;15:906–10. doi: 10.1034/j.1399-3003.2000.15e16.x. [DOI] [PubMed] [Google Scholar]
- Uemura M, et al. Topographical arrangement of hypoglossal motoneurons: an HRP study in the cat. Neurosci Lett. 1979;13:99–104. doi: 10.1016/0304-3940(79)90024-7. [DOI] [PubMed] [Google Scholar]
- van Lunteren E, Dick TE. Motor unit regulation of mammalian pharyngeal dilator muscle activity. J Clin Invest. 1989;84:577–85. doi: 10.1172/JCI114201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lunteren E, Dick TE. Intrinsic properties of pharyngeal and diaphragmatic respiratory motoneurons and muscles. J Appl Physiol. 1992;73:787–800. doi: 10.1152/jappl.1992.73.3.787. [DOI] [PubMed] [Google Scholar]
- van Lunteren E, Dick TE. Heterogeneity within geniohyoid motor unit subpopulations in firing patterns during breathing. Respir Physiol. 2001;124:23–33. doi: 10.1016/s0034-5687(00)00182-1. [DOI] [PubMed] [Google Scholar]
- Van Lunteren E, Strohl KP. Striated muscles of the upper airways. In: Mathew O, Sant’Ambrogio F, editors. Respiratory functionof the Upper Airway. Dekker; New York: 1988. pp. 87–123. [Google Scholar]
- Wheatley JR, et al. Influence of sleep on response to negative airway pressure of tensor palatini muscle and retropalatal airway. J Appl Physiol. 1993;75:2117–24. doi: 10.1152/jappl.1993.75.5.2117. [DOI] [PubMed] [Google Scholar]
- Wiegand L, et al. Collapsibility of the human upper airway during normal sleep. J Appl Physiol. 1989;66:1800–8. doi: 10.1152/jappl.1989.66.4.1800. [DOI] [PubMed] [Google Scholar]
- Wiegand L, et al. Changes in upper airway muscle activation and ventilation during phasic REM sleep in normal men. J Appl Physiol. 1991;71:488–97. doi: 10.1152/jappl.1991.71.2.488. [DOI] [PubMed] [Google Scholar]
- Wilkinson V, et al. Discharge patterns of human genioglossus motor units during sleep onset. Sleep. 2008;31:525–33. doi: 10.1093/sleep/31.4.525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson V, et al. Discharge patterns of human genioglossus motor units during arousal from sleep. Sleep. 2010;33:379–87. doi: 10.1093/sleep/33.3.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams JS, et al. Influence of posture and breathing route on neural drive to upper airway dilator muscles during exercise. J Appl Physiol. 2000;89:590–8. doi: 10.1152/jappl.2000.89.2.590. [DOI] [PubMed] [Google Scholar]
- Withington-Wray DJ, et al. Intracellular analysis of respiratory-modulated hypoglossal motoneurons in the cat. Neuroscience. 1988;25:1041–51. doi: 10.1016/0306-4522(88)90057-7. [DOI] [PubMed] [Google Scholar]
- Younes M. Contributions of upper airway mechanics and control mechanisms to severity of obstructive apnea. Am J Respir Crit Care Med. 2003;168:645–58. doi: 10.1164/rccm.200302-201OC. [DOI] [PubMed] [Google Scholar]