Abstract
Purpose
Periodontal ligament (PDL) cell differentiation into osteoblasts is important in bone formation. Bone formation is a complex biological process and involves several tightly regulated gene expression patterns of bone-related proteins. The expression patterns of bone related proteins are regulated in a temporal manner both in vivo and in vitro. The aim of this study was to observe the gene expression profile in PDL cell proliferation, differentiation, and mineralization in vitro.
Methods
PDL cells were grown until confluence, which were then designated as day 0, and nodule formation was induced by the addition of 50 µg/mL ascorbic acid, 10 mM β-glycerophosphate, and 100 nM dexamethasone to the medium. The dishes were stained with Alizarin Red S on days 1, 7, 14, and 21. Real-time polymerase chain reaction was performed for the detection of various genes on days 0, 1, 7, 14, and 21.
Results
On day 0 with a confluent monolayer, in the active proliferative stage, c-myc gene expression was observed at its maximal level. On day 7 with a multilayer, alkaline phosphatase, bone morphogenetic protein (BMP)-2, and BMP-4 gene expression had increased and this was followed by maximal expression of osteocalcin on day 14 with the initiation of nodule mineralization. In relationship to apoptosis, c-fos gene expression peaked on day 21 and was characterized by the post-mineralization stage. Here, various genes were regulated in a temporal manner during PDL fibroblast proliferation, extracellular matrix maturation, and mineralization. The gene expression pattern was similar.
Conclusions
We can speculate that the gene expression pattern occurs during PDL cell proliferation, differentiation, and mineralization. On the basis of these results, it might be possible to understand the various factors that influence PDL cell proliferation, extracellular matrix maturation, and mineralization with regard to gene expression patterns.
Keywords: Cell differentiation, Gene expression, Periodontal ligament
INTRODUCTION
Periodontal tissue is composed of epithelial tissue and soft and hard connective tissue. Periodontal tissue damage have various reasons, most commonly periodontitis, which is destructive to the connective tissue matrix and its cells, and results in periodontal ligament loss, alveolar bone resorption, and tooth loss. The major challenges in contemporary periodontal therapy are to re-establish soft tissue attachment to the newly formed cementum on the root surface and to restore lost bone. This requires regeneration of the gingival connective tissue destroyed by inflammation, the formation of new cementum and restoration of bone loss, and most importantly, the new attachment of connective tissue fibers to the previously diseased root surfaces. Therefore, it is essential for the differentiation of cells responsible for the cementum, alveolar bone, and fiber production. The periodontal ligament (PDL) is a connective tissue located between the cementum and the alveolar bone. It has been confirmed that undifferentiated mesenchymal cells in the PDL are essential for osteogenesis and cementogenesis in periodontal tissues and can differentiate into osteoblasts or cementoblasts [1,2].
PDL cell differentiation into osteoblasts is important in bone formation. There is some suggestion that PDL cells can differentiate into osteoblasts or cementoblasts, and they are implicated in the regeneration of the alveolar bone, cementum, and PDL [3,4]. Many studies have reported mineralized nodule formation by the PDL cells both in vitro and in vivo [5,6]. Nodules were formed in rat PDL cell cultures, and when 10 mM Na-beta-glycerophosphate was added, these nodules became mineralized. This suggests that the PDL contains osteoprogenitor cells, which differentiate into osteoblasts and produce bone-like tissue [6]. In another study, the mineralized nodules were formed on day 21 in 100 nM or 5 µM dexamethasone treated human PDL (hPDL) cell groups in a dose-dependent manner [7].
Bone formation is a complex biological process and involves several tightly regulated gene expression patterns of bone-related proteins [8]. The expression of bone-related proteins during bone formation has been studied in both in vivo and in vitro systems. The expression patterns of bone related proteins in in vivo and in vitro systems are regulated in a temporal manner. Stein and Lian [8] demonstrated that the expression of cell cycle or growth-related genes (histone, c-fos, c-myc) and extracellular matrix genes (type I collagen [COL I], fibronectin, and transforming growth factor-β1) increased and was followed by mineralization-related genes such as osteocalcin (OC), osteopontin (OPN), and bone sialoprotein (BSP) during bone formation. Therefore, it is reasonable to speculate that there are common expression patterns of bone-related proteins for osteoblast differentiation. However, it is assumed that each osteoblast culture system has its own specific expression patterns of bone-related proteins during osteoblast differentiation [9]. PDL cells also have their own specific expression patterns of bone-related proteins during differentiation.
Accordingly, Zhumabayeva et al. [10] reported gene expression patterns (SPARC, Col I, alkaline phosphate [ALP], OPN, BSP, OC) in relation to the bone morphogenetic proteins (BMP)-7 and dexamethasone. Meanwhile, Strayhorn et al. [11] reported the effects of various growth factors (BMPs, platelet-derived growth factor, and insulin-like growth factor) on osteoblast differentiation-associated genes (OPN, OC, BSP, Osf2/Cbfa1) in vitro using mouse osteoprogenitor cells. BMPs promote regeneration of the bone and also periodontal tissues including alveolar bone, cementum, and PDL [12-14].
However, most of the studies reporting gene expression patterns regarding PDL cell differentiation have the limitation that reported genes are only observed at the restricted stage during mineralization and studied with regard to various growth factors. Hence, the overall gene expression pattern of PDL cells during cell proliferation, bone matrix formation, maturation, and mineralization needs to be elucidated.
Therefore, in the present study, we observed the growth-related (c-myc and c-fos) and differentiation-related [ALP, BMP-2, BMP-4 and OC] gene expression patterns during osteogenic differentiation of hPDL cells in vitro.
MATERIALS AND METHODS
Cell isolation and culture
The PDL tissues were obtained from the PDL of premolar teeth extracted for orthodontic reasons. Premolars were collected from donors, at Department of Periodontology, Kyungpook National University Hospital. The protocols for this study were approved by the Institutional Review Board of Kyungpook National University Hospital (KNUH-74005-745) and informed consent was obtained from donors.
After extraction, the teeth were placed and rinsed in biopsy media (Dulbecco's modified Eagle's medium [DMEM; Gibco BRL, Grand Island, NY, USA] with 500 U/mL penicillin and 500 ug/mL streptomycin). After rinsing, only the PDL attached to the middle third of the root was removed with a curette to avoid contamination with the gingival and apical tissues. The PDL tissues were cut into small pieces and placed in small culture dishes. The tissues were incubated in biopsy medium at 37℃, 5% CO2, and 95% humidity. The medium was replaced with culture medium (DMEM with 10% fetal bovine serum [FBS], 100 U/mL penicillin and 100 µg/mL streptomycin). After reaching confluence, the cells were passaged with 0.25% trypsin/0.1% ethylene diaminotetraacetic acid. Two patients' PDL cells between the fifth and sixth passage were used in the following studies.
Mineralization nodule formation
PDL cells were plated at a density of 1×106 cells/100 mm in a Type I collagen coated petri dish and incubated in DMEM supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were grown until confluent which was designated as day 0, and nodule formation was induced by the addition of 50 µg/mL ascorbic acid, 10 mM β-glycerophosphate (β-GP), and 100 nM dexamethasone to the medium. The medium was changed every 3 days for 21 days. The dishes were stained with Alizarin Red S to identify mineralization nodules on day 1, 7, 14, and 21. The medium was removed and the cells were washed with PBS and fixed in 2% paraformaldehyde for 30 minutes at 4℃. The cells were then gently washed with H2O. The cells were stained in a solution of 1% Alizarin Red S at pH 4.2 for 10 minutes at room temperature and were then washed with H2O, twice. The samples were air dried and were examined and photographed under a light microscope. To determine mineralization, a percentage of the calcified areas of the total area per dish were analyzed using the image analysis program, I-solution (IM Technology Inc., Daegeon, Korea).
Real-time polymerase chain reaction (PCR)
Total RNA extraction
PDL cells were plated at a density of 1×106 cells/100 mm in a COL I coated petri dish and incubated in DMEM supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were grown until confluent which was designated as day 0, and mineralization was induced by the addition of 50 µg/mL ascorbic acid, 10 mM β-GP, and 100 nM dexamethasone to the medium. Media was changed every 3 days for 21 days.
Total RNA was extracted from the cultured cells by using a modified acid phenol method on days 0, 1, 7, 14, and 21. Briefly, the growth medium was removed and the cells were lysed with Trizol (Invitrogen, Carlsbad, CA, USA). The lysate was cleared and extracted with 1/10 the volume of 1-bromo-3-chloporopane. The aqueous layer was collected in a new tube and precipitated with isopropanol. After 75% ethanol washing, the pellet was air-dried and resuspended in diethyl pyrocarbonate-treated water, and quantified by A260/A280 measurements using a UV spectrometer (DU530, Beckman Coulter, Brea, CA, USA).
Real-time PCR
Total RNA was reverse-transcribed to produce cDNA using the ImProm-II reverse transcription system (Promega Inc., Madison, WI, USA) according to the manufacturer's protocol. SYRB Green I-based real-time PCR was done on an MJ Research DNA Engine Opticon Monitor II continuous fluorescence detection system (MJ Research Inc., Walthan, MA, USA). All PCR mixtures contained the following: PCR buffer [final concentration 10 mM Tris-Hcl (pH 9.0), 50 mM KCL, 2 mM MgCl2, and 0.1% Triton X-100), 250 mM deoxy-NTP (Roche, Mannheim, Germany), 0.5 mM of each PCR primer, 0.5X SYBR Green I (Invitrogen), 5% dimethyl sulfoxide, and 1 U taq DNA polymerase (Promega Inc.)] with 2 µL cDNA in a 25 µL final volume reaction mix. The samples were loaded into the wells of Low Profile 96-well microplates. After an initial denaturation step for 1 minute at 94℃, the conditions for cycling were as follows: 35 cycles of 30 second at 94℃, 30 second at 53℃, and 1 minute at 72℃. The fluorescence signal was measured immediately after incubation for 5 seconds at 78℃ following the extension step, which eliminated possible primer dimer detection. At the end of the final PCR cycle, a melting curve was generated to identify the specificity of the PCR product. For each run, serial dilutions of human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) plasmids were used as standards for the quantitative measurement for the amount of amplified DNA. All samples were run in duplicates and the data were presented as a ratio of gene (c-myc, c-fos, ALP, OC, BMP-2, and BMP-4) to GAPDH. The sequences of the primers used for the real-time PCR analysis are presented in Table 1. The primers used for the real-time PCR analysis were purchased from Bioneer Corporation (Daegeon, Korea).
Table 1.
Primers and probes used in real-time polymerase chain reaction.
F: forward, R: reverse.
Statistical analysis
One-way analysis of variance was performed to determine the differences within and between groups with Tukey's test as a post hoc test between day 0 and each day. A P-value of 0.05 or less was considered to be statistically significant.
RESULTS
Nodule formation during PDL cell differentiation
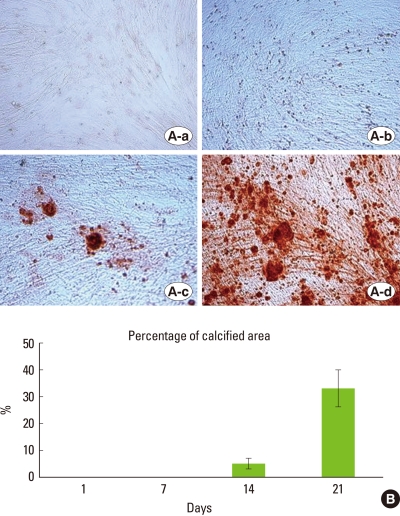
The culture dishes were stained with Alizarin Red S on days 1, 7, 14, and 21. The PDL cells were incubated in mineralized medium over the course of 21 days. On days 1 and 7, no mineralization nodule was observed, but 5% and 33% of the culture dish had calcified by days 14 and 21, respectively (Fig. 1B). In the development of mineralized nodules by PDL cells, distinct stages were identified based on the appearance of the cultures. The first stage was the formation of the confluent monolayer of cells. These cells exhibited spindle or polygonal morphology (Fig. 1A-a). When treated with dexamethasone, ascorbic acid, and β-GP, cell proliferation began to occur in certain areas with the formation of several cell layers appearing like clusters of cells. This is referred to as the multilayer formation stage (Fig. 1A-b). The next stage was associated with the formation of nodules formed by the deposition of the matrix in the clusters of cells and the initiation of nodule mineralization (Fig. 1A-c). In this stage, a few small mineralized nodules started to develop on day 14. During the last stage, referred to as the post-mineralization stage, mineralization of the matrix developed and the mineralization nodule size also increased (Fig. 1A-d).
Figure 1.
Morphology of the various stages and percentage of calcified areas during the formation of mineralized nodule by human periodontal ligament (PDL) cells. (A) shows the visual field at ×200 magnification on days 1(a), 7(b), 14(c), and 21(d). Developed mineralization is observed and size and number of nodules are increased compared to PDL cells on day 14. (B) Measurement of calcified areas by Alizarin Red S stain. Data are shown as the mean±SD of two patients. Percentages of the calcified areas are 5% and 33% of the culture dish on day 14 and 21, respectively.
Expression of mRNA for bone-related proteins during PDL cell differentiation
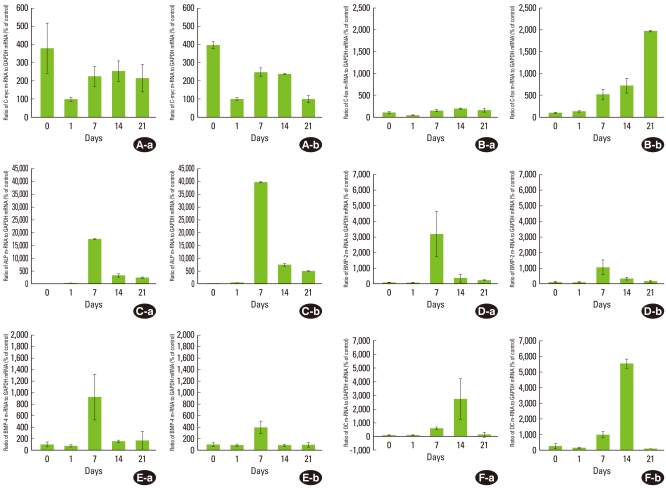
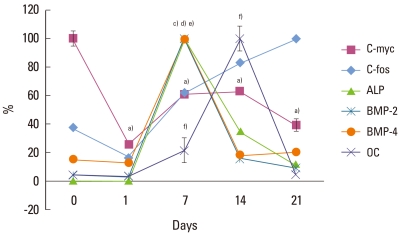
To determine the expression of bone-related proteins during PDL cell differentiation, the expression level of their mRNAs was observed. c-myc is a cell growth-regulated gene and encodes proteins that support proliferation by functioning as transactivation factors. c-myc, as a proliferation marker, showed maximal expression for the confluent cell stage (on day 0), then decreased by 25% of the maximal level on day 1. Thereafter, the c-myc mRNA expression level slightly increased to 60% of the maximal level and was maintained at that level (Fig. 2A). And there was a statistically significant difference as compared to day 0 and each day (P<0.05, Fig. 3).
Figure 2.
c-myc (A), c-fos (B), ALP (C), BMP-2 (D), BMP-4 (E), and OC (F) mRNA expression during mineralization of human periodontal ligament (PDL) cells. The expression of each gene in PDL1 (a) and PDL2 (b) is shown. The graphs show the ratio of mRNA to GAPDH mRNA from the real-time polymerase chain reaction results. Control means the minimal gene expression level during 21 days. Values are means±SD of two cultures.
Figure 3.
Gene expression pattern during mineralization of periodontal ligament cells. Expression of genes was analyzed by real-time PCR and normalized to the levels of GAPDH mRNA. Values are presented as the percent of the maximal expression for each transcript. a)c-myc, b)c-fos, c)ALP, d)BMP-2, e)BMP-4, f)OC. a),c)-f)Statistically significant difference as compared with day 0 (P<0.05). b)(P>0/05).
As proliferation was downregulated, ALP mRNA was highly expressed. ALP mRNA showed a maximal expression level on day 7, and then declined to 32% and 12% of the maximal level on day 14 and 21, respectively (Fig. 2C). ALP is one of the earliest markers of osteoblastic cell differentiation. Thus, the active expression of the ALP gene on day 7 (multilayer formation stage) shows the initiation of PDL cell differentiation. On day 7 and 14, there was a statistically significant difference as compared with day 0 (P<0.05, Fig. 3).
BMPs belong to the TGF-β superfamily and have a variety of functions dependent upon tissue location and developmental stage. Many studies have reinforced these findings demonstrating that BMPs promote the regeneration of bone and also periodontal tissues including alveolar bone, cementum, and PDL [13,14]. It has been reported that both gingival and PDL fibroblasts express mRNA for BMP-2 and -4 [15]. Therefore, the expressions of BMP-2 and -4 were observed to determine their expression patterns during PDL cell differentiation. BMP-2 and -4 showed similar expression patterns to ALP. In the early stage (on days 0 and 1), the expression of BMP-2 was no more than 6% of the maximal level, but showed highly active expression on day 7, and, BMP-2 expression was followed by a decrease to 12% of the maximal level observed over the observational period (Fig. 2D). Similarly, expression of BMP-4 was 18% and 15% of the maximal level on days 0 and 1, respectively and showed a maximal level on day 7. The expression of BMP-4 then decreased to about 20% of the maximal level seen on days 14 and 21 (Fig. 2E). On day 7, there was a statistically significant difference as compared with day 0 (P<0.05, Fig. 3).
The active expression of ALP was followed by the active expression of OC. OC, as a marker for the mineralization stage, was expressed on day 7 (20% of the maximal level) and showed the maximal level on day 14. OC gene expression on day 21 declined to 4% of the maximal level (Fig. 2F). The increase in OC gene expression on day 14 correlates with the formation of bone nodules. On day 14, there was a statistically significant difference as compared with day 0 (P<0.05, Fig. 3).
Like c-myc, c-fos is a cell growth regulated gene [16], and it has been reported that the expression of c-fos appears to precede terminal differentiation and cell death [17] and differentiation of the osteoblast [18]. On day 0, for the active proliferation period, c-fos mRNA was expressed at 32% and declined to 18% of the maximal level. Then, c-fos expression levels gradually increased until day 21 when it showed the maximal level (P<0.05, Figs. 2B, 3).
Other PDL cells were shown to have a similar gene expression pattern with each other.
DISCUSSION
PDL cells in vivo have the capacity to differentiate into osteoblasts or cementoblasts, and to form alveolar bone and cementum, respectively. PDL cells in vitro have also been shown to possess osteoblast-like properties, including high constitutive levels of ALP activity, the production of cAMP in response to parathyroid hormones, and mineralized nodule formation. It was reported that mineralized nodules were formed in vitro by PDL cells [6-8].
The data obtained in this study indicate that the expression patterns of bone-related proteins, which are regulated in a temporal manner during successive developmental stages including proliferation, bone matrix formation/maturation, and mineralization, are unique in the PDL cell culture system as compared to other systems.
The presence of the active proliferation stage is evidence for the temporal expression of bone-related proteins during PDL cell differentiation. Its presence has been supported by two observations. First, the lack of expression of OC and the formation of bone nodules support the presence of the proliferation stage, and this is consistent with other systems such as those of rat calvarial osteoblasts [8] and bone marrow derived osteoblasts [19]. Second, we found that the expression of c-myc gradually decreased during periodontal cell differentiation. The downregulation of c-myc genes during cellular differentiation is similar to other studies done in a variety of cell types [8,20]. c-myc is a cell growth-regulated gene and encodes a protein that supports proliferation by functioning as a transactivation factor. It was reported that more than 95% of the cells proliferate after plating in normal rat diploid osteoblast cultures on day 5, and in that study, the cells showed the maximal expression of the c-myc gene [8]. In the present study, on day 0, for the active proliferation period showing the confluent cell stage, c-myc expression showed a maximal level, but the next day, decreased to 25% of the maximal level. It is suspected that the mineralization medium, added since day 0, has negative effects on periodontal cell proliferation.
There is a transition point early in the developmental sequence that occurs when proliferation ceases and expression of genes related to the differentiated phenotype of osteoblasts is initiated. To determine the post-proliferation period, we observed ALP mRNA expression. ALP is one of the earliest markers for osteoblastic cells. Mineralization of the matrix is initiated by the expression of the membrane-bound glycoprotein ALP. ALP is expressed in large amounts in osteoblasts in vivo and has also been found in differentiation studies with osteoblast-like cell lines in vitro [8,20,21]. We observed the maximal expression of ALP on day 7 for the multilayer formation stage prior to the initiation of nodule mineralization. These results correspond to those of Stein and Lian [8] that ALP expression was maximal for the postproliferative period (on day 16) and decreased during the heavly mineralization stage.
BMPs belong to the TGF-β superfamily and have a variety of functions dependent on tissue location and developmental stage. In vitro studies using recombinant BMPs have demonstrated that BMP-2, -4, -6, and -7 are potent inducers of osteoblast differentiation [22,23]. In vivo studies have reinforced these findings demonstrating that BMPs promote regeneration of the bone and also periodontal tissues including bone, cementum, and PDL [13,14]. Strayhorn et al. [11] reported that BMPs decreased cell proliferation and promoted gene expression of BSP, OPN, and OC in MC3T3-E1 cell cultures. BMP-2, involved in the epithelial-mesenchymal interaction, plays an important role in cell differentiation [24], stimulates osteoblast differentiation, and bone formation [25]. We observed that BMP-2 and -4 had similar expression patterns to ALP with gene expression maximal on day 7 and then declining until day 21. Previous studies have shown that BMP-2 stimulates ALP activity and inhibits the induction of OC by 1,25-dihydroxyvitamin D3 in hPDL cell cultures [26,27]. These results correspond with our observations related to BMP-2 and OC. BMP-2 was highly expressed prior to OC expression, and OC was highly expressed when the expression of BMP-2 declined to 22% of the maximal level. Also, BMP-2 enhanced BMP-4 mRNA expression in cultures of fetal rat calvarial osteoblasts [28]. BMP-4 stimulates OPN, Cbfa1, and BMP-2 expression in hPDL cells [29]. Thus, the enhancement of BMP-2 by BMP-4 or BMP-4 by BMP-2 may be associated with the regulation of bone-related protein expression. However, in another study, BMP-4 mRNA was expressed when the expression of the extracellular matrix proteins was increased and BMP-2 mRNA was expressed when OC appeared during pulp cell differentiation into pre-odontoblasts [30].
The next evidence to support the existence of these stages during PDL cell differentiation was the formation of the nodules and expression of the OC gene. We call this period the mineralization stage. The nodule formation began to be observed on day 14 of this culture system and OC expression showed a maximal level on the same day. This result confirms the separate observations of the studies of MC3T3-E1 [9,31] and several other cells [8,32]. OC expression is well correlated to the time of mineralization of the nodules, and this supports the other studies that reported the major role of OC in mineralization. OC has been shown to contribute to the regulation of the mineral phase in bone both in vitro as a potential inhibitor of mineral nucleation [33], and in vivo as a bone matrix signal that promotes osteoclast differentiation and activation [34]. Thus, expression late in the osteoblast development sequence suggests that OC is a marker of mature osteoblasts, which is consistent with a possible role for the synthesis and binding of OC to minerals in the coupling of bone formation for resorption. Also, OC binds to COL I reversibly [35], and OPN and OC also form complexes in vitro [36]. However, we observed a decrease in OC expression up to 4% of the maximal level on day 21 showing advanced mineralization. This finding is discordant with the results of studies in rat calvarial osteoblasts, bone marrow cells, MC3T3-E1 cells, and PDL cells [9,10,37,38]. They reported that OC expression was maintained once the mineralization of nodules was initiated. We assumed that the decreased expression level of OC on day 21 was related to apoptosis during PDL cell differentiation.
Apoptosis during osteogenesis has not been extensively studied, but it must play an important role in normal bone formation and remodeling. Apoptosis becomes a prominent feature of the in vitro culture in the late mineralization stage during the development of the mature osteoblast phenotype. The absence of ascorbic acid from the media to prevent nodule formation and the absence of β-GP during the culture period to inhibit extracellular matrix (ECM) mineralization inhibit apoptosis. Conversely, the acceleration of differentiation by dexamethasone leads to an increase in the number of apoptotic cells. Lynch et al. [21] reported the occurrence of cell death by apoptosis on day 29 when advanced mineralization developed using a fetal rat calvaria-derived osteoblast culture. Shin et al. [39] observed that Dkk-1 and Nip3, which are apoptosis-inducing agents, were up-regulated, and Btf and TAX1BP1, which have an anti-apoptosis effect, were down-regulated on day 21 when the nodules of the mineralized matrix were strongly stained with Alizarin Red S in the PDL cells cultured in the media with mineralization supplements. Thus, we call this period showing the advanced mineralization and decreased OC expression level the post-mineralization stage or cell death period.
In relation to the decrease of OC expression on day 21, c-fos expression was maximal during the post-mineralization stage. Expression of c-fos appears to precede terminal differentiation and cell death [17] and also precedes differentiation of osteoblasts [18]. Smeyne et al. [40] suggested three possible scenarios regarding the functional role of Fos in the process of cell death. First, expression may have no causal relationship to cell death, but it may merely be a reflection of a breakdown of intracellular signal transduction. Second, the signaling pathway that leads to cell death may result in a coincidental induction of c-fos and perhaps other immediate-early genes. Third, in some circumstances, the c-fos product may be a required component of the gene regulation pathway that leads to cell death.
In conclusion, our results demonstrate that the expression patterns of bone-related proteins are regulated in a temporal manner during the successive developmental stages including proliferation, bone matrix formation/maturation, and mineralization just like the rat calvarial osteoblast system [8]. However, the temporal expression patterns of some bone-related proteins are unique in this culture system compared with other systems. Furthermore, on the basis of our results, we might be able to understand the various factors that influence PDL cell proliferation, extracellular matrix maturation, and mineralization with regard to the gene expression patterns.
ACKNOWLEDGMENTS
This work was supported by Korea Science and Engineering Foundation (KOSEF) grant funded by the Korean Government (MEST) (No. 2010-0020544). The authors report no conflicts of interest related to this study.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Strutz F, Okada H, Lo CW, Danoff T, Carone RL, Tomaszewski JE, et al. Identification and characterization of a fibroblast marker: FSP1. J Cell Biol. 1995;130:393–405. doi: 10.1083/jcb.130.2.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lackler KP, Cochran DL, Hoang AM, Takacs V, Oates TW. Development of an in vitro wound healing model for periodontal cells. J Periodontol. 2000;71:226–237. doi: 10.1902/jop.2000.71.2.226. [DOI] [PubMed] [Google Scholar]
- 3.Nojima N, Kobayashi M, Shionome M, Takahashi N, Suda T, Hasegawa K. Fibroblastic cells derived from bovine periodontal ligaments have the phenotypes of osteoblasts. J Periodontal Res. 1990;25:179–185. doi: 10.1111/j.1600-0765.1990.tb01041.x. [DOI] [PubMed] [Google Scholar]
- 4.Somerman MJ, Young MF, Foster RA, Moehring JM, Imm G, Sauk JJ. Characteristics of human periodontal ligament cells in vitro. Arch Oral Biol. 1990;35:241–247. doi: 10.1016/0003-9969(90)90062-f. [DOI] [PubMed] [Google Scholar]
- 5.Cho MI, Matsuda N, Lin WL, Moshier A, Ramakrishnan PR. In vitro formation of mineralized nodules by periodontal ligament cells from the rat. Calcif Tissue Int. 1992;50:459–467. doi: 10.1007/BF00296778. [DOI] [PubMed] [Google Scholar]
- 6.Mukai M, Yoshimine Y, Akamine A, Maeda K. Bone-like nodules formed in vitro by rat periodontal ligament cells. Cell Tissue Res. 1993;271:453–460. doi: 10.1007/BF02913727. [DOI] [PubMed] [Google Scholar]
- 7.Chung HB, Park JW, Suh JY. The effect of dexamethasone on the gene expression of the bone matrix protein in the periodontal ligament cells. J Korean Acad Periodontol. 2002;32:445–456. [Google Scholar]
- 8.Stein GS, Lian JB. Molecular mechanisms mediating proliferation/differentiation interrelationships during progressive development of the osteoblast phenotype. Endocr Rev. 1993;14:424–442. doi: 10.1210/edrv-14-4-424. [DOI] [PubMed] [Google Scholar]
- 9.Choi JY, Lee BH, Song KB, Park RW, Kim IS, Sohn KY, et al. Expression patterns of bone-related proteins during osteoblastic differentiation in MC3T3-E1 cells. J Cell Biochem. 1996;61:609–618. doi: 10.1002/(SICI)1097-4644(19960616)61:4%3C609::AID-JCB15%3E3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 10.Zhumabayeva BD, Lin WL, Choung PH, Chien HH, Sodek J, Sampath KT, et al. Differential induction of bone sialoprotein by dexamethasone and osteogenic protein-1 (OP-1, BMP-7) in rat periodontal ligament cells in vitro: relationship to the mineralization of tissue nodules. Int J Oral Biol. 1998;23:91–101. [Google Scholar]
- 11.Strayhorn CL, Garrett JS, Dunn RL, Benedict JJ, Somerman MJ. Growth factors regulate expression of osteoblast-associated genes. J Periodontol. 1999;70:1345–1354. doi: 10.1902/jop.1999.70.11.1345. [DOI] [PubMed] [Google Scholar]
- 12.Lian JB, Stein GS. Concepts of osteoblast growth and differentiation: basis for modulation of bone cell development and tissue formation. Crit Rev Oral Biol Med. 1992;3:269–305. doi: 10.1177/10454411920030030501. [DOI] [PubMed] [Google Scholar]
- 13.Ripamonti U. Induction of cementogenesis and periodontal ligament regeneration by bone morphogenetic proteins. In: Lindholm TS, editor. Bone morphogenetic proteins: biology, biochemistry and reconstructive surgery. San Diego: Academic Press; 1996. pp. 189–198. [Google Scholar]
- 14.King GN, King N, Cruchley AT, Wozney JM, Hughes FJ. Recombinant human bone morphogenetic protein-2 promotes wound healing in rat periodontal fenestration defects. J Dent Res. 1997;76:1460–1470. doi: 10.1177/00220345970760080801. [DOI] [PubMed] [Google Scholar]
- 15.Ivanovski S, Li H, Haase HR, Bartold PM. Expression of bone associated macromolecules by gingival and periodontal ligament fibroblasts. J Periodontal Res. 2001;36:131–141. doi: 10.1034/j.1600-0765.2001.360301.x. [DOI] [PubMed] [Google Scholar]
- 16.Shalhoub V, Gerstenfeld LC, Collart D, Lian JB, Stein GS. Downregulation of cell growth and cell cycle regulated genes during chick osteoblast differentiation with the reciprocal expression of histone gene variants. Biochemistry. 1989;28:5318–5322. doi: 10.1021/bi00439a002. [DOI] [PubMed] [Google Scholar]
- 17.Preston GA, Lyon TT, Yin Y, Lang JE, Solomon G, Annab L, et al. Induction of apoptosis by c-Fos protein. Mol Cell Biol. 1996;16:211–218. doi: 10.1128/mcb.16.1.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McCabe LR, Banerjee C, Kundu R, Harrison RJ, Dobner PR, Stein JL, et al. Developmental expression and activities of specific fos and jun proteins are functionally related to osteoblast maturation: role of Fra-2 and Jun D during differentiation. Endocrinology. 1996;137:4398–4408. doi: 10.1210/endo.137.10.8828501. [DOI] [PubMed] [Google Scholar]
- 19.Aubin JE, Turken K, Heersche JNM. Osteoblastic cell lineage. In: Noda M, editor. Cellular and molecular biology of bone. San Diego: Academic Press; 1993. pp. 1–44. [Google Scholar]
- 20.Onyia JE, Hale LV, Miles RR, Cain RL, Tu Y, Hulman JF, et al. Molecular characterization of gene expression changes in ROS 17/2.8 cells cultured in diffusion chambers in vivo. Calcif Tissue Int. 1999;65:133–138. doi: 10.1007/s002239900671. [DOI] [PubMed] [Google Scholar]
- 21.Lynch MP, Capparelli C, Stein JL, Stein GS, Lian JB. Apoptosis during bone-like tissue development in vitro. J Cell Biochem. 1998;68:31–49. [PubMed] [Google Scholar]
- 22.Hughes FJ, Collyer J, Stanfield M, Goodman SA. The effects of bone morphogenetic protein-2, -4, and -6 on differentiation of rat osteoblast cells in vitro. Endocrinology. 1995;136:2671–2677. doi: 10.1210/endo.136.6.7750491. [DOI] [PubMed] [Google Scholar]
- 23.Zegzula HD, Buck DC, Brekke J, Wozney JM, Hollinger JO. Bone formation with use of rhBMP-2 (recombinant human bone morphogenetic protein-2) J Bone Joint Surg Am. 1997;79:1778–1790. doi: 10.2106/00004623-199712000-00003. [DOI] [PubMed] [Google Scholar]
- 24.Thesleff I, Vaahtokari A, Kettunen P, Aberg T. Epithelial-mesenchymal signaling during tooth development. Connect Tissue Res. 1995;32:9–15. doi: 10.3109/03008209509013700. [DOI] [PubMed] [Google Scholar]
- 25.Boden SD, McCuaig K, Hair G, Racine M, Titus L, Wozney JM, et al. Differential effects and glucocorticoid potentiation of bone morphogenetic protein action during rat osteoblast differentiation in vitro. Endocrinology. 1996;137:3401–3407. doi: 10.1210/endo.137.8.8754767. [DOI] [PubMed] [Google Scholar]
- 26.Kobayashi M, Takiguchi T, Suzuki R, Yamaguchi A, Deguchi K, Shionome M, et al. Recombinant human bone morphogenetic protein-2 stimulates osteoblastic differentiation in cells isolated from human periodontal ligament. J Dent Res. 1999;78:1624–1633. doi: 10.1177/00220345990780100701. [DOI] [PubMed] [Google Scholar]
- 27.Zaman KU, Sugaya T, Kato H. Effect of recombinant human platelet-derived growth factor-BB and bone morphogenetic protein-2 application to demineralized dentin on early periodontal ligament cell response. J Periodontal Res. 1999;34:244–250. doi: 10.1111/j.1600-0765.1999.tb02250.x. [DOI] [PubMed] [Google Scholar]
- 28.Chen D, Harris MA, Rossini G, Dunstan CR, Dallas SL, Feng JQ, et al. Bone morphogenetic protein 2 (BMP-2) enhances BMP-3, BMP-4, and bone cell differentiation marker gene expression during the induction of mineralized bone matrix formation in cultures of fetal rat calvarial osteoblasts. Calcif Tissue Int. 1997;60:283–290. doi: 10.1007/s002239900230. [DOI] [PubMed] [Google Scholar]
- 29.Xu WP, Shiba H, Mizuno N, Uchida Y, Mouri Y, Kawaguchi H, et al. Effect of bone morphogenetic proteins-4, -5 and -6 on DNA synthesis and expression of bone-related proteins in cultured human periodontal ligament cells. Cell Biol Int. 2004;28:675–682. doi: 10.1016/j.cellbi.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 30.Nakashima M, Nagasawa H, Yamada Y, Reddi AH. Regulatory role of transforming growth factor-beta, bone morphogenetic protein-2, and protein-4 on gene expression of extracellular matrix proteins and differentiation of dental pulp cells. Dev Biol. 1994;162:18–28. doi: 10.1006/dbio.1994.1063. [DOI] [PubMed] [Google Scholar]
- 31.Franceschi RT, Iyer BS. Relationship between collagen synthesis and expression of the osteoblast phenotype in MC3T3-E1 cells. J Bone Miner Res. 1992;7:235–246. doi: 10.1002/jbmr.5650070216. [DOI] [PubMed] [Google Scholar]
- 32.Gerstenfeld LC, Chipman SD, Glowacki J, Lian JB. Expression of differentiated function by mineralizing cultures of chicken osteoblasts. Dev Biol. 1987;122:49–60. doi: 10.1016/0012-1606(87)90331-9. [DOI] [PubMed] [Google Scholar]
- 33.Romberg RW, Werness PG, Riggs BL, Mann KG. Inhibition of hydroxyapatite crystal growth by bone-specific and other calcium-binding proteins. Biochemistry. 1986;25:1176–1180. doi: 10.1021/bi00353a035. [DOI] [PubMed] [Google Scholar]
- 34.Glowacki J, Lian JB. Impaired recruitment and differentiation of osteoclast progenitors by osteocalcin-deplete bone implants. Cell Differ. 1987;21:247–254. doi: 10.1016/0045-6039(87)90479-9. [DOI] [PubMed] [Google Scholar]
- 35.Prigodich RV, Vesely MR. Characterization of the complex between bovine osteocalcin and type I collagen. Arch Biochem Biophys. 1997;345:339–341. doi: 10.1006/abbi.1997.0254. [DOI] [PubMed] [Google Scholar]
- 36.Ritter NM, Farach-Carson MC, Butler WT. Evidence for the formation of a complex between osteopontin and osteocalcin. J Bone Miner Res. 1992;7:877–885. doi: 10.1002/jbmr.5650070804. [DOI] [PubMed] [Google Scholar]
- 37.Aronow MA, Gerstenfeld LC, Owen TA, Tassinari MS, Stein GS, Lian JB. Factors that promote progressive development of the osteoblast phenotype in cultured fetal rat calvaria cells. J Cell Physiol. 1990;143:213–221. doi: 10.1002/jcp.1041430203. [DOI] [PubMed] [Google Scholar]
- 38.Malaval L, Modrowski D, Gupta AK, Aubin JE. Cellular expression of bone-related proteins during in vitro osteogenesis in rat bone marrow stromal cell cultures. J Cell Physiol. 1994;158:555–572. doi: 10.1002/jcp.1041580322. [DOI] [PubMed] [Google Scholar]
- 39.Shin JH, Park JW, Yeo SI, Noh WC, Kim MK, Kim JC, et al. Identification of matrix mineralization-related genes in human periodontal ligament cells using cDNA microarray. J Korean Acad Periodontol. 2007;37(Suppl):447–463. [Google Scholar]
- 40.Smeyne RJ, Vendrell M, Hayward M, Baker SJ, Miao GG, Schilling K, et al. Continuous c-fos expression precedes programmed cell death in vivo. Nature. 1993;363:166–169. doi: 10.1038/363166a0. [DOI] [PubMed] [Google Scholar]